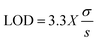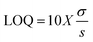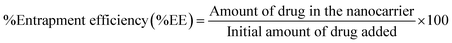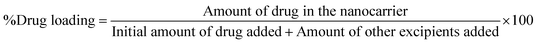Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Design and ICH validation of a stability-indicating green RP-HPLC method for 5,6,7-trihydroxyflavone with multi-metric greenness evaluation and application to nanopharmaceutical systems
Sweta
Acharya
and
Ankit
Jain
 *
*
Industrial Research Laboratories, Department of Pharmacy, Birla Institute of Technology and Science, Pilani Campus, Pilani, Rajasthan 333031, India. E-mail: ankit.j@pilani.bits-pilani.ac.in
First published on 13th April 2026
Abstract
A novel stability-indicating reverse-phase high-performance liquid chromatography (RP-HPLC) method was developed and validated for the quantification of baicalein (5,6,7-trihydroxyflavone) in bulk drug and nanoliposomal formulations. Samples were pretreated via dilution with mobile phase, membrane filtration (0.22 µm), protection from light, and refrigerated storage prior to analysis. Method validation was performed in accordance with ICH Q2(R2) guidelines. Chromatographic separation was achieved on a BDS C18 column using an isocratic mobile phase comprising water (pH 3.0, adjusted with orthophosphoric acid) and acetonitrile (50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50, v/v) at a flow rate of 1.0 mL min−1 with DAD detector. The method exhibited excellent linearity (0.0078–1 µg mL−1; R2 = 0.9998), accuracy (98.25–101.58%), and precision (%RSD < 2%). The limits of detection (LOD) and quantification (LOQ) were 0.0069 µg mL−1 and 0.0208 µg mL−1, respectively. Forced degradation under acidic, alkaline, oxidative, and thermal stress conditions confirmed the stability-indicating capability, with pronounced degradation under acidic conditions and high thermal stability. The method was successfully applied to solubility profiling and quantification in nanoliposomal systems, including entrapment efficiency and drug loading. Greenness assessment using Blue Applicability Grade Index (BAGI), Modified Green Analytical Procedure Index (MoGAPI), Carbon Footprint Reduction Index (CaFRI), Click Analytical Chemistry Index (CACI), Analytical GREEnness (AGREE), and Violet Innovation Grade Index (VIGI) yielded scores of 75, 85, 77, 80, 0.64, and 55, respectively. The method demonstrates key advantages, including buffer-free operation, reduced chemical hazard, and operational simplicity; however, moderate organic solvent usage imposes minor sustainability limitations. Overall, the method is robust, sensitive, and suitable for routine pharmaceutical and nanoformulation analysis.
50, v/v) at a flow rate of 1.0 mL min−1 with DAD detector. The method exhibited excellent linearity (0.0078–1 µg mL−1; R2 = 0.9998), accuracy (98.25–101.58%), and precision (%RSD < 2%). The limits of detection (LOD) and quantification (LOQ) were 0.0069 µg mL−1 and 0.0208 µg mL−1, respectively. Forced degradation under acidic, alkaline, oxidative, and thermal stress conditions confirmed the stability-indicating capability, with pronounced degradation under acidic conditions and high thermal stability. The method was successfully applied to solubility profiling and quantification in nanoliposomal systems, including entrapment efficiency and drug loading. Greenness assessment using Blue Applicability Grade Index (BAGI), Modified Green Analytical Procedure Index (MoGAPI), Carbon Footprint Reduction Index (CaFRI), Click Analytical Chemistry Index (CACI), Analytical GREEnness (AGREE), and Violet Innovation Grade Index (VIGI) yielded scores of 75, 85, 77, 80, 0.64, and 55, respectively. The method demonstrates key advantages, including buffer-free operation, reduced chemical hazard, and operational simplicity; however, moderate organic solvent usage imposes minor sustainability limitations. Overall, the method is robust, sensitive, and suitable for routine pharmaceutical and nanoformulation analysis.
1. Introduction
High-performance liquid chromatography (HPLC) is widely used in pharmaceutical analysis for its efficiency in analyzing multiple compounds, including associated impurities and degradants. The use of this technique has increased in recent years across laboratory and industrial settings due to its precise, accurate determination of analytes of interest. This technique proves itself as an efficient method for determining impurities, especially in Active Pharmaceutical Ingredients (APIs), which is a significant task, as even trace amounts of these can lead to side effects, toxicity, or reduced efficacy of the drug over time.1,2In analytical method development, appropriate sample pretreatment is a critical step to ensure accuracy, reproducibility, and compatibility with chromatographic systems. Common strategies include dilution with suitable solvents, membrane filtration (0.22 µm), and protection from environmental factors such as light and temperature to prevent analyte degradation and matrix interference. These steps are particularly important when dealing with complex pharmaceutical matrices and nanocarrier-based systems.
For any analytical method to be robust, it must be validated to comply with international regulatory standards and demonstrate its suitability for use. According to the International Conference on Harmonization (ICH) Q2(R2) guideline, a validated quantitative analytical procedure is capable not only of quantifying the drug in pharmaceutical formulations but also of detecting changes in product quality during storage, which can be confirmed if a stability-indicating test is able to separate the drug from its degradation products.3
In this context, forced degradation studies are essential to establish the stability-indicating capability of an analytical method. Such studies involve subjecting the analyte to stress conditions, including acidic, alkaline, oxidative, and thermal environments, to evaluate degradation behavior and ensure effective separation of degradation products from the parent compound.
Baicalein (BLN) is a 5,6,7-trihydroxyflavone (Fig. 1(A)), which is obtained from the roots of Scutellaria baicalensis Georgi (Lamiaceae) and is widely used in Traditional Chinese Medicine. Recently, it has been classified as a Biopharmaceutics Classification System (BCS) Class II substance because of its low water solubility (<0.1 mg mL−1), which causes an oral bioavailability (BA) of 36.1% ± 4.4% and restricts its medicinal use.4,5 BLN has a wide range of pharmacological activities, including anticancer, antioxidant, anti-allergic, antiviral, and anti-inflammatory properties.
So far, in vivo studies have shown that BLN can efficiently reduce inflammation associated with cancer, neurodegenerative diseases, and other conditions. As the clinical translation of BLN accelerates, the development and validation of this drug have become increasingly important, underscoring the need for a precise and trustworthy analytical technique (Fig. 1(B)).6
Current HPLC methodologies for quantifying BLN use a phosphate-buffered mobile phase, multi-component gradient elution, or a high percentage of organic solvent to control phenolic compound ionization and improve peak symmetry.6,7 Although these approaches are effective in achieving the desired chromatographic resolution, they are associated with considerable environmental consequences. The use of a buffer-based mobile phase results in the generation of waste containing inorganic salts that need careful treatment and disposal, causes column fouling and reduces the column's lifespan, and increases the solvent consumption and analysis time during column re-equilibration. The use of a multi-component gradient elution approach also increases the environmental impact of the analysis due to excessive solvent consumption during column re-equilibration.
Recent advancements in pharmaceutical analysis emphasize the adoption of sustainable and eco-friendly analytical methodologies. Multi-metric greenness assessment tools such as the Blue Applicability Grade Index (BAGI), Modified Green Analytical Procedure Index (MoGAPI), Analytical GREEnness (AGREE), Carbon Footprint Reduction Index (CaFRI), Click Analytical Chemistry Index (CACI), and Violet Innovation Grade Index (VIGI) enable comprehensive evaluation of environmental impact, method efficiency, and innovation. These tools are increasingly integrated into method development to ensure compliance with Green Analytical Chemistry principles.
In addition, high-performance liquid chromatography coupled with diode-array detection (HPLC-DAD) provides enhanced analytical capabilities through spectral peak purity analysis, improved selectivity, and reliable quantification, making it particularly suitable for stability-indicating studies and complex pharmaceutical formulations. Recent eco-friendly HPLC-DAD methods have demonstrated significant potential in reducing environmental burden while maintaining analytical performance.
In contrast, the current approach uses a mobile phase consisting of Milli-Q water at pH 3, adjusted with orthophosphoric acid, and acetonitrile under isocratic conditions. This approach avoids the generation of inorganic salts and the use of auxiliary reagents. Moreover, the current approach avoids ionic-strength-based control in the analysis of baicalein and instead controls the compound's ionization by adjusting the pH of the mobile phase. As a result, the current approach has a lower environmental impact than the previously published methodologies and can be regarded as a ‘greener’ approach to the analysis of baicalein.
Furthermore, the increasing use of nanopharmaceutical systems, such as liposomes and lipid-based carriers, necessitates robust analytical techniques for accurate quantification, drug loading, entrapment efficiency, and stability assessment, thereby reinforcing the need for sensitive and reliable chromatographic methods.
Therefore, the aim of this work was to develop and validate a sensitive, stability-indicating, and environmentally sustainable RP-HPLC method for the determination of BLN in accordance with ICH guidelines. The proposed method integrates buffer-free chromatographic conditions, multi-metric greenness evaluation, and applicability to nanopharmaceutical systems. The method offers significant advantages, including reduced hazardous waste generation, operational simplicity, and high sensitivity; however, the use of organic solvents poses a minor limitation for overall sustainability.8–10
2. Experimental
2.1. Materials
Baicalein (BLN, purity ≥ 98%, analytical grade, CAS no. 491-67-8), a flavonoid with the IUPAC name 5,6,7-trihydroxy-2-phenylchromen-4-one, was obtained from Density Pharmachem Private Limited (India). The supplier provided a certificate of analysis confirming purity and identity. The compound was stored in a desiccated, light-protected environment at 2–8 °C to prevent degradation. Acetonitrile (ACN, HPLC grade) was obtained from Qualigens Pharma Private Limited (India). De-ionized water was prepared by reverse osmosis using a Millipak system (0.22 µm; Millipore, MA, USA). Reagents such as hydrochloric acid (HCl), sodium hydroxide (NaOH), orthophosphoric acid (OPA), and hydrogen peroxide (H2O2) were obtained from Sisco Research Laboratories (India). Theophylline was purchased from Tokyo Chemical Industry Co., Ltd. The LIPOID S PC-3 (phosphatidylcholine, hydrogenated, content ≥ 98.0%), CAS-no. 97281-48-6, was provided as a gift sample from Lipoid® GmbH, Ludwigshafen, Germany. Cholesterol was purchased from Sigma-Aldrich Corporation.2.2. Instruments and software
The analytical method was developed on the Thermo Scientific™ UltiMate™ 3000 Standard Quaternary Analytical System manufactured by Thermo Fisher Scientific. Data acquisition was performed using Chromeleon 7.2 SR4 Software. The particle size analysis and zeta potential analysis were performed using the Malvern Zetasizer Nano ZS, manufactured by Malvern Panalytical. The data on particle size and zeta potential were obtained using the Zetasizer software. The purification of liposomes was performed by repeated washing and centrifuging the solution using an Eppendorf 5415R centrifuge (Eppendorf AG). The UV spectrum was obtained using the Shimadzu UV-2600 (or the updated UV-2600i) single-monochromator ultraviolet-visible spectrophotometer, manufactured by Shimadzu Corporation. The UV-spectral data were obtained using the UVProbe 2.61 software. Graphical representations and statistical tests were generated using OriginPro 2026 (learning edition). Diagrams were created using Biorender software under an academic license.2.3. Preliminary investigation: ultraviolet (UV) spectroscopic determination of baicalein
The UV spectroscopic method was adapted from previously reported procedures for flavonoid analysis with minor modifications. For the preparation of the working solution aliquot of BLN, the primary stock solution in methanol at 1 mg mL−1 was diluted to 10 µg mL−1. The UV spectra were obtained by placing the prepared solutions into a quartz cuvette. The spectral information was acquired using a Shimadzu 1800 spectrophotometer over a wavelength range of 200–400 nm, with methanol as the blank.112.4. Instrumentation and chromatographic conditions
The Thermo Fisher Ultimate 3000 system, integrated with a quaternary pump, a degasser unit with a 100 µL autosampler loop, and a diode-array detector (DAD), was used to develop the method. Data acquisition was performed using Chromeleon 7.2 SR4 Software.Optimization of chromatographic conditions was performed using a univariate (one-factor-at-a-time) approach, systematically varying mobile-phase composition, pH, flow rate, and detection wavelength to achieve optimal resolution, peak symmetry, and retention time. Method performance was evaluated based on retention time (tR), peak area, tailing factor, and theoretical plate count. Quantification of BLN was carried out using peak area as the analytical response. Matrix effects were minimized through appropriate sample pretreatment and dilution; no significant interference was observed at the BLN retention time, confirming method specificity. The optimized analysis parameters are given in Table 1. The optimization of chromatographic conditions focused on the mobile-phase ratio, isocratic profile, injection volume, analysis time, and column temperature.
| Parameters | Description |
|---|---|
| Instrument | Thermo Fisher Ultimate 3000 HPLC system equipped with a quaternary pump, autosampler, column oven, and DAD |
| Column | BDS C18 Reversed-phase HPLC column, 5 µm, 4.6 mm × 250 mm |
| Mobile phase | Water (pH 3.0 ± 0.05, adjusted with OPA)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ACN, 50 ACN, 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 (v/v) 50 (v/v) |
| Flow rate | 1.0 mL min−1 |
| Detection wavelength | 200–400 nm (λmaxof BLN = 275 nm) |
| Injection volume | 20 µL |
| Column temperature | Ambient or 25 °C |
| Autosampler temperature | 10 °C |
| Run time | 10 min |
| Diluent | Mobile phase (50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 v/v) 50 v/v) |
| Elution | Low-pressure isocratic mode |
| Pressure | 1195–1208 psi |
2.5. Preparation of calibration standards
The previously prepared primary stock solution of BLN was used. The solution was protected from light and refrigerated (4 ± 0.5 °C) until further analysis, which was then diluted to make secondary stocks at 100, 10, and 1 µg mL−1 using the same solvent medium. Working solutions were prepared by diluting the 1 µg mL−1 solution with mobile phase to produce calibration standards at concentrations of 1, 0.5, 0.25, 0.125, 0.0625, 0.0312, 0.0156, and 0.0078 µg mL−1.2.6. Validation of analytical method
The current method was validated for system suitability, specificity, linearity, precision, accuracy, and robustness in accordance with ICH Q2 (R2) (1 November 2023), as described below.where “σ” is the standard deviation of the response and “s” is the slope of the calibration curve.
| Type of degradation | Agent used/condition | Neutralizing agent/2 h |
|---|---|---|
| Acidic hydrolysis | 1 mol L−1 hydrochloric acid (HCl)/37 °C/24 h | 1 mol L−1 NaOH |
| Basic hydrolysis | 1 mol L−1 sodium hydroxide (NaOH)/37 °C/24 h | 1mol L−1 HCl |
| Oxidation | 30% Hydrogen peroxide (H2O2)/37 °C/24 h | 30% Sodium Metabisulfite (Na2S2O5) |
| Thermal stress | 100 °C/3 h |
2.7. Pharmaceutical applications of the developed analytical method
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000×g for 10 minutes. The supernatant was carefully removed, filtered through a 0.22 µm membrane filter, and the first filtrate was discarded to prevent adsorption. The filtrate was diluted with the mobile phase and analyzed by validated RP-HPLC. The drug concentration was calculated from the calibration curve, and solubility was expressed as the mean ± standard deviation (n = 3). All measures were taken to avoid exposure of the samples to light and to maintain a constant temperature throughout the experiment.16
000×g for 10 minutes. The supernatant was carefully removed, filtered through a 0.22 µm membrane filter, and the first filtrate was discarded to prevent adsorption. The filtrate was diluted with the mobile phase and analyzed by validated RP-HPLC. The drug concentration was calculated from the calibration curve, and solubility was expressed as the mean ± standard deviation (n = 3). All measures were taken to avoid exposure of the samples to light and to maintain a constant temperature throughout the experiment.16
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm to pellet the nanoparticles. The pellet was then harvested, resuspended in 1 mL of Milli-Q water, and sonicated for 10 minutes. Samples were diluted with filtered Milli-Q water and equilibrated at 25 ± 0.1 °C prior to analysis. Vesicle size and PDI were measured at a backscattering angle of 173° using a 633 nm laser, and the hydrodynamic diameter was calculated based on the Stokes–Einstein equation. Surface charge was measured using a folded capillary cell and calculated from electrophoretic mobility using the Smoluchowski approximation of Henry's equation. A PDI below 0.3 was considered indicative of a uniform dispersion, while a zeta potential magnitude ≥ ±30 mV indicated good colloidal stability. Measurements were performed in triplicate.17,18
000 rpm to pellet the nanoparticles. The pellet was then harvested, resuspended in 1 mL of Milli-Q water, and sonicated for 10 minutes. Samples were diluted with filtered Milli-Q water and equilibrated at 25 ± 0.1 °C prior to analysis. Vesicle size and PDI were measured at a backscattering angle of 173° using a 633 nm laser, and the hydrodynamic diameter was calculated based on the Stokes–Einstein equation. Surface charge was measured using a folded capillary cell and calculated from electrophoretic mobility using the Smoluchowski approximation of Henry's equation. A PDI below 0.3 was considered indicative of a uniform dispersion, while a zeta potential magnitude ≥ ±30 mV indicated good colloidal stability. Measurements were performed in triplicate.17,18
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm for 10 minutes to remove the lipid debris, and the supernatant was filtered through a 0.22 µm membrane filter. The filtrate was appropriately diluted with the mobile phase and analyzed using the validated RP-HPLC method. The drug loading efficiency at each time point was determined from the measured drug amount, and stability was expressed as the percentage of drug retained (mean ± SD, n = 3).
000 rpm for 10 minutes to remove the lipid debris, and the supernatant was filtered through a 0.22 µm membrane filter. The filtrate was appropriately diluted with the mobile phase and analyzed using the validated RP-HPLC method. The drug loading efficiency at each time point was determined from the measured drug amount, and stability was expressed as the percentage of drug retained (mean ± SD, n = 3).
2.8. Statistical analysis
All experiments were performed in triplicate unless otherwise stated, and results are expressed as mean ± standard deviation (SD). Statistical analysis was performed using OriginPro 2026. Linearity was evaluated using least squares regression analysis. Precision was expressed as a percentage relative standard deviation (%RSD). For solubility studies, statistical significance between groups was assessed using one-way analysis of variance (ANOVA), with p < 0.05 considered statistically significant.3. Results and discussions
3.1. Preliminary investigation: UV spectroscopic determination
In developing an accurate analytical technique, it is crucial to accurately locate the λmax of the target compound. As mentioned above, a scan was performed at 10 µg mL−1 for each drug over the 200–400 nm range. The λmax for BLN was located at 275 nm, as shown in Fig. 2. This value was used for quantification in the development of the RP-HPLC method.3.2. Method development: optimization and validation
3.2.2.1. Specificity. The specificity of the method was demonstrated by injecting the blank (placebo), theophylline (internal standard), baicalein (drug), and a mixture of both. The absence of co-eluting peaks at the BLN retention time confirms the high specificity of the developed method. This underscores the importance of peak purity and spectral confirmation for selectivity in complex matrices. The results demonstrate that the method can accurately distinguish BLN from excipients, internal standards, and degradation products (Fig. 3).
3.2.2.2. Evaluation of system suitability. The HPLC system was subjected to six equal injections of the system suitability solution to evaluate the resolution, retention time, column efficiency, and repeatability. Fig. 3 elucidates the standard chromatogram. The statistical test revealed no significant difference in area or retention time. The % RSD of these parameters was ≤ 2.0%, as presented in Table 3.
| Sample name | Area (mAUa min) | Retention time (tR, min) |
|---|---|---|
| a Values represent mean ± SD (n = 6), as per ICH Q2 (R2) guidelines. | ||
| BLN-SST-1 | 0.1307 | 4.633 |
| BLN-SST-2 | 0.126 | 4.637 |
| BLN-SST-3 | 0.1265 | 4.633 |
| BLN-SST-4 | 0.1273 | 4.637 |
| BLN-SST-5 | 0.1247 | 4.637 |
| BLN-SST-6 | 0.1296 | 4.637 |
| Mean | 0.127466667 | 4.636 |
| SD | 0.002270389 | 0.002065591 |
| % RSD | 1.781162997 | 0.044558664 |
3.2.2.3. Linearity, range, LOD, and LOQ. The developed method for quantifying BLN showed linearity over the concentration range of 0.0078–1 µg mL−1. The QC concentrations were selected to span the full required analytical range for different quantitation experiments, including drug release, drug entrapment, and permeability. The low QC concentration will provide sensitivity for initial burst-release samples, while the high QC concentration accounts for the maximum amount of unentrapped drug. This ensures that all unknown samples fall within the validated range, enabling precise quantitation. Linear regression analysis yielded an R2 value of 0.9998 for BLN, indicating extremely high linearity. The linear regression equation for BLN is y = 1.0292x − 0.0012. The graphical representation of the linear regression equation is presented in Fig. 4. The LOD and LOQ for BLN were 0.0069 and 0.0208 µg mL−1, respectively.
 | ||
| Fig. 4 (A) Linearity curve of the calibration standard samples of BLN and (B) graphical representation of the overlay of the calibration standard samples for linearity. | ||
The obtained linearity and low LOD/LOQ values demonstrate superior sensitivity compared to the reported HPLC methods for baicalein to date, which have reported higher detection limits due to matrix interference or less-optimized chromatographic conditions. The enhanced sensitivity of the present method is attributed to optimized mobile-phase composition and detection conditions, enabling reliable quantification at trace levels.
3.2.2.4. Accuracy. The accuracy of the developed analytical method was evaluated by spiking BLN with 80%, 100%, and 120% levels of 0.125 µg mL−1. Each experiment was performed in triplicate and analyzed for drug recovery (see Fig. 5). The results showed complete recovery of all samples, with an average recovery of 100.00 ± 2.00% and % RSD ≤ 2.00%. The overall mean recovery was 99.26% with a % RSD of 1.32%, indicating high accuracy of the analytical procedure used for quantifying BLN. The results are shown in Table 4. The low recovery variability further confirms the absence of matrix interference and underscores the method's reliability for quantitative analysis in both bulk and formulated systems.
| Day 1 | % Spike | Conc. (µg mL−1) | Conc. (µg mL−1) | % Recovery | % RSD |
|---|---|---|---|---|---|
| a Values represent mean ± SD (n = 3). | |||||
| Morning | 80 | 0.225 | 0.223 ± 0.001 | 99.063 ± 0.561 | 0.567 |
| 100 | 0.25 | 0.247 ± 0.003 | 98.769 ± 1.319 | 1.336 | |
| 120 | 0.275 | 0.275 ± 0.002 | 100.048 ± 0.728 | 0.727 | |
| Noon | 80 | 0.225 | 0.222 ± 0.004 | 98.631 ± 1.796 | 1.821 |
| 100 | 0.25 | 0.251 ± 0.001 | 100.285 ± 0.518 | 0.516 | |
| 120 | 0.275 | 0.275 ± 0.005 | 99.754 ± 1.658 | 1.662 | |
| Evening | 80 | 0.225 | 0.222 ± 0.003 | 98.833 ± 1.517 | 1.535 |
| 100 | 0.25 | 0.247 ± 0.004 | 98.653 ± 1.496 | 1.517 | |
| 120 | 0.275 | 0.272 ± 0.005 | 98.977 ± 1.717 | 1.734 | |
3.2.2.5. Precision. The system precision results demonstrated that the % RSD of the BLN peak area was ≤ 2.00%, well within the acceptance criteria. Table 5 provides the intra- and inter-day precision data for BLN, expressed as % RSD. These findings agree with established analytical standards and demonstrate that the method is robust enough for routine pharmaceutical analysis.
| Conc. (µg mL−1) | Intra-day precision | Inter-day precision | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Morning | Afternoon | Evening | Day 2 | Day 3 | ||||||
| % Rec | % RSD | % Rec | % RSD | % Rec | % RSD | % Rec | % RSD | % Rec | % RSD | |
| a Values represent mean ± SD (n = 9), % Rec. is % recovery. | ||||||||||
| 0.0625 | 100.68 ± 1.18 | 1.89 | 100.64 ± 1.21 | 1.65 | 100.57 ± 1.14 | 1.78 | 100.27 ± 1.34 | 1.58 | 98.77 ± 1.43 | 1.45 |
| 0.25 | 100.51 ± 0.74 | 0.737 | 99.01 ± 0.98 | 0.99 | 101.10 ± 1.91 | 1.94 | 100.71 ± 1.08 | 1.27 | 99.89 ± 0.64 | 0.65 |
| 1 | 101.73 ± 0.80 | 0.788 | 102.68 ± 0.37 | 0.36 | 101.45 ± 0.52 | 0.52 | 102.17 ± 0.19 | 0.187 | 101.99 ± 0.29 | 0.28 |
3.2.2.6. Solution stability. The stability of the stock solution at 1 µg mL−1 was determined over 4 days by comparing the % drug recovery of the QC samples before and after storage at room temperature (Table 6). No significant changes in BLN recovery were observed, and no degradation peaks were observed. This result indicates that BLN is stable in the mobile phase for 4 days at room temperature.
| Time (h) | % Recovery/% RSD |
|---|---|
| a Values represent mean ± SD (n = 3). | |
| 24 | 100.437 ± 0.399/0.397 |
| 48 | 101.937 ± 0.471/0.462 |
| 72 | 102.170 ± 0.191/0.187 |
| 96 | 101.992 ± 0.293/0.287 |
3.2.2.7. Robustness. The robustness of the proposed method was assessed by comparing the results with those of the standard BLN solution, and only slight differences were observed (Table 7 and Fig. 6). Even with intentional parameter changes, the optimized method demonstrated insignificant differences in the retention time, tailing factor, and theoretical plates. The system suitability factors were met under all robustness conditions, and the BLN peak tR remained unchanged. The BLN assay results remained within the acceptance limits. The differences in flow rate (±0.2 mL min−1), mobile phase composition (±2%), and pH (±0.2 pH units) were within the acceptance criteria, demonstrating the method's robustness. The method is robust.
| Flow rate | pH of the mobile phase | Mobile phase composition (ACN![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) water) water) |
||
|---|---|---|---|---|
| 3.0 | 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 50 |
|||
| Parameters | Retention time | Peak area | Tailing factor | Theoretical plates |
| a Values represent mean ± SD (n = 3). | ||||
| Optimized condition | ||||
| 1.0 mL min−1 | 4.636 ± 0.002 | 1.033 ± 0.004 | 1.27 ± 0.016 | 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 718 ± 91 718 ± 91 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||
| Change in flow rate | ||||
| 0.8 mL min−1 | 5.793 ± 0.005 | 1.277 ± 0.014 | 1.3 ± 0.006 | 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 463 ± 73 463 ± 73 |
| 1.2 mL min−1 | 3.868 ± 0.010 | 0.877 ± 0.005 | 1.2 ± 0.021 | 14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 758 ± 87 758 ± 87 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||
| Change in the pH of the mobile phase | ||||
| 2.8 | 4.630 ± 0.008 | 1.032 ± 0.046 | 1.29 ± 0.020 | 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 660 ± 113 660 ± 113 |
| 3.2 | 4.635 ± 0.007 | 1.005 ± 0.009 | 1.24 ± 0.017 | 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 801 ± 33 801 ± 33 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||
Change in mobile phase composition ACN![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) water water
|
||||
48![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 52 52 |
4.940 ± 0.003 | 1.002 ± 0.004 | 1.21 ± 0.021 | 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 358 ± 210 358 ± 210 |
52![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 48 48 |
4.370 ± 0.008 | 1.008 ± 0.005 | 1.29 ± 0.011 | 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 288 ± 45 288 ± 45 |
3.2.2.8. Forced degradation studies. Forced degradation studies were performed to assess the inherent stability of BLN and to understand its degradation characteristics under various chemical and thermal stress conditions. To the best of current knowledge, no previous study has systematically reported the degradation characteristics of BLN under various stress conditions. Although a few previous studies have indicated degradation under alkaline conditions, these views are not supported by any analytical data. The findings of the current study contradict these views. BLN samples were stressed under acidic (1 mol L−1 HCl), basic (1 mol L−1 NaOH), and oxidative (30% H2O2) conditions for 24 h at 37 °C, and under thermal stress at 100 °C for 3 h. Under acidic conditions, complete hydrolysis of BLN was observed, which was evident by the disappearance of the parent chromatographic peak and the absence of the retention time corresponding to the standard. However, partial hydrolysis was observed under alkaline conditions, with the predominant peak resembling the standard, indicating greater stability than previously thought. Moreover, negligible degradation was observed under oxidative and thermal stress conditions, indicating high resistance against peroxide-induced oxidation and elevated temperature (Fig. 7(A) and (B)). Taken together, these observations suggest a high susceptibility of BLN to acidic degradation, suggesting potential instability at gastric pH and, therefore, a limitation for direct oral delivery. However, low degradation even under strong alkaline conditions suggests adequate stability at mildly basic physiological environments. The forced degradation study provides valuable information for formulation development, storage, and handling practices, and also confirms the stability-indicating specificity of the developed chromatographic method for its intended use in analytical and bioanalytical studies. Compared with earlier studies without degradation profiling, the present work provides a more comprehensive evaluation, confirming the method's stability-indicating capability and its applicability in formulation development and stability studies.
3.3. Novelty of the method
The comparative analysis indicates that previously reported HPLC methods for baicalein predominantly employ buffer-based mobile phases, gradient elution, and multi-solvent systems, often resulting in longer retention times (6.6–60.41 min) and increased analytical complexity. While certain methods offer enhanced sensitivity, particularly µHPLC-ECD they require specialized instrumentation or extensive solvent use. In contrast, the present method utilizes a buffer-free, isocratic system with a significantly reduced retention time (∼4.6 min), while maintaining adequate sensitivity (LOD 0.0069 µg mL−1) and excellent linearity. Furthermore, unlike most reported methods focused on phytochemical or pharmacokinetic analysis, the proposed method uniquely integrates stability-indicating capability and applicability to nanopharmaceutical systems, thereby offering a more sustainable, efficient, and application-oriented analytical platform. A comparative analysis of the reported methods has been summarized in Table 8.| Analytes | Mobile phase | Mode | tRBLN (min) | Linearity range (µg mL−1) | LOD/LOQ (µg mL−1) | Detector | Key features | Limitations vs. present method | Ref. |
|---|---|---|---|---|---|---|---|---|---|
| a Where, NR – not reported, FA – formic acid, MeOH – methanol, pg – picograms, ng – nanograms. | |||||||||
| Baicalein |
Water (pH 3, OPA): ACN (50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/i_char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/i_char_2009.gif) 50) 50)
|
Isocratic | ∼4.6 | 0.0078 –1 | 0.0069/0.0208 | HPLC- DAD | Green, stability-indicating, nano-applicable | Moderate sensitivity compared to ECD-based methods | Present method |
| Baicalein, paclitaxel | MeOH![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Milli Q water (with 0.1% FA) (30 Milli Q water (with 0.1% FA) (30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 70) 70) |
Isocratic | 4.3 | 0.019–10 | 0.119/0.361 | HPLC-UV | Simultaneous dual drug estimation | Isocratic elution and higher organic solvent consumption | 6 |
| Baicalein, chrysin, wogonin | MeOH-ACN-Milli Q water: OPA (30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 38 38![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 60 60![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
Isocratic | 6.6 | 0.5–400 | 0.4/1.2 | HPLC-UV | Multi-flavonoid analysis | Complex mobile phase, longer runtime | 7 |
| Baicalein, baicalin | ACN-Milli Q water (40![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 60) (with 0.05% OPA) 60) (with 0.05% OPA) |
Isocratic | 20 | 0.08–10 | 0.08/0.04 | HPLC-UV | Pharmacokinetic analysis | Higher organic solvent consumption, matrix complexity | 8 |
| Baicalein, baicalein-7-glucuronide | MeOH-ACN-phosphate buffer | Gradient | 15 | 1–10 | 0.03/0.05 | HPLC-UV | Metabolite profiling | Gradient system, longer analysis time, buffer use | 9 |
| Baicalein, baicalin | MeOH- Milli Q water (with 0.5% OPA) | Isocratic | 9.1 | 1.4 pg to 2.7 ng | 0.54 pg/14 pg | Micro HPLC (µHPLC)-ECD | High sensitivity detection | Requires specialized detector | 10 |
| Baicalein | ACN/Milli Q water/FA (21/78/1, v/v/v): ACN/Milli Q water/FA (80/19/1, v/v/v) | Gradient | 60.41 | NR | 3.75 ng/12.5 ng | HPLC-DAD | Extremely high sensitivity | Extremely long runtime, high solvent consumption | 20 |
| Baicalein, phytochemicals | MeOH-ACN-Milli Q water (with 1% OPA) | Isocratic | 12.31 | 5.3–37.1 | 0.546/NR | HPLC-UV | Herbal matrix analysis | Longer runtime | 21 |
3.4. Application of the analytical method in pharmaceutical formulation
 | ||
| Fig. 8 Graphical representation of the solubility of BLN in water as compared to various organic solvents. Values represent mean ± SD (n = 3). | ||
The results of the repeated-measures ANOVA showed that solvent selection significantly influenced the analytical response (p < 0.0001), with exceptionally low replicate variance, indicating high experimental reproducibility. The post-hoc analysis revealed that all the organic solvents produced significantly higher responses than water, with the order of magnitude being methanol ≈ ethanol ≫ isopropanol ≫ water.
The solubility profiling has important pharmaceutical implications, establishing that BLN belongs to the poorly water-soluble class of compounds, underscoring the need for solubility enhancement approaches for successful formulation. The solubility data directly support the choice of lipid-based and vesicular carrier systems, such as liposomes, where drug distribution into the lipid bilayer can enhance solubility, stability, and bioavailability. Thus, the developed HPLC method serves not only as a quantitative analytical technique but also as a valuable preformulation tool necessary for rational formulation development.
The observed poor aqueous solubility of BLN is consistent with its classification as a BCS Class II compound and aligns with previously reported literature. The significantly higher solubility in organic solvents further confirms its lipophilic nature. Similar findings have been reported in studies that employed lipid-based and vesicular systems to enhance the solubility and bioavailability of poorly water-soluble drugs. These results strongly justify the selection of nanoliposomal carriers for improving BLN delivery.
| Time (Days) | Drug loading (%) |
|---|---|
| a Values represent mean ± SD (n = 3). | |
| 0 | 5.92 ± 0.016 |
| 7 | 5.65 ± 0.003 |
| 15 | 5.32 ± 0.021 |
| 30 | 5.54 ± 0.046 |
Overall, the developed RP-HPLC method demonstrated excellent analytical performance, including high sensitivity, precision, accuracy, and stability-indicating capability. A comparative evaluation with the existing literature highlights its advantages, including reduced analysis time, elimination of buffer systems, and improved environmental compatibility. Furthermore, the successful application of the method in nanoliposomal systems underscores its versatility in both analytical and formulation domains. The integration of multi-metric greenness assessment further strengthens its relevance in modern sustainable analytical chemistry. Collectively, these findings establish the method as a robust, eco-conscious, and application-oriented analytical tool with significant potential for routine pharmaceutical analysis and advanced drug delivery research.
4. Greenness assessment for the developed analytical method
The environmental sustainability of the analytical procedure before its application in the laboratory is an important consideration aimed at reducing the discharge of hazardous materials. Currently, there has been a strong focus on prioritizing the greenness of analytical procedures, and reliable methods are now available to assess the environmental sustainability. The current study uses six methods to validate the environmental sustainability of the developed method: the MoGAPI,22 VIGI,23 CaFRI,24 CACI,25 BAGI,26 and the AGREE.27–294.1. BAGI assessment
The BAGI tool, used in conjunction with the Perfect Green Indicator, assesses the applicability and functionality of an analytical technique using 10 defined parameters. The pictorial representation in the BAGI tool exemplifies the process, divided into phases: characteristics 1–3 (analytical determination), characteristics 4–5 (sample preparation), and characteristics 6–10 (analytical procedure). A total score of 60 or more is required to define the method's pertinence. The developed method yielded a score of 75 (Fig. 10(a)). | ||
| Fig. 10 Blueness of the developed RP-HPLC method was evaluated by (A) BAGI and Greenness by (B) MoGAPI, (C) VIGI, (D) CaFRI, (E) CACI, and (F) AGREE scores. | ||
4.2. MoGAPI
The Green Analytical Procedure Index (GAPI), launched in 2018, evaluates the sampling and analysis phases of analytical procedures. As GAPI does not have a complete numerical greenness score, it was modified in 2024 to MoGAPI (Modified GAPI). GAPI has a five-point color-coded pictogram for the environmental effect of each step, while MoGAPI gives a numerical pentagram score for easier evaluation. A score of ≥75 is “excellent green”. In the current work, MoGAPI assigned the proposed method a greenness score of 85, indicating a highly environmentally compatible approach (Fig. 10(b)).4.3. VIGI tool
To provide a comprehensive evaluation, in addition to the existing green, blue, and red rankings, VIGI combines ten criteria: sample preparation and instrumentation, data processing and software, white analytical chemistry and derivatives, regulatory compliance, materials and reagents, miniaturization, automation, interdisciplinarity, sensitivity, and approach. The method's innovation score is represented by a star-shaped decagon in a survey. The overall VIGI score, ranging from 1 to 10, is calculated as the geometric mean of all criteria scores. VIGI received a total score of 55, giving emphasis to its distinctiveness, as shown in Fig. 10(c).4.4. CaFRI
The Carbon Footprint Reduction Index (CaFRI) is an online, free tool that evaluates and reduces the carbon footprint of analytical procedures in chemical labs, with carbon footprint as the primary concern and accounting for both method requirements and laboratory-specific information. The result is a value from 0 to 100 (77 at present). The main parameters are energy consumption (total electrical energy and number of samples), energy production emissivity, carbon footprint reduction methods, chemical consumption, sample storage and transportation, employee participation, and waste management/recycling. CaFRI is a questionnaire-based tool that estimates these parameters and displays the results using a human-foot pictogram in red (low), yellow (medium), and green (good) colours to represent performance and encourage emission reduction, connecting analytical chemistry with sustainability (Fig. 10(d)).4.5. CACI
The evaluation is intended to assess the feasibility and usability of established analytical methods. Unlike conventional parameters, the CACI provides a comprehensive analysis of operational factors, including sample size, preparation, feasibility, applicability, portability, sensitivity, and automation. The results are presented using a diagnostic pictogram, where coloured areas indicate excellent performance, grey areas indicate moderate performance, and black areas indicate non-compliance or failure to meet criteria. The resultant score obtained was 80 (Fig. 10(e)).4.6. AGREE
Green designable software was employed to evaluate agreement with the 12 green principles of analytical chemistry. The software program provides a final score ranging from 0 to 1 using a 12-sector pictogram. The colours of the sectors (green, yellow, red) indicate the relative importance and score of each criterion, with darker colours indicating higher importance. For the new RP-HPLC method, the pictogram score was 0.64 (Fig. 10(f)), indicating its eco-friendliness.Overall, the sustainability of the developed RP-HPLC method was assessed using six different tools, and the results confirmed its superior greenness. The method scored 75 on the BAGI, which is adequate for operational feasibility, and has a streamlined analytical process with less sample preparation. The MoGAPI score of 85 categorized the method as “excellent green” due to the solvent's low toxicity, low consumption, and low hazardous-waste generation. The VIGI score of 55 indicated that the method is innovative and advanced while maintaining regulatory compatibility and automation. The CaFRI value of 77 supported the reduction in carbon emissions due to shorter analysis time, ambient conditions, and lower energy use. Additionally, the high CACI score of 80 supported the method's high usability, sensitivity, and applicability in the laboratory. Finally, the AGREE score of 0.64 supported the method's high compliance with the twelve principles of Green Analytical Chemistry. Taken together, the results show that the developed method combines environmental sustainability with analytical efficiency and feasibility, making it a highly efficient green analytical method for pharmaceutical analysis.
5. Conclusions and outlook
An RP-HPLC method has been developed and validated for the quantitative estimation of BLN in accordance with ICH guidelines, covering specificity, linearity, precision, accuracy, LOD/LOQ, robustness, and stability-indicating capability. The forced degradation study under various stress conditions demonstrated the method's ability to selectively separate BLN from its degradation products, thereby establishing its suitability for stability studies. The standard and sample solutions were found to be stable for up to 4 days at room temperature, thus establishing the method's reliability for routine analytical work. The validated method establishes it as a reliable tool for BLN integrity, potency, and consistency testing during its shelf life and for routine use in pharmaceutical quality control laboratories. The method has wider applications in formulation development and preformulation studies. Despite these advantages, certain limitations should be acknowledged. The use of acetonitrile contributes to a moderate environmental footprint. Additionally, the study is limited to laboratory-scale validation and specific nanoformulation systems, and further work is required to assess scalability and broader applicability. The method's stability-indicating capability enables monitoring of BLN degradation across various formulations, including suspensions, nanoparticles, liposomes, and solid dispersions, thereby facilitating optimization of excipient compatibility and storage conditions. The method can also be used in solubility enhancement studies to estimate BLN in various solubilizing agents, such as cosolvents, surfactant systems, complexation systems, and lipid-based carriers, thereby allowing the selection of appropriate drug delivery systems. In the future, the method can be used for dissolution profiling, in vitro release kinetics, degradation pathway studies, and scale-up process monitoring, and can be used as a model analytical method for bioanalytical method adaptation or therapeutic product development for BLN. Currently, our lab is also working on the solubility enhancement strategies of BCS class II drugs, including BLN.Author contributions
Sweta Acharya: data curation, investigation, writing – original draft, writing – review and editing, methodology, software, validation. Ankit Jain: project administration, supervision, writing – original draft, writing – review and editing.Conflicts of interest
There are no conflicts to declare.Abbreviations
| % RSD | % Relative standard deviation |
| ACN | Acetonitrile |
| AGREE | Analytical GREEnness |
| AUC | Area under curve |
| BAGI | Blue applicability grade index |
| BA | Bioavailability |
| BCS | Biopharmaceutical classification system |
| API | Active pharmaceutical ingredient |
| BDS | Base-deactivated silica |
| BLN | Baicalein |
| CACI | Click analytical chemistry index |
| CaFRI | Carbon footprint reduction index |
| DAD | Diode array detector |
| H2O2 | Hydrogen peroxide |
| HCl | Hydrochloric acid |
| HQC | Higher quality control |
| HSPC | Hydrogenated soy phosphatidylcholine |
| ICH | International council for harmonisation of technical requirements for pharmaceuticals for human use |
| LOD | Limit of detection |
| LOQ | Limit of quantification |
| LQC | Lower quality control |
| MoGAPI | Modified green analytical procedure index |
| MQC | Middle quality control |
| Na2S2O5 | Sodium metabisulfite |
| NaOH | Sodium hydroxide |
| OPA | Orthophosphoric acid |
| PDI | Polydispersity index |
| QC | Quality control |
| RP-HPLC | Reversed-phase high-performance liquid chromatography |
| tR | Retention time |
| UV | Ultra-violet |
| VIGI | Violet innovation grade index |
Data availability
The data supporting the findings are presented within the paper, and additional data may be requested from the authors.Acknowledgements
Dr Jain would like to express his sincere gratitude to the New Faculty Seed Grant (NFSG/PIL/2024/P3865) from the Birla Institute of Technology and Science, Pilani. Additionally, Ms Acharya is grateful to BITS-Pilani for awarding her an institutional fellowship for the PhD program. We are also grateful to Lipoid® GmbH, Ludwigshafen, Germany, for providing the gift sample of S PC-3 (Phosphatidylcholine, hydrogenated, content ≥ 98.0%), CAS-No. 97281-48-6. Figures were created using Biorender software under an academic license.References
- T. Arunagiri, H. K. Chanduluru, K. P. Kannaiah and R. H. Obaydo, Microchem. J., 2025, 219, 116016 CrossRef CAS.
- S. Bhujbal, I. D. Rupenthal and P. Agarwal, Methods, 2024, 231, 178–185 CrossRef CAS PubMed.
- Validation of Analytical ProceduresQ2(R2), ICH Harmonised Guideline, 2023 Search PubMed.
- R. Shukla, V. Pandey, G. P. Vadnere and S. Lodhi, in Bioactive Food as Dietary Interventions for Arthritis and Related Inflammatory Diseases, ed. R. R. Watson and V. R. Preedy, Academic Press, 2019, pp. 293–322, DOI:10.1016/b978-0-12-813820-5.00018-0.
- J. Xing, X. Chen, Y. Sun, Y. Luan and D. Zhong, J. Pharm. Pharmacol., 2005, 57, 743–750 CrossRef CAS PubMed.
- P. K. Yadav, A. K. Tiwari, R. Saklani, D. Chauhan, R. Rana, P. Yadav, K. Mishra, A. S. Kedar, Wahajuddin, J. R. Gayen and M. K. Chourasia, Bioanalysis, 2022, 14, 1005–1020 CrossRef CAS PubMed.
- Z. Gharari, K. Bagheri, H. Danafar and A. Sharafi, J. Appl. Res. Med. Aromat. Plants, 2020, 16, 100232 Search PubMed.
- M. Y. Lai, S. L. Hsiu, S. Y. Tsai, Y. C. Hou and P. D. Chao, J. Pharm. Pharmacol., 2003, 55, 205–209 CrossRef CAS PubMed.
- L. Zhang, G. Lin and Z. Zuo, J. Pharm. Biomed. Anal., 2004, 36, 637–641 CrossRef CAS PubMed.
- A. Kotani, S. Kojima, H. Hakamata and F. Kusu, Anal. Biochem., 2006, 350, 99–104 CrossRef CAS PubMed.
- M. C. Oh, M. J. Piao, P. M. Fernando, X. Han, S. R. Madduma Hewage, J. E. Park, M. S. Ko, U. Jung, I. G. Kim and J. W. Hyun, Biomol. Ther., 2016, 24, 616–622 CrossRef CAS PubMed.
- M. Brits, F. Bouwer and J. M. Viljoen, Methods Protoc., 2026, 9, 16 CrossRef PubMed.
- S. K. Prajapati, A. Jain and M. Bajpai, Anal. Methods, 2024, 16, 5280–5287 RSC.
- A. S. de Oliveira, J. L. S. de Oliveira, F. de Souza Dias, L. de Souza, D. de Andrade Santana and A. Júnior, Biointerface Res. Appl. Chem., 2021, 11, 13089–13101 CAS.
- K. B. Vikhe and S. S. Sonawane, J. Bionanosci., 2026, 16, 117 CrossRef.
- W. Ma, F. Du, T. Xu, X. Wang, B. Li, H. Yue, M. Jia and A. Zong, Food Hydrocolloids, 2026, 172, 112023 CrossRef CAS.
- C. L. Putta, H. S. Buddhiraju, D. N. Yadav, A. Basa and A. K. Rengan, ACS Appl. Bio Mater., 2025, 8, 661–675 CrossRef CAS PubMed.
- A. M. Gaber, M. Tsakiri, H. Katifelis, M. Gazouli and C. Demetzos, J. Liposome Res., 2025, 35, 345–356 CrossRef CAS PubMed.
- N. Baltz and R. Scherließ, OpenNano, 2025, 24, 100251 CrossRef CAS.
- M. Navarro Escamilla, F. Rodenas Sanz, H. Li, S. A. Schönbichler, B. Yang, G. K. Bonn and C. W. Huck, Talanta, 2013, 114, 304–310 CrossRef CAS PubMed.
- M. Zaveri, A. Khandhar and S. Jain, Eurasian J. Anal. Chem., 2008, 3(2) CAS.
- F. R. Mansour, J. Płotka-Wasylka and M. Locatelli, Analytica, 2024, 5, 451–457 CrossRef.
- A. Fuente-Ballesteros, V. Martinez-Martinez, A. M. Ares, S. Valverde, V. Samanidou and J. Bernal, Anal. Chem., 2025, 97, 6946–6955 CrossRef CAS PubMed.
- F. R. Mansour and P. M. Nowak, BMC Chem., 2025, 19, 121 CrossRef CAS PubMed.
- F. R. Mansour, A. Bedair and M. Locatelli, Adv. Sample Prep., 2025, 14, 100164 CrossRef.
- N. Manousi, W. Wojnowski, J. Płotka-Wasylka and V. Samanidou, Green Chem., 2023, 25, 7598–7604 RSC.
- D. Mandale, Y. Rana, R. Savaliya and N. Chauhan, Discov. Chem, 2026, 3, 36 CrossRef.
- D. Yallappa, V. K. Yuvaraaj, N. Sharma and A. Jain, Microchem. J., 2026, 220, 116684 CrossRef CAS.
- F. Pena-Pereira, W. Wojnowski and M. Tobiszewski, Anal. Chem., 2020, 92, 10076–10082 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |