Open Access Article
Open Access ArticleSynthesis and in vitro antitumor evaluation of novel 3,4-dihydropyrimidinone-vorinostat hybrids against gastric and breast cancer cell lines
E. A. M. Riosa,
G. M. S. D. M. Pereira b,
M. G. M. D'Oca
b,
M. G. M. D'Oca b,
C. M. Deaa,
E. R. F. B. Santosa,
D. S. Rampona,
L. S. Santos
b,
C. M. Deaa,
E. R. F. B. Santosa,
D. S. Rampona,
L. S. Santos c,
F. M. Nachtigallc,
L. Guzmanc,
R. Moore-Carrascoc,
D. Rebolledo-Mirafc and
C. R. M. D'Oca
c,
F. M. Nachtigallc,
L. Guzmanc,
R. Moore-Carrascoc,
D. Rebolledo-Mirafc and
C. R. M. D'Oca *a
*a
aLaboratory of Polymers and Catalysis (LAPOCA), Department of Chemistry, Federal University of Parana – UFPR, P. O. Box 19032, Curitiba, PR 81531-990, Brazil. E-mail: carolinedoca@ufpr.br
bLaboratory KOLBE of Organic Synthesis, Department of Chemistry, Federal University of Parana – UFPR, P. O. Box 19032, Curitiba, PR 81531-990, Brazil
cInstitute of Natural Resources, Universidad de Talca, Talca 3460000, Chile
First published on 16th March 2026
Abstract
Novel 5-substituted dihydropyrimidinones (DHPMs) bearing a methoxy terminal group, together with DHPM–SAHA molecular hybrids inspired by the clinically approved antitumour agent Vorinostat (SAHA), were synthesised via the Biginelli multicomponent reaction and evaluated for their in vitro antitumour activity against AGS (gastric) and MCF-7 (breast) cancer cell lines. All compounds exhibited low cytotoxicity towards human fibroblasts (SW872), maintaining cell viabilities ≥70%. The derivatives displayed pronounced antiproliferative activity, particularly against MCF-7 cells, consistently showing higher potency than when against AGS cells and, in several cases, superior activity compared to Monastrol. Within the ester DHPM series, three compounds exhibited IC50 values below 5 µM, with derivative 6a emerging as the most active ester analogue (IC50 = 1.33 ± 0.09 µM). Notably, substitution of the terminal ester by a hydroxamic acid moiety—responsible for Zn2+ chelation in HDAC inhibition and central to Vorinostat's antitumour mechanism—resulted in a marked enhancement of antiproliferative activity, yielding highly potent DHPM–SAHA hybrids. Several hybrids displayed submicromolar IC50 values against MCF-7 cells, irrespective of the aromatic substituent, with four compounds exhibiting IC50 values below 1 µM and a further four below 5 µM. Among them, DHPM–SAHA hybrid 7i, derived from 4-methoxybenzaldehyde, emerged as the most potent compound (IC50 = 0.02 ± 0.00 µM). Collectively, these results underscore the successful integration of DHPM and HDAC-inhibitory pharmacophores and highlight DHPM–SAHA hybrids as promising multitarget scaffolds for the development of new breast cancer therapeutics.
Introduction
The search for innovative drug discovery strategies has intensified due to the high costs, extended timelines and low success rates associated with conventional drug development. Traditional approaches typically require 10–15 years of research and exhibit an overall success rate of approximately 2%.1 In this context, molecular hybridisation has emerged as a promising strategy for the identification of new biologically active compounds. This approach involves the rational combination of distinct pharmacophoric moieties with known biological activities into a single molecular framework, enabling the design of new chemical entities with enhanced or synergistic pharmacological profiles, multitarget activity and potentially reduced adverse effects.2By integrating compounds with well-established pharmacodynamic and pharmacokinetic properties, molecular hybridisation has driven substantial advances in medicinal chemistry. A broad range of therapeutic targets has been explored using this strategy, particularly in cancer therapy. Importantly, molecular hybrids have also shown promise in overcoming multidrug resistance, a major limitation of conventional chemotherapy in which tumour cells become unresponsive to multiple anticancer agents.3 Accordingly, hybrid molecules acting as multitarget anticancer agents are regarded as an effective approach to address key challenges in cancer treatment, including therapeutic resistance and the inherent limitations of combination therapies.4 The translational relevance of this strategy is underscored by the growing number of molecular hybrids currently in clinical trials or already approved for therapeutic use.5,6
Among the compound classes exhibiting well-established anticancer activity through the induction of apoptosis are dihydropyrimidinones (DHPMs).7–9 These heterocyclic scaffolds are readily accessible via the Biginelli multicomponent reaction (MCR), first reported by Pietro Biginelli in 1893.8 This one-pot condensation of a nitrogen source (e.g., urea or thiourea), an aldehyde and a 1,3-dicarbonyl compound affords DHPMs as a versatile class of N-heterocycles with a wide range of biological and pharmacological applications.8–11 Notably, Monastrol is one of the most prominent DHPMs and was identified as a selective inhibitor of the mitotic kinesin Eg5. By targeting this motor protein, which is overexpressed in several cancer types including breast, lung, pancreatic, ovarian and bladder cancers, Monastrol disrupts spindle formation, thereby inhibiting cell division and tumour cell proliferation.12,13
In contrast, Vorinostat (1, Fig. 1)14 is a nitrogen-containing compound that exerts its antitumour activity through chelation of the Zn2+ ion located in the catalytic domain of histone deacetylases (HDACs).15 This mechanism underlies its ability to modulate epigenetic regulation and selectively target tumour cells.16 Vorinostat, also known as suberoylanilide hydroxamic acid (SAHA), was approved in 2006 for the treatment of cutaneous T-cell lymphoma and functions as a pan-inhibitor of class I and II HDACs.17
 | ||
| Fig. 1 Structures of Vorinostat/SAHA (1), recognized for their antitumor activity, and novel DHPM–SAHA hybrids proposed. | ||
Recent studies have highlighted hydroxamic acid–based molecular hybrids as particularly effective HDAC inhibitors. Coumarin-derived hydroxamic acids, for example, have emerged as potent HDACis, combining the coumarin scaffold with zinc-binding hydroxamic acid moieties.18 These hybrids exhibited enhanced antiproliferative activity against MDA-MB-231 and MCF-7 breast cancer cell lines (IC50 = 0.36–2.91 µM) compared with Vorinostat (IC50 = 1.33 and 4.15 µM, respectively), while remaining non-toxic toward normal Beas-2B and L-02 cells.19,20 The phenyl group within the coumarin scaffold enables π–π interactions with tyrosine residues in the HDAC active site, contributing to effective Zn2+ chelation by the hydroxamic acid. Ferrocene–hydroxamic acid hybrids have also demonstrated excellent inhibitory activity against HDAC6, with IC50 values in the nanomolar range, as well as significant antiproliferative effects against 4T1 breast cancer cells.21
Despite their structural diversity, most HDAC inhibitors share a common pharmacophoric architecture that reflects the topology of the HDAC active site. As illustrated in Fig. 1, this pharmacophore comprises three key elements: a surface recognition cap group (CAP) that interacts with amino acid residues at the rim of the active site; a zinc-binding group (ZBG) responsible for chelating the catalytic Zn2+ ion; and a linker domain that occupies the hydrophobic channel of the enzyme.22
In this context, and considering the structural diversity of N-heterocycles accessible through the Biginelli multicomponent reaction—together with the reported antitumour performance of monastrol–melatonin hybrids23 and our previous studies demonstrating the antitumoral potential of Biginelli-derived hybrids24–27—this work reports the multistep synthesis and in vitro antitumour evaluation of novel DHPM–SAHA molecular hybrids (Fig. 2). These compounds were evaluated against breast (MCF-7) and gastric (AGS) cancer cell lines, aiming to explore the synergistic integration of DHPM and HDAC-inhibitory pharmacophores.
 | ||
| Fig. 2 Structures of 5-substituted DHPMs (6a–j) bearing a methoxy terminal group obtained via the Biginelli multicomponent reaction. | ||
Results and discussion
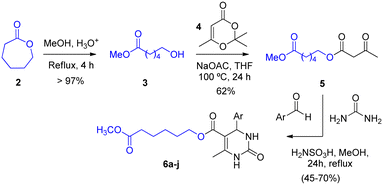
Initially, β-ketoester 5 was synthesised in two steps. In the first step, ring opening of the seven-membered lactone ε-caprolactone (2) was performed in methanol under acidic conditions using sulfamic acid (H2NSO3H, SA, 30 mol%) as catalyst. Sulfamic acid is a stable, inexpensive and environmentally benign solid, which has been widely employed as an efficient green catalyst in organic synthesis.28 After 4 h, the corresponding hydroxyester 3 was obtained in quantitative yield. In the second step, acylation of compound 3 with TMD 4 (2,2,6-trimethyl-4H-1,3-dioxin-4-one) was carried out in the presence of anhydrous NaOAc in THF, affording β-ketoester 5 in 62% yield (Scheme 1).The next step involved a Biginelli multicomponent reaction (MCR) employing the 1,3-dicarbonyl compound 5, a series of aromatic aldehydes bearing either electron-donating or electron-withdrawing substituents, urea, and sulfamic acid (SA, 20 mol%) as the catalyst. The versatility of SA allows its application in a wide range of acid-catalysed organic reactions.29
The reactions were carried out following established literature procedures, affording the 5-substituted DHPMs bearing a methoxy terminal group (6a–i, Fig. 3) in yields ranging from 30% to 70%. The resulting products were purified by recrystallisation.
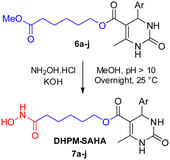
Subsequently, the final step involved the aminolysis of DHPMs 6a–j with hydroxylamine hydrochloride under basic conditions (KOH). The reactions were carried out at 25 °C for 24 h, with the pH maintained above 10 (Scheme 2). The resulting hydroxamic acid–containing hybrids, designated as DHPM–SAHA hybrids 7a–j, were purified by recrystallisation, affording yields in the range of 42–66%. The structures of the hybrids were confirmed by spectroscopic and spectrometric analyses (see SI) and are depicted in Fig. 3.
Both the DHPM–SAHA hybrids 7a–j and their corresponding methoxylated DHPM precursors 6a–j were evaluated for in vitro antitumour activity against AGS and MCF-7 cell lines, representative of gastric and breast cancers, respectively. Initially, MTT assays were performed to evaluate the effects of the compounds on cell viability using the SW872 cell line, a human liposarcoma (malignant) cell model. SW872 has been widely employed in cancer research, particularly for the evaluation of novel anticancer agents, due to its demonstrated sensitivity to several established chemotherapeutic drugs. All experiments were performed in triplicate (Fig. 4).
As shown in Fig. 4, the 5-substituted DHPMs 6a–j and 7a–j, bearing methoxy and hydroxamic acid terminal groups, respectively, exhibited cell viabilities close to or above 70% in human fibroblast cells (SW872), indicating low or negligible cytotoxicity and thus being classified as non-cytotoxic. Consequently, all compounds were selected for further evaluation against the AGS (gastric) and MCF-7 (breast) cancer cell lines. The antiproliferative activities are expressed as IC50 values (µM) and are summarised in Table 1.
| Entry | Compound | IC50 (µM) | ||
|---|---|---|---|---|
| Aromatic moeity | Carboxilic group | AGS | MCF-7 | |
| 1 | 6a, Ph | MeO− | 135.03 ± 20.29 | 1.33 ± 0.09 |
| 2 | 6b, 2-OH-Ph | 106.67 ± 4.21 | 8.79 ± 12.11 | |
| 3 | 6c, 3-OH-Ph | 144.45 ± 21.14 | 13.83 ± 5.83 | |
| 4 | 6d, 4-OH-Ph | 109.94 ± 14.80 | 9.65 ± 8.09 | |
| 5 | 6e, 2-Cl-Ph | 71.12 ± 43.85 | 38.09 ± 12.37 | |
| 6 | 6f, 3-Cl-Ph | 148.13 ± 32.85 | 87.48 ± 125.10 | |
| 7 | 6g, 4-Cl-Ph | 156.43 ± 6.07 | 10.36 ± 2.01 | |
| 8 | 6h, 4-Me-Ph | 125.76 ± 43.32 | 2.33 ± 1.58 | |
| 9 | 6i, 4-MeO-Ph | 120.20 ± 17.45 | 2.75 ± 0.89 | |
| 10 | 6j, furanyl | 138.17 ± 18.18 | 40.17 ± 9.50 | |
| 11 | 7a, Ph | HO-NH− | 64.73 ± 0.48 | 2.36 ± 0.65 |
| 12 | 7b, 2-OH-Ph | 22.12 ± 3.30 | 3.61 ± 0.40 | |
| 13 | 7c, 3-OH-Ph | 14.14 ± 2.50 | 12.85 ± 4.79 | |
| 14 | 7d, 4-OH-Ph | 165.20 ± 4.08 | 18.18 ± 6.12 | |
| 15 | 7e, 2-Cl-Ph | 17.02 ± 0.80 | 3.32 ± 0.57 | |
| 16 | 7f, 3-Cl-Ph | 6.70 ± 2.91 | 0.52 ± 0.20 | |
| 17 | 7g, 4-Cl-Ph | 5.18 ± 0.89 | 0.03 ± 0.01 | |
| 18 | 7h, 4-Me-Ph | 10.83 ± 2.68 | 0.04 ± 0.02 | |
| 19 | 7i, 4-MeO-Ph | 17.12 ± 0.87 | 0.02 ± 0.00 | |
| 20 | 7j, furanyl | 10.19 ± 1.46 | 1.21 ± 0.46 | |
| 21 | Monastrol | 38.2 ± 3.52 | 22.5 ± 6.31 | |
Cells were seeded in 96-well plates at a density of 5000 cells per well and allowed to adhere for 12 h. Subsequently, each DHPM compound was applied at a final concentration of 10 µM and incubated for 48 h. For determination of the half-maximal inhibitory concentration (IC50), the same experimental procedure was followed, using compound concentrations ranging from 0.1 µM to 1000 µM. Monastrol was employed as the reference compound.24
Evaluation of the 5-substituted DHPMs 6a–j, bearing a methoxy terminal group, revealed notable antitumor activity, particularly against the breast cancer cell line (MCF-7). As shown in Table 1 (entries 1–10), all tested DHPMs exhibited significant cytotoxic effects against MCF-7 cells, regardless of the nature of the aromatic substituent, with IC50 values ranging from 1.33 ± 0.09 to 87.48 ± 125.10 µM. Overall, these compounds were more active against MCF-7 than against the gastric cancer cell line (AGS) and, in several cases, displayed superior activity compared to Monastrol (IC50 = 22.5 ± 6.31 µM against MCF-7; Table 1, entry 21).
In contrast, against AGS cells, DHPMs 6a–j exhibited substantially higher IC50 values than Monastrol (38.2 ± 3.52 µM), indicating low cytotoxic activity, with IC50 values ranging from 71.12 ± 43.85 to 156.43 ± 6.07 µM.
Among the evaluated compounds, 6a, 6h, and 6i emerged as the most promising candidates against MCF-7 cells, all exhibiting IC50 values below 5 µM. Notably, the ester derivative 6a (Table 1, entry 1), synthesized from benzaldehyde, displayed the lowest IC50 value (1.33 ± 0.09 µM), representing the most potent compound within the ester DHPM series. Additionally, derivatives 6h and 6i obtained from 4-methylbenzaldehyde and 4-methoxybenzaldehyde (Table 1, entries 8 and 9) also demonstrated high potency, with IC50 values of 2.33 ± 1.58 µM and 2.75 ± 0.89 µM, respectively.
Introduction of a terminal hydroxamic acid group into the side chains (Table 1, entries 11–20), derived from the corresponding ester precursors, resulted in a marked enhancement of in vitro antiproliferative activity for the DHPM–SAHA hybrid derivatives 7a–j.
The IC50 values for this series ranged from 0.02 ± 0.00 to 18.18 ± 6.12 µM against MCF-7 breast cancer cells, and from 5.18 ± 0.89 to 165.20 ± 4.08 µM against AGS gastric cancer cells. Similar to the behaviour observed for the 5-substituted DHPMs 6a–j bearing a methoxy terminal group, the DHPM–SAHA hybrids 7a–j proved to be particularly promising against MCF-7 cells, with all derivatives displaying substantially lower IC50 values than Monastrol (22.5 ± 6.31 µM; Table 1, entry 21).
Against AGS cells, eight compounds (7b, 7c and 7e–j) bearing a terminal hydroxamic acid group exhibited higher activity than Monastrol (38.2 ± 3.52 µM). Notably, derivatives 7f and 7g, obtained from 3-chlorobenzaldehyde and 4-chlorobenzaldehyde, respectively (Table 1, entries 16 and 17), demonstrated good potency, with IC50 values of 5.18 ± 0.89 µM and 6.70 ± 2.91 µM, respectively.
Among the ten compounds evaluated against MCF-7 cells, four derivatives bearing a terminal hydroxamic acid group displayed IC50 values below 1 µM (7f–i), while an additional four compounds exhibited IC50 values below 5 µM (7a, 7b, 7e and 7j), identifying this series as particularly promising for breast cancer inhibition.
In particular, the hydroxamic acid derivative 7i (Table 1, entry 19), synthesised from 4-methoxybenzaldehyde, showed an exceptionally low IC50 value of 0.02 ± 0.00 µM, representing the most potent compound within the DHPM–SAHA hybrid series. Furthermore, derivatives 7g, 7h and 7f, derived from 4-chlorobenzaldehyde, 4-methylbenzaldehyde and 3-chlorobenzaldehyde, respectively (Table 1, entries 16–18), also exhibited significant antiproliferative activity, with IC50 values of 0.03 ± 0.01 µM, 0.04 ± 0.02 µM and 0.52 ± 0.20 µM, respectively.
The DHPM–SAHA hybrid 7j, synthesised from furfuraldehyde, displayed submicromolar activity (IC50 = 1.21 ± 0.46 µM), indicating strong antiproliferative potency. Moreover, derivatives obtained from benzaldehyde, 2-chlorobenzaldehyde, and 2-hydroxybenzaldehyde (Table 1, entries 11, 15 and 12) also exhibited good activity, with IC50 values of 1.21 ± 0.46 µM, 3.32 ± 0.57 µM and 3.61 ± 0.40 µM, respectively.
Compared with our previously reported DHPM fatty acid derivatives, the DHPM–SAHA hybrids exhibited a marked enhancement in antiproliferative activity and a clear structure–activity relationship.24 While the earlier fatty acid derivatives showed moderate activity against AGS and MCF-7 cells (IC50 = 12.1–146.2 µM and 2.3–39.2 µM, respectively), only one derivative derived from 3-hydroxybenzaldehyde displayed an IC50 below 5 µM (2.3 ± 0.4 µM) against MCF-7 cells.
The introduction of a terminal hydroxamic acid moiety at the C5 side-chain of the DHPM core substantially improved potency against the breast cancer cell line MCF-7. Among the ten DHPM–SAHA hybrids evaluated, 7f–i exhibited IC50 values below 1 µM, while 7a, 7b, 7e, and 7j showed IC50 values below 5 µM. These results highlight the crucial role of hydroxamic acid incorporation in enhancing antiproliferative activity.
This profile is consistent with previous reports by Huang and Pardee, who demonstrated that SAHA inhibited proliferation in the breast cancer cell lines MCF-7 and MDA-MB-231 in a dose-dependent manner at low micromolar concentrations (0.5–5 µM), with significant growth suppression observed at approximately 1–2.5 µM.30 MCF-7 cells exhibited the highest sensitivity, confirming the potent antiproliferative effects associated with the hydroxamic acid pharmacophore.
Additionally, the calculated partition coefficient (c![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) log
log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) P) values of the most cytotoxic DHPM–SAHA hybrids were determined to estimate their lipophilicity. Compounds 7e–i exhibited moderate lipophilicity, with c
P) values of the most cytotoxic DHPM–SAHA hybrids were determined to estimate their lipophilicity. Compounds 7e–i exhibited moderate lipophilicity, with c![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) log
log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) P values ranging from 1.3 to 2.1, comparable to that of vorinostat (c
P values ranging from 1.3 to 2.1, comparable to that of vorinostat (c![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) log
log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) P = 1.9).31 Notably, derivatives bearing para-substituted phenyl groups—chloro (7g, c
P = 1.9).31 Notably, derivatives bearing para-substituted phenyl groups—chloro (7g, c![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) log
log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) P = 2.10), methyl (7h, c
P = 2.10), methyl (7h, c![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) log
log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) P = 1.89), and methoxy (7i, c
P = 1.89), and methoxy (7i, c![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) log
log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) P = 1.31)—displayed remarkable potency (Fig. 5), with IC50 values below 0.05 µM, indicating strong antiproliferative activity.
P = 1.31)—displayed remarkable potency (Fig. 5), with IC50 values below 0.05 µM, indicating strong antiproliferative activity.
These findings suggest that the enhanced cytotoxicity observed for these hybrids may be partially associated with their lipophilicity. According to the literature, lipophilicity plays a critical role in histone deacetylase (HDAC) inhibition by influencing both enzyme binding and intracellular activity.32 Classical HDAC inhibitors, such as SAHA, follow a pharmacophoric model consisting of a zinc-binding group (typically a hydroxamic acid), a hydrophobic linker, and a surface-recognition (cap) group with an appropriate degree of lipophilicity. Because the HDAC catalytic site contains a predominantly hydrophobic channel leading to the Zn2+ ion, balanced lipophilicity enhances hydrophobic interactions within the active site and facilitates membrane permeability. However, this relationship is not strictly linear, as excessive lipophilicity may compromise aqueous solubility and pharmacokinetic properties. Therefore, optimal HDAC inhibition requires a well-balanced lipophilic profile that maximizes favorable enzyme interactions and cellular uptake while preserving drug-like characteristics.
Overall, the synthesized DHPM–SAHA hybrids, particularly compound 7i, incorporate structural features characteristic of HDAC inhibitors, notably the presence of a zinc-binding hydroxamic acid capable of coordinating the catalytic Zn2+ ion within the enzyme active site. This modification establishes the classical HDAC pharmacophore architecture—comprising a zinc-binding group, a hydrophobic linker, and a surface-recognition cap group—positioning this hybrid scaffold as a promising platform for breast cancer therapy.
Conclusions
In summary, we synthesized a new series of 5-substituted dihydropyrimidinones (DHPMs) bearing a methoxy terminal group and their corresponding hydroxamic acid-containing DHPM–SAHA hybrids via the Biginelli multicomponent reaction, and evaluated their in vitro antitumor activity against AGS (gastric) and MCF-7 (breast) cancer cell lines.All compounds exhibited low cytotoxicity toward SW872 human fibroblast cells (viability ≥ 70%). The derivatives showed greater activity against MCF-7 than AGS cells, with several compounds outperforming the reference drug Monastrol.
Among the ester derivatives, compound 6a was the most active (IC50 = 1.33 ± 0.09 µM). Importantly, conversion of the terminal ester into hydroxamic acid–bearing DHPM–SAHA hybrids—incorporating a key zinc-binding motif characteristic of HDAC inhibitors such as Vorinostat—significantly enhanced antiproliferative activity.
The resulting hybrids displayed remarkable potency against both cell lines, with most compounds surpassing Monastrol. Compounds 7g, 7h, and 7f exhibited submicromolar activity (IC50 = 0.03 ± 0.01, 0.04 ± 0.02, and 0.52 ± 0.20 µM, respectively), while 7i was the most potent derivative of the series against MCF-7 cells (IC50 = 0.02 ± 0.00 µM).
Overall, hydroxamic acid incorporation and the multitarget DHPM–HDAC design strategy markedly improved anticancer activity. These findings highlight DHPM–SAHA hybrids as promising candidates for breast cancer therapy and warrant further mechanistic and in vivo investigations.
Experimental
General
All commercially available reagents were used without further purification. Sulfamic acid (98 wt%), urea, and thiourea were purchased from Sigma-Aldrich Chemical. The solvents employed in extraction and purification (hexane and ethyl acetate) were distilled before use. The reactions were monitored by TLC (thin-layer chromatography) using pre-coated silica gel 60 F254 plates. The visualization was accomplished with UV light, stained with iodine. Flash chromatography was performed using Merck silica gel with a pore size of 60 Å, 230–400 mesh, with the indicated eluent system. The NMR spectra (1H and 13C) were obtained on a Bruker Nuclear Ascend 400 spectrometer. The spectra were obtained either in CDCl3 or in DMSO-d6 solution. The chemical shifts (δ) are reported in ppm and referenced to tetramethylsilane (TMS) as the internal standard. The coupling constants (J) are reported in hertz (Hz). Hydrogen coupling patterns are described as singlet (s), doublet (d), doublet of doublets (dd), doublet of doublet of doublets (ddd), doublet of triplets (dt), triplet (t), triplet of doublets (td), doublet of quartets (dq), quartet (q), quintet (quint), sextet (sext), broad signal (br), and multiplet (m). The melting points measured for some representative compounds were obtained on an MP90 melting point system.Cell lines
A SW 872 (human fibroblast; HTB-92, ATCC) cell line was used, while AGS (gastric adenocarcinoma; CRL-1739, ATCC) and MCF-7 (mammary carcinoma; HTB-22, ATCC) were used as cancer cell lines. SW 872 and MCF7 cells were cultured in DMEM (Dulbecco's Modified Eagle Medium) medium (HyClone, Thermo Scientific USA); MCF-7′s medium was supplemented with human Insulin (10 µg mL−1; Gibco, Thermo Scientific, USA) and AGS cells were cultured in RPMI-1640 medium (HyClone, Thermo Scientific), both medium were supplemented with 10% fetal bovine serum (FBS) (Hyclone™, GE Healthcare Life Science), 0.25 µg mL−1 amphotericin B (Hyclone™, GE Healthcare Life Science), and 25 µg mL−1 gentamicin (Millipore). Cells were maintained in an incubator at 37 °C, 5% CO2.Cell viability assay (MTT)
5.000 cells per well were seeded in a 96-well plate (SPL Life Sciences, Korea) for 12 hours, and each DHPM compounds were applied at 10 µM as treatment for 48 hours. The cells were washed with 100 µl of 1× PBS for 3 minutes, and then 100 µl of 0.5 mg mL−1 MTT/PBS 1× solution was added. The plate was then incubated for 90 minutes at 37 °C in an incubator. After discarding the MTT solution, 100 µl DMSO was loaded per well and kept stirring for 30 minutes. The plate was agitated for 30 min in darkness, and the absorbance was read at 570 nm using a spectrophotometer. For half maximal inhibitory concentration (IC50) the same previous procedure was followed, but the following concentrations of all the compounds were measured; 1000 µM, 100 µM, 10 µM, 1 µM and 0.1 µM.The MTT assays were conducted in triplicate. Data were saved in an Excel template (Microsoft®, Version 2108) and statistical analyses were performed in GraphPad Prism 8.0.1 (Graphpad Software, San Diego, CA).
Lipophilicity calculations
c![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) log
log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) P values (logarithm of n-octanol/water partition coefficient P) were obtained by the software PerkinElmer ChemDraw®, Level: Professional, Version: 16.0.1.4 (77).
P values (logarithm of n-octanol/water partition coefficient P) were obtained by the software PerkinElmer ChemDraw®, Level: Professional, Version: 16.0.1.4 (77).
General procedure for the synthesis of hydroxyester 3
In a round-bottom flask, ε-caprolactone 2 (1 equivalent), sulfamic acid (NH2SO3H, 30 mol%) and methanol (proportion of 10 mL for 1 mmol of starting material) were added. The reaction was kept under stirring and reflux for 4 hours. After completion of the reaction, the catalyst was filtered and the solvent removed under reduced pressure. Compound 5 was obtained quantitatively and used in the subsequent step without the need of purification.General procedure for the synthesis of β-ketoester 5
In a round bottom flask, methyl 6-hydroxyhexanoate 3 (1 equivalent), TMD 4 (2,2,6-Trimethyl-4H-1,3-dioxin-4-one, 1.3 equivalent) were added, anhydrous sodium NaOAc (1 equivalent) and THF as solvent. The reaction mixture was kept under stirring for 24 hours at 100 °C. Then, a liquid–liquid extraction was carried out with diethyl ether and saturated sodium chloride solution. The collected organic phase was dried with MgSO4, filtered and the solvent removed via rotary evaporation.General procedure for synthesis of 5-substituited DHPM 6a–j
In a round-bottom flask, the aromatic aldehyde (1 equivalent), β-ketoester 3 (1 equivalent), urea (1 equivalent) and the catalyst sulfamic acid (NH2SO3H, 20 mol%) in methanol (MeOH) as solvent. The mixture was kept under reflux and constant stirring for 24 hours. After completion of the reaction, the reaction crude was subjected to purification by column chromatography or recrystallization.General procedure for synthesis of DHPM–SAHA molecular hybrids 7a–j
In a round-bottom flask A, hydroxylamine hydrochloride (18.4 mmol) in methanol was added, kept under stirring at 40 °C. A solution of KOH (18.4 mmol) in methanol was slowly added to the flask containing hydroxylamine hydrochloride to release the hydroxylamine into the reaction medium. After 0.5 hours, the mixture was cooled to 0 °C and filtered (to remove the formed potassium chloride) directly into another flask containing the precursor 6a–j (1 mmol) and methanol (2 mL), previously heated at 85 °C for dissolution. After completion of filtration and addition of the released hydroxylamine, the heating bath was removed so the reaction could occur at room temperature. Subsequently, 0.094 g of KOH was slowly added to reach alkaline pH (pH above 10). The reaction remained under agitation overnight. Afterwards, the mixture was added to 32 mL of water at 0 °C under stirring and the pH was adjusted to 7 with the addition of acetic acid. The precipitate 7a–j formed was filtered under vacuum.Author contributions
Conceptualization: M. G. M. D'Oca and C. D. R Montes D'Oca synthesis and characterization of molecules: E. A. M. Rios, C. M. Dea, E. R. F. B. dos Santos and G. M. S. D. M. Pereira experiment for identification: E. A. M. Rios and G. M. S. D. M. Pereira biological assays: L. S. Santos, F. M. Nachtigall, L. Guzman, R. Moore-Carrasco, D. Rebolledo-Miraf resources: M. G. M. D'Oca, D. S. Rampon and C. D. R Montes D'Oca data curation: E. A. M. Rios and G. M. S. D. M. Pereira writing – original draft: E. A. M. Rios and C. D. R Montes D'Oca writing – review & editing: M. G. M. D'Oca, C. D. R Montes D'Oca, D. S. Rampon and L. S. Santos project administration: Marcelo G. M. D'Oca and Caroline D. R Montes D'Oca funding acquisition: Marcelo G. M. D'Oca, D. S. Rampon, Caroline D. R Montes D'Oca and L. S. Santos.Conflicts of interest
The authors declare no competing interests.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: 1H, 13C NMR spectra and Infrared spectra. See DOI: https://doi.org/10.1039/d6ra00701e.Acknowledgements
The authors are thankful for financial support from Coordenação de Aperfeiçoamento de Pessoal de Nivel Superior (CAPES), and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq). Fellowships from CAPES are also acknowledged.References
- O. M. Soltan, M. E. Shoman, S. A. Abdel-Aziz, A. Narumi, H. Konno and M. Abdel-Aziz, Eur. J. Med. Chem., 2021, 225, 113768 CrossRef CAS PubMed.
- K. Nepali, S. Sharma, M. Sharma, P. M. S. Bedi and K. L. Dhar, Eur. J. Med. Chem., 2014, 77, 422–487 CrossRef CAS PubMed.
- K. Bukowski, M. Kciuk and R. Kontek, Int. J. Mol. Sci., 2020, 21, 3233 CrossRef CAS PubMed.
- E. Marchesi, D. Perrone and M. L. Navacchia, Pharmaceutics, 2023, 15, 2185 CrossRef CAS.
- Shalini and V. Kumar, Expert Opin. Drug Discov., 2021, 16, 335–363 CrossRef CAS PubMed.
- A. K. Singh, A. Kumar, H. Singh, P. Sonawane, H. Paliwal, S. Thareja, P. Pathak, M. Grishina, M. Jaremko and A.-H. Emwas, Pharmaceuticals, 2022, 15, 1071 CrossRef CAS PubMed.
- L. H. S. Matos, F. T. Masson, L. A. Simeoni and M. H. de Mello, Eur. J. Med. Chem., 2018, 143, 1779–1789 CrossRef CAS PubMed.
- P. Biginelli, Ber. Dtsch. Chem. Ges., 1891, 24, 1317–1319 CrossRef.
- H. Nagarajaiah, A. Mukhopadhyay and J. N. Moorthy, Tetrahedron Lett., 2016, 57, 5135–5149 CrossRef CAS.
- S. R. Patil, A. S. Choudhary, V. S. Patil and N. Sekar, Fibers Polym., 2015, 16, 2349–2356 CrossRef CAS.
- A. Fauzi, A. Saifudin and K. Rullah, J. Med. Chem. Sci., 2023, 6, 1810–1821 CAS.
- Z. Maliga, T. M. Kapoor and T. J. Mitchison, Chem. Biol., 2002, 9, 989–996 CrossRef CAS PubMed.
- U. Mayer, T. M. Kapoor, S. J. Haggarty, R. W. King, S. L. Schreiber and T. J. Mitchison, Science, 1999, 286, 971 CrossRef PubMed.
- S. Grant, C. Easley and P. Kirkpatrick, Nat. Rev. Drug Discovery, 2007, 6, 21–22 CrossRef CAS PubMed.
- S. Geurs, D. Clarisse, K. De Bosscher and M. D’hooghe, J. Med. Chem., 2023, 66, 7698–7715 CrossRef CAS PubMed.
- A. Wawruszak, L. Borkiewicz, E. Okon, W. Kukula-Koch, S. Afshan and M. Halasa, Cancers, 2021, 13, 4700 CrossRef CAS PubMed.
- B. S. Mann, J. R. Johnson, M. H. Cohen, R. Justice and R. Pazdur, Oncologist, 2007, 12, 1247–1252 CrossRef CAS PubMed.
- N. Q. Khai and T. K. Vu, Anti-Cancer Agents Med. Chem., 2024, 24, 18–35 CrossRef CAS PubMed.
- N. Zhao, F. Yang, L. Han, Y. Qu, D. Ge and H. Zhang, Molecules, 2020, 25, 717 CrossRef CAS.
- S. García, I. Mercado-Sánchez, L. Bahena, Y. Alcaraz, M. A. García-Revilla, J. Robles, N. Santos-Martínez, D. Ordaz-Rosado, R. García-Becerra and M. A. Vázquez, Molecules, 2020, 25, 5134 CrossRef PubMed.
- J. Yan, K. Yue, X. Fan, X. Xu, J. Wang, M. Qin, Q. Zhang, X. Hou, X. Li and Y. Wang, Eur. J. Med. Chem., 2023, 246, 115004 CrossRef CAS PubMed.
- D. A. Preciado, A. F. Yepes, A. Herrera and W. Cardona, Med. Chem. Res., 2024, 33, 1511–1524 CrossRef.
- Z. Lv, T. Ji, J. Liu, X. Sun and H. Liang, Eur. J. Med. Chem., 2025, 283, 117185 CrossRef CAS PubMed.
- E. A. M. Rios, C. M. Dea, E. R. B. dos Santos, M. G. Montes D'Oca, D. S. Rampon, F. M. Nachtigall, L. S. Santos, L. Guzman, R. Moore-Carrasco, D. Rebolledo-Miraf and C. D. R. Montes D'Oca, RSC Adv., 2024, 14, 22981–22992 RSC.
- V. Vendrusculo, V. P. de Souza, L. A. M. Fontoura, M. G. M. D'Oca, T. P. Banzato, P. A. Monteiro, R. A. Pilli, J. E. de Carvalho and D. Russowsky, Med. Chem. Commun., 2018, 9, 1553–1564 RSC.
- F. S. De Oliveira, P. M. De Oliveira, L. M. Farias, R. C. Brinkerhoff, R. C. M. A. Sobrinho, T. M. Treptow, C. R. Montes D'Oca, M. A. G. Marinho, M. A. Hort, A. P. Horn, D. Russowsky and M. G. Montes D'Oca, Med. Chem. Commun., 2018, 9, 1282–1288 RSC.
- T. G. M. Treptow, F. Figueiró, E. H. F. Jandrey, A. M. O. Battastini, C. G. Salbego, J. B. Hoppe, P. S. Taborda, S. B. Rosa, L. A. Piovesan, C. D. R. Montes D'Oca, D. Russowsky and M. G. Montes D'Oca, Eur. J. Med. Chem., 2015, 95, 552–562 CrossRef CAS PubMed.
- M. G. Montes D'Oca, R. M. Soares, R. R. de Moura and V. F. Granjão, Fuel, 2012, 97, 884–886 CrossRef.
- A. Samiei, M. A. Bodaghifard and M. Hamidinasab, Curr. Green Chem., 2024, 11, 194–203 CrossRef CAS.
- L. Huang and A. B. Pardee, Mol. Med., 2000, 6, 849–866 CAS.
- National Center for Biotechnology Information (NCBI), PubChem Compound Summary for CID 5311, Vorinostat, 2026, https://pubchem.ncbi.nlm.nih.gov/compound/5311 accessed February 2026 Search PubMed.
- P. A. Marks, V. M. Richon and R. A. Rifkind, Nat. Rev. Cancer, 2001, 1, 194–202 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |