 Open Access Article
Open Access ArticleFirst-principles screening of dopants for high-conductivity graphene/copper interfaces
Xinsi Zhaoa,
Boan Zhonga,
Baixue Bian *a,
Jiamiao Ni
*a,
Jiamiao Ni a,
Mingyu Gong*ab,
Yue Liu*a and
Tongxiang Fan
a,
Mingyu Gong*ab,
Yue Liu*a and
Tongxiang Fan a
a
aState Key Lab of Metal Matrix Composites, School of Materials Science and Engineering, Shanghai Jiao Tong University, Shanghai 200240, China. E-mail: baixuebian@sjtu.edu.cn; mingyugong@sjtu.edu.cn; yliu23@sjtu.edu.cn
bInstitute of Medical Robotics, Shanghai Jiao Tong University, Shanghai 200240, China
First published on 28th January 2026
Abstract
Chemical doping is one of the promising approaches for tailoring the electronic properties of graphene/copper (Gr/Cu) composites. However, the diversity of doping elements and their complex bonding configurations result in nuanced effects, such as the competition between increased carrier concentration and defect generation (lattice distortion). Therefore, it is essential to decouple such complex doping effects into the intrinsic contribution of the dopant atom and the extrinsic effects of defects like vacancies. In this work, first-principles calculations, deformation potential theory, and the parabolic band model are combined to investigate the intrinsic mechanisms of various dopants. This approach decouples their contributions to carrier concentration and mobility, enabling the effective selection of dopants with optimal carrier transport properties. The corresponding results reveal that dopants which significantly distort the Dirac cone structure, such as O, S, P, Br, and Si, lead to significant degradation of carrier mobility and are thus excluded. In contrast, N is identified as the optimal dopant of Gr/Cu composites, outperforming B by effectively enhancing carrier concentration while well maintaining high carrier mobility, thereby achieving a superior balance for enhanced conductivity. This work establishes a theoretical framework for dopant selection and provides key insights for the design of high-conductivity Gr/Cu composites.
1 Introduction
With the rapid development of modern technology and emerging fields such as integrated circuits (ICs) and high-frequency communication, the requirements for copper (Cu) conductors with ultra-high electrical conductivity have become increasingly stringent. The development of high conductivity Cu foils is crucial for mitigating conductor losses in high frequency and high speed copper clad laminate applications, thereby supporting the advancement of 5G and next generation electronic technologies.1–4 However, conventional methods for enhancing the electrical conductivity of metals, such as purification and single-crystal growth, have reached their physical limits.5 Recently, graphene/copper (Gr/Cu) composites have emerged as a promising solution.3,4,6–9 Cao et al.7 fabricated multilayered Gr/Cu composites, achieving ultrahigh electrical conductivity (117% IACS). It is well-established that a material's conductivity is governed by both carrier mobility and carrier concentration.10 The enhanced electrical conductivity of Gr/Cu composites originates from two key aspects:Increased carrier concentration: Cu has a higher carrier concentration than Gr.11 When Gr contacts Cu, the difference in their work functions that Gr is higher than Cu drives electron transfer from Cu to Gr to align the Fermi levels,11,12 resulting in n-type doping of Gr and significantly increasing the effective carrier concentration within the Gr layer. This is supported by Kelvin probe force microscopy measurements showing a lower contact potential difference for Gr compared to Cu in Gr/Cu systems.3
Preserved high carrier mobility: Gr exhibits ultra-high carrier mobility due to its linear Dirac-cone band dispersion near the Fermi level, where charge carriers behave as massless Dirac fermions with negligible effective mass.13,14 Owing to the physisorption of Gr on the Cu substrate, where the Gr layer binds weakly to the Cu surface via van der Waals (vdW) interactions, the intrinsic band structure of Gr is preserved with only a rigid Fermi-level shift.15 This minimal disruption allows the Gr's intrinsic ultrahigh carrier mobility to be maximally preserved.
Thus, by optimally combining high carrier concentration from carrier transfer and high mobility from the electronic structure, Gr/Cu composites achieve superior conductivity. However, interfacial charge transfer is limited by the intrinsic work functions difference between the pristine Gr and Cu, which constraining the potential for further increase in carrier concentration. Therefore, it is necessary to regulate the electronic structure of Gr to induce a more substantial Fermi level shift, while simultaneously preserving its characteristic high-mobility band structure.
Chemical doping, is one of the most promising ways to tailor the electronic structure by carrier injection or extraction.16,17 Chemical doping of Gr can be achieved through two primary approaches: surface charge-transfer doping (via physical/chemical adsorption) and substitutional doping. Surface doping preserves Gr's structural integrity but often leads to non-uniformity and poor environmental stability. Moreover, the weak interaction between surface dopants and Gr renders the doping effect vulnerable to external factors. In contrast, substitutional doping, which incorporates heteroatom directly into the Gr lattice, provides stable, controllable, and uniform modulation of properties.16,18,19 It is therefore the preferred method for systematic studies.
Introducing heteroatom as dopants into Gr lattice would effectively modify the electronic structure of Gr and alter the carrier concentration. Such heteroatom dopants can be either electron donors or acceptors. Various nonmetallic elements including halogen (F, Cl, Br and I), N, B, S, P, and Si, have been incorporated into Gr lattice for efficient doping.16 However, assessing the impact of substitutional doping on the Gr/Cu interface is complex due to the variety of doping elements and their multiple bonding configurations within the Gr lattice. For example, N typically forms three types of bonding structures: graphitic, pyridinic, and pyrrolic.20,21 B can form two planar bonding configurations that are equivalent to graphitic and pyridinic.22 Crucially, the different configurations induce different changes in carrier concentration. For instance, graphitic N can contribute 0.5 electrons to the π network of Gr, leading to an n-doping effect.23 Conversely, pyridinic and pyrrolic N induce p-doping in Gr.20 Moreover, defect-associated configurations like pyridinic and pyrrolic N inherently introduce vacancies and structural distortions into the lattice.
To fully elucidate the influence of substitutional doping, it is essential to decouple the doping effect into two components: the intrinsic electronic properties of the dopant atom, and the effect of vacancies or other point defects associated with non-graphitic configurations. Previous studies have demonstrated that the dangling bonds of vacancies would distort or destroy the Dirac point, resulting in a slight decline of the Fermi level and electron deficiency.24,25 In this case, p-type doping is induced, and the carrier scattering would be enhanced.24 Therefore, to design doped Gr/Cu composites with superior electrical transport performance, it is necessary to further investigate the regulation of dopant atom itself on the electronic properties of Gr/Cu.
In this work, we conducted a systematic first-principle study of different doped (O-, N-, B-, S-, P-, Br-, Si-) Gr on Cu (111). The chosen dopants represent commonly reported heteroatoms that have been experimentally demonstrated16,26–32 to modify the electronic and interfacial properties of Gr. Among them, N and B are adjacent to carbon in the periodic table and readily substitute C atoms in graphene. Wei et al.26 has synthesized N-doped Gr on Cu and Pt foils via chemical vapor deposition (CVD) using NH3/CH4 precursors, achieving doping concentrations up to 8–9 at%. B-doped graphene has been reported on Cu, Ni, and Co substrates using precursors such as boron powder or carborane molecules, leading to controllable p-type doping effects.27,28 Other non-metallic dopants have also been experimentally realized.29–32 This study focuses on the graphitic configuration, which allows for a more direct assessment of dopant atom itself and minimizes the confounding negative impacts of other defects. The binding energy and the formation energy were calculated to describe the stability and feasibility of doped Gr/Cu. Then, the charge density difference (CDD) and electron localization (ELF) were used to analyze the effect of different dopant atoms on interfacial bonding. Subsequently, we could screen candidate dopants based on their electronic properties, including band structure and electronic density of states. This screening prioritized elements that effectively modulate the Fermi level while maximally preserving band structure of Gr. The quantitative calculations of carrier concentration and mobility further enabled us to decouple the independent contributions of the dopant elements to these two parameters. Our work provides a systematic, first-principles comparison of the intrinsic interfacial electronic interactions induced by different substitutional dopants at the Gr/Cu interface. While previous studies often focus on single dopants, our unified approach decouples the effects on carrier concentration and mobility across multiple dopant species, offering microscopic insights to guide the optimization of conductivity and interfacial performance. Ultimately, these comprehensive results provide a theoretical guidance for the rational selection of optimal dopants, enabling the synergistic optimization of carrier concentration and carrier mobility, and thereby facilitating the design of high-performance Gr/Cu composite.
2 Computational details in DFT
All DFT calculations were conducted with the Vienna ab initio simulation package (VASP).33 The Gr/Cu interface was modeled by a 4 × 4 × 1 supercell of Gr on Cu (111),34 with a lattice mismatch below 3% after initial optimization. The model, shown in Fig. 1(a) and (b), used top-fcc stacking configuration, which is the most energy-favored arrangement. A dopant atom (O, N, B, S, P, Br, or Si) was introduced by substituting a single C atom in Gr, corresponding to about 3 at% doping concentration. The 4 × 4 lateral cell ensures a ≥10 Å separation between periodic dopant images, thereby minimizing dopant–dopant interactions and preserving the isolated-impurity limit. This concentration was chosen based on our previous measurements of N-doped Gr/Cu, where peak conductivity was observed around ∼3 at%.11 A vacuum layer of ∼20 Å was added along the z-axis to mitigate periodic interactions. The dipole correction35 was neglected due to its negligible impact.The projector augmented wave (PAW)36 method was used to describe the electron–ion interactions, and the generalized gradient approximation (GGA) of Perdew–Burke–Ernzerhof (PBE)37 was adopted for the exchange-correlation functional. The DFT-D3 empirical correction38 was introduced to account for vdW interactions. A plane-wave energy cutoff of 450 eV was used. The convergence accuracy of the system total energy and the force per atom were 1.0 × 10−5 eV per atom and 0.02 eV Å−1, respectively. The Brillouin zone was sampled by a 8 × 8 × 1 Γ-centered k-mesh for optimization relaxation and 24 × 24 × 1 for the electronic structure calculations. VASPKIT39 was used for pre/post-processing, and VESTA40 was used to visualization.
3 Results and discussion
3.1 Interface structure analysis
In order to obtain the energy-favored configuration, structural relaxation of the Gr/Cu interface is necessary. Table 1 shows the structural parameters of Gr/Cu and doped Gr/Cu after relaxation. The equilibrium interfacial distance between Gr and Cu layers (dGr-Cu) is 3.306 Å, agreeing with reported literature.15 At low doping concentration, the equilibrium interfacial distances of all doped Gr/Cu exceed 3 Å, indicating that interfacial binding remains physisorbed interaction. However, doping induces in-plane lattice distortion due to the covalent radius mismatch between dopant and C atoms.41 The magnitude of the lattice parameter after doping changes by about 0.04° in the γ directions. Meanwhile, the lattice constant a and b are changed by −0.02 Å to 0.04 Å depending on the difference radius of the dopant atoms and C atom. In fact, due to the low doping concentration, the doping of foreign atoms will induce more local structure changes rather than all regions. Therefore, we calculated the in-plane distortion of doped Gr/Cu and compared the bond lengths as well as the distance between the dopant atoms and the Cu layer. The results for the in-plane distortion and the corresponding structural parameters are presented in Fig. 2 and Table 1, respectively. The C–C bonds in Gr/Cu is 1.43 Å, which is consistent with the actual.42,43 In this work, the distortion factor (εxy) is employed to quantitatively characterize the in-plane lattice distortion of the doped Gr/Cu system, as defined by eqn (1):
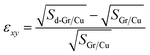 | (1) |
| Interfacial distance/dGr-Cu (Å) | ddopant-Cu (Å) | dC1-dopant (Å) | dC2-dopant (Å) | dC3-dopant (Å) | |
|---|---|---|---|---|---|
| Gr/Cu | 3.306 | 3.306 | 1.430 | 1.430 | 1.430 |
| OGr/Cu | 3.311 | 3.457 | 1.498 | 1.498 | 1.498 |
| NGr/Cu | 3.332 | 3.352 | 1.415 | 1.415 | 1.415 |
| BGr/Cu | 3.068 | 2.729 | 1.500 | 1.500 | 1.500 |
| SGr/Cu | 3.299 | 2.311 | 1.733 | 1.733 | 1.733 |
| PGr/Cu | 3.280 | 2.255 | 1.742 | 1.742 | 1.742 |
| BrGr/Cu | 3.434 | 2.124 | 1.997 | 1.997 | 1.995 |
| SiGr/Cu | 3.268 | 2.149 | 1.791 | 1.791 | 1.792 |
 | ||
| Fig. 2 The in-plane distortion εxy of doped Gr/Cu systems: (a) OGr/Cu, (b) NGr/Cu, (c) BGr/Cu, (d) SGr/Cu, (e) PGr/Cu, (f) BrGr/Cu, and (g) SiGr/Cu. | ||
Based on relaxation configuration, we calculated the formation energy (Ef) and the binding energy (Ebinding). All the results are summarized in Table 2. The formation energy (Ef) was calculated using the following equation:44
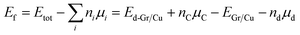 | (2) |
| Weakly-interacting | Strongly-interacting | |||||||
|---|---|---|---|---|---|---|---|---|
| Gr/Cu | OGr/Cu | NGr/Cu | BGr/Cu | SGr/Cu | PGr/Cu | BrGr/Cu | SiGr/Cu | |
| Ef/(eV) | — | 2.76 | 0.78 | −1.07 | 2.98 | −2.03 | 11.12 | 2.06 |
| Ebinding/(eV) | −2.68 | −2.80 | −2.81 | −3.33 | −3.19 | −4.00 | −3.33 | −4.24 |
The binding energy (Ebinding) was calculated by eqn (3):45
| Ebinding = Ed-Gr/Cu − Ed-Gr − ECu | (3) |
3.2 Electronic structure analysis
The observed lattice distortions and the variations in interfacial bonging are fundamentally influenced by the resultant changes in the electronic structure at the Gr/Cu interface. To uncover this electronic level origin, the charge density difference (CDD), the planar-averaged CDD and electron localization function (ELF) were calculated and showed in Fig. 3. The CDD of doped Gr/Cu is calculated by the following equation:47| Δρ = ρd−Gr/Cu − ρGr/Cu − ρCu | (4) |
To comprehensively explore the detailed mechanisms by which doping influences the electrical properties, the band structures and the projected density of states (PDOS) for doped Gr/Cu were calculated as presented in Fig. 4 and 5, respectively. Fig. 4(a) is a schematic of band structure evolution in Gr with Cu substrate and dopant introduction. The band structure of suspended Gr (Fig. 4(b), blue dashed line) exhibits a characteristic Dirac cone with linear dispersion and zero bandgap near the Fermi level,48 which ensures its ultra-high carrier mobility that can reach up to 106 cm2 s−1 V−1.49,50 When supported on Cu, the Fermi level shifts upward (ΔE = −0.25 eV), indicating n-type doping due to electron transfer from the substrate to Gr as depicted in Fig. 4(b). According to the equation 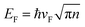 ,7,11,46 the shift of the Fermi level alters the carrier concentration. The introduction of doping significantly influences the electronic state, specially, leading to a shift of the Dirac point, and further modifying the carrier concentration. Moreover, doping causes an obvious bandgap, leads to local distortion, and disrupts the symmetry of Gr,51 resulting in a destroyed Dirac cone due to the enhanced interfacial interaction.15 The full band structures of doped Gr/Cu are shown in Fig. 4(c)–(h). NGr/Cu (Fig. 4(d)) and BGr/Cu (Fig. 4(e)) maintain a relatively intact Dirac cone, but open the bandgap. NGr/Cu shows a strong n-type shift (ΔE = −0.7 eV) compared to Gr/Cu, while BGr/Cu exhibits a p-type shift (ΔE = 0.24 eV) due to the reverse doping effect from B dopant and the Cu layer. In contrast, O, S, P and Si doping severely distort or destroy the Dirac cone as shown in Fig. 4(c) and (f)–(h). Fig. 5 presents the projected density of states (PDOS) for the doped Gr/Cu systems. In addition to the C-2p orbital, the dopant-2p orbital also contribute to the energy bands near the Fermi level. Obviously, The NGr/Cu interface exhibits n-type semiconducting characteristics and BGr/Cu interface exhibits p-type semiconducting characteristics in the doped interface systems, as shown in Fig. 5(b) and (c), respectively. For the BGr/Cu, the C-2p, B-2p and Cu-3d orbitals overlap at −2 eV ∼ −3 eV, which means stronger interfacial interaction compared to NGr/Cu. However, the interface interaction does not significantly change the properties of the doped Gr, both NGr/Cu and BGr/Cu exhibit Dirac cone. For OGr/Cu, the O-2s orbital peaks between 2 eV and 3 eV indicate that the impurity levels appearing in the Fig. 5(a) are mainly contributed by O dopant. Moreover, the peaks of Cu-3d orbitals and O-2p orbitals overlap at −2.5 eV and −4.5 eV, accompanied by orbital hybridization, resulting in the destruction of Dirac cone of OGr/Cu. For SGr/Cu, PGr/Cu and SiGr/Cu as shown in Fig. 5(d)–(f), the Cu-3d and dopant-2p exhibit orbital hybridization in valence band. The destruction of the Dirac cone means the carrier mobility is severely reduced for these systems. Moreover, the total density of states (TDOS) and PDOS for the pristine Gr/Cu system was also calculated in this work, as shown in Fig. S1. No obvious orbital hybridization is observed between Cu and C atoms, indicating the absence of covalent bonding, which is consistent with the analysis of the Gr/Cu models above. It can be seen that in the Gr/Cu interface, the energy bands near the Fermi level are mainly contributed by the C-2p orbital, while the Cu-3d orbital is primarily concentrated in the valence band. The interaction between Gr and the Cu substrate shifts the Fermi level into the conduction band, resulting in n-type semiconductor characteristics, and the Dirac cone of Gr remains intact. The intact Dirac cone preserve the high carrier mobility of Gr, while the destruction of the Dirac cone means the carrier mobility is severely reduced. Consequently, NGr/Cu and BGr/Cu, which preserve the Dirac cone morphology, are expected to maintain higher mobility, with NGr/Cu offering the highest carrier concentration due to the most negative ΔE (Fig. 4(i)).
,7,11,46 the shift of the Fermi level alters the carrier concentration. The introduction of doping significantly influences the electronic state, specially, leading to a shift of the Dirac point, and further modifying the carrier concentration. Moreover, doping causes an obvious bandgap, leads to local distortion, and disrupts the symmetry of Gr,51 resulting in a destroyed Dirac cone due to the enhanced interfacial interaction.15 The full band structures of doped Gr/Cu are shown in Fig. 4(c)–(h). NGr/Cu (Fig. 4(d)) and BGr/Cu (Fig. 4(e)) maintain a relatively intact Dirac cone, but open the bandgap. NGr/Cu shows a strong n-type shift (ΔE = −0.7 eV) compared to Gr/Cu, while BGr/Cu exhibits a p-type shift (ΔE = 0.24 eV) due to the reverse doping effect from B dopant and the Cu layer. In contrast, O, S, P and Si doping severely distort or destroy the Dirac cone as shown in Fig. 4(c) and (f)–(h). Fig. 5 presents the projected density of states (PDOS) for the doped Gr/Cu systems. In addition to the C-2p orbital, the dopant-2p orbital also contribute to the energy bands near the Fermi level. Obviously, The NGr/Cu interface exhibits n-type semiconducting characteristics and BGr/Cu interface exhibits p-type semiconducting characteristics in the doped interface systems, as shown in Fig. 5(b) and (c), respectively. For the BGr/Cu, the C-2p, B-2p and Cu-3d orbitals overlap at −2 eV ∼ −3 eV, which means stronger interfacial interaction compared to NGr/Cu. However, the interface interaction does not significantly change the properties of the doped Gr, both NGr/Cu and BGr/Cu exhibit Dirac cone. For OGr/Cu, the O-2s orbital peaks between 2 eV and 3 eV indicate that the impurity levels appearing in the Fig. 5(a) are mainly contributed by O dopant. Moreover, the peaks of Cu-3d orbitals and O-2p orbitals overlap at −2.5 eV and −4.5 eV, accompanied by orbital hybridization, resulting in the destruction of Dirac cone of OGr/Cu. For SGr/Cu, PGr/Cu and SiGr/Cu as shown in Fig. 5(d)–(f), the Cu-3d and dopant-2p exhibit orbital hybridization in valence band. The destruction of the Dirac cone means the carrier mobility is severely reduced for these systems. Moreover, the total density of states (TDOS) and PDOS for the pristine Gr/Cu system was also calculated in this work, as shown in Fig. S1. No obvious orbital hybridization is observed between Cu and C atoms, indicating the absence of covalent bonding, which is consistent with the analysis of the Gr/Cu models above. It can be seen that in the Gr/Cu interface, the energy bands near the Fermi level are mainly contributed by the C-2p orbital, while the Cu-3d orbital is primarily concentrated in the valence band. The interaction between Gr and the Cu substrate shifts the Fermi level into the conduction band, resulting in n-type semiconductor characteristics, and the Dirac cone of Gr remains intact. The intact Dirac cone preserve the high carrier mobility of Gr, while the destruction of the Dirac cone means the carrier mobility is severely reduced. Consequently, NGr/Cu and BGr/Cu, which preserve the Dirac cone morphology, are expected to maintain higher mobility, with NGr/Cu offering the highest carrier concentration due to the most negative ΔE (Fig. 4(i)).
3.3 Carrier mobility and carrier concentration
To critically evaluate the relative advantages of N doping versus B doping on carrier concentration and mobility, we performed quantitative calculations of these parameters specifically for NGr/Cu and BGr/Cu systems. The carrier transport performance of the doped Gr/Cu at room temperature is estimated by the acoustic phonon limited method, based on the deformation potential theory proposed by Bardeen and Shockley. We first transform the original hexagonal unit cell of Gr/Cu into the rectangular unit cell. Generally, the carrier mobility, µ2D, of the 2D materials, with a view to studying the properties of migration of electrons and holes, can be obtained as follows:52–54
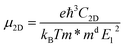 | (5) |
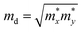 is the average effective mass. E1 = ΔEedge/(Δl/l0) is the deformation potential (DP) constant, defined as the shift of band edges induced by strain. Here, ΔEedge represents the energy difference about conduction band minimum (CBM) or valence band maximum (VBM) with the lattice applied y proper dilatation or compression. l0 and Δl are the lattice constant along the x or y direction and their deformation, corresponding. In the 2D materials, in-plane stiffness, C2D, can be calculated using C2D = [∂2E/∂(Δl/l0)2]/S0, where E and S0 stand for the total energy and the area of the xy plane for the supercell, respectively. Besides, e, ℏ, kB, and T are electronic quantity, reduced Planck constant, Bolzban constant, and 300 K, respectively.
is the average effective mass. E1 = ΔEedge/(Δl/l0) is the deformation potential (DP) constant, defined as the shift of band edges induced by strain. Here, ΔEedge represents the energy difference about conduction band minimum (CBM) or valence band maximum (VBM) with the lattice applied y proper dilatation or compression. l0 and Δl are the lattice constant along the x or y direction and their deformation, corresponding. In the 2D materials, in-plane stiffness, C2D, can be calculated using C2D = [∂2E/∂(Δl/l0)2]/S0, where E and S0 stand for the total energy and the area of the xy plane for the supercell, respectively. Besides, e, ℏ, kB, and T are electronic quantity, reduced Planck constant, Bolzban constant, and 300 K, respectively.
The calculated carrier mobilities of NGr/Cu and BGr/Cu are summarized in Table 3 and Fig. 6(a). The results revealed that the carrier mobilities of NGr/Cu and BGr/Cu show anisotropy between electron and hole. Consistent with their n-type and p-type doping characters, the mobility corresponds to electron transport for NGr/Cu and hole transport for BGr/Cu. NGr/Cu has the electron mobilities of 2.74 × 105 and 2.33 × 105 cm2 s−1 V−1 in the zigzag and armchair directions, respectively, which are both more than the hole mobility of BGr/Cu. According to the formula of µ2D, the greatly enhanced electron mobility in the NGr/Cu mainly results from the lowest the deformation potential (DP) compared to the BGr/Cu. Fig. 2(b) and (c) shows the in-plane distortion of NGr/Cu and BGr/Cu are 7.1% and 14.1% (the sum of all atomic distortions in the plane), respectively. The greater in-plane lattice distortion in BGr/Cu and the localized states induced by B doping (Fig. 3(a)) contribute to enhanced carrier scattering and consequently degraded mobility in BGr/Cu.
| C2D−x (J m−2) | C2D−y | E1−x(J) | E1−y | me | µx | µy | |
|---|---|---|---|---|---|---|---|
| Electrons | |||||||
| NGr/Cu | 275.58 | 283.86 | −3.67 × 10−19 | −4.04 × 10−19 | 0.093 | 27.36 | 23.25 |
| BGr/Cu | 265.37 | 268.98 | −4.73 × 10−19 | −5.29 × 10−19 | 0.064 | 7.49 | 6.08 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
|||||||
| Holes | |||||||
| NGr/Cu | 275.58 | 283.86 | −3.90 × 10−19 | −3.754 × 10−19 | 0.069 | 12.63 | 14.06 |
| BGr/Cu | 265.37 | 268.98 | −6.44 × 10−19 | −6.008 × 10−19 | 0.089 | 7.32 | 8.52 |
 | ||
| Fig. 6 Carrier mobility (µ) and carrier concentration (n) of NGr/Cu, BGr/Cu, and BNGr/Cu along different directions. (b) Electrical conductivity of Gr/Cu, NGr/Cu,11 and BGr/Cu57 in experiment. | ||
According to the equation 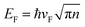 , the carrier concentration is positively correlated with the shift of Fermi level. This relationship, however, assumes Gr's linear dispersion relation. When doping induced bandgap opening, the linearity may be disrupted. To address this nonlinear regime, eqn (6) was derived under parabolic band conditions to calculate carrier concentration. The m* is carrier effective mass. ΔE is a shift of the Fermi level and ℏ is reduced Planck constant. The computational results were detailed in Table 4 and Fig. 6(a), with the full derivation process provided in SI. The results are consistent with the experiment.55,56 NGr/Cu has higher carrier concentration due to the combined doping effects from N dopant and Cu. Although electrons transfer from Cu to Gr occurs in BGr/Cu, B doping induces holes, resulting in a lower carrier concentration in BGr/Cu. This trend is reflected in the experimental conductivities shown in Fig. 6(b).11,57 The peak conductivity of NGr/Cu reaches 104.7% IACS, an 11.14% enhancement over pristine Gr/Cu (94.2% IACS). BGr/Cu also shows a peak conductivity (102.3% IACS) exceeding that of Gr/Cu, its overall performance is inferior to N-doping. Both theoretical and experimental analyses confirm that N doping is the optimal strategy for synergistically regulating carrier concentration and mobility. It should be emphasized, however, that the present calculations are performed under an idealized single-atom substitutional doping. In practical fabrication processes, additional structural infects, such as grain boundaries or non-graphitic N configurations (e.g., pyridinic N), are often present and can introduce extra carrier scattering, thereby reducing the measured conductivity relative to the ideal case. These effects are not explicitly considered in our current framework. Consequently, the conductivity values predicted here should be regarded as the intrinsic upper limit achievable for defect-free Gr/Cu interfaces under substitutional doping, serving as a benchmark for guiding experimental optimization and defect control strategies.
, the carrier concentration is positively correlated with the shift of Fermi level. This relationship, however, assumes Gr's linear dispersion relation. When doping induced bandgap opening, the linearity may be disrupted. To address this nonlinear regime, eqn (6) was derived under parabolic band conditions to calculate carrier concentration. The m* is carrier effective mass. ΔE is a shift of the Fermi level and ℏ is reduced Planck constant. The computational results were detailed in Table 4 and Fig. 6(a), with the full derivation process provided in SI. The results are consistent with the experiment.55,56 NGr/Cu has higher carrier concentration due to the combined doping effects from N dopant and Cu. Although electrons transfer from Cu to Gr occurs in BGr/Cu, B doping induces holes, resulting in a lower carrier concentration in BGr/Cu. This trend is reflected in the experimental conductivities shown in Fig. 6(b).11,57 The peak conductivity of NGr/Cu reaches 104.7% IACS, an 11.14% enhancement over pristine Gr/Cu (94.2% IACS). BGr/Cu also shows a peak conductivity (102.3% IACS) exceeding that of Gr/Cu, its overall performance is inferior to N-doping. Both theoretical and experimental analyses confirm that N doping is the optimal strategy for synergistically regulating carrier concentration and mobility. It should be emphasized, however, that the present calculations are performed under an idealized single-atom substitutional doping. In practical fabrication processes, additional structural infects, such as grain boundaries or non-graphitic N configurations (e.g., pyridinic N), are often present and can introduce extra carrier scattering, thereby reducing the measured conductivity relative to the ideal case. These effects are not explicitly considered in our current framework. Consequently, the conductivity values predicted here should be regarded as the intrinsic upper limit achievable for defect-free Gr/Cu interfaces under substitutional doping, serving as a benchmark for guiding experimental optimization and defect control strategies.
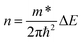 | (6) |
4 Conclusions
Ultra-high-conductivity Cu materials are critically needed in advanced electrical and electronic fields, including high-frequency communication, 5 G infrastructure, integrated circuits, and power transmission. Enhancing Cu conductivity can significantly reduce energy loss, minimize device size and weight, and overcome performance degradation due to electron scattering at scaled-down interconnects. Doped Gr/Cu composites represent a promising strategy to improve interfacial electron coupling and overall conductivity of Cu-based systems. In this work, the effects of various doping elements (O, N, B, S, P, Br and Si) on the interfacial and electrical properties of Gr/Cu composites were investigated systematically by using the first-principles calculations. Our findings reveal competing effects of doping:(i) Interfacial binding enhancement: doping can enhance the interfacial binding strength, where the interaction generally intensifies as the atomic radius increases. However, an overly strong interaction between the dopant and Cu atoms introduces localized electronic states, thereby enhancing carrier scattering and resulting in reduced mobility.
(ii) Carrier concentration modulation: doping effectively modulates the electronic structure of Gr by injecting either electrons or holes, thereby tuning the carrier concentration.
N doping was found to achieve an optimal balance between carrier concentration (1.4 × 1013 cm−2) and carrier mobility (2 × 105 cm2 s−1 V−1). Consequently, nitrogen (N) stands out as the most suitable dopant for achieving a breakthrough in high-conductivity Gr/Cu composites. Future work could include experimental validation of the predicted optimal dopant configurations, along with systematic studies on the influence of doping concentration, common interfacial defects, and interfacial modification on carrier transport. Such efforts would not only substantiate the present theoretical framework but also guide the practical design of high-conductivity Gr/Cu composites.
Author contributions
Xinsi Zhao: data curation (lead); formal analysis (lead); methodology (lead); writing – original draft (lead). Boan Zhong: methodology (equal); validation (equal); visualization (equal). Baixue Bian: methodology (equal); validation (equal); writing – review & editing (equal). Jiamiao Ni: methodology (equal); validation (equal); Mingyu Gong: conceptualization (equal); funding acquisition (lead); methodology (equal); formal analysis (equal); investigation (equal); validation (equal); writing – review & editing (lead). Yue Liu: conceptualization (equal); methodology (equal); validation (equal). Tongxiang Fan: project administration (lead); software (supporting); supervision (supporting).Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting the findings of this study are available from the corresponding authors upon reasonable request.Supplementary information (SI): DOS of the Gr/Cu interface (Fig. S1); and derivation of equation used in carrier concentration in doped Gr/Cu. See DOI: https://doi.org/10.1039/d5ra09101b.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (No. 52201161).Notes and references
- Y. Zhou, Acc. Mater. Res., 2021, 2, 306–310 CrossRef CAS.
- R. A. Maniyara, D. Rodrigo, R. Yu, J. Canet-Ferrer, D. S. Ghosh, R. Yongsunthon, D. E. Baker, A. Rezikyan, F. J. García de Abajo and V. Pruneri, Nat. Photonics, 2019, 13, 328–333 CrossRef CAS.
- S. He and D. Xiong, J.Nonferrous Met., 2023, 33(5), 1349–1377 Search PubMed.
- B. Gwalani, X. Li, A. Nittala, W. Choi, M. Reza-E-Rabby, J. E. Atehortua, A. Bhattacharjee, M. Pole, J. Silverstein, M. Song and K. Kappagantula, Mat. Design, 2024, 237, 112555 CrossRef CAS.
- M. Ajmal, S. Lee, Y. C. Cho, S. J. Kim, S. E. Park, C. R. Cho and S. Y. Jeong, CrystEngComm, 2012, 14, 1463–1467 RSC.
- X. Zhang, N. Zhao and C. He, Prog. Mater. Sci., 2010, 113, 100672 CrossRef.
- M. Cao, D. B. Xiong, L. Yang, S. Li, Y. Xie, Q. Guo, Z. Li, H. Adams, J. Gu, T. Fan, X. Zhang and D. Zhang, Adv. Funct. Mater., 2019, 29, 1806792 CrossRef.
- J. Yu, L. Wang, Y. Guan, B. Shao, Y. Zong and C. Pt, Composites, Part A, 2024, 185, 108345 CrossRef CAS.
- C. Pan, A. P. S. Gaur, M. Lynn, M. P. Olson, G. Ouyang and J. Cui, AIP Adv., 2022, 12, 015310 Search PubMed.
- M. Hofmann, Y. P. Hsieh, K. W. Chang, H. G. Tsai and T. T. Chen, Sci. Rep., 2015, 5, 17393 CrossRef CAS PubMed.
- Y. Wang, B. Zhong, J. Ni, J. Song, Y. Huang, S. Yao, Y. Liu and T. Fan, Scr. Mater., 2024, 239, 115797 Search PubMed.
- G. Giovannetti, P. A. Khomyakov, G. Brocks, V. M. Karpan, J. van den Brink and P. J. Kelly, Phys. Rev. Lett., 2008, 101, 026803 Search PubMed.
- A. H. Castro Neto, F. Guinea, N. M. R. Peres, K. S. Novoselov and A. K. Geim, Rev. Mod. Phys., 2009, 81, 109 CrossRef CAS.
- N. O. Weiss, H. Zhou, L. Liao, Y. Liu, S. Jiang, Y. Huang and X. Duan, Adv. Mater., 2012, 24, 5782–5825 CrossRef CAS PubMed.
- M. Yang, Y. Liu, T. Fan and D. Zhang, Prog. Mater. Sci., 2020, 110, 100652 CrossRef CAS.
- Z. Hu, Y. Zhao, W. Zou, Q. Lu, J. Liao, F. Li, M. Shang, L. Lin and Z. Liu, Adv. Funct. Mater., 2022, 32, 2203179 CrossRef CAS.
- S. Ullah, Q. Shi, J. Zhou, X. Yang, H. Q. Ta, M. Hasan, N. M. Ahmad, L. Fu, A. Bachmatiuk and M. H. Rümmeli, Adv. Mater. interfaces., 2020, 7, 2000999 CrossRef CAS.
- L. Ma, W. Ren and H. Cheng, Acta Phys.-Chim. Sin., 2022, 38(1), 2012080 Search PubMed.
- Y. Zhou, X. He and M. Li, AIP Adv., 2025, 15, 010701 CrossRef CAS.
- S. Jalili and R. Vaziri, Mol. Phys., 2011, 109, 687–694 CrossRef CAS.
- Q. Jia, B. Zhong, S. Wang, Z. Yue, Y. Liu and X. Wang, Phys. Status Solidi RRL, 2025, 19, 2400165 CrossRef CAS.
- S. Agnoli and M. Favaro, J. Mater. Chem. A, 2016, 4, 5002–5025 RSC.
- J. Zhang, C. Zhao, N. Liu, H. Zhang, J. Liu, Y. Q. Fu, B. Guo, Z. Wang, S. Lei and P. Hu, Sci. Rep., 2016, 6, 28330 CrossRef CAS PubMed.
- Y. Liu, L. An and L. Gong, Mod. Phys. Lett. B, 2018, 32, 1850139 CrossRef CAS.
- E. Zaminpayma, M. E. Razavi and P. Nayebi, Appl. Surf. Sci., 2017, 414, 101–106 CrossRef CAS.
- D. Wei, Y. Liu, Y. Wang, H. Zhang, L. Huang and G. Yu, Nano lett., 2009, 9(5), 1752–1758 CrossRef CAS PubMed.
- X. Li, L. Fan, Z. Li, K. Wang, M. Zhong, J. Wei, D. Wu and H. Zhu, Adv. Energy Mater., 2012, 2, 425 CrossRef CAS.
- D. Y. Usachov, A. V. Fedorov, A. E. Petukhov, O. Y. Vilkov, A. G. Rybkin, M. M. Otrokov, A. Arnau, E. V. Chulkov, L. V. Yashina, M. Farjam, V. K. Adamchuk, B. V. Senkovskiy, X. C. Laubschat and D. V. Vyalikh, ACS Nano, 2015, 9(7), 7314–7322 CrossRef CAS PubMed.
- M. Ovezmyradov, I. V. Magedov, L. V. Frolova, G. Chandler, J. Garcia, D. Bethke, E. A. Shaner and N. G. Kalugin, J. Nanosci. Nanotechnol., 2015, 15, 4883 CrossRef CAS PubMed.
- A. Ganguly, S. Sharma, P. Papakonstantinou and J. Hamilton, J. Phys. Chem. C, 2011, 115(34), 17009–17019 CrossRef CAS.
- L. Guo and T. Li, Sens. Actuators, B, 2018, 255, 2258–2263 CrossRef CAS.
- M. Hasan, W. Meiou, Y. Liu, S. Ullah, H. Q. Ta, L. Zhao, R. G. Mendes, Z. P. Malik, N. Ahmad, Z. Liu and M. H. Rummeli, RSC Adv., 2019, 9, 13527 RSC.
- G. Kresse and J. Furthmüller, Phys. Rev. B: Condens. Matter Mater. Phys., 1996, 54, 11169 CrossRef CAS PubMed.
- L. Zhang, J. Dong and F. D, J. Phys. Chem. Lett., 2014, 15, 758–765 CrossRef PubMed.
- L. Bengtsson, Phys. Rev. B: Condens. Matter Mater. Phys., 1999, 59, 12301 CrossRef CAS.
- G. Kresse and D. Joubert, Phys. Rev. B: Condens. Matter Mater. Phys., 1999, 59, 1758 CrossRef CAS.
- J. P. Perdew, K. Burke and M. Ernzerhof, Phys. Rev. Lett., 1996, 77, 3865 CrossRef CAS PubMed.
- S. Grimme, J. Antony, S. Ehrlich and H. Krieg, J. Chem. Phys., 2010, 132, 154104 CrossRef PubMed.
- V. Wang, N. Xu, J. C. Liu, G. Tang and W. T. Geng, Comput. Phys. Commun., 2021, 267, 108033 CrossRef CAS.
- K. Momma and F. Izumi, J. Appl. Crystallogr., 2011, 44, 1272–1276 CrossRef CAS.
- Y. Liao, R. He, W. Pan, Y. Li, Y. Wang, J. Li and Y. Li, Chem.–Eng. J., 2023, 464, 142669 CrossRef CAS.
- P. Avouris, Nano Lett., 2010, 10, 4285–4294 CrossRef CAS PubMed.
- Y. Qu, J. Ding, H. Fu, H. Chen and J. Peng, Appl. Surf. Sci., 2021, 542, 148763 CrossRef CAS.
- C. G. Van de Walle and J. Neugebauer, J. Appl. Phys., 2004, 95, 3851–3879 CrossRef CAS.
- L. Ferrighi, M. I. Trioni and C. Di Valentin, J. Phys. Chem. C, 2015, 119, 6056–6064 CrossRef CAS.
- B. Zhong, J. Ni, Q. Zhang, J. Song, Y. Liu, M. Gong and T. Fan, J. Appl. Phys., 2024, 136, 055107 CrossRef CAS.
- S. He, B. Liu, Z. Pei, X. Zhang, B. Liu and D. Xiong, J. Appl. Phys., 2023, 134, 075104 CrossRef CAS.
- H. Şahin, S. Cahangirov, M. Topsakal, E. Bekaroglu, E. Akturk, R. T. Senger and S. Ciraci, Phys. Rev. B: Condens. Matter Mater. Phys., 2009, 80, 155453 CrossRef.
- N. Weiss, H. Zhou, L. Liao, Y. Liu, S. Jiang, Y. Huang and X. Duan, Adv. Mater., 2012, 24, 5782–5825 CrossRef CAS PubMed.
- Y. Xue, B. Wu, Q. Bao and Y. Liu, Small, 2014, 15, 2975–2991 CrossRef PubMed.
- N. Liu, S. Zhou and J. Zhao, Acta Phys.-Chim. Sin., 2019, 35, 1142–1149 CAS.
- S. Bruzzone and G. Fiori, Appl. Phys. Lett., 2011, 99, 222108 CrossRef.
- T. Liang, C. Hu, M. Lou, Z. Feng, D. Wang, X. Cai and L. Lin, Langmuir, 2023, 39, 10769–10778 CrossRef CAS PubMed.
- Z. Yan, R. Gao, M. Chang, S. Qi and X. Xu, Magn. Magn. Mater, 2020, 496, 165966 Search PubMed.
- L. Zhao, M. Levendorf, S. Goncher, T. Schiros, L. Pálová, A. Zabet-Khosousi, K. T. Rim, C. Gutiérrez, D. Nordlund, C. Jaye, M. Hybertsen, D. Reichman, G. Flynn, J. Park and A. Pasupathy, Nano Lett., 2013, 13, 4659–4665 Search PubMed.
- M. Scardamaglia, C. Struzzi, S. Osella, N. Reckinger, J.-F. Colomer, L. Petaccia, R. Snyders, D. Beljonne and C. Bittencourt, 2D Mater., 2016, 3, 011001 CrossRef.
- M. Yang, T. Li, W. Liu, Y. Wang, H. Hou, CN 112877562 B, 2022.
| This journal is © The Royal Society of Chemistry 2026 |




