 Open Access Article
Open Access ArticleOne-pot synthesis of ketones and symmetrical anhydrides from carboxylic acids
Sayanta Roy ,
Gobinda Dolai,
SK Ariyan and
Bhubaneswar Mandal
,
Gobinda Dolai,
SK Ariyan and
Bhubaneswar Mandal *
*
Department of Chemistry, Laboratory of Peptide and Amyloid Research, Indian Institute of Technology Guwahati, Assam-781039, India. E-mail: bmandal@iitg.ac.in
First published on 26th January 2026
Abstract
A one-pot synthesis of ketones from carboxylic acids is presented, employing 2-nitrobenzenesulfonyl chloride as an activating reagent in the presence of organomagnesium or organolithium nucleophiles. Additionally, activation of the carboxylic acids with 2-nitrobenzenesulfonyl chloride enabled the formation of symmetrical anhydrides. Subsequent conversion of these anhydrides into the desired ketones also proceeded with good yields.
Introduction
Ketones are widely found in natural products, pharmaceuticals, and dyes, playing a significant role in organic chemistry.1,2 Converting carboxylic acid to a ketone is a highly demanding organic transformation.3,4 Numerous commonly employed reactions, such as the Mannich reaction, Baeyer–Villiger oxidation, and Grignard reaction, illustrate a diverse range of ketone transformations.5–7 Developing an efficient pathway for synthesizing ketones remains an enduring area of research interest.8,9 The synthesis of ketones from carboxylic acids requires certain activation steps, such as acid chloride, ester, and amide.10–12 In recent times, Weinreb amides have emerged as prominent acylating agents.13,14 Generally, adding an organometallic reagent to the Weinreb amide intermediate readily transforms it to the desired ketone. The formation of a five-member ring chelate intermediate helps to provide stability and operational efficiency.15 While the procedure for the direct synthesis of ketones using excess organometallic reagents also exists, its application in organic synthesis remains limited.16 Various metal-catalyzed processes have also been established for the construction of ketones, yet most of the methods involve using expensive metal catalysts (Scheme 1).17,18 Very recently, DCC-based activation of carboxylic acids, followed by the addition of organolithium reagents to construct ketones, has been reported.19 The process is limited to organolithium nucleophiles, requires an excess of the nucleophile, and produces a stoichiometric amount of byproduct, such as DCU (N, N′-dicyclohexylurea), complicating the separation of the desired product, making the process less feasible. Construction of ketones from carboxylic acids using tosyl chloride as an activator was also established; the applicability of the process was demonstrated only for nBuLi and methylmagnesium bromide nucleophiles.20 The synthetic process requires an extra stoichiometric amount of additive (DMAP) and low temperature (−20 °C), making the process less viable. Additionally, the process involves two steps: the first step involves activation of the carboxylic acid, and then the filtration of the reaction mixture under a N2 atmosphere. The second step involves the addition of a nucleophile. Filtration of the reaction mixture under a nitrogen atmosphere may be a critical step. Addressing the aforementioned problems and developing a new, straightforward, one-pot synthetic method for constructing ketones from carboxylic acids is highly necessary. Carboxylic anhydrides represent an important class of organic compounds widely used as acylating agents and intermediates in various synthetic pathways.21 Aromatic anhydrides are typically synthesized by reacting acyl chlorides with sodium salts of carboxylic acids or by using pyridine, followed by hydrolysis.22 Another approach involves dehydrating carboxylic acids with reagents such as oxalyl chloride, thionyl chloride, phosphorus pentoxide, triphosgene, dicyclohexylcarbodiimide (DCC).23–27 However, many of these methods often suffer from limitations, including high cost, low efficiency, toxic byproducts, and instability during acid activation. Therefore, developing more practical, economical, and safer alternatives remains a key objective in synthetic chemistry.Results and discussion
