 Open Access Article
Open Access ArticleFabrication of potential environmentally friendly 3D printing biocomposite filament based on poly(lactic acid) and cellulose fibers from agricultural waste
Nguyen Chi Thanh *a,
Pham Duc Thinha,
Bui Phuong Donga,
Tran Hai Cata and
Nguyen Thanh Huy
*a,
Pham Duc Thinha,
Bui Phuong Donga,
Tran Hai Cata and
Nguyen Thanh Huy bc
bc
aFaculty of Applied Sciences, Ho Chi Minh City University of Technology and Engineering, 01 Vo Van Ngan Street, Thu Duc Ward, Ho Chi Minh City, Vietnam. E-mail: thanhnc@hcmute.edu.vn; 20130064@student.hcmute.edu.vn; 20130020@student.hcmute.edu.vn; catth@hcmute.edu.vn
bDepartment of Polymer Materials, Faculty of Materials Technology, Ho Chi Minh City University of Technology (HCMUT), 268 Ly Thuong Kiet Street, Dien Hong Ward, Ho Chi Minh City, Vietnam. E-mail: nthuy.sdh241@hcmut.edu.vn
cVietnam National University Ho Chi Minh City, Thu Duc Ward, Ho Chi Minh City, Vietnam
First published on 23rd February 2026
Abstract
This study focuses on the fabrication of biocomposites based on poly(lactic acid) (PLA) and cellulose fibers (CMF) extracted from corn cob agricultural waste using a simple alkaline hydrogen peroxide (HPK) treatment. The HPK process was optimized by varying the ratio of weight of raw material and volume of used chemicals (wt/V), reaction temperature, and reaction time. The extracted cellulose fibers were used as reinforcing agents in the PLA matrix to improve the mechanical properties of the biocomposites. To enhance compatibility and interfacial adhesion between the cellulose fibers and the polymer matrix, the fiber surface was chemically modified using rice bran oil (RBO), an environmentally friendly modification agent. Scanning electron microscopy (SEM) and Fourier-transform infrared spectroscopy (FTIR) analyses confirmed the effective removal of surface impurities, hemicellulose, and lignin from the corn cobs after HPK treatment. X-ray diffraction (XRD) results showed that the treated cellulose fibers exhibited a higher crystallinity index than the raw material, while thermogravimetric analysis (TGA) demonstrated improved thermal stability of the cellulose fibers. Differential scanning calorimetry (DSC) indicated that the incorporation of RBO-modified cellulose fibers enhanced the crystallization rate of PLA, and rheological measurements revealed higher viscosity of PLA/RBO1 biocomposites compared to neat PLA across the entire shear rate range. Tensile test results demonstrated that biocomposites containing RBO-modified cellulose fibers exhibited significantly improved mechanical properties, with the composite containing 1 wt% CMF achieving the highest performance. Furthermore, PLA/RBO1 composite filaments showed good printability via fused deposition modeling (FDM), highlighting their potential as environmentally friendly materials for 3D printing applications.
Introduction
In the context of increasing environmental pollution and the urgent need for sustainable materials, the development of bio-based composites from renewable resources has garnered significant attention.1 The extraction and utilization of non-renewable resources not only deplete natural reserves but also exacerbate plastic waste and environmental pollution issues. This scenario highlights the critical demand for scientists and industries to explore and develop novel materials derived from renewable, eco-friendly, and biodegradable sources.2Vietnam is an agricultural country with a substantial amount of agricultural waste generated annually.3 Among the by-products, corn cob-a residue from corn processing has attracted growing interest due to its high recyclability and wide-ranging applications.4 The utilization of corn cobs not only reduces agricultural waste but also adds economic value to the circular economy. The primary composition of corn cobs includes cellulose (30–50%), hemicellulose (30–40%), and lignin (5–10%). Due to their high cellulose content, corn cobs serve as an excellent raw material for developing environmentally friendly bio-based materials.5
Cellulose extracted from lignocellulosic biomass was obtained at the nano or micro scale using various methods, including chemical, biological, and mechanical processes, to remove hemicellulose, lignin, pectin, and other amorphous components found in corn cobs.6 Chemical methods commonly used for cellulose extraction involved acid and alkaline treatments. Alkaline solutions such as NaOH, KOH, and Ca(OH)2 were typically employed to remove hemicellulose and lignin from the chemical structure of corn cobs, while inorganic acids (H2SO4, HNO3,…) and organic acids (acetic acid, formic acid,…) were used to isolate cellulose.7 However, chemical treatments generated byproducts that contributed to environmental pollution and degraded the quality of the extracted cellulose. Biological methods, which were considered environmentally friendly, eliminated the need for hazardous chemicals. These methods utilized microorganisms and enzymes to degrade hemicellulose and lignin, yielding high-purity cellulose.8 Despite their advantages-such as sustainability and minimal chemical consumption-biological methods had certain limitations, including high costs, slow processing times, and challenges in process control. Meanwhile, mechanical methods relied on strong physical forces to separate cellulose fibers. Common techniques included grinding, high-pressure homogenization, and ultrasonication.9 However, mechanical processing was highly energy-intensive and was often combined with other methods to enhance extraction efficiency. Each method had its own advantages and limitations, and the selection of an appropriate approach depended on the intended application. In this study, chemical treatment using an H2O2/NaOH system was applied to extract cellulose from agricultural waste, specifically corn cobs.
As a natural polymer, cellulose fibers possess significant advantages such as non-toxicity, biodegradability, renewability, and biocompatibility, making this natural fiber an attractive alternative to synthetic polymers in various applications, including food, pharmaceuticals, cosmetics, and composite materials.10,11
Poly(lactic acid) (PLA) is a bio-based polymer synthesized from renewable feedstocks such as corn starch and sugarcane.2 With the chemical formula (C3H4O2)n, PLA belongs to the poly(α-hydroxy ester) group and features a helical molecular structure with orthorhombic unit cells, forming a tightly packed network.12 This molecular structure significantly influences the physical properties of PLA, including its melting temperature (Tm) and glass transition temperature (Tg).13,14 PLA is widely used due to its biodegradability, excellent biocompatibility, and ease of processing.15 However, PLA exhibits several limitations, such as brittleness, low thermal stability, and poor crystallinity, which hinder its mechanical performance.16 Incorporating cellulose fibers into the PLA matrix had been demonstrated as an effective approach to enhance mechanical strength, thermal stability, and crystallinity while maintaining the material's biodegradability.17
Reported studies explored the reinforcement of PLA-based composites using nanocrystalline cellulose (CNCs) extracted from corn cobs, which significantly improved the tensile strength and crystallinity index of polymer composite.1 Furthermore, surface modification of cellulose fibers had been shown to enhance fiber-matrix interfacial adhesion, leading to superior mechanical properties in biocomposites.18
3D printing technology is revolutionizing industrial manufacturing due to its flexibility, high customization potential, and ability to minimize material waste.19 However, most current 3D printing materials are synthetic polymers, which are non-biodegradable and significantly contribute to environmental pollution.20 Therefore, the development of biodegradable and renewable 3D printing materials has emerged as a critical trend in sustainable manufacturing.15,21
Based on this premise, this study focused on the fabrication of PLA-based biocomposites reinforced with corn cobs-derived cellulose fibers, aiming to develop eco-friendly materials for 3D printing applications.22,23 By optimizing cellulose extraction and surface modification processes, this research not only seeks to reduce environmental pollution but also addresses the growing demand for bio-based 3D printing materials, contributing to the advancement of a circular and sustainable economy.
Materials and methods
Materials
Corn cobs were collected from Giang Dien Commune, Trang Bom District, Dong Nai Province, Vietnam. Hydrogen peroxide (H2O2), sodium hydroxide (NaOH), and ethanol were obtained from Sigma-Aldrich. A commercial grade of PLA (PLA 4043D) was purchased from NatureWorks. Distilled water was supplied by the Material Technology Laboratory, Faculty of Applied Sciences, Ho Chi Minh City University of Technology and Education (HCMUTE).Methods
After the reaction, the mixture was filtered to separate the solid residue (primarily cellulose) from the liquid phase containing degraded components. The solid residue was washed thoroughly with distilled water until a neutral pH was achieved to remove any remaining impurities. The reaction conditions, such as the raw material-to-chemical solution ratio, reaction temperature, and reaction time, were respectively presented in Tables 1–3.
| Reaction parameters | CMF-TL1 | CMF-TL2 | CMF-TL3 |
|---|---|---|---|
| a This table presents the experimental conditions for testing the influence of the raw material-to-chemical solution ratio on the extraction of cellulose from corn cobs. The ratios of 1/10, 1/15, and 1/20 (wt/V) were tested under a constant reaction time of 4 hours at 50 °C. | |||
| Raw material-to-chemical solution ratioa (wt/V) | 1/10 | 1/15 | 1/20 |
| Reaction time (hours) | 4 | 4 | 4 |
| Reaction temperature (°C) | 50 | 50 | 50 |
| Reaction parameters | CMF-T1 | CMF-T2 | CMF-T3 |
|---|---|---|---|
| a The table shows the experimental conditions used to examine the effect of varying reaction temperatures on the extraction process. The reaction temperature was varied at 50 °C, 60 °C, and 70 °C, while the raw material-to-chemical ratio was maintained at 1/10 (wt/V) for 4 hours. | |||
| Raw material-to-chemical solution ratio (wt/V) | 1/10 | 1/10 | 1/10 |
| Reaction time (hours) | 4 | 4 | 4 |
| Reaction temperaturea (°C) | 50 | 60 | 70 |
| Reaction parameters | CMF-T1 | CMF-T2 | CMF-T3 |
|---|---|---|---|
| a This table outlines the experimental conditions used to investigate the influence of reaction time on the cellulose extraction process from corn cobs treated with hydrogen peroxide-alkaline (HPK). The experiment was conducted at three different reaction times (3, 4, and 5 hours) under a constant raw material-to-chemical ratio of 1/10 (wt/V) and reaction temperature of 50 °C. | |||
| Raw material-to-chemical solution ratio (wt/V) | 1/10 | 1/10 | 1/10 |
| Reaction timea (hours) | 3 | 4 | 5 |
| Reaction temperature (°C) | 50 | 50 | 50 |
| Symbol | PLA (wt%) | Unmodified CMF (wt%) | RBO-chemically modified CMF (wt%) |
|---|---|---|---|
| PLA | 100 | — | — |
| PLA/CMF1 | 99.0 | 1.0 | — |
| PLA/CMF5 | 95.0 | 5.0 | — |
| PLA/CMF10 | 90.0 | 10.0 | — |
| PLA/RBO1 | 99.0 | — | 1.0 |
| PLA/RBO5 | 95.0 | — | 5.0 |
| PLA/RBO10 | 90.0 | — | 10.0 |
PLA and cellulose fibers were dried in an oven at 70 °C for 3 hours to remove residual moisture. This step ensures material quality and stability during melting mixing. Subsequently, the determined amounts of dried PLA and cellulose fiber were pre-mixed using a mechanical mixer. All compositions of biocomposites were charged into the mixing chamber at the same time. The fabrication process of the biocomposites based on the extracted cellulose fibers and PLA was illustrated in Fig. 2.
Characterization analysis
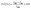 | (1) |
The crystallinity index (CrI) represents the relative degree of crystallinity within the material. The intensity I200 corresponds to the maximum diffraction intensity of the 002 lattice plane, typically observed between 2θ = 22° and 23°, while Iam represents the minimum intensity, located between the 002 and 101 diffraction peaks, generally within the range of 2θ = 18° to 19°. It is assumed that Iam primarily accounts for the amorphous contribution with minimal crystalline influence, whereas I200 encompasses both crystalline and amorphous regions.24
The crystallinity index (CrI) of PLA and PLA/CMF composites was calculated according to eqn (2).
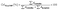 | (2) |
Thermal properties of PLA and PLA/CMF biocomposites were characterized by differential scanning calorimetry (DSC) (model: DSC204F1 Phoenix) technique. The measurements were conducted by heating the specimens from 40 to 200 °C at a heating rate of 5 °C min−1 for the first heating scan to remove thermal history. After keeping the specimens at 200 °C for 5 min, they were cooled to 40 °C at 5 °C min−1. Subsequently, the samples were heated again to 200 °C at 5 °C min−1 for the second heating scan.
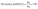 | (3) |
 | (4) |
Results and discussion
Crystalline structure of corn cobs and extracted cellulose fibers
 | ||
| Fig. 3 XRD patterns of the untreated corn cobs sample (CC) and the hydrogen peroxide alkaline treated samples with different raw material-to-chemical solution ratios (CMF-TL1, CMF-TL2, CMF-TL3). | ||
| Sample | Crystallinity index (%) |
|---|---|
| a a: Do not show statistically significant differences among the samples. | |
| CC | 35.42 ± 1.40 |
| CMF-TL1 | 39.03 ± 0.98a |
| CMF-TL2 | 38.56 ± 0.76a |
| CMF-TL3 | 38.93 ± 1.02a |
Compared to the CC sample, all CMF-TL treated samples exhibited higher crystallinity index, indicating that the HPK treatment effectively removed amorphous components such as hemicellulose, lignin, and non-cellulose compounds. Furthermore, as shown in Table 5, decreasing the ratio of raw material to chemical solution from 1/10 (wt/V) to 1/15 (wt/V) resulted in a decrease in crystallinity index from 40.48 ± 0.52% to 38.56 ± 0.76%. When the ratio was changed from 1/15 (wt/V) to 1/20 (wt/V), a slight increase in crystallinity was observed. The higher chemical volume contributed to the removal of impurities like hemicellulose, lignin, and other non-cellulosic substances, however, it may also have slightly reduced the cellulose crystallinity due to partial hydrolysis.28 Among the investigated raw material-to-H2O2 solution ratios, the 1/10 (wt/V) ratio exhibited a higher crystallinity index than the 1/15 (wt/V) and 1/20 (wt/V) ratios, although the differences were not statistically significant, as shown by the ANOVA analysis in Table S1 of the SI. Therefore, the 1/10 (wt/V) ratio was selected for subsequent experiments to optimize the process efficiency.
As observed from Fig. 4, the diffraction peaks remain characteristic at 18.5°, 22.5°, and 34.6°, corresponding to the diffraction planes 101, 002, and 040, respectively. Compared to the CC sample, all CMF-T samples showed higher crystallinity index.
 | ||
| Fig. 4 XRD patterns of the untreated corn cobs sample (CC) and the hydrogen peroxide alkaline treated samples with different reaction temperatures (CMF-T1, CMF-T2, CMF-T3). | ||
Table 6 indicates that the sample treated at 50 °C has the highest crystallinity index compared to other samples. From the results in Fig. 4 and Table 6, it is evident that as the reaction temperature increases from 50 °C to 70 °C, the crystallinity index tends to decrease. In addition, ANOVA analysis showed that the reaction temperature significantly affected the crystallinity index of cellulose (P < 0.05), as presented in Table S2 of the SI. This is because at higher reaction temperatures, hydrolysis occurred, which may remove partially the crystalline regions, leading to a reduction in the crystallinity of cellulose.
| Sample | Crystallinity index (%) |
|---|---|
| a a, b: Show statistically significant differences among the samples. | |
| CC | 35.42 ± 1.40 |
| CMF-T1 | 39.03 ± 0.98a |
| CMF-T2 | 35.42 ± 1.26b |
| CMF-T3 | 35.18 ± 0.53b |
Since the CMF-T1 sample treated at 50 °C exhibited the highest crystallinity index, it was selected as the optimal reaction temperature for further investigation of subsequent reaction condition parameter.
From the results in Fig. 5 and Table 7, it is evident that after treatment, the crystallinity of the CMF-T samples increased compared to the untreated CC sample. As can be seen from Fig. 5 and Table 7, the sample treated for 4 hours (CMF-T2) presented the highest crystallinity (40.48 ± 0.52%), indicating that this reaction time was effective for removing the amorphous components, allowing cellulose molecules to reorganize into a stable crystalline structure.29 Additionally, the CMF-T3 samples showed a slight decrease in crystallinity compared to CMF-T2 sample, suggesting that too long treatment time may adversely affect the crystallinity of cellulose upon HPK treatment. Furthermore, ANOVA analysis indicated that the reaction time significantly affected the crystallinity index of the extracted cellulose (P < 0.05), as shown in Table S3 of the SI. Therefore, the optimal reaction time for obtaining the best cellulose crystallinity is 4 hours.
 | ||
| Fig. 5 XRD patterns of the untreated corn cobs sample (CC) and the hydrogen peroxide alkaline treated samples with different reaction times (CMF-T1, CMF-T2, CMF-T3). | ||
| Sample | Crystallinity index (%) |
|---|---|
| a a, b: Show statistically significant differences among the samples. | |
| CC | 32.97 ± 0.80 |
| CMF-T1 | 31.95 ± 0.58b |
| CMF-T2 | 39.03 ± 0.98a |
| CMF-T3 | 31.44 ± 1.03b |
Thermal stability of cellulose fibers
Specifically, the dTG curves at Fig. 6b show that the CMF-TL1 sample exhibits the highest decomposition temperature, indicating the raw material-to-chemical ratio of 1/10 (wt/V) is optimal for the treatment. The higher thermal degradation temperatures observed in the CMF-TL samples compared to the CC sample can be attributed to the removal of a portion of hemicellulose and lignin after chemical treatment.
These results show that the ratio of raw material to chemical solution directly influenced on the thermal stability of the extracted cellulose fibers. Specifically, increasing the ratio from 1/10 (wt/V) to 1/20 (wt/V) resulted in a slight decrease in thermal stability of the obtained cellulose fibers.
From the Fig. 7, it is evident that upon increasing the reaction temperature from 50 to 70 °C, the thermal stability of extracted cellulose fibers tended to decrease. Compared to raw corn cob sample, the obtained cellulose fibers had higher thermal stability. This result is ascribed to the effective removal of amorphous and non-cellulosic components upon chemical treatment.31 The cellulose fibers extracted with reaction temperature of 50 °C demonstrated the best thermal stability.
Additionally, as observed from Fig. 8, the extracted cellulose fibers indicated the higher thermal stability compared to the raw CC sample. This confirms the effectiveness of the HPK treatment in improving the thermal stability of cellulose fibers.32
Chemical structure of corn cobs and extracted cellulose fibers
The FTIR spectra of raw corn cobs (CC) and cellulose fibers (CMF) was shown in Fig. 9. As observed, a broad band at 3400 cm−1 corresponding to the stretching vibrations of the –OH groups appeared in the FTIR spectrum of both raw corn cobs and cellulose fibers. This peak is characteristic of hydroxyl-containing compounds such as cellulose.33 Besides that, the absorption peak at 2909 cm−1 attributed to the stretching vibrations of C–H bonds in methyl, methylene, or methine groups is presented in the FTIR spectrum of raw corn cobs and extracted cellulose fibers. The peak at 1043 cm−1 is associated with the C–O–C bonds in the chemical structure of cellulose. As shown in Fig. 9, both CC and CMF samples retain this characteristic peak. These results indicate that after chemical treatment, the chemical structure of cellulose remained intact. | ||
| Fig. 9 FTIR spectra of the untreated corn cobs sample (CC) and the hydrogen peroxide alkaline treated sample (CMF). | ||
However, compared to the FTIR spectrum of raw corn cobs, the FTIR spectrum of extracted cellulose fiber revealed a slight increase in the intensity of the peak at 3400 cm−1. This demonstrates more free hydroxyl groups in the chemical structure of treated sample. Notably, there was a clear reduction in the intensity of the absorption peaks at 1732 cm−1 and 1510 cm−1 in the FTIR spectrum of cellulose fiber compared to that of raw corn cobs. These peaks are corresponded to the stretching vibrations of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O and C
O and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C (aromatic rings) bond, respectively. These bonds present in the chemical structure of hemicellulose and lignin component. This result indicates that the non-cellulosic components such as hemicellulose and lignin were effectively removed upon the HPK treatment.34
C (aromatic rings) bond, respectively. These bonds present in the chemical structure of hemicellulose and lignin component. This result indicates that the non-cellulosic components such as hemicellulose and lignin were effectively removed upon the HPK treatment.34
Surface morphology of corn cobs and extracted cellulose fibers
Fig. 10 presented scanning electron microscopy (SEM) images of the untreated corn cob (CC) and the corn cob after HPK treatment (CMF). As shown in Fig. 10a, the untreated corn cob sample exhibited a rough and heterogeneous surface morphology, characterized by a layered structure in which cellulose fibers were tightly bound to amorphous components, mainly lignin and hemicellulose.32,35 This structural feature was typical of untreated agricultural waste.36 In contrast, the corn cob sample after HPK treatment exhibited pronounced morphological changes (Fig. 10b). The original layered structure was disrupted, and internal cellulose cavities became evident.31 These changes were attributed to the effective removal of hemicellulose and lignin from the surface layers of the biomass, resulting in a smoother and more homogeneous surface.37 This observation was consistent with previous studies reporting surface disruption and fiber exposure in corn cob and corn stalk residues after hydrogen peroxide-alkaline treatment.38 | ||
| Fig. 10 SEM images of (a) raw corn cobs sample (CC) and (b) hydrogen peroxide-alkaline treated sample (CMF). | ||
Chemical structure of cellulose fibers after chemically modifying with RBO
The results in Fig. 11 showed that both FTIR spectra of CMF, and CMF/RBO exhibited characteristic peaks at 3400, 2909, 1370, 1043, and 897 cm−1, corresponding to the structural groups of cellulose.39 This confirmed that upon chemical modification treatment, the chemical structure of cellulose was intact. The broad band observed at 3400 cm−1 indicated a reduction in the number of hydroxyl (–OH) groups, suggesting that esterification occurred with the acyl groups of RBO in the CMF/RBO sample.29 The absorption peaks in the region of 2800–3100 cm−1, corresponding to the alkyl groups (2853 and 2924 cm−1) and acyl groups (3006 cm−1), significantly increased in the RBO-CMF sample. Additionally, compared to the FTIR spectrum of CMF sample, the FTIR spectrum of RBO-CMF sample presented the appearance of a new sharp peak at 1740 cm−1, characteristic of the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bond in the carbonyl group of esters. In addition, the FTIR spectrum of the cellulose sample after modification with RBO and subsequent washing three times with ethanol solution and five times with distilled water (CMF/RBO/W) exhibited absorption bands similar to those of the CMF/RBO sample. However, the absorption intensity of the peak at 1740 cm−1 in the CMF/RBO/W sample was lower than that of the CMF/RBO sample. This behavior was attributed to the washing process, which removed excess RBO that did not participate in the reaction. These results confirmed that the chemical modification treatment of cellulose fibers using RBO took place successfully. The reaction mechanism between cellulose fibers and Rice Bran Oil (RBO) was illustrated in Fig. S2 of the SI.
O bond in the carbonyl group of esters. In addition, the FTIR spectrum of the cellulose sample after modification with RBO and subsequent washing three times with ethanol solution and five times with distilled water (CMF/RBO/W) exhibited absorption bands similar to those of the CMF/RBO sample. However, the absorption intensity of the peak at 1740 cm−1 in the CMF/RBO/W sample was lower than that of the CMF/RBO sample. This behavior was attributed to the washing process, which removed excess RBO that did not participate in the reaction. These results confirmed that the chemical modification treatment of cellulose fibers using RBO took place successfully. The reaction mechanism between cellulose fibers and Rice Bran Oil (RBO) was illustrated in Fig. S2 of the SI.
 | ||
| Fig. 11 FTIR spectra of cellulose fibers (CMF), chemically modified cellulose fibers (CMF/RBO), and chemically modified cellulose fibers after washing (CMF/RBO/W). | ||
Energy dispersive X-ray spectroscopy (EDS)
Surface elemental mapping analysis based on EDS of unmodified cellulose fibers, rice bran oil (RBO)-modified cellulose fibers, and RBO-modified cellulose fibers after water washing is presented in Fig. 12. The analysis results showed that the carbon content of the RBO-modified cellulose fibers (69.8%) increased markedly compared to that of the unmodified cellulose fibers (49.9%), while the oxygen content exhibited a decreasing trend. This change indicated that surface modification with rice bran oil enriched the carbon content on the cellulose fiber surface and simultaneously reduced oxygen-containing functional groups. This phenomenon could be explained by an esterification reaction occurring between the hydroxyl groups of cellulose and the carboxyl groups present in rice bran oil, leading to the formation of ester linkages on the surface of the cellulose fibers. In addition, the RBO-modified cellulose fibers after water washing exhibited a relatively lower carbon content (56.9%) than that of the modified cellulose fibers prior to washing, but still higher than that of the unmodified cellulose fibers. This result could be attributed to repeated water washing, which removed unreacted rice bran oil coated on the surface of the cellulose fibers. These results confirmed that the surface modification of cellulose fibers with rice bran oil was successfully achieved. | ||
| Fig. 12 EDS elemental mapping images of (a) cellulose fibers, (b) cellulose fibers after modification, and (c) modified cellulose fibers after washing. | ||
Physicochemical properties of biocomposites based on PLA and cellulose fibers extracted from corn cobs
 | ||
| Fig. 13 SEM images of the fractured surfaces of the biocomposites after mechanical testing: (a) PLA; (b) PLA/RBO biocomposite; (c) PLA/CMF biocomposite. | ||
Fig. 13c indicates that the micro-sized cellulose fiber agglomerations are presented with respect to PLA/CMF composite. This demonstrates the poor dispersion of unmodified cellulose fibers within the PLA matrix. This can be attributed to the incompatibility between hydrophilic cellulose fibers and hydrophobic PLA matrix. When the modified cellulose fibers were incorporated into the PLA matrix, an improvement in fiber dispersion within the PLA matrix was observed (Fig. 13b). Fiber agglomerations was significantly reduced, indicating better interfacial interaction between cellulose fibers and the PLA matrix.40
These results demonstrate that surface modification of cellulose fibers is essential for improving the compatibility between the cellulose fibers and PLA matrix.41 The findings suggest that chemical modification treatment using RBO effectively reduced the polarity of cellulose fibers, facilitating their dispersion and adhesion within the PLA matrix.25
 | ||
| Fig. 14 XRD patterns of the PLA, PLA/CMF biocomposite with different fiber contents (1,5, and 10 wt%). | ||
 | ||
| Fig. 15 XRD patterns of the PLA and PLA/RBO biocomposite with different fiber contents (1,5, and 10 wt%). | ||
As can be seen from the Fig. 14 and Table 8, when the cellulose fiber content increased from 1 wt% to 10 wt%, the crystallinity index of the PLA/CMF biocomposites significantly decreased. This can be explained by the uneven dispersion of non-modified cellulose fibers in the PLA matrix, and the addition of too much non-modified cellulose fibers into the PLA matrix causing severe fiber agglomerations. These agglomerations act as obstacles, preventing the movement and arrangement of PLA chains. As a result, the crystallinity index of the biocomposites decreased significantly, leading to a reduction in mechanical strength.44
| Sample | Crystallinity index (%) |
|---|---|
| PLA | 23.7 |
| PLA/RBO1 | 26.2 |
| PLA/CMF1 | 24.8 |
| PLA/RBO5 | 25.3 |
| PLA/CMF5 | 16.2 |
| PLA/RBO10 | 14.2 |
| PLA/CMF10 | 14.1 |
Furthermore, as observed from Fig. 15 and Table 8, the crystallinity index of PLA/RBO biocomposites was found to be higher than that of PLA/CMF biocomposites, especially for biocomposites with relatively low fiber contents (1 and 5 wt%). The PLA/RBO1 showed the highest crystallinity index of 26.2%, suggesting that the addition of 1 wt% cellulose fibers treated with RBO enhanced the crystallinity index of the biocomposite.
These results highlight the positive impact of surface modification of cellulose fibers on improving fiber-matrix interactions and promoting crystalline alignment.45 Interestingly, as the fiber content increased to 5 wt% and 10 wt%, the CrI values decreased significantly for both biocomposites. These results indicate that higher fiber content potentially disrupts the crystalline order of the PLA matrix.
These findings suggest that while the incorporation of cellulose fibers, particularly those modified with RBO, can enhance the crystallinity of PLA biocomposites at low fiber content, excessive fiber loading negatively impacted crystallinity due to agglomeration and decreased compatibility.
From Fig. 16 and Table 8, it is shown that the addition of 1 wt% cellulose fiber to the PLA matrix increased the crystallinity index of the biocomposite compared to PLA. Notably, when 1 wt% of modified cellulose fibers were added to the PLA matrix, the crystallinity index of the biocomposite was enhanced more effectively than with 1 wt% of unmodified cellulose fibers. This indicates that the chemical modification process improved the interaction between cellulose fibers and the PLA matrix. These findings emphasize the importance of cellulose fiber content and the modification process in enhancing the crystallinity index of the biocomposites.46
The non-isothermal crystallization behavior of PLA and biocomposites prepared by combining with 1 wt% unmodified cellulose fiber (PLA/CMF1) and 1 wt% RBO-modified cellulose fiber (PLA/RBO1) were investigated by DSC technique. DSC thermograms of PLA and biocomposites during the cooling scan cycle are presented in Fig. 17. As observed from Fig. 17, both PLA and PLA/CMF1 biocomposite did not show any melt crystallization peak upon cooling. Conversely, the melt crystallization peak (at around 98 °C) was observed for the PLA/RBO1 biocomposite material. This indicates that with addition of 1 wt% RBO-modified cellulose fiber helped induce the melt crystallization of PLA upon cooling. It is believed that RBO-modified cellulose fiber had good interfacial adhesion with PLA matrix. The well dispersed cellulose fibers within PLA matrix, acting as heterogeneous nucleating agents inducing the lower energy barrier required for the formation of crystal nuclei. This allows the melt crystallization upon cooling of PLA to take place.48 This result is consistent with SEM result mentioned.
DSC thermograms of PLA and its biocomposites during the second heating scan are shown in Fig. 18. It was found that the cold crystallization temperature (the exothermic peaks at 99–102 °C) of the PLA/CMF1 biocomposite did not indicate any significant difference compared to that of PLA. This demonstrates that the addition of unmodified cellulose fiber into PLA matrix did not cause any change in crystallization rate of PLA. It is due to the incompatibility between relative polar cellulose fibers and nonpolar PLA matrix, leading to poor dispersion of fibers in PLA matrix. As a result, the unmodified cellulose fibers exhibited limited efficacy as nucleating agents and did not significantly enhance the crystallization rate of PLA. However, as observed from Fig. 18, the PLA/RBO1 biocomposite indicated a lower cold crystallization temperature compared to PLA and PLA/CMF1 biocomposite. This indicates that the addition of RBO-modified cellulose fiber induced the improvement in the crystallization rate of PLA. This result can be explained that the RBO-modified cellulose fiber dispersed well in PLA matrix allows the heterogeneous nucleation mechanism to occur. This induces a decrease of the free energy barrier and fastens the crystallization rate of PLA.49 Besides that, it was found that the cold crystallization peak became broader for PLA/RBO1 biocomposite (Fig. 18).
Moreover, it can be seen from Fig. 18 that, with the addition of cellulose fibers, the glass transition temperatures (Tg) (the small endothermic peaks at around 55 °C) of PLA/CMF1 and PLA/RBO1 biocomposites are slightly higher than that of PLA. This shift can be explained by the restriction of chain mobility with the presence of cellulose fibers.50 Additionally, the RBO surface chemical modification of cellulose fiber was found to cause a slight decrease of the glass transition temperature in the case of PLA/RBO1 biocomposite.
Fig. 18 also shows that there are two melting temperatures (Tm1 (145 °C) and Tm2 (155 °C)) for both PLA and biocomposites. The lower melting temperature (Tm1) is attributed to the melting of crystals formed during the cooling process from the melt, whereas the higher melting temperature (Tm2) corresponds to the melting of more perfect crystals generated via cold crystallization upon subsequent heating.51 As observed from Fig. 18, the melting temperatures (Tm1 and Tm2) of PLA/CMF1 and PLA/RBO1 biocomposites did not show any significant difference compared to those of PLA.
 | ||
| Fig. 20 (a) Tensile strength; (b) elongation at break; and (c) Young's Modulus of PLA and PLA/RBO1, PLA/RBO5, PLA/RBO10 biocomposites. | ||
From Fig. 21 and 22, it can be observed that the Young's modulus, tensile strength, and elongation at break of the PLA/CMF composites were all lower than those of neat PLA, indicating that the incorporation of unmodified cellulose fibers adversely affected the mechanical properties of the composites.53 Specifically, when the cellulose fiber content increased from 1 wt% to 10 wt%, the elongation at break decreased markedly from 1.73 ± 0.16% to 0.99 ± 0.04%, reflecting a clear brittleness tendency of the material (Fig. 22b). This behavior can be attributed to the inherently rigid nature of cellulose fibers, combined with poor interfacial interactions, which lead to the formation of stress concentration points and non-uniform fiber dispersion within the PLA matrix, thereby creating structurally weak regions in the composite.54–56 Meanwhile, the results in Fig. 22a showed that the tensile strength of PLA/CMF1, PLA/CMF5, and PLA/CMF10 decreased by approximately 49.1%, 37.4%, and 56.3%, respectively, compared to neat PLA, confirming that the insufficient interaction between cellulose fibers and the PLA matrix hindered effective stress transfer, and thus diminished the reinforcing role of the cellulose fiber.53
 | ||
| Fig. 22 (a) Tensile strength; (b) elongation at break; and (c) Young's modulus of PLA, PLA/CMF1, PLA/CMF5, and PLA/CMF10 biocomposites. | ||
Based on the previous results, we selected the biocomposites with an optimal fiber content of 1 wt% for comparison with neat PLA. As shown in Fig. 23 and 24, the biocomposite containing 1 wt% surface-modified cellulose fibers exhibited higher tensile strength, elongation at break, and Young's modulus than both neat PLA and the composite reinforced with unmodified cellulose fibers. In contrast, the biocomposite containing unmodified cellulose fibers showed inferior mechanical properties, even lower than those of neat PLA. This pronounced difference demonstrated that cellulose surface modification played a decisive role in improving fiber–matrix compatibility, thereby enhancing stress transfer efficiency and suppressing premature failure. These findings clearly confirmed that the cellulose modification process was essential for achieving effective mechanical reinforcement in PLA-based biocomposites.54,57 Table S4 of the SI summarizes the mechanical properties of biocomposite samples at different cellulose contents.
Flow curves of PLA and PLA/RBO1 biocomposites with cellulose fiber content of 1 wt% were shown in Fig. 25. It revealed that viscosity at all shear rate ranges of PLA/RBO1 biocomposite was higher than that of PLA. This is attributed to the lower molecular chain mobility of PLA due to the presence of cellulose fibers. Cellulose fibers served as physical crosslinking points. As a result, the higher viscosity of PLA/RBO1 biocomposites was obtained. This result is in good agreement with MFI result as presented in Table 9. PLA/RBO1 biocomposites showed lower MFI than PLA, indicating the higher viscosity. These results indicate that after chemical modification, the interfacial adhesion between cellulose fibers and PLA matrix was improved, which led to lowering the PLA chain flexibility and higher shear viscosity of PLA/RBO1 biocomposites compared to that of PLA at all measured shear rates. In addition, the rheological properties of the biocomposites reinforced with different cellulose contents are presented in Fig. S3 of the SI.
| Sample | MFI (g/10 m) |
|---|---|
| PLA | 2.72 ± 0.05 |
| PLA/RBO1 biocomposite | 2.58 ± 0.03 |
Evaluation of the applicability of PLA/CMF biocomposite as 3D printing material using fused deposition modelling technology
The PLA/RBO1 biocomposite was used to fabricate filaments with a diameter of 1.72 ± 0.06 mm and was subsequently employed as a 3D printing material. The parameters and procedural steps of the 3D printing process were presented in Table S5 and Fig. 26, respectively.Dimensional accuracy
The dimensional accuracy of the 3D-printed specimens was evaluated based on the thickness, width at the gauge length region, and overall length of the samples, in accordance with ASTM D638 Type IV. The design drawing following the ASTM D638 Type IV standard was presented in Fig. S4. In fused deposition modeling (FDM) 3D printing, the actual dimensions of printed parts often deviated from the original design dimensions due to thermal shrinkage and process-related factors. According to previous studies, the dimensional deviation of FDM-printed samples typically ranged from 0.1 to 1.4%, corresponding to the shrinkage behavior of thermoplastic polymers during printing and cooling.26 The average dimensions and dimensional deviations of the 3D-printed samples were summarized in Table 10. The results showed that both material systems exhibited dimensional deviations below 3%. For the width and thickness dimensions, the neat PLA samples exhibited larger dimensional deviations than the PLA/RBO1 samples. This behavior was attributed to the reinforcement effect of cellulose microfibers modified with rice bran oil, which reduced the mobility of PLA chains, thereby limiting filament spreading after extrusion and stabilizing the molten flow during printing.62 In addition, the presence of cellulose acted as physical crosslinking points within the polymer matrix, increasing the viscosity and stiffness of the PLA/RBO1 system compared with neat PLA. This effect contributed to reduced thermal shrinkage and improved dimensional accuracy of the printed samples.63 For the specimen length, the results indicated that neat PLA exhibited a smaller length deviation than PLA/RBO1. This behavior was attributed to the lower melt viscosity of neat PLA, which led to stronger polymer chain orientation along the printing direction. In contrast, the presence of CMF restricted chain orientation, resulting in more uniform shrinkage in different directions and improved overall geometric stability of the composite material. However, dimensional accuracy in FDM printing depended on multiple processing parameters; therefore, a more in-depth discussion of this issue is required.| Sample | Width (mm) | Deviation (%) | Length (mm) | Deviation (%) | Thickness (mm) | Deviation (%) |
|---|---|---|---|---|---|---|
| PLA | 5.90 ± 0.01 | 1.67 | 114.86 ± 0.13 | 0.12 | 3.11 ± 0.06 | 2.88 |
| PLA/RBO1 biocomposite | 5.94 ± 0.11 | 0.97 | 114.59 ± 0.11 | 0.36 | 3.19 ± 0.02 | 0.31 |
In addition, a preliminary evaluation of the mechanical properties of the 3D-printed samples was presented in Fig. S5 of the SI.
Limitations and future development
Furthermore, the interfacial interactions between rice bran oil (RBO)-modified cellulose microfibers and the PLA matrix have only been qualitatively evaluated based on SEM, FTIR, and XRD analyses, while quantitative assessments of interfacial adhesion have not yet been conducted. In addition, a life cycle assessment (LCA) of the fabricated biocomposite has not been performed, although this analysis is essential for evaluating its practical applicability and sustainability. Finally, although the feasibility of the biocomposite as a 3D printing material for fused deposition modeling (FDM) technology has been demonstrated, key printing parameters—such as dimensional accuracy, interlayer adhesion, and the mechanical properties of the final printed parts-have not yet been systematically investigated.
Moreover, systematic studies on surface modification strategies for cellulose microfibers are required to reduce hydrophilicity, enhance dispersion, and improve interfacial compatibility with the PLA matrix. Beyond rice bran oil (RBO), other bio-based modification agents or compatibilizers may be examined to enhance mechanical performance without compromising processability or the quality of printed parts.
Subsequent investigations should also focus on elucidating the relationships among filament extrusion parameters, FDM printing conditions, and the resulting microstructure and mechanical anisotropy of printed components. The integration of rheological modeling with real-time process monitoring may provide deeper insights into melt-flow behavior and interlayer bonding mechanisms, thereby enabling the development of optimized printing protocols for bio-composite materials.
In addition to tensile properties, mechanical characterization should be extended to include flexural strength, impact resistance, and environmental aging behavior under exposure to humidity, heat, and UV radiation in order to assess material suitability for real-world applications. Concurrently, studies on controlled biodegradation under different environmental conditions would yield critical information regarding the end-of-life phase of the printed products.
Finally, life cycle assessment (LCA), recyclability, and integration into circular economy models should be emphasized to clarify the feasibility of reprocessing and reusing printed PLA/cellulose components. Collectively, these research directions will facilitate the transition of bio-based composite materials from laboratory-scale development to environmentally sustainable commercial 3D-printing applications.
Conclusions
The findings of this study have demonstrated the potential of cellulose fibers extracted from corn cobs – an agricultural waste source for fabricating sustainable biocomposite materials using biodegradable poly(lactic acid) as the matrix. The extraction process employed optimal conditions and environmentally friendly extraction methods, using non-toxic chemicals. The use of H2O2 and NaOH was highly effective in removing impurities while preserving the structure and characteristic properties of cellulose fibers. Besides that, H2O2 also functioned as a bleaching agent, effectively removed most of the hemicellulose and lignin components from the fiber structure. Additionally, the surface chemical modification of cellulose fibers using rice bran oil (RBO) significantly improved the compatibility between the cellulose fibers and the PLA matrix. This chemical modification reduced the polarity of the cellulose fibers without harming the environment due to using an environmentally friendly organic acid RBO. The results of FTIR, XRD, SEM, TGA, DSC analyses in combination with rheological and mechanical characterizations provided critical insights into the structure and physicochemical properties of the fabricated biocomposites based on cellulose fibers and poly(lactic acid), particularly its enhanced crystallinity and mechanical performance.With a cellulose fiber content of 1 wt%, the biocomposites obtained optimal mechanical properties with improved tensile strength, elongation at break, and elastic modulus. The 3D printing process by FDM method using the filament fabricated from PLA and cellulose fibers extracted from corn cobs agricultural waste demonstrated its feasibility for application as 3D printing material. The successful development of biocomposites from renewable resources not only paves the way for the production of biodegradable products but also addresses environmental challenges, aligning with the goal of sustainable development of Vietnam in the future.
Author contributions
Nguyen Chi Thanh: conceptualization, data curation, formal analysis, investigation, methodology, supervision, validation, visualization, writing – original draft, writing – review& editing. Pham Duc Thinh: data curation, formal analysis, investigation, visualization, writing – original draft, writing – review & editing. Bui Phuong Dong: formal analysis, writing – review & editing. Tran Hai Cat: formal analysis, writing – review & editing. Nguyen Thanh Huy: formal analysis, writing – review & editing.Conflicts of interest
There are no conflicts to declare.Data availability
The supporting data has been provided as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5ra09705c.Acknowledgements
The authors would like to thank Ho Chi Minh City University of Technology and Engineering for the financial support. This study belongs to the project grant No: T2026-35.References
- H. A. Silvério, W. P. F. Neto, N. O. Dantas and D. Pasquini, Ind. Crop. Prod., 2013, 44, 427–436 Search PubMed.
- W. K. Ng, W. S. Chow and H. Ismail, Polym. Renew. Resour., 2019, 10, 63–76 Search PubMed.
- U. N. Ngoc and H. Schnitzer, Waste Manag., 2009, 29, 1982–1995 Search PubMed.
- T. M. Vu, D. P. Doan, H. T. Van, T. V. Nguyen, S. Vigneswaran and H. H. Ngo, Sci. Total Environ., 2017, 579, 612–619 Search PubMed.
- Y. Zou, J. Fu, Z. Chen and L. Ren, Agriculture, 2021, 11, 556 CrossRef CAS.
- R. S. Abolore, S. Jaiswal and A. K. Jaiswal, Carbohydr. Polym. Technol. Appl., 2024, 7, 100396 CAS.
- L. Chopra, Mater. Today: Proc., 2022, 48, 1265–1270 CAS.
- M. P. Menon, R. Selvakumar and S. Ramakrishna, RSC Adv., 2017, 7, 42750–42773 RSC.
- A. N. Frone, D. M. Panaitescu and D. Donescu, Sci. Bull. - “Politeh.” Univ. Bucharest, Ser. B, 2011, 73, 133–152 Search PubMed.
- J. Wang, R. Zhang, C. Quan, X. Shao, N. Hu, X. Yao and C. Dong, Cellulose, 2022, 29, 7125–7138 Search PubMed.
- H. Chen, Y. Yuan and Q. Li, Starch-Stärke, 2020, 72, 1900209 Search PubMed.
- A. Fouly, A. K. Assaifan, I. A. Alnaser, O. A. Hussein and H. S. Abdo, Polymers, 2022, 14, 5299 Search PubMed.
- S. Farah, D. G. Anderson and R. Langer, Adv. Drug Deliv. Rev., 2016, 107, 367–392 Search PubMed.
- H. Ramezani Dana and F. Ebrahimi, Polym. Eng. Sci., 2023, 63, 22–43 Search PubMed.
- C. A. Murphy and M. N. Collins, Polym. Compos., 2018, 39, 1311–1320 Search PubMed.
- X. Pang, X. Zhuang, Z. Tang and X. Chen, Biotechnol. J., 2010, 5, 1125–1136 Search PubMed.
- N. G. Khouri, J. O. Bahú, C. Blanco-Llamero, P. Severino, V. O. C. Concha and E. B. Souto, J. Mol. Struct., 2024, 138243 CrossRef CAS.
- V. G. Gorade, A. Kotwal, B. U. Chaudhary and R. D. Kale, J. Polym. Res., 2019, 26, 1–12 Search PubMed.
- X. Bi and R. Huang, Mater. Des., 2022, 222, 111065 Search PubMed.
- A. Ji, S. Zhang, S. Bhagia, C. G. Yoo and A. J. Ragauskas, RSC Adv., 2020, 10, 21698–21723 RSC.
- B. B. Mansingh, J. S. Binoj, Z. Q. Tan, W. W. L. Eugene, T. Amornsakchai, S. A. Hassan and K. L. Goh, Polym. Compos., 2022, 43, 6051–6061 CrossRef CAS.
- V. H. M. D. Almeida, R. M. D. Jesus, G. M. Santana, S. Khan, E. F. M. S. Silva, I. S. D. Cruz, I. D. S. Santos and P. N. M. Dos Anjos, Polymers, 2024, 16, 1757 Search PubMed.
- S. Raja, R. M. Ali, S. Karthikeyan, R. Surakasi, R. Anand, N. Devarasu and T. Sathish, Chem. Eng., 2024, 7, 10–59429 Search PubMed.
- L. Segal, J. Creely, Jr., A. E. Martin Jr and C. M. Conrad, Textil. Res. J., 1959, 29, 786–794 CrossRef CAS.
- H. Li, Z. Cao, D. Wu, G. Tao, W. Zhong, H. Zhu, P. Qiu and C. Liu, Plast., Rubber Compos., 2016, 45, 181–187 CrossRef CAS.
- W. Prasong, A. Ishigami, S. Thumsorn, T. Kurose and H. Ito, Polymers, 2021, 13, 740 CrossRef CAS PubMed.
- A. El Oudiani, Y. Chaabouni, S. Msahli and F. Sakli, Carbohydr. Polym., 2011, 86, 1221–1229 CrossRef CAS.
- Z. Wang, A. G. McDonald, R. J. M. Westerhof, S. R. A. Kersten, C. M. Cuba-Torres, S. Ha, B. Pecha and M. Garcia-Perez, J. Anal. Appl. Pyrolysis, 2013, 100, 56–66 CrossRef CAS.
- R. D. Kale, P. S. Bansal and V. G. Gorade, J. Polym. Environ., 2018, 26, 355–364 CrossRef CAS.
- G. Maschio, C. Koufopanos and A. Lucchesi, Bioresour. Technol., 1992, 42, 219–231 CrossRef CAS.
- H. Gao and T. Qiang, Materials, 2017, 10, 624 Search PubMed.
- Y. Su, R. Du, H. Guo, M. Cao, Q. Wu, R. Su, W. Qi and Z. He, Food Bioprod. Process., 2015, 94, 322–330 Search PubMed.
- Y. H. P. Zhang, J. Ind. Microbiol. Biotechnol., 2008, 35, 367–375 CrossRef CAS PubMed.
- X. F. Sun, F. Xu, R. C. Sun, P. Fowler and M. S. Baird, Carbohydr. Res., 2005, 340, 97–106 CrossRef CAS.
- H. Du, K. Liu, T. Xu, C. Xu, M. Lin, Z. Fang, S.-W. Kim, J.-Y. Seo, J. Chen, H. Ma, B. S. Hsiao, L. W. DeVetter, Z. Piao, C. Si, C. Chen, Q. Yang, S.-Y. Lee, Y. Yao and X. Pan, Chem. Rev., 2025, 125, 11666–11814 CrossRef CAS.
- K. Kaur, R. Kaur and H. Kaur, Appl. Surf. Sci. Adv., 2024, 19, 100547 Search PubMed.
- Y. Zhan, X. Liu, C. Huang, X. Zhou, Y. Lyu, Y. Lin, C. Huang, W. Ma, Z. Xie, G. Fang and A. J. Ragauskas, Ind. Crops Prod., 2024, 214, 118533 CrossRef CAS.
- M. J. Selig, T. B. Vinzant, M. E. Himmel and S. R. Decker, Appl. Biochem. Biotechnol., 2009, 155, 94–103 CrossRef PubMed.
- K. O. Reddy, J. Zhang, J. Zhang and A. V. Rajulu, Carbohydr. Polym., 2014, 114, 537–545 CrossRef CAS.
- S. Qian, H. Zhang, W. Yao and K. Sheng, Composites, Part B, 2018, 133, 203–209 CrossRef CAS.
- M. R. Manshor, H. Anuar, M. N. N. Aimi, M. I. A. Fitrie, W. B. W. Nazri, S. M. Sapuan, Y. A. El-Shekeil and M. U. Wahit, Mater. Des., 2014, 59, 279–286 CrossRef CAS.
- T. Lu, M. Jiang, X. Xu, S. Zhang, D. Hui, J. Gou and Z. Zhou, J. Appl. Polym. Sci., 2014, 131, 41077 CrossRef.
- D. Wu, L. Wu, L. Wu, B. I. N. Xu, Y. Zhang and M. Zhang, J. Polym. Sci., Part B: Polym. Phys., 2007, 45, 1100–1113 CrossRef CAS.
- A. Pei, Q. Zhou and L. A. Berglund, Compos. Sci. Technol., 2010, 70, 815–821 CrossRef CAS.
- M. Mohammed, R. Rahman, A. M. Mohammed, T. Adam, B. O. Betar, A. F. Osman and O. S. Dahham, Polym. Test., 2022, 115, 107707 CrossRef CAS.
- T. Lu, S. Liu, M. Jiang, X. Xu, Y. Wang, Z. Wang, J. Gou, D. Hui and Z. Zhou, Composites, Part B, 2014, 62, 191–197 CrossRef CAS.
- S. S. Shazleen, L. Y. Foong Ng, N. A. Ibrahim, M. A. Hassan and H. Ariffin, Polymers, 2021, 13, 3226 CrossRef CAS.
- S. S. Shazleen, T. A. T. Yasim-Anuar, N. A. Ibrahim, M. A. Hassan and H. Ariffin, Polymers, 2021, 13, 389 Search PubMed.
- K. Shi, G. Liu, H. Sun, B. Yang and Y. Weng, Polymers, 2022, 14, 4305 Search PubMed.
- V. de Oliveira, L. N. Horiuchi, A. P. Goncalves, M. De Andrade and R. Polkowski, Nanocomposites Made with Poly (Lactic Acid)/Cellulose Nanofibers for Automotive Applications: The Impact of Annealing on 3D Printed Parts, Report 0148-7191, SAE Technical Paper, 2025 Search PubMed.
- Y. Hu, H. Shen, H. Ni, Y. Liu, L. Zhang and G. Ju, J. Appl. Polym. Sci., 2025, e56796 CrossRef CAS.
- T. Lu, M. Jiang, Z. Jiang, D. Hui, Z. Wang and Z. Zhou, Composites, Part B, 2013, 51, 28–34 CrossRef CAS.
- L. Suryanegara, A. N. Nakagaito and H. Yano, Cellulose, 2010, 17, 771–778 CrossRef CAS.
- A. N. Frone, S. Berlioz, J. F. Chailan, D. M. Panaitescu and D. Donescu, Polym. Compos., 2011, 32, 976–985 CrossRef CAS.
- M. E. Lozano Fernandez and N. Miskolczi, Polymers, 2022, 14, 1887 CrossRef CAS.
- N. Graupner, J. Mater. Sci., 2008, 43, 5222–5229 CrossRef CAS.
- A. K. Trivedi, M. K. Gupta and H. Singh, Adv. Ind. Eng. Polym. Res., 2023, 6, 382–395 CAS.
- K. Badura and P. Bazan, Int. J. Adv. Res. Sci. Commun. Technol., 2025, 19, 184–201 Search PubMed.
- N. J. Hadi, T. Rydzkowski, Z. S. Ali and Q. A. Al-Jarwany, Coatings, 2025, 15, 397 CrossRef CAS.
- M. Lecoublet, M. Ragoubi, N. Leblanc and A. Koubaa, in Sustainable Nanocomposites with Green Biomaterials, Springer, 2025, pp. 445–477 Search PubMed.
- S. Antony Jose, N. Cowan, M. Davidson, G. Godina, I. Smith, J. Xin and P. L. Menezes, Nanomaterials, 2025, 15, 356 CrossRef CAS.
- M. Lecoublet, M. Ragoubi, N. Leblanc and A. Koubaa, Ind. Crops Prod., 2024, 221, 119332 CrossRef CAS.
- F. Burkhardt, V. D. Schmidt, C. Wesemann, C. G. Schirmeister, S. Rothlauf, S. Pieralli, L. S. Brandenburg, L. Kleinvogel, K. Vach and B. C. Spies, Sci. Rep., 2022, 12, 20341 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |














