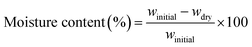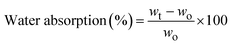Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Eco-sustainable chitosan–PVA biocomposites reinforced with recycled alum: preparation, characterization, and antimicrobial assessment
Mahmoud A. Abdelkawy *a,
Abdelbaset Shokera,
Badr Isamil Badrb and
Yehia A.-G. Mahmoudb
*a,
Abdelbaset Shokera,
Badr Isamil Badrb and
Yehia A.-G. Mahmoudb
aChemistry Department, Faculty of Science, Tanta University, Tanta 31527, Egypt. E-mail: mahmoud.abdelkawy@science.tanta.edu.eg
bBotany and Microbiology Department, Faculty of Science, Tanta University, Tanta 31527, Egypt
First published on 26th January 2026
Abstract
This study presents a sustainable approach to valorize aluminum can waste by converting it into recycled potassium alum (Recy. Al), used as a reinforcing and bioactive filler in chitosan/poly(vinyl alcohol) (CS/PVA) biocomposites. Recy. Al was synthesized through a green precipitation process and incorporated (5–50 wt%) into CS/PVA matrices via solution casting. FTIR and XRD confirmed successful alum integration without altering the matrix's semi-amorphous structure. Thermal analysis revealed enhanced stability, with 5% and 10% weight-loss temperatures (Td5, Td10) increasing from 188 °C and 239 °C for pristine CS/PVA to 223 °C and 240 °C for the 50 wt% composite, along with a higher char yield (38%). Mechanical strength and stiffness improved, reaching 21.1 ± 3.3 MPa and 501.3 ± 15.9 MPa at 30 wt% loading. SEM/EDS confirmed uniform alum dispersion and strong polymer-filler adhesion. Biologically, the CS/PVA–20 wt% composite showed potent antibacterial activity (MIC = 7.8 ± 1.0 µg mL−1; MCD ≈ 15 µg mL−1) against E. coli and S. aureus, while the CS/PVA–10 wt% sample exhibited the highest antifungal effect against S. cerevisiae and A. niger. Cytotoxicity tests confirmed good biocompatibility up to 20 wt% Recy. Al. These findings demonstrate that biocomposite films are thermally stable, antimicrobial, and eco-friendly materials for sustainable applications.
1. Introduction
The rapidly growing global population, combined with the fast pace of industrialization, has caused a significant increase in waste production across domestic, agricultural, and industrial sectors.1 These waste streams pose a significant threat to natural resources, public health, and environmental sustainability. Approximately 2 billion tonnes of municipal solid waste are generated annually, and by 2030, this is projected to grow to 2.59 billion tonnes.2 This burden is compounded by agricultural residues, such as crop straw and husks, and agro-industrial byproducts, like bagasse and fruit peels, especially in developing countries where open burning remains a common but environmentally harmful disposal method.3,4 An extra 20 million tonnes are added each year from industrial waste, which includes both hazardous and non-hazardous materials that leach into land and water ecosystems. This waste encompasses byproducts from mining, manufacturing, and chemical processing.5,6Interest in converting waste into value-added materials has intensified in response to the global shift toward sustainability and waste valorization. For instance, lime sludge has been utilized in polymer composites, fly ash has been repurposed for low-carbon construction materials, and plastic waste has been chemically recycled through depolymerization.7–9 Among industrial byproducts, alum sludge generated during the coagulation–flocculation process of water treatment using aluminum-based coagulants has attracted increasing attention. However, its disposal by landfilling poses significant environmental and economic challenges due to the presence of organic matter, suspended solids, and aluminum hydroxide.10 Although alum sludge has been explored for applications in adsorbents,11 construction materials,12 and agriculture,13,14 its incorporation into polymer matrix composites remains largely unexplored.
Recent studies and reviews on aluminum waste valorization have mainly focused on recycling aluminum scrap into metal matrix composites (MMCs), where recycled aluminum acts as the matrix and ceramic fillers are used to improve mechanical and thermal performance for high-end structural applications.15–17 These approaches generally require high-temperature processing, strict impurity control, and energy-intensive routes, which limit their environmental sustainability and applicability in polymer-based systems. Moreover, most existing studies emphasize aluminum recovery and purification rather than its direct functional utilization as a reinforcing phase in polymer or biopolymer composites. In contrast, the incorporation of recycled aluminum into biodegradable polymer matrices remains largely unexplored, particularly for applications requiring low processing temperatures, antimicrobial functionality, and environmental compatibility. Addressing this gap, the present work proposes a sustainable upcycling strategy that directly integrates aluminum can waste into a chitosan/PVA biopolymer matrix, enabling simultaneous enhancement of mechanical performance and antimicrobial activity through a low-cost and environmentally benign route.
The development of “green polymers” made from renewable resources has accelerated in both academia and industry due to the need for waste valorization.18 Biodegradability, environmental friendliness, and lower energy requirements in production are among the obvious benefits of biopolymers over petroleum-based polymers.19 However, their broader adoption is often limited by intrinsic issues in their mechanical strength, thermal stability, and processability.20–22 Hybrid systems that combine natural and synthetic polymers are actively being researched as solutions to these challenges.
After cellulose, chitosan (CS), a linear polysaccharide derived from the partial deacetylation of chitin, is the second most common natural biopolymer. Its abundant reactive amino and hydroxyl groups provide beneficial qualities such as antimicrobial activity, biodegradability, biocompatibility, and non-toxicity.23,24 Due to these qualities, CS is a versatile option for applications in biomedical fields such as tissue engineering, drug delivery, wound healing, as well as food packaging. Polyvinyl alcohol (PVA), a synthetic water-soluble polymer with a hydroxyl-rich backbone that promotes extensive hydrogen bonding, is well known for its biodegradability, high crystallinity, and strong film-forming ability.25 Its mechanical strength, chemical stability, and compatibility with natural polymers make it suitable for packaging and biomedical uses. When CS and PVA are blended, their combined properties offer a sustainable way to produce functional composite films. However, issues like reduced flexibility and poor thermal stability can occur, especially with higher CS content. The stiff structure of CS limits polymer chain mobility, and studies have shown that increasing chitosan loading tends to lower the elongation at break and thermal degradation temperature of CS–PVA blends.26–28 Despite these drawbacks, CS/PVA composites remain promising for eco-friendly material development. To improve mechanical and thermal properties while maintaining biodegradability, approaches such as adding inorganic fillers or plasticizers have been explored.
The global push toward sustainable materials and waste valorization has spurred increased research into repurposing waste into functional materials. There is an urgent need for antimicrobial agents, antibiotics, and biocides (such as antiseptics, disinfectants, and preservatives) to control human pathogens. Many efforts are underway to develop new antibacterial, antifungal, antiviral, and anti-cancer drugs. It is estimated that around 700![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 people die each year due to infections with resistant bacteria, highlighting the urgent global need for new antimicrobial agents for current and future generations.29 The search for novel antimicrobial compounds from various sources is critical but challenging. Pharmaceutical companies must invest significant time and money into research, involving multiple rigorous phases of synthesis, discovery, and testing of new agents that must pass demanding criteria.30 Therefore, this study aims to develop biocomposites capable of combating microbial contamination, which is a major concern across sectors such as medical devices, healthcare products, water purification, hospitals, dental offices, food packaging, and storage. The study also evaluates both the cidal and static effects of the synthesized agents on test organisms. Additionally, our research focuses on creating a sustainable, high-performance biocomposite by incorporating recycled aluminum particles from cans into a chitosan/PVA matrix. This innovative approach addresses both aluminum waste management and the mechanical limitations of biopolymers while providing an eco-friendly pathway to value-added materials suitable for sustainable packaging, structural films, and environmental coatings. By harnessing the reinforcing properties of recycled aluminum particles and the synergistic interaction between CS and PVA, we aim to develop a biocomposite that is mechanically durable, thermally stable, and environmentally friendly.
000 people die each year due to infections with resistant bacteria, highlighting the urgent global need for new antimicrobial agents for current and future generations.29 The search for novel antimicrobial compounds from various sources is critical but challenging. Pharmaceutical companies must invest significant time and money into research, involving multiple rigorous phases of synthesis, discovery, and testing of new agents that must pass demanding criteria.30 Therefore, this study aims to develop biocomposites capable of combating microbial contamination, which is a major concern across sectors such as medical devices, healthcare products, water purification, hospitals, dental offices, food packaging, and storage. The study also evaluates both the cidal and static effects of the synthesized agents on test organisms. Additionally, our research focuses on creating a sustainable, high-performance biocomposite by incorporating recycled aluminum particles from cans into a chitosan/PVA matrix. This innovative approach addresses both aluminum waste management and the mechanical limitations of biopolymers while providing an eco-friendly pathway to value-added materials suitable for sustainable packaging, structural films, and environmental coatings. By harnessing the reinforcing properties of recycled aluminum particles and the synergistic interaction between CS and PVA, we aim to develop a biocomposite that is mechanically durable, thermally stable, and environmentally friendly.
2. Materials and methods
2.1. Materials
Poly(vinyl alcohol) (PVA) with a purity of (>98.0%), and Chitosan (CS) with a purity of (99.0%) (Mw = 100–300 kDa), with a 90% degree of deacetylation, were provided by Sigma-Aldrich. Glacial acetic acid (99%) was used without further purification from Adwic Co., Egypt.2.2. Experimental methods
2.3. Biological application
2.4. Characterization
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000× at room temperature. Each sample was put on a holder before being coated with a thin platinum layer to avoid the charging effect.
000× at room temperature. Each sample was put on a holder before being coated with a thin platinum layer to avoid the charging effect.3. Results and discussion
3.1. Characterization of recycled potassium alum (Recy. Al)
Potassium alum (KAl(SO4)2·12H2O) is a widely accessible, inexpensive, water-soluble, and environmentally safe compound that is frequently used in laboratory and industrial settings without the need for special handling.38–40 In this study, the successful synthesis of potassium alum from recycled aluminum can waste using a green precipitation method produced a recovery efficiency of approximately 80%, which is consistent with earlier reports on alum regeneration from waste sources. FTIR and XRD analyses of the structural characteristics of the recovered alum verified that the recycled material had a high level of purity and crystallinity, comparable to that of commercial potassium alum.The FTIR spectrum of the recycled alum exhibited several characteristic peaks (Fig. 2), confirming the successful formation of hydrated sulfate salt. Strong absorption bands at approximately 1191 cm−1 and 1095 cm−1 are attributed to the asymmetric stretching of the S![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O groups. Additional peaks at 919 cm−1 and 698 cm−1 correspond to the stretching vibrations of the S–O and Al–O bonds, respectively. In the low-frequency region (750–400 cm−1), multiple bands appeared due to the Al–O lattice vibrations. Moreover, distinctive sulfate ion (SO42−) bands were observed in the ranges of 449–480 cm−1, 595–609 cm−1, 657–698 cm−1, 1095–1110 cm−1, and 1291–1207 cm−1, which are consistent with the symmetric and asymmetric bending and stretching modes of the sulfate tetrahedron.41,42
O groups. Additional peaks at 919 cm−1 and 698 cm−1 correspond to the stretching vibrations of the S–O and Al–O bonds, respectively. In the low-frequency region (750–400 cm−1), multiple bands appeared due to the Al–O lattice vibrations. Moreover, distinctive sulfate ion (SO42−) bands were observed in the ranges of 449–480 cm−1, 595–609 cm−1, 657–698 cm−1, 1095–1110 cm−1, and 1291–1207 cm−1, which are consistent with the symmetric and asymmetric bending and stretching modes of the sulfate tetrahedron.41,42
X-ray diffraction (XRD) analysis further confirmed the crystalline nature of the recycled alum, exhibiting a diffraction pattern closely aligned with that of standard potassium alum (KAl(SO4)2·12H2O). The recycled alum displayed distinct and well-resolved diffraction peaks at 2θ values of 12.6°, 16.2°, 17.8°, 20.5°, 21.9°, 26.4°, 27.3°, 29.2°, 30.2°, 31.1°, 32.0°, 33.8°, 34.6°, 36.1°, 39.9°, and 40.6°, as shown in Fig. 4. The high crystallinity and lack of detectable impurities in the recycled alum were confirmed by the distinct and sharp diffraction peaks. This confirms the effectiveness and dependability of the upcycling process by showing that its structural characteristics closely resemble those of potassium alum made commercially.41,43
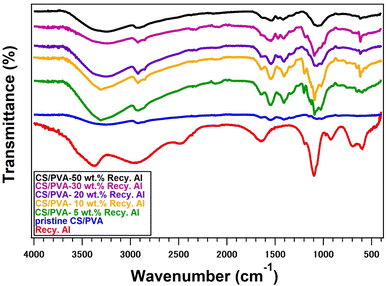
3.2. FTIR analysis of bio-composites
Fig. 2 presents the FTIR spectra of the pristine CS/PVA blend and CS/PVA biocomposites reinforced with different loadings (5, 10, 20, 30, and 50 wt%) of recycled alum. The pristine CS/PVA film exhibited a broad absorption band in the region of 3200–3400 cm−1, arising from overlapping O–H and N–H stretching vibrations. This broadening indicates the formation of strong intermolecular hydrogen bonding between the hydroxyl groups of PVA and the hydroxyl and amino groups of chitosan, confirming good miscibility within the blended polymer matrix (Fig. 3).43 The absorption band near 2917 cm−1 corresponds to C–H stretching vibrations, while the band at approximately 1558 cm−1 is attributed to N–H bending vibrations. Additional bands in the region of 1000–1100 cm−1 are associated with the C–O stretching vibrations of the polymer backbone.44–46The incorporation of recycled alum into the CS/PVA matrix resulted in noticeable changes in the FTIR spectra as a function of the filler content. The broad O–H/N–H stretching band at approximately 3357 cm−1 becomes slightly reduced in intensity and broadens with increasing alum loading, indicating enhanced hydrogen bonding interactions between the polymer functional groups and the alum surface. Moreover, the appearance and gradual intensification of absorption bands in the range of 1081–1190 cm−1 can be attributed to the overlapping of polymer C–O vibrations with the asymmetric stretching modes of sulfate (S![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) groups originating from the recycled alum. In the CS/PVA-Recy. Al composites, this band is shifted to 1540 cm−1. The characteristic band at 1200–1110 cm−1 indicates that the protonated amino groups (–NH3+) of chitosan and the sulfate (–OSO3−) moieties of the recy. Al are forming strong intermolecular interactions.
O) groups originating from the recycled alum. In the CS/PVA-Recy. Al composites, this band is shifted to 1540 cm−1. The characteristic band at 1200–1110 cm−1 indicates that the protonated amino groups (–NH3+) of chitosan and the sulfate (–OSO3−) moieties of the recy. Al are forming strong intermolecular interactions.
Additional bands appearing at approximately 919 cm−1 and 698 cm−1 are assigned to S–O and Al–O stretching vibrations, respectively, confirming the successful incorporation of alum particles into the CS/PVA matrix. In the low-frequency region (750–400 cm−1), multiple bands are associated with Al–O lattice vibrations and sulfate ion bending modes (Fig. 3). These spectral features became more pronounced as the recycled alum content increased from 5 to 50 wt% further evidencing effective filler incorporation.
Importantly, no new absorption bands corresponding to covalent bond formation were detected, indicating that the interaction between the CS/PVA matrix and recycled alum was governed primarily by non-covalent interactions. The observed spectral shifts and intensity variations confirm the presence of hydrogen bonding and electrostatic interactions between the hydroxyl and amino groups of the polymers and sulfate/aluminum species of recycled alum. These interactions facilitated good interfacial compatibility and contributed to the reinforcing effect of the recycled alum within the CS/PVA biocomposites (Fig. 3).
3.3. X-ray diffraction (XRD) analysis of bio-composites
Fig. 4 shows the X-ray diffraction patterns of pristine chitosan–polyvinyl alcohol (CS/PVA) and the corresponding biocomposites containing recycled potassium alum at different loadings (5–50 wt%). In line with earlier reports on conventionally synthesized potassium alum, the recycled alum showed a distinct crystalline profile with multiple sharp reflections, indicating its high crystalline and structural purity. The pristine CS/PVA film, on the other hand, shows a wide, diffuse halo centered at 2θ ≈ 25°, which is consistent with the literature describing hydrogen-bonded CS/PVA networks and a feature of its primarily amorphous nature. The broad amorphous halo still dominates the polymer matrix diffraction pattern when recycled alum is added at low to moderate loadings (5–20 wt%). This suggests that alum is evenly distributed and does not create new crystalline phases or long-range ordering in the polymer backbone. Subtle but significant structural changes became apparent at higher loadings (≥30 wt%). At approximately 2θ ≈ 19°, 24°, 29.7°, 30.9°, and 31.6°, distinct crystalline reflections appeared along with a progressive narrowing of the main diffraction halo. These peaks represent the intrinsic crystalline domains of alum integrated into the matrix. Their appearance suggests localized ordering in the composite, which results from the physical confinement of the CS/PVA matrix remained predominantly amorphous due to restricted polymer chain mobility around alum aggregates.47–49 Although they encourage partial segmental alignment, these interfacial interactions, mostly hydrogen bonding and ionic associations, which remains predominantly amorphous.3.4. Thermal analysis of bio-composites
The thermal properties of pristine CS/PVA and its biocomposites reinforced with varying contents (5–50 wt%) of recycled alum were thoroughly investigated using Differential Scanning Calorimetry (DSC) and Thermogravimetric Analysis (TGA) under a nitrogen atmosphere. The results (Fig. 5, 6 and Table 1) reveal critical insights into the influence of Recy. Al on thermal transitions, decomposition behavior, and residual stability of hybrid materials.| Sample | Tga (°C) | Td5b (°C) | Td10c (°C) | Ycd (%) |
|---|---|---|---|---|
| a Glass transition temperature under nitrogen.b Temperature for 5% weight loss under nitrogen in TGA.c Temperature for 10% weight loss under nitrogen in TGA.d Residual weight of the sample heated at 10 °C min−1 until 700 °C under nitrogen in TGA. | ||||
| Pristine CS/PVA | — | 188 | 239 | 27 |
| CS/PVA-5 wt% Recy. Al | — | 168 | 220 | 33 |
| CS/PVA-10 wt% Recy. Al | — | 172 | 222 | 34 |
| CS/PVA-20 wt% Recy. Al | 102.7 | 176 | 228 | 36 |
| CS/PVA-30 wt% Recy. Al | 98.9 | 185 | 232 | 33 |
| CS/PVA-50 wt% Recy. Al | 96 | 223 | 240 | 38 |
The DSC thermograms (Fig. 5) for the pristine CS/PVA blend did not exhibit a clearly defined glass transition within the measured range, which is consistent with its semi-amorphous hydrogen-bonded structure. Upon the incorporation of recycled alum, distinct endothermic relaxation events emerged, reflecting an increase in the microheterogeneity within the composite. At 20 wt% Recy. Al, a Tg-like transition appeared at 102.7 °C, which decreased to 98.9 °C and 96 °C with further increases to 30 wt% and 50 wt% Recy. Al, respectively. This gradual downward shift is attributed to the disruption of polymer–polymer hydrogen bonding by the growing inorganic phase and the formation of alum–polymer interfacial regions in the gel. The presence of these microdomains weakens the segmental rigidity and promotes easier molecular relaxation, which is consistent with the localized structural ordering observed in the XRD patterns at higher alum loadings.50
The TGA results (Fig. 6) revealed the typical multistep degradation behavior of the CS/PVA systems. The initial mass loss below ∼120 °C corresponds to the evaporation of physically bound moisture and traces of acetic acid in the sample.49,51 Subsequent decomposition between 200 and 300 °C is associated with the cleavage of chitosan glycosidic linkages and the breakdown of PVA hydroxyl-bearing segments, while the third stage (300–550 °C) represents the degradation of intermediate residues and carbonaceous fragments, respectively.52–54
Pristine CS/PVA exhibited Td5 = 188 °C and Td10 = 239 °C, with a char yield of 27%. The addition of 5–20 wt% alum led to a slight decrease in Td5 (168–176 °C) and Td10 (220–228 °C), likely due to the catalytic dehydration effect of alum's sulfate content, which promotes early weight loss. Nevertheless, these composites showed a marked improvement in char yield, increasing to 33–36%, indicating that alum enhances residue formation and interacts favorably with the developing carbonaceous structure during decomposition. As the alum loading increased to 30 wt%, Td5 and Td10 shifted upward to 185 °C and 232 °C, respectively, suggesting reduced catalytic degradation owing to improved filler dispersion and more effective polymer–filler interfacial interactions. At the highest loading of 50 wt% Recy. Al, the biocomposite showed the most pronounced thermal enhancement, with Td5 = 223 °C, Td10 = 240 °C, and a char yield of 38%. This significant improvement is attributed to the dense inorganic network formed by alum, which acts as a thermal barrier, slowing heat transfer and restricting the diffusion of volatile degradation products. The high char yield further supports the formation of a more compact, ceramic-like protective layer at elevated temperatures.
3.5. Mechanical analysis of bio-composites
The mechanical behavior of polymer composites is strongly influenced by the dispersion quality and interfacial interactions of the filler within the matrix. Well-distributed inorganic particles can effectively hinder crack propagation, redistribute stress, and enhance load transfer efficiency, resulting in improved stiffness and tensile resistance.50,55 In contrast, excessive filler content often leads to particle agglomeration, microstructural discontinuities, and stress localization, ultimately diminishing ductility and compromising overall mechanical robustness.56–59The tensile data for pristine CS/PVA and the bio-composites containing 5–50 wt% recycled alum are presented in Table 2 and illustrated in the stress–strain curves of Fig. 7. The pristine CS/PVA film exhibited a tensile strength of 12.2 ± 6.4 MPa, Young's modulus of 84.2 ± 64.1 MPa, and elongation at break of 10.3 ± 4.2%, reflecting the characteristic flexibility and low intrinsic stiffness of the neat biopolymer matrix. Incorporation of 5 wt% recycled alum produced a notable strengthening effect, increasing tensile strength to 16.3 ± 1.3 MPa and stiffness to 129.2 ± 90.4 MPa, while the elongation improved to 15.4 ± 8.6%. This simultaneous enhancement in strength, modulus, and ductility suggests excellent particle dispersion and favorable polymer–filler interactions that facilitate efficient stress transfer without severely restricting chain mobility. At 10 wt% loading, the tensile strength remained high (16.7 ± 4.9 MPa) and the modulus increased further to 180.2 ± 78.6 MPa; however, the elongation decreased to 9.9 ± 4.6%. This reduction in flexibility is likely associated with the onset of localized heterogeneity or the formation of microaggregates that begin to restrict polymer relaxation. The 20 wt% composite exhibited the highest tensile strength among the tested films (19.0 ± 1.7 MPa) and a marked increase in stiffness (243.6 ± 32.6 MPa), confirming the reinforcing capability of alum at moderate loading. Nevertheless, the elongation dropped to 8.3 ± 1.8%, indicating the expected trade-off between stiffness and ductility as the inorganic phase became more dominant. Beyond this optimum range, the mechanical response changes. The 30 wt% film could not be reliably tested due to structural irregularities and premature failure, reflecting excessive filler aggregation and poor stress distribution. At 50 wt% loading, the composite exhibited extremely limited extensibility (4.6 ± 0.5%) and a significantly elevated modulus (501.3 ± 15.9 MPa), demonstrating that the matrix becomes highly rigid and brittle when the inorganic phase overwhelms the polymer network. Although the strength at this level (21.1 ± 3.3 MPa) appears comparatively high, the severe loss of elasticity and predominance of brittle fracture behavior significantly limit its practical applicability. These findings underscore the importance of balancing filler content to optimize mechanical performance and support the use of upcycled alum in developing cost-effective, environmentally friendly biocomposites suitable for packaging, coating, and structural applications.
| Sample | εmaxa (%) | σmaxb (MPa) | Young's modulusc (MPa) |
|---|---|---|---|
| a Ultimate extensibility.b Ultimate tensile stress.c Young's modulus, calculated from the slope of flexural stress–strain curves. | |||
| Pristine CS/PVA | 10.3 ± 4.2 | 12.2 ± 6.4 | 84.2 ± 64.1 |
| CS/PVA-5 wt% Recy. Al | 15.4 ± 8.6 | 16.3 ± 1.3 | 129.2 ± 90.4 |
| CS/PVA-10 wt% Recy. Al | 9.9 ± 4.6 | 16.7 ± 4.9 | 180.2 ± 78.6 |
| CS/PVA-20 wt% Recy. Al | 8.3 ± 1.8 | 19.0 ± 1.7 | 243.6 ± 32.6 |
| CS/PVA-30 wt% Recy. Al | 4.6 ± 0.5 | 21.1 ± 3.3 | 501.3 ± 15.9 |
| CS/PVA-50 wt% Recy. Al | — | — | — |
 | ||
| Fig. 7 Stress–strain analysis for various combinations of pristine CS/PVA and CS/PVA-containing different loading (5–30 wt%) of Recy. Al. | ||
3.6. Surface morphology of bio-composites
The SEM micrographs (Fig. 8a) of the pristine CS/PVA film reveal a smooth, continuous, and homogeneous surface, confirming the excellent miscibility between chitosan and PVA. This uniformity is attributed to strong hydrogen bonding interactions between the hydroxyl and amino functionalities of both polymers, which promote the formation of a cohesive and well-integrated matrix.44,45 In contrast, the surface of the CS/PVA composite containing 20 wt% Recy. Al (Fig. 8b) displayed a rougher texture with distinct elongated, oval-shaped alum particles embedded within the matrix.41,60 The compatibility of recycled alum with the CS/PVA matrix was further facilitated by hydrogen bonding between the polymer functional groups (–OH and –NH2) and the alum surface. These features suggest particle agglomeration and partial alignment during film casting, likely because of the high filler concentration. The observed morphology is consistent with earlier reports on high inorganic filler content in polymer blends.The SEM–EDS spectrum (Fig. 9) further confirms the presence of recycled alum within the composite by detecting the characteristic elemental signals. Complementary elemental mapping analyses (Fig. 10a–f) illustrate the distribution of aluminum, sulfur, and oxygen across the fractured surface, verifying the successful incorporation of recycled alum and its interfacial embedding within the CS/PVA network. The relatively uniform elemental distribution, particularly at 30 wt% loading, indicates that the filler is effectively integrated, even though localized aggregation occurs at higher concentrations. These results confirm that recycled alum acts as a structurally influential filler in the CS/PVA matrix.
 | ||
| Fig. 9 SEM-EDS analysis for the occurrence of Recy. Al in the fracture surface of 20 wt% Recy. Al-CS/PVA biocomposites. | ||
3.7. Moisture and water solubility of bio-composites
Moisture content and water solubility are critical parameters governing the dimensional stability, durability, and environmental performance of biopolymer-based composite films.61 Both chitosan (CS) and poly(vinyl alcohol) (PVA) are inherently hydrophilic due to the presence of abundant hydroxyl (–OH) and amino (–NH2) functional groups, which readily interact with water molecules through hydrogen bonding.The moisture content of pristine CS/PVA and CS/PVA–Recy. Al biocomposite films is presented in Fig. 11. The pristine CS/PVA film exhibited a moisture content of 13.6%, reflecting the strong affinity of the polymer blend toward atmospheric moisture. Upon incorporation of recycled alum, the moisture content increased to 17.18% and 16.97% for composites containing 5 wt% and 10 wt% Recy. Al, respectively. This increase can be attributed to the hydrated nature of potassium alum and the presence of sulfate groups, which enhance moisture retention through additional polar sites. With further increases in recycled alum content, the moisture content slightly decreased to 16.71% and 15.73% for 20 wt% and 30 wt% Recy. Al, respectively, suggesting improved matrix densification and reduced availability of free hydrophilic sites. At the highest filler loading (50 wt% Recy. Al), the moisture content increased again to 17.07%, likely due to the dominance of alum-associated hydration over polymer–polymer interactions.
The water solubility results of pristine CS/PVA and CS/PVA–Recy. Al biocomposites after 24 h immersion in distilled water are illustrated in Fig. 12. The pristine CS/PVA film exhibited a high water solubility of 114%, consistent with its hydrophilic nature. The incorporation of recycled alum at low loadings resulted in a pronounced increase in water solubility, reaching 168% and 171% for 5 wt% and 10 wt% Recy. Al, respectively. This enhancement is attributed to the introduction of hydrated aluminum and sulfate species, which facilitate water diffusion into the polymer matrix and promote partial film disintegration. At higher recycled alum contents, the water solubility progressively decreased to 148% and 119% for 20 wt% and 30 wt% Recy. Al, respectively. This reduction is associated with stronger interfacial interactions between the CS/PVA matrix and alum particles, including hydrogen bonding and electrostatic interactions, which restrict polymer chain mobility and limit water penetration. Notably, the composite containing 50 wt% Recy. Al exhibited a markedly reduced water solubility of 32%, indicating the formation of a highly compact and reinforced structure that significantly suppresses polymer dissolution.
Thus, the moisture content and water solubility results demonstrate that recycled alum content plays a decisive role in regulating water–polymer interactions. At lower filler contents, the hydrophilic nature of recycled alum enhances water uptake, whereas at higher loadings, polymer–filler interactions and structural densification dominate, leading to improved water resistance of the biocomposite films.
3.8. Biological applications
| Compounds | Inhibition zone diameter (mm) | |||||
|---|---|---|---|---|---|---|
| Bacteria | Fungi | |||||
| E. coli | Proteus sp. | S. aureus | B. subtilis | S. crevice | A. niger | |
| a The changes are insignificant at p ≤ 0.05.b The changes are significant at p ≤ 0.01. | ||||||
| Cans waste | Negative | Negative | Negative | Negative | Negative | Negative |
| Recy. Al | 13 ± 1 | 12 ± 1 | 13 ± 1 | 10 ± 1 | 11 ± 1 | 9 ± 1 |
| Pristine CS/PVA | 13 ± 1a | 9 ± 1a | 12 ± 1a | 10 ± 1a | 12 ± 1a | 9 ± 1a |
| CS/PVA-5 wt% Recy. Al | 21 ± 2b | 19 ± 2b | 20 ± 2b | 19 ± 2b | 23 ± 2b | 17 ± 2b |
| CS/PVA-10 wt% Recy. Al | 36 ± 2b | 29 ± 2b | 34 ± 3b | 28 ± 3b | 35 ± 3b | 30 ± 3b |
| CS/PVA-20 wt% Recy. Al | 45 ± 3b | 33 ± 3b | 41 ± 4.0b | 39 ± 3.0b | 45 ± 3b | 35 ± 3b |
| CS/PVA-30 wt% Recy. Al | 16 ± 1b | 15 ± 1a | 15 ± 1a | 12 ± 1a | 13 ± 1a | 14 ± 1a |
| CS/PVA-50 wt% Recy. Al | 10 ± 1a | 11 ± 1a | 11 ± 1a | 10 ± 1a | 13 ± 1a | 10 ± 1a |
| Gentamycin (1 µg mL−1) | 15 ± 1a | 11 ± 1a | — | — | — | — |
| Amoxicillin (2 µg mL−1) | — | — | 15 ± 1a | 13 ± 1a | — | — |
| Fluconazole (4 µg mL−1) | — | — | — | — | 14 ± 1a | 16 ± 1a |
This enhancement is attributed to the synergistic interactions between the alum-derived ionic species and the protonated CS/PVA matrix, which collectively disrupts microbial cell membranes and interferes with metabolic pathways. These results are consistent with reports showing that metal salts and polysaccharide-based matrices exert antimicrobial effects through ion release, electrostatic interactions, and membrane destabilization.62 However, at higher loadings (≥30 wt%), antimicrobial efficacy declined, with the CS/PVA–50 wt% composite showing significantly lower inhibition zones (10–13 mm). This reduction is attributed to alum agglomeration, which decreases the surface accessibility of active sites and possibly limits the diffusion of alum ions through microbial membranes at very high filler contents.
The MIC and MBC assays further supported the antimicrobial trends. The composites containing 10 wt% and 20 wt% Recy. Al demonstrated MBC/MIC tolerance ratios of 2, classifying them as bactericidal/fungicidal agents rather than merely growth-inhibiting (static) agents, following the criteria described by Mogana et al.62 A ratio ≤4 is typically indicative of cidal action, although this may vary depending on the microbial strain, environmental conditions, treatment duration, and agent concentration. These findings confirm that alum-containing CS/PVA biocomposites, particularly at 10–20 wt% Recy. Al, exert strong killing activity rather than simply suppressing microbial growth.
Statistical analysis showed significant differences in the antimicrobial activity of composites containing 5–20 wt% Recy. Al (p ≤ 0.01), confirming that the chemical and morphological modifications induced by alum incorporation play a crucial role in enhancing the biological performance. These results position the optimized composites (10–20 wt% Recy. Al) as promising antimicrobial materials for use in biomedical coatings, food packaging, wound dressings, and water purification systems.
| Cpd. | Bacteria | Fungi | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| E. coli | S. aureus | S. cerevisae | A. niger | |||||||||
| MICs (µg mL−1) | MCD (µg mL−1) | Tolerance value (MCD/MIC) | MICs (µg mL−1) | MCD (µg mL−1) | Tolerance value (MCD/MIC) | MICs (µg mL−1) | MCD (µg mL−1) | Tolerance value (MCD/MIC) | MICs (µg mL−1) | MCD (µg mL−1) | Tolerance value (MCD/MIC) | |
| a Bacteriocidal or fungicidal: tolerance value (MBC/MIC) was ≤4.b Bacteriostatic or fungistatic: tolerance value (MBC/MIC) was >4.c The changes are insignificant at p ≤ 0.05.d The changes are significant at p ≤ 0.01. | ||||||||||||
| Recy. Al | 125 ± 10 | 500 ± 40 | 4 | 125 ± 10 | 500 ± 45 | 4 | 250 ± 25 | 500 ± 55 | 4 | 125 ± 12 | 500 ± 45 | 4 |
| Pristine CS/PVA | 125 ± 10c | 250 ± 23d | 2 | 125 ± 10c | 125 ± 14d | 1 | 125 ± 10d | 500 ± 55c | 4 | 250 ± 12c | 500 ± 45c | 4 |
| CS/PVA-5 wt% Recy. Al | 62.5 ± 7d | 125 ± 12d | 2 | 62.5 ± 7d | 125 ± 14d | 2 | 62.5 ± 6d | 125 ± 14d | 2 | 125 ± 10c | 500 ± 45c | 4 |
| CS/PVA-10 wt% Recy. Al | 31.25 ± 4d | 62.5 ± 4d | 2 | 31.25 ± 4d | 62.5 ± 8d | 2 | 31.25 ± 3d | 62.5 ± 6d | 2 | 31.25 ± 4d | 62.5 ± 4d | 2 |
| CS/PVA-20 wt% Recy. Al | 7.8 ± 1d | 15.626 ± 1d | 2 | 7.8 ± 1d | 15.426 ± 1d | 2 | 62.5 ± 6d | 125 ± 10d | 2 | 62.5 ± 6d | 125 ± 10d | 2 |
| CS/PVA-30 wt% Recy. Al | 62.5 ± 6d | 250 ± 30d | 4 | 125 ± 12c | 500 ± 49c | 4 | 125 ± 12d | 500 ± 50c | 4 | 125 ± 10c | 500 ± 35c | 4 |
| CS/PVA-50 wt% Recy. Al | 125 ± 10c | 500 ± 38c | 4 | 125 ± 12c | 500 ± 48c | 4 | 125 ± 12d | 500 ± 50c | 4 | 125 ± 12c | 500 ± 46c | 4 |
The CS/PVA–10 wt% and CS/PVA–20 wt% Recy. Al composites showed MBC/MIC ratios of 2, confirming strong biocidal activity against both bacteria and fungi. This behavior arises from the integrated mechanisms of alum ion release, protonated CS/PVA interactions, and membrane destabilization. These biocidal composites demonstrate potential for real-world applications requiring rapid microbial eradication, such as disinfection materials, antimicrobial coatings, and biodegradable packaging.
| Compounds | Survival % | |||
|---|---|---|---|---|
| Concentration (mg mL−1) | 0 | 0.01 | 0.1 | 1.00 |
| Recy. Al | 100 | 100 | 100 | 45 |
| Pristine CS/PVA | 100 | 100 | 80 | 40 |
| CS/PVA-5 wt% Recy. Al | 100 | 100 | 80 | 30 |
| CS/PVA-10 wt% Recy. Al | 100 | 100 | 70 | 25 |
| CS/PVA-20 wt% Recy. Al | 100 | 100 | 60 | 23 |
| CS/PVA-30 wt% Recy. Al | 100 | 60 | 40 | 0 |
| CS/PVA-50 wt% Recy. Al | 100 | 50 | 35 | 0 |
4. Conclusion
A new series of sustainable CS/PVA biocomposites was successfully developed by incorporating recycled potassium alum (Recy. Al) derived from aluminum can waste. The addition of alum notably improved both the structural integrity and functional performance of the CS/PVA matrix. The optimum formulation (30 wt% Recy. Al) exhibited a ∼55% increase in tensile strength and a 2.5-fold rise in Young's modulus compared to the pristine film. Thermal analysis revealed a distinct improvement in thermal stability, with Td5 and Td10 values increasing progressively from 188 °C and 239 °C (pristine) to 223 °C and 240 °C (50 wt% Recy. Al), along with an increase in char yield from 27% to 38%. The enhancement in stability is attributed to the presence of inorganic alum domains that restrict polymer chain mobility, facilitate char formation, and act as effective thermal barriers during degradation. XRD patterns suggested improved molecular organization, while SEM and EDS mapping confirmed homogeneous alum distribution and good interfacial compatibility within the polymer matrix. The composites retained high optical transparency and surface uniformity, reflecting strong polymer–filler compatibility. Future studies will include the evaluation of hardness and impact strength to further assess the mechanical durability of the developed biocomposites.Importantly, all alum-reinforced CS/PVA composites exhibited broad-spectrum antimicrobial activity against both Gram-positive and Gram-negative bacteria, verifying their suitability for hygienic, biomedical, and eco-friendly applications. Their demonstrated antimicrobial cidal effect against common pathogens further underscores their potential in medical devices, food packaging, and water purification systems. Future perspectives include performing biodegradation studies to assess the environmental fate of these biocomposites and evaluate their long-term stability and real-world application performance.
Author contributions
Mahmoud A. Abdelkawy: conceptualization, methodology, validation, investigation, formal analysis, writing-original draft, writing – review & editing. Abdelbaset Shoker: investigation, methodology, validation, review & editing, and supervision. Badr Isamil Badr: antimicrobial investigation, data curation, validation, review & editing. Yehia A.-G. mahmoud: microbiological analysis, supervision, and review & editing.Data availability
The data that support the findings of this study are available within the article.Conflicts of interest
The authors declare no competing interests.References
- M. Lu, C. Zhou, C. Wang, R. B. Jackson and C. P. Kempes, Worldwide scaling of waste generation in urban systems, Nat. Cities, 2024, 1, 126–135 Search PubMed.
- A. Maalouf, A. Mavropoulos and M. El-Fadel, Global municipal solid waste infrastructure: Delivery and forecast of uncontrolled disposal, Waste Manage. Res., 2020, 38, 1028–1036 Search PubMed.
- Q. Xu, T. Zhang, Y. Niu, S. Mukherjee, S. F. Abou-Elwafa, N. S. H. Nguyen, N. M. Al Aboud, Y. Wang, M. Pu and Y. Zhang, A comprehensive review on agricultural waste utilization through sustainable conversion techniques, with a focus on the additives effect on the fate of phosphorus and toxic elements during composting process, Sci. Total Environ., 2024, 942, 173567 CrossRef CAS PubMed.
- S. Bhuvaneshwari, H. Hettiarachchi and J. N. Meegoda, Crop residue burning in India: Policy challenges and potential solutions, Int. J. Environ. Res. Public Health, 2019, 16, 832 Search PubMed.
- A. Mesjasz-Lech, Trends of Industrial Waste Generation in Manufacturing Enterprises in the Context of Waste Prevention-Shift-Share Analysis for European Union Countries, Sustainability, 2025, 17, 34 Search PubMed.
- J. Chen, B. Zeng, W. Xu, S. Hu, S. Wang, K. Wang, W. Zhang, S. Wu and Z. Song, Mechanical behaviour and damage constitutive model of semi-circular arch tunnels with straight walls under soaking conditions, Eng. Failure Anal., 2025, 169, 109137 Search PubMed.
- Y. Fang, K. Chao, J. He, Z. Wang and Z. Chen, High-efficiency depolymerization/degradation of polyethylene terephthalate plastic by a whole-cell biocatalyst, Biotech, 2023, 13, 138 Search PubMed.
- M. Lu, W. Ge, Y. Xia, C. Sun, X. Lin, D. C. Tsang, T.-C. Ling, Y. Hu, L. Wang and J. Yan, Upcycling MSWI fly ash into green binders via flue gas-enhanced wet carbonation, J. Cleaner Prod., 2024, 440, 141013 Search PubMed.
- S. Kashyap and D. Datta, Reusing industrial lime sludge waste as a filler in polymeric composites, Mater. Today: Proc., 2017, 4, 946–955 Search PubMed.
- L. Djekoune, A. Maaliou, Z. Salem, D. Ziani, R. Kamel, A. Ouakouak, O. Baigenzhenov, D. O. Bokov, A. Ivanets and A. Hosseini-Bandegharaei, Phosphate adsorption on dried alum sludge: Modeling and application to treatment of dairy effluents, Environ. Res., 2024, 252, 118976 Search PubMed.
- E. M. M. Ewais, R. M. Elsaadany, A. A. Ahmed, N. H. Shalaby and B. E. H. Al-Anadouli, Insulating refractory bricks from water treatment sludge and rice husk ash, Refract. Ind. Ceram., 2017, 58, 136–144 Search PubMed.
- F. R. Ahmed, M. A. Muhammad and R. K. Ibrahim, Effect of alum sludge on concrete strength and two-way shear capacity of flat slabs, Structures, 2022, 40, 991–1001 Search PubMed.
- J. Kluczka, M. Zołotajkin, J. Ciba and M. Staroń, Assessment of aluminum bioavailability in alum sludge for agricultural utilization, Environ. Monit. Assess., 2017, 189, 422 Search PubMed.
- X. Zhao, Y. Zhao, W. Wang, Y. Yang, A. Babatunde, Y. Hu and L. Kumar, Key issues to consider when using alum sludge as substrate in constructed wetland, Water Sci. Technol., 2015, 71, 1775–1782 Search PubMed.
- V. Agarwal, P. Halli, S. Helin, F. Tesfaye and M. Lundström, Electrohydraulic Fragmentation of Aluminum and Polymer Fractions from Waste Pharmaceutical Blisters, ACS Sustain. Chem. Eng., 2020, 8(10), 4137–4145, DOI:10.1021/acssuschemeng.9b06810.
- Y. Cao, S. Shen, L. Li, D. Luo, X. Wang, F. Wang, Z. Di, E. Liu and S. Yang, Upcycling aluminum-plastic packaging waste into high thermal conductivity and fire safety composite, Composites, Part A, 2025, 194, 108887, DOI:10.1016/j.compositesa.2025.108887.
- A. Al Mahmood, M. A. Kader, M. B. Islam and R. Hossain, Sustainable transformation of waste Aluminium into high performance composites: A review, Int. J. Lightweight Mater. Manuf., 2025, 8, 194–204, DOI:10.1016/j.ijlmm.2024.11.002.
- E. Fortunati, M. Peltzer, I. Armentano, L. Torre, A. Jimenez and J. M. Kenny, Effects of modified cellulose nanocrystals on the barrier and migration properties of PLA nano-biocomposites, Carbohydr. Polym., 2012, 90, 948–956 CrossRef CAS PubMed.
- A. L. Goffin, J. M. Raquez, E. Duquesne, G. Siqueira, Y. Habibi, A. Dufresne and P. Dubois, From interfacial ring-opening polymerization to melt processing of cellulose nanowhisker-filled polylactide-based nanocomposites, Biomacromolecules, 2011, 12, 2456–2465 Search PubMed.
- P. Kanmani and J. W. Rhim, Properties and characterization of bionanocomposite films prepared with various biopolymers and ZnO nanoparticles, Carbohydr. Polym., 2014, 106, 190–199 Search PubMed.
- J. W. Rhim and P. K. Ng, Natural biopolymer-based nanocomposite films for packaging applications, Crit. Rev. Food Sci. Nutr., 2007, 47, 411–433 CrossRef CAS PubMed.
- C. González, A. Hernández and V. Castaño, et al., Polysaccharide nanocomposites reinforced with graphene oxide and keratin-grafted graphene oxide, MRS Online Proc. Libr., 2012, 1453, 57–63 Search PubMed.
- L. A. van den Broek, R. J. Knoop, F. H. Kappen and C. G. Boeriu, Chitosan films and blends for packaging material, Carbohydr. Polym., 2015, 116, 237–242 Search PubMed.
- M. Dash, F. Chiellini, R. M. Ottenbrite and E. Chiellini, Chitosan – a versatile semi-synthetic polymer in biomedical applications, Prog. Polym. Sci., 2011, 36, 981–1014 CrossRef CAS.
- J. Bonilla, E. Fortunati, L. Atarés, A. Chiralt and J. M. Kenny, Physical, structural and antimicrobial properties of poly(vinyl alcohol)–chitosan biodegradable films, Food Hydrocolloids, 2014, 35, 463–470 Search PubMed.
- P. C. Srinivasa, M. N. Ramesh, K. R. Kumar and R. N. Tharanathan, Properties and sorption studies of chitosan–poly(vinyl alcohol) blend films, Carbohydr. Polym., 2003, 53, 431–438 Search PubMed.
- K. Vidyalakshmi, K. N. Rashmi, T. M. Pramod Kumar and Siddaramaiah, Studies on formulation and in vitro evaluation of PVA/chitosan blend films for drug delivery, J. Macromol. Sci., Part A:Pure Appl. Chem., 2004, 41, 1115–1122 CrossRef.
- K. Lewandowska, Miscibility and thermal stability of poly(vinyl alcohol)/chitosan mixtures, Thermochim. Acta, 2009, 493, 42–48 Search PubMed.
- WHO, World Health Organization Ten Threats to Global Health, WHO, Geneva, Switzerland, 2019 Search PubMed.
- H. Michael, D. W. Thomas, J. L. Craighead, C. Economides and J. Rosenthal, Clinical development success rates for investigational drugs, Nat. Biotechnol., 2014, 32(1), 40–51 CrossRef PubMed.
- A. U. Birnin-Yauri and M. Aliyu, Synthesis and analysis of potassium aluminium sulphate (alum) from waste aluminium can, Int. J. Adv. Res. Chem. Sci., 2014, 1, 1–6 Search PubMed.
- A. Sionkowska, Prog. Polym. Sci., 2011, 36, 1254–1276, DOI:10.1016/j.progpolymsci.2011.05.003.
- Y. Wang, Y. Li, Z. Zhao, M. Chen, J. Song, J. Chen, Q. Du and B. Zhang, Polymers, 2019, 11, 201, DOI:10.3390/polym11020201.
- A. Farris, K. Schaich, J. Liu, S. Piergiovanni and M. Yam, J. Agric. Food Chem., 2011, 59, 11317–11326, DOI:10.1021/jf202085k.
- H. Chen, L. Zhao, Y. Liu, W. Xu, G. Zhang, H. Li and J. Zhu, Carbohydr. Polym., 2015, 117, 310–317, DOI:10.1016/j.carbpol.2014.09.039.
- R. Jayakumar, M. Prabaharan, P. T. S. Kumar, S. V. Nair and H. Tamura, Biotechnol. Adv., 2011, 29, 322–337, DOI:10.1016/j.biotechadv.2011.01.005.
- D. Gopakumar, M. Pagolu, A. Pasquini, S. Thomas and S. Maria, Carbohydr. Polym., 2019, 211, 207–215, DOI:10.1016/j.carbpol.2019.01.104.
- R. A. Karimi and C. Eslami, Mono- and bis-2-amino-4H-pyrans: alum-catalyzed three- or pseudo five-component reaction of 4-hydroxycoumarin, malononitrile and aldehydes, Lett. Org. Chem., 2011, 8, 150–154 Search PubMed.
- B. Sadeghi, E. Farahzadi and A. Hassanabadi, KAl(SO4)2·12H2O as an eco-friendly and reusable catalyst for the synthesis of amides by the Ritter reaction, J. Chem. Res., 2012, 36, 539–540 Search PubMed.
- M. M. Heravi, M. Zakeri, N. Mohammadi and H. Haghi, KAl(SO4)2·12H2O or KHSO4: efficient and inexpensive catalysts for the one-pot synthesis of β-acetamido ketones by the Dakin–West reaction, Synth. React. Inorg. Met.-Org. Chem., 2012, 42, 178–182 Search PubMed.
- N. Wijayati, L. R. Lestari, L. A. Wulandari, F. W. Mahatmanti, S. K. Rakainsa, E. Cahyono and R. Abdul Wahab, Potassium alum [KAl(SO4)2·12H2O] solid catalyst for effective and selective methoxylation production of α-pinene ether products, Heliyon, 2021, 7, e06058 Search PubMed.
- O. Deveoglu, E. Cakmakcı, T. Taskopru, E. Torgan and R. Karadag, Identification by RP-HPLC-DAD, FTIR, TGA and FESEM-EDAX of natural pigments prepared from Datisca cannabina L, Dyes Pigm., 2012, 94, 437–442 Search PubMed.
- A. M. Abdulwahab, Y. A. A. Al-Magdashi, A. Meftah, D. A. Al-Eryani and A. A. Qaid, Growth, structure, thermal, electrical and optical properties of potassium aluminum sulfate dodecahydrate (potash alum) single crystal, Chin. J. Phys., 2019, 60, 510–521 Search PubMed.
- H. M. P. Naveen Kumar, M. N. Prabhakar, C. Venkata Prasad, K. Madhusudhan Rao, T. V. Ashok Kumar Reddy, K. Chowdoji Rao and M. C. S. Subha, Compatibility studies of chitosan/pva blend in 2% aqueous acetic acid solution at 30 C, Carbohydr. Polym., 2010, 82, 251–255 Search PubMed.
- S. Jasrotia, S. Gupta, M. L. Kudipady and Y. M. Puttaiahgowda, Development and Characterization of Chitosan−PVA−Tannic Acid Film for Extended Shelf Life and Safety of Food Products, ACS Omega, 2025, 10, 19361–19378 Search PubMed.
- E. A. El-Hefian, M. M. Nasef and A. H. Yahaya, The preparation and characterization of chitosan/poly (vinyl alcohol) blended films, J. Chem., 2010, 7, 1212–1219 Search PubMed.
- H. Zheng, Y. Du, J. Yu, R. Huang and L. Zhang, Preparation and characterization of chitosan/poly(vinyl alcohol) blend fibers, J. Appl. Polym. Sci., 2001, 80, 2558–2565 Search PubMed.
- S. Azizi, M. B. Ahmad, N. A. Ibrahim, M. Z. Hussein and F. Namvar, Cellulose nanocrystals/ZnO as a bifunctional reinforcing nanocomposite for poly(vinyl alcohol)/chitosan blend films: fabrication, characterization and properties, Int. J. Mol. Sci., 2014, 15, 11040–11053 CrossRef CAS PubMed.
- K. Choo, Y. C. Ching, C. H. Chuah, S. Julai and N.-S. Liou, Preparation and characterization of poly(vinyl alcohol)–chitosan composite films reinforced with cellulose nanofiber, Materials, 2016, 9, 644 Search PubMed.
- D. Sun, T. Henthorn, C.-M. Popescu and R. Salehiyan, Upcycling alum sludge as a reinforcement in PBAT composites: a sustainable approach to waste valorisation, Appl. Sci., 2025, 15, 2591 CrossRef CAS.
- Y. Yusof, H. Illias and M. Kadir, Incorporation of NH4Br in PVA–chitosan blend-based polymer electrolyte and its effect on the conductivity and other electrical properties, Ionics, 2014, 20, 1235–1245 CrossRef CAS.
- E. A. El-Hefian, M. M. Nasef and A. H. Yahaya, Preparation and characterization of chitosan/poly(vinyl alcohol) blended films: mechanical, thermal and surface investigations, J. Chem., 2011, 8, 91–96 Search PubMed.
- J. Bonilla, E. Fortunati, L. Atarés, A. Chiralt and J. M. Kenny, Physical, structural and antimicrobial properties of poly(vinyl alcohol)–chitosan biodegradable films, Food Hydrocolloids, 2014, 35, 463–470 Search PubMed.
- A. Khan, R. A. Khan, S. Salmieri, C. Le Tien, B. Riedl, J. Bouchard, G. Chauve, V. Tan, M. R. Kamal and M. Lacroix, Mechanical and barrier properties of nanocrystalline cellulose reinforced chitosan-based nanocomposite films, Carbohydr. Polym., 2012, 90, 1601–1608 Search PubMed.
- Q. Meng and T. Wang, An improved crack-bridging model for rigid particle–polymer composites, Eng. Fract. Mech., 2019, 211, 291–302 CrossRef.
- I. Cosmoiu, D. A. Apostol, C. R. Picu, D. M. Constantinescu, M. Sandu and S. Sorohan, Influence of filler dispersion on the mechanical properties of nanocomposites, Mater. Today: Proc., 2016, 3, 953–958 Search PubMed.
- S.-Y. Fu, X.-Q. Feng, B. Lauke and Y.-W. Mai, Effects of particle size, particle/matrix interface adhesion and particle loading on mechanical properties of particulate–polymer composites, Composites, Part B, 2008, 39, 933–961 CrossRef.
- A. Mostovoy, A. Bekeshev, L. Tastanova, M. Akhmetova, P. Bredihin and Y. Kadykova, The effect of dispersed filler on mechanical and physicochemical properties of polymer composites, Polym. Polym. Compos., 2020, 29, 583–590 Search PubMed.
- Q. Shi, P. Cui, M. Hu, F. Wang, H. Xu and X. Zhou, Solid-State Synthesis of SiC Particle-Reinforced AZ91D Composites: Microstructure and Reinforcement Mechanisms, Metals, 2024, 14, 434 Search PubMed.
- H. C. Park, Y. J. Park and R. Stevens, Synthesis of alumina from high purity alum derived from coal fly ash, Mater. Sci. Eng., A, 2004, 367, 166–170 CrossRef.
- K. Bahsaine, B. El Allaoui, H. Benzeid, M. El Achaby, N. Zari, A.-K. Qaissad and R. Bouhfid, Hemp cellulose nanocrystals for functional chitosan/polyvinyl alcohol-based films for food packaging applications, RSC Adv., 2023, 13, 33294–33304, 10.1039/d3ra06586c.
- R. Mogana, A. Adhikari, M. N. Tzar, R. Ramliza and C. Wiart, BMC Complementary Med. Ther., 2020, 20, 1–11 Search PubMed.
- A. Ishak, N. Mazonakis, N. Spernovasilis, K. Akinosoglou and C. Tsioutis, J. Antimicrob. Chemother., 2025, 80, 1–17, DOI:10.1093/jac/dkae380.
- M. O. Fatope, in Free Radicals and Food Additives, ed. O. Aruoma and B. Halliwell, Taylor and Francis Ltd, London, 1995, pp. 77–119 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |