Open Access Article
Open Access ArticleDeprotonation- and TICT-triggered ultra-large Stokes shift of aqueous NIR-emissive hydroxylstryryl-pyridinium derivatives
Xiaolong Luabc,
Liyu Xiongbc,
Zhihan Zhangbc,
Haolong Yangbc,
Enmin Li *def and
Hefeng Zhang
*def and
Hefeng Zhang *bc
*bc
aMarine Science Institute, College of Science, Shantou University, Shantou 515063, China
bKey Laboratory for Preparation and Application of Ordered Structural Materials of Guangdong Province, Department of Chemistry, College of Chemistry and Chemical Engineering, Shantou University, Shantou 515063, China. E-mail: hfzhang@stu.edu.cn
cGuangDong Engineering Technology Research Center of Advanced Polymer Synthesis, College of Chemistry and Chemical Engineering, Shantou University, Shantou 515063, China
dThe Key Laboratory of Molecular Biology for High Cancer Incidence Coastal Chaoshan Area, Department of Biochemistry and Molecular Biology, Shantou University Medical College, Shantou 515041, Guangdong, China. E-mail: nmli@stu.edu.cn
eThe Laboratory for Cancer Molecular Biology, Shantou Academy Medical Sciences, Shantou 515041, China
fChaoshan Branch of State Key Laboratory for Esophageal Cancer Prevention and Treatment, Shantou 515063, China
First published on 14th April 2026
Abstract
Hydroxystyryl-pyridinium derivatives serving as fluorescent dyes with water-soluble conjugated skeletons exhibited remarkably large Stokes shifts of up to 404 nm and tunable deep-red to near-infrared (NIR) emissions, which are attributed to the enhanced charge transferring triggered by excited-state deprotonation of the phenol group and twist intramolecular charge transfer.
Organic fluorescent materials hold considerable promise across diverse fields such as chemical biosensing, optoelectronic devices, and bioimaging.1–6 Among them, a large Stokes shifts is critically helpful for fluorescent dyes as it effectively minimizes self-absorption and background interference, thereby significantly enhancing the signal-to-noise ratio and detection sensitivity in applications such as high-contrast bioimaging and anti-counterfeiting.5–8 However, the widespread adoption of conventional fluorescent dyes including established families like rhodamine,9,10 BODIPY,11–13 and cyanine14–17 are limited by their small Stokes shift, typically less than 50 nm. The narrow gap between excitation and emission wavelengths leads to severe self-absorption and re-excitation, causing signal crosstalk and compromising the sensitivity and accuracy of detection and anti-counterfeiting technologies.18–21 Furthermore, issues such as low luminescence efficiency in aggregate states and poor solubility in aqueous environments significantly restrict their practical utility in biological and environmental settings.13,17,22–25
To address the limitations of small Stokes shifts, several molecular design strategies have been explored. A prominent approach involves the construction of donor–acceptor (D–A) or donor–π–acceptor (D–π–A) systems, where intramolecular charge transfer (ICT) can create a significant difference in electron cloud distribution and dipole moment between the excited and ground states and thus cause moderate increase in the Stokes shift.5,26–30 Moreover, the construction of the D–π–A structure is one of the most powerful and widely used strategy to extend emission wavelengths. More sophisticated mechanisms to enhance Stokes shift, primarily including excited-state intramolecular proton transfer (ESIPT),31–38 which induces a substantial energy loss via rapid proton transfer after excitation, and the formation of a twisted intramolecular charge transfer (TICT)38–44 state in donor–π–acceptor systems, where significant structural reorganization leads to a large Stokes shift. Besides, ICT and ESIPT mechanisms can effectively increase the Stokes shift and have been tremendously important for the development of fluorescence chemical sensors and imaging probes,29–33 the TICT mechanism plays a core role in fields such as aggregation-induced emission (AIE),38 crystallization-induced emission enhancement (CIEE),39 and metal–organic frameworks (MOFs),45 owing to its combination of significant molecular conformational reorganization and charge transfer characteristics. As an example, Peng40 et al. reported a viscosity-responsive endoplasmic reticulum targeting NIR fluorescent probe HBT-PP based on the twisted intramolecular charge transfer (TICT) and the excited state intramolecular proton transfer (ESIPT), which exhibited a Stokes shift up to 320 nm. In addition, to manipulate a single emitter unit, incorporating two fluorophores with short and length emission wavelength, respectively, into fluorescence resonance energy transfer (FRET) cassettes or designing specific molecular skeletons can also achieve a large “pseudo”-Stokes shift determined by the energy difference between the donor's absorption and the acceptor's emission.45–47 Cui45 et al. incorporated the photoisomerizable molecule spiropyran as an energy transfer intermediate, and two fluorescent dyes coumarin 153 (Cou153) and methylene blue (MB) within a metal–organic frameworks (MOFs), which achieving the dual-color dynamic luminescent material with NIR fluorescence at 700 nm and a maximum Stokes shift of 300 nm.
Beyond molecular engineering, strategies operating at the aggregate level, such as forming J-aggregates and incorporating aggregation-induced emission (AIE) moieties, provide effective pathways to amplify the Stokes shift. J-aggregation significantly enhances the Stokes shift primarily through two interconnected mechanisms, the distinctive “head-to-tail” slip-stacked molecular packing and the subsequent facilitation of excited-state structural relaxation and intermolecular charge transfer. Furthermore, J-aggregated state can restrict intramolecular motions that typically dissipate energy non-radiatively in single molecules, thereby channeling more energy into excited-state geometric relaxation, which further enlarges the energy difference between absorption and emission.48–50 Similarly, introduction of aggregation-induced emission (AIE) groups like tetraphenylethene can significantly enhance the Stokes shift by restricting intramolecular motion (RIM).34,37,42,43 However, active intramolecular rotations/vibrations of AIEgenic moieties in solution provide dominant non-radiative decay pathways, resulting in weak emission and a convoluted Stokes shift. In addition, molecular encapsulation strategies, such as the incorporation of fluorescent molecules within metal–organic frameworks (MOFs) or cyclodextrins, can effectively enhance the Stokes shift of the fluorophores by constraining intramolecular motion and promoting excited-state proton transfer processes.45,50,51 Despite sustained efforts, achieving exceptionally large Stokes shifts greater than 400 nm in organic fluorophores, especially in small molecules and UV-visible region, remains a significant challenge, underscoring the need for novel design strategies.
Inspired by our recent reported deprotonation-caused red-shifting of emission in hydroxystyryl-pyridinium (SP-OH) dyes, herein, a series of SP-OH derivatives were developed, and an ultra-large Stokes shift up to 404 nm were discovered.
The SP-OH dyes were synthesized via a Knoevenagel condensation catalysed by ammonium acetate (Scheme 1). As an example, the SP-2-NO2-5-OH was prepared by coupling commercially available 5-hydroxy-2-nitrobenzaldehyde with pre-synthesized 1-(2-hydroxyethyl)-4-methylpyridinium bromide. The same synthetic strategy was applied to afford the full library of SP-X-Py derivatives. All intermediates and final products were fully characterized by 1H and 13C nuclear magnetic resonance (NMR) spectroscopy and high-resolution mass spectrometry (HRMS) (see Experimental section in SI, Fig. S13–S26). In the SP-OH dyes skeleton, the phenol group serves as an electron-donor (D), the nitro group attaching onto the phenyl and the vinyl-bridged pyridinium behave as electron-acceptors (A). The conjugated D–π–A skeleton promised long-wavelength emission due to the intramolecular charge-transfer.
All the SP-OH dyes showed significant absorptions and fluorescent emissions in various organic solvents (c = 10−5 M). The full UV-Vis absorption spectra of all derivatives in different solvents are presented in Fig. S1, which reveals the broad absorption characteristics of SP-OH dyes in the UV to visible range; corresponding fluorescence emission spectra are shown in Fig. S2. The pH-dependent UV-Vis absorption and fluorescence emission spectra of SP-OH dyes in aqueous solution are shown in Fig. S3 and S4, which directly reflect the deprotonation behavior of phenolic hydroxyl group under different pH conditions (Fig. 1, S1, S2, Tables 1 and S1–S4). The absorption and fluorescence emission spectra of SP-OH dyes were first investigated in dimethylformamide (DMF) solutions (c = 10−5 M) (Fig. 1). All derivatives exhibited broad absorption bands in the UV to visible range and red emissions with large Stokes shifts (Table 1). Practically, SP-2-NO2-5-OH-Py showed main absorption at 304 nm, with two side peaks at 331 nm and 422 nm, and the emission at 600 nm with a large Stokes shift up to 296 nm and QY of 6.6%. In contrast, the SP-5-NO2-2-OH-Py also showed 3 absorption peaks, but with the main peak at 513 nm, and red-shifted emission at 642 nm with QY of 9.2%, indicating significant influence of positional effect on their absorption and emission behaviours. To further disclose the role of phenolic hydroxyl group, SP-2-NO2-Py and SP-2-NO2-5-OMe-Py were designed as the blank control in which the hydroxyl group was eliminated and etherified, respectively. SP-2-NO2-Py absorbed at 351 nm and an emission peak at 591 nm, resulting in a Stokes shift of 240 nm. SP-2-NO2-5-OMe-Py showed broad absorption at 317 nm and emitted at 555 nm, with a shift of 238 nm. The molar extinction coefficients (ε) ranged from 4.08 to 5.86 × 104 M−1 cm−1, indicating strong light-absorbing ability, while the absolute fluorescence quantum yields (QY) varied from 2.6% to 14.7%. The low QYs are attributed to non-radiative decay pathways such as molecular vibrations or rotation.
 | ||
| Fig. 1 Normalized absorption and fluorescent emission spectra in DMF (c = 10−5 M). Green: donor (D); red: acceptor (A). | ||
| Samplea | λabs/nm | λex/nm | λem/nm | Stokes shift/nm | ε × 104/M−1 cm−1 | QY/% |
|---|---|---|---|---|---|---|
| a Absorptions, fluorescent emission, molar extinction coefficients (ε) and absolute photoluminescence quantum yields (QY) (integrating sphere method) were measured in DMF solution (c = 10−5 M). | ||||||
| SP-2-NO2-5-OH-Py | 304 | 360 | 600 | 296 | 5.86 | 6.6 |
| SP-5-NO2-2-OH-Py | 513 | 385 | 642 | 129 | 3.04 | 9.2 |
| SP-2-NO2-Py | 351 | 410 | 591 | 240 | 4.08 | 2.6 |
| SP-2-NO2-5-OMe-Py | 317 | 393 | 555 | 238 | 5.64 | 14.7 |
To disclose effect of potential deprotonation of the phenolic hydroxyl group, the absorption and emission of SP-OH dyes were investigated in solvents of varying polarity, including chloroform (CHCl3), dichloromethane (DCM), acetonitrile (MeCN), DMF and DMSO, etc. (Fig. 2). In contrast to SP-5-NO2-2-OH-Py which exhibited absorption at longest wavelength among the dyes, the SP-2-NO2-5-OH-Py showed unexpected absorption at shortest wavelength (<310 nm). Interestingly, both SP-5-NO2-2-OH-Py and SP-2-NO2-5-OH-Py exhibited longer emission wavelengths than that of their analogues without hydroxyl group. The increase of solvent polarity caused red shifting of the emission. For example, emission wavelength SP-2-NO2-5-OH-Py extended from 585 nm to 610 nm. The short-wavelength absorption and long-wavelength emission promised large Stokes shift.
The fluorescence performance in aqueous media is particularly important for biological applications. In water, the SP-OH dyes maintained large Stokes shifts, as detailed in Fig. 3 and Table 2. Especially, SP-2-NO2-5-OH-Py showed absorption at 305 nm (UV region) and NIR emission at 709 nm and an ultra-large Stokes shift of up to 404 nm is achieved which is one of the largest reported for organic fluorophores. Besides, after 180 min of continuous light irradiation, SP-2-NO2-5-OH-Py can still maintain 40% of its fluorescence intensity, indicating that it has good photostability (Fig. S5). SP-5-NO2-2-OH-Py exhibited two absorption peaks at 319 nm and 366 nm, respectively. And it showed red emission at 608 nm and large Stokes shifts of 289 nm and 242 nm. In contrast, SP-2-NO2-Py showed a smaller shift of 155 nm (absorption at 313 nm, emission at 468 nm), emphasizing the critical effect of phenolic hydroxyl group, which further proved the results showed by SP-2-NO2-5-OMe-Py with Stokes shift of 234 nm. The molar extinction coefficients in water remained high (e.g., 7.06 × 104 M−1 cm−1 for SP-2-NO2-5-OH-Py), but quantum yields decreased to 0.8–4.4% due to strong interaction between dyes and water molecules.
 | ||
| Fig. 3 Normalized absorption and fluorescent emission spectra in water (c = 10−5 M). Green: donor (D); red: acceptor (A). | ||
| Samplea | λabs/nm | λex/nm | λem/nm | Stokes shift/nm | ε × 104/M−1 cm−1 | QY/% |
|---|---|---|---|---|---|---|
| a Absorptions, fluorescent emission, molar extinction coefficients (ε) and absolute photoluminescence quantum yields (QY) (integrating sphere method) were measured in aqueous solution (c = 10−5 M). | ||||||
| SP-2-NO2-5-OH-Py | 305 | 450 | 709 | 404 | 7.06 | 1.9 |
| SP-5-NO2-2-OH-Py | 319/366 | 380 | 608 | 289/242 | 2.70/2.53 | 4.4 |
| SP-2-NO2-Py | 313 | 345 | 468 | 155 | 3.09 | 0.8 |
| SP-2-NO2-5-OMe-Py | 319 | 393 | 553 | 234 | 8.29 | 4.3 |
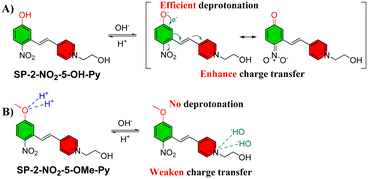
Due to the determinative effect of pH over the deprotonation of phenolic hydroxyl group, the fluorescence behaviors of SP-OHs were investigated in aqueous solutions with varying pH (Fig. 4, S3, S4 and Tables S5–S8). In alkali solutions, deprotonation of phenol group and electrostatic interaction between quaternary ammonium moiety and hydroxide coexist, which enhance and suppress electron transferring across the D–π–A conjugated skeleton of SP-OH, respectively. It is believed that in SP-OH containing a phenolic hydroxyl group, the proton-transfer pathway dominates and thus lead to the long-wavelength emissions.
In case of SP-2-NO2-5-OH-Py and SP-5-NO2-2-OH-Py, due to pH-dependent protonation, both two dyes showed absorption at ∼411 nm and 441 nm at high pH, which significantly blue-shifted to the UV region at ∼305 nm and 350 nm at low pHs. The long-wavelength absorption at high pH is attributed to the neutralization of acidic phenolic hydroxyl group. The deprotonation generated a negatived charge which further greatly prompted charge transfer induced by the positive-charged pyridinium group. In contrast, after elimination of hydroxyl group, the resulting dyes of SP-2-NO2-5-OMe-Py and SP-2-NO2-Py as control showed slightly red-shift with the pH decreasing, which is caused by the weaken charge transfer by the large amount of OH− with strong negative charge (Scheme 2). Surprisingly, the emission of both of SP-2-NO2-5-OMe-Py and SP-2-NO2-5-OH-Py are not effected by the pH, which clearly indicated a consist structure at S1 state, suggesting a deprotonation process at excited state.
Upon photoexcitation, the deprotonation and twisted intramolecular charge transfer (TICT) process of the SP-2-NO2-5-OH-Py molecule form a synergistic effect of mutual promotion, ultimately leading to an ultra-large Stokes shift of 404 nm.
To prove the mechanism for the ultra-large Stokes shifts of SP-OH dyes, especially SP-2-NO2-5-OH-Py, density functional theory (DFT) and time-dependent density functional theory (TD-DFT) calculations were performed at B3LYP/6-311+G(d,p) level52 (Fig. S7–S10). As shown in Fig. 5 and S11, all the SP-OH dyes have significantly distorted dihedral angle between the phenol and pyridinium rings (75.2–83.6°) in the excited state (S1). The large twisted structure endows the SP-OH dyes with the characteristic of exhibiting significant fluorescence emission under solid-state conditions (Fig. S6). In ground state (S0), in contrast to SP-5-NO2-2-OH-Py showing planar structure, due to the large steric hindrance effect of the nitro group, dihedral angles were observed in all SP-2-NO2-5-OH-Py (45.8°), SP-2-NO2-5-OMe-Py (37.7°), and SP-2-NO2-Py (35.5°). The highest occupied molecular orbital (HOMO) localized on the electron donor in both S0 and S1 states, whereas the lowest unoccupied molecular orbital (LUMO) on electron acceptor. The twisted intramolecular charge transfer (TICT) effect caused large Stokes shift of all the nitro-contained dyes.
 | ||
| Fig. 5 The distorted dihedral angle between the phenol and pyridinium rings of SP-OH dyes in the ground state (S0) and the excited state (S1). | ||
In addition to comparison of dihedral angle, the changes of bond length of SP-OH dyes before and after excitation were investigated. Taking SP-2-NO2-5-OH-Py and SP-5-NO2-2-OH-Py as an example, after excitation, the bond length of O–H in phenol group increases from 0.964 Å to 0.970 Å and from 0.965 Å to 0.970 Å (Fig. 6). The deprotonation in excited state leads to increasing of the electron density on oxygen atom and thus enhancement of charge transferring to the pyridinium moiety, which promises these molecules red-shifted emission and a large Stokes.
Based on the above analysis, the dual synergistic effect of the TICT and the deprotonation effect enable SP-2-NO2-5-OH-Py to have the largest Stokes shift (>400 nm).
Conventional invisible fluorescent inks are typically limited to blue emission. Developing long-wavelength versions is challenging because they must remain invisible under daylight, which requires negligible absorption in the visible range. The SP-OH-Py dyes overcome this limitation due to their exceptionally large Stokes shift. This enables the preparation of an invisible ink that shows no color in daylight but emits bright orange fluorescence under UV excitation. This distinct optical signature is difficult to replicate with existing technologies, thereby significantly enhancing anti-counterfeiting security (Fig. S12).
In summary, a series of hydroxystyryl-pyridinium derivatives were facilely synthesized. These dyes show remarkable optical properties in aqueous solution, among which SP-2-NO2-5-OH-Py exhibits an ultra-large Stokes shift up to 404 nm with UV absorption and NIR emission. Mechanistic studies reveal that the large Stokes shift arises from the synergistic effect of excited-state deprotonation and twisted intramolecular charge transfer (TICT). Benefiting from their unique optical features, these dyes can be used as invisible anti-counterfeiting inks with high security. This work provides a new strategy for constructing large Stokes shift fluorophores and offers promising candidates for bioimaging, sensing, and anti-counterfeiting applications.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: materials, experimental details, data details, computational details, NMR spectra, and optical images of anti-counterfeiting. See DOI: https://doi.org/10.1039/d6ra02237e.Acknowledgements
This work was supported by the National Natural Science Foundation of China (82273108) and Guangdong provincial Scientific and Technological Innovation Program (STKJ2023073, 2024A1515012747).References
- J. W. Zhang, K. C. Zhang, K. Wang, B. Wang, S. Y. Zhu, H. P. Qian, Y. M. Ma, M. L. Zhang, T. Y. Liu, P. Chen, Y. Shen, Y. Z. Fu, S. L. Fang, X. X. Zhang, P. Zou, W. L. Deng, Y. Ma and Z. X. Chen, Nat. Methods, 2025, 22, 1276–1287 CrossRef CAS PubMed.
- R. J. Ming, Z. X. Xue, Y. L. Xu, Z. H. Zhang, Z. X. Chen, J. S. Miao and C. L. Yang, Angew. Chem., Int. Ed., 2025, 64, e202516696 CrossRef CAS PubMed.
- J. Y. Wang, H. R. Zheng, X. W. Xiao and X. F. Luo, Chem. Commun., 2025, 61, 7434–7437 RSC.
- Q. N. Liu, Z. Y. Li, Y. J. Huang, Z. N. Lin, X. C. Shen and H. Chen, Chem. Sci., 2025, 16, 23394–23404 RSC.
- S. Y. Wu, Z. C. Ban, H. Tang, N. Ma, X. Y. Ran, Q. Zhou, Y. H. Zhang, Z. Y. Wang and X. Q. Yu, Chem. Commun., 2025, 61, 10574–10577 RSC.
- S. A. H. Ahmed, X. Gao, Y. Zheng, H. T. Wang, S. Zeng, H. Liu and L. H. Qian, Anal. Chim. Acta, 2025, 1367, 344299 CrossRef CAS PubMed.
- R. P. Bhatta, S. Sumit, V. Kachwal, V. Vishwakarma, A. R. Choudhury and I. R. Laskar, J. Mater. Chem. C, 2024, 12, 12906–12918 RSC.
- P. Lu, M. D. Zhou, Y. J. Yuan, Y. Yan and H. L. Zhang, Adv. Opt. Mater., 2024, 12, 2400359 CrossRef CAS.
- J. M. Ritz, A. Khakimzhan, J. J. Dalluge, V. Noireaux and E. M. Puchner, J. Am. Chem. Soc., 2025, 147, 7588–7596 CrossRef CAS PubMed.
- Z. J. Luo, K. R. Bhuskute, Y. X. Cao, J. Tang and A. Kaur, Angew. Chem., Int. Ed., 2026, e19056 CAS.
- J. Y. Zhao, L. Q. Ren, D. R. Lan, B. Q. Zhan, J. Y. Zhang, J. Mu and C. He, Nat. Commun., 2025, 16, 9719 CrossRef PubMed.
- F. Lv, X. Guo, S. S. Zhu, S. Huang, L. Li, S. Z. Wang, L. J. Jiao and E. H. Hao, Chem. Commun., 2025, 61, 4975–4978 RSC.
- C. Schad, C. Ray, C. Diaz Norambuena, S. Serrano-Buitrago, F. Moreno, B. L. Maroto, I. Garcia-Moreno, M. Munoz-Ubeda, I. Lopez-Montero, J. Banuelos and S. D. L. Moya, Chem. Sci., 2025, 16, 8030–8039 RSC.
- W. Z. Ding, M. Jia, S. K. Yao, Z. P. Liu, F. W. Xu, M. F. Li, C. Q. Wang, W. J. He, Y. C. Chen and Z. J. Guo, J. Am. Chem. Soc., 2025, 147, 47835–47847 CrossRef CAS PubMed.
- H. Q. Huang, Y. N. Wu, X. He, Y. H. Liu, J. H. Zhu, M. R. Gu, D. H. Zhou, S. R. Long, Y. H. Chen, L. Wang, M. L. Li, X. Q. Chen and X. J. Peng, Adv. Sci., 2025, 12, 2416905 CrossRef CAS PubMed.
- Y. K. Yue, J. H. Ai, W. J. Chi, X. N. Zhao, F. J. Huo and C. X. Yin, Adv. Mater., 2024, 36, 2408450 CrossRef CAS PubMed.
- Z. P. She, R. Li, F. Zeng and S. Z. Wu, Adv. Healthcare Mater., 2024, 13, 2400791 CrossRef CAS PubMed.
- Y. Yuan, S. Z. Xiong, L. Q. Lv, W. Hu, X. X. Xiong, C. Y. Li and H. P. Deng, Chem. Eng. J., 2023, 471, 144341 CrossRef CAS.
- P. Chowdhury, Z. Y. Lu, S. P. Su, M. H. Liu, C. Y. Lin, M. W. Wang, Y. C. Luo, Y. J. Lee and H. K. Chiang, Adv. Healthcare Mater., 2024, 13, 2400606 CrossRef CAS PubMed.
- X. W. Li, J. H. Ai, Y. R. Zhang, F. J. Huo and C. X. Yin, Nano Today, 2025, 63, 102768 CrossRef CAS.
- Z. P. Chen, L. Yang, W. J. Xu, F. F. Xu, J. R. Sheng, Q. Xiao, X. Z. Song and W. Q. Chen, Anal. Chem., 2023, 95, 3325–3331 CrossRef CAS PubMed.
- F. L. Ma, Q. Jia, Z. W Deng, B. Z. Wang, S. W. Zhang, J. H. Jiang, G. C. Xing, Z. L. Wang, Z. J. Qiu, Z. Zhao and B. Z. Tang, ACS Nano, 2024, 18, 9431–9442 CrossRef CAS PubMed.
- W. L. Tan, Y. Yu, T. Y. Shi, L. T. Zhang, H. L. Gan, B. H. Wang, G. L. Liu, M. K. Li, L. Yang and Y. G. Ma, Adv. Mater., 2024, 36, 2410418 CrossRef CAS PubMed.
- R. M. Jiang, Z. S. Liu, Y. Y. Han, J. W. Long, T. Gao, X. Lan, M. G. Yu, T. Fang, H. J. Ma, Y. Wei, B. Z. Tang and Z. J. Zhao, Chem. Eng. J., 2024, 500, 157575 CrossRef CAS.
- M. L. Wang, X. Q. Wang, D. J. Xu, C. J. Yu and Y. P. Wang, Chem. Commun., 2026, 62, 915–918 RSC.
- Y. X. Sun, Y. F. Lin, Q. Zhou, Y. X. Han, M. J. Mao, L. H. Guo, H. Y. Liu and J. B. Wang, Sens. Actuators, B, 2025, 444, 138527 CrossRef CAS.
- H. Y. Zheng, W. K. Peng, M. M. Liu, M. Li, W. W. Li, J. Y. Xing, P. F. Shi, Q. Wang, S. S. Zhang and L. Yang, Anal. Chem., 2025, 97, 2472–2478 CrossRef CAS PubMed.
- J. Gao, F. Li, J. Chen, Y. K. Gao, C. F. Fan, Y. Huang, H. F. Yu, X. T. Yang and X. C. Wang, Sens. Actuators, B, 2024, 398, 134696 CrossRef CAS.
- A. Pal, M. Karmakar, S. R. Bhatta and A. Thakur, Coord. Chem. Rev., 2021, 448, 214167 CrossRef CAS.
- C. S. Abeywickrama, Chem. Commun., 2022, 58, 9855–9869 RSC.
- A. C. Sedgwick, L. L. Wu, H. H. Han, S. D. Bull, X. P. He, T. D. James, J. L. Sessler, B. Z. Tang, H. Tian and J. Yoon, Chem. Soc. Rev., 2018, 47, 8842–8880 RSC.
- Y. H. Li, D. Dahal, C. S. Abeywickrama and Y. Pang, ACS Omega, 2021, 6, 6547–6553 CrossRef CAS PubMed.
- H. Gu, W. J. Wang, W. Y. Wu, M. L. Wang, Y. R. Liu, Y. J. Jiao, F. Wang, F. Wang and X. Q. Chen, Chem. Commun., 2023, 59, 2056–2071 RSC.
- M. Liu, B. Feng, F. Y. Chu, D. Y. Fan, F. Zhang, F. Chen and W. B. Zeng, Chin. Chem. Lett., 2025, 36, 110043 CrossRef CAS.
- J. K. Wang, C. H. Wang, C. C. Wu, K. H. Chang, C. H. Wang, Y. H. Liu, C. T. Chen and P. T. Chou, J. Am. Chem. Soc., 2024, 146, 3125–3135 CrossRef CAS PubMed.
- S. Z. Yi, B. N. Li, P. Y. Fu, M. Pan and C. Y. Su, ACS Appl. Mater. Interfaces, 2023, 15, 3172–3181 CrossRef CAS PubMed.
- Z. Hu, J. L. Li, L. L. Feng, Y. L. Zhu, R. X. Zhao, C. H. Yu, R. C. Xu, W. Z. Wang, H. Ding and P. P. Yang, Nano Lett., 2024, 24, 16426–16435 CrossRef CAS PubMed.
- M. M. Islam, Z. Hu, Q. S. Wang, C. Redshaw and X. Feng, Mater. Chem. Front., 2019, 3, 762–781 RSC.
- H. Naito, K. Nishino, Y. Morisaki, K. Tanaka and Y. Chujo, Angew. Chem., Int. Ed., 2017, 56, 254–259 CrossRef CAS PubMed.
- Z. Liu, C. Y. Zhang, Z. P. Li, W. Y. Ma, J. Liu, X. Xia, N. Xu, W. Sun, J. J. Du, J. L. Fan and X. J. Peng, Sci. China: Chem., 2025, 68, 360–368 CrossRef CAS.
- X. J. Yan, R. C. Feng, J. J. Fu, Q. Q. Han, Y. L. Liu and Q. Song, Angew. Chem., Int. Ed., 2025, 64, e202516458 CrossRef CAS PubMed.
- L. Zhao, H. Zhu, Y. Y. Duo, D. W. Pang, Z. G. Wang and S. L. Liu, Adv. Healthcare Mater., 2023, 12, 2301584 CrossRef CAS PubMed.
- M. Ojha, M. Banerjee, M. Mandal, T. Singha, S. Ray, P. K. Datta, M. Mandal, A. Anoop and N. D. P. Singh, ACS Appl. Mater. Interfaces, 2024, 16, 21486–21497 CrossRef CAS PubMed.
- C. Wang, W. C. Jiang, D. Tan, L. Huang, J. Li, Q. L. Qiao, P. Yadav, X. G. Liu and Z. C. Xu, Chem. Sci., 2023, 14, 4786–4795 RSC.
- C. Y. Li, H. Q. Zheng, W. L. Xu, Z. Y. Xu, Z. L. Liao, G. D. Qian and Y. J. Cui, Adv. Mater., 2025, 10, e18371 Search PubMed.
- X. L. Zhou, L. Miao, W. Zhou, Q. L. Qiao and Z. C. Xu, Chin. Chem. Lett., 2025, 36, 110984 CrossRef CAS.
- C. J. Yin, Z. A. Yan, R. J. Yan, C. Xu, B. B. Ding, Y. H. Ji and X. Ma, Adv. Funct. Mater., 2024, 34, 2316008 CrossRef CAS.
- K. W. Lee, Y. J. Gao, W. C. Wei, J. H. Tan, Y. P. Wang, Z. Feng, Y. H. Zhang, Y. Liu, X. L. Zheng, C. Cao, H. Chen, P. F. Wang, S. L. Li, K. T. Wong and C. S. Lee, Adv. Mater., 2023, 35, 2211632 CrossRef CAS PubMed.
- N. Zhang, L. Liu, H. X. Chang, K. Liu, T. H. Liu, L. P. Ding and Y. Fang, J. Phys. Chem. Lett., 2023, 14, 7283–7289 CrossRef CAS PubMed.
- M. Orfano, J. Perego, C. X. Bezuidenhout, I. Villa, R. Lorenzi, B. Sabot, S. Pierre, S. Bracco, S. Piva, A. Comotti and A. Monguzzi, Adv. Funct. Mater., 2024, 34, 2404480 CrossRef CAS.
- M. X. Qin, W. L. Ji, P. C. Huang, F. Y. Wu and L. Q. Mao, Anal. Chem., 2024, 96, 14697–14705 CrossRef CAS PubMed.
- T. Lu and F. W. Chen, J. Comput. Chem., 2012, 33, 580–592 CrossRef CAS PubMed; T. Lu, J. Chem. Phys., 2024, 161, 082503 CrossRef PubMed.
| This journal is © The Royal Society of Chemistry 2026 |