 Open Access Article
Open Access ArticleEffect of divalent cation substitution (M = Mn, Ni, Mg, Cu) on the structural, magnetic and electrical properties of MFe2O4 spinel ferrites
Ibtihel Soudani *a,
Nazir Mustaphab and
Mokhtar Hjiri*b
*a,
Nazir Mustaphab and
Mokhtar Hjiri*b
aLaboratory of Multifunctional Materials and Applications (LaMMA), LR16ES18, Faculty of Sciences of Sfax, University of Sfax, BP 1171, 3000 Sfax, Tunisia. E-mail: ebtihel.soudani@gmail.com
bDepartment of Physics, College of Sciences, Imam Mohammad Ibn Saud Islamic University (IMSIU), Riyadh 11623, Saudi Arabia. E-mail: mbhjiri@imamu.edu.sa
First published on 15th May 2026
Abstract
In This study, a systematic investigation of the influence of the nature of the divalent cation (M2+ = Ni, Mg, Cu, Mn) on the multifunctional properties of spinel ferrites MFe2O4 synthesized by solid-state reaction is presented. X-ray diffraction (XRD) analyses validate the formation of single-phase cubic phases (space group Fd![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m). Despite significant disparities between the ionic radii of the M2+ cations (0.69 to 0.80 Å), the lattice retains its integrity, highlighting the high structural robustness of these materials in the face of cationic substitution. The precise determination of the oxidation states and the distribution of metal ions between the tetrahedral and octahedral sites of our prepared samples is carried out by X-ray photoelectron spectroscopy (XPS), while Raman spectroscopy corroborates these results by identifying the five vibrational modes characteristic of the spinel structure. Magnetic characterizations reveals that the ferrites MnFe2O4 and NiFe2O4 exhibit the highest saturation magnetizations (Ms), whereas MgFe2O4 shows a reduced magnetization due to the non-magnetic nature of the Mg2+ ion. The compound CuFe2O4 exhibits intermediate values, influenced by Jahn–Teller distortions and the mixed valence states of copper. The coercivity (Hc) varies depending on the cation, classifying some samples as soft magnetic materials, optimal for low-energy-loss applications. Furthermore, high-frequency analyses demonstrate that substitution engineering allows these ferrites to be optimized for microwave devices. Finally, electrical measurements performed between 400 and 700 K reveal remarkable performance for thermal sensing. Combined with an excellent stability factor, these properties authenticate the reliability of these materials for high-temperature sensor applications.
m). Despite significant disparities between the ionic radii of the M2+ cations (0.69 to 0.80 Å), the lattice retains its integrity, highlighting the high structural robustness of these materials in the face of cationic substitution. The precise determination of the oxidation states and the distribution of metal ions between the tetrahedral and octahedral sites of our prepared samples is carried out by X-ray photoelectron spectroscopy (XPS), while Raman spectroscopy corroborates these results by identifying the five vibrational modes characteristic of the spinel structure. Magnetic characterizations reveals that the ferrites MnFe2O4 and NiFe2O4 exhibit the highest saturation magnetizations (Ms), whereas MgFe2O4 shows a reduced magnetization due to the non-magnetic nature of the Mg2+ ion. The compound CuFe2O4 exhibits intermediate values, influenced by Jahn–Teller distortions and the mixed valence states of copper. The coercivity (Hc) varies depending on the cation, classifying some samples as soft magnetic materials, optimal for low-energy-loss applications. Furthermore, high-frequency analyses demonstrate that substitution engineering allows these ferrites to be optimized for microwave devices. Finally, electrical measurements performed between 400 and 700 K reveal remarkable performance for thermal sensing. Combined with an excellent stability factor, these properties authenticate the reliability of these materials for high-temperature sensor applications.
1 Introduction
Spinel ferrites, with the general formula MFe2O4 (M2+ = Mn, Ni, Mg, Cu), have been extensively investigated for decades due to their unique combination of magnetic, electrical, and structural properties. This multifunctionality makes them highly attractive for a wide range of technological applications.1–5 These materials crystallize in a face-centered cubic structure, where oxygen anions form a close-packed lattice and cations are distributed between tetrahedral (A) and octahedral (B) sites. This flexible cationic distribution plays a crucial role in determining the fundamental properties of spinel ferrites, including saturation magnetization, coercivity, and electrical resistivity.6,7 In particular, partial cationic inversion in nickel ferrite nanostructures has been reported to enhance magnetic hardening, coercive field, and magnetic anisotropy due to changes in the cation occupancy between tetrahedral (A) and octahedral (B) sites and the resulting modification of superexchange interactions.8Spinel ferrites are semiconducting materials widely used in diverse fields such as gas sensing, heterogeneous catalysis, energy storage, wastewater treatment, magnetic resonance imaging (MRI), and high-frequency electromagnetic wave absorption.9,10 Beyond their conventional applications in electronics and microwave devices, these materials have recently gained increasing attention for biomedical applications, particularly in magnetic hyperthermia and cancer therapy.11 In such applications, their performance can be significantly enhanced through precise control of chemical composition and microstructure.12
Recent studies have emphasized the importance of compositional engineering in tuning the structural, magnetic, electrical, and optical properties of spinel ferrites. For instance, Zn–Ni13,14 and Mn–Zn15 ferrites exhibit enhanced magneto-functional and optical properties under optimized processing conditions, while MgFe2O4- and CoFe2O4-based systems have demonstrated improved dielectric performance and microwave absorption efficiency.16 In addition, hybridization strategies, including graphene incorporation and multivalent ion substitution, have been shown to significantly affect AC conductivity, dielectric relaxation, and thermal stability.17,18 These findings highlight that compositional flexibility and microstructural control are key factors in the rational design of next-generation multifunctional ferrite materials.
Despite the large number of studies on individual ferrite compositions such as MnFe2O4, NiFe2O4, MgFe2O4, and CuFe2O4, comparative investigations under strictly identical synthesis conditions remain limited. Such an approach is essential to isolate the intrinsic effect of the divalent cation on structural order, magnetic response, and electrical behavior, while eliminating variations arising from synthesis routes or particle size effects. Moreover, establishing clear correlations between cation nature, crystallographic structure, and functional properties is critical for optimizing ferrites for targeted applications, including sensors, inductors, microwave devices, and multifunctional systems.19–21
In this work, we present a systematic comparative study of MnFe2O4, NiFe2O4, MgFe2O4, and CuFe2O4 ferrites synthesized under identical experimental conditions. Structural (XRD), vibrational (Raman), and surface (XPS) analyses are combined with magnetic and electrical measurements to elucidate the influence of the divalent cation on lattice parameters, cation distribution, electronic states, magnetic behavior, and dielectric properties. This approach allows a direct structure–property correlation, providing deeper insight into the fundamental mechanisms governing spinel ferrites and offering guidance for the design of advanced materials for electronic, magnetic, and energy-related applications.
2 Experimental procedure
2.1 Preparation of samples
Spinel ferrite powders with nominal compositions MFe2O4 (M2+ = Mn, Ni, Mg, Cu) are synthesized by the conventional solid-state reaction method, as illustrated in Fig. 1. High-purity metal oxides are utilized as precursors (99.9%, Sigma-Aldrich): the phases are obtained by combining MnO, CuO, MgO and NiO with Fe2O3, respectively. Each oxide mixture is scrupulously weighted to obtain the desired stoichiometry. Then, it is mechanically ground in an agate mortar for a total of 6 hours. This thorough homogenization process not only reduced particle size but also significantly increased the reactivity of the precursor powders. Afterwards, the homogenized powders are well grinding, then it is compacted into cylindrical pellets in order of 8 mm in diameter and 1 mm in thickness using a uniaxial hydraulic press. To eliminate any residual organic material, the substances produced are subjected to a calcination cycle for 24 h at 1000 °C to enhance grain crystallization. At last, the pellets are annealed in ambient air at 1200 °C for 9 hours. This last step is the sintering which is important step to ensure a high sample densification and promoting the formation of a well-defined spinel phase.2.2 Characterization techniques
The crystalline structure and vibrational properties of the samples are analyzed by X-ray diffraction (XRD) on a Panalytical system using Cu-Kα radiation (0.02° step size) and by Raman spectroscopy using a SENTERRA Spectrometer (Bruker) with green laser excitation of 540 nm. The surface chemical composition is examined by X-ray photoelectron spectroscopy (XPS) on a Thermo Scientific Kα instrument. Magnetic characterizations are performed using an extraction magnetometer, M(H) cycles up to 5 T at room temperature. Finally, the electrical behavior is probed by impedance spectroscopy (Solartron 1260) over a wide thermal range from 400 K to 700 K. For these measurements, platinum electrodes are applied to the sintered disks to obtain stable ohmic contacts, the tests being carried out at thermal equilibrium under ambient air to validate the reproducibility of the results.3 Results and discussion
3.1 Structural analysis
To evaluate the influence of the divalent cation M2+ (M = Mn, Ni, Mg, Cu) on the crystal structure of spinel ferrites, X-ray diffraction (XRD) data collected at room temperature were analyzed using the Rietveld refinement method with FullProf software.5 The XRD patterns (Fig. 2a–d) confirm the formation of a single-phase cubic spinel structure with space group Fd![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m, with no detectable secondary phases, indicating high crystalline purity.
m, with no detectable secondary phases, indicating high crystalline purity.
 | ||
| Fig. 2 X-ray diffraction patterns of MFe2O4 (M2+ = Ni, Mg, Cu, Mn) spinel ferrites with Rietveld analysis. Mn (a), Cu (b), Mg (c), and Ni (d). | ||
The refined profiles show good agreement between the calculated and experimental data, as reflected by the reliability factors and goodness-of-fit (χ2 ≈ 1). The structural parameters obtained from the refinement are summarized in Table 1. The crystal structures, projected along the (011) plane and visualized using VESTA software, are shown in the insets of Fig. 2.
| Formula | MnFe2O4 | CuFe2O4 | MgFe2O4 | NiFe2O4 |
|---|---|---|---|---|
| Crystallin system | Cubic | Cubic | Cubic | Cubic |
| Space group | Fd![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m m |
Fd![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m m |
Fd![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m m |
Fd![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m m |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||
| Lattice parameter (Å) | ||||
| a = b = c | 8.41 | 8.37 | 8.39 | 8.36 |
| α = β = γ (°) | 90 | 90 | 90 | 90 |
| V (Å3) | 594.82 | 586.37 | 590.59 | 584 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||
| Reliability factors (%) | ||||
| Rp (%)/Rwp (%)/Rexp (%) | 6.61/8.68/6.30 | 6.24/8.43/3.81 | 6.94/9.75/7.11 | 6.01/8.28/4.20 |
| Goodness of fit χ2 | 1.64 | 1.48 | 1.54 | 1.18 |
The evolution of lattice parameters reveals a systematic variation correlated with the ionic radius of the M2+ cations (Mn2+: 0.83 Å, Cu2+: 0.73 Å, Mg2+: 0.72 Å, Ni2+: 0.69 Å,.22 A monotonic increase in lattice parameter and unit cell volume is observed with increasing ionic radius, in agreement with Vegard's law.23 This behavior reflects the accommodation of larger cations within the spinel lattice, leading to lattice expansion and local structural relaxation. Similar trends have been reported in the literature, where cation substitution influences crystallinity and cation distribution between tetrahedral (A) and octahedral (B) sites.24–27
Furthermore, the (311) diffraction peak, which is the most intense reflection, exhibits a systematic shift toward lower 2θ values (Fig. 3), indicating an increase in interplanar spacing according to Bragg's law. This shift further confirms the lattice expansion induced by larger M2+ cations.
It is important to note that spinel ferrites are rarely perfectly stoichiometric systems. The presence of intrinsic defects such as oxygen vacancies and cationic disorder can induce local lattice distortions and influence structural properties. These defects may also lead to mixed valence states (e.g., Fe3+/Fe2+), which play a significant role in charge transport mechanisms via polaron hopping.28
To ensure a rigorous evaluation of the microstructural parameters, the crystallite size and lattice strain were decoupled using the Williamson–Hall (W–H) equation. Unlike the simplified Scherrer equation,29 which attributes peak broadening solely to size effects, the W–H approach accounts for the inherent micro-strain ε resulting from lattice distortions and defects. The W–H plots and the extracted parameters are presented in Fig. 4.
The average crystallite size which presented in Fig. 4 is determined by Williamson–Hall (W–H) equation:30
 | (1) |
In which β is a full width at half maximum (FWHM) which was determined by peak fitting using a pseudo-Voigt function after background subtraction. The fitting procedure was performed using FullProf software, allowing accurate extraction of peak position and width. θ is the diffraction angle and 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 416 A is the Cu-Kα wavelength of the radiation.
416 A is the Cu-Kα wavelength of the radiation.
The variation in crystallite size can be attributed to the development of microstrain and structural defects arising from the mismatch between the ionic radii of M2+ cations and Fe3+ ions. This mismatch induces lattice distortion and local disorder, which tend to hinder grain growth. The associated strain energy favors nucleation over crystal growth, resulting in relatively small crystallite sizes.
Although the nature of the M2+ cation influences the crystallite size, the relatively similar values observed for all samples suggest that crystal growth is mainly governed by synthesis conditions, particularly the high-temperature treatment. The slightly larger crystallite size observed for CuFe2O4 can be attributed to the Jahn–Teller effect associated with Cu2+ ions, which induces local distortion and may facilitate cation diffusion, thereby promoting grain growth.31
Overall, these results highlight the strong influence of cation substitution and defect chemistry on the structural properties of spinel ferrites. The interplay between ionic size, lattice distortion, and defect formation provides an effective way to tune the structural and functional properties of these materials.32
3.2 Vibrational analysis
The analysis of the crystal structure and lattice dynamics is further investigated using confocal Raman spectroscopy, a technique known for its sensitivity to atomic vibrations and its ability to probe cationic disorder and phase transitions in ferrites.33 Raman spectra, recorded at room temperature between 120 and 850 cm−1 under 540 nm laser excitation (Fig. 5), validate the chemical purity and phase formation of the spinel ferrite samples. According to factor group analysis for a spinel structure belonging to the Fd![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m space group, the theory predicts five Raman active modes:34
m space group, the theory predicts five Raman active modes:34| ξ = A1g + Eg + 3 T2g | (2) |
![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m space group. This structural correspondence, combined with the absence of spurious peaks in the spectra, corroborates the X-ray diffraction (XRD) analyses and thus confirms the single-phase nature of all the compounds studied. The purity of these spinel phases guarantees that the observed resistivity variations and thermal sensitivities result intrinsically from the lattice dynamics and the cationic distribution specific to each ferrite.37
m space group. This structural correspondence, combined with the absence of spurious peaks in the spectra, corroborates the X-ray diffraction (XRD) analyses and thus confirms the single-phase nature of all the compounds studied. The purity of these spinel phases guarantees that the observed resistivity variations and thermal sensitivities result intrinsically from the lattice dynamics and the cationic distribution specific to each ferrite.37
To extract the frequency and full width at half maximum (FWHM) parameters, the spectra are fitted using Lorentzian profiles. Then, it is tabulated in Table 2. In spinel ferrites, frequencies above 600 cm−1 are associated with oxygen vibrations in tetrahedral sites (AO4), while lower modes correspond to the octahedral sublattice.38 The appearance of “shoulder bands” likely results from a breaking of the momentum conservation rule, linked to the electrical and vibrational behavior of MFe2O4 ferrites (M = Mn, Cu, Mg, Ni) is intrinsically linked to the redistribution of cations between tetrahedral (A) and octahedral (B) sites, a phenomenon governed by the ionic radius and the field stabilization energy of the ligands.39
| Samples | Raman band | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| A1g | T2g(3) | T2g(2) | Eg | T2g(1) | ||||||
| Position (cm−1) | FWHM | Position (cm−1) | FWHM | Position (cm−1) | FWHM | Position (cm−1) | FWHM | Position (cm−1) | FWHM | |
| MnFe2O4 | 615 | 66 | 492 | 66 | 352 | 57 | 270 | 64 | 189 | 35 |
| CuFe2O4 | 623 | 69 | 434 | 84 | 276 | 75 | 193 | 29 | 134 | 31 |
| MgFe2O4 | 625 | 16 | 505 | 61 | 356 | 38 | 287 | 37 | 121 | 35 |
| NiFe2O4 | 645 | 3 | 585 | 8 | 477 | 9 | 396 | 14 | 251 | 11 |
The vibrational behavior of spinel ferrites is strongly influenced by the distribution of cations between A and B sites, which depends on factors such as ionic radius and crystal field stabilization energy.39 For instance, Mn2+ ions (0.83 Å) tend to occupy tetrahedral sites, leading to a predominantly normal spinel structure, whereas Ni2+ ions (0.69 Å) preferentially occupy octahedral sites, resulting in an inverse spinel configuration. This redistribution is quantified by the inversion degree (i), which significantly affects bond lengths, lattice dynamics, and charge transport mechanisms.40
Additional structural distortions, such as the Jahn–Teller effect associated with Cu2+ ions, or the partially inverse character of MgFe2O4, further modify the local symmetry and vibrational behavior. These effects are reflected in the Raman spectra by shifts in peak positions and changes in band widths.
In particular, the A1g mode exhibits a systematic shift toward higher frequencies from MnFe2O4 to NiFe2O4, which can be attributed to the decrease in ionic radius and the corresponding increase in metal–oxygen bond strength.41 Moreover, the broadening of Raman bands, especially in MnFe2O4 and CuFe2O4, suggests enhanced cationic disorder and microstructural effects such as reduced crystallite size or lattice strain.42
Overall, these results demonstrate that Raman spectroscopy is a powerful tool for probing cation distribution, inversion degree, and defect-induced structural distortions in spinel ferrites, providing complementary insights to XRD analysis.43
3.3 X-ray photoelectron spectroscopy (XPS) analysis
X-ray photoelectron spectroscopy (XPS) was employed to investigate the surface chemical composition and oxidation states of MFe2O4 (M = Mn, Ni, Mg, Cu) spinel ferrites. The survey spectra recorded over the binding energy range of 0–1350 eV (Fig. 6) confirm the presence of Fe, O, and the corresponding divalent cations (Mn, Ni, Mg, and Cu). A C 1s peak is also observed, originating from adventitious carbon contamination on the sample surface.44 No additional impurity peaks are detected, indicating the high chemical purity of the samples. | ||
| Fig. 6 X-ray photoelectron spectroscopy (XPS) survey spectra of MFe2O4 (M2+ = Ni, Mg, Cu, Mn) spinels ferrites. | ||
The XPS spectra were fitted after Shirley background subtraction using Gaussian–Lorentzian (pseudo-Voigt) functions. The fitting procedure was performed using Origin, allowing accurate determination of peak positions, intensities, and satellite features.
The high-resolution Fe 2p spectra (Fig. 7) exhibit a characteristic doublet with binding energies located at approximately 710–711 eV (Fe 2p3/2) and 723–725 eV (Fe 2p1/2). In addition, satellite features are clearly observed at about 7.8 eV above the main peaks (as indicated in Fig. 7), which are characteristic of Fe3+ ions and arise from shake-up processes.45 These features confirm that iron predominantly exists in the Fe3+ oxidation state in all samples.
 | ||
| Fig. 7 High resolution X-ray photoelectron spectroscopy (XPS) spectra of the elements of MFe2O4 (M2+ = Ni, Mg, Cu, Mn) spinel ferrites. | ||
Slight variations in peak positions and relative intensities are observed among MnFe2O4, NiFe2O4, MgFe2O4, and CuFe2O4, reflecting differences in local chemical environments and cation distribution within the spinel lattice.
The Mn 2p spectrum of MnFe2O4 shows two main peaks located at approximately 641 eV (Mn 2p3/2) and 653 eV (Mn 2p1/2), consistent with the presence of Mn2+ ions.46 For NiFe2O4, the Ni 2p spectrum exhibits peaks at ∼855–856 eV and ∼872–873 eV, confirming the Ni2+ oxidation state.47 The Mg 2p peak in MgFe2O4 appears around 48–49 eV, indicating the presence of Mg2+ ions. In the case of CuFe2O4, the Cu 2p spectrum shows a characteristic doublet at ∼934 eV and ∼954 eV, along with asymmetry features associated with Cu2+ ions and Jahn–Teller distortion effects.48–50
The O 1s spectra (Fig. 7) can be deconvoluted into two main components. The dominant peak at ∼529.5–530.0 eV corresponds to lattice oxygen (O2−) associated with metal–oxygen bonds (M–O–Fe) in the spinel structure.51 The second component, located in the range 531–532 eV, is attributed to surface oxygen species and/or oxygen vacancies. The variation in the relative intensity of this component among the samples indicates differences in defect concentration, which may significantly influence electrical conductivity and magnetic properties.52
Overall, the XPS results confirm the formation of single-phase spinel ferrites composed of divalent cations (Mn2+, Ni2+, Mg2+, Cu2+) and trivalent Fe3+ ions. The observed variations in binding energies, cation distribution, and oxygen-related defects highlight the strong influence of the M2+ cation on the surface electronic structure, which plays a key role in determining the functional properties of these materials.
3.4 Magnetic properties and hysteresis loop analysis
The magnetic properties of MFe2O4 (M = Mn, Cu, Mg, Ni) spinel ferrites were investigated at room temperature using hysteresis (M–H) measurements (Fig. 8). All samples exhibit typical ferrimagnetic behavior, characterized by a rapid increase in magnetization at low magnetic fields followed by a tendency toward saturation at higher fields. The main magnetic parameters, including saturation magnetization (Ms), remanent magnetization (Mr), and coercive field (Hc), are summarized in Table 3. These parameters strongly depend on the nature of the divalent cation M2+, its magnetic moment, ionic radius, and its distribution between tetrahedral (A) and octahedral (B) sites (Fig. 9).| Samples | MnFe2O4 | CuFe2O4 | MgFe2O4 | NiFe2O4 |
|---|---|---|---|---|
| Ms (emu per g) | 67.17 | 22.06 | 16.69 | 33.07 |
| Mr (emu per g) | 16.65 | 12.04 | 45.46 | 10.44 |
| Hc (Oe) | 113.31 | 1040.60 | 192.91 | 240.01 |
| A (Oe) | 122.84 | 182.04 | 95.12 | 146.56 |
| b 104 (Oe2) | 3.75 | 6.27 | 1.21 | 4.32 |
| k 10−6 (emu per g per Oe) | 2.96 | 5.744 | 1.68 | 3.06 |
| K1 104 (erg per cm3) | 3.96 | 12.66 | 1.51 | 4.46 |
| µ (µB) | 2.77 | 0.95 | 0.6 | 1.38 |
| nth (µB) | 5.00 | 1.00 | 0.00 | 1.99 |
 | ||
| Fig. 9 The evolution of microwave operating frequency versus the Applied magnetic field of MFe2O4 (M2+ = Ni, Mg, Cu, Mn) spinels ferrites. | ||
MnFe2O4 exhibits the highest saturation magnetization, which can be attributed to the high magnetic moment of Mn2+ ions and their contribution to A–B superexchange interactions.53 In contrast, MgFe2O4 shows the lowest Ms due to the non-magnetic nature of Mg2+ ions, which weakens the magnetic coupling between sublattices.54 NiFe2O4 presents intermediate behavior, consistent with its inverse spinel structure where Ni2+ ions preferentially occupy octahedral sites.55 CuFe2O4 is distinguished by a relatively higher coercive field, which is mainly attributed to the Jahn–Teller effect of Cu2+ ions. This effect induces local lattice distortions, enhances magneto-crystalline anisotropy, and increases resistance to magnetization reversal.56,57 Overall, the relatively low Hc values indicate that the studied ferrites behave as soft magnetic materials, although CuFe2O4 shows a tendency toward harder magnetic behavior.58
The magneto-crystalline anisotropy constant (K1) was estimated using the Stoner–Wohlfarth relation:59
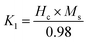 | (3) |
The obtained values indicate that CuFe2O4 exhibits the highest anisotropy, whereas MgFe2O4 shows the lowest value. NiFe2O4 and MnFe2O4 present intermediate values, reflecting the influence of electronic configuration and spin–orbit coupling. In addition to intrinsic factors, microstructural features such as crystallite size and internal strain also contribute to the effective anisotropy.
To improve the accuracy of saturation magnetization values, Ms was extrapolated using the law of approach to saturation:60
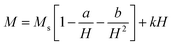 | (4) |
Fitting the experimental data using eqn (4) allowed the extraction of parameters related to magnetic inhomogeneities (a), magneto-crystalline anisotropy (b), and high-field susceptibility (k). The results show that CuFe2O4 exhibits higher magnetic inhomogeneity, while MgFe2O4 and MnFe2O4 display more homogeneous magnetic behavior.33
The magnetic moment per formula unit (µ) was calculated using the following relation:44
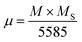 | (5) |
| nth = MB − MA | (6) |
The calculated magnetic moments show qualitative agreement with experimental results, confirming the dominant role of cation distribution in determining magnetic behavior.61
Finally, the microwave resonance frequency (ωm) was evaluated using the following relation:62
| ωm = 8π2Msγ | (7) |
3.5 Electrical characterization
This behavior is attributed to thermally activated charge transport governed by small polaron hopping between Fe2+ and Fe3+ ions located at octahedral (B) sites.63,64 In this mechanism, charge carriers are initially localized and require thermal energy to overcome potential barriers, leading to a decrease in resistivity as temperature increases. The resistivity follows the Arrhenius relation:63,64
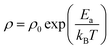 | (8) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) (ρ) versus 1/T (inset of Fig. 10). The obtained values are 1.25 eV (MnFe2O4), 1.05 eV (CuFe2O4), 1.01 eV (NiFe2O4), and 1.59 eV (MgFe2O4).
(ρ) versus 1/T (inset of Fig. 10). The obtained values are 1.25 eV (MnFe2O4), 1.05 eV (CuFe2O4), 1.01 eV (NiFe2O4), and 1.59 eV (MgFe2O4).
The relatively high activation energy observed for MgFe2O4 is associated with the non-magnetic nature of Mg2+ ions, which reduces the number of available hopping sites and weakens the Fe2+/Fe3+ exchange interactions. In contrast, lower Ea values in MnFe2O4 and NiFe2O4 indicate more favorable charge transport due to stronger electronic interactions within the spinel lattice.65
Fig. 11 presents the temperature coefficient of resistance (TCR) as a function of temperature. The obtained values confirm pronounced NTC behavior, with maximum sensitivities of −4.69%/K (MnFe2O4), −4.49%/K (CuFe2O4), −4.73%/K (NiFe2O4), and −5.31%/K (MgFe2O4). These values fall within the typical range for high-performance NTC thermistors, demonstrating good thermal sensitivity.66–68
 | ||
| Fig. 11 Variation of the Temperature Coefficient of Resistance (TCR) parameter as a function of the temperature of MFe2O4 (M = Mn, Cu, Mg, Ni) spinel ferrites samples. | ||
The thermistor constant β was determined from the slope of the ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) (ρ) versus 1/T plots (Fig. 12), according to:69–71
(ρ) versus 1/T plots (Fig. 12), according to:69–71
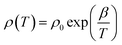 | (9) |
 | ||
Fig. 12 Evolution of ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) (ρ) vs. 1/(kBT) of MFe2O4 spinel ferrites samples: M = Mn (a), Cu (b), Mg(c), and Ni(d). (ρ) vs. 1/(kBT) of MFe2O4 spinel ferrites samples: M = Mn (a), Cu (b), Mg(c), and Ni(d). | ||
The stability factor (SF), defined as the ratio between maximum and minimum resistivity values over the studied temperature range, was also evaluated:69–71
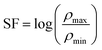 | (10) |
The obtained values indicate good thermal stability and reproducibility of the electrical response. In particular, the relatively low SF values observed for MnFe2O4, CuFe2O4, and NiFe2O4 confirm stable resistive behavior, which is essential for reliable sensor performance.71
Finally, the temperature sensitivity coefficient (α) was calculated using the standard relation derived from thermistor models:66,68
 | (11) |
The obtained α values range between −4%/K and −5.5%/K, which is consistent with typical NTC thermistor materials. This strong temperature dependence confirms that the studied ferrites are suitable for high-temperature sensing applications.67,68
Overall, the electrical properties of MFe2O4 ferrites are governed by thermally activated hopping conduction, strongly influenced by cation nature, defect concentration, and cation distribution within the spinel structure. These results highlight the potential of these materials for thermistor and high-temperature sensing applications.72–75
3.6 Outlook and challenges for industrial applications
Despite the promising multifunctional properties of MFe2O4 spinel ferrites, several scientific and technological challenges still limit their large-scale industrial deployment. One of the primary issues lies in the precise control of cation distribution between tetrahedral (A) and octahedral (B) sites. This distribution critically governs magnetic anisotropy, electrical conductivity, and dielectric response through A–B superexchange interactions. However, achieving a controlled degree of inversion remains challenging due to thermodynamic equilibrium constraints and kinetic limitations during high-temperature synthesis.76,77Defect engineering represents another key challenge. Intrinsic defects such as oxygen vacancies and cationic disorder strongly influence charge transport through small polaron hopping (Fe2+/Fe3+). While a controlled defect concentration can enhance electrical conductivity and sensing performance, uncontrolled defect formation often leads to instability, aging effects, and poor reproducibility.77 This remains a major bottleneck for industrial reliability. Advanced approaches such as controlled atmosphere processing, aliovalent doping, and defect compensation strategies are therefore required.77
From a processing standpoint, scalability and reproducibility remain critical barriers. Conventional solid-state synthesis typically involves high sintering temperatures, leading to excessive grain growth, microstructural inhomogeneity, and increased energy consumption. These factors not only affect magnetic losses and electrical performance but also hinder compatibility with emerging technologies such as miniaturized and flexible electronic systems.78
Another major challenge concerns high-frequency applications. Although spinel ferrites are widely used in microwave devices, achieving an optimal balance between high magnetic permeability and low dielectric/magnetic losses remains difficult, particularly for inverse spinel ferrites such as NiFe2O4 and CoFe2O4. This limitation is directly related to intrinsic material parameters (anisotropy, domain wall dynamics) and extrinsic factors (grain size, porosity, defects).79
In addition, long-term stability, thermal aging, and environmental robustness are often overlooked but are essential for real industrial applications, especially in high-temperature sensors and thermistors. The lack of standardized protocols for stability evaluation further complicates material qualification. Nevertheless, significant opportunities exist. By tailoring the nature of the divalent cation and optimizing cation distribution, it is possible to engineer ferrites with tunable magnetic softness, enhanced thermal sensitivity, and controlled electrical conductivity. These properties make spinel ferrites strong candidates for next-generation technologies, including high-frequency components, NTC thermistors, gas sensors, and electromagnetic interference (EMI) shielding systems.80
Future research should focus on advanced synthesis strategies such as sol–gel, hydrothermal, and thin-film deposition techniques, which offer improved control over microstructure and defect chemistry. Furthermore, integrating experimental approaches with theoretical modeling (e.g., density functional theory) will be essential to better understand cation distribution, defect formation, and structure–property relationships.80 Such combined approaches can accelerate the rational design and industrial optimization of high-performance spinel ferrite materials.
4 Conclusion
In this work, clear structure–property correlations in MFe2O4 (M = Mn, Cu, Mg, Ni) spinel ferrites have been established, highlighting the key role of cation engineering in tuning their multifunctional properties. X-ray diffraction combined with Rietveld refinement confirms the formation of a single-phase cubic spinel structure (space group Fd![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m) for all compositions, with lattice parameters systematically influenced by the ionic radius of the divalent cation (Fig. 13).
m) for all compositions, with lattice parameters systematically influenced by the ionic radius of the divalent cation (Fig. 13).
 | ||
| Fig. 13 Temperature dependence of the coefficient α (T) of MFe2O4 (M2+ = Ni, Mg, Cu, Mn) spinel ferrites. | ||
XPS analysis confirms the presence of the expected oxidation states (Fe3+ and M2+) and provides insight into the surface chemical environment, while Raman spectroscopy further confirms the characteristic vibrational fingerprint of the spinel structure through the observation of five active modes associated with tetrahedral and octahedral sites.
Magnetic measurements reveal a strong dependence of the magnetic properties on the nature of the divalent cation. MnFe2O4 exhibits the highest saturation magnetization due to the large magnetic moment of Mn2+, whereas MgFe2O4 shows reduced magnetization because of the non-magnetic character of Mg2+. NiFe2O4 displays intermediate behavior, while CuFe2O4 is characterized by enhanced coercivity attributed to Jahn–Teller-induced lattice distortions and increased magnetocrystalline anisotropy. The relatively low coercivity values observed for most samples indicate soft magnetic behavior, suitable for applications requiring low energy losses.
Electrical measurements confirm the semiconducting nature of all compositions, governed by thermally activated hopping conduction. The significant negative temperature coefficient of resistance (NTCR) and high TCR values highlight the strong potential of these materials for thermistor and temperature sensing applications.
Overall, this study demonstrates that the controlled selection of the divalent cation is an effective strategy to tailor the structural, magnetic, and electrical properties of spinel ferrites. These findings provide useful guidelines for the design of high-performance materials for electronic, sensing, and energy-related applications.
Author contributions
Ibtihel Soudani: writing – original draft, visualization, methodology, investigation, formal analysis. Nazir Mustapha: validation, supervision, resources, project administration, methodology. Mokhtar Hjiri: writing – original draft, supervision, methodology.Conflicts of interest
The authors declare that there is no conflict of interest.Data availability
The authors confirm that the data used to support the findings of this study are included within the article and are available from the corresponding author upon reasonable request.Acknowledgements
This work was supported and funded by the Deanship of Scientific Research at Imam Mohammad Ibn Saud Islamic University (IMSIU) (grant number IMSIU-DDRSP2602).References
- A. Zaman, A. Ali, H. Shahid, S. H. Alrefaee, S. Knani, V. Tirth and N. Elboughdiri, RSC Adv., 2025, 15(44), 37050–37061 RSC.
- R. Sharma, S. Bansal and S. Singhal, RSC Adv., 2015, 5(8), 6006–6018 RSC.
- T. N. Pham, T. Q. Huy and A. T. Le, RSC Adv., 2020, 10(52), 31622–31661 RSC.
- T. Alammar, M. A. Bedair, N. A. Andreeva, F. Al-Odail, M. A. Alkhalifah, M. E. Owda and A. M. Abuelela, RSC Adv., 2026, 16(14), 12737–12758 RSC.
- Z. Ali, J. A. Baig, H. I. Afridi, K. Akhtar, S. Perveen and N. Raza, RSC Adv., 2025, 15(58), 50470–50480 RSC.
- M. M. Hussein, S. A. Saafan, H. F. Abosheiasha, D. Zhou, D. S. Klygach, M. G. Vakhitov and M. A. Darwish, RSC Adv., 2023, 13(38), 26879–26891 RSC.
- M. D. Hossain, A. T. M. K. Jamil, M. S. Hossain, S. J. Ahmed, H. N. Das, R. Rashid and M. N. I. Khan, RSC Adv., 2022, 12(8), 4656–4671 RSC.
- J. M. Li, X. L. Zeng and Z. A. Xu, Appl. Phys. Lett., 2013, 103, 23–30 Search PubMed.
- M. Asif, W. Irshad, M. I. Asif, R. S. Almufarij, Y. M. Alanazi, R. U. Hassan and F. Atamurotov, J. Mater. Sci.: Mater. Electron., 2025, 36(29), 1–19 CrossRef.
- T. Prabhakaran and J. Hemalatha, Ceram. Int., 2016, 42(12), 14113–14120 CrossRef CAS.
- M. A. Almessiere, A. V. Trukhanov, F. A. Khan, Y. Slimani, N. Tashkandi, V. A. Turchenko and A. Baykal, Ceram. Int., 2020, 46(6), 7346–7354 CrossRef CAS.
- N. Mott, Appl. Phys., 1984, 56, 169–193 CAS.
- A. Manohar, K. Chintagumpala and K. H. Kim, Ceram. Int., 2021, 47(5), 7052–7061 CrossRef CAS.
- A. Miller and E. Abrahams, Phys. Rev., 1960, 120(3), 745–755 CrossRef CAS.
- M. B. Mohamed and A. M. Wahba, Ceram. Int., 2014, 40(8), 11773–11780 CrossRef CAS.
- S. A. Habib, S. A. Saafan, T. M. Meaz, M. A. Darwish, D. Zhou, M. U. Khandaker and M. K. Omar, Nanomaterials, 2022, 12, 931–950 CrossRef CAS PubMed.
- S. K. Kiran and N. Thakur, Ceram. Int., 2022, 48(4), 4444–4455 CrossRef CAS.
- Z. Wang, R. Gao, G. Chen, X. Deng, W. Cai and C. Fu, Ceram. Int., 2019, 46(7), 9154–9160 CrossRef.
- R. I. Shakirzyanov, A. L. Kozlovskiy, M. V. Zdorovets, A. L. Zheludkevich, D. Shlimas, N. V. Abmiotka and A. V. Trukhanov, J. Alloys Compd., 2023, 954, 170083 CrossRef CAS.
- M. M. Salem, L. V. Panina, E. L. Trukhanova, M. A. Darwish, A. T. Morchenko, T. I. Zubar and A. V. Trukhanov, Compos. Part B Eng., 2019, 174, 107054 CrossRef CAS.
- Y. Moualhi, H. Rahmouni, M. Gassoumi and K. Khirouni, Ceram. Int., 2020, 46(15), 24710–24717 CrossRef CAS.
- R. D. Shannon, Acta Crystallogr. A, 1976, 32(5), 751–767 CrossRef.
- A. V. Trukhanov, X. Zhao, V. G. Kostishin, D. I. Tishkevich, E. L. Trukhanova, M. A. Almessiere and Z. Sun, J. Alloys Compd., 2024, 986, 174048 CrossRef CAS.
- A. V. Trukhanov, M. A. Almessiere, A. Baykal, S. V. Trukhanov, Y. Slimani, D. A. Vinnik and M. Zdorovets, J. Alloys Compd., 2019, 788, 1193–1202 CrossRef CAS.
- D. A. Rayan, A. M. Elseman and M. M. Rashad, Appl. Phys. A, 2018, 124(9), 659 CrossRef.
- C. Iacovita, G. F. Stiufiuc, R. Dudric, N. Vedeanu, R. Tetean, R. I. Stiufiuc and C. M. Lucaciu, Magnetochemistry, 2020, 6, 2–20 CrossRef.
- S. B. Somvanshi, M. V. Khedkar, P. B. Kharat and K. M. Jadhav, Ceram. Int., 2020, 46(7), 8640–8650 CrossRef CAS.
- K. Pubby, K. Vijay Babu and S. Bindra Narang, Mater. Sci. Eng. B, 2020, 255, 114513 CrossRef CAS.
- U. Holzwarth and N. Gibson, Nat. Nanotechnol., 2011, 6(9), 534 CrossRef CAS PubMed.
- T. T. N. Nha, P. H. Nam, T. D. Thanh and P. T. Phong, RSC Adv., 2023, 13(36), 25007–25017 RSC.
- K. H. Maria, S. Choudhury and M. A. Hakim, Int. Nano Lett., 2013, 3(1), 42 CrossRef.
- F. Sun, L. Huang, R. Zhang, S. Wang, S. Jiang, Y. Sun and L. J. Geng, J. Alloys Compd., 2020, 834, 155248 CrossRef CAS.
- J. Massoudi, M. Smari, K. Nouri, E. Dhahri, K. Khirouni, S. Bertaina and E. K. Hlil, RSC Adv., 2020, 10(57), 34556–34580 RSC.
- J. P. Singh, R. C. Srivastava, H. M. Agrawal and R. Kumar, J. Raman Spectrosc., 2011, 42(7), 1510–1517 CrossRef CAS.
- M. Tan, Y. Köseoğlu, F. Alan and E. Şentürk, J. Alloys Compd., 2011, 509(39), 9399–9405 CrossRef CAS.
- P. Chirawatkul, S. Khoonsap, S. Phumying, C. Kaewhan, S. Pinitsoontorn and S. Maensiri, J. Alloys Compd., 2017, 697, 249–256 CrossRef CAS.
- S. Aliyeva, S. Babayev and T. Mehdiyev, J. Raman Spectrosc., 2018, 49(2), 271–278 CrossRef CAS.
- D. K. Pradhan, S. Kumari, V. S. Puli, P. T. Das, D. K. Pradhan, A. Kumar and R. S. Katiyar, Phys. Chem. Chem. Phys., 2017, 19(1), 210–218 RSC.
- B. K. Chatterjee, K. Bhattacharjee, A. Dey, C. K. Ghosh and K. K. Chattopadhyay, Dalton Trans., 2014, 43(21), 7930–7944 RSC.
- K. R. Sanchez-Lievanos, J. L. Stair and K. E. Knowles, Inorg. Chem., 2021, 60(7), 4291–4305 CrossRef CAS PubMed.
- H. L. Andersen, C. Granados-Miralles, K. M. Ø. Jensen, M. Saura-Múzquiz and M. Christensen, ACS Nano, 2024, 18(14), 9852–9870 CrossRef CAS.
- S. Wen, B. Chen, J. Zhang, W. Zhan, Z. He and L. Gao, Crystals, 2023, 13, 10–14 Search PubMed.
- N. Nazari, M. M. Golzan and K. Mabhouti, Sci. Rep., 2025, 15(1), 25169 CrossRef CAS PubMed.
- F. Tiss, S. Hcini, K. Khirouni, E.-K. Hlil and M. Gassoumi, Ceram. Int., 2025, 51(23), 38979–38989 CrossRef CAS.
- K. K. Jani, P. Y. Raval, N. H. Vasoya, M. Nehra, M. Singh, N. Jakhar and R. K. Singhal, Ceram. Int., 2022, 48(21), 31843–31849 CrossRef CAS.
- B. Yang, Y. Feng, X. Deng, S. Ta, D. Xiong, K. Wen and M. He, Ceram. Int., 2021, 47(2), 2107–2114 CrossRef CAS.
- S. V. Nekipelov, О. V. Petrova, A. V. Koroleva, M. G. Krzhizhanovskaya and N. A. Zhuk, Ceram. Int., 2025, 51(3), 3343–3349 CrossRef CAS.
- L. Chen, G. Ye, W. Zhou, J. Dijkmans, B. Sels, A. Malfliet and M. Guo, Ceram. Int., 2015, 41(10), 12651–12657 CrossRef CAS.
- S. A. Hosseini, A. Niaei, D. Salari, M. C. Alvarez-Galvan and J. L. G. Fierro, Ceram. Int., 2014, 40(4), 6157–6163 CrossRef CAS.
- F. A. Al-Agel, E. Al-Arfaj, A. A. Al-Ghamdi, B. D. Stein, Y. Losovyj, L. M. Bronstein and W. E. Mahmoud, Ceram. Int., 2015, 41(1), 1115–1119 CrossRef CAS.
- W. Kong, J. Wang, J. Yao and A. Chang, Ceram. Int., 2018, 44(2), 1455–1460 CrossRef CAS.
- C. Murugesan, K. Ugendar, L. Okrasa, J. Shen and G. Chandrasekaran, Ceram. Int., 2021, 47(2), 1672–1685 CrossRef CAS.
- Z. Peng, X. Fu, H. Ge, Z. Fu, C. Wang, L. Qi and H. Miao, J. Magn. Magn. Mater., 2011, 323(20), 2513–2518 CrossRef CAS.
- S. M. Seyedian, A. Ghaffari, A. Mirkhan, G. Ji, S. Tan, S. Ghorbanian-Gezaforodi and R. Peymanfar, Ceram. Int., 2024, 50(8), 13447–13458 CrossRef CAS.
- Q. Ni, L. Sun, E. Cao, W. Hao, Y. Zhang and L. Ju, Ceram. Int., 2020, 46(7), 9722–9728 CrossRef CAS.
- A. H. Cahyana, A. R. Liandi, Y. Yulizar, Y. Romdoni and T. P. Wendari, Ceram. Int., 2021, 47(15), 21373–21380 CrossRef CAS.
- M. A. Wahba, Ceram. Int., 2025, 51(4), 4329–4342 CrossRef CAS.
- H. Shokrollahi and K. Janghorban, J. Mater. Process. Technol., 2007, 189(1), 11–12 Search PubMed.
- M. Kaiser, Appl. Phys. A, 2023, 129(12), 840 CrossRef CAS.
- F. A. Alfehaid, R. Charguia, S. Hcini, K. Khirouni and M. L. Bouazizi, Opt. Mater., 2024, 154, 115642 CrossRef CAS.
- F. Fakhry, E. Shaheen, H. El-Dosoky, T. M. Meaz, M. Mubark and R. El-Shater, Sci. Rep., 2024, 14(1), 29407 CrossRef CAS PubMed.
- M. L. Bouazizi, S. Hcini, K. Khirouni, M. H. Dhaou, F. Najar and A. H. Alshehri, J. Electron. Mater., 2023, 52(4), 2878–2893 CrossRef CAS.
- N. F. Mott, Philos. Mag., 1971, 24(190), 935–958 CrossRef CAS.
- N. F. Mott, J. Non-Cryst. Solids, 1978, 28(2), 147–158 CrossRef CAS.
- N. Ponpandian, P. Balaya and A. Narayanasamy, J. Phys. Condens. Matter, 2022, 14(12), 3221 CrossRef.
- E. Stefan, P. A. Connor, A. K. Azad and J. T. S. Irvine, J. Mater. Chem. A, 2014, 2, 18106–18114 RSC.
- Y. Moualhi and H. Rahmouni, Mater. Chem. Phys., 2025, 332, 130185 CrossRef CAS.
- A. Ghodhbani, Y. Moualhi, W. Dimassi, R. M’nassri, H. Rahmouni and K. Khirouni, Phys. Rev. B Condens. Matter, 2024, 683, 415964 CrossRef CAS.
- C. Yuan, X. Liu, M. Liang, C. Zhou and H. Wang, Sens. Actuators, A Phys., 2011, 167(2), 291–296 CrossRef CAS.
- Z. P. Nenova and T. G. Nenov, IEEE Trans. Instrum. Meas., 2009, 58(2), 441–449 Search PubMed.
- G. M. Gouda and C. L. Nagendra, Sens. Actuators, A Phys., 2009, 155(2), 263–271 CrossRef CAS.
- Y. Q. Gao, Z. M. Huang, Y. Hou, J. Wu, W. Zhou, C. OuYang and J. H. Chu, Mater. Sci. Eng. B, 2014, 185, 74–78 CrossRef CAS.
- S. Aleksić, N. Obradović, N. S. Mitrović and M. D. Luković, Sci. Sinter., 2024, 56(3), 299–308 CrossRef.
- J. E. Kang, J. Ryu, G. Han, J. J. Choi, X. H. Yoon, B. D. Hahn and D. S. J. Park, J. Alloys Compd., 2012, 534, 70–73 CrossRef CAS.
- L. Wu, J. Wang, G. Lan, W. Li, P. Meng and S. Jia, J. Alloys Compd., 2025, 1011, 178396 CrossRef CAS.
- C. A. Hall, P. Ferrer, D. C. Grinter, S. Kumar, I. da Silva, J. Rubio-Zuazo and R. Grau-Crespo, J. Mater. Chem. A, 2024, 12(43), 29645–29656 RSC.
- M. Baričić, P. Maltoni, G. Barucca, N. Yaacoub, A. Omelyanchik, F. Canepa and D. Peddis, Phys. Chem. Chem. Phys., 2024, 26(7), 6325–6334 RSC.
- P. Dolcet, S. Diodati, F. Zorzi, P. Voepel, C. Seitz, B. M. Smarsly and S. Gross, Green Chem, Green Chem., 2018, 20(10), 2257–2268 RSC.
- N. Guijarro, P. Bornoz, M. Prévot, X. Yu, X. Zhu, M. Johnson and K. Sivula, Sustain. Energy Fuels, 2018, 2(1), 103–117 RSC.
- M. D. Hossain, A. T. M. K. Jamil, M. S. Hossain, S. J. Ahmed, H. N. Das, R. Rashid and M. N. I. Khan, RSC Adv., 2022, 12(8), 4656–4671 RSC.
| This journal is © The Royal Society of Chemistry 2026 |






