Open Access Article
Open Access ArticleDeep eutectic solvents as recyclable media for metal removal from crude oil
Eminemi Mirinn ,
Andrew P. Abbott
,
Andrew P. Abbott and
Alexander F. R. Kilpatrick
and
Alexander F. R. Kilpatrick *
*
School of Chemistry, University of Leicester, University Road, LE1 7RH, Leicester, UK. E-mail: sandy.kilpatrick@leicester.ac.uk
First published on 23rd March 2026
Abstract
Crude oil contains trace metal species that impair refinery performance, poison catalysts, and contribute to environmental contamination. Conventional metal-removal processes, such as hydrotreating and solvent extraction, are energy-intensive and costly. In this study, deep eutectic solvents (DESs) were evaluated as alternative media for extracting metal-containing complexes from crude oil. Model experiments using copper, iron, and nickel phthalocyanines dissolved in toluene showed that all complexes were transferred intact into the DES phase. Thermodynamic analysis revealed that extraction is exothermic and driven by protonation of the phthalocyanine ring within acidic DESs, leading to enhanced metal uptake at lower temperatures. Across the formulations tested, more acidic DES systems consistently delivered higher extraction efficiencies.The methodology was then applied to a Nigerian crude oil sample to assess performance under realistic conditions. Acidified DESs effectively extracted metalloporphyrins and other metal species while maintaining good phase separation. A simple filtration-based workflow enabled recovery and reuse of the DES phase with minimal energy input, demonstrating the potential for operationally straightforward and recyclable metal-removal processes. Overall, this work establishes acidified DESs as efficient, mechanistically understood, and potentially low-energy media for the extraction of metal species from crude oil, offering a promising basis for next-generation upgrading technologies.
Introduction
Crude oil is composed predominantly of aliphatic and aromatic hydrocarbons, but it also contains a diverse array of more polar constituents incorporating sulfur, nitrogen, and oxygen heteroatoms, together with trace metals such as nickel, vanadium and iron, and small quantities of ionic species (Na+, Ca2+, Cl−).1,2 Even at low concentrations, these heteroatom-containing and metal-bearing impurities adversely affect downstream refining and utilisation.3,4 Metals may enter the crude either through the biological precursors of the oil – for example, iron originating from haem proteins – or via contact with mineral phases during geological processing. The high natural abundance of N- and S-containing macrocycles such as porphyrins and phthalocyanines within crude oil is particularly significant, as these ligands effectively solubilise a wide range of metal ions including Al, Fe, Ti, Mn, Mg and V.5,6 Once present, metal complexes can deactivate cracking catalysts, promote inorganic deposition that compromises heat transfer, and accelerate the degradation of lubricants.7–9At present, the most widely applied method for removing sulfur-, nitrogen-, oxygen- and metal-containing species is catalytic hydrotreating.10–12 In this process, crude oil fractions are contacted with hydrogen over a solid catalyst at elevated temperatures and moderate pressures.13 Hydrotreating converts heteroatom-containing and metal-associated impurities into forms more amenable to separation, but it is energy-intensive and carries a substantial operational and carbon cost.14 Alternative separation strategies—including solid- or liquid-phase extraction, alkylation, distillation and adsorption—have therefore been explored as complementary or lower-impact methods.15
Among non-hydrogenative approaches, liquid–liquid extraction remains an attractive option, provided that cross-contamination between phases is minimised. The extractant must be immiscible with the hydrocarbon matrix, which favours polar solvents; however, conventional aqueous systems introduce complications if water becomes incorporated into fuels or lubricants. Ionic liquids (ILs) and deep eutectic solvents (DESs) have thus emerged as promising alternatives.16–20
DESs are mixtures of Brønsted or Lewis acids and bases that form eutectic liquids through strong hydrogen-bonding interactions.21 Both ILs and DESs have been deployed for the extraction of complex organic molecules, including proteins, natural products and reaction products, and the field has been comprehensively reviewed.22–27 Challenges associated with the high viscosity of ILs and DESs have been addressed through liquid-phase microextraction,28 microwave-assisted extraction,29 and ultrasound-assisted microextraction.30 Notably, DESs have proved effective for the extraction of phenolic, sulfur-containing and nitrogen-containing compounds.31,32
While much early work focused on natural products, DESs have also been applied to the extraction of metals from diverse matrices, most commonly from aqueous media using hydrophobic DESs.26 Shishov et al. demonstrated the extraction of metals from vegetable oil and used motor oil employing choline chloride-based DESs with various hydrogen-bond donors.33 Mohammed et al. applied a choline chloride DES to Iraqi crude oil and observed appreciable extraction of Fe and Zn; they also employed ultrasound-assisted oxidative demetalisation, though extraction of vanadium and nickel remained only moderate.34
Thermodynamic studies have provided further insight into extraction processes. Abbott et al. investigated the transfer of thiophene from cyclohexane, used as an oil analogue, into DESs.35 Thiophene was preferentially solvated by the non-polar phase, resulting in a slightly positive free energy of extraction. The transfer into the DES was endothermic, and extraction proceeded through a favourable entropy change; heating the biphasic system enhanced extraction. The most effective DESs were those with the lowest surface tension, indicating that the principal energetic barrier was the formation of a cavity within the DES to accommodate the solute. By contrast, extraction of more polar solutes such as polyphenols and aromatic carboxylic acids from olive oil exhibited negative free energies and enthalpies of extraction, consistent with strong hydrogen-bonding interactions between solute and DES and proton-transfer processes occurring during extraction.36
The aim of the present study is to establish whether acidic DESs can be used to extract metals directly from crude oil via liquid–liquid extraction. Our working hypothesis is that protonation of the macrocyclic ligand framework—specifically porphyrinato metal complexes—facilitates efficient metal transfer by rendering the coordinated species more compatible with the DES phase. To develop and test this mechanistic model, we first employ metallophthalocyanines dissolved in toluene as a tractable molecular system that mimics the behaviour of porphyrinic metal complexes in a simplified hydrocarbon environment. We then extend the optimised extraction conditions to Nigerian crude oil, a well-characterised petroleum system whose trace metal composition is representative of many terrestrially derived crude oils.37–39
Experimental section
Materials
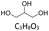
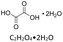
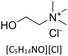
All reagents and solvents used in this study are listed in Table 1 together with their suppliers and stated purities. All materials were used as received without further purification. Crude oil samples were obtained from the Samabiri/Biseni flow station in Bayelsa State, Nigeria.Preparation of deep eutectic solvents
DESs were prepared following standard procedures described in the literature.40,41 Briefly, the hydrogen-bond donor and hydrogen-bond acceptor components were combined in the appropriate molar ratio and heated with stirring until a homogeneous liquid was formed. All DESs were stored in sealed containers to prevent moisture uptake prior to extraction experiments.Extraction experiments with metallophthalocyanines
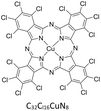
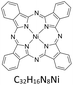
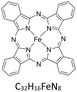
Copper(II) phthalocyanine green ([Cu(PcCl16)]), iron(II) phthalocyanine ([Fe(Pc)]), nickel(II) phthalocyanine ([Ni(Pc)]) and free-base phthalocyanine (H2Pc) were dissolved individually in toluene at 25 °C to give stock solutions. Liquid–liquid extraction was performed by contacting the metallophthalocyanine solution with the acidic DES under controlled mixing conditions.The concentration of metallophthalocyanines remaining in the toluene phase after extraction was determined using a Shimadzu UV-1601 spectrophotometer. Calibration curves for each compound were prepared over the range 0.01–0.10 µmol dm−3. All samples were diluted as necessary to ensure measurements fell within this linear range.
Determination of metal content in crude oil
Metal concentrations in crude oil before and after extraction were quantified using the emulsion-breaking method developed by de Sousa et al.42 Untreated crude oil (0.5 g) was mixed with 2.5 mL of paraffin wax (mineral oil) to reduce viscosity. An extractant solution comprising 20% (m/v) Triton X-114 and 6.5 mol dm−3 HNO3 (3 mL) was then added, and the mixture was vigorously shaken to form a stable emulsion.The emulsion was broken by heating at 90 °C for 40 min, followed by centrifugation at 5000 rpm for 15 min. This yielded two distinct phases; the denser lower phase was isolated and submitted for ICP-MS analysis.
For ICP-MS preparation, an aliquot (0.1 g) of the bottom phase was diluted with xylene to a total mass of 10 g. A subsample (0.5 g) of this solution was further diluted to 5.0 g with xylene, and indium was added as an internal standard to obtain a final concentration of 50 µg kg−1. Calibration standards were prepared by dilution of an organic metal standard in xylene.
ICP-MS analyses were performed using a PerkinElmer NexION 300X instrument equipped with a micro-nebuliser and cooled spray chamber. Oxygen was supplied as an auxiliary gas to facilitate direct introduction of the xylene-diluted samples into the plasma.
Physical characterisation of crude oil
Thermogravimetric analysis (TGA) of untreated and DES-treated Samabiri/Biseni crude oil samples was conducted from 25 to 1100 °C under controlled heating conditions. Dynamic Light Scattering (DLS) measurements were performed using a Zetasizer Nano-ZS (Malvern Instruments). Surface tension measurements were obtained using a Krüss K9 tensiometer (model K9MK1) equipped with a Pt–Ir plate (part no. PL21). Density was determined gravimetrically by measuring the mass of a known volume of oil in a calibrated volumetric flask, with reported values representing the average of five measurements.Specific gravity was determined using the standard American Petroleum Institute (API) gravity method (API gravity = 141.5–131.5 per SG).43
Results and discussion
Establishing a molecular basis for metalloporphyrin extraction
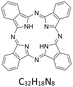
To design an effective strategy for removing metalloporphyrins from crude oil, it is first necessary to understand the underlying molecular chemistry of metal ion transfer from aromatic media into deep eutectic solvents (DESs). A key question is whether the metal centre remains coordinated to the macrocycle during the extraction, or whether protonation under acidic conditions leads to demetallation (Scheme 1). A further consideration is whether chloride ions present in the DES compete effectively with the phthalocyanine donor set for coordination to the metal centre. To address these questions, we first investigated the speciation and extraction behaviour of metallophthalocyanines dissolved in toluene, used here as a simple aromatic mimic for crude oil.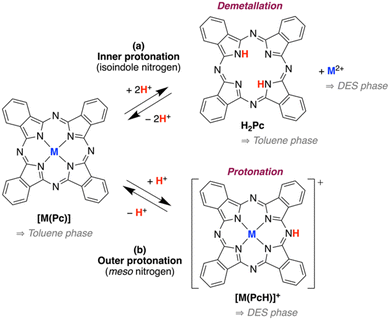 | ||
| Scheme 1 Possible protonation routes of metallophthalocyanines via (a) isoindole-nitrogen and (b) meso-nitrogen (adapted from ref. 44), and their predicted solubilities in toluene and DES. | ||
Copper(II) phthalocyanine green, [Cu(PcCl16)], was selected as an initial test solute due to its strong colour contrast and relatively high solubility in toluene. Although phthalocyanato complexes are generally sparingly soluble in organic solvents (<1 mmol dm−3), toluene provides adequate solubility and serves as a reasonable model for the aromatic fraction of crude oil.
Speciation of [Cu(PcCl16)] in toluene and DESs
Fig. 1 shows [Cu(PcCl16)] dissolved in toluene, in binary DES ethaline (1ChCl:2 EG) (Ethaline), and in a ternary DES based on ethaline (1ChCl:2 EG) acidified with methanesulfonic acid (MSA). In both DESs, the compound exhibited minimal solubility, but the colour remained characteristic of intact copper phthalocyanine. No yellow coloration was observed, indicating that chloride ions in the DES do not displace the macrocycle to generate [CuCl4]2−, in agreement with prior speciation studies.28,45 This conclusion was supported by UV-visible spectroscopy (Fig. S2). Calibration curves for metallophthalocyanines and H2Pc were constructed over 0.01–0.10 µmol dm−3 (Fig. S4 and S7), and all analytical samples were diluted to fall within this range.Extraction of [Cu(PcCl16)] from toluene into DESs
Fig. 2 shows that copper phthalocyanine initially forms a homogeneous blue-green solution in toluene. Upon contact with either 1ChCl:2 EG or 1ChCl:2 EG + MSA, the majority of the metallophthalocyanine is transferred into the DES phase. After 40 min, the extraction efficiency of [Cu(PcCl16)] from toluene was 96.9% at 25 °C, but decreased to 59.4% at 45 °C, consistent with an exothermic extraction process.In both DESs, the extracted material forms a cloudy dispersion or colloidal suspension rather than a clear solution. Although this complicates strict thermodynamic analysis, such behaviour is advantageous for practical separations: centrifugation or filtration can recover the solid material, enabling straightforward regeneration of the DES. Previous extraction systems—for example, thiophene extraction into DESs—experienced difficulties in regenerating the solvent.35 Here the formation of a removable dispersed phase is beneficial.
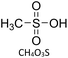
Acidifying 1ChCl:2 EG with MSA substantially enhances extraction. MSA has previously been shown to behave as a strong acid in DES media.46–48 UV-visible spectra of both phases (Fig. S3) were used to quantify partitioning.
Thermodynamics of extraction
The thermodynamic parameters governing extraction were examined using the partition coefficients measured at different temperatures. For fully homogeneous solutions, a partition coefficient (Kp) is the ratio of concentrations of a solute in a mixture consists of immiscible layers at equilibrium,35 defined as:
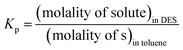 | (1) |
 | (2) |
 | ||
| Fig. 3 (a) 0.08 mmol of [Fe(Pc)], [Ni(Pc)] and H2Pc in toluene (top layer) and BTMAC:EG + 10 mmol MSA (bottom layer) before extraction and (b) after extraction for 40 min at 25 °C. | ||
Table 2 summarises the apparent thermodynamic parameters. The extraction is exothermic in all cases, with apparent enthalpy changes ( ) between −30 and −50 kJ mol−1, similar in magnitude to exothermic protonation reactions of amines.50 These values are consistent with protonation of the phthalocyanine macrocycle leading to formation of a cationic species that partitions into (or aggregates within) the ionic DES phase.
) between −30 and −50 kJ mol−1, similar in magnitude to exothermic protonation reactions of amines.50 These values are consistent with protonation of the phthalocyanine macrocycle leading to formation of a cationic species that partitions into (or aggregates within) the ionic DES phase.
| DES | Kp,app | ΔGapp (kJ mol−1) | (kJ mol−1) | (J mol−1 K−1) | ||
|---|---|---|---|---|---|---|
| 25 °C | 45 °C | 25 °C | 45 °C | 25 °C | 25 °C | |
| 1ChCl:2 EG + MSA | 4.23 | 1.36 | −3.57 | −0.88 | −44.7 | −138 |
| 1ChCl:EG | 1.05 | 1.05 | −0.12 | −0.12 | −29.2 | −97.9 |
| BTMAC:EG + MSA | 3.71 | 1.67 | −3.25 | −1.36 | −35.4 | −108 |
| [Bmim]Cl:EG | 3.44 | 1.67 | −3.06 | −1.36 | −33.6 | −102.3 |
| 1ChCl:2Gly + OA | 3.71 | 3.00 | −3.25 | −2.90 | −35.4 | −108 |
Phthalocyanato metal complexes typically undergo “outer” mono-protonation at the meso-nitrogen atoms to form [M(PcH)]+ species.44,51–55 Our results are consistent with this mechanism. No evidence was obtained for isoindole-nitrogen protonation, which would be expected to promote demetallation, and no spectral signatures consistent with free Cu2+ or [CuCl4]2− were observed.45
The apparent partition coefficient is highest for 1ChCl:2 EG + MSA, the most acidic system studied. Replacing the cation (e.g. [Bmim]Cl), the hydrogen-bond donor (e.g. glycerol in glyceline 1ChCl:2Gly), or the acid (oxalic acid) has relatively little effect, indicating that acidity is the dominant factor controlling extraction efficiency.
The strongly negative  values show that transfer from toluene into the DES is spontaneous. Increasing temperature decreases Kp, consistent with an exothermic process, as predicted by Le Chatelier's principle. The negative entropy changes suggest increased ordering associated with formation of an ionic or aggregated species.
values show that transfer from toluene into the DES is spontaneous. Increasing temperature decreases Kp, consistent with an exothermic process, as predicted by Le Chatelier's principle. The negative entropy changes suggest increased ordering associated with formation of an ionic or aggregated species.
Interestingly, 1ChCl:2 EG without added acid gives Kp ≈ 1 and only slightly negative  , although this DES is mildly acidic (pH ≈ 6).56 This suggests some degree of protonation or functionalisation of the phthalocyanato complex may still occur, albeit to a much lesser extent.
, although this DES is mildly acidic (pH ≈ 6).56 This suggests some degree of protonation or functionalisation of the phthalocyanato complex may still occur, albeit to a much lesser extent.
Previous extraction studies have shown that hydrogen-bond donors (e.g. alcohols) exhibit exothermic extraction into DESs, whereas non-polar solutes such as thiophene show endothermic extraction.35 The larger exothermicity observed here for [Cu(PcCl16)] indicates that ionic or strongly interacting solutes extract more favourably than neutral hydrogen-bonding solutes, in line with expectations.
Extraction of [Fe(Pc)], [Ni(Pc)] and H2Pc
To assess whether the trends observed for [Cu(PcCl16)] extend to other phthalocyanines, the extraction of iron(II) phthalocyanine ([Fe(Pc)]), nickel(II) phthalocyanine ([Ni(Pc)]) and the free-base phthalocyanine (H2Pc) was investigated using benzyltrimethylammonium chloride-based DES acidified with methanesulfonic acid (BTMAC:EG + MSA). These complexes were selected to probe the influence of metal identity and the absence of a coordinated metal centre on extraction behaviour.Fig. 3 shows the appearance of the toluene and DES phases before and after extraction. Initially, all three phthalocyanines formed homogeneous solutions in toluene, consistent with their moderate solubility in aromatic solvents. After 40 min of contact with BTMAC:EG + MSA at 25 °C, the majority of each species was transferred into the DES phase, as evidenced by the near-complete decolourisation of the toluene layer.
Quantitative analysis revealed transfer efficiencies of 99.9% for [Fe(Pc)], 76.2% for [Ni(Pc)] and 77.6% for H2Pc under these conditions. These values are comparable to those reported for other DES-mediated extractions, such as lidocaine with decanoic acid and hexylethylenediamine/menthol systems,57–59 highlighting the versatility of DESs as extraction media. The particularly high efficiency for [Fe(Pc)] suggests that iron phthalocyanine is more readily protonated or stabilised in the ionic DES environment, whereas [Ni(Pc)] and H2Pc exhibit slightly lower but still significant extraction.
As with the copper analogue, the DES phase forms a cloudy emulsion containing colloidal particles. Dynamic light scattering (DLS) revealed particle diameters of 250–2500 nm (broad size distribution). Such particles are easily removed by filtration or centrifugation.
Fig. 4 presents a schematic of the separation process. After extraction, centrifugation at 5000 rpm for 5 min gives a biphasic mixture in which the DES forms a clear lower layer while solid phthalocyanine aggregates remain suspended or collect at the interface. Filtration produces a clear DES filtrate and a solid residue of extracted macrocycle.
Application to Nigerian crude oil
Having established the feasibility of extracting metallophthalocyanines from an aromatic model system, the optimised conditions were applied to real crude oil from the Samabiri/Biseni flow station (Bayelsa State, Nigeria). The oil is classified as a heavy crude (API gravity 22.81).60 ICP-MS analysis of untreated samples (Table S2) revealed metal contents of Mg (13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 307 ppb), V (972 ppb), Fe (104
307 ppb), V (972 ppb), Fe (104![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 340 ppb), Ni (10
340 ppb), Ni (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 116 ppb) and Cu (2914 ppb).
116 ppb) and Cu (2914 ppb).
Extraction with a range of DESs (Table 3) showed that iron- and nickel-containing species in the crude oil are not present predominantly as metallophthalocyanines, as the extracts are yellow-brown rather than blue-green. DESs such as 1ChCl:2 EG and 1ChCl:1OA phase-separate cleanly from the oil, whereas 1ChCl:2Gly is unexpectedly miscible. Literature suggests that glycerol coordinates strongly to chloride, reducing the polarity of the resulting liquid and enabling mixing with hydrocarbons.61
Acidifying 1ChCl:2Gly with 10 wt% MSA restores phase separation and produces a dense, transparent ionic phase. A coloured solid collects at the interface, consistent with extraction of metal-containing components. Similar behaviour was observed for 1ChCl:2EG + MSA and 1ChCl:2EG + OA.
Overall extraction efficiencies exceed ca. 95% for all systems investigated, with 1ChCl:1OA giving the highest value (99.5%). Thermogravimetric analysis of crude oil before and after extraction shows negligible differences, indicating that ethylene glycol is not back-extracted into the oil phase; otherwise a characteristic exotherm at around 180 °C would be expected.
From a practical standpoint, the use of a liquid extracting phase offers advantages over solid acids, enabling efficient phase separation and avoiding blockage by precipitated solids. Preliminary reuse experiments (five consecutive cycles) show that the extraction efficiency remains essentially unchanged for both 1ChCl:2EG + MSA and 1ChCl:1OA (Fig. S13 and S14), supporting the potential recyclability of the DES phase.
Potential process design
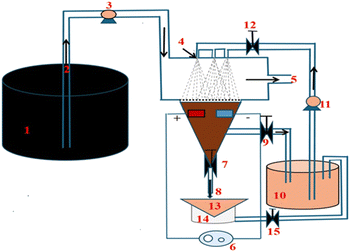
To translate the laboratory results into an operational metals-extraction process suitable for crude oil treatment, a conceptual liquid–liquid extraction system using DESs is proposed (Fig. 5). The design aims to maximise interfacial contact between the crude oil and the DES phase without the need for ultrasound-assisted oxidative demetallisation, relying instead on efficient mixing and density-driven phase separation.The system consists of a crude oil feed tank (1) supplying oil to a pipeline reactor via an inlet (2) and a regulated pump (3). The DES is introduced into the reactor through multiple spray nozzles (4) positioned at the top of the contact zone to generate fine droplets, thereby increasing the liquid–liquid interfacial area. The droplet size distribution is critical for mass transfer, and the DES flow is controlled by a recirculation pump (11), allowing continuous reuse of the DES as long as its metal loading remains below saturation.
Following dispersion and agitation through an electrically driven mixing system (6), the two phases enter a conical separation chamber where phase disengagement occurs due to the density difference. Metal-free crude oil exits through the upper outlet (5), while the DES enriched in metal complexes is withdrawn via valve (7).
Regeneration of the DES is envisioned at stage (14), where metal-containing particles are removed. In principle, this could be achieved by simple filtration (13), taking advantage of the tendency of protonated phthalocyanines and their metal complexes to aggregate. If the complexes exhibit sufficient paramagnetism (e.g., Fe-containing species), magnetic separation could be employed, although this would require validation since magnetic susceptibility will depend on the oxidation and spin states of the metal centres. Once regenerated, the DES is returned to the circulation tank (10) for reuse. Should the DES become saturated after repeated cycles, valve (9) is closed and valve (15) opened to introduce fresh DES into the system.
The primary advantage of using a DES rather than a solid acid is its pumpability and ease of mixing with the crude oil, ensuring that protonation (and subsequent displacement or aggregation of the metalloporphyrin) occurs uniformly throughout the reactor volume. The experimental data presented above suggest that re-acidification of the DES and precipitation of the metalloporphyrin species are the key chemical steps required for regeneration.
Approximate costs of the acids used in the DES formulations are listed in Table 4. Estimated recovery costs assume an average metal content of 131 ppm and a stoichiometry of two moles of acid per metalloporphyrin extracted. These values indicate that the basic chemical cost of the extraction step is typically below 1 cent per litre of crude oil. This suggests that the process could be economically viable at scale, provided that DES recycling efficiency and metal-complex handling are optimised.
| Acid | Cost per US$ tonne | Recovery cost per US$ tonne oil |
|---|---|---|
| Methanesulfonic | 2000–3000 | 0.76–1.15 |
| Oxalic | 350–500 | 0.13–0.18 |
| Lactic | 1300–2800 | 0.47–1.01 |
| Citric | 600–1900 | 0.46–1.45 |
Conclusions
This study demonstrates that deep eutectic solvents (DESs), particularly when acidified, are effective media for extracting phthalocyanato metal complexes from both model solvent systems (toluene) and crude oil. Copper, iron, and nickel phthalocyanines were all transferred intact into the DES phase, confirming that the central metal ion remains coordinated to the macrocycle throughout extraction. The combined spectroscopic and thermodynamic analyses indicate that extraction is driven by protonation of the phthalocyanine amine functionality at the DES–organic interface, yielding the corresponding ammonium phthalocyanine species. This protonation step underpins the observed exothermicity of the process and explains why partitioning into the DES phase is favoured at lower temperatures.Across the solvent systems explored, the apparent partition coefficients at 25 and 45 °C decrease in the order:
| 1ChCl:2 EG + MSA > 1ChCl:2Gly + OA > BTMAC:2 EG + MSA > [Bmim]Cl:EG > 1ChCl:2 EG. |
In many cases, extraction resulted in colloidal suspensions consistent with the formation of protonated metal phthalocyanine salts within the acidic DES phase. Importantly, the DESs could be cleanly recovered through simple filtration, enabling physical separation of the extracted metal phthalocyanines and highlighting the potential recyclability of these systems.
Application of this methodology to a Nigerian crude oil sample confirmed that acidified DESs effectively extract metal-containing species under realistic conditions. All acidic DESs showed high extraction efficiencies and good phase behaviour. Considering reagent cost and performance, oxalic acid emerged as the most economically advantageous acidifying agent.
Overall, this work provides mechanistic and practical insight into the use of acidified DESs for metal phthalocyanine extraction, demonstrating an exothermic, protonation-driven process that is operationally simple, scalable, and potentially recyclable. These findings offer a foundation for developing DES-based strategies for crude oil upgrading and contaminant removal.
Author contributions
conceptualisation, E. M., A. P. A., A. F. R. K.; methodology, E. M., A. P. A., A. F. R. K.; validation, E. M., A. P. A., A. F. R. K.; formal analysis, E. M., A. P. A.; investigation, E. M., A. P. A.,; writing - original draft, E. M., A. P. A., A. F. R. K.; writing - review & editing, E. M., A. P. A., A. F. R. K.; visualisation, E. M., A. P. A., A. F. R. K.; supervision, A. P. A., A. F. R. K.; project administration, A. P. A., A. F. R. K.; funding acquisition, E. M.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). The raw data for all the figures are openly available on figshare: https://doi.org/10.25392/leicester.data.29504831. Supplementary information: including UV-vis spectra and calibration curves, dynamic light scattering (DLS), FT-IR spectra, thermogravimetric analysis (TGA) and differential scanning calorimetry (DSC), and ICP-MS analysis. See DOI: https://doi.org/10.1039/d5ra09189f.Acknowledgements
We thank the Isaac Jasper Boro College of Education, Bayelsa State and The Tertiary Education Trust Fund, Nigeria for financial support and a PhD scholarship to E.M. We thank the Engineering and Physical Sciences Research Council (EPSRC) for the award of an Early Career Researcher International Collaboration Grant (EP/Y002695/1) to A.F.R.K.References
- B. P. Tissot and D. H. Welte, in. Petroleum Formation and Occurrence, Springer Berlin Heidelberg, 1984 Search PubMed.
- J. S. S. Damsté, J. W. De Leeuw, A. C. Kock-Van Dalen, M. A. De Zeeuw, F. De Lange, W. Irene, C. Rijpstra and P. A. Schenck, Geochim. Cosmochim. Acta, 1987, 51, 2369–2391 CrossRef.
- J. G. Reynolds, in. Metals and Heteroatoms in Heavy Crude Oils, Taylor & Francis Publishers Washington, DC, 1998 Search PubMed.
- M. Larocca, H. Farag, H. de Lasa and S. Ng, Ind. Eng. Chem. Res., 1990, 29, 2181–2191 CrossRef.
- Z. Niu, W. Meng, Y. Wang, X. Wang, Z. Li, J. Wang, H. Liu and X. Wang, J. Pet. Sci. Eng., 2022, 209, 109833 CrossRef.
- L. López and S. Lo Mónaco, Fuel, 2004, 83, 365–374 CrossRef.
- A. C. Jenifer, P. Sharon, A. Prakash and P. C. Sande, Energy Fuels, 2015, 29, 7743–7752 CrossRef.
- R. N. Magomedov, A. Z. Popova, T. A. Maryutina, K. M. Kadiev and S. N. Khadzhiev, Pept. Chem., 2015, 55, 423–443 CrossRef.
- M. A. Alabdullah, T. Shoinkhorova, A. Dikhtiarenko, S. Ould-Chikh, A. Rodriguez-Gomez, S. Chung, A. O. Alahmadi, I. Hita, S. Pairis and J. Hazemann, Catal. Sci. Technol., 2022, 12, 5657–5670 RSC.
- A. Hart, Int. J. Pet. Sci. Technol., 2012, 6, 79–96 Search PubMed.
- G. H. C. Prado, Y. Rao and A. de Klerk, Energy Fuels, 2017, 31, 14–36 CrossRef.
- A. Marafi, H. Albazzaz and M. S. Rana, Catal. Today, 2019, 329, 125–134 CrossRef.
- J. Ancheyta, A. Alvarez-Majmutov and C. Leyva, in Multiphase Catalytic Reactors, 2016, pp. 295–329 Search PubMed.
- P. Kokayeff, S. Zink and P. Roxas, in Handbook of Petroleum Processing, Springer, 2015, pp. 361–434 Search PubMed.
- A. Kayode Coker, in. Petroleum Refining Design and Applications Handbook, Mechanical Separations, Distillation, Packed Towers, Liquid-Liquid Extraction, Process Safety Incidents, Wiley, vol. 3, 2022 Search PubMed.
- F. Lima, M. Dave, A. J. D. Silvestre, L. C. Branco and I. M. Marrucho, ACS Sustain. Chem. Eng., 2019, 7, 11341–11349 CrossRef.
- S. E. E. Warrag, A. S. Darwish, F. O. S. Abuhatab, I. A. Adeyemi, M. C. Kroon and I. M. AlNashef, Ind. Eng. Chem. Res., 2020, 59, 11723–11733 CrossRef.
- Z. Zhu, H. Lü, M. Zhang and H. Yang, Phys. Chem. Chem. Phys., 2021, 23, 785–805 RSC.
- X. Wang, Z. Li, X. Wang, C. Wu, I. D. Gates, S. Guo, B. Wu, W. Zhu, M. Gu and M. Gao, J. Mol. Liq., 2022, 366, 120159 CrossRef.
- M. Y. Mohammed, A. M. Ali and T. M. Albayati, Chem. Afr., 2023, 6, 1595–1603 CrossRef.
- E. L. Smith, A. P. Abbott and K. S. Ryder, Chem. Rev., 2014, 114, 11060–11082 CrossRef PubMed.
- S. Oppermann, F. Stein and U. Kragl, Appl. Microbiol. Biotechnol., 2011, 89, 493–499 CrossRef.
- S. P. M. Ventura, F. A. e Silva, M. V Quental, D. Mondal, M. G. Freire and J. A. P. Coutinho, Chem. Rev., 2017, 117, 6984–7052 CrossRef PubMed.
- J. Xiao, G. Chen and N. Li, Molecules, 2018, 23, 1765 Search PubMed.
- Y. Hao, F. Pei, J. Huang, G. Li and C. Zhong, J. Sep. Sci., 2024, 47, 2300925 Search PubMed.
- S. C. Cunha and J. O. Fernandes, TrAC, Trends Anal. Chem., 2018, 105, 225–239 CrossRef.
- Y. H. Choi and R. Verpoorte, Curr. Opin. Food Sci., 2019, 26, 87–93 CrossRef.
- B. Tang, W. Bi, H. Zhang and K. H. Row, Chromatographia, 2014, 77, 373–377 CrossRef.
- J. Chen, M. Liu, Q. Wang, H. Du and L. Zhang, Molecules, 2016, 21, 1383 CrossRef PubMed.
- T. Khezeli, A. Daneshfar and R. Sahraei, Talanta, 2016, 150, 577–585 CrossRef PubMed.
- D. Chandran, M. Khalid, R. Walvekar, N. M. Mubarak, S. Dharaskar, W. Y. Wong and T. C. S. M. Gupta, J. Mol. Liq., 2019, 275, 312–322 CrossRef.
- P. H. Helou de Oliveira, P. J. Boaler, G. Hua, N. M. West, R. T. Hembre, J. M. Penney, M. H. Al-Afyouni, J. D. Woollins, A. García-Domínguez and G. C. Lloyd-Jones, Chem. Sci., 2024, 15, 11875–11883 RSC.
- A. Shishov, S. Savinov, N. Volodina, I. Gurev and A. Bulatov, Microchem. J., 2022, 179, 107456 Search PubMed.
- M. Y. Mohammed, A. M. Ali and T. M. Albayati, Chem. Eng. Res. Des., 2022, 182, 659–666 CrossRef.
- A. P. Abbott, A. Y. M. Al-Murshedi, O. A. O. Alshammari, R. C. Harris, J. H. Kareem, I. B. Qader and K. Ryder, Fluid Phase Equilib., 2017, 448, 99–104 Search PubMed.
- O. A. O. Alshammari, G. A. A. Almulgabsagher, K. S. Ryder and A. P. Abbott, Green Chem., 2021, 23, 5097–5105 Search PubMed.
- A. Akinlua, N. Torto and T. R. Ajayi, Fuel, 2008, 87, 1469–1477 CrossRef.
- O. L. Faboya, A. A. Ojo, H. Bello and I. Osasona, Pet. Sci. Technol., 2018, 36, 130–135 Search PubMed.
- I. O. Shotonwa, A. O. Giwa-Ajeniya and G. O. Mekuleyi, in The Political Ecology of Oil and Gas Activities in the Nigerian Aquatic Ecosystem, Academic Press, 2018, pp. 33–46 Search PubMed.
- A. P. Abbott, R. C. Harris and K. S. Ryder, J. Phys. Chem. B, 2007, 111, 4910–4913 Search PubMed.
- A. P. Abbott, D. Boothby, G. Capper, D. L. Davies and R. K. Rasheed, J. Am. Chem. Soc., 2004, 126, 9142–9147 CrossRef.
- J. M. de Sousa, R. J. Cassella and F. G. Lepri, Energy Fuels, 2019, 33, 10435–10441 Search PubMed.
- M. Kumar and M. P. Sharma, Egypt. J. Pet., 2016, 25, 375–382 CrossRef.
- S. Fukuzumi, T. Honda and T. Kojima, Coord. Chem. Rev., 2012, 256, 2488–2502 CrossRef.
- J. M. Hartley, C.-M. Ip, G. C. H. Forrest, K. Singh, S. J. Gurman, K. S. Ryder, A. P. Abbott and G. Frisch, Inorg. Chem., 2014, 53, 6280–6288 Search PubMed.
- M. Rollo, F. Raffi, E. Rossi, M. Tiecco, E. Martinelli and G. Ciancaleoni, Chem. Eng. J., 2023, 456, 141092 CrossRef CAS.
- B. Bae, Y. Kwon, S. Kang, Y. Kang, T. H. Won and J. Lee, Sep. Purif. Technol., 2025, 134869 CrossRef CAS.
- A. A. N. M. Al-Haimi, F. Yehia, F. Liu, X. Zhen, S. Zhu and Z. Wang, Fuel Process. Technol., 2025, 277, 108310 CrossRef CAS.
- P. W. Atkins, in. Concepts in Physical Chemistry, Royal Society of Chemistry, 2024 Search PubMed.
- P. Paoletti, R. Barbucci and A. Vacca, J. Chem. Soc., Dalton Trans., 1972, 2010–2013 RSC.
- P. A. Bernstein and A. B. P. Lever, Inorg. Chim. Acta, 1992, 198–200, 543–555 CrossRef CAS.
- T. T. Lu, M. Xiang, H. L. Wang, T. J. He and D. M. Chen, J. Mol. Struct. THEOCHEM, 2008, 860, 141–149 CrossRef CAS.
- T. Honda, T. Kojima, N. Kobayashi and S. Fukuzumi, Angew. Chem., Int. Ed., 2011, 50, 2725–2728 CrossRef CAS PubMed.
- A. S. Malyasova, E. A. Kostrova, I. G. Abramov, V. E. Maizlish and O. I. Koifman, Russ. Chem. Bull., 2021, 70, 2405–2415 CrossRef CAS.
- M. J. Lin, X. Fang, M. B. Xu and J. D. Wang, Spectrochim. Acta, Part A Mol. Biomol. Spectrosc., 2008, 71, 1188–1192 Search PubMed.
- A. P. Abbott, S. S. M. Alabdullah, A. Y. M. Al-Murshedi and K. S. Ryder, Faraday Discuss., 2018, 206, 365–377 RSC.
- Z. Yuan, H. Liu, W. F. Yong, Q. She and J. Esteban, Green Chem., 2022, 24, 1895–1929 Search PubMed.
- C. Wang and E. Hua, J. Solution Chem., 2024, 53, 1340–1352 Search PubMed.
- N. Schaeffer, M. A. R. Martins, C. M. S. S. Neves, S. P. Pinho and J. A. P. Coutinho, Chem. Commun., 2018, 54, 8104–8107 Search PubMed.
- R. E. C. Pabón and C. R. de Souza Filho, Fuel, 2019, 237, 1119–1131 Search PubMed.
- S. L. Perkins, P. Painter and C. M. Colina, J. Chem. Eng. Data, 2014, 59, 3652–3662 Search PubMed.
- Prices were obtained as a range from, https://www.alibaba.com, accessed August 2024.
| This journal is © The Royal Society of Chemistry 2026 |