Open Access Article
Open Access ArticleMolecular and materials design for efficient solar energy conversion: a review of photochemical technologies
Abdulrahman A. Alsimaree
a,
Saeed S. Samman
b,
Abdulaziz M. Almohyawi
 c,
Hatem M. Altassc,
Jan Mohammad Mir
c,
Hatem M. Altassc,
Jan Mohammad Mir
 *d and
Saleh A. Ahmed
*c
*d and
Saleh A. Ahmed
*c
aDepartment of Chemistry, College of Science and Humanities, Shaqra University, Shaqra, Saudi Arabia
bDepartment of Basic Science and Technologies, Applied College, Taibah University, 42353 Madina, Saudi Arabia
cDepartment of Chemistry, Faculty of Science, Umm Al-Qura University, 21955 Makkah, Saudi Arabia. E-mail: saahmed@uqu.edu.sa; saleh_63@hotmail.com
dMultidisciplinary Chemistry Laboratory, Department of Chemistry, Islamic University of Science and Technology, Kashmir 192122, India. E-mail: mirjanmohammad@gmail.com
First published on 28th January 2026
Abstract
The inexorable rise in global energy demand, coupled with the pressing imperative to mitigate anthropogenic climate change, has catalyzed unprecedented research effort into renewable energy sources. Photochemistry, the study of chemical reactions initiated by light, is fundamentally shaping this landscape, particularly in solar energy conversion. This review provides a comprehensive and critical analysis of current trends in photochemistry that are directly enabling the development of next-generation renewable energy technologies. We delve into the operational principles, recent advances in materials, and persistent challenges across three pivotal photochemical systems: photoelectrochemical (PEC) devices, artificial photosynthetic systems for solar fuel production, and dye-sensitized solar cells (DSSCs). The discourse highlights the strategic shift from scarce, noble-metal-based components towards earth-abundant alternatives, the integration of molecular and solid-state systems in hybrid architectures, and the critical pursuit of long-term operational stability. While significant progress has been made in understanding charge transfer dynamics and tailoring material properties at the nanoscale, the path to widespread commercialization necessitates continued interdisciplinary innovation to overcome efficiency, durability, and scalability hurdles. This critical evaluation of the current state of the art aims to illuminate both the remarkable achievements and the fundamental scientific questions that remain at the forefront of photochemical energy research.
1 Introduction
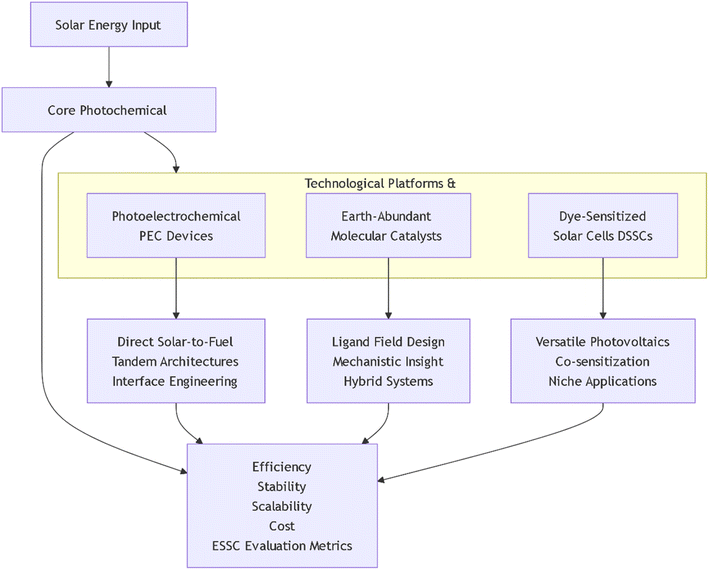
The Sun delivers a continuous and prodigious flux of energy to Earth, dwarfing humanity's total power consumption. Harnessing this resource through photochemical conversion, the transformation of light energy into storable chemical fuels or electricity, represents a paramount scientific challenge in the world. The process begins when a photon is absorbed by a material, promoting an electron to a higher-energy, excited state. This excited state is a crucial, energy-rich intermediate. The core objective of artificial energy conversion is to harness this transient energy to drive thermodynamically uphill chemical reactions, such as water splitting or carbon dioxide (CO2) reduction, before it is lost as heat or light.1–4 Modern photochemical research is characterized by a powerful convergence of disciplines, including synthetic chemistry, materials science, nanoscience, and electrochemistry. These fields are directed at optimizing three sequential steps involving broad-spectrum light absorption, efficient charge separation, and rapid catalytic turnover.5–7 These steps manifest in several promising technological platforms, each with unique trade-offs between efficiency, stability, and cost. The overarching goal across all platforms is to develop technologies that are not only efficient but also economically viable and sustainable, requiring a holistic assessment from material sourcing to end-of-life recycling.8,9The fundamental photochemical process involves three sequential steps, viz, the absorption of a photon to create an excited state, the efficient separation of charge carriers (electrons and holes), and the directed use of these energetic charges to drive useful chemical reactions, such as water splitting or CO2 reduction. Optimizing each step, while minimizing parasitic energy losses, requires a deep, synergistic understanding of molecular design, materials science, and interface engineering. A dominant and necessary trend is the strategic shift from scarce, noble-metal-based components (e.g., Ru, Ir, Pt) toward catalysts and sensitizers made from earth-abundant elements (e.g., Cu, Fe, Ni, Co).10,11 Also, the convergence of disciplines has given rise to several promising technological platforms. Photoelectrochemical (PEC) cells aim to directly produce hydrogen or hydrocarbons from sunlight and water.12–15 DSSCs offer a versatile, lower-cost photovoltaic alternative with unique aesthetic and diffuse-light capabilities.16–18 Concurrently, the field of artificial photosynthesis seeks to orchestrate molecular components into integrated systems that mimic nature's efficiency.19–22 While significant progress has been made, the path to commercialization hinges on simultaneously optimizing efficiency, stability, scalability, and cost, a complex, multi-parameter challenge.23–25
While numerous reviews exist on individual technologies,26–29 such as PEC cells, artificial photosynthesis, or DSSCs, this review article provides a unified and critical perspective that bridges device-level engineering with molecular-level design (Scheme 1). The work is a critical evaluation of the progression from fundamental discovery to integrated system performance across these platforms, employing a consistent framework of efficiency, stability, scalability, and cost (ESSC) metrics to assess commercial viability. The article is structured to first examine advances at both the device architecture and molecular catalyst levels. It then explores applications in solar fuels and photovoltaics, before synthesizing the prevailing challenges and future research vectors. This holistic narrative, which moves from materials to devices to systems-level analysis, explicitly connects macroscopic engineering with molecular science under a common evaluative lens.
2 Advances in photochemical technologies: from materials to devices
The field of photochemical technology is undergoing a profound transformation, driven by a convergence of innovative materials design and sophisticated device engineering. This evolution progresses from the molecular-scale synthesis of novel photoactive compounds to their integration into functional, scalable architectures for energy conversion and catalysis.30–34 Therefore, this section discusses advances at the device architecture level, focusing on integrated systems for direct solar-to-fuel conversion, followed by an examination of progress at the molecular and materials level, detailing the design of next-generation photocatalysts.35–382.1 Device-level architectures for direct solar fuel production
The photoanode, responsible for the OER, is often the performance-limiting component due to the reaction's kinetic sluggishness and thermodynamic demands.44 Among visible-light-responsive metal oxides, Bismuth Vanadate (BiVO4, bandgap ∼2.4 eV) has emerged as a benchmark photoanode material. However, its performance is hampered by poor charge carrier mobility and rapid surface recombination. Research strategies to overcome these limitations involve nanostructuring to shorten charge transport paths and the creation of heterojunctions (e.g., BiVO4/WO3) to enhance charge separation via built-in fields. Crucially, surface modification with OER co-catalysts like cobalt phosphate (Co-Pi) or nickel-iron oxyhydroxide (NiFeOOH) is essential to passivate surface traps and accelerate the reaction kinetics.45–48 Recent insights emphasize the need for kinetic synergy, where the hole transfer rate from the semiconductor bulk to the catalyst must outpace recombination (Fig. 2).
| Material/system | Type | Bandgap (eV) | Function | STH efficiency (%) | Key features/challenges | Ref. |
|---|---|---|---|---|---|---|
| BiVO4/WO3 heterojunction with Co-Pi | Photoanode | ∼2.4 | OER | ∼6.2 (with bias) | Good visible light absorption; requires co-catalyst; modest stability | 97 and 98 |
| Ta3N5 with NiFeOOH | Photoanode | ∼2.1 | OER | ∼3.5 (with bias) | Narrow bandgap; suffers from severe self-oxidation | 99 |
| p-Si with Pt/TiO2 protection | Photocathode | 1.1 | HER | ∼10 (in tandem) | High efficiency; requires complex protection layers | 51 |
| Sb2Se3 with Pt | Photocathode | ∼1.2 | HER | ∼3.5 | Earth-abundant; emerging material; stability under study | 51 |
| Perovskite/BiVO4 – Si tandem | Tandem cell | Varies | Full WS | >20 (PEC-PV) | Record efficiency; complex multi-layer fabrication; long-term stability is a major concern | 100 |
| CIGS-based tandem | Tandem cell | Varies | Full WS | ∼10–15 | Good stability potential; scalable thin-film technology | 101 |
2.2 Molecular and materials design for photocatalysis
3 Applications in renewable energy: from laboratory concepts to integrated systems
3.1 Artificial photosynthesis: closing the carbon cycle
Artificial photosynthesis is a technological process designed to mimic the natural reactions by which plants convert sunlight, water, and carbon dioxide into energy-storing molecules. Its primary goal is to use solar energy to chemically reduce captured CO2, combining it with protons from water to synthesize sustainable fuels and chemical feedstocks. By sourcing carbon from atmospheric or industrial CO2 emissions, this process aims to create a circular economy, effectively closing the anthropogenic carbon cycle. A key objective is to achieve solar-to-chemical conversion efficiencies that significantly surpass the relatively low efficiency of natural photosynthesis in plants.67 The technology fundamentally relies on advanced light-induced molecular transformations, typically driven by specialized photocatalysts or photoelectrochemical systems (Fig. 6).These systems often employ semiconductor materials or molecular complexes to absorb light, generate charges, and catalyze the critical reactions of water oxidation and CO2 reduction. Target outputs include energy-dense fuels such as hydrogen (from water splitting), methane, or methanol, which serve as renewable carriers for storing intermittent solar energy. As such, artificial photosynthesis represents a direct application of photochemical principles for sustainable energy storage and is a major focus of contemporary research in renewable energy technologies.68,69
| Catalyst (metal) | Major Product(s) | Selectivity (%) | Turnover number (TON) | Key design feature | Ref. |
|---|---|---|---|---|---|
| [Re(bpy)(CO)3Cl] | CO | >90 | ∼300 | Seminal noble-metal catalyst; operates at low overpotential | 71 |
| [Mn(bpy)(CO)3Br] | CO | >95 | ∼1100 | Earth-abundant alternative to Re; similar bipyridine ligand | 82 |
| Co-porphyrin with pendant phenol groups | CO/HCOOH | 80 (CO) | ∼5000 | Second-sphere proton relay enhances rate and selectivity | 71 and 74 |
| Ni(cyclam)2+ derivatives | CO/H2 | Varies | ∼105 | High durability; product selectivity sensitive to conditions | 68 |
| Fe-porphyrin/covalent organic framework (COF) hybrid | CO | >99 | ∼1500 | Heterogenized system; combines molecular specificity with solid-state stability | 75 and 76 |
3.2 DSSCs: versatility and aesthetic appeal
In 1991, Brian O'Regan and Michael Grätzel achieved a ground-breaking development by inventing the DSSC, a new type of photovoltaic device.77 Their key innovation was the use of a nanostructured, wide-bandgap titanium dioxide semiconductor coated with light-absorbing dye molecules, which effectively separated the processes of light absorption and charge transport.78,79 This biomimetic “artificial leaf” architecture offered a lower-cost alternative to conventional silicon solar cells and established foundational principles for modern photoelectrochemical and photocatalytic research. Such advancements have occupied a unique niche, offering advantages in manufacturing cost, performance under diffuse light, and design flexibility (color, transparency) compared to conventional silicon PV.80The components of dye-sensitized solar cells have undergone significant and focused development. Early dye sensitizers were primarily ruthenium-based polypyridyl complexes, such as N3 and N719. Research later shifted toward metal-free organic dyes featuring a donor-π-acceptor, or D–π–A, molecular structure. Another important class of advanced sensitizers is porphyrin-based dyes, which have helped achieve record power conversion efficiencies exceeding 14%.81,83 Alongside dyes, the electrolyte system has also evolved substantially from its original form. The traditional liquid electrolyte relied on the iodide/triiodide (I−/I3−) redox couple as a charge mediator. To address issues like corrosion and durability, alternative redox mediators such as cobalt (Co3+/2+) complexes were introduced. Another major advancement has been the move toward solid-state hole-transport materials, like 2,2′,7,7′-tetrakis(N,N-di-p-methoxyphenylamine)-9,9′-spirobifluorene (spiro-OMeTAD), to replace liquid electrolytes. These changes in electrolyte design improve the long-term stability of the solar cell. They also simplify the sealing process of the device, which is crucial for practical manufacturing and deployment84,85
| Component | Early generation (∼1991–2005) | State-of-the-art/emerging | Key advantages/status | Ref. |
|---|---|---|---|---|
| Sensitizer | Ru-polypyridyl complexes (N3, N719) | Metal-free organic D–π–A dyes (e.g., Y123); porphyrins (e.g., SM315) | Lower cost, higher molar absorptivity; PCE >14% achieved | 81 and 83 |
| Redox mediator | Iodide/triiodide (I−/I3−) | Cobalt complexes (e.g., [Co(bpy)3]3+/2+); copper complexes; solid HTMs (spiro-OMeTAD) | Higher photovoltage; better stability; enables solid-state DSSCs | 88 and 85 |
| Counter electrode | Platinum (Pt) on FTO | Carbon materials (graphene, CNTs); conductive polymers (PEDOT) | Lower cost, comparable catalytic activity for I3− reduction | 85 |
| Primary application focus | General outdoor PV | BIPV; indoor light harvesting for IoT | Leverages aesthetic flexibility and superior diffuse-light performance | 84 and 86 |
4 Challenges and future perspectives
Despite the compelling progress detailed herein, the path to the widespread deployment of photochemical energy technologies is fraught with scientific and engineering challenges that demand continued focused research. The paramount challenge across all systems is the simultaneous optimization of efficiency, stability, and cost, the pivot of energy technology. For PEC water splitting, corrosion of semiconductor materials in harsh aqueous electrolytes remains a fundamental issue, necessitating the development of more robust protection layers that are both conductive and optically transparent. In artificial photosynthesis, the selectivity of CO2 reduction towards a single, high-value product remains elusive, and the scaling of these systems to industrially relevant levels is a monumental task that involves not only chemical engineering but also the development of efficient product separation and purification processes. For DSSCs and molecular systems, long-term photostability and thermal stability of the organic and molecular components under continuous operational stress are critical concerns.87–89Future research directions will likely involve several convergent strategies. The use of advanced computational modeling, including density functional theory (DFT) and machine learning, will accelerate the discovery and design of new materials with tailored optoelectronic properties. The exploration of novel nanoscale and heterostructured architectures, such as core–shell nanoparticles, nanowire arrays, and two-dimensional materials, will provide new avenues for controlling light–matter interactions and charge transport pathways.90–93 The integration of biological components, such as enzymes, with synthetic photochemical systems is an emerging frontier that could lead to highly selective and efficient hybrid biocatalysts for fuel production. Finally, a greater emphasis on life-cycle analysis (LCA) and techno-economic assessment (TEA) at the research stage will be crucial for guiding the development of technologies that are not only scientifically impressive but also economically viable and environmentally sustainable on a global scale.94–96
5. Conclusions
In conclusion, the dynamic field of photochemistry is in the midst of a profoundly transformative era. This transformation is largely propelled by the world's urgent and escalating need for sustainable energy solutions. The collective research we have surveyed represents a strategic and coordinated front against energy challenges. One dominant trend is the meticulous refinement of photoelectrochemical (PEC) devices. These devices are engineered for the direct production of solar fuels, such as hydrogen. Through PEC systems, sunlight directly facilitates the splitting of water molecules. This process aims to store solar energy in a clean, chemical form. Concurrently, a significant paradigm shift is moving research away from rare, expensive catalysts. The new focus is squarely on earth-abundant molecular photocatalysts. Catalysts based on iron, copper, or cobalt are now at the forefront of this investigation. This shift is crucial for ensuring the economic viability and widespread adoption of future technologies. Furthermore, scientists are engaging in sophisticated mimicry of natural photosynthesis. This bio-inspired approach seeks to replicate the elegance and efficiency of plant-based systems. Researchers are designing synthetic complexes that emulate natural light-harvesting antennae. They are also constructing molecular assemblies that mimic catalytic reaction centers found in nature. The goal is to achieve the high selectivity and low energy waste characteristic of biological systems. Alongside this, the persistent optimization of dye-sensitized architectures continues unabated. DSSCs are being steadily improved for better performance and stability. Novel organic and perovskite-sensitized materials are broadening the spectral absorption of these devices. Engineering of mesoporous semiconductor scaffolds enhances electron injection and transport. These four interconnected trends collectively represent a powerful, multifaceted assault on solar energy conversion. Each approach complements the others, creating a rich and diverse research landscape. Underpinning all these advancements is a deepening, atom-level understanding of fundamental mechanisms. Scientists are now unraveling complex photophysical pathways with unprecedented detail. The precise sequence of events following photon absorption—from excitation to charge separation is being mapped. Similarly, the intricate catalytic mechanisms that facilitate fuel-forming reactions are being decoded. This fundamental knowledge is critically enabled by a suite of cutting-edge characterization tools. Ultrafast laser spectroscopy captures events occurring on femtosecond timescales. Advanced X-ray techniques provide atomic-resolution snapshots of catalysts in action. Sophisticated computational modeling predicts properties and guides the design of new materials. However, the path forward is not without its significant and persistent hurdles. Improving the overall efficiency of photon-to-fuel conversion remains a paramount challenge. The long-term durability of materials under constant photochemical stress is a major concern. Scaling laboratory breakthroughs to industrially relevant, manufacturable dimensions is a formidable task. The integration of components into robust, working systems adds another layer of complexity. Despite these challenges, the relentless pace of innovation in the field is undeniable. New discoveries and incremental improvements are reported in the literature virtually every day. This progress is fueled by a collaborative spirit that unites the global research community. Chemists, physicists, materials scientists, and engineers are working in concert across disciplines. This collaboration bridges the gap between molecular discovery and functional device engineering. Therefore, there is strong and justified cause for optimism about the future. The continued interplay between fundamental molecular science and applied research will be pivotal. Insights from quantum mechanics directly inform the synthesis of new light-absorbing molecules. Advanced materials engineering then translates these molecules into stable, functional films and devices. Finally, systems-level integration assembles these components into practical, operational technology. This virtuous cycle of discovery and application will undoubtedly yield transformative results. It will lead to the next generation of efficient and affordable photochemical technologies. These future technologies will move beyond the laboratory to impact the global energy landscape. They are poised to solidify the role of sunlight as a cornerstone of our energy portfolio. Ultimately, the work in photochemistry today is lighting the path toward a sustainable, solar-powered future.Author contributions
Abdulrahman A. Alsimaree: writing – original draft, visualization, data curation. Saeed S. Samman: writing – original draft, visualization, data curation. Jan Mohammad Mir: writing – review & editing, supervision, conceptualization. Hatem M. Altass: investigation, formal analysis, validation. Abdulaziz M. Almohyawi: investigation, conceptualization, writing – review & editing. Saleh A. Ahmed: supervision, project administration, funding acquisition, writing – review & editing.Conflicts of interest
No potential conflict of interest was reported by the authors.Data availability
No primary research results, software, or code have been included in this review article, and no new data were generated or analysed as part of this work.Acknowledgements
The authors extend their appreciation to Umm Al-Qura University, Saudi Arabia for funding this research work through grant number: 26UQU4320545GSSR03.References
- J. H. Montoya, L. C. Seitz, P. Chakthranont, A. Vojvodic, T. F. Jaramillo and J. K. Nørskov, Nat. Mater., 2017, 16, 70–81 Search PubMed
.
- I. Rodriguez-Gutierrez, F. L. Souza and O. K. Varghese, in Conversion of Water and CO2 to Fuels Using Solar Energy: Science, Technology and Materials, 2024, pp. 1–8 Search PubMed
.
- M. Ashraf, M. Ayaz, M. Khan, S. F. Adil, W. Farooq, N. Ullah and M. Nawaz Tahir, Energy Fuels, 2023, 37, 6283–6301 CrossRef CAS
.
- C. Casadevall, A. Call, Z. Codolà, F. Acuña-Parés and J. L. Fillol, An. Quim. RSEQ, 2016, 112, 133–140 Search PubMed
.
- Y. Wang, H. Suzuki, J. Xie, O. Tomita, D. J. Martin, M. Higashi, D. Kong, R. Abe and J. Tang, Chem. Rev., 2018, 118, 5201–5241 Search PubMed
.
- K. Fan, L. Wang and L. Tong, in Conversion of Water and CO2 to Fuels Using Solar Energy: Science, Technology and Materials, 2024, pp. 19–45 Search PubMed
.
- V. Balzani, A. Credi and M. Venturi, ChemSusChem, 2008, 1, 26–58 CrossRef CAS PubMed
.
- X. Xia, J. H. Pan, X. Pan, L. Hu, J. Yao, Y. Ding, D. Wang, J. Ye and S. Dai, ACS Energy Lett., 2019, 4, 405–410 Search PubMed
.
- A. V. Emeline, A. V. Rudakova, V. K. Ryabchuk and N. Serpone, Curr. Opin. Green Sustain. Chem., 2022, 34, 100588 CrossRef CAS
.
- G. Granqvist, Adv. Mater., 2003, 15, 1789–1803 CrossRef
.
- C. G. Granqvist, Appl. Phys. A, 1991, 52, 83–93 CrossRef
.
- G. P. Smestad, F. C. Krebs, C. M. Lampert, C. G. Granqvist, K. L. Chopra, X. Mathew and H. Takakura, Sol. Energy Mater. Sol. Cells, 2008, 92, 371–373 CrossRef CAS
.
- J. Gong, C. Li and M. R. Wasielewski, Chem. Soc. Rev., 2019, 48, 1862–1864 RSC
.
- G. W. Crabtree and N. S. Lewis, Phys. Today, 2007, 60, 37–42 CrossRef CAS
.
- N. S. Lewis, Chem. Rev., 2015, 115, 12631–12632 Search PubMed
.
- Y. W. Su, S. C. Lan and K. H. Wei, Mater. Today, 2012, 15, 554–562 CrossRef CAS
.
- G. Zhang, F. R. Lin, F. Qi, T. Heumüller, A. Distler, H. J. Egelhaaf, N. Li, P. C. Chow, C. J. Brabec, A. K. Jen and H. L. Yip, Chem. Rev., 2022, 122, 14180–14274 Search PubMed
.
- K. A. Mazzio and C. K. Luscombe, Chem. Soc. Rev., 2015, 44, 78–90 Search PubMed
.
- B. Ehrler, E. Alarcón-Lladó, S. W. Tabernig, T. Veeken, E. C. Garnett and A. Polman, ACS Energy Lett., 2020, 5, 3029–3033 CrossRef CAS
.
- W. Shockley and H. J. Queisser, J. Appl. Phys., 1961, 32, 510–519 CrossRef CAS
.
- D. Gust and T. A. Moore, Science, 1989, 244, 35–41 CrossRef CAS PubMed
.
- M. Freemantle, Chem. Eng. News, 1998, 76, 37–46 CrossRef
.
- A. M. Al-Amri, Polymers, 2023, 15, 3234 CrossRef CAS PubMed
.
- Z. Peng, Y. Zhang, C. L. Choi, P. Zhang, T. Wu and Y. K. Chan, Nanoscale, 2023, 15, 11403–11421 RSC
.
- N. Palavesam, S. Marin, D. Hemmetzberger, C. Landesberger, K. Bock and C. Kutter, Flex. Print. Electron., 2018, 3, 014002 CrossRef
.
- Y. Magazov, A. Aliyev, N. Zhumabay, Z. Taubaldiyeva, G. Zhigerbayeva and N. Nuraje, RSC Adv., 2026, 16, 1643–1661 RSC
.
- V. Dhiman, S. Singh, V. Srivastava, S. Garg and A. D. Saran, Environ. Sci. Pollut. Res., 2025, 32, 13299–13312 CrossRef CAS PubMed
.
- B. D. Nguyen, I. H. Choi and J. Y. Kim, Nanomaterials, 2025, 15, 1494 CrossRef CAS PubMed
.
- T. Hisatomi, T. Yamada, H. Nishiyama, T. Takata and K. Domen, Nat. Rev. Mater., 2025, 10, 769–782 CrossRef
.
- P. Heinstein, C. Ballif and L. E. Perret-Aebi, Green, 2013, 3, 125–156 Search PubMed
.
- E. Biyik, M. Araz, A. Hepbasli, M. Shahrestani, R. Yao, L. Shao, E. Essah, A. C. Oliveira, T. Del Cano, E. Rico and J. L. Lechón, Eng. Sci. Technol., Int. J., 2017, 20, 833–858 Search PubMed
.
- R. J. Yang and P. X. W. Zou, Int. J. Constr. Manage., 2016, 16, 39–53 Search PubMed
.
- A. V. Emeline, R. V. Mikhaylov, P. E. Lavrik, A. V. Kozhevina, K. M. Bulanin, P. D. Murzin, N. I. Glazkova, V. N. Kuznetsov, A. V. Rudakova, M. V. Mayevskaia and Y. M. Artemiev, Rev. Adv. Chem., 2020, 10, 73–111 Search PubMed
.
- F. Schmidt, J. Wenzel, N. Halland, S. Güßregen, L. Delafoy and A. Czich, Chem. Res. Toxicol., 2019, 32, 2338–2352 Search PubMed
.
- X. Li, P. M. Maffettone, Y. Che, T. Liu, L. Chen and A. I. Cooper, Chem. Sci., 2021, 12, 10742–10754 RSC
.
- C. Rodríguez-Seco, Y. S. Wang, K. Zaghib and D. Ma, Nanophotonics, 2022, 11, 1443–1484 Search PubMed
.
- C. Y. Toe, J. Pan, J. Scott and R. Amal, ACS ES&T Eng., 2022, 2, 1130–1143 Search PubMed
.
- X. Yzeiri, J. Res. Appl. Chem., 2026, 8, 45–67 Search PubMed
.
- Q. Wang, J. Zhao and J. Luo, in Water Photo- and Electro-Catalysis: Mechanisms, Materials, Devices, and Systems, 2024, pp. 207–239 Search PubMed
.
- K. H. Bevan, B. Miao and A. Iqbal, in Conversion of Water and CO2 to Fuels Using Solar Energy: Science, Technology and Materials, 2024, pp. 183–202 Search PubMed
.
- K. Fan, L. Wang and L. Tong, in Conversion of Water and CO2 to Fuels Using Solar Energy: Science, Technology and Materials, 2024, pp. 19–45 Search PubMed
.
- W. Yang, R. R. Prabhakar, J. Tan, S. D. Tilley and J. Moon, Chem. Soc. Rev., 2019, 48, 4979–5015 RSC
.
- W. H. Cheng, A. De La Calle, H. A. Atwater, E. B. Stechel and C. Xiang, ACS Energy Lett., 2021, 6, 3096–3113 Search PubMed
.
- Q. Wang, J. Zhao and J. Luo, in Water Photo- and Electro-Catalysis: Mechanisms, Materials, Devices, and Systems, 2024, pp. 207–239 Search PubMed
.
- G. McClea, J. Kennedy and A. T. Marshall, J. Electrochem. Soc., 2025, 172, 086501 Search PubMed
.
- S. Corby, R. R. Rao, L. Steier and J. R. Durrant, Nat. Rev. Mater., 2021, 6, 1136–1155 Search PubMed
.
- M. B. Vishlaghi, A. Kahraman, N. Österbacka, E. Usman, E. Erdem, A. Sennaroğlu, J. Wiktor and S. Kaya, J. Mater. Chem. A, 2023, 11, 16648–16658 Search PubMed
.
- I. Grigioni, A. Polo, M. V. Dozzi, K. G. Stamplecoskie, D. H. Jara, P. V. Kamat and E. Selli, ACS Appl. Energy Mater., 2022, 5, 13142–13148 CrossRef CAS PubMed
.
- Y. J. Jang and J. S. Lee, ChemSusChem, 2019, 12, 1835–1845 CrossRef CAS PubMed
.
- A. C. Salomao, M. dos Santos Araujo, H. L. Dos Santos, M. Medina, L. H. Mascaro and M. A. Andrade Junior, ChemSusChem, 2021, 14, 4671–4679 Search PubMed
.
- W. Yang, J. Park, H. C. Kwon, O. S. Hutter, L. J. Phillips, J. Tan, H. Lee, J. Lee, S. D. Tilley, J. D. Major and J. Moon, Energy Environ. Sci., 2020, 13, 4362–4370 Search PubMed
.
- A. M. Fehr, A. Agrawal, F. Mandani, C. L. Conrad, Q. Jiang, S. Y. Park, O. Alley, B. Li, S. Sidhik, I. Metcalf and C. Botello, Nat. Commun., 2023, 14, 3797 Search PubMed
.
- C. B. Larsen and O. S. Wenger, Chem.–Eur. J., 2018, 24, 2039–2058 CrossRef CAS PubMed
.
- C. Förster and K. Heinze, Chem. Soc. Rev., 2020, 49, 1057–1070 RSC
.
- V. Ferraro, C. Bizzarri and S. Bräse, Adv. Sci., 2024, 11, 2404866 Search PubMed
.
- P. Jin and C. Wang, Phys. Chem. Chem. Phys., 2025, 27, 14055–14072 Search PubMed
.
- C. Bizzarri, in Electrochemical Society Meeting Abstracts 247, the Electrochemical Society, Inc., 2025, p. 2730 Search PubMed
.
- C. Li, X. Y. Kong, Z. H. Tan, C. T. Yang and H. S. Soo, Chem. Phys. Rev., 2022, 3, 021305 CrossRef
.
- K. Behm and R. D. McIntosh, ChemPlusChem, 2020, 85, 2611–2618 Search PubMed
.
- D. Li, F. Fan, C. Li and X. Wang, in Water Photo- and Electro-Catalysis: Mechanisms, Materials, Devices, and Systems, 2024, pp. 397–447 Search PubMed
.
- K. T. Arul, T. T. Nga, C. L. Dong and W. C. Chou, in Water Photo- and Electro-Catalysis: Mechanisms, Materials, Devices, and Systems, 2024, pp. 363–395 Search PubMed
.
- K. Fan, L. Wang and L. Tong, in Conversion of Water and CO2 to Fuels Using Solar Energy: Science, Technology and Materials, 2024, pp. 19–45 Search PubMed
.
- D. Arifin and A. W. Weimer, Sol. Energy, 2018, 160, 178–185 Search PubMed
.
- F. M. Hadi, R. C. Al-Zuhairy and H. S. Majdi, Al-Rafidain J. Eng. Sci., 2025, 2, 167–190 Search PubMed
.
- Y. Zhao, W. Gao, S. Li, G. R. Williams, A. H. Mahadi and D. Ma, Joule, 2019, 3, 920–937 CrossRef CAS
.
- P. T. Cesana, B. X. Li, S. G. Shepard, S. I. Ting, S. M. Hart, C. M. Olson, J. I. Alvarado, M. Son, T. J. Steiman, F. N. Castellano and A. G. Doyle, Chem, 2022, 8, 174–185 CAS
.
- B. Zhou, P. Zhou, W. Dong and Z. Mi, in Conversion of Water and CO2 to Fuels Using Solar Energy: Science, Technology and Materials, 2024, pp. 309–339 Search PubMed
.
- S. Fukuzumi, Artif. Photosynth., Royal Society of Chemistry, 2025 Search PubMed
.
- M. L. Lee, P. H. Liao, G. L. Li, H. W. Chang, C. W. Lee and J. K. Sheu, Sol. Energy Mater. Sol. Cells, 2019, 202, 110153 Search PubMed
.
- D. Arifin and A. W. Weimer, Sol. Energy, 2018, 160, 178–185 Search PubMed
.
- B. Krammer and T. Verwanger, in Applied Photochemistry: when Light Meets Molecules, ed. G. Bergamini and S. Silvi, Springer, Cham, 2016, pp. 978–3 Search PubMed
.
- O. S. De La Pena, V. Antonio, L. Collado, P. Renones, E. Alfonso, F. Fresno, M. Liras and M. Barawi, Acta Crystallogr., Sect. A: Found. Adv., 2014, 70, C134 Search PubMed
.
- R. Kathpalia and A. K. Verma, in Plant Physiology Annual Volume 2023, IntechOpen, 2023 Search PubMed
.
- S. Zhao, Q. Liu, Y. Li, L. Feng, S. Fu, M. Mazarji and J. Pan, Biofuel Res. J., 2024, 11, 2082–2098 CrossRef CAS
.
- J. Liu, Y. Y. Ren, W. J. Xia, B. Y. Deng and F. Wang, J. Mater. Chem. A, 2021, 9, 19346–19368 RSC
.
- M. E. Louis, T. G. Fenton, J. Rondeau, T. Jin and G. Li, Comments Inorg. Chem., 2016, 36, 38–60 CrossRef CAS
.
- B. O'Regan and M. Grätzel, Nature, 1991, 353, 737–740 Search PubMed
.
- S. K. Deb, S. Ferrere, A. J. Frank, B. A. Gregg, S. Y. Huang, A. J. Nozik, G. Schlichthörl and A. Zaban, in Conference Record of the Twenty Sixth IEEE Photovoltaic Specialists Conference, IEEE, 1997, pp. 507–510 Search PubMed
.
- A. K. Pandey, V. V. Tyagi, A. Jeyraj, L. Selvaraj, N. A. Rahim and S. K. Tyagi, Renewable Sustainable Energy Rev., 2016, 53, 859–884 CrossRef
.
- J. Castillo-Seoane, L. Contreras-Bernal, A. J. Riquelme, S. Fauvel, Y. Kervella, J. Gil-Rostra, G. Lozano, A. Barranco, R. Demadrille, J. R. Sánchez-Valencia and A. Borrás, Mater. Today Energy, 2025, 49, 101851 Search PubMed
.
- P. G, S. C. Pradhan and S. Soman, in Advances in Solar Energy Research, Springer, Singapore, 2018, pp. 281–316 Search PubMed
.
- F. Santos, D. Ivanou and A. Mendes, Sol. RRL, 2024, 8, 2300574 CrossRef CAS
.
- B. K. Korir, J. K. Kibet and S. M. Ngari, Energy Sci. Eng., 2024, 12, 3188–3226 CrossRef
.
- P. Mariani, L. Vesce and A. Di Carlo, in 2018 IEEE 4th International Forum on Research and Technology for Society and Industry (RTSI), IEEE, 2018, pp. 1–5 Search PubMed
.
- M. Dhonde, K. Sahu, M. Das, A. Yadav, P. Ghosh and V. V. Murty, J. Electrochem. Soc., 2022, 169, 066507 CrossRef CAS
.
- N. Pugazhenthiran, M. Kandasamy, P. Sathishkumar, S. Suresh, S. Karthick Kumar and S. Murugesan, RSC Adv., 2021, 11, 30566–30588 Search PubMed
.
- J. Li and N. Wu, Catal. Sci. Technol., 2015, 5, 1360–1384 RSC
.
- T. Abbas, H. S. Yahya and N. A. Amin, Energy Fuels, 2023, 37, 18330–18368 CrossRef CAS
.
- A. V. Raghu and T. Tachikawa, in Conversion of Water and CO2 to Fuels Using Solar Energy: Science, Technology and Materials, 2024, pp. 285–307 Search PubMed
.
- G. Segev, J. Kibsgaard, C. Hahn, Z. J. Xu, T. G. Deutsch, C. Xiang, J. Z. Zhang, L. Hammarström, D. G. Nocera, A. Z. Weber and P. Agbo, J. Phys. D: Appl. Phys., 2022, 55, 323003 Search PubMed
.
- S. A. Chowdhury, National Solar Energy Roadmap, 2021–2041, Sustainable and Renewable Energy Development Authority (SREDA), 2020 Search PubMed
.
- N. Taylor, O. G. Joanney, O. Eulaerts and M. Grabowska, Clean Energy Technology Observatory: Direct Solar Fuels in the European Union – 2022 Status Report on Technology Development, Trends, Value Chains and Markets, Publications Office of the European Union, 2022 Search PubMed
.
- S. Zei, Energy Convers., 2025, 1, 100025 Search PubMed
.
- A. Patra, R. Samal and C. S. Rout, Catal. Today, 2023, 424, 113853 CrossRef CAS
.
- S. Mohd, L. Balan, T. Gries, H. Alem, M. Lebrun, V. Falk and R. Schneider, Catal. Sci. Technol., 2026, 16, 123–135 Search PubMed
.
- Y. Liu, X. Pan, Z. Qiu, L. Sun, P. Zhang, R. He, Z. Han, J. Ma, Z. Zhang and Y. Wang, Fuel, 2025, 393, 135036 Search PubMed
.
- J. Chi, Z. Wei, W. Fang, J. Yan, J. Wang, H. Zu, J. Cheng, H. Luo, Z. Ye, J. Liu and Z. Jiang, Appl. Catal., B, 2025, 365, 124973 Search PubMed
.
- I. Grigioni, A. Polo, M. V. Dozzi, K. G. Stamplecoskie, D. H. Jara, P. V. Kamat and E. Selli, ACS Appl. Energy Mater., 2022, 5, 13142–13148 Search PubMed
.
- A. Kafizas, R. Godin and J. R. Durrant, in Semiconductors and Semimetals, ed. S. Hu and C. Xiang, Elsevier, 2017, vol. 97, pp. 3–46 Search PubMed
.
- A. C. Salomao, M. dos Santos Araujo, H. L. Dos Santos, M. Medina, L. H. Mascaro and M. A. Andrade Junior, ChemSusChem, 2021, 14, 4671–4679 CrossRef CAS PubMed
.
- H. Kobayashi, N. Sato, M. Orita, Y. Kuang, H. Kaneko, T. Minegishi, T. Yamada and K. Domen, Energy Environ. Sci., 2018, 11, 3003–3009 RSC
.
| This journal is © The Royal Society of Chemistry 2026 |