 Open Access Article
Open Access ArticleMitigating heavy metals leachability from CO2 carbonated coal-based solid waste backfill in high-salinity environments via biochar-clay-nanomaterial synergistic modification
Zhishang Zhang abcd,
Liqiang Ma*abcd,
Ichhuy Ngo*a,
Kunpeng Yua,
Jiangtao Zhaia,
Zezhou Guoa,
Zhiyang Zhaoa,
Chengkun Penga and
Ruizhi Yanga
abcd,
Liqiang Ma*abcd,
Ichhuy Ngo*a,
Kunpeng Yua,
Jiangtao Zhaia,
Zezhou Guoa,
Zhiyang Zhaoa,
Chengkun Penga and
Ruizhi Yanga
aSchool of Mines, China University of Mining and Technology, Xuzhou, 221116, China. E-mail: 4225@cumt.edu.cn; ngoichhuy@cumt.edu.cn
bXinjiang Key Laboratory of Coal-bearing Resources Exploration and Exploitation, Xinjiang Institute of Engineering, Urumqi, 830023, China
cXinjiang Engineering Research Center of Green Intelligent Coal Mining, Xinjiang Institute of Engineering, Urumqi, 830023, China
dKey Laboratory of Xinjiang Coal Resources Green Mining (Xinjiang Institute of Engineering), Ministry of Education, Urumqi, 830023, China
First published on 22nd April 2026
Abstract
High-salinity mine water exerts an uncertain influence on the leaching behavior of heavy metals in CO2-carbonated backfill material (CCB), posing potential environmental risks. To address this, this study develops a novel CCB using locally sourced solid waste and high-salinity mine water from a certain mining area, and proposes a control strategy involving rice husk biochar (RHB), natural clinoptilolite (NCP), sepiolite (SEP), and γ-phase nano-Al2O3 (NA) as a synergistic modifier. Through static and dynamic leaching tests, combined with ICP-MS, SEM, XRD, and FTIR analysis, this study investigates the leaching behavior of heavy metals before and after modification, while evaluating the mechanical properties and other parameters to assess the engineering applicability of the modified CCB. The results indicated that high-salinity environments (rich in SO42− and Cl−) promoted the expansion and cracking of CCB, increasing the risk of heavy metals leaching. However, the introduction of the modifiers effectively suppressed the leaching of Cr, Ni, Mn, and other heavy metals through mechanisms such as electrostatic attraction, precipitation, ion exchange and complexation. Among the single-modifier systems, SEP demonstrated the most significant effect, reducing the average leaching of heavy metals by approximately 75.78%, though limitations remained for elements like Cr. The synergistic modification system ensures all detected heavy metals concentrations are below the Class IV groundwater quality standard (based on TCLP results), while enhancing porosity and adsorption capacity without sacrificing mechanical properties. The addition of NA enhanced mechanical strength but its effectiveness in controlling heavy metals leaching varied depending on the accompanying modifiers.
1. Introduction
Coal-fired power generation, blast furnace ironmaking, and other industrial processes produce large amounts of solid waste, such as fly ash (FA) and blast furnace slag (SG).1 Given the pozzolanic potential of these waste materials, some researchers have suggested using them as substitutes for cement to produce backfill materials that can address surface subsidence issues caused by coal mining, while also reducing the high carbon emissions associated with cement production.2 Additionally, the disposal of power plant flue gases, represented by CO2 emissions, is also a pressing concern.3 As the carbon neutrality strategy progresses, attention has turned to the feasibility of using cementitious backfill materials to create an alkaline environment for capturing and storing CO2, such as the preparation of CO2 carbonated coal-based solid waste backfill (CCB).4 However, the use of such backfill materials faces limitations due to locally sourced raw materials (such as solid waste, flue gases, and mine water), resulting in significant uncertainty regarding their physicochemical properties and stability. This makes it challenging to accurately assess their potential environmental impact. In terms of raw materials, both FA and SG are solid waste products of high-temperature pyrolysis processes and generally present a high risk of heavy metals leaching (e.g., Cr, Ni, Cu).5 Notably, the leaching behavior of heavy metals from different coal types varies significantly after high-temperature pyrolysis. Since these heavy metal pollutants are difficult to degrade in the natural environment, they may migrate and accumulate through water cycles and the food chain, ultimately entering the human body. Long-term accumulation could pose health risks, including carcinogenesis or respiratory system damage.6Prior to this study, a significant amount of research has been conducted on the environmental stability of backfill materials before CO2 carbonation. For instance, Yang et al.7 employed the horizontal vibration method to investigate the leachability of heavy metals from magnesium-coal slag solid waste backfill materials, finding that the hardening of the slurry effectively controlled the leaching to levels below the Class III groundwater quality standard. However, in a study of similar coal-based solid waste backfills, Long et al.8 discovered that the leaching of Ni exceeded the Class III standard limit. Their findings also suggested that pH played a critical role in the leaching of heavy metals. Additionally, Hu et al.9 studied the leaching behavior of coal fly ash-based geopolymers and found that although the geopolymer effectively immobilized heavy metals such as Fe, Cr, As, Cd, and Ti in FA, its control over Mn and Ni was limited. These results indicate that for cementitious backfill systems, differences in raw materials, reaction conditions, and even testing methods can lead to significant variations in the leaching behavior of heavy metals. Building on this, the introduction of coal-fired flue gases, represented by CO2, is likely to reduce the overall pH of the reaction system,10 increasing the uncertainty of heavy metals leaching from the CCB. Furthermore, in major mining areas in northwestern China, particularly in Xinjiang, the salinity of mine water is exceptionally high, with mineralization levels often exceeding 5000 mg L−1 and reaching up to 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 mg L−1 in some regions, classifying it as high-salinity mine water.11 Whether the continuous exposure of backfill materials to high-salinity mine water will exacerbate or mitigate the leaching risk of heavy metals requires further investigation.
000 mg L−1 in some regions, classifying it as high-salinity mine water.11 Whether the continuous exposure of backfill materials to high-salinity mine water will exacerbate or mitigate the leaching risk of heavy metals requires further investigation.
Currently, traditional methods for controlling heavy metals leaching from solid waste mainly rely on chemical stabilizers or physical coating technologies, such as direct precipitation.12–14 However, these methods not only have limited control effectiveness but may also pose new environmental risks.15 Particularly in high-salinity environments, the effectiveness of heavy metals fixation relying on precipitation is easily disturbed, leading to reduced stability efficiency. In contrast, porous materials, such as biochar and clay, have shown great potential in the field of heavy metals immobilization due to their excellent adsorption properties, ion exchange capacity, and structural stability.16 Biochar, a carbon-rich solid produced by the anaerobic pyrolysis of plant biomass, is particularly effective in adsorbing and immobilizing heavy metals due to its rich functional groups and porous structure. However, its high carbon content may suppress the expression of pozzolanic activity, thus hindering the improvement of backfill strength.17 This contradiction limits its widespread use in cementitious backfill materials. Notably, the pozzolanic activity of biochar is closely related to its raw material. For example, Liu et al.18 found that bamboo-derived biochar exhibited poor pozzolanic reactivity, while Chen et al.19 reported that rice husk biochar, even with a high fixed carbon content, still demonstrated pozzolanic activity. Moreover, rice husk biochar is abundantly available, with an estimated global annual production of 28 million tons, making it a promising material for application.20 Clay materials are another class of widely studied adsorbents.21 Natural zeolites, such as clinoptilolite, are abundant and inexpensive, with their regular pore structure and exchangeable cations (e.g., Na+, K+, Ca2+) providing excellent ion adsorption and exchange capacity. Studies show that their adsorption performance and structural stability are closely related to the Si/Al ratio.22 A lower Si/Al ratio typically corresponds to higher adsorption capacity but poorer structural stability, while a higher Si/Al ratio improves stability but may result in some loss of adsorption capacity, making it difficult to balance both properties. Furthermore, sepiolite (Mg4[Si6O15](OH)2·3H3O), a fibrous hydrated magnesium silicate, has also been widely studied for its adsorption capacity. Unlike the regular pore structure of natural clinoptilolite, sepiolite has a layered structure where interlayer cations and water molecules can participate in ion exchange. Its surface is also rich in hydroxyl functional groups, which contribute to its strong adsorption ability.23
It is important to note that the introduction of porous materials often alters the pore structure and pore size distribution of the backfill, which can lead to a decrease in mechanical strength and overall stability.24 This degradation compromises the structural integrity and long-term durability of the backfill, thus limiting the widespread application of porous materials in backfill modification. Studies have shown that nanoparticles, due to their nanoscale particle size and extremely high specific surface area, exhibit significant nanofilling, bridging, and nucleation effects,25 which may help to compensate for the structural deficiencies of porous materials. Among various nanoparticles, nano-Al2O3 has attracted significant attention in the field of cement-based material modification due to its high stability and excellent mechanical properties.26 However, its pore-filling behavior may reduce the availability of active sites, thus weakening its adsorption capacity for heavy metals and limiting its applicability. It is also important to distinguish that the performance of nanoparticles is closely related to their crystalline structure. Compared to α and δ phases, γ-phase nano-Al2O3 has a higher specific surface area and more abundant surface active sites due to its loose and porous mesoporous structure.27,28 This suggests that γ-phase nano-Al2O3 may not only maintain the mechanical strength and structural stability of the backfill but also retain excellent adsorption properties for heavy metals.
Therefore, this study aims to take the Hami Dananhu No. 1 Mine in Xinjiang as a case study. By using locally sourced materials—fly ash, slag, and high-salinity mine water, the study will employ CO2 carbonation technology to prepare CO2 carbonated coal-based solid waste backfill (CCB) and systematically evaluate their heavy metals leaching behavior. On this basis, the study will explore the control effects of synergistic modifiers, including rice husk biochar, natural clinoptilolite, sepiolite, and γ-phase nano-Al2O3, on the leaching behavior of the CCB. Additionally, the mechanical properties and long-term stability of the modified backfill will be comprehensively assessed to evaluate its engineering applicability. This research will provide a theoretical basis and research methodology for controlling heavy metals pollution and promoting the resource utilization of solid waste in CO2 carbonated coal-based solid waste backfill under high-salinity mine water conditions.
2. Materials and methods
2.1. Materials
To further investigate the fundamental properties of the raw materials and modifiers, X-ray diffractometry (XRD, Puxi General XD-6), laser particle size analysis (PSA, Malvern Mastersizer 3000), and field emission scanning electron microscopy (SEM, ZEISS Sigma 300) were used to analyze the phase composition, particle size distribution, and microstructure of them, respectively. All test results are presented in Fig. 1. Additionally, the specific surface area, total pore volume, and average pore diameter were measured by an automated specific surface area and porosity analyzer (BET, Micromeritics ASAP 2460) (The N2 adsorption–desorption isotherms are shown in SI Fig. S1). The oxide composition of the material was determined via X-ray fluorescence spectroscopy (XRF, Rigaku ZSX Primus III+). The XRF results, together with the materials' basic characteristics including morphology, mesoporous structure, and particle size, are summarized in Table 1.
 | ||
| Fig. 1 Characterization of the raw materials and modifiers. (a) XRD; (b) particle size distribution; (c) appearance morphology; (d) SEM; (i) FA; (ii) SG; (iii) RHB; (iv) NCP; (v) SEP (vi) NA. | ||
| FA | SG | RHB | NCP | SEP | NA | |
|---|---|---|---|---|---|---|
| a LOI: loss on ignition. | ||||||
| Basic properties | ||||||
| Color | Dark grey | Light grey | Black | Light yellow | Yellowish white | White |
| Morphology | Spherical | Blocky | Porous | Laminar | Fibrous | Spherical |
| BET surface area (m2 g−1) | 0.97 | 1.06 | 86.11 | 19.69 | 229.29 | 140.59 |
| Single point adsorption total pore volume of pores (cm3 kg−1) | 2.19 | 3.73 | 57.91 | 45.42 | 438.13 | 837.47 |
| t-Plot micropore volume (cm3 kg−1) | 0.21 | 0.07 | 35.42 | 4.53 | 42.68 | 6.74 |
| BJH desorption average pore diameter (4V/A) (nm) | 11.16 | 14.84 | 4.15 | 13.30 | 12.10 | 19.30 |
| Proportion of particle sizes below 10 µm (%) | 31.73 | 46.58 | 7.45 | 30.14 | 6.68 | 100.00 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||||
| Chemical composition (wt%) | ||||||
| CaO | 2.08 | 39.49 | 0.60 | 4.59 | 0.38 | — |
| SiO2 | 53.43 | 26.92 | 41.12 | 64.61 | 58.01 | — |
| Al2O3 | 31.93 | 16.23 | 0.21 | 12.78 | 1.74 | 99.99 |
| Fe2O3 | 3.36 | 0.99 | 0.15 | 1.65 | 0.42 | — |
| K2O | 1.29 | 0.51 | 0.96 | 3.80 | 0.56 | — |
| TiO2 | 1.18 | 1.14 | 0.01 | 0.20 | 0.08 | — |
| MgO | 0.53 | 9.93 | 0.33 | 1.01 | 24.15 | — |
| P2O5 | — | 2.12 | 0.36 | 0.02 | — | — |
| SO3 | 0.53 | 2.12 | 0.07 | 0.04 | — | — |
| P2O3 | 0.18 | 0.05 | — | — | — | — |
| Na2O | — | — | 0.17 | 0.65 | 0.24 | — |
| Others (including other trace oxides and LOI) | 5.49 | 2.62 | 56.02 | 10.65 | 14.42 | 0.01 |
The results indicate that the phase composition of FA primarily consists of mullite, quartz, and a small amount of hematite, which is consistent with the higher contents of Al2O3, SiO2, and Fe2O3 observed in the XRF analysis. SG mainly consists of quartz, calcite, and dicalcium silicate (C2S), and a typical broad diffraction peak is observed around 2θ = 30°, suggesting the presence of a large amount of amorphous glassy phase. Its oxides are predominantly CaO, SiO2, Al2O3, and a small amount of MgO. The main structure of RHB is amorphous, with only the crystal diffraction peak corresponding to quartz observed around 44°, indicating the presence of crystalline SiO2. The main phases of NCP, SEP, and NA correspond to clinoptilolite, sepiolite, and γ-Al2O3, respectively, while the other small peaks are attributed to quartz and calcite. XRF results show that NCP is rich in SiO2 and Al2O3, while SEP is mainly composed of SiO2 and MgO. The particle size distribution follows the trend of RHB > SEP > NCP > FA > SG, with SG having the finest particle size, where D90 is only 28.10 µm, and NCP also contains a considerable amount of smaller particles. In terms of microstructure, FA exhibits typical spherical particles, and NA is also spherical. SG and NCP mainly exhibit blocky or laminar structures, while SEP presents a clear fibrous morphology. The RHB particles are relatively larger, with a large number of pores of about 10 µm visible on the surface, exhibiting a loose and porous structure. As shown in Table 1, except for RHB, the micropore content (t-plot method) of the other modifiers is below 10%, confirming that their pore structure is primarily mesoporous. This also indicates that the mesopores in RHB are relatively scarce compared to its macropores and micropores.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 017 mg L−1, with an electrical conductivity of 46
017 mg L−1, with an electrical conductivity of 46![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 100 µS cm−1, characteristic of typical high-salinity mine water. Other relevant ion concentrations and parameters are detailed in the table.
100 µS cm−1, characteristic of typical high-salinity mine water. Other relevant ion concentrations and parameters are detailed in the table.
| Test item (common ions) | Test result (mg L−1) | Test item (heavy metals) | Test result (mg L−1) | Other test items | Test result | |
|---|---|---|---|---|---|---|
| Cations | K+ | 32.10 | Cr | — | pH | 7.33 |
| Na+ | 9061.00 | Ni | — | TDS (mg L−1) | 29![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 017.00 017.00 |
|
| Ca2+ | 705.00 | Cu | — | Alkalinity (mg L−1) | 258.00 | |
| Mg2+ | 404.00 | Zn | — | Total hardness (mg L−1) | 3427.00 | |
| NH4+ | 6.20 | As | — | Conductivity (µS cm−1) | 46![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 100.00 100.00 |
|
| Anions | HCO3− | 315.20 | Cd | — | Suspended solids (mg L−1) | — |
| Cl− | 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 210.00 210.00 |
Pb | — | Turbidity (NTU) | — | |
| SO42− | 6437.00 | Be | — | Color | None | |
| CO32− | 0.00 | Ag | — | Odor & taste | None | |
| NO2− | 0.04 | Mn | — | Visible substances | Precipitate | |
2.2. Preparation
The preparation of CCB was optimized following the GB/T 17671-2021,29 with material ratios detailed in Table 3. Initially, the raw materials and modifiers were thoroughly dry blended at low speed for 30 seconds to ensure uniform solid-phase distribution. Separately, SS and SH were dissolved in the deionized water and stirred at 500 rpm for 10 min to prepare the alkaline activator. The solid mixture was then combined with the liquid phase and transferred to a reactor, with continuous CO2 injection at a rate of 1 L min−1. After reaching the designated time, the fresh slurry was poured into a Φ 50 mm × 100 mm cylindrical mold and allowed to cure at room temperature for 48 h before being demolded. The sample was then transferred to a standard curing chamber (temperature 20 ± 2 °C; humidity 95 ± 2%) for curing.| No. | Solid mass percentage | Solid/wt% | Liquid mass percentage | Liquid/wt% | CO2/min | Modifier | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| FA/wt% | SG/wt% | DW/wt% | HSW/wt% | SS/wt% | SH/wt% | RHB/wt% | NCP/wt% | SEP/wt% | NA/wt% | ||||
| CCB | 80 | 20 | 70 | 46.25 | 46.25 | 5 | 2.5 | 30 | 20 | — | — | — | — |
| CCB-R | 80 | 20 | 67 | 46.25 | 46.25 | 5 | 2.5 | 30 | 20 | 3.00 | — | — | — |
| CCB-N | 80 | 20 | 67 | 46.25 | 46.25 | 5 | 2.5 | 30 | 20 | — | 3.00 | — | — |
| CCB-S | 80 | 20 | 67 | 46.25 | 46.25 | 5 | 2.5 | 30 | 20 | — | — | 3.00 | — |
| CCB-RN | 80 | 20 | 67 | 46.25 | 46.25 | 5 | 2.5 | 30 | 20 | 1.50 | 1.50 | — | — |
| CCB-RS | 80 | 20 | 67 | 46.25 | 46.25 | 5 | 2.5 | 30 | 20 | 1.50 | — | 1.50 | — |
| CCB-Rn | 80 | 20 | 67 | 46.25 | 46.25 | 5 | 2.5 | 30 | 20 | 2.90 | — | — | 0.10 |
| CCB-RNn | 80 | 20 | 67 | 46.25 | 46.25 | 5 | 2.5 | 30 | 20 | 1.45 | 1.45 | — | 0.10 |
| CCB-RSn | 80 | 20 | 67 | 46.25 | 46.25 | 5 | 2.5 | 30 | 20 | 1.45 | — | 1.45 | 0.10 |
2.3. Methods
Following filtration, the leachate samples were immediately subjected to aqua regia digestion (HNO3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) HCl = 1
HCl = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3) to prevent metal precipitation and match the calibration standards. Specifically, the samples were heated in a water bath with 5 mL of aqua regia until clear, cooled to 60 °C, and diluted to 25 mL with deionized water. The concentrations of target heavy metals were determined using an ICP-MS (Agilent 7800, USA) operating in kinetic energy discrimination (KED) mode to effectively eliminate polyatomic interferences. Quantification was performed using an external standard method with internal standard correction. Multi-element standard solutions, ranging from 0 to 500 µg L−1, were used to establish calibration curves, all of which exhibited excellent linearity (R2 > 0.999). To compensate for matrix effects and instrumental drift, 103Rh and 185Re were employed as internal standards. Detailed methodological parameters, including the standard series, limits of detection (LOD), limits of quantification (LOQ), and raw calibration data, are provided in SI Table S1.
3) to prevent metal precipitation and match the calibration standards. Specifically, the samples were heated in a water bath with 5 mL of aqua regia until clear, cooled to 60 °C, and diluted to 25 mL with deionized water. The concentrations of target heavy metals were determined using an ICP-MS (Agilent 7800, USA) operating in kinetic energy discrimination (KED) mode to effectively eliminate polyatomic interferences. Quantification was performed using an external standard method with internal standard correction. Multi-element standard solutions, ranging from 0 to 500 µg L−1, were used to establish calibration curves, all of which exhibited excellent linearity (R2 > 0.999). To compensate for matrix effects and instrumental drift, 103Rh and 185Re were employed as internal standards. Detailed methodological parameters, including the standard series, limits of detection (LOD), limits of quantification (LOQ), and raw calibration data, are provided in SI Table S1.
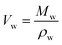 | (1) |
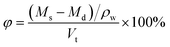 | (2) |
The microstructure and elemental composition were examined by field emission scanning electron microscopy (SEM, ZEISS Sigma 300) equipped with energy dispersive spectroscopy (EDS, OXFORD X-max 50). Samples were dried and mounted on conductive adhesive tape. Observations were made under high vacuum at 2 kV accelerating voltage.
3. Results
3.1. Immobilization behavior of heavy metals in modified samples
Comparative analysis revealed that FA had significantly higher leaching concentrations of Cr, Ni, Cu, Zn, As, Cd, Pb, and Be than SG (p < 0.05). Excluding Cd, the average leaching levels of these elements in FA were approximately 57 times higher than in SG, with more elements exceeding regulatory limits. Although only Ag and Mn in SG exhibited higher leaching than in FA—Mn being roughly four times higher—the absolute leaching concentrations of these two elements in SG were substantially greater by 2238.20 µg L−1 and 5703.39 µg L−1, respectively. These differences likely arise from the distinct origins and physicochemical properties of the materials. Overall, considering both the number of elements and concentrations exceeding III-GQS and IV-GQS thresholds, FA poses a higher leaching risk than SG.
In the single-modifier systems, the leaching concentrations of most heavy metals are lower than those of the unmodified group. However, its control of certain elements remains a worse effect. For example, RHB failed to reduce the Mn concentration below the IV-GQS limit, and the leaching concentration of As even rose to 34.42 ± 2.54 µg L−1.33 Only the introduction of NCP exhibited a notably weaker immobilization capacity for Ag. By contrast, SEP used alone showed the most pronounced improvement. The leaching concentrations of Ni, Cu, Zn, As, Cd (below the LOD), Pb, Be (below the LOD), Ag, and Mn decreased by approximately 74.29%, 84.21%, 89.80%, 35.09%, 97.95%, 81.67%, 100.00%, 88.17%, and 76.68%, respectively (p < 0.05). Nevertheless, SEP alone still showed a limitation in immobilizing Cr, with its leaching concentration of 101.22 ± 4.25 µg L−1 slightly exceeding the IV-GQS limit for Cr (100 µg L−1). Overall, SEP alone reduced the average leaching of heavy metals by approximately 75.78% (p < 0.05). On this basis, although the combined use of modifiers to some extent reduces the control efficiency for certain heavy metals compared with the best-performing single modifier, it still maintains their leaching concentrations below the IV-GQS limits, while compensating for the inadequate control of specific heavy metals by individual modifiers. For instance, in the CCB-RN system, the synergistic use of RHB and NCP overcame the insufficient control of Mn, As, and Ag observed when each is used alone, and further enhanced the immobilization of Be, reducing its leaching concentration to below the III-GQS limit. In the CCB-RS group, although the fixation of Ni, Cu, and Zn was slightly inferior to that achieved with SEP alone, a synergistic enhancement was observed for Cr and Ag. The leaching concentration of Cr decreased by 48.52% and 83.18% relative to CCB-R and CCB-S, respectively (p < 0.05), and was reduced from exceeding the IV-GQS limit to within the III-GQS range.
Previous studies have shown that the adsorption capacity of materials is closely related to the adsorption sites provided by their rich pore structure.34 Considering that the addition of NA fills the pores within the material, it is likely to weaken the adsorption capacity of the CCB for heavy metals. However, when comparing CCB-R and CCB-Rn, the introduction of NA further reduced the leaching concentrations of Cr, Cu, Zn, As, Cd, Pb, Be, and Mn in CCB-RN by 52.36%, 59.17%, 44.72%, 21.98%, 7.96%, 28.18%, 90.13%, and 28.52% respectively on average (p < 0.05). However, in the CCB-RNn and CCB-RSn systems, the leaching concentrations of Cr, Ni, Ag, and Mn increase compared to CCB-RN and CCB-RS, with the increase being more significant in CCB-RNn. This suggests that when NA is combined with RHB, it not only does not weaken the immobilization effect but actually enhances the suppression of certain heavy metals. However, when NA is used in combination with clay materials (such as NCP and SEP), the immobilization effect is weakened. This may be because NA has less impact on the overall pore structure when combined with RHB, but when combined with clay materials, the pores between the different materials are more likely to fill each other, leading to a reduction in the total porosity. The damage to the pore structure of NCP is particularly noticeable, which weakens the overall immobilization capacity of the system for heavy metals.
3.2. Evolution of macroscopic properties and solution pH of modified backfill
Fig. 4 shows the changes in the UCS, porosity, and solution pH over time for different modified backfill during high-salinity water soaking. Detailed experimental data are provided in SI Tables S3–S5, respectively. | ||
| Fig. 4 Evolution of macroscopic properties and solution pH of modified backfill: (a) UCS; (b) porosity; (c) solution pH. | ||
According to Fig. 4a, all samples initially experienced a strength decrease due to slight surface dissolution.35 As continued soaking, most samples showed gradual strength recovery, suggesting that cations like Ca2+ and Mg2+ provided by high-salinity water promoted further hydration. Notably, CCB and CCB-N underwent significant strength loss after 60 d (−11.62% and −7.84%, respectively, p < 0.05), likely caused by expansive products formed from high ion concentrations, leading to micro-cracking.36 In contrast, samples modified with RHB, SEP, or NA maintained strength growth, reflecting superior long-term durability. The impact of RHB and NCP on strength is more complex. At 3 wt% RHB content, the backfill showed an average strength decrease of 5.91% across all soaking periods compared with CCB (p < 0.05), whereas reducing the dosage to 1.5 wt% led to a strength increase. This can be attributed to the aggregation effect caused by excessive biochar, which creates weak zones within the CCB.37 NCP similarly reduced strength, further influenced by its low pozzolanic reactivity due to higher stable crystalline SiO2 content.38 The introduction of SEP significantly increased the backfill UCS by an average of 51.3% across all soaking periods compared with CCB (p < 0.05). Furthermore, its UCS continued to increase even after 60 d of soaking, reaching 10.07 ± 0.16 MPa. This remarkable long-term durability is owing to its high surface area that supports continuous hydration, and its fibrous straw-bundle structure which forms an interwoven network to disperse stress.39 Introducing NA further improved the strength of the three sample groups (e.g., CCB-RSn averaged 15.98% higher than CCB-RS across all soaking periods, p < 0.05). This enhancement is likely due to the nanomaterial's filling effect and its direct participation in hydration to promote gel formation.40
Porosity tests (Fig. 4b) show that all samples experienced a temporary increase in porosity during the initial immersion phase due to the rapid dissolution of soluble salts. Subsequently, as hydration gels continuously formed, the structure became denser, leading to a gradual decrease in porosity.41 Compared to the CCB, all modified samples showed a significant increase in porosity. SEP had the most significant impact on porosity of CCB. As shown in Table 1, the total pore volume of SEP is second only to that of NA, and its particles are relatively coarse (with only 6.68% of particles below 10 µm), making it difficult to embed within the larger pores of RHB, leading to a relatively independent distribution. Additionally, its average pore diameter is only 12.10 nm, much smaller than the average particle size of the raw materials and NA (average particle size: 40 nm), which would require larger filling space. This allows the high pore volume of SEP to be effectively retained, significantly increasing the overall porosity. When NCP was used alone for modification, a similar mechanism was observed. However, its finer particles (30.14% below 10 µm) could more easily fill the larger pores of RHB, reducing its overall contribution to the porosity. After the NA introduction, the porosity of the samples (CCB-Rn, CCB-RNn, CCB-RSn) decreased by an average of 1.50%, 1.63%, and 1.78% across all soaking periods compared with non-NA groups (p < 0.05) due to the nanoparticles filling pores and promoting densification.42
The changes in solution pH (Fig. 4c) further reveal the reaction process within the system. All samples showed a rapid pH increase during the initial soaking period (0–7 d), rising from 7.33 to about 11.00, primarily due to the rapid dissolution of cementitious materials releasing a large amount of OH−.35 Subsequently, the pH gradually declined due to the ongoing consumption of OH− by hydration. To verify that this pH decline was not caused by atmospheric CO2 interference, supplementary acid–base-titration experiments were conducted, which confirmed that internal hydration dominated the OH− consumption (titration curves and detailed analyses are provided in SI Fig. S1). Additionally, when pH fell within the range of 8–10, part of carbonates transformed into more soluble bicarbonates that dissociated to release H+, further lowering the pH.43 For FA and SG, the initial pH increase was driven by the rapid dissolution of Ca2+ and Mg2+, forming hydroxides and subsequently releasing OH−. As the reaction progressed, the ionization of OH− reached a limit, and as the pH increased, SiO2 and Al2O3 within the system began to react and consume OH−, forming SiO(OH)3− and Al(OH)4− (key precursors for pozzolanic reactions), which again lowered the pH. The reaction equations are given in eqn (3)–(6). XRF analysis reveals that FA contains less CaO and MgO but more SiO2 and Al2O3 than SG, therefore its pH was lower and declined more rapidly later. In contrast, the different modifiers had little impact on the overall pH evolution of the system.
| CaO + H2O → Ca(OH)2 → Ca2+ + 2OH− | (3) |
| MgO + H2O → Mg(OH)2 →Mg2+ + 2OH− | (4) |
| SiO2 + H2O + OH− → SiO(OH)3− | (5) |
| Al2O3 + 3H2O + 2OH− → 2Al(OH)4− | (6) |
3.3. Heavy metal immobilization mechanisms in modified backfill
 | ||
| Fig. 5 XRD patterns of each group of samples. (a) CCB; (b) CCB-R; (c) CCB-N; (d) CCB-S; (e) CCB-RN; (f) CCB-RS; (g) CCB-Rn; (h) CCB-RNn; (i) CCB-RSn. | ||
Hydration gels, mainly C-S-H, are the primary products from the CCB hydration. After carbonation, the decreased Ca/Si ratio promotes Al incorporation into the gel network, yielding the C-A-S-H gel phase (2θ = 54.5°).41 In high-salinity water, ions exchange and adsorption with cations (e.g., Na+, Mg2+) further transform the gels into N-A-S-H and N-M-A-S-H,35 with characteristic peaks at 2θ = 31.1°, 33.1°, and 50.1°. After modification, the peak intensities of the two new gel phases increased slightly, especially in the SEP group, due to its high Mg content. A broad hump peak at 2θ = 20–35° indicates the presence of amorphous silica gel, which exhibits higher reactivity SiO2. Carbonate phases—magnesian calcite, magnesite, and calcite—appear at 2θ = 29.5°, 43.0°, and 39.3°, respectively. Gypsum (2θ = 20.7°) and Friedel's salt (2θ = 11.3°), a layered double hydroxide (LDH), also arise form under high Cl− and SO42− concentrations. Additionally, meixnerite, identified at 2θ = 60.8°, is another Mg–Al hydrotalcite with a bilayer structure.44 No significant phase differences were observed with different modifiers, likely owing to their high stability and low addition.
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C/C
C/C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O and C–H bonds,51 respectively, suggesting the introduction of oxygen-containing functional groups such as –COOH. These functional groups are beneficial for mitigating Heavy metals leaching.
O and C–H bonds,51 respectively, suggesting the introduction of oxygen-containing functional groups such as –COOH. These functional groups are beneficial for mitigating Heavy metals leaching.
 | ||
| Fig. 6 FTIR spectral of each group of samples. (a) CCB; (b) CCB-R; (c) CCB-N; (d) CCB-S; (e) CCB-RN; (f) CCB-RS; (g) CCB-Rn; (h) CCB-RNn; (i) CCB-RSn. | ||
Two absorption peaks within 400–800 cm−1 are assigned to M–O vibrations, where M may include Mg, Al, Ca, or heavy metals (HMs). Specifically, a comparison between the Dried group and the Immer-3 or Immer-60 groups reveals that these M–O peaks undergo corresponding shifts in position and intensity throughout the immersion period. This phenomenon indicates that Mg(Al, Ca)–O bonds are being transformed into HMs–O bonds through chemical adsorption and surface complexation,49 thereby providing direct evidence for the “re-adsorption” of heavy metals. This process is further facilitated by the abundant oxygen-containing functional groups (e.g., –OH and –COOH) introduced by the modifiers, particularly biochar, which serve as active sites for trapping heavy metal ions. These slight shifts in peak positions and intensities after modification and immersion in high-salinity solutions reflect changes in stretching or bending vibrations, indicative of the active immobilization of heavy metals within the CCB matrix.
 | ||
| Fig. 7 SEM-EDS images of each group of samples under dry conditions. (a) CCB; (b) CCB-R; (c) CCB-N; (d) CCB-S; (e) CCB-RN; (f) CCB-RS; (g) CCB-Rn; (h) CCB-RNn; (i) CCB-RSn. | ||
| Reaction type | Reaction equations |
|---|---|
| Hydration reaction | XCa2+ + Y[SiO(OH)3]− + H2O + (Y − 2X)OH− → C-S-H |
| XCa2+ + Y[Al(OH)4]− + Z[SiO(OH)3]− + H2O + (Y + Z − 2X)OH− → C-A-S-H | |
| XNa+ + Y[Al(OH)4]− + Z[SiO(OH)3]− + H2O + (Y + Z − X)OH− → N-M-A-S-H | |
| XNa+ + YMg2+ + Z[Al(OH)4]− + W[SiO(OH)3]− + H2O + (Z + W − X − 2Y)OH− → N-M-A-S-H | |
| Carbonation reaction | Ca2+ + 2OH− + CO2 → CaCO3 + H2O |
| Mg2+ + 2OH− + CO2 → MgCO3 + H2O | |
| Reaction under high-salinity conditions | Ca2+ + SO42− + 2H2O → CaSO4·2H2O |
| 2[Al(OH)4]− + 4Ca2+ + 2Cl− + 4H2O + 4OH− → 3CaO·Al2O3·CaCl2·10H2O |
Fig. 8 illustrates the microstructural evolution of five sample groups (unmodified, –RHB, –NCP, –SEP–, and –NA) to clarify modifier effects. The overall trend shows progressive pore closure with soaking, consistent with the previous porosity evolution. During immersion, C-S-H gels interact with cations to form more complete hydration phases. Simultaneously, carbonates formed via carbonation, along with gypsum and Friedel's salt generated under anionic erosion, tend to aggregate through phase transformation-driven processes. These compounds nucleate and precipitate in crystalline forms, further developing within the porous structure.53 Interestingly, although gypsum expansion is often associated with reduced durability and cracking in cement-based materials,54 the UCS results indicate that the strength of the most samples increased with soaking time. This is attributed to the porous property of the modifiers, where abundant pore space accommodates expansion stresses. Nevertheless, microcracks observed in CCB and CCB-N at 60 d (Fig. 8(a-3) and (c-3)) correspond to their strength drop, indicating that the mesoporous structure of NCP provides insufficient space to accommodate expansion. In contrast, SEP-modified samples maintained strength improvement due to their robust fibrous structure (Fig. 8d).
 | ||
| Fig. 8 Microstructure evolution of samples with soaking duration. (a) CCB; (b) CCB-R; (c) CCB-N; (d) CCB-S; (e) CCB-Rn; (1) dried; (2) Immer-3; (3) Immer-60. | ||
It should be noted that although the reduction in porosity improves the strength of the sample, it also reduces the contact opportunities between the porous material and heavy metals in the solution, which may potentially lower its adsorption effect on heavy metals. At this point, the superiority of substances with rich mesoporous structures represented by NCP and SEP becomes prominent. Compared with macroporous structures which are more readily filled, mesoporous structures often exhibit more outstanding performance in reducing diffusion resistance, increasing adsorption rate and providing greater adsorption capacity.55 This well explains the better adsorption effect of NCP and SEP compared to RHB when facing most heavy metals.
What's more, although the macroscopic porosity slightly decreased by 1.49–1.78% due to the filling effect of nano-Al2O3 (Fig. 4b), this physical loss is functionally compensated by chemical enhancement. Specifically, by comparing SEM images in Fig. 8e and b, the introduction of NA further refined the pore structure. EDS surface scans (Fig. 7) also reveal the distribution of Al in the NA-modified groups is more uniform and denser. Moreover, under the influence of Al, the distribution of Na, Mg, Si, Ca, and O also shows this uniform distribution. These observations quantitatively indicate that NA acts as effective nucleation sites, facilitating the continuous formation and homogenization of abundant hydration gels (e.g., C-A-S-H). Consistently, TCLP results confirmed that NA addition does not diminish overall heavy metals adsorption. The increase of these active gel phases provides abundant negatively charged sites, which further contribute to heavy metals adsorption via electrostatic attraction.56 Crucially, as corroborated by the FTIR spectra, this gel proliferation also introduces a massive number of oxygen-containing functional groups (e.g., hydroxyls and silanols) that act as robust chemical complexation sites. Together, these chemical enhancements effectively resolving the apparent paradox between the reduced total porosity and the sustained adsorption capacity.
3.4. Synergistic mechanisms for controlling heavy metal leaching
Under the continuous influence of high-salinity mine water ions, chemical reactions occur within the filling body, resulting in the formation of various reaction products, with gypsum as a representative phase. During gypsum nucleation and crystallization, its intrinsic expansive properties may induce cracking of the sample upon prolonged exposure to SO42−. This cracking increases the contact area between the sample and high-salinity mine water, thereby enhancing the leaching concentration of heavy metals such as Mn, Cr, and Ni. Moreover, cations such as Na+, present at the highest concentration, actively participate in cations exchange processes by replacing Ca2+ in the gel system. This substitution weakens the cementation ability of the gel, further promoting heavy metals leaching.52 Fig. 9 reveals the mechanism behind the enhanced leaching of heavy metals under high-salinity conditions.Under normal conditions, the CCB can initially achieve the immobilization of heavy metals through CO2 sequestration and the formation of gels. SEM results indicate that this immobilization is primarily achieved through the physical “encapsulation” effect, where a substantial amount of gels and carbonates enclose the heavy metals within the matrix. However, other characterization results, such as XRD and FTIR, further demonstrate that these materials also exert a “re-adsorption” effect on the leached heavy metals through chemical pathways. On the one hand, under continuous high-salinity water immersion conditions, some leached heavy metals react with OH−, SO42−, or CO32− introduced into the system,14 forming insoluble precipitates (such as Cr(OH)3, PbCO3, etc.) (eqn (7)–(9)). Notably, the actual solubility of these heavy metal precipitates encompasses not only simple ionic dissolution but also the dissolution of various mononuclear and polynuclear hydroxyl complexes as well as neutral molecules.57,58 Since these dissolution modes exhibit different dependencies on the solution pH, the alkaline environment established at varying pH levels, influenced by the CCB hydration process, exerts a crucial impact on the solubility of these heavy metal precipitates. Simultaneously, various gels produced in the reaction system form negatively charged Si–O− groups under alkaline conditions, which can physically attract positively charged heavy metals through electrostatic attraction. Furthermore, metal cations carried by the gels can undergo ions exchange with heavy metals (eqn (10)).59
However, the TCLP results indicate that relying solely on the above mechanisms is still insufficient for effective immobilization of heavy metals. The introduction of modifiers for synergistic modification significantly enhances the overall immobilization effect. As shown by XRD and SEM results, the introduction of modifiers, with their abundant pore structures and larger specific surface areas, further increases CO2 adsorption and gel formation.60 The aforementioned processes—precipitation, ions exchange, and electrostatic attraction—are all effectively enhanced. Additionally, FTIR results show that the introduction of modifiers also brings a variety of functional group structures, such as –OH and –COOH. These surfaces generally carry negative charges, and such negatively charged functional groups (–COO−, –O−) can effectively adsorb cationic heavy metals like Cr3+ and Ni2+. Moreover, the functional groups exhibit strong complexation capabilities with heavy metals in the solution, forming stable chemical bonds through oxygen-containing functional groups on the surface (eqn (11) and (12)). Among them, RHB (rich in carboxyl and hydroxyl groups) shows a significant effect, while the introduction of NCP and SEP also increases the hydroxyl content of the structure, enhancing the surface complexation potential.
| nOH− + Mn+ → M(OH)n↓ | (7) |
| nSO42− + 2Mn+ → nM2SO4↓ | (8) |
| nCO32− + 2Mn+ → nM2CO3↓ | (9) |
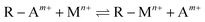 | (10) |
| nR–OH + Mn+ → [(R–O)nM] + nH+ | (11) |
| nR–COOH + Mn+ → [(R–COO)nM] + nH+ | (12) |
In addition to the above mechanisms, the study have also found that the high-salinity environment rich in Mg2+ and Cl− promotes the formation of layered double hydroxides (LDHs) such as meixnerite and Friedel's salt. These LDHs feature bilayer metal hydroxide frameworks composed of divalent and trivalent cations (e.g., Mg2+ and Al3+ in meixnerite; Ca2+ and Al3+ in Friedel's salt), with interlayer anions (OH− or Cl−) and water molecules, all stabilized by ionic and hydrogen bonds. LDHs structures exhibit excellent adsorption properties arising from their unique physicochemical characteristics.61 Generally, both exchangeable cations and anions within LDHs are capable of participating in ions exchange actively. Therefore, the heavy metal cations can replace the cations (such as Mg2+, Al3+, and Ca2+, Al3+) within the LDH framework. For elements with multiple valence states like Cr, its exact form in the solution—whether Cr(III) or Cr(VI)—dictates its potential immobilization pathway. Any present Cr(III) would be favored to precipitate as hydroxides due to the highly alkaline nature of the CCB, whereas the formation of LDHs provides a mechanistically plausible structural pathway for capturing anionic Cr(VI) species (such as CrO42−) via interlayer anion exchange. Additionally, the interlayer anions can also precipitate and complex with heavy metals. The heavy metals control mechanism of modified filling materials in high-salinity environment is illustrated in Fig. 10.
Overall, within the modified CO2-carbonated cementitious backfill system, the immobilization of heavy metals involves both the physical “encapsulation” and the “re-adsorption” of leached heavy metals, forming a dynamic “release-and-readsorption” equilibrium process. Based on the synergistic effect of these multiple mechanisms, the modified CCB, with its unique structure, demonstrates excellent adsorption performance and can effectively suppress the leaching of heavy metals.
4. Discussions
4.1. Evaluation of heavy metals immobilization effect
To further contextualize the effectiveness of the proposed synergistic modification system, SI Table S6 presents the percentage reduction in heavy metal leaching for each modified group compared to the unmodified CCB. Meanwhile, SI Table S7 presents a quantitative comparison of heavy metal immobilization performance between the other representative methods published recently. It is worth noting that while biochar, clay minerals (like sepiolite) and nanomaterials have been extensively utilized in soil remediation and wastewater treatment, their specific application for heavy metal control in solid waste backfill, particularly the CO2-carbonated solid waste backfills under high-salinity conditions, remains a largely unexplored domain. As shown in Table S7, traditional methods in soil or water treatment typically achieve removal or immobilization efficiencies ranging from 21% to nearly 100%, or exhibit specific adsorption capacities (e.g., 12.23–833.33 mg g−1) depending on the material dosage and target environment. In comparison, despite the harsh, continuous high-salinity immersion in our study—a condition that typically exacerbates heavy metal leaching via ion exchange and expansive cracking—our modified CCB system exhibits a better immobilization capability. The synergistic modification method ensures that all the tested heavy metals are controlled within the IV-GQS limit, achieving reduction efficiencies that parallel to those of traditional soil/water treatments (e.g., maintaining 80% to over 95% reduction for key toxic elements like Cr, Ag, and Be in synergistic groups). The results not only highlight the adsorption capacity of the modifiers but also demonstrate the potential of this biochar-clay-nanomaterial synergistic modification strategy in complex, high-salinity underground engineering environments.Furthermore, when evaluating these reduction efficiencies, it is important to emphasize that the rates currently measured for the biochar-modified groups (e.g., CCB-R) actually represent a conservative estimate. Owing to the trace heavy metals inherently present in the raw biochar, its true immobilization efficacy is likely even higher. As evidenced by the experimental results, even within the RHB-inclusive CCB-R group, a substantial decrease in the leaching of heavy metals (with the exception of As) was still observed—for instance, a 77.11% reduction for Cr and a 96.71% reduction for Ag. These findings provide sufficient qualitative evidence that biochar effectively mitigates pollutant leaching from CO2 carbonated coal-based solid waste backfill in high-salinity environments. However, the inevitable presence of inherent heavy metals in biochar poses a methodological challenge. Achieving an accurate quantitative assessment of its pure immobilization capacity without compromising its critical physicochemical properties (e.g., oxygen-containing functional groups) remains a key issue to be addressed in our future research.
4.2. Limitations and future work
While the 60 d static leaching test in this study provides some insights into the short-to medium-term physical durability and heavy metals immobilization behavior of the modified CCB, it has certain limitations regarding long-term ecological risk assessment. In actual underground backfill applications, the backfill is subjected to continuous and dynamic high-salinity groundwater penetration over decades. A static immersion experiment cannot fully simulate these complex hydrodynamic conditions and the consequent long-term geochemical evolution of the hydration products. Therefore, to more accurately simulate environmental exposure, future research should incorporate long-term dynamic leaching tests to assess the release kinetics of heavy metals under continuous flow conditions. Furthermore, coupling these experimental data with geochemical modeling software (e.g., PHREEQC) will be highly recommended to predict the multi-decadal phase transformations and long-term ecological impacts of the synergistic modified CCB in high-salinity environments.Furthermore, while the TCLP method provides a reliable assessment of the maximum leachable toxicity, it cannot identify the specific chemical speciation of the immobilized heavy metals. To address this, future research could consider employing the Tessier 5-step sequential extraction method to analyze the specific occurrence forms of heavy metals. Alongside this speciation analysis, deeper mechanistic investigations are also required. Although Section 3.4 qualitatively classifies the control mechanisms of the modified CCB into physical “encapsulation” and the “re-adsorption” of leached heavy metals, quantitatively distinguishing the exact contribution of each effect to the overall immobilization efficiency remains highly challenging without isotopic-level evidence. Therefore, future work should consider employing isotopic tracing techniques to determine the specific distribution coefficient (Kd) of heavy metal migration, which would allow for a rigorous and quantitative decoupling of the heavy metal migration pathways.
5. Conclusions
This study investigated the leaching of heavy metals from CO2 carbonated coal-based solid waste backfill (CCB) in high-salinity mine water environments, with a focus on the synergistic modification effects of rice husk biochar (RHB), natural clinoptilolite (NCP), sepiolite (SEP), and γ-phase nano-Al2O3 (NA). The leaching behavior and control mechanisms of heavy metals in the modified CCB were elucidated, and the mechanical properties were evaluated to assess its engineering applicability. The main findings are summarized as follows:(1) Mechanical performance was enhanced by over 50% with SEP due to its fibrous structure, whereas RHB exhibited a concentration-dependent effect: it weakened strength at 3 wt% but slightly improved it at 1.5 wt%. NCP reduced strength owing to its low pozzolanic reactivity, which further declined after 60 d of immersion. NA also contributed to strength improvement.
(2) The leaching of heavy metals from CCB under high-salinity environment is primarily driven by the formation of expansive gypsum under high pore water salinity, which induces micro-cracking and facilitates heavy metals release.
(3) Among single-modifier systems, SEP achieved the most pronounced reduction in heavy metals leaching (by an average of 75.78%), though limitations remained for certain elements such as Cr. Synergistic modification effectively compensated for these deficiencies, lowering all heavy metals concentrations below the IV-GQS (based on TCLP results).
(4) The control of heavy metals leaching by synergistic modifiers was attributed to multiple mechanisms, including physical encapsulation, electrostatic attraction, ions exchange, precipitation, and complexation.
Author contributions
Zhishang Zhang: data curation, writing – original draft, visualization, methodology, formal analysis. Liqiang Ma: conceptualization, funding acquisition, project administration. Ichhuy Ngo: supervision, resources. Kunpeng Yu: formal analysis. Jiangtao Zhai: formal analysis. Zezhou Guo: visualization, formal analysis. Zhiyang Zhao: validation, investigation. Chengkun Peng: data curation, conceptualization. Ruizhi Yang: investigation, software.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included in the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5ra09892k.Acknowledgements
The authors appreciate the Xinjiang Uygur Autonomous Region Key R&D Programme Projects (Grant No. 2023B03009-1) for financial support.References
- E. Kaya, B. Ciza, Ç. Yalçınkaya, B. Felekoğlu, H. Yazıcı and O. Çopuroğlu, A comparative study on the effectiveness of fly ash and blast furnace slag as partial cement substitution in 3D printable concrete, J. Build. Eng., 2025, 108, 112841 CrossRef.
- J. Zhang, K. Yang, X. He, X. Zhao, Z. Wei and S. He, Research status of comprehensive utilization of coal-based solid waste (CSW) and key technologies of filling mining in China: A review, Sci. Total Environ., 2024, 926, 171855 CrossRef CAS PubMed.
- M. Fang, N. Yi, W. Di, T. Wang and Q. Wang, Emission and control of flue gas pollutants in CO2 chemical absorption system – A review, Int. J. Greenh. Gas Control, 2020, 93, 102904 CrossRef CAS.
- X. Cao, Q. Wang, M. Li, C. Ma and S. Wang, Research progress on strengthening mechanism and industrial application of CO2 mineralization technology for coal electricity solid waste, Results Eng., 2025, 27, 106593 CrossRef CAS.
- G. V. P. Bhagath Singh and V. Durga Prasad, Environmental impact of concrete containing high volume fly ash and ground granulated blast furnace slag, J. Clean. Prod., 2024, 448, 141729 CrossRef CAS.
- L. Velarde, M. S. Nabavi, E. Escalera, M. Antti and F. Akhtar, Adsorption of heavy metals on natural zeolites: A review, Chemosphere, 2023, 328, 138508 CrossRef CAS PubMed.
- P. Yang, L. Liu, Y. Suo, H. Qu, G. Xie, C. Zhang, S. Deng and Y. Lv, Basic characteristics of magnesium-coal slag solid waste backfill material: Part I. preliminary study on flow, mechanics, hydration and leaching characteristics, J. Environ. Manage., 2023, 329, 117016 CrossRef CAS PubMed.
- K. Long, B. Li, J. Ma, H. Liu and J. Li, Ecological risk analysis of leakage caused by coal-based solid waste backfill slurry bleeding: An experimental study, J. Clean. Prod., 2025, 494, 144993 CrossRef CAS.
- Y. Hu, J. Tang, Y. Fei, J. Shang, P. Liu and Y. Liu, Enhancement of Zn adsorption on coal fly ash-based geopolymer with steel slag incorporation: Leaching behavior and performance insights, Environ. Pollut., 2025, 367, 125639 CrossRef CAS PubMed.
- J. A. Meima, R. D. van der Weijden, T. T. Eighmy and R. N. J. Comans, Carbonation processes in municipal solid waste incinerator bottom ash and their effect on the leaching of copper and molybdenum, Appl. Geochem., 2002, 17, 1503–1513 CrossRef CAS.
- Y. Wang, L. Hu, Y. Zhang and L. Dong, Experimental study on the scaling law of the heat exchange tube surface in the process of low-temperature single-effect distillation of high-mineralized mine water, Desalination Water Treat., 2024, 319, 100571 CrossRef CAS.
- Q. Y. Chen, M. Tyrer, C. D. Hills, X. M. Yang and P. Carey, Immobilisation of heavy metal in cement-based solidification/stabilisation: A review, Waste Manage., 2009, 29, 390–403 CrossRef CAS PubMed.
- L. Chen, K. Nakamura and T. Hama, Review on stabilization/solidification methods and mechanism of heavy metals based on OPC-based binders, J. Environ. Manage., 2023, 332, 117362 CrossRef CAS PubMed.
- B. Guo, B. Liu, J. Yang and S. Zhang, The mechanisms of heavy metal immobilization by cementitious material treatments and thermal treatments: A review, J. Environ. Manage., 2017, 193, 410–422 CrossRef CAS PubMed.
- K. Polipalli, S. Suraboyina, M. Kashimalla and A. Polumati, A review on value addition of Agricultural Residues by Chemical and Bio-chemical Processes to abate environmental pollution, Green Technol. Sustainability, 2025, 3, 100241 CrossRef.
- R. Hou, B. Zhu, L. Wang, S. Gao, R. Wang and D. Hou, Mechanism of clay mineral modified biochar simultaneously immobilizes heavy metals and reduces soil carbon emissions, J. Environ. Manage., 2024, 361, 121252 CrossRef CAS PubMed.
- S. S. Senadheera, S. Gupta, H. W. Kua, D. Hou, S. Kim, D. C. W. Tsang and Y. S. Ok, Application of biochar in concrete – A review, Cem. Concr. Compos., 2023, 143, 105204 CrossRef CAS.
- W. Liu, S. Tan, L. Qing and Y. Li, Pozzolanic activity of biochar with high carbon content and its influence on comprehensive strength-emission performance of biochar-cement composite paste, Constr. Build. Mater., 2025, 478, 141427 CrossRef CAS.
- L. Chen, L. Wang, Y. Zhang, S. Ruan, V. Mechtcherine and D. C. W. Tsang, Roles of biochar in cement-based stabilization/solidification of municipal solid waste incineration fly ash, Chem. Eng. J., 2022, 430, 132972 CrossRef CAS.
- I. Khan, H. A. Saddiqi, F. Ahmad and A. Ullah, Mortar performance enhancement by partially replacement of cement with hybrid biochar: An experimental evaluation and machine learning predictions of compressive strength, water absorption and thermal conductivity, Mater. Chem. Phys. Sustain. Energy, 2025, 3, 100024 Search PubMed.
- S. Xie, L. Huang, C. Su, J. Yan, Z. Chen, M. Li, M. Du and H. Zhang, Application of clay minerals as adsorbents for removing heavy metals from the environment, Green Smart Min. Eng., 2024, 1, 249–261 CrossRef CAS.
- J. Li, M. Gao, W. Yan and J. Yu, Regulation of the Si/Al ratios and Al distributions of zeolites and their impact on properties, Chem. Sci., 2023, 14, 1935–1959 RSC.
- K. Gu, M. Zhu, Q. Song and Z. Jiang, Hydration and microstructure formation of magnesium oxysulfate (MOS) cement regulated by sepiolite with high adsorption ability, Cem. Concr. Compos., 2024, 153, 105728 CrossRef CAS.
- C. Sánchez-Mendieta, J. J. Galán-Díaz and I. Martinez-Lage, Relationships between density, porosity, compressive strength and permeability in porous concretes: Optimization of properties through control of the water-cement ratio and aggregate type, J. Build. Eng., 2024, 97, 110858 CrossRef.
- M. Seifan, S. Mendoza and A. Berenjian, Mechanical properties and durability performance of fly ash based mortar containing nano- and micro-silica additives, Constr. Build. Mater., 2020, 252, 119121 CrossRef CAS.
- S. Said, S. Mikhail and M. Riad, Recent processes for the production of alumina nano-particles, Mater. Sci. Energy Technol., 2020, 3, 344–363 CAS.
- D. Kim, C. Lee, B. G. Lee, J. Park, K. C. Kim, J. K. Choe, P. Westerhoff and H. Rho, γ-Al2O3 selectively adsorbs transition group metals from contaminated waters to produce bi-metallic catalysts for efficient nitrate reduction, J. Hazard. Mater., 2025, 493, 138428 CrossRef CAS PubMed.
- R. Prins, On the structure of γ-Al2O3, J. Catal., 2020, 392, 336–346 CrossRef CAS.
- AQSIQ and SAC, Test Method of Cement Mortar Strength (ISO Method), 2021 Search PubMed.
- EPA, Synthetic Precipitation Leaching Procedure, 1994 Search PubMed.
- AQSIQ and SAC, Methods for Determining the Uniaxial Compressive Strength and Counting Softening Coefficient, 2009 Search PubMed.
- AQSIQ and SAC, Standard for Groundwater Quality, 2017 Search PubMed.
- Z. Li, Y. Huang, Z. Zhu, H. Shi, Y. Xiao, H. Song, W. Zuo, H. Zhou, S. Wang and L. Dong, Research on the stepwise treatment of phytoremediation residue: Combining leaching pretreatment with pyrolysis, Process Saf. Environ. Prot., 2025, 194, 74–82 CrossRef CAS.
- Z. A. AlDhawi, I. I. BinSharfan and M. A. Abdulhamid, Carboxyl-functionalized polyimides for efficient bisphenol A removal: Influence of wettability and porosity on adsorption capacity, Chemosphere, 2023, 313, 137347 CrossRef PubMed.
- I. Ngo, L. Ma, J. Zhai, Y. Wang and T. Wei, Durability of CO2-fly ash-based backfill materials in cation water deterioration, Int. J. Min. Reclamat. Environ., 2023, 37, 544–567 CrossRef CAS.
- C. Cai, F. Li, S. Fan, D. Yan, Q. Sun, H. Jin, J. Shao, Z. Chen and M. Liu, Investigation on deterioration mechanism of geopolymer cemented coal Gangue-Fly ash backfill under combined action of high temperature and salt corrosion environment, Constr. Build. Mater., 2023, 398, 132518 CrossRef CAS.
- L. Qing, H. Zhang and Z. Zhang, Effect of biochar on compressive strength and fracture performance of concrete, J. Build. Eng., 2023, 78, 107587 CrossRef.
- M. S. Islam, B. J. Mohr and D. VandenBerge, Performance of natural clinoptilolite zeolite in the cementitious materials: A comparative study with metakaolin, fly ash, and blast furnace slag, J. Build. Eng., 2022, 53, 104535 CrossRef.
- D. Salehzadeh, B. Elahi, J. E. Ten Elshof, G. Brem and M. Mehrali, Synergistic effect of sepiolite as an active binder for enhancing mechanical stability of porous K2CO3 granules in thermochemical heat storage, Chem. Eng. J. Adv., 2025, 23, 100783 CrossRef CAS.
- K. Sargunan, M. Venkata Rao, A. Alex Rajesh, R. Babu, P. Prasanthni, K. Jagadeep and M. L. Rinawa, Experimental investigations on mechanical strength of concrete using nano-alumina and nano-clay, Mater. Today: Proc., 2022, 62, 5420–5426 CAS.
- Z. Zhang, L. Ma, I. Ngo, C. Peng, J. Zhai and Z. Guo, Effect of Mine Water Environment on Durability of Solid Backfilling Based on Carbonated Coal-Based Waste, ACS Omega, 2025, 10, 17626–17641 CrossRef CAS PubMed.
- W. Wang, B. Wang and S. Zhang, Dispersion, properties, and mechanisms of nanotechnology-modified alkali-activated materials: A review, Renewable Sustainable Energy Rev., 2024, 192, 114215 CrossRef CAS.
- Z. X. Chen, N. T. Zhang and S. H. Chu, Role of alkalinity in CO2 sequestration of γ-belite, Constr. Build. Mater., 2024, 432, 136508 CrossRef CAS.
- O. Yazici and S. Yilmaz, Production and characterisation of LDH meixnerite powders, Adv. Appl. Ceram., 2010, 109, 341–345 CrossRef CAS.
- Y. Guan, Y. Xia, L. Chen, B. Guo and L. Wang, Efficient adsorption of heavy metals using Friedel's salt synthesized from municipal solid waste incineration fly ash leachate, Environ. Res., 2025, 285, 122330 CrossRef CAS PubMed.
- T. Hou, L. Yan, S. Yang, J. Li, X. Li and Y. Zhao, Efficient removal of graphene oxide by Fe3O4/MgAl-layered double hydroxide and oxide from aqueous solution, J. Mol. Liq., 2019, 284, 300–306 CrossRef CAS.
- S. Liu, C. Zhang, C. Hu, K. Mei and X. Cheng, Study on low thermal conductivity cement reinforced by sepiolite fiber for deep geothermal well, Geothermics, 2025, 129, 103290 CrossRef.
- I. Ngo, L. Ma, J. Zhai, Y. Wang, Y. Xu, T. Wei and K. Yu, Effect of the co-activation of sodium silicate and CO2 on setting and mechanical properties of coal gangue-fly ash backfill (CGFB), Environ. Earth Sci., 2023, 82, 190 CrossRef CAS.
- D. Liang, F. Wang, S. Xue, J. Yan, Y. Xu, G. Lv and J. Yan, Purification of waste liquids from alkaline washing pretreatment of municipal solid waste incineration fly ash using Friedel's salt, J. Environ. Manage., 2025, 380, 125020 CrossRef CAS PubMed.
- U. A. Birnin-Yauri and F. P. Glasser, Friedel's salt, Ca2Al(OH)6(Cl,OH)·2H2O: its solid solutions and their role in chloride binding, Cem. Concr. Res., 1998, 28, 1713–1723 CrossRef CAS.
- S. Fan, M. Zhao, J. Luo, W. Li, X. Fan, N. Zhou, H. Xu and Y. Shi, Facile preparation of N/P co-doped mesoporous biochar for efficient removal of methylene blue from aqueous solutions: A 2D-FTIR-COS, adsorption mechanism analysis, and fixed-bed column study, J. Water Process Eng., 2025, 72, 107479 CrossRef.
- Y. Wang, Y. Cao, Z. Zhang, P. Zhang, Y. Ma, A. Wang and H. Wang, Intrinsic sulfuric acid resistance of C-(N)-A-S-H and N-A-S-H gels produced by alkali-activation of synthetic calcium aluminosilicate precursors, Cem. Concr. Res., 2023, 165, 107068 CrossRef CAS.
- M. Cantaluppi, V. Diella, A. Pavese, M. Marchi and N. Marinoni, Crystallisation and phase transformation processes during clinkering in portland cement (pc) by means of in situ and ex situ analyses, Constr. Build. Mater., 2023, 394, 132261 CrossRef CAS.
- Q. Dong, B. Liang, L. Jia and L. Jiang, Effect of sulfide on the long-term strength of lead-zinc tailings cemented paste backfill, Constr. Build. Mater., 2019, 200, 436–446 CrossRef CAS.
- Y. Mei, S. Zhuang and J. Wang, Adsorption of heavy metals by biochar in aqueous solution: A review, Sci. Total Environ., 2025, 968, 178898 CrossRef CAS PubMed.
- N. Gineys, G. Aouad and D. Damidot, Managing trace elements in Portland cement – Part I: Interactions between cement paste and heavy metals added during mixing as soluble salts, Cem. Concr. Compos., 2010, 32, 563–570 CrossRef CAS.
- Y. F. Orlov, E. I. Maslov and E. I. Belkina, Solubilities of metal hydroxides, Russ. J. Inorg. Chem., 2013, 58, 1306–1314 CrossRef CAS.
- F. Scholz and H. Kahlert, The calculation of the solubility of metal hydroxides, oxide-hydroxides, and oxides, and their visualisation in logarithmic diagrams, ChemTexts, 2015, 1, 7 CrossRef.
- M. L. D. Gougar, B. E. Scheetz and D. M. Roy, Ettringite and C-S-H Portland cement phases for waste ion immobilization: A review, Waste Manage., 1996, 16, 295–303 CrossRef CAS.
- T. Wang, X. Quan, Z. Sun and Z. Sun, Sustainable biochar synthesis via synergistic H2O2-KOH modification for enhanced CO2 physisorption, Carbon Capture Sci. Technol., 2025, 15, 100438 CAS.
- L. Chi, C. Huang, Z. Li, S. Ruan, B. Peng, M. Li, Q. Liang, K. Yin and S. Lu, Heavy metals immobilization of LDH@biochar-containing cementitious materials: Effectiveness and mechanisms, Cem. Concr. Compos., 2024, 152, 105667 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |




