Open Access Article
Open Access ArticleRecent advances in functional materials for uranium monitoring through optical and electrochemical techniques
KaiZeng Quan†
a,
YuMin Wang†b,
Meng Qia,
Peng Xua,
YaXing Wang b and
GuoXun Ji
b and
GuoXun Ji *a
*a
aRocket Force University of Engineering, Xi'an 710025, PR China. E-mail: ji_guoxun@sina.com
bState Key Laboratory of Radiation Medicine and Protection, School for Radiological and Interdisciplinary Sciences (RAD-X) and Collaborative Innovation Center of Radiation Medicine of Jiangsu Higher Education Institutions, Soochow University, Suzhou 215123, PR China
First published on 24th April 2026
Abstract
Uranium is a naturally occurring element widely employed in nuclear energy production and various industrial applications. However, it poses substantial risks to the environment and human health due to its combined radiological and chemical toxicity, thereby making rapid, accurate, and field-deployable detection technologies critical. Conventional detection methods, including atomic absorption or fluorescence spectroscopy and inductively coupled plasma spectroscopy, are limited by their high costs, complicated operational procedures, and dependence on specialized laboratory facilities, which severely restrict their practical on-site applications. To address these limitations, significant advances have been made in recent years in the development of functional materials for uranium sensing, especially metal–organic frameworks (MOFs), covalent organic frameworks (COFs), nanomaterials, DNAzymes, and other emerging materials. These materials support versatile optical and electrochemical detection strategies, such as fluorescence, colorimetry, and electrochemical sensing, which offer high sensitivity, fast response, and considerable potential for real-time field monitoring. This review highlights recent advances in the detection mechanisms, sensing performance, and practical advantages and limitations of various sensing systems. Finally, a comparative analysis is presented to identify key challenges and provide perspectives for the future development of efficient and reliable uranium detection technologies.
1. Introduction
Uranium, a naturally occurring radioactive element, plays a critical role in both the energy and defense sectors as a primary nuclear fuel.1–3 As a high-energy-density energy source, uranium is indispensable for nuclear power generation, whereas its involvement in nuclear weapons testing, atomic bombings, and the deployment of depleted uranium munitions has led to extensive nuclear pollution. Major nuclear accidents, including those at Chernobyl and Fukushima, have further exacerbated environmental contamination.4–6 The radiological and chemical toxicity of uranium presents considerable risks to ecological systems and human health. In aqueous environments, uranium mainly exists in the form of the uranyl ion (UO22+), and its excessive accumulation results in persistent environmental contamination. Radiation emitted during uranium decay can cause cellular damage and elevate cancer risks, while its chemical toxicity exerts adverse effects on vital organs, including the kidneys, liver, and bones.7–9 The World Health Organization has established a maximum contaminant limit of 30 µg L−1 for uranium in drinking water, underscoring the urgent demand for effective monitoring techniques. Accordingly, the development of rapid, highly selective detection materials for UO22+ in aqueous systems has emerged as a major research direction.10,11Traditional methods for detecting UO22+ in aqueous solutions, including atomic absorption spectroscopy,12 atomic fluorescence spectroscopy,13 and inductively coupled plasma spectroscopy,14 are limited in their widespread application by high costs and complex operational requirements.15 In contrast, newer techniques such as fluorescence, colorimetry, and electrochemical detection provide more practical alternatives.16–19 Fluorescence methods are particularly recognized for their high sensitivity and rapid response,20 while colorimetric detection is valued for its simplicity and visual readout capability.21 Electrochemical detection, characterized by its low detection limits, is especially well-suited for complex environmental samples.22 In recent years, advanced materials, including metal–organic frameworks (MOFs), covalent organic frameworks (COFs), nanomaterials, and DNAzymes, have attracted significant attention for their potential in uranium detection.23–26 MOFs and COFs stand out due to their porous structures, high specific surface areas, and tunable functional groups, which make them ideal candidates for UO22+ detection.27,28 Nanomaterials, with their excellent optical and electrical properties, can be surface-functionalized to achieve the selective detection of UO22+.25 DNAzymes, which leverage the biomolecular recognition capabilities of DNA, further enhance the selectivity of UO22+ detection.29 These functional materials offer substantial advantages, rendering them highly promising for the future development of uranium detection technologies.
Over the past few years, several reviews focusing on UO22+ detection have been published. Specifically, in 2022, Liu et al.30 reviewed the advances in the adsorption, detection, and synchronous application of MOF-based materials for U(VI) in aqueous solutions. In 2023, Rani et al.31 summarized the research progress of various nanomaterials (e.g., metal nanoparticles and carbon-based materials) in uranium detection and removal. In 2024, Sharma et al.32 analyzed the advantages and limitations of electrochemical techniques based on chemically modified electrodes for the detection of radionuclides, such as uranium and cesium. In 2025, Sun et al.33 elaborated on the preparation and characterization of carbon dots and their composites, along with their applications, mechanisms, and challenges in the detection and adsorption of uranium and other radioactive elements. In the same year, Poulin-Ponnelle et al.34 reviewed the research progress on fluorescent probes for toxic elements associated with the nuclear industry (e.g., uranium and cesium), covering multiple sensing platforms, fluorescence mechanisms, and application prospects. However, most of these reviews tend to focus on specific material categories or individual detection methods rather than providing a comprehensive overview. In view of this, the present review summarizes the recent progress in UO22+ detection by covering a wide spectrum of representative sensing materials, including MOFs,35 COFs,36 nanomaterials,31 DNAzymes,37 and other functional systems. Notably, this work provides the first comprehensive cross-material and cross-technique comparative analysis of UO22+ sensing platforms. We systematically examine the sensing mechanisms, analytical performance, and structure–property relationships of various material systems, critically evaluate their respective advantages and limitations, and further discuss key challenges that hinder their practical deployment, as well as feasible pathways for future improvement. Through this integrated and forward-looking perspective, the review aims to deliver a holistic understanding of UO22+ detection and provide valuable guidance for the rational design of next-generation high-performance uranium sensing platforms.
2. Fluorescence
Fluorescence is an analytical technique based on photoluminescence, with its mechanism varying depending on the type of material used.38 By tailoring the chemical structures or surface properties of fluorescent probes, these probes can be engineered to interact specifically with a target analyte, thereby inducing a distinct change in fluorescence. This allows for the detection of the target substance through the monitoring of corresponding signal variations (Fig. 1).392.1 MOFs
MOFs are a new class of porous materials that have emerged rapidly over the past few decades. They are constructed from metal ions or clusters connected to organic ligands through coordination bonds, forming crystalline structures with periodic networks, high porosity, and large specific surface areas.40 Due to these distinctive properties, MOFs have been widely applied in numerous fields, among which the design of fluorescent sensors based on their fluorescent characteristics has become a prominent research focus.41In addition to metal-centered luminescence, luminescence from ligand centers primarily arises from π → π* or n → π* electronic transitions within the π-conjugated system of the organic ligands.44 The rigid crystalline framework of MOFs significantly suppresses non-radiative relaxation processes, such as molecular vibrations and rotations of the ligands, through spatial confinement effects, thereby substantially enhancing the fluorescence quantum yield. In certain systems, additional mechanisms, such as intermolecular charge transfer (CT) or aggregation-induced emission (AIE), are also involved. Furthermore, by tuning the energy-level alignment, electronic properties, and geometric structure between the metal and organic ligands, a variety of luminescence mechanisms can be achieved, including ligand-to-ligand charge transfer, metal-to-metal charge transfer, and metal-to-ligand or ligand-to-metal charge transfer.45
Fluorescence quenching primarily operates through three mechanisms: photoinduced electron transfer (PET), fluorescence resonance energy transfer (FRET), and the inner filter effect (IFE).48 In PET, excited-state electrons from the fluorescent MOF are transferred to UO22+, creating a non-radiative relaxation pathway back to the ground state and resulting in significant fluorescence reduction.49 FRET occurs when the emission spectrum of the MOF overlaps considerably with the absorption spectrum of UO22+, allowing non-radiative energy transfer to UO22+ and subsequent fluorescence quenching.50 IFE arises when the fluorescent material itself or other light-absorbing species absorb either the excitation or emitted light, leading to decreased fluorescence intensity.51 Qin et al.52 synthesized a Zn-based MOF (HNU-50), whose fluorescence originates from the π-conjugated system of the pyromellitic acid (PMA) ligand. The uncoordinated oxygen atoms on the ligand serve as binding sites for UO22+ (Fig. 2A). A notable spectral overlap was observed between the emission spectrum of the PMA ligand and the absorption spectrum of uranyl nitrate in the 335–476 nm region. Consequently, UO22+ absorbs the emission from the PMA ligand, leading to fluorescence quenching of HNU-50 via the FRET mechanism.
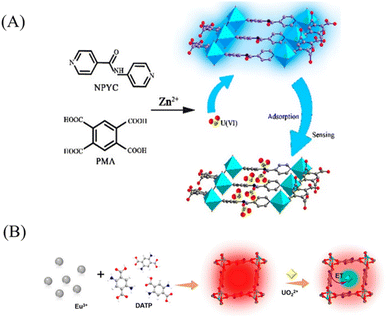 | ||
| Fig. 2 (A) Mechanism of HNU-50 fluorescence detection of uranyl ions. Reproduced with permission from ref. 52. Copyright 2020, the American Chemical Society. (B) Schematic of the preparation of Eu-DATP and its application for UO22+ analysis. Reproduced with permission from ref. 55. Copyright 2024, the American Chemical Society. | ||
In contrast to fluorescence quenching, fluorescence enhancement is primarily achieved through two distinct mechanisms: inhibition of non-radiative energy transfer and sensitization of the intrinsic luminescence of uranyl ions. For MOFs with lanthanide-centered emission, coordination of UO22+ to the MOF can induce structural rigidification, which significantly reduces non-radiative energy loss and enhances the efficiency of the “antenna effect”, thereby markedly boosting the characteristic fluorescence of the lanthanide ions. Alternatively, when UO22+ coordinates with the organic ligands in the MOF, energy transfer from the ligands to the uranyl ion may occur, which sensitizes and activates the intrinsic emission of UO22+ itself. Both of these mechanisms can be effectively utilized to construct fluorescence-enhanced sensors for UO22+ detection.
Ratiometric sensing for UO22+ detection utilizes the ratio of emission intensities at two distinct wavelengths from a fluorescent probe as the response signal.53 This relative signal change enables the sensitive and accurate quantification of UO22+. Although UO22+ exhibits weak intrinsic photoluminescence, its coordination with organic ligands can significantly enhance its green emission at 509 nm via the “antenna effect”, wherein the ligand transfers energy to UO22+. Concurrently, this coordination process may alter the fluorescence emitted from the metal nodes or dopants within the MOF. By monitoring changes in the ratio of these dual emission signals, ratiometric sensing achieves highly precise and sensitive UO22+ detection.54 For example, Tong et al.55 developed a Eu-MOF with pore environments capable of simultaneously activating the intrinsic fluorescence of UO22+ and quenching the emission of Eu3+, thereby constructing a dual-signal, inversely varying ratiometric fluorescent sensor (Fig. 2B). In this system, the organic ligand 2,5-diaminoterephthalic acid (DATP) absorbs light and transfers energy to Eu3+ via the antenna effect, resulting in its characteristic strong red emission. When UO22+ is captured and immobilized by the free amino groups within the MOF channels, the energy absorbed by DATP is distributed to both Eu3+ and UO22+. This leads to reduced energy transfer to Eu3+, which attenuates its red emission, while the enhanced energy transfer to UO22+ intensifies its intrinsic green fluorescence. By calculating the intensity ratio at these two emission wavelengths, a concentration-dependent signal for UO22+ is established. This built-in self-referencing mechanism renders the ratiometric approach more reliable and resistant to interference than single-intensity-based detection.
2.2 COFs
Similar to MOFs, COFs are crystalline materials composed of light elements connected by covalent bonds, forming two- or three-dimensional topologies.56 They are primarily constructed from aromatic organic ligands, such as phenyl, furan, and bipyridine units, which form extended covalent networks. Compared with MOFs, COFs are composed entirely of nonmetallic light elements, which endow them with superior thermal and chemical stability.57 Additionally, COFs possess high specific surface areas, porous structures, strong fluorescence, and tunable chemical functionalities, making them versatile materials for a wide range of applications.58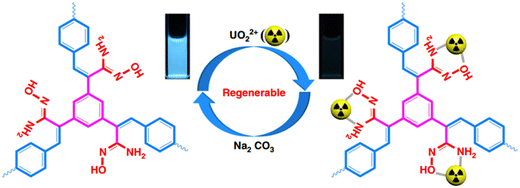 | ||
| Fig. 3 Schematic of TFPT-BTAN-AO regeneration detection and extraction of UO22+. Reproduced with permission from ref. 62. Copyright 2020, Springer. | ||
2.3 Nanomaterials
In addition to MOFs and COFs, nanomaterials have emerged as promising candidates for the detection and remediation of radioactive waste, owing to their unique physicochemical properties, including an exceptionally high surface area, ultrasmall size, quantum effects, and intrinsic fluorescence.64 In recent years, their application in fluorescence-based detection of UO22+ has grown significantly, with metal nanoclusters and quantum dots as the primary examples.Another key type of nanomaterial for fluorescence-based UO22+ detection is quantum dots (QDs), which are semiconductor nanocrystals typically ranging in size from 2 to 10 nm. Their ultrasmall dimensions confine electrons and holes within the nanoscale domain, giving rise to quantum confinement effects that generate discrete and tunable electronic energy levels. Fluorescence emission from QDs arises from radiative transitions between these quantized energy states.67
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O and P–O groups on the AuNP surface, triggering the aggregation of AuNPs. This aggregation event causes a redshift and the broadening of the AuNP SPR absorption peak. Initially, the limited spectral overlap between AuNP absorption and AuNC emission results in a weak IFE, leading to strong red fluorescence from the AuNCs. Following AuNP aggregation, the enhanced spectral overlap induces significant IFE-mediated quenching of AuNC fluorescence, resulting in a pronounced decrease in the red emission intensity (Fig. 4). Thus, the ratio of the fluorescence intensity of red-emitting AuNCs to that of blue-emitting B-CDs serves as a reliable readout for quantifying the UO22+ concentration.
O and P–O groups on the AuNP surface, triggering the aggregation of AuNPs. This aggregation event causes a redshift and the broadening of the AuNP SPR absorption peak. Initially, the limited spectral overlap between AuNP absorption and AuNC emission results in a weak IFE, leading to strong red fluorescence from the AuNCs. Following AuNP aggregation, the enhanced spectral overlap induces significant IFE-mediated quenching of AuNC fluorescence, resulting in a pronounced decrease in the red emission intensity (Fig. 4). Thus, the ratio of the fluorescence intensity of red-emitting AuNCs to that of blue-emitting B-CDs serves as a reliable readout for quantifying the UO22+ concentration.
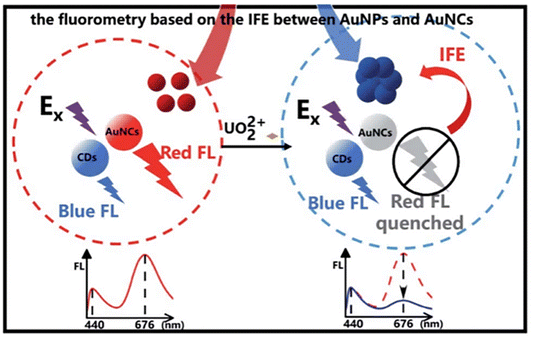 | ||
| Fig. 4 Schematic of the IFE-based ratiometric fluorescence method for UO22+ detection. Reproduced with permission from ref. 69. Copyright 2022, the Royal Society of Chemistry. | ||
Quantum dots inherently lack affinity for UO22+ ions, necessitating surface modification with functional groups, such as carboxyl, pyridyl, or thiol, to enable selective binding. While quantum dots alone exhibit strong fluorescence, coordination with UO22+ leads to the formation of a non-emissive ground-state complex, resulting in fluorescence quenching through a static mechanism.70 Zheng et al.71 introduced abundant oxygen- and nitrogen-containing groups onto molybdenum oxide (MoOx) quantum dot surfaces, producing dopamine-functionalized molybdenum oxide quantum dots (DA-MoOx QDs) with high affinity for UO22+. Upon coordination, these DA-MoOx QDs aggregated into larger clusters, which cause significant fluorescence quenching.
2.4 DNAzymes
DNAzymes are catalytic DNA molecules capable of specifically cleaving substrate strands upon activation by target metal ions, exhibiting high selectivity in recognition. This property makes DNAzymes ideal for the development of sensitive and selective sensors for UO22+ detection.72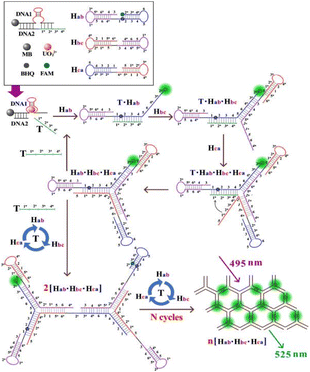 | ||
| Fig. 5 Detection mechanism of the DNA network biosensor for the ultrasensitive detection of UO22+ based on the double-loop hairpin probe assembly. Reproduced with permission from ref. 75. Copyright 2024, the American Chemical Society. | ||
2.5 Comparison of properties between different materials
Table 1 summarizes the performance of representative materials selected from different categories for fluorescence-based UO22+ detection. Different materials exhibit distinct properties, which are inherently associated with variations in their molecular and structural characteristics. Analyzing the structure–property relationship enables us to design materials with superior performance.| Material type | Material name | pH | Response time | Linear range (µM) | LOD (nM) | Ref. |
|---|---|---|---|---|---|---|
| MOFs | Tb-MOF | 4 | — | 0.74–1296 | 3.33 | 80 |
| HNU-50 | 3 | — | 0.01–1000 | 12 | 52 | |
| CSMCRI-21 | 5 | 20 s | 1.26–1680 | 130 | 81 | |
| Eu3+@UiO-67-bpydc | 6 | — | 0.01–0.1 | 3 | 54 | |
| IRMOF-3-DPC | 4 | — | 26.25–1260 | 1510 | 82 | |
| Eu-DATP | — | — | 0.01–100 | 2.7 | 55 | |
| COFs | TFPT-BTAN-AO | 6 | 2 s | 0.02–6 | 6.7 | 62 |
| TP-COF-AO | 6 | 2 s | 0–10 | 8.3 | 83 | |
| PyTT-Tp | 5 | 2 s | 0.015–4.5 | 4.92 | 84 | |
| TAPM-DHBD | 5 | 2 s | 0.01–3.7 | 4.08 | 28 | |
| GC-TFPB-AO | 7 | — | 0.05–0.5 | 21.25 | 63 | |
| Nanomaterials | AuNCs@AuNPs@B-CDs | 2.5 | 12 min | 0.2–5 | 8.4 | 69 |
| DA-MoOx QDs | 6 | 5 min | 0.005–1 | 3.85 | 71 | |
| CMH-NCDs | 4–5 | 0.5 s | 0–20 | 8.4 | 85 | |
| DNAzymes | DNA network biosensor | 6.6 | 100 min | 0.00001–1 | 0.002 | 75 |
| DNA@SYBR green I | 7.4 | 20 min | 0.01–1 | 4.6 | 86 | |
| DNA hairpin@39E DNAzyme | 5.5 | 50 min | 0.00005–0.02 | 0.017 | 76 |
Under acidic to neutral conditions, UO22+ remains the dominant form that can undergo cation exchange and directly coordinate with most binding sites. At pH > 7, it predominantly exists as the highly stable anionic complex [UO2(CO3)3]4−, which can cause materials and sensors originally designed to capture UO22+ to lose function entirely, especially those with anionic frameworks, such as MOFs and COFs.77 As illustrated in Table 1, the optimal detection pH for most materials ranges from 3 to 7, which corresponds to the favorable speciation of UO22+. Anionic MOFs perform well under mildly acidic conditions; however, extreme acidity or alkalinity can impair detection performance and damage MOF structures, attributed to the inherent lability of coordination bonds. Many MOF ligands contain nitrogen-containing moieties (e.g., imidazole or pyridine) or oxygen-containing groups (e.g., carboxyl groups). Excessive proton concentrations compete with UO22+ for binding sites and can protonate the ligands, thereby disrupting the coordination framework. Under alkaline conditions, hydroxide ions induce hydrolysis of metal nodes (e.g., zinc or copper ions), resulting in framework collapse.78 Compared to MOFs, COFs exhibit superior stability, due to their entirely organic frameworks and strong covalent bonds.79 Fluorescent nanomaterials, such as metal nanoclusters and quantum dots, are susceptible to structural damage under extreme pH conditions: ligands on metal nanoclusters may undergo protonation or hydrolysis, while quantum dot nanocrystals are prone to corrosion, both of which lead to irreversible fluorescence loss. DNAzymes are particularly sensitive to extreme pH, as hydrolysis of purine bases or phosphodiester bonds disrupts their precisely folded three-dimensional structures, resulting in denaturation and the loss of both catalytic activity and specific UO22+ recognition capability.
The fluorescence response time is a key parameter that reflects the sensing efficiency of a material. As shown in Table 1, few studies on MOFs report clear fluorescence response times, whereas COF-based sensors generally exhibit rapid responses, with the majority achieving signal stabilization within 2 seconds. Both COFs and MOFs detect UO22+ through specific coordination between the uranyl ion and functional groups on their pore walls; this coordination event triggers electron transfer and energy-level transitions, which in turn alter the fluorescence properties of the materials. This rapid physicochemical interaction typically reaches equilibrium within a few seconds to one minute. The fluorescence of nanomaterials is intrinsic to their structure, and fluorescence quenching occurs promptly once UO22+ diffuses to the nanomaterial surface via collisions or weak coordination interactions. However, reported response times vary widely, ranging from 0.5 seconds to several minutes, depending on the experimental setup. For instance, UO22+-induced the aggregation of AuNPs requires a certain amount of time to form stable clusters—a process that is essential for achieving significant and stable ratiometric fluorescence detection based on the IFE. Thus, the response speed of such sensors is constrained by the kinetics of AuNP aggregation. In contrast, DNAzyme-based sensors exhibit longer response times, ranging from tens of minutes to hours, due to their multistep catalytic processes, which involve UO22+ binding, conformational changes of the DNAzyme, and catalytic activity. Unlike porous materials (e.g., MOFs and COFs) that possess large specific surface areas and multiple binding sites, a single DNAzyme molecule typically contains only a few catalytic sites, resulting in relatively lower sensing efficiency.
The detection limit is defined as the lowest concentration of UO22+ that can be reliably quantified, with lower detection limits indicating higher sensing sensitivity of the material. As shown in Table 1, MOFs exhibit linear detection ranges spanning one to five orders of magnitude, with detection limits ranging from several nanomolar to several micromolar. Their porous structure provides abundant binding sites that resist rapid saturation, thereby yielding relatively wide linear ranges; however, the upper limit of these ranges is constrained by the diffusion rate of UO22+ within the pores. The strong coordination between UO22+ and high-affinity functional groups on MOFs enables these materials to achieve low detection limits. In comparison, COFs typically exhibit linear detection ranges of one to two orders of magnitude and detection limits ranging from several to tens of nanomolar. Despite possessing more stable covalent bonds and open pore structures than MOFs, the performance of COFs is similarly limited by the diffusion of UO22+ within their pores. Nanomaterials display linear detection ranges of one to three orders of magnitude, with detection limits generally in the single-digit nanomolar range. Their nanoscale dimensions confer high fluorescence efficiency and a large specific surface area, leading to excellent sensing sensitivity; however, the limited number of surface binding sites restricts their linear ranges due to facile saturation. DNAzymes offer linear detection ranges spanning two to five orders of magnitude and detection limits ranging from picomolar to nanomolar levels. The catalytic amplification effect and high specificity of aptamer-UO22+ binding allow a single UO22+ ion to trigger significant signal changes, resulting in ultralow detection limits.
For ligand-based luminescent MOFs, the selection of ligands with AIE properties offers distinct advantages. Such ligands exhibit weak fluorescence in their monomeric state but display significantly enhanced emission when confined within the fixed, ordered framework of MOFs. For metal-centered luminescent MOFs, the careful selection of organic ligands is critical to match their triplet energy levels with those of lanthanide ions (e.g., Eu3+ and Tb3+), thereby enabling efficient energy transfer. Alternatively, strong fluorophores, such as organic dyes or quantum dots, can be post-synthetically grafted onto MOF pores or frameworks to serve as new emission centers. For instance, Ahmad et al.88 utilized ZIF-8 as a porous host to uniformly disperse quantum dots within its channels, which effectively suppressed aggregation, maintained high fluorescence efficiency, and consequently enhanced the photoluminescence quantum yield. Cui et al.89 fabricated a composite of Tb-MOF and terbium alginate hydrogel (Tb-AG); this composite improved material stability and provided additional coordination sites for uranyl ions via the carboxyl oxygen atoms in Tb-AG, thereby greatly enhancing fluorescence quenching efficiency and achieving both high sensitivity and stability. COFs generally exhibit weaker fluorescence compared to MOFs, as dynamic covalent bonds facilitate nonradiative decay pathways. The introduction of lanthanide ions coordinated to COFs can act as antenna moieties, transferring absorbed energy from the organic framework to amplify the characteristic emission of lanthanides and dramatically improve the fluorescence performance. Mao et al.90 coordinated Eu3+ to the nitrogen atoms in the bipyridine units within the COF, which restricted the free rotation of bipyridine and suppressed nonradiative energy dissipation caused by intramolecular rotation. This modification significantly enhanced the intrinsic fluorescence of the COF and enabled turn-on fluorescence detection of uranyl ions via resonance energy transfer. Similar to MOFs, post-synthetic modification can incorporate highly emissive molecules into COF pores, leveraging confinement effects to protect and enhance luminescence. For quantum dots, surface modification with organic molecules or polymers can improve their dispersibility and stability while reducing nonspecific interactions that induce fluorescence quenching. The introduction of heteroatom dopants (e.g., nitrogen or sulfur) modifies their electronic structure, allowing for the tuning of emission color and enhancement of fluorescence intensity. Yang et al.91 employed a ligand exchange strategy to functionalize originally hydrophobic CdSe/CdS/ZnS quantum dots, successfully converting them into water-soluble fluorescent probes with excellent dispersibility and fluorescence properties. In the design of DNAzyme-based fluorescent sensors, the selection of high-performance fluorophores or integration with fluorescent materials enables highly specific detection. Zhang et al.87 developed a “switch-on/off” fluorescent sensing platform by integrating upconversion nanoparticles (UCNPs), AuNPs, DNAzyme, and hybridization chain reaction (HCR). Specifically, UCNPs serve as energy donors and AuNPs as energy acceptors: in the absence of UO22+, AuNPs remain dispersed and quench UCNPs' fluorescence via FRET, putting the system in an “off” state. Upon the introduction of UO22+, the target ion triggers DNAzyme hydrolysis to produce single-stranded DNA (ssDNA), which further initiates HCR for the formation of long-chain double-stranded DNA (dsDNA). The electrostatic repulsion between dsDNA and AuNPs then induces AuNP aggregation, which weakens FRET and enables the subsequent fluorescence recovery of UCNPs (i.e., an “on” state) (Fig. 6). This system synergistically combines the high specificity of DNAzyme-based recognition, the robust signal amplification of HCR, the low-background advantage of UCNPs, and the efficient quenching capability of AuNPs, thereby achieving ultrasensitive, highly selective, and rapid detection of UO22+.
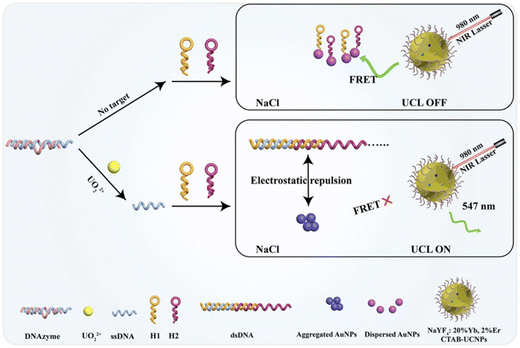 | ||
| Fig. 6 Upconversion fluorescence platform for the sensitive quantification of UO22+ based on specific DNAzyme-mediated signal amplification and fluorescence resonance energy transfer. Reproduced with permission from ref. 87. Copyright 2024, Springer. | ||
In summary, single materials often struggle to simultaneously achieve high fluorescence intensity and excellent stability. The key to performance enhancement lies in the strategic integration of complementary material properties through sophisticated composite design.
3. Colorimetry
Colorimetric detection is an analytical technique based on detectable color changes in a solution. It enables direct visual readout, facilitating rapid on-site screening and straightforward semi-quantitative assessment (Fig. 7).923.1 MOFs
The colorimetric detection of UO22+ based on MOFs relies on the catalytic reaction between MOF-enzyme composites and substrates, which induces a color change. The binding of UO22+ to the MOF-enzyme composite inhibits the enzyme's catalytic activity, preventing substrate oxidation and enabling colorimetric detection of uranyl ions.93 In 2022, Zhou et al.94 embedded hemin into two-dimensional ZIF-L nanosheets to form a biomimetic peroxidase, ZIF-L-hemin (Fig. 8). In the presence of H2O2, the composite catalyzes the oxidation of 3,3′,5,5′-tetramethylbenzidine (TMB), producing blue-colored oxidized TMB (oxTMB). Upon UO22+ introduction, coordination between U–N and the electron-rich 2-methylimidazole in ZIF-L disrupts the axial electron donation from imidazole to hemin-Fe, inhibiting the catalytic cycle. This results in a decreased oxTMB yield and a color shift from deep blue to light blue or colorless.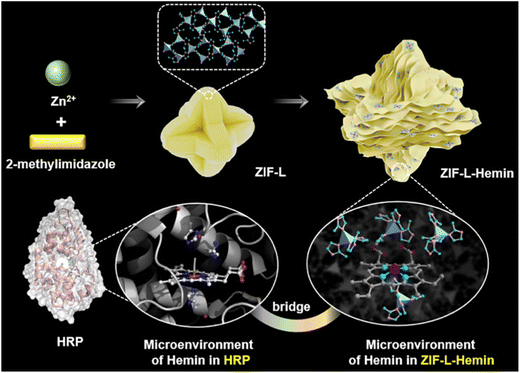 | ||
| Fig. 8 Schematic of the synthesis procedure of ZIF-L-hemin and the structures of HRP and ZIF-L-hemin. Reproduced with permission from ref. 94. Copyright 2022, the American Chemical Society. | ||
3.2 COFs
The colorimetric method is based on the generation of highly reactive free radicals or reactive oxygen species on the surface of COF materials under light irradiation, which catalyze the substrate's colorimetric reaction.95 The introduction of UO22+ inhibits this catalytic process through interactions with the COFs or the substrate. Xu et al.96 developed a visible-light-responsive sulfone-based COF (TAS-COF) that exhibits oxidase-like activity upon visible-light excitation, catalyzing the conversion of the colorless substrate TMB into blue oxTMB. The imine groups in oxTMB coordinate with UO22+ to form a colorless complex, causing the fading of the blue color in the presence of uranyl ions (Fig. 9). Thus, monitoring the significant absorbance changes of the TAS-COF-TMB system enables sensitive colorimetric detection of UO22+.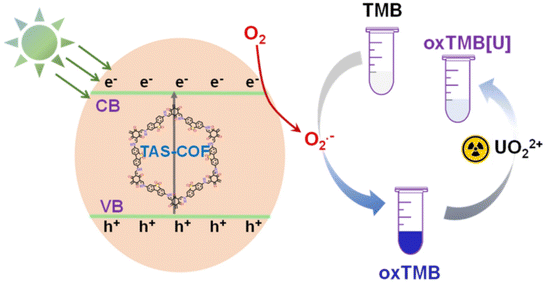 | ||
| Fig. 9 Illustration of the colorimetric detection of UO22+ using visible-light-responsive TAS-COF. Reproduced with permission from ref. 96. Copyright 2022, MDPI. | ||
3.3 Nanomaterials
AuNPs can be employed to design colorimetric sensors for UO22+ detection. When dispersed, AuNPs exhibit strong absorption at a characteristic wavelength and reflect or scatter the complementary light, resulting in a distinct color. Upon aggregation, the plasmon coupling between particles induces a red shift in the localized surface plasmon resonance (LSPR) absorption peak, causing the solution color to change due to the absorption of longer-wavelength light.97 Since AuNPs lack inherent specificity, their surfaces are functionalized with ligands that selectively coordinate to UO22+. The binding of UO22+ to these ligand crosslinks dispersed AuNPs into larger aggregates, triggering a visible color transition. Zhang et al.98 functionalized AuNPs with phosphate groups, imparting a negative charge and yielding a stable wine-red colloidal solution. Specific coordination between UO22+ and the phosphate groups reduces the electrostatic repulsion among the AuNPs, inducing nanoparticle aggregation. This shifts the SPR peak from 519 nm to approximately 650 nm, with the solution color changing from wine red to blue, enabling sensitive colorimetric detection of UO22+.3.4 DNAzymes
In the presence of UO22+, DNAzymes specifically cleave their substrate and form peroxidase-mimicking DNAzyme complexes that catalyze the oxidation of chromogenic ions in solution, causing a visible color change for detection.29 Zhang et al.99 developed a colorimetric assay combining DNAzyme-modified agarose microbeads with cascade signal amplification for UO22+ detection. A bifunctional DNA probe integrates a uranyl-specific DNAzyme substrate as the recognition element and a rolling circle amplification (RCA) primer as the signal transducer, immobilized on MBs via the biotin–streptavidin interaction. Upon UO22+ addition, the DNAzyme cleaves the substrate strand, releasing the primer to initiate RCA, producing abundant guanine quadruplex (G-quadruplex) repeats. The G-quadruplexes complex with hemin to form peroxidase-like catalysts that oxidize ABTS2− with H2O2, generating a blue-green color detectable by the naked eye or UV-vis spectroscopy (Fig. 10).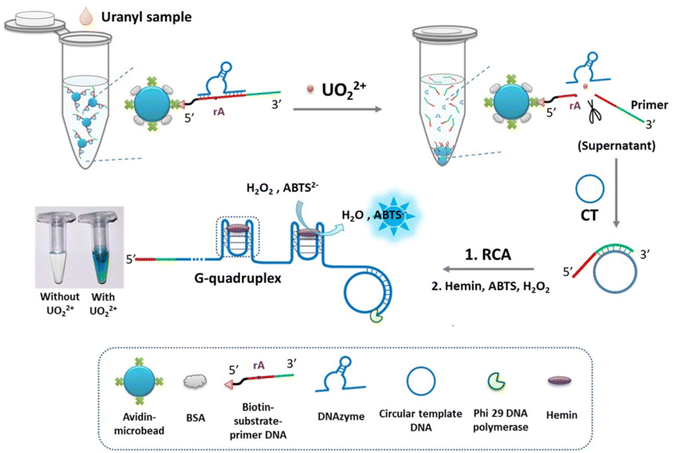 | ||
| Fig. 10 Schematic of the DNAzyme colorimetric detection of UO22+. Reproduced with permission from ref. 99. Copyright 2022, the Royal Society of Chemistry. | ||
3.5 Comparison of properties between different materials
For the currently reported colorimetric detection of UO22+ using MOFs, COFs, and DNAzymes, the underlying principle is uniformly based on enzyme-catalyzed oxidation or inhibition events that modulate substrate conversion, thereby inducing a color change. The final colorimetric signal is derived from the oxidized product of the substrate, whose chemical structure and absorption maximum are typically governed by the pH condition. Colorimetric detection is usually performed under fixed pH conditions, as pH affects not only the structural stability of the materials but also the catalytic activity of enzymes and the stability of substrates involved in the colorimetric reaction. Therefore, maintaining an optimal pH to ensure maximum catalytic activity and the formation of a stable, intensely colored product is crucial for achieving high-sensitivity detection.As shown in Table 2, MOF-based colorimetric sensors exhibit the fastest response, whereas DNAzyme-based assays require the longest response time. This striking difference in response kinetics can be fundamentally attributed to the distinct nature of their catalytic systems. Although MOF, COF, and DNAzyme-based methods all rely on enzyme-mimetic catalyses, the catalytic activity of MOFs and COFs originates from the materials themselves, which act as abiotic nanozymes rather than fragile biological enzymes. Unlike natural enzymes that require strict conformational maintenance to retain activity, these porous framework materials possess inherent, structure-dependent catalytic active sites (e.g., metal nodes and functionalized organic linkers) that enable direct, unimpeded substrate access and rapid redox reactions. Consequently, their catalytic efficiency and reaction rate are generally more stable than those of biological enzymes, leading to significantly faster colorimetric responses. In contrast, DNAzyme-based colorimetric detection involves a multistep biological catalytic process, including UO22+-dependent substrate cleavage, G-quadruplex formation, and subsequent peroxidase-mimetic catalysis for substrate oxidation. Each step in this cascade requires sufficient time for molecular recognition, conformational rearrangement, and catalytic turnover, which inherently prolongs the overall reaction time and results in the slowest response among the studied methods. Notably, AuNP-based colorimetric sensors do not rely on enzymatic catalysis; instead, they leverage LSPR changes induced by physical and chemical interactions, enabling rapid color transitions driven by direct nanoparticle aggregation without catalytic lag.
Regarding detection sensitivity, the DNAzyme-based method achieves an exceptionally low limit of detection (LOD), far outperforming MOF-, COF-, and AuNP-based sensors, while AuNP-based sensors exhibit the highest LOD. This performance gap arises directly from the fundamental differences in their sensing mechanisms. The DNAzyme-based colorimetric method for UO22+ detection capitalizes on its high specificity to UO22+, which enables precise, signal-amplified catalytic turnover even at ultratrace analyte concentrations. Each UO22+ ion can trigger the cleavage of multiple DNAzyme substrates, initiating a cascade of G-quadruplex formation and peroxidase-mimetic catalysis that amplifies the colorimetric signal exponentially, thus enabling detection at sub-nanomolar levels. In contrast, the use of AuNPs results in a relatively higher detection limit. This is because the underlying mechanism requires a sufficient concentration of UO22+ to neutralize the abundant negative charge on the AuNP surface, thereby overcoming electrostatic repulsion and triggering nanoparticle aggregation, which is accompanied by a subsequent color change. At low UO22+ concentrations, only limited aggregation occurs, producing a color change too weak to be reliably detected by either the naked eye or analytical instruments. Additionally, AuNP aggregation is a non-catalytic, stoichiometric process with no signal amplification, further limiting its sensitivity compared to catalytically amplified methods.
For MOF and COF-based sensors, their LOD values fall between those of DNAzyme- and AuNP-based sensors, which can be explained by their intermediate catalytic amplification and binding affinity. As porous framework nanozymes, MOFs and COFs offer moderate catalytic activity and UO22+ adsorption capacity, enabling signal amplification through catalytic substrate oxidation; however, they lack the extreme specificity and exponential amplification capability of DNAzyme systems. This balance results in sensitivity that is higher than those of AuNP-based aggregation sensors but lower than those of DNAzyme-based assays, aligning with the trend observed in Table 2.
The sensitivity of UO22+ detection can be improved by enhancing the intrinsic selectivity of the sensing material. Materials, such as MOFs, COFs, and nanomaterials, can be modified through post-synthetic incorporation of specific functional groups, including amidoxime or phosphate groups, which exhibit strong coordination affinity toward UO22+. These optimized materials can be integrated with smartphone platforms, yielding more objective and quantifiable results compared to visual observation and enabling a transition from qualitative to semiquantitative analysis. This integrated approach facilitates rapid, on-site, and equipment-free quantitative detection, significantly improving the practicality and accessibility of UO22+ monitoring. For instance, Lei et al.100 developed a convenient and portable smartphone-assisted colorimetric method for UO22+ detection. Rifampicin and Br-PADAP were used as chromogenic agents, which undergo coordination reactions with UO22+ separately, resulting in distinct color changes of the solutions: from orange to dark red in the rifampicin system and from yellow to light pink in the Br-PADAP system. As illustrated in Fig. 11, a custom colorimetric viewfinder was employed to standardize the smartphone's shooting conditions; digital images of the reacted solutions were then imported into the built-in “Detect” application to extract the S value (saturation) of HSV and the L value (lightness) of HSL. Leveraging the linear relationships between these color parameters and the UO22+ concentration, on-site rapid estimation of the concentration range was achieved. Meanwhile, combined with the characteristic peak detection using a UV-vis spectrophotometer at 375 nm (rifampicin system) and 562 nm (Br-PADAP system), precise quantification with a low limit of detection of 0.89 µM was realized. This platform integrates a dual-mode of “on-site preliminary screening and laboratory precise determination”, which not only addresses the timeliness requirement of on-site emergency detection but also ensures the accuracy of laboratory analysis, achieving the unification of “portability” and “precision”.
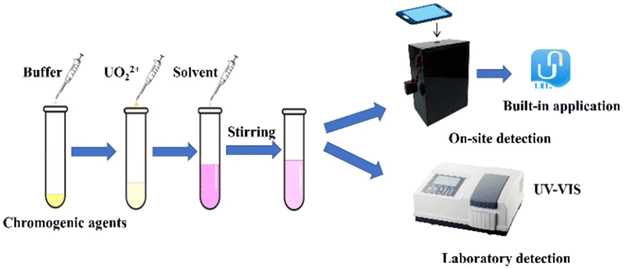 | ||
| Fig. 11 Schematic of the chromogenic reactions for the detection of UO22+. Reproduced with permission from ref. 100. Copyright 2023, the Royal Society of Chemistry. | ||
4. Electrochemical detection
Electrochemical detection is a sensitive and selective detection method for trace UO22+, which involves measuring the current signal generated by the redox reaction of UO22+ on the electrode surface, facilitated by high-sensitivity pulse voltammetry and a functional electrode-modified interface (Fig. 12).324.1 MOFs
MOFs provide well-defined pores and high specific surface areas, and, upon carbonization, they form conductive three-dimensional networks.101 The resulting MOF-derived porous carbon materials possess negatively charged surfaces that strongly attract the positively charged UO22+ ions in solution, enabling efficient enrichment of UO22+ at the electrode interface. This facilitates the electrochemical reduction of UO22+ on the electrode surface, producing measurable reduction currents for uranium detection.102 In 2020, Cao et al.103 used UiO-66-NH2 as a precursor to embed PtRu bimetallic nanoparticles onto MOF-derived porous carbon to create a composite for electrochemical UO22+ detection (Fig. 13). The embedded PtRu nanoparticles serve as key catalytic centers, significantly lowering the overpotential required for UO22+ reduction and accelerating electron transfer. The porous carbon provides a large surface area and rapid electron- and ion-transport channels, greatly amplifying the current signal. The synergistic interaction between PtRu nanoparticles and MOF-derived porous carbon enhances the overall electrochemical performance.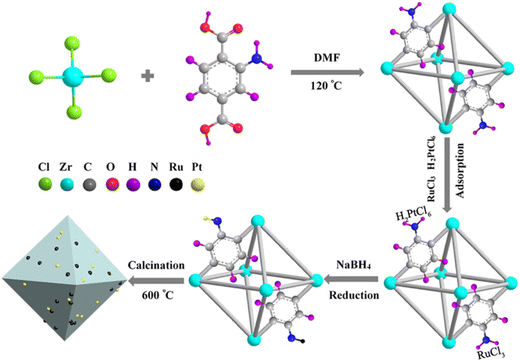 | ||
| Fig. 13 Schematic of the synthesis of Pt1Ru2-PC. Reproduced with permission from ref. 103. Copyright 2020, Springer. | ||
4.2 COFs
COFs can be employed for electrochemiluminescence (ECL) detection of UO22+, a technique that integrates electrochemical reactions with optical signal measurement. By rational design and the selection of electron donor and acceptor units within COFs, efficient intramolecular charge transfer (ICT) can be achieved, exciting electrons in the COF to produce luminescence. Specific binding of UO22+ to functional groups in the COF alters its electronic structure, thereby modulating the ECL intensity.104 Cui et al.105 developed an alkene-linked COF, TBTN-COF, and synthesized a BCBA-TBTN-AO complex by converting cyano groups in the COF into amidoxime groups, enabling selective UO22+ coordination. Although the monomers are non-luminescent, the ICT within the framework induces strong ECL, where TBTN serves as the electron acceptor and BCBA and DAFB as electron donors, facilitating effective charge transfer. Under applied potential, the electrons in the COF are excited to form radical anions, which emit light upon collision with superoxide anions, generating a robust ECL signal (Fig. 14). The coordination of UO22+ with BCBA-TBTN-AO transfers electrons from the donor units to TBTN, enhancing the ECL response and establishing a highly selective “turn-on” ECL sensing system with ultralow detection limits.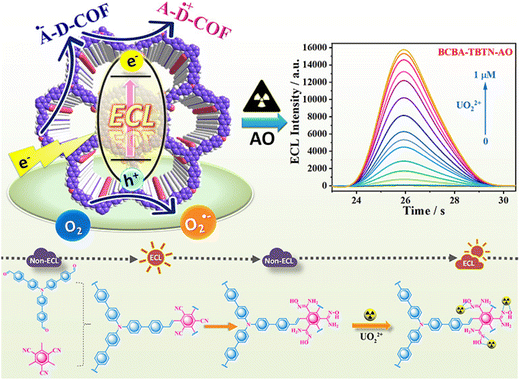 | ||
| Fig. 14 Schematic of the COF-based electrochemiluminescence detection of UO22+. Reproduced with permission from ref. 105. Copyright 2021, the American Chemical Society. | ||
4.3 Nanomaterials
Nanomaterials, featuring a high specific surface area, abundant active sites, and excellent conductivity, serve as carriers to immobilize numerous recognition elements and establish efficient electron-transfer channels with electrodes, enabling highly sensitive detection of low-concentration uranyl ions.106 Han et al.107 modified a glassy carbon electrode (GCE) with AuNPs for uranium detection using differential pulse adsorptive stripping voltammetry. Distinct redox peaks were observed on all electrodes. After the AuNP modification, the oxidation peak current increased from 454 µA to 579 µA, corresponding to a 27.53% enhancement, which clearly demonstrates the superior electron-transfer capability of the AuNPs/GCE. In this method, UO22+ is first reduced and deposited onto the electrode at negative potentials. A subsequent positive potential scan then oxidizes and strips the deposited uranium, generating an oxidation peak current corresponding to the UO22+ concentration. The high surface area of AuNPs significantly increases the active electrode area, which amplifies the stripping signal and enhances the detection sensitivity.4.4 DNAzymes
DNA itself is non-conductive; thus, to realize electrochemical detection, electroactive molecules are covalently attached at specific positions on the DNA strand, along with UO22+ binding aptamers.108 When UO22+ binds to the aptamer, it induces conformational changes in the DNA structure. This conformational shift affects the electron-transfer efficiency between the electroactive molecule and the electrode, resulting in measurable current changes.109Jett et al.110 reported a DNA-based electrochemical biosensor for detecting UO22+ in water. The sensor employs a modified uranyl-binding aptamer embedded within an oligonucleotide scaffold, forming a reusable, conformation-switching electrochemical DNA biosensor (Fig. 15A). Upon UO22+ binding, the DNAzyme activity is triggered, causing cleavage at a specific site and a conformational switch from an unbound to a bound state (Fig. 15B). This structural change alters the position and environment of the methylene blue electroactive reporter attached to the DNA, thus modulating electron transfer at the electrode surface and generating a corresponding electrochemical signal change.
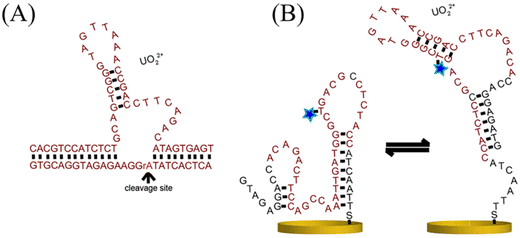 | ||
| Fig. 15 (A) Schematic of the DNAzyme–UO22+ binding. Reproduced with permission from ref. 110. Copyright 2017, Wiley. (B) Schematic of the conformation-switching E-DNA uranyl biosensor. Reproduced with permission from ref. 110. Copyright 2017, Wiley. | ||
4.5 Comparison of properties between different materials
As shown in Table 3, distinct variations in electrochemical detection performance are observed across different material systems for UO22+ analysis. Specifically, COFs and nanomaterials outperform MOFs and DNAzymes in terms of linear ranges and LOD values, while the latter two material categories are relatively less reported and typically exhibit inferior sensing metrics. This performance disparity stems fundamentally from the intrinsic electronic and structural properties of each material class, as well as their compatibility with electrochemical transduction mechanisms.| Material type | Material name | pH | Response time | Linear range (µM) | LOD (nM) | Ref. |
|---|---|---|---|---|---|---|
| MOFs | PtRu-PCs | 4.6 | 3 min | 0.168–3.528 | 24 | 103 |
| COFs | AuNCs@COF | 4 | 60 min | 0.0005–25 | 0.347 | 111 |
| BCBA-TBTN-AO/GCE | 5.5 | 150 s | 0.000001–0.001 | 0.00036 | 105 | |
| Nanomaterials | AuNPs/GCE | 3 | 600 s | 0.0084–0.042 | 0.44 | 107 |
| AuNPs/GCE | 1.6 | 400 s | 0.010–0.20 | 1.26 | 112 | |
| PNPs | 7.4 | — | 0.00000037–0.00037 | 0.0021 | 113 | |
| DNAzymes | SG@dsDNA@[Fe(CN)6]3−/4− | 5.5 | 75 min | 0.001–1 | 0.13 | 114 |
| Electrochemical biosensor | 5 | — | 0.1–500 | 1200 | 110 |
Most MOFs and COFs are inherently semiconductors or insulators with low electrical conductivity, which severely restricts efficient electron transfer between the electrode interface and the bulk of the material. This limitation directly translates into sluggish electron kinetics and weak signal generation, ultimately resulting in the relatively high LOD (24 nM) and narrow linear range (0.168–3.528 µM) observed for MOF-based sensors like PtRu–PCs (Table 3). In contrast, conductive nanomaterials, such as AuNPs and PNPs, possess exceptional electron mobility, large specific surface areas, and robust electrocatalytic activity. These features facilitate rapid electron transfer at the electrode surface, enabling wider linear ranges (e.g., PNPs cover 0.00000037–0.00037 µM) and lower LOD values (e.g., AuNPs/GCE at pH 3 achieves 0.44 nM). However, unmodified conductive nanomaterials lack specific recognition sites for uranyl ions, making them vulnerable to interference from coexisting ions and compromising detection selectivity—a critical drawback reflected in their limited practical applicability.
COFs bridge the gap between the low conductivity of pristine MOFs and the non-selectivity of bare nanomaterials. Their porous framework structure not only provides abundant active sites for UO22+ adsorption but also, when integrated with conductive components (e.g., AuNCs@COF), increases electron-transfer efficiency. This synergy explains the superior performance of COF-based sensors: AuNCs@COF achieves an ultra-low LOD of 0.347 nM across a wide linear range (0.0005–25 µM), while BCBA-TBTN-AO/GCE sets an impressive LOD of 0.00036 nM (0.000001–0.001 µM). These results validate that combining COFs with conductive nanomaterials addresses the conductivity limitation of pure frameworks while retaining their specific recognition capability.
For DNAzyme-based electrochemical sensors, their poor performance (e.g., SG@dsDNA@[Fe(CN)6]3−/4− has an LOD of 0.13 nM and a narrow linear range of 0.001–1 µM) arises from the intrinsic electro inactivity of DNAzymes. Unlike catalytically active nanomaterials or semiconducting frameworks, DNAzymes lack inherent electron-transfer capacity; their signal transduction relies on UO22+-induced DNA cleavage, which must be coupled with electroactive probes (e.g., [Fe(CN)6]3−/4−) or conductive substrates to generate a measurable electrochemical response. This multi-step coupling process introduces additional barriers to electron transfer and signal amplification, resulting in relatively higher LODs and narrower linear ranges compared with those for COF and nanomaterial-based sensors. Even the electrochemical biosensor reference shows a high LOD of 1200 nM, further highlighting the challenges of direct electrochemical detection with unmodified DNAzymes.
In summary, the performance differences in Table 3 are tightly governed by the electronic conductivity, specific recognition ability, and structural synergy of each material system. Composite structures, combining porous MOFs/COFs with conductive nanomaterials or coupling DNAzymes with electroactive transducers, have emerged as a universal strategy to overcome intrinsic limitations, enabling high sensitivity and wide linear ranges for UO22+ electrochemical detection.
5. Comparison of different detection methods
As shown in Table 4, a comprehensive performance comparison reveals that each detection method possesses distinct advantages and limitations, which are systematically summarized below.| Detection method | Linear range | LOD | Selectivity | Convenience | Response time |
|---|---|---|---|---|---|
| Fluorescence | Ordinary | Low | High | Ordinary | Rapid |
| Colorimetry | Narrow | High | Ordinary | Excellent | Fast |
| Electrochemical detection | Wide | Extremely low | Ordinary | Limited | Moderate |
Fluorescence-based detection exhibits a balanced profile, with superior sensitivity and selectivity. Through sophisticated probe design (e.g., DNAzymes) and signal amplification strategies, it can achieve ultra-low LODs at the nanomolar to picomolar level. The high selectivity is inherently derived from the specific molecular recognition between uranyl ions and functional probes (e.g., G-quadruplex structures or amidoxime groups), which effectively minimizes interference from coexisting ions. Operationally, this method offers convenience as it typically requires only sample-probe mixing without complex pretreatment, enabling quantitative analysis via fluorescence intensity changes. Kinetic studies benefit from the rapid response as fluorescence quenching or enhancement occurs instantaneously upon target binding, usually within minutes, facilitating real-time monitoring. However, its primary constraint lies in the reliance on sophisticated optical instrumentation (e.g., fluorometers), which restricts its applicability for field-deployed, on-site rapid testing and confines it mainly to laboratory settings.
Colorimetric detection excels in terms of operational convenience and speed, marked as “Excellent” and “Fast” in Table 4, making it the preferred choice for naked-eye, on-site analysis. It delivers results within seconds to minutes through visible color changes. However, this convenience comes at the cost of sensitivity, characterized by a “High” LOD and a “Narrow” linear range. This performance trade-off stems from the fundamental mechanism: colorimetric signals rely on the accumulation of sufficient chromophores to produce a measurable absorbance change. At low concentrations, the signal is too weak to be detected, while at high concentrations, color saturation occurs, leading to a non-linear relationship between intensity and concentration. Furthermore, the method is highly susceptible to environmental interferences. Its “Ordinary” selectivity is attributed to the high pH sensitivity of the chromogenic reaction; even minor changes in pH or direct competition from other metal ions can alter the reaction equilibrium, resulting in false signals or background interference.
Electrochemical detection stands out with the highest sensitivity, featuring an “Extremely low” LOD and a “Wide” linear range (Table 4), making it ideal for trace-level analysis in complex matrices. This high sensitivity stems from its direct signal-transduction mechanism, in which trace UO22+ undergoes rapid redox reactions at the electrode surface and generates measurable current signals, while efficient electron transfer and interfacial signal amplification further lower the detection limit and extend the linear range. Despite these advantages, the method suffers from notable practical limitations: electrode modification typically involves complex, time-consuming assembly steps that demand skilled operation and yield unstable interfaces, resulting in “Limited” convenience. Its “Moderate” response time can be attributed to the necessary adsorption and accumulation of UO22+ prior to effective signal generation, and its reliance on costly, specialized potentiostats prevents truly portable and real-time field applications. Moreover, its “Ordinary” selectivity arises from the inherent nature of electrochemical transduction, as other redox-active metal ions can produce overlapping interfering currents or compete for active sites, making it difficult to achieve exclusive recognition of uranyl ions without highly specific functional modification.
In summary, Table 4 clearly delineates the performance spectrum: fluorescence offers high sensitivity and speed but requires lab equipment; colorimetry provides unmatched on-site convenience but sacrifices linear range and sensitivity; electrochemistry delivers ultra-sensitivity but lacks portability and simplicity. This comparison underscores the importance of tailoring method selection to specific application scenarios, whether prioritizing laboratory accuracy, field portability, or trace-level detection.
To address these performance trade-offs and achieve comprehensive detection capabilities, integrating multiple complementary techniques into a single platform has emerged as a promising strategy. This dual-modal approach not only leverages the strengths of individual methods but also mitigates their respective limitations, thereby significantly enhancing the overall reliability and practical applicability of the sensor. Chen et al.111 developed an innovative dual-mode colorimetric and electrochemical sensor for UO22+ detection using AuNCs@COF composites (Fig. 16A and B). This material combines excellent electrocatalytic activity with optical properties, embedding nanoclusters with peroxidase-mimicking activity that catalyze substrates, inducing a color change in the presence of UO22+. Simultaneously, the composite on the electrode catalyzes electrochemical reactions with UO22+ to generate current signals. Dual measurement of electrochemical and colorimetric signals allows mutual validation, improving accuracy and reliability. Xiong et al.117 designed a fluorescence/colorimetric dual-mode sensing strategy based on cerium-sulfonated calix[4]arene (SC4A@Ce). This method cleverly exploits SC4A coordination with Ce3+ and Ce4+: Ce3+ fluorescence is quenched when bound to SC4A but restored upon the addition of UO22+, which displaces Ce3+. Meanwhile, Ce4+'s oxidase-like activity catalyzes TMB oxidation to oxTMB, changing the solution color from blue to colorless. By combining highly sensitive fluorescence with visible colorimetric analysis, this approach achieves enhanced sensitivity, accuracy, and reliability. Wu et al.118 developed a dual-mode colorimetric and photothermal sensor based on self-assembled TMB-CuO2 nanosheets for UO22+ detection. In acidic environments, the nanosheets spontaneously decompose to generate Cu2+, H2O2, and TMB in situ. A Fenton reaction then oxidizes TMB to blue oxTMB, which produces both a strong colorimetric signal and an efficient photothermal effect. UO22+ selectively coordinates with the imine groups of oxTMB. This coordination disrupts the conjugated structure of oxTMB, resulting in color fading, and simultaneously suppresses its photothermal conversion efficiency. Together, these effects produce a concentration-dependent dual signal response. By integrating the substrate, catalyst, and signal units into a single nanosheet without the need for external H2O2, this sensor achieves highly selective detection. It enables straightforward visual screening with the naked eye and on-site quantification based on thermal signal changes.
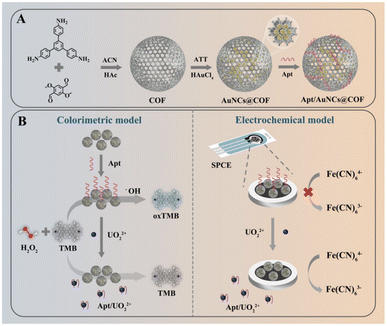 | ||
| Fig. 16 (A) Preparation of COF, AuNCs@COF, and Apt/AuNCs@COF. Reproduced with permission from ref. 111. Copyright 2025, Springer. (B) Schematic of the principles of colorimetric and electrochemical dual-mode sensor detection of UO22+. Reproduced with permission from ref. 111. Copyright 2025, Springer. | ||
6. Summary and outlook
In summary, this review provides a comprehensive examination of recent advancements in materials research for the optical and electrochemical detection of UO22+, with particular focus on MOFs, COFs, nanomaterials, and DNA-based materials. These materials offer distinct advantages in the selective and sensitive detection of UO22+, contributing to the development of efficient monitoring platforms. Despite the remarkable breakthroughs achieved in this field, several challenges persist for the practical application of UO22+ detection.6.1 Challenges in practical deployment
In summary, the practical deployment of UO22+ detection technologies is constrained not merely by analytical performance, but by a combination of matrix interference, material instability, instrumentation and operational barriers, performance–practicality trade-offs and scalability issues. Addressing these challenges requires a paradigm shift from lab-scale optimization of detection limits and selectivity to design optimization for real-world applications. This entails integrating material engineering, matrix adaptation and device miniaturization to develop sensing systems that are stable, portable, low-cost and robust in complex environmental conditions, a critical focus for translating the remarkable advances in functional materials outlined in this review into actionable, field-deployable uranium monitoring technologies.
6.2 Future improvement directions
Looking ahead, future research on UO22+ detection should focus on the development of multifunctional composite materials that integrate optical, electrochemical or multimodal sensing capabilities with enhanced selectivity, sensitivity and environmental stability. Such materials will address the core practical challenges, including complex matrix interference, poor material stability in harsh field settings, instrumentation and operational limitations, as well as scalability and cost constraints. Leveraging advances in computational chemistry and machine learning will yield valuable atomic-level insights into UO22+ recognition mechanisms and material degradation pathways, enabling the rational, predictive design of high-performance sensing materials, moving beyond empirical lab-scale optimization. More importantly, the seamless integration of such advanced materials with miniaturized, portable and field-adaptable detection devices, together with simplified, automated operational workflows and pretreatment-free sensing systems, will be pivotal to realizing real-time, on-site and reliable uranium contamination monitoring. Ultimately, the translation of these lab-scale innovations into efficient, cost-effective and robust UO22+ detection technologies will play a vital role in safeguarding the environment and human health, supporting the sustainable development of global nuclear energy and facilitating effective pollution control and environmental remediation at uranium-contaminated sites. By addressing the practical deployment challenges outlined in this review, we can advance uranium monitoring technologies that deliver long-term value for environmental protection and a more sustainable future.
Author contributions
KaiZeng Quan: writing – review & editing, writing – original draft, investigation. YuMin Wang: writing – review & editing, writing – original draft, investigation. Meng Qi: writing – original draft. Peng Xu: supervision, methodology. YaXing Wang: supervision, methodology. GuoXun Ji: writing – review & editing, investigation.Conflicts of interest
There are no conflicts to declare.Data availability
No primary research results, software or codes have been included, and no new data were generated or analysed as part of this review.Acknowledgements
This work was supported by the National Natural Science Foundation of China (22476144).Notes and references
- L. Zhan, Y. Bo, T. Lin and Z. Fan, Development and outlook of advanced nuclear energy technology, Energy Strategy Rev., 2021, 34, 100630 CrossRef.
- C. Degueldre, Uranium as a renewable for nuclear energy, Prog, Nucl. Energy, 2017, 94, 174–186 CAS.
- F. E. Arhouni, M. A. Saleh Abdo, S. Ouakkas, M. L. Bouhssa and A. Boukhair, Uranium recovery from Moroccan Phosphate rock for nuclear-powered desalination and water security: A review, Radiat. Phys. Chem., 2025, 234, 112802 CrossRef CAS.
- A. Durakovic, Medical effects of internal contamination with actinides: further controversy on depleted uranium and radioactive warfare, Environ. Health Prev. Med., 2016, 21, 111–117 CrossRef CAS PubMed.
- G. Steinhauser, A. Brandl and T. E. Johnson, Comparison of the Chernobyl and Fukushima nuclear accidents: A review of the environmental impacts, Sci. Total Environ., 2014, 470–471, 800–817 CrossRef CAS PubMed.
- S. Chandrakar, S. Sahu, S. Sharma and M. Pandey, A Review on the Efficiency of Adsorbents for Uranium Pollutant Remediation from Wastewater, Trans. Indian Inst. Met., 2025, 78, 55 CrossRef CAS.
- G. Bjørklund, Y. Semenova, L. Pivina, M. Dadar, M. M. Rahman, J. Aaseth and S. Chirumbolo, Uranium in drinking water: a public health threat, Arch. Toxicol., 2020, 94, 1551–1560 CrossRef PubMed.
- E. Schnug and B. G. Lottermoser, Fertilizer-Derived Uranium and its Threat to Human Health, Environ. Sci. Technol., 2013, 47, 2433–2434 CrossRef CAS PubMed.
- A. K. Bairwa, K. Saini, S. K. Sahoo, S. N. Tiwari, T. Menaria and K. Gupta, Health risk study of groundwater uranium and terrestrial gamma radiation in Sawai Madhopur, Rajasthan (India), J. Radioanal. Nucl. Chem., 2025, 334, 6077–6089 CrossRef CAS.
- W. T. Yang, Q. H. Pan, S. Y. Song and H. J. Zhang, Metal-organic framework-based materials for the recovery of uranium from aqueous solutions, Inorg. Chem. Front., 2019, 6, 1924–1937 RSC.
- W. W. He and D. B. Hua, Spectrographic sensors for uranyl detection in the environment, Talanta, 2019, 201, 317–329 CrossRef CAS PubMed.
- P. Panwar, A. Joshi, K. P. Singh, M. Prasad, R. Mehra, S. K. Sahoo and R. C. Ramola, Distribution of uranium and selected toxic heavy metals in drinking water of Garhwal Himalaya, India, J. Radioanal. Nucl. Chem., 2024, 333, 2927–2935 CrossRef CAS.
- X. K. Shen and Y. F. Lu, Detection of uranium in solids by using laser-induced breakdown spectroscopy combined with laser-induced fluorescence, Appl. Opt., 2008, 47, 1810–1815 CrossRef CAS PubMed.
- M. Rezaee and F. Khalilian, Determination of uranium in water samples using homogeneous liquid-liquid microextraction via flotation assistance and inductively coupled plasma-optical emission spectrometry, J. Radioanal. Nucl. Chem., 2015, 304, 1193–1200 CrossRef CAS.
- H. Peng, H. Nan, S. Li, Y. Wang, Z. Dong, Z. Zhang, X. Cao and Y. Liu, Covalently construction of C-GSH-AuNCs@UiO-66-NH2 composites as an efficient turn-on fluorescence sensor for UO22+, J. Solid State Chem., 2024, 334, 124650 CrossRef CAS.
- Y. Zhan, X. Lu, T. Lan, Q. Tian, J. Shen and W. He, An AIE fluorescence sensor based on amidoxime for rapid detection of uranyl ions in aqueous solutions, Dyes Pigm., 2023, 216, 111299 CrossRef CAS.
- S. J. Xiao, J. Huang, A. T. Qiu, G. Z. Liu, L. Zhang, T. Wu, Y. D. Shi and J.-D. Qiu, Advanced “turn-on” colorimetric uranium platform based on the enhanced nanozyme activity of a donor-acceptor structured covalent organic framework, Anal. Chim. Acta, 2024, 1302, 342503 CrossRef CAS PubMed.
- X. Wu, Q. Huang, Y. Mao, X. Wang, Y. Wang, Q. Hu, H. Wang and X. Wang, Sensors for determination of uranium: A review, TrAC, Trends Anal. Chem., 2019, 118, 89–111 CrossRef CAS.
- K. Wu, Y. Zhou, Z. Zhou, X. Liang, J. Ma and L. Zhou, Electrochemical sensor modified with poly(toluidine blue) for monitoring trace uranium in natural water by stripping voltammetry, J. Radioanal. Nucl. Chem., 2023, 332, 3893–3901 CrossRef CAS.
- L. Zhang, M. Jia, X. Wang, L. Gao, B. Zhang, L. Wang, J. Kong and L. Li, A novel fluorescence sensor for uranyl ion detection based on a dansyl-modified peptide, Spectrochim. Acta, Part A, 2023, 292, 122403 CrossRef CAS PubMed.
- S. H. Park, H. S. Kim and B. S. You, Improving the tensile strength of Mg-7Sn-1Al-1Zn alloy through artificial cooling during extrusion, Mater. Sci. Eng., A, 2015, 625, 369–373 CrossRef CAS.
- X. Tang, H. Han, L. Li and H. Wang, Electrodes functionalized with advanced recognition materials for trace electrochemical sensing of uranyl ion, Microchem. J., 2024, 199, 109924 CrossRef CAS.
- J. Luo, X. Luo, Y. Gan, X. Xu, B. Xu, Z. Liu, C. Ding, Y. Cui and C. Sun, Advantages of Bimetallic Organic Frameworks in the Adsorption, Catalysis and Detection for Water Contaminants, Nanomaterials, 2023, 13(15), 2194 CrossRef CAS PubMed.
- Q. Liu, Y. Yang, Y. Zou, L. Wang, Z. Li, M. Wang, L. Li, M. Tian, D. Wang and D. Gao, Fluorescent covalent organic frameworks for environmental pollutant detection sensors and enrichment sorbents: a mini-review, Anal. Methods, 2023, 15, 5919–5946 RSC.
- L. Farzin, M. Shamsipur, S. Sheibani, L. Samandari and Z. Hatami, A review on nanomaterial-based electrochemical, optical, photoacoustic and magnetoelastic methods for determination of uranyl cation, Microchim. Acta, 2019, 186, 289 CrossRef PubMed.
- L. Sun, J. Liu, L. Li, D. Zhen, Z. Dai, S. Tang, B. Zhu, L. Chen, H. Chen, M. Gong, Z. Tang and Y. Hu, Advances of biosensors for UO22+ detecting based on specific DNAzyme, Inorg. Chem. Commun., 2022, 138, 109234 CrossRef CAS.
- J. Perego, I. Villa, A. Pedrini, E. C. Padovani, R. Crapanzano, A. Vedda, C. Dujardin, C. X. Bezuidenhout, S. Bracco, P. E. Sozzani, A. Comotti, L. Gironi, M. Beretta, M. Salomoni, N. Kratochwil, S. Gundacker, E. Auffray, F. Meinardi and A. Monguzzi, Composite fast scintillators based on high-Z fluorescent metal-organic framework nanocrystals, Nat. Photonics, 2021, 15, 393–400 CrossRef CAS.
- W. Cui, Y. Chen, W. Xu, K. Liu, W. Qiu, Y. Li and J. Qiu, A three-dimensional luminescent covalent organic framework for rapid, selective, and reversible uranium detection and extraction, Sep. Purif. Technol., 2023, 306, 122726 CrossRef CAS.
- Y. Bai, L. Xu, H. Chai, L. Zhou, G. Jiang and G. Zhang, Recent Advances on DNAzyme-Based Biosensors for Detection of Uranyl, Front. Chem., 2022, 10, 882250 CrossRef CAS PubMed.
- H. Liu, T. Fu and Y. Mao, Metal-Organic Framework-Based Materials for Adsorption and Detection of Uranium(VI) from Aqueous Solution, ACS Omega, 2022, 7, 14430–14456 CrossRef CAS PubMed.
- N. Rani, P. Singh, S. Kumar, P. Kumar, V. Bhankar, N. Kamra and K. Kumar, Recent advancement in nanomaterials for the detection and removal of uranium: A review, Environ. Res., 2023, 234, 116536 CrossRef CAS PubMed.
- M. Sharma, P. Taneja, L. Yadav, P. Sharma, V. C. Janu and R. Gupta, Current insights into the implementation of electrochemical sensors for comprehensive detection and analysis of radionuclides, Trends Anal. Chem., 2024, 178, 117845 CrossRef CAS.
- J. Sun, D. Lin, Q. Wu, Z. Hong, X. Chen, Z. Liu, X. Liu, J. Cui, Z. Yuan and X. Xiao, Advances in CDs composites toward detection and adsorption of uranium and other radioactive elements: A review, J. Environ. Chem. Eng., 2025, 13, 118004 CrossRef CAS.
- C. Poulin-Ponnelle, D. Boudreau and D. Lariviere, Fluorescent Probes for Monitoring Toxic Elements from the Nuclear Industry: A Review, Sensors, 2025, 25, 5835 CrossRef CAS PubMed.
- A. Amini, M. Khajeh, A. R. Oveisi, S. Daliran, M. Ghaffari-Moghaddam and H. S. Delarami, A porous multifunctional and magnetic layered graphene oxide/3D mesoporous MOF nanocomposite for rapid adsorption of uranium(VI) from aqueous solutions, J. Ind. Eng. Chem., 2021, 93, 322–332 CrossRef CAS.
- Y. Luo, N. Li, J. Tian, J. Pan, R. Su, V. Turkevich and L. Wang, Two birds with one stone strategy: selective uranium extraction and detection by a fluorescent covalent organic framework with high-affinity binding sites, J. Mater. Chem. A, 2025, 13, 37458–37468 RSC.
- S. Sauge-Merle, M. Recuerda, M. R. Beccia, D. Lemaire, R. Cherif, N. Bremond, F. Merola, Y. Bousmah and C. Berthomieu, Development of an Efficient FRET-Based Ratiometric Uranium Biosensor, Biosensors, 2023, 13(5), 561 CrossRef CAS PubMed.
- S. Jampasa, W. Khamcharoen, S. Wirojsaengthong, A. Suea-Ngam, S. Traipop, T. Ozer, F. Unob, P. Puthongkham and O. Chailapakul, Recent advances and trends in the applications of nanomaterials in optical sensing platforms, TrAC, Trends Anal. Chem., 2024, 180, 117914 CrossRef CAS.
- X. Jiang, R. Yang, X. Lei, S. Xue, Z. Wang, J. Zhang, L. Yan, Z. Xu, Z. Chen, P. Zou and G. Wang, Design, Synthesis, Application and Research Progress of Fluorescent Probes, J. Fluoresc., 2023, 34, 965–975 CrossRef PubMed.
- L. Jiao, J. Y. R. Seow, W. S. Skinner, Z. U. Wang and H.-L. Jiang, Metal-organic frameworks: Structures and functional applications, Mater. Today, 2019, 27, 43–68 CrossRef CAS.
- L. Huang, Z. Liu and C. Tao, Strategies for constructing metal-organic frameworks-based biosensors and their applications in environmental pollutant detection, J. Environ. Chem. Eng., 2025, 13(3), 116806 CrossRef CAS.
- Q. Yu, M. Wang, X. Feng and X. Li, Research Progress of Rare Earth Metal-Organic Frameworks on Pollutant Monitoring, Chemosensors, 2025, 13(5), 184 CrossRef CAS.
- M. D. Allendorf, C. A. Bauer, R. K. Bhakta and R. J. T. Houk, Luminescent metal-organic frameworks, Chem. Soc. Rev., 2009, 38, 1330–1352 RSC.
- R. Huang, Z. Yu, Z. Li, X. Lin, J. Hou, Z. Hu and J. Zou, Photoluminescent metal-organic frameworks and their composites: Fundamentals, applications, and outlooks, Coord. Chem. Rev., 2025, 526, 216358 CrossRef CAS.
- X. Guo, L. Zhou, X. Liu, G. Tan, F. Yuan, A. Nezamzadeh-Ejhieh, N. Qi, J. Liu and Y. Peng, Fluorescence detection platform of metal-organic frameworks for biomarkers, Colloids Surf., B, 2023, 229, 113455 CrossRef CAS PubMed.
- L. Feng, H. Wang, T. Feng, B. Yan, Q. Yu, J. Zhang, Z. Guo, Y. Yuan, C. Ma, T. Liu and N. Wang, In Situ Synthesis of Uranyl-Imprinted Nanocage for Selective Uranium Recovery from Seawater, Angew. Chem., Int. Ed., 2022, 61, 82–86 Search PubMed.
- H. Zhang, Y. Wang, W. Yan, J. Jin, Y. Wang, Y. Wang, J. Zhang, G. Wang, H. Dong and S. Zhang, Research progress of LMOFs containing aromatic carboxylic acid in anions recognition, Microchem. J., 2024, 200, 110453 CrossRef CAS.
- X. Li, H. Hao, L. Liu, H. Ren, J. Yin, J. Wang, L. Zhang, X. Zhang and L. Fan, Recent Advances in Metal-Organic Framework-Based Luminescent Sensors toward 3-Nitrotyrosine Biomarker, Cryst. Growth Des., 2025, 25, 7880–7902 CrossRef CAS.
- W. Hu, Y. Yang, S. Qie, Z. Mu, R. Xu, W. Li, Y. Chen, J. Qiao, L. Yue and M. Hu, The post-synthetic modification of a Cd-MOF and its fluorescence sensing properties, Spectrochim. Acta, Part A, 2026, 346, 126867 CrossRef CAS PubMed.
- S. Jain, M. Nehra, R. Kumar, N. Dilbaghi, K. Kim and S. Kumar, Development of a FRET aptasensor based on MoS2-doped Zn-MOF as luminophore for selective detection of cadmium in aqueous solutions, Microchim. Acta, 2024, 191, 324 CrossRef CAS PubMed.
- L. Zhang, Y. He, Y. Wu, J. Zhang, S. Li and Z. Zhang, Highly sensitive ratiometric fluorescence detection of tetracycline residues in food samples based on Eu/Zr-MOF, Food Chem., 2024, 436, 137717 CrossRef CAS PubMed.
- X. Qin, W. Yang, Y. Yang, D. Gu, D. Guo and Q. Pan, A Zinc Metal–Organic Framework for Concurrent Adsorption and Detection of Uranium, Inorg. Chem., 2020, 59, 9857–9865 CrossRef CAS PubMed.
- L. Chen, D. Liu, J. Peng, Q. Du and H. He, Ratiometric fluorescence sensing of metal-organic frameworks: Tactics and perspectives, Coord. Chem. Rev., 2020, 404, 213113 CrossRef CAS.
- J. Zhang, Y. Ren, T. Xia, Y. Du, L. Shao, H. Tang and S. Yang, Post-synthesis metal-organic framework for turn-on ratiometric fluorescence sensing of UO22+, Z. Anorg. Allg. Chem., 2021, 648, 41–46 Search PubMed.
- Y. Tong, L. Yu, X. Gong, L. Wu, Y. Chen, D. Wang, Y. Ye, F. Zhu, Z. Gong, J. Xu and G. Ouyang, On-Site Ratiometric Analysis of UO22+ with High Selectivity, Anal. Chem., 2024, 96, 3070–3076 CAS.
- N. Huang, P. Wang and D. Jiang, Covalent organic frameworks: a materials platform for structural and functional designs, Nat. Rev. Mater., 2016, 1, 16068 CrossRef CAS.
- X. Li, S. Cai, B. Sun, C. Yang, J. Zhang and Y. Liu, Chemically Robust Covalent Organic Frameworks: Progress and Perspective, Matter, 2020, 3, 1507–1540 CrossRef.
- S. Raghav, J. Raghav, M. a. A. Muhammad, M. B. Singh, P. Jain and D. Kumar, Recent advancements in synthesis and applications of covalent organic frameworks, Appl. Mater. Today, 2025, 44, 102694 CrossRef.
- S. Liu, G. Liang, S. Wang and Q. Hu, Recent multifunctional applications of AIE-MOF/COF porous materials, CrystEngComm, 2025, 27, 3643–3658 RSC.
- L. Guo, L. Yang, M. Li, L. Kuang, Y. Song and L. Wang, Covalent organic frameworks for fluorescent sensing: Recent developments and future challenges, Coord. Chem. Rev., 2021, 440, 213957 CrossRef CAS.
- D. Zhen, C. Liu, Q. Deng, S. Zhang, N. Yuan, L. Li and Y. Liu, A review of covalent organic frameworks for metal ion fluorescence sensing, Chin. Chem. Lett., 2024, 35(8), 109249 CrossRef CAS.
- W. Cui, C. Zhang, W. Jiang, F. Li, R. Liang, J. Liu and J. Qiu, Regenerable and stable sp2 carbon-conjugated covalent organic frameworks for selective detection and extraction of uranium, Nat. Commun., 2020, 11, 436 CrossRef CAS PubMed.
- D. Zhen, C. Liu, Q. Deng, L. Li, C. A. Grimes, S. Yang, Q. Cai and Y. Liu, Novel Olefin-Linked Covalent Organic Framework with Multifunctional Group Modification for the Fluorescence/Smartphone Detection of Uranyl Ion, ACS Appl. Mater. Interfaces, 2024, 16, 27804–27812 CrossRef CAS PubMed.
- T. A. Saleh, Trends in nanomaterial types, synthesis methods, properties and uses: Toxicity, environmental concerns and economic viability, Nano-Struct. Nano-Objects, 2024, 37, 101109 CrossRef CAS.
- Y. Pan, Z. Han, S. Chen, K. Wei and X. Wei, Metallic nanoclusters: From synthetic challenges to applications of their unique properties in food contamination detection, Coord. Chem. Rev., 2023, 478, 214964 CrossRef CAS.
- R. Jin and T. Higaki, Open questions on the transition between nanoscale and bulk properties of metals, Commun. Chem., 2021, 4, 28 CrossRef CAS PubMed.
- D. Feng, G. Zhang and Y. Li, Semiconductor Quantum Dots: Synthesis, Properties and Applications, Nanomaterials, 2024, 14(22), 1825 CrossRef CAS PubMed.
- M. R. Kumalasari, R. Alfanaar and A. S. Andreani, Gold nanoparticles (AuNPs): A versatile material for biosensor application, Talanta Open, 2024, 9, 100327 CrossRef.
- H. Nan, Y. Liu, W. Gong, H. Peng, Y. Wang, Z. Zhang and X. Cao, An inner-filter-effect based ratiometric fluorescent sensor for the detection of uranyl ions in real samples, Anal. Methods, 2022, 14, 532–540 RSC.
- M. B. Cánchig, F. López, Z. Morales-Navarro, A. Debut, K. Vizuete, T. Terencio, M. Caetano and J. P. Saucedo-Vázquez, Enhanced selectivity of carbon quantum dots for metal ion detection through surface modification by heteroatom doping: A study on optical properties and theoretical approach, Carbon Trends, 2025, 18, 100445 CrossRef.
- Z. J. Zheng, L. Zhang, L. Z. Wang, Z. Q. Zhong, Y. T. Xiong, J. Guo, Z. B. Zhang, X. H. Cao and S. J. Xiao, Ultrasensitive detection of UO22+ based on dopamine-functionalized MoOx quantum dots, Luminescence, 2021, 37, 127–133 CrossRef PubMed.
- M. Hollenstein, DNA Catalysis: The Chemical Repertoire of DNAzymes, Molecules, 2015, 20, 20777–20804 CrossRef CAS PubMed.
- B. Liu, G. Chen, A. M. Abd El-Aty, R. Zhai, G. Liu, X. Xu, Y. Zhang, L. Li, J. Zhang and D. Xu, Advances of functional nucleic acids based on specific recognition:A review, Int. J. Biol. Macromol., 2025, 304, 140828 CrossRef CAS PubMed.
- Z. Xiong, Q. Wang, J. Zhang, W. Yun, X. Wang, X. Ha and L. Yang, A simple and programmed DNA tweezer probes for one-step and amplified detection of UO22+, Spectrochim. Acta, Part A, 2020, 229, 118017 CrossRef CAS PubMed.
- J. Chen, X. Wang, J. Guo, Y. Lv, M. Chen, H. Tong and C. Liu, Heavy Metal-Induced Assembly of DNA Network Biosensor from Double-Loop Hairpin Probes for Ultrasensitive Detection of UO22+ in Water and Soil Samples, Anal. Chem., 2024, 96, 3153–3159 CAS.
- F. Lin, Y. Cheng, M. Li, Z. Li and J. Dai, Detection of uranyl ions by single-hairpin based self-hybridization chain reaction, Talanta, 2025, 285, 127374 CrossRef CAS PubMed.
- A. Kalintsev, A. Migdisov, J. Brugger and H. Xu, Uranium(VI) hydrolysis up to 250 °C and its geological implications, Geochim. Cosmochim. Acta, 2024, 377, 68–83 CrossRef CAS.
- B. Pramanik, R. Sahoo and M. C. Das, pH-stable MOFs: Design principles and applications, Coord. Chem. Rev., 2023, 493, 215301 CrossRef CAS.
- R. Wei, X. Luo, G. Ning and D. Li, Covalent Metal-Organic Frameworks: Fusion of Covalent Organic Frameworks and Metal-Organic Frameworks, Acc. Chem. Res., 2025, 58, 746–761 CrossRef CAS PubMed.
- W. Liu, X. Dai, Z. Bai, Y. Wang, Z. Yang, L. Zhang, L. Xu, L. Chen, Y. Li, D. Gui, J. Diwu, J. Wang, R. Zhou, Z. Chai and S. Wang, Highly Sensitive and Selective Uranium Detection in Natural Water Systems Using a Luminescent Mesoporous Metal-Organic Framework Equipped with Abundant Lewis Basic Sites: A Combined Batch, X-ray Absorption Spectroscopy, and First Principles Simulation Investigation, Environ. Sci. Technol., 2017, 51, 3911–3921 CrossRef CAS PubMed.
- N. Seal, A. Karmakar, S. Kundu and S. Neogi, Pendent carboxylic acid-fuelled high-performance uranium extraction in a hydrogen-bonded framework and prolifically improved water oxidation via post-metalation-actuated composite fabrication, J. Mater. Chem. A, 2024, 12, 3501–3512 RSC.
- V. Venkata Sravani, S. Tripathi, B. Sreenivasulu, S. Kumar, S. Maji, C. V. S. Brahmmananda Rao, A. Suresh and N. Sivaraman, Post synthetically modified IRMOF-3 for efficient recovery and selective sensing of U(vi) from aqueous medium, RSC Adv., 2021, 11, 28126–28137 RSC.
- C. Zhang, W. Cui, W. Jiang, F. Li, Y. Wu, R. Liang and J. Qiu, Simultaneous sensitive detection and rapid adsorption of UO22+ based on a post-modified sp2 carbon-conjugated covalent organic framework, Environ. Sci.: Nano, 2020, 7, 842–850 RSC.
- W. Cui, W. Xu and W. Qiu, Constructing an ultrastable imidazole covalent organic framework for concurrent uranium detection and recovery, Ecotoxicol. Environ. Saf., 2023, 252, 114639 CrossRef CAS PubMed.
- Q. Wang, T. Han, C. Miao, W. Qin and X. Wu, A fluorescent N-doped carbon dot-hydrogel composite for concurrent selective detection and local hot spot promoted adsorption of uranium(vi), Environ. Sci.:Water Res. Technol., 2023, 9, 2680–2691 RSC.
- R. Lu, Y. Luo, L. Su, S. Ye, X. Wang, W. Ren, J. Zhang, F. Zhao and C. Zheng, Field Detection of Uranyl in Coastal Water of China Using a Portable Device via DNA Photocleavage, Anal. Chem., 2024, 96, 11525–11532 CrossRef CAS PubMed.
- X. Zhang, Y. Wang, M. Gong, L. Xiong, J. Song, S. Chen, Y. Tong, Y. Liu, L. Li and D. Zhen, Engineering an upconversion fluorescence sensing platform with “off-on” pattern through specific DNAzyme-mediated signal amplification for supersensitive detection of uranyl ion, Microchim. Acta, 2024, 191, 503 CrossRef CAS PubMed.
- I. Ahmad, T. Muhmood, A. Rehman, M. Zahid, M. Abohashrh, S. Nishat, Y. Raharjo, Z. Zhou and X. Yang, Zeolite imidazole framework entrapped quantum dots (QDs@ZIF-8): encapsulation, properties, and applications, J. Taiwan Inst. Chem. Eng., 2023, 149, 104993 CrossRef CAS.
- A. Cui, X. Wu, J. Ye, G. Song, D. Chen, J. Xu, Y. Liu, J. Lai and H. Sun, “Two-in-one” dual-function luminescent MOF hydrogel for onsite ultra-sensitive detection and efficient enrichment of radioactive uranium in water, J. Hazard. Mater., 2023, 448, 130864 CrossRef CAS PubMed.
- X. Mao, Y. Cai, Q. Luo, X. Liu, Q. Jiang, C. Zhang, L. Zhang, R. Liang and J. Qiu, Europium(III) Functionalized Covalent Organic Framework as Sensitive and Selective Fluorescent Switch for Detection of Uranium, Anal. Chem., 2024, 96, 5037–5045 CrossRef CAS PubMed.
- S. Yang, W. Li, Z. Li, X. Zeng, Q. Wang, C. Yan, L. Pan, Y. Chen, M. Mu, C. Li and W. Yang, Water-soluble, SDS-functionalized CdSe/CdS/ZnS quantum dots for uranium detection in aqueous environments, Ceram. Int., 2024, 50, 23493–23499 CrossRef CAS.
- F. Yang and L. Ge, Colorimetric Sensors: Methods and Applications, Sensors, 2023, 23(24), 9887 CrossRef PubMed.
- S. Lee, H. Yoo and J. Y. Kim, Recent advances on MOF-based colorimetric sensors, Bull. Korean Chem. Soc., 2025, 46, 918–939 CrossRef CAS.
- Z. Zhou, W. He, H. Chao, H. Wang, P. Su, J. Song and Y. Yang, Insertion of Hemin into Metal-Organic Frameworks: Mimicking Natural Peroxidase Microenvironment for the Rapid Ultrasensitive Detection of Uranium, Anal. Chem., 2022, 94, 6833–6841 CrossRef CAS PubMed.
- Y. Feng, Y. Xu, S. Liu, D. Wu, Z. Su, G. Chen, J. Liu and G. Li, Recent advances in enzyme immobilization based on novel porous framework materials and its applications in biosensing, Coord. Chem. Rev., 2022, 459, 214414 CrossRef CAS.
- Y. Xu, J. Wei and X. Chen, Visible Light-Responsive Sulfone-Based Covalent Organic Framework as Metal-Free Nanoenzyme for Visual Colorimetric Determination of Uranium, Chemosensors, 2022, 10(7), 248 CrossRef CAS.
- S. Sangwan and R. Seth, Synthesis, Characterization and Stability of Gold Nanoparticles (AuNPs) in Different Buffer Systems, J. Cluster Sci., 2021, 33, 749–764 CrossRef.
- L. Zhang, D. Huang, P. Zhao, G. Yue, L. Yang and W. Dan, Colorimetric detection for uranyl ions in water using vinylphosphonic acid functionalized gold nanoparticles based on smartphone, Spectrochim. Acta, Part A, 2022, 269, 120748 CrossRef CAS PubMed.
- L. Zhang, S. Shi, P. Xiong, L. Chen, J. Xu, J. Jiang, S. Yang and H. Wu, DNAzyme recognition triggered cascade signal amplification for rapid and highly sensitive visual detection of uranyl ions, Analyst, 2022, 147, 4158–4166 RSC.
- Y. Lei, Y. Li, C. Liu, L. He, Q. Li, L. Liao and X. Xiao, Smartphone-assisted colorimetric determination of uranyl ions in aqueous solutions, New J. Chem., 2023, 47, 4667–4673 RSC.
- S. A. Hashemi, A. Ghaffarkhah, A. A. Isari, M. Panahi-Sarmad, F. Jiang, O. J. Rojas, S. Wuttke, M. Dincă and M. Arjmand, Advancing from MOFs and COFs to Functional Macroscopic Porous Constructs, Adv. Mater., 2025, 37, 2411617 CrossRef CAS PubMed.
- J. Song, L. Chai, M. Zhao, Y. Sun, X. Li and J. Pan, J. Research progress on metal organic framework derived porous carbon though interfacial engineering and synergistic effect, Power Sources, 2024, 604, 234471 CrossRef CAS.
- X. Cao, Y. Sun, Y. Wang, Z. Zhang, Y. Dai, Y. Liu, Y. Wang and Y. Liu, PtRu bimetallic nanoparticles embedded in MOF-derived porous carbons for efficiently electrochemical sensing of uranium, J. Solid State Electrochem., 2020, 25, 425–433 CrossRef.
- B. Sun and L. Cui, Rational Design of Covalent Organic Frameworks for Enhanced Reticular Electrochemiluminescence and Biosensing Applications, Biosensors, 2025, 15(11), 760 CrossRef CAS PubMed.
- W. Cui, Y. Li, Q. Jiang, Q. Wu, Q. Luo, L. Zhang, R. Liang and J. Qiu, Covalent Organic Frameworks as Advanced Uranyl Electrochemiluminescence Monitoring Platforms, Anal. Chem., 2021, 93, 16149–16157 CrossRef CAS PubMed.
- E. Kianfar and H. Sayadi, Recent advances in properties and applications of nanoporous materials and porous carbons, Carbon Lett., 2022, 32, 1645–1669 CrossRef.
- J. Han, J. Gao, J. Chen, G. Qi, Y. Gui, Y. Wang, Y. Yan, F. Ma, Q. Chen and Y. Xue, Electrochemical measurements of uranyl ions in real nuclear wastewater using nanogold-modified electrodes, Microchem. J., 2025, 212, 113340 CrossRef CAS.
- J. Wang, H. Zhou, J. Liu, J. He, J. Liu and W. Yang, Electrochemical detection of DNA by formation of efficient electron transfer pathways through adsorbing gold nanoparticles to DNA modified electrodes, Microchem. J., 2021, 169, 106581 CrossRef CAS.
- S. Guo, J. Lin, L. Lin, W. Xu, Y. Guo, Z. Xu, C. Lu, X. Shi, L. Chen and H. Yang, Selecting small molecule DNA aptamers with significant conformational changes for constructing transcriptional switches and biosensors, Sci. China:Chem., 2023, 66, 1529–1536 CAS.
- S. E. Jett and A. J. Bonham, Reusable Electrochemical DNA Biosensor for the Detection of Waterborne Uranium, ChemElectroChem, 2017, 4, 843–845 CrossRef CAS.
- Z. Chen, J. Liu, W. Wang, G. Qin, S. Liu, W. Zhang, C. Peng, Y. Tan, Z. Dai, D. Zhen and L. Li, Aptamer-regulated colorimetric and electrochemical dual-mode sensor for the detection of uranyl ions utilizing AuNCs@COF composite, Microchim. Acta, 2025, 192, 295 CrossRef CAS PubMed.
- S. Shi, H. Wu, L. Zhang, S. Wang, P. Xiong, Z. Qin, M. Chu and J. Liao, Gold nanoparticles based electrochemical sensor for sensitive detection of uranyl in natural water, J. Electroanal. Chem., 2021, 880, 114884 CrossRef CAS.
- Z. Wang, H. Gao, P. Liu, X. Wu, Q. Li, J. Xu and D. Hua, Visualized uranium rapid monitoring system based on self-enhanced electrochemiluminescence-imaging of amidoxime functionalized polymer nanoparticles, Chin. Chem. Lett., 2022, 33, 3456–3460 CrossRef CAS.
- M. Xiang, C. Yin, Q. Chen, G. Zhu, Z. Chen, H. Li, R. Antwi-Baah, J. Tang, K. Yang and S. Xu, A label-free electrochemical biosensor for detection of uranyl ions in water based on nucleic acid dye-inducing turn-off of photosensitization, Microchem. J., 2025, 213, 113811 CrossRef CAS.
- C. Jiang, X. Tang, B. He, Y. Ouyang, R. Xiang and L. Li, Amidoxime-grafted fluorinated graphene nanosheets as a trace electrochemical sensing platform of uranyl ion, Microchem. J., 2024, 205, 111371 CrossRef CAS.
- L. Chen, J. Liu, C. Cao, S. Tang, C. Lv, X. Xiao, S. Yang, L. Liu, L. Sun, B. Zhu and L. Li, Dual-signal amplification electrochemical sensing for the sensitive detection of uranyl ion based on gold nanoparticles and hybridization chain reaction-assisted synthesis of silver nanoclusters, Anal. Chim. Acta, 2021, 1184, 338986 CrossRef CAS PubMed.
- L. Xiong, Y. Tong, J. Song, S. Chen, Y. Liu, J. Liu, L. Li and D. Zhen, Smartphone-assisted fluorescence/colorimetric dual-mode sensing strategy for uranium ion detection using cerium-sulfonyl calix[4]arene, Microchim. Acta, 2025, 192, 158 CrossRef CAS PubMed.
- Y. Wu, X. Tan, H. Gong, M. Wang, G.-l. Wu, N. Li, F. Liu, H. Xia, L. Tang and Q. Yang, Self-assembled TMB-CuO2 nanosheets for dual-mode colorimetric and NIR-II photothermal detection of uranyl ion, Anal. Chim. Acta, 2025, 1356, 344041 CrossRef CAS PubMed.
Footnote |
| † These authors contributed equally. |
| This journal is © The Royal Society of Chemistry 2026 |