 Open Access Article
Open Access ArticleMechanosynthesis of a bismuth nanoparticle-decorated palm kernel mesocarp fiber (Bi-PKMF): adsorption of Pb(II), kinetics and thermodynamics
Amarachi Udoka Nkwoada *a,
Simeon Chukwudozie Nwanonenyi
*a,
Simeon Chukwudozie Nwanonenyi b,
Callistus Izunna Iheme
b,
Callistus Izunna Iheme c,
Ali Bilar
c,
Ali Bilar a,
Ijeoma Akunna Duru
a,
Ijeoma Akunna Duru a,
Tochukwu Ifeanyi Nwakile
a,
Tochukwu Ifeanyi Nwakile a,
Divinefavour Anioke
a,
Divinefavour Anioke a,
Janefrances Oluebube Onwuemeodo
a,
Janefrances Oluebube Onwuemeodo a,
Chukwuemeka Fortunatus Nnadozie
a,
Chukwuemeka Fortunatus Nnadozie a,
Celestine Chidubem Chukwunonyerem
a,
Celestine Chidubem Chukwunonyerem a,
Favour Ogadinma Izuagba
a,
Favour Ogadinma Izuagba a and
Uchenna Gift Nwaneri
a and
Uchenna Gift Nwaneri a
a
aDepartment of Chemistry, Federal University of Technology, Owerri, Nigeria. E-mail: amarachi.nkwoada@futo.edu.ng
bDepartment of Polymer and Textile Engineering, Federal University of Technology, Owerri, Nigeria
cDepartment of Biochemistry, Federal University of Technology, Owerri, Nigeria
First published on 5th May 2026
Abstract
A new bismuth-decorated palm kernel mesocarp fiber (Bi-PKMF) adsorbent was prepared through green liquid-assisted mechano-chemical grinding, followed by thermal crystallization. Detailed characterization (SEM-EDX, TEM, BET, FTIR, and AFM) was performed to verify the uniform dispersion of bismuth oxide nanoparticles in the lignocellulosic matrix and the production of a high-surface-area mesoporous material (387 m2 g−1 and a pore size of 12.5 nm). Control experiments of the ball-milled PKMF indicated that bismuth functionalization provides chemical enhancement rather than mechanical enhancement. Bi-PKMF exhibited excellent Pb(II) adsorption capacity (112.3 mg g−1), which was 231 times higher than that of pristine PKMF (48.5 mg g−1) with more than 90% removal in 60 min at pH 6. The kinetics followed the pseudo-second-order model (R2 = 0.999), and the obtained Dubinin–Radushkevich adsorption energy (12.6 kJ mol−1) confirmed chemisorption. The mesoporous structure reduces the resistance to intraparticle diffusion, allowing access to the active sites (Bi–O, –OH, and –COO–). Spontaneous (ΔG° < 0), exothermic (ΔH° = −29.02 kJ mol−1), and ordered adsorption were indicated by the thermodynamic parameters. The reusability of Bi-PKMF was good, retaining over 90% of its adsorption capacity after five regeneration cycles. The key to the increased Pb(II) uptake is surface chemistry, rather than physical structure, which makes Bi-PKMF a high-performance biosorbent for sustainable water purification.
1 Introduction
One of the most urgent environmental issues of the twenty-first century is water quality degradation by heavy metals, particularly mercury (Hg), arsenic (As), cadmium (Cd), and lead (Pb).1,2 These industrial effluent, mining, and agricultural runoff toxicants disrupt aquatic ecosystems and bioaccumulate in the food chain, ultimately threatening human health through dermal contact, ingestion, and inhalation.3 New developments in the treatment of wastewater have seen a paradigm shift from traditional adsorbents towards novel functionalized biosorbents and nanoparticle-modified materials, such as polystyrene/periwinkle shells,4 zeolite/periwinkle shells,5 bismuth-based materials,6 and functionalized palm kernel fruit fibers and palm kernel shell fibers,7 as innovative and functionalized biosorbents and nanoparticle-decorated materials.Among the heavy metals, lead (Pb(II)) is especially toxic, has no known biological role, and causes harmful effects even at minute concentrations. Epidemiological studies have proven that blood lead levels (BLLs) of 5 µg dL−1 in children are linked with reduced intelligence quotient, neurodevelopmental impairment, and behavioural disorders, while chronic exposure to lead in adulthood is related to hypertension, kidney dysfunction, and reproductive toxicity.8 Importantly the U.S. Centers for Disease Control and Prevention (CDC) claims that there is no safe BLL; the neurological effects are evident at 5.0 g dL−1.9 Lead concentrations exceeding 400 mg kg−1 in contaminated soils can cause up to 42% root growth retardation10 and pose long-term hazards through food chain transfer and ecosystem degradation; thus, there is an urgent need to develop effective lead remediation techniques.11
The palm oil industry also produces large amounts of lignocellulosic biomass waste, especially the palm kernel shell (PKS) and palm kernel mesocarp fiber (PKMF). These substances are commonly underused or burnt in the air, which leads to greenhouse gas emissions and particulate pollution.12 Recent studies have demonstrated that PKS and PKMF can be employed as inexpensive biosorbents to remove heavy metals because they possess abundant active functional groups (hydroxyl, carboxyl, and phenolic) on their surfaces.13,14 Under optimized conditions, unmodified PKS exhibits Pb(II) adsorption capacities of about 43.12 mg g−1 and removal efficiencies over 99%.15 Further functionalization assists chemical removal; for example, sulfonation can remove about 95% of Pb(II) by improving surface chemistry and increasing the active-site density.14 Despite these developments, the following severe constraints still remain: heterogeneous surface chemistry, inadequate nanoscale active sites, and absence of integrated surface-active agents in lignocellulosic matrices.16,17 These deficiencies limit the performance in complicated wastewater matrices where several pollutants are present. Moreover, the majority of the current literature is devoted to the use of oil palm waste derivatives as palm kernel shell activated carbon,16 biocatalysts,18 and modified PKS adsorbents19,20 without addressing these critical challenges. Thus, in this work, we made a direct attempt to investigate the synthesis of functionalized materials from palm kernel mesocarp fiber (PKMF) using bismuth for Pb(II) removal.
This is due to the ability of bismuth (Bi) to form a wide range of interfacial bonds, provide a large surface area, and create defect sites in adsorbent materials as a result of nano-bismuth oxide (Bi–O) doping and incorporation into heterostructures.21 Nanomaterials made of bismuth have emerged as promising environmental remediation agents due to their distinct physicochemical characteristics, including low toxicity, high chemical stability, and affinity to heavy metal oxyanions and cations.22 Bismuth-doped nanocomposites exhibit improved adsorption rates due to the synergistic nature of the adsorption process, namely surface complexation, ion exchange, and electrostatic interaction. An example is the use of bismuth-impregnated sludge-derived biochar, which showed a high uranium(VI) adsorption capacity (516.5 mg g−1 and 93.8% removal) through the affinity of bismuth towards oxyanions, which was 21 times higher compared to undoped biochar (average capacity of 100–300 mg g−1).23 Similarly, bismuth-doped metal oxides (PBCuO) have been found to exhibit excellent activity for the photocatalytic degradation (93.10%) of organic pollutants owing to their low band gaps (2.43 eV) and larger surface areas (88.90 m2 g−1).24 These achievements indicate that the incorporation of bismuth species in PKMF matrices is likely to overcome the drawbacks of pure biosorbents, producing hybrid products that possess enhanced adsorptive capacity. Therefore, herein, we present the synthesis of nanoparticle bismuth-doped palm kernel mesocarp fiber (PKMF) via mechano-synthesis, which enables uniform doping, nanoscale integration, and improved interfacial properties to remove Pb(II).
Traditional techniques for producing nanocomposites (hydrothermal and sol–gel) normally require high temperatures (>200 °C), long reaction times, and toxic solvents, which are incompatible with the concept of green chemistry.25 Alternatively, mechanochemical synthesis, especially liquid-assisted grinding (LAG), is another promising option because it uses mechanical energy to induce solid–state reactions at ambient temperature with low solvent concentrations (η < 1–2 µL mg−1).26 This facilitates the homogenous integration of bismuth species in biomass. The obtained materials possess enhanced surface areas, designed porosity, and high concentration of active sites, qualities that are vital in effective Pb(II) sequestration. Furthermore, LAG mechano-synthesis can be economically scaled, is environmentally friendly, and can be applied to agricultural waste, aligning with the principles of a circular economy while producing materials with nanoscale dispersion without aggregation or disruption of the lignocellulosic structure.27
Although palm kernel shell adsorbents and bismuth-based materials have been studied extensively as individual adsorbents, no reported study employed bismuth-functionalized palm kernel mesocarp fiber in the removal of Pb(II), which has been a significant missed opportunity to capitalize on the low-cost nature of the PKMF with the high adsorption capability of bismuth nanoparticles through green mechanochemical synthesis. In response to this omission, the current study was guided by the following questions: can bismuth oxide nanoparticles be selectively functionalised on PKMF via a solution-minimised mechanochemical-thermal process? How would bismuth functionalisation affect the physicochemical properties of PKMF? Would the adsorption capacity of Bi-PKMF be enhanced compared with that of pristine PKMF? How would the operating parameters affect its performance? Which kinetic, isotherm, and thermodynamic models best describe the adsorption process? What drives the mechanism, and would Bi-PKMF be regenerable for multiple adsorption–desorption cycles28? To this end, the detailed aims of this study were to synthesize Bi-PKMF via liquid-assisted mechanochemical grinding and subsequent thermal crystallization, to thoroughly characterize it using the SEM-EDX, TEM, XRD, AFM, FTIR, and BET techniques, to evaluate its performance for Pb(II) adsorption under varying conditions, to model its adsorption kinetics, isotherms, and thermodynamics, and to assess its reusability. Bi-PKMF was found to be an excellent adsorbent for the removal of heavy metal ions from water, requiring a very short time of only 1 h to remove Pb(II), with a removal efficiency of ≥90.02% compared with that of ≤85.01% for PKMF.
2 Materials and methods
2.1 Chemicals and reagents
Elaeis guineensis (palm kernel) is derived from the oil palm tree and was sourced from a local oil mill in Orlu, Imo State, Nigeria. Analytical grade bismuth(II) nitrate pentahydrate (Bi(NO3)3·5H20, 98%, CAS no. 10035-06-0) and lead nitrate (Pb(NO3)2, 98%, CAS: 10099-74-8) were supplied by Gate Lab Chemicals Ltd and were used as received without further purification. A stock solution of Pb2+ (1000 mg L−1) was formulated by dissolving 1.598 g of Pb(NO3)2 in a small amount of deionized water (DI water, resistivity 18.2 MΩ cm) with the addition of 1 mL of concentrated HNO3 to inhibit hydrolysis and make the solution stable. It was then transferred to a volumetric flask and quantitatively diluted to 1000 mL with deionized water. Serial dilution of the stock solution using DI water was used to prepare working solutions of desired concentrations, which were prepared daily to ensure accuracy and limit degradation. The pH of the solution was kept at 7.0 by adding 0.1 M HCl or 0.1 M NaOH accordingly. All solutions were made in DI water, and glassware was thoroughly rinsed with deionized water before use.2.2 Palm kernel mesocarp fiber (PKSF) preparation
300 g of palm kernel mesocarp fiber (PKMF) was collected from a local oil mill after red palm oil was completely extracted from the mesocarp, and the material was sundried for 6 h a day over 3 continuous days at temperatures ≥28 °C.29,30 The palm kernel mesocarp fiber was dried in the sun, then solvent-extracted in ethanol with a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 solid-to-solvent ratio, and stirred at 50–60 °C to extract the remaining oils. The fiber was recovered by filtration, and further washing with hot deionized water at 80–90 °C was repeated to remove traces of the solvent and any water-soluble impurities in the filtrate until a clear filtrate was obtained. The palm kernel mesocarp fiber (PKMF) was dried in an oven at 105 °C to a constant weight of 267.5 g and placed in a desiccator before proceeding to the preparation of the nano-adsorbent. The PKMF was manually cut with sanitized scissors to 1–2 cm and then milled using an IKA MF 10 basic grinder with a 2 mm sieve to a size of ≤250 µm.
10 solid-to-solvent ratio, and stirred at 50–60 °C to extract the remaining oils. The fiber was recovered by filtration, and further washing with hot deionized water at 80–90 °C was repeated to remove traces of the solvent and any water-soluble impurities in the filtrate until a clear filtrate was obtained. The palm kernel mesocarp fiber (PKMF) was dried in an oven at 105 °C to a constant weight of 267.5 g and placed in a desiccator before proceeding to the preparation of the nano-adsorbent. The PKMF was manually cut with sanitized scissors to 1–2 cm and then milled using an IKA MF 10 basic grinder with a 2 mm sieve to a size of ≤250 µm.
2.3 Synthesis of the bismuth-decorated palm kernel mesocarp fiber (Bi-PKMF)
The functionalization of the palm kernel mesocarp fiber (PKMF) was carried out through a two-step mechanochemical-thermal process on the cleaned and milled fibers.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. The mixture was mixed in a planetary ball mill (Retsch PM 100) at 350 rpm over a period of 4 h. This is a high-energy reaction necessary to physically mix, reduce the particle size, and chemically initiate the interaction between the bismuth precursor and the fibrous substrate, forming a slurry.32
1. The mixture was mixed in a planetary ball mill (Retsch PM 100) at 350 rpm over a period of 4 h. This is a high-energy reaction necessary to physically mix, reduce the particle size, and chemically initiate the interaction between the bismuth precursor and the fibrous substrate, forming a slurry.322.4 Materials and analytical methods
2.5 Adsorption experiments
The amount of aqueous Pb(II) ions in the adsorption experiments was determined by atomic absorption spectroscopy (AAS) on a PinAAcle 900F spectrometer (PerkinElmer Inc., USA). The samples were analyzed using instrument calibration with freshly prepared Pb(II) standard solutions in the concentration range of 10–100 mg L−1 to provide a calibration curve with a correlation coefficient (R2) greater than 0.999. All measurements were repeated three times and, to ensure the reliability of the analysis, procedural blanks were measured and compared to the samples to confirm that there was no contamination.| qe = (C0 − Ce)V/m | (1) |
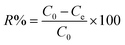 | (2) |
3 Results and discussion
3.1 Bi-PKMF characterization
In addition, the elemental composition of the pristine PKMF and Bi-PKMF was measured using energy-dispersive X-ray spectroscopy at five randomly chosen locations on each sample to ensure representative and statistically significant results. The elemental compositions of both samples are presented in the form of mean values with standard deviations to reflect the homogeneity of the elemental distribution across the surfaces of the samples. As shown in Table 1, pristine PKMF is dominated by 58.7 ± 2.8 wt% and 38.2 ± 2.1 wt% of carbon and oxygen, respectively, which is typical of lignocellulosic biomass, with trace amounts of naturally occurring mineral elements, such as silicon, aluminium, potassium, and calcium, introduced into the biomass during the growth and processing of palm kernels. After mechanochemical bismuth functionalization, as shown in Table 2, Bi-PKMF still contained carbon and oxygen as its main components with contents of 52.4 ± 3.2 wt% and 31.8 ± 2.5 wt%, respectively, while the bismuth content of 12.6 ± 1.8 wt% is relatively high and uniformly distributed across the Bi-PKMF structure. This also confirms the successful homogeneous distribution of bismuth species across the fiber structure based on TEM and EDX mapping analysis. The minor decrease in the weight percentage of carbon and oxygen in Bi-PKMF relative to pristine PKMF is due to the dilution effect caused by the addition of bismuth in the lignocellulosic structure. It is important to note that the trace rhenium signals detected in both samples, with contents of about 0.7–0.8 wt%, are artefacts resulting from the rhenium-tungsten sputter coating target used in the preparation of the SEM samples to increase their conductivity, and thus do not reflect the elemental components in the adsorbent materials. The overall elemental analysis verifies that PKMF was successfully functionalized with bismuth, and that compositional reproducibility was achieved using the liquid-assisted mechanochemical synthesis methodology.
| Element | Weight% (mean ± SD) | Atomic% (mean ± SD) |
|---|---|---|
| a Rhenium (Re) signals (originally ∼0.7 wt%) arising from the sputter coating were excluded from these calculations; values are normalized to Re-free composition. | ||
| Carbon (C) | 59.1 ± 2.7 | 67.5 ± 2.5 |
| Oxygen (O) | 38.5 ± 2.0 | 33.0 ± 1.8 |
| Silicon (Si) | 1.6 ± 0.4 | 0.8 ± 0.2 |
| Aluminium (Al) | 0.8 ± 0.3 | 0.4 ± 0.2 |
| Potassium (K) | 0.4 ± 0.2 | 0.1 ± 0.1 |
| Calcium (Ca) | 0.3 ± 0.1 | 0.1 ± 0.1 |
| Element | Weight% (mean ± SD) | Atomic% (mean ± SD) |
|---|---|---|
| a Rhenium (Re) signals (originally ∼0.8 wt%) arising from the sputter coating were excluded from these calculations; values are normalized to Re-free composition. | ||
| Carbon (C) | 52.8 ± 3.1 | 65.1 ± 2.8 |
| Oxygen (O) | 32.1 ± 2.4 | 29.7 ± 2.0 |
| Bismuth (Bi) | 12.7 ± 1.7 | 3.2 ± 0.5 |
| Silicon (Si) | 1.8 ± 0.4 | 1.0 ± 0.2 |
| Aluminium (Al) | 0.9 ± 0.3 | 0.5 ± 0.2 |
| Calcium (Ca) | 0.5 ± 0.2 | 0.2 ± 0.1 |
This contrast arises from the substantial difference in electron density between the high-atomic-number bismuth (Z = 83) and the low-atomic-number constituents (carbon, oxygen, and hydrogen) in the lignocellulosic biomass. The micrographs display the bismuth-based nanoparticles, where the sizes are about 10 to 50 nm in diameter and uniformly distributed and attached to the fibrous palm kernel mesocarp matrix. This confirms the effectiveness of the mechanochemical and thermal synthesis in the realization of nanoscale integration with minimal aggregation, which is typical of biomass-based composites. The proximity of the high-aspect-ratio particles to the fiber substrate reduces the interfacial resistance, thereby maximizing the available active sites.22 Although some micron-scale agglomerates are visible in the SEM images (likely due to drying), high-resolution TEM confirms that the size of the primary bismuth oxide nanoparticles remains in the 10–50 nm range and they are uniformly distributed on the nanoscale.
Nanostructures are conducive to the adsorption kinetics because the small particle size reduces the internal diffusion barriers to Pb(II) ions and thus enables surface complexation of these ions within a short time. The suggested large surface area and clearly exposed bismuth oxide phases are associated with the predicted excellent adsorption ability, as similar nano-structuring of bismuth-decorated biochar has been demonstrated to increase the rapid uptake of pollutants.35 This engineered nanodispersion is a key structural feature that explains the superior removal performance of Bi-PKMF compared with the pristine fiber or micron-scale composites.36
Furthermore, to validate the incorporation of bismuth into the palm kernel mesocarp fibers, as observed by SEM and TEM analyses, energy-dispersive X-ray (EDX) elemental mapping was conducted, as shown in Fig. 3. The representative SEM image of Bi-PKMF (Fig. 3a) illustrates a fibrous morphology, with visible bismuth-containing nanoparticles (lighter-contrast regions) decorating the surface.
This morphology is typical of the regions analysed by EDX mapping (Fig. 3a–e), confirming that the bismuth distribution shown in the maps corresponds to the fiber substrate rather than isolated agglomerates. The EDX elemental mapping presents convincing and straightforward visual evidence of the effective and uniform incorporation of bismuth into the palm kernel mesocarp fiber structure, confirming the synthesis and innovative structure of this material. The individual elemental maps clearly show the distribution of carbon and oxygen throughout the fibrous structure, which is characteristic of lignocellulosic biomass, whereas the bismuth map (Fig. 3c) clearly shows uniformly distributed red hotspots that are characteristic of the decorated nanoparticles. Importantly, the composite overlay (Fig. 3d) indicates a consistent combination in which bismuth signals (yellow) are closely entangled with the carbonaceous fiber structure (green), and there is no evidence of macroscopic phase separation. Quantitative line profile analysis (Fig. 3e) was performed across the composite overlay to assess the spatial correlation between bismuth and the fiber matrix. The overlapping Bi, C and O peaks along the transect confirm that the bismuth-containing nanoparticles are not merely deposited on the surface but are intimately embedded within the lignocellulosic framework, consistent with the TEM observations.
This co-localization was determined using the line profile analysis, which gives a quantitative measure by revealing areas of high carbon and oxygen content through simultaneous peaks in bismuth intensity. This spatial correlation directly supports the TEM data on extensively dispersed nanoparticles and the explanation of the high adsorption capacity, where the homogeneous distribution results in the maximum number of active sites for complexing Pb(II). This directly demonstrates the effectiveness of liquid-assisted mechanochemical synthesis to achieve nanoscale doping and interfacial bonding, as proposed.37 The combination of TEM imaging and quantitative EDX elemental mapping herein presents strong and direct proof regarding the homogeneous assembly of bismuth oxide nanoparticles in the palm kernel mesocarp fiber matrix, visually supporting the idea of functionalization and mechanochemical synthesis of homogeneous biosorbents.23,36,38
Low intensity, weak, and broadened diffraction peaks may arise from trace mineral components (i.e., iron or silica-containing impurities) naturally occurring in agro-waste biomass, but these reflections lack insufficient intensity and resolution to obtain any reliable quantitative phase information.37 As a result, Rietveld refinement or percentage determination of phases was not performed. Interestingly, crystalline bismuth oxide (e.g., α-Bi2O3 or β-Bi2O3) phases are not associated with any specific diffraction peaks.40 This suggests that the bismuth species can either be amorphous or present as ultra-fine nanoparticles below the XRD detection limit, as identified by the TEM micrographs, which showed their effective nanoscale dispersion in the biomass matrix. These results show that XRD is a qualitative method to confirm structural disorder and nanoscale integration, rather than for quantifying the crystalline phase.22 This interpretation is consistent with the literature,41 which reports that amorphous bismuth oxide (A-BiO) prepared via a hydrothermal method exhibits a broad featureless XRD pattern with no sharp peaks, even though bismuth is present in significant quantity.39 In the XRD pattern of Bi-PKMF, the broad halo from the lignocellulosic matrix overlaps with and dominates the scattering from any amorphous or nanocrystalline bismuth oxide. The TEM analysis (Fig. 2) confirms the presence of 10–50 nm bismuth oxide nanoparticles, which may be polycrystalline or poorly crystalline, leading to peak broadening below the detection limit. Alternatively, the nanoparticles observed by TEM could be aggregates of even smaller crystallites. Therefore, XRD does not contradict the successful bismuth functionalisation but rather indicates that the bismuth oxide is either amorphous or nanocrystalline with very small domain sizes.
These results reveal a nanoscale surface topography with varying degrees of roughness.38 The monotonic increase in surface roughness correlates with the BET surface areas (85 → 245 → 387 m2 g−1) and supports the conclusion that ball milling physically activates the fiber surface, while bismuth functionalisation introduces additional nanoscale textural features. The AFM data do not directly resolve individual cellulose microfibrils; rather, they quantify the overall nanoscale roughness that enhances the density of accessible adsorption sites.42
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching of carbonyl groups appears at 1627 cm−1. The peak at 1034 cm−1 is attributed to polysaccharide stretching in the lignocellulosic palm kernel fiber matrix, consistent with a similar study.40
O stretching of carbonyl groups appears at 1627 cm−1. The peak at 1034 cm−1 is attributed to polysaccharide stretching in the lignocellulosic palm kernel fiber matrix, consistent with a similar study.40
 | ||
| Fig. 6 (a) FTIR spectrum and (b) identification of functional groups in Bi-PKMF and confirmation of bismuth oxide integration for enhanced adsorption. | ||
Importantly, the sharp peak at 534 cm−1 corresponds to Bi–O stretching vibrations, providing direct spectroscopic evidence for the integration of bismuth oxide (Bi2O3) nanoparticles into the fibrous structure. In aqueous environments, hydroxylation of these Bi–O sites produces bismuthol groups (Bi–OH), which are high-affinity binding sites for Pb(II) via ligand exchange and inner-sphere complexation. This represents a distinct type of active site that is not found in pure PKMF.38
This chemical fingerprint indicates that the material surface contains not only the inherent oxygen-containing functional groups of the biomass, but it also has newly formed bismuth-related sites.32 These hybridized sites play a key role in enhancing the adsorption of Pb(II) ions via synergistic adsorption by surface complexation and ion exchange. The incorporation of bismuth generates more and highly active binding sites compared to native hydroxyl and carboxyl groups, which form the basis of palm kernel shell adsorbents and are found in other bismuth-decorated bio-sorbents that display better affinity to heavy metal cations.22 This detailed surface chemistry correlates with the synthesis strategy and the high removal efficiency of the material for lead in contaminated water.
These features have direct and important implications for adsorption. Both high surface area and the mesoporous network provide a high density of available active sites, low diffusion resistance, and rapid kinetics for Pb(II) ion transport to the binding sites.43 The excellent performance of this material can be explained by this synergistic structure with the high surface area of activated carbons and customizable functional sites of bismuth nanomaterials. It has a higher surface area than raw palm kernel shell (<100 m2 g−1)30 and is comparable to other developed bismuth-functionalized adsorbents,32 where surface areas of 300–500 m2 g−1 are associated with outstanding heavy metal uptake rates. Therefore, the BET data confirm that the synthesis strategy is effective, demonstrating that Bi-PKMF is a high-surface-area, porous adsorbent, not merely surface-modified fibers.
As observed in Table 3, the as-received PKMF exhibits a moderate specific surface area of 85 m2 g−1 and a total pore volume of 0.021 cm3 g−1, which are typical of untreated lignocellulosic biomass with low porosity and compressed fiber structure, consistent with the values in a prior study on raw palm kernel materials.44 After ball milling, BM-PKMF exhibited an impressive 188% increase in surface area to 245 m2 g−1 and an over twofold increase in pore volume to 0.045 cm3 g−1, indicating that the high-energy mechanochemical process efficiently alters the hierarchical lignocellulosic architecture, exposing internal surfaces, creating new pore networks by fragmenting fibres, and delaminating them, as often seen in the literature.31,34 This physical modification directly translated into an enhanced adsorption performance, where BM-PKMF showed a Langmuir maximum adsorption capacity of 78.3 mg g−1 for Pb(II), which is 61% higher than that of pristine PKMF, resulting from the increased accessibility of native oxygen-containing functional groups, including hydroxyl and carboxyl moieties exposed on the newly formed surfaces.
| Parameter | PKMF (as-received, washed) | BM-PKMF (ball-milled only) | Bi-PKMF (ball-milled with Bi) |
|---|---|---|---|
| Surface area (m2 g−1) | 85 ± 8 | 245 ± 12 | 387 ± 15 |
| Pore volume (cm3 g−1) | 0.021 ± 0.003 | 0.045 ± 0.004 | 0.068 ± 0.005 |
| Pb(II) Q0 (mg g−1) | 48.5 ± 2.1 | 78.3 ± 3.2 | 112.3 ± 4.1 |
| Removal at 50 mg L−1 (%) | 85.0 ± 1.5 | 91.2 ± 1.2 | 96.3 ± 1.1 |
| Time to 90% removal (min) | 180 | 120 | 60 |
Interestingly, the introduction of bismuth via liquid-assisted grinding using bismuth nitrate and subsequent thermal crystallization produced Bi-PKMF, with further improved textural characteristics, achieving a surface area of 387 m2 g−1 and pore volume of 0.068 cm3 g−1. These values correspond to enhancements of 58% and 51% relative to BM-PKMF, respectively, indicating that the bismuth species present during mechanochemical processing inhibit agglomeration of the particles and pore collapse and can further increase interfacial interactions.
Importantly, the adsorption capacity of Bi-PKMF is 43% and 231% higher than that of BM-PKMF and pristine PKMF, respectively, indicating that bismuth functionalization contributes a significant chemical improvement to the physical advantages of milling. The synergistic effect of increased surface area from mechanochemical activation and the incorporation of bismuth oxide species to provide additional high-affinity binding sites through surface complexation mechanisms is responsible for the improved performance of Bi-PKMF. This is supported by the presence of Bi–O stretching vibrations at 534 cm−1 in its FTIR spectrum,38 along with the homogenous dispersion of nanoscale bismuth particles observed in the TEM images and EDX elemental mapping.37 Moreover, Bi-PKMF exhibits a kinetic advantage, with the shortest time to reach 90% removal of 60 min compared to BM-PKMF and pristine PKMF, which take 120 and 180 min, respectively. These control studies clearly demonstrate that although mechanochemical processing alone has a significant beneficial effect on the adsorptive ability of palm kernel mesocarp fiber in terms of physical activation and increased surface area, further bismuth functionalization introduces a distinct chemical contribution, resulting in a hybrid material with an overall performance unattainable by either mechanical or chemical modification alone.
3.2 Effects of adsorption parameters
At equilibrium, Bi-PKMF (∼96%) exhibits a higher percentage removal (than PKMF, at the same point, of 92%), owing to its increased surface area and the presence of numerous active sites (supplied by bismuth oxide nanoparticles), thus leading to faster and more complete adsorption. These findings validate that Bi-PKMF has greater capacity and better kinetics, making it more efficient for time-sensitive water treatment systems.22
3.3 Adsorption isotherms of PKMF and Bi-PKMF
Table 4 shows the adsorption isotherm parameters (Langmuir, Freundlich, Temkin, and Dubinin–Radushkevich (D–R)) for Pb(II) ion removal by the PKMF and Bi-PKMF adsorbents. The adsorption isotherms were constructed using the mean equilibrium uptake (qe) from triplicate experiments at each initial concentration.| Models | Parameters | PKMF | Bi-PKMF |
|---|---|---|---|
| Experimental data (at C0 = 100 mg L−1) | Ce (mg L−1) | 24.8 | 8.6 |
| Langmuir model | Q0 (mg g−1) | 48.5 | 112.3 |
| bL (L mg−1) | 0.085 | 0.142 | |
| R2 | 0.992 | 0.998 | |
| Freundlich model | 1/n | 0.62 | 0.54 |
| kf (mg−1/n L1/n g−1) | 12.8 | 32.5 | |
| R2 | 0.978 | 0.985 | |
| Temkin model | B (J mol−1) | 985 | 645 |
| bT (J mol−1) | 125 | 198 | |
| Kt (L g−1) | 2.45 | 5.67 | |
| R2 | 0.965 | 0.974 | |
| Dubinin–Radushkevich model | qD (mg g−1) | 44.2 | 105.8 |
| E (kJ mol−1) | 9.2 | 12.6 | |
| R2 | 0.960 | 0.970 |
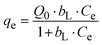 | (3) |
The higher Q0 value for Bi-PKMF (112.3 mg g−1) compared with PKMF (48.5 mg g−1) indicates significantly enhanced maximum adsorption capacity due to bismuth oxide incorporation, which provides additional active sites.22 The bL values reflect strong affinity between Pb(II) ions and the adsorbent surfaces, with Bi-PKMF showing greater affinity. The close agreement between the Langmuir maximum capacity (Q0) and experimental uptake at the initial highest concentration (100 mg L−1) proves that the studies in the batch were under near-saturation conditions.
These results confirm the experimental layout and the credibility of the obtained Langmuir parameters. A comparison was performed to categorize the heavy metal removal capacity of the as-synthesized Bi-PKMF and PKMF in this work, as tabulated in Table 5. The comparative study in Table 5 clearly shows that the novel nanoparticle Bi-PKMF performed better, with a Pb(II) adsorption capacity of 112.3 mg g−1 is exceptional. This is more than twice that of the unmodified PKMF (48.5 mg g−1) and is much higher than that of the previously reported palm kernel shell-derived adsorbents, including functionalized derivatives, which commonly exhibit capacities of less than 50 mg g−1.
| S/no | Adsorbate | Adsorbent | Adsorption capacity (qe) | References |
|---|---|---|---|---|
| 1 | Pb2+ | Bi-PKMF | 112.3 mg g−1 | Current study |
| 2 | Pb2+ | PKMF | 48.50 mg g−1 | Current study |
| 3 | Pb2+ | Palm kernel shell (PKS) | 49.62 mg g−1 | 15 |
| 4 | Cd2+ | Palm kernel shell (PKS) | 42.12 mg g−1 | 15 |
| 5 | Cr3+ | Palm kernel shell (PKS) | 49.65 mg g−1 | 15 |
| 6 | Zn2+ | Palm kernel shell (PKS) | 41.72 mg g−1 | 15 |
| 7 | Cd2+ | Functionalized PKS | 39.00 mg g−1 | 14 |
| 8 | Pb2+ | Functionalized PKS | 40.00 mg g−1 | 14 |
| 9 | Zn2+ | Functionalized PKS | 37.00 mg g−1 | 14 |
| 10 | Cr6+ | Functionalized PKS | 40.00 mg g−1 | 14 |
This outstanding improvement is directly linked to the novel liquid-assisted mechanochemical synthesis, which enables the molar-scale uniform integration of bismuth oxide.27 The resulting material is a high-surface-area, multifunctional material with numerous active sites for surface complexation and ion exchange. Moreover, the enhanced adsorption capacity of the base PKMF was better than that reported in a previous study, demonstrating the efficiency of the optimized cleaning and milling protocol in maintaining25 and accessing the native lignocellulosic structure of the fiber as a superior foundational matrix, which was further doped with bismuth.17
| qe = Kf·C1/ne | (4) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) qe and log
qe and log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ce can be used to determine Kf and n. Consequently, 1/n values smaller than 1 indicate normal adsorption with considerable contact between the adsorbent and the metal, whereas 1/n values greater than one indicate cooperative adsorption. The 1/n values were <1 for both materials, indicating favorable adsorption. The lower 1/n value for Bi-PKMF (0.54) suggests a more heterogeneous surface with stronger binding sites introduced by bismuth doping. The higher Kf for Bi-PKMF (32.5) confirms its superior adsorption intensity and capacity compared with PKMF;15 the higher R2 value of Bi-PKMF also indicates a good fit to the adsorption isotherm.
Ce can be used to determine Kf and n. Consequently, 1/n values smaller than 1 indicate normal adsorption with considerable contact between the adsorbent and the metal, whereas 1/n values greater than one indicate cooperative adsorption. The 1/n values were <1 for both materials, indicating favorable adsorption. The lower 1/n value for Bi-PKMF (0.54) suggests a more heterogeneous surface with stronger binding sites introduced by bismuth doping. The higher Kf for Bi-PKMF (32.5) confirms its superior adsorption intensity and capacity compared with PKMF;15 the higher R2 value of Bi-PKMF also indicates a good fit to the adsorption isotherm.
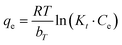 | (5) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) qe versus E2. The mean adsorption energy (E) provides information about chemical and physical adsorption.
qe versus E2. The mean adsorption energy (E) provides information about chemical and physical adsorption.
ln = qe = ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) qm − βε2 qm − βε2
| (6) |
The E values for PKMF (9.2 kJ mol−1) and Bi-PKMF (12.6 kJ mol−1) are both >8 kJ mol−1, indicating that the adsorption process is governed by chemisorption.49 This supports the findings of the Langmuir and pseudo-second-order models. The higher E value for Bi-PKMF indicates a stronger chemisorption process, which is consistent with bismuth oxide-integrated nanoparticles, enhancing surface complexation with Pb(II) ions.38 In addition, the data for Bi-PKMF fitted the D–R isotherm better than PKMF, which agrees with the “physisorption isotherm of N2 (Fig. 7) that has a Type IV(a) shape and distinctive H3-type hysteresis loop between the relative pressure (P/P0) of 0.45 to 0.95, typical of mesoporous materials having slit-shaped pores being formed by aggregation of plate-like particles or fibrous structures”, as confirmed in Section 3.1.6. The D–R model offers a very important understanding of the nature of the active sites. It is worth noting that the E value is 12.6 kJ mol−1 in the case of Bi-PKMF, indicating that the added bismuth oxide sites cause greater chemical bonding with Pb(II) and the oxygen functionalities present in the original compound. This energetic difference is evidence that the bismuth sites are not just extra surface area but are structurally different, higher-energy active sites.
3.4 Adsorption kinetics on PKMF and Bi-PKMF
We employed four adsorption kinetic models including pseudo-first-order kinetics (Lagergren model), pseudo-second-order kinetics (Ho–McKay model), Elovich model, and intraparticle diffusion model (Weber–Morris model) to explain the adsorption mechanism of Pb(II) ions on PKMF and Bi-PKMF. Each data point in the isotherm (Ce, qe) and kinetic (t, qt) curves represents the mean of replicate measurements (typically n = 3). According to pseudo-first-order kinetics, adsorption is proportional to the amount of vacant adsorption sites, indicating that adsorption mostly occurs through physical interactions (physisorption) and that its rate is governed by mass transfer at the solid–liquid interface, and its formula is shown as follows:
log(qe − qt) = log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) qe − (K1/2.303)t qe − (K1/2.303)t
| (7) |
| Models | Parameters | PKMF | Bi-PKMF |
|---|---|---|---|
| Pseudo-first order | K1 (min)−1 | 0.0042 | 0.065 |
| qe (mg g−1) | 22.50 | 23.80 | |
| R2 | 0.981 | 0.989 | |
| Pseudo-second order | K2 (g mg−1min−1) | 0.0028 | 0.0045 |
| qe (mg g−1) (calculated) | 23.00 | 24.00 | |
| qe (mg g−1) (experimental) | 23.12 | 24.44 | |
| R2 | 0.998 | 0.999 | |
| Elovich model | α (mg min−1) | 4.20 | 8.60 |
| β (g mg−1) | 0.28 | 0.22 | |
| R2 | 0.963 | 0.972 | |
| Intra particle diffusion | K1 | 1.85 | 2.40 |
| C (mg g−1) | 5.2 | 8.1 | |
| R2 | 0.958 | 0.966 |
The pseudo-second-order model is frequently utilized in the study of adsorption systems, in which the rate-limiting step can be chemisorption through either exchange or sharing of valence forces, such as electron sharing or exchange.46 Hence, the exceptionally good fit of our data to this model (Table 6) is consistent with a chemisorption-mediated process.
This rate depends on the square of the number of unoccupied sites and its equation is shown in eqn (8), where K2 is the rate constant (g mg−1 min−1). By plotting t/qt vs. time (t), with this equation, qe and K2 can be determined from the intercept and slope.
| t/qt = 1/K2qe2 + 1/qet | (8) |
A chemisorptive mechanism is further substantiated by FTIR spectroscopy (Section 3.1.5, Fig. 6), where the decrease or shift in the BiO vibration band at approximately 534 cm−1 after Pb(II) adsorption suggests direct interactions between the bismuth oxide sites on the surface and the metal ions.45,48
Furthermore, the adsorption kinetics were investigated using the Elovich model. The Elovich model assumes that adsorption occurs on a heterogeneous surface and that the activation energy of adsorption increases exponentially with surface coverage, as is typical of chemisorption on non-uniform sites of varying energy, as expressed in eqn (9). In this model, α is the initial adsorption rate and β is the adsorption coefficient, and the plot of qt vs. ln (t) is used to determine the constants α and β, as shown in Table 6.
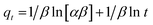 | (9) |
The higher α for Bi-PKMF reflects a higher initial adsorption rate than PKMF, while the lower β suggests a more heterogeneous surface with abundant active sites due to bismuth incorporation.32
To characterize solute transfer in solid–liquid adsorption, intraparticle diffusion was implemented to determine the adsorption mechanism. The intraparticle diffusion Weber–Morris model assumes that adsorption is controlled, at least in part, by diffusion of adsorbate molecules within the pores of the adsorbent rather than solely by surface reactions. The theoretical equation is given in eqn (10).
| qt = Kidt21 + C | (10) |
In brief, the high rate of Pb(II) adsorption by Bi-PKMF (90% in 60 min) and its Dubinin–Radushkevich adsorption energy of 12.6 kJ mol−1 offer a seemingly paradoxical situation as chemisorption is expected to occur more slowly when diffusion is the rate-limiting step. This inconsistency is resolved by the mesoporous architecture of the material. The BET analysis showed a pore size of 12.5 nm (Section 3.1.6), which can easily accommodate hydrated Pb(II) ions (≈0.4–0.5 nm) with very low steric hindrance. As a result, the resistance to intraparticle diffusion is greatly reduced, including a high Weber–Morris intraparticle diffusion rate constant (Kid = 2.40 mg g−1 min−1/2, Table 6). The diffusion barrier has been eliminated, and the rate-limiting step is now the surface chemisorption reaction at the numerous abundant Bi–O and –COO− sites. This is exactly the case with the pseudo-second-order kinetic model (R2 > 0.999), where the adsorption rate is determined by the square of the concentration of unoccupied sites, a rate law that is surface reaction-controlled. Therefore, the combination of chemical affinity (large E value) and physical accessibility (mesoporous structure) helps in understanding the strength and rate of Pb(II) sequestration by Bi-PKMF.
3.5 Thermodynamics of adsorption
Entropy (ΔS0), free energy (ΔG0), and enthalpy (ΔH0) are important thermodynamic parameters. The thermodynamics of Pb(II) adsorption on PKMF and Bi-PKMF were evaluated at different temperatures (298.15 K, 308.15 K, 318.15 K and 328.15 K) using the following equation:
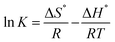 | (11) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) K−1. The free energy of specific adsorption, ΔG0 (kJ mol−1), was calculated using the following equation:
K−1. The free energy of specific adsorption, ΔG0 (kJ mol−1), was calculated using the following equation:| ΔG° = ΔH° − TΔS° | (12) |
Negative ΔG° values at all temperatures in Table 7 confirm that the adsorption of Pb(II) on PKMF and Bi-PKMF is spontaneous. The decrease in the ΔG° value with increasing temperature indicates reduced spontaneity at higher temperatures, consistent with the observed decrease in removal efficiency. The negative ΔH° value confirms that it is an exothermic adsorption reaction, where stronger adsorption occurs at lower temperatures. The more negative values of ΔH for Bi-PKMF indicate a higher binding energy associated with bismuth oxide nanoparticles, which provide more active sites to participate in surface complexation and ion exchange. The −ΔS° value indicates that the randomness at the solid–liquid interface decreases in the process of adsorption, since the Pb(II) ions are bound to the adsorbent surface. The larger negative −ΔS° value of Bi-PKMF (−77.07 J mol−1 K−1) than that of PKMF (−49.32 J mol−1 K−1) suggest an ordered adsorption process, probably because of the organized nanoscale structure and enhanced site heterogeneity arising from bismuth doping. These thermodynamic findings verify the strong performance of Bi-PKMF for the remediation of Pb(II)-contaminated water at low temperatures, supporting its practical usability and economic viability.
| Adsorbent | T (K) = T (°C) + 273.15 | K−1 (L g−1) | ΔG° (kJmol−1) | ΔH° (kJmol−1) | ΔS° (J mol−1 K−1) | R2 |
|---|---|---|---|---|---|---|
| PKMF | 25 | 5.75 | −4.33 | −19.01 | −49.32 | 0.991 |
| PKMF | 35 | 4.50 | −3.86 | −19.01 | −49.32 | 0.991 |
| PKMF | 45 | 3.35 | −3.20 | −19.01 | −49.32 | 0.991 |
| PKMF | 55 | 2.83 | −2.84 | −19.01 | −49.32 | 0.991 |
| Bi-PKMF | 25 | 12.00 | −6.16 | −29.02 | −77.07 | 0.987 |
| Bi-PKMF | 35 | 7.83 | −5.27 | −29.02 | −77.07 | 0.987 |
| Bi-PKMF | 45 | 5.06 | −4.29 | −29.02 | −77.07 | 0.987 |
| Bi-PKMF | 55 | 4.05 | −3.81 | −29.02 | −77.07 | 0.987 |
3.6 Desorption and regeneration studies
To evaluate the practical reusability and economic viability of the synthesized adsorbent, desorption and multi-cycle regeneration experiments were conducted.• Adsorption: 50 mL of 50 mg L−1 Pb(II) at pH 6 was treated with 0.3 g of adsorbent for 2 h.
• Separation washing: The adsorbent was dried, rinsed using DI water, and filtered.
• Desorption/regeneration: 50 mL of 0.1 M HNO3 was stirred with the loaded adsorbent for 4 h.
• Recovery and neutralization: The regenerated adsorbent was filtered, flushed with DI water until the filtrate became neutral pH (∼6), and dried overnight at 60 °C. The efficiency of Pb(II) removal% per adsorption cycle was calculated and compared. The control experiment involved a parallel experiment with DI water as a desorption agent.
3.7 Regeneration and reusability studies
The usefulness of an adsorbent is not limited to its original capacity but also its regenerability. The screening of the eluent showed that acidic and chelating agents were effective for desorbing Pb(II) from Bi-PKMF. The desorption efficiencies were evaluated with 0.1 M HNO3 (92.5%), 0.1 M HCl (88.1%), 0.1 M Na2-EDTA (81.7%), and 0.1 M CH3COOH (65.3%). The superior performance of HNO3 could be explained by its high ionic strength and capacity to protonate the active sites on the adsorbent (e.g., Bi–O and –COO–), reversing the surface complexation and ion exchange interactions between Pb(II) and the adsorbent. The DI water control had negligible desorption (≤5%), which proved that the binding was strong and non-physical. All regeneration experiments were conducted in triplicate to ensure statistical reliability, and the results are presented as the mean ± standard deviation (Fig. 9). The amount of Pb(II) adsorbed (q, mg g−1) was determined at the end of each cycle to measure the capacity retention in excess of percentage removal. Pristine PKMF exhibited a gradual decrease in adsorption capacity, with a value in cycle 1 of 7.09 ± 0.15 mg g−1, which decreased to 5.71 ± 0.23 mg g−1 in cycle 5, indicating a 19.5% loss and cumulative degradation and irreversible binding of the sites. The increase in standard deviation (between ± 0.15 to ± 0.23) indicates the increasing heterogeneity of the material performance in terms of lignocellulosic matrix degradation, probably because of the uneven lignocellulosic matrix degradation over time as a result of repeated exposure to acid. Conversely, Bi-PKMF exhibited a significantly more stable performance, and its capacity only declined from 7.51 ± 0.14 mg g−1 to 8.03 0.10 mg g−1 throughout the five cycles, just a 6.5% change. The standard deviations (±0.10–0.14) are low, which indicates the synthesis reproducibility and the strength of the bismuth-functionalized material. The high capacity retention of Bi-PKMF is attributed to the fact that bismuth oxide nanoparticles are anchored on the fiber matrix, which prevents their leaching in acids and preserves the lignocellulosic structure beneath them. These results indicate that Bi-PKMF is not only a high-capacity adsorbent but also a highly regenerable and durable material that can be used in real water treatment. The findings verify that Bi-PKMF is not only a high-capacity adsorbent but also a robust and recyclable adsorbent, which is essential in cost-effective and sustainable water treatment processes.3.8 Proposed mechanism of adsorption
There are two sources of active sites in Bi-PKMF, the palm kernel mesocarp fiber lignocellulosic matrix and the incorporated bismuth oxide nanoparticles, as shown in Fig. 10. Fourier transform infrared (FTIR) spectroscopy gives direct evidence of these features. The broad band at 3282 cm−1 corresponds to the O–H stretching band of the alcoholic and phenolic hydroxyl groups of cellulose and lignin. These hydroxyl groups can engage in metal binding by proton exchange and the formation of monodentate or bidentate surface complexes. The peak at 1627 cm−1 is attributed to the carbonyl stretching, which is mainly caused by carboxylic acid groups.The carboxylic acid moieties (–COOH) in lignocellulosic materials typically have pKa values in the range of 4.0–5.0. At the working pH of 6, these groups are predominantly, but not completely, deprotonated to form carboxylate anions (–COO−). The partially deprotonated surface provides negatively charged sites that can electrostatically attract cationic Pb(II) ions and participate in chelation. Additionally, the remaining protonated –COOH groups can engage in hydrogen bonding or ligand exchange with Pb(II) after deprotonation induced by metal binding. Thus, the adsorption mechanism involves dynamic equilibrium between protonated and deprotonated forms rather than a single fully deprotonated state.
This assignment is directly supported by the pH-dependent adsorption profile (Fig. 8a), where the removal efficiency is low when conditions are highly acidic, and when the conditions are not acidic but deprotonation is preferential (i.e., at pH 6). Most importantly, the broad peak at 534 cm−1 in the spectrum of Bi-PKMF is associated with Bi–O stretching vibrations, which is clear spectroscopic evidence of the incorporation of bismuth oxide. These Bi–O sites are surface-hydroxylated to bismuthol groups (Bi–OH) in aqueous environments, which are a unique and high-affinity class of binding sites lacking in the pristine PKMF. These bismuthol complexes are capable of ligand–exchange reactions with Pb(II) ions to produce inner-sphere surface complexes of the form ![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) Bi–O–Pb or (
Bi–O–Pb or (![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) Bi–O)2–Pb, which are stronger covalently directed than the interactions of the native oxygen functionalities alone. The predominant role of surface chemistry or physical structure in the process of metal adsorption can now be resolved quantitatively using experimental data. The mean free energy of adsorption (E) is the quantity obtained from the Dubinin–Radushkevich (D–R) isotherm model, which represents the difference between physical and chemical sorption. In the case of pristine PKMF, the E value of 9.2 kJ mol−1 is greater than the set limit of 8 kJ mol−1, and thus, chemisorption prevails. Upon bismuth functionalization, the E value significantly increases to 12.6 kJ mol−1. This increase is not just incremental but reflects a qualitatively stronger chemical interaction, which can be directly attributed to the greater bond energy in Pb(II) complexation at the Bi–O positions than in natural oxygen groups. The doubling of the maximum adsorption capacity (from 48.5 to 112.3 mg g−1) cannot be attributed solely to the effect of surface area, but instead suggests some new, stronger binding processes. In addition, the pseudo-second-order kinetic model, which shows excellent agreement with the experimental data with correlation coefficients greater than 0.999, is fundamentally based on the assumption that the rate-limiting step involves chemisorption through valence forces via electron sharing or exchange.
Bi–O)2–Pb, which are stronger covalently directed than the interactions of the native oxygen functionalities alone. The predominant role of surface chemistry or physical structure in the process of metal adsorption can now be resolved quantitatively using experimental data. The mean free energy of adsorption (E) is the quantity obtained from the Dubinin–Radushkevich (D–R) isotherm model, which represents the difference between physical and chemical sorption. In the case of pristine PKMF, the E value of 9.2 kJ mol−1 is greater than the set limit of 8 kJ mol−1, and thus, chemisorption prevails. Upon bismuth functionalization, the E value significantly increases to 12.6 kJ mol−1. This increase is not just incremental but reflects a qualitatively stronger chemical interaction, which can be directly attributed to the greater bond energy in Pb(II) complexation at the Bi–O positions than in natural oxygen groups. The doubling of the maximum adsorption capacity (from 48.5 to 112.3 mg g−1) cannot be attributed solely to the effect of surface area, but instead suggests some new, stronger binding processes. In addition, the pseudo-second-order kinetic model, which shows excellent agreement with the experimental data with correlation coefficients greater than 0.999, is fundamentally based on the assumption that the rate-limiting step involves chemisorption through valence forces via electron sharing or exchange.
All these pieces of evidence together prove that the determining factor in Pb(II) uptake is surface chemistry. Nevertheless, the outstanding physical characteristics of Bi-PKMF cannot be neglected. BET analysis confirmed a high specific surface area of 387 m2 g−1, and the well-developed mesoporous network with the major pore diameter of 12.5 nm plays an indispensable role. The large surface area serves as an amplifier to maximize the concentration of accessible active sites per unit mass of adsorbent, while the mesoporous architecture reduces the diffusion barriers and provides speedy routes for the transport of Pb(II) ions to the internal binding sites. In the absence of a high surface area, most of the chemical sites would be buried and inaccessible; conversely, without chemical sites, the surface would bind metals with only moderate, non-specific affinity via physisorption. Thus, the overall adsorption process on Bi-PKMF can be described as a multi-step process that combines both physical transport and chemical binding.
Initially, the diffusion of Pb(II) ions to the surface of the adsorbent occurs via film diffusion, which is aided by the large surface area and the good concentration gradient that causes the movement. The ions then gradually enter the interior of the adsorbent particles via the mesopore network, in which the pore size of probably 12.5 nm is large enough to allow access to all the active sites within the material without significant steric hindrance. Upon approaching the vicinity of a binding site, the prevailing retention process is determined by the local surface functionality. On the surface of bismuth oxide, the surface-hydroxylated Bi–OH groups participate in ligand exchange with Pb(II), releasing protons and forming stable inner-sphere complexes. This is the most energetically favourable and strongest interaction, which is consistent with the high D–R adsorption energy and the exothermic enthalpy change of −29.02 kJ mol−1. Simultaneously, at the carboxylate sites, the negatively charged –COO− groups will electrostatically attract Pb(II) cations and may form chelate rings via coordination with the metal centre. The hydroxyl groups are involved in proton exchange of and the formation of weaker coordinate bonds. It is important to note that at the optimal pH of 6, carboxylic acid groups are predominantly deprotonated (approximately 97% based on an assumed pKa of 4.5), but some protonated –COOH sites remain and may also contribute to Pb(II) binding via hydrogen bonding or ligand exchange. The net reaction is spontaneous, as demonstrated by the negative Gibbs free energy values (between −6.16 and −3.81 kJ mol−1), and entails a change in entropy (−77.07 J mol−1K−1), which represents the arrangement of hydrated Pb(II) ions into regular surface complexes.
The basis of this mechanistic system is the spectroscopic identification of active sites, quantitative energy estimation based on isotherm modeling, and kinetic analysis of the rate-determining steps, which fully explain the high adsorption activity of Bi-PKMF and provide a rational background for the use of this system in the purification of lead-contaminated water.
3.9 Study implications and limitations
Additional desorption experiments using selective eluents would differentiate reversible and irreversible bound fractions of lead in the ion-exchangeable and irreversible complexes, respectively, which would further limit the binding mechanism. These sophisticated spectroscopic and surface characterization studies will be done in future research to propose the final mechanistic explanation, whereas the present research can make conjectures only depending on the experimental evidence.
4 Conclusion
A novel bismuth-decorated palm kernel mesocarp fiber (Bi-PKMF) was successfully produced through green mechanochemical-thermal processing. Extensive characterization (SEM-EDX, TEM, BET, and FTIR) revealed the uniform dispersion of bismuth oxide nanoparticles in the lignocellulosic matrix to form a high-surface-area mesoporous material (387 m2 g−1 and pore size of 12.5 nm). Neutral control experiments with ball-milled PKMF (BM-PKMF) showed that bismuth functionalization is not a replication of the physical advantages of mechanical activation. Bi-PKMF showed superior Pb(II) adsorption capacity (112.3 mg g−1), which is 231% higher than that of pristine PKMF (48.5 mg g−1), and more than 90% removal was observed in 60 min at pH 6. The pseudo-second-order model and kinetic data fit (R2 > 0.999) and the Dubinin-Radushkevich adsorption energy (12.6 kJ mol−1) proved chemisorption as the dominant process. Bi-PKMF has a mesoporous structure that reduces the resistance to intraparticle diffusion, which enables rapid access to the active sites (Bi–O, –OH, and –COO−). The change in thermodynamic parameters was spontaneous (ΔG0 < 0), exothermic (ΔH0 = −29.02 kJ mol−1), with ordered adsorption. Bi-PKMF has very high reusability, retaining more than 90% of its original capacity after 5 regeneration cycles. This study confirms that Bi-PKMF is a high-performing, sustainable biosorbent for remediating lead, showing that surface chemistry, rather than physical structure, is the critical element in the uptake of metals.Author contributions
Amarachi Udoka Nkwoada: conceptualization, methodology, formal analysis, investigation, writing – original draft, project administration, funding acquisition. Simeon Chukwudozie Nwanonenyi: validation, resources, writing – review & editing, supervision. Callistus Izunna Iheme: data curation, investigation, writing – review & editing, visualization. Ali Bilar: validation, investigation, formal analysis, writing – review & editing. Ijeoma Akunna Duru: methodology, investigation, data curation. Tochukwu Ifeanyi Nwakile: resources, validation, writing – review & editing. Divinefavour Anioke: methodology, investigation, and formal analysis. Janefrances Oluebube Onwuemeodo: methodology, investigation, and formal analysis. Chukwuemeka Fortunatus Nnadozie: investigation, data curation, validation. Celestine Chidubem Chukwunonyerem: software, formal analysis, visualization. Favour Ogadinma Izuagba: methodology, investigation. Uchenna Gift Nwaneri: writing – review & editing, project administration, supervision.Conflicts of interest
There are no conflicts to declare.Data availability
Data availability will be made available on reasonable request.References
- P. Zhang, M. Yang, J. Lan, Y. Huang, J. Zhang, S. Huang, Y. Yang and J. Ru, Toxics, 2023, 11, 828 CrossRef CAS PubMed.
- O. Azubuike, A. Basil, N. Amarachi, E. Uchechi, B. Ali and O. Cynthia, Water, Air, Soil Pollut., 2025, 236, 773 CrossRef CAS.
- M. Meena, P. Sonigra and G. Yadav, Environ. Sci. Pollut. Res., 2021, 28, 2485–2508 CrossRef CAS PubMed.
- A. U. Nkwoada, I. C. Iwu, S. A. Kalu, C. A. Nweze and B. N. Anukam, Phys. Chem. Res., 2025, 13, 129–138 CAS.
- P. Elechi, A. U. Nkwoada, C. S. Okpara, T. I. Nwakile, D. P. Fapojuwo, C. P. Chikezie, S. A. Kalu, C. I. Iheme, C. A. Nweze, S. C. Ukanero, C. K. Oguzie and C. I.-A. Nwoko, Next Mater., 2026, 11, 101581 CrossRef CAS.
- Z. Saddique, M. Imran, A. Javaid, S. Latif, T. H. Kim, M. Janczarek, M. Bilal and T. Jesionowski, Environ. Res., 2023, 229, 115861 CrossRef CAS PubMed.
- J. Idris, S. Osman, E. Gaius and C. Christian, Environ. Eng. Manag. J., 2017, 16, 26–57 Search PubMed.
- D. A. Axelrad, E. Coffman, E. F. Kirrane and H. Klemick, Environ. Int., 2022, 169, 107475 CrossRef CAS PubMed.
- A. Carocci, A. Catalano, G. Lauria, M. S. Sinicropi and G. GenchiFood Toxicology, ed. P. de Voogt and F. A. Gunther, Springer International Publishing, Cham, 2016, pp. 45–67 Search PubMed.
- S. Collin, A. Baskar, D. M. Geevarghese, M. N. V. S. Ali, P. Bahubali, R. Choudhary, V. Lvov, G. I. Tovar, F. Senatov, S. Koppala and S. Swamiappan, J. Hazard. Mater. Lett., 2022, 3, 100064 CrossRef CAS.
- P. Mishra, S. Ali, R. Kumar, S. Shekhar and J. Trace, Elem. Miner., 2025, 14, 100259 Search PubMed.
- O. B. Abogunrin-Olafisoye, O. Adeyi, A. J. Adeyi and E. O. Oke, Waste Manag. Bull., 2024, 2, 214–228 CrossRef.
- I. Uchegbulam, E. O. Momoh and S. A. Agan, Clean. Mater., 2022, 6, 100154 CrossRef.
- R. Baby, M. Z. Hussein, Z. Zainal and A. H. Abdullah, J. Hazard. Mater. Adv., 2023, 10, 100253 CAS.
- R. Baby, B. Saifullah and M. Z. Hussein, Sci. Rep., 2019, 9, 18955 CrossRef CAS PubMed.
- B. Osei Bonsu, M. Takase and J. Mantey, Heliyon, 2020, 6, e05266 CrossRef CAS PubMed.
- C. Marcuello, B. Chabbert, F. Berzin, N. B. Bercu, M. Molinari and V. Aguié-Béghin, Materials (Basel), 2023, 16, 2440 CrossRef CAS PubMed.
- E. O. Babatunde, S. Enomah, O. M. Akwenuke, M. A. Ibeh, C. O. Okwelum, M. M. Mundu, P. O. Adepoju, A. O. Aki, O. D. Oghenejabor, T. F. Adepoju, C. O. Ifedora and K. Mabel, South African J. Chem. Eng., 2025, 52, 200–210 CrossRef.
- M. S. M. Zaini, M. Arshad and S. S. A. Syed-Hassan, J. Bioresour. Bioprod., 2023, 8, 66–77 CAS.
- C. L. Lee, P. S. H’ng, M. T. Paridah, K. L. Chin, U. Rashid, M. Maminski, W. Z. Go, R. A. R. Nazrin, S. N. A. Rosli and P. S. Khoo, R. Soc. Open Sci., 2018, 5, 180775 CrossRef CAS PubMed.
- J. Liu, S. Du, Q. Zhu, A. Labidi, H. Wang and C. Wang, J. Environ. Chem. Eng., 2024, 12, 114790 CrossRef CAS.
- S. Jiang, Y. Zhang and J. Gong, Environ. Sci. Nano, 2024, 11, 1332–1367 RSC.
- J. Liao, X. He, Y. Zhang, W. Zhu, L. Zhang and Z. He, Sci. Total Environ., 2022, 819, 153145 CrossRef CAS PubMed.
- K. H. Thamir, H. A. Ahmed, S. Z. Al-Ashoor, P. Kanjariya, M. Akku, R. Thakur, G. D. A. Jebaselvi, S. Choudhury, M. A. Rusho and H. M. Alkahtani, J. Inorg. Organomet. Polym. Mater., 2025, 35, 8249–8267 CrossRef CAS.
- W. A. Ali, S. E. Richards and R. H. Alzard, J. Ind. Eng. Chem., 2025, 149, 63–93 CrossRef CAS.
- C. Zhao, L. Xiao, Z. Chen and Y. Zhang, Accounts Mater. Res., 2024, 5, 1583–1597 CrossRef CAS.
- J. Choma, B. Szczęśniak and M. Jaroniec, Molecules, 2025, 30, 3125 CrossRef CAS PubMed.
- R. Singh and R. Bhateria, ACS Omega, 2020, 5, 10826–10837 CrossRef CAS PubMed.
- A. R. Katte, J. Mwero, M. Gibigaye and D. O. Koteng, Results Eng, 2023, 17, 100903 CrossRef CAS.
- K. S. Ukanwa, K. Patchigolla, R. Sakrabani and E. Anthony, Molecules, 2020, 25 DOI:10.3390/molecules25215028.
- J.-L. Do and T. Friščić, ACS Cent. Sci., 2017, 3, 13–19 CrossRef CAS PubMed.
- H. Yan, X. Jiang, H. Xia, L. Zhang, Q. Zhang, W. Zhang, C. Xin and K. Zeng, Desalin. Water Treat., 2021, 230, 372–383 CrossRef CAS.
- J. Han, J. Shao, X. He, N. Li, Y. Qian, D. Chen and J. Lu, J. Environ. Chem. Eng., 2025, 13, 116485 CrossRef CAS.
- D. Szczerba, J.-L. Do, D. Tan, H. M. Titi, N. Geoffroy, M. del C. M. de Lucas, J. Boudon, I. Halasz, T. Friščić and S. A. J. Kimber, RSC Mechanochemistry, 2024, 1, 255–262 RSC.
- H. Arshad, M. U. Tahir, F. Rehman, L. Wang, J. Wang, X. Su and C. Yang, Appl. Surf. Sci., 2022, 574, 151678 CrossRef CAS.
- C. Gomez, G. Hallot and M. Port, S. N. Grumezescu, Inorganic Frameworks as Smart Nanomedicines, William Andrew Publishing, 2018, pp. 449–487 Search PubMed.
- C. Wang, H. Hu, M. Chen, Q. Wang, C. Liu, M. Chen, S. Yan and Q. Zhang, J. Clean. Prod., 2022, 340, 130747 CrossRef CAS.
- M. Prakash, H. P. Kavitha, S. Abinaya, J. P. Vennila and D. Lohita, Sustain. Chem. Pharm., 2022, 25, 100547 CrossRef CAS.
- N. F. Abu Bakar, N. Abd Rahman, M. B. Mahadi, S. A. Mohd Zuki, K. N. Mohd Amin, M. Z. Wahab and I. Wuled Lenggoro, Mater. Today Proc., 2022, 48, 1899–1904 CrossRef CAS.
- G. N. Sambo, A. O. Adeola and S. A. Muhammad, Appl. Water Sci., 2024, 14, 113 CrossRef CAS.
- X. Li, Y. Sun, T. Xiong, G. Jiang, Y. Zhang, Z. Wu and F. Dong, J. Catal., 2017, 352, 102–112 CrossRef CAS.
- P. Ying, J. Yu and W. Su, Adv. Synth. Catal., 2021, 363, 1246–1271 CrossRef CAS.
- R. J. Allenbaugh and A. Shaw, Results Chem., 2023, 5, 100827 CrossRef CAS.
- P. W. Huisken Mejouyo, E. M. Tiaya, N. R. Sikame Tagne, S. T. Tiwa and E. Njeugna, Results Mater, 2022, 14, 100284 CrossRef CAS.
- B. Haider, M. Imran, A. Naeem, H. S. El-Beltagi, H. Arshad, A. Hussain, U. Zulfiqar, N. Y. Rebouh, P. V. V. Prasad and I. Djalovic, Sci. Rep., 2025, 15, 22823 CrossRef PubMed.
- S. Mohamed Nasser, M. Abbas and M. Trari, Prog. React. Kinet. Mech., 2024, 49, 14686783241226858 CrossRef.
- Sheetal and J. Pal, Environ. Monit. Assess., 2024, 196, 570 CrossRef CAS PubMed.
- J. M. Jose, E. H. E. Gladis and J. J, ECS Trans., 2022, 107, 18753 CrossRef CAS.
- K. Y. Foo and B. H. Hameed, Chem. Eng. J., 2010, 156, 2–10 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |









