Open Access Article
Open Access ArticleA comprehensive review of the medicinal properties of Terminalia arjuna and its major constituent arjunolic acid: a natural product source for new drug leads
Manohar Bhujel a,
Sai Giridhar Sarma Kandanur
a,
Sai Giridhar Sarma Kandanur b,
Lakshminath Sripada
b,
Lakshminath Sripada a and
Nageswara Rao Golakoti
a and
Nageswara Rao Golakoti *a
*a
aDepartment of Chemistry, Sri Sathya Sai Institute of Higher Learning, Prasanthi Nilayam, Puttaparthi, A.P. 515134, India. E-mail: gnageswararao@sssihl.edu.in
bIndependent Researcher, Hyderabad-500094, Telangana, India
First published on 5th May 2026
Abstract
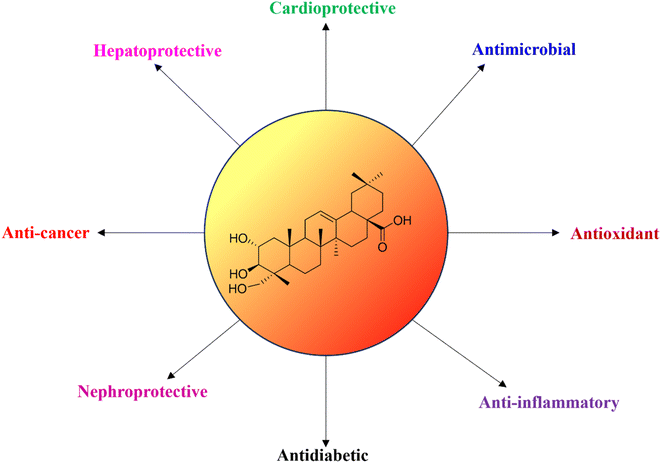
Plants have long been a primary source of natural products for medicine. Terminalia arjuna, a deciduous tree native to the Indian subcontinent, is renowned for its diverse biological properties, such as anti-cancer, anti-diabetic, and cardioprotective effects. Various parts of this tree have been used to treat numerous ailments such as cancer, diabetes, inflammation, and microbial infections, as mentioned in ancient medicinal texts such as Ayurveda and Charaka Samhita. Arjunolic acid, a pentacyclic triterpene, is a major constituent of T. arjuna, and exhibits a plethora of biological properties such as anti-cancer, anti-diabetic, anti-inflammatory, and antioxidant activities. Diverse research groups have utilized arjunolic acid as a template for synthesizing various semi-synthetic derivatives, which have been evaluated for their anti-cancer, anti-diabetic, and antioxidant properties. This review comprehensively compiles and discusses the literature related to T. arjuna, arjunolic acid and its derivatives, focusing on their medicinal properties and the mechanism of their biological activities.
1. Introduction
Natural products, classified into alkaloids, carbohydrates, flavonoids, lipids, steroids, terpenes, etc., have been a rich and consistent source of biologically important and chemically diverse molecules.1,2 In view of the recent surge in FDA approvals for natural-product-based drug leads,3,4 terpenes present a promising opportunity to identify new drug candidates, as they are the largest class among natural products. The major pentacyclic triterpene constituent of T. arjuna, arjunolic acid, has received widespread attention from various research groups5,6 for its diverse biological properties. The structural feasibility of arjunolic acid to undergo various semi-synthetic transformations has resulted in analogues with interesting in vitro and in vivo biological activity.7–9 Although efforts have been made to review the literature on T. arjuna10–16 and arjunolic acid,5,6,17 the present review aims to comprehensively compile and discuss the following areas: (a) T. arjuna and its medicinal properties, (b) the isolation of arjunolic acid from various sources and its biological activities, and (c) various semi-synthetic derivatives of arjunolic acid, along with their biological properties and action mechanisms, reported to date.1.1 Morphology of Terminalia arjuna (T. arjuna)
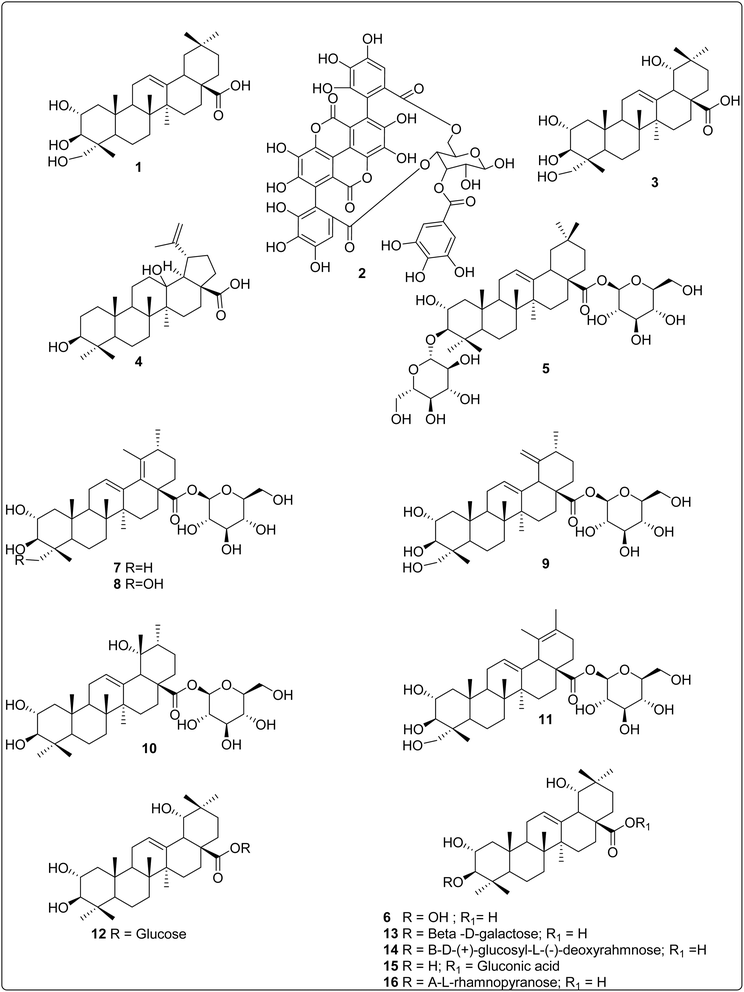
Terminalia arjuna is a species in the genus Terminalia of the family Combretaceae, which includes approximately 20 genera and 600 species.18 The genus name Terminalia refers to the terminal arrangement of leaves on many species in this group. T. arjuna is found throughout the sub-Himalayan regions of Bihar, Delhi, Madhya Pradesh, Uttar Pradesh, and the Deccan Plateau in India, as well as in the forests of Sri Lanka, Bangladesh, Mauritius, and Myanmar.19,20 It is a deciduous tree with buttressed roots, a sturdy trunk, and horizontally spreading branches and can reach heights of 60–70 feet (Fig. 1). The bark has a single-layered epidermis with hair-like projections, along with periderm and secondary phloem in mature bark. Its bark is usually white-grey, turning pink depending on the season and age, and it flakes off in large, flat pieces.21The leaves are conical, oblong or elliptic, typically 10–15 cm long and 4–7 cm wide, with a green or dark-green upper surface and a light-brown underside. The petioles have one or two prominent glands at the top.21,22 The flowers are pale yellow to milky white, bisexual, and grow in dense clusters at the branch tips or as axillary spikes. Each flower has 10 stamens and an ovary covered with reddish or yellow hairs, while the calyx is glabrous. The woody fruit is 2.5–3.5 cm long with five angled wings that curve upward. T. arjuna flowers from March to June, with fruit appearing between September and November.21 T. arjuna contains many phytochemicals that have been reported to exhibit biological properties (Table 1 and Fig. 2). Among them, arjunolic acid is the major constituent, and this compound has been the focus of diverse research groups as it was reported to exhibit a plethora of biological properties.5,6
| Part | Phytochemicals | Medicinal properties |
|---|---|---|
| Stem bark | Arjunolic acid (1)23,24 | Cardioprotective, anti-cancer7,25 |
| Arjunin (2)23,24,26 | Anti-cancer27 | |
| Arjungenin (3)23,24 | Cytotoxic28 | |
| Terminic acid (4)29 | — | |
| Terminolitin (5)23,24 | — | |
| Arjunic acid (6)26 | Antioxidant30 | |
| 2α,3β-Dihydroxyurs-12,18-dien-28-oic acid- 28-O-β-D-glucopyranosyl ester (7)31 | — | |
| 2α,3β,23-Trihydroxyurs-12,18-dien-28-oic acid- 28-O-β-D-glucopyranosyl ester (8)31 | — | |
| Quadranoside VIII (9)31 | — | |
| Kajiichigoside F1 (10)31 | — | |
| 2α,3β,23-Trihydroxyurs-12,19-dien-28-oic acid-28-O-β-D-glucopyranosyl ester (11)31 | — | |
| Arjunetin (12)23,24 | Anti-viral,32 antioxidant33 | |
| Arjunoside I (13), arjunoside II (14), arjunoside III(15), arjunoside IV (16)34,35 | — | |
| Arjunolone (17)36 | — | |
| Arjunolitin (18)37 | — | |
| Arjunaphthanoloside (19)38,39 | — | |
| Arjunoglycoside I (20), arjunoglycoside II (21), arjunoglycoside III (22), arjunoglycoside IV (23), arjunoglycoside V (24)40,41 | — | |
| Olean-3β,22β-diol-12-en-28-oic acid-28-β-D-glucopyranoside (25)42 | — | |
| Terminarjunoside I (26) and terminarjunoside II (27)43 | — | |
| Terminoside A (28)44 | Inhibits NO production45 | |
| Terminoic acid (29)44 | — | |
| Arjunone (30)36 | — | |
| Luteolin (31)36 | Antioxidant, antimicrobial, anticancer46 | |
| Baicalein (32)36 | Anticancer, antimicrobial, anti-biofilm47 | |
| Ethyl gallate (33)36 | ||
| Gallic acid (34)36 | Antioxidant48–50 | |
| Kempferol (35)36 | Anti-cancer, anti-inflammatory51,52 | |
| Oligomeric proanthocyanidin (36)36 | — | |
| Pelargonidin (37)36 | — | |
| Quercetin (38)36 | Antimicrobial, anticancer47 | |
| (+) catechin (39), (+) gallocatechin (40) and (−) epigallocatechin (41)53 | Antioxidant54 | |
| Gallic acid (34), ellagic acid (42)53 | Antioxidant, hepatoprotective.48–50 | |
| 3-O-Methyl-ellagic acid-4-O-β-D-xylopyranoside (43)53 | — | |
| 3-O-Methyl ellagic acid-3-O-β-D-rhamnoside (44)53 | — | |
| 3-O-Methyl ellagic acid-4′-O-α-L-rhamnopyranoside (45)31 | — | |
| (−)-Epicatechin (46)31 | Antioxidant54 | |
| Pyrocatechol (47)55 | Antioxidant54 | |
| Punicallin (48)56 | Anti-cancer57 | |
| Castalagin (49)58 | Antibacterial, antiviral59 | |
| Casuariin (50)58 | Anti-cancer, antioxidant60,61 | |
| Casuarinin (51)58 | Anti-cancer, anti-inflammatory60,61 | |
| Punicalagin (52)58 | Antioxidant57 | |
| Terchebulin (53)58 | Antibacterial62 | |
| Terflavin C (54)58 | Antimicrobial63 | |
| β-Sitosterol (55)29 | Antimicrobial64 | |
| Roots | Arjunoside I–IV (13–16)29 | |
| Arjunolic acid (1)29 | Cardioprotective, anti-cancer7,25 | |
| Oleanolic acid (56)29 | Antimicrobial, anti-cancer65 | |
| Terminic acid (4)29 | — | |
| 2α,19α-Dihydroxy-3oxo-olean-12-en-28-oic acid −28-O-β-D-glucopyranoside (57)66 | — | |
| Arjunic acid (6)23,24 | Antioxidant30 | |
| Arjunetoside (3-O-β-D-glucopyranosyl-2α,3β,19α-trihydroxyolean-12-en-28-oic acid-28-O-β-D-glucopyranoside) (58)67 | — | |
| Fruits | Arjunic acid (6)68 | Antioxidant30 |
| Arjunone (30)68 | — | |
| Arachidic acid (59) and stearic acid (60)68 | Anti-inflammatory69 | |
| Cerasidin (61)68 | — | |
| Ellagic acid (42)68 | Antioxidant.48–50 | |
| Friedelin (62)68 | Anti-inflammatory, antibacterial70 | |
| Gallic acid (34)68 | Antioxidant48–50 | |
| Hentriacontane (63)68 | — | |
| Methyl oleate (64)68 | — | |
| Myristyl oleate (65)68 | — | |
| β-Sitosterol (55)68 | Anti-inflammatory64 | |
| Leaves | Gallic acid (34)71 | Antioxidant48–50 |
| Luteolin (31)71 | Antimicrobial, anticancer.46 | |
| Quercetin (38)71 | Antimicrobial, anticancer47 | |
| Catechin (39)71 | Antioxidant54 | |
| Epicatechin (46)71 | Antioxidant54 | |
| Apigenin (66)71 | Antibacterial, anticancer72 |
Apart from the major constituent arjunolic acid, many phytochemicals, as described in Fig. 2, have been reported to exhibit biological activities. This further affirms that T. arjuna has been a source of bioactive molecules, and it would be interesting to explore the potential of these other phytochemicals in various drug discovery programs.
1.2 Uses of T. arjuna in traditional medicine
T. arjuna has been valued since the Vedic era, referred to as Arjun in Ayurvedic texts. It appears in ancient medicinal literature, including the Astang Hridayam, Charaka Samhita, and Sushruta Samhita. The Ayurvedic scholar Vagbhattacharya was the first to mention the use of bark powder prepared from its stem for heart-related ailments, a use later supported by Chakradattam and Bhavasisram. In Ayurveda, T. arjuna is considered a cardiotonic and is used to treat conditions such as kshata (wounds), kshaya (emaciation), visham (poison), raktavikara (styptic), medoroga (diabetes), prameha (urinary disorders), and vrana (ulcers). It is also known to relieve painful urination in diabetic conditions. A decoction of T. arjuna bark in conjunction with white sandalwood is used to boost sperm count in men and to relieve menstrual pain and leukorrhea in women. It is applied for skin ailments, fever, and general vitality enhancement.73Root paste is applied to the forehead to alleviate headaches. Leaf paste mixed with milk and sugar is given to treat spermatorrhoea, while water-soaked leaves are used for dyspepsia and burning sensations. In traditional veterinary medicine, leaves combined with Syzygium cumini and Acacia catechu are used to treat cattle diarrhoea.74,75 Healers in Tamil Nadu's Kanchipuram district use fruit paste topically for wounds and inhale boiled bark for headaches and toothache relief.76 The Malamalasar tribe in Parambikulam uses fresh leaf juice for earache, while the powdered bark is employed for heart issues.77 In Madhya Pradesh's Surguja district, bark is used for treating fever and hypertension. In the Malkangiri district of Orissa, fresh bark is chewed as an antacid.78 Rajasthan communities combine bark powder with ghee and milk to improve heart health.79
Throughout India, pastes of the bark are applied to wounds for faster healing and to stop bleeding. The bark decoction is useful for treating asthma, dysentery, ulcers, and acne (when mixed with honey).80 Bark ash is used to treat snakebites and scorpion stings, while a bark paste is applied to the area of a fracture for quicker recovery and to reduce swelling from injuries.77,81 It is also employed for gleet, urinary issues, as an expectorant, aphrodisiac, tonic, and diuretic.82
1.3 Medicinal properties of T. arjuna
The herbal formulation “Lipistat,” which includes the bark of the arjuna tree, reduced the loss of myocardial high-energy phosphate stores and lactate accumulation in myocardial tissue in cases of isoproterenol-induced myocardial necrosis, also offering protection in ischemic heart disease. Additionally, T. arjuna bark extract improved symptoms and signs in New York Heart Association (NYHA) class patients, reducing left ventricular end-diastolic and end-systolic volume indices, while increasing left ventricular ejection fractions. Long-term treatment and evaluation of NYHA class patients showed improved symptoms, signs, and exercise tolerance.86 It was observed that the ethanolic extract of the bark reduced total cholesterol, HDL cholesterol, triglyceride levels, LDL cholesterol, and LDL/HDL ratios, without impacting renal or hepatic function.87
The hypolipidemic activity of T. arjuna was evaluated in experimentally induced atherosclerosis in rabbits fed on a cholesterol-rich diet, where it acted as a hypolipidemic agent, partially inhibiting atheroma formation and indicating an antiatherogenic role.88 A study on “Hartone,” an arjuna-based herbal formulation, found that 80% of patients receiving Hartone treatment experienced relief from stable angina pectoris compared to those on isosorbide mononitrate (ISMN), with a higher reduction in heart attack rates than ISMN. This formulation also improved blood pressure response to stress without disturbing renal or hepatic function.89
In rabbits, oral administration of a suspension of bark powder of T. arjuna for 12 weeks increased myocardial antioxidants, superoxide dismutase, catalase, and glutathione levels, while inducing heat-shock protein-72. In cases of ischemic reperfusion injury, oxidative stress and heart tissue injury were prevented in bark powder-administered rabbit hearts. In these treated rabbits, left ventricular end-diastolic pressure normalized within 10 minutes of reperfusion, and myocardial lipid peroxidation levels were lower.90 Bark powder treatment significantly reduced total cholesterol and LDL cholesterol levels and decreased lipid peroxide levels.91 The bark powder of T. arjuna reduced ischemic mitral regurgitation (IMR), improved the E/A ratio (indicating early and late phases of ventricular filling), and reduced angina frequency.90 In acute myocardial infarction, a 500 mg oral dose of T. arjuna bark powder administered for three months decreased ischemic mitral regurgitation and significantly reduced angina frequency.92
Another study based on the hypothesis that the aqueous stem bark extract of T. arjuna (TAE) could inhibit IL-18-induced atherosclerosis through the NF-κB/PPAR-γ-mediated pathway in ApoE-/mice was conducted. Twelve-week-old male ApoE−/− mice, fed with a standard chow diet, were divided into four groups (n = 6 per group): Group I (control): Phosphate-buffered saline (PBS) for 2 months. Group II: Recombinant IL-18 (rIL-18) for 1 month, followed by PBS for 1 month. Group III: rIL-18 for 1 month, followed by TAE for 1 month. Group IV: rIL-18 for 1 month, followed by atorvastatin for 1 month. Treatment with IL-18 significantly elevated pro-inflammatory IL-18 levels (1178.66 ± 8.08 pg mL−1 vs. 170 ± 9.16 pg mL−1 in controls, p < 0.001) and downregulated the cholesterol efflux gene PPAR-γ (0.6-fold vs. 1.00 in control mice). Administration of TAE significantly reduced IL-18 levels (281.66 ± 9.60 pg mL−1 vs. 1178.66 ± 8.08 pg mL−1) and upregulated PPAR-γ expression (1.5-fold vs. 0.6-fold in the IL-18-treated group). TAE also decreased atherogenic lipids and reduced the percentage of atherosclerotic lesion area, showing comparable efficacy to atorvastatin. The findings suggest that TAE mitigates IL-18-induced atherosclerosis via the NF-κB/PPAR-γ-mediated pathway, highlighting its potential as a therapeutic agent.93
Another study investigated the therapeutic and prophylactic potential of T. arjuna bark extract in a rat model of isoproterenol (ISO)-induced chronic heart failure (CHF). Rats were administered isoproterenol (ISO) at a dose of 85 mg kg−1 subcutaneously, twice at an interval of 24 hours, to induce CHF. Fifteen days post-injection, the rats exhibited reduced cardiac function, including declines in the maximal rate of rise and fall of left ventricular pressure (LV (dP/dt) max and LV (dP/dt) min), cardiac contractility index (LV (dP/dt) max/LVP), and cardiac output. Additionally, an increase in left ventricular end-diastolic pressure was observed. The CHF rats also showed significantly elevated serum levels of creatine kinase isoenzyme-MB (CK-MB) and malondialdehyde (MDA), along with reduced activities of antioxidant enzymes, such as superoxide dismutase (SOD), and reduced glutathione (GSH). Alterations in lipid profiles, elevated levels of the pro-inflammatory cytokine tumor necrosis factor-alpha (TNF-α), and histological heart damage were also evident. Treatment with T. arjuna bark extract (500 mg kg−1, orally) both before and for 15 days after ISO administration significantly improved cardiac function and reduced myocardial injury. The cardioprotective effects of T. arjuna were comparable to those of fluvastatin, a synthetic drug. The bark extract effectively attenuated cardiac dysfunction and myocardial damage by restoring endogenous antioxidant enzyme activity, reducing lipid peroxidation, and lowering cytokine levels.94
An alcoholic bark extract at a dose of 0.75 mg kg−1 body weight normalized in vivo myocardial injury in rats. In a double-blind, placebo-controlled study on chronic stable angina patients, T. arjuna bark extract improved treadmill and clinical exercise parameters compared to placebo, yielding results similar to isosorbide mononitrate therapy.95 A 6 mg kg−1 dose of a 70% alcoholic extract of the bark produced a hypotensive effect in anesthetized dogs, which was blocked by propranolol, suggesting that active constituents in the extract may have adrenergic beta-2 receptor potential, directly affecting heart muscle and supporting its cardiovascular use in Ayurveda.73
Dilated cardiomyopathy with reduced left ventricular ejection fraction (LVEF), whether due to idiopathic or ischemic causes, showed significant improvement in left ventricular parameters and functional capacity with standard therapy supplemented by 50% ethanol extract of T. arjuna bark. In diabetic rats, 50% ethanol bark extract significantly mitigated cardiac dysfunction and myocardial injury, reducing oxidative stress, endothelin-1 (ET-1), and inflammatory cytokine levels. However, the decreased body weight, heart rate, blood pressure, and elevated blood sugar levels in diabetic rats did not improve with 50% ethanol bark extract therapy. These findings suggest that T. arjuna bark extract may enhance altered myocardial function in diabetic rats, possibly by maintaining endogenous antioxidant enzyme activities and reducing ET-1 and cytokine levels.96
Shreya et al. studied the bark extract of T. arjuna for its anti-microbial properties. Methanolic extracts of bark exhibited a stronger inhibitory effect against Gram-negative bacteria compared to Gram-positive bacteria.107
The ethanolic bark extract inhibited Vibrio cholera growth at all concentrations, with zone diameters increasing proportionally to concentration. Regression coefficients for the relationship between concentration and zone diameter ranged from 0.75 to 0.984 for most isolates, suggesting a linear relationship.108
Methanolic and water extracts of T. arjuna bark showed significant inhibition zones against 22 tested bacteria, including eight uropathogens, with minimum inhibitory concentration (MIC) values between 0.16 and 2.56 mg mL−1. The chloroform extract showed no antibacterial activity. The aqueous extract of T. arjuna bark also demonstrated strong antifungal effects against eight Candida species, with MIC values ranging from 0.16 to 0.64 mg mL−1. The high antimicrobial efficacy of polar extracts correlated with their elevated polyphenol content, in contrast to the non-polar chloroform fraction.109
Aqueous extracts of Terminalia arjuna (fruit, bark, root, and leaves) demonstrated notable antibacterial properties. The bark extract effectively inhibited the growth of both Gram-positive and Gram-negative bacteria. In contrast, the inhibitory effect of the leaf extract varied depending on the microorganism. Aqueous extracts from the leaves and fruits showed greater activity against Gram-negative bacteria compared to Gram-positive bacteria. Among the microorganisms tested, the highest inhibition was observed in Escherichia coli, followed by Pseudomonas aeruginosa, Bacillus cereus, Bacillus subtilis, and Lactobacillus bulgaricus. However, Micrococcus glutamicus and Micrococcus luteus exhibited lower sensitivity to the bark extract. Overall, the bark extract showed the strongest antibacterial activity across most strains, except for M. glutamicus. However, leaf and fruit extracts were not active toward any of the Gram-positive strains used in the study.110
Meena et al. evaluated the antimicrobial efficacy of sequentially fractionated solvent extracts from T. arjuna (Roxb) against key pathogens in aquaculture. The study examined solvent extracts from the bark, fruit, and leaves of T. arjuna against 17 bacterial isolates from various species and the fungus Aphanomyces invadans. The minimum inhibitory concentration (MIC) for the bacterial isolates ranged from 0 to 25 µg mL−1, with an average of 11.39 ± 0.48 µg mL−1. Among the extracts, the ethanolic bark extract demonstrated the largest inhibition zone (20.83 ± 0.10 mm), followed by the ethanolic fruit extract (19.50 ± 0.06 mm) against Edwardsiella tarda. The solvent extracts showed efficacy at concentrations of 0.5 mg mL−1 and 1 mg mL−1 for bacterial isolates and fungal strains, respectively. The chemical analysis identified prominent bioactive compounds, including ellagic acid (m/z 302.01), 18-glycyrrhetinic acid (m/z 470.34), azelaic acid (m/z 188.10), and caffeic acid (m/z 180.04), with a fit of over 80% in mzCloud analysis. Additionally, naringenin (m/z 272.06) was detected in the acetone bark extract, while kanamycin (m/z 426.35) and tetracycline (m/z 444.415) were present in the ethanol bark extract. Orientin (m/z 448.10) and phloretin (m/z 274.08) were identified in the leaf extracts. Results show that the ethanolic bark extract exhibited the most significant antimicrobial activity, followed by the methanolic bark extract.111
The stem bark alcoholic extract of T. arjuna (ALTA) was also tested for antioxidant and antimutagenic (anticlastogenic) activities. ALTA displayed potent antioxidant properties with EC50 values of 2.491 ± 0.160 µM, 50.110 ± 0.150 µM, and 71.00 ± 0.250 µM in DPPH, lipid peroxidation, and superoxide radical scavenging assays, respectively, comparable to ascorbic acid (EC50 of 2.471 ± 0.140, 40.500 ± 0.390, and 63.00 ± 0.360). In a micronucleus test, ALTA (100 and 200 mg kg−1, orally) significantly reduced the percentage of micronuclei in both normochromatic erythrocytes (NCE) and polychromatic erythrocytes (PCE) and showed a significant reduction in the P/N ratio. These findings suggest that ALTA has notable antioxidant and antimutagenic effects.113
The free-radical scavenging capacity and antioxidant activity of bark extracts of T. arjuna (aqueous![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ethanol (20
ethanol (20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 80 v/v) and aqueous
80 v/v) and aqueous![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) methanol (20
methanol (20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 80 v/v)) were also evaluated, with extraction yields ranging from 6.66–19.09 g/100 g (w/w) on a dry-weight basis. The extracts contained substantial amounts of total phenolic content (TPC) at 6.02–11.00 g/100 g (as gallic acid equivalent) and total flavonoid content (TFC) at 1.75–5.96 g/100 g (as catechin equivalent). DPPH radical-scavenging activity had IC50 values of 2.71–7.68 µg mL−1, inhibition of peroxidation ranged from 64.79 to 71.43%, and reducing power ranged from 0.001 to 1.584 mg mL−1. These results indicate that T. arjuna bark extracts are a naturally rich source of antioxidants.114 An in vitro model using goat red blood cells subjected to Cu2+-ascorbate-induced oxidative stress demonstrated that aqueous T. arjuna bark extract reduced lipid peroxidation, increased the reduced glutathione levels, and decreased protein carbonyl content in treated RBCs. The aqueous extract also protected the activity of antioxidant enzymes catalase and superoxide dismutase (SOD) and exhibited scavenging effects on hydroxyl and superoxide anion radicals.115 The effects of ethanolic extract (at doses of 250 and 500 mg kg−1 body weight) of T. arjuna stem bark were evaluated in alloxan-induced diabetic rats, specifically examining lipid peroxidation and enzymatic and non-enzymatic antioxidant activity in liver and kidney tissues. The extract significantly (P < 0.05) reduced lipid peroxidation (LPO), with the 500 mg kg−1 dose showing a greater effect than the 250 mg kg−1 dose. Additionally, the extract significantly (P < 0.05) increased levels of superoxide dismutase, catalase, glutathione peroxidase, glutathione-S-transferase, glutathione reductase, glucose-6-phosphate dehydrogenase, reduced glutathione, vitamins A, C, and E, as well as total sulfhydryl groups (TSH) and non-protein sulfhydryl groups (NPSH) in the liver and kidney of diabetic rats. These findings suggest that the extract exhibits antioxidant activity by mitigating oxidative stress, supporting the traditional use of this plant in managing diabetes.116
80 v/v)) were also evaluated, with extraction yields ranging from 6.66–19.09 g/100 g (w/w) on a dry-weight basis. The extracts contained substantial amounts of total phenolic content (TPC) at 6.02–11.00 g/100 g (as gallic acid equivalent) and total flavonoid content (TFC) at 1.75–5.96 g/100 g (as catechin equivalent). DPPH radical-scavenging activity had IC50 values of 2.71–7.68 µg mL−1, inhibition of peroxidation ranged from 64.79 to 71.43%, and reducing power ranged from 0.001 to 1.584 mg mL−1. These results indicate that T. arjuna bark extracts are a naturally rich source of antioxidants.114 An in vitro model using goat red blood cells subjected to Cu2+-ascorbate-induced oxidative stress demonstrated that aqueous T. arjuna bark extract reduced lipid peroxidation, increased the reduced glutathione levels, and decreased protein carbonyl content in treated RBCs. The aqueous extract also protected the activity of antioxidant enzymes catalase and superoxide dismutase (SOD) and exhibited scavenging effects on hydroxyl and superoxide anion radicals.115 The effects of ethanolic extract (at doses of 250 and 500 mg kg−1 body weight) of T. arjuna stem bark were evaluated in alloxan-induced diabetic rats, specifically examining lipid peroxidation and enzymatic and non-enzymatic antioxidant activity in liver and kidney tissues. The extract significantly (P < 0.05) reduced lipid peroxidation (LPO), with the 500 mg kg−1 dose showing a greater effect than the 250 mg kg−1 dose. Additionally, the extract significantly (P < 0.05) increased levels of superoxide dismutase, catalase, glutathione peroxidase, glutathione-S-transferase, glutathione reductase, glucose-6-phosphate dehydrogenase, reduced glutathione, vitamins A, C, and E, as well as total sulfhydryl groups (TSH) and non-protein sulfhydryl groups (NPSH) in the liver and kidney of diabetic rats. These findings suggest that the extract exhibits antioxidant activity by mitigating oxidative stress, supporting the traditional use of this plant in managing diabetes.116
The aqueous extract of T. arjuna bark demonstrated protective effects on liver and kidney tissues against CCl4-induced oxidative stress.117 Methanol and ethanol extracts of T. arjuna seeds showed high antioxidant activity by inhibiting DPPH (88% ± 1.52% and 87% ± 0.057% inhibition, respectively), which was significant, although slightly lower than that of standard ascorbic acid (93.73% ± 1.12%).118 Alcoholic extracts of the bark exhibited notable radical-scavenging activity against DPPH and nitric oxide radicals.119
In another study, oral administration of CCl4 at a dose of 1 mL kg−1 body weight for 2 days significantly reduced the activities of antioxidant enzymes—catalase (CAT), glutathione-S-transferase (GST), and superoxide dismutase (SOD)—and depleted reduced glutathione (GSH) levels in cardiac tissue. Lipid peroxidation and oxidized glutathione (GSSG) levels were increased under these conditions. However, oral administration of the active constituents of T. arjuna (50 mg kg−1 body weight) for 7 days before CCl4 exposure significantly restored antioxidant enzyme activities, increased GSH levels, and reduced lipid peroxidation products. The FRAP assay showed that the active constituents also enhanced intracellular antioxidant activity in cardiac tissue. Histological studies further supported the cardioprotective effects of these active constituents, which were comparable to those of vitamin C, a known antioxidant.120
Shreya et al. used thin-layer chromatography to separate the flavonoid components of T. arjuna and evaluated the phytochemical composition, antimicrobial activity, antioxidant activity, and total flavonoid content. Phytochemical screening was conducted using standard and commonly available tests. Antioxidant activity and the presence of flavonoid compounds were analyzed using thin-layer chromatography (TLC). Total antioxidant capacity was determined using the 2,2-diphenyl-1-picrylhydrazyl (DPPH) colorimetric assay. The methanolic extract showed significant antioxidant activity, as evidenced by a decrease in DPPH radical absorption during the scavenging assay. Flavonoid components with antioxidant properties were present in the methanolic extract at a concentration of 199.00 mg quercetin equivalent per gram of dried extract, as determined by the colorimetric method.107
Meena et al. investigated the antioxidant, free-radical scavenging, and DNA-nicking inhibition activities of 21 solvent extracts of T. arjuna under hydrogen peroxide (H2O2) conditions. The study revealed a strong positive correlation and interaction (explaining 84.54% variation in PCA 1) between 2,2-diphenyl-1-picrylhydrazyl (DPPH), ferric reducing antioxidant power (FRAP), 2,2′-azino-bis-3-ethylbenzothiazoline-6-sulphonic acid, and nitrous oxide scavenging (%) with ethanol bark extract (EBE) and methanol fruit extract (MFE). The total phenolic content (TPC) ranged from 144.67 to 1794 µg mL−1 (gallic acid equivalent, GAE), and total flavonoid content (TFC) ranged from 2.5 to 34 µM Fe(II)/g, with EBE exhibiting the highest values. Among the solvent extracts, EBE demonstrated the greatest antioxidant capacity, followed by ELE, MBE, and DWFE. The DNA nicking inhibition analysis showed significant positive correlations (p < 0.01) for relative front, relative quantity, band intensity (%), and lane intensity (%), with an R2 value of 0.94. EBE was found to nick supercoiled, circular plasmid DNA effectively, showing comparable activity to standard antioxidants such as butylated hydroxyanisole, butylated hydroxytoluene, ascorbic acid, and gallic acid. Notably, gallic acid (m/z 170.0208) and ellagic acid (m/z 302.0063) were present in high concentrations in the extracts, with an effective concentration of 0.48 mg required for DNA nicking inhibition. This work also reports higher steroid concentrations in acetone, ethanol, and methanol bark fractions. It provides significant insights into the phytochemical composition, antioxidant potential, and DNA nicking inhibition properties of T. arjuna solvent extracts, which could aid in developing remedies for geriatric chronic diseases.121
In vitro studies suggested that methanol extracts of T. arjuna leaves and fruits had a stronger anti-inflammatory effect than the methanol extract of the bark, potentially due to high levels of saponins, tannins, and flavonoids, which may contribute to HRBC membrane stabilization and anti-inflammatory activity.123 Oral administration of T. arjuna fruit aqueous extract demonstrated an acute anti-inflammatory effect in carrageenan-induced paw oedema, along with analgesic effects in acetic acid-induced writhing and heat-induced pain tests, indicating both central and peripheral mechanisms by inhibiting the biosynthesis and/or release of pain mediators.124
Studies found that bark powder of T. arjuna in formulations such as Arjuna Ksheera Paka (AKP) and hydroalcoholic (HA) extracts exhibited significant anti-inflammatory activity (p < 0.05) in reducing paw oedema in mice. HA was more effective in the early phase of inflammation, while AKP demonstrated greater efficacy in the late phase, potentially due to the presence of milk solids in AKP.125
Pre-treatment with methanolic extracts of T. arjuna leaves (200 and 400 mg kg−1) reduced paw oedema by 38.05% and 64.95%, respectively, and decreased granulomatous tissue weight to 115.33 mg and 73.67 mg. Haematological analysis of pre-treated animals showed decreased white blood cells (WBC) and platelet counts with increased Hb and RBC levels. Methanolic leaf extract pre-treatment also reduced nitrite and lipid peroxidation (LPO) levels while increasing antioxidant enzymes, such as superoxide dismutase (SOD), glutathione synthetase (GSH), and glutathione peroxidase (GPx). Histopathology revealed a decrease in inflammatory cell accumulation with methanolic leaf extract treatment.126 Additionally, T. arjuna bark powder (400 mg kg−1, orally) significantly minimized formalin-induced paw oedema at 24 hours.127
Further evaluation of T. arjuna's anti-diabetic effects showed that its root extract has potent antidiabetic effects in alloxan-induced diabetic rats. A 50% ethanol extract of the stem bark, administered at 1.25 g kg−1 body weight for 21 days to type 2 diabetic Long-Evans rats, significantly (p < 0.05) improved glucose tolerance by the end of this study compared to controls. Fasting serum glucose levels also decreased significantly (p < 0.05) after 21 days of treatment, though no changes were observed in liver glycogen content or serum insulin levels. Additionally, T. arjuna stem bark 50% ethanolic extract showed beneficial effects on lipid profiles, significantly reducing serum total cholesterol and triglyceride levels (p < 0.01 and p < 0.001, respectively). Glibenclamide (5 mg kg−1) similarly produced a significant reduction (p < 0.01) in serum glucose in type 2 diabetic rats.132
The hydroethanolic extract of T. arjuna bark exhibited α-amylase inhibitory activity, with an IC50 of 145.90 µg mL−1 compared to the IC50 of acarbose 62.35 µg mL−1, suggesting that α-amylase inhibition might contribute to its antidiabetic effect through the synergistic action of its phytochemicals.133
T. arjuna also showed efficacy in managing diabetes with cardiovascular complications, helping to restore body weight in treated rats compared to controls. The hydroalcoholic extract of T. arjuna bark (HATAB) treatment significantly reduced hyperglycaemia compared to the diabetic ISP control (D-ISP) group, though Vildagliptin (VIL) showed a superior effect. Rats treated with both VIL and (HATAB) had a significantly lower heart-to-body weight ratio compared to the D-ISP group. Combined VIL and HATAB extract treatment reversed STZ/ISP-induced increases in CPK-MB and hs-CRP levels. Serum CPK-MB isoenzyme and hs-CRP levels were significantly lower in treated rats, indicating reduced cardiac damage. Moreover, VIL and treatment with HATAB extract significantly reduced serum DPP-4 levels in D-ISP rats, with a positive correlation between cardiac marker CPK-MB and serum DPP-4 confirmed by histopathology (VIL, r = 0.899; p < 0.01; bark, r = 0.848; p < 0.05).134
T. arjuna bark aqueous extract also stimulated insulin release alone (p < 0.001) and in combination with other modulators, though this effect required extracellular Ca2+. The extract increased intracellular calcium without affecting depolarized cells and significantly enhanced glucose uptake (P < 0.001). At higher concentrations, it inhibited both starch digestion and protein glycation (p < 0.001).135
Meenakshi et al. prepared a 50% aqueous alcohol extract of T. arjuna bark and fractionated this extract to obtain a tannin-rich fraction. The effects of topical application of this tannin-rich fraction were also assessed on rat dermal wounds in vivo. The findings showed a statistically significant increase in tensile strength in incision wounds and in epithelialization percentage in excision wounds compared to the control (p < 0.05).144
Additionally, wound-healing effects of a 50% ethanolic extract of T. arjuna bark and tannins isolated from the bark were investigated in incision and excision models following oral or topical application as a hydrogel. The results showed a statistically significant increase in tensile strength in incision wounds and a greater reduction in wound size in excision wounds compared to the control. Topical treatment with tannins was particularly effective in both models, with an increase in hydroxyproline content in the granulation tissue of excision wounds, indicating enhanced collagen turnover and accelerated wound healing.145
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DNA ratio were positively correlated with growth rates and nutrient retention (P < 0.05). The study concluded that TABP, at an optimal inclusion level of 12.3 g kg−1 feed, can effectively produce healthy, high-quality fish for human consumption, termed “Green Fish.” This research also provides a foundation for developing TABP-based medicated feeds on a larger scale, which could be tested under field conditions alone or in combination with commercial feeds.146
DNA ratio were positively correlated with growth rates and nutrient retention (P < 0.05). The study concluded that TABP, at an optimal inclusion level of 12.3 g kg−1 feed, can effectively produce healthy, high-quality fish for human consumption, termed “Green Fish.” This research also provides a foundation for developing TABP-based medicated feeds on a larger scale, which could be tested under field conditions alone or in combination with commercial feeds.146Meena et al. also assessed the impact of processing and pre-treatment on the biochemical composition, mineral profiling, and yield (%) of selected solvent extracts of T. arjuna (Arjuna). The analysis revealed that the maximum yield was obtained from the ethanolic bark extract in both individual (23.6% ± 0.026%) and serial (22.23% ± 0.017%) fractions. The yield of ethanolic and methanolic bark extracts showed no significant difference (p ≥ 0.05), whereas non-polar solvent extracts exhibited significant differences (p ≤ 0.05). Mineral profiling demonstrated substantial variation between dry powders and their solvent extracts. Fractionation increased the zinc (Zn) content in fruit extracts, with the methanolic extract recording the highest Zn level (45.29 mg L−1). An inverse relationship between ash and moisture content was observed across all solvent extracts. The maximum ash content was recorded in the bark powder, with values of 28.95% ± 0.001% (serial fraction) and 28.19% ± 0.008% (individual fraction). However, ash content did not directly correlate with mineral profiling, suggesting higher acid-insoluble ash levels in bark compared to leaves and fruit. This may contribute to the bio-efficacy of the solvent extracts.
The findings indicated that the inherent medicinal properties of T. arjuna bark can be leveraged, and its solvent extracts could be utilized in drug formulation. The study highlights the potential to enhance mineral profiling, particularly zinc—a potent neurotransmitter—which could be used to develop suitable feeds for livestock and fisheries. Furthermore, these findings provide a pathway for addressing nervous disorders in human health through dietary supplementation or therapeutic formulations.147
Pankaj et al. developed a process utilizing T. arjuna bark extract to produce herbal ghee from buffalo milk. Three types of extracts were evaluated: commercially available aqueous arjuna extract powder, laboratory-prepared aqueous extract, and alcoholic extract. Among these, ghee produced with the alcoholic extract (at a 4% fat-to-extract ratio) was found to be superior in quality. To optimize the level of the alcoholic extract to maximize phytosterol retention in ghee, samples were prepared using extract concentrations of 5%, 6%, and 7% relative to the weight of fat. No significant difference in sensory acceptability scores was observed among these levels. However, phytosterol content was highest (0.38 mg g−1 with cream as the fat source and 0.47 mg g−1 with butter) at the 7% extract level. The optimized herbal ghee was prepared using 7% alcoholic arjuna extract with the creamery butter method. Its chemical composition included 99.92% fat, 0.08% moisture, 0.22% free fatty acid (as oleic acid), a butyro-refractometer reading of 41.5 at 40 °C, a Reichert-Meissl value of 31.5, and a phytosterol content of 0.39 mg g−1. This process demonstrates the potential of incorporating arjuna extract into ghee for enhanced phytosterol content and associated health benefits.148
Suman et al. developed buffalo meat rolls by incorporating extracts from T. arjuna bark and Aloe vera gel at varying levels (2%, 4%, and 6%) to determine the optimal incorporation rate and evaluate their effects on the texture profile of the products. Meat rolls containing 4% Aloe vera gel scored slightly higher in juiciness and tenderness compared to those with 2% T. arjuna bark extract. However, no significant differences were observed between these two samples in overall sensory scores, which were comparable to the control. Based on sensory evaluations, 4% Aloe vera gel and 2% T. arjuna bark extract were identified as the optimal levels for incorporation and were selected for further study. The texture attributes of the developed products, such as hardness, springiness, and cohesiveness, were comparable with those of the control samples. The study concluded that buffalo meat rolls with desirable sensory and textural properties can be successfully developed by incorporating 4% Aloe vera gel and 2% T. arjuna bark extract, enhancing the product's overall quality.149
Pravin et al. evaluated the hypolipidemic and antioxidant properties of a vanilla-chocolate dairy drink fortified with encapsulated T. arjuna bark powder (1.8%) in Wistar rats fed a high-cholesterol diet for 60 days. By the conclusion of the experimental period, a significant lowering in body weight gain was observed in rats administered with the encapsulated herb extract when compared to those on a high-cholesterol diet without supplementation. Additionally, microencapsulation of the herb resulted in a significant reduction in organ weights, particularly epididymal fat and liver. The supplemented group exhibited notable reductions in serum lipid levels, including total cholesterol, triglycerides, low-density lipoprotein (LDL), very-low-density lipoprotein (VLDL), and the atherogenic index. Encapsulation also enhanced antioxidant activity, evidenced by increased reduced glutathione levels and decreased thiobarbituric acid reactive substances (TBARS) in liver tissues and red blood cell lysates. The findings highlighted that the bioactive compounds in encapsulated T. arjuna—such as phytosterols, flavonoids, saponins, and tannins—remained stable during processing and were effectively released in the intestine. These compounds contributed to its hypolipidemic and antioxidant effects, offering therapeutic potential for managing cardiovascular diseases.150
Various groups have also formulated herbal tea with the extracts of T. arjuna, along with other herbal extracts. These products are commercially available and known to have several health benefits.151,152
In a related study by the same group, a nutritional fish feed incorporating TABP was administered to Labeo rohita at varying levels: 0% (0 g kg−1, CT), 0.5% (5 g kg−1, T1), 1% (10 g kg−1, T2), and 1.5% (15 g kg−1, T3). A comprehensive biomarker response strategy was used to assess the genomic impact of TABP inclusion. Following bacterial infection, the expression of immunogenic genes—STAT1 (signal transducer and activator of transcription 1), ISG15 (interferon-stimulated gene 15), and Mx (myxovirus resistance gene)—was elevated. Densitometry analysis revealed a dose-dependent increase in STAT1 and ISG15, with Mx reaching its highest levels at 10 g kg−1 TABP.154
Meena et al. conducted an in vitro toxicity study to validate, standardize, and supplement data on the toxicity assessment of various solvent extracts of T. arjuna using Artemia salina as a model organism. Experimental conditions were optimized, including the use of 100 W yellow light, 5% salinity, pH 8.0–8.5, and a temperature of 30 °C, over 48 hours. Nauplii were collected using a modified dropper and incubated for 24 hours under the same conditions before being observed with a 50× magnifying glass. Functional screening of solvent extracts and their respective mother solvents revealed LC50 values for hexane (118.50 ppm), ethyl acetate (101.75 ppm), chloroform (93.36 ppm), acetone (278.32 ppm), ethanol (528.78 ppm), and methanol (477.67 ppm). These values categorized the solvents as medium, medium, high, medium, low, and medium toxicity, respectively. Interestingly, the toxicity of the solvent extracts differed from their mother solvents, demonstrating the extracts' effectiveness. Universal solvents, such as DMSO and distilled water (DW), were classified as non-toxic according to Meyers' toxicity index and Clarkson's toxicity criterion. Among the solvent extracts of T. arjuna, all were toxic per Meyers' toxicity index but varied in toxicity classification under Clarkson's criterion, ranging from low to high toxicity. Principal Component Analysis (PCA) revealed strong correlations, with PCA1 and PCA2 explaining 69.46% and 19.74% of the variation, respectively. The study concluded that LC50 values could be categorized as relative LC50 (linked to the mother solvent) or absolute LC50 (actual extract potential). Relationships between relative and absolute LC50 values were linear, with their percentage fractions and inversely related to their counterparts.156
Anuradha and colleagues reported the biosynthesis of copper (CuNPs) and zinc (ZnNPs) nanoparticles using aqueous T. arjuna bark extract as a reducing and stabilizing agent. The nanoparticles, characterized using UV-visible spectroscopy, FTIR, and SEM, were found to be spherical, with CuNPs ranging from 10 to 26 nm and ZnNPs from 15 to 25 nm. XRD analysis confirmed their structure. The antibacterial activity of the CuNPs was observed to be superior to that of ZnNPs against the tested organisms, although Klebsiella pneumoniae showed higher resistance. Both nanoparticles demonstrated antihaemolytic activity, with CuNPs being more effective.157
Amol and collaborators described the synthesis of gold nanoparticles (AuNPs) using T. arjuna leaf extract, with UV-visible spectroscopy showing a characteristic peak at 530 nm. TEM analysis revealed AuNPs with sizes of between 15 and 30 nm. The synthesized nanoparticles exhibited antibacterial activity against Staphylococcus aureus, Pseudomonas aeruginosa, and Salmonella typhimurium.158
Rakhi et al. demonstrated the synthesis of AuNPs using bark extract under mild conditions at room temperature. Characterization through HRTEM, AFM, and XRD confirmed their structural properties. These nanoparticles were successfully used to catalyze the reduction of 4-nitrophenol to 4-aminophenol.159
Gopinath and his team synthesized spherical AuNPs (20–50 nm) using leaf extract and reported their characterization by UV-visible spectroscopy, FTIR, and TEM. These nanoparticles promoted mitotic cell division and pollen germination without cytotoxic effects.160
Natarajan et al. utilized ethanolic bark extract to synthesize AuNPs (20–50 nm), which were found to possess neuroprotective properties. Characterization techniques, including UV-visible spectroscopy and TEM, confirmed their stability and size. The nanoparticles demonstrated antioxidant, anticholinesterase, and anti-amyloidogenic activities, highlighting their potential for treating neurodegenerative disorders such as Alzheimer's disease.161
Akther and colleagues developed silver nanoparticles (AgNPs) through microwave-assisted green synthesis using bark extract. These nanoparticles showed significant antibacterial and anti-biofilm activity against multidrug-resistant Escherichia coli, including strains harbouring the CTXM-15 gene. Docking studies revealed strong interactions of phytochemicals with CTXM-15, inhibiting its expression.162
Koparde et al. synthesized AgNPs (40–50 nm) using bark extract and confirmed their formation through colorimetric and SEM analyses. These nanoparticles exhibited strong antibacterial activity, especially against Pseudomonas aeruginosa.163
Yallappa et al. employed a microwave-assisted method to produce AgNPs (10–15 nm) using bark extract. The synthesized nanoparticles demonstrated excellent antioxidant and antibacterial properties against bacteria and yeast.164
Sakheel and colleagues reported a simple “one-pot” synthesis of AgNPs using leaf extract, without additional stabilizers. FTIR analysis confirmed bio-capping by plant biomolecules. The nanoparticles exhibited antibacterial efficacy, making them suitable for biomedical applications such as wound treatment.165
Chabbi and collaborators synthesized palladium nanoparticles (PdNPs) using T. arjuna bark extract. Characterized by UV-visible spectroscopy and TEM, these PdNPs demonstrated catalytic efficiency in C–C coupling reactions and dye degradation.166
These findings collectively highlight the ability of T. arjuna extracts to facilitate the environmentally friendly synthesis of nanoparticles with significant applications in medicine, catalysis, and environmental remediation.
Sobia et al. developed and characterized a pH-triggered in situ gelling system using moxifloxacin HCl (MOX-HCl). The gelling system utilized gum extracted from T. arjuna bark resin, blended with sodium alginate as the gelling agent. Sterilized formulations were prepared and assessed for their physicochemical properties, microbiological activity, and eye irritation potential. The drug-loaded in situ gel appeared as a clear solution, which transformed into a gel in the presence of tear fluid. The optimized formulation was stable, therapeutically effective, non-irritating, and exhibited sustained drug release for up to 12 hours. In rabbit studies, the gel caused no irritation symptoms such as redness, inflammation, or excessive tearing compared to controls. The study concluded that MOX-HCl-loaded in situ gel could serve as an alternative to conventional eye drops, offering extended precorneal retention, enhanced corneal permeability, and improved ocular bioavailability.168
In the following section, the various isolation methods of arjunolic acid, its biological activities, and its mechanism of action are presented and thoroughly discussed.
2. Arjunolic acid
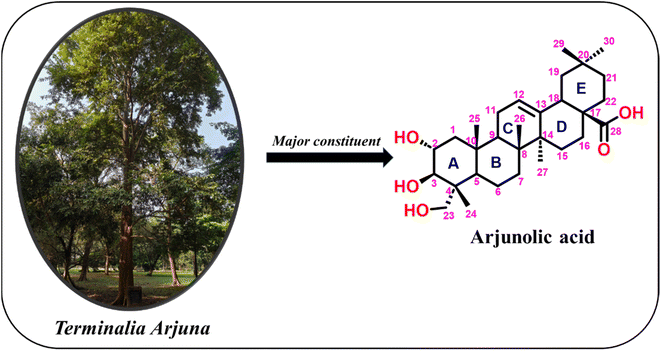
Arjunolic acid (AA) (Fig. 3) is a pentacyclic triterpene acid, a major constituent of T. arjuna with the IUPAC name 2α,3β,23α-trihydroxy-olean-12-en-28-oic acid. It is reported to exhibit a plethora of biological activities (Fig. 4). It was first isolated by King et al. from the heartwood of T. arjuna.5,6,169Although arjunolic acid is readily available in T. arjuna, it has also been isolated from various other plant sources (Table 2). Along with arjunolic acid, other known reported minor constituents (Fig. 2 and Table 1) of T. arjuna include arjunic acid, arjuntenin, baicalein, and quercetin (Fig. 2). Due to the diverse biological profile exhibited by arjunolic acid, many groups have focused their research efforts on this compound.6,17
| S. No | Source | Part | Method | Qty. of raw material used | Isolated quantity (% yield) | Ref. |
|---|---|---|---|---|---|---|
| a Mixture of arjunolic acid and asiatic acid. | ||||||
| 1 | T. arjuna | Heartwood | Column chromatography | — | — | 169 |
| 2 | T. arjuna | Stem bark | Column chromatography | 1.5 kg | 400 mg (0.04%) | 170 |
| 3 | T. arjuna | Stem bark | Column chromatography | 2 kg | 210 mg (0.01%) | 171 |
| 4 | T. arjunaa | Heartwood | Flash column chromatography | 1 kg | 15 g | 172 |
| 5 | T. arjuna | Bark | HPTLC | — | — | 173 |
| 6 | T. arjuna | Bark | HPLC-PDA | — | — | 174 |
| 7 | T. arjuna | Stem bark | Column chromatography | 1 kg | 250 mg (0.025%) | 175 |
| 8 | T. arjuna | Heartwood | Flash column chromatography | 1 kg | 11.5 g (1.15%) | 7 and 176 |
| 9 | Akebiae fructus | — | UHPLC-QTOF-MS | — | — | 177 |
| 10 | Tilia kiusiana | Leaves | Column chromatography | 187 g | 3.5 mg (0.002%) | 178 |
| 11 | T. catappa | Stem bark | MPLC | — | — | 179 |
| 12 | Combretum griffithii | Stem bark | Column chromatography | 4.1 kg | 127 mg (0.003%) | 180 |
| 13 | Lagerstroemia indica | Stem | Column chromatography | 5 kg | 2 mg (0.00004%) | 181 |
| 14 | Shuang-Huang-Lian | — | UHPLC -QTOF | — | — | 182 |
| 15 | Wuzi-Yanzong-Wan | — | UPLC-ESI-LTQ-orbitrap-MS | — | — | 183 |
| 16 | Combretum racemosum | Air dried pericarp | Column chromatography | 9 kg | 184 mg (0.002%) | 184 |
| 17 | Premna odorata | Leaves | LC–HRESIMS | — | — | 185 |
| 18 | Folium eriobotryae | — | — | — | — | 186 |
| 19 | Bixa orellana L | Root | Extraction/Separation | — | — | 187 |
| 20 | Quercus serrata Thunba | Seeds/acorn | Column chromatography | 30 kg | 8.6 mg (0.0003%) | 188 |
| 21 | Quince (Cydonia vulgaris) | Peels | Column chromatography | 3.1 kg | 7.9 mg (0.002%) | 189 |
| 22 | Phlomis umbrosa | Root | Column chromatography | 15 kg | 9.4 mg (0.006%) | 190 |
| 23 | Juglans sinensis | Leaves and twigs | Column chromatography | 20 kg | 500 mg (0.002%) | 191 |
| 24 | Cleistocalyx operculatusa | Leaves | Column chromatography | 3.3 kg | 60 mg (0.002%) | 192 |
| 25 | Stauntonia hexaphylla | Fruit | Column chromatography | — | — | 193 |
| 26 | T. albida | Root | Column chromatography | 470.9 g | 7.4 mg (0.002%) | 194 |
| 27 | Poraqueiba sericea | Stems | Column chromatography | 856 mg | 2 mg (0.23%) | 195 |
| 28 | Combretum nelsoniia | Leaves | Column chromatography | 12.38 g | 81 mg (0.65%) | 196 |
| 29 | Myrica adenophora | Root | MPLC | 7.2 kg | 110 mg (0.002%) | 197 |
| 30 | Myrcia rubella Cambess | Leaves | Extraction/HPLC | 320 g | 0.3 mg (0.0009%) | 198 |
| 31 | T. bentzoe L | Leaves | Flash column chromatography | 1.5 kg | 20 mg (0.0013%) | 199 |
| 32 | Clidemia hirta | Root | HPLC | — | — | 200 |
| 33 | Psidium guajava La | Leaves | Column chromatography | — | — | 201 |
| 34 | Mosla dianthera | Whole herb | Column chromatography | 4.4 kg | 80 mg (0.0018%) | 202 |
| 35 | Eugenia jambolana Lam | Berry/fruit pulp | Prep HPLC/column chromatography | 500 g | 8.2 mg (0.0016%) | 203 |
| 36 | Glochidion assamicum | Aerial parts | Column chromatography | 14 kg | 3 mg (0.00002%) | 204 |
| 37 | Melandrium firmum | Areial parts | Column chromatography | 10 kg | 13.5 mg (0.00013%) | 205 |
| 38 | Duabanga grandiflora L | Stem | Column chromatography | 5 kg | 162 mg (0.003%) | 206 |
| 39 | Gentiana scarba | Root and rhizomes | Column chromatography | 2.5 kg | 45.9 mg (0.0018%) | 207 |
| 40 | Cyclocarya paliurus | Leaves | HPLC-QTOF-MS | — | — | 208 |
| 41 | Lagerstroemia. guilinensis | Branches | Column chromatography | — | — | 209 |
| 42 | Melaleuca alternifolia | Bark and stem | Column chromatography | 700 g | 3.9 mg (0.0005%) | 210 |
| 43 | Combretum leprosum | Root | Column chromatography | — | — | 211 |
| 44 | Vaccinium emarginatum | Whole plant | Column chromatography | 17.4 kg | 36.4 mg (0.0002%) | 212 |
| 45 | Juglans mandshuricaa | Green walnut husk | Column chromatography | 10 kg | 5.2 mg (0.0005%) | 213 |
| 46 | Akebia trifoliata | Areial parts | Column chromatography | — | — | 214 |
| 47 | Siphoneugena densiflora | Stem | Column chromatography | 60.1 g | 12 mg (0.019%) | 215 |
| 48 | Eucalyptus globulusa | Stem bark | GC-MS | — | — | 216 |
| 49 | Osmanthus fragransa | Fruits | UHPLC-TQUAD-MS | — | — | 217 |
| 50 | Miconia trailii | Twig/leaves | Column chromatography | 370 g | 5 mg (0.0013%) | 218 |
| 51 | Psidium guajavaa | Leaves | Column chromatography | 20 kg | 3 mg (0.00001%) | 219 |
| 52 | Combretum nelsoniia | Leaves | Column chromatography | 500 g | 81 mg (0.016%) | 220 |
| 53 | Rubus sanctus and Rubus ibericus | Leaves | UHPLC-ESI/HRMS | — | — | 221 |
| 54 | Sapium haematospermum | Aerial parts | HPLC | 850 g | 3 mg (0.0003%) | 222 |
| 55 | Carissa opaca | Whole plant | Column chromatography | 10 kg | 17 mg (0.00017%) | 223 |
| 56 | Cola rostara K. Schum | Root | Column chromatography | 2.5 kg | 10 mg (0.004%) | 224 |
| 57 | Heteropyxis canescens | Stems and root | Column chromatography | 1.5 kg | 17 mg (0.001%) | 225 |
| 58 | Durio zibethinus | Fruit shell | Column chromatography | 20 kg | 5 mg (0.00002%) | 226 |
| 59 | Hawthorn berries | Berries | Column chromatography | 8 kg | 4.7 mg (0.0006%) | 227 |
| 60 | Potentilla chinensis | Whole plant | CC/MPLC | 100 g | 5.2 mg (0.05%) | 228 |
| 61 | Terminalia mollisa | Stem bark | Column chromatography | — | — | 229 |
| 62 | Myrtus communisa | Leaves | HPLC/HRMS | — | — | 230 |
| 63 | Solanum melongena L | Root | Column chromatography | 12 kg | 6.5 mg (0.005%) | 231 |
| 64 | Eucalyptus globulus Wooda | Wood | GC-MS | — | — | 232 |
| 65 | Cochlospermum tinctorium | Rhizomes | Column chromatography | 75 g | 75 mg (0.1%) | 233 |
| 66 | Symplocos lancifolia | Leaves | HPLC/Column chromatography/prep HPLC | 600 g | 17 mg (0.002%) | 234 |
| 67 | Syzygium samarangense | Areial parts | Column chromatography | — | — | 235 |
| 68 | Myrica arborea | Twigs | Column chromatography | 1 kg | 70 mg (0.007%) | 236 |
| 69 | Cecropia schreberiana | Leaves | Column chromatography | 590 g | 2 mg (0.003%) | 237 |
| 70 | Premna fulva | Stalks | Column chromatography | 238 | ||
| 71 | Rhododendron hainanense | Areial parts | Column chromatography | 1 kg | 2 mg (0.0002) | 239 |
| 72 | Campsis grandiflora K. Schum | Flower | Column chromatography | 1 kg | 33 mg (0.003) | 240 |
| 73 | Isodon yuennanensis | Rhizomes | Column chromatography | — | — | 241 |
| 74 | Momordica cochinchinensis | Seeds | Column chromatography | 50 kg | 86 mg (0.001%) | 242 |
| 75 | Rhodomyrtus tomentosa | Roots | Column chromatography | — | — | 243 |
| 76 | Averrhoa carambola | Fruit | Column chromatography | 6.8 kg | 13.3 mg (0.001%) | 244 |
| 77 | Rhododendron collettianum | Areial parts | Column chromatography | 20 kg | 15 mg (0.00007%) | 245 |
| 78 | Datisca cannabina linn | Whole plant | Column chromatography | 15 kg | 8.4 mg (0.00005%) | 246 |
| 79 | Cornus capita | Leaves | Column chromatography | 247 | ||
2.1 Isolation of arjunolic acid from T. arjuna
The limited number of literature reports on the isolation of arjunolic acid from T. arjuna encouraged us to explore novel isolation processes that could furnish arjunolic acid from T. arjuna in higher yields.176 In general, the isolation process adopted by various groups involved the extraction of the plant material with various organic solvents (ethyl acetate, acetone, etc.), followed by either chromatographic purification or a recrystallisation process to furnish the pure product.25,169,170,248,249 However, it was seen that the aforementioned isolation processes were low-yielding (milligram quantities). In another study, Bag et al.250 reported the extraction and separation of arjunolic acid and asiatic acid from the heartwood of T. arjuna by employing cumbersome chemical methods involving several synthetic transformations followed by crystallisation. However, Manohar et al.176 were able to efficiently separate arjunolic acid and asiatic acid by a simple flash chromatography method, affording the former in high purity and in large quantities (gram quantities, Table 2, Entry 8). Thus, an efficient process for the isolation of arjunolic acid from the crude plant material was developed in-house by our co-worker Manohar et al., affording arjunolic acid in high yields when compared with the isolated yields reported to date.1763. Biological activities of arjunolic acid
3.1 Cardioprotective activity
Arjunolic acid (AA) has been extensively studied for its cardioprotective effects across various in vitro and in vivo models. Research by Ghosh et al. suggests that doxorubicin-induced cardiac damage results primarily from two mechanisms: first, doxorubicin exposure generates H2O2 in myocytes, which regulates the p53 and p38-JNK signaling pathways in cardiomyocytes; second, activation of p38 and JNK promotes Bax translocation to the mitochondria, disrupting the balance between proapoptotic and antiapoptotic factors. These imbalances trigger mitochondrial permeabilization, leading to cytochrome c release, caspase activation, and cell death. Arjunolic acid supplementation (25 mg kg−1 body weight every two days for three treatments) offers protection by detoxifying reactive oxygen species and inhibiting p38 and JNK MAPKs phosphorylation, preventing Bax translocation to the mitochondria and mitochondrial permeabilization.251Sumitra et al. further demonstrated the cardioprotective effects of arjunolic acid, showing that it reduces platelet aggregation, coagulation, and myocardial necrosis. In isoproterenol-induced myocardial necrosis, arjunolic acid restored elevated serum enzyme levels (creatine kinase and lactate dehydrogenase) and normalized electrocardiographic parameters, preserving heart architecture pre- and post-isoproterenol exposure.171
Trisha et al. explored the mechanisms of arjunolic acid and identified its intracellular target in ameliorating hemodynamic load-induced cardiac fibrosis. Results show that AA binds and stabilizes the ligand-binding domain of peroxisome proliferator-activated receptor gamma (PPARγ), increasing its expression during cardiac hypertrophy. In hypertrophic samples, PPARγ knockdown reveals that AA's cardioprotective effects are primarily due to PPARγ agonism. This upregulation of PPARγ inhibits TGF-β signaling by suppressing TAK1 phosphorylation. AA-bound PPARγ interacts with TAK1, masking its kinase domain and decreasing TAK1 phosphorylation, which reduces p38 MAPK and NF-κBp65 activation, ultimately reducing excess collagen synthesis in cardiac hypertrophy.252
Prasenjit et al. studied the cardioprotective property of arjunolic acid in arsenic-induced oxidative cardiac damage. NaAsO2 exposure (10 mg kg−1 body weight) increased arsenic levels in cardiac tissue and reduced antioxidant enzyme activities (superoxide dismutase, catalase, glutathione-S-transferase, glutathione reductase, and glutathione peroxidase). Treatment with arjunolic acid (20 mg kg−1 body weight for four days before NaAsO2 exposure) protected cardiac tissues by maintaining antioxidant enzyme activities and normalizing oxidative stress markers. Additionally, arjunolic acid prevented arsenic-induced hyperlipidemia and preserved cardiac architecture, as shown in histological studies.253
Hassan et al. investigated AA's effects on TLR4 downstream signaling in response to lipopolysaccharide (LPS) treatment. LPS exposure upregulated TLR4, MyD88, MAPK, JNK, and NF-κB markers (2–6 times higher than the control), whereas AA treatment downregulated MyD88, NF-κB, p38, and JNK in H9C2 myotubes. AA also decreased TLR4 expression in both H9C2 and C2C12 myotubes, suggesting potential cardioprotective effects through modulation of TLR4 expression.254
Gayyar et al. highlighted AA's protective effects on sodium nitrite-induced cardiac toxicity. Sodium nitrite increased pro-inflammatory cytokines (TNF-α, IL-1β), C-reactive protein, and caspase activity while reducing anti-inflammatory cytokines (IL-4, IL-10) and cytochrome C oxidase levels. AA treatment reduced pro-inflammatory cytokines and apoptosis markers while increasing anti-inflammatory cytokines and antioxidant enzymes. AA also improved sodium nitrite-induced histopathological changes in cardiomyocytes, suggesting protection against both intrinsic and extrinsic cell death pathways.255
Hany et al. reported that AA attenuated LPS-induced cardiotoxicity. Pretreatment with AA reduced serum cardiac troponin I, lactate dehydrogenase, and creatine kinase levels in Swiss albino mice. AA pretreatment also increased antioxidant levels, reduced lipid peroxidation, decreased C-reactive protein, and normalized pro- and anti-inflammatory cytokine balance. These findings collectively indicate that AA offers cardioprotection through antioxidant activity, inflammation inhibition, and prevention of cardiac cell death.256
3.2 Anti-cancer activity
Arjunolic acid has demonstrated anticancer properties in various in vitro and in vivo studies. Ramesh et al. studied anti-cancer properties of arjunolic acid. Ehrlich ascites carcinoma (EAC) and Dalton's lymphoma (DAL) cell lines were incubated with different concentrations of arjunolic acid, and the results showed that a 100 µg concentration induced approximately 66% cell death in DAL cells and 70% in EAC cells, primarily through membrane disruption.257Arjunolic acid (AA) was also seen to exhibit significant anticancer activities against MCF-7 and HeLa cell lines, with minimal toxicity observed in normal lymphocytes. Studies have highlighted the self-assembly properties of AA, which contribute to its anticancer effects. Fluorescence-based analyses, such as ROS generation and EtBr/AO and DAPI staining, showed that AA at 50 µg mL−1 disrupts redox balance in cancer cells by generating reactive oxygen species. This ROS-induced apoptosis is mediated by regulatory proteins TNF-α and NF-κB, with elevated levels of pro-inflammatory cytokines indicating the involvement of a ROS-driven apoptotic pathway.258
In another study reported by Elsherbiny et al.,89 AA showed antitumor effects by enhancing cytotoxicity and apoptosis, partly by blocking TGF-βR1 and modulating inflammatory cytokine levels. In this study, Ehrlich Ascites Carcinoma (EAC) was induced in fifty female Swiss albino mice, and AA was administered at doses of 100 mg kg−1 and 250 mg kg−1. AA significantly reduced tumor volume and cell count, decreased cell viability, and increased cytotoxicity in EAC cells. Additionally, AA lowered the levels of TNF-α, IL-1β, TGF-β, TGF-β type I receptor, and latency-associated peptide, while increasing the IL-10 levels in both in vitro and in vivo conditions.259
3.3 Anti-diabetic activity
Arjunolic acid (AA) has been widely studied for its antidiabetic effects across several models. Prasenjit et al. investigated AA's prophylactic effects against streptozotocin (STZ)-induced diabetes in the pancreatic tissue of Swiss albino rats. In this study, it was observed that STZ administration (65 mg kg−1 body weight via tail vein) increased reactive oxygen and nitrogen species (ROS and RNS) in pancreatic tissue. This oxidative stress reduced intracellular antioxidant defense, heightened lipid peroxidation, protein carbonylation, serum glucose, and TNF-α levels. The signaling pathway analysis showed that STZ activated phospho-ERK1/2, phospho-p38, and NF-κB, and disrupted mitochondrial transmembrane potential, causing cytochrome c release and caspase-3 activation without significantly affecting total ERK1/2 and p38 levels. AA treatment (20 mg kg−1 body weight, orally), both before and after STZ administration, effectively mitigated these adverse effects by reducing excessive ROS and RNS formation and down-regulating phospho-ERK1/2, phospho-p38, NF-κB, and mitochondria-dependent apoptosis pathways.260AA's protective effects on hyperglycaemia-induced liver dysfunction were evaluated in experimental rats with type 1 hyperglycaemia induced by STZ. AA, administered orally at 20 mg kg−1 before and after diabetes induction, was compared to an insulin-treated positive control. Hyperglycaemia led to body weight loss, reduced serum insulin, increased HbA1C, and elevated advanced glycation end products (AGEs). Markers of liver dysfunction included raised serum alanine transaminase (ALT), alkaline phosphatase (ALP), ROS, RNS, lipid peroxidation, and oxidative DNA damage (8-OHdG/2-dG ratio) along with decreased GSH content and antioxidant defenses. Hyperglycaemia also triggered iNOS, IκBα/NF-κB, and MAPK pathways and mitochondrial signals, which promoted apoptotic cell death. Increased PARP expression, NAD and ATP depletion, and DNA fragmentation in liver tissue confirmed apoptosis. Immunofluorescence with anti-caspase-3 and anti-Apaf-1, DAPI/PI staining, DNA laddering, and FACS analysis corroborated apoptotic death. Histology supported these findings, and AA treatment reduced diabetic liver complications, apoptosis, ROS, RNS, HbA1C, AGEs, and PARP-mediated DNA damage, highlighting AA's potential in diabetic care.261
Gonçalves-Neto et al. examined whether AA could offset dexamethasone (DEX)-induced glucose imbalance in adult Wistar rats. Groups included vehicle-treated (Ctrl), DEX-treated (1 mg kg−1 body weight intraperitoneally for 5 days) (DEX), AA-treated (30 mg kg−1 by oral gavage twice daily) (AA s), AA plus DEX (AA DEX), and AA following DEX (DEX AA). AA treatment significantly improved glucose intolerance when administered before and during DEX (AA DEX) but did not ameliorate it when given after (DEX AA). AA failed to reduce the DEX-induced rise in hepatic glycogen and triacylglycerol, and prolonged AA with DEX increased hepatic steatosis. Liver function and oxidative stress markers were not notably altered among groups, suggesting that AA could partly prevent DEX-induced glucose intolerance but was less effective after DEX initiation.262
The antidiabetic potential of AA isolated from T. arjuna was tested in type 2 diabetic Sprague Dawley rats, with T2DM induced via a single STZ-nicotinamide injection. After 10 days, fasting and random blood glucose, body weight, food and water intake, serum C-peptide, insulin, and glycated haemoglobin (HbA1c) levels confirmed T2DM development. Over four weeks, AA (25 and 50 mg kg−1 day−1) significantly normalized fasting and random glucose, restored body weight, controlled excessive eating and drinking, and improved glucose tolerance. AA also reduced HbA1c, total cholesterol (TC), triglycerides (TG), and LDL levels, while raising HDL slightly. Serum and pancreatic insulin and C-peptide levels increased with AA, while GDF-15 levels were stable, and AA lowered serum and pancreatic pro-inflammatory cytokines, including TNF-α, IL-1β, and IL-6, in a dose-dependent manner.170
In another study, AA isolated from T. arjuna selectively inhibited glucagon secretion in palmitate-induced αTC1-6 cells and isolated islets, improving hyperglycaemia and pancreatic islet morphology in STZ-induced type 2 diabetic mice. AA's selective inhibition of α-cell glucagon secretion involved ephrin-A1 and EphA4 interaction, which activated the PI3K and Akt pathways, suggesting AA as a promising therapeutic agent for T2DM.263
In an STZ-induced diabetic model, AA dosed at 10 and 30 mg kg−1 orally over ten weeks was examined for its effect on retinal damage from diabetic retinopathy (DR). H2O2-induced ARPE-19 cells were used to assess anti-apoptotic and antioxidant effects. The results showed that AA reduced STZ-induced weight loss, increased retinal thickness and nuclei, and upregulated HO-1 protein levels in vitro and in vivo. AA protected retinal cells from apoptosis via the AMPK-mTOR-regulated autophagy pathway, with dorsomorphin dihydrochloride blocking AA's anti-apoptotic effects, indicating AA's potential as a treatment for diabetic retinal oxidative stress and inflammation.264
AA's effects on insulin signaling and TLR-4-Wnt crosstalk in the pancreas of type 2 diabetic rats were investigated by Khurram et al. using histology, immunohistochemistry, and RT-PCR. AA treatment reversed T2DM-induced pancreatic apoptosis and necrosis, reduced TLR-4, MyD88, NF-κB, p-JNK, and Wnt/β-catenin levels by inhibiting TLR-4/MyD88 and Wnt signaling, while IRS-1, PI3K, and pAkt were upregulated through NF-κB and β-catenin crosstalk alteration. These findings indicate AA's potential as a therapeutic agent for T2DM-associated meta-inflammation.265
3.4 Antioxidant activity
Arjunolic acid is well-documented for its antioxidant activity. It plays an important role in maintaining levels of key antioxidant enzymes, such as catalase (CAT), glutathione-S-transferase (GST), glutathione reductase (GR), glutathione peroxidase (GPx), and superoxide dismutase (SOD), along with cellular metabolites such as reduced glutathione (GSH) and oxidized glutathione (GSSG), thereby reducing oxidative stress.Ramesh et al. evaluated the antioxidant properties of arjunolic acid (AA) using four different in vitro methods: beta-carotene bleaching assay, DPPH radical scavenging assay, reducing power assay, and hydroxyl radical scavenging assay. The findings were compared with those of standard antioxidants, including ascorbic acid, butylated hydroxytoluene (BHT), and catechin. All four assays confirmed the antioxidant potential of AA, though it showed slightly lower efficacy than the standards.266
Maity et al. assessed the protective effect of arjunolic acid against arsenic-induced oxidative stress and reproductive dysfunction in female Wistar rats. Sodium arsenite (10 mg kg−1) was administered along with arjunolic acid (10 mg kg−1) for two estrous cycles. Electrozymographic analysis indicated that arjunolic acid co-treatment reduced arsenic-induced ROS production in uterine tissue by enhancing endogenous antioxidant enzyme activities. Arjunolic acid also helped protect against DNA damage, necrosis, and ovarian and uterine tissue damage in arsenic-treated rats by promoting ovarian steroidogenesis. These protective mechanisms may be related to an enhanced antioxidant defense system, partly facilitated by the elimination of arsenic via the S-adenosyl methionine pathway, where levels of vitamin B12, folic acid, and homocysteine play crucial roles.267
Sinha et al. investigated the preventive effects of arjunolic acid on arsenic-induced oxidative damage in the murine brain. Sodium arsenite was used as the arsenic source. The in vitro free-radical scavenging activity and in vivo antioxidant power of arjunolic acid were assessed using the 2,2-diphenyl-1-picryl hydrazyl (DPPH) radical scavenging and ferric reducing/antioxidant assays, respectively. Oral administration of sodium arsenite (10 mg kg−1) for two days significantly reduced the activity of antioxidant enzymes, including CAT, GST, GR, SOD, and GPx, as well as cellular metabolites, such as reduced glutathione and total thiols, while increasing oxidized glutathione levels. Lipid peroxidation products and protein carbonyl content were also elevated. Treatment with arjunolic acid (20 mg kg−1) for four days before arsenic exposure nearly normalized these levels. Histological findings supported these biochemical results, showing that arjunolic acid mitigates arsenic-induced oxidative damage in the brain. The effects of vitamin C were included as a positive control. Overall, the results suggest that arjunolic acid can protect against arsenic-induced oxidative damage in the brain, likely due to its antioxidant properties.268
Arsenic-induced tissue damage is a major concern, particularly because oxidative stress and impaired antioxidant defense are primary causes of arsenic toxicity, which can lead to reproductive failure. Prasenjit et al. examined the protective role of arjunolic acid, isolated from T. arjuna bark, against arsenic-induced testicular damage in mice. Arsenic administration (10 mg kg−1) over two days significantly reduced antioxidant enzyme activity and cellular metabolite levels, while increasing testicular arsenic content, lipid peroxidation, protein carbonylation, and glutathione disulfide (GSSG) levels. Arsenic exposure also caused degeneration in the seminiferous tubules, leading to necrosis and spermatocyte detachment. Pretreatment with arjunolic acid (20 mg kg−1) for four days helped prevent oxidative stress and structural damage in the testes. Arjunolic acid demonstrated both in vitro free radical scavenging activity and in vivo antioxidant efficacy. Overall, the study suggests that arjunolic acid's antioxidant properties help prevent arsenic-induced testicular toxicity.269
In another study, rats pretreated with arjunolic acid showed significant reductions in neurological deficits and infarct size. Arjunolic acid mitigated ischemia/reperfusion (I/R)-induced neuronal damage by regulating levels of malondialdehyde (MDA), reduced glutathione (GSH), nitric oxide (NO), protein carbonyl content, and mitochondria-generated reactive oxygen species (ROS). It also maintained the activities of Na+-K+ ATPase, SOD, CAT, GPx, and GR enzymes, effectively preventing I/R-induced cerebral oxidative damage due to its antioxidant potential.270
Sumitra et al. examined the modulation of neutrophil function and ROS generation by arjunolic acid in vitro. Neutrophils were isolated from normal and myocardial infarction (MI) mice to evaluate AA's efficacy in reducing oxidative stress. Stimulation of neutrophils with phorbol-12-myristate-13-acetate (PMA) resulted in an oxidative burst of superoxide anions (O2−) and increased release of lysosomal enzymes. Treatment with arjunolic acid significantly inhibited phosphorylation of p47phox and ERK in both stimulated control and MI neutrophils, reduced oxidative phosphorylation activity, and lowered intracellular oxidative stress. This study highlights potential targets for arjunolic acid in ROS regulation through the MAPK pathway in neutrophils.271
Prasenjit et al. investigated the benefits of arjunolic acid in modulating cytokine levels and oxidative stress signaling pathways in the spleen under hyperglycaemic conditions. Diabetes was induced by streptozotocin (STZ) injection (70 mg kg−1), which increased IL-2 and IFN-γ levels and decreased TNF-α in diabetic animals. Hyperglycaemia was associated with elevated production of intracellular reactive intermediates, lipid peroxidation, and protein carbonylation, leading to reduced antioxidant defense. Increased expression of phosphorylated p65, its inhibitor phospho IκBα, and phosphorylated MAPKs was also observed in diabetic spleen tissue. Studies on isolated splenocytes showed mitochondrial membrane potential disruption, cytochrome c release, and caspase 3 activation, leading to apoptotic cell death. Histological examination indicated a depletion of white pulp, consistent with reduced immune response.272
3.5 Hepatoprotective activity
The hepatoprotective properties of arjunolic acid have been documented by multiple studies.6 Prasenjit et al. assessed arjunolic acid for its protective effects against arsenic-induced oxidative stress in mouse liver tissue. Administering sodium arsenite (10 mg kg−1 body weight for two days) notably decreased the activity of antioxidant enzymes—superoxide dismutase, catalase, glutathione-S-transferase, glutathione reductase, and glutathione peroxidase—and depleted reduced glutathione and total thiols. It also elevated the levels of serum markers (alanine transaminase and alkaline phosphatase), DNA fragmentation, protein carbonylation, lipid peroxidation products, and oxidized glutathione. Arjunolic acid, known for its in vitro radical-scavenging and in vivo antioxidant properties, demonstrated protective effects when administered at 20 mg kg−1 for four days before arsenic exposure. This pretreatment normalized antioxidant enzyme activities and minimized tissue damage. Histology confirmed reduced liver necrosis with arjunolic acid compared to arsenic alone. Vitamin C served as a positive control, reinforcing arjunolic acid's role in reducing arsenic-induced liver oxidative stress.249Additionally, this study investigated arjunolic acid's potential to protect against acetaminophen (APAP)-induced acute liver toxicity. Exposure to a hepatotoxic APAP dose (700 mg kg−1) increased oxidative stress biomarkers, reactive oxygen species (ROS), and cell death. Pretreatment with arjunolic acid (80 mg kg−1) provided significant liver protection, preventing glutathione depletion and reducing APAP metabolite formation, without affecting hepatic glutathione levels. The findings suggest that arjunolic acid inhibits APAP bioactivation by cytochrome P450 and reduces APAP-induced JNK and Bcl-2 phosphorylation, thus preventing mitochondrial damage.175
In another model, arjunolic acid was tested for its protective effects against arsenic trioxide (ATO)-induced oxidative stress in mouse liver and kidney tissues. Administering ATO (30 mg kg−1 day−1 for eight weeks) elevated serum markers, ROS, and disrupted the prooxidant-antioxidant balance, triggering apoptotic pathways. Post-treatment with arjunolic acid (20 mg kg−1 for four days) mitigated oxidative stress and apoptosis markers, including mitochondrial membrane potential loss, caspase activation, and cytochrome c release.273
In a study on cisplatin-induced liver toxicity, arjunolic acid demonstrated hepatoprotective effects by reducing oxidative stress, inflammation, and apoptosis. Rats pretreated with arjunolic acid (20 mg kg−1 for 10 days) showed improved liver function, histology, and suppressed malondialdehyde and nitric oxide levels. Arjunolic acid significantly enhanced antioxidant enzyme activities, reduced tumor necrosis factor α, and decreased caspase-3 expression.274
Arjunolic acid also reduced lipid accumulation in free fatty acid (FFA)-challenged hepatocytes without toxicity. In vivo, arjunolic acid reduced adiposity, hepatic inflammation, and lipid disorders in the non-alcoholic fatty liver disease (NAFLD) model, activating Sirt1/AMPK pathways, enhancing autophagy, and restoring gut barrier integrity.275
Finally, in NAFLD models, arjunolic acid showed promising results by reducing triglyceride accumulation, ALT, and AST levels. In vivo, it lowered liver enzyme markers and modified PPAR and FXR expression, indicating reduced liver steatosis and inflammation. Collectively, these studies suggest arjunolic acid as a promising agent for liver protection across various hepatic damage models due to its antioxidant, anti-inflammatory, and anti-apoptotic effects.172
3.6 Anti-inflammatory activity
Arjunolic acid has been widely studied for its anti-inflammatory properties. The anti-arthritic effects of arjunolic acid were evaluated in a rat model of arthritis induced by complete Freund's adjuvant (CFA). Arthritis was induced in male Sprague Dawley rats through an intradermal injection of 0.1 mL CFA into the right footpad. Following arthritis induction, arjunolic acid was administered orally at doses of 40 and 80 mg kg−1 once daily for 25 consecutive days. Indomethacin, a reference drug, was given at 3 mg kg−1 twice weekly for the same duration. Standard procedures were used to assess paw swelling, serum haematology, antioxidant enzymes, inflammatory mediators, and histopathology. The results showed that arjunolic acid significantly (p < 0.01) reduced paw swelling and weight loss in arthritic rats. It also lowered malondialdehyde (MDA) levels, spleen index, and thymus index, while enhancing the activities of superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GPx). Furthermore, arjunolic acid downregulated TNF-α, IL-1β, and IL-6 serum levels (p < 0.01), improving haematological and histopathological changes in CFA-induced arthritis due to its anti-inflammatory effects.276Atopic dermatitis (AD), a chronic, pruritic, eczematous skin disorder associated with allergic inflammation, involves oxidative stress and immune imbalance. The anti-pruritic effects of oral arjunolic acid (AA) were studied in a mouse model of AD induced by 2,4-dinitrochlorobenzene (DNCB). Mice were treated with DNCB on their ears and shaved dorsal skin to induce AD-like symptoms, followed by oral administration of AA at 250 mg kg−1 for three weeks. The results indicated that AA significantly improved AD symptoms, reducing skin lesions, dermatitis scores, ear thickness, and scratching behaviour. AA also decreased reactive oxygen species and modulated inflammatory markers, lowering serum TNF-α, IL-6, IgE, and caspase-3 while maintaining IL-4 and IL-10 levels, thus mitigating DNCB-induced AD symptoms.277
Zining et al. investigated arjunolic acid's effect on a Crohn's disease (CD) model. CD was simulated in IL-10 knockout mice, with arjunolic acid's impact assessed through weight monitoring, histopathology, and intestinal barrier function. Arjunolic acid improved colitis symptoms, reducing weight loss, inflammation scores, and enhancing intestinal permeability. AA also inhibited apoptosis in intestinal epithelial cells by reducing Bax and C-caspase-3 levels. Mechanistic analysis revealed that AA suppressed TLR4 signaling and altered gut microbiome composition, increasing short-chain fatty acid-producing bacteria. Fecal transplantation from AA-treated mice alleviated CD-like colitis in recipient mice, highlighting AA's potential to inhibit key inflammatory pathways and promote gut microbiota balance in CD therapy.278
Edozie et al. evaluated the protective effects of arjunolic acid on fluoxetine (FXT)-induced testicular dysfunction in male rats. Thirty-six rats were assigned to six groups and treated with combinations of saline, arjunolic acid (1.0 or 2.0 mg kg−1), fluoxetine (10 mg kg−1), or fluoxetine plus arjunolic acid. The results showed that FXT treatment disrupted testicular steroidogenic enzymes (3β-HSD and 17β-HSD), ATPase enzymes, and testicular structure. FXT also increased oxidative inflammation markers (MDA, MPO, TNF-α, IL-1β, caspase-3, and p53) and promoted apoptosis. Arjunolic acid treatment counteracted these effects, restoring steroidogenic enzyme activity, improving testicular structure, and increasing antioxidant levels (SOD, CAT, GSH) while reducing oxidative stress and inflammation.279
Ying et al. investigated the effects of arjunolic acid on depression and its potential mechanisms. In vivo and in vitro models were established by administering intraperitoneal injections of lipopolysaccharide (LPS) to mice and stimulating BV2 microglia with LPS. Behavioral tests, H&E staining, and ELISA were conducted to assess arjunolic acid's effect on depression, while RT-qPCR, immunofluorescence, molecular docking, and Western blot analyses were performed to explore the molecular mechanisms. Arjunolic acid significantly improved depressive behaviors in LPS-treated mice, elevated BDNF and 5-HT levels in the hippocampus, decreased iNOS + IBA1 + cells, and increased Arg1 + IBA1 + positive cells in the brain. Furthermore, arjunolic acid facilitated the polarization of BV2 microglia from the M1 to M2 phenotype. Notably, DARTS, CETSA, and molecular docking techniques identified SIRT1 as a target of arjunolic acid. Inhibition of SIRT1 with EX-527 blocked arjunolic acid's effects on reducing LPS-induced depressive behavior and promoted M2 microglia polarization. Additionally, arjunolic acid activated AMPK and reduced Notch1 expression; however, inhibiting AMPK weakened its effect on Notch1 downregulation. This study revealed that arjunolic acid mitigates neuroinflammation by modulating the SIRT1/AMPK/Notch1 signaling pathway, suggesting its potential as an antidepressant agent.280
3.7 Anti-asthmatic activity
Arjunolic acid has been reported to exhibit anti-asthmatic properties. In a study with rats, treatment with arjunolic acid at doses of 50 and 100 mg kg−1 body weight provided significant protection against mast cell disruption, showing 42% and 33% degranulation, respectively, compared to the standard degranulation level induced by disodium chromoglycate. Additionally, arjunolic acid at these doses offered 49% and 64% protection against histamine challenges and 40% and 51% protection against acetylcholine challenges. These anti-asthmatic effects of arjunolic acid may be attributed to its ability to stabilize mast cells and inhibit the release of histamine and acetylcholine in response to antigens.2813.8 Anti-microbial activity
Arjunolic acid has demonstrated moderate antimicrobial activity. Isolated from the leaves of Syzygium guineense, arjunolic acid exhibited antibacterial effects against Bacillus subtilis (0.5 µg per spot), Escherichia coli (3 µg per spot), and Shigella sonnei (30 µg per spot).282 It also showed inhibitory activity against Cryptococcus neoformans, with an IC50 of 20 µg mL−1, and demonstrated significant antibacterial activity against Brucella melitensis, with a zone of inhibition of 9 mm and a minimum inhibitory concentration (MIC) of 30 µg mL.283,284 Furthermore, a mixture of arjunolic acid and asiatic acid exhibited moderate antifungal effects against Candida albicans and Candida parapsilosis.2293.9 Insecticidal activity
Bhakuni et al. identified the insecticidal activity of arjunolic acid. Arjunolic acid, isolated from the stem of Cornus capitata (Wall. ex Roxb.) showed significant inhibitory effects on the fourth instar larvae of Spilarctia obliqua. A dose-dependent effect was observed, with optimal concentrations for reducing larval feeding and growth found to be 617 ppm and 666 ppm, respectively.2473.10 Physicochemical properties
Bag et al. studied the potential of arjunolic acid as a robust and functional molecular framework suitable for creating nanoscale supramolecular architectures and nanomaterials. This nanosized triterpenoid demonstrated effective gelation of various organic solvents (methanol, ethanol, propanol, butanol, dichloromethane, chloroform, cyclohexane, benzene, cyclohexanone, diethylene glycol, and triethylene glycol) at low concentrations. In these solvents, low molecular weight gelator molecules self-assembled to form nanofibers, creating a fibrous network that restricted solvent movement, resulting in gel formation.285 Arjunolic acid also exhibited hierarchical self-assembly in aqueous solutions, forming vesicular structures on nano-to micrometer scales that contribute to gel formation and have proven effective for encapsulating and releasing anticancer drugs such as doxorubicin at physiological pH.Bag et al. also highlighted the self-assembly properties of sodium and potassium salts of arjunolic acid in water and aliphatic alcohols, showing that, at lower concentrations, the molecules form vesicular structures, while, at higher concentrations, they transition into large fibrillar assemblies to create stable gels. These vesicular assemblies are further utilized for the controlled release of various set fluorophores, including doxorubicin, at physiological pH.286,287
4. Analogues of arjunolic acid and their biological activities
Diverse research groups7,9,233,288,289 have subjected arjunolic acid to semi-synthetic modifications by carrying out either selective or simultaneous transformations to various rings and functional groups present in arjunolic acid.Bag et al. first reported the synthesis of an 18-crown-6 methyl ester derivative of arjunolic acid. Arjunolic acid was converted into its methyl ester (67). The primary hydroxy group of compounds (67) was protected using trityl chloride to yield compound (68). This compound was further reacted with sodium hydride and pentaethyleneglycol ditosylate in dry THF to yield 23-[(triphenylmethyl)oxy]– 28-(methoxycarbonyl)-arjuna-18-crown-6 (69). This compound was further reacted with dichloro acetic acid to obtain compound (70), which was further treated with benzoyl chloride to yield the final product (71, Fig. 5). In the same study this group also reported the synthesis of benzylidene derivatives (72–73, Fig. 5) prepared by reacting arjunolic acid methyl ester with benzaldehyde in the presence of perchloric acid as a catalyst. The hydroxy group at the 2-position of the compound was esterified with 3,5-dinitrobenzoyl chloride, and these derivatives were examined for organogelator properties in various organic solvents.285,290
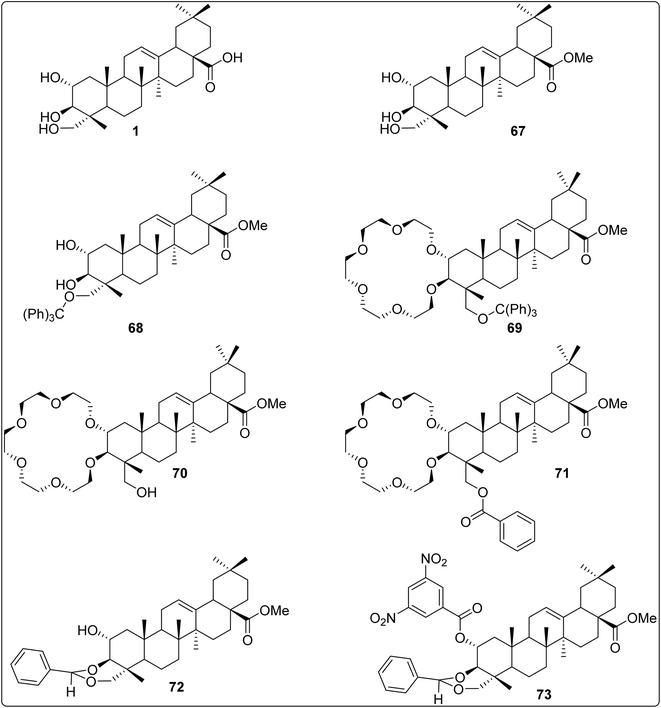 | ||
| Fig. 5 Arjunolic acid analogues synthesized by Bag et al.285,286,291 | ||
Subsequently Bag's team synthesized ten aliphatic and aromatic acetals and ketals of arjunolic acid (Fig. 6) and studied their self-assembly properties in diverse organic solvents. Except for the p-nitrobenzylidene derivative, these ketals, at low concentrations, formed gel-like dispersions in several organic solvents. Using optical, electron, and atomic-force microscopies, the morphologies were found to consist of fibrillar networks and vesicles capable of entrapping 5(6)-carboxyfluorescein. X-ray diffractograms showed that the fibrillar structures were crystalline. Additionally, a charge–transfer complex was formed from a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture of ketal derivatives with electron-donating and electron-accepting groups, and the fibrillar network of the 9-anthrylidene derivative dimerized under irradiation. This work demonstrates that minor structural variations in the ketals significantly influence their aggregation pathways.292
1 mixture of ketal derivatives with electron-donating and electron-accepting groups, and the fibrillar network of the 9-anthrylidene derivative dimerized under irradiation. This work demonstrates that minor structural variations in the ketals significantly influence their aggregation pathways.292
 | ||
| Fig. 6 Aromatic and aliphatic acetals/ketals of arjunolic acid synthesized by Bag et al.292 | ||
Reddy et al. reported the synthesis and HPLC validation of acetylated arjunolic acid (84, Fig. 7), which showed higher antioxidant activity than arjunolic acid, BHT, and vitamin E, as measured by the thiobarbituric acid method (TBA).289
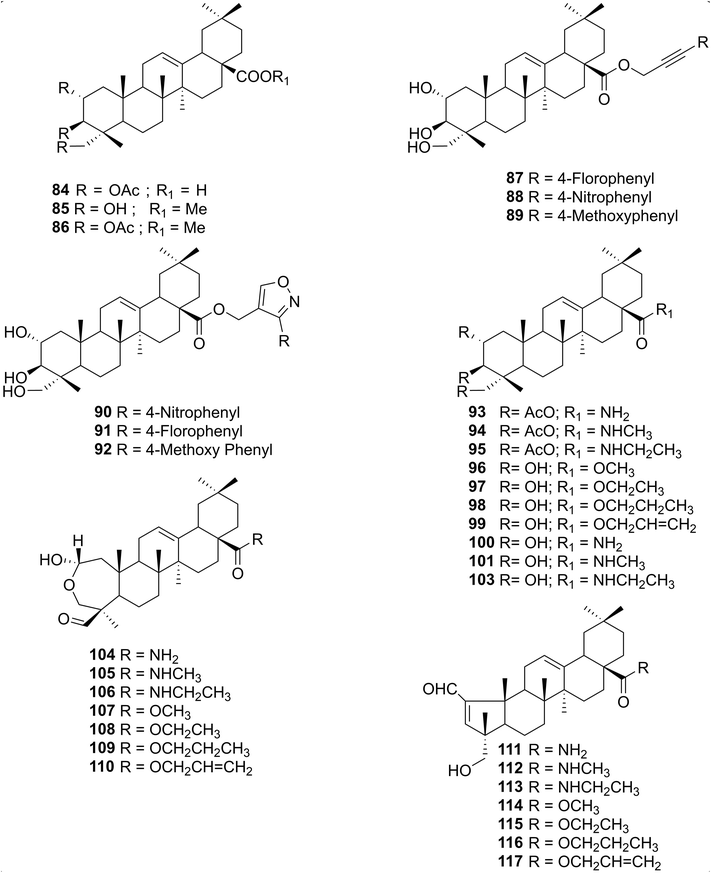 | ||
| Fig. 7 Arjunolic acid analogues synthesized by various groups exhibiting anti-oxidant, anti-cancer, anti-viral, and α-glucosidase inhibitory activities.9,233,288,289 | ||
Diallo and co-workers reported the synthesis of the triacetate of arjunolic acid and its methyl ester. Compound 1 was treated with acetic anhydride and pyridine at room temperature for 12 hours to yield compound 84. Methylation of 1 and 84 was achieved by treating them with diazomethane in diethyl ether at room temperature to yield compounds 85 and 86 (Fig. 7). These compounds were tested for Epstein–Barr virus (EBV) inhibition and further assessed in a two-stage carcinogenesis assay in mouse skin. The results indicated that mice treated with the triacetate and triacetate methyl ester of arjunolic acid showed delayed papilloma development compared to the controls.293,294
Bolatito et al. synthesized new phenyl acetylene and isoxazole analogues of arjunolic acid. The acid functional group was converted into the propargyl ester using propargyl bromide. This propargyl ester of arjunolic acid was reacted with substituted aryl iodide in the presence of a palladium catalyst to obtain compounds 87–89. Simultaneously, this propargyl ester was reacted with various bromoaldoximes to yield compounds 90–92. These analogues were evaluated for their tyrosinase and alpha-glucosidase inhibitory activity. All the tested analogues showed stronger inhibitory effects than the standard drug acarbose. One compound demonstrated tyrosinase inhibition with an IC50 of 14.3 ± 7.6 µM, about three times more potent than kojic acid (IC50 of 41.5 ± 1.0 μM). Additionally, compound 89 exhibited significant alpha-glucosidase inhibition activity with an IC50 close to that of the standard acarbose (10.4 ± 0.06).9
Bruno et al. synthesized novel arjunolic acid derivatives containing a five-membered A-ring with an enal moiety, combined with additional modifications of COOH (at C-28) as esters and aliphatic amides. Compounds 93–103 were reacted with NaIO4 at room temperature to yield compounds 104–110. These derivatives were further treated with acetic acid and piperidine and refluxed in dry benzene before being subjected to reflux with anhydrous magnesium sulphate to yield compounds 111–117. These derivatives were tested for cytotoxicity on cancer and non-tumor human cell lines, aiming to identify promising compounds. Compound 115 showed the highest selectivity between malignant cells and non-malignant fibroblasts. This compound also induced cell cycle arrest in the G0/G1 phase and significantly inhibited the wound closure rate of PANC-1 cancer cells. At a concentration of 0.24 µM, compound 115 synergistically enhanced gemcitabine cytotoxicity and showed no toxicity at lower doses in preliminary pharmacological studies.8
Manohar et al. synthesized various acetals (118–136, Fig. 8) and amides (137–159, Fig. 8) of arjunolic acid and evaluated their anticancer properties. All the synthesized compounds (118–159) were tested against 60 cancer cell lines, representing nine different cancer types, at the National Cancer Institute (NCI), USA. These acetals and amides of arjunolic acid exhibited significant activity, particularly against colon, leukemia, breast, and melanoma cancer types. From the library of acetals (118–136), compounds 119, 121, 126, and 135 demonstrated activity against all nine cancer types, including breast cancers, colon, CNS, leukemia, non-small cell lung cancer, melanoma, ovarian, renal, and prostate cancer. Among these, compound 126 exhibited the most potent IC50 value of 1.56 µM against the CT-26 cancer cell line. Further studies on compound 126 revealed that it induces ROS generation, causing cell necrosis by arresting cells in the G2/M phase of the cell cycle.
 | ||
| Fig. 8 Arjunolic acid derivatives synthesized by Manohar et al., exhibiting anti-cancer activities.7 | ||
Tri-hydroxy amides (151–155, Fig. 8) showed greater activity than tri-acetate amides in the series of aniline-substituted amides, Fig. 8. Conversely, in the case of amides derived from heterocyclic amines, tri-acetate amides (147–150) were more active compared to tri-hydroxy amides (156–159). Substituted benzylamine amides were inactive against all cancer cell lines. Among the amides, compound 147 exhibited the best IC50 values, with 1.88 µM against the CT-26 cell line and 0.4 µM against the HCT-116 colon cancer cell line. Further investigations revealed that compound 147 induces apoptosis through ROS generation and arrests cells in the G0/G1 phase of the cell cycle. In vivo studies also showed that compound 147 inhibited cancer growth in the case of the mouse model.7
Analysis of the structure–activity relationship (SAR) and modifications responsible for the therapeutic profile of arjunolic acid derivatives is tabulated in Table 3. These studies could help identify future hits, optimize lead molecules with drug-like properties and support future drug discovery efforts. We have also made efforts to capture the mechanisms of action of biological activities exhibited by arjunolic acid and its derivatives in Fig. 9.
| Therapeutic action | Rings of arjunolic acid | Modifications |
|---|---|---|
| Anti-cancer | A | Selective conversion of hydroxyls (R–OH) attached at C-2, C-3 and C-23 to acetals and acetates |
| A | Conversion of the A ring to lactol | |
| A | Simultaneous conversion of the A ring to a five-membered ring containing an α,β-unsaturated carbonyl moiety, and conversion of the acid (R–COOH) at C-28 to alkyl esters or alkyl amides | |
| D/E | Selective conversion of acid (R–COOH) at C-28 to amides | |
| Anti-diabetic | D/E | Selective conversion of acid (R–COOH) at C-28 to aryl propargyl esters and isoxazole carbinol esters |
| Anti-viral | A | Simultaneous conversion of hydroxyls (R–OH) attached at C-2, C-3 and C-23 to acetates |
| D/E | Selective conversion of acid (R–COOH) at C-28 to methyl ester | |
| Anti-oxidant | A | Selective conversion of hydroxyls (R–OH) attached at C-2, C-3 and C-23 to acetates |
5. Conclusion
In summary, natural products continue to be a constant source of interesting biologically active molecules. T. arjuna, a traditional medicinal plant native to the Indian subcontinent, was shown to exhibit several medicinal properties and was thus used in traditional medicine. Arjunolic acid, the major triterpenoid constituent of T. arjuna, has been reported to exhibit a plethora of biological activities. The structural amenability of arjunolic acid for diverse modifications has attracted the attention of various research groups, affording semi-synthetic derivatives with interesting therapeutic value. To date, many investigations have focused their research efforts on exploring the multi-therapeutic potential of arjunolic acid and delineating its corresponding mechanism of action. However, the limited reports on arjunolic acid analogues and their corresponding biological profile reveal that its therapeutic potential as a source of new drug leads has not yet been thoroughly explored. The continuous rise in FDA approvals for natural-product-based drugs further augments the need to look at arjunolic acid as a potential new source of natural-product-based drug leads. Hence, natural-product-based drug-discovery programs focused on arjunolic acid would build on the existing armamentarium of desirable molecules with drug-like properties and with a potential for clinical trials.Author contributions
Manohar Bhujel: methodology, investigation, writing – original draft preparation. Sai Giridhar Sarma Kandanur: conceptualization, methodology, investigation, writing – original draft Preparation. Lakshminath Sripada: conceptualization, supervision, writing – reviewing and editing. Nageswara Rao Golakoti: conceptualization, supervision, writing – reviewing and editing.Conflicts of interest
Authors M. B., L. S. and N. R. G. have a patent “A Process For Separating Arjunolic Acid And Asiatic Acid From Heartwood of Terminalia Arjuna” pending to the Sri Sathya Sai Institute of Higher Learning, Prasanthi Nilayam-515134, India. The authors declare no additional conflicts of interest.Data availability
No primary research results, software or codes have been included, and no new data were generated or analysed as part of this review.Acknowledgements
The authors would like to express their deep sense of gratitude to the Founder Chancellor, Bhagawan Sri Sathya Sai Baba, for His constant guidance and inspiration throughout the work.References
- S. B. Bharate and C. W. Lindsley, Natural Products Driven Medicinal Chemistry, J. Med. Chem., 2024, 67, 20723–20730 CrossRef CAS PubMed.
- S. G. S. Kandanur, N. Tamang, N. R. Golakoti and S. Nanduri, Andrographolide: A natural product template for the generation of structurally and biologically diverse diterpenes, Eur. J. Med. Chem., 2019, 176, 513–533 CrossRef CAS PubMed.
- D. J. Newman and G. M. Cragg, Natural Products as Sources of New Drugs over the Nearly Four Decades from 01/1981 to 09/2019, J. Nat. Prod., 2020, 83, 770–803 CrossRef CAS PubMed.
- D. Domingo-Fernández, Y. Gadiya, A. J. Preto, C. A. Krettler, S. Mubeen, A. Allen, D. Healey and V. Colluru, Natural Products Have Increased Rates of Clinical Trial Success throughout the Drug Development Process, J. Nat. Prod., 2024, 87, 1844–1851 CrossRef PubMed.
- T. Hemalatha, S. Pulavendran, C. Balachandran, B. M. Manohar and R. Puvanakrishnan, Arjunolic acid: A novel phytomedicine with multifunctional therapeutic applications, Indian J. Exp. Biol., 2010, 48, 238–247 CAS.
- J. Ghosh and P. C. Sil, Arjunolic acid: A new multifunctional therapeutic promise of alternative medicine, Biochimie, 2013, 95, 1098–1109 CrossRef CAS PubMed.
- M. Bhujel, L. Sripada, K. Buvanesvaragurunathan, P. Perumal, D. Jain, N. Pandey, A. Bajaj and N. R. Golakoti, Synthesis and Anti-cancer activity of Acetals of Arjunolic acid, New J. Chem., 2024, 48, 16957–16967 RSC.
- B. M. F. Gonçalves, V. I. S. Mendes, S. M. Silvestre and J. A. R. Salvador, Design, synthesis, and biological evaluation of new arjunolic acid derivatives as anticancer agents, RSC Med. Chem., 2022, 14, 313–331 RSC.
- B. E. Olanipekun, M. G. Ponnapalli, H. K. Patel, K. Munipalle and K. Shaik, Design, synthesis of new phenyl acetylene and isoxazole analogues of arjunolic acid as potent tyrosinase and alpha glucosidase inhibitors, Nat. Prod. Res., 2023, 37, 1092–1097 CrossRef CAS PubMed.
- S. Gupta, J. P. Bishnoi, N. Kumar and H. Kumar, Terminalia arjuna ( Roxb .) Wight & Arn: Competent source of bioactive components in functional food and drugs, Pharma Innov. J., 2018, 7, 223–231 CAS.
- S. Thakur, H. Kaurav and G. Chaudhary, Terminalia arjuna: A Potential Ayurvedic Cardio Tonic, Int. J. Res. Appl. Sci. Biotechnol., 2021, 8, 227–236 CrossRef.
- N. Mishra and A. Kumar, Pharmacological Approach of Terminalia arjuna: A Review, Plant Cell Biotechnol. Mol. Biol., 2021, 22, 1–5 Search PubMed.
- N. Soni and V. K. Singh, Efficacy and Advancement of Terminalia arjuna in Indian Herbal Drug Research: A Review, Trends Appl. Sci. Res., 2019, 14, 233–242 CrossRef CAS.
- A. Amalraj and S. Gopi, Medicinal properties of Terminalia arjuna (Roxb.) Wight & Arn.: A review, J. Tradit. Complement. Med., 2017, 7, 65–78 CrossRef PubMed.
- S. K. Maulik and K. K. Talwar, Therapeutic Potential of Terminalia Arjuna in Cardiovascular Disorders, Am. J. Cardiovasc. Drugs, 2012, 12, 157–163 CrossRef PubMed.
- P. Ramesh and A. Palaniappan, Terminalia arjuna , a Cardioprotective Herbal Medicine – Relevancy in the Modern Era of Pharmaceuticals and Green Nanomedicine — A Review, Pharmaceuticals, 2023, 16, 2–24 CrossRef PubMed.
- A. Nanda, S. H. Ansari and S. Khatkar, Arjunolic Acid: A Promising Antioxidant Moiety with Diverse Biological Applications, Curr. Org. Chem., 2017, 21, 287–293 CrossRef CAS.
- G. R. De Morais Lima, I. R. P. De Sales, M. R. D. C. Filho, N. Z. T. De Jesus, H. De Sousa Falcão, J. M. Barbosa-Filho, A. G. S. Cabral, A. L. Souto, J. F. Tavares and L. M. Batista, Bioactivities of the genus Combretum (Combretaceae): A review, Molecules, 2012, 17, 9142–9206 CrossRef PubMed.
- P. K. Warrier, V. P. K. Nambiar and C. Ramankutty, Indian Medicinal Plants: A Compendium of 500 Species, Orient Blackswan, 1st edn, 1993, vol. 5 Search PubMed.
- S. Sharma, D. Sharma and N. Agarwal, Diminishing effect of arjuna tree (Terminalia arjuna) bark on the lipid and oxidative stress status of high fat high cholesterol fed rats and development of certain dietary recipes containing the tree bark for human consumption, Res. Pharm., 2012, 2, 22–30 CAS.
- C. Orwa, A. Mutua, R. Kindt, R. Jamnadass and S. Anthony, in World Agroforestry Database, 2009, vol. 4, pp. 1–5 Search PubMed.
- M. R. Wani and S. S. Singh, Variation in leaf morphological traits of Terminalia arjuna Roxb. in natural population of lower parts of Achanakmar Amarkantak Biosphere Reserve (AABR) of Central India, Int. J. Adv. Res., 2016, 4, 484–491 Search PubMed.
- D. V. Singh, R. K. Verma, S. C. Singh and M. M. Gupta, RP-LC determination of oleane derivatives in Terminalia arjuna, J. Pharm. Biomed. Anal., 2002, 28, 447–452 CrossRef CAS PubMed.
- D. V. Singh, R. K. Verma, M. M. Gupta and S. Kumar, Quantitative determination of oleane derivatives in Terminalia arjuna by high performance thin layer chromatography, Phytochem. Anal., 2002, 13, 207–210 CrossRef CAS PubMed.
- M. Sumitra, P. Manikandan, D. A. Kumar, N. Arutselvan, K. Balakrishna, B. M. Manohar and R. Puvankrishnan, Experimental myocardial necrosis in rats : Role of arjunolic acid on platelet aggregation , coagulation and antioxidant status, Mol. Cell. Biochem., 2001, 224, 135–142 CrossRef CAS PubMed.
- L. R. Row, P. S. Murty, G. S. Rao, C. P. Sastry and K. V. J. Rao, Chemical examination of Terminalia species. XII. Isolation \& structure determination of arjunic acid, a new trihydroxytriterpene carboxylic acid from Terminalia arjuna bark, Indian J. Chem., 1970, 8, 772–775 CAS.
- F. E. Kandil and M. I. Nasar, A Tannin Anti-Cancer Promoter From Terminalia arjuna, Phytochemistry, 1998, 47, 1567–1568 CrossRef CAS PubMed.
- M. Saxena, U. Faridi, R. Mishra, M. M. Gupta, M. P. Darokar, S. K. Srivastava, D. Singh, S. Luqman and S. P. S. Khanuja, Cytotoxic agents from Terminalia arjuna, Planta Med., 2007, 73, 1486–1490 CrossRef CAS PubMed.
- A. S. R. Anjaneyulu and A. V. R. Prasad, Structure of terminic acid, a dihydroxytriterpene carboxylic acid from Terminalia arjuna, Phytochemistry, 1983, 22, 993–998 CrossRef CAS.
- F. Y. Sun, X. P. Chen, J. H. Wang, H. L. Qin, S. R. Yang and G. H. Du, Arjunic acid, a strong free radical scavenger from Terminalia arjuna, Am. J. Chin. Med., 2008, 36, 197–207 CrossRef CAS PubMed.
- W. Wang, Z. Ali, Y. Shen, X. C. Li and I. A. Khan, Ursane triterpenoids from the bark of Terminalia arjuna, Fitoterapia, 2010, 81, 480–484 CrossRef CAS PubMed.
- G. S. Arumugam, A. Sen, S. S. Dash, K. Mitra, M. Doble, G. Rajaraman and S. N. Gummadi, Arjunetin as a promising drug candidate against SARS-CoV-2: molecular dynamics simulation studies, J. Biomol. Struct. Dyn., 2022, 40, 12358–12379 CrossRef CAS PubMed.
- M. S. Kim, D. Y. Lee, S. H. Sung and W. K. Jeon, Anti-cholinesterase activities of hydrolysable tannins and polyhydroxytriterpenoid derivatives from Terminalia chebula Retz. fruit, Rec. Nat. Prod., 2018, 12, 284–289 CrossRef CAS.
- M. Ishimatsu, T. Tanaka, G. Nonaka and I. N. J. Lin, The Structure of Arjungenin. A New Sapogenin from Terminalia arjuna, Chem. Pharm. Bull., 1976, 24, 178–180 CrossRef.
- T. Honda, T. Murae, T. Tsuyuki, T. Takahashi and M. Sawai, Arjungenin, Arjunglucoside I, and Arjunglucoside II. A new Triterpene and New Triterpene Glucosides from Terminalia arjuna, Bull. Chem. Soc. Jpn., 1976, 49, 3213–3218 CrossRef CAS.
- P. N. Sharma, S. AB, K. RS and P. SP, Arjunolone–a new flavone from stem bark of Terminalia arjuna, Indian J. Chem. B, 1982, 21, 263 Search PubMed.
- V. K. Tripathi, V. B. Pandey, K. N. Udupa and G. Rücker, Arjunolitin, a triterpene glycoside from Terminalia arjuna, Phytochemistry, 1992, 31, 349 CrossRef CAS.
- A. Ali, G. Kaur, H. Hamid, T. Abdullah, M. Ali, M. Niwa and M. S. Alam, Terminoside A, a new triterpene glycoside from the bark of Terminalia arjuna inhibits nitric oxide production in murine macrophages, J. Asian Nat. Prod. Res., 2003, 5, 137–142 CrossRef CAS PubMed.
- A. Ali, G. Kaur, K. Hayat, M. Ali and M. Ather, A novel naphthanol glycoside from Terminalia arjuna with antioxidant and nitric oxide inhibitory activities, Pharmazie, 2003, 58, 932–934 CAS.
- W. Wang, Z. Ali, X. C. Li, Y. Shen and I. A. Khan, Triterpenoids from two terminalia species, Planta Med., 2010, 76, 1751–1754 CrossRef CAS PubMed.
- R. Hossain, R. Sultana, M. Din Islam, S. Zaman and M. I. Choudhary, Isolation of two new triterpene glycoside from the fruits of Terminalia arjuna and their in vitro and in silico studies, Nat. Prod. Res., 2022, 37, 1–9 Search PubMed.
- T. Patnaik, R. K. Dey and P. Gouda, Isolation of triterpenoid glycoside from bark of Terminalia arjuna using chromatographic technique and investigation of pharmacological behavior upon muscle tissues, E-J. Chem., 2007, 4, 474–479 CAS.
- M. S. Alam, G. Kaur, A. Ali, H. Hamid, M. Ali and M. Athar, Two new bioactive oleanane triterpene glycosides from Terminalia arjuna, Nat. Prod. Res., 2008, 22, 1279–1288 CrossRef CAS PubMed.
- A. MU, M. K. JK and N. T. Ulla, Terminoic acid, A new trihydroxytriterpene carboxylic-acid from bark of Terminalia-arjuna, Indian J. Chem. Sect. B-Organic Chem. Incl. Med. Chem., 1983, 22, 738–740 Search PubMed.
- I. E. Cock, The medicinal properties and phytochemistry of plants of the genus Terminalia (Combretaceae), Inflammopharmacology, 2015, 23, 203–229 CrossRef CAS PubMed.
- M. F. Manzoor, N. Ahmad, Z. Ahmed, R. Siddique, X. A. Zeng, A. Rahaman, R. Muhammad Aadil and A. Wahab, Novel extraction techniques and pharmaceutical activities of luteolin and its derivatives, J. Food Biochem., 2019, 43, 1–19 CrossRef PubMed.
- G. Chaudhari and R. T. Mahajan, Bioprospecting of Terminalia arjuna stem bark and its flavonoids for antimicrobial and anti-biofilm potential, J. Chem. Pharm. Res., 2015, 7, 834–841 CAS.
- W. R. García-Niño and C. Zazueta, Ellagic acid: Pharmacological activities and molecular mechanisms involved in liver protection, Pharmacol. Res., 2015, 97, 84–103 CrossRef PubMed.
- N. A. AL Zahrani, R. M. El-Shishtawy and A. M. Asiri, Recent developments of gallic acid derivatives and their hybrids in medicinal chemistry: A review, Eur. J. Med. Chem., 2020, 204, 112609 CrossRef CAS PubMed.
- D. A. Vattem and K. Shetty, Biological Functionality Of Ellagic Acid: A Review, J. Food Biochem., 2005, 29, 234–266 CrossRef CAS.
- S. P. Bangar, V. Chaudhary, N. Sharma, V. Bansal, F. Ozogul and J. M. Lorenzo, Kaempferol: A flavonoid with wider biological activities and its applications, Crit. Rev. Food Sci. Nutr., 2023, 63, 9580–9604 CrossRef CAS.
- J. K. Kim and S. U. Park, Recent Studies on Kaempferol and its Biological and Pharmacological Activities, EXCLI J., 2020, 19, 627–634 Search PubMed.
- A. Saha, V. M. Pawar and S. Jayaraman, Characterisation of polyphenols in Terminalia arjuna bark extract, Indian J. Pharm. Sci., 2012, 74, 339–347 CrossRef CAS.
- T. W. Sirk, E. F. Brown, M. Friedman and A. K. Sum, Molecular binding of catechins to biomembranes: relationship to biological activity, J. Agric. Food Chem., 2009, 57, 6720–6728 CrossRef CAS PubMed.
- S. Takahashi, H. Tanaka, Y. Hano, K. Ito, T. Nomura and K. Shigenobu, Hypotensive effect in rats of hydrophilic extract from Terminalia arjuna containing tannin-related compounds, Phyther. Res., 1997, 11, 424–427 CrossRef CAS.
- T. C. Lin, S. l. Chien, H. F. Chen and F. L. Hsu, Tannins and related compounds from Combretaceae plants, Chinese Pharm. J., 2000, 52, 1–26 CAS.
- A. P. Kulkarni, H. S. Mahal, S. Kapoor and S. M. Aradhya, In vitro studies on the binding, antioxidant, and cytotoxic action of punicalagin, J. Agric. Food Chem., 2007, 55, 1491–1500 CrossRef CAS PubMed.
- P. L. Kuo, Y. L. Hsu, T. C. Lin, L. T. Lin, J. K. Chang and C. C. Lin, Casuarinin from the bark of Terminalia arjuna induces apoptosis and cell cycle arrest in human breast adenocarcinoma MCF-7 cells, Planta Med., 2005, 71, 237–243 CrossRef CAS PubMed.
- A. R. ArauÌjo, A. C. ArauÌjo, R. L. Reis and R. A. Pires, Vescalagin and Castalagin Present Bactericidal Activity toward Methicillin-Resistant Bacteria, ACS Biomater. Sci. Eng., 2021, 7, 1022–1030 CrossRef.
- T. M. Souza-Moreira, J. A. Severi, K. Lee, K. Preechasuth, E. Santos, N. A. R. Gow, C. A. Munro, W. Vilegas and R. C. L. R. Pietro, Anti-Candida targets and cytotoxicity of casuarinin isolated from Plinia cauliflora leaves in a bioactivity-guided study, Molecules, 2013, 18, 8095–8108 CrossRef CAS PubMed.
- D. J. Kwon, Y. S. Bae, S. M. Ju, A. R. Goh, S. Y. Choi and J. Park, Casuarinin suppresses TNF-α-induced ICAM-1 expression via blockade of NF-κB activation in HaCaT cells, Biochem. Biophys. Res. Commun., 2011, 409, 780–785 CrossRef CAS PubMed.
- A. M. Muddathir, K. Yamauchi and T. Mitsunaga, Anti-acne activity of tannin-related compounds isolated from Terminalia laxiflora, J. Wood Sci., 2013, 59, 426–431 CrossRef CAS.
- Z. Chang, Q. Zhang, W. Liang, K. Zhou, P. Jian, G. She and L. Zhang, A Comprehensive Review of the Structure Elucidation of Tannins from Terminalia Linn, Evid. Based Complement. Alternat. Med., 2019, 2019, 1–26 Search PubMed.
- A. Sen, P. Dhavan, K. K. Shukla, S. Singh and G. Tejovathi, Analysis of IR, NMR and Antimicrobial Activity of β-Sitosterol Isolated from Momordica charantia, Sci Secur. J Biotech, 2012, 1, 9–13 CAS.
- J. M. Castellano, S. Ramos-Romero and J. S. Perona, Oleanolic Acid: Extraction, Characterization and Biological Activity, Nutrients, 2022, 14, 1–29 CrossRef PubMed.
- B. K. Chouksey and S. K. Srivasava, Antifungal agent from Terminalia arjuna, Indian J. Chem. B., 2001, 40, 354–356 Search PubMed.
- R. K. Upadhyay, M. B. Pandey, R. N. Jha, V. P. Singh and V. B. Pandey, Triterpene glycoside from terminalia arjuna, J. Asian Nat. Prod. Res., 2001, 3, 207–212 CrossRef CAS.
- R. M. Rastogi, Compendium of Indian medicinal plants, Cent. Drug Res. Inst., 1990, 1, 388–389 Search PubMed.
- R. M. Lopes, T. Da Silveira Agostini-Costa, M. A. Gimenes and D. Silveira, Chemical composition and biological activities of Arachis species, J. Agric. Food Chem., 2011, 59, 4321–4330 CrossRef CAS PubMed.
- M. H. Radi, R. A. El-Shiekh, A. M. El-Halawany and E. Abdel-Sattar, Friedelin and 3β-Friedelinol: Pharmacological Activities, Rev. Bras. Farmacogn., 2023, 33, 886–900 CrossRef CAS.
- P. P. S. Ambika and S. M. S. Chauhan, Activity-guided isolation of antioxidants from the leaves of Terminalia arjuna, Nat. Prod. Res., 2014, 28(10), 760–763 CrossRef PubMed.
- R. Liu, B. Zhao, D. E. Wang, T. Yao, L. Pang, Q. Tu, S. M. Ahmed, J. J. Liu and J. Wang, Nitrogen-containing apigenin analogs: Preparation and biological activity, Molecules, 2012, 17, 14748–14764 CrossRef CAS PubMed.
- P. Udaysing and D. K. Gaikwad, Comprehensive Review of Arjuna: A Potent Drug in the Management of Cardiac Disorder, Int. J. Drug Formul. Res., 2014, 2, 166–184 Search PubMed.
- R. L. S. Sikarwar, B. Pathak and A. Jaiswal, Some unique ethnomedicinal perceptions of tribal communities of Chitrakoot, Madhya Pradesh, Indian J. Tradit. Knowl., 2008, 7, 613–617 Search PubMed.
- M. Anisuzzaman, A. H. M. M. Rahman, M. Harun-Or-Rashid, A. T. M. Naderuzzaman and A. K. M. R. Islam, An ethnobotanical study of Madhupur, Tangail, J. Appl. Sci. Res., 2007, 3, 519–530 Search PubMed.
- C. Muthu, M. Ayyanar, N. Raja and S. Ignacimuthu, Medicinal plants used by traditional healers in Kancheepuram District of Tamil Nadu, India, J. Ethnobiol. Ethnomed., 2006, 2, 1–10 Search PubMed.
- K. Yesodharan and K. A. Sujana, Ethnomedicinal knowledge among Malamalasar tribe of Parambikulam wildlife sanctuary, Kerala, Indian J. Tradit. Knowl., 2007, 6, 481–485 Search PubMed.
- A. B. Prusti and K. K. Behera, Ethnobotanical Exploration of Malkangiri District of Orissa, India, Ethnobot. Leafl., 2007, 11, 122–140 Search PubMed.
- L. Sharma and A. Kumar, Traditional medicinal practices of Rajasthan, Indian J. Tradit. Knowl., 2007, 06, 531–533 Search PubMed.
- K. Bone, Clinical Applications of Ayurvedic and Chinese Herbs. Monographs for the Western Herbal Practitioner, Phyther. Press, Australia, 1996, pp. 137–141 Search PubMed.
- S. Nesamony, Oushadha Sasyangal – 2, State Institute of Languages, Tiruvanantapuram, 2001 Search PubMed.
- L. D. Kapoor, CRC Handbook of Ayurvedic Medicinal Plants, CRC press, 1st edn, 2018 Search PubMed.
- N. Singh, K. K. Kapur, S. P. Singh, K. Shanker, J. N. Sinha and R. P. Kohli, Mechanism of cardiovascular action of Terminalia arjuna, Planta Med., 1982, 45, 102–104 CrossRef CAS PubMed.
- S. Dwivedi and M. P. Agarwal, Antianginal and cardioprotective effects of Terminalia arjuna, an indigenous drug, in coronary artery disease, J. Assoc. Physicians India, 1994, 42(4), 287–289 CAS.
- S. Dwivedi and R. Jauhari, Beneficial effects of Terminalia arjuna in coronary artery disease, Indian Heart J, 1997, 49(5), 507–510 CAS.
- A. Bharani, A. Ganguly and K. D. Bhargava, Salutary effect of Terminalia Arjuna in patients with severe refractory heart failure, Int. J. Cardiol., 1995, 49, 191–199 CrossRef CAS.
- A. Ram, P. Lauria, R. Gupta, P. Kumar and V. N. Sharma, Hypocholesterolaemic effects of Terminalia arjuna tree bark, J. Ethnopharmacol., 1997, 55, 165–169 CrossRef CAS PubMed.
- H. P. Shaila, S. L. Udupa and A. L. Udupa, Hypolipidemic activity of three indigenous drugs in experimentally induced atherosclerosis, Int. J. Cardiol., 1998, 67, 119–124 CrossRef CAS PubMed.
- P. U. Kumar, P. Adhikari, P. Pereira and P. Bhat, Safety and efficacy of Hartone in stable angina pectoris—an open comparative trial, J. Assoc. Physicians India, 1999, 47, 685–689 CAS.
- K. Gauthaman, M. Maulik, R. Kumari, S. C. Manchanda, A. K. Dinda and S. K. Maulik, Effect of chronic treatment with bark of Terminalia arjuna: a study on the isolated ischemic-reperfused rat heart, J. Ethnopharmacol., 2001, 75, 197–201 CrossRef CAS.
- R. Gupta, S. Singhal, A. Goyle and V. N. Sharma, Antioxidant and hypocholesterolaemic effects of Terminalia arjuna tree-bark powder: a randomised placebo-controlled trial, J. Assoc. Physicians India, 2001, 49, 231–235 CAS.
- B. Chopra, Antianginal and cardiopretective effects of Terminalia arjuna, J. Assoc. Physicians India, 1994, 42, 756 CAS.
- O. M. Bhat, P. U. Kumar, K. R. Rao, A. Ahmad and V. Dhawan, Terminalia arjuna prevents Interleukin-18-induced atherosclerosis via modulation of NF-κB/PPAR-γ-mediated pathway in Apo E−/− mice, Inflammopharmacology, 2018, 26, 583–598 CrossRef CAS PubMed.
- A. Parveen, R. Babbar, S. Agarwal, A. Kotwani and M. Fahim, Mechanistic clues in the cardioprotective effect of Terminalia arjuna bark extract in isoproterenol-induced chronic heart failure in rats, Cardiovasc. Toxicol., 2011, 11, 48–57 CrossRef PubMed.
- N. K. Mary, B. H. Babu and J. Padikkala, Antiatherogenic effect of Caps HT2, a herbal Ayurvedic medicine formulation, Phytomedicine, 2003, 10, 474–482 CrossRef CAS PubMed.
- F. Khaliq, A. Parveen, S. Singh, R. Gondal, M. E. Hussain and M. Fahim, Improvement in myocardial function by Terminalia arjuna in streptozotocin-induced diabetic rats: Possible mechanisms, J. Cardiovasc. Pharmacol. Ther., 2013, 18, 481–489 CrossRef PubMed.
- S. J. Kaur, I. S. Grover and S. Kumar, Modulatory effects of a tannin fraction isolated from Terminalia arjuna on the genotoxicity of mutagens in Salmonella typhimurium, Food Chem. Toxicol., 2000, 38, 1113–1119 CrossRef CAS.
- A. Nagpal, L. S. Meena, S. Kaur, I. S. Grover, R. Wadhwa and S. C. Kaul, Growth suppression of human transformed cells by treatment with bark extracts from a medicinal plant, Terminalia arjuna, Vitr. Cell. Dev. Biol. – Anim., 2000, 36, 544–547 CrossRef CAS.
- G. Scassellati-Sforzolini, L. M. Villarini, L. M. Moretti, L. M. Marcarelli, R. Pasquini, C. Fatigoni, L. S. Kaur, S. Kumar and I. S. Grover, Antigenotoxic properties of Terminalia arjuna bark extracts, J. Environ. Pathol. Toxicol. Oncol., 1999, 18, 119–125 CAS.
- T. K. Reddy, P. Seshadri, K. K. R. Reddy, G. C. Jagetia and C. D. Reddy, Effect of Terminalia arjuna Extract on Adriamycin-induced DNA Damage, Phyther. Res., 2008, 22, 1188–1194 CrossRef.
- S. Singh, S. K. Verma and S. K. Singh, Analysis of anti-cancer potential of Terminalia arjuna, Int. J. Adv. Sci. Res. Manag., 2017, 2, 82–87 Search PubMed.
- P. Rani and N. Khullar, Antimicrobial evaluation of some medicinal plants for their anti-enteric potential against multi-drug resistant Salmonella typhi, Phyther. Res., 2004, 18, 670–673 CrossRef PubMed.
- K. R. Aneja, C. Sharma and R. Joshi, Antimicrobial activity of Terminalia arjuna Wight & Arn: An ethnomedicinal plant against pathogens causing ear infection, Braz. J. Otorhinolaryngol., 2012, 78, 68–74 CrossRef PubMed.
- J. P. Jethinlalkhosh and A. Antony, Antibacterial and cytotoxic activity of aqueous and methanolic extracts of Terminalia arjuna, Int. J. Res. Pharm. Sci., 2013, 4, 36–39 Search PubMed.
- T. Javed, S. Riaz, M. Uzair, G. Mustafa, A. Mohyuddin and B. A. Ch., Biological activity of Terminalia arjuna on Human Pathogenic Microorganisms, Pakistan J. Pharm. Res., 2016, 2, 23 CrossRef CAS.
- V. Kumar, N. Sharma, A. Sourirajan, P. K. Khosla and K. Dev, Comparative evaluation of antimicrobial and antioxidant potential of ethanolic extract and its fractions of bark and leaves of Terminalia arjuna from north-western Himalayas, India, J. Tradit. Complement. Med., 2018, 8, 100–106 CrossRef PubMed.
- S. Mandal, A. Patra, A. Samanta, S. Roy, A. Mandal, T. Das Mahapatra, S. Pradhan, K. Das and D. K. Nandi, Analysis of phytochemical profile of Terminalia arjuna bark extract with antioxidative and antimicrobial properties, Asian Pac. J. Trop. Biomed., 2013, 3, 960–966 CrossRef CAS PubMed.
- F. Murad, K. A. Alam, R. M. Mazumdar, S. Islam, M. N. Nipa, A. Iqbal and H. R. Bhuiyan, Anti-bacterial Activity of the Extract of Terminalia arjuna Against Multi Antibiotic Resistant Vibrio cholerae, J. Sci. Res., 2010, 3, 129 CrossRef.
- S. Debnath, D. Dey and S. Hazra, Antibacterial and antifungal activity of Terminalia arjuna Wight & Arn. bark against multi-drug resistant clinical isolates, J. Coast. Life Med, 2014, 4(4), 312–318 Search PubMed.
- S. Ramya, T. Kalaivani, C. Rajasekaran, P. Jepachanderamohan, N. Alaguchamy, M. Kalayansundaram and R. Jayakumararaj, Antimicrobial Activity of Aqueous Extracts of Bark, Root, Leaves and Fruits of Terminalia arjuna Wight & Arn, Ethnobot. Leafl., 2008, 12, 1192–1197 Search PubMed.
- D. K. Meena, N. P. Sahu, P. P. Srivastava, M. Jadhav, R. Prasad, R. C. Mallick, A. K. Sahoo, B. K. Behera, D. Mohanty and B. K. Das, Effective valorisation of facile extract matrix of Terminalia arjuna (Roxb) against elite microbes of aquaculture industry–a credence to bioactive principles: Can it be a sustainability paradigm in designing broad spectrum antimicrobials?, Ind. Crops Prod., 2021, 171, 113905 CrossRef CAS.
- G. Phani Kumar, K. Navya, E. M. Ramya, M. Venkataramana, T. Anand and K. R. Anilakumar, DNA damage protecting and free radical scavenging properties of Terminalia arjuna bark in PC-12 cells and plasmid DNA, Free Radic. Antioxid., 2013, 3, 35–39 CrossRef.
- G. L. shastry Viswanatha, S. K. Vaidya, C. Ramesh, N. Krishnadas and S. Rangappa, Antioxidant and antimutagenic activities of bark extract of Terminalia arjuna, Asian Pac. J. Trop. Med., 2010, 3, 965–970 CrossRef.
- S. A. Shahid Chatha, Bioactive Components and Antioxidant Properties of Terminalia arjuna L.Extracts, J. Food Process. Technol., 2014, 5(2), 1–5 Search PubMed.
- A. K. Ghosh, E. Mitra, M. Dutta, D. Mukherjee, A. Basu, S. B. Firdaus, D. Bandyopadhyay and A. Chattopadhyay, Protective effect of aqueous bark extract of Terminalia arjuna on Cu2+-ascorbate induced oxidative stress in vitro: Involvement of antioxidant mechanism(S), Asian J. Pharm. Clin. Res., 2013, 6, 196–200 Search PubMed.
- B. Raghavan and S. K. Kumari, Effect of Terminalia arjuna stem bark on antioxidant status in liver and kidney of alloxan diabetic rats, Indian J. Physiol. Pharmacol., 2006, 50, 133–142 CAS.
- P. Manna and P. C. Sil, Arjunolic acid: benificial role in type 1 diabetes and its associated organ pathophysiology, Free Radic. Res., 2012, 46, 815–830 CrossRef CAS PubMed.
- N. Aslam, M. Ahmad, K. Barkat and M. Abbas, Antioxidant and Anti-urease Activity of Various Solvent Extracts of “Terminalia arjuna” Seeds, J. Pharm. Res. Int., 2018, 20, 1–5 CrossRef.
- N. Anbalagan, M. Mallika, S. Kuruvilla, M. V. V. Prasad, A. Patra and K. Balakrishna, Anti-oxidant and hepatoprotective activities of alcoholic extract of Terminalia arjuna, Nat. Prod. Sci., 2007, 13(2), 105–109 CAS.
- P. Manna, M. Sinha and P. C. Sil, Phytomedicinal activity of Terminalia arjuna against carbon tetrachloride induced cardiac oxidative stress, Pathophysiology, 2007, 14, 71–78 CrossRef CAS PubMed.
- D. K. Meena, A. K. Sahoo, P. P. Srivastava, N. P. Sahu, M. Jadhav, M. Gandhi, H. S. Swain, S. Borah and B. K. Das, On valorization of solvent extracts of Terminalia arjuna (arjuna) upon DNA scission and free radical scavenging improves coupling responses and cognitive functions under in vitro conditions, Sci. Rep., 2021, 11, 1–20 Search PubMed.
- M. A. Morshed, M. A. Uddin, T. Hasan, T. Ahmed, F. Uddin, M. Zakaria, A. Haque and A. K. Parvez, Evaluation of Analgesic and Anti-inflammatory Effects of Terminalia arjuna Ethanol Extract, Int. J. Pharm. Sci. Res., 2011, 2, 2577–2585 Search PubMed.
- F. Abo-Elghiet, A. Abd-elsttara, A. Metwaly and A.-E. Mohammad, Antioxidant, anti-inflammatory, and antimicrobial evaluation of Terminalia arjuna leaves, fruits, and bark, Azhar Int. J. Pharm. Med. Sci., 2022, 2, 148–158 Search PubMed.
- L. Chularojanamontri, N. Chiruntanat, K. Jaijoy, N. Soonthornchareonnon and S. Sireeratawong, Anti-inflammatory, Analgesic and Antipyretic Activity of Terminalia arjuna Roxb. Extract in Animal Models, J. Med. Assoc. Thail., 2017, 100, S92–S97 Search PubMed.
- N. Dube, C. Nimgulkar and D. K. Bharatraj, Validation of therapeutic anti-inflammatory potential of Arjuna Ksheera Paka – A traditional Ayurvedic formulation of Terminalia arjuna, J. Tradit. Complement. Med., 2017, 7, 414–420 CrossRef.
- R. M. Sarma, B. Subramanian, P. Tamilmaran and G. Ramakrishnan, Anti-inflammatory and Anti-granuloma effect of the extract of the leaf of Terminalia arjuna (Roxb.) Wight & Arn, J. Pharm. Sci. Res., 2019, 11, 3579–3586 Search PubMed.
- S. Halder, N. Bharal, P. K. Mediratta, I. Kaur and K. K. Sharma, Anti-inflammatory, immunomodulatory and antinociceptive activity of Terminalia arjuna roxb bark powder in mice and rats, Indian J. Exp. Biol., 2009, 47, 577–583 Search PubMed.
- S. Shweta Shaligram, P. Tabassum Arif, K. Surekha Babasaheb and P. Scholar, Review on Arjuna (Terminalia Arjuna Roxb.) With Special Reference To Prameha (Diabetes), World J. Pharm. Res., 2019, 8, 1477–1489 Search PubMed.
- A. Gupta and S. R. Chaphalkar, Haemolytic Activities and Antidiabetic Effect of Terminalia arjuna and Emblica officinalis, Eur. J. Pharm. Med. Res., 2016, 3, 334–338 Search PubMed.
- C. Kumar, R. Kumar and S. Nehar, Phytochemical properties, total antioxidant status of acetone and methanol extract of Terminalia arjuna Roxb. bark and its hypoglycemic effect on Type-II diabetic albino rats, J. Pharmacogn. Phytochem., 2013, 2, 199–208 Search PubMed.
- S. Singh, N. Shukla, M. Bhowmick and J. Rathi, Evaluation of Anti-Diabetic Activity of Terminalia Arjuna Root Extract in Alloxan Induced Diabetic Rats, J. Biomed. Pharm. Res., 2019, 8(1), 20–29 CAS.
- M. Alam Morshed, A. Haque, B. Rokeya and L. Ali, Anti-Hyperglycemic and Lipid Lowering Effect of Terminalia arjuna Bark Extract on Streptozotocin induced Type 2 Diabetic Model Rats, Int. J. Pharm. Pharm. Sci., 2011, 3, 449–453 Search PubMed.
- S. A. Shengule, S. Mishra and S. Bodhale, Inhibitory effect of a standardized hydroethanolic extract of Terminalia arjuna bark on alpha-amylase enzyme, Asian J. Pharm. Clin. Res., 2018, 11, 366–369 CrossRef.
- M. K. Borde, I. R. Mohanty, U. Maheswari, R. K. Suman and Y. Deshmukh, Natural dipeptidyl peptidase-4 inhibitor Terminalia arjuna mitigates myocardial infarction co-existing with diabetes in experimental rats, J. Diabetes, Metab. Disord. Control, 2018, 5, 48–56 CrossRef.
- H. A. J. Thomson, O. O. Ojo, P. R. Flatt and Y. H. A. Abdel-Wahab, Aqueous Bark Extracts of Terminalia arjuna Stimulates Insulin Release, Enhances Insulin Action and Inhibits Starch Digestion and Protein Glycation in vitro, Austin J. Endocrinol. Diabetes, 2014, 1(1) Search PubMed.
- S. P. Sangamithira, J. Revathy, S. Sheik Abdullah and P. Sampath Kumar, The hepatoprotective effect of ethanolic bark extract of Terminalia arjuna on paracetamol induced liver damage, Biosci. Biotechnol. Res. Asia, 2011, 8, 777–781 CrossRef.
- M. Biswas, K. Biswas, T. K. Karan, S. Bhattacharya, A. K. Ghosh and P. K. Haldar, Evaluation of Analgesic and Anti-inflammatory Activities of Terminalia arjuna Leaf, J. Phytol., 2011, 3, 33–38 CAS.
- S. Sivalokanathan, M. Ilayaraja and M. P. Balasubramanian, Anticancer potency of Terminalia arjuna bark on N-nitrosodiethylamine- induced hepatocellular carcinoma in rats, Nat. Prod. Sci., 2004, 10, 190–195 CAS.
- G. M. Chaudhari and R. T. Mahajan, In Vitro Hepatoprotective Activity of Terminalia arjuna Stem Bark and Its Flavonoids Against CCl4 Induced Hepatotoxicity in Goat Liver Slice Culture, Asian J. Plant Sci. \& Res., 2016, 6, 10–17 Search PubMed.
- A. P. S. Vishwakarmȧ, A. Vishwe, P. K. Sahu and A. K. Chaurasiya, Screening of Hepatoprotective Potential of Ethanolic and Aqueous Extract of Terminalia arjuna bark against paracetamol/CCl4 Induced Liver Damage in Wistar Albino Rats, Int. J. Pharm. Arch., 2013, 10, 243–250 Search PubMed.
- P. Manna, M. Sinha and P. C. Sil, Aqueous extract of Terminalia arjuna prevents carbon tetrachloride induced hepatic and renal disorders, BMC Complement. Altern. Med., 2006, 6, 1–10 CrossRef PubMed.
- S. Gomathi and K. M. S. Rathinam, Evaluation of repellency and antifeedant activities of Saraca asoca and Terminalia arjuna Bark extracts against Sitophilus oryzae, Int. J. Zool. Stud., 2017, 2, 282–285 Search PubMed.
- P. K. Mukherjee, K. Mukherjee, M. R. Kumar, M. Pal and B. P. Saha, Evaluation of wound healing activity of herbal formulation, Asian J. Chem., 2007, 19, 1273–1276 Search PubMed.
- M. Chaudhari and S. Mengi, Evaluation of phytoconstituents of Terminalia arjuna for wound healing activity in rats, Phyther. Res., 2006, 20, 799–805 CrossRef CAS PubMed.
- M. M. Rane and S. A. Mengi, Comparative effect of oral administration and topical application of alcoholic extract of Terminalia arjuna bark on incision and excision wounds in rats, Fitoterapia, 2003, 74, 553–558 CrossRef PubMed.
- D. K. Meena, A. K. Sahoo, M. Jayant, N. P. Sahu, P. P. Srivastava, H. S. Swain, B. K. Behera, K. Satvik and B. K. Das, Bioconversion of Terminalia arjuna bark powder into a herbal feed for Labeo rohita: Can it be a sustainability paradigm for Green Fish production?, Anim. Feed Sci. Technol., 2022, 284, 115132 CrossRef CAS.
- D. K. Meena, A. K. Sahoo, H. Chowdhury, H. S. Swain, N. P. Sahu, B. K. Behera, P. P. Srivastava and B. K. Das, Effects of extraction methods and solvent systems on extract yield, proximate composition and mineral profiling of Terminalia arjuna (Arjuna) dry powders and solvent extracts, J. Innov. Pharm. Biol. Sci, 2020, 7, 22–31 CAS.
- P. Parmar and K. Khamrui, Development of process for the production of arjuna herbal ghee from buffalo milk, Indian J. Anim. Sci., 2017, 87, 203–207 CAS.
- S. Bishnoi and S. S. Ahlawat, Development of Buffalo Meat Rolls Incorporated with Aloe Vera Gel and Arjun Tree Bark Extract, Haryana Vetnary, 2015, 54, 174–177 Search PubMed.
- P. D. Sawale, R. Pothuraju, S. Abdul Hussain, A. Kumar, S. Kapila and G. R. Patil, Hypolipidaemic and anti-oxidative potential of encapsulated herb (Terminalia arjuna) added vanilla chocolate milk in high cholesterol fed rats, J. Sci. Food Agric., 2016, 96, 1380–1385 CrossRef CAS PubMed.
- K. Batta and H. Rajput, Production, Consumption and Benefits of Different Herbal Tea: A Review, J. Food Ind. Microbiol., 2021, 7, 128 Search PubMed.
- S. Verma, S. R. Murti, D. L. Singh, R. K. Tiwari and N. Quadri, Formulation of Herbal Green Tea Using Terminalia Arjuna Bark,Glycyrrhiza Glabra Root, Gymnema Sylvester Leaf And Green CardamomSeeds: Its Nutritional & Phytochemical Analysis, Br. J. Pharm. Med. Res., 2020, 4, 2202–2211 Search PubMed.
- D. K. Meena, B. K. Das, A. K. Sahoo, N. P. Sahu, P. P. Srivastava and S. Borah, Terminalia arjuna Bark Powder as a Potential Immunomodulator in Labeo rohita: Enhanced Hematological, Adaptive, and Humoral Responses against Bacterial Pathogens and Concordant Liver Histomorphology, Pathogens, 2024, 13(4), 295 CrossRef CAS PubMed.
- D. K. Meena, S. P. Panda, A. K. Sahoo, P. P. Srivastava, N. P. Sahu, M. Kumari, S. Samantaray, S. Borah and B. K. Das, Immunogenic Effects of Dietary Terminalia arjuna Bark Powder in Labeo rohita, a Fish Model: Elucidated by an Integrated Biomarker Response Approach, Animals, 2023, 13(1), 39 CrossRef PubMed.
- K. Aamir, H. U. Khan, C. F. Hossain, M. RejinaAfrin, I. Shaik, N. Salleh, N. Giribabu and A. Arya, Oral toxicity of arjunolic acid on hematological, biochemical and histopathological investigations in female Sprague Dawley rats, PeerJ, 2019, 7, e8045 CrossRef PubMed.
- D. K. Meena, A. K. Sahoo, H. S. Swain, S. Borah, P. P. Srivastava, N. P. Sahu and B. K. Das, Prospects and perspectives of virtual in-vitro toxicity studies on herbal extracts of Terminalia arjuna with enhanced stratagem in Artemia salina model: A panacea to explicit the credence of solvent system in brine shrimp lethality bioassay, Emirates J. Food Agric., 2020, 32, 25–37 CrossRef.
- V. Anuradha, P. Shankar, P. Bhuvana, M. Syedali and N. Yogananth, Terminalia Arjuna Bark Assisted Biosynthesis , Characterization and Bioactivity of Metal oxide Nanoparticles, J. Chem. Pharm. Res., 2017, 9, 34–46 CAS.
- A. A. Dudhane, S. R. Waghmode, L. B. Dama and P. Vaibhav, Synthesis and Characterization of Gold Nanoparticles using Plant Extract of Terminalia arjuna with Antibacterial Activity, Int. J. Nanosci. Nanotechnol., 2019, 15, 75–82 CAS.
- M. Rakhi and B. B. Gopal, Terminalia Arjuna Bark Extract Mediated Size Controlled Synthesis of Polyshaped Gold Nanoparticles and Its Application in Catalysis, Int. J. Res. Chem. Environ. Search PubMed.
- K. Gopinath, K. S. Venkatesh, R. Ilangovan, K. Sankaranarayanan and A. Arumugam, Green synthesis of gold nanoparticles from leaf extract of Terminalia arjuna , for the enhanced mitotic cell division and pollen germination activity, Ind. Crop. Prod., 2013, 50, 737–742 CrossRef CAS.
- N. Suganthy, V. S. Ramkumar and A. Pugazhendhi, Biogenic synthesis of gold nanoparticles from Terminalia arjuna bark extract : assessment of safety aspects and neuroprotective potential via antioxidant, anticholinesterase, and antiamyloidogenic effects, Environ. Sci. Pollut. Res., 2018, 25, 10418–10433 CrossRef CAS PubMed.
- T. Akther, S. Priya, S. K. Sah, M. S. Khan and S. Hemalatha, Ta-AgNps are Potential Antimicrobial Resistance Breakers, J. Nanostructure, 2019, 9, 376–383 CAS.
- S. B. Koparde and D. K. Gaikwad, Antibacterial Potential of Green Silver Nanoparticles Synthesized from Medicinal Plant Terminalia arjuna, Int. J. Res. Biosci. Agric. Technol., 2017, 258–260 Search PubMed.
- S. Yallappa and J. Manjanna, Biological Evaluation of Silver Nanoparticles Obtained from T. arjuna Bark Extract as Both Reducing and Capping Agent, J. Clust. Sci., 2014, 1449–1462 CrossRef CAS.
- S. Ahmed and S. Ikram, Silver Nanoparticles : One Pot Green Synthesis Using Terminalia arjuna Extract for Biological Application, J. Nanomed. Nanotechnol., 2015, 6(4), 1–6 Search PubMed.
- C. Garai, S. Nurul, H. Abir, C. Barai, S. Ghorai, S. Kumar, P. Braja and G. Bag, Green synthesis of Terminalia arjuna - conjugated palladium nanoparticles ( TA - PdNPs ) and its catalytic applications, J. Nanostructure Chem., 2018, 8, 465–472 CrossRef CAS.
- D. Gaikwad and N. Jadhav, Terminalia arjuna Transdermal Matrix Formulation Containing Different Polymer Components, Asian J. Pharm. Clin. Res., 2019, 12, 1–6 Search PubMed.
- S. Noreen, S. A. Ghumman, F. Batool, B. Ijaz, M. Basharat, S. Noureen, T. Kausar and S. Iqbal, Terminalia arjuna gum/alginate in situ gel system with prolonged retention time for ophthalmic drug delivery, Int. J. Biol. Macromol., 2020, 152(4), 1056–1067 CrossRef PubMed.
- F. E. King, J. R. Housley and T. J. King, The Chemistry of Extractives from Hardwoods, J. Chem. Soc., 1954, 16, 1392–1399 RSC.
- K. Aamir, H. Ullah, C. Faiz, M. Rejina, P. Regina, I. Waheed, G. Sethi and A. Arya, Arjunolic acid downregulates elevated blood sugar and pro-inflammatory cytokines in streptozotocin ( STZ ) -nicotinamide induced type 2 diabetic rats, Life Sci., 2022, 289, 120232 CrossRef CAS.
- M. Sumitra, P. Manikandan, D. A. Kumar, N. Arutselvan, K. Balakrishna, B. M. Manohar and R. Puvanakrishnan, Experimental myocardial necrosis in rats: Role of arjunolic acid on platelet aggregation, coagulation and antioxidant status, Mol. Cell. Biochem., 2001, 224, 135–142 CrossRef CAS PubMed.
- E. Toppo, S. Sylvester Darvin, S. Esakkimuthu, K. Buvanesvaragurunathan, T. P. Ajeesh Krishna, S. Antony Caesar, A. Stalin, K. Balakrishna, P. Pandikumar, S. Ignacimuthu and N. A. Al-Dhabi, Curative effect of arjunolic acid from Terminalia arjuna in non-alcoholic fatty liver disease models, Biomed. Pharmacother., 2018, 107, 979–988 CrossRef CAS PubMed.
- S. Khatkar, A. Nanda and S. H. Ansari, Microwave assisted extraction, optimization using central composite design, quantitative estimation of arjunic acid and arjunolic acid using HPTLC and evaluation of radical scavenging potential of stem bark of Terminalia arjuna, Nat. Prod. Sci., 2017, 23, 75–83 CrossRef CAS.
- S. C. Verma, C. L. Jain, M. M. Padhi and R. B. Devalla, Microwave extraction and rapid isolation of arjunic acid from Terminalia arjuna (Roxb. ex DC.) stem bark and quantification of arjunic acid and arjunolic acid using HPLC-PDA technique, J. Sep. Sci., 2012, 35, 1627–1633 CrossRef CAS PubMed.
- J. Ghosh, J. Das, P. Manna and P. C. Sil, Arjunolic acid, a triterpenoid saponin, prevents acetaminophen (APAP)-induced liver and hepatocyte injury via the inhibition of APAP bioactivation and JNK-mediated mitochondrial protection, Free Radic. Biol. Med., 2010, 48, 535–553 CrossRef CAS PubMed.
- M. Bhujel, G. Nageswara Rao and L. Sripada, Development of an efficient chromatographic method for the extraction and separation of arjunolic acid and asiatic acid from the heartwood of Terminalia arjuna, 2024 Search PubMed.
- B. Ma, S. Yang, J. Li, H. Ouyang, M. He, Y. Feng and T. Tan, A four-step filtering strategy based on ultra-high-performance liquid chromatography coupled to quadrupole-time-of-flight tandem mass spectrometry for comprehensive profiling the major chemical constituents of Akebiae fructus, Rapid Commun. Mass Spectrom., 2019, 33, 1464–1474 CrossRef CAS PubMed.
- M. Shimada, M. Ozawa, K. Iwamoto, Y. Fukuyama, A. Kishida and A. Ohsaki, A lanostane triterpenoid and three cholestane sterols from Tilia kiusiana, Chem. Pharm. Bull., 2014, 62, 937–941 CrossRef CAS PubMed.
- D. Pertuit, A. C. Mitaine-Offer, T. Miyamoto, C. Tanaka, S. Delemasure, P. Dutartre and M. A. Lacaille-Dubois, A new aromatic compound from the stem bark of Terminalia catappa, Nat. Prod. Commun., 2015, 10, 1005–1007 CrossRef PubMed.
- L. Wang, M. Wu, J. Wang, J. Jiang, Y. Chen and T. Li, A new chalcone glycoside from Combretum griffithii, Chem. Nat. Compd., 2014, 50, 258–260 CrossRef CAS.
- K. W. Woo, J. M. Cha, S. U. Choi and K. R. Lee, A new triterpene glycoside from the stems of Lagerstroemia indica, Arch. Pharm. Res., 2016, 39, 631–635 CrossRef CAS PubMed.
- F. X. Zhang, M. Li, Z. H. Yao, C. Li, L. R. Qiao, X. Y. Shen, K. Yu, Y. Dai and X. S. Yao, A target and nontarget strategy for identification or characterization of the chemical ingredients in Chinese herb preparation Shuang-Huang-Lian oral liquid by ultra-performance liquid chromatography–quadrupole time-of-flight mass spectrometry, Biomed. Chromatogr., 2018, 32(3), e4110 CrossRef PubMed.
- D. Zou, J. Wang, B. Zhang, S. Xie, Q. Wang, K. Xu, R. Lin and C. W. K. Lam, Analysis of chemical constituents in Wuzi-Yanzong-Wan by UPLC-ESI-LTQ-Orbitrap-MS, Molecules, 2015, 20, 21373–21404 CrossRef CAS PubMed.
- D. P. A. Gossan, A. Alabdul Magid, P. A. Yao-Kouassi, J. Josse, S. C. Gangloff, H. Morjani and L. Voutquenne-Nazabadioko, Antibacterial and cytotoxic triterpenoids from the roots of Combretum racemosum, Fitoterapia, 2016, 110, 89–95 CrossRef CAS PubMed.
- J. Wang, H. Ren, Q. L. Xu, Z. Y. Zhou, P. Wu, X. Y. Wei, Y. Cao, X. X. Chen and J. W. Tan, Antibacterial oleanane-type triterpenoids from pericarps of Akebia trifoliata, Food Chem., 2015, 168, 623–629 CrossRef CAS PubMed.
- J. Zhang, Y. Li, S. S. Chen, L. Zhang, J. Wang, Y. Yang, S. Zhang, Y. Pan, Y. Wang and L. Yang, Systems pharmacology dissection of the anti-inflammatory mechanism for the medicinal herb Folium Eriobotryae, Int. J. Mol. Sci., 2015, 16, 2913–2941 CrossRef CAS PubMed.
- B. Zhai, J. Clark, T. Ling, M. Connelly, F. Medina-Bolivar and F. Rivas, Antimalarial evaluation of the chemical constituents of hairy root culture of Bixa orellana l, Molecules, 2014, 19, 756–766 CrossRef PubMed.
- Y. Mai, Z. Wang, Y. Wang, J. Xu and X. He, Anti-neuroinflammatory triterpenoids from the seeds of Quercus serrata Thunb, Fitoterapia, 2020, 142, 104523 CrossRef CAS PubMed.
- D. Alesiani, A. Canini, B. D'Abrosca, M. DellaGreca, A. Fiorentino, C. Mastellone, P. Monaco and S. Pacifico, Antioxidant and antiproliferative activities of phytochemicals from Quince (Cydonia vulgaris) peels, Food Chem., 2010, 118, 199–207 CrossRef CAS.
- D. H. Nguyen, D. D. Le, B. T. Zhao, E. S. Ma, B. S. Min and M. H. Woo, Antioxidant compounds isolated from the roots of Phlomis umbrosa Turcz, Nat. Prod. Sci., 2018, 24, 119–124 CrossRef CAS.
- H. Yang, E. J. Jeong, J. Kim, S. H. Sung and Y. C. Kim, Antiproliferative triterpenes from the leaves and twigs of juglans sinensis on HSC-T6 cells, J. Nat. Prod., 2011, 74, 751–756 CrossRef CAS.
- C. Wang, P. Wu, S. Tian, J. Xue, L. Xu, H. Li and X. Wei, Bioactive Pentacyclic Triterpenoids from the Leaves of Cleistocalyx operculatus, J. Nat. Prod., 2016, 79, 2912–2923 CrossRef CAS.
- L. Ba Vinh, H. J. Jang, N. Viet Phong, G. Dan, K. Won Cho, Y. Ho Kim and S. Young Yang, Bioactive triterpene glycosides from the fruit of Stauntonia hexaphylla and insights into the molecular mechanism of its inflammatory effects, Bioorganic Med. Chem. Lett., 2019, 29, 2085–2089 CrossRef CAS.
- M. A. Baldé, E. Tuenter, A. Matheeussen, M. S. Traoré, P. Cos, L. Maes, A. Camara, M. S. T. Diallo, E. S. Baldé, A. M. Balde, L. Pieters and K. Foubert, Bioassay-guided isolation of antiplasmodial and antimicrobial constituents from the roots of Terminalia albida, J. Ethnopharmacol., 2021, 267, 113624 CrossRef PubMed.
- I. Zebiri, M. Haddad, L. Duca, M. Sauvain, L. Paloque, B. Cabanillas, E. Rengifo, J. B. Behr and L. Voutquenne-Nazabadioko, Biological activities of triterpenoids from Poraqueiba sericea stems, Nat. Prod. Res., 2017, 31, 1333–1338 CrossRef CAS PubMed.
- P. Masoko, L. K. Mdee, L. J. Mampuru and J. N. Eloff, Biological activity of two related triterpenes isolated from Combretum nelsonii (Combretaceae) leaves, Nat. Prod. Res., 2008, 22, 1074–1084 CrossRef CAS PubMed.
- Y. C. Ting, H. H. Ko, H. C. Wang, C. F. Peng, H. S. Chang, P. C. Hsieh and I. S. Chen, Biological evaluation of secondary metabolites from the roots of Myrica adenophora, Phytochemistry, 2014, 103, 89–98 CrossRef CAS PubMed.
- R. de Cássia Lemos Lima, L. Kato, K. T. Kongstad and D. Staerk, Brazilian insulin plant as a bifunctional food: Dual high-resolution PTP1B and α-glucosidase inhibition profiling combined with HPLC-HRMS-SPE-NMR for identification of antidiabetic compounds in Myrcia rubella Cambess, J. Funct. Foods, 2018, 45, 444–451 CrossRef.
- H. E. Elsayed, M. R. Akl, H. Y. Ebrahim, A. A. Sallam, E. G. Haggag, A. M. Kamal and K. A. El Sayed, Discovery, optimization, and pharmacophore modeling of oleanolic acid and analogues as breast cancer cell migration and invasion inhibitors through targeting Brk/Paxillin/Rac1 axis, Chem. Biol. Drug Des., 2015, 85, 231–243 CrossRef CAS PubMed.
- S. El Abdellaoui, E. Destandau, I. Krolikiewicz-Renimel, P. Cancellieri, A. Toribio, V. Jeronimo-Monteiro, L. Landemarre, P. André and C. Elfakir, Centrifugal partition chromatography for antibacterial bio-guided fractionation of Clidemia hirta roots, Sep. Purif. Technol., 2014, 123, 221–228 CrossRef CAS.
- Y. Li, J. Xu, D. Li, H. Ma, Y. Mu, D. Zheng, X. Huang and L. Li, Chemical Characterization and Hepatoprotective Effects of a Standardized Triterpenoid-Enriched Guava Leaf Extract, J. Agric. Food Chem., 2021, 69, 3626–3637 CrossRef CAS PubMed.
- Y.-H. Kuo and S.-L. Lin, Chemical Components of the Whole Herb of Mosla dianthera, Chem. Pharm. Bull., 1999, 47, 1152–1153 CrossRef CAS.
- Y. Li, J. Xu, C. Yuan, H. Ma, T. Liu, F. Liu, N. P. Seeram, Y. Mu, X. Huang and L. Li, Chemical composition and anti-hyperglycaemic effects of triterpenoid enriched Eugenia jambolana Lam. berry extract, J. Funct. Foods, 2017, 28, 1–10 CrossRef CAS.
- J. M. Tian, X. Y. Fu, Q. Zhang, H. P. He, J. M. Gao and X. J. Hao, Chemical constituents from Glochidion assamicum, Biochem. Syst. Ecol., 2013, 48, 288–292 CrossRef CAS.
- C. H. Zhang, J. Luo, T. Li, Y. Cui, M. Jin, D. L. Yao, M. S. Zheng, Z. H. Lin, J. M. Cui and G. Li, Chemical constituents from the aerial parts of Melandrium firmum, Arch. Pharm. Res., 2015, 38, 1746–1751 CrossRef CAS PubMed.
- W. Auamcharoen, A. Chandrapatya, A. Kijjoa, A. M. S. Silva and W. Herz, Chemical constituents of Duabanga grandiflora (Lythraceae), Biochem. Syst. Ecol., 2009, 37, 535–537 CrossRef CAS.
- W. Li, W. Zhou, S. H. Shim and Y. H. Kim, Chemical constituents of the rhizomes and roots of Gentiana scabra (Gentianaceae), Biochem. Syst. Ecol., 2015, 61, 169–174 CrossRef CAS.
- Y. Cao, S. Fang, Z. Yin, X. Fu, X. Shang, W. Yang and H. Yang, Chemical fingerprint and multicomponent quantitative analysis for the quality evaluation of Cyclocarya paliurus leaves by HPLC–Q–TOF–MS, Molecules, 2017, 22, 1–16 Search PubMed.
- H. Dou, R. Zhang, X. Lou, J. Jia, C. Zhou and Y. Zhao, Constituents of three species of Lagerstroemia, Biochem. Syst. Ecol., 2005, 33, 639–642 CrossRef CAS.
- T. R. Vieira, L. C. A. Barbosa, C. R. A. Maltha, V. F. Paula and E. A. Nascimento, Constituintes químicos de Melaleuca alternifolia (Myrtaceae), Quim. Nova, 2004, 27, 536–539 CrossRef CAS.
- F. F. A. Fernandes, M. A. Tomaz, C. Z. El-Kik, M. Monteiro-Machado, M. A. Strauch, B. L. Cons, M. S. Tavares-Henriques, A. C. O. Cintra, V. A. Facundo and P. A. Melo, Counteraction of Bothrops snake venoms by Combretum leprosum root extract and arjunolic acid, J. Ethnopharmacol., 2014, 155, 552–562 CrossRef CAS PubMed.
- P. C. Tu, Y. C. Liang, G. J. Huang, M. K. Lin, M. C. Kao, T. L. Lu, P. J. Sung and Y. H. Kuo, Cytotoxic and Anti-inflammatory Terpenoids from the Whole Plant of Vaccinium emarginatum, Planta Med., 2020, 86, 1313–1322 CrossRef CAS PubMed.
- Y. Zhou, B. Yang, Z. Liu, Y. Jiang, Y. Liu, L. Fu, X. Wang and H. Kuang, Cytotoxicity of triterpenes from green walnut husks of Juglans mandshurica Maxim in HepG-2 cancer cells, Molecules, 2015, 20, 19252–19262 CrossRef CAS PubMed.
- R. S. Pawar, P. Balachandran, D. S. Pasco and I. A. Khan, Cytotoxicity studies of triterpenoids from Akebia trifoliata and Clematis ligusticifolia, Acta Hortic, 2006, 720, 171–178 CrossRef CAS.
- M. B. C. Gallo, A. S. F. Marques, P. C. Vieira, M. F. D. G. F. da Silva, J. B. Fernandes, M. Silva, R. V. Guido, G. Oliva, O. H. Thiemann, S. Albuquerque and A. H. Fairlamb, Enzymatic Inhibitory Activity and Trypanocidal Effects of Extracts and Compounds from Siphoneugena densiflora O. Berg and Vitex polygama Cham, Zeitschrift fur Naturforsch. - Sect. C J. Biosci., 2008, 63, 371–382 CrossRef CAS PubMed.
- J. Gominho, R. A. Costa, A. Lourenço, T. Quilhó and H. Pereira, Eucalyptus globulus Stumps Bark: Chemical and Anatomical Characterization Under a Valorisation Perspective, Waste Biomass Valoriz., 2021, 12, 1253–1265 CrossRef CAS.
- F. Hu, X. Liao, Y. Guo, S. Yamaki, X. Li, N. Hamada, Y. Hashi and Z. Chen, Fast determination of isomeric triterpenic acids in Osmanthus fragrans (Thunb.) Lour. fruits by UHPLC coupled with triple quadrupole mass spectrometry, Food Chem., 2020, 322, 126781 CrossRef CAS PubMed.
- Z. Zhang, H. N. ElSohly, X. C. Li, S. I. Khan, S. E. Broedel, R. E. Raulli, R. L. Cihlar and L. A. Walker, Flavanone glycosides from Miconia trailii, J. Nat. Prod., 2003, 66, 39–41 CrossRef CAS.
- M. Shao, Y. Wang, X. J. Huang, C. L. Fan, Q. W. Zhang, X. Q. Zhang and W. C. Ye, Four new triterpenoids from the leaves of Psidium guajava, J. Asian Nat. Prod. Res., 2012, 14, 348–354 CrossRef CAS PubMed.
- P. Masoko, J. Picard, R. L. Howard, L. J. Mampuru and J. N. Eloff, In vivo antifungal effect of Combretum and Terminalia species extracts on cutaneous wound healing in immunosuppressed rats, Pharm. Biol., 2010, 48, 621–632 CrossRef CAS PubMed.
- G. Zengin, C. Ferrante, I. Senkardes, R. Gevrenova, D. Zheleva-Dimitrova, L. Menghini, G. Orlando, L. Recinella, A. Chiavaroli, S. Leone, L. Brunetti, C. M. N. Picot-Allain, K. R. Rengasamy and M. F. Mahomoodally, Multidirectional biological investigation and phytochemical profile of Rubus sanctus and Rubus ibericus, Food Chem. Toxicol., 2019, 127, 237–250 CrossRef CAS.
- G. M. Woldemichael, M. T. Gutierrez-Lugo, S. G. Franzblau, Y. Wang, E. Suarez and B. N. Timmermann, Mycobacterium tuberculosis Growth Inhibition by Constituents of Sapium haematospermum, J. Nat. Prod., 2004, 67, 598–603 CrossRef CAS PubMed.
- S. Parveen, M. Saleem, N. Riaz, M. Ashraf, Q. Ul-Ain, M. F. Nisar and A. Jabbar, New norterpenoids and a sphingolipid from Carissa opaca, J. Asian Nat. Prod. Res., 2016, 18, 222–231 CrossRef CAS PubMed.
- W. D. Tékapi Tsopgni, A. G. Blaise Azebaze, J. E. Mbosso Teinkela, B. L. Ndjakou, F. F. Bosyom, B. N. Tchaleu and J. C. Vardamides, New unsaturated fatty acid and other chemical constituents from the roots of Cola rostrata K. Schum. (Malvaceae), Biochem. Syst. Ecol., 2019, 86 DOI:10.1016/j.bse.2019.103913.
- A. M. A. Mohammed, P. H. Coombes, N. R. Crouch and D. A. Mulholland, Non-volatile isolates of two Heteropyxis species: A first chemotaxonomic assessment of subfamily Psiloxyloideae (Myrtaceae), Biochem. Syst. Ecol., 2009, 37, 241–243 CrossRef CAS.
- J. Feng, X. Yi, W. Huang, Y. Wang and X. He, Novel triterpenoids and glycosides from durian exert pronounced anti-inflammatory activities, Food Chem., 2018, 241, 215–221 CrossRef CAS PubMed.
- A. Qiao, Y. Wang, L. Xiang, Z. Zhang and X. He, Novel triterpenoids isolated from hawthorn berries functioned as antioxidant and antiproliferative activities, J. Funct. Foods, 2015, 13, 308–313 CrossRef CAS.
- J. S. Han, J. G. Kim, T. P. L. Le, Y. B. Cho, M. K. Lee and B. Y. Hwang, Pentacyclic triterpenes with nitric oxide inhibitory activity from Potentilla chinensis, Bioorg. Chem., 2021, 108, 104659 CrossRef CAS PubMed.
- M. Liu, D. R. Katerere, A. I. Gray and V. Seidel, Phytochemical and antifungal studies on Terminalia mollis and Terminalia brachystemma, Fitoterapia, 2009, 80, 369–373 CrossRef CAS PubMed.
- C. Liang, D. Staerk and K. T. Kongstad, Potential of Myrtus communis Linn. as a bifunctional food: Dual high-resolution PTP1B and α-glucosidase inhibition profiling combined with HPLC-HRMS and NMR for identification of antidiabetic triterpenoids and phloroglucinol derivatives, J. Funct. Foods, 2020, 64 DOI:10.1016/j.jff.2019.103623.
- B. Y. Yang, X. Yin, Y. Liu, Y. Sun, W. Guan, Y. Y. Zhou and H. X. Kuang, Terpenes and lignans from the roots of Solanum melongena L, Nat. Prod. Res., 2020, 34, 359–368 CrossRef CAS PubMed.
- A. Lourenço, A. V. Marques and J. Gominho, The identification of new triterpenoids in Eucalyptus globulus wood, Molecules, 2021, 26, 1–13 CrossRef PubMed.
- B. Diallo, R. Vanhaelen-Fastré, M. Vanhaelen, T. Konoshima, M. Takasaki and H. Tokuda, In vivo inhibitory effects of arjunolic acid derivatives on two-stage carcinogenesis in mouse skin, Phyther. Res., 1995, 9(6), 444–447 CrossRef CAS.
- I. L. Acebey-Castellon, L. Voutquenne-Nazabadioko, H. Doan Thi Mai, N. Roseau, N. Bouthagane, D. Muhammad, E. Le Magrex Debar, S. C. Gangloff, M. Litaudon, T. Sevenet, N. Van Hung and C. Lavaud, Triterpenoid saponins from Symplocos lancifolia, J. Nat. Prod., 2011, 74, 163–168 CrossRef CAS PubMed.
- R. Srivastava, A. K. Shaw and D. K. Kulshreshtha, Triterpenoids and chalcone from Syzygium samarangense, Phytochemistry, 1995, 38, 687–689 CrossRef CAS.
- M. Tene, P. Tane and J. D. Connolly, Triterpenoids and diarylheptanoids from Myrica arborea, Biochem. Syst. Ecol., 2008, 36, 872–874 CrossRef CAS.
- J. Li, C. M. Coleman, H. Wu, C. L. Burandt, D. Ferreira and J. K. Zjawiony, Triterpenoids and flavonoids from Cecropia schreberiana Miq, Biochem. Syst. Ecol., 2013, 48, 96–99 CrossRef CAS PubMed.
- Y. S. Wang, H. Zhang, K. W. Wang, T. T. Zhang and X. F. Fan, Triterpenoids and Steroids from Premna fulva, Chem. Nat. Compd., 2020, 56, 373–375 CrossRef CAS.
- J. Zhao, Triterpenoids from Rhododendron hainanense, Chem. Nat. Compd., 2013, 49, 467–469 CrossRef CAS.
- D. H. Kim, K. M. Han, I. S. Chung, D. K. Kim, S. H. Kim, B. M. Kwon, T. S. Jeong, M. H. Park, E. M. Ahn and N. I. Baek, Triterpenoids from the flower of Campsis grandiflora K. Schum. as human Acyl-CoA: Cholesterol acyltransferase inhibitors, Arch. Pharm. Res., 2005, 28, 550–556 CrossRef CAS PubMed.
- Z. Y. Huang, B. Huang, C. J. Xiao, X. Dong and B. Jiang, Two new labdane diterpenoids from the rhizomes of Isodon yuennanensis, Nat. Prod. Res., 2015, 29, 628–632 CrossRef CAS PubMed.
- R. Fan, R. R. Cheng, H. T. Zhu, D. Wang, C. R. Yang, M. Xu and Y. J. Zhang, Two new oleanane-type triterpenoids from methanolyzed saponins of Momordica cochinchinensis, Nat. Prod. Commun., 2016, 11, 725–728 Search PubMed.
- Y. B. Zhang, W. Li, Z. M. Zhang, N. H. Chen, X. Q. Zhang, J. W. Jiang, G. C. Wang and Y. L. Li, Two new triterpenoids from the roots of rhodomyrtus tomentosa, Chem. Lett., 2016, 45, 368–370 CrossRef CAS.
- D. Yang, H. Xie, B. Yang and X. Wei, Two tetrahydroisoquinoline alkaloids from the fruit of Averrhoa carambola, Phytochem. Lett., 2014, 7, 217–220 CrossRef CAS.
- F. Ullah, H. Hussain, J. Hussain, I. A. Bukhari, M. Tareq, H. Khan, M. I. Choudhary, A. H. Gilani and V. U. Ahmad, Tyrosinase Inhibitory Pentacyclic Triterpenes and Analgesic and Spasmolytic Activities of Methanol Extracts of Rhododendron collettianum, Phyther. Res., 2008, 21, 1076–1081 CrossRef PubMed.
- M. Ahmad, N. Muhammad, M. Ahmad, M. A. Lodhi, N. Jehan, Z. Khan, R. Ranjit, F. Shaheen and M. I. Choudhary, Urease inhibitor from Datisca cannabina Linn, J. Enzyme Inhib. Med. Chem., 2008, 23, 386–390 CrossRef CAS PubMed.
- R. S. Bhakuni, Y. N. Shukla, A. K. Tripathi, V. Prajapati and S. Kumar, Insect growth inhibitor activity of arjunolic acid isolated from Cornus capitata, Phyther. Res., 2002, 16, 2000–2002 Search PubMed.
- E. Toppo, S. Sylvester Darvin, S. Esakkimuthu, K. Buvanesvaragurunathan, T. P. Ajeesh Krishna, S. Antony Caesar, A. Stalin, K. Balakrishna, P. Pandikumar, S. Ignacimuthu and N. A. Al-Dhabi, Curative effect of arjunolic acid from Terminalia arjuna in non-alcoholic fatty liver disease models, Biomed. Pharmacother., 2018, 107, 979–988 CrossRef CAS PubMed.
- P. Manna, M. Sinha and P. C. Sil, Protection of arsenic-induced hepatic disorder by arjunolic acid, Basic Clin. Pharmacol. Toxicol., 2007, 101, 333–338 CrossRef CAS PubMed.
- B. G. Bag, P. P. Dey, S. K. Dinda, W. S. Sheldrick and I. M. Oppel, A simple route for renewable nano-sized arjunolic and asiatic acids and self-assembly of arjuna-bromolactone, Beilstein J. Org. Chem., 2008, 4, 2–6 Search PubMed.
- J. Ghosh, J. Das, P. Manna and P. C. Sil, The protective role of arjunolic acid against doxorubicin induced intracellular ROS dependent JNK-p38 and p53-mediated cardiac apoptosis, Biomaterials, 2011, 32, 4857–4866 CrossRef CAS PubMed.
- T. Bansal, E. Chatterjee, J. Singh, A. Ray, B. Kundu, V. Thankamani, S. Sengupta and S. Sarkar, Arjunolic acid, a peroxisome proliferator-activated receptor agonist, regresses cardiac fibrosis by inhibiting non-canonical TGF- signaling, J. Biol. Chem., 2017, 292, 16440–16462 CrossRef CAS PubMed.
- P. Manna, M. Sinha and P. C. Sil, Arsenic-induced oxidative myocardial injury: Protective role of arjunolic acid, Arch. Toxicol., 2008, 82, 137–149 CrossRef CAS PubMed.
- M. M. Hasan, P. Madhavan, N. A. Ahmad Noruddin, W. K. Lau, Q. U. Ahmed, A. Arya and Z. A. Zakaria, Cardioprotective effects of arjunolic acid in LPS-stimulated H9C2 and C2C12 myotubes via the My88-dependent TLR4 signaling pathway, Pharm. Biol., 2023, 61, 1135–1151 CrossRef CAS PubMed.
- M. M. H. Al-Gayyar, A. Al Youssef, I. O. Sherif, M. E. E. Shams and A. Abbas, Protective effects of arjunolic acid against cardiac toxicity induced by oral sodium nitrite: Effects on cytokine balance and apoptosis, Life Sci., 2014, 111, 18–26 CrossRef CAS PubMed.
- H. Elsawy, M. Almalki, O. Elmenshawy and A. Abdel-Moneim, In vivo evaluation of the protective effects of arjunolic acid against lipopolysaccharide-induced septic myocardial injury, PeerJ, 2022, 10, 1–16 CrossRef PubMed.
- A. S. Ramesh, J. G. Christopher, R. Radhika, C. R. Setty and V. Thankamani, Isolation, characterisation and cytotoxicity study of arjunolic acid from Terminalia arjuna, Nat. Prod. Res., 2012, 26, 1549–1552 CrossRef CAS PubMed.
- S. Manna, A. Dey, R. Majumdar, B. G. Bag, C. Ghosh and S. Roy, Self assembled arjunolic acid acts as a smart weapon against cancer through TNF- α mediated ROS generation, Heliyon, 2020, 6, 1–9 CrossRef PubMed.
- N. M. Elsherbiny and M. M. H. Al-gayyar, Anti-tumor activity of arjunolic acid against Ehrlich Ascites Carcinoma cells in vivo and in vitro through blocking TGF- b type 1 receptor, Biomed. Pharmacother., 2016, 82, 28–34 CrossRef CAS.
- P. Manna, M. Sinha and P. C. Sil, Protective role of arjunolic acid in response to streptozotocin-induced type-I diabetes via the mitochondrial dependent and independent pathways, Toxicology, 2009, 257, 53–63 CrossRef CAS PubMed.
- P. Manna, J. Das, J. Ghosh and P. C. Sil, Contribution of type 1 diabetes to rat liver dysfunction and cellular damage via activation of NOS, PARP, IκBα/NF-κB, MAPKs, and mitochondria-dependent pathways: Prophylactic role of arjunolic acid, Free Radic. Biol. Med., 2010, 48, 1465–1484 CrossRef CAS PubMed.
- L. M. Goncalves-Neto, F. B. Ferreira, L. Souza, C. dos Santos, A. C. Boschero, V. A. Facundo, A. R. S. Santos, E. A. Nunes and A. Rafacho, Disruption of glucose tolerance caused by glucocorticoid excess in rats is partially prevented, but not attenuated, by arjunolic acid, Indian J. Experiemental Biol., 2014, 52, 972–982 Search PubMed.
- C. Fang, Y. Teng, Y. Wang, Y. Zhao and X. Zheng, Arjunolic acid from Cyclocarya paliurus selectively inhibits glucagon secretion from α cells and ameliorates diabetes via ephrin-A1 and EphA4 interaction, J. Funct. Foods, 2022, 99, 105323 CrossRef CAS.
- X. Zhang, Y. Ji, L. Zhu, Z. Wang and C. Fang, Arjunolic acid from Cyclocarya paliurus ameliorates diabetic retinopathy through AMPK/mTOR/HO-1 regulated autophagy pathway, J. Ethnopharmacol., 2022, 284, 1–15 Search PubMed.
- K. Aamir, G. Sethi, M. Rejina, C. Faiz, P. Regina, S. D. Sarker and A. Arya, Arjunolic acid modulate pancreatic dysfunction by ameliorating pattern recognition receptor and canonical Wnt pathway activation in type 2 diabetic rats, Life Sci., 2023, 327, 121856 CrossRef CAS PubMed.
- S. R. A, G. Christopher and V. Thankamani, In-vitro study on antioxidant property of Arjunolic acid, J. Pharmacogn. Herb. Formul., 2010, 1, 1–11 Search PubMed.
- M. Maity, H. Perveen, M. Dash, S. Jana, S. Khatun, A. Dey and A. K. Mandal, Arjunolic Acid Improves the Serum Level of Vitamin B 12 and Folate in the Process of the Attenuation of Arsenic Induced Uterine Oxidative Stress, Biol. Trace Elem. Res., 2018, 182, 78–90 CrossRef CAS PubMed.
- M. Sinha, P. Manna and P. C. Sil, Protective Effect of Arjunolic Acid against Arsenic-Induced Oxidative Stress in Mouse Brain, J. Biochen Mol. Toxicol., 2008, 22, 15–26 CrossRef CAS PubMed.
- P. Manna, M. Sinha and P. C. Sil, Protection of arsenic-induced testicular oxidative stress by arjunolic acid, Redox Rep., 2008, 13, 67–77 CrossRef CAS PubMed.
- L. Yaidikar and S. Thakur, Arjunolic acid, a pentacyclic triterpenoidal saponin of Terminalia arjuna bark protects neurons from oxidative stress associated damage in focal cerebral ischemia and reperfusion, Pharmacol. Reports, 2015, 67, 890–895 CrossRef CAS PubMed.
- S. Miriyala, M. Chandra, B. Maxey, A. Day, D. K. St and M. Panchatcharam, Arjunolic acid ameliorates reactive oxygen species via inhibition of p47 phox -serine phosphorylation and mitochondrial dysfunction, Int. J. Biochem. Cell Biol., 2015, 68, 70–77 CrossRef CAS PubMed.
- P. Manna, J. Ghosh, J. Das and P. C. Sil, Streptozotocin induced activation of oxidative stress responsive splenic cell signaling pathways : Protective role of arjunolic acid, Toxicol. Appl. Pharmacol., 2010, 244, 114–129 CrossRef CAS PubMed.
- S. Pal, A. Sarkar, P. B. Pal and P. C. Sil, Protective effect of arjunolic acid against atorvastatin induced hepatic and renal pathophysiology via MAPK, mitochondria and ER dependent pathways, Biochimie, 2015, 112, 20–34 CrossRef CAS PubMed.
- I. O. Sherif, Hepatoprotective effect of arjunolic acid against cisplatin-induced hepatotoxicity: Targeting oxidative stress, inflammation, and apoptosis, J. Biochem. Mol. Toxicol., 2021, 35, 1–8 CrossRef PubMed.
- X. Zheng, X. G. Zhang, Y. Liu, L. P. Zhu, X. S. Liang, H. Jiang, G. F. Shi, Y. Y. Zhao, Z. W. Zhao, Y. Teng, K. Pan, J. Zhang and Z. Q. Yin, Arjunolic acid from Cyclocarya paliurus ameliorates nonalcoholic fatty liver disease in mice via activating Sirt1/AMPK, triggering autophagy and improving gut barrier function, J. Funct. Foods, 2021, 86, 104686 CrossRef CAS.
- Y. Fan, C. Zhang, G. Zheng, S. Wu, Y. Wang and J. Bian, Ameliorative and anti-arthritic potential of arjunolic acid against complete Freund's adjuvant-induced arthritis in rats, Trop. J. Pharm. Res., 2020, 19, 1933–1939 CrossRef CAS.
- A. Alyoussef, Arjunolic acid protects against DNCB-induced atopic dermatitis-like symptoms in mice by restoring a normal cytokine balance, Eur. Cytokine Netw., 2015, 26, 38–45 Search PubMed.
- Z. Zhang, L. Zuo, X. Song, L. Wang, Y. Zhang, Y. Cheng, J. Huang, T. Zhao, Z. Yang, H. Zhang, J. Li, X. Zhang, Z. Geng, Y. Wang, S. Ge and J. Hu, Arjunolic acid protects the intestinal epithelial barrier, ameliorating Crohn's disease-like colitis by restoring gut microbiota composition and inactivating TLR4 signalling, Phytomedicine, 2024, 123, 155223 CrossRef CAS PubMed.
- E. O. Lynda, N. E. Kingsley, O. M. Obukohwo, B. A. Benneth, E. Victor, O. I. Simon, E. Agbonifo-Chijiokwu and O. B. Oghenetega, Arjunolic acid reverses fluoxetine-induced alterations in testicular steroidogenic enzymes and membrane bound ionic pump imbalance through suppression of oxido-inflammatory stress and apoptosis, J. Bras. Reprod. Assist., 2024, 28, 66–77 Search PubMed.
- Y. Yang, Y. Lai, X. Tong, Z. Li, Y. Cheng and L. Tian, Arjunolic acid ameliorates lipopolysaccharide-induced depressive behavior by inhibiting neuroinflammation via microglial SIRT1/AMPK/Notch1 signaling pathway, J. Ethnopharmacol., 2024, 330, 118225 CrossRef CAS PubMed.
- M. V. V. Prasad, N. Anbalagan, A. Patra, G. Veluchamy and K. Balakrishna, Antiallergic and anti-asthmatic activities of the alcoholic extract of Terminalia arjuna and arjunolic acid, Nat. Prod. Sci., 2004, 10, 240–243 CAS.
- J. D. Djoukeng, E. Abou-Mansour, R. Tabacchi, A. L. Tapondjou, H. Bouda and D. Lontsi, Antibacterial triterpenes from Syzygium guineense (Myrtaceae), J. Ethnopharmacol., 2005, 101, 283–286 CrossRef CAS PubMed.
- M. Yasasve, P. M. Kumaran, D. Manoj, M. Kanmani and A. S. Ramesh, Evaluation of arjunolic acid against Brucella melitenis and in vitro cytotoxic study of lung adenocarcinomic cell line (A549), Indian J. Exp. Biol., 2022, 60, 510–513 CAS.
- Q. Zhang, H. N. ElSohly, X. C. Li and L. A. Walker, A new triterpene from Leandra chaetodon, Planta Med., 2003, 69, 582–584 CrossRef CAS PubMed.
- B. G. Bag, G. C. Maity and S. R. Pramanik, A terpenoid-based gelator: The first arjunolic acid-derived organogelator for alcohols and mixed solvents, Supramol. Chem., 2005, 17, 383–385 CrossRef CAS.
- B. G. Bag and R. Majumdar, Vesicular self-assembly of a natural triterpenoid arjunolic acid in aqueous medium: study of entrapment properties and in situ generation of gel-gold nanoparticle hybrid material, RSC Adv., 2015, 95, 53327–53334 Search PubMed.
- R. Majumdar and B. G. Bag, Evolution of Vesicular Self-Assemblies of the Salts of a Natural Triterpenoid Arjunolic Acid into Superstructured Ambidextrous Gels and Study of Their Entrapment Properties, ChemistrySelect, 2018, 3, 951–957 CrossRef.
- B. M. F. Gonçalves, J. A. R. Salvador, S. Marín and M. Cascante, Synthesis and biological evaluation of novel asiatic acid derivatives with anticancer activity, RSC Adv., 2016, 6, 3967–3985 RSC.
- K. N. Devaraj Reddy, M. P. Sadashiva, M. Mahesh, B. K. Bettadaiah and N. P. Geetha, HPLC method for determination of acetylated arjunolic acid-a derivative of arjunolic acid from Terminalia arjuna and their antioxidant activity, Int. J. Res. Phytochem. Pharmacol., 2012, 2, 188–193 Search PubMed.
- B. G. Bag, S. R. Pramanik and G. C. Maity, Arjunolic acid in molecular recognition: First synthesis and cation binding studies of a novel arjuna-18-crown-6, Supramol. Chem., 2005, 17, 297–302 CrossRef CAS.
- B. G. Bag and S. K. Dinda, Arjunolic acid: A renewable template in supramolecular chemistry and nanoscience, Pure Appl. Chem., 2007, 79, 2031–2038 CrossRef CAS.
- B. G. Bag, R. Majumdar, S. K. Dinda, P. P. Dey, G. C. Maity, V. A. Mallia and R. G. Weiss, Self-assembly of ketals of arjunolic acid into vesicles and fibers yielding gel-like dispersions, Langmuir, 2013, 29, 1766–1778 CrossRef CAS PubMed.
- B. Diallo, M. Vanhaelen, R. Vanhaelen-Fastre, T. Konoshima and M. Kozuka, Studies on inhibitors of skin tumour promotion. Inhibitory effects of triterpenes from Cochlospermum tznctorzum on Epstein-Barr virus activation., J. Nat. Prod., 1989, 52, 879–881 CrossRef CAS PubMed.
- B. Diallo, R. Vanhaelen-Fastré, M. Vanhaelen, T. Konoshima, M. Takasaki and H. Tokuda, In vivo inhibitory effects of arjunolic acid derivatives on two-stage carcinogenesis in mouse skin, Phyther. Res., 1995, 9, 444–447 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |