 Open Access Article
Open Access ArticleLandfill leachate treatment by the symbiotic system of bacteria and microalgae: high-efficiency and low-consumption green biological treatment system
Di Wanga,
Salma Tabassum *bc,
Jun Li*a,
Guanglei Lia,
Hüseyin Altundagbc,
Ningwen Zhanga and
Imran Khan
*bc,
Jun Li*a,
Guanglei Lia,
Hüseyin Altundagbc,
Ningwen Zhanga and
Imran Khan d
d
aSchool of Municipal and Environmental Engineering, Shenyang Jianzhu University, Shenyang 110168, China. E-mail: junlee@sjzu.edu.cn
bDepartment of Chemistry, Faculty of Science, Sakarya University, Sakarya 54187, Turkey. E-mail: tsalma@sakarya.edu.tr; salmazenith@gmail.com; Tel: +90 5011085451
cBiomedical, Magnetic and Semiconductor Materials Research Center (BIMAS-RC), Sakarya University, Sakarya 54187, Turkey
dDepartment of Chemistry, College of Science, Sultan Qaboos University, Muscat, Oman
First published on 7th April 2026
Abstract
This study uses domestic sewage to dilute landfill leachate, conserve freshwater resources, and supplement phosphorus. The proportion of landfill leachate is increased, and the microalgae photosynthesis is coupled with the SBR system to process the diluted leachate. A bacterial and algal symbiotic photobioreactor (PBR) was constructed to improve the efficiency of sewage treatment by optimizing parameters (aeration rate, light) for investigating the synergy of microalgae and bacteria, and the effect of treating Landfill leachate. The long-term operational impact of the reactor under two different inoculation conditions was investigated: one group was inoculated only with activated sludge and controlled light to promote the spontaneous growth of microalgae (Rc), and the other group was inoculated with activated sludge and Chlorella (Rs). The highest pollutant removal efficiencies were observed in 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (microalgae/sludge) cultures, with COD at 96.5%, NH4+-N at 97.4%, and PO43−-P at 92.3%. Synergistic growth of bacteria and microalgae was observed, with a total biomass concentration of 2.32 g L−1. Pollutant removal effect was best at an aeration rate of 0.6 L min−1, with removal efficiencies of 72.7% for COD, 78.3% for NH4+-N, and 62.5% for PO43−-P. A low-aeration method, mechanical aeration and microalgae photosynthesis cooperated to reduce operating costs. When light intensity was 108 µmol m−2 s−1, pollutant removal efficiencies of COD 82.6%, NH4+-N 84.9% and PO43−-P 75.9% were achieved. Treatment effect of the Rs system: average volume load of 36.22 mg per L per h COD, 8.53 mg per L per h NH4+-N, 0.44 mg per L per h PO43−-P. This provides new ideas for achieving high-efficiency, low-consumption green biological treatment of landfill leachate.
1 (microalgae/sludge) cultures, with COD at 96.5%, NH4+-N at 97.4%, and PO43−-P at 92.3%. Synergistic growth of bacteria and microalgae was observed, with a total biomass concentration of 2.32 g L−1. Pollutant removal effect was best at an aeration rate of 0.6 L min−1, with removal efficiencies of 72.7% for COD, 78.3% for NH4+-N, and 62.5% for PO43−-P. A low-aeration method, mechanical aeration and microalgae photosynthesis cooperated to reduce operating costs. When light intensity was 108 µmol m−2 s−1, pollutant removal efficiencies of COD 82.6%, NH4+-N 84.9% and PO43−-P 75.9% were achieved. Treatment effect of the Rs system: average volume load of 36.22 mg per L per h COD, 8.53 mg per L per h NH4+-N, 0.44 mg per L per h PO43−-P. This provides new ideas for achieving high-efficiency, low-consumption green biological treatment of landfill leachate.
1. Introduction
With the acceleration of urbanization and industrialization, environmental pollution from urban solid waste is becoming increasingly severe. The primary methods of urban waste disposal are landfill, incineration and composting. The landfill method is widely used because of its simple operation and low cost. However, landfills also generate landfill leachate. Landfill leachate is a high-concentration, refractory liquid formed by various factors, such as the infiltration of external moisture and the microbial decomposition of the garbage itself after landfill treatment. The main sources of landfill leachate are inherent moisture, atmospheric precipitation, groundwater infiltration, and microbial degradation.1 The composition and volume of landfill leachate are highly variable and non-uniform, depending on many factors, including the nature of the waste itself, landfill's climate, the degree of compaction, the stage of waste degradation, and more. The COD concentration is exceptionally high, 10–100 times that of domestic sewage,2 and the water quality is quite complex.3 Leachate water quality is classified according to landfill age:4 landfill age <1 year is characterized by low pH and high COD. The pH value increases when landfill leachate is >5 years old, and the COD concentration decreases. NH4+-N concentration is high.5 When pH is high, PO43−-P precipitates, resulting in insufficient phosphorus (P) for microbial growth. BOD5:P is generally >300, which is far from the optimal ratio (BOD5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) N
N![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) P = 100
P = 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5
5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) required by microorganisms.6 Therefore, it must be treated to meet regulatory standards before being discharged into sewers or surface waters. The physicochemical, adsorption, and coagulation–sedimentation methods cannot be used alone to treat landfill leachate; most are used for pretreatment.7–9 In membrane treatment processes, the interception efficiency of small-molecular substances is low. The subsequent treatment of the concentrated solution still requires integration with other methods, and it is prone to membrane fouling and to increased processing costs.5,10 The effectiveness of incorporating quorum-quenching bacteria, specifically Brucella sp. ZJ1, as a biofouling control strategy in membrane bioreactor systems treating synthetic landfill leachate was demonstrated by Zhu et al.11 Compared with other treatment technologies, biological treatment is considered efficient and inexpensive.12,13 However, the aerobic biological treatment is not suitable for treating leachate with long-term residence time in landfill, and its BOD/COD is low, which can easily reduce the activity of sludge bacteria, and the effluent is unstable. Higher concentrations of organic matter and toxic compounds inhibit microorganisms' growth. The nutrients required for microbial growth are missing, and phosphates must be added to balance nutrient proportions. Compared with aerobic treatment, anaerobic biological treatment can effectively treat high-concentration organic wastewater (COD > 10 g L−1). It is worth noting that if the biological method is used alone to treat landfill leachate, the final treatment effect cannot meet the wastewater discharge standard.14 The Sun et al.15 study shed light on the conversion of various fractions of organic matter during landfill leachate treatment and offered strategies to reduce N2O emissions. There is an increasing need for sustainable treatment technologies that focus on resource recovery and minimize environmental impact. There is an urgent need for practical, sustainable, and cost-effective on-site leachate treatment technologies to reduce the burden of water pollution and energy shortages. Compared with other biotechnologies,16,17 bacteria–microalgae symbiosis technology has the advantages of energy saving and consumption reduction, improves the efficiency of sewage treatment,18 promotes the recycling of microalgal biomass resources19,20 and is conducive to the effective sedimentation of microalgae cells.21 This technology of bacteria and microalgae has been widely reported, such as its application in the treatment of urban wastewater,22 swine-raising wastewater,23 aquaculture wastewater,24 and so on. In the field of landfill leachate, however, it is still relatively new. The mechanism of the symbiotic system of bacteria and microalgae in sewage treatment is essentially the synergistic growth of bacteria and microalgae, including the assimilation and absorption of microalgae, the oxidative degradation of bacteria, and the removal of physicochemical effects caused by pH changes in the symbiotic environment. Bacteria and microalgae can utilize each other's metabolites. The oxidative degradation of bacteria is well known, and microalgae's assimilation, absorption and physicochemical effects on nitrogen and phosphorus.25–28 There are three primary forms used in sewage treatment: suspended bacteria and microalgae systems,29,30 immobilized bacteria and microalgae systems,31,32 and bacteria and microalgae biofilm systems.23,33 The effects of pollutants are still closely related to microbial growth in the suspended bacteria–microalgae symbiotic system. Due to the shading effect of sludge, the light factor becomes the most significant factor affecting the system's treatment performance, and algal biomass promotes bacterial proliferation and further sludge production. The unavoidable mixing and stirring in the suspension reactor resulted in poor sedimentation of microorganisms, leading to unsatisfactory, unstable final effluent quality. In immobilized bacteria and microalgae systems, immobilized material will block sunlight and reduce the photosynthesis efficiency of microalgae; the cost of the immobilized carrier is high and needs to be replaced after some time, otherwise it will cause secondary pollution after cracking34 and limit the development and utilization in practical applications. Compared with the suspension system, the bacteria and microalgae biofilm system has certain advantages35 but suffers from biofilm shedding problems.23 The effect of the symbiotic system of bacteria and microalgae in wastewater treatment is affected by many aspects, such as the bacteria and microalgae ratio,36,37 light,38,39 DO,40,41 temperature,41 pH,42 organic load,43 wastewater concentration and so on. The reactor's external environment and operating conditions differ, and the treatment effects and mechanisms of the bacteria–microalgae system are also different.
1) required by microorganisms.6 Therefore, it must be treated to meet regulatory standards before being discharged into sewers or surface waters. The physicochemical, adsorption, and coagulation–sedimentation methods cannot be used alone to treat landfill leachate; most are used for pretreatment.7–9 In membrane treatment processes, the interception efficiency of small-molecular substances is low. The subsequent treatment of the concentrated solution still requires integration with other methods, and it is prone to membrane fouling and to increased processing costs.5,10 The effectiveness of incorporating quorum-quenching bacteria, specifically Brucella sp. ZJ1, as a biofouling control strategy in membrane bioreactor systems treating synthetic landfill leachate was demonstrated by Zhu et al.11 Compared with other treatment technologies, biological treatment is considered efficient and inexpensive.12,13 However, the aerobic biological treatment is not suitable for treating leachate with long-term residence time in landfill, and its BOD/COD is low, which can easily reduce the activity of sludge bacteria, and the effluent is unstable. Higher concentrations of organic matter and toxic compounds inhibit microorganisms' growth. The nutrients required for microbial growth are missing, and phosphates must be added to balance nutrient proportions. Compared with aerobic treatment, anaerobic biological treatment can effectively treat high-concentration organic wastewater (COD > 10 g L−1). It is worth noting that if the biological method is used alone to treat landfill leachate, the final treatment effect cannot meet the wastewater discharge standard.14 The Sun et al.15 study shed light on the conversion of various fractions of organic matter during landfill leachate treatment and offered strategies to reduce N2O emissions. There is an increasing need for sustainable treatment technologies that focus on resource recovery and minimize environmental impact. There is an urgent need for practical, sustainable, and cost-effective on-site leachate treatment technologies to reduce the burden of water pollution and energy shortages. Compared with other biotechnologies,16,17 bacteria–microalgae symbiosis technology has the advantages of energy saving and consumption reduction, improves the efficiency of sewage treatment,18 promotes the recycling of microalgal biomass resources19,20 and is conducive to the effective sedimentation of microalgae cells.21 This technology of bacteria and microalgae has been widely reported, such as its application in the treatment of urban wastewater,22 swine-raising wastewater,23 aquaculture wastewater,24 and so on. In the field of landfill leachate, however, it is still relatively new. The mechanism of the symbiotic system of bacteria and microalgae in sewage treatment is essentially the synergistic growth of bacteria and microalgae, including the assimilation and absorption of microalgae, the oxidative degradation of bacteria, and the removal of physicochemical effects caused by pH changes in the symbiotic environment. Bacteria and microalgae can utilize each other's metabolites. The oxidative degradation of bacteria is well known, and microalgae's assimilation, absorption and physicochemical effects on nitrogen and phosphorus.25–28 There are three primary forms used in sewage treatment: suspended bacteria and microalgae systems,29,30 immobilized bacteria and microalgae systems,31,32 and bacteria and microalgae biofilm systems.23,33 The effects of pollutants are still closely related to microbial growth in the suspended bacteria–microalgae symbiotic system. Due to the shading effect of sludge, the light factor becomes the most significant factor affecting the system's treatment performance, and algal biomass promotes bacterial proliferation and further sludge production. The unavoidable mixing and stirring in the suspension reactor resulted in poor sedimentation of microorganisms, leading to unsatisfactory, unstable final effluent quality. In immobilized bacteria and microalgae systems, immobilized material will block sunlight and reduce the photosynthesis efficiency of microalgae; the cost of the immobilized carrier is high and needs to be replaced after some time, otherwise it will cause secondary pollution after cracking34 and limit the development and utilization in practical applications. Compared with the suspension system, the bacteria and microalgae biofilm system has certain advantages35 but suffers from biofilm shedding problems.23 The effect of the symbiotic system of bacteria and microalgae in wastewater treatment is affected by many aspects, such as the bacteria and microalgae ratio,36,37 light,38,39 DO,40,41 temperature,41 pH,42 organic load,43 wastewater concentration and so on. The reactor's external environment and operating conditions differ, and the treatment effects and mechanisms of the bacteria–microalgae system are also different.
Current research often uses freshwater dilution to mitigate the toxicity of high-concentration pollutants to microorganisms. However, the large-scale use of freshwater to dilute leachate exacerbates water scarcity. Typical dilution factors range from 10–50%, requiring an additional 50–90 Liters of water to treat 10–50 Liters of leachate. This can limit the large-scale implementation of this process. To address this issue, this study used domestic sewage with lower pollutant concentration as a dilution medium, mixing it with landfill leachate. Because healthy microalgae growth requires phosphorus, and landfill leachate's phosphorus content is limited, external phosphorus supplementation is necessary to achieve optimal biomass growth and nutrient removal. In this case, domestic sewage dilutes the landfill leachate and replenishes nutrients it lacks. Sequencing Batch Reactor (SBR) still faces technical bottlenecks, such as low nitrogen and phosphorus removal rates. Coupling microalgae photosynthesis with traditional SBRs for synergistic wastewater treatment leverages the bacteria's efficient organic matter degradation and the microalgae's efficient nitrogen and phosphorus assimilation to achieve high-quality effluent. This technology offers advantages such as simultaneous denitrification and dephosphorization, reducing energy consumption costs. It is a sustainable and low-cost treatment technology with excellent settling performance and biomass resource recycling. Therefore, this study focuses on removing carbon, nitrogen, and phosphorus from landfill leachate. By sequentially increasing the leachate ratio and by studying different bacterial/microalgae dosing ratios and optimizing the synergistic conditions between bacteria and microalgae, we aim to identify optimal conditions for wastewater treatment by bacteria and microalgae, construct a highly efficient bacterial–algal symbiotic system, and contribute to the practical application of the bacterial–algal symbiotic PBR process. The main objectives are: (i) effects of different bacterial and algal inoculation ratios on treatment efficiency and system stability were investigated through batch experiments, in preparation for the subsequent construction of bacterial and algal photobioreactors; (ii) investigation on the comprehensive optimization and control of the parameters of the bacteria–microalgae PBR symbiotic system. The effects of aeration and light on the wastewater treatment were investigated. The differences in effluent quality and bacteria–microalgae characteristics under different conditions of the same type of reactor were examined to study the changes in the system's overall efficiency. The optimal operating parameters were selected. A traditional activated sludge SBR reactor was used as a control to investigate the synergistic effect between bacteria and microalgae, providing a scientific theoretical basis for optimizing design parameters, and (iii) based on the determination of optimal operating parameters, one approach involves using light to induce the natural growth of microalgae in the sludge to form a bacterial–algal symbiotic system. The other involves artificially inoculating microalgae to form a bacterial–algal symbiotic system. Then, long-term operations were conducted to examine the operating performance, community structure, and differences between the spontaneously formed bacterial–algal symbiotic system and the microalgal symbiotic system within the reactor.
2. Materials and methods
2.1 Sources of bacteria and microalgae
Chlorella vulgaris (FACHB-08), microalgae were purchased from the Institute of Freshwater Hydrobiology, Chinese Academy of Sciences, China. It's a spherical unicellular freshwater microalga (diameter 3–8 µm). The activated sludge was taken from the aeration tank of the sewage treatment plant. Raw landfill leachate and simulated domestic sewage were used as the test influents, and the pH was adjusted with a small amount of NaOH (Table 1). The experimental water used is the original solution of simulated domestic sewage and garbage leachate. The composition of trace elements are mentioned in Table 2. The leachate from the garbage comes from the concentration tank at the Shenyang Daxin garbage treatment plant in China. The concentration of pollutants in the mixed wastewater is detailed in Table 3.| Concentration (mg L−1) | |
|---|---|
| CH3COONa | 448.7 |
| KH2PO4 | 47 |
| NH4Cl | 133.6 |
| NaHCO3 | 300 |
| MgSO4·7H2O | 75 |
| CaCl2·2H2O | 36 |
| Ammonium ferric citrate | 6 |
| EDTANa2 | 1 |
| Reagent | Concentration (µg L−1) |
|---|---|
| H3BO3 | 2.86 |
| MnCl2·4H2O | 1.86 |
| ZnSO4·7H2O | 0.22 |
| Na2MoO4·2H2O | 0.39 |
| CuSO4·5H2O | 0.08 |
| Co(NO3)2·6H2O | 0.05 |
| Water quality index (mg L−1) | COD | NH4+-N | NO2−-N | NO3−-N | PO43−-P |
|---|---|---|---|---|---|
| Simulated sewage | 350 | 35 | — | — | 10 |
| Landfill leachate | 6748.27 | 2145.68 | 4.36 | 57.59 | 5.8 |
2.2 Test method
2.2.1.1 Inoculation. BG11 culture solution was prepared in an Erlenmeyer flask. The bottle mouth was wrapped in multiple layers of gauze and kraft paper and sterilized in a high-pressure steam sterilizer at 121 °C for 22 minutes. After sterilization, it was cooled to room temperature on a clean workbench (Tables 4, 5 and Fig. 1a). The outer surface of the microalgae liquid container was wiped with 75% alcohol, placed on a clean workbench, and the sterile air was turned on for 15 minutes. The pure microalgae liquid was transferred to the sterile medium, and the inoculation ratio was 1
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. Sterile air was maintained throughout the inoculation period. After inoculation, the mouth of the conical flask was tightly wrapped with multiple layers of gauze. This prevents contamination from other impurities while allowing CO2 from the air to enter. The inoculated Chlorella was placed in a shaking incubator at a low temperature and low light to reduce its growth and metabolism. The light intensity was set to 36 µmol m−2 s−1, and the light–dark ratio was 12 h light
1. Sterile air was maintained throughout the inoculation period. After inoculation, the mouth of the conical flask was tightly wrapped with multiple layers of gauze. This prevents contamination from other impurities while allowing CO2 from the air to enter. The inoculated Chlorella was placed in a shaking incubator at a low temperature and low light to reduce its growth and metabolism. The light intensity was set to 36 µmol m−2 s−1, and the light–dark ratio was 12 h light![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 12 h darkness (20 °C and 20 rpm). Subculture was performed every 2–3 months.
12 h darkness (20 °C and 20 rpm). Subculture was performed every 2–3 months.
| Components | Dosage | Mother liquor concentration |
|---|---|---|
| NaNO3 | 10 mL L−1 | 15 g/100 mL dH2O |
| K2HPO4 | 10 mL L−1 | 2 g/500 mL dH2O |
| MgSO4·7H2O | 10 mL L−1 | 3.75 g/500 mL dH2O |
| CaCl2·2H2O | 10 mL L−1 | 1.8 g/500 mL dH2O |
| Citric acid | 10 mL L−1 | 0.3 g/500 mL dH2O |
| Ammonium citrate | 10 mL L−1 | 0.3 g/500 mL dH2O |
| EDTANa2Na2CO3 | 10 mL L−1 | 0.05 g/500 mL dH2O |
| Na2CO3 | 10 mL L−1 | 1 g/500 mL dH2O |
| A5 | 1 mL L−1 | — |
| Concentration | Concentration | ||
|---|---|---|---|
| H3BO3 | 2.86 g per L dH2O | Na2MoO4·2H2O | 0.39 g per L dH2O |
| MnCl2·4H2O | 1.86 g per L dH2O | CuSO4·5H2O | 0.08 g per L dH2O |
| ZnSO4·7H2O | 0.22 g per L dH2O | Co(NO3)2·6H2O | 0.05 g per L dH2O |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 (medium). The inoculation operation was the same as for preserving microalgae. Culture conditions were changed, and the light intensity was 54 µmol m−2 s−1, with a light–dark ratio of 12 h light
4 (medium). The inoculation operation was the same as for preserving microalgae. Culture conditions were changed, and the light intensity was 54 µmol m−2 s−1, with a light–dark ratio of 12 h light![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 12 h dark (25 °C and 130 rpm). When the growth reached the logarithmic growth phase (end), the microalgae were transferred to a new medium under the same culture conditions. The culture medium used in this test must be prepared fresh and cannot be stored. The stock solution can be stored at 4 °C.
12 h dark (25 °C and 130 rpm). When the growth reached the logarithmic growth phase (end), the microalgae were transferred to a new medium under the same culture conditions. The culture medium used in this test must be prepared fresh and cannot be stored. The stock solution can be stored at 4 °C.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 12 h dark cycle (Fig. 1b). 888.9 mL and 111.1 mL of acclimatized Chlorella and sludge (800 mL and 200 mL, 500 mL and 500 mL, and 200 mL and 800 mL, respectively) were taken to obtain Chlorella/sludge ratios of 8
12 h dark cycle (Fig. 1b). 888.9 mL and 111.1 mL of acclimatized Chlorella and sludge (800 mL and 200 mL, 500 mL and 500 mL, and 200 mL and 800 mL, respectively) were taken to obtain Chlorella/sludge ratios of 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 4
1, 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 1
1, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, and 1
1, and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4. The inoculum was centrifuged. The resulting pellet was placed in the reactor. The water used was a combined wastewater solution containing 4% landfill leachate and 96% domestic sewage. 2 L of the combined wastewater was added to the beaker to assess nutrient removal efficiency (Table 6).
4. The inoculum was centrifuged. The resulting pellet was placed in the reactor. The water used was a combined wastewater solution containing 4% landfill leachate and 96% domestic sewage. 2 L of the combined wastewater was added to the beaker to assess nutrient removal efficiency (Table 6).
| Water quality | COD | NH4+-N | PO43−-P | NO2−-N | NO3−-N |
|---|---|---|---|---|---|
| Conc. (mg L−1) | 605.93 | 119.42 | 9.83 | 0.17 | 2.30 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) was Rs. Each group of four reactors had a different aeration rate: 0, 0.3, 0.6, and 1 L min−1. The test water was mixed with 5% leachate (Table 7). As shown in Fig. 1c, eight reactors were placed on a magnetic stirrer (25 °C, 120 rpm). Fluorescent tubes were installed at the top to provide sufficient lighting (54 µmol m−2 s−1) and to simulate sunlight conditions, achieving a 12 h
1) was Rs. Each group of four reactors had a different aeration rate: 0, 0.3, 0.6, and 1 L min−1. The test water was mixed with 5% leachate (Table 7). As shown in Fig. 1c, eight reactors were placed on a magnetic stirrer (25 °C, 120 rpm). Fluorescent tubes were installed at the top to provide sufficient lighting (54 µmol m−2 s−1) and to simulate sunlight conditions, achieving a 12 h![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 12 h light–dark ratio. Aeration was controlled by adjusting the gas flow meter to achieve 0, 0.3, 0.6, and 1 L min−1. The reactor operated twice daily (influent: 10 min, aeration: 640 min, settling: 60 min, and effluent: 10 min). The periodic water exchange rate was 80%, and the hydraulic retention time (HRT) was 15 h. Influent is poured into the reactor using a container until the liquid level reaches the 2 L mark (effluent was manually discharged). After the solution in the reactor was completely settled, the bottom of the outlet pipe was located near the 400 mL mark, and the other end was suctioned using a bulb syringe. Compared with the oxygen produced by algae, that from atmospheric diffusion was negligible.
12 h light–dark ratio. Aeration was controlled by adjusting the gas flow meter to achieve 0, 0.3, 0.6, and 1 L min−1. The reactor operated twice daily (influent: 10 min, aeration: 640 min, settling: 60 min, and effluent: 10 min). The periodic water exchange rate was 80%, and the hydraulic retention time (HRT) was 15 h. Influent is poured into the reactor using a container until the liquid level reaches the 2 L mark (effluent was manually discharged). After the solution in the reactor was completely settled, the bottom of the outlet pipe was located near the 400 mL mark, and the other end was suctioned using a bulb syringe. Compared with the oxygen produced by algae, that from atmospheric diffusion was negligible.
| Water quality | COD | NH4+-N | PO43−-P | NO2−-N | NO3−-N |
|---|---|---|---|---|---|
| Conc. (mg L−1) | 669.91 | 140.53 | 9.79 | 0.21 | 2.88 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) was Rs. The test was conducted for 80 days and divided into four phases (Table 8). The pollutant concentration in the test water was increased based on the previous stage, using 6% landfill leachate and 94% domestic sewage (Table 9). As shown in Fig. 1d, to ensure more uniform heating, the two reactors were placed in a constant-temperature water bath at 26 °C, and fluorescent lamps were installed above them. The lighting conditions were adjusted by changing the number and distance of the lamps. The reactor operated for two cycles per day; each cycle lasted 12 h (water inlet 10 min, aeration 640 min (stage 2 without aeration), settling 60 min, water outlet 10 min, aeration rate 0.6 L min−1, periodic water exchange rate 80%, and HRT 15 h). Peristaltic pumps controlled the inlet and outlet water.
1) was Rs. The test was conducted for 80 days and divided into four phases (Table 8). The pollutant concentration in the test water was increased based on the previous stage, using 6% landfill leachate and 94% domestic sewage (Table 9). As shown in Fig. 1d, to ensure more uniform heating, the two reactors were placed in a constant-temperature water bath at 26 °C, and fluorescent lamps were installed above them. The lighting conditions were adjusted by changing the number and distance of the lamps. The reactor operated for two cycles per day; each cycle lasted 12 h (water inlet 10 min, aeration 640 min (stage 2 without aeration), settling 60 min, water outlet 10 min, aeration rate 0.6 L min−1, periodic water exchange rate 80%, and HRT 15 h). Peristaltic pumps controlled the inlet and outlet water.
| Stage | Lighting time (h) | Aeration intensity (L min−1) | Light intensity (µmol m−2 s−1) |
|---|---|---|---|
| 1(D1–20) | 12 | 0.6 | 54 |
| 2(D21–40) | 24 | 0 | 54 |
| 3(D41–60) | 12 | 0.6 | 108 |
| 4(D61–80) | 12 | 0.6 | 180 |
| Water quality | COD | NH4+-N | PO43−-P | NO2−-N | NO3−-N |
|---|---|---|---|---|---|
| Conc. (mg L−1) | 733.89 | 161.64 | 9.75 | 0.26 | 3.45 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). After inoculating 1 L of seed sludge into each reactor, 4 L of Chlorella concentrate was inoculated into Rs at a microalgae–bacteria ratio of 4
1). After inoculating 1 L of seed sludge into each reactor, 4 L of Chlorella concentrate was inoculated into Rs at a microalgae–bacteria ratio of 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.
1.Two reactors were placed in a constant-temperature water bath, with small fluorescent lamps spirally wound around them to achieve a light intensity of 108 µmol m−2 s−1. The reactors were illuminated for 12 h to simulate sunlight conditions, achieving a 12 h![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 12 h light–dark ratio. The reactors were wrapped with tin foil to reduce the influence of the external environment. The aeration rate was 0.6 L min−1. A peristaltic pump and a timer control the influent and effluent. The reactor operated for two cycles per day, each cycle was 12 h (influent 10 min, aeration 640 min, settling 60 min, effluent 10 min, volume exchange rate 60%, and HRT 20 h). pH 7–8, and the reactions run for 100 days. The device is the same as shown in Fig. 1d. The pollutant concentration in the test water increased based on the previous stage. The wastewater used was a mixture of 7% landfill leachate and 93% domestic sewage (Table 10).
12 h light–dark ratio. The reactors were wrapped with tin foil to reduce the influence of the external environment. The aeration rate was 0.6 L min−1. A peristaltic pump and a timer control the influent and effluent. The reactor operated for two cycles per day, each cycle was 12 h (influent 10 min, aeration 640 min, settling 60 min, effluent 10 min, volume exchange rate 60%, and HRT 20 h). pH 7–8, and the reactions run for 100 days. The device is the same as shown in Fig. 1d. The pollutant concentration in the test water increased based on the previous stage. The wastewater used was a mixture of 7% landfill leachate and 93% domestic sewage (Table 10).
| Water quality | COD | NH4+-N | PO43−-P | NO2−-N | NO3−-N |
|---|---|---|---|---|---|
| a Conc. is concentration. | |||||
| Conc. (mg L−1) | 797.88 | 182.74 | 9.71 | 0.34 | 4.03 |
2.3 Analytical methods
The analysis methods were based on the water monitoring specifications and method standards promulgated by the Ministry of Environmental Protection.44 NH4+-N (Nessler's reagent spectrophotometry: UV spectrophotometer), NO2−-N (N-(1-naphthyl)ethylene diamine method), NO3−-N (UV spectrophotometry), TSS (filter paper weighing method), PO43−-P (potassium persulfate oxidation-UV spectrophotometry), Chl-a (acetone spectrophotometer method), pH by portable pH meter (instrument: PHS-29A type) and dissolved oxygen (DO) by HACH Sension 8 portable Dissolved Oxygen analyzer. The soluble EPS (S-EPS) was extracted using NaOH heating.45 The determination method of polysaccharides (PS) by the anthrone colorimetric method,46 and the Coomassie Brilliant Blue standard method was used for protein (PN) determination.47Due to the turbidity caused by sludge biomass, the OD680 of the culture medium cannot accurately reflect microalgae biomass, so ultraviolet spectrophotometry and hemocytometry are not suitable for measuring Chlorella biomass. The concentration of chlorophyll a can be used to characterize changes in microalgae growth.48 Chlorella chlorophyll a was determined using the acetone-spectrophotometric method.49 The black-and-white bottle method was used to measure the photosynthetic oxygen production rate of algae.50
 | (1) |
N, P volume load eqn (2):
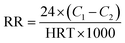 | (2) |
3. Results and discussion
3.1 Effect of bacteria and microalgae ratio on the bacteria–microalgae system in batch test
There are complex relationships between bacteria and algae, including mutualistic symbiosis and competition for nutrients. The biomass of bacteria and microalgae may affect their cooperation. If the microalgae ratio is too high, microalgae photosynthesis is stronger, leading to excessive O2 production, which may inhibit microalgae growth. If the bacteria ratio is too high. In that case, insufficient O2 cannot be provided to the bacteria, affecting bacterial growth. The synergistic effect is poor, which also inhibits microalgae growth, thus affecting reaction performance and community structure. Therefore, it is necessary to study the effect of the bacterial–microalgae ratio on the treatment efficiency of combined wastewater.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 1
1, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and 1
1 and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4, thereby saving operating costs. In reactors with activated sludge and microalgae, microalgal photosynthesis provides oxygen to facilitate heterotrophic bacteria's mineralization, thereby improving COD removal efficiency.
4, thereby saving operating costs. In reactors with activated sludge and microalgae, microalgal photosynthesis provides oxygen to facilitate heterotrophic bacteria's mineralization, thereby improving COD removal efficiency.
 | ||
| Fig. 2 Effect of different inoculation ratios: (a) COD, (b) NH4+-N, (c) PO43−-P (d) Chl-a, (e) TSS content, (f) DO, (g) turbidity content, and (h) EPS secretion. | ||
DO concentration is a factor in the respiration of heterotrophic aerobic bacteria and the degradation of COD by nutrients but DO concentrations of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 and 1
4 and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 is not the highest. Therefore, there may be a mutually beneficial interaction between microalgae and activated sludge that extends beyond CO2/O2 gas exchange (improving activated sludge mineralization). When the ratio was 8
1 is not the highest. Therefore, there may be a mutually beneficial interaction between microalgae and activated sludge that extends beyond CO2/O2 gas exchange (improving activated sludge mineralization). When the ratio was 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, although the COD decreased from 604.5 to 84.4 mg L−1, the COD removal efficiency was low due to insufficient activated sludge and limited COD removal by microalgae. Roudsari et al.51 believed that activated sludge played a significant role in the COD removal in the mixed system. Due to the accumulation of metabolites over time and the disintegration of microorganisms, the COD concentration increased. Wang et al.52 also found a similar phenomenon. Wang et al.52 suggested that after Chlorella treats the wastewater in the secondary sedimentation tank, the activity of organic matter decreases, and microalgae cannot utilize it. Chlorella will then use CO2 as a carbon source to secrete organic matter. Since there are no microorganisms in the environment to degrade organic matter, COD levels increase. NH4+-N removal trend in the reactor was similar, with levels below 20 mg L−1 at the end of the test and removal efficiencies of 88.9%, 97.4%, 93.2%, and 94.9%, respectively. The 4
1, although the COD decreased from 604.5 to 84.4 mg L−1, the COD removal efficiency was low due to insufficient activated sludge and limited COD removal by microalgae. Roudsari et al.51 believed that activated sludge played a significant role in the COD removal in the mixed system. Due to the accumulation of metabolites over time and the disintegration of microorganisms, the COD concentration increased. Wang et al.52 also found a similar phenomenon. Wang et al.52 suggested that after Chlorella treats the wastewater in the secondary sedimentation tank, the activity of organic matter decreases, and microalgae cannot utilize it. Chlorella will then use CO2 as a carbon source to secrete organic matter. Since there are no microorganisms in the environment to degrade organic matter, COD levels increase. NH4+-N removal trend in the reactor was similar, with levels below 20 mg L−1 at the end of the test and removal efficiencies of 88.9%, 97.4%, 93.2%, and 94.9%, respectively. The 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio is especially notable for its highest and fastest removal efficiency. NH4+-N can be directly absorbed by algal cells, promoting algal growth. The high NH4+-N removal efficiency was not only related to the algal cells' assimilation but also contributed to the sludge nitrification caused by the increase in DO concentration (Fig. 2b). The PO43−-P removal efficiency changes similarly to that of NH4+-N, with removal efficiencies of 63.7%, 92.3%, 84.9% and 75.5%, respectively (Fig. 2c).
1 ratio is especially notable for its highest and fastest removal efficiency. NH4+-N can be directly absorbed by algal cells, promoting algal growth. The high NH4+-N removal efficiency was not only related to the algal cells' assimilation but also contributed to the sludge nitrification caused by the increase in DO concentration (Fig. 2b). The PO43−-P removal efficiency changes similarly to that of NH4+-N, with removal efficiencies of 63.7%, 92.3%, 84.9% and 75.5%, respectively (Fig. 2c).
Reactors inoculated with the highest sludge concentrations had the lowest removal efficiency, which may be related to the lowest DO concentrations resulting from bacterial respiration. Abiotic nitrogen and phosphorus removal at pH > 8 is also an important mechanism. The Chlorella concentration determines its photosynthetic efficiency, which absorbs nitrogen and phosphorus through photosynthesis, thereby affecting pollutant removal efficiency.53 When the microalgae concentration was low, photosynthetic efficiency was directly proportional to it. When the microalgae concentration is too high, the treatment efficiency begins to decline due to the competitive consumption of oxygen by microalgae.54 Microalgae play a leading role in the bio assimilation process.37 However, this test found that the removal effect was poor in the 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 reactor due to a high microalgae inoculation ratio. Once they reach a specific density, the microalgae that overlap will be limited by light, inhibiting photosynthesis. This test simulates natural light conditions, and the respiration of high-density microalgae also competed with activated sludge for O2 under dark conditions.55 An microalgae/sludge ratio that is too high or too low in the inoculum is not conducive to the removal of pollutants.
1 reactor due to a high microalgae inoculation ratio. Once they reach a specific density, the microalgae that overlap will be limited by light, inhibiting photosynthesis. This test simulates natural light conditions, and the respiration of high-density microalgae also competed with activated sludge for O2 under dark conditions.55 An microalgae/sludge ratio that is too high or too low in the inoculum is not conducive to the removal of pollutants.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 reactor did not show the greatest increase in chlorophyll concentration because it had less activated sludge and an insufficient CO2 supply, both of which are detrimental to photosynthesis. High-density microalgae compete for nutrients, and the dead microalgae are degraded as organic matter, decreasing chlorophyll. Reactors with high sludge concentrations also blocked sunlight, reducing photosynthetic efficiency and, in turn reduced algal growth. After inoculation with Chlorella, TSS initially increased by 451, 515, 279, and 207 mg L−1, respectively, followed by a roughly similar decrease in the later stages (Fig. 2e). The final TSS concentration for each inoculation ratio was 1967, 2314, 2924, and 3692 mg L−1. However, chlorophyll a showed a noticeable upward trend, indicating that the reduction in sludge volume was the main driver of the decline in TSS in the symbiotic system. Lack of nutrients and oxygen reduces sludge activity.
1 reactor did not show the greatest increase in chlorophyll concentration because it had less activated sludge and an insufficient CO2 supply, both of which are detrimental to photosynthesis. High-density microalgae compete for nutrients, and the dead microalgae are degraded as organic matter, decreasing chlorophyll. Reactors with high sludge concentrations also blocked sunlight, reducing photosynthetic efficiency and, in turn reduced algal growth. After inoculation with Chlorella, TSS initially increased by 451, 515, 279, and 207 mg L−1, respectively, followed by a roughly similar decrease in the later stages (Fig. 2e). The final TSS concentration for each inoculation ratio was 1967, 2314, 2924, and 3692 mg L−1. However, chlorophyll a showed a noticeable upward trend, indicating that the reduction in sludge volume was the main driver of the decline in TSS in the symbiotic system. Lack of nutrients and oxygen reduces sludge activity.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio, the low sludge ratio resulted in insufficient CO2 supply, possibly due to CO2 absorption from the air in the later stage. The pH stabilized between 7 and 8, indicating that the growth and metabolism of microalgae and sludge reached a balance. The diversity of bacteria and algae in the reactor can resist external changes and achieve a stable pH value (Fig. SI1).
1 ratio, the low sludge ratio resulted in insufficient CO2 supply, possibly due to CO2 absorption from the air in the later stage. The pH stabilized between 7 and 8, indicating that the growth and metabolism of microalgae and sludge reached a balance. The diversity of bacteria and algae in the reactor can resist external changes and achieve a stable pH value (Fig. SI1).Initially, DO was increased to about 3 mg L−1 by aeration (Fig. 2f). In the reactor with a higher sludge ratio, DO dropped to almost 0 on the first day. This indicates that the activated sludge was in a state of vigorous respiration and metabolism in the early stage, and the O2 released by microalgae photosynthesis was completely consumed.54 Bacterial heterotrophic activity was higher than the microalgae's photosynthetic activity. Later, DO recovered. The low concentration of microalgae at a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 ratio could not provide sufficient O2 for the activated sludge, resulting in a prolonged low DO state. The DO level in the high microalgae ratio reactor remained at 3–4 mg L−1. Due to the reduction in microalgae biomass, it gradually decreases around the 10th day. In the early stage, the turbidity of the supernatant was high, and stable bacteria–microalgae symbionts had not yet formed in the reactor (Fig. 2g). Suspended Chlorella caused the supernatant to turn green. At the end of the test, the supernatant turbidity was below 5 NTU value, indicating good settling performance. Only turbidity 8
4 ratio could not provide sufficient O2 for the activated sludge, resulting in a prolonged low DO state. The DO level in the high microalgae ratio reactor remained at 3–4 mg L−1. Due to the reduction in microalgae biomass, it gradually decreases around the 10th day. In the early stage, the turbidity of the supernatant was high, and stable bacteria–microalgae symbionts had not yet formed in the reactor (Fig. 2g). Suspended Chlorella caused the supernatant to turn green. At the end of the test, the supernatant turbidity was below 5 NTU value, indicating good settling performance. Only turbidity 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 initially had an around 60 NTU, decreased slowly, and eventually remained at 22 NTU.
1 initially had an around 60 NTU, decreased slowly, and eventually remained at 22 NTU.
It showed that the high concentration of Chlorella is mostly suspended in the water and does not bind well with activated sludge. Chlorella has a diameter of 3–8 µm, is suspensible, and has poor settling performance. Sludge can capture microalgae cells, preventing them from being washed away. EPS are macromolecular gel networks with adsorption and aggregation effects, formed by proteins, polysaccharides, and other substances secreted by bacteria. Their viscosity is related to the formation of microbial aggregates. By binding with activated sludge, it can form more tightly bound EPS to adsorb algae, thereby enhancing settling performance.58 The settleability of bacteria and microalgae is also affected by the surface properties of microalgae cells and the amount of cations.59
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and 8
1 and 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (Fig. 2h). EPS positively affects nutrient uptake,61 so in the 4
1 (Fig. 2h). EPS positively affects nutrient uptake,61 so in the 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 reactor, the highest nutrient removal efficiency may be associated with the highest EPS concentration. Studies have shown that PN promotes the growth of bacteria and microalgae, as it has a surface electronegativity and high hydrophobicity, which positively affect the aggregation and flocculation of bacteria and algae.62 This study also found that the PN concentration difference among different bacteria and microalgae ratios was significantly between 43.8 and 63.8 mg gSS−1, and the PS difference was not significant, indicating that microalgae might convert influent N to PN.63 PN/PS64 can indicate the quality of sludge hydrophobicity. Similarly, the PN/PS ratio of 4
1 reactor, the highest nutrient removal efficiency may be associated with the highest EPS concentration. Studies have shown that PN promotes the growth of bacteria and microalgae, as it has a surface electronegativity and high hydrophobicity, which positively affect the aggregation and flocculation of bacteria and algae.62 This study also found that the PN concentration difference among different bacteria and microalgae ratios was significantly between 43.8 and 63.8 mg gSS−1, and the PS difference was not significant, indicating that microalgae might convert influent N to PN.63 PN/PS64 can indicate the quality of sludge hydrophobicity. Similarly, the PN/PS ratio of 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 is 1.94. The bacteria and microalgae symbionts have good hydrophobicity, the microorganisms and the water phase are easily separated, the aggregation effect is good, and the effluent turbidity is the lowest.
1 is 1.94. The bacteria and microalgae symbionts have good hydrophobicity, the microorganisms and the water phase are easily separated, the aggregation effect is good, and the effluent turbidity is the lowest.3.2 Process parameters optimization for bacteria–microalgae symbiotic reactor
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 was selected for the next test. A bacterial and microalgae photobioreactor (PBR) was constructed to increase the landfill leachate ratio further.
1 was selected for the next test. A bacterial and microalgae photobioreactor (PBR) was constructed to increase the landfill leachate ratio further.
3.2.1.1 Pollutant removal. COD removal efficiency of the Rs reactor is significantly higher than that of the Rc, indicating that the bacterial microalgae system has the potential advantage of removing COD under limited aeration conditions (Fig. 3a). Sludge activity higher and microalgae growth absorbs organic matter,54 contributing to the COD removal. Increasing the aeration rate increases the COD removal efficiency. Except for the un-aerated pure sludge reactor, COD removal efficiencies were 41.4%, 57.8%, and 64.9%, while for the bacteria and microalgae reactors, they were 60.7%, 67.3%, 72.7%, and 74.6%, respectively. The low removal efficiency of the un-aerated bacteria–microalgae reactor is due to the oxygen generated by the algae's photosynthesis being insufficient to maintain bacterial respiration in the activated sludge. Compared with the two groups of reactors with the same aeration rate, the COD removal efficiency increased by 181.3%, 62.5%, 25.7%, and 14.9%, respectively. At lower aeration rates, the additional oxygen produced by microalgae supplemented the sludge demand, improved the gas exchange rates and O2 utilization between the two, thereby enhancing organic matter removal.
 | ||
| Fig. 3 Effects of different aeration rates on: (a) COD removal, (b) NH4+-N removal, (c) PO43−-P removal, (d) TSS content, (e) Chl-a content, and (f) Chl-a/TSS content. | ||
The comparison of reactors with the same aeration rate indicates that adding microalgae significantly improved the NH4+-N removal efficiency (Fig. 3b). At lower aeration rates (0, 0.3 L min−1), NH4+-N removal efficiencies in the Rc system were 21.4% and 43.3%, and in the Rs system were 50.8% and 60.3%, respectively, increases of 137.3% and 39.2%. This indicates that lower aeration conditions can better promote bacteria–microalgae cooperation. Mechanical aeration significantly compensated for the insufficient O2 produced by microalgae, met the sludge's O2 demand, and further enhanced NH4+-N removal. At higher aeration rates (0.6 and 1 L min−1), the removal efficiencies were about 80%, with increases of 27.3% and 17.2%, respectively. It is inferred that a higher aeration rate of 1 L min−1 leads to higher DO content in the water, which will limit the photosynthetic rate, inhibit algal growth and reduce the growth rate of microalgae66 (affecting NH4+-N assimilation). The CO2 escape via aeration affects pollutant removal efficiency.67 Therefore, the similar NH4+-N removal efficiency at aeration rates of 0.6 and 1 L min−1 may be due to the higher DO and CO2 escape caused by the higher aeration rate of 1 L min−1, which inhibits microalgae reproduction and affects photosynthesis. Considering both economic cost and water treatment effect, an aeration rate of 0.6 L min−1 was preferred. The PO43−-P removal efficiency in the Rc reactor was directly proportional to the aeration rate, which was 15.1%, 33.5%, 40.6% and 52%, respectively (Fig. 3c). The Rs removal efficiencies for the four bacteria–microalgae reactors were 67.5%, 64%, 62.5%, and 57.9%. The Rs reactor without an external oxygen supply has the best phosphorus removal effect due to the dual impact of microalgae assimilation, anaerobic phosphorus release and aerobic phosphorus uptake by polyphosphate-accumulating bacteria. Polyphosphates mainly accumulated in microalgae rather than bacteria,68 indicating that microalgae played a significant role in phosphorus removal. Therefore, the removal efficiencies were similar at 0.3 and 0.6 L min−1, suggesting comparable algae growth. Microalgal cells have been reported to remove at least 70% of soluble phosphorus.68 Increased aeration intensity leads to greater carbon dioxide emissions and reduces the inorganic carbon required for microalgae growth, thus limiting growth. Excessive DO also inhibits microalgae growth, resulting in poorer removal efficiency in reactors with high aeration volumes.
3.2.1.2 Changes in bacterial and microalgae biomass. Both the bacteria and microalgae group and the sludge group reactors showed good growth in suspended solids within the range of 2–3.2 g L−1, except for the non-aerated sludge group reactors (Fig. 3d). The total suspended solid concentration in the aerated pure sludge group was 2.31, 2.58, and 2.87 g L−1, which increased with the increase of aeration.
The TSS concentrations in the bacteria and microalgae reactors were 2.61, 2.74, 2.72, and 3.04 g L−1, respectively. From the 10th to 15th day, the TSS fluctuated slightly within a specific range and gradually stabilized. Chlorella has a diameter of only a few microns and extremely light (its weight is negligible), this suggests that adding Chlorella did not negatively affect sludge growth and reproduction but instead promoted them. The two combined to form a stable symbiotic system of bacteria and microalgae. However, since the Rc(0) reactor lacked both additional mechanical aeration and oxygen provided by Chlorella, the activated sludge experienced poor growth conditions due to oxygen deficiency during the test. TSS dropped from 1.92 g L−1 to about 0.9 g L−1 at the end of the test. Microalgae photosynthesis can also provide O2 to the system, thereby reducing the sludge's dependence on external oxygen supply and maintaining sludge activity. Chlorophyll content increased throughout the operation, driven by Chlorella growth. The initial inoculated Chl-a concentration was around 2 mg L−1, increased to 5.6, 4.4, 4.2 and 3.6 mg L−1 at the end of the operation (Fig. 3e).
The reactor with the largest aeration had the lowest chlorophyll content. It may be due to the high DO concentration.69 The large aeration volume may also cause more CO2 to be blown off, resulting in inorganic carbon loss. Both reasons affect photosynthesis and inhibit algal growth. Low aeration rates promoted microalgal growth but negatively impacted system stability. With reduced aeration, Chl-a concentration in the suspension increases significantly and becomes extremely unstable. Effluent Chl-a concentration increased significantly, especially in the non-aerated reactor with the best Chlorella growth. Effluent Chl-a was high at 0.2–0.4 mg L−1 and fluctuated greatly. In the first 4 days, effluent Chl-a concentration in other reactors was 0.15–0.3 mg L−1. Chlorella is a unicellular green alga with good buoyancy and can be discharged with wastewater. The stability of the bacterial–algal symbiotic system depends on microalgae attaching to bacterial flocs or being captured by activated sludge flocs, which then settle together.58 In the later stage, Chl-a concentration in the effluent was low and stable, and fluctuated slightly (0–0.12 mg L−1). It has been reported that lower aeration intensity can increase the lipid concentration ratio in microalgae, thereby promoting the recycling of microalgae resources.70
Chl-a/TSS initial ratio was between 1 and 1.5 mg g−1, the ratio was stable after 10 days, slightly increased, and increased by 2.79, 1.86, 1.67, and 1.56 mg g−1, indicating that the bacteria and microalgae are in good condition (Fig. 3f). The decreasing Chl-a/TSS ratio with the increasing aeration rate suggested that the increase in aeration rate disrupts the synergy and balance between bacteria and microalgae. Increased aeration inhibits microalgae growth. O2 is essential for activated sludge. Increased aeration rate promotes sludge reproduction.69 Therefore, appropriately reducing the aeration intensity can promote cooperation between bacteria and microalgae, leading bacteria to rely on O2 produced by microalgae for survival, which explains why Chl-a/TSS increases under low aeration conditions. The aeration rate of 0.6 L min−1 was taken as the operating parameter of the subsequent test (Fig. 3a–f). This conclusion is not solely based on Fig. 3e. To determine the optimal aeration rate, the pollutant removal effect and the growth characteristics of bacteria and microalgae should be considered together.
3.2.1.3 Chlorella activity. POER represents the amount of oxygen produced per unit time per unit amount of chlorophyll a and can be used to measure algal cell activity. The POER value decreases with the increase of aeration intensity (Fig. SI2), which are 47.2, 38.6, 33.5 and 28.7 O2 per mg Chl-a per h, respectively. The aerated reactor showed decreases of 22.2%, 40.8%, and 64.4%. This trend is similar to that of chlorophyll content, further indicating that increasing aeration intensity significantly affects microalgal photosynthesis. This may be due to the high DO concentration or CO2 release during aeration, leading to a lack of inorganic carbon and inhibiting microalgal activity.
3.2.2.1 Pollutant removal. The synergistic effect of bacteria and microalgae improved the gas exchange rate and oxygen utilization rate, which was beneficial to COD removal (Fig. 4a). At the beginning of the I stage, the COD removal efficiency of Rs fluctuated around 40%, stabilizing after 13 days, with a removal efficiency of 78%. The COD removal efficiency of Rc fluctuated greatly, and its shock load resistance capacity was not as good as that of Rs, reaching 57.5% at the end of the stage. In the II stage, there was a significant difference in carbon removal efficiency under natural conditions (12 h
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 12 h light–dark cycle) and prolonged light conditions (24 h full light). The COD concentration removal efficiency dropped to 53.5% as activated sludge contributed significantly to COD removal. Experimental conditions of stage II are favorable for the growth of microalgae rather than sludge; O2 required for the degradation of organic matter by activated sludge can only come from microalgae. Lee et al.73 found that carbon removal was positively correlated with the length of the dark cycle, and bacterial growth conditions were consistent with COD removal. The RC reactor's COD removal efficiency decreased significantly after aeration was stopped. Aeration and light–dark cycles were restored in the III stage, and light intensity was increased simultaneously. The COD removal efficiency of the Rc reactor recovered to 53.4% at the end. COD removal efficiency (Rs reactor) rose to 82.6%, indicating that the bacteria can utilize more oxygen to degrade organic matter. Chlorella has a heterotrophic metabolic pathway74 and thus consumes COD during dark periods. In the IV stage, the light intensity increased to 180 µmol m−2 s−1, and the COD removal efficiency of the Rs reactor significantly different from that in the previous stage. The effect of light intensity on bacteria needs to be further studied. Chlorophyll was detected at the end of the test, as stronger light promoted microalgae growth in the sludge, leading to spontaneous symbiosis between bacteria and the microalgae.
12 h light–dark cycle) and prolonged light conditions (24 h full light). The COD concentration removal efficiency dropped to 53.5% as activated sludge contributed significantly to COD removal. Experimental conditions of stage II are favorable for the growth of microalgae rather than sludge; O2 required for the degradation of organic matter by activated sludge can only come from microalgae. Lee et al.73 found that carbon removal was positively correlated with the length of the dark cycle, and bacterial growth conditions were consistent with COD removal. The RC reactor's COD removal efficiency decreased significantly after aeration was stopped. Aeration and light–dark cycles were restored in the III stage, and light intensity was increased simultaneously. The COD removal efficiency of the Rc reactor recovered to 53.4% at the end. COD removal efficiency (Rs reactor) rose to 82.6%, indicating that the bacteria can utilize more oxygen to degrade organic matter. Chlorella has a heterotrophic metabolic pathway74 and thus consumes COD during dark periods. In the IV stage, the light intensity increased to 180 µmol m−2 s−1, and the COD removal efficiency of the Rs reactor significantly different from that in the previous stage. The effect of light intensity on bacteria needs to be further studied. Chlorophyll was detected at the end of the test, as stronger light promoted microalgae growth in the sludge, leading to spontaneous symbiosis between bacteria and the microalgae.
 | ||
| Fig. 4 The effect of (a) COD removal, (b) NH4+-N removal, (c) PO43−-P removal, (d) TSS changes (e) Chl-a; Chl-a/TSS changes, and (f) POER changes under different lighting condition. | ||
In the early stage (I stage), the Rs reactor NH4+-N removal efficiency reached 77.1% after stabilization. The trend of Rc in the control group was the same, and the removal efficiency was 60.6% (Fig. 4b). In the II stage, additional aeration was stopped. At the same time, light duration was extended to 24 h to investigate whether the increased light duration could promote microalgae photosynthesis replacing the need for additional aeration. The Rs reactor removal efficiency dropped to 65.4%. Due to the insufficient oxygen supply to the Rc reactor, the removal efficiency was only 31.7%. This indicates that increasing the light duration does not completely replace aeration for algal photosynthesis. Low DO levels inhibit nitrifying bacteria activity in Rs, resulting from reduced DO content due to aeration cessation and excessive microalgae growth from prolonged light duration, which hinders photosynthesis. Prolonged exposure to light may inhibit the activity of ammonia-oxidizing bacteria (AOB) and nitrite-oxidizing bacteria (NOB).75 Almost complete nitrification inhibition was observed at a light intensity of 300 µmol m−2 s−1.76 Merbt et al.77 studied the effects of light–dark cycles on different nitrifying bacteria (Nitrosopumilus maritimus, Nitrosotalea deanaterra, Nitrosomonas europaea and Nitrosopira multiformis) under continuous light and 8 h light/16 h of darkness. Light/dark cycling was associated with lower light inhibition than continuous illumination. In the III stage, aeration and the normal light–dark cycle were restored, with light intensity increased. Both reactors were recovered well; removal efficiencies were 60.5% and 84.9%, respectively. The results show that microalgae can efficiently use intense light, provided there is a reasonable dark period that allows the electron transporters of the photosynthetic apparatus to re-oxidize. If the alternation of light and dark is not optimal, microalgae suffer radiation damage, and photosynthetic productivity significantly reduced due to the inhibition of photosystem II (PSII) by reactive oxygen species (ROS).78 Alternation of light and dark cycles enhances photosynthetic efficiency. The Rs reactor in the IV stage may have reached light saturation. The average removal efficiency of the Rc reactor was 63.6%, higher than that of the previous stage.
The PO43−-P concentrations and removal efficiencies of the three stages of the Rc and Rs reactors are shown in Fig. 4c. Similar to the NH4+-N results, the overall performance of the Rs reactor was better than that of the Rc. The removal efficiency of the Rs reactor in the II stage decreased slightly to 64.8%, indicating that although a lack of aeration promoted microalgae growth, excessive light was detrimental to it. The Rc reactor had the worst reaction due to insufficient oxygen supply, with only 26.8% removal. Compared with the I stage, the Rs reactor removal efficiency in the III stage increased to 75.9%. Increasing light intensity can give microalgae an edge over competitors. The light intensity is conducive to the phosphorus assimilation by Chlorella. Algal cells can also take up excessive phosphorus,70 and the phosphorus intake was higher than that of bacteria. Rs can utilize more phosphorus for growth and metabolism. Biomass uptake can be considered the primary mechanism for phosphorus removal. Fan et al.22 found a strong correlation between total chlorophyll content and PO43−-P (r = 1.0). The PO43−-P removal in the fourth stage was similar to that of NH4+-N. However, the fluctuation in removal efficiency was not significant compared with the previous stage, which may be due to the gradual stabilization of the reactor during the test operation.
3.2.2.2 Changes in microbial and microalgae biomass. Biomass growth was predicted from changes in total suspended solids (TSS) content in the reactor over time (Fig. 4d). The Rc biomass showed a downward trend in the early stage, indicating that the Rc reactor had poor shock resistance. Rs biomass first decreased and then increased slowly in the second stage. Similar to the previous experiment, the Rc biomass concentration decreased when the external oxygen supply was stopped. In the III stage, resumption of aeration and light exposure were beneficial to Chlorella's growth and promoted activated sludge reproduction. The biomass concentrations of the Rc and Rs reactors finally increased to 2.68 g L−1 and 3.58 g L−1, respectively, indicating that bacteria can use more oxygen to degrade organic matter,18 which explains the bacterial growth at this stage. Huang et al.79 Observed that natural sunlight promoted algal growth, contributing to pollutant removal. The increase in light intensity during the IV stage favored microalgal growth, thereby enhancing synergistic effects between bacteria and microalgae and promoting sludge growth. However, in the second half of the stage, TSS concentration decreased, possibly due to the high microalgae density, which caused mutual shading and reduced photosynthetic rate, disrupting the balance between bacteria and microalgae from the previous stage. In the Rc system, due to the lower microalgal content, the above phenomenon did not occur.
The accumulation of algal biomass and algal cell composition is largely dependent on the supply of light. In the I stage, the chlorophyll concentration fluctuated within a small range, with an upward trend (Fig. 4e). In the II stage, sufficient light during the initial 24 hours promoted microalgae growth. The ratio subsequently decreased, likely because a high microalgae density increased the dark area within the reactor, thereby attenuating light. Exposure of microalgae to excessive light leads to the formation of harmful reactive oxygen species (ROS) and oxidative stress.72 Up to 80% of photons are emitted to avoid radiation damage and oxidative stress, reducing light utilization efficiency.80 Although microalgae biomass is high in the Rs reactor and is expected to produce more oxygen, this may not be the case, as increased biomass also reduces light transmittance through shading, thereby inhibiting oxygen production,54 affecting pollutant removal. Similar results were obtained from previous studies.81 Light intensity is related to microalgae growth, as reported by Ho et al.82 Biomass productivity increases with light intensity in the 7560–23520 lux range, with 23![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 520 lux as the light saturation point. Chl-a/TSS was relatively stable at around 2, indicating that the light–dark cycle was suitable for the microalgal proliferation. In the IV stage, the light saturation point was reached.
520 lux as the light saturation point. Chl-a/TSS was relatively stable at around 2, indicating that the light–dark cycle was suitable for the microalgal proliferation. In the IV stage, the light saturation point was reached.
3.2.2.3 Chlorella activity. The algal cell POER was measured at the end of each test period, and the results are shown in Fig. 4f. The POERs of the four stages were 36.2, 32.5, 46.7, and 47.4 O2 per mg Chl-a per h, respectively. The exposure time (light) was doubled, and POER decreased by 10.2%. When cells are exposed to excessive light, photosystem II (PSII) is constantly damaged and must be repaired by resynthesizing damaged components. Photosynthetic organisms exposed to saturated light can reduce oxidative damage by dissipating excess energy through heat.80 Algae's photosynthesis consists of light and dark reactions.18 Increasing the light exposure time is equivalent to shortening the dark time, affecting the dark reaction of algal cells. In the third stage, with increased light, the oxygen production capacity of microalgae increases, and the POER increases by 29% compared to the first stage. Using the oxygen produced by microalgal photosynthesis, bacteria can degrade organic matter and release carbon dioxide back to the microalgae, promoting photosynthesis. In the fourth stage, light intensity increases but reaches saturation, and the increase in POER is minimal. This explains why the pollutant removal effect in this stage is not significantly different from the previous stage.
3.3 Two strategies to build a bacterial–microalgal symbiosis system and long-term operation
Under sufficient light, microalgae grow spontaneously during the cultivation and operation of an activated sludge system. However, the difference between the bacteria–microalgae systems cultivated under these two inoculation conditions is unclear. This section compares the long-term operation of the bacteria–microalgae and sludge systems under light conditions. Two groups of photobioreactors were constructed under the optimized operating conditions (light intensity 108 µmol m−2 s−1, light cycle 12 h light![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 12 h darkness, aeration intensity 0.6 L min−1). In this section, only one group was inoculated with activated sludge. The light was controlled to promote the spontaneous growth of algae, and the other group was inoculated with activated sludge and Chlorella.
12 h darkness, aeration intensity 0.6 L min−1). In this section, only one group was inoculated with activated sludge. The light was controlled to promote the spontaneous growth of algae, and the other group was inoculated with activated sludge and Chlorella.
3.3.1.1 Sludge growth. The TSS trends in the two reactors were similar during cultivation (Fig. 5a). The initial sludge concentration was 3150 mg L−1 and subsequently decreased, as the inoculated sludge was not adapted to the new environment. Rc and Rs decreased to 2214 and 2743 mg L−1 after 15 days and 10 days, respectively (the system inoculated with microalgae had a strong resistance to shock loads). After the adaptation period, the reactor entered the rapid symbiont formation period. On the 60th day, Rc and Rs rose to 3648 and 3779 mg L−1, respectively, maintaining a steady, slow growth. Finally stabilized at 3502 and 4170 mg L−1, respectively, indicating that the microalgae and bacteria entered the mature stage. Growth reached equilibrium, and the two competed for nutrients, resulting in slow biomass growth.83 The interaction between bacteria and microalgae promotes biomass growth.84 Microalgae assimilate CO2 and convert it into substances needed for their growth. Bacteria degrade macromolecular organic matter into small molecular organic matter that microalgae can use, and microalgae provide the O2 needed for bacterial reproduction through photosynthesis. The bacteria and microalgae promote synergistic growth. With the formation of symbionts, the development and expansion of the internal anaerobic microenvironment were promoted (facultative anaerobic bacteria could grow and reproduce). The synergistic effect of bacteria and microalgae was more substantial in Rs.
 | ||
| Fig. 5 Changes of (a) MLSS, (b) Chl-a, (c) COD removal, (d) NH4+-N removal, (e) NO2−-N, NO3−-N, (f) PO43−-P removal, (g) PN component, and (h) PS in the reactor. | ||
3.3.1.2 Microalgae growth. For pure microalgae, chlorophyll accounted for about 15% of the dry weight.85 Chlorophyll concentration (Chl-a) can be used to indicate algal growth. Fig. 5b shows the variation of Chl-a in the Rc and Rs reactors with time. On the 0th day, the concentrations of the two reactors were 266 and 2743 µg L−1, respectively. There is a small amount of microalgae in the inoculated activated sludge. During the first 15 days (Rs reactor), the microalgae were not attached to the sludge and easily effluented, causing Chl-a to decrease rapidly from 2743 µg L−1 to 2016 µg L−1. Rc slowly increased to 923 µg L−1 because it was not initially inoculated with microalgae, and the microalgae content in the sludge itself was very low, which affected microalgae biomass accumulation. The microalgae and activated sludge flocs with strong adsorption capacity tightly bound together to form a bacteria–microalgae symbiosis, ensuring microalgae were not flushed out of the reactor while still allowing for large-scale reproduction.
With the formation of symbionts, the flocs continued to grow, and the sludge wrapped part of the microalgae in the core, blocking light transmission and reducing the photosynthetic activity of the internal microalgae. Chl-a in the Rs reactor was reduced to 4526 µg L−1. A Chl-a concentration of microalgae below 4600 µg L−1 is optimal.86 Rc and Rs reactors' Chl-a reached 3454 and 4303 µg L−1. The Chl-a concentration during Rs formation was higher than that during Rc formation. The initial proliferation of Rc was slow due to its low microalgae content. Rs achieved rapid growth after adapting to the environment, due to its large initial microalgal biomass. It shows that the inoculation of microalgae to cultivate bacterial–algal symbionts is beneficial for accelerating microalgal enrichment.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). Rc relies on sunlight to induce spontaneous growth of microalgae. The accumulation of microalgae in the reactor is slow. Rs is directly inoculated with microalgae and activated sludge. During the reaction period, the symbiotic relationship between bacteria and microalgae gradually reaches equilibrium. During the first 15 days, the COD effluent of the two was relatively high, 237.6 and 273.8 mg L−1, respectively, which was likely to be caused by the adaptation period of activated sludge and the loss of biomass. COD removal efficiency reached more than 90% on the 40th and 25th day, respectively (COD removal load 36.14 and 36.10 mg L−1 h−1; effluent concentrations 51.1 and 75.8 mg L−1). In the second half of the test, the average COD removal efficiency for the two was 86.3% and 90.7%, respectively (effluent concentrations of 108.8 and 73.5 mg L−1). Rs reached the second level of urban sewage discharge standard (COD < 100 mg L−1), the average removal load was 34.45, 36.22 mg L−1 h−1. Qunying and Yushu87 indicated that the COD degradation rate of the traditional activated sludge method is about 73–80%, which showed that the bacteria and microalgae symbionts performed excellently in removing organic matter. In the stable removal stage, microalgae concentration in Rc and Rs: Rc < Rs. The high concentration of inoculated microalgae in Rs facilitates rapid enrichment of subsequent microalgae, promotes bacterial and algal cooperation, provides more DO for sludge, and enhances sludge-mediated COD degradation. Roudsari et al.51 found that bacterial growth is the main driving force for COD removal, and its removal capacity was much greater than that of microalgae (the proportion of microalgae increased, and the COD removal effect improved, indicating that microalgae had a positive impact on the ability of activated sludge to remove COD). Mineralization by heterotrophic bacteria plays a significant role in COD removal. Microalgae play an auxiliary role in the COD removal process, as reflected in two aspects. Firstly, in a eutrophic environment, microalgae absorb CO2 generated by sludge respiration during the day and perform photosynthesis, providing O2 and various organic substances to the sludge. The mutually beneficial symbiotic relationship between the two improves the O2 utilization rate during gas exchange and enhances sludge activity. Secondly, photosynthesis also stops at night when the light stops. Microalgae assimilate organic matter for heterotrophic reproduction (consuming part of COD).74
1). Rc relies on sunlight to induce spontaneous growth of microalgae. The accumulation of microalgae in the reactor is slow. Rs is directly inoculated with microalgae and activated sludge. During the reaction period, the symbiotic relationship between bacteria and microalgae gradually reaches equilibrium. During the first 15 days, the COD effluent of the two was relatively high, 237.6 and 273.8 mg L−1, respectively, which was likely to be caused by the adaptation period of activated sludge and the loss of biomass. COD removal efficiency reached more than 90% on the 40th and 25th day, respectively (COD removal load 36.14 and 36.10 mg L−1 h−1; effluent concentrations 51.1 and 75.8 mg L−1). In the second half of the test, the average COD removal efficiency for the two was 86.3% and 90.7%, respectively (effluent concentrations of 108.8 and 73.5 mg L−1). Rs reached the second level of urban sewage discharge standard (COD < 100 mg L−1), the average removal load was 34.45, 36.22 mg L−1 h−1. Qunying and Yushu87 indicated that the COD degradation rate of the traditional activated sludge method is about 73–80%, which showed that the bacteria and microalgae symbionts performed excellently in removing organic matter. In the stable removal stage, microalgae concentration in Rc and Rs: Rc < Rs. The high concentration of inoculated microalgae in Rs facilitates rapid enrichment of subsequent microalgae, promotes bacterial and algal cooperation, provides more DO for sludge, and enhances sludge-mediated COD degradation. Roudsari et al.51 found that bacterial growth is the main driving force for COD removal, and its removal capacity was much greater than that of microalgae (the proportion of microalgae increased, and the COD removal effect improved, indicating that microalgae had a positive impact on the ability of activated sludge to remove COD). Mineralization by heterotrophic bacteria plays a significant role in COD removal. Microalgae play an auxiliary role in the COD removal process, as reflected in two aspects. Firstly, in a eutrophic environment, microalgae absorb CO2 generated by sludge respiration during the day and perform photosynthesis, providing O2 and various organic substances to the sludge. The mutually beneficial symbiotic relationship between the two improves the O2 utilization rate during gas exchange and enhances sludge activity. Secondly, photosynthesis also stops at night when the light stops. Microalgae assimilate organic matter for heterotrophic reproduction (consuming part of COD).74NH4+-N removal by the reactor first increased, then remained stable, and later fluctuated at a high level, similar to the overall trend in COD removal (Fig. 5d and e). On the 20th day, the rapid increase in NH4+-N removal efficiency may be due to microbial biomass growth in the reactor. With increase in the biomass in the reactor, the NH4+-N removal effect in the Rs reactor was relatively ideal and stable, at 93.1% (effluent NH4+-N < 10 mg L−1), meeting the secondary discharge requirements for urban sewage. Average ARR in the stable period was 8.53 mg L−1 h−1. This was similar to the results reported by Arun et al.:88 a removal load of 8.92 mg L−1 h−1 was achieved with an NH4+-N concentration of 100 mg L−1 at 12 h HRT. Marcilhac et al.89 showed that the maximum nitrogen removal efficiency (8.5 mg L−1 d−1) was detected under the light intensity of 244 µmol m−2 s−1. The removal efficiency of Rc exceeded 90% on the 60th day, but there were significant fluctuations. ARR was 8.31 mg L−1 h−1 at the end of the test. The variation trends of NO2−-N and NO3−-N in the Rc and Rs reactors are similar. Both reactors were under light conditions, and nitrite oxidation was inhibited.39 Microalgae in Rc were not enriched, and light inhibition completely affected bacteria, resulting in a higher NO2−-N concentration in Rc than in Rs. However, NO2−-N concentration dropped to below 0.3 mg L−1 in about 25 days, while the NO3−-N concentration gradually increased to 23.5 and 25.4 mg L−1, which indicated that with the growth of microalgae, the photoinhibition effect of nitrifying bacteria was reduced. After the symbiont formation period, NO2−-N slowly increased to about 1 mg L−1, while NO3−-N concentration decreased slowly and stabilized at 12.2 and 11.4 mg L−1. It showed that denitrification occurred in the system. As symbiotic flocs gradually increase, the anaerobic space formed within also becomes larger and more stable, providing favorable conditions for anaerobic bacteria such as denitrifying bacteria and improving their activity, thereby enhancing denitrification capacity. There are three main ways to remove nitrogen in the symbiotic system of bacteria and microalgae: assimilation, nitrification, and denitrification. Assimilation means that microalgae absorb combined nitrogen, such as NH4+-N, NO2−-N, and NO3−-N into nitrogen metabolism and finally synthesize amino acids to support their continuous proliferation. During the dark stage, microalgae and bacteria simultaneously consume system DO, accelerating the formation of an anoxic environment and promoting denitrification.
As shown in Fig. 5f, the PO43−-P removal by Rc and Rs was consistent with the trends in COD and NH4+-N. Overall, the efficiency of the Rc reactor was lower than that of Rs, consistent with the results for COD and NH4+-N. Rc relies on sunlight to induce the spontaneous growth of microalgae, resulting in a difference in biomass concentration (a lag period). In the later stage, the average PO43−-P removal efficiency in the Rc and Rs systems was 87.1% and 90.9%, respectively, and the average volume loads were 0.42 and 0.44 mg L−1 h−1, which were close to those reported by Rezvani et al.90 The results showed that the microalgae–bacteria symbiotic system could achieve a removal efficiency of 10 mg per L PO43−-P of 6.34 mg L−1 d−1 in 6 days. Higher than that of monoculture microalgae (4.5 mg L−1 d−1) and bacteria (2.6 mg L−1 d−1). The removal of P by the system primarily includes biological and chemical phosphorus removal. Phosphorus removal by chemical precipitation mainly occurred at pH 9–11. The pH was controlled at 7–8 during the test, and the reactor's phosphorus removal mechanism was mainly biological. Phosphorus-accumulating bacteria (PAOs) and microalgae in sludge contributed to phosphorus removal. Second, microalgae can absorb phosphorus, assimilate it, and over-absorb it. Assimilate PO43−-P to synthesize the required phosphorus-containing organic matter such as phospholipids and ATP, thus completing their own reproduction.25 Similar to PAO under eutrophic conditions, microalgae can also over-absorb phosphorus, synthesize polyphosphates, and store them in their bodies for metabolism. Their phosphorus uptake was higher than that of bacteria of the same biomass.70
Nitrifying bacteria in the Nitrospira can oxidize nitrite to nitrate. Its abundance decreased to 2.36% and 1.92% in the two reactors, respectively, indicating that microalgae growth inhibited Nitrospira. Flavobacteria and Cytophagia (Bacteroidetes phylum) gradually became the dominant bacterial classes in the bacteria–microalgae reactor. The abundance of Cyanobacteria also increased significantly, especially in the Rc system, reaching 16.1%. Flavobacterium has a strong tendency to self-aggregate, and its increased abundance enhances integration between bacteria and microalgae. Cytophagia (Bacteroidetes phylum) degrade macromolecular organic matter.92 Sphingobacteria (Bacteroidetes phylum) remove nitrogen and phosphorus.100 It also includes filamentous bacteria, which easily lead to sludge bulking,18 as reported by Meng et al.,39 who believed that microalgae are detrimental to Sphingobacteria reproduction. However, its abundance was lower in both sets of reactors. Cyanobacteria are photosynthetic prokaryotes whose growth is driven by light. Its abundance in the Rs system was significantly lower than that of Rc. Chlorophyceae growth limits the overgrowth of Cyanobacteria. The phylum Bacteroidetes degrades cyanobacteria, preventing their massive growth and associated toxicity. The presence of Verrucomicrobia was positively correlated with environmental phosphorus content,92 suggesting lower Verrucomicrobia abundance in the two sets of reactors indicating higher phosphorus removal efficiency. Kazamia et al.101 found that Chloroflexia was closely related to phosphorus removal from activated sludge. Notably, the detected Anaerolineae within the Chloroflexia phylum are facultative anaerobes, indicating an anaerobic zone in the reactor. Similar to granular sludge, there may be an anaerobic space in the centre of the bacterial–microalgae symbiotic floc. Additional microalgae dosing altered the bacterial community abundance in the system, and spontaneous microalgae growth also affected the microbial community. Microalgae and bacteria interacted and selected each other, forming a symbiotic system that influenced their performance and stability.
Although high dissolved oxygen levels promote bacterial activity, they limit microalgae photosynthesis. CO2 escape reduces the inorganic carbon available to microalgae and inhibits their activity. The utilization efficiency of bacteria for O2 produced by microalgae is higher than that of external mechanical aeration. Therefore, additional oxygen supply is an energy-consuming and limiting factor for the microalgae's growth. The aeration intensity should be appropriately reduced to promote bacterial and microalgal cooperation and maintain system stability. Light promotes microalgae growth, but excessive light can also have adverse effects. Exposure of microalgae to excessive light leads to the formation of harmful ROS and oxidative stress. Microalgae will self-regulate and emit up to 80% of the photons, thereby reducing light-use efficiency. Light also directly affects bacteria, such as AOB and NOB, thereby influencing nitrification. Therefore, the selection of various parameters should be comprehensively considered to maintain the growth balance between bacteria and algae, ensure the system's stable operation, and ultimately achieve effective nitrogen and phosphorus removal.
4. Conclusions
This study aims to enhance the removal of carbon, nitrogen, and phosphorus and to ensure the system's stable operation by establishing an environment conducive to the symbiotic relationship between bacteria and microalgae and to the formation of symbiotic flocs of bacteria and algae. Gradient inoculation experiments revealed a nonlinear relationship between the microalgae–bacterial biomass ratio (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 to 8
4 to 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) and pollutant removal efficiency. The study found that the optimal inoculation ratio (4
1) and pollutant removal efficiency. The study found that the optimal inoculation ratio (4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 algae/sludge) achieved the highest pollutant removal efficiency, with peak chlorophyll a, PN, and total biomass concentrations. This promoted the synergistic growth of bacteria and algae, improved the biomass settleability, and reduced effluent turbidity. This study proposed an optimal ratio for bacterial–microalgae symbiosis, providing a design basis for photobioreactor construction. The operating parameters of aeration and lighting conditions within the multi-parameter coupled control system were optimized by constructing a bacterial–algal symbiotic PBR system. Furthermore, single-factor experiments were conducted to analyze the interactive effects of light intensity (54–180 µmol m−2 s−1) and aeration rate (0–1.0 L min−1) to determine the optimal operating conditions. Analysis of bacterial–algal biomass and Chlorella vulgaris activity revealed a synergistic mechanism: higher dissolved oxygen levels inhibited microalgae photosynthesis and activity, promoted bacterial growth, and allowed CO2 to escape, disrupting the synergistic balance between the biological relationship and physical structure between microalgae and bacteria. The addition of microalgae facilitated biomass accumulation and accelerated the establishment of the symbiotic process, whereas the reactor formed using spontaneous light-induced microalgae generation in sludge was slower. The pollutant removal rate in the artificial system was higher than in the natural enrichment system. This study provides new ideas for achieving highly effective low consumption green biological landfill leachate treatment.
1 algae/sludge) achieved the highest pollutant removal efficiency, with peak chlorophyll a, PN, and total biomass concentrations. This promoted the synergistic growth of bacteria and algae, improved the biomass settleability, and reduced effluent turbidity. This study proposed an optimal ratio for bacterial–microalgae symbiosis, providing a design basis for photobioreactor construction. The operating parameters of aeration and lighting conditions within the multi-parameter coupled control system were optimized by constructing a bacterial–algal symbiotic PBR system. Furthermore, single-factor experiments were conducted to analyze the interactive effects of light intensity (54–180 µmol m−2 s−1) and aeration rate (0–1.0 L min−1) to determine the optimal operating conditions. Analysis of bacterial–algal biomass and Chlorella vulgaris activity revealed a synergistic mechanism: higher dissolved oxygen levels inhibited microalgae photosynthesis and activity, promoted bacterial growth, and allowed CO2 to escape, disrupting the synergistic balance between the biological relationship and physical structure between microalgae and bacteria. The addition of microalgae facilitated biomass accumulation and accelerated the establishment of the symbiotic process, whereas the reactor formed using spontaneous light-induced microalgae generation in sludge was slower. The pollutant removal rate in the artificial system was higher than in the natural enrichment system. This study provides new ideas for achieving highly effective low consumption green biological landfill leachate treatment.
Conflicts of interest
There are no conflicts to declare.Data availability
The authors confirm that the data supporting the findings of this study are available within the article.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d6ra02058e.
Acknowledgements
The authors thank the School of Municipal and Environmental Engineering, Shenyang Jianzhu University, for providing the research facilities. The program for Liaoning Innovative Research Team in University-LNIRT supports this research. Dr Tabassum is also thankful to the Department of Chemistry, Sakarya University.References
- A. Irto, F. Crea, C. Alessandrello, C. De Stefano, R. Somma and G. Zaffino, et al., Landfill leachate from Municipal Solid Waste: Multi-technique approach for its fine characterization and determination of the thermodynamic and sequestering properties towards some toxic metals, Sci. Total Environ., 2024, 917, 170311 CrossRef CAS PubMed.
- L. Zhang, J. Han and A. Shengji ea, Pollution and removal of organic pollutants in landfill leachate, China Environ. Sci., 1998, 02, 184–188 Search PubMed.
- L. Lindamulla, N. Nanayakkara, M. Othman, S. Jinadasa, G. Herath and V. Jegatheesan, Municipal solid waste landfill leachate characteristics and their treatment options in tropical countries, Curr. Pollut. Rep., 2022, 8(3), 273–287 CrossRef CAS.
- T. Nawaz, A. Rahman, S. Pan, K. Dixon, B. Petri and T. Selvaratnam, A review of landfill leachate treatment by microalgae: Current status and future directions, Processes, 2020, 8(4), 384 CrossRef CAS.
- I. Dogaris, E. Ammar and G. P. Philippidis, Prospects of integrating algae technologies into landfill leachate treatment, World J. Microbiol. Biotechnol., 2020, 36(3), 39 CrossRef CAS PubMed.
- L. Jiang, C. Niu, L. Chen, L. Jia, Z. Yan and Z. Zhuo, et al., Engineering practice of two-stage DTRO process for treating landfill leachate, Environ. Sci. Technol., 2020, 26(04), 13–7+30 Search PubMed.
- I. K. Erabee, A. Ahsan, B. Jose, M. M. A. Aziz, A. W. M. Ng and S. Idrus, et al., Adsorptive Treatment of Landfill Leachate using Activated Carbon Modified with Three Different Methods, KSCE J. Civ. Eng., 2018, 22(4), 1083–1095 CrossRef.
- M. S. Yusoff, H. A. Aziz, M. F. M. A. Zamri, A. Z. Abdullah and N. E. A. Basri, Floc behavior and removal mechanisms of cross-linked Durio zibethinus seed starch as a natural flocculant for landfill leachate coagulation-flocculation treatment, Waste Manage., 2018, 74, 362–372 CrossRef CAS PubMed.
- J. Liu, X. Qiu, Q. Shi, L. Dong, Y. Yang and S. Chunlong, Pilot study on treatment of landfill leachate by modified mineral adsorption method, Guangdong Chem. Ind., 2019, 46(6), 68–70 Search PubMed.
- G. Yali, Analysis of Landfill Leachate Treatment Process in a Certain City, Chang'an University, China, 2015 Search PubMed.
- J. Zhu, W. Xu, Y. Yang, X. Su, X. Xiao and F. Dong, et al., Quorum quenching driven microbial community to biofouling control in membrane bioreactor for landfill leachate treatment, J. Membr. Sci., 2025, 722, 123899 CrossRef CAS.
- F. Cha, M. Zhang, G. Yan and Y. Fang, Study on treatment of chemical waste leachate by biological ponds, Water Treat. Technol., 2010, 36(11), 104–6+12 Search PubMed.
- J. Boonnorat, R. Honda, P. Panichnumsin, N. Boonapatcharoen, N. Yenjam and C. Krasaesueb, et al., Treatment efficiency and greenhouse gas emissions of non-floating and floating bed activated sludge system with acclimatized sludge treating landfill leachate, Bioresour. Technol., 2021, 330, 124952 CrossRef CAS PubMed.
- H. Yao, L. Zhang and L. Shugeng ea, Study on influencing factors of anaerobic iron-ammonia oxidation treatment of simulated landfill leachate, J. Environ. Sci., 2019, 39(09), 2953–2963 CAS.
- F. Sun, W. Zhang, G. Jiang, Y. Liu, S. Wu and D. Wu, et al., Effective biological nitrogen process and nitrous oxide emission characteristics for the treatment of landfill leachate with low carbon-to-nitrogen ratio, J. Clean. Prod., 2020, 268, 122289 CrossRef CAS.
- R. Katiyar, B. R. Gurjar, S. Biswas, V. Pruthi, N. Kumar and P. Kumar, Microalgae: An emerging source of energy based bio-products and a solution for environmental issues, Renew. Sustain. Energy Rev., 2017, 72, 1083–1093 CrossRef CAS.
- H. Chang, Q. Fu, N. Zhong, X. Yang, X. Quan and S. Li, et al., Microalgal lipids production and nutrients recovery from landfill leachate using membrane photobioreactor, Bioresour. Technol., 2019, 277, 18–26 CrossRef CAS PubMed.
- Z. Bing, Formation Mechanism of Aerobic Granular Sludge with Bacterial and Algal Symbiosis and its Enhancement Mechanism Based on QS, Harbin Institute of Technology, China, 2020 Search PubMed.
- R. Gutiérrez, I. Ferrer, E. Uggetti, C. Arnabat, H. Salvado and J. García, Settling velocity distribution of microalgal biomass from urban wastewater treatment high rate algal ponds, Algal Res., 2016, 16, 409–417 CrossRef.
- E. Vulsteke, S. Van Den Hende, L. Bourez, H. Capoen, D. P. L. Rousseau and J. Albrecht, Economic feasibility of microalgal bacterial floc production for wastewater treatment and biomass valorization: a detailed up-to-date analysis of up-scaled pilot results, Bioresour. Technol., 2017, 224, 118–129 CrossRef CAS PubMed.
- S. Huasheng, Effects of Bacterial-Algal Symbiosis on the Aggregation Behavior of Unicellular Microalgae in Aquatic Environments, Hefei University of Technology, China, 2019 Search PubMed.
- S. Fan, L. Zhu and B. Ji, Deciphering the effect of light intensity on microalgal-bacterial granular sludge process for non-aerated municipal wastewater treatment, Algal Res., 2021, 58, 102437 CrossRef.
- C. González, J. Marciniak, S. Villaverde, C. Leon, P. A. García and R. Munoz, Efficient nutrient removal from swine manure in a tubular biofilm photo-bioreactor using algae-bacteria consortia, Water Sci. Technol., 2008, 58(1), 95–102 CrossRef PubMed.
- Y. Gao, L. Guo, Q. Liao, Z. Zhang, Y. Zhao and M. Gao, et al., Mariculture wastewater treatment with Bacterial-Algal Coupling System (BACS): Effect of light intensity on microalgal biomass production and nutrient removal, Environ. Res., 2021, 201, 111578 CrossRef CAS PubMed.
- M. E. Martinez, J. M. Jiménez and F. El Yousfi, Influence of phosphorus concentration and temperature on growth and phosphorus uptake by the microalga Scenedesmus obliquus, Bioresour. Technol., 1999, 67(3), 233–240 CrossRef CAS.
- N. C. Boelee, H. Temmink, M. Janssen, C. J. N. Buisman and R. H. Wijffels, Balancing the organic load and light supply in symbiotic microalgal–bacterial biofilm reactors treating synthetic municipal wastewater, Ecol. Eng., 2014, 64, 213–221 CrossRef.
- J. J. Schmidt, G. A. Gagnon and R. C. Jamieson, Microalgae growth and phosphorus uptake in wastewater under simulated cold region conditions, Ecol. Eng., 2016, 95, 588–593 CrossRef.
- E. Segev, T. P. Wyche, K. H. Kim, J. Petersen, C. Ellebrandt and H. Vlamakis, et al., Dynamic metabolic exchange governs a marine algal-bacterial interaction, eLife, 2016, 5, e17473 CrossRef PubMed.
- Z. Song, Q. Wang, L. Li, Y. Gao and H. Zhao, Optimization of culture medium for Scenedesmus obliquus and its symbiotic system for wastewater treatment., J. Heilongjiang Univ. Sci. Technol., 2015, 3, 1–6 CrossRef.
- K. Jinda, T. Koottatep, C. Chaiwong and C. Polprasert, Performance evaluation of novel attached-growth high rate algal pond system with additional artificial illumination for wastewater treatment and nutrient recovery, Water Sci. Technol., 2020, 82(1), 97–106 Search PubMed.
- G. Mujtaba, M. Rizwan and K. Lee, Removal of nutrients and COD from wastewater using symbiotic co-culture of bacterium Pseudomonas putida and immobilized microalga Chlorella vulgaris, J. Ind. Eng. Chem., 2017, 49, 145–151 Search PubMed.
- X. Hu, Y. E. Meneses and A. A. Hassan, Integration of sodium hypochlorite pretreatment with co-immobilized microalgae/bacteria treatment of meat processing wastewater, Bioresour. Technol., 2020, 304, 122953 CrossRef CAS PubMed.
- I. De Godos, C. González, E. Becares, P. A. García-Encina and R. Muñoz, Simultaneous nutrients and carbon removal during pretreated swine slurry degradation in a tubular biofilm photobioreactor, Appl. Microbiol. Biotechnol., 2009, 82(1), 187–194 Search PubMed.
- Q. Yan, X. Gao and P. Xuya, Advanced treatment of sewage treatment plant effluent by immobilized bacteria-algae system, Chin. J. Environ. Eng., 2012, 6(10), 3629–3634 CAS.
- D. Zhang, Z. Yongsheng and P. Xiangliang, Role of extracellular polymeric substances (EPS) in the removal of Cd from wastewater by algae-bacteria biofilms, Res. Environ. Sci., 2004, 17(5), 52–55 Search PubMed.
- R. Du, S. Cao, B. Li, M. Niu, S. Wang and Y. Peng, Performance and microbial community analysis of a novel DEAMOX based on partial-denitrification and anammox treating ammonia and nitrate wastewaters, Water Res., 2017, 108, 46–56 Search PubMed.
- T. T. Nguyen, Q. A. Binh, X. T. Bui, H. H. Ngo, H. N. P. Vo and K. Y. A. Lin, et al., Co-culture of microalgae-activated sludge for wastewater treatment and biomass production: exploring their role under different inoculation ratios, Bioresour. Technol., 2020, 314, 123754 CrossRef CAS PubMed.
- V. Ashok, A. Shriwastav, P. Bose and S. K. Gupta, Phycoremediation of wastewater using algal-bacterial photobioreactor: effect of nutrient load and light intensity, Bioresour. Technol. Rep., 2019, 7, 100205 CrossRef.
- F. Meng, L. Xi, D. Liu, W. Huang, Z. Lei and Z. Zhang, et al., Effects of light intensity on oxygen distribution, lipid production and biological community of algal-bacterial granules in photo-sequencing batch reactors, Bioresour. Technol., 2019, 272, 473–481 Search PubMed.
- C. C. Tang, W. Zuo, Y. Tian, N. Sun, Z. W. Wang and J. Zhang, Effect of aeration rate on performance and stability of algal-bacterial symbiosis system to treat domestic wastewater in sequencing batch reactors, Bioresour. Technol., 2016, 222, 156–164 CrossRef CAS PubMed.
- D. Manhaeghe, S. Michels, D. P. L. Rousseau and S. W. H. Van Hulle, A semi-mechanistic model describing the influence of light and temperature on the respiration and photosynthetic growth of Chlorella vulgaris, Bioresour. Technol., 2019, 274, 361–370 CrossRef CAS PubMed.
- Z. Liang, Y. Liu, F. Ge, Y. Xu, N. Tao and F. Peng, et al., Efficiency assessment and pH effect in removing nitrogen and phosphorus by algae-bacteria combined system of Chlorella vulgaris and Bacillus licheniformis, Chemosphere, 2013, 92(10), 1383–1389 CrossRef CAS PubMed.
- H. Pan, Z. Xiong and S. Wei, Study on the removal of nitrogen and phosphorus from municipal wastewater by co-immobilized bacteria and algae, Environ. Sci. Technol., 2006, 29(1), 14–15 Search PubMed.
- The editorial board of State Bureau of Environmental Protection, Water and Wastewater Monitoring Analysis Method, China Environmental Science Press, Beijing, 4th edn, 2002, vol. 448, pp. 88–439 Search PubMed.
- J. W. Morgan, C. F. Forster and L. Evison, A comparative study of the nature of biopolymers extracted from anaerobic and activated sludges, Water Res., 1990, 24(6), 743–750 CrossRef CAS.
- A. F. Gaudy, Colorimetric determination of protein and carbohydrate, Ind. Water Wastes, 1962, 7, 17–22 CAS.
- J. J. Sedmak and S. E. Grossberg, A rapid, sensitive, and versatile assay for protein using Coomassie brilliant blue G250, Anal. Biochem., 1977, 79(1–2), 544–552 CrossRef CAS PubMed.
- S. Ni, Efficiency and Characteristics of Bacterial-Algal Symbiotic SBR in Treating Domestic Sewage, Harbin Institute of Technology, Harbin, 2016 Search PubMed.
- Laboratory measurement: Chlorophyll-a concentration measurement with acetone method using spectrophotometer, 2014 IEEE International Conference on Industrial Engineering and Engineering Management, ed. F. Johan, M. Z. Jafri, H. S. Lim and W. O. W. Maznah, 2014 Search PubMed.
- Q. Yuntian, Study on Microwave Enhancement of Algae Oxygen Production in Municipal Sewage Culture Algae System, Harbin Institute of Technology, China, 2015 Search PubMed.
- F. P. Roudsari, M. R. Mehrnia, A. Asadi, Z. Moayedi and R. Ranjbar, Effect of microalgae/activated sludge ratio on cooperative treatment of anaerobic effluent of municipal wastewater, Appl. Biochem. Biotechnol., 2014, 172(1), 131–140 CrossRef CAS PubMed.
- L. Wang, M. Min, Y. Li, P. Chen, Y. Chen and Y. Liu, et al., Cultivation of green algae Chlorella sp. in different wastewaters from municipal wastewater treatment plant, Appl. Biochem. Biotechnol., 2010, 162(4), 1174–1186 CrossRef CAS PubMed.
- L. Sun, W. Zuo, Y. Tian, J. Zhang, J. Liu and N. Sun, et al., Performance and microbial community analysis of an algal-activated sludge symbiotic system: effect of activated sludge concentration, J. Environ. Sci., 2019, 76, 121–132 CrossRef CAS PubMed.
- B. Guieysse, X. Borde, R. Muñoz, R. Hatti-Kaul, C. Nugier-Chauvin and H. Patin, et al., Influence of the initial composition of algal-bacterial microcosms on the degradation of salicylate in a fed-batch culture, Biotechnol. Lett., 2002, 24(7), 531–538 CrossRef CAS.
- Y. Su, A. Mennerich and B. Urban, Synergistic cooperation between wastewater-born algae and activated sludge for wastewater treatment: influence of algae and sludge inoculation ratios, Bioresour. Technol., 2012, 105, 67–73 CrossRef CAS PubMed.
- R. Muñoz and B. Guieysse, Algal–bacterial processes for the treatment of hazardous contaminants: a review, Water Res., 2006, 40(15), 2799–2815 CrossRef PubMed.
- B. T. Higgins, I. Gennity, P. S. Fitzgerald, S. J. Ceballos, O. Fiehn and J. S. VanderGheynst, Algal–bacterial synergy in treatment of winery wastewater, npj Clean Water, 2018, 1(1), 6 CrossRef CAS.
- D. Vandamme, I. Foubert and K. Muylaert, Flocculation as a low-cost method for harvesting microalgae for bulk biomass production, Trends Biotechnol., 2013, 31(4), 233–239 CrossRef CAS PubMed.
- G. Gutzeit, D. Lorch, A. Weber, M. Engels and U. Neis, Bioflocculent algal–bacterial biomass improves low-cost wastewater treatment, Water Sci. Technol., 2005, 52(12), 9–18 CrossRef CAS PubMed.
- M. F. Dignac, V. Urbain, D. Rybacki, A. Bruchet, D. Snidaro and P. Scribe, Chemical description of extracellular polymers: implication on activated sludge floc structure, Water Sci. Technol., 1998, 38(8–9), 45–53 CrossRef CAS.
- B. Chen, F. Li, N. Liu, F. Ge, H. Xiao and Y. Yang, Role of extracellular polymeric substances from Chlorella vulgaris in the removal of ammonium and orthophosphate under the stress of cadmium, Bioresour. Technol., 2015, 190, 299–306 CrossRef CAS PubMed.
- G. Chen, N. Zhu, Z. Tang, P. Ye, Z. Hu and L. Liu, Resource availability shapes microbial motility and mediates early-stage formation of microbial clusters in biological wastewater treatment processes, Appl. Microbiol. Biotechnol., 2014, 98(3), 1459–1467 CrossRef CAS PubMed.
- B. Ji, S. Wang, M. R. U. Silva, M. Zhang and Y. Liu, Microalgal-bacterial granular sludge for municipal wastewater treatment under simulated natural diel cycles: Performances-metabolic pathways-microbial community nexus, Algal Res., 2021, 54, 102198 CrossRef.
- A. Ding, W. Pronk, F. Qu, J. Ma, G. Li and K. Li, et al., Effect of calcium addition on sludge properties and membrane fouling potential of the membrane-coupled expanded granular sludge bed process, J. Membr. Sci., 2015, 489, 55–63 CrossRef CAS.
- M. Wang, H. Yang, S. J. Ergas and P. van der Steen, A novel shortcut nitrogen removal process using an algal-bacterial consortium in a photo-sequencing batch reactor (PSBR), Water Res., 2015, 87, 38–48 CrossRef CAS PubMed.
- E. Posadas, M. del Mar Morales, C. Gomez, F. G. Acién and R. Muñoz, Influence of pH and CO2 source on the performance of microalgae-based secondary domestic wastewater treatment in outdoors pilot raceways, Chem. Eng. J., 2015, 265, 239–248 CrossRef CAS.
- H. Zhang, W. Gong, L. Bai, R. Chen, W. Zeng and Z. Yan, et al., Aeration-induced CO2 stripping, instead of high dissolved oxygen, have a negative impact on algae–bacteria symbiosis (ABS) system stability and wastewater treatment efficiency, Chem. Eng. J., 2020, 382, 122957 CrossRef CAS.
- B. Ji, M. Zhang, L. Wang, S. Wang and Y. Liu, Removal mechanisms of phosphorus in non-aerated microalgal-bacterial granular sludge process, Bioresour. Technol., 2020, 312, 123531 CrossRef CAS PubMed.
- J. L. Mouget, A. Dakhama, M. C. Lavoie and J. de la Noüe, Algal growth enhancement by bacteria: is consumption of photosynthetic oxygen involved?, FEMS Microbiol. Ecol., 1995, 18(1), 35–43 CrossRef CAS.
- L. Liu, H. Fan, Y. Liu, C. Liu and X. Huang, Development of algae-bacteria granular consortia in photo-sequencing batch reactor, Bioresour. Technol., 2017, 232, 64–71 CrossRef CAS PubMed.
- J. U. Grobbelaar and C. J. Soeder, Respiration losses in planktonic green algae cultivated in raceway ponds, J. Plankton Res., 1985, 7(4), 497–506 CrossRef.
- Z. Li, S. Wakao, B. B. Fischer and K. K. Niyogi, Sensing and responding to excess light, Annu. Rev. Plant Biol., 2009, 60, 239–260 CrossRef CAS PubMed.
- C. S. Lee, S. A. Lee, S. R. Ko, H. M. Oh and C. Y. Ahn, Effects of photoperiod on nutrient removal, biomass production, and algal-bacterial population dynamics in lab-scale photobioreactors treating municipal wastewater, Water Res., 2015, 68, 680–691 CrossRef CAS PubMed.
- D. M. Eny, Respiration studies on Chlorella. II. Influence of various organic acids on gas exchange, Plant Physiol., 1951, 26(2), 268 CrossRef CAS PubMed.
- J. E. Alleman, V. Keramida and L. Pantea-Kiser, Light induced Nitrosomonas inhibition, Water Res., 1987, 21(4), 499–501 CrossRef CAS.
- F. Lipschultz, S. C. Wofsy and L. E. Fox, The effects of light and nutrients on rates of ammonium transformation in a eutrophic river, Mar. Chem., 1985, 16(4), 329–341 CrossRef CAS.
- S. N. Merbt, D. A. Stahl, E. O. Casamayor, E. Martí, G. W. Nicol and J. I. Prosser, Differential photoinhibition of bacterial and archaeal ammonia oxidation, FEMS Microbiol. Lett., 2012, 327(1), 41–46 CrossRef CAS PubMed.
- S. Akizuki, M. Kishi, G. Cuevas-Rodríguez and T. Toda, Effects of different light conditions on ammonium removal in a consortium of microalgae and partial nitrifying granules, Water Res., 2020, 171, 115445 CrossRef CAS PubMed.
- W. Huang, B. Li, C. Zhang, Z. Zhang, Z. Lei and B. Lu, et al., Effect of algae growth on aerobic granulation and nutrients removal from synthetic wastewater by using sequencing batch reactors, Bioresour. Technol., 2015, 179, 187–192 CrossRef CAS PubMed.
- E. Sforza, D. Simionato, G. M. Giacometti, A. Bertucco and T. Morosinotto, Adjusted light and dark cycles can optimize photosynthetic efficiency in algae growing in photobioreactors, PLoS One, 2012, 7(6), e38975 CrossRef CAS PubMed.
- H. Jia and Q. Yuan, Ammonium removal using algae–bacteria consortia: the effect of ammonium concentration, algae biomass, and light, Biodegradation, 2018, 29(2), 105–115 CrossRef CAS PubMed.
- S. H. Ho, C. Y. Chen and J. S. Chang, Effect of light intensity and nitrogen starvation on CO2 fixation and lipid/carbohydrate production of an indigenous microalga Scenedesmus obliquus CNW-N, Bioresour. Technol., 2012, 113, 244–252 CrossRef CAS PubMed.
- D. J. Lee, Y. Y. Chen, K. Y. Show, C. G. Whiteley and J. H. Tay, Advances in aerobic granule formation and granule stability in the course of storage and reactor operation, Biotechnol. Adv., 2010, 28(6), 919–934 CrossRef CAS PubMed.
- Y. Liu and J. H. Tay, State of the art of biogranulation technology for wastewater treatment, Biotechnol. Adv., 2004, 22(7), 533–563 CrossRef CAS PubMed.
- A Simple in Vitro Fluorescence Method for Biomass Measurements in Algal Growth Inhibition Tests, ed. P. Mayer and N. Nyholm, (Abstract No. PWP187), Society of Environmental Toxicology and Chemistry, 1997 Search PubMed.
- J. Ye, J. Liang, L. Wang, G. Markou and Q. Jia, Operation optimization of a photo-sequencing batch reactor for wastewater treatment: study on influencing factors and impact on symbiotic microbial ecology, Bioresour. Technol., 2018, 252, 7–13 CrossRef CAS PubMed.
- Z. Qunying and W. Yushu, Application of batch activated sludge process (SBR process) in domestic sewage treatment, Water Purif. Technol., 2011, 30(06), 36–38 Search PubMed.
- S. Arun, S. Ramasamy and K. Pakshirajan, Mechanistic insights into nitrification by microalgae-bacterial consortia in a photo-sequencing batch reactor under different light intensities, J. Clean. Prod., 2021, 321, 128752 CrossRef CAS.
- C. Marcilhac, B. Sialve, A.-M. Pourcher, C. Ziebal, N. Bernet and F. Béline, Digestate color and light intensity affect nutrient removal and competition phenomena in a microalgal-bacterial ecosystem, Water Res., 2014, 64, 278–287 CrossRef CAS PubMed.
- F. Rezvani, M. H. Sarrafzadeh and H. M. Oh, Hydrogen producer microalgae in interaction with hydrogen consumer denitrifiers as a novel strategy for nitrate removal from groundwater and biomass production, Algal Res., 2020, 45, 101747 CrossRef.
- L. Meile, Study on the Role of Extracellular Proteins in Aerobic Sludge Granulation, Zhejiang University, 2013 Search PubMed.
- S. Li, Enhanced removal of carbon, nitrogen and phosphorus and membrane fouling mitigation mechanism in bacterial-algal symbiotic, MBR System, China, Harbin Institute of Technology, 2019 Search PubMed.
- M. Zhao, Study on the Enhanced Cultivation and Characteristics of Algae-Bacteria Granular Sludge, China, Shandong University, 2013 Search PubMed.
- L. Mei, Y. Julin and H. Haisheng ea, Growth and purification effect of microalgae in wastewater from Penaeus vannamei aquaculture, Chin. J. Appl. Environ. Biol., 2018, 24(04), 866–872 Search PubMed.
- J. Wang, H. Yang and F. Wang, Mixotrophic cultivation of microalgae for biodiesel production: status and prospects, Appl. Biochem. Biotechnol., 2014, 172(7), 3307–3329 CrossRef CAS PubMed.
- E. Isanta, T. Bezerra, I. Fernández, M. E. Suárez-Ojeda, J. Pérez and J. Carrera, Microbial community shifts on an anammox reactor after a temperature shock using 454-pyrosequencing analysis, Bioresour. Technol., 2015, 181, 207–213 CrossRef CAS PubMed.
- W. Zeng, A. Wang, J. Zhang, L. Zhang and Y. Peng, Enhanced biological phosphate removal from wastewater and clade-level population dynamics of “Candidatus Accumulibacter phosphatis” under free nitrous acid inhibition: linked with detoxication, Chem. Eng. J., 2016, 296, 234–242 CrossRef CAS.
- A. Rivadeneyra Torres, M. V. Martinez-Toledo, A. Gonzalez-Martinez, J. Gonzalez-Lopez, D. Martin-Ramos and M. A. Rivadeneyra, Precipitation of carbonates by bacteria isolated from wastewater samples collected in a conventional wastewater treatment plant, Int. J. Environ. Sci. Technol., 2013, 10(1), 141–150 CrossRef CAS.
- X. Chen, Z. Hu, Y. Qi, C. Song and G. Chen, The interactions of algae-activated sludge symbiotic system and its effects on wastewater treatment and lipid accumulation, Bioresour. Technol., 2019, 292, 122017 CrossRef CAS PubMed.
- C. Zhongjun, Z. Haiqin, W. Yaoqi, Y. Xuliang, W. Jianfang and C. Yaoliang, Analysis of bacterial community characteristics in each compartment of ABR anaerobic ammonium oxidation reactor based on high-throughput sequencing, Environ. Sci., 2016, 7, 2652–2658 Search PubMed.
- E. Kazamia, D. C. Aldridge and A. G. Smith, Synthetic ecology–a way forward for sustainable algal biofuel production?, J. Biotechnol., 2012, 162(1), 163–169 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |



