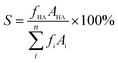Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Efficient selective hydrogenation of N,N-dimethylaniline in a continuous fixed-bed reactor over a Cu/Ni–Al2O3 catalyst
Jingdong Nong ,
Zhonghua Sun*,
Weiyou Zhou
,
Zhonghua Sun*,
Weiyou Zhou ,
Zhong Wu,
Junfeng Qian
,
Zhong Wu,
Junfeng Qian ,
Qun Chen and
Mingyang He
,
Qun Chen and
Mingyang He
Jiangsu Key Laboratory of Advanced Catalytic Materials and Technology, Changzhou University, 213164 Changzhou, China. E-mail: sunzhonghua@cczu.edu.cn
First published on 13th April 2026
Abstract
A series of Cu-modified Ni–Al2O3 catalysts with various Cu/Ni ratios (xCu/Ni–Al2O3) were prepared, and systematically characterized using BET, XRD, SEM, H2-TPR, H2-TPD, TG, and XPS. The catalyst exhibited excellent catalytic performance in the selective hydrogenation of N,N-dimethylaniline to N,N-dimethylcyclohexylamine in a continuous fixed-bed reactor. When the Cu/Ni molar ratio was 0.1, the active components were highly dispersed on the Al2O3 support, which possessed a high specific surface area and well-developed porous structure. Furthermore, the catalyst demonstrated strong hydrogen desorption capacity and enhanced thermal stability. Synergistic electronic interactions between Ni and Cu improved the adsorption and activation of both reactant and H2 molecules. The incorporation of Cu near the Ni active sites effectively modulated the selectivity toward the desired product N,N-dimethylcyclohexylamine. Under the optimal reaction conditions: H2 pressure of 5 MPa, reaction temperature of 130 °C, liquid hourly space velocity 0.1 h−1 and hydrogen-amine volume ratio of 5500![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, the 0.1Cu/Ni–Al2O3 catalyst achieved superior performance, with a N,N-dimethylaniline conversion rate of 99.5% and a selectivity for N,N-dimethylcyclohexylamine of 97.6%.
1, the 0.1Cu/Ni–Al2O3 catalyst achieved superior performance, with a N,N-dimethylaniline conversion rate of 99.5% and a selectivity for N,N-dimethylcyclohexylamine of 97.6%.
1 Introduction
Amines, as a class of important industrial organic compounds, play an indispensable role in the production of pharmaceutical intermediates, resin precursors, rubber stabilizers, corrosion inhibitors, and plastics.1 Among these, N,N-dimethylcyclohexylamine (DMCHA), a significant fine chemical within the tertiary amine family, functions as an efficient industrial catalyst in the processing and molding of rigid polyurethane (RPUR) plastics.2,3 Furthermore, DMCHA exhibits tunable hydrophilicity, making it a commercially available and irreplaceable switchable hydrophilic solvent (SHS).4 These attributes underscore the promising market prospects of DMCHA, whose diverse application domains and unique physicochemical properties establish it as a critical raw material across multiple industries.Traditionally, DMCHA is synthesized via the reductive amination of cyclohexanone with dimethylamine. However, this method suffers from several inherent limitations, including low selectivity, susceptibility to side reactions, dehalogenation or over-reduction,5–7 Recently, Yu et al.8 developed a supported Pd/C catalyst that achieved relatively high yields of DMCHA in a continuous-flow micro-packed bed reactor system. While this approach mitigates some drawbacks of conventional reductive amination, the high cost of the Pd/C catalyst poses economic and sustainability challenges, restricting its potential for large-scale industrial application. The palladium and ruthenium catalysts supported on alumina, investigated by Palkovics et al.,9 effectively catalyzed the hydrogenation of ethyl anisate and dimethyl anisate in a continuous flow system, achieving a 90% yield of DMCHA. Similarly, the raw materials for its catalyst are highly costly, which hinders large-scale production, and the reaction yield remains relatively low. Therefore, compared to the conventional reductive amination approach, the synthesis of cyclohexylamine and its derivatives via hydrogenation of aromatic amines offers distinct advantages in terms of synthetic steps and cost efficiency.
The one-step hydrogenation of N,N-dimethylaniline (N,N-DMA) for DMCHA synthesis is characterized by its straightforward pathway, environmental compatibility, and high efficiency.10–13 In previous studies, various catalysts, including noble metals (Rh, Ru, Pd) and non-noble metals (Fe, Co, Ni, Cu), have been employed for the hydrogenation of aniline and its derivatives.10–13 The hydrogenation of aromatic amines to alicyclic amines represents a well-established and strategically important transformation in organic synthesis.14,15 Considerable efforts have been devoted to achieving complete benzene ring hydrogenation in aromatic amines. For example, J. Wu et al. developed a Pt–Ru/C3N4 bimetallic catalyst capable of accomplishing tandem nitro and benzene ring hydrogenation under base-free conditions, elucidating the synergistic mechanism of bimetallic co-hydrogenation.16 X. Lu et al. utilized nickel-based catalysts doped with 3‰ Rh for the hydrogenation of nitrobenzene, achieving 100% conversion and 91.6% selectivity toward cyclohexylamine.10 Currently, research on the hydrogenation synthesis of DMCHA from N,N-DMA has primarily been conducted through batch-type reactor experiments. For example, Mao et al.17 developed a NiRe/HAP catalyst by incorporating Re as a promoter, which significantly enhanced the hydrogenation activity of Ni. This catalytic system demonstrated high efficiency in converting aromatic and heteroaromatic compounds, achieving a DMCHA yield of up to 99%. Nevertheless, this approach suffers from limited production capacity, low separation efficiency, and reliance on Re, thus posing challenges for large-scale industrial application. Similarly, Murugesan and Kuma et al.18,19 reported nano-cobalt and nano-oxidized Ru catalysts, respectively, both of which exhibited efficient and selective hydrogenation of aromatic hydrocarbons, yielding DMCHA at 90% and 94%. However, these powdered catalysts suffer from poor recyclability and separation. Therefore, developing efficient catalysts for selective N,N-DMA hydrogenation in a continuous-flow process is highly desirable for industrial applications.
In this study, a series of Cu-modified Ni–Al2O3 catalysts with various Cu/Ni ratios (xCu/Ni–Al2O3) were prepared, systematically characterized, and investigated in the selective hydrogenation of N,N-DMA towards DMCHA in a continuous fixed-bed reactor. The effect of different metal promoters (Fe, Co, Ru, Cu, Sn) on catalytic performance was systematically investigated.20–23 Cu was identified as the most effective promoter, and synergistic effect was observed between them. Subsequently, the effect of varying Cu/Ni molar ratios on the catalytic hydrogenation of N,N-DMA was investigated. Furthermore, key reaction parameters-including hydrogen pressure, reaction temperature, liquid hourly space velocity (LHSV) and hydrogen-amine volume ratio were optimized to maximize catalytic performance. Under the refined conditions, the 0.1Cu/Ni–Al2O3 catalyst exhibited superior activity, achieving high substrate conversion while significantly enhancing DMCHA selectivity. Notably, the catalyst demonstrates excellent mechanical strength and rapid mass transfer properties, making it highly suitable for industrial-scale implementation.
2 Experimental
2.1 Materials and chemicals
N,N-dimethylaniline (C8H11N, AR) and methanol (CH3OH, AR) were purchased from Shanghai Aladdin Biochemical Technology Co., Ltd. RuCl3·xH2O (Ru: 37.0wt%), SnCl4 (AR), CoN2O6·6H2O (AR), FeN3O9·6H2O (AR) and CuN2O6·6H2O (AR), NiN2O6·6H2O and Al2O3 (99.9% purity) were purchased from Sinopharm Chemical Reagent Co., Ltd (Shanghai, China). All reagents were of analytical reagent grade and used without further treatment. High purity hydrogen (>99.9%) and high purity nitrogen (>99.9%) were purchased from Jiang Su Shenlian Industrial Gas Co., Ltd.2.2 Preparation of catalyst
2.3 Catalyst characterization
2.4 Evaluation of catalytic performance
The catalytic performance of the xCu/Ni–Al2O3 catalyst for N,N-DMA hydrogenation was evaluated in a high-pressure fixed-bed reactor. 20 mL of the catalyst sample was filled into the middle section of a three-stage stainless steel reaction tube, with quartz sand filled in the upper and lower ends for fixation. A thermocouple was inserted from the bottom of the stainless-steel tube. The thermosensitive element to extend to the core area of the catalyst bed to monitor the temperature changes during the hydrogenation reaction in real time.Pressurized hydrogen was flowed downward through the reactor containing the catalyst bed, using an electronic mass flow controller to control the hydrogen gas flow rate. The temperature of the fixed bed was programmed using a temperature control system. Typically, the heating rate of the fixed-bed reactor was set at 10 °C min−1. Since the reaction was performed under high pressure, N,N-DMA was fed using a reciprocating pump, and liquid product samples were collected at regular intervals (every 2 h). The reaction scheme for the hydrogenation of N,N-DMA to DMCHA is illustrated in Scheme 1.
The obtained liquid samples were monitored using a gas chromatography (GC, Shimadzu GCMS-QP2010) equipped with a flame ionization detector (FID). The temperature of FID detector was set at 280 °C. An HP-5 capillary column (30 m × 0.32 mm × 1.00 µm) was used with nitrogen as the carrier gas at a flow rate of 1 mL min−1. The injection volume was 0.2 µL with a split ratio of 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. Maintain the sample port temperature at 240 °C and the column oven temperature at 100 °C for 2 minutes. The temperature was raised to 240 °C at a rate of 20 °C min−1 and maintained for 10 minutes. The conversion of N,N-DMA (C) and the selectivity of DMCHA (S) were calculated using the following equations:
1. Maintain the sample port temperature at 240 °C and the column oven temperature at 100 °C for 2 minutes. The temperature was raised to 240 °C at a rate of 20 °C min−1 and maintained for 10 minutes. The conversion of N,N-DMA (C) and the selectivity of DMCHA (S) were calculated using the following equations:
where AMA, AHA, and Ai are the peak areas of N,N-DMA, DMCHA, and each product, respectively. fMA, fHA, and fi are the correction factor of N,N-DMA, DMCHA, and each product, respectively.
3 Results and discussion
3.1 Synthesis and characterization of xCu/Ni–Al2O3 catalysts
 | ||
| Fig. 1 (a) N2 adsorption–desorption isotherms and (b) pore size distribution of the xCu/Ni–Al2O3 catalysts and Ni–Al2O3 precursor. | ||
As shown in Fig. 1, with increasing copper loading, both the N2 adsorption–desorption isotherms and pore size distribution curves decrease, reflecting a reduction in specific surface area and pore volume. This trend can be attributed to the partial blockage and occupation of alumina support pores by an increased amount of copper species. This variation in the specific surface area and pore structure of the catalyst is evident from Table 1. Notably, When the Cu/Ni molar ratio is 0.1, the catalyst exhibits the smallest specific surface area and pore volume, indicating that the active species influence the pore structure as well as the surface filling and coverage of the support. Meanwhile, the appropriate incorporation of Cu reduces the continuity of Ni active sites, thereby exerting minimal impact on the main reaction while significantly suppressing the side reaction.
| Catalyst | Specific surface area (m2 g−1) | Average pore diameter (nm) | Total pore volume (mL g−1) |
|---|---|---|---|
| Ni–Al2O3 | 129.1 | 6.9 | 0.25 |
| 0.05Cu/Ni–Al2O3 | 105.7 | 7.0 | 0.24 |
| 0.1Cu/Ni–Al2O3 | 101.6 | 7.1 | 0.22 |
| 0.3Cu/Ni–Al2O3 | 102.3 | 7.3 | 0.23 |
| 0.5Cu/Ni–Al2O3 | 104.3 | 6.7 | 0.24 |
 | ||
| Fig. 2 (a) XRD patterns of Ni–Al2O3 and xCu/Ni–Al2O3 precursor. (b) TG profiles of Ni–Al2O3 and xCu/Ni–Al2O3 precursor. | ||
The XRD diffraction peak intensity gradually increased as the introduction of Cu, and the half-peak width of NiO became narrower, revealing that the crystallinity and crystal size of NiO crystals increased correspondingly, mainly because the addition of CuO induced lattice distortion.27 With the increase of the Cu/Ni ratio, the diffraction peak intensity of CuO increased correspondingly because more CuO lattices accumulated on the surface of the catalyst, and excess CuO would cover the position of NiO on the carrier. At the same time, the half-peak width of NiO diffraction peak gradually widened and the intensity decreased, which would lead to the reduction of the contact between Ni2+ and H+,28 excessive inhibition of hydrogenation performance, and reduction of reactant conversion. Therefore, the improved catalytic performance should be related to the appropriate Cu/Ni ratio.
 | ||
| Fig. 3 (a) H2-TPR profiles of xCu/Ni–Al2O3 and Ni–Al2O3 precursor samples. (b) H2-TPD profiles of xCu/Ni–Al2O3 and Ni–Al2O3 precursor samples. | ||
With increasing Cu content, peak α progressively shifted toward lower temperatures, while its area increased, suggesting that Cu promoted the reduction of NiO species. Additionally, more surface CuO species are reduced at lower temperatures, reflecting improved H2 adsorption capacity and enhanced low-temperature reducibility in the CuNi bimetallic system compared to the monometallic counterpart, thereby favoring hydrogenation reactions. For the 0.05Cu/Ni–Al2O3, 0.1Cu/Ni–Al2O3, 0.3Cu/Ni–Al2O3 and 0.5Cu/Ni–Al2O3 precursors, peak β initially shifted to higher temperatures and then shifted back to lower temperatures as the Cu/Ni ratio increased. The highest reduction temperature for peak β is observed at 345.9 °C for 0.1Cu/Ni–Al2O3. This phenomenon can be attributed to the optimal Cu/Ni molar ratio facilitating the formation of structurally CuNi alloy clusters, whose stability delays reduction and necessitates higher temperatures,31 the strong interaction between Cu and Ni attenuates Ni's ability for dissociative adsorption of H2 and activation of C–N bonds, thereby exerting a more pronounced inhibitory effect on the demethylation side reaction. Furthermore, the area of peak β increases with Cu loading, owing to the greater amount of CuO deposited on the support. Compared to NiO, CuO exhibits higher reducibility under similar conditions.32
Upon incorporation of Cu into the Ni–Al2O3 precursor, an additional H2 desorption peak (designated as peak ε) emerged in the H2-TPD profile of xCu/Ni–Al2O3. With increasing Cu/Ni molar ratio, peak ε broadens progressively, suggesting that Cu enhances the dispersion of Ni species. Concurrently, peak γ shifts toward lower temperatures while peak δ shifts toward higher temperatures, indicating the influence of electronic interactions in the CuNi bimetallic system on the catalyst's hydrogen adsorption behavior. The appearance of three distinct reduction peaks in the xCu/Ni–Al2O3 precursor reflects heterogeneous hydrogen adsorption sites arising from intermetallic electron interactions,34,35 When the Cu/Ni molar ratio is 0.1, the ε area of the low-temperature H2 desorption peak is the largest, indicating that the number of weakly chemically adsorbed hydrogen species on the catalyst surface is the highest. This is mainly attributed to the strong bimetallic synergistic interaction between Cu and Ni, resulting in the formation of a Cu–Ni alloy phase, continuous and regular crystal plane spacing stripes can be seen in the HR-TEM images (Fig. S3), no individual lattice stripes of Cu (d = 0.208 nm, 111 crystal plane) or Ni (d = 0.203 nm, 111 crystal plane) were observed.36,37 Instead, characteristic lattice stripes (d = 0.205 nm) that were between the interplanar spacings of the Cu and Ni crystal planes were present, indicating that Cu atoms had successfully been incorporated into the Ni lattice, forming a Cu–Ni substitutional alloy phase. This phenomenon is confirmed by SEM elemental mapping and XPS electron transfer analysis This ensures the hydrogenation of the aromatic rings in the main reaction. The weakly adsorbed hydrogen activity is insufficient to attack the strong C–N bonds, thereby inhibiting the formation of by-products and improving the selectivity.
 | ||
| Fig. 5 In situ XPS spectra of Ni 2p for the Ni–Al2O3, xCu/Ni–Al2O3 catalyst (outside the dashed area) and Cu2p for the xCu/Ni–Al2O3 catalyst (within the dashed line area). | ||
The Cu 2p XPS spectrum displays distinct spin–orbit splitting, as shown in Fig. 5 (within the dashed line area). The Cu 2p3/2 peak for Cu0 is observed at 931.9–932.6 eV, with the corresponding Cu 2p1/2 peak appearing at 951.8–952.5 eV. When the Cu/Ni molar ratio increases from 0.05 to 0.1, a slight decrease in the Cu 2p3/2 binding energy is observed, reflecting the electronic interaction between Ni and Cu. Metallic Ni has a higher density of d-band holes than Cu, promoting the transfer of d electrons from Cu0 to vacant d orbitals of Ni0 and resulting in electron-deficient Cu species.39 This synergistic electronic effect facilitates the formation of well-dispersed and highly adsorptive bimetallic Cu–Ni species on the Al2O3 support, thereby enhancing the catalytic performance of the 0.1Cu/Ni–Al2O3 catalyst. However, at higher copper loadings (Cu/Ni = 0.3 and 0.5), excessive copper leads to surface enrichment of Cu species, which may mask or block certain Ni active sites, disrupting the balance between the two metals and ultimately reducing overall catalytic efficiency.
3.2 Catalytic activity test
As presented in Table 2, when the molar ratio of Cu/Ni increased from 0.05 to 0.5, the conversion rate of N,N-DMA declined gradually, suggesting that Ni serves as the primary active site. A higher Ni loading enhances the adsorption and activation of H2 molecules, thereby generating more active hydrogen species. The exposed Ni sites on the catalyst surface facilitate the attack of hydrogen atoms on the aromatic ring, promoting the cleavage of C–C bonds within the benzene ring.40 This process enables effective hydrogen addition to the aromatic ring in N,N-DMA, leading to the formation of DMCHA and maintaining a high conversion. However, excessive Cu incorporation reduces the availability of Ni active sites, resulting in diminished conversion efficiency. Consequently, by optimizing the Cu loading, specifically at a Cu/Ni molar ratio of 0.1, not only is a high N,N-DMA conversion rate preserved, but the selectivity toward DMCHA is also enhanced. This improvement is attributable to the moderating effect of Cu on excessive hydrogenation. A side reaction commonly observed with highly active Ni-based catalysts. Under such catalysis, over-hydrogenation may lead to C–N bond cleavage in DMCHA, yielding undesirable by-products (e.g., N-Me-CyH, cyclohexanol, and cyclohexane) that compromise product selectivity. The introduction of Cu with appropriate amount helps reduce the d band electrons of Ni, thereby weakening electron occupancy in the antibonding orbitals of the C–N bond in DMCHA,41 suppressing its premature cleavage and minimizing side reactions. As a result, the 0.1Cu/Ni–Al2O3 catalyst demonstrates superior performance in the hydrogenation of N,N-DMA, achieving both high activity and optimal selectivity.
| Catalyst | N,N-DMA conversion (%) | DMCHA selectivity (%) | N-Me-CyH selectivity (%) |
|---|---|---|---|
| a Reaction conditions: 20 mL catalyst, P = 5 MPa, T = 120 °C, LHSV = 0.2 h−1 and H2/N,N-DMA (v/v) = 3500. | |||
| Ni–Al2O3 | 89.5 | 89.1 | 8.6 |
| 0.05Cu/Ni–Al2O3 | 89.3 | 90.4 | 7.4 |
| 0.1Cu/Ni–Al2O3 | 89.2 | 94.5 | 3.9 |
| 0.3Cu/Ni–Al2O3 | 81.6 | 95.6 | 3.5 |
| 0.5Cu/Ni–Al2O3 | 77.4 | 95.9 | 3.0 |
On the monometallic Ni-based catalyst, the strong adsorption of Ni toward both the reactant (N,N-DMA) and protons (H+), coupled with the presence of extensive contiguous Ni active sites on the surface, facilitates multipoint adsorption. This configuration promotes over-hydrogenation, manifesting as undesired demethylation or deamination side reactions. Introducing an optimal amount of Cu (e.g., Cu/Ni ratio = 0.1) modulates the electronic structure of Ni: owing to Cu's broader d-band center and its greater energetic separation from the Fermi level relative to Ni, the adsorption strength of Ni toward unsaturated bonds is attenuated.42 This electronic effect between Cu and Ni is consistent with the XPS results. Meanwhile, this moderated adsorption energy favors end-on (terminal) adsorption of the reactant, thereby accelerating the initial H atom insertion (i.e., primary hydrogenation) while suppressing flat-lying or multipoint adsorption modes.43,44 Consequently, over-hydrogenation is effectively inhibited, and selectivity toward the target product (DMCHA) is maximized. Furthermore, the prominent high-temperature reduction peak β observed in H2-TPR for the 0.1Cu/Ni–Al2O3 catalyst provides direct evidence for the formation of strongly interacting Cu–Ni alloy clusters. Such alloying not only enhances structural stability but also induces electron redistribution at Ni sites via ligand effects, lowering the binding energy of DMCHA and facilitating its timely desorption. This mitigates secondary reactions arising from prolonged surface residence, a critical factor in boosting product selectivity.
In contrast, excessive Cu loading (e.g., 0.5Cu/Ni–Al2O3) further improves low-temperature reducibility, yet catalytic evaluation reveals that surplus Cu species may physically occlude Ni active sites and diminish the density of synergistic Ni–Cu interfacial sites. Although primary hydrogenation activity remains appreciable, the number of sites available for the reactants to undergo specific configuration adsorption decreases, resulting in disordered reaction pathways and decreased conversion rate, reflecting the volcano-type relationship between catalytic structure and activity.
 | ||
| Fig. 6 Effects of (a) H2 pressure, (b) reaction temperature, (c) LHSV and (d) hydrogen-amine volume ratio on the N,N-DMA hydrogenation to DMCHA. | ||
Reaction temperature significantly influences catalytic activity. Elevating the reaction temperature enhances the diffusion rate of N,N-DMA, thereby promoting stronger interaction between the reactant molecules and the metallic Ni sites within the catalyst. This leads to improved conversion of N,N-DMA and enhanced selectivity toward DMCHA. As illustrated in Fig. 6(b), the effect of temperature on the performance of the 0.1Cu/Ni–Al2O3 catalyst was investigated by increasing the temperature from 110 °C to 150 °C. The results indicate that the conversion of N,N-DMA increases progressively with temperature and reaches a maximum of 99.6% at 150 °C. However, the selectivity toward DMCHA exhibits a non-linear trend, it initially increases and then decreases with further temperature elevation, peaking at 95.8% at 130 °C. In contrast, the selectivity toward N-Me-CyH exhibits an initial decline followed by an increase, with a minimum observed at 130 °C. These observations suggest that while higher temperatures accelerate the primary reaction, they also promote side reactions that negatively affect catalytic selectivity. Therefore, a reaction temperature of 130 °C is determined to be optimal for subsequent reaction optimization studies.
The feed rate of reactants exerts a significant influence on the outcome of the hydrogenation reaction. Liquid hourly space velocity (LHSV) is a critical parameter reflecting the residence time of reactants on the catalyst surface. The effect of LHSV on the catalytic performance in the hydrogenation of N,N-DMA was systematically investigated, and with the results illustrated in Fig. 6(c). When the LHSV increased from 0.05 to 0.25 h−1, the conversion of N,N-DMA decreased from 99.5% to 84.8%. Elevated LHSV indicates higher liquid throughput per unit time, thereby reducing the contact duration between reactant molecules and the active sites on the catalyst surface. This shortened interaction time limits the catalyst's ability to fully facilitate the reaction, resulting in lower conversion efficiency. These findings underscore LHSV as a key determinant of hydrogenation conversion rates. However, DMCHA selectivity remained relatively stable across the tested LHSV range, with no significant variation. The highest selectivity toward the desired product DMCHA, along with the lowest formation of the main by-product N-Me-CyH, was achieved at an LHSV of 0.1 h−1. Therefore, an LHSV of 0.1 h−1 is identified as the optimal operational condition for this catalytic system.
The influence of the hydrogen-amine volume ratio was also investigated. Hydrogen functions not only as a reactant in the hydrogenation process but also as a carrier gas, maintaining system pressure stability and ensuring consistent reaction progression. Under selected conditions, the optimal hydrogen-amine volume ratio was experimentally determined. As illustrated in Fig. 6(d), increasing the hydrogen-amine ratio led to higher N,N-DMA conversion rates, achieving a maximum of 99.8%. This enhancement is attributed to the efficient hydrogenation of N,N-DMA in a hydrogen-rich environment. However, the selectivity toward the desired product DMCHA peaked at 97.6% when the hydrogen-amine ratio reached 5500![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. Further increases in the ratio resulted in decreased DMCHA selectivity, accompanied by increased formation of the by-product N-Me-CyH. This trend suggests excessive hydrogenation under such conditions, promoting side reactions and reducing selectivity for DMCHA. Consequently, a hydrogen-amine volume ratio of 5500
1. Further increases in the ratio resulted in decreased DMCHA selectivity, accompanied by increased formation of the by-product N-Me-CyH. This trend suggests excessive hydrogenation under such conditions, promoting side reactions and reducing selectivity for DMCHA. Consequently, a hydrogen-amine volume ratio of 5500![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 was identified as optimal for achieving high conversion and selectivity simultaneously. Following systematic optimization of various reaction parameters, the substrate conversion rate reached 99.5%, with a corresponding yield of the target product DMCHA of 97.6%. This result represents a notable improvement compared to previous studies,9 which reported a maximum yield of 90% under similar conditions in a continuous-flow fixed-bed reactor system.
1 was identified as optimal for achieving high conversion and selectivity simultaneously. Following systematic optimization of various reaction parameters, the substrate conversion rate reached 99.5%, with a corresponding yield of the target product DMCHA of 97.6%. This result represents a notable improvement compared to previous studies,9 which reported a maximum yield of 90% under similar conditions in a continuous-flow fixed-bed reactor system.
4 Conclusions
In conclusion, the synthesis of DMCHA has been successfully achieved via the hydrogenation of N,N-DMA in a fixed-bed reactor employing a non-noble metal catalytic system. By incorporating Cu as a promoter into the Ni–Al2O3 precursor via an impregnation method, the resulting 0.1Cu/Ni–Al2O3 catalyst demonstrates excellent catalytic performance and stability, as well as high selectivity toward DMCHA. The well-developed specific surface area and mesoporous structure of the Al2O3 support facilitate highly dispersed distribution of active metal components. This structural feature promotes more efficient adsorption and activation of H2 molecules into reactive hydrogen species, thereby enhancing overall reaction conversion. Furthermore, an electronic interaction between nickel and copper is observed in the xCu/Ni–Al2O3 catalyst system, which influences the primary hydrogenation pathways and product selectivity. The relative contents of nickel and copper play a critical role in determining catalytic behavior; specifically, at a Cu/Ni molar ratio of 0.1, the catalyst effectively suppresses C–N bond cleavage, minimizes side reactions, and significantly improves selectivity toward the desired product. The effects of reaction parameters-including H2 pressure, reaction temperature, LHSV and hydrogen-amine volume ratio on the hydrogenation process were systematically investigated. Ultimately, this study demonstrates a highly selective hydrogenation process for converting N,N-DMA to DMCHA, underscoring its significant potential for practical application.Author contributions
Jingdong Nong: conducting experiments, data curation, and writing original draft. Zhonghua Sun, Weiyou Zhou: writing – review & editing. Zhong Wu: investigation. Junfeng Qian: supervision, project management, and funding acquisition. Qun Chen, Mingyang He: resources, supervision, and funding acquisition.Conflicts of interest
There are no conflicts to declare.Data availability
The data obtained from this research experiment are fully presented in the article and supplementary information (SI). The authors will provide any additional requested data upon reasonable request. Supplementary information: the SI of this research experiment includes physical illustrations of the main experimental equipment (fixed-bed reactor), hydrogenation synthesis schemes, catalyst stability assessment, elemental content table of 0.1Cu/Ni–Al2O3 catalyst, hydrogenation performance of different additives, gas phase spectra and gas chromatographic spectra of the products, for reference. See DOI: https://doi.org/10.1039/d6ra01119e.Acknowledgements
This work was supported by Jiangsu Key Laboratory of Advanced Catalytic Materials and Technology (BM2012110), Thanks to Sinopec Nanjing Chemical Industries Co., Ltd and Changzhou University for their support and help in this experiment.References
- S. Gomez, J. A. Peters and T. Maschmeyer, Adv. Synth. Catal., 2002, 344, 1037–1057 CrossRef CAS.
- A. L. Silva and J. C. Bordado, Catal. Rev.-Sci. Eng., 2004, 46, 31–51 CrossRef.
- D. Sridaeng, W. Jitaree, P. Thiampanya and N. Chantarasiri, e-Polymers, 2016, 16, 265–275 CrossRef CAS.
- C. Samori, L. Pezzolesi, D. L. o. Barreiro, P. Galletti, A. Pasteris and E. Tagliavini, RSC Adv., 2014, 4, 5999–6008 RSC.
- A. Villa, K. Dumoleijn, C. Evangelisti, K. Moonen and L. Prati, RSC Adv., 2018, 8, 15202–15206 RSC.
- A. R. Jadhav, H. A. Bandal and H. Kim, Chem. Eng. J., 2016, 295, 376–383 CrossRef CAS.
- F. Mao, D. Sui, Z. Qi, H. Fan, R. Chen and J. Huang, RSC Adv., 2016, 6, 94068–94073 RSC.
- H. Yu, W. Ming, W. Wei, Y. Zhao, X. Dong, X. Wu, G. Qin and D. Yan, React. Chem. Eng., 2026, 188–195 RSC.
- I. Palkovics, I. Gemes and P. Szonyi, Hung. J. Ind. Chem., 1989, 17, 113–120 CAS.
- X. Lu, Y. chen, Z. Zhao, H. Deng, D. Zhou, C. Wei, R. Nie and Q. Xia, RSC Adv., 2016, 6, 15354–15361 RSC.
- H. Hagihara and E. Echigoya, Bull. Chem. Soc. Jpn., 1965, 38, 2094–2100 CrossRef CAS.
- P. Kar and B. G. Mishra, J. Environ. Chem. Eng., 2016, 4, 1962–1969 CrossRef CAS.
- D. Xie, X. Liu, H. Lv and Y. Guo, J. Supercrit. Fluids, 2022, 182, 105509 CrossRef CAS.
- T.-C. Huang and B.-C. Kang, Ind. Eng. Chem. Res., 1995, 34, 2955–2963 CrossRef CAS.
- L. Zuo, J. Cai, Z. Guo, Y. Fu and J. Shen, Catal. Commun., 2022, 170, 106496 CrossRef CAS.
- J. Wu, J. Zhang, L. Wang, Z. Han, X. Hui, Y. Cao, J. Shi, S. Xu, P. He and H. Li, Chem. Eng. J., 2024, 488, 151083 CrossRef CAS.
- W. Mao, Y. Sun, M. Liu, X. Gao, X. Liu, Z. Bao, Q. Yang, Q. Ren and Z. Zhang, ACS Appl. Nano Mater., 2025, 8, 11131–11139 CrossRef CAS.
- K. Murugesan, T. Senthamarai, A. S. Alshammari, R. M. Altamimi, C. Kreyenschulte, M.-M. Pohl, H. Lund, R. V. Jagadeesh and M. Beller, ACS Catal., 2019, 9, 8581–8591 CrossRef CAS.
- A. Kumar, V. Goyal, N. Sarki, B. Singh, A. Ray, T. Bhaskar, A. Bordoloi, A. Narani and K. Natte, ACS Sustain. Chem. Eng., 2020, 8, 15740–15754 CrossRef CAS.
- J. Duan, H. Zhu, F. Xu and J. Zhao, Chem. Eng. J., 2016, 304, 282–288 CrossRef CAS.
- Z. Ruijie, J. Haibo, M. Lei and Y. Suohe, RSC Adv., 2023, 13, 27036–27045 RSC.
- Q. Guo, H. Lv, J. Mao and J. Zhou, Mol. Catal., 2024, 554, 113857 CAS.
- Y. Shao, M. Guo, L. Kong, M. Fan, K. Sun, C. Li, L. Zhang, S. Zhang, T. Wei and X. Hu, Fuel, 2025, 391, 134782 CrossRef CAS.
- O. Arbeláez, T. R. Reina, S. Ivanova, F. Bustamante, A. L. Villa, M. A. Centeno and J. A. Odriozola, Appl. Catal., A, 2015, 497, 1–9 CrossRef.
- S. Lin, Z. Sun, Z. Wang, Q. Zhou, A. Wang, Z. Wu, J. Qian, M. He and Q. Chen, New J. Chem., 2025, 49, 5783–5790 RSC.
- X. Li, L. Geng, H. Shi, S. Ye, L. B. Sim, J. Zheng, N. Zhang and B. Chen, Appl. Catal., A, 2024, 671, 119564 CrossRef CAS.
- C. Miao, G. Zhou, S. Chen, H. Xie and X. Zhang, Renewable Energy, 2020, 153, 1439–1454 CrossRef CAS.
- M. Kimi, M. M. H. Jaidie and S. C. Pang, J. Phys. Chem. Solids, 2018, 112, 50–53 CrossRef CAS.
- M. C. Biesinger, B. P. Payne, A. P. Grosvenor, L. W. M. Lau, A. R. Gerson and R. S. C. Smart, Appl. Surf. Sci., 2011, 257, 2717–2730 CrossRef CAS.
- Y. Fu, F. Devred, P. Eloy, T. Haynes, M. L. Singleton and S. Hermans, Appl. Catal., A, 2022, 644, 118833 CrossRef CAS.
- T. V. Reshetenko, L. B. Avdeeva, Z. R. Ismagilov, A. L. Chuvilin and V. A. Ushakov, Appl. Catal., A, 2003, 247, 51–63 CrossRef CAS.
- J.-H. Lin, P. Biswas, V. V. Guliants and S. Misture, Appl. Catal., A, 2010, 387, 87–94 CrossRef CAS.
- S. Chen, C. Miao, Y. Luo, G. Zhou, K. Xiong, Z. Jiao and X. Zhang, Renewable Energy, 2018, 115, 1109–1117 CrossRef CAS.
- N. Hu, C. Yang, L. He, Q. Guan and R. Miao, New J. Chem., 2019, 43, 18120–18125 RSC.
- S. Zhang, J. Li, N.-Z. Shang, S.-T. Gao, C. Wang and Z. Wang, New J. Chem., 2020, 44, 15671–15676 RSC.
- A. A. A. Ahmed, T. S. Aldeen, S. A. Al-Aqil, Z. M. Alaizeri and S. Megahed, ACS Omega, 2022, 7, 37340–37350 CrossRef CAS PubMed.
- J. Dutkiewicz, L. Litynska, W. Maziarz, R. Kocisko, M. Molnarova and A. Kovacova, Micron, 2009, 40, 1–5 CrossRef CAS PubMed.
- C. Yang, W. Xue, H. Yin, Z. Lu, A. Wang, L. Shen and Y. Jiang, New J. Chem., 2017, 41, 3358–3366 RSC.
- X. Du, J. Huang, Y. Feng and Y. Ding, Chin. J. Catal., 2016, 37, 123–134 CrossRef CAS.
- X. Wang and J. Chen, Chin. J. Catal., 2017, 38, 1818–1830 CrossRef CAS.
- S. Jin, Z. Xiao, C. Li, X. Chen, L. Wang, J. Xing, W. Li and C. Liang, Catal. Today, 2014, 234, 125–132 CrossRef CAS.
- Z.-P. Fu, Y.-P. Zhao, Y.-F. Wu, Q.-J. Zhou, S.-T. Hu, L.-L. Qiu, F.-J. Liu, M. Zhong, J. Liang and J.-P. Cao, J. Anal. Appl. Pyrolysis, 2026, 195, 107623 CrossRef CAS.
- X. Ge, J. Pan, X. Chen, C. Qian and S. Zhou, RSC Adv., 2016, 6, 53188–53194 RSC.
- Z. Lv, Z. Hong, D. Ke, C. Qian, X. Chen and S. Zhou, J. Colloid Interface Sci., 2025, 683, 247–261 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |