Open Access Article
Open Access ArticleDesign, synthesis and bioactivity of indobufen derivatives
Danli Yuan†
b,
Liping Chen†c,
Xin Huang†b,
Jinquan Zhang *b and
Ziyan Meng*a
*b and
Ziyan Meng*a
aKey Laboratory of Green and Healthy Livestock and Poultry Breeding Technology of Jiangxi Education Institutes, Ganzhou Polytechnic College, Ganzhou, 341000, China. E-mail: mengzy@gzpt.edu.cn
bGannan Medical University, Ganzhou, 341000, China
cGanzhou People's Hospital, Ganzhou, 341000, China
First published on 24th April 2026
Abstract
Osteoporosis is characterized by excessive osteoclast-mediated bone resorption, a process that can be amplified by inflammatory cues within the bone microenvironment. Targeting the inflammation–osteoclast axis has therefore emerged as a promising strategy for preventing pathological bone loss. Here, a series of indobufen-modified derivatives were rationally designed and synthesized, and their biological activities were evaluated through a stepwise in vitro screening workflow. Early inflammatory profiling in fibroblast-like synoviocytes (FLS) identified compound A1 as a lead candidate with pronounced suppression of pro-inflammatory mediators and matrix-degrading enzymes. In a RANKL-induced RAW264.7 osteoclastogenesis model, A1 inhibited osteoclast maturation at non-cytotoxic concentrations, markedly reducing the expression of osteoclast functional proteins (MMP9 and cathepsin K) and disrupting F-actin ring formation and multinucleation. Collectively, these findings demonstrate that indobufen scaffold modification can yield small molecules with dual anti-inflammatory and anti-osteoclastogenic activities and highlight A1 as a promising lead compound for the development of therapeutics targeting osteoporosis and inflammation-associated bone loss.
1 Introduction
As a systemic skeletal disease, osteoporosis is defined by decreased bone mass and disrupted bone microstructure, directly heightening bone fragility and fracture risk.1,2 Globally, its prevalence stands at around 18.3%, and incidence increases exponentially with age.3–5 Bone integrity relies on the dynamic balance of bone remodeling, a process driven by osteoblast-mediated bone formation and osteoclast-mediated bone resorption.6 Disruption of this balance—with bone resorption predominating—triggers osteoporosis. Multiple studies confirm that excessive activation and abnormal proliferation of osteoclasts are the primary drivers of pathological bone loss.7 Osteoclasts derive from hematopoietic monocyte/macrophage precursors, and their maturation is tightly regulated by microenvironmental signaling molecules.8 The binding of receptor activator of nuclear factor κB ligand (RANKL) to its receptor RANK on the surface of osteoclast precursors represents a key step in initiating osteoclast differentiation, fusion, and functional activation.9 This interaction activates downstream transcription factors (NF-κB, NFATc1), thereby upregulating the expression of critical bone resorption-related enzymes such as tartrate-resistant acid phosphatase (TRAP) and cathepsin K. Targeting and inhibiting RANKL signaling pathway-mediated osteoclastogenesis is therefore a core strategy for the prevention and treatment of osteoporosis.10–13Clinically used anti-osteoporotic drugs are primarily categorized into two classes: anti-resorptive agents and bone-forming agents. Anti-resorptive drugs, including bisphosphonates and denosumab, inhibit osteoclast-mediated bone resorption and have been proven effective in reducing fracture risk, but long-term administration carries safety concerns.14 For instance, long-term bisphosphonate use may lead to rare adverse effects such as atypical femoral fractures and osteonecrosis of the jaw, while certain regimens also increase infection risk.15 Bone-forming agents, such as parathyroid hormone analogs, exert their effects by stimulating osteoblast-mediated new bone formation. Despite demonstrated efficacy, their high cost, limited treatment duration, and potential side effects16 underscore the urgent need for a novel therapeutic approach with high efficacy, excellent safety, and affordability. Because of these drawbacks, there is a clear need for novel therapies that can safely reduce osteoclast-driven bone resorption without incurring serious adverse effects.
Drug repurposing and structural modification of existing drugs offer a fast-track route in the search for new osteoporosis treatments. Using known drugs as starting points is efficient since their pharmacological and safety profiles are already well documented.17 In this study, we explored the repurposing of indobufen, a drug originally developed as an antiplatelet cyclooxygenase inhibitor for cardiovascular conditions.
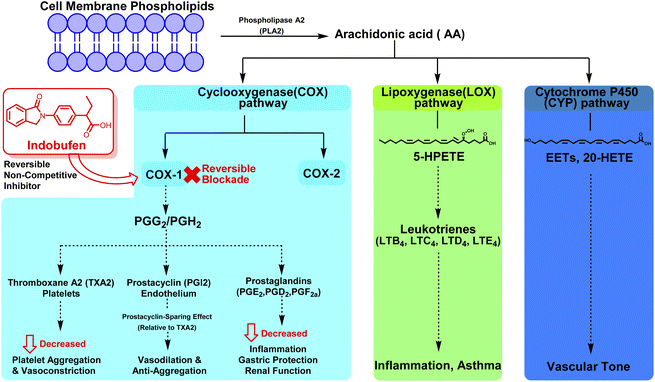
Indobufen, as a reversible cyclooxygenase inhibitor, exerts significant anticoagulant and anti-inflammatory activities by blocking the biosynthesis of thromboxane and prostaglandins.18 Scheme 1 details the arachidonic acid (AA) biochemical pathway and the selective, reversible inhibitory action of indobufen, a non-steroidal antiplatelet agent. Arachidonic acid is first released from cell membrane phospholipids via the enzyme phospholipase A2 (PLA2). Free AA then enters three major metabolic branches: the cyclooxygenase (COX) pathway (cyan), the lipoxygenase (LOX) pathway (green), and the cytochrome P450 (CYP) pathway (blue). Indobufen acts exclusively on the COX pathway: it functions as a reversible, non-competitive inhibitor of cyclooxygenase-1 (COX-1), blocking the conversion of AA to prostaglandin G2/H2 (PGG2/PGH2) in platelets, while sparing COX-2 activity.19 Epidemiological and pathological evidence indicates a close “immune–bone” interaction between chronic inflammation and osteoporosis. Particularly in pathological conditions such as rheumatoid arthritis, elevated levels of prostaglandin E2 and inflammatory cytokines (e.g., IL-1β, IL-6) in the synovial microenvironment upregulate the expression of Receptor Activator of Nuclear Factor-κB Ligand (RANKL) on fibroblast-like synoviocytes. This disruption of bone remodeling directly drives abnormal osteoclast differentiation and bone erosion.20 Based on the potential of indobufen to inhibit the COX/PGE2 axis, we propose a medicinal chemistry optimization strategy: to develop a class of dual-acting bone protective agents that simultaneously target the inflammatory microenvironment and osteoclastogenesis through structural modification of the indobufen scaffold.
To validate this structure–activity relationship hypothesis, this study designed and synthesized 10 novel indobufen derivatives and established a dual in vitro screening model encompassing the “inflammatory microenvironment” and the “bone resorption endpoint.” In a co-culture system of FLS cells and RANKL-induced RAW264.7 macrophages, we precisely evaluated the transcriptional inhibitory activity of candidate molecules on osteoclast-specific genes (Ctsk, Acp5, Mmp9) using quantitative PCR (qPCR). The screening results identified compound A1 as the most promising lead compound. Cell cytotoxicity experiments confirmed that A1 exhibited excellent safety within its effective pharmacological concentration range without significant cytotoxicity and dose-dependently inhibited osteoclast phenotypic formation. We further elucidated the molecular targets of A1 through follow-up mechanistic studies. Western blot analysis revealed that A1 potently blocks RANKL-induced phosphorylation cascades in the MAPK pathway—encompassing the p-ERK, p-JNK, and p-p38 subgroups—and simultaneously inhibits the NF-κB signaling pathway.21 Laser confocal microscopy experiments confirmed that A1 effectively suppresses the cytoplasmic-to-nuclear translocation of NFATc1 (nuclear factor of activated T-cells, cytoplasmic 1), a key transcription factor orchestrating osteoclast differentiation.22,23 Collectively, this study characterizes A1, a novel indobufen derivative, as exerting robust anti-osteoclastogenic activity in vitro by targeting the NF-κB/MAPK/NFATc1 signaling axis. These findings support targeted modification of the indobufen scaffold as a viable strategy for developing therapies against inflammatory bone loss diseases. Notably, A1, a highly promising candidate, warrants rigorous in vivo pharmacodynamic and pharmacokinetic evaluations.
2 Results and discussion
2.1 Synthesis of Indobufen derivatives A1–A10
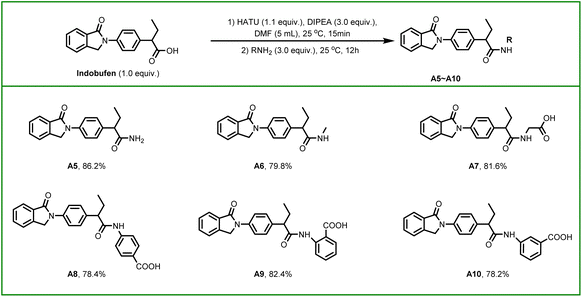
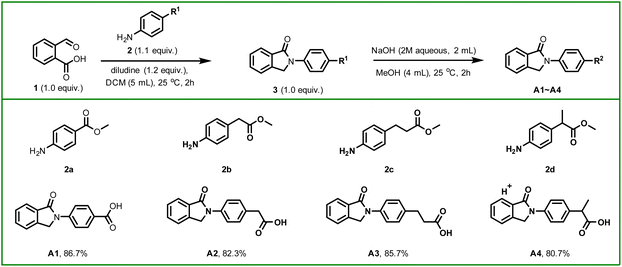
In the present study, a series of Indobufen derivatives was rationally designed and synthesized. Compounds A1–A4 were obtained via a cyclization reaction between stoichiometric amounts of o-carboxybenzaldehyde (1 equiv.) and substituted anilines (1.1 equiv.), followed by ester hydrolysis, as illustrated in Scheme 2. Additionally, indobufen derivatives A5–A10 were prepared through a condensation reaction between Indobufen and corresponding amine derivatives, as depicted in Scheme 3. The yield of synthesized derivatives ranged from 78% to 87%.2.2 Screening of indobufen derivatives
To preliminarily evaluate the anti-inflammatory potential of the synthesized indobufen-derived compounds, fibroblast-like synoviocytes (FLS) were treated with derivatives A1–A10 (20 µM) during inflammatory stimulation for 6 h, followed by RT-qPCR analysis of cytokines and matrix/remodeling-associated genes. As shown in Fig. 1, inflammatory stimulation markedly increased the mRNA expression of interleukin-1β (IL-1β, Fig. 1A and B) and interleukin-6 (IL-6, Fig. 1C and D), as well as intercellular adhesion molecule 1 (ICAM1, Fig. 1E and F) and matrix metalloproteinase 9 (MMP9, Fig. 1G and H), compared with the NC group, confirming successful induction of an inflammatory response in FLS cells. Among the tested compounds, several derivatives exhibited varying degrees of inhibitory effects on inflammatory gene expression. Notably, treatment with A1 resulted in a consistent and significant reduction in the mRNA levels of IL-1β and IL-6 compared with the stimulated group, whereas most other derivatives showed limited or inconsistent effects. In addition, A1 markedly suppressed the expression of MMP9, a key mediator associated with extracellular matrix degradation, while exerting minimal influence on ICAM1 expression, suggesting a selective anti-inflammatory profile. In contrast, other indobufen-modified derivatives displayed either marginal inhibitory activity or no statistically significant effects on the examined inflammatory markers. Based on its superior and consistent inhibitory effects on pro-inflammatory cytokines and matrix-degrading enzymes, A1 was identified as a lead candidate for further evaluation. Collectively, these results demonstrate that A1 exhibits a favorable anti-inflammatory profile in FLS cells during early inflammatory stimulation. Therefore, A1 was selected for subsequent mechanistic investigations in RAW264.7 cells to further explore its therapeutic potential in inflammation-associated osteoclastogenesis and bone resorption.2.3 Extended inflammatory profiling
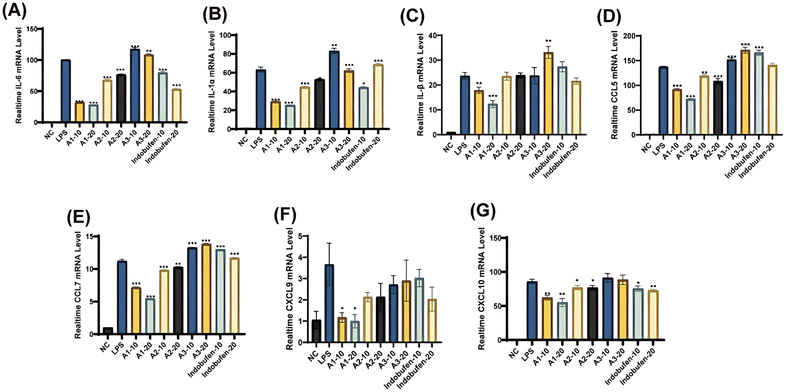
To further prioritize lead candidates after the initial screen (Fig. 1), we expanded transcriptional profiling to three representative derivatives (A1–A3) and a broader panel of cytokine and chemokine genes. FLS cells were exposed to A1–A3 at 10 and 20 µM, and gene expression changes were benchmarked against a negative control (NC) and an LPS-treated positive control processed in parallel. This design allowed us to distinguish compounds that are intrinsically pro-inflammatory from those with a comparatively low inflammatory liability. As shown in Fig. 2A–C, LPS markedly increased the transcription of IL-6, IL-1α, and IL-1β relative to NC, confirming robust inflammatory activation. At both concentrations tested, A1 resulted in minimal induction of the measured cytokines. In contrast, A3 triggered a strong inflammatory response, particularly at 20 µM, at which IL-6 and IL-1α transcript levels reached or surpassed those observed in LPS-treated cells. The effects of A2 were moderate relative to A1 and A3. This pattern suggests that cytokine responses differ in a structure-dependent manner among the three compounds.A similar pattern was observed for chemokine-related genes (CCL5, CCL7, CXCL9, and CXCL10; Fig. 2D–G). LPS significantly elevated CCL5, CCL7, and CXCL10 compared with NC. Among the derivatives, A3 again produced the strongest and most dose-dependent induction of CCL5 and CCL7, whereas A2 and A3 more prominently increased CXCL10. In contrast, A1 triggered comparatively modest chemokine responses. CXCL9 showed only limited variation across treatments, suggesting selective regulation of chemokine subtypes rather than a uniform amplification of all chemotactic signals. Indobufen (10 and 20 µM) was included as a reference compound and induced only moderate transcriptional changes across the cytokine and chemokine panel, remaining consistently below the responses observed with A3 at equivalent concentrations. Collectively, this extended profiling indicates that A1 has the lowest inflammatory liability among A1–A3 in FLS cells, supporting its selection for subsequent osteoclast-focused assays, therefore we focused on A1 to avoid confounding pro-inflammatory activation in downstream osteoclastogenesis assays.2.4 A1 exhibits favorable cellular safety and suppresses osteoclast functional protein expression
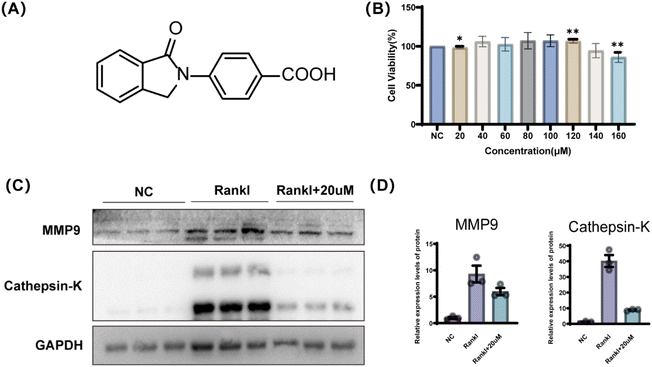
To investigate the biological activity of the indobufen-modified compound A1, its chemical structure is shown in Fig. 3A. The cytotoxicity of A1 was first evaluated using a CCK-8 assay to determine an appropriate concentration range for subsequent experiments. As shown in Fig. 3B, treatment with A1 at concentrations ranging from 20 to 120 µM did not result in significant cytotoxic effects, with cell viability remaining above 90% compared with the NC group. A1 did not compromise RAW264.7 cell viability at concentrations up to 120 µM, whereas exposure to 140 or 160 µM resulted in a pronounced viability loss, consistent with dose-dependent cytotoxicity. Non-cytotoxic concentrations of compound A1 were therefore selected for mechanistic analysis, given that osteoclast-mediated bone resorption is closely linked to functional protease expression. We employed western blotting to determine how A1 modulates the expression levels of RANKL-induced matrix metalloproteinase 9 (MMP9) and cathepsin K. As shown in Fig. 3C, RANKL stimulation markedly increased the levels of both proteins relative to the negative control (NC) group, whereas treatment with 20 µmol L−1 A1 substantially attenuated this induction. Densitometric analysis further confirmed reduced MMP9 and cathepsin K expression in A1-treated cells compared with RANKL-only controls (Fig. 3D). These findings indicate that A1 suppresses osteoclast functional protein expression under non-cytotoxic conditions, supporting a mechanism independent of non-specific cytotoxic effects.2.5 A1 disrupts F-actin ring formation during RANKL-induced osteoclast differentiation
To further investigate the effects of A1 on osteoclast maturation, F-actin organization was examined by immunofluorescence staining in RAW264.7 cells subjected to RANKL-induced osteoclastogenesis. As shown in Fig. 4A, cells in the NC group exhibited a relatively uniform cortical distribution of F-actin without the formation of distinct actin ring structures. In contrast, RANKL stimulation (RANKL group) induced the formation of large, well-defined F-actin rings accompanied by multinucleated cells, which are characteristic features of mature osteoclasts. Treatment with A1 resulted in a marked disruption of RANKL-induced F-actin ring formation in a concentration-dependent manner. At 10 µM, A1 partially impaired the integrity of F-actin rings, with fewer and less continuous ring structures observed compared with the RANKL group. Increasing the concentration of A1 to 20 µM further attenuated F-actin ring formation, accompanied by markedly reduced cell fusion and disorganized cytoskeletal structures. | ||
| Fig. 4 Inhibition of F-Actin Ring Assembly by Indobufen Derivative A1 During RANKL-Mediated Osteoclast Differentiation. | ||
3 Materials and methods
3.1 Chemical synthesis of indobufen derivatives
3.2 Cell culture
Fibroblast-like synoviocytes (FLS) cells were cultured in Dulbecco's Modified Eagle Medium (DMEM, high glucose) supplemented with 10% fetal bovine serum (FBS), 100 U mL−1 penicillin, and 100 µg mL−1 streptomycin. RAW264.7 cells were maintained in DMEM containing 10% FBS and antibiotics under the same conditions. All cells were incubated at 37°Cin a humidified atmosphere with 5% CO2. Culture media were refreshed every 2–3 days, and cells were passaged upon reaching approximately 80% confluence. RAW264.7 cells were selected due to their well-established capacity to differentiate into osteoclast-like cells upon receptor activator of nuclear factor-κB ligand (RANKL) stimulation,24 making them a widely accepted in vitro model for osteoclastogenesis studies.25 RAW264.7 mouse monocyte/macrophage cell (PubMed ID: 23430347) and Human primary spondylitis hip joint synovial fibroblasts (Cat No. HUM-iCell-s010RA-1) were purchased from iCell Bioscience Inc. (Shanghai, China). Recombinant mouse RANKL (Cat No. PRP1114) was purchased from Proteintech Group, Inc. (Wuhan, China). The following primary antibodies were used in this study: anti-cathepsin K (Cat No. 11239-1-AP, Proteintech), anti-MMP-9 (Cat No. 10375-2-AP, Proteintech), and anti-GAPDH (Cat No. 60004-1-Ig, Proteintech).3.3 Inflammatory stimulation and compound screening in FLS cells
FLS were seeded in plates and allowed to adhere overnight. Cells were pretreated with each indobufen-derived compound (A1–A10, 20 µM; final DMSO ≤ 0.1%) for 1 h and then exposed to lipopolysaccharide (LPS) to induce an inflammatory response. After 6 h stimulation, total RNA was harvested for RT-qPCR analysis of Il1b, Il6, Mmp9 and Icam1. For the extended cytokine/chemokine panel, FLS were treated with A1–A3 or indobufen at 10 and 20 µM, and LPS-stimulated in parallel. LPS alone served as the positive control and vehicle-treated cells served as the negative control.3.4 Quantitative PCR analysis
Total RNA was extracted from FLS or RAW264.7 cells using TRIzol (or an equivalent commercial reagent) according to the manufacturer's instructions. RNA concentration and purity were assessed by spectrophotometry, and equal amounts of RNA were reverse-transcribed to cDNA using a commercial reverse transcription kit. RT-qPCR was performed using SYBR Green chemistry on a real-time PCR system. For FLS, inflammatory and matrix-related genes (Il1b, Il6, Il1a, Mmp9, Icam1, Ccl5, Ccl7, Cxcl9 and Cxcl10) were quantified. For RAW264.7 cells, osteoclast-related genes (Acp5/TRAP, Nfatc1 and Ctsk) were quantified when indicated. GAPDH was used as the internal reference gene. Relative expression was calculated using the 2-ΔΔCt method, normalized to the vehicle control group. Each sample was run in technical triplicate, and all experiments were repeated in at least three independent biological replicates.3.5 Cell viability assay (CCK-8)
Cell viability was evaluated using the Cell Counting Kit-8 (CCK-8) assay. FLS or RAW264.7 cells were seeded in 96-well plates and allowed to adhere overnight, then treated with test compounds (vehicle control: DMSO ≤ 0.1%). For the A1 dose-range assessment, RAW264.7 cells were exposed to A1 (20–160 µM) for 24 h before CCK-8 measurement. Following treatment, 10 µL of CCK-8 reagent was added to each well and incubated at 37 °C for 1–2 h. Absorbance was measured at 450 nm using a microplate reader. Cell viability was calculated as a percentage relative to the untreated control group. Experiments were performed in triplicate and repeated independently at least three times. The CCK-8 assay was chosen due to its high sensitivity and reproducibility in assessing cellular metabolic activity.263.6 RANKL-induced osteoclastogenesis in RAW264.7 cells
RAW264.7 cells were used to establish an in vitro osteoclastogenesis model. Cells were seeded in culture plates (e.g., 6-well plates for protein analysis and coverslips in 24-well plates for imaging) and stimulated with recombinant mouse RANKL (typically 50 ng mL−1) to induce osteoclast differentiation. Test compounds were added at the start of induction and maintained throughout differentiation with medium changes every 48 h. For the mechanistic evaluation of A1, cells were treated with A1 at the indicated concentrations (10, 20 or 40 µM for F-actin ring analysis; 20 µM for western blotting unless otherwise stated). Differentiation was monitored by the appearance of multinucleated osteoclast-like cells and by the induction of osteoclast markers at the mRNA and/or protein level.3.7 Western blot analysis
Western blotting was performed to analyze osteoclast functional proteins and signaling molecules. Following treatment, RAW264.7 cells were lysed in RIPA buffer supplemented with protease and phosphatase inhibitors. Protein concentrations were determined using a BCA assay. Membranes were probed with primary antibodies against MMP9, cathepsin-K (CTSK) and GAPDH (loading control). When indicated, antibodies against NF-κB/MAPK pathway components and NFATc1 were also used. Equal amounts of protein were separated by SDS–PAGE and transferred onto polyvinylidene fluoride (PVDF) membranes. After blocking with 5% non-fat milk, membranes were incubated overnight at 4 °Cwith primary antibodies targeting osteoclast-related signaling proteins, including components of the NF-κB pathway, MAPK pathway, and NFATc1. Membranes were then incubated with appropriate horseradish peroxidase–conjugated secondary antibodies and visualized using an enhanced chemiluminescence detection system. Protein band intensities were quantified using ImageJ software and normalized to the corresponding loading control. Western blot analysis was conducted following established protocols for investigating osteoclast-related signaling mechanisms.273.8 Immunofluorescence and confocal microscopy
For cytoskeletal visualization, F-actin ring formation was assessed by phalloidin staining. RAW264.7 cells cultured on glass coverslips were induced with RANKL in the presence or absence of A1, fixed with 4% paraformaldehyde, permeabilized with 0.1% Triton X-100, and blocked with bovine serum albumin. Cells were stained with fluorescent phalloidin to label F-actin and counterstained with DAPI to visualize nuclei. Images were acquired using a confocal laser scanning microscope under identical settings for all groups. F-actin ring integrity and the number of multinucleated cells were quantified using ImageJ in a blinded manner from multiple random fields per condition. Where indicated, immunofluorescence staining for protein targets was performed using primary antibodies followed by fluorophore-conjugated secondary antibodies. Immunofluorescence provides spatial confirmation of molecular and morphological changes during osteoclast differentiation and drug intervention.283.9 Statistical analysis
All experimental assays were conducted with a minimum of three independent repetitions. Results are presented as the arithmetic mean the standard deviation. Statistical evaluations were carried out employing appropriate software packages. For comparisons involving multiple experimental groups, a one-way analysis of variance was performed, followed by suitable post hoc tests as indicated. A probability value of was considered to denote a statistically significant difference.4 Conclusions
In conclusion, this study designed and synthesized a series of novel indobufen derivatives based on drug repurposing and scaffold hopping medicinal chemistry strategies. Through the establishment of a hierarchical phenotypic screening model encompassing inflammatory microenvironment regulation and bone resorption endpoint blockade, we successfully identified the lead compound A1. Experimental data demonstrate that A1 exhibits unique dual pharmacological activities in in vitro models. On one hand, A1 significantly inhibited the secretion of pro-inflammatory factors, including IL-1 and IL-6, from fibroblast-like synoviocytes, thereby interrupting the pathological drive of inflammation on bone remodeling. On the other hand, A1 directly targeted the RANKL-induced osteoclast differentiation program. Within a clear therapeutic window, A1 not only downregulated the expression of NFATc1 and its downstream effector proteins but also markedly disrupted cytoskeletal F-actin ring assembly, thereby blocking bone resorption from both transcriptional and structural dimensions. These findings confirm the potential of the indobufen scaffold, following rational chemical modification, to effectively target the immuno–skeletal interface. As a small-molecule inhibitor with a novel dual-target mechanism, A1 extends beyond the simple cyclooxygenase–inhibitory activity of its parent compound and provides a valuable candidate and experimental foundation for clinical intervention in secondary osteoporosis associated with rheumatoid arthritis or postmenopausal inflammatory bone loss.Author contributions
D. Yuan, L. Chen and X. Huang performed experiments and synthesis of indobufen derivatives. Z. Meng and J. Zhang supervised the project and prepared the manuscript. All authors discussed the results and commented on the manuscript.Conflicts of interest
The authors declare no competing financial interest.Data availability
The data underlying this study are available in the published article and supporting information (SI). Supplementary information: materials, experimental details and NMR spectra for the synthesis of indobufen derivatives. See DOI: https://doi.org/10.1039/d6ra01985d.Acknowledgements
This work was supported by the Project of Natural Science Foundation of Jiangxi Province (20252BAC200103), (20242BAB21036) and (20232BAB216135).Notes and references
- S. Khosla and L. C. Hofbauer, Osteoporosis treatment: recent developments and ongoing challenges, Lancet Diabetes Endocrinol., 2017, 5, 898–907 CrossRef PubMed.
- B. Liang, G. Burley, S. Lin and Y. C. Shi, Osteoporosis pathogenesis and treatment: existing and emerging avenues, Cell. Mol. Biol. Lett., 2022, 27, 72 CrossRef PubMed.
- N. Salari, H. Ghasemi and L. Mohammadi, et al., The global prevalence of osteoporosis in the world: a comprehensive systematic review and meta-analysis, J. Orthop. Surg. Res., 2021, 16, 609 CrossRef PubMed.
- J. Wang, B. Shu, D. Z. Tang, C. G. Li, X. W. Xie, L. J. Jiang, X. B. Jiang, B. L. Chen, X. C. Lin, X. Wei, X. Y. Leng, Z. Y. Liao, B. L. Li, Y. Zhang, X. J. Cui, Q. Zhang, S. Lu, Q. Shi and Y. J. Wang, The prevalence of osteoporosis in China, a community based cohort study of osteoporosis, Front. Public Health, 2023, 11, 1084005 CrossRef PubMed.
- Y. Wang, Y. Tao, M. E. Hyman, J. Li and Y. Chen, Osteoporosis in china, Osteoporosis Int., 2009, 20, 1651–1662 CrossRef CAS PubMed.
- S. Meng, M. Tong, Y. Yu, Y. Cao, B. Tang, X. Shi and K. Liu, The prevalence of osteoporotic fractures in the elderly in China: a systematic review and meta-analysis, J. Orthop. Surg. Res., 2023, 18, 536 CrossRef PubMed.
- Y. Wu, H. Ai, Y. Xi, P. Yin, Y. Qu, J. Xu, C. Dou and F. Luo, Reduced osteoclast-derived apoptotic bodies in bone marrow characterizes the pathological progression of osteoporosis, Cell Death Discovery, 2023, 9, 135 CrossRef PubMed.
- D. S. Amarasekara, H. Yun, S. Kim, N. Lee, H. Kim and J. Rho, Regulation of Osteoclast Differentiation by Cytokine Networks, Immune Netw, 2018, 18, e8 CrossRef PubMed.
- J. H. Park, N. K. Lee and S. Y. Lee, Current Understanding of RANK Signaling in Osteoclast Differentiation and Maturation, Mol. Cells, 2017, 40, 706–713 CrossRef CAS PubMed.
- T. Matsumoto and I. Endo, RANKL as a target for the treatment of osteoporosis, J. Bone Miner. Metab., 2021, 39, 91–105 CrossRef CAS PubMed.
- S. Khosla and D. G. Monroe, Regulation of Bone Metabolism by Sex Steroids, Cold Spring Harb. Perspect. Med., 2018, 8, a031211 CrossRef.
- W. J. Boyle, W. S. Simonet and D. L. Lacey, Osteoclast differentiation and activation, Nature, 2003, 423, 337–342 CrossRef CAS PubMed.
- H. Takayanagi, Osteoimmunology and the effects of the immune system on bone, Nat. Rev. Rheumatol., 2009, 5, 667–676 CrossRef CAS PubMed.
- E. Shane, D. Burr, P. R. Ebeling, B. Abrahamsen, R. A. Adler, T. D. Brown, A. M. Cheung, F. Cosman, J. R. Curtis, R. Dell, D. Dempster, T. A. Einhorn, H. K. Genant, P. Geusens, K. Klaushofer, K. Koval, J. M. Lane, F. McKiernan, R. McKinney, A. Ng, J. Nieves, R. O'Keefe, S. Papapoulos, H. T. Sen, M. C. H. van der Meulen, R. S. Weinstein and M. Whyte, American Society for Bone and Mineral Research. Atypical subtrochanteric and diaphyseal femoral fractures: report of a task force of the American Society for Bone and Mineral Research, J. Bone Miner. Res., 2010, 25, 2267–2294 CrossRef PubMed.
- I. R. Reid, Short-term and long-term effects of osteoporosis therapies, Nat. Rev. Endocrinol., 2015, 11, 418–428 CrossRef CAS.
- R. M. Neer, C. D. Arnaud, J. R. Zanchetta, R. Prince, G. A. Gaich, J. Y. Reginster, A. B. Hodsman, E. F. Eriksen, S. Ish-Shalom, H. K. Genant, O. Wang and B. H. Mitlak, Effect of parathyroid hormone (1-34) on fractures and bone mineral density in postmenopausal women with osteoporosis, N. Engl. J. Med., 2001, 344, 1434–1441 CrossRef CAS PubMed.
- S. Pushpakom, F. Iorio, P. A. Eyers, K. J. Escott, S. Hopper, A. Wells, A. Doig, T. Guilliams, J. Latimer, C. McNamee, A. Norris, P. Sanseau, D. Cavalla and M. Pirmohamed, Drug repurposing: progress, challenges and recommendations, Nat. Rev. Drug Discovery, 2019, 18, 41–58 CrossRef CAS PubMed.
- N. Bhana and K. J. McClellan, Indobufen: an updated review of its use in the management of atherothrombosis, Drugs Aging, 2001, 18, 369–388 CrossRef CAS.
- A. R. Brash, Arachidonic acid as a bioactive molecule, J. Clin. Invest., 2001, 107, 1339–1345 CrossRef CAS.
- Q. Lu, J. Xu, H. Jiang, Q. Wei, R. Huang and G. Huang, The bone-protective mechanisms of active components from TCM drugs in rheumatoid arthritis treatment, Front. Pharmacol, 2022, 13, 1000865 CrossRef CAS PubMed.
- T. Yang, W. W. Chen, K. Gan, C. F. Wang, X. X. Xie, Y. G. Su, H. Y. Lian, J. Xu, J. M. Zhao and Q. Liu, Myrislignan targets extracellular signal-regulated kinase (ERK) and modulates mitochondrial function to dampen osteoclastogenesis and ovariectomy-induced osteoporosis, J. Transl. Med., 2023, 21, 839 CrossRef CAS PubMed.
- H. Huynh and Y. Wan, mTORC1 impedes osteoclast differentiation via calcineurin and NFATc1, Commun. Biol., 2018, 1, 29 CrossRef PubMed.
- T. S. Suda, K. Kobayashi, E. Jimi, N. Udagawa and N. Takahashi, The molecular basis of osteoclast differentiation and activation, Novartis Found. Symp., 2001, 232, 235–247 CAS.
- P. Collin-Osdoby and P. Osdoby, RANKL-mediated osteoclast formation from murine RAW 264.7 cells, Methods Mol. Biol., 2012, 816, 187–202 CrossRef CAS PubMed.
- W. J. Boyle, W. S. Simonet and D. L. Lacey, Osteoclast differentiation and activation, Nature, 2003, 423, 337–342 CrossRef CAS PubMed.
- M. Ishiyama, Y. Miyazono, K. Sasamoto, Y. Ohkura and K. Ueno, A highly water-soluble disulfonated tetrazolium salt as a chromogenic indicator for NADH as well as cell viability, Talanta, 1997, 44, 1299–1305 CrossRef CAS PubMed.
- Z. H. Lee and H. H. Kim, Signal transduction by receptor activator of nuclear factor kappa B in osteoclasts, Biochem. Biophys. Res. Commun., 2003, 305, 211–214 CrossRef CAS PubMed.
- S. Epsley, S. Tadros, A. Farid, D. Kargilis, S. Mehta and C. S. Rajapakse, The Effect of Inflammation on Bone, Front. Physiol., 2021, 11, 511799 CrossRef PubMed.
Footnote |
| † These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |