 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
A novel magnetic AC/bentonite/Fe3O4/CeO2 nanocomposite catalyst for photocatalytic degradation of persistent chlorinated pesticides in water
Saad S. M. Hassan*a,
Mahmoud Abdelwahab Fathy *ab,
Eslam I. El-Aswarc,
Sabah S. Ibrahimd,
A. M. Ismaelc,
M. M. Yehiac and
M. M. Rashadd
*ab,
Eslam I. El-Aswarc,
Sabah S. Ibrahimd,
A. M. Ismaelc,
M. M. Yehiac and
M. M. Rashadd
aDepartment of Chemistry, Faculty of Science, Ain Shams University, Abbasia, Cairo 11566, Egypt. E-mail: saadsmhassan@yahoo.com; Mahmoud.abdelwahab@sci.asu.edu.eg
bDepartment of Chemistry, School of Engineering Sciences in Chemistry, Biotechnology and Health, KTH Royal Institute of Technology, Teknikringen 30, Stockholm, SE-100 44, Sweden. E-mail: msaye@kth.se
cNational Water Research Center, P.O. Box 74, Shoubra El-Kheima, 13411, Egypt
dCentral Metallurgical Research and Development Institute (CMRDI), P.O. Box 87, Helwan, Cairo, 11422, Egypt
First published on 6th March 2026
Abstract
Persistent organic pesticides (POPs), particularly chlorinated pesticides, represent a critical environmental threat due to their chemical stability, bioaccumulation potential, and resistance to conventional water-treatment methods. Herein, a rapid and cost-effective photocatalytic strategy is developed for the degradation of seventeen structurally diverse POPs in water under ultraviolet (UV) irradiation at ambient temperature. A magnetic heterostructured nanocomposite consisting of activated carbon, derived from sugarcane bagasse, and bentonite, coupled with a magnetite/cerium oxide (Fe3O4/CeO2) nanocomposite, is fabricated via a facile ball-milling approach and tested. The material exhibits a strong light absorption, high adsorption capability, high surface activity, and excellent magnetic recoverability. Comprehensive characterization is performed using ultraviolet-visible (UV-vis) spectroscopy, X-ray diffraction (XRD), scanning electron microscopy (SEM), transmission electron microscopy (TEM), and Brunauer–Emmett–Teller (BET) surface analysis. Under optimized conditions (0.1 g L−1 catalyst, 20 °C, UV 365 nm, 20 min), removal efficiencies exceeding 90% are achieved for δ-benzene hexachloride (δ-BHC), heptachlor, dichlorodiphenyl trichloroethane (DDT), endrine aldehyde, and methoxychlor, while 73.9–86.6% degradation is recorded for α-BHC, β-BHC, γ-BHC, aldrin, heptachlor epoxide, dichlorodiphenyldichloroethylene (DDE), endrin, dieldrin, dichlorodiphenyldichloroethane (DDD), and endosulfan sulfate. The developed AC/Bentonite/Fe3O4/CeO2 nanocomposite demonstrates some key advantages including high adsorption affinity, fast reaction kinetics, broad pollutant applicability, magnetic separability, and significantly shorter treatment time compared with many of those previously reported using cerium-based photocatalysts. These results highlight the potential of this sustainable catalyst system for efficient remediation of POP-contaminated water.
1 Introduction
Persistent organic pollutants (POPs), particularly chlorinated pesticides, constitute one of the most hazardous groups of environmental contaminants. Owing to their strong chemical stability, hydrophobicity, and resistance to chemical, biological, and photolytic degradation, these compounds persist and accumulate in various environmental compartments for long periods. Their lipophilic nature facilitates both bioaccumulation in living organisms and biomagnification along the food chain, resulting in severe ecological and public-health concerns. Chronic exposure to chlorinated POPs has been associated with carcinogenicity, endocrine disruption, reproductive dysfunction, immunotoxicity, neurobehavioral disorders, genotoxicity, and developmental abnormalities.1,2 Due to their tendency to adsorb onto suspended particulate matter, chlorinated POPs frequently occur and accumulate in aquatic environments. Their widespread presence in surface water, groundwater, and wastewater has prompted strict international regulatory actions. Global frameworks such as the Stockholm Convention and European POP regulations mandate strict control measures including elimination, restriction, and progressive reduction of these pollutants.3,4Several physico-chemical and biological methods have been explored to remove chlorinated pesticides from aquatic systems, including adsorption on activated carbon5 and metal–organic frameworks (MOFs),6 electrocoagulation,7 photocatalytic oxidation,8 advanced oxidation technologies (AOTs),9 membrane filtration,10 ionizing radiation,11 and bioremediation using microorganisms.12 Although each method offers specific advantages, challenges persist, such as long processing time, high operational cost, secondary waste formation, incomplete mineralization, and limited efficiency toward highly chlorinated and recalcitrant species. Therefore, there remains a critical need for highly efficient, economical, and environmentally benign technologies capable of rapidly degrading many chlorinated POPs under mild operating conditions.
Semiconductor-based photocatalysis has emerged as one of the most promising and sustainable approaches for degrading persistent organic contaminants. Our previous work demonstrated the efficiency of TiO2/GO/CuFe2O4 nanocomposites for the degradation of chlorinated pesticides.13 Among semiconductor materials, titanium dioxide (TiO2) and cerium dioxide (CeO2) were widely employed due to their stability, cost-effectiveness, and low toxicity.13,14 CeO2 exhibited distinctive physicochemical properties including a tunable band gap (2.6–3.4 eV depending on synthesis method),15 remarkable oxygen storage capacity, high redox activity, excellent thermal stability, and facile Ce4+/Ce3+ transition capability, which enhance photocatalytic oxidation reactions.16,17 However, practical limitations such as unfavorable band-edge positions and rapid recombination of charge carriers restrict its standalone photocatalytic efficiency, necessitating structural modification and composite formation.
Previous studies have been investigated with CeO2-based photocatalysts for the removal of various pollutants (Table 1), including phenols, dyes, pharmaceuticals, and selected pesticides.18–27 Nevertheless, most reported systems suffer from some limitations such as long irradiation periods (30–240 min), high catalyst dosage, narrow target applicability (often one or two analytes), or low mineralization rates. However, the present work offers some significant advantages over many of those reported including the low dose of the catalyst, the short time for the removal process, the removal efficiency, removal selectivity and applicability at ambient temperature.
| Photocatalyst | Organic pollutant | Removal time, min | Removal efficiency, % | Ref. |
|---|---|---|---|---|
| CeO2/TiO2 | Nitrophenol/phenol red | 60 | 97 | 18 |
| PANI/g-C3N4/CeO2 | Diazinon | 180 | 97 | 19 |
| Fe3O4/CeO2 | 4-Chlorophenol | 60 | 100 | 20 |
| CeO2–ZnO | Endosulfan | 130 | 55 | 21 |
| SnO2/CeO2 | Alizarine dye | 30 | 97 | 22 |
| CeO2/Bi2WO6 | Organic dyes | 75 | 54 | 23 |
| AC/Fe3O4/CeO2 | Sulfamethoxazole | 120 | 80 | 24 |
| Ss/ZnO/CeO2 | Pesticides | 240 | 75 | 25 |
| Ag/CeO2 | Organic dyes | 180 | 96 | 26 |
| CeO2 | 2,4-Dichloro-phenoxyacetic acid | 100 | 47 | 27 |
In parallel to CeO2 literature, iron oxide-based photocatalysts—particularly α-Fe2O3 and Fe3O4—have been extensively studied due to their natural abundance, low cost, visible-light absorption, and magnetic separability.28,29 Several Fe-based photocatalyst systems have demonstrated promising performance for wastewater treatment and for degradation of organic pollutants. α-Fe2O3 synthesized by sol–gel autocombustion was used for dye degradation under sunlight30 and Ag–Fe2O3 nanohybrids displayed an enhanced visible-light photocatalytic performance.31 Moreover, Fe-doped hydroxyapatite encapsulated with alginate has been investigated for wastewater treatment applications.32 Fe3O4-based magnetic composites have attracted attention due to their ability to combine photocatalytic degradation with rapid magnetic recovery and recyclability, which is essential for sustainable operation.33 Fe3O4 has also been reported to exhibit a narrower band gap (1.9–2.2 eV),34 which was relevant to its electronic interaction with wide-band-gap semiconductors in composite photocatalysts.
Unlike most reported CeO2 or Fe-based photocatalysts that typically dealt with only one or two model contaminants and required extended irradiation periods, the present work introduced a more efficient ternary magnetic nanocomposite. Comprising activated carbon, produced from sugarcane bagasse, bentonite, magnetite (Fe3O4), and cerium oxide (CeO2). In this design, bio-derived activated carbon provided high porosity and strong adsorption affinity, bentonite enhanced pollutant anchoring and structural stability, Fe3O4 enabled magnetic separation and Fe-mediated redox synergy, while CeO2 provided powerful oxidation capability for degrading chlorinated POPs. The SCBB/Fe3O4/CeO2 heterostructured photocatalyst was synthesized via a facile ball-milling process and evaluated for the rapid degradation of seventeen different chlorinated POP pesticides in water under UV irradiation at ambient temperature. The system demonstrated fast kinetics (≤20 min), high removal efficiency, operational sustainability, magnetic recovery, cost-effectiveness and environmentally sustainable approach for chlorinated POP remediation.
2 Experimental
2.1. Equipment
A NOVA touch® LX2 surface area and pore-size analyzer (Quantachrome Instruments, USA) was used to determine the surface area and pore characteristics of the nanocomposite using the Brunauer–Emmett–Teller (BET) method with N2 adsorption–desorption isotherms at 77 K. UV-vis-NIR diffuse-reflectance spectra were recorded using a Jasco V-570 spectrophotometer (Jasco, Japan). The surface morphology and microstructure were examined using field-emission scanning electron microscopy (FE-SEM, FEI, USA) and transmission electron microscopy (TEM, JEOL 2010F, Japan). Crystal structures were characterized using a Philips PW/103 X-ray diffractometer with Cu-Kα radiation (λ = 1.5406 Å) operated at 40 kV and 40 mA over a 2θ range of 0–70°. Room-temperature magnetic hysteresis measurements were obtained using a vibrating sample magnetometer (VSM, MDK, Germany) over an applied magnetic field of ±10 kOe. Photocatalytic experiments were performed under UV illumination using a 150 W xenon arc lamp (CHF-XQ-500W, Beijing Changtuo Co., Ltd, China) at 365 nm.Chemical analysis of pesticide residues was carried out using an Agilent 7890A GC system coupled to a triple-quadrupole mass spectrometer (5975C) and controlled by MassHunter software. A 7693 autosampler and an HP-5MS Ultra Inert capillary column (30 m × 250 µm × 0.25 µm) were employed. Helium served as the carrier gas at 1.0 mL min−1. The oven program was set as follows: 80 °C for 1 min, ramp to 175 °C at 30 °C min−1 and hold for 4 min, then increased to 225 °C at 3 °C min−1 with a 6-min hold, giving a 20-min effective runtime and a 30-min total analysis time.
2.2. Materials and reagents
Sugarcane bagasse (SCB) was collected from a local market in Cairo, Egypt. Bentonite was obtained from CMB Company (Wadi El-Natroun Industrial Zone, Egypt). Hydrochloric acid (HCl, 33%, ADWIC, Egypt) and sodium hydroxide (NaOH) were used for pH adjustment. A certified organochlorine pesticide (OCP) standard mixture was purchased from AccuStandard Inc. (New Haven, USA). Dichloromethane (98%) and n-hexane (98%) of HPLC grade (LiChrosolv) were supplied by Merck (Darmstadt, Germany). Magnetite (Fe3O4, < 20 µm particle size), cerous nitrate [Ce(NO3)3·6H2O, 99%], hydrogen peroxide (H2O2, 30% w/w), phosphoric acid (H3PO4, 40% w/v), and glycine (≥99%, HPLC grade) were obtained from Sigma-Aldrich.2.3. Preparation of sugarcane-bagasse/bentonite composite (SCBB)
The sugarcane bagasse was thoroughly washed with distilled water to remove surface impurities, then dried at 105 °C overnight to eliminate moisture. The dried biomass was ground and mixed with bentonite and polyurethane in a 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 weight ratio, respectively to obtain (SCBBPU precursor). The mixture was homogenized and heated at 200 °C for 30 min.
1 weight ratio, respectively to obtain (SCBBPU precursor). The mixture was homogenized and heated at 200 °C for 30 min.
A 50 g portion of the dried SCBBPU was soaked in 200 mL of 40% (w/v) H3PO4 for 24 h under anaerobic conditions. The mixture was then heated to 85 °C with stirring for 1 h and subsequently carbonized in a closed system. The temperature was increased to 500 °C at a ramp rate of 4 °C min−1 and held for 3 h. After cooling, the product was washed with distilled water to remove excess acid, dried at 120 °C for 3 h, and finally activated at 650 °C in a muffle furnace. The resulting SCBB composite was stored in airtight glass containers until use.
2.4. Synthesis of cerium oxide (CeO2) nanoparticles
CeO2 nanoparticles were synthesized via a sol–gel auto-combustion route.35 Cerous nitrate was dissolved in distilled water and mixed with a stoichiometric amount of glycine, followed by the addition of 10% (w/v) hydrogen peroxide as an oxidizing agent. The solution was gently heated with continuous stirring until a viscous gel was formed. The resulting gel was dried at 100 °C overnight, then calcined in a muffle furnace at 400 °C for 2 h to obtain crystalline CeO2 nanoparticles.2.5. Preparation of SCBB/Fe3O4/CeO2 nanocomposite
The nanocomposite catalyst was prepared by combining SCBB (1.5 g), magnetite (0.5 g), and CeO2 nanoparticles (3.5 g) with zirconia grinding media (20 g) in a milling chamber. The ball-to-powder weight ratio was maintained at the ratio 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. The mixture was subjected to high-energy ball milling at 550 rpm for 3 h to obtain a homogeneous SCBB/Fe3O4/CeO2 nanocomposite.
1. The mixture was subjected to high-energy ball milling at 550 rpm for 3 h to obtain a homogeneous SCBB/Fe3O4/CeO2 nanocomposite.
2.6. Photocatalytic degradation of chlorinated pesticides
The photocatalytic performance of the prepared nanocomposite was evaluated using a standard organochlorine pesticide mixture containing 17 compounds. In a typical experiment, 0.1 g of the catalyst was dispersed in 50 mL of pesticide test solution, to which 1.0 mL of the standard mixture was added. The pH was adjusted to 3.0 ± 0.1 and the suspension was stirred in the dark for 10 min to ensure uniform dispersion and to allow adsorption–desorption pre-equilibrium between the pesticides and the catalyst surface, in line with commonly adopted photocatalytic degradation protocols.36,37 The reaction mixture was then irradiated under UV light using a xenon arc lamp (365 nm) for 20 min. Photocatalytic experiments were conducted under ambient air without oxygen injection, and continuous magnetic stirring was maintained during irradiation at approximately 600 rpm to ensure homogeneous suspension. The 150 W xenon lamp was positioned vertically above the reactor at a fixed distance of approximately 10 cm, with an irradiance of approximately 50 mW cm−2 at 365 nm. A blank experiment was carried out under identical conditions without the standard pesticide mixture. A photolysis control test (pesticide solution + UV only) was additionally performed under the same irradiation conditions in the absence of the SCBB/Fe3O4/CeO2 nanocomposite to assess the contribution of direct photolysis. In all photocatalytic experiments, the initial pesticide concentrations in the reaction medium were dictated by the certified standard mixture, resulting in analyte levels in the range of 0.5–2.0 mg L−1. Reaction aliquots were collected at t = 20 min and analyzed by GC-MS/MS for quantification.Following irradiation, residual pesticides were extracted by liquid–liquid extraction according to method 6630B, and quantified by GC-MS/MS as previously described.13 The degradation efficiency was calculated using eqn (1):38
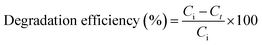 | (1) |
3 Results and discussion
A novel nanocomposite photocatalyst consisting of activated carbon, derived from sugarcane bagasse (SCB), bentonite (B), Fe3O4, and CeO2 was successfully synthesized, characterized, and evaluated for the degradation of chlorinated pesticides.3.1. Characterization of SCBB/Fe3O4/CeO2 nanocomposite
 | ||
| Fig. 1 TEM images of (a) CeO2 nanoparticles39 and (b–d) SCBB/Fe3O4/CeO2 nanocomposite at different magnifications, showing nanoscale particle morphology and uniform distribution of metal oxides within the carbon–bentonite matrix. | ||
 | ||
| Fig. 2 SEM micrographs of (A) raw SCB and (B) SCBB composite illustrating the development of a porous surface structure following chemical activation. | ||
The formation of this porous network was attributed to phosphoric acid activation and bentonite incorporation, which enhance surface area and improve the material's capacity for pesticide adsorption—an essential step prior to photocatalytic degradation.
 | ||
| Fig. 3 XRD patterns of (a) raw sugarcane bagasse (SCB) and activated SCBB composite, (b) CeO2 nanoparticles, and (c) SCBB/Fe3O4/CeO2 nanocomposite. | ||
The crystalline phase of CeO2 nanoparticles was confirmed by the presence of distinct diffraction peaks at 2θ ≈ 28.8°, 32.9°, 47.6°, and 56.6°, which correspond to (111), (200), (220), and (311) planes of the fluorite cubic CeO2 structure, as shown in Fig. 3b and consistent with JCPDS Card No. 34-0394.41 In the SCBB/Fe3O4/CeO2 nanocomposite (Fig. 3c), characteristic peaks of Fe3O4 at 2θ ≈ 30.2°, 35.5°, 43.3°, 57.2°, and 62.8°, along with CeO2 reflections, were developed and clearly observed, confirming their successful incorporation. The Fe3O4 reflections appeared at (220), (311), (400), (511), and (440) planes, were in a good agreement with JCPDS Card No. 19-0629,42 confirming the presence of the magnetite phase in the prepared composite. Additional reflections attributed to carbon and aluminosilicate phases of bentonite further supported the formation of a multi-component heterostructure.
These results verified the coexistence of CeO2, Fe3O4, carbon, and bentonite phases within the composite, demonstrating successful synthesis without compromising the crystalline integrity of the incorporated metal oxides. The clear peak definition also indicated nanoscale crystallite dimensions and maintained structural stability, supporting the photocatalytic activity and durability of the composite.
 | ||
| Fig. 4 UV-vis-NIR absorption spectrum of CeO2 nanoparticles recorded in the wavelength range of 200–2000 nm. | ||
In addition, a slight absorption tail extending toward the visible region and associated with defect-related sub-bandgap states, was commonly linked to oxygen vacancies and Ce3+ centres in ceria-based systems.44 Such defects are known to play a key role in enhancing photocatalytic performance by facilitating charge separation and broadening the light-absorption range.39 These optical features further supported the applicability of CeO2 in photocatalytic systems operating under UV illumination, and indicating the possible contribution of defect states to light absorption.
 | ||
| Fig. 5 N2 adsorption–desorption analysis of SCBB/Fe3O4/CeO2 nanocomposite: (a) adsorption–desorption isotherm, (b) BJH pore-size distribution, and (c) BET linear plot. | ||
| Parameter | Value ± SD |
|---|---|
| BETanalysis | |
| Vm (cm3 STP g−1) | 1.5034 ± 0.0752 |
| BET surface area, as (m2 g−1) | 6.5435 ± 0.3272 |
| C constant | 86.539 ± 4.327 |
| Total pore volume, Vt (cm3 g−1) | 0.04229 ± 0.00211 |
| Average pore diameter (nm) | 25.853 ± 1.293 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
|
| BJH analysis | |
| Pore volume, Vp (cm3 g−1) | 0.09815 ± 0.00491 |
| Peak pore diameter (nm) | 1.0454 ± 0.0523 |
| Pore surface area (m2 g−1) | 8.5861 ± 0.4293 |
| Average pore diameter (nm) | 45.726 ± 2.286 |
| Median pore diameter (nm) | 85.672 ± 4.284 |
The BJH pore-size distribution (Fig. 5b) further confirmed the predominantly mesoporous nature of the composite, with an average pore diameter of ≈45.7 nm. Such mesoporous architecture enhanced the adsorption and diffusion of pesticide molecules toward active sites, thereby supporting efficient photocatalytic degradation. The combined effect of carbonaceous porosity and bentonite templating provided a favourable textural framework for improved pollutant uptake and photocatalytic activity.
 | ||
| Fig. 6 VSM hysteresis loop of the SCBB/Fe3O4/CeO2 nanocomposite measured at room temperature within an applied magnetic field of ±10 kOe. | ||
Despite the modest Ms, the nanocomposite responded rapidly to an external magnet, allowing complete separation from aqueous suspension within ∼15–20 s. This rapid magnetic recovery eliminated the need for centrifugation or filtration during catalyst harvesting and facilitated solid-phase recycling, offering a practical advantage for sustainable water-treatment applications.
3.2. Photodegradation of chlorinated pesticides using SCBB/Fe3O4/CeO2 nanocomposite
The efficiency of the synthesized SCBB/Fe3O4/CeO2 nanocomposite toward the removal and photocatalytic degradation of seventeen chlorinated pesticides (0.5–2.0 mg L−1) was evaluated under dark and UV-irradiation conditions. A recent review reported that the concentrations of organochlorine pesticides in rivers worldwide during 2014–2024 ranged from 0.4 to 37 × 105 ng L−1 (i.e., up to 3.7 mg L−1), depending on location, pollution sources, and seasonal variation.49 In addition, concentrations as low as 0.4 µg L−1 have been detected in certain local wastewater samples.50 Therefore, the selected concentration range (0.5–2.0 mg L−1) falls within the upper levels reported for highly contaminated river systems and allows reliable analytical quantification and performance evaluation.Preliminary adsorption–equilibrium tests indicated that extending the dark stirring period from 10 to up to 30 min did not lead to noticeable changes in removal efficiencies, suggesting that the adsorption–desorption equilibrium was reached rapidly under the applied conditions. As presented in Table 3, the catalyst exhibited a noticeable removal capability in the absence of light, where adsorptive removal efficiencies ranged from 13.5 ± 0.6% to 75.2 ± 3.9% after 20 min. This behaviour highlighted the strong affinity of the sugarcane-bagasse-derived activated carbon, in combination with bentonite, toward chlorinated pesticides due to the presence of accessible adsorption sites and surface functional groups,51 which collectively facilitated pre-concentration of the pollutants at the catalyst surface prior to photocatalytic activation.
| Pesticide | Retention time (Rt), min | Removal % (dark) | Photocatalytic degradation, % (UV) |
|---|---|---|---|
| α-BHC | 8.96 | 40.3 ± 2.1 | 78.8 ± 3.4 |
| β-BHC | 9.87 | 39.1 ± 1.9 | 79.9 ± 2.8 |
| γ-BHC | 10.11 | 36.2 ± 1.7 | 83.4 ± 3.1 |
| δ-BHC | 11.02 | 40.8 ± 2.3 | 74.2 ± 2.6 |
| Heptachlor | 13.03 | 37.9 ± 1.5 | 86.1 ± 3.7 |
| Aldrin | 14.60 | 60.9 ± 3.4 | 84.6 ± 2.5 |
| Heptachlor epoxide | 16.58 | 29.2 ± 1.2 | 57.3 ± 1.8 |
| Endosulfan I | 18.42 | 75.2 ± 3.9 | 97.3 ± 2.9 |
| Dieldrin | 19.79 | 51.9 ± 2.4 | 93.0 ± 3.6 |
| DDE | 19.97 | 35.7 ± 1.6 | 77.9 ± 2.3 |
| Endrin | 20.92 | 68.5 ± 3.1 | 92.3 ± 2.7 |
| Endosulfan II | 21.46 | 66.5 ± 2.8 | 60.0 ± 1.9 |
| DDD | 22.17 | 56.0 ± 2.0 | 86.6 ± 3.2 |
| Endrin aldehyde | 22.52 | 25.2 ± 1.1 | 78.9 ± 2.4 |
| Endosulfan sulfate | 23.83 | 13.5 ± 0.6 | 90.5 ± 3.8 |
| DDT | 24.21 | 36.2 ± 1.4 | 73.9 ± 2.6 |
| Methoxychlor | 28.05 | 26.6 ± 1.3 | 93.7 ± 4.1 |
It should be noted that the degradation values presented in Table 3 represent end-point measurements at t = 20 min, which was selected based on preliminary GC-MS/MS monitoring. Extending the irradiation beyond this interval did not substantially change the residual pesticide profiles under the applied conditions.
Upon exposure to UV irradiation (365 nm), the composite catalyst demonstrated a significant enhancement in performance, achieving photocatalytic degradation efficiencies ranging from 57.3 ± 1.8% (for heptachlor epoxide) to 97.7 ± 4.1% (for methoxychlor) under the same conditions. Many of the tested chlorinated pesticides were decomposed with efficiencies > 90% (endosulfan-I 97.3 ± 2.9%, dieldrin, 93.0 ± 3.6%, endrin, 92.3 ± 2.7%, endosulfan sulfate 90.5 ± 3.8%). This pronounced increase in the decomposition under UV illumination confirmed that the SCBB/Fe3O4/CeO2 composite operated via a dual-stage mechanism, where the initial adsorption enriched pesticide molecules on the catalyst surface, followed by rapid photodegradation through electron–hole-driven oxidative pathways.
The synergistic contribution of the nanocomposite components was central to this enhanced performance. The activated carbon matrix provided extensive adsorption sites,52,53 whereas bentonite enhanced structural stability and surface interaction.51,54 Fe3O4 enabled magnetic recovery and participated in redox mediation, which not only facilitated catalyst separation but also allowed the material to be reused while maintaining its photocatalytic performance for up to four consecutive cycles. On the other hand, CeO2 facilitated efficient charge separation and promoted reactive oxygen species (ROS) generation through Ce4+/Ce3+ cycling. Such synergy accelerated radical-mediated oxidation and minimized charge recombination. This behaviour aligned with a previously reported mechanisms where adsorption-driven enrichment enhanced photocatalytic decomposition efficiency in hybrid carbon-ceria materials.55
GC-MS chromatograms (Fig. 7) further validated the photocatalytic degradation process, showing a substantial reduction or disappearance of characteristic pesticide peaks following treatment, while exposure to the UV-only (photolysis control) did not lead to measurable attenuation of pesticide peaks, confirming that direct photolysis was negligible under the applied irradiation conditions. This negligible photolysis was consistent with the classification of these chlorinated pesticides as persistent organic pollutants (POPs), which are known for their chemical and photochemical stability due to extensive chlorination and limited absorption in the UVA range.
3.3. Comparison of photocatalytic degradation performance with other nanocomposites
Although various photocatalysts have been previously investigated for pesticide removal, comprehensive degradation studies covering a wide range of structurally different chlorinated pesticides remained scarce; most reported works frequently focus on one or two compounds only. Accordingly, a comparative photocatalytic evaluation was conducted for the degradation of seventeen common chlorinated pesticides using three nanocomposite catalysts synthesized and used in our laboratory under identical UV-irradiation conditions for 20 min. The performance of the present SCBB/Fe3O4/CeO2 nanocomposite was benchmarked against TiO2/GO/CuFe2O4 (ref. 13) and Mo-TiO2/GO/Fe3O4.56 The results, presented in Table 4, revealed that each catalyst exhibited a preferential degradation pattern toward specific pesticide classes.| Pesticide | Retention time (Rt), min | Photocatalytic degradation % (UV) | ||
|---|---|---|---|---|
| TiO2/GO/CuFe2O4 (ref. 13) | Mo-TiO2/GO/Fe3O4 (ref. 56) | SCBB/Fe3O4/CeO2 [present work] | ||
| α-BHC | 8.96 | 92.2 | 83.8 | 78.8 ± 3.4 |
| β-BHC | 9.87 | 93.4 | 80.9 | 79.9 ± 2.8 |
| γ-BHC | 10.11 | 85.4 | 82.4 | 83.4 ± 3.1 |
| δ-BHC | 11.02 | 70.6 | 90.7 | 74.2 ± 2.6 |
| Heptachlor | 13.03 | 71.7 | 90.7 | 86.1 ± 3.7 |
| Aldrin | 14.60 | 95.9 | 81.5 | 84.6 ± 2.5 |
| Heptachlor epoxide | 16.58 | 89.2 | 82.8 | 57.3 ± 1.8 |
| Endosulfan I | 18.42 | 86.5 | 93.5 | 97.3 ± 2.9 |
| Dieldrin | 19.79 | 84.8 | 69.9 | 93.0 ± 3.6 |
| DDE | 19.97 | 96.5 | 79.9 | 77.9 ± 2.3 |
| Endrin | 20.92 | 89.2 | 94.3 | 92.3 ± 2.7 |
| Endosulfan II | 21.46 | 79.1 | 55.6 | 60.0 ± 1.9 |
| DDD | 22.17 | 76.1 | 83.6 | 86.6 ± 3.2 |
| Endrin aldehyde | 22.52 | 70.4 | 58.4 | 78.9 ± 2.4 |
| Endosulfan sulfate | 23.83 | 32.4 | 88.1 | 90.5 ± 3.8 |
| DDT | 24.21 | 59.6 | 89.1 | 73.9 ± 2.6 |
| Methoxychlor | 28.05 | 42.7 | 93.6 | 93.7 ± 4.1 |
It can be seen that TiO2/GO/CuFe2O4 nanocomposite demonstrated superior activity toward α-BHC, β-BHC, γ-BHC, aldrin, heptachlor epoxide, endosulfan II, and DDE, giving degradation efficiencies in the range of 79.1–96.5%. In contrast, Mo-TiO2/GO/Fe3O4 system showed enhanced degradation of δ-BHC, heptachlor, endrin, and DDT, with efficiencies ranging between 89.1–94.3%. The present suggested nanocomposite catalyst (SCBB/Fe3O4/CeO2) exhibited the highest efficiency (78.9–97.3%) toward endosulfan I, dieldrin, DDD, endrin aldehyde, endosulfan sulfate, and methoxychlor.
The preferential degradation patterns observed in Table 4 can be rationalized by considering structure-performance correlations. For instance, highly hydrophobic and heavily chlorinated pesticides such as endosulfan I, dieldrin and DDD tend to exhibit stronger affinity toward carbonaceous and clay-containing systems due to non-polar and van der Waals interactions, promoting pre-adsorption and facilitating subsequent photocatalytic conversion. In contrast, TiO2-based photocatalysts, which possess more hydrophilic surfaces and different band edge positions, have been reported to preferentially degrade less hydrophobic or sterically smaller organochlorines. Therefore, both the physicochemical properties of the pesticides (e.g. hydrophobicity, molecular size and degree of chlorination) and the surface chemistry and band structure of the catalysts collectively contributed to the observed differences in degradation selectivity among the three systems.
3.4. Mechanism of action of SCBB/Fe3O4/CeO2 nanocomposite
The photocatalytic activity of the SCBB/Fe3O4/CeO2 nanocomposite toward chlorinated pesticides under UV irradiation proceeded through a combined photocatalytic and Fenton-like pathway, as illustrated in Scheme 1. Upon UV exposure, electrons were promoted from the valence band (VB) to the conduction band (CB), leaving their corresponding holes in the VB. The generated charge carriers migrated efficiently between Fe3O4 and CeO2 due to their favourable band potentials, thereby suppressing electron–hole recombination. The holes reacted with water molecules or surface hydroxyl groups to generate hydroxyl radicals (˙OH),57 while the electrons reduced dissolved oxygen to superoxide radicals (O2˙−). Both species initiated oxidation and cleavage of chlorinated pesticide molecules. O2˙− can further react with protons to generate additional ˙OH, intensifying the oxidative environment.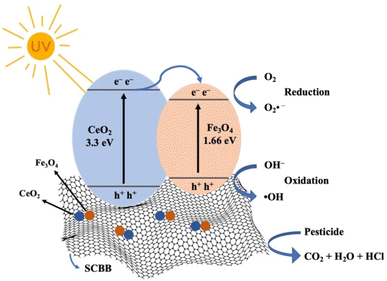 | ||
| Scheme 1 Schematic mechanism for photocatalytic degradation of chlorinated pesticides using SCBB/Fe3O4/CeO2 nanocomposite under UV irradiation. | ||
Simultaneously, Fe2+/Fe3+ species in the Fe3O4 phase and Ce4+/Ce3+ cycling in CeO2 (E° Ce4+/Ce3+ = 1.44 V vs. E° Fe2+/Fe3+ = 0.77 V) promoted Fenton-like reactions in which Fe2+ reacted with H2O2 to produce surface-bound hydroxyl radicals (˙OH_ads).58 CeO2 facilitated regeneration of Fe2+ from Fe3+, sustaining the catalytic cycle. A fraction of Fe ions may also diffuse into solution, contributing to homogeneous Fenton reactions that generate free hydroxyl radicals (˙OH_free). These reactive oxygen species mineralized pesticide molecules into CO2, H2O, and HCl.
4 Conclusion
In this study, a cost-effective and sustainable magnetic AC/Bentonite/Fe3O4/CeO2 nanocomposite was successfully synthesized from sugarcane bagasse and evaluated as a multifunctional adsorbent-photocatalyst for the removal of seventeen persistent chlorinated pesticides from water. Comprehensive characterization confirmed the formation of a mesoporous, heterostructured material with enhanced surface properties and efficient light-absorption capability. Under mild conditions (0.1 g L−1 catalyst, UV 365 nm, ambient temperature), the nanocomposite demonstrated rapid degradation performance, achieving up to ∼97% removal within only 20 min and showing significant adsorption activity in dark conditions. The superior efficiency can be attributed to the synergistic contribution of activated carbon, bentonite, Fe3O4, and CeO2, enabling strong pollutant adsorption, efficient charge separation, and enhanced radical generation via Ce4+/Ce3+ and Fe3+/Fe2+ redox cycling. Compared to previously reported systems, the proposed catalyst offered faster kinetics, broader applicability, and magnetic recoverability. These characteristics demonstrated the strong potential of the suggested catalyst for the treatment of POPs-contaminated water.Ethical approval
Ethical approval was obtained from the Ethical Committee (EC) of the Faculty of Science, Ain Shams University, Cairo, Egypt, for the experimental procedures conducted in this study.Author contributions
Saad S.M. Hassan: supervision, conceptualization, validation, writing reviewing & editing. Mahmoud Abdelwahab Fathy: conceptualization, resources, methodology, investigation, formal analysis, data curation, visualization, validation, writing – original draft, writing – reviewing & editing. Eslam I. El-Aswar: resources, investigation. Sabah S. Ibrahim: resources, investigation. A. M. Ismael: investigation, formal analysis, visualization. M. M. Yehia: resources, investigation. M. M. Rashad: supervision, conceptualization, validation.Conflicts of interest
The authors declare no competing interests.Data availability
Data will be made available on request.Acknowledgements
The authors declare no funding or financial support was received for the preparation or conduct of this study, and no organizations were involved in the submission of this work.References
- O. M. Alharbi, R. A. Khattab and I. Ali, Health and environmental effects of persistent organic pollutants, J. Mol. Liq., 2018, 263, 442–453 CrossRef CAS.
- K. Kumari, S. Balbudhe and A. Singh, Effects of Persistent Organic Pollutants (POPs) on Human Health, Persistent Organic Pollutants, CRC Press, 2021, pp. 91–122 Search PubMed.
- H. Fiedler, R. Kallenborn, J. D. Boer and L. K. Sydnes, The Stockholm convention: a tool for the global regulation of persistent organic pollutants, Chem. Int., 2019, 41, 4–11 CAS.
- E. Regulation, 1021 of the European Parliament and of the Council of 20 June 2019 on Persistent Organic Pollutants, European Union, 2019 Search PubMed.
- J. O. Ighalo, P.-S. Yap, K. O. Iwuozor, C. O. Aniagor, T. Liu, K. Dulta, F. U. Iwuchukwu and S. Rangabhashiyam, Adsorption of persistent organic pollutants (POPs) from the aqueous environment by nano-adsorbents: A review, Environ. Res., 2022, 212, 113123 CrossRef CAS PubMed.
- S. Naghdi, M. M. Shahrestani, M. Zendehbad, H. Djahaniani, H. Kazemian and D. Eder, Recent advances in application of metal-organic frameworks (MOFs) as adsorbent and catalyst in removal of persistent organic pollutants (POPs), J. Hazard. Mater., 2023, 442, 130127 CrossRef CAS PubMed.
- F. E. Titchou, H. Zazou, H. Afanga, J. El Gaayda, R. Ait Akbour and M. Hamdani, Removal of persistent organic pollutants (POPs) from water and wastewater by adsorption and electrocoagulation process, Groundw. Sustain. Dev., 2021, 13, 100575 CrossRef.
- P. Pascariu, C. Cojocaru, V. Ciornea, C. Romanitan and A. B. Serban, Visible light-responsive Ce-doped ZnO ceramic nanostructures as effective photocatalysts for removal of persistent organic pollutants from contaminated waters, Mater. Today Sustain., 2024, 26, 100719 Search PubMed.
- I. L. Clímaco Cunha, P. G. Machado, C. de Oliveira Ribeiro and L. Kulay, Bibliometric analysis of Advanced Oxidation Processes studies with a focus on Life Cycle Assessment and Costs, Environ. Sci. Pollut. Res., 2024, 31, 22319–22338 CrossRef PubMed.
- S. A. Mhlongo, L. L. Sibali and P. P. Ndibewu, Some aspects of the synthesis, characterization and modification of poly (ether) sulfone polymeric membrane for removal of persistent organic pollutants in wastewater samples, J. Water Chem. Technol., 2023, 45, 388–401 CrossRef.
- M. Trojanowicz, Removal of persistent organic pollutants (POPs) from waters and wastewaters by the use of ionizing radiation, Sci. Total Environ., 2020, 718, 134425 CrossRef CAS PubMed.
- C. Mateescu, E.-M. Lungulescu and N.-O. Nicula, Effectiveness of Biological Approaches for Removing Persistent Organic Pollutants from Wastewater: A Mini-Review, Microorganisms, 2024, 12, 1632 CrossRef CAS PubMed.
- A. Ismael, A. El-Shazly, S. Gaber, M. Rashad, A. Kamel and S. S. M. Hassan, Novel TiO2/GO/CuFe2O4 nanocomposite: a magnetic, reusable and visible-light-driven photocatalyst for efficient photocatalytic removal of chlorinated pesticides from wastewater, RSC Adv., 2020, 10, 34806–34814 RSC.
- T. Montini, M. Melchionna, M. Monai and P. Fornasiero, Fundamentals and catalytic applications of CeO2-based materials, Chem. Rev., 2016, 116, 5987–6041 CrossRef CAS PubMed.
- D. Sharma and B. Mehta, Nanostructured TiO2 thin films sensitized by CeO2 as an inexpensive photoanode for enhanced photoactivity of water oxidation, J. Alloys Compd., 2018, 749, 329–335 CrossRef CAS.
- E. Kusmierek, A CeO2 semiconductor as a photocatalytic and photoelectrocatalytic material for the remediation of pollutants in industrial wastewater: a review, Catalysts, 2020, 10, 1435 CrossRef CAS.
- A. Fauzi, A. Jalil, N. Hassan, F. Aziz, M. Azami, I. Hussain, R. Saravanan and D.-V. Vo, A critical review on relationship of CeO2-based photocatalyst towards mechanistic degradation of organic pollutant, Chemosphere, 2022, 286, 131651 CrossRef CAS PubMed.
- A. Ahlawat, T. K. Dhiman, P. R. Solanki and P. S. Rana, Enhanced photocatalytic degradation of p-nitrophenol and phenol red through synergistic effects of a CeO2-TiO2 nanocomposite, Catal., 2022, 2, 1–13 Search PubMed.
- A. Hussen, PANI/g-C3N4/CeO2 Nanocomposite for Photodegradation of Diazinon in Aqueous Solution, Middle East J. appl. Sci. Technol., 2021 Search PubMed.
- L. Xu and J. Wang, Magnetic nanoscaled Fe3O4/CeO2 composite as an efficient Fenton-like heterogeneous catalyst for degradation of 4-chlorophenol, Environ. Sci. Technol., 2012, 46, 10145–10153 CrossRef CAS PubMed.
- A. Imtiaz, M. A. Farrukh and H. Latif, pH-dependent synthesis of CeO2-ZnO nanocomposite for enhanced environmental remediation of endosulfan and dimethoate pesticides, J. Mol. Struct., 2024, 1300, 137174 CrossRef CAS.
- S. S. M. Hassan, A. H. Kamel, A. A. Hassan, A. E.-G. E. Amr, H. A. El-Naby and E. A. Elsayed, A SnO2/CeO2 nano-composite catalyst for alizarin dye removal from aqueous solutions, Nanomaterials, 2020, 10, 254 CrossRef CAS PubMed.
- S. Issarapanacheewin, K. Wetchakun, S. Phanichphant, W. Kangwansupamonkon and N. Wetchakun, Photodegradation of organic dyes by CeO2/Bi2WO6 nanocomposite and its physicochemical properties investigation, Ceram. Int., 2016, 42, 16007–16016 CrossRef CAS.
- J. Akhtar, N. S. Amin and A. Aris, Combined adsorption and catalytic ozonation for removal of sulfamethoxazole using Fe2O3/CeO2 loaded activated carbon, Chem. Eng. J., 2011, 170, 136–144 CrossRef CAS.
- X. Wang, L. Huang, N. Yuan, P. Huang, X. Du and X. Lu, Facile fabrication of a novel SPME fiber based on silicone sealant/hollow ZnO@CeO2 composite with super-hydrophobicity for the enhanced capture of pesticides from water, Microchem. J., 2022, 183, 108118 Search PubMed.
- G. Murugadoss, D. D. Kumar, M. R. Kumar, N. Venkatesh and P. Sakthivel, Silver decorated CeO2 nanoparticles for rapid photocatalytic degradation of textile rose bengal dye, Sci. Rep., 2021, 11, 1080 CrossRef CAS PubMed.
- E. A. A. Elhussein, S. Şahin and Ş. S. Bayazit, Preparation of CeO2 nanofibers derived from Ce-BTC metal-organic frameworks and its application on pesticide adsorption, J. Mol. Liq., 2018, 255, 10–17 CrossRef.
- A. Bagade, P. Nagwade, A. Nagawade, S. Thopate, V. Pandit and S. Pund, Impact of Mg2+ substitution on structural, magnetic and optical properties of Cu-Cd ferrites, Mater. Today: Proc., 2022, 53, 144–152 CAS.
- S. S. M. Hassan, H. H. El-Shalakany, M. A. Fathy and A. H. Kamel, A magnetic macroporous α-Fe2O3/Mn2O3 nanocomposite as an efficient adsorbent for simple and rapid removal of Pb (II) from wastewater and electronic waste leachate, Environ. Sci. Pollut. Res., 2024, 31, 65648–65660 Search PubMed.
- V. R. U. Pandit, G. K. P. Jadhav, V. M. S. Jawale, R. Dubepatil, R. Gurao and D. J. Late, Synthesis and characterization of micro-/nano-α-Fe2O3 for photocatalytic dye degradation, RSC Adv., 2024, 14, 29099–29105 RSC.
- A. AlZubaidi, V. Pandit, V. Jawale, M. Gupta and D. Late, Ag-Fe2O3 nanohybrids for photocatalytic degradation and antibacterial activity, Front. Catal., 2025, 5, 1655324 CrossRef.
- A. Grouli, Y. Bachra, F. Damiri, V. U. Pandit and M. Berrada, Removal of Pollutants from Wastewater Using Fe-Doped Hydroxyapatite Encapsulated with Alginate, Biointerface Res. Appl. Chem., 2023, 13, 438 CAS.
- R. Chalasani and S. Vasudevan, Cyclodextrin-functionalized Fe3O4@TiO2: reusable, magnetic nanoparticles for photocatalytic degradation of endocrine-disrupting chemicals in water supplies, ACS Nano, 2013, 7, 4093–4104 CrossRef CAS PubMed.
- S. Delice, M. Isik and N. Gasanly, Temperature-dependent tuning of band gap of Fe3O4 nanoparticles for optoelectronic applications, Chem. Phys. Lett., 2024, 840, 141139 CrossRef CAS.
- A. Younis, D. Chu and S. Li, Cerium oxide nanostructures and their applications, Funct. Nanomater., 2016, 3, 53–68 Search PubMed.
- D. Bhuyan, S. S. Arbuj and L. Saikia, Template-free synthesis of Fe3O4 nanorod bundles and their highly efficient peroxidase mimetic activity for the degradation of organic dye pollutants with H2O2, New J. Chem., 2015, 39, 7759–7762 RSC.
- V. U. Pandit, S. S. Arbuj, Y. B. Pandit, S. D. Naik, S. B. Rane, U. P. Mulik, S. W. Gosavi and B. B. Kale, Solar light driven dye degradation using novel organo–inorganic (6,13-pentacenequinone/TiO2) nanocomposite, RSC Adv., 2015, 5, 10326–10331 RSC.
- M. A. Fathy, A. H. Kamel and S. S. M. Hassan, Novel magnetic nickel ferrite nanoparticles modified with poly (aniline-co-o-toluidine) for the removal of hazardous 2, 4-dichlorophenol pollutant from aqueous solutions, RSC Adv., 2022, 12, 7433–7445 RSC.
- F. Hossam, A. M. Elseman, M. Rasly, R. Mahani, S. Sayed and M. Rashad, Observation of structural, optical, electrical and magnetic properties of ternary copper-doped CeO2/GO/SrTiO3 nanocomposites, J. Mater. Sci.: Mater. Electron., 2023, 34, 1513 CrossRef CAS.
- E. de Morais Teixeira, T. J. Bondancia, K. B. R. Teodoro, A. C. Corrêa, J. M. Marconcini and L. H. C. Mattoso, Sugarcane bagasse whiskers: extraction and characterizations, Ind. Crops Prod., 2011, 33, 63–66 CrossRef CAS.
- A. Xie, W. Liu, S. Wang, X. Liu, J. Zhang and Y. Yang, Template-free hydrothermal synthesis and CO oxidation properties of flower-like CeO2 nanostructures, Mater. Res. Bull., 2014, 59, 18–24 CrossRef CAS.
- W. Zhao, J. Chen, X. Chang, S. Guo, C. Srinivasakannan, G. Chen and J. Peng, Effect of microwave irradiation on selective heating behavior and magnetic separation characteristics of Panzhihua ilmenite, Appl. Surf. Sci., 2014, 300, 171–177 CrossRef CAS.
- M. Farahmandjou, M. Zarinkamar and T. Firoozabadi, Synthesis of Cerium Oxide (CeO2) nanoparticles using simple CO-precipitation method, Rev. Mex. Fis., 2016, 62, 496–499 CAS.
- J. Calvache-Muñoz, F. A. Prado, L. Tirado, L. C. Daza-Gomez, G. Cuervo-Ochoa, H. L. Calambas and J. E. Rodríguez-Páez, Structural and optical properties of CeO2 nanoparticles synthesized by modified polymer complex method, J. Inorg. Organomet. Polym. Mater., 2019, 29, 813–826 CrossRef.
- M. Thommes, K. Kaneko, A. V. Neimark, J. P. Olivier, F. Rodriguez-Reinoso, J. Rouquerol and K. S. Sing, Physisorption of gases, with special reference to the evaluation of surface area and pore size distribution (IUPAC Technical Report), Pure Appl. Chem., 2015, 87, 1051–1069 CrossRef CAS.
- X. Cao, Z. Meng, E. Song, X. Sun, X. Hu, W. Li, Z. Liu, S. Gao and B. Song, Co-adsorption capabilities and mechanisms of bentonite enhanced sludge biochar for de-risking norfloxacin and Cu2+ contaminated water, Chemosphere, 2022, 299, 134414 CrossRef CAS PubMed.
- J. Xu, H. Lin, Y. Su and S. Tang, An Experimental Investigation on the Barrier Performance of Complex-Modified Bentonite, Appl. Sci., 2025, 16, 299 CrossRef.
- J. Liu, J. Cui, T. Zhao, S. Fan, C. Zhang, Q. Hu and X. Hou, Fe3O4-CeO2 loaded on modified activated carbon as efficient heterogeneous catalyst, Colloids Surf., A, 2019, 565, 59–69 CrossRef CAS.
- A. López-Benítez, A. Guevara-Lara, M. A. Domínguez-Crespo, J. A. Andraca-Adame and A. M. Torres-Huerta, Concentrations of organochlorine, organophosphorus, and pyrethroid pesticides in rivers worldwide (2014–2024): A review, Sustainability, 2024, 16, 8066 CrossRef.
- H. El-Mekkawi, M. Diab, M. Zaki and A. Hassan, Determination of chlorinated organic pesticide residues in water, sediments, and fish from private fish farms at Abbassa and Sahl Al-Husainia, Shakia Governorate, Aust. J. Basic Appl. Sci., 2009, 3(4), 4376–4383 CAS.
- X.-L. Gong, H.-Q. Lu, K. Li and W. Li, Effective adsorption of crystal violet dye on sugarcane bagasse–bentonite/sodium alginate composite aerogel: Characterisation, experiments, and advanced modelling, Sep. Purif. Technol., 2022, 286, 120478 CrossRef CAS.
- S. S. M. Hassan, N. R. Mohamed, M. M. Saad, A. M. Salem, Y. H. Ibrahim, A. A. Elshakour and M. A. Fathy, A novel non-woven fabric sandwich filter with activated carbon/polypyrrole nanocomposite for the removal of CO, SO2 and NOx emitted from gasoline engines, Fuel, 2025, 401, 135838 CrossRef CAS.
- S. S. M. Hassan, N. R. Mohamed, M. M. Saad, Y. H. Ibrahim, A. A. Elshakour and M. A. Fathy, Eco-Friendly Removal and IoT-Based Monitoring of CO2 Emissions Released from Gasoline Engines Using a Novel Compact Nomex/Activated Carbon Sandwich Filter, Polymers, 2025, 17, 1447 CrossRef CAS PubMed.
- A. K. Singh, V. U. Pandit, S. L. Sonawane, Bio-based raw materials for preparation of carbon nanostructures, Bio-derived Carbon Nanostructures, Elsevier, 2024, pp. 25–63 Search PubMed.
- M. Šťastný, D. Bavol, J. Tolasz, P. Bezdička, J. Čundrle, M. Kormunda, I. Dimitrov, P. Janoš, K. Kirakci and J. Henych, Interfacial behavior of ceria grown on graphene oxide and its use for hydrolytic and photocatalytic decomposition of bisphenols A, S, and F, Environ. Sci.: Nano, 2025, 12, 502–513 RSC.
- S. S. M. Hassan, A. El-Shazly, A. Ismael, M. Yehia, A. Kamel and M. Rashad, Enhanced photocatalytic degradation of chlorinated pesticides and polychlorinated biphenyls using Mo–TiO2/GO/MS nanocomposite, Opt. Mater., 2023, 142, 114084 CrossRef CAS.
- Y. Nosaka and A. Nosaka, Understanding hydroxyl radical (•OH) generation processes in photocatalysis, ACS Energy Lett., 2016, 1, 356–359 CrossRef CAS.
- N. S. Rizi, A. Shahzeydi, M. Ghiaci and L. Zhang, Photocatalytic degradation of cationic and anionic organic pollutants in water via Fe-g-C3N4/CF as a macroscopic photo-Fenton catalyst under visible light irradiation, J. Environ. Chem. Eng., 2020, 8, 104219 CrossRef.
| This journal is © The Royal Society of Chemistry 2026 |

