 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Boosting PVC reactivity through iodination for CO2-derived functionalization
Juliette Delcorps,
Emna Ben Ayed and
Olivier Coulembier *
*
Laboratory of Polymeric and Composite Materials, Centre of Innovation and Research in Materials and Polymers, University of Mons, Place du Parc 23, 7000 Mons, Belgium. E-mail: olivier.coulembier@umons.ac.be
First published on 6th March 2026
Abstract
Aqueous phase-transfer catalysis enables selective iodination of PVC under mild and sustainable conditions. Subsequent reaction with a DBU-based CO2-binding amine (CO2BAM) achieves efficient SN2 substitution at CH–X centers, improving CO2 incorporation fourfold relative to previous CO2BAL strategies. The polymer remains fully soluble, demonstrating a greener, safer, and effective approach to PVC structural modification.
Introduction
Among the many challenges facing sustainable materials science, the simultaneous management of carbon dioxide (CO2) and halogenated plastics stands out as both urgent and conceptually rich. Poly(vinyl chloride) (PVC), the third most produced polymer worldwide, combines large-scale utility with a notorious resistance to circularity.1–5 Its chlorine content hinders conventional recycling, while its incineration generates corrosive and toxic emissions.6 In parallel, the chemical community continues to seek viable strategies for CO2 utilization, not merely for capture, but for its integration into value-added materials.7–13 Bringing these two issues together, the chemical modification of PVC through CO2-based chemistry represents a promising step toward genuine carbon circularity.Our group recently explored this concept using CO2-binding alcohols (CO2BALs), a subclass of CO2-binding organic liquids (CO2BOLs).14 These dual-function reagents acted both as CO2 carriers and as nucleophiles capable of reacting with PVC through substitution, enabling the partial incorporation of carbonate functionalities into the polymer backbone.15 In our previous study, the CO2BAL was generated from 1-decanol and 1,5,7-triazabicyclo[4.4.0]dec-5-ene (TBD) by bubbling CO2 at 0.2 bar and 90 °C, reaching a conversion of approximately 80%. While this approach elegantly demonstrated the feasibility of CO2-mediated PVC functionalization, the overall degree of incorporation remained limited to a 7 wt% mass substitution corresponding to only ca. 2.5 mol% of the methine sites along the chain, after 2 hours of reaction. In terms of CO2 loading, this corresponds to a maximum of 17.6 g per kg of PVC (Fig. 1a).
 | ||
| Fig. 1 (A) Previously reported reactivity of CO2BALs in SN2 with pristine PVC. See ref. 14; (B) this work: enhancement of substitution rate via PVC iodination and the use of CO2BAMs. With TBD = 1,5,7-triazabicyclo[4.4.0]dec-5-ene, DBU = 1,8-diazabicyclo[5.4.0]undec-7-ene, SN2 = second-order nucleophilic substitution and E2 = second-order elimination. | ||
Reasoning that a greener and more efficient CO2-mediated functionalization of PVC would benefit from the use of safer and renewable reagents while improving reaction selectivity, we turned our attention to amine-based CO2-binding agents (CO2BAMs). In contrast to the carbonate linkages formed with CO2BALs, the carbamate bonds formed by CO2BAMs are thermodynamically more stable due to resonance delocalization between the nitrogen lone pair and the carbonyl group. This higher stability is expected to improve the durability of the grafted structure,16,17 its compatibility with further transformations, and ultimately the sustainability of an expected upcycling process.
In this framework, primary amines were identified as the most suitable nucleophilic partners for CO2 activation.18 Among them, 1-hexylamine was selected for its favorable green chemistry profile and potential renewability. Linear aliphatic amines such as 1-hexylamine can be synthesized from biomass-derived alcohols via catalytic “hydrogen-borrowing” amination.19 This transformation is fully atom-economic, producing only water as a by-product and aligning with principles of green chemistry. In parallel, we sought to replace the costly and moisture-sensitive TBD guanidine used in CO2BAL synthesis with a more affordable and robust organic base. 1,8-Diazabicyclo[5.4.0]undec-7-ene (DBU) was selected for its economic advantage (100 ml costs approximately €109, whereas 5 g of TBD amounts to €55.4) as well as for its distinct mechanistic benefits.20,21 As reported by McGhee et al.,22 DBU efficiently promotes carbamate anion formation and enhances SN2 selectivity in urethane-type bond formation under mild CO2 pressure, a feature recently exploited in continuous flow conditions by Bica-Schröder and co-workers.23
To further enhance substitution kinetics while addressing one of the major safety and sustainability issues of PVC recycling, i.e. the release of corrosive hydrogen chloride, we reasoned that a partial iodination of PVC prior to CO2BAM treatment could mitigate acid-gas formation. Although hydrogen iodide is a stronger acid than hydrogen chloride, its vapours are less corrosive toward metallic equipment and form less volatile metal halides.24–26 In addition, iodinated PVC displays higher SN2 reactivity, facilitating more efficient CO2 incorporation under mild conditions.
Overall, as presented herein, this dual approach, combining CO2BAMs with a greener preparation of iodinated PVC (Fig. 1b), provides a safer and more sustainable pathway for covalent CO2 upcycling into polymeric materials. Under conditions directly comparable to those previously employed for CO2BAL-mediated functionalization, this strategy increases the efficiency of SN2 substitution by roughly fourfold (10 mol% substitution rate and max. CO2 content ∼70.5 g per kg of polymer), yielding a polymer that remains fully soluble in organic media despite the formation of some vinyl groups via E2 elimination. Together, these features highlight a clear improvement in both the efficiency and versatility of CO2 incorporation into PVC (Table 1).
| Parameter | CO2BAL Strategy | CO2BAM Strategy |
|---|---|---|
| Previous work15 | This work | |
| Polymer support | PVC | Partially iodinated PVC |
| CO2 carrier activation | 1-Decanol + TBD | 1-Hexylamine + DBU |
| Purity of activated species | ∼80% (20 mol% free base) | ∼98% |
| Nucleophile | Carbonate | Carbamate |
| Bond formed | Carbonate linkage | Carbamate linkage |
| SN2/E2 behaviour | Limited substitution | Enhanced SN2 selectivity |
| Degree of substitution | ∼2.5 mol% | ∼10 mol% |
| Max CO2 content | ∼17.6 g per kg of PVC | ∼70.5 g per kg of PVC |
| Solubility | Soluble | Fully soluble |
| Base | TBD (moisture sensitive, costly) | DBU (robust, affordable) |
Results and discussion
PVC iodination
In the literature, the iodination of PVC is most frequently carried out through the Conant–Finkelstein reaction using NaI in THF/acetone (Scheme 1a).27–29 However, this protocol relies on a problematic solvent system30 and does not consistently deliver high iodination efficiency. Although bio-based THF has been reported,31–33 replacing this mixture with safer and more sustainable media remains an important objective. An alternative route proposed by Lakshmi and Jayakrishnan uses water as solvent under phase-transfer catalysis (PTC, Scheme 1b),34 offering a greener and underexplored approach. | ||
| Scheme 1 PVC iodination via the Conant–Finkelstein reaction (A) and phase-transfer catalysis (B). TBAB = tetrabutylammonium bromide. | ||
To establish a clear baseline for comparison, the conventional Conant–Finkelstein reaction was first reproduced on a smaller scale following the protocol of Rusen et al.29 This benchmark experiment not only provided direct insight into the substitution pattern and material response under classical conditions but also served as a reference for assessing the efficiency, selectivity, and environmental performance of the PTC-based alternative developed herein. After 24 hours at 40 °C, the polymer exhibited a faint yellow coloration, consistent with partial chlorine-to-iodine substitution (Fig. 2). Size exclusion chromatography (SEC) revealed a moderate increase in number-average molar mass (Mn), while thermogravimetric analysis (TGA) showed a 32 °C decrease of the maximal degradation temperature (MDT), both supporting a PVC backbone substitution (Fig. S3 and S4). However, conventional spectroscopic techniques such as 1H and 13C NMR or FT-IR failed to provide unambiguous structural evidence, as the spectral features of PVC remain largely unaffected (Fig. S2).
 | ||
| Fig. 2 Color evolution of PVC after 24 h via the Conant–Finkelstein (C–F) reaction, and after 0, 6, 17, 24, and 48 h of iodination via PTC. | ||
To obtain quantitative insight, X-ray photoelectron spectroscopy (XPS) was employed to determine the surface iodine content, within the 1–10 nm sampling depth (Fig. S5 and S6). Although this technique does not probe the bulk of the film (ca. 70 µm thick), it offers a reliable benchmark for comparing different iodination methods. The XPS spectrum indicated an iodine content of 0.10 at%, confirming a moderated successful chlorine substitution ≤3% (Fig. S6 and Table S1 – entry 2). A minor sodium signal was also detected, suggesting the presence of residual NaI despite purification. Consequently, this value likely represents a slight overestimation of the true iodine incorporation. Overall, this experiment establishes the Conant–Finkelstein reaction as an effective yet limited benchmark, providing a quantitative baseline for evaluating greener iodination strategies under comparable conditions.
Building on the promising report by Lakshmi and Jayakrishnan,34 the phase-transfer-catalyzed (PTC) iodination of PVC was then reinvestigated. The optimized conditions reported in their original study were maintained, while reaction times were increased to enhance substitution efficiency. Reactions were thus conducted with tetrabutylammonium bromide (TBAB) used as transfer agent at 80 °C for durations of 6, 17, 24, and 48 h ([KI]0 = 6 M, [KI]0/[TBAB]0 = 40). After completion, the samples were thoroughly washed, dried, and solvent-casted into thin films suitable for subsequent analysis. A simple visual inspection immediately reflected the progress of iodination; while pristine PVC films remained transparent, the treated samples displayed a distinct yellow coloration that deepened with increasing reaction time (Fig. 2). Importantly, all materials remained soluble in conventional PVC solvents (THF and DMF), and none exhibited the darkening typical of extensive dehydrohalogenation. 1H NMR and FT-IR analyses further confirmed that elimination reactions were minimal, with the fraction of unsaturated sites remaining below 1 mol% even after 48 h (Fig. S1 and S2). This negligible degradation attests to the mildness and selectivity of the PTC approach, which proceeds efficiently under aqueous conditions without compromising polymer integrity. With the exception of the 6 h sample, all PTC-treated films exhibited a deeper yellow hue than the Conant–Finkelstein analogue, suggesting higher iodination levels. This trend was confirmed by TGA, which revealed a more pronounced MDT decrease upon increasing reaction time (Fig. S3). The relatively modest MDTs variation in TGA thermograms among the PTC samples aligns with earlier observations by Lakshmi et al., who reported that substitution efficiency plateaued after prolonged exposure.34
SEC data showed an initial increase in Mn during the first 6 hours of reaction, followed by a gradual decrease at longer times (Fig. S4). Since SEC determines apparent molar masses based on hydrodynamic volume rather than absolute molecular weight, these variations reflect conformational changes in solution rather than backbone scission. Attractive intramolecular interactions are known to reduce the hydrodynamic radius of polymer chains in solution, leading to lower apparent molar masses when calibrated against polystyrene standards.35 Given the negligible extent of elimination, the progressive decrease in Mn at longer reaction times is therefore attributed to a reduction in hydrodynamic volume as the polymer backbone becomes increasingly iodinated. As the most polarizable and least electronegative halogen, iodine exhibits the strongest σ-hole donor character and forms particularly robust halogen bonds.36 Increasing iodine content thus enhances the likelihood of intra- and intermolecular halogen-bonding interactions, which promote chain compaction in solution and consequently lower the apparent Mn.
XPS measurements substantiated these findings, revealing a steady increase in iodine surface content up to 0.84 at% after 48 h, suggesting a chlorine substitution ≤24.5% (Fig. 3, S5–S9 and Table S1 – entries 3–6). In parallel, water static contact-angle measurements showed no statistically significant change between pristine PVC (85.6 ± 1.7°) and PTC-iodinated films (83.4 ± 4.3°, after 48 h), indicating that the surface wettability remains essentially unchanged despite iodine incorporation. In accord, DSC analysis showed a pronounced decrease in the glass-transition temperature, Tg (from 86 °C for pristine PVC to 50 °C after 48 h of iodination), reflecting local structural relaxation induced by the introduction of bulky iodinated groups (Fig. S10 and Table S1). Residual TBAB still detected by 1H NMR after purification was found to be negligible and therefore cannot account for the observed decrease in Tg. Instead, this reduction is attributed to a self-plasticization effect arising from the incorporation of iodinated substituents, consistent with previous reports on bulky-substituted PVC derivatives.37,38
CO2BAM selection and reaction with iodinated PVC
Primary and secondary alkylamines react readily with CO2 at ambient pressure and temperature, forming carbamate or carbamic acid adducts,18 depending on the nature of the medium. Early reports also highlighted that the addition of organic superbases, such as amidines or guanidines, is critical for stabilizing certain amine carboxylations, enhancing both yield and nucleophilicity.18,39,40 Guided by this principle, three CO2-binding amines (CO2BAMs) were prepared from the representative primary amine, 1-hexylamine, for comparative evaluation. When CO2 was bubbled into the pristine liquid alkylamine, 1H NMR analysis of the medium indicated that approximately 43% of the amine reacted (Fig. S11). In this polar protic medium, and mirroring reactions performed in water,41 the reaction proceeds via formation of the carbamic acid, which is immediately deprotonated by another molecule of free amine (Scheme 2). By adding one equivalent of strong base such as 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU) or 1,5,7-triazabicyclo[4.4.0]dec-5-ene (TBD), only the 1-hexylamine acts as a Lewis base while the superbase acts as Brønsted base pushing conversions in carbamate salts to 95 and 90%, respectively (Fig. S12 and S13).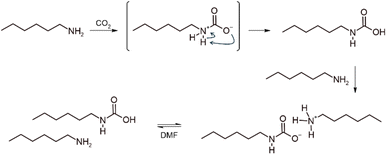 | ||
| Scheme 2 Preparation of a CO2BAM of 1-hexylamine through CO2 activation. In DMF, the carbamate form should be in equilibrium with its carbamic homologue. | ||
Performing nucleophilic substitution directly in the bulk iodinated PVC represents the greenest approach to avoid the use of solvents entirely. Pristine PVC softens around 92 °C,42 whereas iodination lowers its Tg to 50–55 °C, suggesting a softening temperature near 60 °C. The three CO2BAMs were thus subjected to a TGA analysis to estimate their degradation onset temperatures (T5%, Fig. S15). Even the most thermally robust DBU-based CO2BAM (T5% = 65 °C) leaves only a narrow margin, precluding safe bulk processing, making the solvent-free strategy impractical, and the choice of a solvent necessary.
DMF was selected to ensure solubility of both iodinated PVC and carbamate salts. Although DMF is not ideal from a toxicity standpoint, the reaction conditions (see Experimental Section) produce only aqueous DMF effluents, which can be efficiently recovered via membrane-based techniques (pervaporation, nanofiltration), avoiding high-energy distillation or incineration.43 The use of DMF also informs the choice of CO2BAM for SN2 functionalization of iodinated PVC. Under a saturated CO2 atmosphere, the carbamate form of CO2BAM obtained without an organic strong base exists in equilibrium with its carbamic acid form (Scheme 2),18 precluding its use as a nucleophile and making it ineffective for SN2 substitution of the iodinated PVC. For CO2BAMs prepared with DBU or TBD, we previously showed that residual free base in CO2-binding alcohols (CO2BALs) promotes dehydrochlorination, forming carbon–carbon double bonds and provoking precipitation of the modified PVC. Among the CO2BAMs tested here, DBU provides the highest conversion in carbamate form (95%), whereas TBD reaches only 90% conversion. Considering its lower basicity (pKaDBU.H+ = 24.3 vs. pKaTBD.H+ = 26.0 in ACN), as well as the greater accessibility and lower cost of DBU, the DBU-based CO2BAM was selected as the optimal reagent for nucleophilic substitution on iodinated PVC. This choice is further supported by the excellent stability of the DBU-based CO2BAM in DMF over time (Fig. S14). Under identical experimental conditions as those used for the subsequent reaction with iodinated PVC ([CO2BAM]0 = 0.5 M, 21 °C, CO2-saturated atmosphere), the carbamate salt remained indeed fully stable for at least 24 h, with very little to no sign of CO2 release.
Considering the limitations of our analytical methods, it was not possible to reliably determine the iodine content of PVC-I. TGA of the iodinated material after 48 h shows an initial mass-loss event of 11–12 wt% between 90 and 140 °C, followed by a broader 53–54 wt% degradation from 140 to 400 °C (Fig. S3). For comparison, unmodified PVC typically exhibits a two-step thermal decomposition, with a major initial mass loss of ∼63 wt% and a plateau associated with char formation.44 This first step is often attributed to HCl elimination, but the overall mechanism is highly complex, defect-dependent, and involves overlapping processes such as dehydrohalogenation and backbone scission.45 In PVC-I, the splitting of the initial mass loss into two distinct events likely reflects the same intrinsic complexity, and the early 12 wt% loss cannot be unambiguously assigned to iodine elimination alone. Notably, this initial mass loss is substantially higher than the iodine surface content inferred from XPS (0.84 at%), further emphasizing the fundamentally different information depth of both techniques and precluding any direct quantitative comparison. Consequently, neither TGA, XPS nor NMR spectroscopy provides a reliable estimate of the degree of iodination. As a result, the absolute molar mass of PVC-I remains inaccessible, preventing accurate determination of a defined [halogen]0/[CO2BAM]0 ratio. To allow a meaningful comparison with the reactivity of pristine PVC toward CO2BALs reported in our previous study,15 we therefore elected to use the absolute number-average molar mass of unmodified PVC (Mn,SEC = 41![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 400 g mol−1, ĐM = Mw/Mn = 2.2), determined in THF at 25 °C using PS calibration and Mark–Houwink corrections, as the reference value to compute the amount of CO2BAM. The reaction in DMF at 21 °C with the DBU-based CO2BAM derived from 1-hexylamine was thus conducted using this Mn,SEC to impose an initial [halogen]0/[CO2BAM]0 molar ratio of 1, enabling direct comparison with the corresponding SN2 reactivity observed in the CO2BAL series. In our previous study, we demonstrated that the degree of carbonatation reached 2.5 mol% after 2 hours at 21 °C.15 As a proof-of-concept of our new strategy, the reaction between PVC-I and the DBU-based CO2BAM was then reiterated in DMF for 2 hours only ([PVC-I]0 = 0.7 mmol L−1). The reaction medium was subsequently precipitated in cold water, washed, and dried to ensure complete removal of CO2BAM derivatives. The structural changes induced by the reaction between PVC-I and the DBU-based CO2BAM were first evaluated by FTIR spectroscopy (Fig. S16). The appearance of a weak band at 1724 cm−1 is consistent with the presence of carbonyl groups, in line with the partial formation of carbamate-type motifs upon nucleophilic substitution. Concomitantly, a distinct absorption at 1643 cm−1 clearly evidences the formation of C
400 g mol−1, ĐM = Mw/Mn = 2.2), determined in THF at 25 °C using PS calibration and Mark–Houwink corrections, as the reference value to compute the amount of CO2BAM. The reaction in DMF at 21 °C with the DBU-based CO2BAM derived from 1-hexylamine was thus conducted using this Mn,SEC to impose an initial [halogen]0/[CO2BAM]0 molar ratio of 1, enabling direct comparison with the corresponding SN2 reactivity observed in the CO2BAL series. In our previous study, we demonstrated that the degree of carbonatation reached 2.5 mol% after 2 hours at 21 °C.15 As a proof-of-concept of our new strategy, the reaction between PVC-I and the DBU-based CO2BAM was then reiterated in DMF for 2 hours only ([PVC-I]0 = 0.7 mmol L−1). The reaction medium was subsequently precipitated in cold water, washed, and dried to ensure complete removal of CO2BAM derivatives. The structural changes induced by the reaction between PVC-I and the DBU-based CO2BAM were first evaluated by FTIR spectroscopy (Fig. S16). The appearance of a weak band at 1724 cm−1 is consistent with the presence of carbonyl groups, in line with the partial formation of carbamate-type motifs upon nucleophilic substitution. Concomitantly, a distinct absorption at 1643 cm−1 clearly evidences the formation of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C double bonds, confirming that elimination processes also occur under these conditions. Additional features, namely the irregular envelope between 2000–2400 cm−1 and the broad, intense band between 3100–3600 cm−1, are characteristic of overlapping O–H, N–H and weak overtone/combination contributions.
C double bonds, confirming that elimination processes also occur under these conditions. Additional features, namely the irregular envelope between 2000–2400 cm−1 and the broad, intense band between 3100–3600 cm−1, are characteristic of overlapping O–H, N–H and weak overtone/combination contributions.
More detailed insight was obtained from the 13C NMR spectrum of the modified polymer (Fig. 4). While the characteristic triplet-type CH–X pattern of PVC (δ = 56.87, 57.76 and 58.75 ppm, with X = Cl or I) is preserved, three new resonances emerge at δ = 57.08, 58.03 and 59.05 ppm. Their chemical-shift proximity and analogous triplet-like distribution strongly suggest that the modification remains localized at the CH–X carbons of the polymer backbone. The slight downfield displacement of these new signals (∼0.2–0.3 ppm) is fully consistent with partial nucleophilic substitution by CO2BAM-derived species or by hexylamine liberated in situ, although the relative contributions of carbamate retention and CO2 loss cannot be unambiguously established at this stage. Nevertheless, the appearance of these carbon resonances provides direct spectroscopic evidence for covalent grafting at the CH–X positions, supporting the occurrence of SN2-type substitution in competition with elimination.
The 1H NMR spectrum corroborates this mixed reactivity (Fig. S17). Integration of the vinylic protons (δ = 5.5–6.0 ppm) indicates 4.6 mol% of unsaturation, while signals attributable to the grafted hexyl group (δ = 0.7–1.5 ppm) correspond to 10.1 mol% of covalent incorporation. Taken together, these spectroscopic data depict a reaction manifold in which CO2BAM-derived nucleophiles engage in efficient substitution at the CH–X centers of PVC-I while a concomitant fraction of sites undergo base-promoted elimination, ultimately yielding a functionalized material with dual substitution–elimination character. Importantly, despite the formation of unsaturations, the polymer remained fully soluble throughout the reaction, in contrast to our previous observations with TBD-derived CO2BALs.15 This preserved solubility strongly suggests that the substantially higher degree of substitution achieved under CO2BAM/DBU conditions effectively outcompetes the elimination-inducing pathways observed in our earlier study.
Conclusions
We report a sustainable, dual-strategy approach for covalent CO2 incorporation into PVC. Aqueous PTC-mediated iodination enhances SN2 reactivity, while DBU-based CO2BAMs provide stable, efficient nucleophiles for functionalization. This method increases substitution efficiency compared to CO2BALs, preserves polymer solubility and highlights an efficient and greener pathway for PVC chemical modification. Systematic studies on different CO2BAM alkyl types and reaction times are currently underway, and a comparative evaluation of the resulting polymer properties will be presented in a forthcoming paper.Experimental
Materials
Poly(vinyl chloride) (low molecular weight, Mnth = 48 kg mol−1, MnSEC = 41.4 kg mol−1),15 tetrabutylammonium bromide (TBAB), and N,N-dimethylformamide (anhydrous, 99.8%) were purchased from Sigma-Aldrich. Potassium iodide (KI), acetone p., and methanol p. were supplied by Chemlab. Sodium iodide (NaI) as well as deuterated tetrahydrofuran (99.5% D) were acquired from VWR. 1-hexylamine (99%) and 1,8-diazabicyclo[5.4.0]undec-7-ene (98+%) were supplied by Thermo Scientific. Finally, carbon dioxide N50 (≥99.999%), 1,5,7-triazabicycl[4.4.0]dec-5-ene, and deuterated chloroform (99.8%D + 0.03% TMS V/V, for NMR spectroscopy) were purchased from Air Liquide, Apollo Scientific, and Eurisotop, respectively.Experimental procedures
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) mixture. The reaction mixture was then stirred under nitrogen at 40 °C for 24 hours. Once the 24 hours were up, the reaction medium was precipitated in a methanol/water (2
2) mixture. The reaction mixture was then stirred under nitrogen at 40 °C for 24 hours. Once the 24 hours were up, the reaction medium was precipitated in a methanol/water (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) mixture. The precipitate was then dried overnight under vacuum at 40 °C.
1) mixture. The precipitate was then dried overnight under vacuum at 40 °C.Analytical methods
Thermogravimetric analysis (TGA)
The CO2BAMs were subjected to thermogravimetric analysis to determine their degradation temperature. Measurements were performed under nitrogen flow at a rate of 5 °C per minute using a TA Q500 TGA. All PVC-based samples were subjected to TGA as well. Measurements were performed under nitrogen flow at a rate of 2.5 °C per minute using a TA Q500 TGA.Fourier transform infraRed (FT-IR) spectroscopy
FT-IR spectroscopy was performed using a Bruker Tensor 27 spectrometer to give an indication regarding the success of the substitution reaction between PVC and CO2BAM.Nuclear magnetic resonance (NMR) spectroscopy
CO2 conversion was followed time-to time through 1H NMR using a Spinsolve 60 Ultrabenchtop NMR spectrometer (Magritek, Aachen, Germany). All spectra were acquired according to the following parameters: aq = 6.4 s, repetition time = 10 s, pulse angle = 90, ns = 32. Final CO2BAM and substitution products were all characterized by 1H NMR using a Bruker AVANCEII 500 MHz apparatus at room temperature in chloroform-d1 (CDCl3) and THF-d8, respectively. All spectra were acquired according to the following parameters: aq = 2.75 s, d1 = 1 s, sw = 20, ns = 32 for small molecules and 512 for PVC-based compounds. 13C NMR spectra were also acquired using the a Bruker AVANCEII 500 MHz apparatus at room temperature in THF-d8 according to the following parameters: aq = 1.1 s, d1 = 2 s, sw = 240, ns = 4096.Size exclusion chromatography (SEC)
All PVC-based samples were subjected to SEC in THF at 25 °C using a Triple Detection Polymer Laboratories liquid chromatograph equipped with a refractive index (ERMA 7517), a UV detector (254 nm), a capillary viscometer, a light scattering RALS (Viscotek T-60) (Polymer Laboratories GPC-RI/CV/RALS), an automatic injector (Polymer Laboratories GPC-RI/UV), and four columns: a PL gel 10 µm guard column and three PL gel mixed-B 10 µm columns (linear columns for separation of Mw PS ranging from 500 to 106 daltons). All samples for SEC analyses were prepared according to the following concentration: 1 mg of polymer sample in 1 mL of THF.Differential scanning calorimetry (DSC)
DSC analysis was conducted using a DSC Q2000 from TA instruments under inert atmosphere (N2/50 ml min−1). Samples of 3–4 mg were sealed in aluminum DSC pans, placed in the DSC cell and heated from −20 to 80 or 100 °C with a heating rate of 10 °C min−1. The glass transition temperature (Tg) was measured on the second heating scan.X-ray photoelectron spectroscopy (XPS)
The sample composition was evaluated using XPS with a VERSAPROBE PHI 5000 instrument from Physical Electronics. The instrument was equipped with a monochromatic Al Kα X-ray source. X-ray photoelectron spectra were collected at a takeoff angle of 45°, and the electron energy analyzer was operated in the CAE (constant analyser energy) mode. To compensate for the built-up charge on the sample surface during measurements, a dual beam charge neutralization system consisting of an electron gun (approximately 1 eV) and an Ar ion gun (less than or equal to 10 eV) was employed. The binding energies were referenced to the C1s peak at 284.6 eV. Atomic concentration percentages were determined using CasaXPS software based on the XPS data.Contact angle
To perform contact angle measurements, films were made out of each sample by solvent casting. These films were casted directly onto microscope slides for easier handling. These measurements were performed using a Krüss Drop Shape Analysis System DSA 10 Mk2. 20 µl droplets of demineralized water were deposited in various places on the polymer film and contact angles were measured using the Drop Shape Analysis software.Author contributions
conceptualization: O. C.; methodology: J. D.; validation: J. D. and E. B. A.; investigation: J. D. and E. B. A.; writing original draft: J. D.; writing– review & editing: O. C., J. D., and E. B. A.; visualization: J. D., E. B. A., and O. C.; supervision: O. C.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this study, including spectroscopic, thermal, and chromatographic analyses, are available in the supplementary information (SI) of this article. No additional datasets were generated or analyzed beyond those included in the main text and SI. Supplementary information is available. See DOI: https://doi.org/10.1039/d6ra00749j.Acknowledgements
J. D. and E. B. A. acknowledge the support of the AXA Research Fund for the funding of this project. O. C. acknowledges support for his position as a Research Director for the F.R.S.-FNRS of Belgium and AXA Professor in Chemistry.References
- The Circular Economy for Plastics - A European Analysis, Plastics Europe, 2024.
- A. Buekens and A. Sevenster, J. Mater. Cycles Waste Manag., 2010, 12, 184–192 CrossRef CAS.
- K. Lewandowski and K. Skórczewska, Polymers, 2022, 14, 3035 CrossRef CAS PubMed.
- Z. Ait-Touchente, M. Khellaf, G. Raffin, N. Lebaz and A. Elaissari, Polym. Adv. Tech., 2024, 35, e6228 CrossRef CAS.
- M. Sadat-Shojai and G.-R. Bakhshandeh, Polym. Degrad. Stab., 2011, 96, 404–415 CrossRef CAS.
- X. Jiang, B. Zhu and M. Zhu, Green Chem., 2023, 25, 6971–7025 RSC.
- S. H. Kim, K. H. Kim and S. H. Hong, Angew. Chem. Int. Ed., 2014, 53, 771–774 CrossRef CAS PubMed.
- Q. Liu, L. Wu, R. Jackstell and M. Beller, Nat. Commun., 2015, 6, 5933 CrossRef PubMed.
- B. Grignard, S. Gennen, C. Jérôme, A. W. Kleij and C. Detrembleur, Chem. Soc. Rev., 2019, 48, 4466–4514 RSC.
- C. Hepburn, E. Adlen, J. Beddington, E. A. Carter, S. Fuss, N. Mac Dowell, J. C. Minx, P. Smith and C. K. Williams, Nature, 2019, 575, 87–97 CrossRef CAS PubMed.
- Y. Liu and X.-B. Lu, Macromolecules, 2023, 56, 1759–1777 CrossRef CAS.
- X. Xiaoding and J. A. Moulijn, Energy Fuels, 1996, 10, 305–325 Search PubMed.
- Z. Li, R. Shi, Y. Ma, J. Zhao and T. Zhang, J. Phys. Chem. Lett., 2022, 13, 5291–5303 CrossRef CAS PubMed.
- J. Delcorps, K. S. Rawat, M. Wells, E. Ben Ayed, B. Grignard, C. Detrembleur, B. Blankert, P. Gerbaux, V. Van Speybroeck and O. Coulembier, Cell Rep. Phys. Sci., 2024, 5, 102057 Search PubMed.
- J. Delcorps, E. Ben Ayed and O. Coulembier, Polym. Chem., 2025, 16, 2923–2932 RSC.
- L. W. Dittert and T. Higuchi, J. Pharm. Sci., 1963, 52, 852–857 Search PubMed.
- M. Takahashi, S. Sakamoto, N. Sakamoto, S. Nishimura, S. Hwang, J. Kim and M. Hosokawa, J. Pharm. Sci., 2026, 115, 104146 Search PubMed.
- J. K. Mannisto, L. Pavlovic, T. Tiainen, M. Nieger, A. Sahari, K. H. Hopmann and T. Repo, Catal. Sci. Technol., 2021, 11, 6877–6886 RSC.
- A. M. Afanasenko, N. Deak, J. October, R. Sole and K. Barta, Green Chem., 2025, 27, 5947–5981 RSC.
- D. J. Heldebrant, C. R. Yonker, P. G. Jessop and L. Phan, Energy Environ. Sci., 2008, 1, 487–493 Search PubMed.
- D. J. Heldebrant, P. G. Jessop, C. A. Thomas, C. A. Eckert and C. L. Liotta, J. Org. Chem., 2005, 70, 5335–5338 Search PubMed.
- W. McGhee, D. Riley, K. Christ, Y. Pan and B. Parnas, J. Org. Chem., 1995, 60, 2820–2830 Search PubMed.
- K. Stagel, L. Ielo and K. Bica-Schröder, ACS Omega, 2023, 8, 48444–48450 CrossRef CAS PubMed.
- Y. Imai, Y. Kanda, H. Sasaki and H. Togano, Corrosion Eng., 1982, 31, 714–721 Search PubMed.
- J. Y. Choi, Y. S. Kim, I. Sah, H. C. No and C. Jang, Int. J. Hydrogen Energy, 2014, 39, 14557–14564 CrossRef CAS.
- R. Kaczmarczyk and S. Gurgul, Energies, 2024, 17, 3223 CrossRef CAS.
- S. Moulay and Z. Zeffouni, J. Polym. Res., 2006, 13, 267–275 CrossRef CAS.
- N. A. El-Ghamaz and H. A. Ghaly, Chem. Phys. Lett., 2016, 648, 66–74 CrossRef CAS.
- E. Rusen, R. Şomoghi, C. Busuioc and A. Diacon, RSC Adv., 2020, 10, 35692–35700 RSC.
- O. Clavilier, D. Foy and F. Byrne, Green Chem., 2025, 27, 12151–12159 Search PubMed.
- Y. Zhu, J. Yang, F. Mei, X. Li and C. Zhao, Green Chem., 2022, 24, 6450–6466 RSC.
- J. Lange and S. H. Wadman, ChemSusChem, 2020, 13, 5329–5337 CrossRef CAS PubMed.
- V. M. Shinde, G. N. Patil, A. Katariya and Y. S. Mahajan, Chem. Eng. Process. Process Intensif., 2015, 95, 241–248 CrossRef CAS.
- S. Lakshmi and A. Jayakrishnan, J. Appl. Polym. Sci., 2002, 84, 493–499 CrossRef CAS.
- A. M. Striegel, Anal. Bioanal. Chem., 2012, 402, 77–81 CrossRef CAS PubMed.
- G. Cavallo, P. Metrangolo, R. Milani, T. Pilati, A. Priimagi, G. Resnati and G. Terraneo, Chem. Rev., 2016, 116, 2478–2601 Search PubMed.
- P. Jia, M. Zhang, L. Hu, R. Wang, C. Sun and Y. Zhou, Polymers, 2017, 9, 621 CrossRef PubMed.
- L. Lu, S. Kumagai, T. Kameda, L. Luo and T. Yoshioka, RSC Adv., 2019, 9, 28870–28875 RSC.
- W. D. McGhee, D. P. Riley, M. E. Christ and K. M. Christ, Organometallics, 1993, 12, 1429–1433 CrossRef CAS.
- M. Costa, G. P. Chiusoli and M. Rizzardi, Chem. Commun., 1996, 1699 RSC.
- P. V. Kortunov, M. Siskin, L. S. Baugh and D. C. Calabro, Energy Fuels, 2015, 29, 5919–5939 CrossRef CAS.
- Heat distortion temperature, https://pvc.org/about-pvc/pvcs-physical-properties/heat-distortion-temperature/, (accessed 9 August 2024).
- Z. Zhang, F. Ruan, S. Xu, W. Wu, S. Shi and G. Xiao, Process Saf. Environ. Prot., 2022, 159, 779–794 CrossRef CAS.
- N. Choudhury, A. Kim, M. Kim and B.-S. Kim, Adv. Mater., 2023, 35, 2304113 CrossRef CAS PubMed.
- K. G. Papanikolaou, J. Wu, G. W. Huber and M. Mavrikakis, J. Polym. Res., 2023, 30, 83 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |


