Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Nitrogen-containing FDA-approved drugs in 2025: synthesis, significance and therapeutic applications
Chetna Jadalaa,
Shweta Mishrabc,
Gal Reddy Potuganti d,
Savio Cardozae and
Ganga Reddy Velma
d,
Savio Cardozae and
Ganga Reddy Velma *e
*e
aIndiana University School of Medicine, Indiana University (IU), Indianapolis, 46202, IN, USA
bDr. D.Y. Patil Institute of Pharmaceutical Sciences and Research, Sant Tukaram Nagar, Pimpri, Pune, 411018, India
cDPGU - School of Pharmacy and Research, Sant Tukaram Nagar, Pimpri, Pune, 411018, India
dDepartment of Chemistry and Biochemistry, The University of Oklahoma, Norman, Oklahoma 73019, USA
eDepartment of Pharmacology and Toxicology, R. Ken Coit College of Pharmacy, University of Arizona, Tucson, 85721, AZ, USA. E-mail: vgreddy@arizona.edu; velmagangareddy47@gmail.com
First published on 27th April 2026
Abstract
Nitrogen-containing heterocycles (N-Hets) are among the most prevalent and versatile structural motifs in pharmaceuticals, serving as key scaffolds in over 85% of biologically active small molecules. Their structural diversity and functional versatility have made them integral components of both U.S. Food and Drug Administration (FDA) approved and investigational drug molecules, modulating biological activity across a range of molecular targets. In 2025, the FDA approved 26 small-molecule drugs featuring N-Het frameworks, spanning multiple therapeutic areas, including oncology, metabolic disorders, infectious diseases, and rare/orphan conditions. This review offers a comprehensive overview of these newly approved compounds, emphasizing their biological activity and synthetic approaches. Special attention is given to drug–target interactions, focusing on receptor binding and highlighting the important role of N-Hets in medicinal chemistry. By exploiting the intrinsic chemical properties of N-Hets and leveraging modern synthetic methodologies, these scaffolds continue to drive the discovery of therapeutically relevant molecules, highlighting their sustained significance in modern drug discovery.
1. Introduction
Heterocyclic compounds, in general, constitute an important class of naturally occurring molecules with favorable physicochemical and biological properties, conferring substantial pharmaceutical relevance. Their adaptable structures serve as biomimetic frameworks, often incorporating essential pharmacophoric elements, making them indispensable motifs in modern drug discovery.1–3 Among the diverse heterocyclic systems,4 nitrogen-containing heterocycles (N-Hets) are particularly valued for their structural diversity, chemical stability, and ability to modulate biological targets with high specificity.5,6 N-Het represents one of the most prominent structural motifs in pharmaceuticals and remains a key fragment in drug discovery.7 Their versatile frameworks provide structural flexibility, enabling fine-tuning of the pharmacokinetic properties and biological efficacy of both investigational and approved drug molecules.8 Over the past two decades, these scaffolds have attracted extensive research, driving the design of novel small molecules and enabling the development of innovative synthetic strategies. Beyond advancing organic synthesis, N-Hets have played a central role in the discovery and development of therapeutically relevant compounds.9–12This prominent feature of N-Het is further supported by a comprehensive database analysis of approved pharmaceuticals, underscoring their widespread presence in clinically successful therapeutics. The N-heterocyclic skeletons span a broad range of therapeutic applications, including anticancer,13–16 anti-HIV,17 anti-inflammatory,18 anti-convulsant,19 anti-diabetic,13 anti-bacterial20 and anti-depressants.21 They serve as key building blocks for numerous drug candidates, largely owing to the ability of nitrogen atoms to readily participate in hydrogen-bonding interactions with biological targets, thereby enhancing target recognition and binding affinity. Among the diverse heterocyclic frameworks, triazoles,22 tetrazoles,23 imidazoles,24 benzimidazoles,25 pyrimidines,26,27 and quinolines26 have received particular attention, emerging as highly prevalent scaffolds contributing to a wide range of therapeutic applications across diverse disease areas.
A review of FDA-approved small molecules from 2021 to 2025 reveals a variable trend in N-Het approvals (Fig. 1). In 2021, 32 of 50 small molecules (64%) contained N-heterocyclic scaffolds. Approval declined in 2022 and 2023, with 14 of 37 (37%) and 13 of 55 (23%) N-heterocyclic small molecules, respectively. This trend reversed in 2024 and 2025, with approvals increasing to 23 of 50 (46%) and 26 of 46 (56%), respectively, highlighting the sustained relevance of N-Het in medicinal chemistry and drug discovery. In 2025, the FDA approved 46 drugs, of which 29 were small molecules.28 Among these, N-Het drugs accounted for 56.5% of approvals, reflecting their predominance among small molecules. Monoclonal antibodies, biologics, and antibody–drug conjugates (ADCs) together accounted for 30.4%, while oligonucleotide- and siRNA-based therapies accounted for the remaining 6.5% (Fig. 2).
 | ||
| Fig. 1 Trends in FDA drug approvals and nitrogen-containing heterocyclic small molecules from 2021 to 2025. | ||
In 2025, FDA approvals spanned a broad range of therapeutic areas, reflecting ongoing efforts to address unmet medical needs in oncology, rare genetic disorders, metabolic diseases, infectious diseases, and cardiovascular conditions.15,29,30 Notably, 16 drugs (∼35%) targeted various cancers, including 9 N-Het small molecules (Fig. 3). Key approvals include Romvimza™ (vimseltinib) for symptomatic tenosynovial giant cell tumor (TGCT); Avmapki Fakzynja Co-Pack™ for KRAS-mutated recurrent low-grade serous ovarian cancer (LGSOC); Komzifti™ (ziftomenib) for relapsed or refractory acute leukemia; and Hyrnuo™ (sevabertinib) for HER2-mutated non-small cell lung cancer, highlighting ongoing advances in targeted oncology therapies and the continued prominence of N-Het scaffolds. Beyond oncology, approvals addressed genetic, rare, and metabolic disorders (Fig. 4), such as Kygevvi™ (doxecitine and doxribtimine) for thymidine kinase 2 deficiency, Sephience™ (sepiapterin) for hyperphenylalaninemia in phenylketonuria, and Palsonify™ (paltusotine) for adult acromegaly. Cardiovascular innovations included Myqorzo™ (aficamten) for obstructive hypertrophic cardiomyopathy. Infectious disease approvals included novel oral antibiotics Blujepa™ (gepotidacin) and Nuzolvence™ (zoliflodacin) for urinary tract infections and uncomplicated gonorrhea, reflecting efforts against antimicrobial-resistant pathogens.
 | ||
| Fig. 4 Nitrogen-containing non-anticancerous drugs approved by the FDA in 2025 across different therapeutic areas. | ||
Additional approvals encompassed lung diseases, including non-cystic fibrosis bronchiectasis (NCFB) with Brinsupri™ (brensocatib) and idiopathic pulmonary fibrosis (IPF) with Jascayd™ (nerandomilast), rare blood disorders were addressed by Wayrilz™ (rilzabrutinib), while endocrine and menopausal symptoms were treated with Lynkuet™ (elinzanetant), ophthalmic conditions such as presbyopia were managed by Vizz™, (aceclidine). Pain management included Journavx™ (suzetrigine), dermatological conditions encompassed chronic hand eczema with Anzupgo™ (delgocitinib), and for chronic spontaneous urticaria with Rhapsido™ (remibrutinib), and Nereus™ (tradipitant) was approved for motion sickness. Importantly, 2025 approvals emphasized treatments for rare and orphan diseases, including Ekterly™ (sebetralstat), Vanrafia™ (atrasentan), and Romvimza™ (vimseltinib) etc., highlighting a strategic focus on addressing unmet needs in less common conditions.
This review focuses on the synthetic strategies and clinical applications of FDA-approved nitrogen-containing heterocycles in FDA-approved drugs in 2025, with the aim of providing insights into their mechanisms of action and therapeutic potential. To facilitate review and interpretation, nitrogen-containing heterocyclic molecules are classified into anticancer and non-anticancer categories. We further examine the synthetic pathways used in their development, emphasizing innovative approaches that could guide future drug design efforts.
In light of the increasing complexity of modern drug discovery, the efficient and sustainable synthesis of these molecules remains a critical component for advancing pharmaceutical research and accelerating the translation of novel therapies to the clinic. In this context, understanding the structure–activity relationships and pharmacophoric features of nitrogen-containing scaffolds is essential.31
1.1 Pharmacophore features, rational design, and SAR considerations
Although the FDA-approved drugs discussed in this review are structurally diverse, several general structure–activity relationship (SAR) trends can be identified for nitrogen-containing scaffolds. The presence and placement of nitrogen atoms significantly impact target binding through hydrogen bonds and ionic interactions, thus influencing potency and selectivity. Aromatic nitrogen heterocycles often contribute to π–π stacking and electronic modulation, while aliphatic amines enhance solubility and pharmacokinetic properties.32 Additionally, substitution patterns on heterocyclic frameworks are crucial for enhancing biological activity and target specificity.Nitrogen-containing heterocycles play a central role in shaping the pharmacophoric structure of modern drugs. Incorporating nitrogen atoms as amines, amides, and within heterocyclic frameworks enables key interactions with biological targets, such as hydrogen bonding and ionic interactions, thereby improving binding affinity and selectivity. In many relevant clinical situations, protonated nitrogen centers help facilitate electrostatic interactions with negatively charged residues in enzyme active sites or receptor binding pockets.33
From a rational design perspective, nitrogen atoms are incorporated to adjust physicochemical properties such as pKa, aqueous solubility, and membrane permeability. Aromatic nitrogen-containing heterocycles like pyridine, imidazole, triazole, and pyrimidine serve as adaptable scaffolds for modifying electronic properties and enhancing metabolic stability. Conversely, saturated nitrogen-containing motifs such as piperidine and morpholine are often added to increase solubility and improve pharmacokinetic profiles. These design principles are evident in many FDA-approved drugs discussed in this review, where nitrogen-containing pharmacophores enhance target specificity and therapeutic effectiveness. The dominance of nitrogen-containing frameworks among FDA-approved drugs in 2025 further emphasizes their importance in balancing potency, selectivity, and favorable pharmacokinetic properties.34
2. FDA-approved nitrogen-containing drugs in 2025
2.1 Anticancer drugs
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) to receive either vimseltinib or placebo. Approval of vimseltinib underscores the successful translation of selective CSF1R inhibition from molecular design to clinical efficacy. Vimseltinib acts as a selective CSF1R inhibitor by binding to the kinase switch-control region and stabilizing the receptor in an inactive conformation (PDB ID: 7MFC). At the molecular level, the inhibitor forms key hydrogen-bonding interactions with residues such as Asp-796 and Cys-712 within the active site. Additional interactions with residues, including Lys-662, further contribute to stabilization of the drug–target complex. These interactions collectively promote the DFG-out conformation and enhance binding affinity and selectivity.36 Limitations of the drug: treatment may cause elevated Aspartate Aminotransferase (AST)/Alanine Aminotransferase (ALT) and fetal risk, so liver tests and contraception are advised.
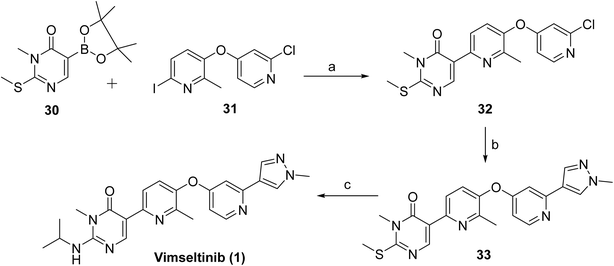
1) to receive either vimseltinib or placebo. Approval of vimseltinib underscores the successful translation of selective CSF1R inhibition from molecular design to clinical efficacy. Vimseltinib acts as a selective CSF1R inhibitor by binding to the kinase switch-control region and stabilizing the receptor in an inactive conformation (PDB ID: 7MFC). At the molecular level, the inhibitor forms key hydrogen-bonding interactions with residues such as Asp-796 and Cys-712 within the active site. Additional interactions with residues, including Lys-662, further contribute to stabilization of the drug–target complex. These interactions collectively promote the DFG-out conformation and enhance binding affinity and selectivity.36 Limitations of the drug: treatment may cause elevated Aspartate Aminotransferase (AST)/Alanine Aminotransferase (ALT) and fetal risk, so liver tests and contraception are advised.Schemes 1 and 2 outlines the synthetic route used for the preparation of vimseltinib (1).35 Methylation of the commercially available pyrimidone 27, using iodomethane and LiHMDS afforded intermediate 28, which was subsequently brominated to give 29. Intermediate 29 was then converted to corresponding boronate ester 30 via palladium-mediated cross-coupling reaction with bis(pinacolato)diboron. Suzuki–Miyaura cross-coupling of 30 with the 2-chloropyridine derivative 31 provided the intermediate 32, which was further coupled with the appropriately substituted pyrazole boronate derivative, under classical Suzuki-type coupling conditions, to afford the pyrimidone 33. Final amination of 33 with isopropylamine provided vimseltinib (1).
 | ||
| Scheme 1 Synthesis of intermediate 30. aReaction conditions: (a) LiHMDS, MeI, DMF (62%); (b) Br2, DCM (97%); (c) bis(pinacolato)diboron, KOAc, PdCl2(dppf), DCM, 1,4-dioxane, 85 °C (100%). | ||
LGSOC38 is a rare, slow-growing, and highly recurrent ovarian cancer that predominantly affects younger women and shows limited responsiveness to standard platinum-based chemotherapy, approximately 30% of cases harbor KRAS mutations. On May 8, 2025, Avmapki Fakzynja Co-Pack (avutometinib in combination with defactinib) became the first FDA-approved therapy for KRAS-mutated recurrent LGSOC, receiving accelerated approval based on results from the Phase 2 RAMP 201 trial (NCT04625270), which demonstrated a 44% overall response rate and a duration ranging from 3.3 to 31.1 months. Prior to this approval, no FDA-approved treatments were available for LGSOC, which is a biologically distinct subtype that differs from high-grade serous ovarian cancer in both disease pathology and therapeutic response. The combination was granted breakthrough therapy and orphan drug designations. Avutometinib functions as a RAF/MEK pathway inhibitor by modulating kinase signaling and stabilizing the inactive conformation of MEK, thereby preventing downstream MAPK activation. Defactinib acts as an ATP-competitive inhibitor of focal adhesion kinase (FAK), binding within the catalytic domain and disrupting integrin-mediated signaling pathways involved in tumor cell survival and migration. These complementary mechanisms enable dual-pathway inhibition and contribute to enhanced antitumor efficacy. A limitation of this therapy is that approval was granted under the accelerated approval pathway based on response rate, and continued approval is contingent upon confirmation of clinical benefit in a confirmatory trial.
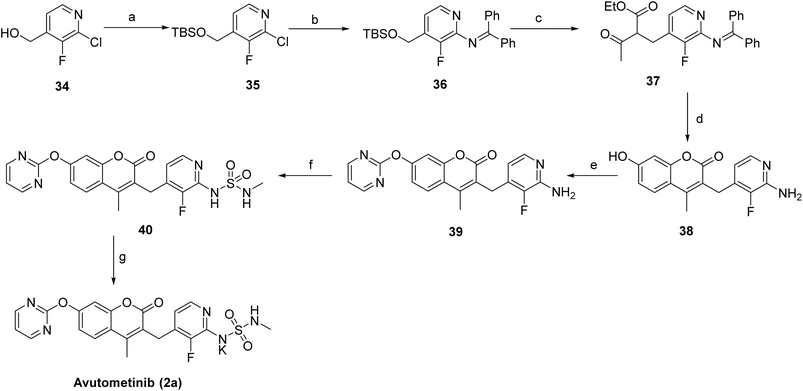
Scheme 3 outlines the synthesis of avutometinib (2a).39 The route begins with the protection of the hydroxy group in 34 using tert-butyldimethylsilyl chloride, affording the protected intermediate 35, followed by palladium-catalyzed C–N coupling with benzophenone imine to give 36. C–C bond formation with ethyl 3-oxobutanoate generated 37, which then underwent acid-mediated cyclization with resorcinol to furnish 38. The pyrimidine moiety was installed via nucleophilic substitution with 2-bromopyrimidine, producing 39, and the sulfonamide functionality was introduced by reaction with N-methylsulfamoyl chloride to give 40. Final deprotection and/or saponification using KOH afforded the target compound, avutometinib (2a).
The target compound defactinib (2b)40 was synthesized as shown in Scheme 4. The synthesis began with the introduction of the N-methylsulfonamide group on 41 using N-methyl-methanesulfonamide in the presence of Cs2CO3, affording intermediate 42. The resulting intermediate 42 was then reduced with H2 and Pd/C to provide 43. Subsequent coupling of 43 with 4-((4-chloro-5-(trifluoromethyl)pyrimidin-2-yl)amino)-N-methylbenzamide in the presence of DIEA in DCE/t-BuOH installed the substituted pyrimidine amide functionality. Finally, treatment with HCl in MeOH resulted in deprotection, yielding the desired compound, defactinib (2b).
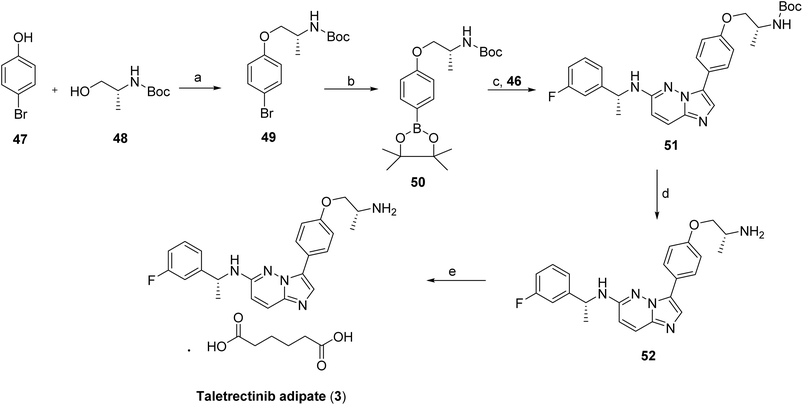
The synthesis of taletrectinib (3)44 begins with a Mitsunobu coupling between p-bromophenol (47) and N-Boc-D-alaninol (48) using PPh3 and DIAD in THF, affording N-Boc-1-(4-bromophenoxy)-2(R)-propanamine (49) as shown in Scheme 6. This intermediate is then subjected to Miyaura borylation with bis(pinacolato)diboron in the presence of PdCl2(dppf)·CH2Cl2 and KOAc in 1,4-dioxane at 80 °C to yield the corresponding boronate ester (50). In a parallel sequence, nucleophilic aromatic substitution between 1(R)-(3-fluorophenyl)ethanamine (45) and 3-bromo-6-chloroimidazo[1,2-b]pyridazine, conducted in the presence of KF in DMSO at 120 °C, furnishes the secondary amine intermediate (46), as shown in Scheme 5. This intermediate is subsequently coupled with boronate (50) via a Suzuki–Miyaura cross-coupling using PdCl2(dppf)·CH2Cl2 and K2CO3 in refluxing 1,4-dioxane/water to afford N-Boc-protected taletrectinib (51). Final carbamate deprotection using HCl in methanol/1,4-dioxane yields taletrectinib free base (52), which is then converted to the final product by salt formation with adipic acid in n-propanol affording taletrectinib adipate (3).
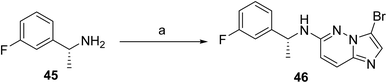 | ||
| Scheme 5 Synthesis of intermediate (46). aReaction conditions: (a) 3-bromo-6-chloroimidazo[1,2-b]pyridazine KF, DMSO at 120 °C. | ||
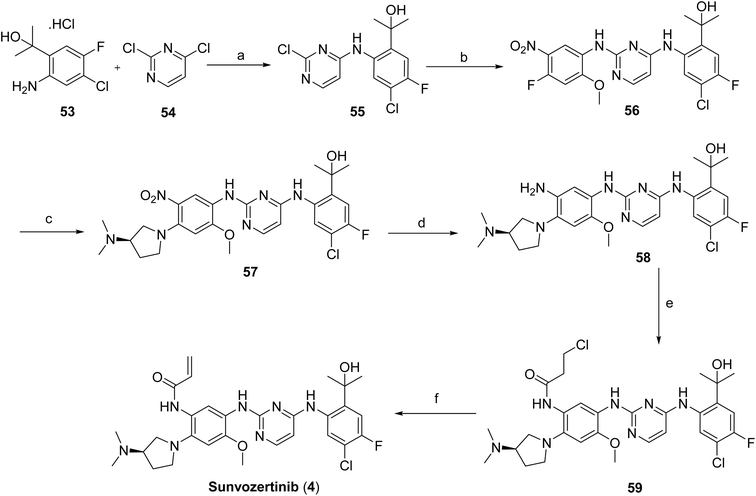
The synthesis of sunvozertinib (4) is depicted in Scheme 7. Compounds 53 and 54 undergo nucleophilic substitution under alkaline conditions to afford intermediate 55, which reacts with 4-fluoro-2-methoxy-5-nitroaniline to yield intermediate 56. Subsequent nucleophilic substitution of 56 with (R)-N,N-dimethylpyrrolidin-3-amine provides intermediate 57. Reduction of the nitro group in 57 using hydrogen and platinum on carbon furnishes the corresponding amino intermediate 58. Condensation of 58 with an acyl chloride generates amide 59, which undergoes base-mediated elimination to afford the final product, sunvozertinib (4).48
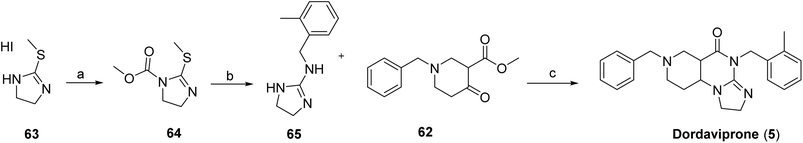
The synthesis of dordaviprone (5)51 begins with the formation of intermediate 64 from commercially available 2-methylthio-2-imidazoline hydroiodide (63) through reaction with methyl chloroformate as shown in Scheme 9. Intermediate 64 is then coupled with 2-methylbenzylamine to generate the corresponding guanidine derivative 65. In parallel, 1-benzyl-4-oxopiperidine-3-carboxylate (62) is prepared from benzylamine (60) and methyl acrylate via dimethyl 3,3′-(benzylazanediyl)dipropionate (61), which is subsequently treated with sodium hydride in THF to yield 62 as shown in Scheme 8. The final step involves condensation of 65 with 62 under reflux in methanol in the presence of sodium methoxide, affording dordaviprone (5).
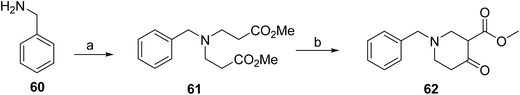 | ||
| Scheme 8 Synthesis of intermediate (62). aReaction conditions: (a) methyl acrylate, MeOH; (b) NaH, THF. | ||
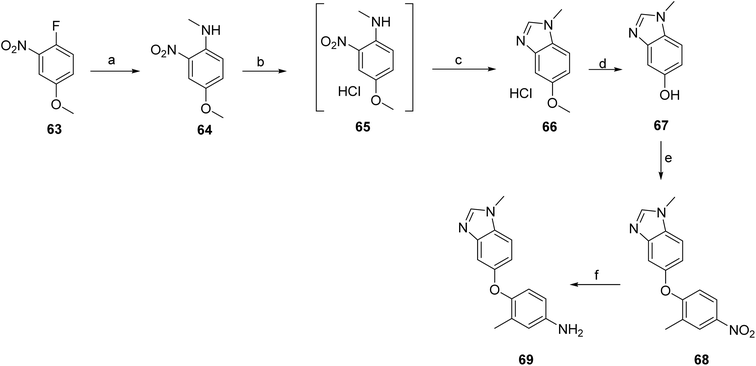
The synthesis for zongertinib proceeds via the initial preparation of intermediate 69. The Intermediate 69 was synthesized from starting material 63 via a nucleophilic aromatic substitution (SNAr) with methylamine, affording intermediate 64 as shown in Scheme 10. Catalytic hydrogenation of 64 yielded the corresponding 65, which was subsequently cyclized with methyl formate to construct the benzimidazole core, giving intermediate 66. Demethylation of 66 produced compound 67, which then underwent a further SNAr reaction to furnish intermediate 68. Final hydrogenation under mild conditions provided compound 69 after purification by crystallization.
The synthesis of zongertinib was carried out using intermediate 69. The piperazine fragment was installed first via an SNAr reaction after the oxidation of methyl sulfide 70 to sulfoxide 71 using m-CPBA to make intermediate 72, as shown in Scheme 11. Subsequent chlorination of 72 with oxalyl chloride yielded the compound 73, followed by amination with 69, which provided intermediate 74. Boc-deprotection of 74 formed the precursor intermediate 75, which was used to make compound 6 using amidation with acryloyl chloride.55,56
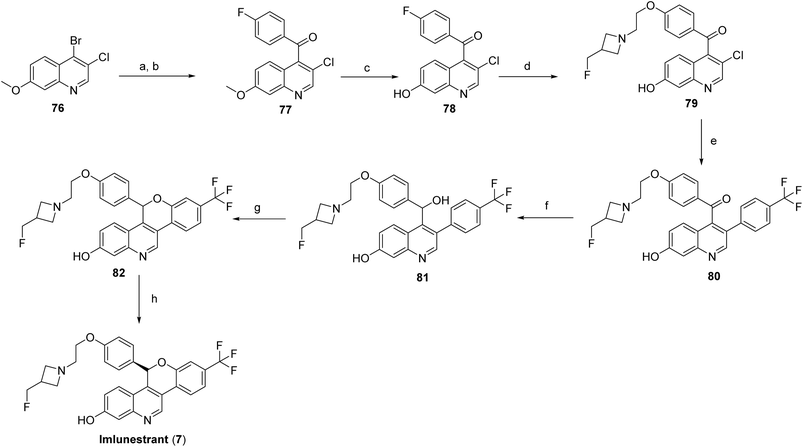
Imlunestrant (7) is synthesized from 4-bromo-3-chloro-7-methoxyquinoline (76) via a metal–halogen exchange with i-PrMgCl, followed by Grignard acylation with 4-fluorobenzoyl chloride to yield (3-chloro-7-methoxy-4-quinolyl)(4-fluorophenyl)methanone (77). The 7-methoxy group is then selectively demethylated using BBr3 to provide the 7-hydroxyquinoline derivative (78), which undergoes ether formation with 2-[3-(fluoromethyl)azetidin-1-yl]ethanol via NaH-mediated alkylation, affording intermediate (79). The fluoroaryl intermediate (79) is further elaborated through a Suzuki–Miyaura cross-coupling with [4-(trifluoromethyl)phenyl]boronic acid to generate the diaryl ketone (80), which is reduced with LiBHEt3 to the corresponding secondary alcohol (81). Intramolecular cyclization of this alcohol under basic conditions furnishes the racemic chromeno[4,3-c]quinolin-2-ol core (82), which is finally resolved by chiral supercritical fluid chromatography (SFC) to yield enantiomerically pure imlunestrant (7) as shown in Scheme 12.
On November 13, 2025, the U.S. FDA approved ziftomenib (Komzifti) for the treatment of adult patients with relapsed or refractory acute myeloid leukemia (AML) harboring a susceptible nucleophosmin 1 (NPM1) mutation and lacking satisfactory alternative treatment options. Approval was based on efficacy results from the KO-MEN-001 trial (NCT04067336), an open-label, multicenter, single-arm study evaluating 112 adults with relapsed or refractory NPM1-mutant AML, including tumors with type A, B, D, and other NPM1 variants associated with cytoplasmic localization of the NPM1 protein, as determined by next-generation sequencing or PCR. Ziftomenib was granted breakthrough therapy and orphan drug designations. The prescribing information includes warnings for differentiation syndrome, QTc interval prolongation, and embryo-fetal toxicity. Differentiation syndrome,61 which may be fatal, has been reported and requires prompt interruption of therapy, initiation of corticosteroids and appropriate clinical monitoring, followed by resumption of treatment upon symptom resolution.
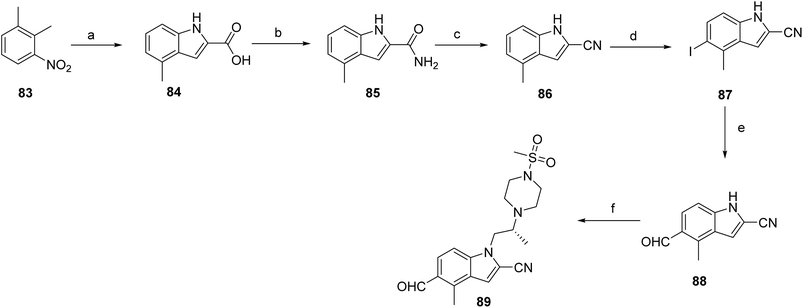
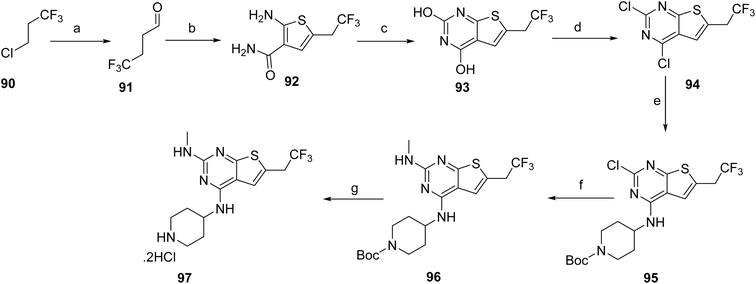
The synthesis of ziftomenib (8) proceeds through first synthesizing the intermediates (89 & 97). Compound 84 was prepared via base-mediated condensation of starting material 83 with diethyl oxalate in the presence of tert-amyl alcohol. Activation of 84 followed by amidation furnished 85, which was subsequently converted to the intermediate 86 by reacting with phosphorus oxychloride. Electrophilic iodination of this intermediate using boron trifluoride etherate and N-iodosuccinimide afforded compound 87. The iodinated intermediate was then elaborated through a palladium-catalyzed carbonylative coupling to generate 88. Final functionalization involved conversion of the alcohol moiety of 88 to the corresponding triflate, followed by base-mediated coupling with (R)-2-(4-(methylsulfonyl)piperazin-1-yl)propan-1-ol to deliver intermediate 89 as shown in Scheme 13.
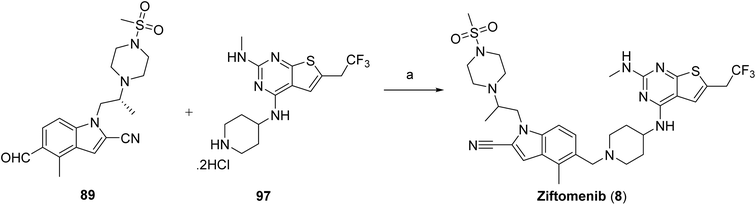
The synthesis of intermediate 97 begins with the formation of an alkyl Grignard reagent from a halogenated trifluoropropane precursor (90), which is subsequently converted to the corresponding trifluorinated aldehyde (91). This aldehyde undergoes a multicomponent thiophene-forming reaction with 2-cyanoacetamide and elemental sulfur under basic conditions to furnish the substituted aminothiophene carboxamide (92). Cyclization of this intermediate using a carbonyl-activating reagent affords the corresponding thieno[2,3-d]pyrimidine-2,4-diol core (93). The heterocyclic core is then activated through chlorination to generate a dichlorothienopyrimidine intermediate (94), enabling subsequent nucleophilic aromatic substitution with a protected amine fragment to give compound 95. Further functionalization is achieved via amine displacement using methylamine to introduce the desired amino substituent, affording compound 96. Final deprotection under acidic conditions yields the target intermediate 97, Scheme 14. The final assembly of ziftomenib (8) was achieved through a reductive coupling strategy, Scheme 15. An activated hydride reducing system, generated from sodium borohydride and isobutyric acid, was used to mediate the reductive transformation of a preassembled mixture of intermediates 89 and 97 in the presence of a base. This reduction furnished the target ziftomenib (8) as the final product.
On November 19, 2025, the U.S. FDA granted accelerated approval to sevabertinib (Hyrnuo™) for adult patients with locally advanced or metastatic, non-squamous NSCLC harboring HER2 (ERBB2) tyrosine kinase domain-activating mutations,54,62,63 as detected by an FDA-approved test, who had received prior systemic therapy. The approval was based on efficacy data from the SOHO-01 trial (NCT05099172), an open-label, multicenter, single-arm, multi-cohort study evaluating sevabertinib in previously treated patients with HER2-mutant NSCLC. Sevabertinib was granted priority review and received both breakthrough therapy and orphan drug designations, highlighting its clinical significance in a molecularly defined patient population with high unmet medical need. The prescribing information includes warnings and precautions for diarrhea, hepatotoxicity, interstitial lung disease (ILD)/pneumonitis, ocular toxicity, pancreatic enzyme elevations, and embryo-fetal toxicity.
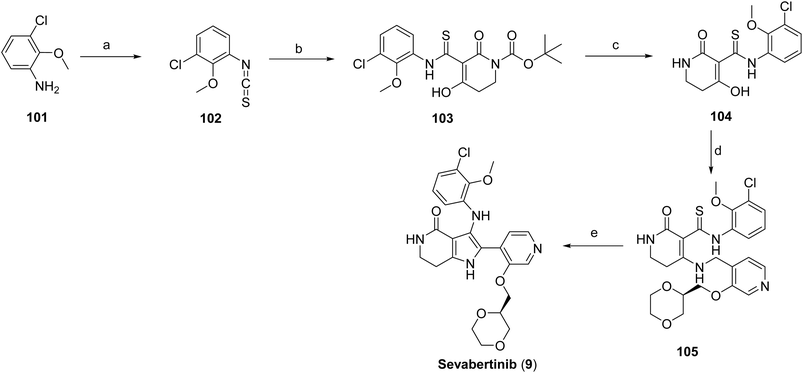
The synthesis of sevabertinib (9) proceeds via the synthesis of intermediate 100. The starting material 98 was reacted with (1,4-dioxan-2-yl)methanol to afford 99 which was subjected to catalytic hydrogenation to give the intermediate 100, Scheme 16. 3-Chloro-2-methoxyaniline (101) was converted to the corresponding isothiocyanate (102) under standard thiophosgene conditions at low temperature. The resulting intermediate was Boc-protected to afford intermediate 103, which was subsequently deprotected under acidic conditions to give intermediate 104. Intermediate 104 was then coupled with intermediate 100 at elevated temperature to furnish compound 105. Final oxidative transformation of compound 105 in methanol under acidic conditions completed the synthesis of the target compound, sevabertinib (9) (Scheme 17).
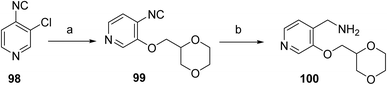 | ||
| Scheme 16 Synthesis of intermediate (100). aReaction conditions: (a) (1,4-dioxan-2-yl)methanol; (b) LiAlH4, THF. | ||
2.2 Non-anticancerous drugs
The U.S. FDA approved suzetrigine as the first drug in this new class of peripherally acting sodium channel-targeted analgesics. Acute pain, typically arising from tissue injury such as trauma or surgery, is commonly managed with analgesics that may include opioids, highlighting the clinical need for effective non-opioid alternatives. The efficacy of suzetrigine was demonstrated in two randomized, double-blind, placebo- and active-controlled clinical trials of acute postoperative pain following abdominoplasty and bunionectomy. In both studies, suzetrigine produced a statistically significant reduction in pain compared with placebo, with ibuprofen permitted as rescue medication for inadequate pain control. The prescribing information for Journavx includes warnings for pruritus, muscle spasms, elevated creatine phosphokinase, and rash, contraindication with strong CYP3A inhibitors, avoidance of grapefruit, and use in severe hepatic impairment.
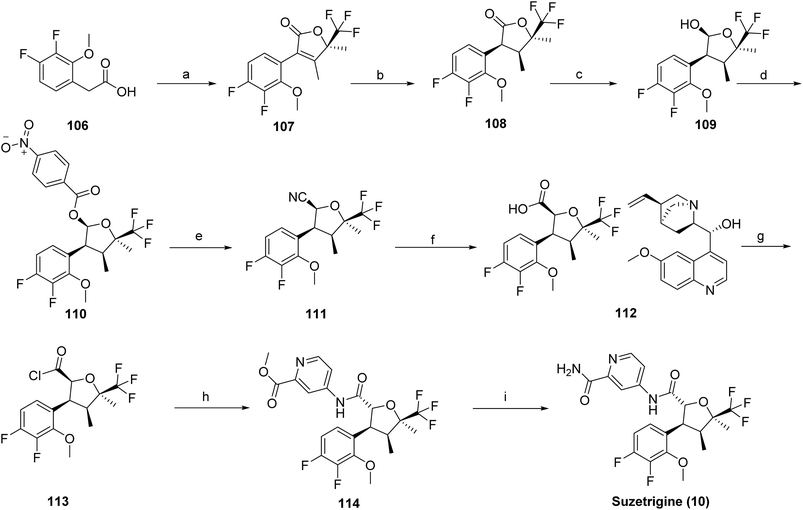
The chemical synthesis of suzetrigine (10)66 was reported by Vertex Pharmaceuticals through a series of patents and involves a multistep sequence, as outlined in Scheme 18. The synthesis begins with (R)-4,4,4-trifluoro-3-hydroxy-3-methylbutan-2-one, which is activated using 1,1′-carbonyldiimidazole (CDI) to generate the corresponding activated intermediate (107). Catalytic hydrogenation over Pd/C furnished the reduced product (108), which was then selectively reduced with diisobutylaluminium hydride (DIBAL-H) to afford intermediate (109). Acylation with 4-nitrobenzoyl chloride in the presence of triethylamine produced the ester derivative (110). Introduction of the nitrile functionality was achieved via trimethylsilyl cyanide in the presence of boron trifluoride diethyl etherate (111). Enantioenrichment was accomplished through quinine-mediated resolution (112), followed by acidic deprotection to yield the free intermediate (113). Coupling with methyl 4-aminopicolinate provided intermediate (114), which upon aminolysis with ammonia furnished the final product, suzetrigine (10).
Blujepa is approved for the treatment of uncomplicated urinary tract infections (uUTIs) in adult and pediatric female patients (≥12 years, ≥40 kg) caused by susceptible organisms, including Escherichia coli, Klebsiella pneumoniae, Citrobacter freundii complex, Staphylococcus saprophyticus, and Enterococcus faecalis. The safety of BLUJEPA was evaluated in 2 double-blind, active-controlled, randomized trials in female adult and pediatric patients 12 years of age and older with uUTI (Trial 1 and Trial 2). On 25 March 2025, the U.S. FDA approved Blujepa (Gepotidacin) oral tablets for patients aged 12 years and older weighing at least 45 kg, addressing an important unmet need in the management of gonorrhea. Gepotidacin is indicated for patients with limited alternative treatment options due to constrained clinical safety data. To limit the emergence of antimicrobial resistance and preserve antibacterial efficacy, BLUJEPA should be used only for infections that are confirmed or strongly suspected to be bacterial in origin.
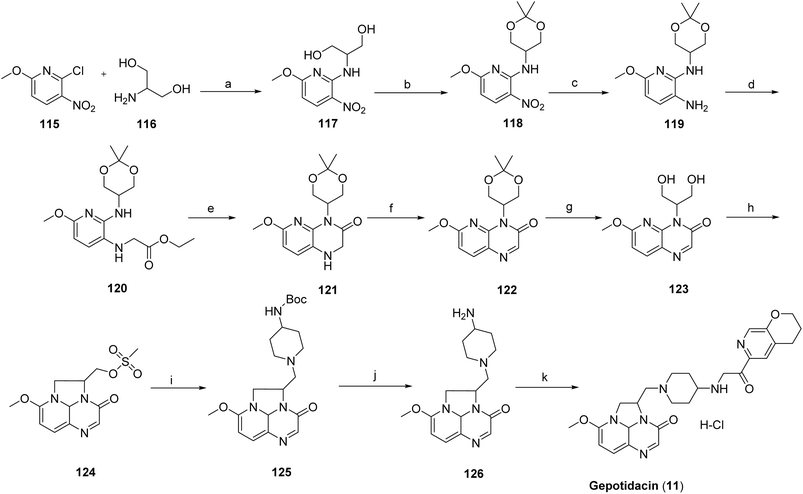
The synthesis of gepotidacin70 has been described in two patents in 2008 (ref. 71) and 2016.72 The synthesis of Gepotidacin (11) proceeds with a reaction between 2-chloro-6-methoxy-3-nitropyridine (115) and 2-aminopropane-1,3-diol (116), affording the corresponding amino-substituted diol intermediate (117). Protection of the diol as its acetal using 2,2-dimethoxypropane yielded compound 118, followed by catalytic hydrogenation of the nitro group over 10% Pd/C to furnish the aniline derivative 119. Subsequent alkylation of the aniline with ethyl bromoacetate afforded intermediate 120. Base-mediated cyclization using sodium hydride generated the bicyclic intermediate 121, which was oxidized with manganese dioxide to provide compound 122. Acidic cleavage of the acetal protecting group released the free diol 123, which upon treatment with methanesulfonic anhydride, underwent intramolecular cyclization to construct the triazaacenaphthylene core (124). Introduction of the piperidine moiety was achieved via substitution with Boc-protected aminopiperidine to afford intermediate 125. Subsequent Boc deprotection and chiral chromatographic purification yielded the enantiomerically enriched primary amine 126. Final reductive amination with the appropriate aldehyde delivered gepotidacin as the free base, which was converted to the mono-hydrochloride salt by treatment with one equivalent of 1 M HCl in diethyl ether to give gepotidacin salt (11), Scheme 19.73
Vanrafia was evaluated in a randomized, double-blind, placebo-controlled trial (ALIGN) with IgAN. Patients with IgAN and protein in the urine were randomly assigned to receive either Vanrafia or placebo once daily. The trial also included a subset of subjects on sodium–glucose cotransporter-2 inhibitors (SGLT2i) at baseline who were excluded from the primary efficacy analysis. The primary endpoint for accelerated approval was the percent reduction in urine protein at week 36 compared to baseline. This indication was granted accelerated approval based on reductions in proteinuria; however, it has not yet been established whether Vanrafia slows kidney function decline in patients with IgA nephropathy. Continued approval is contingent upon confirmation of clinical benefit in ongoing confirmatory trials. Vanrafia is contraindicated during pregnancy due to the risk of major birth defects, and pregnancy must be excluded prior to treatment initiation; effective contraception is required before, during, and for two weeks following therapy, and treatment should be discontinued if pregnancy occurs.
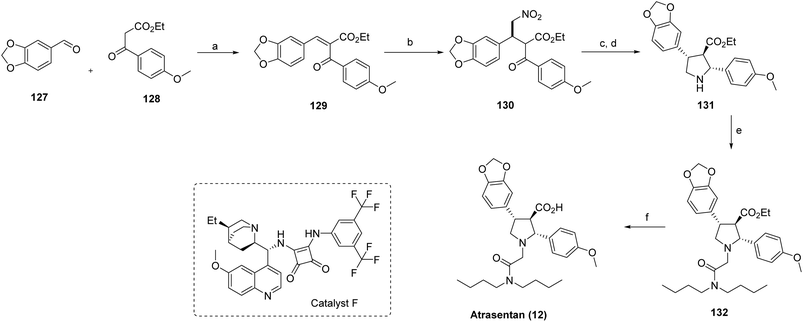
Previously, the total synthesis of atrasentan (12) had been accomplished using chiral auxiliary-mediated acylation,76 aldol reactions,77 and a hetero-Diels-Alder strategy.78 In contrast, the current synthetic approach employs a nitromethane addition product (130) as a key intermediate. As outlined in Scheme 20, intermediate 129 is first generated via condensation of compounds 127 and 128. Subsequent stereoselective addition of nitromethane under optimized conditions, using catalyst F, affords the desired adduct 130 with controlled stereochemistry. Hydrogenation of 130 using Raney® Nickel produces a transient imine intermediate, which is directly reduced with sodium cyanoborohydride to furnish pyrrolidine 131 in 84% yield over two steps. The crude reaction mixture exhibited a diastereomeric ratio of 2.6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1; purification by column chromatography provided the major diastereoisomer of 131 in 56% isolated yield. Alkylation of pyrrolidine 131 yielded intermediate 132 in 63% yield. Final saponification of 132 in ethanol/water completed the synthesis of atrasentan (12), delivering the target compound in an overall yield of 15.7% and 67% enantiomeric excess.
1; purification by column chromatography provided the major diastereoisomer of 131 in 56% isolated yield. Alkylation of pyrrolidine 131 yielded intermediate 132 in 63% yield. Final saponification of 132 in ethanol/water completed the synthesis of atrasentan (12), delivering the target compound in an overall yield of 15.7% and 67% enantiomeric excess.
Sebetralstat (13)81 was synthesized as shown in Scheme 21, via a multistep sequence involving benzyl-heterocycle functionalization and late-stage amide coupling. Nucleophilic substitution of 4-(chloromethyl)benzyl alcohol (133) with 2-hydroxypyridine afforded the benzyl-linked pyridinone 134, which was converted to the corresponding benzyl electrophile 135 via mesylation. Alkylation of 135 with methyl 3-(methoxymethyl)-1H-pyrazole-4-carboxylate furnished regioisomeric pyrazole derivatives 136a/136b. 136b was a byproduct in the reaction, and the desired regioisomer 136a was isolated. Hydrolysis of the ester functionality in 136a yielded the carboxylic acid 137a, which underwent amide coupling with a substituted aminopyridine fragment to afford the target compound sebetralstat (13).
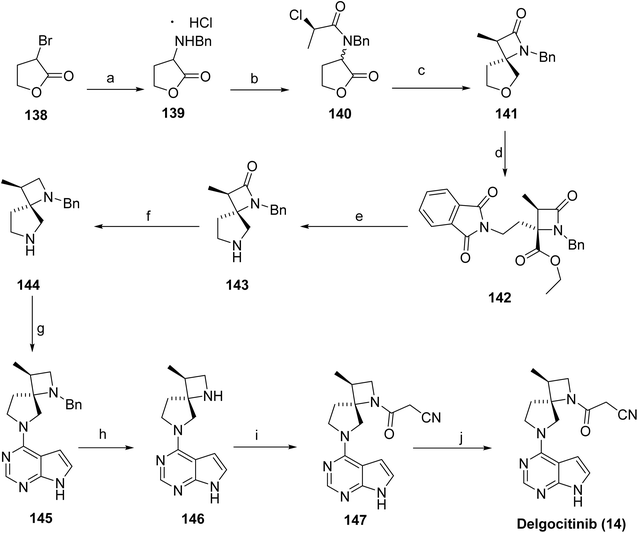
The stereocontrolled synthesis of delgocitinib (14)86 was accomplished via a modular sequence (Scheme 22). Initial SN2 coupling of bromolactone 138 with benzylamine furnished the α-amino lactone 139 as the HCl salt. Acylation of 139 with an enantiomerically pure acid chloride afforded lactone 140. Treatment of 140 with LHMDS generated the corresponding enolate, which underwent intramolecular SN2 displacement of the chloride, forming the spirocyclic lactone 141 with high stereocontrol (dr 98![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, 96% ee). The lactone ring of 141 was subsequently opened by nucleophilic attack of potassium phthalimide at the γ-carbon, and the resulting carboxylic acid was converted to the ethyl ester. Subsequent treatment with diethylenetriamine released the phthalimide, enabling cyclization to the spirolactam 142 via the ethyl ester intermediate (80% yield over four steps, >99% de). Reduction of the carbonyls in 143 with LiAlH4/AlCl3 in THF provided diamine 144, isolated as the succinic acid salt in 86% yield. Finally, SNAr reaction of 144 with chloropyrrolopyrimidine resulted in 145, followed by hydrogenolytic removal of the benzyl group, furnished amine 146 (92% yield over 2 steps). Acylation of 146 with cyanoacetyl pyrazole afforded 147 and recrystallization from n-butanol containing 3 wt% BHT provided delgocitinib (15) in 86% yield, >99% ee, and >99% de.
2, 96% ee). The lactone ring of 141 was subsequently opened by nucleophilic attack of potassium phthalimide at the γ-carbon, and the resulting carboxylic acid was converted to the ethyl ester. Subsequent treatment with diethylenetriamine released the phthalimide, enabling cyclization to the spirolactam 142 via the ethyl ester intermediate (80% yield over four steps, >99% de). Reduction of the carbonyls in 143 with LiAlH4/AlCl3 in THF provided diamine 144, isolated as the succinic acid salt in 86% yield. Finally, SNAr reaction of 144 with chloropyrrolopyrimidine resulted in 145, followed by hydrogenolytic removal of the benzyl group, furnished amine 146 (92% yield over 2 steps). Acylation of 146 with cyanoacetyl pyrazole afforded 147 and recrystallization from n-butanol containing 3 wt% BHT provided delgocitinib (15) in 86% yield, >99% ee, and >99% de.
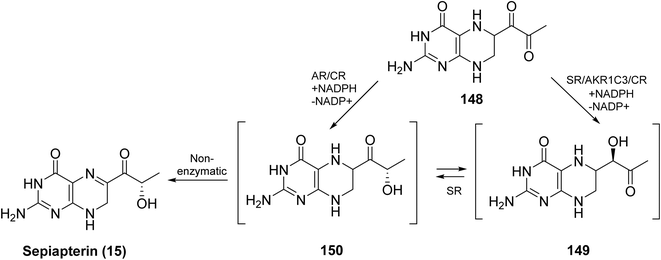
6-Pyruvoyl tetrahydropterin (148) can be converted into two intermediates: 2′-oxo-tetrahydropterin (149) and 1′-oxo-tetrahydropterin, also called lactoyl tetrahydropterin (150), with formation of 150 being more favorable (Scheme 23). 3α-Hydroxysteroid dehydrogenase type 2 efficiently converts 148 into 149, while aldose reductase (AR) can transform 148 into either 150 or 149, which can then be further processed to tetrahydrobiopterin (BH4). In the absence of sepiapterin reductase (SR), BH4 can be synthesized via two alternative routes: one through 149, involving the sequential action of 3α-hydroxysteroid dehydrogenase type 2 and AR and consuming 2 NADPH equivalents; and another through 150, proceeding through 148 → 150 →15, with the participation of AR, CR, and dihydrofolate reductase, including a nonenzymatic reduction to Sepiapterin, consuming 3 NADPH equivalents.
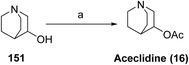
Aceclidine is synthesized as shown in Scheme 24. Subsequent O-acetylation of this secondary alcohol 3-quinuclidinol (151) using acetic anhydride in the presence of an appropriate base affords 3-acetoxyquinuclidine (16).
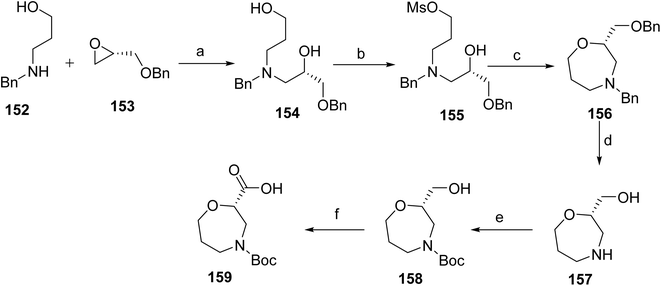
The synthesis of brensocatib (17) proceeds via the synthesis of an intermediate 159, Scheme 25. The benzylamino propanol (155) and the oxirane derivative (156) were stirred in i-PrOH to afford the corresponding intermediate (154). The reaction mixture is treated with MsCl to form the mesylate derivative (155). Subsequent treatment with NaH promoted the cyclization 156. The resulting intermediate was subjected to catalytic hydrogenation to afford 157, which was then protected using Boc2O to give 158. Finally, oxidation was carried out using Bu4NHSO4 to furnish the target intermediate (159) after standard aqueous workup and purification.
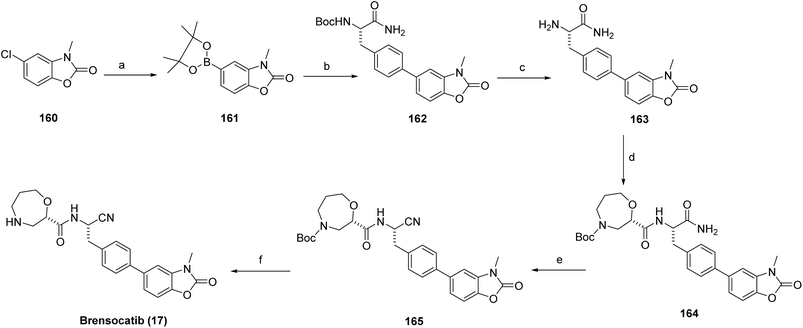
The synthesis of brensocatib (17) commenced with a palladium-catalyzed borylation of the 160 to afford the corresponding boronate intermediate (161), which was subsequently subjected to Suzuki–Miyaura cross-coupling with a protected chiral aryl amide (tert-butyl(S)-(1-amino-3-(4-iodophenyl)-1-oxopropan-2-yl)carbamate) under standard palladium catalysis to generate the biaryl intermediate (162). Acidic deprotection furnished the corresponding amine (163), which was coupled with the oxazepane carboxylic acid intermediate (159) using a peptide-coupling reagent to form the desired amide linkage (164). The amide is efficiently converted to a nitrile via T3P mediated dehydration to give 165. Final acidic treatment provided the target compound brensocatib (17), Scheme 26.
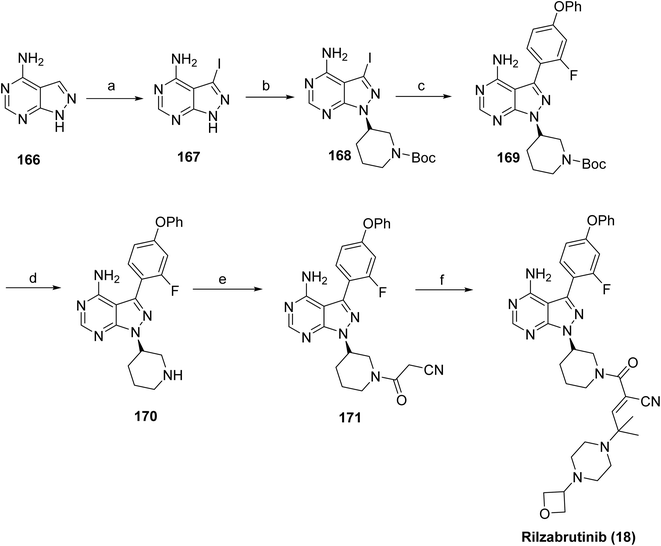
Sanofi reported a synthetic route to rilzabrutinib in 2022 (Scheme 27).93 The synthesis commenced with 4-aminopyrazolo[3,4-d]pyrimidine 166 as the starting material, which was sequentially transformed into cyanoacetamide derivative 171 via five steps: iodination (167), Mitsunobu reaction (168), Suzuki–Miyaura coupling (169), Boc deprotection (170), and amidation with cyanoacetic acid, affording a total yield of 3% of 171. The final step involved a Knoevenagel condensation between 171 and 2-methyl-2-(4-(oxetan-3-yl)piperazin-1-yl)propanal, yielding a 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 E/Z mixture, from which the Z isomer was removed by chiralpak IC HPLC purification, yielding rilzabrutinib (18). Overall, the reported route is limited by low cumulative yields (0.5% over eight steps), high cost, and labor-intensive procedures, emphasizing the need for a more efficient and economical synthetic strategy. A recent study describes an alternative approach to rilzabrutinib using an E-configured cyanoacrylic acid intermediate.96
1 E/Z mixture, from which the Z isomer was removed by chiralpak IC HPLC purification, yielding rilzabrutinib (18). Overall, the reported route is limited by low cumulative yields (0.5% over eight steps), high cost, and labor-intensive procedures, emphasizing the need for a more efficient and economical synthetic strategy. A recent study describes an alternative approach to rilzabrutinib using an E-configured cyanoacrylic acid intermediate.96
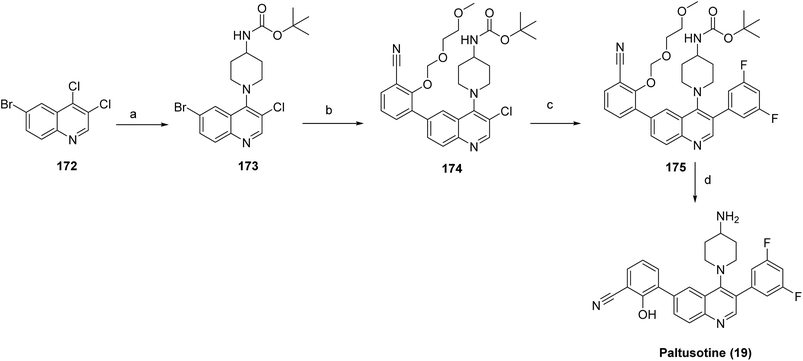
The target compound paltusotine (19)99 was synthesized through a modular, stepwise cross-coupling strategy (Scheme 28). Nucleophilic aromatic substitution of 6-bromo-3,4-dichloroquinoline (172) with tert-butyl piperidin-4-ylcarbamate provided the corresponding quinoline–piperidine intermediate (173). Suzuki–Miyaura cross-coupling of 173 with a protected 2-(2-methoxy-ethoxymethoxy)-3-boronate benzonitrile generated the aryl-substituted intermediate 174, which underwent a second Suzuki coupling with 3,5-difluorophenylboronic acid to furnish the fully substituted quinoline derivative 175. Finally, deprotection of the tert-butyl carbamate under acidic conditions followed by isolation as the hydrochloride salt afforded the final target compound paltusotine (19) in high purity.
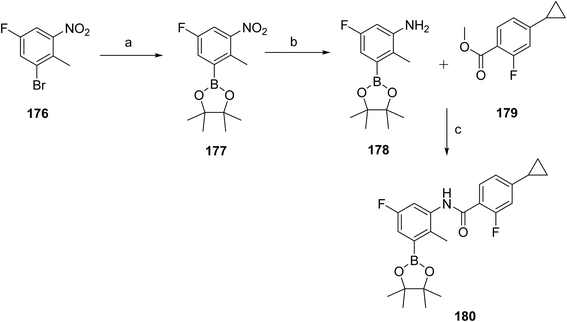
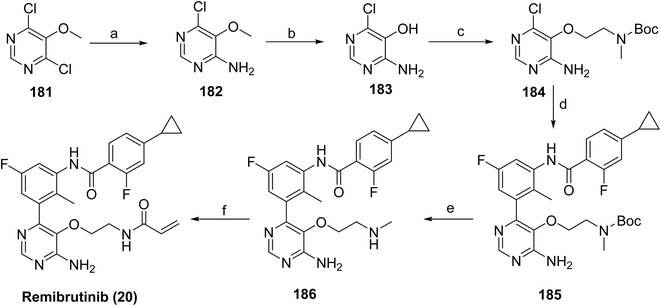
The synthesis of remibrutinib (20)103 is outlined in Schemes 29 and 30 where Scheme 29 depicts the synthesis of the upper portion of the molecule, intermediate 180. Borylation of 1-bromo-5-fluoro-2-methyl-3-nitrobenzene (176) with bis(pinacolato)diboron in the presence of Pd(dppf)Cl2·DCM and potassium acetate gave boronic ester 177, which upon catalytic hydrogenation provided aniline 178. Boronic ester 181 was then accessed via a NaHMDS mediated amide formation of aniline 178 and cyclopropyl intermediate 179. The synthesis of 20 was completed as outlined in Scheme 30. The lower portion of the molecule was assembled starting from 2,4-dichloro-3-methoxypyrimidine (181). Treatment with ammonia in a pressure reactor yielded in aminopyrimidine 182, which was subjected to boron tribromide treatment to give aminopyrimidinol 183. The linker moiety was introduced via a Mitsunobu reaction with N-Boc-N-methyl-2-hydroxyethylamine in the presence of DIAD and Smopex-301, a convenient polymer-supported version of triphenylphosphine, to obtain 184. Boc-protected intermediate 185 was assembled via Suzuki coupling of 184 and 180. Finally, Boc deprotected 186 was followed by amide coupling with acrylic acid in the presence of propylphosphonic anhydride (T3P) provided remibrutinib (20).
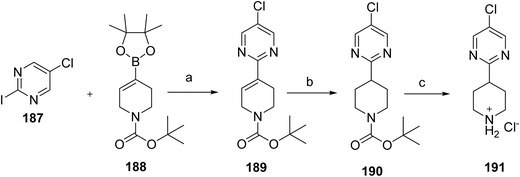
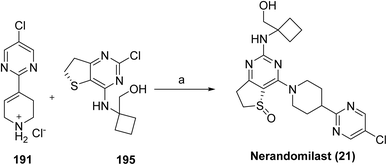
The synthesis for nerandomilast (21) proceeds via coupling of intermediates synthesized from Schemes 31 and 32 shown in Scheme 33.106 Intermediate 191 is synthesized via a key Suzuki coupling using commercially available iodo-pyrimidine 187 and boronic ester 188 with 0.05 mol% PdCl2(AmPhos)2 as the catalyst, upon completion, N-acetylcysteine was added (to reduce residual Pd in the product), and the product was crystallized by adding water as the antisolvent to give 189. Subsequently, 189 was reduced by a Sponge (Raney) Nickel catalyzed (10 mol%) hydrogenation (4.00 bar H2 pressure), which gave 190. Subsequent cleavage of the BOC protecting group in 190 was accomplished, followed by crystallization from MTBE to its hydrochloride salt (191).
 | ||
| Scheme 33 Synthesis of nerandomilast (21). aReaction conditions: (a) DIPEA, THF, H2O, 65 °C for 14 h. | ||
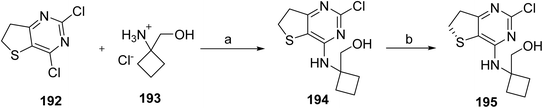
The synthesis of chiral sulfoxide intermediate 195 was carried out as shown in Scheme 32. The synthesis of 195 was accomplished by carrying out an SNAr displacement of the chlorine at the C4 of 192 with commercially available 1-aminocyclobanemethanol hydrochloride (193). This transformation was not completely regioselective, and approximately 10% of the product resulted from C2-chlorine the displacement was also observed. However, this by-product was completely removed during the isolation of 194. Intermediate 194 was then recrystallized from ethanol to remove traces of triethylamine or other impurities that could affect the subsequent asymmetric oxidation to chiral sulfoxide 195, which was carried out using a modification of the procedure reported by Uemura and co-workers107 for the general asymmetric oxidation of sulfides. Accordingly, oxidation of 194 in the presence of a catalyst made from (S)-BINOL (2 mol%) and (i-PrO)4Ti (1 mol%), and with tert-BuOOH as the stoichiometric oxidant, sulfoxide 195 in good yield (87.4%) and enantioselectivity (>99.0 %ee).
With robust and scalable processes for the synthesis of intermediates 191 and 195 at hand, the final chemical step in the synthesis, which was carried out as shown in Scheme 33 by the SNAr reaction between 191 and 195 in the presence of Hünig's base and a solvent system consisting of a mixture of THF and water. Under these conditions, nerandomilast (21) was obtained with good yield (>88%) and purity (>99%) as a product.
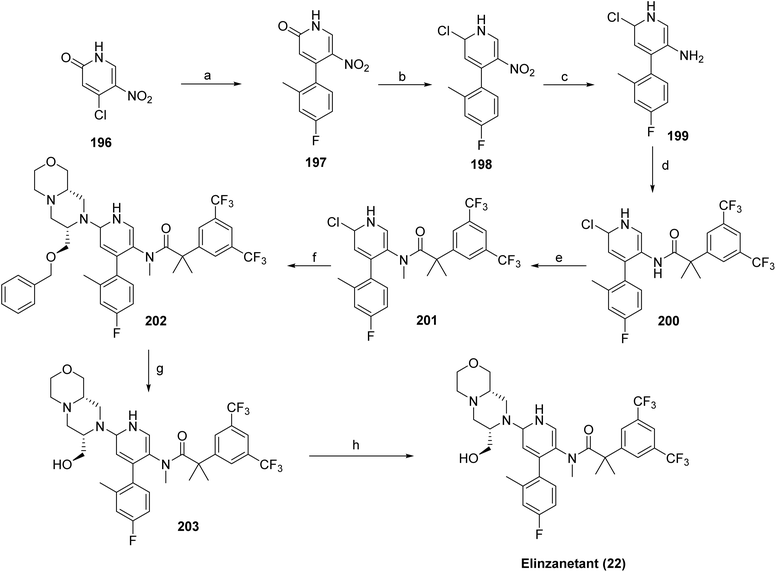
The synthesis of elinzanetant (22) shown in Scheme 34, proceeds with a palladium-catalyzed cross-coupling of the 196 and arylboronic acid under basic conditions in 1,4-dioxane to afford the coupled intermediate (197). Subsequent formylation using POCl3/DMF generated the corresponding chlorine derivative (198) where the nitro group was then reduced over Pt/C in ethyl acetate to yield the amine derivative (199). Amide intermediate (200) was generated by acid and amine (199) coupling. Later methylation was achieved under basic conditions in DMF to introduce the desired methyl group, followed by a palladium-catalyzed coupling of oxazine derivative to afford intermediate 202. Final hydrogenation over Pd/C in isopropanol under acidic conditions caused the selective hydrogenolytic cleavage of the benzyl ether protecting group (203), and the crude product was purified by basic workup with NaOH in MTBE/IPA to afford the target compound elinzanetant (22).
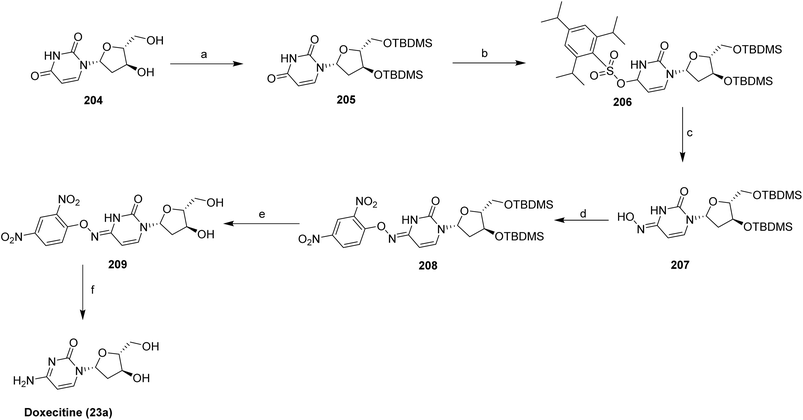
The synthesis of doxecitine (23a) and related compounds has been reported in the literature.112,113 As shown in Scheme 35, doxecitine (23a) was synthesized via a sequential protection, activation, and substitution strategy starting from the nucleoside deoxyuridine (204). Initial silylation of the ribose hydroxyl groups with TBDMSCl in DMF/pyridine in the presence of imidazole afforded intermediate 205, which was then converted to sulfonate 206 by treatment with NaH and 2,4,6-triisopropylbenzenesulfonyl chloride in THF. Displacement of the sulfonate with hydroxylamine hydrochloride under DBU-mediated conditions yielded intermediate 207, which was then subjected to nucleophilic aromatic substitution with 1-fluoro-2,4-dinitrobenzene to produce compound 208. Subsequent removal of the silyl protecting groups using triethylamine trihydrofluoride in THF afforded diol 209, and the final transformation under mild DMSO/MeOH conditions at near-neutral pH provided the target compound doxecitine (23a).
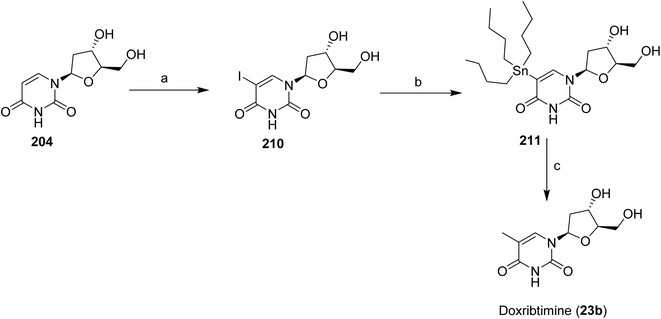
Similarly, compound doxribtimine (23b)114,115 was synthesized from nucleoside 204 via halogenation, an organometallic transformation, and a final coupling reaction to afford the desired product (Scheme 35). Treatment of 204 with iodine and silver nitrate in MeOH at 40 °C afforded the iodinated intermediate 210, which was then converted to the organotin derivative 211 by reaction with bis(tributyltin) in the presence of tetrakis(triphenylphosphine)palladium in DMF. Final conversion of 211 using methyl iodide, tris(dibenzylideneacetone)dipalladium, tri(o-tolyl)phosphine, copper bromide, and cesium fluoride in DMF under controlled heating conditions afforded the target compound doxribtimine (23b) (Scheme 36).
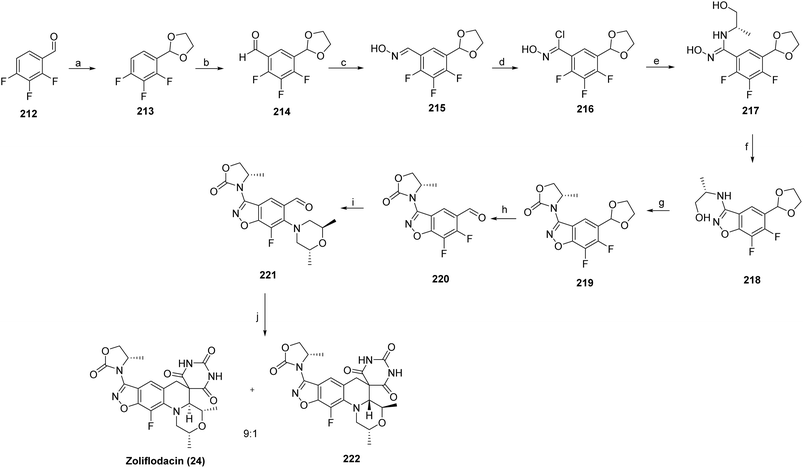
The synthesis was initiated by protecting the starting material (212) with ethylene glycol in the presence of p-TsOH in refluxing toluene to afford the corresponding acetal (213) lithiation with n-BuLi in THF at −70 °C, followed by DMF quenching, provided the formylated intermediate (214), which was subsequently converted to oxime using hydroxylamine in ethanol (215). Treatment with NCS in DMF furnished the chlorinated derivative (216), which was then reacted with D-alaninol in DMF to yield the corresponding amine intermediate (217). Further transformation with Cs2CO3 at room temperature afforded the coupled product (218), which was activated using CDI and DIEA in DMF at 70 °C (219). Acidic deprotection with HCl in THF/water gave the key intermediate, (220) which was then reacted with (2R,6R)-2,6-dimethylmorpholine in the presence of K2CO3 or DIEA in CH3CN/water at 80 °C (221). Finally, condensation with pyrimidine-2,4,6 (1H,3H,5H)-trione in AcOH/water at 120 °C afforded a 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture of diastereomers, from which the major isomer was separated to furnish the target compound zoliflodacin (24) (Scheme 37).
1 mixture of diastereomers, from which the major isomer was separated to furnish the target compound zoliflodacin (24) (Scheme 37).
The synthesis of aficamten (25)123 was accomplished via a multistep route (Scheme 38). Reductive amination of 5-bromo-1-indanone (223) with ammonium formate and sodium cyanoborohydride furnished the corresponding amine (224), which was protected as the tert-butyloxycarbonyl (Boc) derivative. The resulting aryl bromide (225) underwent palladium-mediated cyanation using potassium ferrocyanide to afford the corresponding nitrile (226). Treatment of this nitrile with hydroxylamine generated a hydroxycarbamimidoyl intermediate (22), which is cyclized with propionyl chloride in the presence of pyridine to give the oxadiazole derivative. Finally, Boc deprotection of the amide followed by amide coupling with methyl pyrazole carboxylic acid furnished the target compound, aficamten (25).
Tradipitant was synthesized as shown in Scheme 39. The synthesis commences with a nucleophilic aromatic substitution of 2-chloropyridine with thiophenol (228) under basic conditions, affording the corresponding pyridyl sulfide (229). Subsequent oxidation of the sulfide furnishes the sulfone intermediate, 2-(benzenesulfonyl)pyridine (230). Directed lithiation of sulfone 230 using n-butyllithium in the presence of diisopropylamine, followed by electrophilic trapping with 2-chlorobenzaldehyde, yields ketone intermediate 231. This intermediate undergoes condensation with the enolate derived from 4-acetylpyridine, generated in situ with potassium tert-butoxide in DMSO. The resulting adduct is subjected to base-mediated cyclization in the presence of lithium hydroxide and benzoic acid to produce the pyridine benzoate intermediate 232. Finally, nucleophilic substitution with 3,5-bis(trifluoromethyl)benzyl azide affords the target molecule, tradipitant (26).
To provide a comparative overview of pharmacological profiles, Table 1 summarizes representative activity data (IC50/EC50 or equivalent potency values) for selected FDA-approved drugs against their primary biological targets. It should be noted that standardized IC50 and CC50 values are not consistently reported, as pharmacological evaluations often involve different assay systems and endpoints. Therefore, representative potency data have been included where available to ensure accuracy and consistency, offering a clear overview of their pharmacological characteristics.
| S. no | Brand name | Active ingredient | Activity and target | References |
|---|---|---|---|---|
| 1 | Romvimza™ | Vimseltinib | IC50: <0.01 µM (c-FMS/CSF-IR), 0.1–1 µM (c-Kit) | 127 |
| 2 | Avmapki Fakzynja Co-Pack™ | Avutometinib and defactinib | Avutometinib: (IC50): MEK 160 nM; BRafV600E 8.2 nM; Braf 190 nM; CRAF 56 nM | 128 |
| Defactinib: FAK (IC50): 0.6 nM | ||||
| 3 | Ibtrozi™ | Taletrectinib | IC50: (1–1000 nM; 72 hours) against Ba/F3-TPM3-NTRK1, Ba/F3-ETV6-NTRK1, –NTRK2, –NTRK3, or KM12 cells is ∼3–20 nM | 129 |
| 4 | Zegfrovy™ | Sunvozertinib | IC50: EGFR exon 20 insertion: 20.4 nM | 130 |
| EGFRL858R/T790M: 1.1 nM | ||||
| Her2 Exon20 YVMA: 7.5 nM | ||||
| 5 | Modeyso™ | Dordaviprone | hERG IC50 is reported as 2.4 µM | 131 |
| 6 | Hernexeos™ | Zongertinib | IC50: HER2 (YVMA) 16 nM; EGFR wild type 1540 nM | 132 |
| 7 | Inluriyo™ | Imlunestrant | IC50: MCF7: 17 nM | 133 |
| 8 | Komzifti™ | Ziftomenib | IC50: (4–31 nM) for KMT2A-rearranged and NPM1-mutant acute myeloid leukemia (AML) cell lines | 59 |
| 9 | Hyrnuo™ | Sevabertinib | IC50: <0.5 nM for wild-type HER2, HER2 A775insYVMA, wild-type EGFR, and EGFR D770_N771insSVD. | 62 |
| 10 | Journavx™ | Suzetrigine | Highly selective NaV1.8 inhibitor | 65 |
| 11 | Blujepa™ | Gepotidacin | The MIC50 and MIC90 for gepotidacin against the 25 N. gonorrhoeae isolates tested are 0.12 and 0.25 µg mL−1, respectively | 134 |
| 12 | Vanrafia™ | Atrasentan | IC50: 0.055 nM (ETA) | 135 |
| 13 | Ekterly™ | Sebetralstat | IC50: 6.0 nM plasma kallikrein | 81 |
| 14 | Anzupgo™ | Delgocitinib | IC50: JAK2:2.6 nM; JAK1:2.8 nM; JAK3:13 nM; Tyk2: 58 nM | 136 |
| 15 | Sephience™ | Sepiapterin | Human endogenous metabolite; BH4 precursor (eNOS cofactor) | 87 |
| 16 | Vizz™ | Aceclidine | Aceclidine is a modulator of M3 muscarinic acetylcholine receptor and a M1 receptor agonist (EC50: 40 µM) | 137 |
| 17 | Brinsupri™ | Brensocatib | DPP1 inhibitor with pIC50s of 6.85, 7.6, 7.7, 7.8, and 7.8 in human, mouse, rat, dog and rabbit, respectively | 138 |
| 18 | Wayrilz™ | Rilzabrutinib | IC50: BTK: 1.3 nM; BMX: 1.0 nM; ITK: 440 nM; TEC:0.8 nM etc. | 138 and 139 |
| 19 | Palsonify™ | Paltusotine | Human SST2 EC50: 0.25 nM | 99 |
| Rat SST2 EC50: 1.2 nM | ||||
| 20 | Rhapsido™ | Remibrutinib | IC50: 1 nM (BTK) | 103 |
| 21 | Jascayd™ | Nerandomilast | PDE4B IC50: 7.2 nM | 140 |
| 22 | Lynkuet™ | Elinzanetant | Orally active and selective NK-1 and NK-3 receptor antagonist | 108 |
| 23 | Kygevvi™ | Doxecitine and Doxribtimine | Nucleoside therapy; restores mtDNA in TK2 deficiency | 141 |
| 24 | Nuzolvence™ | Zoliflodacin | S. aureus with the MIC90 of 0.25 µg mL−1; HepG2: IC50: > 50 µM | 142 |
| 25 | Myqorzo™ | Aficamten | IC50 of 1.4 µM | 123 |
| 26 | Nereus™ | Tradipitant | NK1 receptor antagonist; inhibits substance P-mediated signaling | 125 |
2.3 Highlights of FDA-approved drugs in 2025
In Table 2, the key highlights of FDA-approved drugs in 2025 are summarized, including their active pharmaceutical ingredients, therapeutic indications, recommended dosages, and approval dates as reported by the U.S. FDA. The table emphasizes the structural diversity of nitrogen-containing motifs present in these agents and illustrates their contributions to target selectivity and overall therapeutic performance, underscoring the importance of nitrogen functionalities in contemporary drug discovery and design.| S. no | Drug name | Active ingredient | FDA-approved use, dosage, and administration on approval date | FDA approval date |
|---|---|---|---|---|
| I. Anticancer drugs | ||||
| 1 | Romvimza™ | Vimseltinib | Use: for the treatment of symptomatic TGCT for which surgical resection will potentially cause worsening functional limitation or severe morbidity | 2/14/2025 |
| Recommended dosage and administration: 30 mg orally taken twice weekly, with a minimum of 72 hours between doses | ||||
| 2 | Avmapki Fakzynja Co-Pack™ | Avutometinib and defactinib | Use: for the treatment of KRAS-mutated recurrent low-grade serous ovarian cancer (LGSOC) after prior systemic therapy | 5/8/2025 |
| Recommended dosage and administration: AVMAPKI 3.2 mg administered orally twice weekly (day 1 and day 4) for the first 3 weeks of each 4 weeks cycle | ||||
| FAKZYNJA 200 mg administered orally twice daily for the first 3 weeks of each 4 weeks cycle | ||||
| 3 | Ibtrozi™ | Taletrectinib | Use: for the treatment of locally advanced or metastatic ROS1-positive non-small cell lung cancer | 6/11/2025 |
| Recommended dosage and administration: 600 mg orally once daily on an empty stomach (no food intake at least 2 hours before and 2 hours after taking IBTROZI | ||||
| 4 | Zegfrovy™ | Sunvozertinib | Use: for the treatment of locally advanced or metastatic non-small cell lung cancer with epidermal growth factor receptor exon 20 insertion mutations, as detected by an FDA-approved test, with disease progression on or after platinum-based chemotherapy | 7/2/2025 |
| Recommended dosage and administration: 200 mg orally once daily taken with food | ||||
| 5 | Modeyso™ | Dordaviprone | Use: to treat diffuse midline glioma harboring an H3 K27M mutation with progressive disease following prior therapy | 8/6/2025 |
| Recommended dosage and administration: in pediatric patients weighing ≥10 kg is based on body weight | ||||
| Taken orally once weekly on an empty stomach (at least 1 hour before or 3 hours after food intake) | ||||
| 6 | Hernexeos™ | Zongertinib | Use: to treat adults with unresectable or metastatic non-squamous non-small cell lung cancer whose tumors have HER2 tyrosine kinase domain activating mutations, as detected by an FDA-approved test, and who have received prior systemic therapy | 8/8/2025 |
Recommended dosage and administration: it is based on body weight < 90 kg![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 120 mg; ≥ 90 kg 120 mg; ≥ 90 kg![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 180 mg. Orally once daily with or without food until disease progression or unacceptable toxicity 180 mg. Orally once daily with or without food until disease progression or unacceptable toxicity |
||||
| 7 | Inluriyo™ | Imlunestrant | Use: for the treatment of estrogen receptor-positive, human epidermal growth factor receptor 2-negative, estrogen receptor-1-mutated advanced or metastatic breast cancer with disease progression following at least one line of endocrine therapy | 9/25/2025 |
| Recommended dosage and administration: 400 mg orally once daily, on an empty stomach | ||||
| 8 | Komzifti™ | Ziftomenib | Use: for the treatment of adults with relapsed or refractory acute myeloid leukemia with a susceptible nucleophosmin 1 mutation who have no satisfactory alternative treatment options | 11/13/2025 |
| Recommended dosage and administration: 600 mg taken orally once daily until disease progression or unacceptable toxicity | ||||
| 9 | Hyrnuo™ | Sevabertinib | Use: for the treatment of locally advanced or metastatic non-squamous non-small cell lung cancer with tumors that have activating HER2 tyrosine kinase domain activating mutations in patients who received a systemic therapy | 11/19/2025 |
| Recommended dosage and administration: 20 mg orally twice daily with food until disease progression or unacceptable toxicity | ||||
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||
| II. Non-anticancerous drugs | ||||
| 10 | Journavx™ | Suzetrigine | Use: for the treatment of moderate to severe acute pain | 1/30/2025 |
| Recommended dosage and administration: it is 100 mg. Take the starting dose on an empty stomach at least 1 hour before or 2 hours after food | ||||
| 11 | Blujepa™ | Gepotidacin | Use: to treat uncomplicated urinary tract infections | 3/25/2025 |
| Recommended dosage and administration: 1500 mg (two 750 mg tablets) orally twice daily after meals, approximately 12 hours apart, for 5 days | ||||
| 12 | Vanrafia™ | Atrasentan | Use: to reduce proteinuria in adults with primary immunoglobulin A nephropathy at risk of rapid disease progression | 4/02/2025 |
| Recommended dosage and administration: 0.75 mg orally once daily with or without food | ||||
| 13 | Ekterly™ | Sebetralstat | Use: to treat acute attacks of hereditary angioedema | 7/3/2025 |
| Recommended dosage and administration: one dose of 600 mg (2 tablets) taken orally at the earliest recognition of an HAE attack | ||||
| 14 | Anzupgo™ | Delgocitinib | Use: to treat moderate-to-severe chronic hand eczema when topical corticosteroids are not advisable or produce an inadequate response | 7/23/2025 |
| Recommended dosage and administration: apply to affected areas of the hands and wrists twice daily. Not more than 30 g per 2 weeks or 60 g per month. For topical use only | ||||
| 15 | Sephience™ | Sepiapterin | Use: to treat hyperphenylalaninemia in patients with sepiapterin-responsive phenylketonuria, in conjunction with a phenylalanine-restricted diet. | 7/28/2025 |
| Recommended dosage and administration: it is taken orally once daily with food. The starting dosage is | ||||
| Less than 6 months 7.5 mg kg−1 | ||||
| 6 months to less than 1 year 15 mg kg−1 | ||||
| 1 year to less than 2 years 30 mg kg−1 | ||||
| 2 years and older 60 mg kg−1 | ||||
| 16 | Vizz™ | Aceclidine | Use: to treat presbyopia | 7/31/2025 |
| Recommended dosage and administration: instill one drop in each eye, wait 2 minutes, and instill a second drop in each eye once daily | ||||
| 17 | Brinsupri™ | Brensocatib | Use: to treat non-cystic fibrosis bronchiectasis | 8/12/2025 |
| Recommended dosage and administration: 10 mg or 25 mg orally once daily with or without food | ||||
| 18 | Wayrilz™ | Rilzabrutinib | Use: to treat persistent or chronic immune thrombocytopenia that has not sufficiently responded to immunoglobulins, anti-D therapy, or corticosteroids | 8/29/2025 |
| Recommended dosage and administration: 400 mg orally twice daily; swallow whole with water, with or without food. Do not cut, crush, or chew tablets | ||||
| 19 | Palsonify™ | Paltusotine | Use: to treat acromegaly in adults who had an inadequate response to surgery and/or for whom surgery is not an option | 9/25/2025 |
| Recommended dosage and administration: orally once daily with water on an empty stomach (at least 6 hours after a meal) and at least 1 hour before the next meal. Initial dose of 40 mg once daily; may reduce to 20 mg temporarily for tolerability, then resume 40 mg | ||||
| 20 | Rhapsido™ | Remibrutinib | Use: to treat chronic spontaneous urticaria in adults who remain symptomatic despite H1 antihistamine treatment | 9/30/2025 |
| Recommended dosage and administration: it is taken 25 mg orally twice daily with or without food | ||||
| Swallow tablets whole. Do not split, crush, or chew | ||||
| 21 | Jascayd™ | Nerandomilast | Use: to treat idiopathic pulmonary fibrosis | 10/7/2025 |
| Recommended dosage and administration: 18 mg orally twice daily approximately 12 hours apart with or without food | ||||
| 22 | Lynkuet™ | Elinzanetant | Use: to treat moderate-to-severe vasomotor symptoms due to menopause | 10/24/2025 |
| Recommended dosage and administration: 120 mg (two 60 mg capsules) orally once daily at bedtime with or without food. Swallow tablets whole. Do not split, crush, or chew | ||||
| 23 | Kygevvi™ | Doxecitine and doxribtimine | Use: to treat thymidine kinase 2 deficiency in patients who start to show symptoms when they are 12 years old or younger | 11/3/2025 |
| Recommended dosage and administration: starting 260 mg kg−1 day−1 (consisting of 130 mg doxecitine and 130 mg doxribtimine), intermediate 520 mg kg−1 day−1 (consisting of 260 mg doxecitine and 260 mg doxribtimine), maintenance 800 mg kg−1 day−1 (consisting of 400 mg doxecitine and 400 mg doxribtimine) | ||||
| 24 | Nuzolvence™ | Zoliflodacin | Use: to treat uncomplicated urogenital gonorrhea due to Neisseria gonorrhoeae | 12/12/2025 |
| Recommended dosage and administration: adults and pediatric patients 12 years of age and older, weighing at least 35 kg: recommended dose is 3 g (one packet) administered as a single dose orally | ||||
| 25 | Myqorzo™ | Aficamten | Use: to treat symptomatic obstructive hypertrophic cardiomyopathy | 12/19/2025 |
| Recommended dosage and administration: starting dose is 5 mg orally once daily | ||||
| 26 | Nereus™ | Tradipitant | Use: to treat vomiting associated with motion | 12/30/2025 |
| Recommended dosage and administration: 5 mg or 170 mg orally, single dose, ∼60 minutes before motion, on an empty stomach | ||||
3. Conclusion
The analysis of FDA-approved drugs in 2025 shows a strong dominance of nitrogen-containing heterocyclic small molecules, reaffirming their central role in modern medicinal chemistry. Of the 46 drugs approved in 2025, 26 (∼56.5%) are nitrogen-containing small molecules, the largest chemical category, compared with monoclonal antibodies/biologics (14; ∼30.4%), non-nitrogen small molecules (3; ∼6.5%), and oligonucleotide/ASO/siRNA therapeutics (3; ∼6.5%). This trend underscores the sustained preference for nitrogen-rich scaffolds in the design of clinically effective and drug-like molecules.A defining structural characteristic of these nitrogen-containing drugs is the broad use of heterocyclic frameworks, including pyridines, pyrimidines, imidazoles, triazoles, indoles, and fused polycyclic nitrogen systems, which enable precise tuning of electronic properties, hydrogen-bonding potential, solubility, metabolic stability, and receptor-binding affinity. In particular, oncology-focused approvals, such as Vimseltinib (1), Taletrectinib (3), Sunvozertinib (4), Dordaviprone (5), Zongertinib (6), Ziftomenib (8), and Sevabertinib (9), illustrate the strategic use of nitrogen heterocycles for kinase inhibition, mutation-selective targeting, and enhanced therapeutic precision. Similarly, CNS- and rare-disease-directed agents, including Sepiapterin (15), Doxecitine/Doxribtimine (23), and Tradipitant (26), highlight the role of nitrogen-centered scaffolds in improving brain penetration, pharmacokinetics, and pathway-specific modulation.
Beyond oncology, nitrogen-containing frameworks underpin advances in anti-infective, immunological, metabolic, cardiovascular, and pain-related therapies, exemplified by Gepotidacin (11), Zoliflodacin (24), Remibrutinib (20), Nerandomilast (21), Paltusotine (19), Aficamten (25), and Suzetrigine (10). A notable feature of these molecules is their increased stereochemical complexity and structural refinement, reflecting regulatory trends that prioritize enantioselectivity, safety, and target specificity, particularly in kinase inhibitors, immune modulators, and CNS-active drugs.
From a medicinal chemistry and synthetic perspective, the 2025 FDA approvals underscore the growing reliance on nitrogen heterocycles as privileged scaffolds in modern drug design. Advances in heterocycle-focused synthetic methodologies, including late-stage functionalization, modular cross-coupling, asymmetric synthesis, and scalable green chemistry, have enabled efficient construction of structurally complex and stereochemically rich nitrogen frameworks. Collectively, these trends underscore the indispensable role of N-heterocycles in modulating biological activity, optimizing pharmacokinetic profiles, and enhancing target engagement. As synthetic innovation and structure-guided design continue to evolve, nitrogen-containing scaffolds are poised to remain key drivers of next-generation therapeutic discovery.
Conflicts of interest
There are no conflicts of interest to declare.Data availability
No primary research results, software, or code have been included, and no new data were generated or analyzed as part of this review.References
- N. Bansal, A. Dadwal and B. Kumar, Future Med. Chem., 2025, 17, 2657–2659 CrossRef CAS PubMed.
- T. Biswas, R. K. Mittal, V. Sharma, Kanupriya and I. Mishra, Med. Chem., 2024, 20, 369–384 CrossRef CAS PubMed.
- E. Vitaku, D. T. Smith and J. T. Njardarson, J. Med. Chem., 2014, 57, 10257–10274 CrossRef CAS PubMed.
- O. Ebenezer, M. A. Jordaan, G. Carena, T. Bono, M. Shapi and J. A. Tuszynski, Int. J. Mol. Sci., 2022, 23, 8117 CrossRef CAS PubMed.
- A. Mermer, T. Keles and Y. Sirin, Bioorg. Chem., 2021, 114, 105076 CrossRef CAS PubMed.
- A. Amin, T. Qadir, P. K. Sharma, I. Jeelani and H. Abe, Open Med. Chem. J., 2022, 16, e187410452202280 CrossRef.
- J. Liu, J. Jiang, L. Zheng and Z.-Q. Liu, Adv. Synth. Catal., 2020, 362, 4876–4895 CrossRef CAS.
- J. Jampilek, Molecules, 2019, 24, 3839 CrossRef CAS PubMed.
- X. Li, L. He, H. Chen, W. Wu and H. Jiang, J. Org. Chem., 2013, 78, 3636–3646 CrossRef CAS PubMed.
- C. M. M. Santos, M. Freitas and E. Fernandes, Eur. J. Med. Chem., 2018, 157, 1460–1479 CrossRef CAS PubMed.
- P. N. Kalaria, S. C. Karad and D. K. Raval, Eur. J. Med. Chem., 2018, 158, 917–936 CrossRef CAS PubMed.
- N. Kerru, L. Gummidi, S. Maddila, K. K. Gangu and S. B. Jonnalagadda, Molecules, 2020, 25, 1909 CrossRef CAS PubMed.
- G. L. Khatik, A. K. Datusalia, W. Ahsan, P. Kaur, M. Vyas, A. Mittal and S. K. Nayak, Curr. Drug Discov. Technol., 2018, 15, 163–177 CrossRef CAS PubMed.
- V. G. Reddy, T. S. Reddy, C. Jadala, M. S. Reddy, F. Sultana, R. Akunuri, S. K. Bhargava, D. Wlodkowic, P. Srihari and A. Kamal, Eur. J. Med. Chem., 2019, 182, 111609 CrossRef PubMed.
- S. Mishra, C. Jadala, S. Cardoza, G. R. Potuganti and G. R. Velma, RSC Adv., 2026, 16, 13830–13850 RSC.
- A. Kumar, A. K. Singh, H. Singh, V. Vijayan, D. Kumar, J. Naik, S. Thareja, J. P. Yadav, P. Pathak, M. Grishina, A. Verma, H. Khalilullah, M. Jaremko, A.-H. Emwas and P. Kumar, Pharmaceuticals, 2023, 16, 299 CrossRef CAS PubMed.
- R. V. Patel, Y.-S. Keum and S. W. Park, Eur. J. Med. Chem., 2015, 97, 649–663 CrossRef CAS PubMed.
- A. K. Dhingra, B. Chopra, J. S. Dua and D. N. Prasad, Anti-Inflammatory Anti-Allergy Agents Med. Chem., 2017, 16, 136–152 CrossRef CAS PubMed.
- G. Grover, R. Nath, R. Bhatia and M. J. Akhtar, Bioorg. Med. Chem., 2020, 28, 115585 CrossRef CAS PubMed.
- V. Antoci, D. Cucu, G. Zbancioc, C. Moldoveanu, V. Mangalagiu, D. Amariucai-Mantu, A. Aricu and I. I. Mangalagiu, Future Med. Chem., 2020, 12, 207–222 CrossRef CAS PubMed.
- D. Choudhary, B. Kumar and R. Kaur, Chem. Biol. Drug Des., 2024, 103, e14479 CrossRef CAS PubMed.
- R. Kharb, P. C. Sharma and M. S. Yar, J. Enzyme Inhib. Med. Chem., 2011, 26, 1–21 CrossRef CAS PubMed.
- Y. Yuan, M. Li, V. Apostolopoulos, J. Matsoukas, W. M. Wolf, M. A. T. Blaskovich, J. Bojarska and Z. M. Ziora, Eur. J. Med. Chem., 2024, 279, 116870 CrossRef CAS PubMed.
- S. S. Alghamdi, R. S. Suliman, K. Almutairi, K. Kahtani and D. Aljatli, Drug Des., Dev. Ther., 2021, 15, 3289–3312 CrossRef PubMed.
- R. Kumar, A. B. Marianesan and S. Pathak, Curr. Top. Med. Chem., 2024, 24, 1504–1528 CrossRef PubMed.
- M. Albratty and H. A. Alhazmi, Arab. J. Chem., 2022, 15, 103846 CrossRef CAS.
- P. R. Adiyala, K. N. V. Sastry, J. Kovvuri, A. Nagarajan, V. G. Reddy, I. B. Sayeed, V. L. Nayak, R. A. Maurya and A. Kamal, ChemistrySelect, 2017, 2, 8158–8161 CrossRef CAS.
- https://www.fda.gov/drugs/novel-drug-approvals-fda/novel-drug-approvals-2025.
- B. G. de la Torre and F. Albericio, Molecules, 2026, 31, 419 CrossRef CAS PubMed.
- S. Mishra, C. Jadala, S. Cardoza, G. R. Potuganti and G. R. Velma, RSC Adv., 2026, 16, 13830–13850 RSC.
- D. C. Blakemore, L. Castro, I. Churcher, D. C. Rees, A. W. Thomas, D. M. Wilson and A. Wood, Nat. Chem., 2018, 10, 383–394 CrossRef CAS PubMed.
- R. D. Taylor, M. MacCoss and A. D. G. Lawson, J. Med. Chem., 2014, 57, 5845–5859 CrossRef CAS PubMed.
- N. A. Meanwell, J. Med. Chem., 2011, 54, 2529–2591 CrossRef CAS PubMed.
- D. G. Brown and J. Boström, J. Med. Chem., 2016, 59, 4443–4458 CrossRef CAS PubMed.
- T. M. Caldwell, Y. M. Ahn, S. L. Bulfer, C. B. Leary, M. M. Hood, W.-P. Lu, L. Vogeti, S. Vogeti, M. D. Kaufman, S. C. Wise, B. Le Bourdonnec, B. D. Smith and D. L. Flynn, Bioorg. Med. Chem. Lett., 2022, 74, 128928 CrossRef CAS PubMed.
- B. D. Smith, M. D. Kaufman, S. C. Wise, Y. M. Ahn, T. M. Caldwell, C. B. Leary, W. P. Lu, G. Tan, L. Vogeti, S. Vogeti, B. A. Wilky, L. E. Davis, M. Sharma, R. Ruiz-Soto and D. L. Flynn, Mol. Cancer Ther., 2021, 20, 2098–2109 CrossRef CAS PubMed.
- S. Banerjee, M. G. Krebs, A. Greystoke, A. I. Garces, V. S. Perez, A. Terbuch, R. Shinde, R. Caldwell, R. Grochot, M. Rouhifard, R. Ruddle, B. Gurel, K. Swales, N. Tunariu, T. Prout, M. Parmar, S. Symeonides, J. Rekowski, C. Yap, A. Sharp, A. Paschalis, J. Lopez, A. Minchom, J. S. de Bono and U. Banerji, Nat. Med., 2025, 31, 3074–3080 CrossRef CAS PubMed.
- R. Kabirian, T. Gaillard, D. Bello-Roufai, C. Malhaire, V. Huchet, C. Callens, B. Pham, E. Bentivegna, B. Borghese, E. Laas, V. Cockenpot, A. Leary, J. Alexandre, F. Selle and M. Rodrigues, Bull. Cancer, 2026, 113, 533–540 CrossRef PubMed.
- T. Aoki, I. Hyohdoh, N. Furuichi, S. Ozawa, F. Watanabe, M. Matsushita, M. Sakaitani, K. Morikami, K. Takanashi, N. Harada, Y. Tomii, K. Shiraki, K. Furumoto, M. Tabo, K. Yoshinari, K. Ori, Y. Aoki, N. Shimma and H. Iikura, ACS Med. Chem. Lett., 2014, 5, 309–314 CrossRef CAS PubMed.
- K. Moore and A. Walter, Drugs Future, 2014, 39, 767–773 CrossRef.
- M. Nagasaka, D. Brazel and S.-H. I. Ou, Expert Opin. Invest. Drugs, 2024, 33, 79–84 CrossRef CAS PubMed.
- I. Khan, A. Sahar, S. Numra, N. Saha, Nidhi and R. Parveen, Expet Opin. Pharmacother., 2025, 26, 765–772 CrossRef CAS PubMed.
- M. Pérol, W. Li, N. A. Pennell, G. Liu, Y. Ohe, F. De Braud, M. Nagasaka, E. Felip, A. Xiong, Y. Zhang, H. Fan, X. Wang, S. Li, R. K. Lai, F. Ran, X. Zhang, W. Chen, L. Bazhenova and C. Zhou, J. Clin. Oncol., 2025, 43, 1920–1929 CrossRef PubMed.
- C. G. Zifan Zhang, X. Ma, Z. Li, R. A. Jr. Charles, L. Wei and Z.-S. Chen, Drugs Future, 2022, 47, 489–499 CrossRef.
- S. Dhillon, Drugs, 2023, 83, 1629–1634 CrossRef PubMed.
- M. Wang, J. C.-H. Yang, P. L. Mitchell, J. Fang, D. R. Camidge, W. Nian, C.-H. Chiu, J. Zhou, Y. Zhao, W.-C. Su, T.-Y. Yang, V. W. Zhu, M. Millward, Y. Fan, W.-T. Huang, Y. Cheng, L. Jiang, D. Brungs, L. Bazhenova, C. K. Lee, B. Gao, Y. Xu, W.-H. Hsu, L. Zheng and P. A. Jänne, Cancer Discov., 2022, 12, 1676–1689 CrossRef CAS PubMed.
- H. Hayashi, Lancet Respir. Med., 2024, 12, 185–186 CrossRef CAS PubMed.
- Y.-T. Wang, P.-C. Yang, J.-Y. Zhang and J.-F. Sun, Molecules, 2024, 29, 1448 CrossRef CAS PubMed.
- S. O. Pathak and S. M. Manohar, Biomolecules, 2025, 15, 463 CrossRef CAS PubMed.
- Y. Odia, C. Koschmann, N. A. Vitanza, P. de Blank, D. Aguilera, J. Allen, D. Daghistani, M. Hall, Z. Khatib, C. Kline, T. MacDonald, S. Mueller, S. L. Faison, J. E. Allen, O. J. Naderer, S. C. Ramage, R. S. Tarapore, S. L. McGovern, S. Khatua, W. Zaky and S. L. Gardner, Neuro Oncol., 2024, 26, S155–s164 CrossRef PubMed.
- J. M. Honnanayakanavar, J. B. Nanubolu and S. Suresh, Org. Biomol. Chem., 2021, 19, 8497–8501 RSC.
- D. Brazel, C. J. Park and M. Nagasaka, Crit. Rev. Oncol. Hematol., 2025, 215, 104896 CrossRef PubMed.
- A. Ismail, A. Desai and Y. Boumber, Front. Oncol., 2025, 15, 1624124 CrossRef CAS PubMed.
- P. Trillo Aliaga, G. Spitaleri, I. Attili, C. Corvaja, E. Battaiotto, P. A. Angelopoulos, E. Del Signore, A. Passaro and F. de Marinis, Molecules, 2025, 30, 2645 CrossRef CAS PubMed.
- B. Wilding, D. Scharn, D. Böse, A. Baum, V. Santoro, P. Chetta, R. Schnitzer, D. A. Botesteanu, C. Reiser, S. Kornigg, P. Knesl, A. Hörmann, A. Köferle, M. Corcokovic, S. Lieb, G. Scholz, J. Bruchhaus, M. Spina, J. Balla, B. Peric-Simov, J. Zimmer, S. Mitzner, T. N. Fett, A. Beran, L. Lamarre, T. Gerstberger, D. Gerlach, M. Bauer, A. Bergner, A. Schlattl, G. Bader, M. Treu, H. Engelhardt, S. Zahn, J. E. Fuchs, J. Zuber, P. Ettmayer, M. Pearson, M. Petronczki, N. Kraut, D. B. McConnell, F. Solca and R. A. Neumüller, Nat. Cancer, 2022, 3, 821–836 CrossRef CAS PubMed.
- E. Chong, R. Li, W. Dong, M. Chevliakov, T. G. Tampone, Y. Zhang, B. Qu, N. Haddad, J. C. Lorenz, M. Sarvestani, T. Omar, H. Li, J. J. Gao, D. A. Gao, S. Pennino, L. Wu, E. Spinelli, S. Yao, H. Lee, F. Buono, J. J. Song and B. Wilding, Org. Process Res. Dev., 2025, 29, 1606–1616 CrossRef CAS.
- S. V. Bhagwat, C. Mur, M. Vandekopple, B. Zhao, W. Shen, C. Marugán, A. Capen, L. Kindler, J. R. Stephens, L. Huber, M. A. Castanares, D. Garcia-Tapia, J. D. Cohen, J. Bastian, B. Mattioni, E. Yuen, T. K. Baker, V. Rodriguez Cruz, D. Fei, J. R. Manro, N. Pulliam, M. S. Dowless, M. J. Ortiz Ruiz, C. Yu, L. Puca, A. Klippel, F. Bacchion, R. Ismail-Khan, V. Rodrik-Outmezguine, S. B. Peng, M. J. Lallena, X. Gong and A. de Dios, Cancer Res., 2025, 85, 777–790 CrossRef CAS PubMed.
- E. S. Wang, P. Montesinos, J. Foran, H. Erba, E. Rodríguez-Arbolí, K. Fedorov, M. Heiblig, F. H. Heidel, J. K. Altman, M. R. Baer, L. Ades, K. Pettit, P. Peterlin, C. Papayannidis, C. Berthon, R. B. Walter, M. V. Shah, S. Balasubramanian, M. Khawandanah, O. Salamero Garcia, J. Bergeron, Y. F. Madanat, G. J. Roboz, M. Ulrickson, R. L. Redner, J. McCloskey, A. Pigneux, A. de la Fuente Burguera, A. Mitra, H. S. Soifer, M. Tabachri, Z. Zhang, M. Riches, D. Corum, M. Leoni, G. C. Issa and A. T. Fathi, J. Clin. Oncol., 2025, 43, 3381–3390 CrossRef CAS PubMed.
- J. Rausch, M. M. Dzama, N. Dolgikh, H. L. Stiller, S. R. Bohl, C. Lahrmann, K. Kunz, L. Kessler, H. Echchannaoui, C. W. Chen, T. Kindler, K. Döhner, F. Burrows, M. Theobald, D. Sasca and M. W. M. Kühn, Haematologica, 2023, 108, 2837–2843 CrossRef CAS PubMed.
- A. Mitra, J. M. Ahsan, M. Tabachri, T. El-Shahat, M. Leoni and S. Dale, Clin. Transl. Sci., 2025, 18, e70153 CrossRef CAS PubMed.
- G. Reyhanoglu, B. Hughes, K. E. King and R. Cambridge, Cureus, 2020, 12, e12042 Search PubMed.
- F. Siegel, S. Siegel, K. Kotýnková, G. Karsli Uzunbas, D. Korr, H. Tomono, S. Andersen, D. Denney, M. Berger, V. K. Schulze, T. A. Lewis, B. Kaplan, S. Golfier, J. Mortier, R. C. Hillig, U. Boemer, K. Petersen, K. Eis, S. Williams, D. Rüttinger, A. D. Cherniack, H. H. Loong, K. Goto, P. Grassi, M. Meyerson and H. Greulich, Cancer Discov., 2026, 16, 81–94 CrossRef PubMed.
- X. Le, T. M. Kim, H. H. Loong, A. Prelaj, B. C. Goh, L. Li, Y. Fang, S. Lu, X. Dong, L. Wu, Y. Shinno, G. Daniele, T. Y. Yang, H. R. Kim, G. Ruiter, J. Zhao, S. Novello, L. Miao, P. A. Jänne, K. Goto, D. Rüttinger, T. Descamps, J. C. Brase, W. Bao, R. Li, N. Brega, P. Grassi, N. Girard and D. S. Tan, N. Engl. J. Med., 2025, 393, 1819–1832 CrossRef CAS PubMed.
- J. D. Osteen, S. Immani, T. L. Tapley, T. Indersmitten, N. W. Hurst, T. Healey, K. Aertgeerts, P. A. Negulescu and S. M. Lechner, Pain Ther., 2025, 14, 655–674 CrossRef PubMed.
- J. Jones, D. J. Correll, S. M. Lechner, I. Jazic, X. Miao, D. Shaw, C. Simard, J. D. Osteen, B. Hare, A. Beaton, T. Bertoch, A. Buvanendran, A. S. Habib, L. J. Pizzi, R. A. Pollak, S. G. Weiner, C. Bozic, P. Negulescu and P. F. White, N. Engl. J. Med., 2023, 389, 393–405 CrossRef CAS PubMed.
- M. Jones, A. Demery and R. A. Al-Horani, Drugs Drug Candidates, 2025, 4, 32 CrossRef PubMed.
- J. E. Ross, N. E. Scangarella-Oman, R. K. Flamm and R. N. Jones, J. Clin. Microbiol., 2014, 52, 2629–2632 CrossRef PubMed.
- J. A. Collins and N. Osheroff, ACS Infect. Dis., 2024, 10, 1097–1115 CrossRef CAS PubMed.
- E. G. Gibson, B. Bax, P. F. Chan and N. Osheroff, ACS Infect. Dis., 2019, 5, 570–581 CrossRef CAS PubMed.
- F. Ruggieri, N. Compagne, K. Antraygues, M. Eveque, M. Flipo and N. Willand, Eur. J. Med. Chem., 2023, 256, 115413 CrossRef CAS PubMed.
- D. E. Davies, D. T. Davies, I. Giordano, A. J. Hennessy and N. D. Pearson, WO2008128942A1, 2008.
- K. L. Widdowson, US pat., 20170304301A1, 2017 Search PubMed.
- C. W. Barfoot, P. Brown, S. Dabbs, D. T. Davies, A. J. Hennessy, T. J. Miles and N. D. Pearson, Tetrahedron Lett., 2010, 51, 5038–5040 CrossRef CAS.
- A. Rajasekaran, B. A. Julian and D. V. Rizk, Am. J. Med. Sci., 2021, 361, 176–194 CrossRef PubMed.
- H. J. L. Heerspink, H. H. Parving, D. L. Andress, G. Bakris, R. Correa-Rotter, F. F. Hou, D. W. Kitzman, D. Kohan, H. Makino, J. J. V. McMurray, J. Z. Melnick, M. G. Miller, P. E. Pergola, V. Perkovic, S. Tobe, T. Yi, M. Wigderson and D. de Zeeuw, Lancet, 2019, 393, 1937–1947 CrossRef CAS PubMed.
- M. Winn, T. W. von Geldern, T. J. Opgenorth, H.-S. Jae, A. S. Tasker, S. A. Boyd, J. A. Kester, R. A. Mantei, R. Bal and B. K. Sorensen, J. Med. Chem., 1996, 39, 1039–1048 CrossRef CAS PubMed.
- S. J. Wittenberger and M. A. McLaughlin, Tetrahedron Lett., 1999, 40, 7175–7178 CrossRef CAS.
- M. Buchholz and H. U. Reißig, Eur. J. Org Chem., 2003, 2003, 3524–3533 CrossRef.
- H. A. Blair, Drugs, 2025, 85, 1499–1505 CrossRef CAS PubMed.
- H. Farkas and Z. Balla, Expet Opin. Pharmacother., 2025, 26, 685–693 CrossRef PubMed.
- R. L. Davie, H. J. Edwards, D. M. Evans, S. T. Hodgson, M. J. Stocks, A. J. Smith, L. J. Rushbrooke, S. J. Pethen, M. B. Roe, D. E. Clark, P. A. McEwan and S. L. Hampton, J. Med. Chem., 2022, 65, 13629–13644 CrossRef CAS PubMed.
- M. Worm, J. P. Thyssen, S. Schliemann, A. Bauer, V. Y. Shi, B. Ehst, S. Tillmann, S. Korn, K. Resen and T. Agner, Br. J. Dermatol., 2022, 187, 42–51 CrossRef CAS PubMed.
- S. Dhillon, Drugs, 2020, 80, 609–615 CrossRef CAS PubMed.
- S. Noji, Y. Hara, T. Miura, H. Yamanaka, K. Maeda, A. Hori, H. Yamamoto, S. Obika, M. Inoue, Y. Hase, T. Orita, S. Doi, T. Adachi, A. Tanimoto, C. Oki, Y. Kimoto, Y. Ogawa, T. Negoro, H. Hashimoto and M. Shiozaki, J. Med. Chem., 2020, 63, 7163–7185 CrossRef CAS PubMed.
- Z. Jawaid, M. Khalid and A. Waafira, Ann. Med. Surg., 2025, 87, 6916–6917 CrossRef PubMed.
- H. Takiguchi, A. Higashi, T. Watanabe, T. Takeichi, T. Shimazaki and T. Inaba, Org. Process Res. Dev., 2021, 25, 342–348 CrossRef CAS.
- B. Thöny, G. Auerbach and N. Blau, Biochem. J., 2000, 347(Pt 1), 1–16 CrossRef.
- L. Gao, D. Kaushik, K. Ingalls, N. Smith and R. Kong, Pharmaceuticals, 2024, 17, 1411 CrossRef CAS PubMed.
- A. Grzybowski, L. Kapitanovaite and R. Zemaitiene, Adv. Ophthalmol. Pract. Res., 2024, 4, 220–225 CrossRef PubMed.
- A. C. Kruse, J. Hu, A. C. Pan, D. H. Arlow, D. M. Rosenbaum, E. Rosemond, H. F. Green, T. Liu, P. S. Chae, R. O. Dror, D. E. Shaw, W. I. Weis, J. Wess and B. K. Kobilka, Nature, 2012, 482, 552–556 CrossRef CAS PubMed.
- A. Renna, J. L. Alió and L. F. Vejarano, Eye Vis., 2017, 4, 3 CrossRef PubMed.
- J. D. Chalmers, R. Kettritz and B. Korkmaz, Front. Immunol., 2023, 14 CAS.
- C. L. Langrish, J. M. Bradshaw, M. R. Francesco, T. D. Owens, Y. Xing, J. Shu, J. LaStant, A. Bisconte, C. Outerbridge, S. D. White, R. J. Hill, K. A. Brameld, D. M. Goldstein and P. A. Nunn, J. Immunol., 2021, 206, 1454–1468 CrossRef CAS PubMed.
- D. J. Kuter, M. Efraim, J. Mayer, M. Trněný, V. McDonald, R. Bird, T. Regenbogen, M. Garg, Z. Kaplan, N. Tzvetkov, P. Y. Choi, A. J. G. Jansen, M. Kostal, R. Baker, J. Gumulec, E. J. Lee, I. Cunningham, I. Goncalves, M. Warner, R. Boccia, T. Gernsheimer, W. Ghanima, O. Bandman, R. Burns, A. Neale, D. Thomas, P. Arora, B. Zheng and N. Cooper, N. Engl. J. Med., 2022, 386, 1421–1431 CrossRef CAS PubMed.
- D. J. Kuter and W. Ghanima, Immunotherapy, 2025, 17, 767–782 CrossRef CAS PubMed.
- D. Wei, J. Guo, F. Zhu, E. M. Bonku, C. Sun, P. Yang, H. Qin and J. Shen, J. Org. Chem., 2025, 90, 15065–15073 CrossRef CAS PubMed.
- M. R. Gadelha, M. B. Gordon, M. Doknic, E. Mezősi, M. Tóth, H. Randeva, T. Marmon, T. Jochelson, R. Luo, M. Monahan, A. Madan, C. Ferrara-Cook, R. S. Struthers and A. Krasner, J. Clin. Endocrinol. Metab., 2023, 108, e148–e159 CrossRef PubMed.
- J. Zhao, H. Fu, J. Yu, W. Hong, X. Tian, J. Qi, S. Sun, C. Zhao, C. Wu, Z. Xu, L. Cheng, R. Chai, W. Yan, X. Wei and Z. Shao, Nat. Commun., 2023, 14, 962 CrossRef CAS PubMed.
- J. Zhao, S. Wang, S. Markison, S. H. Kim, S. Han, M. Chen, A. K. Kusnetzow, E. Rico-Bautista, M. Johns, R. Luo, R. S. Struthers, A. Madan, Y. Zhu and S. F. Betz, ACS Med. Chem. Lett., 2023, 14, 66–74 CrossRef CAS PubMed.
- B. Nuesslein-Hildesheim, E. Ferrero, C. Schmid, C. Huck, P. Smith, S. Tisserand, J. Rubert, F. Bornancin, D. Eichlisberger and B. Cenni, J. Neuroinflammation, 2023, 20, 194 CrossRef CAS PubMed.
- T. Dörner, M. Kaul, A. Szántó, J. C. Tseng, A. S. Papas, I. Pylvaenaeinen, M. Hanser, N. Abdallah, A. Grioni, A. Santos Da Costa, E. Ferrero, P. Gergely, R. Hillenbrand, A. Avrameas, B. Cenni and R. M. Siegel, Ann. Rheum. Dis., 2024, 83, 360–371 CrossRef PubMed.
- M. Kaul, P. End, M. Cabanski, C. Schuhler, A. Jakab, M. Kistowska, A. Kinhikar, A. Maiolica, A. Sinn, R. Fuhr and B. Cenni, Clin. Transl. Sci., 2021, 14, 1756–1768 CrossRef CAS PubMed.
- D. Angst, F. Gessier, P. Janser, A. Vulpetti, R. Wälchli, C. Beerli, A. Littlewood-Evans, J. Dawson, B. Nuesslein-Hildesheim, G. Wieczorek, S. Gutmann, C. Scheufler, A. Hinniger, A. Zimmerlin, E. G. Funhoff, R. Pulz and B. Cenni, J. Med. Chem., 2020, 63, 5102–5118 CrossRef CAS PubMed.
- R. Keith and A. M. Nambiar, Ther. Adv. Respir. Dis., 2025, 19, 17534666241309795 CrossRef CAS PubMed.
- T. M. Maher, S. Assassi, A. Azuma, V. Cottin, A. M. Hoffmann-Vold, M. Kreuter, J. M. Oldham, L. Richeldi, C. Valenzuela, M. S. Wijsenbeek, E. Clerisme-Beaty, C. Coeck, H. Gu, I. Ritter, A. Schlosser, S. Stowasser, F. Voss, G. Weimann, D. F. Zoz and F. J. Martinez, N. Engl. J. Med., 2025, 392, 2203–2214 CrossRef CAS PubMed.
- R. P. Frutos, T. G. Tampone, F. Gerstmann, D. Weber, T. Brodmann, R. Hagenkötter, J. Abella, B.-S. Yang, J. Mulder, S. Rodriguez, H. Lee, J. Gao and J. J. Song, Org. Process Res. Dev., 2024, 28, 3745–3751 CrossRef CAS.
- N. Komatsu, M. Hashizume, T. Sugita and S. Uemura, J. Org. Chem., 1993, 58, 4529–4533 CrossRef CAS.
- J. Sassarini and R. A. Anderson, Expert Opin. Invest. Drugs, 2024, 33, 19–26 CrossRef CAS PubMed.
- M. Hager, T. Goldstein, V. Fitz and J. Ott, Expet Opin. Pharmacother., 2024, 25, 783–789 CrossRef CAS PubMed.
- J. A. Simon, R. A. Anderson, E. Ballantyne, J. Bolognese, C. Caetano, H. Joffe, M. Kerr, N. Panay, C. Seitz, S. Seymore, M. Trower, L. Zuurman and S. Pawsey, Menopause, 2023, 30, 239–246 CrossRef PubMed.
- F. Hassan, A. Saleem, S. S. Samuel, Z. Sarfraz, A. Sarfraz, M. Sarfraz and M. Kc, Medicine, 2023, 102, e33978 CrossRef CAS PubMed.
- M. Münzel, C. Szeibert, A. F. Glas, D. Globisch and T. Carell, J. Am. Chem. Soc., 2011, 133, 5186–5189 CrossRef PubMed.
- H. Peng, J. Jie, I. P. Mortimer, Z. Ma, H. Su and M. M. Greenberg, J. Am. Chem. Soc., 2021, 143, 14738–14747 CrossRef CAS PubMed.
- B. Huy Le, V. T. Nguyen and Y. J. Seo, Chem. Commun., 2019, 55, 2158–2161 RSC.
- H. Koyama, Siqin, Z. Zhang, K. Sumi, Y. Hatta, H. Nagata, H. Doi and M. Suzuki, Org. Biomol. Chem., 2011, 9, 4287–4294 RSC.
- H. Morgan, M. Lipka-Lloyd, A. J. Warren, N. Hughes, J. Holmes, N. P. Burton, E. Mahenthiralingam and B. D. Bax, Int. J. Mol. Sci., 2023, 24, 1634 CrossRef CAS PubMed.
- A. R. Raccagni, M. Ranzenigo, E. Bruzzesi, C. Maci, A. Castagna and S. Nozza, J. Clin. Med., 2023, 12, 7767 CrossRef CAS PubMed.
- S. Abdellati, J. G. E. Laumen, T. de Block, I. De Baetselier, D. Van Den Bossche, C. Van Dijck, S. S. Manoharan-Basil and C. Kenyon, Sci. Rep., 2024, 14, 1179 CrossRef CAS PubMed.
- A. Masri, R. N. Cardoso, T. P. Abraham, B. L. Claggett, C. J. Coats, S. M. Hegde, I. J. Kulac, M. M. Y. Lee, M. S. Maron, B. Merkely, M. Michels, I. Olivotto, A. Oreziak, D. L. Jacoby, S. B. Heitner, S. Kupfer, F. I. Malik, L. Meng, S. D. Solomon, A. Wohltman, R. Y. Kwong and C. M. Kramer, J. Am. Coll. Cardiol., 2024, 84, 1806–1817 CrossRef CAS PubMed.
- S. A. Sebastian, I. Padda, E. J. Lehr and G. Johal, Am. J. Cardiovasc. Drugs, 2023, 23, 519–532 CrossRef CAS PubMed.
- D. Saleh, M. Eskandari and L. Choudhury, Future Cardiol., 2025, 21, 685–691 CrossRef CAS PubMed.
- J. J. Hartman, D. T. Hwee, J. Robert-Paganin, C. Chuang, E. R. Chin, S. Edell, K. H. Lee, R. Madhvani, P. Paliwal, J. Pernier, S. S. Sarkar, J. Schaletzky, K. Schauer, K. D. Taheri, J. Wang, E. Wehri, Y. Wu, A. Houdusse, B. P. Morgan and F. I. Malik, Nat. Cardiovasc. Res., 2024, 3, 1003–1016 CrossRef CAS PubMed.
- C. Chuang, S. Collibee, L. Ashcraft, W. Wang, M. Vander Wal, X. Wang, D. T. Hwee, Y. Wu, J. Wang, E. R. Chin, P. Cremin, J. Zamora, J. Hartman, J. Schaletzky, E. Wehri, L. A. Robertson, F. I. Malik and B. P. Morgan, J. Med. Chem., 2021, 64, 14142–14152 CrossRef CAS PubMed.
- V. M. Polymeropoulos, L. Kiely, M. L. Bushman, E. B. Sutherland, A. R. Goldberg, A. X. Pham, C. R. Miller, R. Mourad, T. R. Davis, N. V. Pham, D. B. Morgan, A. K. Giles, C. Xiao, C. M. Polymeropoulos, G. Birznieks and M. H. Polymeropoulos, Front. Neurol., 2025, 16, 1550670 CrossRef PubMed.
- V. M. Polymeropoulos, M. Czeisler, M. M. Gibson, A. A. Anderson, J. Miglo, J. Wang, C. Xiao, C. M. Polymeropoulos, G. Birznieks and M. H. Polymeropoulos, Front. Neurol., 2020, 11, 563373 CrossRef PubMed.
- J. L. Carlin, C. Polymeropoulos, M. Camilleri, A. Lembo, M. Fisher, C. Kupersmith, D. Madonick, P. Moszczynski, S. Smieszek, C. Xiao, G. Birznieks and M. H. Polymeropoulos, Clin. Gastroenterol. Hepatol., 2024, 22, 2506–2516 CrossRef CAS PubMed.
- Y. Ahn, L. Vogeti, T. M. Caldwell, M. D. Kaufman and D. L. Flynn, WO2014145025A2, 2014.
- M. Martinez-Garcia, U. Banerji, J. Albanell, R. Bahleda, S. Dolly, F. Kraeber-Bodéré, F. Rojo, E. Routier, E. Guarin, Z. X. Xu, R. Rueger, J. J. Tessier, E. Shochat, S. Blotner, V. M. Naegelen and J. C. Soria, Clin. Cancer Res., 2012, 18, 4806–4819 CrossRef CAS PubMed.
- R. Katayama, B. Gong, N. Togashi, M. Miyamoto, M. Kiga, S. Iwasaki, Y. Kamai, Y. Tominaga, Y. Takeda, Y. Kagoshima, Y. Shimizu, Y. Seto, T. Oh-Hara, S. Koike, N. Nakao, H. Hanzawa, K. Watanabe, S. Yoda, N. Yanagitani, A. N. Hata, A. T. Shaw, M. Nishio, N. Fujita and T. Isoyama, Nat. Commun., 2019, 10, 3604 CrossRef PubMed.
- Z. Li, H. Zou, W. Zhu, C. Shen, R. Wang, W. Liu, X. Chen, H. Tsui, Z. Yang and X. Zhang, WO2019149164A1, 2019.
- S. L. Faison, J. Batonga, T. Arumugham, A. Bartkus, M. Morrison, M. J. Mullin, T. Tippin and O. Naderer, Clin. Pharmacol. Drug Dev., 2025, 14, 382–390 CrossRef CAS PubMed.
- B. Wilding, D. Boese, H. Engelhardt, J. Fuchs, R. Neumueller, M. Petronczki, D. Scharn and M. Treu, WO2021213800, 2021.
- R. K. Rej, J. E. Thomas, R. K. Acharyya, J. M. Rae and S. Wang, J. Med. Chem., 2023, 66, 8339–8381 CrossRef CAS PubMed.
- D. J. Farrell, H. S. Sader, P. R. Rhomberg, N. E. Scangarella-Oman and R. K. Flamm, Antimicrob. Agents Chemother., 2017, 61, e02047 CrossRef CAS PubMed.
- H. Yuyama, Y. Noguchi, A. Fujimori, M. Ukai, N. Fujiyasu, A. Ohtake, S. Sato, K. Sudoh, M. Sasamata and K. Miyata, Eur. J. Pharmacol., 2004, 498, 171–177 CrossRef CAS PubMed.
- A. Tanimoto, Y. Ogawa, C. Oki, Y. Kimoto, K. Nozawa, W. Amano, S. Noji, M. Shiozaki, A. Matsuo, Y. Shinozaki and M. Matsushita, Inflamm. Res., 2015, 64, 41–51 CrossRef CAS PubMed.
- Y. Cui, D. Wang, W. Si, W. Lv, Y. Niu, X. Lei, Y. Hu and X. Cao, Cell Res., 2008, 18, 1151–1153 CrossRef CAS PubMed.
- K. Doyle, H. Lönn, H. Käck, A. Van de Poël, S. Swallow, P. Gardiner, S. Connolly, J. Root, C. Wikell, G. Dahl, K. Stenvall and P. Johannesson, J. Med. Chem., 2016, 59, 9457–9472 CrossRef CAS PubMed.
- P. F. Smith, J. Krishnarajah, P. A. Nunn, R. J. Hill, D. Karr, D. Tam, M. Masjedizadeh, J. O. Funk and S. G. Gourlay, Br. J. Clin. Pharmacol., 2017, 83, 2367–2376 CrossRef CAS PubMed.
- F. E. Herrmann, C. Hesslinger, L. Wollin and P. Nickolaus, Front. Pharmacol, 2022, 13, 838449 CrossRef CAS PubMed.
- C. Domínguez-González, M. Madruga-Garrido, F. Mavillard, C. Garone, F. J. Aguirre-Rodríguez, M. A. Donati, K. Kleinsteuber, I. Martí, E. Martín-Hernández, J. P. Morealejo-Aycinena, F. Munell, A. Nascimento, S. G. Kalko, M. D. Sardina, C. Álvarez del Vayo, O. Serrano, Y. Long, Y. Tu, B. Levin, J. L. P. Thompson, K. Engelstad, J. Uddin, J. Torres-Torronteras, C. Jimenez-Mallebrera, R. Martí, C. Paradas and M. Hirano, Ann. Neurol., 2019, 86, 293–303 CrossRef PubMed.
- P. Govender, R. Müller, K. Singh, V. Reddy, C. J. Eyermann, S. Fienberg, S. R. Ghorpade, L. Koekemoer, A. Myrick, D. Schnappinger, C. Engelhart, J. Meshanni, J. A. W. Byl, N. Osheroff, V. Singh, K. Chibale and G. S. Basarab, J. Med. Chem., 2022, 65, 6903–6925 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |