Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Synthesis of sulfanyl derivatives of 1,2,4-triazoles via an acid catalyzed intramolecular cyclization of isothiosemicarbazones: structural characterization, E/Z isomerism, mechanistic insights and in vitro cytotoxicity
Kallivalappil Snishaa,
Mano Chitra Karthikeyanb,
Nattamai Bhuvaneshc,
Antony Joseph Velanganni Arockiamb and
Ramasamy Karvembu *a
*a
aDepartment of Chemistry, National Institute of Technology, Tiruchirappalli 620 015, India. E-mail: kar@nitt.edu
bMolecular Oncology Laboratory, Department of Biochemistry, School of Life Sciences, Bharathidasan University, Tiruchirappalli 620 024, India
cDepartment of Chemistry, Texas A & M University, College Station, TX 77842, USA
First published on 10th March 2026
Abstract
Sulfanyl derivatives of 1,2,4-triazoles (CL1–CL3) were synthesized via a Lewis or Brønsted acid catalyzed intramolecular cyclization of corresponding isothiosemicarbazones (TL1–TL3). All the synthesized isothiosemicarbazones and their cyclized sulfanyl 1,2,4-triazole derivatives were well characterized by spectroscopic techniques and single crystal XRD analyses. The E/Z isomerism of the isothiosemicarbazones was elucidated using NMR spectroscopy and single crystal XRD analysis. Mechanistic investigations, supported by controlled experiments and spectroscopic evidence, revealed that the cyclization proceeded via an ionic pathway with the evolution of hydrogen. The cytotoxic effect of the cyclized sulfanyl 1,2,4-triazole derivatives (CL1–CL3) was evaluated by MTT assay against MDA-MB-231 (breast), MCF-7 (breast), and HeLa (cervical) cancer cell lines, as well as HEK-293 (kidney) normal cell line, taking 5-fluorouracil (5-FU) as a reference drug. All the substituted sulfanyl-1,2,4-triazoles showed higher cytotoxicity toward MDA-MB-231 and HeLa cells than 5-FU, while exhibiting low toxicity toward HEK-293 cells, indicating good selectivity toward cancer cells. Substituted compounds [CL2 (p-OCH3) and CL3 (p-NO2)] displayed enhanced activity compared to the unsubstituted one (CL1), indicating the influence of the substituents on cytotoxicity. Fluorescence staining assays (AO/EB, Hoechst 33342, Rhodamine 123 and DCFH-DA) further supported the observed cytotoxic effects, and suggested that the compounds promoted apoptotic cell death via intracellular reactive oxygen species (ROS) generation, depletion of mitochondrial membrane potential (MMP) and damage to nuclear material.
1. Introduction
Heterocycle-based compounds constitute an important class of molecules in pharmaceutical research owing to their wide spectrum of therapeutic properties.1,2 Among these, nitrogen-containing heterocycles, particularly triazoles, have garnered more attention due to their promising biological activities.1 Triazoles consist of a five-membered aromatic ring incorporating three nitrogen atoms, and based on the relative positioning of these nitrogen atoms within the ring, triazoles exist in two isomeric forms, namely 1,2,3-triazoles and 1,2,4-triazoles (Fig. 1).2,3 Among these, 1,2,4-triazoles2,4 are of greater significance due to their well-documented and diverse pharmacological properties, including anticancer,3,5 antibacterial,2 antifungal,4,6,7 antitubercular,8 antiviral,9 antihypertensive,10 antidepressant,11 anti-inflammatory,12 antimalarial,13 antidiabetic,14 analgesic15 and antimigraine16 activities. The pronounced biological efficacy of 1,2,4-triazoles is largely attributed to their rigidity,7 stability,7 moderate dipole character7 and ability to engage in diverse non-covalent interactions17 with biological targets. The therapeutic relevance of the 1,2,4-triazole scaffold is further underscored by its presence in several clinically approved drugs such as anastrozole, letrozole and vorozole, which are extensively used in the treatment of hormone-dependent breast cancer7,18 (Fig. 2). Other drugs containing a 1,2,4-triazole core are voriconazole, itraconazole, ravuconazole, fluconazole (all antifungal drugs),2,6,7 taribavirin (antiviral),2,9 rizatriptan (antimigraine),2,16 trazodone (antidepressant),2 etc. Sulfanyl derivatives of 1,2,4-triazoles represent a relatively understudied class of heterocycles. The thioether linkage present in these compounds is known to enhance biological activity by increasing aqueous solubility, reducing lipophilicity and providing hydrogen bond acceptor sites.19,20 According to previous reports, these derivatives exhibit diverse biological activities such as anticancer,21 antimicrobial,22,23 antidepressant,24 anti-tuberculosis,25 antifungal,23 antibacterial,23 diuretic, neuroleptic and anti-inflammatory effects.19,26 The drug ufiprazole, which contains a thioether functionality, is active against cancer.19There are numerous approaches for the synthesis of 1,2,4-triazole-based compounds from various nitrogen sources like amidines,27 imidates,28 amidrazones,29 aryldiazoniums30 and hydrazones.31 But there are very few reports available for the synthesis of its sulfanyl derivatives.19,26,32,33 Kaldrikyan et al. reported the synthesis of substituted sulfanyl-1,2,4-triazoles starting from thiosemicarbazide, involving cyclization to form the 1,2,4-triazole core followed by S-alkylation.34 The synthesis of sulfanyl derivatives of 1,2,4-triazole from isothiosemicarbazone was found to be quite interesting, and to the best of our knowledge, there is no report for the acid-catalyzed cyclization of isothiosemicarbazones into sulfanyl derivatives of 1,2,4-triazole. Isothiosemicarbazones are a class of Schiff base compounds,35 which have been less explored.36 Isothiosemicarbazones are the enol form of classical thiosemicarbazones,36 and the reactions of thiosemicarbazones with alkyl/aryl halide (RX/ArX) in a suitable solvent yield isothiosemicarbazones incorporated with HX.35,37,38 Structurally, isothiosemicarbazones are prone to E/Z isomerism38 around the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N bond, often resulting in inseparable isomeric mixtures in solution.35 This inherent isomerism poses a significant challenge for their biological evaluation, as the presence of multiple interconverting species complicates the establishment of reliable structure–activity relationships. In the course of recrystallization of isothiosemicarbazone derivatives (HX form), an unexpected cyclization was observed under the employed conditions, leading to the formation of crystals of sulfanyl-1,2,4-triazole derivatives rather than those of isothiosemicarbazones (Fig. 3). This drove us to conduct a deeper research into the formation of sulfanyl derivatives of 1,2,4-triazoles from isothiosemicarbazones.
N bond, often resulting in inseparable isomeric mixtures in solution.35 This inherent isomerism poses a significant challenge for their biological evaluation, as the presence of multiple interconverting species complicates the establishment of reliable structure–activity relationships. In the course of recrystallization of isothiosemicarbazone derivatives (HX form), an unexpected cyclization was observed under the employed conditions, leading to the formation of crystals of sulfanyl-1,2,4-triazole derivatives rather than those of isothiosemicarbazones (Fig. 3). This drove us to conduct a deeper research into the formation of sulfanyl derivatives of 1,2,4-triazoles from isothiosemicarbazones.
 | ||
| Fig. 3 General structures of thiosemicarbazone, isothiosemicarbazone and sulfanyl derivative of 1,2,4-triazole. | ||
Given the established relevance of heterocycles, particularly nitrogen-containing systems in anticancer drug discovery, the incidental formation of stable sulfanyl derivatives of 1,2,4-triazole from isothiosemicarbazones motivated further exploration. Tuning of a compound by the introduction of a new moiety, varying the substituent and position of the substituent, also significantly matters in medicinal chemistry. Based on these considerations, the present work describes the synthesis of a series of isothiosemicarbazones and their cyclized sulfanyl 1,2,4-triazole derivatives bearing electron withdrawing or electron donating substituent, with the aim of elucidating the influence of electronic effects on their cytotoxicity. Cyclized sulfanyl 1,2,4-triazole derivatives were synthesized by the Lewis or Brønsted acid catalyzed intramolecular cyclization of isothiosemicarbazones. Isothiosemicarbazones are comparatively less explored; E/Z isomerism exhibited by isothiosemicarbazones was studied in detail using NMR spectroscopy and single crystal XRD. A good attempt was made to explore the mechanism of acid-catalyzed intramolecular cyclization of isothiosemicarbazones with some controlled experiments and spectroscopic techniques. Cytotoxicity of cyclized sulfanyl 1,2,4-triazole compounds was evaluated by MTT assay and fluorescence staining assays against cancer cell lines such as MDA-MB-231, MCF-7 and HeLa, which were selected in consideration of their high global prevalence.39
2. Experimental section
2.1. Materials and methods
All the chemicals used in this work were purchased from Sigma Aldrich, Alfa-Aesar or Avra. The melting points were determined using a Lab India instrument and are uncorrected. Fourier transform-infrared (FT-IR) spectra were recorded with a Thermo Scientific Nicolet iS5 FT-IR spectrometer by using KBr pellets of the compounds. Ultraviolet-visible (UV-vis) spectra of the compounds were recorded using a Shimadzu UV-2600 instrument. Nuclear magnetic resonance (NMR) spectra were recorded in dimethyl sulfoxide-d6 (DMSO-d6)/CDCl3/CD3OD using tetramethylsilane (TMS) as an internal standard on a Bruker 500 MHz spectrometer. High resolution mass spectra (HRMS) were recorded with an Agilent QTOF G6545 XT spectrometer at 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 resolutions using ESI mode.
000 resolutions using ESI mode.
2.2. Synthesis
2.2.1.1. Benzyl-N′-benzylidene-N-methylcarbamohydrazonothioate (TL1). Yield: 74%. Off white solid. M.p.: 96 °C. FT-IR [KBr, ν (cm−1)]: 3441 (N–H), 1606 (C1
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N1), 1570 (C2
N1), 1570 (C2![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N2), 1161 (N1–N2), 752 (C2–S1). UV-vis [CH3CN, λmax (nm)]: 232 (π → π*), 318 (n → π*). 1H NMR [500 MHz, DMSO-d6, δ (ppm)]: 8.22 (s, 1H, imine CH), 7.64 (d, J = 8.3 Hz, 2H, aromatic CH), 7.41 (d, J = 7.1 Hz, 2H, aromatic CH), 7.38–7.32 (m, 5H, aromatic CH), 7.31 (d, J = 6.5 Hz, 1H, aromatic CH), 6.69 (s, 1H, exchangeable with D2O, NH), 4.18 (s, 2H, benzylic CH2), 2.87 (d, J = 4.5 Hz, 3H, terminal NCH3). 13C NMR [126 MHz, DMSO-d6, δ (ppm)]: 162.98 (C2
N2), 1161 (N1–N2), 752 (C2–S1). UV-vis [CH3CN, λmax (nm)]: 232 (π → π*), 318 (n → π*). 1H NMR [500 MHz, DMSO-d6, δ (ppm)]: 8.22 (s, 1H, imine CH), 7.64 (d, J = 8.3 Hz, 2H, aromatic CH), 7.41 (d, J = 7.1 Hz, 2H, aromatic CH), 7.38–7.32 (m, 5H, aromatic CH), 7.31 (d, J = 6.5 Hz, 1H, aromatic CH), 6.69 (s, 1H, exchangeable with D2O, NH), 4.18 (s, 2H, benzylic CH2), 2.87 (d, J = 4.5 Hz, 3H, terminal NCH3). 13C NMR [126 MHz, DMSO-d6, δ (ppm)]: 162.98 (C2![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N2), 150.80 (C1
N2), 150.80 (C1![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N1), 137.16, 136.12, 129.61, 129.57, 129.10, 129.04, 128.94, 128.85, 127.96, 127.77, 127.58, 127.28 (aromatic carbons), 33.87 (benzylic CH2), 30.77 (terminal NCH3). HRMS (m/z): found (calcd.) 284.1229 (284.1221) {[M + H]+ = [C16H18N3S]+}.
N1), 137.16, 136.12, 129.61, 129.57, 129.10, 129.04, 128.94, 128.85, 127.96, 127.77, 127.58, 127.28 (aromatic carbons), 33.87 (benzylic CH2), 30.77 (terminal NCH3). HRMS (m/z): found (calcd.) 284.1229 (284.1221) {[M + H]+ = [C16H18N3S]+}.
2.2.1.2. Benzyl-N′-(4-methoxybenzylidene)-N-methylcarbamohydrazonothioate (TL2). Yield: 72%. Off white solid. M.p.: 118 °C. FT-IR [KBr, ν (cm−1)]: 3373 (N–H), 1601 (C1
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N1), 1509 (C2
N1), 1509 (C2![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N2), 1166 (N1–N2), 714 (C2–S1), 2926 (aliphatic C–H of OCH3), 1245 (asymmetric C–O–C stretching), 1029 (symmetric C–O–C stretching). UV-vis [CH3CN, λmax (nm)]: 257 (π → π*), 332 (n → π*). 1H NMR [500 MHz, DMSO-d6, δ (ppm)]: 8.16 (s, 1H, imine CH), 7.58 (d, J = 8.8 Hz, 2H, aromatic CH), 7.40 (d, J = 7.0 Hz, 2H, aromatic CH), 7.36 (t, J = 7.5 Hz, 2H, aromatic CH), 7.30 (d, J = 7.2 Hz, 1H, aromatic CH), 6.94 (d, J = 8.8 Hz, 2H, aromatic CH), 6.56 (q, J = 4.3 Hz, 1H, exchangeable with D2O, NH), 4.17 (s, 2H, benzylic CH2), 3.77 (s, 3H, OCH3), 2.84 (d, J = 4.5 Hz, 3H, terminal NCH3). 13C NMR [126 MHz, DMSO-d6, δ (ppm)]: 161.95 (C2
N2), 1166 (N1–N2), 714 (C2–S1), 2926 (aliphatic C–H of OCH3), 1245 (asymmetric C–O–C stretching), 1029 (symmetric C–O–C stretching). UV-vis [CH3CN, λmax (nm)]: 257 (π → π*), 332 (n → π*). 1H NMR [500 MHz, DMSO-d6, δ (ppm)]: 8.16 (s, 1H, imine CH), 7.58 (d, J = 8.8 Hz, 2H, aromatic CH), 7.40 (d, J = 7.0 Hz, 2H, aromatic CH), 7.36 (t, J = 7.5 Hz, 2H, aromatic CH), 7.30 (d, J = 7.2 Hz, 1H, aromatic CH), 6.94 (d, J = 8.8 Hz, 2H, aromatic CH), 6.56 (q, J = 4.3 Hz, 1H, exchangeable with D2O, NH), 4.17 (s, 2H, benzylic CH2), 3.77 (s, 3H, OCH3), 2.84 (d, J = 4.5 Hz, 3H, terminal NCH3). 13C NMR [126 MHz, DMSO-d6, δ (ppm)]: 161.95 (C2![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N2), 160.66 (C1
N2), 160.66 (C1![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N1), 150.64, 137.27, 129.59, 129.50, 129.08, 128.84, 128.79, 127.73, 114.57 (aromatic carbons), 55.67 (OCH3), 33.85 (benzylic CH2), 30.75 (terminal NCH3). HRMS (m/z): found (calcd.) 314.1326 (314.1327) {[M + H]+ = [C17H20N3OS]+}.
N1), 150.64, 137.27, 129.59, 129.50, 129.08, 128.84, 128.79, 127.73, 114.57 (aromatic carbons), 55.67 (OCH3), 33.85 (benzylic CH2), 30.75 (terminal NCH3). HRMS (m/z): found (calcd.) 314.1326 (314.1327) {[M + H]+ = [C17H20N3OS]+}.
2.2.1.3. Benzyl-N-methyl-N′-(4-nitrobenzylidene)carbamohydrazonothioate (TL3). Yield: 78%. Orange solid. M.p.: 162 °C. FT-IR [KBr, ν (cm−1)]: 3434 (N–H), 1592 (C1
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N1), 1519 (C2
N1), 1519 (C2![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N2), 1168 (N1–N2), 765 (C2–S1), 1497 (asymmetric NO2 vibration), 1328 (symmetric NO2 vibration). UV-vis [CH3CN, λmax (nm)]: 215 (π → π*), 250 (n → π*), 382 (n → π*). 1H NMR [500 MHz, DMSO-d6, δ (ppm)]: 8.33 (s, 1H, imine CH), 8.23 (d, J = 8.9 Hz, 2H, aromatic CH), 7.87 (d, J = 8.9 Hz, 2H, aromatic CH), 7.45–7.40 (m, 2H, aromatic CH), 7.37 (t, J = 7.5 Hz, 2H, aromatic CH), 7.33–7.30 (m, 1H, aromatic CH), 7.04 (q, J = 4.5 Hz, 1H, exchangeable with D2O, NH), 4.21 (s, 2H, benzylic CH2), 2.91 (d, J = 4.5 Hz, 3H, terminal NCH3). 13C NMR [126 MHz, DMSO-d6, δ (ppm)]: 165.26 (C2
N2), 1168 (N1–N2), 765 (C2–S1), 1497 (asymmetric NO2 vibration), 1328 (symmetric NO2 vibration). UV-vis [CH3CN, λmax (nm)]: 215 (π → π*), 250 (n → π*), 382 (n → π*). 1H NMR [500 MHz, DMSO-d6, δ (ppm)]: 8.33 (s, 1H, imine CH), 8.23 (d, J = 8.9 Hz, 2H, aromatic CH), 7.87 (d, J = 8.9 Hz, 2H, aromatic CH), 7.45–7.40 (m, 2H, aromatic CH), 7.37 (t, J = 7.5 Hz, 2H, aromatic CH), 7.33–7.30 (m, 1H, aromatic CH), 7.04 (q, J = 4.5 Hz, 1H, exchangeable with D2O, NH), 4.21 (s, 2H, benzylic CH2), 2.91 (d, J = 4.5 Hz, 3H, terminal NCH3). 13C NMR [126 MHz, DMSO-d6, δ (ppm)]: 165.26 (C2![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N2), 148.35 (C1
N2), 148.35 (C1![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N1), 147.58, 142.64, 136.78, 129.68, 129.63, 129.16, 128.89, 128.59, 127.87, 124.46, 124.20 (aromatic carbons), 33.89 (benzylic CH2), 30.88 (terminal NCH3). HRMS (m/z): found (calcd.) 329.1072 (329.1072) {[M + H]+ = [C16H17N4O2S]+}.
N1), 147.58, 142.64, 136.78, 129.68, 129.63, 129.16, 128.89, 128.59, 127.87, 124.46, 124.20 (aromatic carbons), 33.89 (benzylic CH2), 30.88 (terminal NCH3). HRMS (m/z): found (calcd.) 329.1072 (329.1072) {[M + H]+ = [C16H17N4O2S]+}.
2.2.2.1. 3-(Benzylthio)-4-methyl-5-phenyl-4H-1,2,4-triazole (CL1). Yield: 75%. Off white solid. M.p.: 134 °C. FT-IR [KBr, ν (cm−1)]: 1467 (C
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N), 1148 (N–N), 775 (C2–S1). UV-vis [CH3CN, λmax (nm)]: 219, 252 (π → π*). 1H NMR [500 MHz, CDCl3, δ (ppm)]: 7.51–7.47 (m, 2H, aromatic CH), 7.44–7.40 (m, 2H, aromatic CH), 7.25–7.20 (m, 5H, aromatic CH), 4.32 (s, 2H, benzylic CH2), 3.20 (s, 3H, terminal NCH3). 13C NMR [126 MHz, CDCl3, δ (ppm)]: 156.05 (C2
N), 1148 (N–N), 775 (C2–S1). UV-vis [CH3CN, λmax (nm)]: 219, 252 (π → π*). 1H NMR [500 MHz, CDCl3, δ (ppm)]: 7.51–7.47 (m, 2H, aromatic CH), 7.44–7.40 (m, 2H, aromatic CH), 7.25–7.20 (m, 5H, aromatic CH), 4.32 (s, 2H, benzylic CH2), 3.20 (s, 3H, terminal NCH3). 13C NMR [126 MHz, CDCl3, δ (ppm)]: 156.05 (C2![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N2), 150.91 (C1
N2), 150.91 (C1![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N1), 136.97, 130.13, 129.09, 128.91, 128.71, 128.59, 127.83, 127.15 (aromatic carbons), 38.98 (benzylic CH2), 31.49 (NCH3). HRMS (m/z): found (calcd.) 282.1067 (282.1065) {[M + H]+ = [C16H16N3S]+}.
N1), 136.97, 130.13, 129.09, 128.91, 128.71, 128.59, 127.83, 127.15 (aromatic carbons), 38.98 (benzylic CH2), 31.49 (NCH3). HRMS (m/z): found (calcd.) 282.1067 (282.1065) {[M + H]+ = [C16H16N3S]+}.
2.2.2.2. 3-(Benzylthio)-5-(4-methoxyphenyl)-4-methyl-4H-1,2,4-triazole (CL2). Yield: 73%. Off white solid. M.p.: 128 °C. FT-IR [KBr, ν (cm−1)]: 1468 (C
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N), 1175 (N–N), 700 (C2–S1), 2922 (aliphatic C–H of OCH3), 1247 (asymmetric C–O–C stretching), 1033 (symmetric C–O–C stretching). UV-vis [CH3CN, λmax (nm)]: 212, 258 (π → π*). 1H NMR [500 MHz, DMSO-d6, δ (ppm)]: 7.62 (d, J = 8.9 Hz, 2H, aromatic CH), 7.36–7.26 (m, 5H, aromatic CH), 7.12 (d, J = 11.8 Hz, 2H, aromatic CH), 4.38 (s, 2H, benzylic CH2), 3.84 (s, 3H, OCH3), 3.42 (s, 3H, NCH3). 13C NMR [126 MHz, DMSO-d6, δ (ppm)]: 161.14 (C2
N), 1175 (N–N), 700 (C2–S1), 2922 (aliphatic C–H of OCH3), 1247 (asymmetric C–O–C stretching), 1033 (symmetric C–O–C stretching). UV-vis [CH3CN, λmax (nm)]: 212, 258 (π → π*). 1H NMR [500 MHz, DMSO-d6, δ (ppm)]: 7.62 (d, J = 8.9 Hz, 2H, aromatic CH), 7.36–7.26 (m, 5H, aromatic CH), 7.12 (d, J = 11.8 Hz, 2H, aromatic CH), 4.38 (s, 2H, benzylic CH2), 3.84 (s, 3H, OCH3), 3.42 (s, 3H, NCH3). 13C NMR [126 MHz, DMSO-d6, δ (ppm)]: 161.14 (C2![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N2), 155.53 (C2
N2), 155.53 (C2![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N2), 150.44, 137.62, 130.45, 129.46, 128.97, 128.02, 119.17, 114.87 (aromatic carbons), 55.85 (OCH3), 37.85 (benzylic CH2), 32.10 (NCH3). HRMS (m/z): found (calcd.) 312.1172 (312.1171) {[M + H]+ = [C17H18N3OS]+}.
N2), 150.44, 137.62, 130.45, 129.46, 128.97, 128.02, 119.17, 114.87 (aromatic carbons), 55.85 (OCH3), 37.85 (benzylic CH2), 32.10 (NCH3). HRMS (m/z): found (calcd.) 312.1172 (312.1171) {[M + H]+ = [C17H18N3OS]+}.
2.2.2.3. 3-(Benzylthio)-4-methyl-5-(4-nitrophenyl)-4H-1,2,4-triazole (CL3). Yield: 79%. yellow solid. M.p.: 168 °C. FT-IR [KBr, ν (cm−1)]: 1467 (C
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N), 1139 (N–N), 774 (C2–S1), 1512 (asymmetric NO2 vibration), 1343 (symmetric NO2 vibration). UV-vis [CH3CN, λmax (nm)]: 203, 222 (π → π*), 308 (n → π*). 1H NMR [500 MHz, DMSO-d6, δ (ppm)]: 8.39 (d, J = 8.9 Hz, 2H, aromatic CH), 8.01 (d, J = 8.9 Hz, 2H, aromatic CH), 7.38 (d, J = 6.9 Hz, 2H, aromatic CH), 7.35–7.28 (m, 3H, aromatic CH), 4.43 (s, 2H, benzylic CH2), 3.53 (s, 3H, terminal NCH3). 13C NMR [126 MHz, DMSO-d6, δ (ppm)]: 154.25 (C2
N), 1139 (N–N), 774 (C2–S1), 1512 (asymmetric NO2 vibration), 1343 (symmetric NO2 vibration). UV-vis [CH3CN, λmax (nm)]: 203, 222 (π → π*), 308 (n → π*). 1H NMR [500 MHz, DMSO-d6, δ (ppm)]: 8.39 (d, J = 8.9 Hz, 2H, aromatic CH), 8.01 (d, J = 8.9 Hz, 2H, aromatic CH), 7.38 (d, J = 6.9 Hz, 2H, aromatic CH), 7.35–7.28 (m, 3H, aromatic CH), 4.43 (s, 2H, benzylic CH2), 3.53 (s, 3H, terminal NCH3). 13C NMR [126 MHz, DMSO-d6, δ (ppm)]: 154.25 (C2![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N2), 151.99 (C1
N2), 151.99 (C1![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N1), 148.53, 137.59, 133.61, 129.95, 129.48, 128.98, 128.05, 124.53 (aromatic carbons), 37.58 (benzylic CH2), 32.36 (NCH3). HRMS (m/z): found (calcd.) 327.0918 (327.0916) {[M + H]+ = [C16H15N4O2S]+}.
N1), 148.53, 137.59, 133.61, 129.95, 129.48, 128.98, 128.05, 124.53 (aromatic carbons), 37.58 (benzylic CH2), 32.36 (NCH3). HRMS (m/z): found (calcd.) 327.0918 (327.0916) {[M + H]+ = [C16H15N4O2S]+}.
2.3. Single crystal X-ray diffraction (XRD) analyses
A suitable crystal was selected and mounted on a MITIGEN holder on a XtaLAB Synergy, Dualflex, HyPix diffractometer. The crystal was kept at a steady T = 100.00(10) K during data collection. The structure was solved with the ShelXT 2018/2 solution program using dual methods and by using Olex2 1.5 as the graphical interface.40,41 The model was refined with ShelXL 2019/1 using full matrix least squares minimization on F2.422.4. Solution stability
Solution stability of the cyclized sulfanyl 1,2,4-triazoles (CL1–CL3) was evaluated in different media, like DMSO, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 99 (v/v) DMSO-water, and phosphate-buffered saline (PBS) solution (pH = 7.4) over a period of 24 h using UV-vis spectroscopy.43 Stock solutions of the compounds were prepared in DMSO at a concentration of 3 × 10−3 M. From each stock solution, 10 µL was added into a cuvette containing 2990 µL of either DMSO, water or PBS, and UV-vis spectra were recorded immediately (0 min) and subsequently at 2 min, 5 min, 15 min, 1 h, 4 h, 8 h and 24 h in all the media at room temperature.
99 (v/v) DMSO-water, and phosphate-buffered saline (PBS) solution (pH = 7.4) over a period of 24 h using UV-vis spectroscopy.43 Stock solutions of the compounds were prepared in DMSO at a concentration of 3 × 10−3 M. From each stock solution, 10 µL was added into a cuvette containing 2990 µL of either DMSO, water or PBS, and UV-vis spectra were recorded immediately (0 min) and subsequently at 2 min, 5 min, 15 min, 1 h, 4 h, 8 h and 24 h in all the media at room temperature.
2.5. In vitro cytotoxicity
3. Results and discussion
3.1. Synthesis
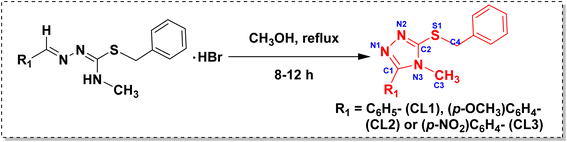
The syntheses of the isothiosemicarbazones and their cyclized sulfanyl 1,2,4-triazole derivatives are depicted in Scheme 1 and 2, respectively. The isothiosemicarbazones (TL1–TL3) were synthesized according to a procedure reported in the literature.35 Initially, N4-substituted thiosemicarbazide was treated with benzyl bromide in ethanol solvent under reflux condition to get isothiosemicarbazide. All the isothiosemicarbazones (TL1–TL3) were obtained by the condensation of isothiosemicarbazide with the desired aldehyde under the same conditions. It was obtained as a hydrobromide salt. To get the isothiosemicarbazone, the precipitate of hydrobromide salt was worked up with a dichloromethane-water mixture. The cyclized sulfanyl 1,2,4-triazole derivatives (CL1–CL3) were obtained by the intramolecular cyclization of hydrobromide salts of isothiosemicarbazones in methanol under reflux condition. The compound TL1 was already reported by Yamazaki et al. in 1975.383.2. Spectroscopic characterization of isothiosemicarbazones and their cyclized sulfanyl 1,2,4-triazole derivatives
The synthesized isothiosemicarbazones and their cyclized sulfanyl 1,2,4-triazole derivatives were well characterized by various spectroscopic techniques such as UV-vis, FT-IR, 1H NMR and 13C NMR, and HRMS.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N stretching frequencies in the range 1592–1606 and 1509–1570 cm−1 which corresponded to azomethine C1
N stretching frequencies in the range 1592–1606 and 1509–1570 cm−1 which corresponded to azomethine C1![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N1 and C2
N1 and C2![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N2 groups, respectively.46 In addition to the above, TL2 displayed peaks at 1245, 1029 and 2926 cm−1 which could be assigned to asymmetric C–O–C, symmetric C–O–C and aliphatic C–H stretching vibrations of the OCH3 group, respectively. Similarly, TL3 exhibited additional peaks, apart from the characteristic bands of isothiosemicarbazones, at 1497 and 1328 cm−1, which corresponded to the asymmetric and symmetric stretching vibrations of the NO2 group, respectively.47 Upon cyclization of isothiosemicarbazones, there was a reduction in the C
N2 groups, respectively.46 In addition to the above, TL2 displayed peaks at 1245, 1029 and 2926 cm−1 which could be assigned to asymmetric C–O–C, symmetric C–O–C and aliphatic C–H stretching vibrations of the OCH3 group, respectively. Similarly, TL3 exhibited additional peaks, apart from the characteristic bands of isothiosemicarbazones, at 1497 and 1328 cm−1, which corresponded to the asymmetric and symmetric stretching vibrations of the NO2 group, respectively.47 Upon cyclization of isothiosemicarbazones, there was a reduction in the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N stretching frequency, and it appeared in the range 1467–1468 cm−1, and the N–N bond stretching frequency shifted to 1139–1175 cm−1. Absorption bands observed in the range 700–775 cm−1 were attributed to the C2–S1 stretching vibrations in the cyclized sulfanyl 1,2,4-triazole derivatives.47 Absence of N–H stretching frequency confirmed the cyclization of isothiosemicarbazones. Additional peaks at 1247, 1033 and 2922 cm−1 in the FT-IR spectrum of CL2 corresponded to asymmetric C–O–C, symmetric C–O–C and aliphatic C–H stretching vibrations of the OCH3 group, respectively. The cyclized sulfanyl 1,2,4-triazole compound CL3 exhibited peaks at 1497 and 1328 cm−1 which were ascertained to the asymmetric and symmetric vibrations of NO2, respectively (Fig. S1, S6, S11, S16, S21 and S26).
N stretching frequency, and it appeared in the range 1467–1468 cm−1, and the N–N bond stretching frequency shifted to 1139–1175 cm−1. Absorption bands observed in the range 700–775 cm−1 were attributed to the C2–S1 stretching vibrations in the cyclized sulfanyl 1,2,4-triazole derivatives.47 Absence of N–H stretching frequency confirmed the cyclization of isothiosemicarbazones. Additional peaks at 1247, 1033 and 2922 cm−1 in the FT-IR spectrum of CL2 corresponded to asymmetric C–O–C, symmetric C–O–C and aliphatic C–H stretching vibrations of the OCH3 group, respectively. The cyclized sulfanyl 1,2,4-triazole compound CL3 exhibited peaks at 1497 and 1328 cm−1 which were ascertained to the asymmetric and symmetric vibrations of NO2, respectively (Fig. S1, S6, S11, S16, S21 and S26).UV-vis spectra of the isothiosemicarbazones (TL1–TL3) and their cyclized sulfanyl 1,2,4-triazole derivatives CL1–CL3 were recorded in acetonitrile. It was observed that the electronic spectra of the isothiosemicarbazones showed strong absorption bands in the range 215–232 nm which corresponded to π → π* transitions of the aromatic rings, and absorption bands in the range 318–382 nm represented n → π* transitions of the conjugated azomethine groups.35,46,48 The absorption maxima displayed in the UV-vis spectra of cyclized sulfanyl 1,2,4-triazole derivatives in the range 222–258 nm could be assigned to π → π* transitions of the 1,2,4-triazole ring.49 Nitro-substituted isothiosemicarbazone (TL3) and its cyclized sulfanyl 1,2,4-triazole derivative (CL3) exhibited an additional absorption band at 250 and 308 nm, respectively, which corresponded to the n → π* transition of the nitro group (Fig. S2, S7, S12, S17, S22 and S27).
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N carbons (C1 and C2) were observed in the most deshielded region (165.26–148.35 ppm). Aliphatic carbons of the terminal N-methyl and benzylic CH2 groups were found to resonate in the shielded regions 30.75–30.88 and 33.85–33.89 ppm, respectively. Further, the methoxy carbon of TL2 was observed at 55.67 ppm (Fig. S3, S4, S8, S9, S13 and S14).50 As the compound exists as an E/Z isomeric mixture in the solution state, all major peaks were accompanied by minor ones.35,38 The integrations corresponding to the minor isomer were excluded to ensure better interpretation of the spectra. The E/Z isomerism of isothiosemicarbazones is discussed in detail in Section 3.4.
N carbons (C1 and C2) were observed in the most deshielded region (165.26–148.35 ppm). Aliphatic carbons of the terminal N-methyl and benzylic CH2 groups were found to resonate in the shielded regions 30.75–30.88 and 33.85–33.89 ppm, respectively. Further, the methoxy carbon of TL2 was observed at 55.67 ppm (Fig. S3, S4, S8, S9, S13 and S14).50 As the compound exists as an E/Z isomeric mixture in the solution state, all major peaks were accompanied by minor ones.35,38 The integrations corresponding to the minor isomer were excluded to ensure better interpretation of the spectra. The E/Z isomerism of isothiosemicarbazones is discussed in detail in Section 3.4.In the 1H NMR spectra of cyclized sulfanyl 1,2,4-triazole derivatives, the disappearance of the signals corresponding to imine and NH protons confirmed the cyclization of isothiosemicarbazones. Upon ring closure, the benzylic CH2 and N-methyl protons were deshielded (4.32–4.43 and 3.25–3.53 ppm, respectively). In the 13C NMR spectra, the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N carbons (C1 and C2) were observed to be shifted to 150.91–161.14 ppm. There is no significant shift in the chemical shift value of the methoxy carbon in the spectrum of CL2 on cyclization. Aliphatic carbons of the N-methyl (31.49–32.36 ppm) and benzylic CH2 (37.58–38.98 ppm) groups were found to be deshielded when compared to those of the corresponding isothiosemicarbazones (Fig. S18, S19, S23, S24, S28 and S29).
N carbons (C1 and C2) were observed to be shifted to 150.91–161.14 ppm. There is no significant shift in the chemical shift value of the methoxy carbon in the spectrum of CL2 on cyclization. Aliphatic carbons of the N-methyl (31.49–32.36 ppm) and benzylic CH2 (37.58–38.98 ppm) groups were found to be deshielded when compared to those of the corresponding isothiosemicarbazones (Fig. S18, S19, S23, S24, S28 and S29).
3.3. Crystallographic characterization of isothiosemicarbazones and their cyclized sulfanyl 1,2,4-triazole derivatives
The molecular structures derived from the single crystal XRD analyses are shown in Fig. 4, which are consistent with the structures proposed from spectroscopic data for the isothiosemicarbazones and their cyclized sulfanyl 1,2,4-triazole derivatives. A summary of crystallographic data and refinement parameters is given in Tables S1 and S2. TL1 was obtained as yellow block-shaped crystals on recrystallization, and it was crystallized as a monoclinic crystal system with a space group of P21/c. The E isomer of TL3 was crystallized as orange needle-shaped crystals, and the Z isomer of TL3·HBr as yellow block-shaped crystals. TL3 and TL3·HBr were crystallized in a monoclinic and triclinic fashion with a space group of P21 and P-1, respectively. For TL1, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N bond length was found to be 1.2777 (C1
N bond length was found to be 1.2777 (C1![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N1) and 1.2997 (C2
N1) and 1.2997 (C2![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N2) Å, comparable to the standard C
N2) Å, comparable to the standard C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N length (1.27 Å). N1–N2 and C2–S1 had a single bond character with the bond lengths of 1.4043 and 1.7699 Å, respectively, in accordance with the standard bond length values of N–N (1.447 Å) and C–S (1.82 Å). In TL3 and TL3·HBr, C
N length (1.27 Å). N1–N2 and C2–S1 had a single bond character with the bond lengths of 1.4043 and 1.7699 Å, respectively, in accordance with the standard bond length values of N–N (1.447 Å) and C–S (1.82 Å). In TL3 and TL3·HBr, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N bond length was observed as 1.281 and 1.278 Å (C1
N bond length was observed as 1.281 and 1.278 Å (C1![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N1), and 1.312 and 1.336 Å (C2
N1), and 1.312 and 1.336 Å (C2![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N2), respectively. The small increment in the C2
N2), respectively. The small increment in the C2![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N2 bond length in the hydrobromide salt was due to the interaction of H+ with the N2 atom. There is no significant difference in the N1–N2 bond length of TL3 and TL3·HBr, which were 1.387 and 1.3907 Å, respectively. However, a slight shortening of the C2–S1 bond was noted in TL3·HBr (1.7411 Å) compared to TL3 (1.7681 Å).
N2 bond length in the hydrobromide salt was due to the interaction of H+ with the N2 atom. There is no significant difference in the N1–N2 bond length of TL3 and TL3·HBr, which were 1.387 and 1.3907 Å, respectively. However, a slight shortening of the C2–S1 bond was noted in TL3·HBr (1.7411 Å) compared to TL3 (1.7681 Å).
 | ||
| Fig. 4 Molecular structures of isothiosemicarbazones [TL1, TL3 (E isomer) and TL3·HBr (Z isomer)] and cyclized sulfanyl 1,2,4-triazole derivatives (CL1·HBr, CL2 and CL3). | ||
The cyclized sulfanyl 1,2,4-triazole compounds crystallized in a monoclinic system with a space group P21/c (CL1·HBr and CL3) or C2/c (CL2). CL1·HBr and CL2 formed colourless block-shaped crystals, whereas CL3 yielded yellow block-shaped crystals. CL1 was recrystallized with HBr, in which H+ was interacting with N2. Upon cyclization of isothiosemicarbazones, the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N bond length got increased from 1.277–1.281 to 1.307–1.319 Å, which implied the partial double bond character of C
N bond length got increased from 1.277–1.281 to 1.307–1.319 Å, which implied the partial double bond character of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N as a consequence of electron delocalisation within the triazole ring. A new C1–N3 bond was formed during the ring closure, whose length was observed in the range 1.368–1.374 Å.
N as a consequence of electron delocalisation within the triazole ring. A new C1–N3 bond was formed during the ring closure, whose length was observed in the range 1.368–1.374 Å.
3.4. E/Z isomerization of isothiosemicarbazones
Isothiosemicarbazones exist in two isomeric forms (E and Z) concerning the C2![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N2 bond as shown in Fig. 4. The Z isomer of TL3 was recrystallized as its hydrobromide salt (yellow block-shaped), whereas the corresponding E isomer was recrystallized as orange needle-shaped crystal from its saturated acetonitrile solution with a few drops of methanol. The crystals were mechanically separated and analyzed by single crystal XRD. 1H NMR spectrum was recorded for the E isomer, which was same as that of synthesized TL3 consisting of both the isomers, suggesting the isomerization in the solution state.
N2 bond as shown in Fig. 4. The Z isomer of TL3 was recrystallized as its hydrobromide salt (yellow block-shaped), whereas the corresponding E isomer was recrystallized as orange needle-shaped crystal from its saturated acetonitrile solution with a few drops of methanol. The crystals were mechanically separated and analyzed by single crystal XRD. 1H NMR spectrum was recorded for the E isomer, which was same as that of synthesized TL3 consisting of both the isomers, suggesting the isomerization in the solution state.
To study the E/Z isomerization in the solution state,38 a time-dependent 1H NMR analysis of TL1 was performed in CDCl3 for a period of 24 h. Immediately following the sample preparation, 1H NMR spectrum was acquired. The same sample was reanalyzed after 24 h under identical conditions. No change was observed between the spectra recorded at 0 and 24 h, indicating that the E/Z equilibrium was attained rapidly (Fig. 5). Additionally, the 1H NMR spectrum shown in Fig. S31 clearly demonstrates the coexistence of E/Z isomers, as evidenced by the duplicated signals and corresponding integration values of the characteristic protons, such as the imine proton, N-methyl proton and benzylic CH2 protons. To understand the ratio of E/Z isomers in different solvents,38 1H NMR spectra of TL1 were recorded in CD3OD, DMSO-d6 and CDCl3. The ratio of E/Z isomers was calculated by considering the integration of imine protons of E and Z isomers in the NMR spectra. As the polarity of the solvent increases, its ability to interrupt intramolecular hydrogen bonding present in the Z isomer also increases. Therefore, it was clearly seen that the E/Z ratio changed significantly in different solvents. The E/Z ratio was found to be 21.25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 78.74, 62.89
78.74, 62.89![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 37.10, 76.33
37.10, 76.33![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 23.66 in CDCl3, CD3OD and DMSO-d6, respectively (Fig. 6).
23.66 in CDCl3, CD3OD and DMSO-d6, respectively (Fig. 6).
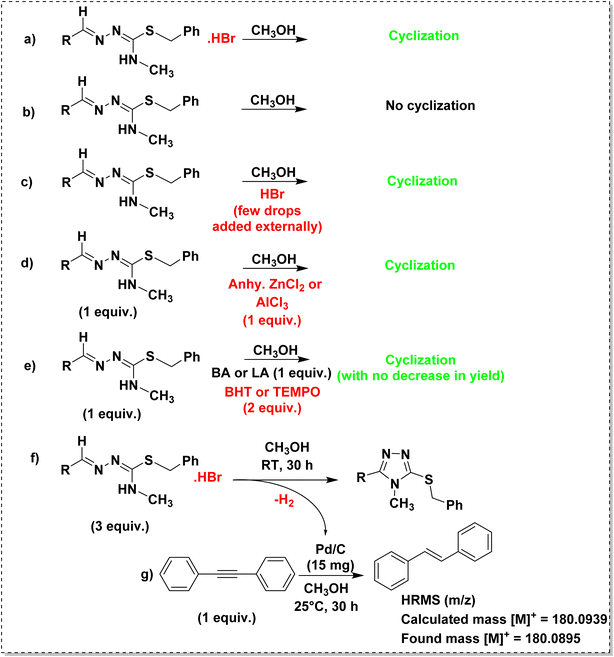
3.5. Mechanism of cyclization
To understand the mechanism of cyclization, a few controlled experiments were performed as shown in Scheme 3. The hydrobromide salt of isothiosemicarbazone underwent cyclization in methanol under reflux condition (Scheme 3a). The same reaction was performed for the isothiosemicarbazone extracted by a dichloromethane-water workup (Scheme 3b), but no cyclization was observed. To get to know the role of HBr in cyclization, when a few drops of hydrobromic acid was added externally to the above reaction mixture (Scheme 3c), the cyclization occurred, which indicated that HBr acted as an acid catalyst for the reaction. The same reaction proceeded even in the presence of Lewis acid (LA), like anhydrous ZnCl2 or anhydrous AlCl3 in the place of Brønsted acid (BA), HBr (Scheme 3d). To explore the possible pathway of cyclization, reactions were performed in the presence of either a Brønsted acid or Lewis acid and radical scavenger like butylated hydroxytoluene (BHT) or 2,2,6,6-tetramethylpiperidin-1-oxyl (TEMPO) (Scheme 3e).51 The reactions proceeded with no decrease in yield, which implied that the cyclization was not proceeding via a free radical mechanism. So, it might have been followed an ionic mechanism. The crystal structure of TL3·HBr validated the first step [(ii) in Scheme 4] of the mechanism proposed for the acid-catalyzed cyclization of isothiosemicarbazones. Further, the intramolecular nucleophilic attack of the terminal nitrogen on the electrophilic azomethine carbon led to ring closure. The Brønsted/Lewis acid facilitates this process by activating the substrate toward intramolecular nucleophilic attack. In the following step, the LA/BA dissociates, resulting in the formation of a neutral intermediate [(v) in Scheme 4]. To achieve aromatic stabilization, the intermediate undergoes dehydrogenation with the loss of molecular hydrogen, thereby forming the aromatic triazole ring. To confirm the evolution of hydrogen during the cyclization of isothiosemicarbazones (Scheme 3d), a H-tube experiment52 was performed to trap the formed hydrogen, and used the same for the reduction of diphenylacetylene in the connected tube (Scheme 3g), as shown in Fig. S32. HRMS spectrum was recorded for the crude mixture in the right tube, and confirmed the presence of reduced product, further confirming the H2 evolution (Fig. S33). | ||
| Scheme 4 Plausible mechanistic pathway for the acid-catalyzed intramolecular cyclization of isothiosemicarbazone. | ||
3.6. Solution stability of sulfanyl derivatives of 1,2,4-triazoles
Solution stability studies for CL1–CL3 were performed in DMSO, DMSO-water (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 99 v/v), and PBS over a period of 24 h using UV-vis spectroscopy at room temperature. No new peaks or significant shifts in the absorption maxima were observed even after 24 h, indicating that the cyclized sulfanyl 1,2,4-triazole compounds were sufficiently stable to carry out their biological functions within the cell and are suitable for further in vitro studies (Fig. S34–S36).
99 v/v), and PBS over a period of 24 h using UV-vis spectroscopy at room temperature. No new peaks or significant shifts in the absorption maxima were observed even after 24 h, indicating that the cyclized sulfanyl 1,2,4-triazole compounds were sufficiently stable to carry out their biological functions within the cell and are suitable for further in vitro studies (Fig. S34–S36).
3.7. Cytotoxic ability of the compounds
| Compound | HEK-293 (IC50 ± SD) | MDA-MB-231 (IC50 ± SD) | MCF-7 (IC50 ± SD) | HeLa (IC50 ± SD) |
|---|---|---|---|---|
| CL1 | 61.9 ± 1.8 | 34.3 ± 1.5 | 50.2 ± 1.7 | 41.7 ± 1.6 |
| CL2 | 44.4 ± 1.6 | 26.2 ± 1.4 | 33.8 ± 1.5 | 35.2 ± 1.5 |
| CL3 | 46.5 ± 1.7 | 32.3 ± 1.5 | 40.3 ± 1.6 | 40.4 ± 1.6 |
| 5-FU | 47.5 ± 1.7 | 74.5 ± 1.9 | 25.4 ± 1.4 | 101.2 ± 2.0 |
4. Conclusions
In conclusion, a series of isothiosemicarbazones (TL1–TL3) and their corresponding sulfanyl-1,2,4-triazoles (CL1–CL3) were synthesized by varying the substituents from electron withdrawing to electron donating to assess their influence on cytotoxicity. The sulfanyl derivatives of 1,2,4-triazoles were obtained via an acid-catalyzed intramolecular cyclization, and all the compounds were comprehensively characterized by FT-IR, UV-vis, 1H NMR and 13C NMR spectroscopic and HRMS techniques, and further validated with single crystal XRD. E/Z isomerism exhibited by the isothiosemicarbazones was investigated through detailed NMR spectroscopic studies and single crystal XRD. Moreover, mechanistic aspects of the acid-catalyzed intramolecular cyclization were explored, and it was concluded that cyclization proceeded via an ionic mechanism with the evolution of hydrogen as a byproduct. Biological evaluation revealed that the cyclized sulfanyl 1,2,4-triazole derivatives exhibited significant cytotoxicity against MDA-MB-231, MCF-7 and HeLa cancer cell lines, with lower toxicity toward normal cell line HK-293, suggesting good selectivity of the compounds to the cancer cells. Substituted derivatives CL2 and CL3, bearing p-OCH3 and p-NO2, respectively, exhibited enhanced activity across all the tested cancer cell lines than the unsubstituted analogue. The present study suggests that electronic modulation influences cytotoxicity, and the lipophilicity and hydrogen bonding ability of the substituents may also play an important role in enhancing the biological activity of the drug candidates. The mechanism of cell death was investigated using dual AO/EB, Hoechst 33342, Rh-123 and DCFH-DA fluorescence staining assays. The studies suggested that apoptosis might be induced through intracellular ROS generation and depletion of MMP. Overall, these findings highlight sulfanyl 1,2,4-triazoles as promising anticancer scaffolds. Further studies focusing on detailed mechanistic investigations, expanded structure–activity relationship analysis, and in vivo evaluation are warranted to advance their potential therapeutic development. Isothiosemicarbazones are a relatively underexplored class of compounds, despite being rich in coordination sites. In future work, we plan to synthesize organometallic compounds using isothiosemicarbazones as ligand systems, considering the growing importance of organometallic compounds in anticancer drug development.Author contributions
Kallivalappil Snisha: conceptualization, methodology, investigation, data curation, formal analysis, writing – original draft, writing – review & editing. Mano Chitra Karthikeyan: methodology, investigation, data curation, formal analysis, writing – review & editing. Nattamai Bhuvanesh: data curation, formal analysis. Antony Joseph Velanganni Arockiam: data curation, formal analysis, writing – review & editing. Ramasamy Karvembu: supervision, conceptualization, writing – review & editing.Conflicts of interest
There are no conflicts of interest to declare.Data availability
CCDC 2506364 (TL1), 2506370 (TL3·HBr), 2506371 (TL3), 2506359 (CL1·HBr), 2506360 (CL2) and 2506361 (CL3) contain the supplementary crystallographic data for this paper.57a–fSupplementary information (SI): experimental procedures and spectral data (FT-IR, UV-vis, 1H & 13C NMR and HRMS). See DOI: https://doi.org/10.1039/d6ra00822d.
Acknowledgements
K. S. thanks the National Institute of Technology, Tiruchirappalli, for the facilities provided, and the Ministry of Education, Government of India, for the financial assistance. M. C. K. acknowledges the Department of Science and Technology (DST)–INSPIRE and RUSA 2.0 Biological Sciences, Bharathidasan University, for the financial support for performing biological studies. R. K. greatly acknowledges the Anusandhan National Research Foundation (ANRF) for the financial support (CRG/2022/003145).References
- Z. Hosseinzadeh, A. Ramazani and N. Razzaghi-Asl, Curr. Org. Chem., 2018, 22, 2256–2279 CrossRef CAS.
- M. Strzelecka and P. Świątek, Pharmaceuticals, 2021, 14, 224 CrossRef CAS PubMed.
- D. S. Bhagat, G. S. Bumbrah, P. A. Chawla, W. B. Gurnule and S. K. Shejul, Anti Cancer Agents Med. Chem., 2022, 22, 2852–2875 CrossRef CAS PubMed.
- A. M. Mohamed, W. A. El-Sayed, A. A. Ibrahim, N. A. Abdel-Hafez, K. A. K. Ali and S. F. Mohamed, Org. Prep. Proced. Int., 2021, 53, 211–239 CrossRef CAS.
- H. E. Hashem, A. E. E. Amr, E. S. Nossier, M. M. Anwar and E. M. Azmy, ACS Omega, 2022, 7, 7155–7171 CrossRef CAS PubMed.
- E. M. Carmona and A. H. Limper, Clin. Chest Med., 2017, 38, 393–402 CrossRef PubMed.
- H. Aziz, A. Saeed, A. U. Rehman, F. Jabeen, B. Nasir, A. U. Khan and I. U. Khan, J. Iran. Chem. Soc., 2021, 18, 1965–1977 CrossRef CAS.
- N. D. Rode, A. D. Sonawane, L. Nawale, V. M. Khedkar, R. A. Joshi, A. P. Likhite, D. Sarkar and R. R. Joshi, Chem. Biol. Drug Des., 2017, 90, 1206–1214 CrossRef CAS PubMed.
- A. A. Aly, A. A. Hassan, M. M. Makhlouf and S. Bräse, Molecules, 2020, 25, 3036 CrossRef CAS PubMed.
- J. Liu, Q. Liu, X. Yang, S. Xu, H. Zhang, R. Bai, H. Yao, J. Jiang, M. Shen, X. Wu and J. Xu, Bioorg. Med. Chem., 2013, 21, 7742–7751 CrossRef CAS PubMed.
- C. Radhika, A. Venkatesham and M. Sarangapani, Med. Chem. Res., 2012, 21, 3509–3513 CrossRef CAS.
- S. Li, S. Tsai, C. Chiang, C. Chung, T. Chuang, C. Tseng, W. Jiang, G. Huang, C. Lin, Y. Yang, M. Fuh and F. Wong, Bioorg. Chem., 2020, 104, 104333 CrossRef CAS PubMed.
- N. Boechat, L. C. S. Pinheiro, O. A. Santos-Filho and I. C. Silva, Molecules, 2011, 16, 8083–8097 CrossRef CAS PubMed.
- F. Hichri, A. Omri, A. S. M. Hossan and H. B. Jannet, Pharm. Biol., 2019, 57, 564–570 CrossRef CAS PubMed.
- S. G. Khanage, A. Raju, P. B. Mohite and R. B. Pandhare, Adv. Pharm. Bull., 2013, 3, 13–18 Search PubMed.
- D. J. Williamson, R. G. Hill, S. L. Shepheard and R. J. Hargreaves, Br. J. Pharmacol., 2001, 133, 1029–1034 CrossRef CAS PubMed.
- J. Dai, S. Tian, X. Yang and Z. Liu, Front. Chem., 2022, 10, 891484 CrossRef CAS PubMed.
- L. Emami, S. Sadeghian, A. Mojaddami, S. khabnadideh, A. Sakhteman, H. Sadeghpour, Z. Faghih, M. Fereidoonnezhad and Z. Rezaei, BMC Chem., 2022, 16, 91 CrossRef CAS PubMed.
- M. Shkoor, H. Tashtoush, M. Al-Talib, I. Mhaidat, Y. Al-Hiari, V. Kasabri and S. Alalawi, Russ. J. Org. Chem., 2021, 57, 1141–1151 CrossRef CAS.
- Y. Li, B. Zhang, H. Yang, Q. Li, P. Diao, W. You and P. Zhao, Eur. J. Med. Chem., 2017, 125, 1098–1106 CrossRef CAS PubMed.
- A. T. A. Boraei, M. S. Gomaa, E. S. H. E. Ashry and A. Duerkop, Eur. J. Med. Chem., 2017, 125, 360–371 CrossRef CAS PubMed.
- S. Ersan, S. Nacak and R. Berkem, Il Farmaco, 1998, 53, 773–776 CrossRef CAS PubMed.
- G. Turan-Zitouni, Z. A. Kaplancıklı, M. T. Yıldız, P. Chevallet and D. Kaya, Eur. J. Med. Chem., 2005, 40, 607–613 CrossRef CAS PubMed.
- R. Chelamalla, V. Akena and S. Manda, Med. Chem. Res., 2017, 26, 1359–1366 CrossRef CAS.
- I. Mir, M. T. Siddiqui and A. Comrie, Tetrahedron, 1970, 26, 5235–5238 CrossRef CAS PubMed.
- B. I. Buzykin, E. V Mironova, V. N. Nabiullin, N. M. Azancheev, L. V Avvakumova, I. K. Rizvanov, A. T. Gubaidullin, I. A. Litvinov and V. V Syakaev, Russ. J. Gen. Chem., 2008, 78, 461–479 CrossRef CAS.
- G. M. Castanedo, P. S. Seng, N. Blaquiere, S. Trapp and S. T. Staben, J. Org. Chem., 2011, 76, 1177–1179 CrossRef CAS PubMed.
- M. Nakka, M. B. Gajula, R. Tadikonda, S. Rayavarapu, P. Sarakula and S. Vidavalur, Tetrahedron Lett., 2014, 55, 177–179 CrossRef.
- V. Siddaiah, G. M. Basha, R. Srinuvasarao and S. K. Yadav, Catal. Lett., 2011, 141, 1511–1520 CrossRef CAS.
- Y. Tian, F. Zhang, J. Nie, C. W. Cheung and J. Ma, Adv. Synth. Catal., 2021, 363, 227–233 CrossRef CAS.
- Z. Chen, H. Li, W. Dong, M. Miao and H. Ren, Org. Lett., 2016, 18, 1334–1337 CrossRef CAS PubMed.
- M. D. Semenova, S. A. Popov, T. S. Golubeva, D. S. Baev, E. E. Shults and M. Turks, ChemistrySelect, 2021, 6, 6472–6477 CrossRef CAS.
- K. Sarac, Russ. J. Org. Chem., 2020, 56, 119–128 CrossRef CAS.
- M. A. Kaldrikyan, N. S. Minasyan and R. G. Melik-Ogandzhanyan, Russ. J. Gen. Chem., 2015, 85, 622–627 CrossRef CAS.
- M. Ahmadi, A. Fasihizad, B. Machura, R. Kruszynski and T. Barak, Polyhedron, 2014, 81, 115–122 CrossRef CAS.
- V. B. Arion, Coord. Chem. Rev., 2019, 387, 348–397 CrossRef CAS.
- R. Takjoo, S. M. M. Moghadam, H. A. Rudbari and G. Bruno, Transition Met. Chem., 2019, 44, 525–534 CrossRef CAS.
- C. Yamazaki, Can. J. Chem., 1975, 53, 610 CrossRef CAS.
- R. L. Siegel, T. B. Kratzer, A. N. Giaquinto, H. Sung and A. Jemal, Ca-Cancer J. Clin., 2025, 75, 10–45 CrossRef PubMed.
- G. M. Sheldrick, Acta Crystallogr. A, 2015, 71, 3–8 CrossRef PubMed.
- O. V. Dolomanov, L. J. Bourhis, R. J. Gildea, J. A. K. Howard and H. Puschmann, J. Appl. Crystallogr., 2009, 42, 339–341 CrossRef CAS.
- G. M. Sheldrick, Acta Crystallogr. C, 2015, 71, 3–8 CrossRef PubMed.
- J. Jayadharini, S. Swaminathan, M. S. M. Kasim, N. Bhuvanesh and R. Karvembu, Organometallics, 2025, 44, 1545–1557 CrossRef CAS.
- M. C. Karthikeyan, C. Srinivasan, K. Prabhakar, P. Manogar, A. Jayaprakash and A. J. V. Arockiam, Med. Oncol., 2024, 41, 220 CrossRef CAS PubMed.
- A. Arunachalam, R. Rengan, D. Umapathy and A. J. V. Arockiam, Organometallics, 2022, 41, 2474–2486 CrossRef CAS.
- B. İ. Ceylan, Y. D. Kurt and B. Ülküseven, J. Coord. Chem., 2009, 62, 757–766 CrossRef CAS.
- O. Pintilie, L. Profire, V. Sunel, M. Popa and A. Pui, Molecules, 2007, 12, 103–113 CrossRef CAS PubMed.
- R. Takjoo, R. Centore, A. Akbari and M. Ahmadi, Polyhedron, 2014, 80, 243–249 CrossRef CAS.
- B. A. Varynskyi, M. A. Scherback, A. G. Kaplaushenko and I. A. Yurchenko, J. Chem. Pharm. Res., 2014, 6, 1342–1350 Search PubMed.
- N. Balakrishnan, J. Haribabu, M. Dharmasivam, J. P. Jayadharini, D. Anandakrishnan, S. Swaminathan, N. Bhuvanesh, C. Echeverria and R. Karvembu, Organometallics, 2023, 42, 259–275 CrossRef CAS.
- A. H. A. Mercy, P. P. V R, N. K, R. Kataria and G. C. Nandi, J. Org. Chem., 2025, 90, 830–839 CrossRef PubMed.
- N. E. A. Ravindran, R. J. Deepak, N. Bhuvanesh and R. Karvembu, Inorg. Chem. Commun., 2024, 168, 112963 CrossRef.
- G. M. Reddy, J. R. Garcia, V. H. Reddy, A. M. de Andrade, A. Camilo Jr., R. A. P. Ribeiro and S. R. de Lazaro, Eur. J. Med. Chem., 2016, 123, 508–513 CrossRef CAS PubMed.
- K. Nepali, H. Lee and J. Liou, J. Med. Chem., 2019, 62, 2851–2893 CrossRef CAS PubMed.
- M. J. Strauss, Ind. Eng. Chem. Prod. Res. Dev., 1979, 18, 158–166 CrossRef CAS.
- S. Swaminathan, J. Haribabu, N. K. Kalagatur, R. Konakanchi, N. Balakrishnan, N. Bhuvanesh and R. Karvembu, ACS Omega, 2019, 4, 6245–6256 CrossRef CAS PubMed.
- (a) CCDC2506364: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q42gf; (b) CCDC2506370: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q42nm; (c) CCDC2506371: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q42pn; (d) CCDC2506359: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q4298; (e) CCDC2506360: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q42b9; (f) CCDC2506361: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q42cb.
| This journal is © The Royal Society of Chemistry 2026 |