Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Functionalisation of homopropargyl boronic esters via hydrozirconation followed by Pd-catalysed cross coupling reaction
Patrick Schäfer and
Uli Kazmaier
and
Uli Kazmaier *
*
Organic Chemistry, Saarland University, P. O. Box 151150, 66041 Saarbrücken, Germany. E-mail: u.kazmaier@mx.uni-saarland.de
First published on 24th March 2026
Abstract
Hydrozirconation of homopropargyl boronic esters accessible via Matteson homologation allows their selective functionalisation while retaining the boronic ester functionality. In this study, reactions of vinyl zirconium reagents derived from boronic esters in Negishi coupling, in particular with benzyl halides and aryl iodides, are presented. Different synthesis routes for the stereoselective assembly of (E)- and (Z)-alkenes are discussed.
Introduction
Polyketides form a wide-spread and diverse class of natural products with various biological activities.1 Therefore, the asymmetrical overall synthesis of the members of this class is of great importance, and it is often anything but trivial.2 In most cases, the polyketide chain is synthesized via aldol reactions3 or allylations/crotylations followed by ozonolysis.4 While these methods are suitable for multiple hydroxylated polyketide chains, specially substituted alkyl chains can often be better obtained via the Matteson homologation approach.5 This protocol was first described in the 1980s by Donald Matteson et al. in its asymmetrical version.6 Using alkylboronic esters of chiral diols, a carbon chain can be highly stereoselectively extended by reacting with a halogenated lithium carbenoid.7 The resulting α-haloboronic ester can then be subjected to a variety of C-, O- and N-nucleophiles such as Grignard6b,8 or organolithium reagents,9 alkoxides10 and azides (Scheme 1A).11 This allows each carbon atom of the chain to be substituted individually without getting into matched/mismatched situations, as the configuration of newly formed stereocentres is almost exclusively controlled by the chiral boronic ester. Functional groups that are not compatible with the homologation conditions, e.g. carbonyl groups, can be inserted via placeholders, which can later on be converted into the desired functionality (Scheme 1B).12 Therefore, Matteson homologation is increasingly used in natural product synthesis13 and for the synthesis of pharmaceuticals,14 whereby it also attracted the attention of our group.15While saturated alkyl chains can be easily obtained using the Matteson protocol, the stereoselective introduction of double bonds or even conjugated double bonds, such as those found in the natural products like (5Z)-7-oxozeaenol,16 papulacandin D17 and (−)-dictyostatin18 (Fig. 1) is not possible.
To solve this problem, cross-coupling reactions offer a good alternative to extend the carbon chain by introducing a double bond. In this context, zirconium is the metal of choice for this approach because it provides readily reactive vinyl nucleophiles via hydrozirconation of alkynes, has functional group tolerance and is non-toxic (Scheme 1C).19 In addition, Srebnik et al. were able to demonstrate the compatibility of these two different metals in the same molecule by hydrozirconation of vinyl boranes20 and vinyl boronic esters (Scheme 1D).21 The vinyl zirconium reagents can be functionalised either by reactions with electrophiles such as protons, halides and acid chlorides22 or by Ni- or Pd-catalysed Negishi couplings (Scheme 1C).23 Negishi couplings allow for the introduction of various aryl,24 vinyl,25 allyl26 and even alkyl groups.27 Therefore, these reactions have become common tools for the synthesis of natural products.28
Herein, we report an application of this approach using selective functionalisations of homopropargyl boronic esters via hydrozirconation and subsequent Negishi couplings.
Results and discussion
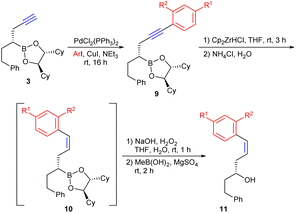
Initially, the homopropargyl boronic ester 3 required for hydrozirconation was synthesised as a model compound starting from boronic ester 1 (Scheme 2). For this purpose, 1 was first reacted in a Matteson homologation with LiCHCl2 and the α-chloroboronic ester formed was immediately subjected to a nucleophilic substitution with the TMS-protected propargylzinc reagent.15f The primary formed boronic ester 2 was then deprotected with K2CO3 in methanol/ether, which proved to be the method of choice (see SI).29 The addition of diethyl ether was necessary because of the poor solubility of boronic ester 2 in methanol. It should be mentioned that commonly used fluoride-containing cleavage reagents such as TBAF or KF were not compatible with the boronic ester functionality because the fluoride binds to boron rather than silicon, resulting in at least partial protodeboration.Starting from 3, Negishi couplings with benzyl halides and allyl bromide were first investigated (Scheme 3).30 The hydrozirconation of 3 went smoothly, but no complete turnover could be observed in subsequent cross couplings. Thus, after workup, the desired coupling product 5 was usually contaminated with allyl-substituted boronic ester 4.12 It originates from the vinyl zirconium intermediate formed via hydrozirconation of 3, which did not react in the following cross coupling, and was thus hydrolysed during aqueous workup. Changes in the reaction conditions as minimisation of the excess of Schwartz reagent to 1.0 eq. or increasing the amount of benzyl halide as well as extended reaction times did not improve the turnover of the cross-coupling reaction. Since 4 could not be separated from the desired substitution product 5 due to almost equal chromatographic retention, the mixture of boronic esters 4 and 5 was directly oxidised to the corresponding alcohols, which could be easily separated by column chromatography. Methylboronic acid was added after the workup to remove the chiral auxiliary.15a The corresponding chiral methylboronic ester formed can easily be separated and reused in Matteson reactions. The yield given in Scheme 3 for the desired alcohol 6 corresponds to the isolated and purified alcohol after 4 steps.
 | ||
| Scheme 3 Negishi couplings of the vinyl zirconium reagent prepared from 3 with benzyl and allyl halides. | ||
The ratio 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 was determined by 1H NMR spectroscopy of the crude product mixture. In addition to the unsubstituted benzyl residue (6a), benzyl residues containing both electron-withdrawing (6b and 6c) and electron-donating (6g, 6e, 6f and 5h) substituents were also tested. The electronic properties of the respective substituents showed no significant influence on the results of the reactions, nor did different positions of the substituents on the aromatic ring. If allyl bromide was used instead of a benzyl halide, 1,4-dienes (6g) became available. In the case of the p-methoxy-substituted derivative 5h, the boronic ester could be obtained in high yield by chromatography, since 4 was formed only in trace amounts in this case.
5 was determined by 1H NMR spectroscopy of the crude product mixture. In addition to the unsubstituted benzyl residue (6a), benzyl residues containing both electron-withdrawing (6b and 6c) and electron-donating (6g, 6e, 6f and 5h) substituents were also tested. The electronic properties of the respective substituents showed no significant influence on the results of the reactions, nor did different positions of the substituents on the aromatic ring. If allyl bromide was used instead of a benzyl halide, 1,4-dienes (6g) became available. In the case of the p-methoxy-substituted derivative 5h, the boronic ester could be obtained in high yield by chromatography, since 4 was formed only in trace amounts in this case.
As illustrated with 6c, p-bromobenzyl bromide reacts regioselectively at the benzylic position, which allows further modifications of the aryl bromide, e.g. by Sonogashira coupling (Scheme 4).
Although benzyl halides are obviously more reactive than aryl halides, these can still be used in Negishi couplings (Scheme 5). However, since the vinyl zirconium reagent formed in situ was not reactive enough to enable coupling with vinyl and aryl halides, transmetallation to zinc was necessary in this case.23,24b The addition of zinc chloride should take place last, since transmetallation in the absence of a Pd catalyst leads to a partial decomposition of the respective boronic ester.
Under the optimised conditions, Negishi couplings were performed with different aryl iodides, which carried electron-donating and -withdrawing groups in ortho and para position, respectively, as well as an electron-rich (8g) and an electron-poor vinyl iodide (8h). As already observed in the synthesis of boronic esters 5, allylboronic ester 4 was also obtained as a by-product in most cases. The products could be easily separated after oxidation by flash chromatography, whereby also here the yields given refer to the complete reaction sequence (from 3). Depending on the substituents on the aromatic ring, slightly different oxidation protocols were used. Interestingly, in the case of the nitrile-substituted derivative 8f, complete turnover and no formation of 4 was observed.
Due to the highly regioselective syn-hydrozirconation, a large number of (E)-configured unsaturated alcohols and boronic esters could be obtained without any problems. However, direct access to the corresponding (Z)-alkenes is not possible via this protocol. However, this can be achieved by reversing the order of the synthetic steps. If the substituent is introduced at the alkyne level via Sonogashira coupling, the subsequent hydrozirconation of 9 represents an alternative to the Lindlar hydrogenation of internal alkynes (Table 1). For easier purification, the boronic esters 10 were again oxidised to the corresponding alcohols 11.
| Entry | R1 | R2 | 9 | 9 [yield] | 10 [yield] | 11 [yield] | Overall yield |
|---|---|---|---|---|---|---|---|
| a Base on recovered starting material.b No conversion.c Not investigated. | |||||||
| 1 | H | H | 9a | 99% | 88% | 66% | 57% 11a |
| 2 | Me | H | 9b | 100% | 84% | 76% | 64% 11b |
| 3 | OMe | H | 9c | 100% | 71% | 90% | 64% 11c |
| 4 | F | H | 9d | 97% | 87% | 76% | 64% 11d |
| 5 | H | OMe | 9e | 78% | 39% (62%)a | n.i.c | 30% 10e (48%)a |
| 6 | NO2 | H | 9f | 92% | —b | — | 92% 9f |
As shown in Table 1, a number of different aryl substituents, both with electron-donating and -withdrawing groups, were introduced via the Sonogashira coupling, but no significant influence on the yields was observed. The introduction of an ortho-substituent was possible without problems, whereby the steric interaction between the o-MeO group and the zirconocene group during hydrozirconation only leads to an incomplete transformation. Here, unreacted alkyne 9e could be recovered, since 9e and 10e could be separated by flash chromatography.
Only in case of 9f no hydrozirconation was observed, which might be caused by the strong electron-withdrawing effect of the nitro group, which obviously makes the alkyne too electron-poor for coordinating to the Schwartz reagent. Interestingly, however, the nitro group was also not reduced.
Therefore, by simply changing the order of hydrozirconation and cross coupling both, (E)- and (Z)-alkenes become available.
Concerning the yields obtained, the presented method will certainly not be able to compete with Lindlar hydrogenation, but it would be an interesting approach if the hydrozirconation is regioselective and the formed disubstituted vinyl zirconium intermediate can be converted into triple-substituted alkenes via cross couplings. Therefore, the hydrozirconation was investigated in more detail using the aryl-substituted alkyne 9b. The formed vinyl zirconium compound was converted with N-iodosuccinimide into the corresponding vinyl iodide.31 According to the mechanism of hydrozirconation of internal alkynes, as proposed by Schwartz et al.,32 Cp2ZrHCl was used in excess to achieve better regioselectivity. But nevertheless, in case of 9b, the regioisomeric iodides 12b and 13b were obtained only as a 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 mixture, but in a high yield (Scheme 6). Therefore, optimisations of regioselectivity could also open up new synthetic possibilities here.
2 mixture, but in a high yield (Scheme 6). Therefore, optimisations of regioselectivity could also open up new synthetic possibilities here.
Conclusions
In summary, we have shown that the hydrozirconation of homopropargyl boronic esters enables the selective functionalisation of the carbon chain established by Matteson homologation. The respective organozirconium reagents can react with electrophiles such as halides or participate in Negishi couplings with either benzyl halides or aryl and vinyl iodides. Further applications of this method in total syntheses are currently under investigation.Conflicts of interest
There are no conflicts to declare.Data availability
Supplementary information (SI): copies of 1H and 13C NMR spectra and experimental details. See DOI: https://doi.org/10.1039/d6ra02170k.Acknowledgements
Financial support from Saarland University and the DFG (grants: Ka 880/13-1; Bruker Neo 500 – 447298507; INST 256/472-1 FUGG) is gratefully acknowledged. The authors also thank Dr Stefan Boettcher (Pharmaceutical and Medicinal Chemistry, Saarland University) for ESI-HRMS measurements.Notes and references
- (a) J. Staunton and K. J. Weissman, Polyketide biosynthesis: a millennium review, Nat. Prod. Rep., 2001, 18, 380–416 RSC; (b) F. von Nussbaum, M. Brands, B. Hinzen, S. Weigand and D. Häbich, Antibacterial natural products in medicinal chemistry – exodus or revival?, Angew. Chem., Int. Ed., 2006, 45, 5072–5129 CrossRef CAS PubMed; (c) M. S. Butler, A. A. B. Robertson and M. A. Cooper, Natural product and natural product derived drugs in clinical trials, Nat. Prod. Rep., 2014, 31, 1612–1661 RSC; (d) Z. Liu, H. Liu and W. Zhang, Natural polypropionates in 1999–2020: An overview of chemical and biological diversity, Mar. Drugs, 2020, 18, 569 CrossRef CAS PubMed.
- I. Paterson and N. Y. S. Lam, Challenges and discoveries in the total synthesis of complex polyketide natural products, J. Antibiot., 2018, 71, 215–233 CrossRef CAS PubMed.
- (a) H. Steinmetz, K. Gerth, R. Jansen, R. Dehn, S. Reinecke, A. Kirschning and R. Müller, Elansolid A, a unique macrolide antibiotic from Chitinophaga sancti isolated as two stable atropisomers, Angew. Chem., Int. Ed., 2011, 50, 532–536 CrossRef CAS PubMed; (b) S. Scheeff and D. Menche, Total synthesis of archazolid F, Org. Lett., 2019, 21, 271–274 CrossRef CAS PubMed; (c) N. Y. S. Lam and I. Paterson, Stereocontrolled synthesis as an enabling tool for the configurational assignment of marine polyketide natural products, Eur. J. Org Chem., 2020, 2310–2320 CrossRef CAS; (d) N. Y. S. Lam, T. P. Stockdale, M. J. Anketell and I. Paterson, Conquering peaks and illuminating depths: developing stereocontrolled organic reactions to unlock nature's macrolide treasure trove, Chem. Commun., 2021, 57, 3171–3189 RSC.
- (a) W. R. Roush, A. D. Palkowitz and K. Ando, Acyclic diastereoselective synthesis using tartrate ester-modified crotylboronates. Double asymmetric reactions with α-methyl chiral aldehydes and synthesis of the C(19)-C(29) segment of rifamycin S, J. Am. Chem. Soc., 1990, 112, 6348–6359 CrossRef CAS; (b) A. Arefolov and J. S. Panek, Crotylsilane reagents in the synthesis of complex polyketide natural products: total synthesis of (+)-discodermolide, J. Am. Chem. Soc., 2005, 127, 5596–5603 CrossRef CAS PubMed; (c) A. M. R. Dechert-Schmitt, D. C. Schmitt, X. Gao, T. Itoh and M. J. Krische, Polyketide construction via hydrohydroxyalkylation and related alcohol C–H functionalizations: reinventing the chemistry of carbonyl addition, Nat. Prod. Rep., 2014, 31, 504–513 RSC; (d) K. Spielmann, G. Niel, R. M. De Figueiredo and J. M. Campagne, Catalytic nucleophilic ‘umpoled’ π-allyl reagents, Chem. Soc. Rev., 2018, 47, 1159–1173 RSC.
- (a) D. S. Matteson, α-Halo boronic esters: intermediates for stereodirected synthesis, Chem. Rev., 1989, 89, 1535–1551 CrossRef CAS; (b) D. S. Matteson, Boronic esters in asymmetric synthesis, J. Org. Chem., 2013, 78, 10009–10023 CrossRef CAS PubMed; (c) D. S. Matteson, B. S. L. Collins, V. K. Aggarwal and E. Ciganek, The Matteson reaction, Org. React., 2021, 105, 427–860 Search PubMed.
- (a) D. S. Matteson and D. Majumdar, α-Chloro boronic esters from homologation of boronic esters, J. Am. Chem. Soc., 1980, 102, 7588–7590 CrossRef CAS; (b) D. S. Matteson and R. Ray, Directed chiral synthesis with pinanediol boronic esters, J. Am. Chem. Soc., 1980, 102, 7590–7591 CrossRef CAS.
- (a) D. S. Matteson, K. M. Sadhu and M. L. Peterson, 99% Chirally selective synthesis via pinanediol boronic esters: insect pheromones, diols, and an amino alcohol, J. Am. Chem. Soc., 1986, 108, 810–819 CrossRef CAS; (b) E. J. Corey, D. Barnes-Seeman and T. W. Lee, The mechanistic basis for diastereoselectivity in the Matteson rearrangement, Tetrahedron: Asymmetry, 1997, 8, 3711–3713 CrossRef CAS.
- (a) D. S. Matteson and J. J. Yang, Stereoselective chain extension of (R,R)- or (S,S)-1,2-dicyclohexylethane-1,2-diol trityloxymethylboronate to compounds having three stereogenic centers, Tetrahedron: Asymmetry, 1997, 8, 3855–3861 CrossRef CAS; (b) D. Leonori and V. K. Aggarwal, Lithiation–borylation methodology and its application in synthesis, Acc. Chem. Res., 2014, 47, 3174–3183 CrossRef CAS PubMed.
- W. C. Hiscox and D. S. Matteson, Asymmetric synthesis of the Japanese beetle pheromone via boronic esters, J. Organomet. Chem., 2000, 614–615, 314–317 CrossRef CAS.
- D. S. Matteson and M. Peterson, Synthesis of L-(+)-ribose via (S)-pinanediol (αS)-α-bromoboronic esters, J. Org. Chem., 1987, 52, 5116–5121 CrossRef CAS.
- (a) D. S. Matteson and E. C. Beedle, A directed chiral synthesis of amino acids from boronic esters, Tetrahedron Lett., 1987, 28, 4499–4502 CrossRef CAS; (b) A. Horn, E. Papadopoulos, T. Kinsinger, J. Greve, E. Bickel, S. Pachoula and U. Kazmaier, Stereoselective synthesis of α-azido esters and α-amino acid derivatives via Matteson homologation of boronic esters, Z. Anorg. Allg. Chem., 2024, 650, e202400113 CrossRef CAS.
- O. Andler and U. Kazmaier, Allylzinc reagents: versatile nucleophiles in Matteson homologations, Org. Lett., 2021, 23, 8439–8444 CrossRef CAS PubMed.
- (a) D. S. Matteson, H. W. Man and O. C. Ho, Asymmetric synthesis of stegobinone via boronic ester chemistry, J. Am. Chem. Soc., 1996, 118, 4560–4566 CrossRef CAS; (b) S. M. Bauer and R. W. Armstrong, Total synthesis of motuporin (nodularin-V), J. Am. Chem. Soc., 1999, 121, 6355–6366 CrossRef CAS; (c) J. Janetzko and R. A. Batey, Organoboron-based allylation approach to the total synthesis of the medium-ring dilactone (+)-antimycin A1b, J. Org. Chem., 2014, 79, 7415–7424 CrossRef CAS PubMed; (d) A. Noble, S. Roesner and V. K. Aggarwal, Short enantioselective total synthesis of tatanan A and 3-epi-tatanan A using assembly-line synthesis, Angew. Chem., Int. Ed., 2016, 55, 15920–15924 CrossRef CAS PubMed; (e) U. Kazmaier, Syntheses of marine natural products via Matteson homologations and related processes, Mar. Drugs, 2025, 23, 20 CrossRef CAS PubMed.
- (a) A. S. Gorovoy, O. Gozhina, J. S. Svendsen, G. V. Tetz, A. Domorad, V. V. Tetz and T. Lejon, Syntheses and anti-tubercular activity of β-substituted and α,β-disubstituted peptidyl β-aminoboronates and boronic acids, J. Pept. Sci., 2013, 19, 613–618 CrossRef CAS PubMed; (b) X. Li and D. G. Hall, Synthesis and applications of β-aminoalkylboronic acid derivatives, Adv. Synth. Catal., 2021, 363, 2209–2223 CrossRef CAS.
- (a) J. Gorges and U. Kazmaier, Matteson homologation-based total synthesis of lagunamide A, Org. Lett., 2018, 20, 2033–2036 CrossRef CAS PubMed; (b) M. Tost, O. Andler and U. Kazmaier, A Matteson homologation-based synthesis of doliculide and derivatives, Eur. J. Org Chem., 2021, 6459–6471 CrossRef CAS; (c) O. Andler and U. Kazmaier, U. Total synthesis of apratoxin A and B using Matteson's homologation approach, Org. Biomol. Chem., 2021, 19, 4866–4870 RSC; (d) O. Andler and U. Kazmaier, Stereoselective synthesis of a protected side chain of meliponamycin A, Org. Lett., 2022, 24, 2541–2545 CrossRef CAS PubMed; (e) M. Kempf, O. Andler and U. Kazmaier, Total Synthesis of salviachinensine A using a Matteson homologation approach, Helv. Chim. Acta, 2023, 106, 1–9 CrossRef; (f) M. Tost and U. Kazmaier, Synthesis and late-stage modification of (−)-doliculide derivatives using Matteson's homologation approach, Mar. Drugs, 2024, 22, 165 CrossRef CAS PubMed.
- S. Ayers, T. N. Graf, A. F. Adcock, D. J. Kroll, S. Matthew, E. J. Carcache De Blanco, Q. Shen, S. M. Swanson, M. C. Wani, C. J. Pearce and N. H. Oberlies, Resorcylic acid lactones with cytotoxic and NF-κB inhibitory activities and their structure-activity relationships, J. Nat. Prod., 2011, 74, 1126–1131 CrossRef CAS PubMed.
- (a) P. Traxler, J. Gruner and J. A. L. Auden, Papulacandins, a new family of antibiotics with antifungal activity – I. fermentation, isolation, chemical and biological characterization of papulacandins A, B, C, D and E, J. Antibiot., 1977, 30, 289–296 CrossRef CAS PubMed; (b) P. Traxler, H. Fritz, H. Fuhrer and W. J. Richter, Papulacandins, a new family of antibiotics with antifungal activity – structures of papulacandins A, B, C and D, J. Antibiot., 1980, 33, 967–978 CrossRef CAS PubMed.
- (a) G. R. Pettit, Z. A. Cichacz, F. Gao, M. R. Boyd and J. M. Schmidt, Isolation and structure of the cancer cell growth inhibitor dictyostatin 1, J. Chem. Soc. Chem. Commun., 1994, 1111–1112 RSC; (b) I. Paterson, R. Britton, O. Delgado, A. Meyer and K. G. Poullennec, Total synthesis and configurational assignment of (−)-dictyostatin, a microtubule-stabilizing macrolide of marine sponge origin, Angew. Chem., Int. Ed., 2004, 43, 4629–4633 CrossRef CAS PubMed.
- (a) P. Wipf, W. Xu, H. Takahashi, H. Jahn and P. D. G. Coish, Synthetic applications of organozirconocenes, Pure Appl. Chem., 1997, 69, 639–644 Search PubMed; (b) P. Wipf and C. Kendall, Novel applications of alkenyl zirconocenes, Chem. Eur. J., 2002, 8, 1778–1784 CrossRef CAS PubMed; (c) P. Wipf, Hydrozirconation and Its applications, Top. Organomet. Chem., 2004, 8, 1–25 CrossRef; (d) Z. Song and T. Takahashi, Hydrozirconation of Alkenes and Alkynes, Elsevier Ltd, 2014 Search PubMed.
- B. Zheng and M. Srebnik, Preparation and selective cleavage reactions of boron-zirconium 1,1-bimetalloalkanes, Tetrahedron Lett., 1993, 34, 4133–4136 CrossRef CAS.
- (a) B. Zheng and M. Srebnik, Synthesis of a new class of bidentate Lewis acids based on boronic esters and zirconocene, J. Organomet. Chem., 1994, 474, 49–53 CrossRef CAS; (b) B. Zheng and M. Srebnik, Reactions of N-halosuccinimides with 1,1-bimetallics based on zirconocene and boronic esters: A new synthesis of α-haloboronic esters, Tetrahedron Lett., 1994, 35, 1145–1148 CrossRef CAS; (c) E. Skrzypczak-Jankun, B. V. Cheesman, B. Zheng, R. M. Lemert, S. Asthanab and M. Srebnik, First example of a stable 1,1-bidentate Lewis acid based on boron and zirconium: synthesis, reactivity, X-ray analysis and NMR studies, J. Chem. Soc. Chem. Commun., 1994, 127–128 RSC; (d) L. Deloux, E. Skrzypczak-Jankun, B. V Cheesman, M. Srebnik and M. Sabat, First example of stable 1,1-bimetalloalkenes of boron and zirconium: Synthesis, reactivity, X-ray analysis, and NMR studies, J. Am. Chem. Soc., 1994, 116, 10302–10303 CrossRef CAS.
- (a) D. W. Hart and J. Schwartz, Hydrozirconation. Organic synthesis via organozirconium intermediates. Synthesis and rearrangement of alkylzirconium(IV) complexes and their reaction with electrophiles, J. Am. Chem. Soc., 1974, 96, 8115–8116 CrossRef CAS; (b) S. Karlsson, A. Hallberg and S. Gronowitz, Hydrozirconation of (E)-3-methoxy-1-phenyl-1-propene and (E)-3-phenyl-2-propenol, J. Organomet. Chem., 1991, 403, 133–144 CrossRef CAS.
- E. I. Negishi, N. Okukado, A. O. King, D. E. Van Horn and B. I. Spiegel, Selective carbon-carbon bond formation via transition metal catalysts. 9. Double metal catalysis in the cross-coupling reaction and its application to the stereo- and regioselective synthesis of trisubstituted olefins, J. Am. Chem. Soc., 1978, 100, 2254–2256 CrossRef CAS.
- (a) E. I. Negishi and D. E. Van Horn, Selective carbon-carbon bond formation via transition metal catalysis. 4. A novel approach to cross-coupling exemplified by the nickel-catalyzed reaction of alkenylzirconium derivatives with aryl halides, J. Am. Chem. Soc., 1977, 99, 3168–3170 CrossRef CAS; (b) E. I. Negishi, T. Takahashi, S. Baba, D. E. Van Horn and N. Okukado, Nickel- or palladium-catalyzed cross coupling. 31. Palladium- or nickel-catalyzed reactions of alkenylmetals with unsaturated organic halides as a selective route to arylated alkenes and conjugated dienes: scope, limitations, and mechanism, J. Am. Chem. Soc., 1987, 109, 2393–2401 CrossRef CAS.
- (a) N. Okukado, D. E. Van Horn, W. L. Klima and E. I. Negishi, A highly stereo-, regio-, and chemoselective synthesis of conjugated dienes by the palladium-catalyzed reaction of (E)-1-alkenylzirconium derivatives with alkenyl halides, Tetrahedron Lett., 1978, 19, 1027–1030 CrossRef; (b) P. Vincent, J.-P. Beaucourt and L. Pichat, (E)-alcenyl-5-desoxy-2-uridines par couplages d'organozirconiens ethyleniques avec l'iodo-5 o-3,5-bis(trimethyl) desoxyuridine, catalyses par des complexes organopallades, Tetrahedron Lett., 1982, 23, 63–64 CrossRef CAS; (c) F. Zeng and E. I. Negishi, A Highly efficient, selective, and general method for the synthesis of conjugated (all-E)-oligoenes of the (CH=CH)n type via iterative hydrozirconation-palladium-catalyzed cross-Coupling, Org. Lett., 2002, 4, 703–706 CrossRef CAS PubMed.
- Y. Hayasi, M. Riediker, J. S. Temple and J. Schwartz, Ligand control in palladium-catalyzed coupling reactions between organozirconium compounds and allylic species, Tetrahedron Lett., 1981, 22, 2629–2632 CrossRef CAS.
- S. L. Wiskur, A. Korte and G. C. Fu, Cross-couplings of alkyl electrophiles under “ligandless” conditions: Negishi reactions of organozirconium reagents, J. Am. Chem. Soc., 2004, 126, 82–83 Search PubMed.
- (a) M. Riediker and J. Schwartz, A new synthesis of 25-hydroxycholesterol, Tetrahedron Lett., 1981, 22, 4655–4658 CrossRef CAS; (b) A. M. G. Barrett, M. Peña and J. A. Willardsen, Total synthesis and structural elucidation of the antifungal agent papulacandin D, J. Org. Chem., 1996, 61, 1082–1100 Search PubMed; (c) T. Hu and J. S. Panek, Enantioselective synthesis of the protein phosphatase inhibitor (−)-motuporin, J. Am. Chem. Soc., 2002, 124, 11368–11378 Search PubMed.
- V. Navickas, C. Rink and M. E. Maier, Synthetic studies towards leiodermatolide: rapid stereoselective syntheses of key fragments, Synlett, 2011, 191–194 Search PubMed.
- S. Xu, H. M. Holst, S. B. McGuire and N. J. Race, Reagent control enables selective and regiodivergent opening of unsymmetrical phenonium ions, J. Am. Chem. Soc., 2020, 142, 8090–8096 CrossRef CAS PubMed.
- N. F. O'Rourke, K. A. Davies and J. E. Wulff, Cascading radical cyclization of bis-vinyl ethers: mechanistic investigation reveals a 5-exo/3-exo/retro-3-exo/5-exo pathway, J. Org. Chem., 2012, 77, 8634–8647 CrossRef PubMed.
- D. W. Hart, T. F. Blackburn and J. Schwartz, Hydrozirconation. III. Stereospecific and regioselective functionalization of alkylacetylenes via vinylzirconium(IV) intermediates, J. Am. Chem. Soc., 1975, 97, 679–680 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |