 Open Access Article
Open Access ArticleVisible-light-responsive Fe2O3–TiO2–carbon dot nanocomposite coatings for enhanced biofouling mitigation in aquaculture cage nets
R. Gopikaab and
P. Muhamed Ashraf *a
*a
aICAR-Central Institute of Fisheries Technology, Matsyapuri PO, Cochin 682 029, Kerala, India. E-mail: ashrafp2008@gmail.com; Fax: +91 484 2668212; Tel: +91 484 2412300 Tel: +91 9746236477
bCochin University of Science and Technology, Cochin 682 022, Kerala, India
First published on 30th January 2026
Abstract
A photocatalytic antifouling coating was designed for aquaculture cage nets, as biofouling is a persistent challenge in aquaculture that substantially reduces operational efficiency and compromises the economic performance of the systems. A nanostructured composite was developed using nano-titanium dioxide (TiO2), iron(III) oxide (Fe2O3), and carbon dots (CDs). The antifouling efficacy of the composite was evaluated by applying it onto a (3-glycidyloxypropyl)trimethoxysilane (silane)-pretreated polyethylene substrate, followed by immersion in natural estuarine waters. This ternary photocatalytic composite, formed by the interaction between the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond of CDs and inorganic oxides, exhibited a reduced band gap from 4 eV to 1.7 eV by suppressing electron–hole recombination. The uniform distribution in the composite was evidenced by XRD, the characteristic peaks of TiO2 and Fe2O3 suggested nanoscale crystallinity, and the 2θ = 21° peak suggested the presence of low-crystalline CDs in the matrix, which was confirmed by transmission electron microscopy. The nanocomposite uniformly coated over silane-pretreated polyethylene exhibited strong interfacial bonding between CD aromatic moieties and glycidyloxy C–O–C groups. Nets coated with a 0.05% FeTiCD nanocomposite exhibited excellent biofouling resistance in a six-month field trial. Fe(III) doping introduced mid-gap states, enabling efficient electron transfer to CDs and sustained reactive oxygen species (ROS) production, thereby disrupting the attachment of biofouling organisms. This nanocomposite offers a scalable, low-impact antifouling solution that supports more sustainable aquaculture operations.
C bond of CDs and inorganic oxides, exhibited a reduced band gap from 4 eV to 1.7 eV by suppressing electron–hole recombination. The uniform distribution in the composite was evidenced by XRD, the characteristic peaks of TiO2 and Fe2O3 suggested nanoscale crystallinity, and the 2θ = 21° peak suggested the presence of low-crystalline CDs in the matrix, which was confirmed by transmission electron microscopy. The nanocomposite uniformly coated over silane-pretreated polyethylene exhibited strong interfacial bonding between CD aromatic moieties and glycidyloxy C–O–C groups. Nets coated with a 0.05% FeTiCD nanocomposite exhibited excellent biofouling resistance in a six-month field trial. Fe(III) doping introduced mid-gap states, enabling efficient electron transfer to CDs and sustained reactive oxygen species (ROS) production, thereby disrupting the attachment of biofouling organisms. This nanocomposite offers a scalable, low-impact antifouling solution that supports more sustainable aquaculture operations.
1 Introduction
Photocatalysts are materials that enhance the rate of a chemical reaction upon exposure to light by initiating active oxidation and reduction reactions.1 These materials produce reactive oxygen species (ROS), which act as potent oxidizing agents capable of degrading toxic organic compounds and disinfecting bacteria.2 Titanium dioxide (TiO2) is a widely recognized semiconductor and a potential photocatalyst owing to its unique properties, such as photostability, low cost and nontoxic nature.3 The anatase phase of bulk TiO2 has a band gap of 3.2 eV, restricting its response to UV radiation and thus limiting sunlight absorption (5–7%).3,4 Additionally, it exhibits rapid electron–hole (e−/h+) recombination, a low quantum yield and reduced adsorption efficiency, resulting in poor photocatalytic efficiency.5The photocatalytic efficiency of TiO2 can be enhanced by integrating it with transition metals and carbon nanomaterials such as carbon nanotubes (CNTs), fullerenes and graphene,6–8 which delay e−/h+ recombination. Transition metals induce Schottky and plasmonic effects, while carbon nanomaterials offer a high specific surface area and electrical mobility.9–14 Carbon nanomaterials can also form additional energy levels, reduce the band gap and increase sensitivity to visible light.3 Carbon dots (CDs) are synthesized from natural raw materials, are resistant to photobleaching, and possess low toxicity, high chemical and photostability, excellent biocompatibility, and cost-effectiveness.15–17 Thus, CDs can be integrated into TiO2 to enhance the photocatalytic activity of TiO2. Fe3+ oxide, possessing a 3d5 configuration, initiates photocatalysis by releasing and facilitating the migration of trapped carriers. It also narrows the band gap, creating intermediate energy levels, and delays e−/h+ recombination. The doping of Fe3+ into TiO2 extends its photocatalytic activity from the UV to visible region.18,19 Not many studies have reported the integration of CDs, iron oxide and TiO2 to produce photocatalytic materials with reduced band gaps and delayed e−/h+ recombination.
The accumulation of undesirable micro- and macro-organisms on submerged structures, such as ship hulls and offshore platforms, is referred to as biofouling. In aquaculture, the biofouling of cage nets is a significant and widely recognized issue, impacting farm productivity, fish health, and maintenance costs. According to the FAO (2024), the global aquaculture production reached 130.9 million tonnes in 2022, of which 94.4 million tonnes comprised aquatic animals. To meet the growing demand for aquatic food, the FAO emphasizes the need for sustainable intensification and expansion of aquaculture. Implementing effective antifouling strategies by employing nanomaterial-based advanced coatings is crucial for maintaining optimal aquaculture operations while minimizing environmental impact. To combat biofouling, the surface modification of submerged surfaces with antimicrobial agents is among the most effective methods. Nano-sized biocides employed for the antifouling of aquaculture cage nets include cuprous oxide,20 ZnO,21 ZnO–SiO composites,22 nanocarbon–inorganic biocide composites, nitrogen-doped CDs,23 and photocatalytic nano-TiO2-based composites,24,25 all of which have shown significantly enhanced antifouling properties.
The nonpolar surface of polyethylene does not adsorb biocides effectively without prior surface modification.25 Organosilicon-based sol–gel materials like silane are widely used for surface modification of textiles due to their hydrophobicity, chemical stability, self-cleaning ability, nontoxicity, enhanced surface durability, and resistance to ice formation.26–31 These properties make them potential materials for surface modification of nonpolar surfaces like PE. Ashraf et al. reported the utilization of organosilicon molecules to modify the polyalkene surfaces.25 In recent years, photocatalytic activity and its application are attracting attention for the development of biocidal molecules. TiO2 codoped with Ag, carbon and sulfur; ZnO with concrete; graphitic carbon nitrite; Cd sulfide with reduced graphene oxide; Zr6Ti4-based metal–organic frameworks; and ZnO reduced graphene oxide coatings have been widely utilized as biocides across various sectors including healthcare, construction, and water treatment plants.31–37 Very few studies have been attempted to develop antifouling strategies for aquaculture cage nets using photocatalytic materials as biocides. Synthesis of a Fe2O3–TiO2–CD (FeTiCD) nano composite with improved biocidal properties, expected to produce more reactive oxygen species (ROS) for inhibiting microorganism growth, and its application as an effective antifouling biocide in aquatic environment. The photocatalytic efficiency of TiO2 doped with Fe2O3 degrade organic biofouling agents, antimicrobial and light harvesting activity of CD prevent microbial colonization even in natural condition. Ashraf et al. (2024) reported the CuO–TiO2–CD composite, which has a similar synthetic route as the FeTiCD composite.25 Copper oxide nanoparticles are toxic in nature, and they can cause oxidative stress, inflammation, cytotoxicity, genotoxicity and immunotoxicity in living systems.38 Therefore, Fe2O3 is selected instead of CuO for the synthesis of the FeTiCD ternary system owing to its lower toxicity, economic viability, and ecofriendly nature. Excellent photocatalytic activity and a reduced band gap are obtained when Fe2O3 is doped into TiO2.39,40 Fe2O3 is considered an ideal dopant for TiO2 owing to its magnetic properties and low band gap energy, and Fe2O3–TiO2 shows excellent persistent organic pollutant removal and better photocatalyst recovery.41 The band gaps reported for the CuO–TiO2 nanocatalyst and carbon-doped TiO2/CuO photocatalyst are 2.8 and 2.2–2.5 eV, respectively.42,43 The band gap energy of the photocatalyst proposed here is much better than that of the CuO-doped TiO2 photocatalyst. Most of the existing photocatalytic systems of Fe and Ti are used for the removal of pollutants in water treatment plants, organic dye degradation, and so forth, but here, we use them as an antifouling coating for aquaculture cage nets to prevent biofouling. In contrast to biocide coatings like CuO, the presented coating is a sustainable and nontoxic approach. Hence, this synergistic coating can enhance the antifouling property under sunlight through the continuous supply of reactive oxygen species (ROS).
Herein, we propose the facile synthesis of a FeTiCD nanocomposite through hydrothermal carbonization using in-house-synthesized fish skin-derived carbon dots.44 Capitalizing on the efficient light-harvesting capabilities of CDs, the resulting FeTiCD composite coated on a silane base shows significant promise as an antifouling agent for polyethylene aquaculture cage nets. This innovative approach aims to enhance the efficacy of antifouling strategies, addressing the critical issue of biofouling in aquaculture systems.
2 Materials and methods
2.1. Materials used
Nano-iron oxide (Fe2O3) (5 wt% solution), a titanium dioxide (anatase) photocatalyst nanopowder having a size of approximately 7 nm, dimethyl sulfoxide and (3-glycidyloxypropyl)trimethoxysilane (silane), respectively, were procured from Plasma Chem GmbH, Berlin, SRL Chemicals, India, Qualigens, India, and Sigma-Aldrich. High-density polyethylene (HDPE) was obtained from the Government's Matsyafed Net Factory, Kochi, India. MilliQ type 1 water was used throughout the analysis. The polyethylene cage nets were pretreated using organosilane as per the method described by Ashraf et al.252.2. Synthesis and characterization of the photocatalytic Fe2O3–TiO2–CD nanocomposite
Initially, the nano-Fe2O3 and TiO2 composite (FeTi) was synthesized through a hydrothermal method.0.20 g of nano-Fe2O3, 0.10 g of TiO2 (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), 1 mL of dimethyl sulfoxide (DMSO) and 20 mL of water were taken in a 100 mL Teflon vessel. DMSO acted as a stabilizing agent, preventing nanoparticle aggregation and maintaining dispersion stability.45 The mixture was then subjected to hydrothermal synthesis with a temperature program of 1 hour to reach 180 °C, followed by 2 hours at 180 °C. After allowing it to cool naturally, the formed FeTi composite was centrifuged and washed thoroughly with MilliQ type 1 water at least three times. The residue was separated and dried. To incorporate CDs into this synthesized FeTi composite, the composite and CDs were mixed in a 2
1), 1 mL of dimethyl sulfoxide (DMSO) and 20 mL of water were taken in a 100 mL Teflon vessel. DMSO acted as a stabilizing agent, preventing nanoparticle aggregation and maintaining dispersion stability.45 The mixture was then subjected to hydrothermal synthesis with a temperature program of 1 hour to reach 180 °C, followed by 2 hours at 180 °C. After allowing it to cool naturally, the formed FeTi composite was centrifuged and washed thoroughly with MilliQ type 1 water at least three times. The residue was separated and dried. To incorporate CDs into this synthesized FeTi composite, the composite and CDs were mixed in a 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (w/w) weight ratio, and the total volume was made up to 25 mL with water. This mixture was stirred using a magnetic stirrer at 55 °C for 30 minutes. The solution was then centrifuged and washed thoroughly, and the residue Fe2O3–TiO2–CD (FeTiCD) was separated and dried. The preliminary characterization and photocatalytic efficiency studies of the FeTiCD nanocomposite were conducted using a Shimadzu 2450 double-beam UV-visible spectrophotometer equipped with an integrating sphere accessory. Also, the band gap energy (Eg) of TiO2, FeTi, and FeTiCD was obtained using UV-visible absorbance data using the Tauc relation46 (see SI, Fig. S1). A Shimadzu RF6000 spectrofluorometer was used for fluorescence spectral analysis at the excitation wavelengths of 230 nm and 260 nm.
1 (w/w) weight ratio, and the total volume was made up to 25 mL with water. This mixture was stirred using a magnetic stirrer at 55 °C for 30 minutes. The solution was then centrifuged and washed thoroughly, and the residue Fe2O3–TiO2–CD (FeTiCD) was separated and dried. The preliminary characterization and photocatalytic efficiency studies of the FeTiCD nanocomposite were conducted using a Shimadzu 2450 double-beam UV-visible spectrophotometer equipped with an integrating sphere accessory. Also, the band gap energy (Eg) of TiO2, FeTi, and FeTiCD was obtained using UV-visible absorbance data using the Tauc relation46 (see SI, Fig. S1). A Shimadzu RF6000 spectrofluorometer was used for fluorescence spectral analysis at the excitation wavelengths of 230 nm and 260 nm.
2.3. Treatment of PE nets with the photocatalytic Fe2O3–TiO2–CD nanocomposite
Acid-washed high-density polyethylene (HDPE) aquaculture cage nets were dipped in a 0.02 M silane solution for 3 hours and dried at 60 °C for 3 hours in a hot-air oven.25 Surface-modified PE netting samples (15 × 8 meshes) were immersed in various concentrations of the FeTiCD composite for 24 hours, and on the next day, they were retrieved and dried in air. This dried net sample was weighed and numbered using an aluminum token on each panel. The FeTiCD composite treatment concentrations were as follows: untreated PE (P0), silane-modified PE (P1), silane + 0.01% FeTiCD-coated PE (P2), silane + 0.02% FeTiCD-coated PE (P3), silane + 0.05% FeTiCD-coated PE (P4), and silane + 0.1% FeTiCD-coated PE (P5). Four replicates of six treatments were included in a random fashion on a PVC frame, as shown in Fig. 1, to ensure uniform exposure for each treatment. The PVC frames were submerged at a depth of 1 m from the surface during low tide at test sites at the institute in the Cochin estuary for a period of 6 months. The FTIR spectra of the composite and composite-coated net samples were obtained using a PerkinElmer Spectrum 100 spectrophotometer attached with a horizontal attenuated total reflectance (HATR) accessory. The X-ray diffraction pattern of the FeTiCD composite was analysed using a Bruker D8 advanced diffractometer with Cu Kα radiation operating at 40 kV and 35 mA. Scanning electron microscopy (SEM) images of the net panels were captured using a JEOL 6390LA at a working distance of 5 µm using an accelerating voltage of 20 kV, with magnifications ranging from 300× to 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 00×. Contact angle measurements were performed using a Rame-Hart 290-U1 contact angle goniometer.
00×. Contact angle measurements were performed using a Rame-Hart 290-U1 contact angle goniometer.
 | ||
| Fig. 1 Schematic of the PVC frame displaying four replicates of each treatment arranged in a random pattern. | ||
Each frame was retrieved at the end of one month (December 2023 to January 2024), two months (December 2023 to February 2024), four months (December 2023 to April 2024) and six months (February 2024 to August 2024). The retrieved samples were washed with running water to remove adhered debris, sediments and plastic waste. Cleaned samples were dried in air after removing the token. Microscopic images of the dried net samples were taken at 25× magnification using a Leica MZ16A stereomicroscope.
3 Result and discussion
3.1. Synthesis of the FeTiCD composite
The nano-Fe2O3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) TiO2 composite (FeTi) was synthesized hydrothermally, and the optimum ratio of its components was 2
TiO2 composite (FeTi) was synthesized hydrothermally, and the optimum ratio of its components was 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (w/w basis). The FeTi–carbon dot composite (FeTiCD) was synthesized by dispersing FeTi in a nano-CD solution and heating at 55 °C for 30 minutes in a hot-air oven. The optimum ratio of FeTi
1 (w/w basis). The FeTi–carbon dot composite (FeTiCD) was synthesized by dispersing FeTi in a nano-CD solution and heating at 55 °C for 30 minutes in a hot-air oven. The optimum ratio of FeTi![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CD was 2
CD was 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. The composite was subjected to morphological and physicochemical characterizations and field evaluations after coating onto silane-pretreated aquaculture cage nets.
1. The composite was subjected to morphological and physicochemical characterizations and field evaluations after coating onto silane-pretreated aquaculture cage nets.
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C carbon core and the n–π* transition from functional groups like C
C carbon core and the n–π* transition from functional groups like C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, OH, and C–N, respectively.44 The nano-TiO2 displayed characteristic absorption peaks at 278 and 323 nm, both within the UV region. Nano-Fe2O3 exhibited distinct absorption maxima at 206, 283, 370, and 501 nm. The Fe–Ti demonstrated absorption maxima at 287, 325, 450, 481 and 547 nm. The FeTiCD exhibited absorption at 217, 278, 320, 437 and 518 nm. FeTiCD exhibited prominent broader peaks in the 280–330 nm region compared with FeTi. The peak intensity ratio of 280/330 nm increased from 0.97 for FeTi to 1.01 for FeTiCD. Similarly, in the visible range, the absorption peak of FeTiCD at 437 nm became sharper and better defined compared to FeTi. The disappearance of the absorption at 481 nm for FeTiCD and a blue shift in the absorption from 547 nm (FeTi) to 518 nm (FeTiCD) with a lower intensity were observed. The successful integration of CDs into FeTi was evidenced in FeTiCD by the presence of peaks at 217 and 278 nm. The broader absorption at 278 nm had a higher intensity ratio (217/278 nm), which was contributed by TiO2 and the functional groups responsible for the n–π* transition. The extended absorption into the visible range suggests that the composite can effectively utilize sunlight, overcoming the inherent UV-region limitation of pristine TiO2.47,48
O, OH, and C–N, respectively.44 The nano-TiO2 displayed characteristic absorption peaks at 278 and 323 nm, both within the UV region. Nano-Fe2O3 exhibited distinct absorption maxima at 206, 283, 370, and 501 nm. The Fe–Ti demonstrated absorption maxima at 287, 325, 450, 481 and 547 nm. The FeTiCD exhibited absorption at 217, 278, 320, 437 and 518 nm. FeTiCD exhibited prominent broader peaks in the 280–330 nm region compared with FeTi. The peak intensity ratio of 280/330 nm increased from 0.97 for FeTi to 1.01 for FeTiCD. Similarly, in the visible range, the absorption peak of FeTiCD at 437 nm became sharper and better defined compared to FeTi. The disappearance of the absorption at 481 nm for FeTiCD and a blue shift in the absorption from 547 nm (FeTi) to 518 nm (FeTiCD) with a lower intensity were observed. The successful integration of CDs into FeTi was evidenced in FeTiCD by the presence of peaks at 217 and 278 nm. The broader absorption at 278 nm had a higher intensity ratio (217/278 nm), which was contributed by TiO2 and the functional groups responsible for the n–π* transition. The extended absorption into the visible range suggests that the composite can effectively utilize sunlight, overcoming the inherent UV-region limitation of pristine TiO2.47,48
 | ||
| Fig. 2 UV-visible spectra of (a) nano-iron oxide, nano-TiO2, FeTiCD and FeTi (nano-iron oxide (green line) is shown in the secondary axis) and (b) FeTiCD and FeTi in the visible range (400–600 nm). | ||
The band gap was calculated based on Tauc's equation. For bulk anatase TiO2, the band gap is typically around 3.2 eV. Quantum confinement effects can cause the band gap of TiO2 to increase, if it is in nanoscale range. The observed increase in the band gap can be attributed to the reduction in the crystallite size compared to the bulk material. As the crystallite size decreases, electrons become spatially confined within a smaller region, leading to an expansion of the energy separation between the valence band (VB) and conduction band (CB). Consequently, a higher energy input is required for electrons to transition from the VB to CB. This phenomenon is well explained by the quantum confinement effect (QCE), which states that the band gap energy increases with decreasing crystallite size, and vice versa.49,50 In bulk semiconductors, electrons and holes occupy continuous allowed and forbidden energy bands. When the material dimensions approach the nanoscale, quantum confinement results in discrete, size-dependent energy levels rather than continuous bands. Therefore, the VB and CB edges shift relative to those of the bulk counterpart, thereby widening the band gap energy.51 Results showed that the incorporation of Fe3+ and CDs into TiO2 introduced an intermediate energy level below the conduction band, narrowing the band gap from 4 eV (TiO2) to 2 eV in FeTi and further to 1.7 eV in FeTiCD. This modification enabled the visible light absorption of the composite (detailed description is given in SI, Fig. S1(a–c)). CDs exhibited fluorescence with the highest emission maxima at 445 nm at an excitation wavelength of 220 nm.38 The fluorescence of CDs was quenched in the FeTiCD composite upon the incorporation of CDs into FeTi (SI, Fig. S2). The introduction of CDs into the metal oxide matrix altered the bandgap, inducing charge separation and delayed e−/h+ recombination, leading to fluorescence quenching.52 The results clearly establish that CDs interact with metal oxides through conjugated C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds and other functional groups to form FeTiCD. The composite is expected to exhibit increased photocatalytic properties and delayed e−/h+ recombination.
C bonds and other functional groups to form FeTiCD. The composite is expected to exhibit increased photocatalytic properties and delayed e−/h+ recombination.
Narrowing the band gap of TiO2 is expected to decrease the reducing and oxidizing power, respectively, due to the less negative valence band maximum and less positive conduction band maximum.53,54 The introduction of iron oxide and carbon dots in the FeTiCD composite reduced the bandgap and significantly increased the visible light absorption, leading to enhanced population of e−/h+ pairs and hence increased photocatalytic reactions. Carbon dot improved the photocatalytic activity of iron oxide, while formation of the FeTiCD composite through coupling with TiO2 decreased the bandgap of TiO2 in the composite.55,56
The UV-visible spectroscopy of FeTiCD + dye in seawater was performed at hourly intervals to monitor the changes in the absorbance of the seawater samples exposed to sunlight with and without the MB dye. The absorbance intensities at 290 nm (UV region) and 665 nm (visible region) were recorded for both control (MB in water) and MB + FeTiCD-treated solutions. The photocatalytic efficiency was then calculated based on the variation in the absorbance intensity. Fig. 3(d) presents the photocatalytic efficiency under UV irradiation, where the synthesized photocatalyst showed enhanced performance compared to the control. Initially, the photocatalyst exhibited adsorption or partial degradation of the dye, followed by a significant increase in activity, which eventually stabilized, whereas the control displayed fluctuations and a considerable decrease in efficacy. This indicates the efficient utilization of UV light for the generation of reactive species and effective photocatalytic degradation.
Fig. 3(e) shows the photocatalytic efficiency under visible light, where the photocatalyst consistently outperformed the control throughout the experiment. Initially, the photocatalyst demonstrated efficient absorption of visible light and higher degradation rates compared to the control. Although a slight decline in activity was observed at later stages, possibly due to reduced availability of active sites, the photocatalyst maintained superior performance. The enhanced activity of the synthesized photocatalyst can be attributed to the efficient separation of e−/h+ pairs, as well as modifications and interactions among the composite components, which resulted in an improved surface area, thus leading to enhanced light harvesting and increased photocatalytic efficiency.59,60 The degradation of the MB dye by the composite established the composite's photocatalytic characteristics.
The Ti–O bond peaks at 669 and 896 cm−1 in FeTi and FeTiCD shifted to 675 and 902 cm−1, respectively, and the latter peak was wider and more suppressed. This clearly showed that the CDs interacted with the oxygen atom of the FeTi composite. The absorption corresponding to the O–H bond stretching of phenols/tertiary alcohol of CDs at 1390 cm−1 became more defined in FeTiCD, indicating the strong coordination bond-type interaction of O–H with metal oxides, and this was evidence by the wider absorption around 3600 cm−1. The aromatic C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C–C stretching vibration of fish skin-derived CDs was observed at 1454 and 1515 cm−1,44 and the absorptions in FeTiCD at 1507 and 1515 cm−1 indicated the interaction of aromatic C
C–C stretching vibration of fish skin-derived CDs was observed at 1454 and 1515 cm−1,44 and the absorptions in FeTiCD at 1507 and 1515 cm−1 indicated the interaction of aromatic C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C with metal oxide bonds. The triplet at ∼1640 cm−1, related to Fe–O and Ti–O bond bending vibrations, changed to a doublet upon the modification of the metal oxide composite with CDs. A shift in the aromatic C–H stretching frequency of CDs was observed from 3125 to 3115 cm−1 after FeTiCD formation. The peaks corresponding to C–H stretching vibrations of CH2 and CH3 groups introduced by CDs were observed as nicks and were masked by the dominating metal oxide absorptions around 3000 cm−1. The hydrogen bonded OH stretching vibration of metal oxides with CD exhibited wider absorption peak spanning from 3100 to 3600 cm−1 in FeTiCD composite. The conjugated C
C with metal oxide bonds. The triplet at ∼1640 cm−1, related to Fe–O and Ti–O bond bending vibrations, changed to a doublet upon the modification of the metal oxide composite with CDs. A shift in the aromatic C–H stretching frequency of CDs was observed from 3125 to 3115 cm−1 after FeTiCD formation. The peaks corresponding to C–H stretching vibrations of CH2 and CH3 groups introduced by CDs were observed as nicks and were masked by the dominating metal oxide absorptions around 3000 cm−1. The hydrogen bonded OH stretching vibration of metal oxides with CD exhibited wider absorption peak spanning from 3100 to 3600 cm−1 in FeTiCD composite. The conjugated C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C system of CDs and OH groups mainly interacted with the Ti–O and Fe–O bonds in the composite through van der Waals, coordination-type or hydrogen bonding interactions, which led to the successful formation of the composite.
C system of CDs and OH groups mainly interacted with the Ti–O and Fe–O bonds in the composite through van der Waals, coordination-type or hydrogen bonding interactions, which led to the successful formation of the composite.
3.2. FeTiCD composite-coated HDPE cage nets
Silane-pretreated polyethylene aquaculture cage nets were immersed overnight in varied concentrations of FeTiCD solutions (0.01% to 0.10%), and the retrieved nets were characterized and exposed to an estuarine environment to study the biofouling resistance of aquaculture cage nets after the biocide treatment.3.2.1.1. Scanning electron microscopy (SEM). Scanning electron microscopy with energy-dispersive X-ray analysis (SEM-EDAX) was employed to analyze the surface morphology and elemental composition of the synthesized material. The surface of PE (Fig. 7(a)) exhibited noticeable unevenness and roughness. However, after silane treatment (Fig. 7(b)), no particulate or crystalline structure was observed, implying the uniform coating of silane over PE. Coating FeTiCD over the silane-treated polyethylene (Fig. 7(c)) resulted in a more granular and particulate morphology and homogenously distributed FeTiCD on the surface. The presence of FeTiCD in the matrix may influence antimicrobial properties, potentially due to its previously established increased activity under visible light. The quantitative elemental composition of silane-pretreated HDPE nets was determined by SEM-EDX analysis, as given in Fig. 7(d), and the weight percentages (wt%) of C, Si, and O, respectively, were 84.08, 0.34 and 15.58 wt%. This clearly established the formation of the silane coating over the polyethylene net. In contrast, the FeTiCD-coated silane-pretreated HDPE net showed a dominant carbon peak at 0.27 keV (100%), along with a weak Si signal at 1.74 keV, as given in Fig. 7(e). The Fe and Ti peaks were expected between 6 and 7 keV but only appeared as weak signals, probably due to the nanoscale concentration of Fe and Ti, which was below the detection limit and further obscured by the strong carbon absorption originating from the polyethylene substrate and carbon dots. Such situations are expected when analytes are present in either very high or very low concentrations within the material. The results indicated that the FeTiCD is strongly embedded on the silane-coated PE surface, and its FTIR characteristics will determine the nature of the interaction of the biocide with silane.
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C aromatic stretching, C
C aromatic stretching, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C conjugation, and C
C conjugation, and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C aryl vibrations, respectively, provided clear evidence of the successful incorporation of CDs into the composite matrix. The additional peaks at 3633 cm−1 and 3651 cm−1, associated with hydroxyl (–OH) functional groups from CDs, indicated potential hydrogen bonding and π–π interactions between CDs and the silane-treated PE matrix. These spectral modifications confirmed that the FeTiCD composite was effectively integrated into the PE-silane network through epoxy bonds, π–π interactions and hydrogen bonding, leading to enhanced structural interactions that contributed to the stability and functional properties of the composite.
C aryl vibrations, respectively, provided clear evidence of the successful incorporation of CDs into the composite matrix. The additional peaks at 3633 cm−1 and 3651 cm−1, associated with hydroxyl (–OH) functional groups from CDs, indicated potential hydrogen bonding and π–π interactions between CDs and the silane-treated PE matrix. These spectral modifications confirmed that the FeTiCD composite was effectively integrated into the PE-silane network through epoxy bonds, π–π interactions and hydrogen bonding, leading to enhanced structural interactions that contributed to the stability and functional properties of the composite.
 | ||
| Fig. 10 Biomass accumulation (g g−1 of net) on nets exposed to the estuarine environment for 6 months. | ||
The salinity in the estuary during 1, 2, 4 and 6 months, respectively, was 19.77, 28.39, 29.81 and 12.8 psu. Similarly, the pH levels in 1, 2, 4, and 6 months, respectively, were 7.93, 7.59, 7.99 and 7.02. The dissolved oxygen levels in 1, 2, 4 and 6 months, respectively, were 1.6, 2.4, 2.0, and 2.4 mg L−1. The elevated salinity in the 4th month might have contributed to the higher biomass accumulation in the treated cage nets, as higher salinity accelerates biofouling development.70 The fourth-month retrieval coincided with the premonsoon season, during which the fouling intensity was high. In contrast, during the sixth-month retrieval, which occurred during the monsoon season, the low pH and salinity and high DO levels supported the growth of hydroids, which became the dominant fouling organisms attached to the net surfaces during this period. The results clearly established that the 0.05% FeTiCD-coated silane-pretreated polyethylene surface has the highest biofouling inhibition.
In this study, TiO2's surface is modified with nano-Fe2O3 and CDs. The incorporation of Fe(III) introduces mid-gap states between the valence and conduction bands, effectively reducing the bandgap of the composite. This shift is confirmed by the observed redshift in the absorption spectrum, indicating enhanced absorption in the visible region. Consequently, the composite can harness visible light photons to generate ROS. Fe(III) ions act as electron acceptors, suppressing e−/h+ recombination and prolonging charge carrier lifetimes.71
The narrowing of the bandgap can also be attributed to Ti–O–C bonding, which facilitates efficient charge transfer.72 According to John and Achari,3 in the presence of graphene, photogenerated electrons transfer from the conduction band (CB) of TiO2 (d-orbital) to the Fermi level of graphene (π-orbital), enhancing charge separation and further promoting ROS generation.3
The proposed mechanism for the enhanced photocatalytic activity of the composite is provided in Fig. 11. Fe(III) doping introduces a new energy level within the TiO2 bandgap, allowing photogenerated electrons to transfer to the modified conduction band, while holes remain in the valence band. The electrons in the conduction band are further accepted by CDs, which facilitate their transport to the surface, where they interact with water and oxygen to generate ROS, including superoxide (O2−˙), hydroperoxyl 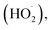 and hydroxyl radicals (˙OH). The reduced recombination rate ensures continuous ROS generation, effectively counteracting fouling agents. This study demonstrates that the synergistic effects of Fe(III) and CD incorporation significantly enhance the photocatalyst-mediated antifouling properties of TiO2, making it a promising material for environmental applications.
and hydroxyl radicals (˙OH). The reduced recombination rate ensures continuous ROS generation, effectively counteracting fouling agents. This study demonstrates that the synergistic effects of Fe(III) and CD incorporation significantly enhance the photocatalyst-mediated antifouling properties of TiO2, making it a promising material for environmental applications.
4 Conclusion
A photocatalytic iron oxide–titanium dioxide–carbon dot nanocomposite was synthesized; and results revealed that FeTiCD was successfully formed, and FeTi interacted with conjugated C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds of CDs, resulting in a reduced bandgap. FTIR spectra confirmed that the conjugated C
C bonds of CDs, resulting in a reduced bandgap. FTIR spectra confirmed that the conjugated C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds in CD interacted with oxygen moieties of the Fe–O and Ti–O bonds, most likely via hydrogen bonding, coordination, or van der Waals interactions involving the amine and hydroxyl groups introduced by CD into the matrix. It expanded visible-light absorption by reducing the bandgap to 1.7 eV, and iron oxide's and CDs' interaction with TiO2 enhanced photocatalytic efficiency by delaying e−/h+ recombination, resulting in the generation of ROS. The FeTiCD composite was coated onto silane-pretreated polyethylene cage nets, exhibiting strong adhesion via C–O–C bonds, π–π interactions, and hydrogen bonding, forming a stable organic–inorganic system between organosilane and the composite. The 0.05% composite-coated nets demonstrated superior antifouling performance after six months of exposure, making them a promising antifouling material. The present study emphasized the use of organosilane compounds as surface modifiers of nonpolar polyethylene and their effectiveness in holding biocides to deter the biofoulers in the aquatic environment. The contamination of the environment due to the nanomaterial is of little concern because the net is immersed in a 0.05% composite solution, making its impact very limited.
C bonds in CD interacted with oxygen moieties of the Fe–O and Ti–O bonds, most likely via hydrogen bonding, coordination, or van der Waals interactions involving the amine and hydroxyl groups introduced by CD into the matrix. It expanded visible-light absorption by reducing the bandgap to 1.7 eV, and iron oxide's and CDs' interaction with TiO2 enhanced photocatalytic efficiency by delaying e−/h+ recombination, resulting in the generation of ROS. The FeTiCD composite was coated onto silane-pretreated polyethylene cage nets, exhibiting strong adhesion via C–O–C bonds, π–π interactions, and hydrogen bonding, forming a stable organic–inorganic system between organosilane and the composite. The 0.05% composite-coated nets demonstrated superior antifouling performance after six months of exposure, making them a promising antifouling material. The present study emphasized the use of organosilane compounds as surface modifiers of nonpolar polyethylene and their effectiveness in holding biocides to deter the biofoulers in the aquatic environment. The contamination of the environment due to the nanomaterial is of little concern because the net is immersed in a 0.05% composite solution, making its impact very limited.
Author contributions
P. M. Ashraf: conceptualization and planning, writing the manuscript and data interpretation. Gopika R.: synthesis and application of the composite, laboratory experiments, data compilation, and manuscript writing.Conflicts of interest
The authors declare no conflicts of interest.Data availability
The data supporting this research paper are available from the corresponding author and will be shared upon reasonable request.Supplementary information (SI): the application of Tauc equation (Fig S1), fluorescence spectra of FeTiCD composite (Fig S2), detailed FTIR absorption frequencies of FeTi and FeTiCD composite (Table S1) and spectral data of silane pretreated and FeTiCD coated silane pretreated polyethylene aquaculture cage net (Table S2). See DOI: https://doi.org/10.1039/d5ra09273f.
Acknowledgements
DST INSPIRE India provided financial support to the research scholar in the form of a scholarship and contingencies. The authors sincerely thank the Director, ICAR-Central Institute of Fisheries Technology and the Head of Fishing Technology and Microbiology Fermentation and Biotechnology Division for providing facilities. Gratitude is also extended towards the Sophisticated Technology Instrumentation Centre, Cochin, for providing SEM facilities for samples analysis. The DST-INSPIRE programme is acknowledged for providing financial support.References
- R. Ameta, M. S. Solanki, S. Benjamin and S. C. Ameta, in Advanced Oxidation Processes for Waste Water Treatment, Academic Press, 2018, pp. 135–175, DOI:10.1016/B978-0-12-810499-6.00006-1.
- R. Ullah and J. Dutta, J. Hazard. Mater., 2008, 156, 194–200, DOI:10.1016/j.jhazmat.2007.12.033.
- D. John and V. S. Achari, Curr. Sci., 2022, 123, 1089–1100, DOI:10.18520/cs/v123/i9/1089-1100.
- D. I. Anwar and D. Mulyadi, Procedia Chem., 2015, 17, 49–54, DOI:10.1016/j.proche.2015.12.131.
- Z. Zhan, H. Wang, Q. Huang, S. Li, X. Yi, Q. Tang, J. Wang and B. Tan, Small, 2022, 18, 2105083, DOI:10.1002/smll.202105083.
- T. S. Natarajan, J. Y. Lee, H. C. Bajaj, W. K. Jo and R. J. Tayade, Catal. Today, 2017, 282, 13–23, DOI:10.1016/j.cattod.2016.03.018.
- Y. Pan, X. Liu, W. Zhang, Z. Liu, G. Zeng, B. Shao, Q. Liang, Q. He, X. Yuan, D. Huang and M. Chen, Appl. Catal., B, 2020, 265, 118579, DOI:10.1016/j.apcatb.2019.118579.
- A. Nasir, S. Khalid, T. Yasin and A. Mazare, Energies, 2022, 15, 6248, DOI:10.3390/en15176248.
- N. R. Khalid, A. Majid, M. B. Tahir, N. A. Niaz and S. Khalid, Ceram. Int., 2017, 43, 14552–14571, DOI:10.1016/j.ceramint.2017.08.143.
- D. Wang, N. B. Saleh, W. Sun, C. M. Park, C. Shen, N. Aich, W. J. Peijnenburg, W. Zhang, Y. Jin and C. Su, Environ. Sci. Technol., 2019, 53, 7265–7287, DOI:10.1021/acs.est.9b01453.
- L. G. Devi and R. Kavitha, Appl. Surf. Sci., 2016, 360, 601–622, DOI:10.1016/j.apsusc.2015.11.016.
- L. L. Tan, S. P. Chai and A. R. Mohamed, ChemSusChem, 2012, 5, 1868–1882, DOI:10.1002/cssc.201200480.
- Q. Xiang, J. Yu and M. Jaroniec, J. Am. Chem. Soc., 2012, 134, 6575–6578, DOI:10.1021/ja302846n.
- H. Yi, D. Huang, L. Qin, G. Zeng, C. Lai, M. Cheng, S. Ye, B. Song, X. Ren and X. Guo, Appl. Catal., B, 2018, 239, 408–424, DOI:10.1016/j.apcatb.2018.07.068.
- J. Peng, W. Gao, B. K. Gupta, Z. Liu, R. Romero-Aburto, L. Ge, L. Song, L. B. Alemany, X. Zhan, G. Gao, S. A. Vithayathil, B. A. Kaipparettu, A. A. Marti, T. Hayashi, J. J. Zhu and P. M. Ajayan, Nano Lett., 2012, 12, 844–849, DOI:10.1021/nl2038979.
- Y. Li, Y. Hu, Y. Zhao, G. Shi, L. Deng, Y. Hou and L. Qu, Adv. Mater., 2011, 23, 776–780, DOI:10.1002/adma.201003819.
- S. Sahu, Y. Liu, P. Wang, C. E. Bunker, K. S. Fernando, W. K. Lewis, E. A. Guliants, F. Yang, W. Jinping and Y. P. Sun, Langmuir, 2014, 30(28), 8631–8636, DOI:10.1021/la5010209.
- K. Zhou, Y. Zhu, X. Yang, X. Jiang and C. Li, New J. Chem., 2011, 35, 353–359, 10.1039/C0NJ00623H.
- T. S. Rao, in Biofouling in Industrial Water Systems, ed. Z. Amjad and K. D. Demadis, Elsevier, 2015, pp. 123–140, DOI:10.1016/B978-0-444-63228-9.00006-1.
- P. M. Ashraf, N. M. Lekshmi, S. Chinnadurai, S. Anjitha, M. Archana, C. M. Vineeth Kumar, K. M. Sandhya and A. P. Gop, Aquacult. Fish., 2022, 8, 538–543, DOI:10.1016/j.aaf.2022.01.002.
- P. Sathe, K. Laxman, M. T. Z. Myint, S. Dobretsov, J. Richter and J. Dutta, Sci. Rep., 2017, 7, 3624, DOI:10.1038/s41598-017-03636-6.
- N. M. Lekshmi, P. M. Ashraf, A. K. Keerthana, S. N. Thomas and L. Edwin, SN Appl. Sci., 2020, 2, 1–9, DOI:10.1007/s42452-020-03788-7.
- X. Ren, X. Wang, X. Yang, M. Hu, H. H. Xie and C. Wang, Polym. Compos., 2023, 44, 5121–5131, DOI:10.1002/pc.27478.
- K. Woan, G. Pyrgiotakis and W. Sigmund, Adv. Mater., 2009, 21, 2233–2239, DOI:10.1002/adma.200802738.
- P. M. Ashraf, C. S. Anjana and N. M. Lekshmi, Int. Biodeterior. Biodegrad., 2024, 193, 105856, DOI:10.1016/j.ibiod.2024.105856.
- S. Sfameni, T. Lawnick, G. Rando, A. Visco, T. Textor and M. R. Plutino, Nanomaterials, 2022, 12, 3404, DOI:10.3390/nano12193404.
- E. Richard, R. V. Lakshmi, S. T. Aruna and B. J. Basu, Appl. Surf. Sci., 2013, 277, 302–309, DOI:10.1016/j.apsusc.2013.04.052.
- B. Xu, Z. Cai, W. Wang and F. Ge, Surf. Coat. Technol., 2010, 204, 1556–1561, DOI:10.1016/j.surfcoat.2009.09.086.
- M. R. Plutino, C. Colleoni, I. Donelli, G. Freddi, E. Guido, O. Maschi, A. Mezzi and G. Rosace, J. Colloid Interface Sci., 2017, 506, 504–517, DOI:10.1016/j.jcis.2017.07.048.
- I. Ielo, M. Galletta, G. Rando, S. Sfameni, P. Cardiano, G. Sabatino, D. G. Drommi and M. R. Plutino, IOP Conf. Ser.: Mater. Sci. Eng., 2020, 777, 012003, DOI:10.1088/1757-899X/777/1/012003.
- S. Sfameni, G. Rando, A. Marchetta, C. Scolaro, S. Cappello, C. Urzì, A. Visco and M. R. Plutino, Gels, 2022, 8, 528, DOI:10.3390/gels8090528.
- D. B. Hamal, J. A. Haggstrom, G. L. Marchin, M. A. Ikenberry, K. Hohn and K. J. Klabunde, Langmuir, 2010, 26, 2805–2810, DOI:10.1021/la902844r.
- B. O. Bica and J. V. S. de Melo, Constr. Build. Mater., 2020, 252, 119120, DOI:10.1016/j.conbuildmat.2020.119120.
- N. F. F. Moreira, M. J. Sampaio, A. R. Ribeiro, C. G. Silva, J. L. Faria and A. M. T. Silva, Appl. Catal., B, 2019, 248, 184–192, DOI:10.1016/j.apcatb.2019.02.001.
- P. Devendran, D. Selvakumar, G. Ramadoss, R. Sivaramakrishnan, T. Alagesan, R. Jayavel and K. Pandian, Chemosphere, 2022, 287(1), 132091, DOI:10.1016/j.chemosphere.2021.132091.
- X. Wang, K. Ma, T. Goh, M. R. Mian, H. Xie, H. Mao, J. Duan, K. O. Kirlikovali, A. E. Stone, D. Ray and M. R. Wasielewski, J. Am. Chem. Soc., 2022, 144, 12192–12201, DOI:10.1021/jacs.2c03060.
- L. Valenzuela, A. Iglesias-Juez, B. Bachiller-Baeza, M. Faraldos, A. Bahamonde and R. Rosal, J. Mater. Chem. B, 2020, 8, 8294–8304, 10.1039/D0TB01428A.
- S. Naz, A. Gul and M. Zia, IET Nanobiotechnol., 2020, 14, 1–13 Search PubMed.
- A. Banisharif, A. A. Khodadadi, Y. Mortazavi, A. A. Firooz, J. Beheshtian, S. Agah and S. Menbari, Appl. Catal., B, 2015, 165, 209–221 CrossRef CAS.
- S. Zhan, J. Yang, Y. Liu, N. Wang, J. Dai, H. Yu, X. Gao and Y. Li, J. Colloid Interface Sci., 2011, 355, 328–333, DOI:10.1016/j.jcis.2010.11.026.
- O. F. S. Khasawneh and P. Palaniandy, Environ. Technol. Innovation, 2021, 21, 101230 CrossRef.
- A. Manivel, S. Naveenraj, P. S. Sathish Kumar and S. Anandan, Sci. Adv. Mater., 2010, 2, 51–57 Search PubMed.
- X. Peng, M. Wang, F. Hu, F. Qiu, H. Dai and Z. Cao, J. Alloys Compd., 2019, 770, 1055–1063 Search PubMed.
- R. Gopika, P. M. Ashraf and P. K. Binsi, Arabian J. Sci. Eng., 2023, 1–19, DOI:10.1007/s13369-023-08569-z.
- S. V. Gopal, R. Mini, V. B. Jothy and I. H. Joe, Mater. Today: Proc., 2015, 2, 1051–1055, DOI:10.1016/j.matpr.2015.06.036.
- P. Makuła, M. Pacia and W. Macyk, J. Phys. Chem. Lett., 2018, 9, 6814–6817, DOI:10.1021/acs.jpclett.8b02892.
- M. V. Shandrikov, A. S. Bugaev, A. V. Vizir, K. P. Savkin and E. M. Oks, J. Phys.: Conf. Ser., 2018, 1115, 032055, DOI:10.1088/1742-6596/1115/3/032055.
- X. F. Fan and J. M. Liu, J. Air Waste Manage. Assoc., 2015, 65, 50–58, DOI:10.1080/10962247.2014.962647.
- M. Podder, M. F. Ahmed, M. R. Moni, M. L. Rahman, B. Biswas and N. Sharmin, Arabian J. Chem., 2023, 16, 5 Search PubMed.
- S. Fatima, M. Anwar, A. S. Almalki, A. Alhadhrami, M. F. Warsi and Z. M. El-Bahy, Ceram. Int., 2024, 50, 29201–29212 Search PubMed.
- P. Borah, D. Siboh, P. K. Kalita, J. K. Sarma and N. M. Nath, Phys. B, 2018, 530, 208–214 Search PubMed.
- A. Sciortino, F. Ferrante, G. Gonçalves, G. Tobias, R. Popescu, D. Gerthsen, N. Mauro, G. Giammona, G. Buscarino, F. M. Gelardi and S. Agnello, ACS Appl. Mater. Interfaces, 2021, 13, 49232–49241, DOI:10.1021/acsami.1c14299.
- S. Na-Phattalung, D. J. Harding, P. Pattanasattayavong, H. Kim, J. Lee, D.-W. Hwang, T. D. Chung and J. Yu, J. Phys. Chem. Solids, 2022, 162, 110503, DOI:10.1016/j.jpcs.2021.110503.
- J. Huang, S. Wen, J. Liu and G. He, J. Nat. Gas Chem., 2012, 21, 302–307, DOI:10.1016/S1003-9953(11)60368-X.
- M. M. Mohamed, W. A. Bayoumy, M. E. Goher, M. H. Abdo and T. Y. Mansour El-Ashkar, Appl. Surf. Sci., 2017, 412, 668–682, DOI:10.1016/j.apsusc.2017.03.200.
- Y. Li, W. Xiang, T. Zhou, M. Huang, C. Wang, X. Wu, J. Mao and P. Wang, Chin. Chem. Lett., 2020, 31, 2757–2761, DOI:10.1016/j.cclet.2020.01.032.
- F. M. Dukes, E. Iuppa, B. Meyer and M. J. Shultz, Langmuir, 2012, 28, 16933–16940, DOI:10.1021/la303848g.
- G. Žerjav, M. S. Arshad, P. Djinović, I. Junkar, J. Kovač, J. Zavašnik and A. Pintar, Nanoscale, 2017, 9, 4578–4592, 10.1039/C7NR00704C.
- M. Shaban, A. M. Ashraf and M. R. Abukhadra, Sci. Rep., 2018, 8, 1–17, DOI:10.1038/s41598-018-19172-w.
- M. Kocijan, L. Ćurković, D. Ljubas, K. Mužina, I. Bačić, T. Radošević, M. Podlogar, I. Bdikin, G. Otero-Irurueta and M. J. Hortigüela, Appl. Sci., 2021, 11, 3966, DOI:10.3390/app11093966.
- D. T. Cromer and K. Herrington, J. Am. Chem. Soc., 1955, 77, 4708–4709, DOI:10.1021/ja01623a004.
- S.-C. Yu, J.-S. Lee, S.-F. Tung and C.-L. Lan, Golden Book of Phase Transitions, Wroclaw, 2002, vol. 1, p. 1 Search PubMed.
- S.-C. Yu, J.-S. Lee, S.-F. Tung and C.-L. Lan, J. Geol. Soc. China, 1999, 42, 349–358 Search PubMed.
- A. V. Vorontsov and S. V. Tsybulya, Ind. Eng. Chem. Res., 2018, 57, 2526–2536, DOI:10.1021/acs.iecr.7b04480.
- K. J. Mintz, M. Bartoli, M. Rovere, Y. Zhou, S. D. Hettiarachchi, S. Paudyal, J. B. Chen, P. Y. Liyanage, R. Sampson and D. Khadka, Carbon, 2021, 173, 433–447, DOI:10.1016/j.carbon.2020.11.017.
- F. Dai, Q. Zhuang, G. Huang, H. Deng and X. Zhang, ACS Omega, 2023, 8, 17064–17076, DOI:10.1021/acsomega.3c01336.
- R. Ravinesh, R. L. Laju, J. K. P. Edward and A. Biju Kumar, J. Aquat. Biol. Fish., 2023, 11, 28–34 Search PubMed.
- L. Chen and P.-Y. Qian, Mar. Drugs, 2017, 15, 264, DOI:10.3390/md15090264.
- M. S. Selim, S. A. El-Safty, M. A. Shenashen, S. A. Higazy and A. Elmarakbic, J. Mater. Chem. B, 2020, 8, 3701–3732, 10.1039/C9TB02119A.
- S. Mhaddolkar, A. Dineshbabu, T. Sujitha and L. Jayasree, Int. J. Life Sci., 2019, 7, 655–664 Search PubMed.
- P. M. Álvarez, J. Jaramillo, F. Lopez-Pinero and P. K. Plucinski, Appl. Catal., B, 2010, 100, 338–345, DOI:10.1016/j.apcatb.2010.08.010.
- Z. Wu, F. Dong, W. Zhao, H. Wang, Y. Liu and B. Guan, Nanotechnology, 2009, 20, 235701, DOI:10.1088/0957-4484/20/23/235701.
| This journal is © The Royal Society of Chemistry 2026 |








