 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Stability and hydrogen storage potential of zirconium-based A2ZrH6 (A = Na, K) hydrides: a DFT and AIMD investigation
Muhammad Kaleem *a,
Amna Nasira,
Zahid Sarfrazb,
Sanober Kanwalc,
Asif Nawaz Khan
*a,
Amna Nasira,
Zahid Sarfrazb,
Sanober Kanwalc,
Asif Nawaz Khan d,
A. F. Abd El-Rehime and
Heba Y. Zahrane
d,
A. F. Abd El-Rehime and
Heba Y. Zahrane
aMaterial Research Laboratory (MRL), Department of Physics, International Islamic University H-10, Islamabad 44000, Pakistan. E-mail: mkaleemphy@gmail.com
bAdvanced Materials Processing Laboratory, Department of Physics, Air University, Pakistan
cResearch in Modeling and Simulation Group, COMSATS University Islamabad, Pakistan
dMaterials Modeling and Simulation Lab, Department of Physics, University of Science & Technology, Bannu 28100, Khyber Pakhtunkhwa, Pakistan
ePhysics Department, Faculty of Science, King Khalid University, P. O. Box 9004, Abha 61413, Saudi Arabia
First published on 6th March 2026
Abstract
Perovskite-based materials offer considerable potential for efficient, stable and environmentally sustainable hydrogen storage technologies. In this work an inclusive density functional theory (DFT) investigation was conducted to evaluate the structural, mechanical, optoelectronic and thermodynamic features of A2ZrH6 (A = Na, K) perovskite hydrides. Structural analysis reveals the stable cubic Fm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m symmetry, supported by favorable tolerance factors (0.92–0.99) and negative formation energies endorsing thermodynamic stability. Ab initio molecular dynamics (AIMD) simulations assure thermal stability at 300 K without significant structural distortion. Na2ZrH6 and K2ZrH6 exhibit notable hydrogen storage characteristics, achieving 4.22 and 3.45 wt% capacities with desorption temperatures of 441.39 and 258.91 K respectively. Mechanical analysis confirms elastic and Born stability with Poisson's ratios of 0.14 (Na2ZrH6) and 0.25 (K2ZrH6) and B/G ratios of 1.06 and 1.66 indicating brittle behavior. Electronic structure calculations confirm the band gaps of 1.25 and 1.87 eV while optical investigations indicate the suitability of these hydrides for photovoltaic applications. These results highlight A2ZrH6 (A = Na, K) perovskite hydrides as viable and efficient materials for next-generation hydrogen storage and energy conversion systems.
m symmetry, supported by favorable tolerance factors (0.92–0.99) and negative formation energies endorsing thermodynamic stability. Ab initio molecular dynamics (AIMD) simulations assure thermal stability at 300 K without significant structural distortion. Na2ZrH6 and K2ZrH6 exhibit notable hydrogen storage characteristics, achieving 4.22 and 3.45 wt% capacities with desorption temperatures of 441.39 and 258.91 K respectively. Mechanical analysis confirms elastic and Born stability with Poisson's ratios of 0.14 (Na2ZrH6) and 0.25 (K2ZrH6) and B/G ratios of 1.06 and 1.66 indicating brittle behavior. Electronic structure calculations confirm the band gaps of 1.25 and 1.87 eV while optical investigations indicate the suitability of these hydrides for photovoltaic applications. These results highlight A2ZrH6 (A = Na, K) perovskite hydrides as viable and efficient materials for next-generation hydrogen storage and energy conversion systems.
Introduction
Concerns about the enduring viability of fossil fuel based energy systems have grown as a result of the sharp increase in the world energy demand brought on by industrialization and population expansion.1–3 Elongated reliance on coal and oil and natural gas has a negative impact on the environment, contributing to climate change and deteriorating air quality.4 These difficulties have sped up the worldwide hunt for sustainable and clean energy sources that can provide future energy needs with little negative influence on the environment.5 Due to its high gravimetric energy density, toxic free nature and carbon free combustion products, hydrogen has become a particularly appealing alternative.6,7 Despite these benefits, the absence of safe, portable and effective hydrogen storage technology continues to be a major barrier to the widespread use of hydrogen energy. It is commonly acknowledged that resolving this issue is one of the most important obstacles to achieving a hydrogen-based energy economy.8,9Solid-state storage of complex metal hydrides has attracted a lot of attention due to its improved safety, high volumetric density and advantageous thermodynamic features as compared to gaseous and liquid hydrogen storage methods. Because of their highly adjustable physicochemical characteristics and structurally flexible frameworks, perovskite and double perovskite hydrides in this family have attracted increased attention.10,11 The perovskite design enables significant compositional engineering through cation substitution which enables systematic control over hydrogen concentration, lattice stability and electrical performance. Specifically organized octahedral networks made of double perovskite hydrides with the general formula A2BB′H6 are intriguing options for solid state H2 storage applications because they can tolerate high H2 concentrations without losing structural integrity.12
Various double perovskite hydrides have shown aptitude for H2 storage in recent theoretical and experimental studies. Research showed that XAlH6 (X = Ca, Sr, Ba)13 and Na2LiXH6 (X = Al, Sc, Ga)2 and K2LiAlH6 (ref. 6) demonstrate encouraging H2 storage capacities and structural stability. Other related systems such as AeSiH3 (Ae = Li, Na, K, Mg),14 XSrH3 (ref. 15) and A2BH6 type hydrides have also been examined, revealing diverse electronic, mechanical and thermodynamic behaviors. Recent first-principles studies have further highlighted the potential of double perovskite hydrides for hydrogen storage. For instance Bakar et al. investigated double hydrides Na2YH6 (Y = Ca, Ti) using density functional theory and reported gravimetric H2 storage capacities of 6.17 wt% for Na2CaH6 and 5.72 wt% for Na2TiH6.16 Their electronic structure analysis indicated metallic behavior in Na2CaH6 whereas Na2TiH6 exhibited semiconducting character with a band gap of 0.921 eV. Similarly Hakami et al. examined the H2 storage, mechanical and optoelectronic properties of A2FeH6 (A = Be, Mg) double perovskite hydrides reporting gravimetric hydrogen capacities of 7.50 wt% for Be2FeH6 and 5.43 wt% for Mg2FeH6 along with wide band gaps of 2.89 and 3.08 eV respectively.17 In another related study, Ahmed et al. explored X2FeH6 (X = Ca, Sr) hydrides and demonstrated thermodynamic stability and semiconducting behavior with indirect band gaps of 1.67 eV for Ca2FeH6 and 1.37 eV for Sr2FeH6 while reporting gravimetric H2 densities of 4.28 wt% and 2.54 wt% respectively.18
Despite these advances many reported perovskite hydrides continue to face intrinsic limitations including elevated hydrogen desorption temperatures, sluggish sorption kinetics, limited gravimetric capacity or structural instability under operating conditions.19 In several instances improvements in H2 capacity have been achieved at the expense of mechanical or dynamical stability underscoring the persistent challenge of balancing hydrogen density with material robustness.20,21 Motivated by these considerations zirconium based perovskite hydrides represent an attractive yet relatively underexplored class of materials. Zirconium as a tetravalent transition metal provides favorable charge balance and strong metal–hydrogen interactions that can stabilize corner-sharing [ZrH6] octahedra within a perovskite lattice. Alkali metal substitution at the A-site offers an additional degree of freedom to tune lattice dimensions, electronic structure and hydrogen binding strength. In particular the A2ZrH6 (A = Na, K) family is of significant interest as replacing Na+ with the larger K+ cation is expected to systematically influence structural parameters, thermodynamic stability and hydrogen storage performance. Nevertheless a comprehensive investigation of the structural, mechanical, AIMD stability, H2 storage, optoelectronic and thermodynamic properties of Na2ZrH6 and K2ZrH6 remains absent from the literature.
In this work a detailed first-principles investigation of A2ZrH6 (A = Na, K) perovskite hydrides is performed using density functional theory. The study systematically examines their crystal structure, AIMD stability, elastic and mechanical behavior, electronic band structure and density of states, optical, thermodynamic characteristics and H2 storage performance to establish clear composition-property relationships. By elucidating how alkali metal substitution impacts lattice stability, electrical responsiveness and hydrogen binding behavior this work identifies crucial factors regulating H2 absorption and release in zirconium based perovskite hydrides. The results provide new theoretical insights into this mostly unexplored material class as well as a rational foundation for the design and experimental development of superior solid state H2 storage materials. Future studies will also explore the influence of other alkali metals on these material properties, further broadening the scope of perovskite hydride research.
Computational methodology
The structural and physical characteristics of the A2ZrH6 (A = Na, K) perovskite hydrides were investigated using first principles calculations carried out within the context of DFT. The CASTEP code which uses a plane wave pseudopotential method to solve the KohnSham equations was used to carry out the simulations. Vanderbilt ultrasoft pseudopotentials were used to represent electron and ion interactions allowing for an accurate and effective handling of valence electrons. The generalized gradient approximation (GGA) was used to tackle the exchange correlation effects in the Perdew–Burke–Ernzerhof (PBE) formulation.22–24 The valence electronic configurations explicitly considered in the calculations were Na (2s2 2p6 3s1), K (3s2 3p6 4s1), Zr (4s2 4p6 4d2 5s2) and H (1s1). To guarantee consistent convergence of total energies and stress tensors a plane wave kinetic energy cutoff of 600 eV was used. A Monkhorst–Pack k-point mesh of 6 × 6 × 6 was used for Brillouin zone integrations and it was confirmed to yield converged structural and electrical properties.25 The Broyden–Fletcher–Goldfarb–Shanno (BFGS) minimization approach was used for structural optimizations enabling the simultaneous relaxation of internal atomic coordinates and lattice parameters. The convergence criteria for geometry optimization were set to 2.0 × 10−5 eV per atom for total energy 0.05 eV Å−1 for maximum force and 2 × 10−3 Å for maximum atomic displacement and 0.01 GPa for residual stress.26 All calculations were conducted under zero-temperature and zero-pressure conditions to determine the ground-state configurations of the investigated hydrides.To assess thermal stability beyond static calculations ab initio molecular dynamics (AIMD) simulations were performed using the pw.x module of Quantum ESPRESSO. The simulations were conducted within the canonical NVT ensemble with temperature control achieved through a Berendsen thermostat set at 300 K. Atomic trajectories were propagated using the velocity-Verlet algorithm with a time step of 0.97 fs over 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 steps corresponding to a total simulation time of approximately 9.7 ps. Ultrasoft pseudopotentials consistent with the PBE exchange-correlation functional were employed with plane-wave and charge-density cutoffs of 60 Ry and 400 Ry respectively. The self-consistent field convergence threshold was set to 1.0 × 10−8 Ry while total energy and force convergence criteria were fixed at 1.0 × 10−5 Ry and 1.0 × 10−4 Ry/Bohr respectively.27
000 steps corresponding to a total simulation time of approximately 9.7 ps. Ultrasoft pseudopotentials consistent with the PBE exchange-correlation functional were employed with plane-wave and charge-density cutoffs of 60 Ry and 400 Ry respectively. The self-consistent field convergence threshold was set to 1.0 × 10−8 Ry while total energy and force convergence criteria were fixed at 1.0 × 10−5 Ry and 1.0 × 10−4 Ry/Bohr respectively.27
Results and discussion
Structural properties
The structural characteristics and geometric stability of the A2ZrH6 (A = Na, K) perovskite hydrides were systematically investigated using first-principles DFT calculations. Both compounds crystallize in a highly symmetric cubic structure with space group Fm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m (No. 225) preserving the ideal perovskite framework.28 The atomic arrangements within the cubic unit cell follow distinct Wyckoff positions in which the A-site cations (Na or K) occupy the 8c sites (0.25, 0.25, 0.25) while Zr atoms are positioned at the 4a sites (0, 0, 0) forming the backbone of the perovskite lattice. The hydrogen atoms be vested at the 24e sites (x, 0, 0) where the internal parameter x = 0.245 resulting in marginally distorted ZrH6 octahedra as displayed in Fig. 1. This octahedral coordination directly affects hydrogen binding and is essential for maintaining the crystal structure. The optimized lattice constants were found to be 8.31 Å for Na2ZrH6 and 8.95 Å for K2ZrH6 with lattice parameters satisfying a = b = c as summarized in Table 1. The observed lattice expansion upon substitution of Na with the larger K cation is further reflected in the computed unit cell volumes which increase from 573.87 Å3 for Na2ZrH6 to 718.86 Å3 for K2ZrH6 consistent with the ionic size difference between Na+ and K+.
m (No. 225) preserving the ideal perovskite framework.28 The atomic arrangements within the cubic unit cell follow distinct Wyckoff positions in which the A-site cations (Na or K) occupy the 8c sites (0.25, 0.25, 0.25) while Zr atoms are positioned at the 4a sites (0, 0, 0) forming the backbone of the perovskite lattice. The hydrogen atoms be vested at the 24e sites (x, 0, 0) where the internal parameter x = 0.245 resulting in marginally distorted ZrH6 octahedra as displayed in Fig. 1. This octahedral coordination directly affects hydrogen binding and is essential for maintaining the crystal structure. The optimized lattice constants were found to be 8.31 Å for Na2ZrH6 and 8.95 Å for K2ZrH6 with lattice parameters satisfying a = b = c as summarized in Table 1. The observed lattice expansion upon substitution of Na with the larger K cation is further reflected in the computed unit cell volumes which increase from 573.87 Å3 for Na2ZrH6 to 718.86 Å3 for K2ZrH6 consistent with the ionic size difference between Na+ and K+.
| Parameters | Na2ZrH6 | K2ZrH6 | Sr2FeH6 (ref. 18) | Ca2FeH6 (ref. 18) | Sr2VH6 (ref. 30) |
|---|---|---|---|---|---|
| Lattice constant a = b = c (Å) | 8.31 | 8.95 | 5.30 | 4.87 | 7.12 |
| Volume (Å)3 | 573.87 | 718.86 | 148.88 | 115.50 | 59.56 |
| τG | 0.92 | 0.99 | — | — | 0.99 |
| µ | 0.47 | 0.47 | — | — | 0.47 |
| ΔHf | −1.33 | −0.78 | −4.23 | −6.93 | −0.284 |
| ΔHf(kJ mol−1 H2) | −57.69 | −33.84 | — | — | −33.84 |
| Cwt% | 4.22 | 3.45 | 2.54 | 4.28 | 2.60 |
| Tdes (K) | 441.39 | 258.91 | — | — | 210.7 |
To further assess geometric stability, the Goldschmidt tolerance factor (τG) and octahedral factor (µ) were evaluated using the eqn (1) and (2):29
 | (1) |
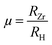 | (2) |
Moreover the thermodynamic stability of the investigated compounds was further examined through the calculation of the formation energy (ΔHf) given by eqn (3):31
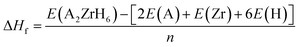 | (3) |
Hydrogen storage
Hydrogen is largely considered as a vital energy vector for achieving a sustainable and low carbon energy future owing to its high gravimetric energy density and environmentally benign utilization. However a significant obstacle to its widespread use is still the absence of safe and portable and effective H2 storage systems. In this sense solid state H2 storage based on complex metal hydrides has garnered a lot of interest since it provides better safety and volumetric efficiency when compared to traditional gaseous and liquid storage techniques. Perovskite derived hydrides particularly A2ZrH6 (A = Na, K) systems represent a promising class of materials due to their structural robustness and intrinsically high hydrogen content. The gravimetric hydrogen capacity which measures the mass fraction of hydrogen in relation to the total mass of the storage medium is a crucial metric for assessing the efficacy of hydrogen storage. The eqn (4) was used to get the theoretical gravimetric hydrogen capacity:32
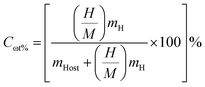 | (4) |
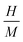 denotes the hydrogen to metal ratio. Using this formulation the gravimetric hydrogen capacities of Na2ZrH6 and K2ZrH6 were determined to be 4.22 wt% and 3.45 wt% respectively as listed in Table 1. The higher capacity of Na2ZrH6 originates from the lower atomic mass of sodium which enhances the hydrogen-to-host mass ratio. These values are competitive among complex hydride systems and show a clear possibility for more optimization through compositional adjustment even if they are still below the U.S. DOE 2025 gravimetric target of 5.5 wt%. In addition to storage capacity the hydrogen release behaviour plays a crucial role in practical applications and is commonly assessed through the desorption temperature (Tdes). Desorption temperature can be estimated from thermodynamic considerations based on the Gibbs free energy eqn (5): 33,34
denotes the hydrogen to metal ratio. Using this formulation the gravimetric hydrogen capacities of Na2ZrH6 and K2ZrH6 were determined to be 4.22 wt% and 3.45 wt% respectively as listed in Table 1. The higher capacity of Na2ZrH6 originates from the lower atomic mass of sodium which enhances the hydrogen-to-host mass ratio. These values are competitive among complex hydride systems and show a clear possibility for more optimization through compositional adjustment even if they are still below the U.S. DOE 2025 gravimetric target of 5.5 wt%. In addition to storage capacity the hydrogen release behaviour plays a crucial role in practical applications and is commonly assessed through the desorption temperature (Tdes). Desorption temperature can be estimated from thermodynamic considerations based on the Gibbs free energy eqn (5): 33,34
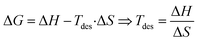 | (5) |
Ab Initio molecular dynamics (AIMD) calculations
AIMD simulations conducted at room temperature were used to investigate the dynamical and thermal stability of the A2ZrH6 (A = Na, K) perovskite hydrides as illustrated in Fig. 2(a and b). AIMD simulations provide a realistic assessment of finite temperature behavior by merging first principles electronic structure computations with classical atomic motion and directly evaluating total energy development and thermodynamic stability under thermal agitation.36 The total energy profile for Na2ZrH6 is well controlled over the 9.7 ps simulation frame with extremely small amplitude fluctuations around an equilibrium value as seen in Fig. 2(a). Importantly no systematic drift or sudden fluctuation is seen indicating that there is no structural instability during heat stimulation. The temperature evolution continuously varies about the target value of 300 K while remaining within a narrow physically attainable range. This behavior shows that under ambient conditions the Na2ZrH6 framework does not undergo phase transformation or breakdown and retains its structural integrity. As seen in Fig. 2(b) K2ZrH6 reacts similarly. A dynamically stable lattice is suggested by the total energy small fluctuations and lack of abrupt discontinuities throughout the trial. With brief fluctuations typical of constant temperature AIMD simulations the associated temperature swings stay centered around room temperature. The mechanical system of K2ZrH6 and thermal endurance are further supported by the lack of abnormal pressure or temperature variations. These findings which consistently show limited energy routes and steady temperature profiles offer compelling evidence of the thermal and dynamical stability of both Na2ZrH6 and K2ZrH6. These perovskite hydrides resilience under practical operating settings is demonstrated by the AIMD simulations lack of structural degradation which further supports their applicability for H2 storage and energy related applications.Mechanical properties
Mechanical characteristics exhibit a decisive role in prevailing the elastic response and structural reliability of perovskite hydrides mainly under the cyclic stresses associated with H2 absorption and release.37 A quantitative evaluation of elastic constants is therefore essential to assess mechanical stability and deformation behavior.38 In this work the second-order elastic constants (Cij) of A2ZrH6 (A = Na, K) perovskite hydrides were calculated from which key mechanical parameters were derived. The calculated elastic constants and related mechanical properties are summarized in Table 2. Due to the cubic crystal symmetry only three independent elastic constants C11, C12 and C44 are required to describe the elastic behavior of the studied compounds.39 These constants represent resistance to uniaxial deformation, transverse deformation and shear deformation respectively. The calculated values for both Na2ZrH6 and K2ZrH6 satisfy the Born-Huang mechanical stability criteria for cubic crystals: C11 − C12 > 0, C11 + 2C12 > 0, C11 > 0 and C44 > 0 confirming that both compounds are mechanically stable under small strains. As shown in Table 2 Na2ZrH6 exhibits relatively lower elastic stiffness with C11 = 19.48 GPa, C12 = 4.41 GPa and C44 = 9.96 GPa whereas K2ZrH6 displays higher longitudinal rigidity with C11 = 54.75 GPa indicating stronger resistance to uniaxial deformation. The larger C11 value of K2ZrH6 reflects enhanced stiffness along principal crystallographic directions consistent with its expanded lattice framework.| Elastic moduli | Na2ZrH6 | K2ZrH6 | Sr2FeH6 (ref. 18) | Ca2FeH6 (ref. 18) | Sr2VH6 (ref. 30) |
|---|---|---|---|---|---|
| C11 | 19.48 | 54.75 | 95.38 | 117.19 | 67.75 |
| C12 | 4.41 | 6.69 | 17.32 | 26.99 | 12.32 |
| C44 | 9.96 | 9.11 | 30.14 | 38.78 | 21.56 |
| Born's stability | Stable | Stable | Stable | Stable | Stable |
| B (GPa) | 9.43 | 22.71 | 43.34 | 57.06 | 30.79 |
| G (GPa) | 8.90 | 13.60 | 33.42 | 41.19 | 23.84 |
| E (GPa) | 20.32 | 34.02 | 79.75 | 99.60 | 56.86 |
| B/G | 1.06 | 1.66 | 1.29 | 1.38 | 1.23 |
| ν | 0.14 | 0.25 | — | — | 0.19 |
| AU | 0.09 | 1.22 | 0.77 | 0.86 | 0.778 |
| Cp | −5.55 | −2.42 | — | — | −9.24 |
The bulk modulus (B) and shear modulus (G) were calculated using the Voigt–Reuss–Hill (VRH) approximation in order to assess macroscopic elastic behavior.40 The shear modulus shows resistance to shape deformation whereas the bulk modulus shows resistance to homogeneous compression. The calculated B values are 9.43 GPa for Na2ZrH6 and 22.71 GPa for K2ZrH6 indicating that K2ZrH6 possesses a higher resistance to volume compression. In a similar vein the shear modulus rises from 8.90 GPa for Na2ZrH6 to 13.60 GPa for K2ZrH6 indicating that the K based combination is more stiff. The Young's modulus (E) which measures stiffness under uniaxial stress and has values of 20.32 GPa for Na2ZrH6 and 34.02 GPa for K2ZrH6 further supports this tendency. These findings show that Na2ZrH6 is relatively more compliant than K2ZrH6 which may be helpful for adapting to volume variations during H2 absorption and desorption. However, the comparatively low elastic moduli, particularly for Na2ZrH6, also indicate mechanical softness, meaning that the material can deform more readily under external or internally generated cycling stresses.
Additionally the ductile or brittle nature of the materials was assessed using Pugh ratio (B/G) and Poisson's ratio (ν). According to criteria materials with B/G > 1.75 and ν > 0.26 exhibit ductile nature.41 The computed B/G ratios of 1.06 (Na2ZrH6) and 1.66 (K2ZrH6) along with Poisson's ratios of 0.14 and 0.25 respectively indicate that both compounds exhibit a predominantly brittle mechanical character with K2ZrH6 lying close to the brittle or ductile transition. Elastic anisotropy was quantified using the universal anisotropy index (AU). The low value of AU = 0.09 for Na2ZrH6 suggests near isotropic elastic behavior whereas the higher value of AU = 1.22 for K2ZrH6 indicates pronounced elastic anisotropy and direction dependent deformation characteristics. Additional insight into bonding nature was obtained from the Cauchy pressure (Cp = C12 − C44) which yields negative values of −5.55 GPa for Na2ZrH6 and −2.42 GPa for K2ZrH6. The unfavorable brittle mechanical behavior in hydride systems is frequently linked to a directed and covalent bonding character, as suggested by Cauchy pressures. From a practical perspective, the combination of low stiffness and brittleness presents both advantages and challenges for hydrogen cycling. While the low moduli can help absorb volumetric strain during repeated H2 absorption and desorption, reducing stress buildup, the brittle nature may lead to crack formation, particle fragmentation, and loss of structural integrity over time, especially under uneven stresses or microstructural defects. Thus, although the current elastic analysis confirms stability under small strains, a full assessment of cycling durability would require further studies focused on fracture behavior and microstructural impacts, which are beyond the scope of this work. Overall, the calculated elastic parameters indicate that A2ZrH6 (A = Na, K) perovskite hydrides are mechanically stable under small strains, exhibit moderate stiffness, and predominantly brittle behavior, which should be considered when interpreting their ability to withstand structural stresses during repeated hydrogen absorption and release cycles, as shown in the elastic constants and derived mechanical parameters in Table 2.
Electronic properties
Electronic characteristics play a central role in governing hydrogen adsorption–desorption kinetics, charge redistribution and chemical stability in complex hydrides. In perovskite hydrides the magnitude and nature of the electronic band gap together with the distribution of electronic states near the Fermi level directly influence hydrogen binding strength and diffusion pathways.42,43 To elucidate these effects the electronic band structures and DOS of A2ZrH6 (A = Na, K) were systematically analyzed using first principles calculations. The calculated electronic band structures of Na2ZrH6 and K2ZrH6 along the high symmetry directions X–R–M–Γ–R are presented in Fig. 3(a and b) with the Fermi level (EF) aligned at 0 eV. A substantial separation between the valence and conduction bands characterizes these materials semiconducting nature. Na2ZrH6 has an indirect band gap of about 1.25 eV because the valence band maximum and conduction band minimum occur at distinct symmetry sites. This restrained band gap validates that there is enough electronic insulation to prevent undesired electronic leakage while permitting charge redistribution during H2 absorption and release. K2ZrH6 on the other hand exhibits a broader indirect band gap of roughly 1.87 eV which is a result of the greater K+ ionic radius expanding the lattice and decreasing orbital overlap inside the Zr–H framework. During repeated hydrogen cycling, the increasing band gap suggests improved electronic stability which may be advantageous for preserving structural integrity.44 The fact that both hydrides are semiconducting implies that electron transport is regulated rather than metallic. Improved reversibility and fewer parasitic processes in H2 storage materials are often associated with this feature.The total density of states (TDOS) displayed in Fig. 4(a and b) provides further information on the electrical response. The TDOS shows a significant depletion of states at the Fermi level in both Na2ZrH6 and K2ZrH6 supporting the band structure findings and verifying their semiconducting nature. The lack of finite DOS at EF suggests electronic passivation which reduces electronic contributions to structural degradation under operating conditions and aids in stabilizing the H2 rich lattice. The TDOS peaks pronounced asymmetry near the Fermi level indicates that the effective masses of electrons and holes are different. Reaction kinetics may be impacted by this asymmetry potential to affect charge redistribution during hydrogen uptake and release.
The partial density of states (PDOS) for Na2ZrH6 and K2ZrH6 displayed in Fig. 4(c and d) reveals the orbital origins of the electronic bands. In both compounds the valence band region below EF is dominated by H-s states with appreciable hybridization from Zr-d and alkali metal s/p orbitals. This hybridization reflects mixed ionic-covalent bonding within the ZrH6 octahedra which is essential for reversible hydrogen binding. Above the Fermi level the conduction bands are primarily governed by Zr-4d states with secondary contributions from Na-3p or K-4p orbitals. The stronger Zr-d character in the conduction manifold indicates that electronic excitation and charge transfer during hydrogen release are mainly mediated through the Zr–H network. Compared to Na2ZrH6, K2ZrH6 shows a slightly reduced overlap between Zr-d and H-s states near the band edges consistent with its larger band gap and expanded lattice.
Both Na2ZrH6 and K2ZrH6 are indirect gap semiconductors with band gaps falling in a region that combines electrical stability and bonding flexibility according to the combined band structure and DOS investigations. The ZrH6 octahedra play a crucial role in regulating H2 retention and release as evidenced by the preponderance of H(s) states in the valence band and Zr(d) states in the conduction band. While retaining enough electronic flexibility to enable reversible sorption processes these electronic characteristics promote stable H2 accommodation.
Optical properties
The optical response provides direct insight into photon matter interaction in semiconducting hydride perovskites and is closely linked to interband transitions across the band gap. In H2 storage hydrides such photoexcited carrier generation and associated dielectric screening might promote charge redistribution and localized heating supporting surface reaction steps and influencing absorption desorption rates.45 This connection between optical properties and hydrogen storage/release performance is important, as the photo-induced carrier excitation can enhance charge redistribution, which supports the surface reaction steps crucial for hydrogen release.To clarify these effects the frequency dependent optical properties of A2ZrH6 (A = Na, K) were evaluated. The computed dielectric function and the derived optical spectra are presented in Fig. 5(a–f). The optical response is designated by the complex dielectric function ε(ω) = ε1(ω) + ε2(ω) where ε1(ω) reflects dispersion (polarization) and ε2(ω) represents absorption due to interband transitions.46 As shown in Fig. 5(a) both compounds exhibit finite static dielectric constants indicating measurable low-energy electronic polarizability. Na2ZrH6 displays a higher static dielectric constant of approximately ε1(0) 4.6 whereas K2ZrH6 shows a lower value of about ε1(0) 3.3 reflecting stronger dielectric screening and enhanced polarization in the Na-based hydride. With increasing photon energy ε1(ω) decreases gradually and exhibits pronounced dispersion features before crossing zero in the 3.5–4.0 eV range for Na2ZrH6 and slightly above 4.2 eV for K2ZrH6 indicating the effective plasma frequency and the transition from reflective to transparent behavior. At higher energies ε1(ω) attains weakly negative values followed by stabilization, a response characteristic of semiconductors governed by interband transitions rather than free-carrier (Drude) contributions.
The imaginary part ε2(ω) also shown in Fig. 5(a) exhibits an earlier onset for Na2ZrH6 at 1.3 eV consistent with its smaller electronic band gap (Eg = 1.25 eV) while K2ZrH6 shows a delayed onset near 1.9 eV in agreement with its wider gap (Eg = 1.87 eV). For Na2ZrH6 ε2(ω) displays prominent absorption peaks centered around 2.6 eV and 5.8 eV with a maximum intensity approaching ε2 of 5.0 whereas K2ZrH6 exhibits comparatively weaker peaks near 3.0 eV and 6.2 eV reaching a maximum of ε2 4.0. These dominant spectral features originate from interband transitions between H-derived valence states and Zr-d dominated conduction states as corroborated by the DOS analysis.47 The stronger ε2(ω) response of Na2ZrH6 indicates higher transition probability and enhanced photon-induced carrier excitation which can facilitate charge redistribution and support hydrogen desorption processes under optical or thermal stimulation. This enhanced carrier excitation directly influences hydrogen release, with Na2ZrH6 showing a stronger capability to drive the desorption process through photon stimulation. The absorption coefficient is obtained from the extinction coefficient k(ω) through equation 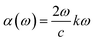 .48 In Fig. 5(b) both materials show negligible absorption at very low energies followed by a clear rise after the band-edge region. Na2ZrH6 exhibits the stronger absorption across the spectrum, including a low-energy feature around 2–3 eV (peak α ∼ 3.0 × 104 cm−1) and a dominant high-energy band in the near-UV where the absorption reaches α ≈ 1.7 × 105 cm−1 (around 7–8 eV). In parallel, K2ZrH6 peaks at α ≈ 1.2–1.3 × 105 cm−1 in the same region. The stronger α(ω) for Na2ZrH6 indicates more intense photon–electron coupling which can support photo-assisted carrier excitation and localized thermal effects relevant to hydrogen release. This stronger coupling can directly facilitate more efficient photon-driven hydrogen desorption.
.48 In Fig. 5(b) both materials show negligible absorption at very low energies followed by a clear rise after the band-edge region. Na2ZrH6 exhibits the stronger absorption across the spectrum, including a low-energy feature around 2–3 eV (peak α ∼ 3.0 × 104 cm−1) and a dominant high-energy band in the near-UV where the absorption reaches α ≈ 1.7 × 105 cm−1 (around 7–8 eV). In parallel, K2ZrH6 peaks at α ≈ 1.2–1.3 × 105 cm−1 in the same region. The stronger α(ω) for Na2ZrH6 indicates more intense photon–electron coupling which can support photo-assisted carrier excitation and localized thermal effects relevant to hydrogen release. This stronger coupling can directly facilitate more efficient photon-driven hydrogen desorption.
Optical conductivity σ(ω) describes the frequency-dependent response of charge carriers under an external electromagnetic field and directly reflects photon-induced electronic transitions that govern photo assisted charge transport and hydrogen desorption dynamics. The optical conductivity can be expressed as  .49 In Fig. 5(c) both compounds show conductivity features that track the absorption bands. Na2ZrH6 reaches a higher peak optical conductivity (4.0 in the plotted units) near 6–7 eV whereas K2ZrH6 peaks at a lower magnitude (2.8) in the same region confirming stronger photoinduced carrier activity in the Na-based hydride.
.49 In Fig. 5(c) both compounds show conductivity features that track the absorption bands. Na2ZrH6 reaches a higher peak optical conductivity (4.0 in the plotted units) near 6–7 eV whereas K2ZrH6 peaks at a lower magnitude (2.8) in the same region confirming stronger photoinduced carrier activity in the Na-based hydride.
The refractive index and extinction coefficient provide further insight into light propagation and attenuation inside the material and are particularly relevant for assessing light-matter interaction strength in semiconducting hydrides. As evident from Fig. 5(d) both Na2ZrH6 and K2ZrH6 exhibit relatively high refractive indices in the low-energy region, consistent with their semiconducting nature and finite band gaps. Na2ZrH6 shows a higher static refractive index, with n(0) ≈ 2.1–2.3 compared to n(0) 1.7–1.9 for K2ZrH6 indicating stronger electronic polarization and enhanced photon coupling at low photon energies. The refractive index n(ω) and extinction coefficient k(ω) are consequent from the real and imaginary parts of the dielectric function using eqn (6).50
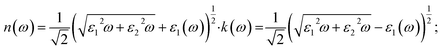 | (6) |
As shown in Fig. 5(d) the refractive index of both compounds gradually decreases with increasing photon energy and approaches unity beyond 7 eV reflecting reduced light-matter interaction in the high energy ultraviolet region. The consistently larger n(ω) values for Na2ZrH6 across the visible range indicate stronger dispersion and improved optical confinement which can assist photon-assisted charge excitation relevant for hydrogen release. Significant absorption features in the visible range are revealed by the extinction coefficient spectra which are also shown in Fig. 5(d). While K2ZrH6 displays a relatively weaker peak with kmax 0.5 and Na2ZrH6 displays a prominent extinction peak about 2–3 eV with kmax 1.0. Both materials exhibit wider extinction bands that extend into the UV region at higher energies which are linked to interband electronic transitions involving Zr-d and H-s states. Stronger electromagnetic wave attenuation and improved absorption efficiency which might encourage localized heating and speed up hydrogen desorption processes are confirmed by the greater extinction coefficient of Na2ZrH6.
Reflectivity R(ω) quantifies the fraction of incident electromagnetic radiation reflected from the material surface and provides insight into surface optical response absorption efficiency and photon matter coupling which are relevant for light assisted hydrogen absorption and desorption processes can be calculated using eqn (7).51
 | (7) |
As shown in Fig. 5(e) both materials exhibit low-to-moderate reflectance with a pronounced minimum around 3 eV (down to 0.03–0.05) coincident with strong absorption. At higher energies Na2ZrH6 attains a larger reflectivity maximum of 0.28 near 7–8 eV while K2ZrH6 peaks at 0.20 in a similar range indicating a comparatively stronger reflective response of the Na compound in the near-UV. The energy-loss function provides insight into collective electronic excitations and plasmonic behavior describing the energy dissipated by fast electrons traversing the material and serving as a sensitive indicator of dielectric screening and electronic stability and calculated by eqn (8).52
 | (8) |
In Fig. 5(f) both compounds show a clear low-energy loss peak near ≈2–3 eV (Na2ZrH6: Lmax 0.40 and K2ZrH6: Lmax 0.25) followed by a gradual increase at higher energies. Toward 10 eV K2ZrH6 rises more strongly (approaching 0.85) than Na2ZrH6 (0.65) suggesting comparatively higher high energy loss in the K-based hydride.
Overall both A2ZrH6 (A = Na, K) compounds exhibit semiconducting optical responses with absorption onsets governed by their band gaps and dominant interband activity in the visible-to-UV range. Across Fig. 5(a–f) Na2ZrH6 consistently shows stronger dielectric screening, higher absorption and larger optical conductivity indicating more efficient photon-driven electronic excitation which can be beneficial for photo-assisted processes and thermally supported hydrogen cycling.
Thermodynamic properties
Thermodynamic properties play a central role in determining the feasibility and kinetic performance of hydrogen storage materials. Parameters such as sound velocities, Debye temperature (θD) and melting temperature (Tm) provide indirect yet reliable insight into lattice stiffness, bond strength, phonon transport and thermal robustness. In H2 storage systems these features directly influence hydrogen diffusion kinetics and absorption desorption reversibility and resistance to structural degradation during repeated thermal cycling. In this context elastic constants serve as a fundamental basis for evaluating the thermodynamic stability of A2ZrH6 (A = Na, K) perovskite hydrides. The longitudinal (vl) and transverse (vt) sound velocities were derived from the bulk modulus (B) or shear modulus (G) and were used to calculate the average sound velocity (vm). The Debye temperature and melting temperature were subsequently estimated using definite eqn (9)–(11):53,54
 | (9) |
 | (10) |
| Tm = [553 + 5.911(C11)] ± 300 | (11) |
| Compounds | vl (km s−1) | vt (km s−1) | vm (km s−1) | θD (K) | Tm (K) |
|---|---|---|---|---|---|
| Na2ZrH6 | 3.04 | 2.32 | 2.48 | 293 | 668 |
| K2ZrH6 | 4.43 | 2.90 | 3.18 | 348 | 877 |
Conclusion
This work presents a comprehensive first principles investigation of A2ZrH6 (A = Na, K) double perovskite hydrides employing DFT calculations within the CASTEP framework to evaluate their potential for H2 storage and clean energy applications. The structural stability of both compounds is confirmed by their cubic Fm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m symmetry crystallization and their thermodynamic stability is further supported by negative formation enthalpies. By proving that both compounds retain structural stability at 300 K AIMD provides more evidence for thermal stability. According to mechanical analysis both compounds have a mechanical character that is primarily brittle and satisfy the Born stability requirements. The optical response shows high static dielectric constants and significant absorption in the ultraviolet area while the electronic study shows semiconducting band gaps of 1.25 and 1.87 eV for Na2ZrH6 and K2ZrH6 respectively. The calculated gravimetric H2 storage capacities of 4.22 wt% for Na2ZrH6 and 3.45 wt% for K2ZrH6 approach the U.S. DOE targets while their corresponding desorption temperatures of 441.39 K and 258.91 K respectively underscore their promising potential for next-generation solid state H2 storage technologies. These results suggest that A2ZrH6 hydrides offer a promising combination of high H2 storage capacity, thermodynamic viability and structural integrity making them great candidates for future H2 storage and energy delivery systems.
m symmetry crystallization and their thermodynamic stability is further supported by negative formation enthalpies. By proving that both compounds retain structural stability at 300 K AIMD provides more evidence for thermal stability. According to mechanical analysis both compounds have a mechanical character that is primarily brittle and satisfy the Born stability requirements. The optical response shows high static dielectric constants and significant absorption in the ultraviolet area while the electronic study shows semiconducting band gaps of 1.25 and 1.87 eV for Na2ZrH6 and K2ZrH6 respectively. The calculated gravimetric H2 storage capacities of 4.22 wt% for Na2ZrH6 and 3.45 wt% for K2ZrH6 approach the U.S. DOE targets while their corresponding desorption temperatures of 441.39 K and 258.91 K respectively underscore their promising potential for next-generation solid state H2 storage technologies. These results suggest that A2ZrH6 hydrides offer a promising combination of high H2 storage capacity, thermodynamic viability and structural integrity making them great candidates for future H2 storage and energy delivery systems.
Conflicts of interest
All authors declare that they have no conflicts of interest.Data availability
Data relevant to this study is available upon request.Acknowledgements
The authors extend their appreciation to the Deanship of Research and Graduate Studies at King Khalid University for funding this work through Large Research Project under grant number RGP2/140/46.References
- A. Züttel, Hydrogen storage methods, Naturwissenschaften, 2004, 91(4), 157–172 CrossRef PubMed.
- A. Ayyaz, Q. Mahmood, N. D. Alkhaldi, G. Murtaza, A. Usman, M. A. Ullah and H. Albalawi, Exploring hydrogen storage potential, thermodynamic, and optoelectronic characteristics of novel double perovskite hydrides Na2LiXH6 (X= Al, Sc, and Ga): DFT analysis, J. Energy Storage, 2025, 122, 116650 CrossRef CAS.
- A. Gencer, G. Surucu and S. Al, MgTiO3Hx and CaTiO3Hx perovskite compounds for hydrogen storage applications, Int. J. Hydrogen Energy, 2019, 44(23), 11930–11938 CrossRef CAS.
- C. Zou, Q. Zhao, G. Zhang and B. Xiong, Energy revolution: From a fossil energy era to a new energy era, Nat. Gas Ind. B, 2016, 3(1), 1–11 Search PubMed.
- M. A. Ullah, M. Kaleem, A. Nasir, Z. Sarfraz, M. M. A. Iqbal, M. Rizwan, K. N. Riaz and M. Tanzeel, An approach towards next-generation hydrogen storage: a DFT study on A 2 LiTiH 6 (A= K, Ca) perovskite hydrides, RSC Adv., 2025, 15(46), 38714–38728 RSC.
- M. Baaddi, R. Chami, O. Baalla, S. E. Quaoubi, A. Saadi, L. E. H. Omari and M. Chafi, The effect of strain on hydrogen storage characteristics in K 2 NaAlH 6 double perovskite hydride through first principle method, Environ. Sci. Pollut. Res., 2024, 31(53), 62056–62064 CrossRef CAS PubMed.
- M. Tahir, M. Usman, J. U. Rehman and M. B. Tahir, A first-principles study to investigate the physical properties of Sn-based hydride perovskites XSnH3 (X= K, Li) for hydrogen storage application, Int. J. Hydrogen Energy, 2024, 50, 845–853 CrossRef CAS.
- S. Niaz, T. Manzoor and A. H. Pandith, Hydrogen storage: Materials, methods and perspectives, Renew. Sustain. Energy Rev., 2015, 50, 457–469 CrossRef CAS.
- B. Chen, L. J. Conway, W. Sun, X. Kuang, C. Lu and A. Hermann, Phase stability and superconductivity of lead hydrides at high pressure, Phys. Rev. B, 2021, 103(3), 035131 Search PubMed.
- G. Surucu, A. Gencer, A. Candan, H. H. Gullu and M. Isik, CaXH3 (X= Mn, Fe, Co) perovskite-type hydrides for hydrogen storage applications, Int. J. Energy Res., 2020, 44(3), 2345–2354 Search PubMed.
- R. Song, Y. Chen, S. Chen, N. Xu and W. Zhang, First-principles to explore the hydrogen storage properties of XPtH3 (X= Li, Na, K, Rb) perovskite type hydrides, Int. J. Hydrogen Energy, 2024, 57, 949–957 Search PubMed.
- A. Ayyaz, M. A. Ullah, M. Zaman, N. D. Alkhaldi, Q. Mahmood, I. Boukhris, M. Al-Buriahi and M. mana Al-Anazy, Investigation of hydrogen storage and energy harvesting potential of double perovskite hydrides A2LiCuH6 (A= Be/Mg/Ca/Sr): A DFT approach, Int. J. Hydrogen Energy, 2025, 102, 1329–1339 Search PubMed.
- M. Umer, G. Murtaza, N. Ahmad, A. Ayyaz, H. H. Raza, A. Usman, A. Liaqat and S. Manoharadas, First principles investigation of structural, mechanical, thermodynamic, and electronic properties of Al-based perovskites XAlH3 (X= K, Rb, Cs) for hydrogen storage, Int. J. Hydrogen Energy, 2024, 61, 820–830 Search PubMed.
- A. Mera and M. A. Rehman, First-principles investigation for the hydrogen storage properties of AeSiH3 (Ae= Li, K, Na, Mg) perovskite-type hydrides, Int. J. Hydrogen Energy, 2024, 50, 1435–1447 CrossRef CAS.
- H. H. Raza, G. Murtaza, N. Muhammad and S. M. Ramay, First-principle investigation of XSrH3 (X= K and Rb) perovskite-type hydrides for hydrogen storage, Int. J. Quantum Chem., 2020, 120(24), e26419 CrossRef CAS.
- A. Bakar, M. Ahmed, Q. Xia, J. Yang, A. Afaq and Y. Yang, Conceptual design and stand-alone multiscale simulation for hydrogen storage energy: A rigorous way forward for double hydride perovskites Na2YH6 (Y= Ca, Ti), Int. J. Hydrogen Energy, 2026, 197, 152679 CrossRef CAS.
- O. Hakami and H. J. Alathlawi, Study of mechanical, optoelectronic, and thermoelectric characteristics of Be/Mg ions Based double perovskites A2FeH6 (A= Be, Mg) for hydrogen storage applications, Int. J. Hydrogen Energy, 2024, 83, 307–316 CrossRef CAS.
- B. Ahmed, M. B. Tahir, A. Ali and M. Sagir, DFT insights on structural, electronic, optical and mechanical properties of double perovskites X2FeH6 (X= Ca and Sr) for hydrogen-storage applications, Int. J. Hydrogen Energy, 2024, 50, 316–323 CrossRef CAS.
- S. Al, M. Yortanlı and E. Mete, Lithium metal hydrides (Li2CaH4 and Li2SrH4) for hydrogen storage; mechanical, electronic and optical properties, Int. J. Hydrogen Energy, 2020, 45(38), 18782–18788 Search PubMed.
- A. Hosen, A. Sadeghi, H. A. Abdulhussein, E. Nemati-Kande, A. N. Khan, H. Bekkali, A. Akremi and I. Boukhris, First-principles insights into NaScQH6 (Q= Fe, Ru, Os): Promising high-density hydrogen storage materials, Int. J. Hydrogen Energy, 2025, 177, 151392 CrossRef CAS.
- M. mana Al-Anazy, G. M. Mustafa, O. Zayed, B. Younas, T. M. Al-Daraghmeh, N. D. Alkhaldi, A. S. Alofi, A. K. Alqorashi and Q. Mahmood, Study of alkaline metals hydrides RbXH3 (X= Mg/Ca/Sr/Ba) for green energy and hydrogen storage applications, Int. J. Hydrogen Energy, 2024, 78, 927–937 Search PubMed.
- W. Khan, Computational screening of BeXH3 (X: Al, Ga, and In) for optoelectronics and hydrogen storage applications, Mater. Sci. Semicond. Process., 2024, 174, 108221 Search PubMed.
- M. Z. Abbasi, A. U. Rehman, M. Sheraz, W. U. K. Tareen, M. Kaleem, S. T. Jan, H. Ali and T. C. Chuah, Enhancing the performance of lead-free CsInCl3 perovskite solar cells with Ag and Au plasmonic nanoparticles: A DFT and SCAPS-1D analysis, Results Eng., 2025, 105043 Search PubMed.
- M. Ali, Z. Bibi, S. Kanwal, T. Fatima, M. Raheel, A. F. Khan and M. Kaleem, A comprehensive investigation of structural, mechanical and optoelectronics attributes of M2AsC (M= Zr, Hf, Ta, W) MAX phase carbides: A DFT investigation, J. Mol. Graph. Model., 2025, 109102 Search PubMed.
- R. J. Meier, On the effectiveness of ultra-soft pseudopotentials in plane-wave based molecular electronic structure calculations, J. Mol. Struct., 1999, 467(1), 79–83 CrossRef CAS.
- A. Hussain, N. Jabeen, A. Yaqoob, A. Smerat, M. A. Qaiser and N. A. Aldawsari, Hydrogen Storage Capacity and Optoelectronic Response of Mechanically and Thermally Stable Lithium-Based Tetrahydrates (LiXH4, X= B, Al, Mn), a DFT Approach, Crystals, 2025, 15(11), 990 CrossRef CAS.
- A. N. Khan, M. Kaleem, N. U. Khan, A. Nasir, A. Khan and M. Z. Abbasi, Multi-functional DFT and SCAPS-1D analysis of lead-free Z2MgGeI6 (Z= Na, K) double perovskites for optoelectronic, photo-catalytic, and photovoltaic applications, Sol. Energy Mater. Sol. Cells, 2026, 294, 113922 CrossRef CAS.
- Z. Abbas, A. Alqahtani, K. Bibi, A. Parveen, Z. Bayhan, A. El-Rayyes and M. T. Khan, First-principles study of structural, electronic, optical, and hydrogen storage properties of Li2 TiXH6 (X= Cr, Mn) hydrides for an advanced hydrogen storage system, Int. J. Mod. Phys. B, 2025, 39(29n30), 2550261 Search PubMed.
- A. N. Khan, N. U. Khan, M. Kaleem, M. Tanzeel, A. Nasir, A. Hosen, A. Akremi and I. Boukhris, Lead-Free X2MgGeI6 (X= Rb, Cs) Double Perovskites for Multi-functional Energy Applications: A DFT and SCAPS-1D Perspective, Solid State Sci., 2025, 108049 CrossRef CAS.
- K. El Kihel, Z. El Fatouaki, A. Tahiri, M. Idiri and M. El Bouziani, Electronic structure, Optical, Phonon, Mechanical, Thermodynamic, and Hydrogen Storage properties of A2BH6 (A= Ca, Sr, Ba) perovskite hydrides by DFT calculations, Phys. B, 2026, 418219 CrossRef CAS.
- S. S. Raman, D. Davidson, J.-L. Bobet and O. Srivastava, Investigations on the synthesis, structural and microstructural characterizations of Mg-based K2PtCl6 type (Mg2FeH6) hydrogen storage material prepared by mechanical alloying, J. Alloys Compd., 2002, 333(1–2), 282–290 CrossRef CAS.
- S. Al, Mechanical and electronic properties of perovskite hydrides LiCaH 3 and NaCaH 3 for hydrogen storage applications, Eur. Phys. J. B, 2021, 94(9), 182 CrossRef CAS.
- A. Züttel, P. Wenger, S. Rentsch, P. Sudan, P. Mauron and C. Emmenegger, LiBH4 a new hydrogen storage material, J. Power Sources, 2003, 118(1–2), 1–7 Search PubMed.
- A. Obeidat, A. Alrousan and S. Abu-Rajouh, First-principles study of structural, hydrogen storage, mechanical, electronic, and optical properties of K2NaXH6 (X= Al, As, Bi, Ga, In) double perovskite hydrides, J. Power Sources, 2025, 642, 236944 CrossRef CAS.
- S. Bahou, H. Labrim, M. Lakhal, M. Bhihi, B. Hartiti and H. Ez-Zahraouy, Magnesium vacancies and hydrogen doping in MgH2 for improving gravimetric capacity and desorption temperature, Int. J. Hydrogen Energy, 2021, 46(2), 2322–2329 CrossRef CAS.
- A. Hosen, A. A. Mousa, E. Nemati-Kande, A. N. Khan, M. S. Abu-Jafar, E. Benassi, E. Elghmaz, N. Abd EL-Gawaad and J. Asad, Systematic Computational Screening and Design of Double Perovskites Q2LiMH6 (Q= K, Rb; M= Ga, In, Tl) for Efficient Hydrogen Storage: A DFT and AIMD Approach, Surf. Interfaces, 2025, 106608 Search PubMed.
- M. Kaleem, M. M. A. Iqbal and A. N. Khan, Stability and hydrogen storage performance of Na 2 LiXH 6 (X= Zr, V, Cr) double perovskite hydrides via DFT and AIMD, RSC Adv., 2026, 16(2), 995–1007 RSC.
- A. Ayyaz, M. Kaleem, A. Nasir, N. D. Alkhaldi, M. mana Al-Anazy, I. Boukhris, Q. Mahmood and M. Z. Abbasi, Investigation of optoelectronic and photovoltaic characteristics of A2NaAlI6 (A= Rb, Cs)-based perovskite solar cells with different charge transport layers: DFT and SCAPS-1D simulation, Sol. Energy, 2025, 299, 113738 CrossRef CAS.
- R. Song, N. Xu, Y. Chen, S. Chen, J. Zhang, W. Dai and W. Zhang, Insight into the mechanical, electronic, kinetic, thermodynamic, and hydrogen storage properties of XFeH3 (X= Ca, Sr, Ba) perovskites for hydrogen storage applications: first-principle calculations, Chin. J. Phys., 2024, 89, 1152–1163 Search PubMed.
- B. Rehmat, M. Rafiq, Y. Javed, Z. Irshad, N. Ahmed and S. Mirza, Elastic properties of perovskite-type hydrides LiBeH3 and NaBeH3 for hydrogen storage, Int. J. Hydrogen Energy, 2017, 42(15), 10038–10046 CrossRef CAS.
- M. Ali, M. Raheel, Z. Bibi, M. A. Rusho, D. Khan, W. Al-Azzawi and R. A. Alshgari, Atomistic simulations for electronic structure, mechanical stability and optical responses of sodium-based NaAH3 (A= Sc, Ti and V) metal hydride perovskites for hydrogen storage applications, Int. J. Hydrogen Energy, 2025, 128, 749–759 CrossRef CAS.
- X. Yang, Y. Li, Y. Liu, Q. Li, T. Yang and H. Jia, Crystal structure prediction and performance assessment of hydrogen storage materials: Insights from computational materials science, Energies, 2024, 17(14), 3591 CrossRef CAS.
- A. Es-Smairi, S. Al-Qaisi, N. Sfina, A. Boutramine, H. R. Jappor, H. S. Alzahrani, A. H. Alfaifi, H. Rached, A. S. Verma and M. Archi, DFT insights into the structural, stability, elastic, and optoelectronic characteristics of Na2LiZF6 (Z= Ir and Rh) double perovskites for sustainable energy, J. Comput. Chem., 2025, 46(8), e70097 CrossRef CAS PubMed.
- S. Takagi, H. Saitoh, N. Endo, R. Sato, T. Ikeshoji, M. Matsuo, K. Miwa, K. Aoki and S.-i. Orimo, Density-functional study of perovskite-type hydride LiNiH 3 and its synthesis: Mechanism for formation of metallic perovskite, Phys. Rev. B Condens. Matter, 2013, 87(12), 125134 CrossRef.
- F. Qureshi, M. Yusuf, S. Ahmed, M. Haq, A. M. Alraih, T. Hidouri, H. Kamyab, D.-V. N. Vo and H. Ibrahim, Advancements in sorption-based materials for hydrogen storage and utilization: A comprehensive review, Energy, 2024, 309, 132855 CrossRef CAS.
- Y. Tang, J. Zhang, X. Zhong, Q. Wang, H. Zhang, C. Ren and J. Wang, Revealing the structural, electronic and optical properties of lead-free perovskite derivatives of Rb2SnX6 (X= Cl, Br and I): a theory calculation, Sol. Energy, 2019, 190, 272–277 CrossRef CAS.
- S. Hayat, R. A. Khalil, M. I. Hussain, A. M. Rana and F. Hussain, First-principles investigations of the structural, optoelectronic, magnetic and thermodynamic properties of hydride perovskites XCuH3 (X= Co, Ni, Zn) for hydrogen storage applications, Optik, 2021, 228, 166187 CrossRef CAS.
- M. Tanzeel, M. Kaleem, A. N. Khan, A. Nasir, M. Ali, A. Alshamari, A. Ayyaz and A. Almohammedi, A computational investigation of novel double perovskite oxides Ba2XReO6 (X= Li, Na) for optoelectronic and photocatalytic applications, Int. J. Mod. Phys. B, 2025, 2650012 Search PubMed.
- A. Ayyaz, M. Kaleem, A. Shakoor, H. D. Alkhaldi, A. Nasir, A. Akremi, H. Albalawi and Q. Mahmood, Investigation of promising stable halide double perovskites K2LiXI6 (X= Al, Ga) for solar cells and thermoelectric device applications: DFT and SCAPS-1D simulation approach, Chem. Phys. Lett., 2025, 142433 CrossRef CAS.
- H. Murtaza, J. Munir, Q. Ain, A. S. Aldwayyan, H. M. Ghaithan, A. A. A. Ahmed and S. M. Qaid, First-principles prediction of the optoelectronic, mechanical, thermodynamic and hydrogen storage attributes of double perovskite Rb2NaXH6 (X= Al, In) hydrides, J. Inorg. Organomet. Polym. Mater., 2025, 35(4), 2877–2888 CrossRef CAS.
- S. Zhang, S. Chen, Y. Chen, J. Hou, D. Xu, W. Luo and Z. Shi, First principles study on the structure, hydrogen storage, and physical properties of X2VH6 (X= Mg, Ca, Sr, Ba) perovskite hydrides for hydrogen storage applications, Int. J. Hydrogen Energy, 2025, 166, 150903 CrossRef CAS.
- N. Al-Zoubi, A. Almahmoud, A. Almahmoud and A. Obeidat, Theoretical assessment of a novel NaXH3 and KXH3 (X= Tc, Ru and Rh) perovskite hydrides for hydrogen storage, Int. J. Hydrogen Energy, 2024, 93, 822–831 CrossRef CAS.
- S. Al-Qaisi, N. Iram, N. Sfina, A. Boutramine, H. R. Jappor, A. H. Alfaifi, H. S. Alzahrani, H. Rached, M. A. Ali and G. Murtaza, Comprehensive DFT study of K2TlZI6 (Z= Al, In) double perovskites: Structural stability and potential for optoelectronic and thermoelectric energy harvesting, Phys. B, 2025, 710, 417239 CrossRef CAS.
- O. L. Anderson, A simplified method for calculating the Debye temperature from elastic constants, J. Phys. Chem. Solids, 1963, 24(7), 909–917 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |