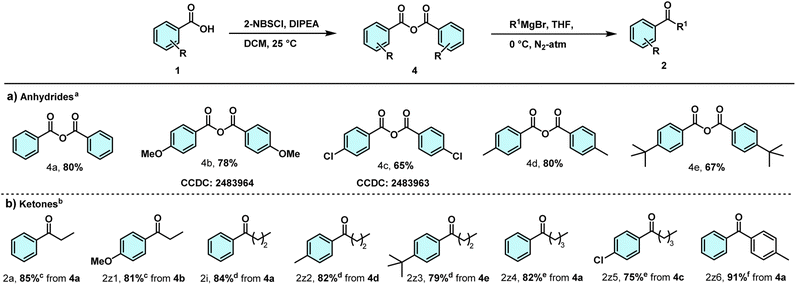
We envisioned that substituted nitrobenzenesulfonyl (Ns) chloride could activate the carboxylic acids efficiently, which can be easily converted to ketones with the treatment of organometallic reagents. Initially, benzoic acid (1a, 1 mmol) was activated using 2-nitrobenzenesulfonyl chloride (2-NBSCl, 1 mmol) and N,N-diisopropylethylamine (DIPEA, 1.3 mmol) in dry tetrahydrofuran (THF, 2 mL) under a nitrogen atmosphere at room temperature. After 30 minutes, TLC revealed the appearance of new spots in the reaction medium, possibly due to the formation of a mixed anhydride and other reactive intermediates. Next, we added CH3CH2MgBr (1.2 mmol, 1 M in THF) in a dropwise manner at −20 °C. A total 60% (46% ketone and 14% ester) yield was obtained in 6 h (entry 1, Table S1). The formation of ketone and ester products was confirmed by 1D (1H, 13C) NMR. Based on the primary result, we proceeded to optimize the reaction conditions for ketone synthesis in different solvents and temperatures. An improved yield (2a, 64%) of the ketone was obtained when the reaction was performed in dry toluene (entry 8, Table S1). Extending the reaction time did not significantly affect the ketone yield. The reaction was also tested with other substituted nitrobenzenesulfonyl (Ns) chlorides (4-nitrobenzenesulfonyl chloride, 4-chloro-2-nitrobenzenesulfonyl chloride, and 2,4-dinitrobenzenesulfonyl chloride) as activating reagents; however, no further improvement in yield was observed (entries 12–14, Table S1). Reaction with 2,4-dinitrobenzenesulfonyl chloride did not offer any ketone formation. Therefore, the conditions used in entry 8 of Table S1 were considered optimal. Under optimized conditions, various ketones were synthesized from aromatic and aliphatic acids and different Grignard reagents (Table 1). Aromatic acids reacted with aliphatic Grignard reagents to give ketones in moderate yields. The aromatic acids bearing electron-donating group (m-OMe, o-OMe, m-, p-OMe) reacted with CH3CH2MgBr to afford the desired ketones (2b–2d) with satisfactory yields (52–66%). The electron-withdrawing groups (p-Br, p-I) containing aromatic acids also responded under optimized conditions (2e, 2f). Alkyl groups (p-Me, p-tBu) containing aromatic acids underwent smooth reaction, offering the desired ketones (2g, 2h) with good yields. Increasing the number of methylene units in the Grignard reagent also satisfies the reaction conditions smoothly. The reaction of CH3CH2CH2MgBr with various aromatic acids offered the desired ketones with satisfactory yields. Aromatic acids bearing electron-donating groups (such as p-OMe, m-OMe, and m-Me) successfully met the reaction conditions and yielded the desired ketones (2j–2l) with satisfactory yields (60–62%). Further using CH3CH2CH2CH2MgBr, the reaction also responded well. The reaction of 4-methoxybenzoic acid and 3-methoxybenzoic acid under optimized conditions offered the desired ketones (2m, 2n) with satisfactory yields (63%, 58%). Halogen bearing benzoic acids, such as 4-I, 4-Br, and 3-Br, after activation, underwent reactions with CH3CH2CH2CH2MgBr, offering the desired ketones (2o–2q). Alkyl-substituted benzoic acids (p-Me, m-Me) reacting with CH3CH2CH2CH2MgBr offered desired ketones (2r, 2s) with satisfactory yields. The applicability of the process is not restricted to the Grignard reagent; Organo-lithium also satisfies the reaction conditions. Activated 4-methoxy and 4-methyl benzoic acids reacted with nBuLi readily, producing the desired ketones (2m, 2r). The yields in these specific cases were very low, possibly due to the higher basic character of nBuLi compared to its nucleophilic character. Heterocyclic acid and an aromatic drug ((S)-Naproxen (NSAID)) containing an aliphatic carboxylic acid also satisfied the reaction conditions, affording the desired ketones (2t, 2u) in moderate yields. In some cases, a small percentage of esters was formed along with the ketone product. We also performed the ketone synthesis with aromatic Grignards, which satisfy the reaction conditions and offer the desired ketones (2v–2y) with satisfactory yields. In the case of aromatic Grignard reagents, no significant ester formation was observed.| a Reaction conditions: 1 (1 mmol, 1 equiv), 2-NBSCl (1 mmol, 1 equiv), DIPEA (1.3 mmol, 1.3 equiv), toluene, 0 °C, N2-atm; aCH3CH2MgBr (1.2 mmol, 1.2 equiv, 1 M in THF); bCH3(CH2)2MgBr (1.2 mmol, 1.2 equiv, 1 M in THF); cCH3(CH2)3MgBr (1.2 mmol, 1.2 equiv, 1 M in THF); dCH3(CH2)3Li (1.2 mmol, 1.2 equiv, 2 M in cyclohexane); eArMgBr (1.2 mmol, 1.2 equiv, 1 M in THF), solvent: THF; 35 min-6 h, Isolated yields. |
|---|
 |
A lower yield percentage of ketone was observed in most cases. We carefully observed the reaction to determine the cause of the lower yield percentage. During the activation of the benzoic acid with 2-nitrobenznesulfonyl chloride, some new spots were found in TLC. When we allowed the intermediate formation for quite a long time (∼2 h), the reaction offered the formation of symmetrical anhydride (benzoic anhydride 4a, Table 2a) of benzoic acid, which was characterized by NMR (1H, 13C) and HRMS analysis. Thus, the formation of such symmetrical anhydrides lowered the amounts of activated acids, hence lowering the ketone yields. Also, the formation of ester byproducts in the reactions lowered the desired yield percentage of ketones. After successfully identifying the symmetrical anhydride formation, we proceeded with reactions to synthesize some symmetrical anhydrides with different aromatic acids (Table 2a). Electron-donating group containing benzoic acid, such as 4-methoxybenzoic acid, readily underwent a reaction, offering good yield of anhydride formation (4b, 78%). Electron-withdrawing group containing benzoic acid (4-Cl benzoic acid) also smoothly participated in the reaction, allowing the formation of symmetrical anhydride 4c with satisfactory yield. The crystal structure of 4b (CCDC: 2483964, SI, Fig. S1 and Table S2) and 4c (CCDC: 2483963, SI, Fig. S1 and Table S2) further confirmed the symmetrical anhydride formation. Alkyl groups (p-Me, p-tBu) containing acids offered the desired anhydrides (4d, 4e) with satisfactory yields. After synthesizing some symmetrical anhydrides, Next, we proceeded to synthesize the ketones using these symmetrical anhydrides (Table 2b). Benzoic anhydride (1 mmol) reacted with CH3CH2MgBr (1.2 mmol), offering the desired ketone 2a with good yield. The reaction of 4-methoxybenzoic anhydride with CH3CH2MgBr offered ketone 2z1 satisfactorily. 4-Methyl and 4-(tert-butyl) benzoic anhydrides readily underwent the reactions with CH3CH2CH2MgBr, offering the desired ketones (2z2, 2z3) with good yields. Additionally, the reaction of benzoic anhydride with a relatively long-chain Grignard reagent (CH3CH2CH2CH2MgBr) yielded the targeted ketone 2z4 with a good yield. 4-Chlorobenzoic anhydride also offered the targeted ketone 2z5 with a moderate yield. p-Tolylmagnesium bromide reacted efficiently with benzoic anhydride to afford the target ketone 2z6 in excellent yield (91%).
| a Reaction conditions: a1 (1 mmol, 1 equiv.), 2-NBSCl (0.5 mmol, 0.5 equiv), DIPEA (1 mmol, 1 equiv), DCM, 25 °C, yields were calculated w.r.t. 0.5 mmol (2-NBSCl); b4 (1 mmol, 1 equiv, Isolated), R1MgBr (1.2 mmol, 1.2 equiv, 1 M in THF), THF (2 mL), 0 °C, N2-atm; cEtMgBr (1.2 mmol, 1.2 equiv, 1 M in THF); dCH3(CH2)2MgBr (1.2 mmol, 1.2 equiv, 1 M in THF); eCH3(CH2)3MgBr (1.2 mmol, 1.2 equiv, 1 M in THF); f4-MePhMgBr (1.2 mmol, 1.2 equiv, 1 M in THF); 2–6 h, Isolated yields. |
|---|
 |
A proposed mechanism for ketone formation is illustrated in Scheme 2a.20 The reaction of a carboxylic acid with 2-nitrobenzenesulfonyl chloride leads to the in situ formation of a mixture of mixed anhydride A and symmetrical anhydride C. Subsequent slow addition of an organometallic nucleophile may result in the formation of intermediate B and D. These intermediates can be stabilized through chelation with metal ions, forming a six-membered chair-like intermediate. Direct elimination of the aryl sulfonate or carboxylate group from the intermediate may then yield the corresponding ketone. The strong electron-withdrawing nature of the nitro group makes the sulfonyl sulfur electron-deficient, thereby enhancing the leaving-group ability of the resulting sulfonate moiety.
 | ||
| Scheme 2 (a) Plausible mechanistic pathway for ketone formation. (b) Plausible mechanistic pathway for ester byproduct formation. | ||
The electron-deficient sulfonyl sulfur withdraws electron density from the ester oxygen, which in turn increases the electrophilic character of the carbonyl carbon, allowing nucleophilic attack to the carbonyl carbon more readily. The reaction of small amounts of the Grignard reagent with the nitro group of intermediate A may lead to the formation of N-hydroxylamine derivative with the elimination of alkoxide F.28,29 The reaction of alkoxide F with activated carboxylic acid may lead to the formation of ester side product in some cases (Scheme 2b). In addition, the presence of unreacted alkyl bromide can react with the carboxylate and may also form small amounts of ester side product in some cases.
Conclusions
In conclusion, we have successfully developed a one-pot synthesis of ketones directly from carboxylic acids. The method involves activation of carboxylic acids using 2-nitrobenzenesulfonyl chloride, followed by nucleophilic addition of organomagnesium or organolithium reagents to yield the desired ketones. Notably, the extended activation time of acids promotes the formation of symmetrical anhydrides. These protocols offer significant advantages, including near-stoichiometric use of organometallic reagents, broad substrate scopes, and straightforward methods.Conflicts of interest
The authors declare no conflicts of interest.Data availability
Supporting data are available in the supplementary information (SI). Supplementary information: experimental details, characterization data and spectra (NMR) for all compounds. See DOI: https://doi.org/10.1039/d5ra08605a.CCDC 2483964 (4b) and 2483963 (4c) contain the supplementary crystallographic data for this paper.30a,b
Acknowledgements
We are thankful to the Central Instruments Facility (CIF), IITG for NMR, Department of Chemistry, IIT Guwahati for SC-XRD, NMR, and other instrumental facilities, and SERB, Govt. of India, for financial support (CRG/2023/004467).References
- R. Ruzi, K. Liu, C. Zhu and J. Xie, Nat. Commun., 2020, 11, 3312 CrossRef CAS PubMed.
- D. J. Foley and H. Waldmann, Chem. Soc. Rev., 2022, 51, 4094–4120 Search PubMed.
- A. Whyte and T. P. Yoon, Angew. Chem., Int. Ed., 2022, 61, e202213739 Search PubMed.
- R. Murata, K. Hirano and M. Uchiyama, Chem.–Asian J., 2015, 10, 1286–1290 CrossRef CAS PubMed.
- H. Wu, Y. Shen, L.-y. Fan, Y. Wan, P. Zhang, C.-f. Chen and W.-x. Wang, Tetrahedron, 2007, 63, 2404–2408 CrossRef CAS.
- C.-H. Liu, Z. Wang, L.-Y. Xiao, Mukadas, D.-S. Zhu and Y.-L. Zhao, Org. Lett., 2018, 20, 4862–4866 CrossRef CAS PubMed.
- P. Sureshbabu, S. Azeez, N. Muniyappan, S. Sabiah and J. Kandasamy, J. Org. Chem., 2019, 84, 11823–11838 CrossRef CAS PubMed.
- M.-C. Fu, R. Shang, B. Zhao, B. Wang and Y. Fu, Science, 2019, 363, 1429–1434 CrossRef CAS PubMed.
- R. Shi and X. Hu, Angew. Chem., Int. Ed., 2019, 58, 7454–7458 CrossRef CAS PubMed.
- J. Amani and G. A. Molander, J. Org. Chem., 2017, 82, 1856–1863 CrossRef CAS PubMed.
- T. Ben Halima, W. Zhang, I. Yalaoui, X. Hong, Y.-F. Yang, K. N. Houk and S. G. Newman, J. Am. Chem. Soc., 2017, 139, 1311–1318 CrossRef CAS PubMed.
- J. A. Murphy, A. G. J. Commeureuc, T. N. Snaddon, T. M. McGuire, T. A. Khan, K. Hisler, M. L. Dewis and R. Carling, Org. Lett., 2005, 7, 1427–1429 Search PubMed.
- S. Hirao, R. Saeki, T. Takahashi, K. Iwai, N. Nishiwaki and Y. Ohga, ACS Omega, 2022, 7, 48476–48483 CrossRef CAS PubMed.
- M. Miele, A. Citarella, N. Micale, W. Holzer and V. Pace, Org. Lett., 2019, 21, 8261–8265 CrossRef CAS PubMed.
- B. Qu and D. B. Collum, J. Org. Chem., 2006, 71, 7117–7119 CrossRef CAS PubMed.
- H. Gilman and P. R. V. Ess, J. Am. Chem. Soc., 1933, 55, 1258–1261 CrossRef CAS.
- J. Ni, X. Xia, D. Gu and Z. Wang, J. Am. Chem. Soc., 2023, 145, 14884–14893 Search PubMed.
- J. Zhao, C. O. Hughes and F. D. Toste, J. Am. Chem. Soc., 2006, 128, 7436–7437 CrossRef CAS PubMed.
- H. G. Mekonnen and S. Jana, Tetrahedron Lett., 2019, 60, 1382–1384 CrossRef CAS.
- D. Sahoo, S. Sarkar and S. Jana, Tetrahedron Lett., 2019, 60, 151084 Search PubMed.
- P. Tung and N. P. Mankad, J. Am. Chem. Soc., 2023, 145, 9423–9427 CrossRef CAS PubMed.
- P. Rambacher and S. Mäke, Angew. Chem., Int. Ed. Engl., 1968, 7, 465 CrossRef CAS.
- G. Kantin, E. Chupakhin, D. Dar'in and M. Krasavin, Tetrahedron Lett., 2017, 58, 3160–3163 CrossRef CAS.
- F. Kazemi, A. R. Kiasat and B. Mombaini, Synth. Commun., 2007, 37, 3219–3223 Search PubMed.
- S. G. Burton and P. T. Kaye, Synth. Commun., 1989, 19, 3331–3335 Search PubMed.
- R. Kocz, J. Roestamadji and S. Mobashery, J. Org. Chem., 1994, 59, 2913–2914 Search PubMed.
- T. Hata, K. Tajima and T. Mukaiyama, Bull. Chem. Soc. Jpn., 2006, 41, 2746–2747 Search PubMed.
- Z. Huang, J. Lv and Y. Jia, ChemistrySelect, 2016, 1, 5892–5894 Search PubMed.
- A. Ricci and M. Fochi, Angew. Chem., Int. Ed., 2003, 42, 1444–1446 CrossRef CAS PubMed.
- (a) CCDC 2483964: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2pcrwq; (b) CCDC 2483963: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2pcrvp.
| This journal is © The Royal Society of Chemistry 2026 |

