 Open Access Article
Open Access ArticleEngineering Ti3C2Tx (titanium carbide) MXene-based composites and emerging applications: a mini review
Ly Tan Nhiem *
*
Faculty of Chemical and Food Technology, Ho Chi Minh City University of Technology and Engineering, 01 Vo Van Ngan Street, Thu Duc Ward, Ho Chi Minh City, Vietnam. E-mail: nhiemlt@hcmute.edu.vn
First published on 12th May 2026
Abstract
Two-dimensional Ti3C2Tx MXene has attracted considerable attention owing to its metallic conductivity, tunable surface terminations, and structural versatility, which underpin its broad functional applicability. Although numerous review articles have addressed Ti3C2Tx MXene-based composites, most have primarily focused on energy storage and energy conversion systems. In contrast, this review provides a fundamental and up-to-date overview of Ti3C2Tx MXene research in recent years, emphasizing the relationship between synthesis strategies, surface termination control, structural and electronic properties, and application-specific performance. First, various synthesis routes are discussed with particular emphasis on how etching conditions, post-treatment processes, and delamination strategies regulate the surface termination groups (–O, –OH, and –F) and defect structures of Ti3C2Tx. These surface chemistries critically influence the electronic structure, interlayer spacing, hydrophilicity, and charge transport characteristics of the material. Subsequently, the resulting structure–property relationships are analyzed to explain how these physicochemical features govern functional performance in different technological applications. Particular attention is devoted to integrating experimental observations with density functional theory (DFT) calculations to elucidate reaction mechanisms and interfacial interactions. Finally, applications in sensing, energy harvesting, water splitting, water remediation, and biomedical technologies are systematically discussed. By correlating synthesis parameters with surface chemistry and electronic structure, this review aims to provide a fundamental framework for understanding current challenges and guiding the rational design and scalable deployment of Ti3C2Tx MXene-based systems.
Introduction
Nowadays, nanoscience and engineering, particularly of two-dimensional (2D) nanomaterials, have become a central focus of both fundamental research and industrial commercialization due to their unique physicochemical properties.1–5 Among them, the exploration of 2D Ti3C2Tx in the year of 2011 introduced a novel class of 2D materials known as MXene.6,7 MXene possesses diverse elemental compositions, natural hydrophilicity, unique 2D structures with multilayered shapes, large surface areas with abundant surface termination groups, good conductivity, and excellent optoelectronic properties.8,9 To date, more than 35 stoichiometric phases have been identified with hexagonal P63/mmc symmetry. MXene sandwiched layers are produced by removing A layers from the MAX phase precursor (A represents Ga, Si, Al, etc.).10 There are several methods to break the chemically bonded A layer in MAX phases, such as top–down synthesis techniques and wet chemical etching.11 These methods can be summarized into several categories, including alkaline hydrothermal treatments, hydrofluoric acid (HF) and HF-containing etchant treatment, HF-forming etchant treatment, and molten-salt etching methods.In general, the MXene class of materials is composed of transition metal carbides, nitrides, and carbonitrides, with the general formula Mn+1XnTx (n = 1, 2, or 3), where M, X, and Tx represent a transition metal, N and/or C atoms, and surface termination groups, respectively.10,12,13 MXenes with n > 3 were only explored as minor impurity phases or in radiofrequency sputtered thin films.6 Recently, Deysher and co-workers successfully synthesized an (Mo0.8V0.2)5AlC4 MAX phase with the molar ratio of Mo![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) V
V![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) V2O3
V2O3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Al
Al![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) C of ∼4
C of ∼4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.9
0.9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.05
0.05![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.2
1.2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3.5 under an Ar flow via a powder metallurgy technique. Then, (Mo0.8V0.2)C4Tx was also etched using HF acid at 50 °C in an oil bath for 8 days.14 It is important to note that HF was utilized for etching Al layers, thereby creating additional space for electrochemical Li intercalation in battery research.15 The etching conditions were tuned to optimize the yields and minimize the destruction of the MXene nanosheets in liquid acid.16 The surface termination groups of the Ti3AlC2 MXene depend on the etching conditions. When HF acid was used for etching, F terminations dominate, while when molten chloride salts were used, Cl-rich surfaces are obtained.17
3.5 under an Ar flow via a powder metallurgy technique. Then, (Mo0.8V0.2)C4Tx was also etched using HF acid at 50 °C in an oil bath for 8 days.14 It is important to note that HF was utilized for etching Al layers, thereby creating additional space for electrochemical Li intercalation in battery research.15 The etching conditions were tuned to optimize the yields and minimize the destruction of the MXene nanosheets in liquid acid.16 The surface termination groups of the Ti3AlC2 MXene depend on the etching conditions. When HF acid was used for etching, F terminations dominate, while when molten chloride salts were used, Cl-rich surfaces are obtained.17
In addition, as synthesis routes and surface terminations critically influence reproducibility, scalability, and application-specific performance, it is necessary not only to select high-quality MAX phases and precisely control the synthesis strategy, including appropriate etchants and dosages, but also to employ encapsulation strategies, construct MXene-derived composites, or utilize suitable surfactants to stabilize MXenes. One of the main drawbacks of MXene materials is their high susceptibility to oxidation under environmental conditions such as elevated temperature, high humidity, and exposure to air or oxygen (1–2 months). In particular, structural degradation of Ti3C2Tx can occur even in the absence of external oxidizing agents at elevated temperatures, which is attributed to the interaction between molecular hydrogen and surface functional groups trapped between the interlayers. It has been reported that oxidation preferentially occurs at defective sites or –OH/–O-terminated sites rather than at –F-terminated sites, owing to the diffusion of Ti4+ and O2− ions, which participate in the oxidation process. Thermal heat treatment and storing samples under refrigerated or deoxygenated conditions are considered effective strategies to mitigate the oxidation of Ti3C2Tx nanostructures. Moreover, representative stabilizing agents include 1H,1H,2H,2H-perfluoroalkylsilane, sodium L-ascorbate, sodium dodecyl sulfate, and (3-chloropropyl) trimethoxysilane.18,19 It is essential to recognize and address the long-term stability issues of MXenes during storage and utilization, as they are strongly influenced by the preparation methods employed. Under humid or elevated-temperature conditions, MXenes are prone to degradation and oxidation into TiO2 and amorphous C, which significantly restricts their large-scale commercialization and practical applications. On the one hand, current storage strategies, such as dispersing MXenes in organic solvents, storing them at low or cryogenic temperatures, or sealing them under inert atmospheres, can effectively extend their storage lifetime. However, these approaches primarily delay degradation rather than fundamentally preventing it. On the other hand, more intrinsic stabilization strategies have been proposed, including minimizing defect sites on MXene surfaces and edges through surface functionalization, edge or surface passivation, encapsulation, and hydrogen-based thermal treatments.20 These approaches aim to directly suppress oxidation pathways and offer more sustainable solutions for improving the durability of MXenes.
As mentioned previously, owing to the surface functional groups (typically –Cl, –F, ![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, and –OH), Ti3C2Tx MXenes have excellent properties such as hydrophilicity, desirable optical, scalability, mechanical and electrical properties.6,21 Since their discovery, Ti3C2Tx MXenes have attracted increasing research interest thanks to their broad range of applications in energy storage, gas and pressure sensors, and electromagnetic interference shielding, depending on their surface termination groups and compositions.22 Different from their parent MAX phases, Ti3C2Tx MXenes can be easily intercalated between layers using sonication and polar organic solvents such as dimethyl sulfoxide (DMSO), tetrabutylammonium hydroxide (TBAOH), and n-butylamine.23 Ti3C2Tx MXene materials can be well dispersed in liquids without any surfactant thanks to their hydrophilicity. Following this, Ti3C2Tx MXene's properties can be enhanced by forming composites with metal oxide semiconductors.24 There are many reports predicting the properties of these types of heterostructures and their potential applications, thereby expanding opportunities for the scientific community.25
O, and –OH), Ti3C2Tx MXenes have excellent properties such as hydrophilicity, desirable optical, scalability, mechanical and electrical properties.6,21 Since their discovery, Ti3C2Tx MXenes have attracted increasing research interest thanks to their broad range of applications in energy storage, gas and pressure sensors, and electromagnetic interference shielding, depending on their surface termination groups and compositions.22 Different from their parent MAX phases, Ti3C2Tx MXenes can be easily intercalated between layers using sonication and polar organic solvents such as dimethyl sulfoxide (DMSO), tetrabutylammonium hydroxide (TBAOH), and n-butylamine.23 Ti3C2Tx MXene materials can be well dispersed in liquids without any surfactant thanks to their hydrophilicity. Following this, Ti3C2Tx MXene's properties can be enhanced by forming composites with metal oxide semiconductors.24 There are many reports predicting the properties of these types of heterostructures and their potential applications, thereby expanding opportunities for the scientific community.25
In recent years, numerous review articles have summarized MXene-based materials, with a predominant focus on energy-related applications such as supercapacitors, batteries, and electrocatalysis.26–29 However, rapid developments in synthesis strategies, surface functionalization, and emerging multifunctional applications have not been updated or compared across different research directions. In particular, the integration of experimental observations with theoretical studies, such as DFT calculations, remains underrepresented in existing reviews, despite its importance for understanding structure–property relationships and guiding rational material design. Moreover, DFT calculations can be used to predict composite configurations, estimate electrocatalytic reaction performance, and elucidate the relationships between material composition, properties, and performance, thereby providing guidance for material synthesis as well as a better understanding of plausible reaction mechanisms.26
This review aims to provide a focused and updated perspective on metallic Ti3C2Tx MXene research by critically analyzing recent progress reported primarily over the past five years. Unlike many existing reviews that predominantly emphasize energy storage and electrochemical applications, this work highlights the broader multifunctionality of Ti3C2Tx MXenes, including emerging roles in photonics, sensing, catalysis, and environmental technologies. To clearly distinguish this review from the prior literature, we adopt a structure–property–performance framework that systematically links synthesis strategies and surface termination engineering with the resulting electronic and physicochemical properties governing application-specific performance. In addition, this review incorporates technological readiness levels (TRLs), cost considerations, and realistic commercialization perspectives, supported by recent global MXene market forecasts (e.g., MXene Market Insights, with projections up to 2033). A comparative overview of representative 2D materials (MXenes, borophene, and graphene) is also provided to highlight their respective properties, strengths, and limitations. Furthermore, the selected studies emphasize advances in surface chemistry control, scalable synthesis approaches, and performance mechanisms validated through both experimental investigations and theoretical calculations, including density functional theory (DFT). By integrating experimental observations with theoretical understanding, this review aims to clarify key design principles for Ti3C2Tx-based composites and highlight opportunities for scalable production and practical implementation. Finally, we discuss current limitations and future research directions that may accelerate the industrial translation of Ti3C2Tx MXene technologies.
Synthesis and properties of Ti3C2Tx MXenes
Although MXene synthesis is often associated with HF, fluorine-containing compounds are well known for their severe toxicity and hazardous effects, including potential impacts on the nervous, skeletal, and reproductive systems, as well as dental health. However, despite these risks, relatively few studies have quantitatively examined fluorine-related safety issues and laboratory accidents during MXene synthesis, which remain a significant bottleneck for their large-scale commercialization. According to a clinical report, the serum calcium level of a patient exposed to HF acid was approximately 4.9 mg dL−1, which is substantially lower than the normal physiological range of 8.4–10.2 mg dL−1.27 Therefore, strict safety protocols and emergency response guidelines are essential when handling fluorine-containing aqueous solutions, regardless of their concentration, and appropriate laboratory protective equipment must always be employed.Fluoride-based methods
Ti3C2Tx MXenes are synthesized via the wet chemical technique from Ti3AlC2 MAX phases in HF solution or via in situ HF generation using a mixture of LiF and HCl as well as NH4HF2, as shown in Fig. 1.30,31 In particular, typically F-containing etchants are utilized to selectively eliminate the Al layer, releasing termination groups Tx. These methods are easy to perform; however, the acute toxicity of fluoride-containing acidic solutions poses a challenge in aqueous processing and hinders scale-up. Moreover, the wastewater after the experiment should be treated well before its release into the natural environment. Each technique has its own pros as well as cons, which can be briefly expressed through the following chemical reactions for Ti3AlC2:32| Ti3AlC2 + 3HF → AlF3 + 3/2H2 + Ti3C2Tx | (1.1) |
| Ti3C2Tx + 2HF → Ti3C2F2 + H2 | (1.2) |
| Ti3C2Tx + 2H2O → Ti3C2(OH)2 + H2 | (1.3) |
 | ||
| Fig. 1 Schematic of (a and c) cost consideration, adapted/reproduced from ref. 22 with permission from Elsevier, Copyright 2026. (b) General method for Ti3C2Tx MXene synthesis from Ti3AlC2 MAX phase, adapted/reproduced from ref. 35 with permission from the American Chemical Society, Copyright 2017. | ||
The quality of Ti3C2Tx MXenes depends not only on the choice of etchant, synthesis scale, and molar ratios, but also on the external forces applied during delamination and post-treatment processes. For instance, sonication-assisted delamination typically yields small-sized flakes (∼200–500 nm), whereas manual shaking produces significantly larger flakes, with lateral sizes ranging from ∼4 to 15 µm.33 Furthermore, centrifugation speed strongly influences size selection: at 5000 rpm, the collected MXene supernatant predominantly contains small flakes with an average size of 0.11 ± 0.07 µm, while centrifugation at 1000 rpm yields larger flakes with an average size of 0.8 ± 0.3 µm.34
Halogen etching of Ti3AlC2 MAX phase
In 2021, a new method to etch the Ti3AlC2 MAX phase was explored by Richard and colleagues using elemental halogens, as shown in Fig. 2a. In this study, the aluminum halide was generated during the etching process, along with halogenated Ti3C2Tx with 1% yield MXene (at 1 mg mL−1). The Ti3C2Tx crude was decontaminated by washing with a nonpolar solvent (CHCl3) several times.36 As shown in Fig. 2b, the plasmonic resonance exhibits a gradual red shift across different surface terminations, which may correlate with variations in the electronegativity of the functional groups (F > Cl > Br > I) and the resulting polarization of the Ti–halogen bonds. Although electron density redistribution is likely involved, the underlying mechanisms governing the observed plasmonic trends remain insufficiently clarified and warrant further systematic experimental and theoretical investigations.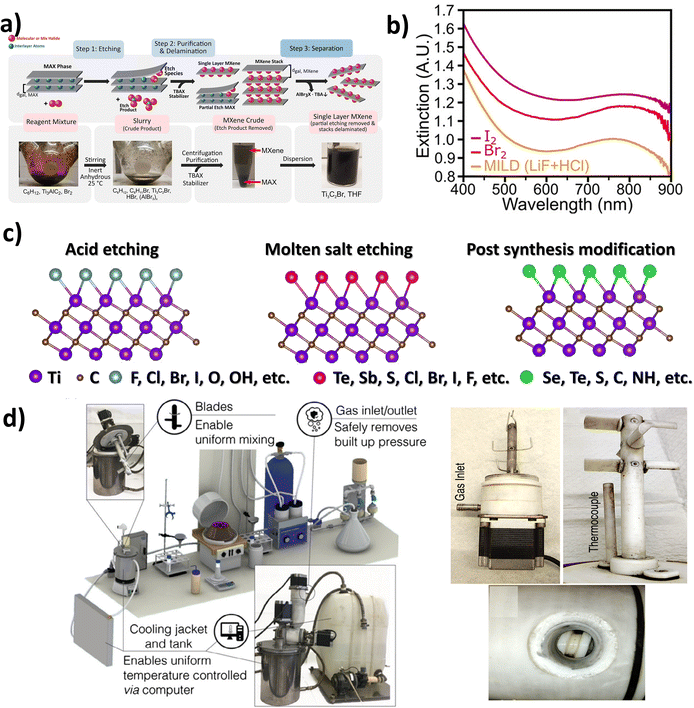 | ||
| Fig. 2 (a) Schematic of the halogen etching of Ti3AlC2 MAX phase and (b) extinction spectra of conventional Ti3C2Tx MXene and halogen-terminated Ti3C2Tx MXenes synthesized via different etching routes, adapted/reproduced from ref. 36 with permission from the American Chemical Society, Copyright 2021. (c) Highlighting the dependence of surface functional groups on the synthesis technique, adapted/reproduced from ref. 37 with permission from Elsevier, Copyright 2025. (d) Photograph of the 1 L batch MXene synthesis system integrated with a cooling tank, adapted/reproduced from ref. 32 with permission from John Wiley and Sons, Copyright 2020. | ||
These approaches remain largely academic due to significant challenges related to cost, safety, scalability, and reproducibility. Beyond the highly reactive and toxic nature of halogen species, a major limitation is that the entire process must be conducted under an inert atmosphere within an isolated glovebox. Moreover, Br2 poses severe corrosion risks to metallic parts of glovebox systems, resulting in unacceptable operational hazards. In addition, the treatment of halogenated waste and the continuous consumption of inert gases further increase operational costs and safety concerns.
Despite significant achievements in the synthesis of Ti3C2Tx MXenes, scaling up their production remains a major challenge that limits their large-scale industrial deployment. Over the past 15 years, several advanced etching strategies have been developed. Among them, in situ HF generation via the minimally intensive layer delamination (MILD) method has been widely adopted due to its relatively simple implementation, high yield, and improved flake quality compared to direct HF etching. However, this approach still generates corrosive wastewater and may lead to surface termination heterogeneity and structural inconsistencies. In contrast, fluoride-free alkaline etching strategies, which aim to synthesize Ti3C2Tx MXenes without fluorine terminations, offer enhanced control over surface chemistry and intrinsic properties while reducing fluorine-related environmental and safety concerns; nevertheless, these methods often require stringent reaction conditions and currently suffer from limited etching efficiency and material yield. Molten salt synthesis represents another promising route that meets basic requirements for MXene formation and enables the production of fluorine-free or termination-tunable MXenes (Fig. 2c); however, its practical application is constrained by high operating temperatures, significant energy consumption, and complex post-treatment processes.
According to the list of precursors required for Ti3AlC2 MAX phase preparation and their corresponding prices from Merck, the estimated cost of the raw precursors for Ti3AlC2 is approximately USD 421 per 100 g, excluding processing, manufacturing, and other associated variables (Table S1). Overall, each conventional synthesis technique presents inherent advantages and drawbacks, including issues related to energy consumption, hazardous byproducts, equipment complexity, and safety concerns. Therefore, the development of green, scalable, and sustainable synthesis strategies is critically important to align Ti3C2Tx MXene production with sustainable development goals and to enable large-scale commercialization for diverse applications (Table 1).
| Synthesis method | Typical yield | Flake size | Electrical conductivity | Defect density | Oxidation stability | Cost | Environmental burden | Scalability |
|---|---|---|---|---|---|---|---|---|
| HF etching | High (>80%) | Large (5–20 µm) | Moderate (5000–10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 S cm−1) 000 S cm−1) |
Moderate | Low–moderate | Low | High (toxic HF) | Good (lab–pilot) |
| Mild (LiF/HCl) | High | Large (5–25 µm) | High (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000–15 000–15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 S cm−1) 000 S cm−1) |
Low–moderate | Moderate | Moderate | Medium | Excellent |
| F-free etching | Moderate | Medium (1–10 µm) | Moderate | Moderate–high | Moderate | Moderate–high | Low | Developing |
| Halogen etching | Moderate | Small–medium | High (low –F terminations) | Low | High | High | High (corrosive gases) | Limited |
| Molten salt etching | Moderate–high | Small–medium | High (low defect, –Cl terminations) | Low | High | Moderate–high | Medium | Promising (industrial potential) |
An overview of the techno-economic analysis of Ti3C2Tx is essential, as this material has attracted significant attention in recent years, accompanied by continuous efforts to develop novel preparation methods, enhance scalability, and promote commercialization and industrial applications that were not comprehensively addressed in earlier assessments. Wet chemical etching routes appear to remain the most cost-effective at the laboratory scale due to their relatively low energy consumption and high yield. However, their industrial translation is constrained by hazardous waste management requirements and regulatory compliance costs. In contrast, fluoride-free and molten-salt strategies reduce environmental burdens but introduce higher energy demands and greater process complexity. In fact, no single synthesis route currently satisfies all sustainability criteria (Table S2). Therefore, efforts should focus on balancing chemical intensity, energy input, operational safety, and product quality through systematic process optimization and green engineering approaches.
Properties of metallic Ti3C2Tx MXene
It is well-known that Ti3C2Tx MXenes have excellent electrical conductivity (11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 S cm−1; monolayer Ti3C2Tx MXene) compared with Ti3C2Tx flakes (4600 S cm−1) due to the d–d orbitals of the Ti atom and rich Tx surface functional groups in Ti3C2Tx MXenes. These electrical properties could help Ti3C2Tx MXene become a good candidate for applications in the fields of energy storage, gas sensing, and EMI shielding.38 Besides, owing to the strong quantum confinement of Ti3C2Tx MXene quantum dots (MQDs) with an average size of around 3–6 nm, they exhibited excitation-dependent PL with high quantum yields (10%). Ti3C2Tx MQDs displayed excellent solubility in both ethanol and DI water due to the strong hydrophilic properties.39 As shown in Fig. 3, MXene-based films could be deformed in various shapes, such as membranes, stamping, and a paper airplane. It is important to know that pure Ti3C2Tx MXenes with a 3D macroporous architecture exhibit superior flexibility and strong moldability.40 Moreover, the electrochemical properties of Ti3C2Tx MXenes in a flexible micro-supercapacitor toward wearable flexible electronics were investigated under bending during operation.41 In particular, Ti3C2Tx MXenes are not stable in the natural environment for long times. The oxidation process was accelerated in the presence of dissolved oxygen, exclusively in water. Ti3C2Tx MXenes can be degraded into TiO2 and methane after a long time of storage, which poses a challenge to the research community. They should be kept in a refrigerator or an inert environment at low temperatures.42
000 S cm−1; monolayer Ti3C2Tx MXene) compared with Ti3C2Tx flakes (4600 S cm−1) due to the d–d orbitals of the Ti atom and rich Tx surface functional groups in Ti3C2Tx MXenes. These electrical properties could help Ti3C2Tx MXene become a good candidate for applications in the fields of energy storage, gas sensing, and EMI shielding.38 Besides, owing to the strong quantum confinement of Ti3C2Tx MXene quantum dots (MQDs) with an average size of around 3–6 nm, they exhibited excitation-dependent PL with high quantum yields (10%). Ti3C2Tx MQDs displayed excellent solubility in both ethanol and DI water due to the strong hydrophilic properties.39 As shown in Fig. 3, MXene-based films could be deformed in various shapes, such as membranes, stamping, and a paper airplane. It is important to know that pure Ti3C2Tx MXenes with a 3D macroporous architecture exhibit superior flexibility and strong moldability.40 Moreover, the electrochemical properties of Ti3C2Tx MXenes in a flexible micro-supercapacitor toward wearable flexible electronics were investigated under bending during operation.41 In particular, Ti3C2Tx MXenes are not stable in the natural environment for long times. The oxidation process was accelerated in the presence of dissolved oxygen, exclusively in water. Ti3C2Tx MXenes can be degraded into TiO2 and methane after a long time of storage, which poses a challenge to the research community. They should be kept in a refrigerator or an inert environment at low temperatures.42
 | ||
| Fig. 3 (a–c and g) Photographs of stamped MXene microsupercapacitors with different device architectures. (d–f) SEM images of the interdigitated electrodes on the corresponding substrates. (h) Photographs of the device under four different bending states and the corresponding resistance changes, adapted/reproduced from ref. 53 with permission from John Wiley and Sons, Copyright 2018. | ||
DFT calculations indicate that the work function of metallic MXene is approximately −5.35 eV, which decreases to −6.88 eV after a light oxidation process. The HOMO and LUMO levels are located at approximately −7.23 eV and −3.26 eV, respectively. The DFT-based calculations of the energy levels of MXenes after their oxidation confirm that they exhibit semiconducting behavior.26
Tables 2 and S3 summarize the physical and chemical properties of MXenes, which are compared with those of other representative 2D materials. Although several experimental techniques are available to characterize the structure, morphology, and composition of materials, these methods are often time-consuming and costly. Therefore, theoretical calculations are expected to provide efficient and accurate analyses that support a deeper understanding of the physicochemical properties of MXenes and their derivatives, particularly with respect to interfacial properties and relative stability.
| Category | Property | Typical values |
|---|---|---|
| Chemical stability | pH stability | Stable in mild acidic/neutral media43 |
| Oxidation sensitivity | Degrades in O2, light, high humidity44 | |
| Adsorption properties | Surface area | ∼10–100 m2 g−1 (ref. 45) |
| Adsorption sites | Ti–O, Ti–OH, defects46 | |
| Chemical properties | Hydrophilicity | Strongly hydrophilic47 |
| Chemical reactivity | Easily oxidized (TiO2 formed in air/water)48 | |
| Redox activity | Ti4+/Ti3+ surface states49 | |
| Mechanical properties | Young's modulus | ∼330 GPa (monolayer, theoretical)50 |
| Flexibility | High (bendable films)50 | |
| Thermal properties | Thermal conductivity | ∼10–100 W m−1 K−1 (ref. 51) |
| Electronic properties | Conductivity | ∼103–104 S cm−1 (ref. 52) |
| Band structure | Metallic to semi-metallic52 | |
| Work function | ∼4.6–5.2 eV (Tx dependent)52 | |
| Surface chemistry | Terminal groups | –O, –OH, –F, –Cl, –Br, –I…37 |
| Surface charge | Negative zeta potential (−35 mV)43 |
A recent study by Mostafa's group evaluated the environmental impacts and cumulative energy demand associated with MXene synthesis from gram- to kilogram-scale production. Their results indicate that more than 70% of the total environmental impact arises from electricity consumption. At the laboratory scale, the production of 1000 g of MXene releases approximately 428.1 kg of CO2, whereas the industrial-scale manufacture of 1 kg of copper foil results in only ∼8.75 kg of CO2 emissions.54 This substantial discrepancy is primarily attributed to intensive chemical consumption and the associated wastewater treatment processes. To mitigate these impacts, the authors proposed synthesizing secondary MXenes from MAX phases derived from secondary precursors, such as tire-recycled carbon, aluminum scrap, and titanium dioxide. Notably, the resulting secondary MXenes exhibit competitive electrical conductivity (5857 ± 680 S cm−1), comparable to that of conventionally synthesized MXenes.55 Furthermore, the integration of artificial intelligence and computational modeling is expected to play a critical role in optimizing synthesis parameters for secondary MXenes. The millions of tons of titanium and aluminum scrap worldwide offer promising precursors and a pathway toward the scalable, low-cost, and sustainable production of high-quality MXenes.
Applications of Ti3C2Tx MXenes and Ti3C2Tx MXene-based composites
Scientists worldwide have increasingly focused on the development of multifunctional materials capable of delivering high performance across diverse application domains. In this context, Ti3C2Tx MXene-based nanocomposites have emerged as a potential material platform due to their unique properties. These attributes enable their effective deployment in strategic applications, including energy storage and conversion, gas sensing, and water splitting, which are closely linked to national energy security and technological competitiveness. Simultaneously, Ti3C2Tx MXene nanocomposites offer substantial benefits for societal applications, such as soft tissue regeneration and environmental remediation, addressing pressing challenges related to sustainability, public health, and environmental protection. Therefore, systematic investigation of Ti3C2Tx MXenes and their nanocomposites is crucial to fully exploit their multifunctionality and to bridge strategic technological advancement with societal needs.Ti3C2Tx based platforms for soft tissue regeneration
The versatility of Ti3C2Tx nanostructures allows for their composites to be multifunctional systems, especially in bone tissue regeneration and engineering.56,57 For example, Ti3C2Tx-based platforms can be combined with biomaterials to construct composite scaffolds with tailored functionalities and improved mechanical behaviors. These optimized nanostructures offer synergistic effects, integrating the strength of each compound to provide specific tissue regeneration demands.58–60 Ti3C2Tx-based composites can also be integrated with drug delivery systems, improving the controlled release of therapeutics to enhance tissue regeneration results.Although Ti3C2Tx nanostructures have many unique properties and inherent hydrophilic behavior,61 tailoring these properties to suit the specific demands of bone and soft tissue regeneration is a challenging and sophisticated goal. It is important to fully understand the fundamentals of the structural properties of Ti3C2Tx MXenes and how to control their operational parameters. In other words, the integrated bone-soft tissue regeneration facilitated by Ti3C2Tx MXenes offers several advantages, such as improved healing, reduced risk of infection, and targeted drug delivery. The integrated thermal effects of photoactivated Ti3C2Tx MXenes have been shown to enhance tissue regeneration, providing a potential technique for tissue repair. As shown in Fig. 4, Xiaoyan and co-workers have demonstrated that a photoactivated Ti3C2Tx MXene exerts positive effects on the improvement of soft and bone tissue regeneration. The photoactivated Ti3C2Tx MXene displayed not only a strong thermal effect but also a robust antibacterial performance, effectively modulating the expression of inflammation factors (methicillin-resistant Staphylococcus aureus).62 Moreover, this technique enables the regulation of the osteogenic differentiation of adipose-derived stem cells through heat shock protein 70 and an extracellular signal-regulated kinase signaling pathway. However, despite these good outcomes, their study has some limitations. On the one hand, scale-up of high-quality Ti3C2Tx nanostructures remains a challenge for the science community.63 Also, another difficulty is controlling the surface termination groups (–OH, –O, and –Cl) and preventing their rapid accumulation in biological media.64 On the other hand, long-term storage of non-oxidized Ti3C2Tx nanosheets is difficult to control,65,66 and the toxicological investigation of Ti3C2Tx nanostructures in vivo must be performed systematically.
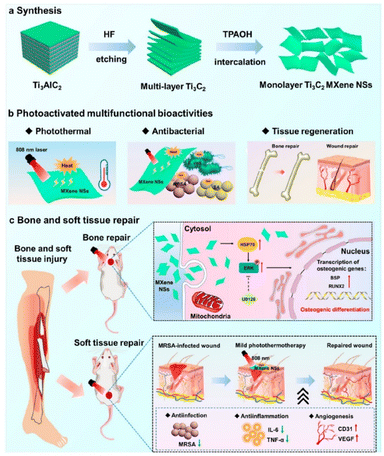 | ||
| Fig. 4 (a) Synthesis procedure, (b) multifunctional bioactivities, and (c) photoactivated Ti3C2Tx nanostructures for biomedical applications, adapted/reproduced from ref. 62 with permission from the American Chemical Society, Copyright 2023. | ||
Based on the reported achievements of MXene-based composites in soft and bone tissue regeneration (Table 3), several key research directions remain to be addressed to further enhance osteogenic differentiation and cell adhesion within scaffolds. These include (i) mimicking the native bone architecture to provide appropriate structural and biochemical cues; (ii) improving mechanical properties to better match those of natural bone; (iii) enhancing osteoinductive capacity to promote stem cell differentiation; (iv) facilitating vascularization and efficient nutrient and waste transport; (v) ensuring long-term stability and seamless integration with host tissue; (vi) achieving scalable and cost-effective fabrication; and (vii) advancing clinical translation through standardized evaluation and regulatory approval.
| Ti3C2Tx-based platform | Applications and representative functions | Main advantages | Ref. |
|---|---|---|---|
| Ti3C2Tx MXene | - Skin wound healing | - Good cytocompatibility and photothermal conversion | 67 and 68 |
| - Accelerated wound closure; infection suppression | - High antibacterial activity | ||
| Ti3C2Tx-incorporated hydrogel | - Skin and soft tissue repair | - Injectable and flexible | 69 and 70 |
| - Promotes angiogenesis and tissue remodeling | - Enhanced cell adhesion and proliferation | ||
| Ti3C2Tx-polymer composite | - Tissue engineering | - High electrical conductivity | 71 and 72 |
| - Enhances fibroblast migration and epithelialization | - Mechanical robustness | ||
| - Moisture retention | |||
| Ti3C2Tx-poly(lactic acid) | - Bone tissue regeneration and engineering | - Optimization NO release | 73 |
| - High photothermal effects | |||
| - Suitable tensile strength | |||
| Ti3C2Tx-PLCL/collagen | - Spontaneous osteogenic differentiation. Bone tissue regeneration and engineering | - The growth of preosteoblasts | 74 |
| - NIR photon absorption | |||
| - High biocompatibility stability |
Ti3C2Tx MXene for energy harvesting
Recently, Shrabani and colleagues reported the synthesis of a WS2-covered Ti3C2Tx MXene/boron nitride (BN) composite for supercapacitors using the hydrothermal technique at 180 °C for 12 h, as shown in Fig. 5.75 Here, the multilayered Ti3C2Tx MXene was composed of stacked layers of BN and formed homogeneous dispersions of the MBN binary composite. The optimum electrode revealed a high specific capacitance (SC) of 1318 F g−1 in 1 M KOH at 1 A g−1, which displayed an excellent cyclic life after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 charge and discharge runs (retaining around 84% of its initial capacitance). The synthesized composite showed specific energy density and capacitance values of 19.5 Wh kg−1 and 140 F g−1, respectively, using commercial PVA-KOH-KI gel as the electrolyte. The strong interactions between individual components were the main factor contributing to these excellent results.
000 charge and discharge runs (retaining around 84% of its initial capacitance). The synthesized composite showed specific energy density and capacitance values of 19.5 Wh kg−1 and 140 F g−1, respectively, using commercial PVA-KOH-KI gel as the electrolyte. The strong interactions between individual components were the main factor contributing to these excellent results.
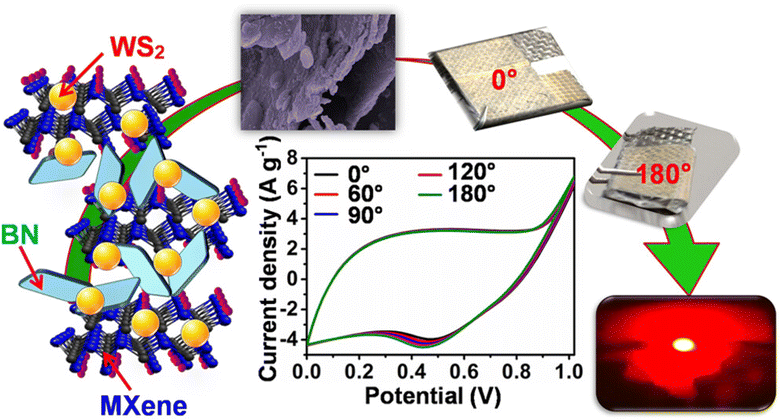 | ||
| Fig. 5 WS2/boron nitride/Ti3C2Tx MXene binary composite nanostructure for supercapacitor applications, adapted/reproduced from ref. 75 with permission from the American Chemical Society, Copyright 2023. | ||
To support and interpret the experimental results, Xiaofeng employed first-principles calculations to optimize the band energy, density of states, and crystal structure of Ti3C2Tx MXene.76 As depicted in Fig. 6, the optimized structure of MXene is Ti3C2Tx (Tx = O), with the main band structures of p-orbitals originating from C and O atoms, while d-orbitals originate from Ti.77 These optimized crystal structures indicate that Ti3C2Tx possesses higher conductivity, making it suitable for high cyclability and good rate capability. Fig. 6d illustrates the favourable adsorption sites for Na+ ions based on the charge density difference. This calculation demonstrates that Na+ ions adsorbed on the surface of MXene lose charges, which contributes to the charge acceptance of the electrode system. The results of the charge density difference reveal that Na+ ions are favorable for facilitating the flexible loss of charges in the electrode.
 | ||
| Fig. 6 (a–c) Optimized crystal structure, density of states and band structure of Ti3C2Tx (Tx = O). (d) Crystal structure with suitable adsorption sites. (e and f) Charge density difference and adsorption energy of Na+ ion on the surface of Ti3C2Tx. (g–i) Diffusion paths and diffusion energy plots, adapted/reproduced from ref. 76 with permission from Elsevier, Copyright 2024. | ||
Overall, MXenes exhibit competitive performance relative to graphene and TMDs due to their unique combination of physicochemical properties and hybrid charge-storage mechanisms involving both electric double-layer capacitance and pseudocapacitance (Table 4).78 Among these materials, graphene currently offers advantages in terms of cost and long-term stability, making it more suitable for near-term commercial applications. At the laboratory scale, the production cost of MXenes is estimated to be approximately USD 20.33 per gram in 2024,79 indicating that further cost reduction is required before their large-scale commercialization. In contrast, the global graphite market is projected to expand from USD 8.32 billion in 2025 to USD 13.35 billion by 2032.80 Nevertheless, the MXene market is expected to reach approximately USD 121.5 million by 2027,81 reflecting growing industrial interest and the potential for MXenes to emerge as a competitive class of materials once scalable and cost-effective synthesis strategies are fully matured.
| Property | Ti3C2Tx MXenes | Graphene | TMDs (MoS2) |
|---|---|---|---|
| Charge storage mechanism | Hybrid EDLC and pseudocapacitance | Primarily EDLC | Predominantly pseudocapacitance and ion intercalation |
| Gravimetric capacitance | 400–1500 F g−1 | 100–550 F g−1 | 100–400 F g−1 |
| Volumetric capacitance | Up to 1500 F cm−3 | Up to 550 F cm−3 | Typically, lower than MXenes |
| Electrolyte accessibility | Interlayer ion transport | Surface-limited | Limited by slow intercalation kinetics |
Ti3C2Tx MXenes for gas sensors
In 2023, Seyed and colleagues reported the excellent gas sensing performance of Ti3C2Tx MXenes and their composites with PDS-Cl conjugated polymers.82 The strong electrostatic interactions between Ti3C2Tx MXenes and polymers formed several heterojunction interfaces via hydrogen bonding and ion–dipole interactions, which are beneficial for gas-sensing activities. In particular, the optimized composite displayed the highest sensing signal of around 2% upon exposure to 5 ppm of H2S gas in comparison to pure Ti3C2Tx MXene, as shown in Fig. 7. Moreover, the noise level in the optimal composite was higher than that in the pure Ti3C2Tx MXene, which is attributed to the fact that the polymer modifies the charge transport between each layer.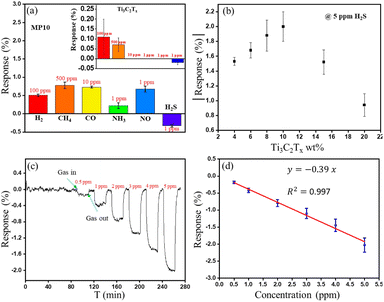 | ||
| Fig. 7 (a–d) H2S sensing response values of Ti3C2Tx MXene and Ti3C2Tx MXene-organic heterostructure (the inset presents the gas selectivity of pure MXene), adapted/reproduced from ref. 82 with permission from the American Chemical Society, Copyright 2023. | ||
Moreover, Ta et al. reported the most stable configuration of a Ti3C2Tx/MoS2 sensor composite through density functional theory calculation using the Vienna Ab initio Simulation Package, as shown in Fig. 8. Ti3C2Tx (Tx = OH) and Ti3C2Tx (Tx = F) are less stable than Ti3C2Tx (Tx = O) according to the DFT calculations of the Ti3C2Tx MXene surfaces.83 In particular, the stability of these configurations differs depending on the types of gas molecules. The adsorption of NO2 molecules on the composite surface was estimated to be stronger than that of CH4 or CO2, which explains the good sensing performance of the composite toward NO2 molecules (response 40%; 20 ppm of NO2). Specifically, the stronger adsorption of NO2 compared to CO2 and CH4 originates from the formation of strong interactions at the composite interface, which are absent or significantly weaker for other gases. These interactions promote pronounced charge redistribution at the MoS2 surface, leading to a larger modulation of the electronic structure and consequently a stronger sensing response. In general, beyond identifying adsorption configurations with weak or strong interactions, DFT analysis elucidates the binding mechanisms between the target gas molecules and the composite surface, distinguishing between physisorption and chemisorption. Gas selectivity is predicted by comparing adsorption energy values, the formation of localized active adsorption sites, and the density of states near the Fermi level of the sensor composites. These insights provide fundamental guidance for rational material design and the selection of suitable sensing gases.
 | ||
| Fig. 8 (ai–iv) Optimized structures and (bi–iv) stable configurations of the as-prepared Ti3C2Tx/MoS2 sensor composite when exposed to NO2 molecules, adapted/reproduced from ref. 84 with permission from Elsevier, Copyright 2022. | ||
Ti3C2Tx MXenes for photo-electrochemical water splitting and triboelectric nanogenerator
Adem et al.85 prepared a novel binary Ti3C2Tx/WO3/ZnO composite using post-annealing procedures and radio frequency sputtering and studied its photoelectrochemical (PEC) performance under solar light irradiation, demonstrating superior photoconversion efficiency. According to the results, at +0.4 V vs. RHE in 0.1 M KOH electrolyte, the optimal Ti3C2Tx/WO3/ZnO binary heterostructure enhanced the photocurrent generation (1.4 × 10−3 A cm−2) in comparison with pristine ZnO (7.8 × 10−4 A cm−2) and the WO3/ZnO composite (1.1 × 10−3 A cm−2). As shown in Fig. 9, the superior PEC performance achieved by the Ti3C2Tx/WO3/ZnO binary composite can be explained by the formation of a Schottky barrier and its high charge-carrier separation efficiency.85 | ||
| Fig. 9 ZnO/WO3 composites interfaced with metallic Ti3C2Tx MXene for enhanced photoelectrochemical water splitting, adapted/reproduced from ref. 85 with permission from Elsevier, Copyright 2023. | ||
Recently, Shaochun et al. reported that the strength of the Ti3C2Tx MXene/substrate interface substantially affects the electrical and mechanical properties of Ti3C2Tx MXene films (Fig. 10). First, the Ti3C2Tx MXene was covered on a leather substrate, and then, polyimide (PI) with a thickness of 100 µm was deposited as the electrode via laser technology.86 Finally, a flexible triboelectric tactile sensor was obtained after forming an Au circuit in HAuCl4 solution (5 mg in 1 mL). Interestingly, the as-prepared MXene film displayed good activity in the TENG with the highest output voltage and output power density of 199.56 V and 0.469 mW cm−2, respectively. During the application of external force during movement, charges were generated and induced on PI owing to the different electron affinities. The electrons in the negatively charged Ti3C2Tx MXene move to the load when the force is released owing to the edge electric-field leakage effect.87 This process is gradually repeated in two friction layers and continuously releases electrons to the load, resulting in an alternating voltage. The flexible robust Ti3C2Tx MXene/leather films showed excellent cycling performance with long-time operation (more than 1 hour), which can be used in smart wearable and health monitoring devices.88,89
 | ||
| Fig. 10 (a–d) Schematic of a flexible TENG based on a robust Ti3C2Tx MXene/leather PI film for human-machine movement, adapted/reproduced from ref. 86 with permission from the American Chemical Society, Copyright 2023. (e) Global MXene market forecast to 2033 (adapted from the MXene Market Insights research report https://www.24chemicalresearch.com/reports/283167/global-mxene-market). | ||
In general, the design of MXene-based catalysts for overall energy harvesting has mainly focused on heterostructure engineering, alloying, and electronic structure optimization at the laboratory scale. However, studies addressing the influence of the electric double layer at the catalyst–electrolyte interface and ion transfer across different components in composite catalysts remain limited. A deeper understanding of these interfacial processes is essential for achieving performance breakthroughs and enabling large-scale applications. In fact, current techniques still face substantial challenges that hinder practical scale-up. In detail, hydrogen storage during water-splitting processes often relies on ultra-low-temperature tanks, which are costly and energy-intensive. In addition, freeze-drying procedures commonly used for MXene processing are time-consuming, and the high batch-scale production cost of MXenes remains a critical concern. Furthermore, MXene powders used in photo- and electrocatalytic applications must typically be redispersed into colloidal solutions, where oxidation and stability issues arise. The use of organic solvents further complicates storage and necessitates post-treatment and waste-liquid recovery. It is anticipated that with continued technological advancements, these challenges can be gradually addressed, enabling MXene-based water-splitting systems to reach pilot-scale operation by 2050.
Ti3C2Tx MXenes for photocatalysis and water remediation
Industry-released wastewater commonly contains a wide range of toxic contaminants, including pesticides, organic dyes, pharmaceuticals, and other harmful aqueous pollutants. Ti3C2Tx MXenes have been widely recognized as promising materials for photocatalysis and water remediation due to their outstanding physicochemical properties, such as high adsorption capacity, unique layered structure, and abundant surface functional groups. In water treatment systems, Ti3C2Tx MXenes play a crucial role as membrane materials, adsorbents, and catalytic supports. For example, the multilayered Ti3C2Tx MXene structure, featuring interlayer channels of approximately 6 Å, enables the efficient removal of organic contaminants.90 Several studies have reported the development of MXene-based membranes for methylene blue removal, achieving exceptionally high adsorption capacities of up to 28![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 403.7 mg g−1.91 Furthermore, reinforcement with cellulose nanofibers effectively suppresses membrane swelling, thereby enhancing structural stability and enabling the efficient removal of antibiotics such as tetracycline, azithromycin, and penicillin G potassium salt.92
403.7 mg g−1.91 Furthermore, reinforcement with cellulose nanofibers effectively suppresses membrane swelling, thereby enhancing structural stability and enabling the efficient removal of antibiotics such as tetracycline, azithromycin, and penicillin G potassium salt.92
In addition to adsorption-based remediation, MXenes exhibit an excellent photocatalytic degradation performance under light irradiation. Zeng and co-workers demonstrated the highly efficient degradation of Congo red (98%) and tetracycline hydrochloride (86%) using hierarchical g-C3N4/Ti3C2Tx/polyethersulfone composites.93 The enhanced degradation efficiency was primarily attributed to the improved light-harvesting capability and the effective suppression of electron–hole recombination achieved through optimized interfacial coupling between g-C3N4 and MXene/polyethersulfone.93 Overall, water remediation via adsorption and photo-induced radical pathways using Ti3C2Tx MXenes and their composites has shown significant progress recently, which is systematically summarized in Table 5.
| Catalysts | Pollutants | Reaction time | Proposed mechanism | Efficiency | Ref. |
|---|---|---|---|---|---|
| Ti3C2Tx | Ciprofloxacin (10 ppm) | 20 min | Adsorption/electrochemical regeneration | 208.2 mg g−1 | 94 |
| Al2O3/Ti3C2Tx | Rhodamine B (15 ppm) | 60 min | Adsorption | 99.8% | 95 |
| Methylene blue (15 ppm) | 60 min | 99.9% | |||
| Bi4Ti3O12/Ti3C2Tx | Rhodamine B (5 ppm) | 50 min | Photocatalytic degradation | 100% | 96 |
| Methyl orange (5 ppm) | 50 min | 100% | |||
| Fe/C/Ti3C2Tx | Methylene blue (4000 ppm) | 30 min | Photocatalytic degradation | 81.2% | 97 |
| CuNiAl/Ti3C2Tx | Tetracycline (20 ppm) | 80 min | Photocatalytic degradation | 90.3% | 98 |
Conclusions
In this review, we have basically examined the synthesis strategies and multifunctional applications of Ti3C2Tx MXenes across energy, environmental, biomedical, and sensing-related fields. Fluoride-based etching approaches generally enable the production of Ti3C2Tx MXenes with relatively large lateral flake sizes and preserved mechanical integrity, which are advantageous for flexible electrodes and structural composites. In contrast, fluoride salt-based and HF aqueous etching methods typically yield smaller flakes and richer surface terminations, which can enhance electrochemical activity but often compromise oxidation stability and long-term durability. These synthesis-dependent trade-offs underscore the necessity of tailoring Ti3C2Tx MXene preparation routes to specific application requirements rather than pursuing a general fabrication strategy.Since the initial discovery of Ti3C2Tx MXenes, substantial progress has been achieved in both synthesis control and composite design, establishing Ti3C2Tx MXenes as a versatile two-dimensional platform. Nevertheless, several fundamental challenges remain. In particular, oxidation instability under humid or ambient conditions, limited control over surface terminations, batch-to-batch reproducibility, and safety and sustainability concerns associated with fluorine-containing etchants continue to restrict reliable performance and large-scale deployment. Addressing these issues requires a deeper mechanistic understanding of surface chemistry, defect evolution, and interfacial and financial processes under realistic operating environments. According to the MXene Materials Market Insights report, the global MXene market is projected to reach approximately USD 244.38 million by 2032, corresponding to a compound annual growth rate of 22.7% (Fig. 10e).
From an application perspective, Ti3C2Tx MXenes have demonstrated notable potential in biomedical regeneration, energy harvesting, and environmental remediation, especially where high electrical conductivity and surface reactivity are critical. The translation of laboratory-scale demonstrations into practical technologies remains constrained by energy-intensive processing steps, high production costs, and stability challenges during storage and device integration. In this context, emerging strategies such as fluoride-free or green synthesis, secondary Ti3C2Tx MXene production from recycled precursors, and machine learning-assisted modelling to optimize synthesis and composite architectures represent promising pathways toward more sustainable and scalable manufacturing.
Controlling product development through stepwise phases of technological advancement remains both an opportunity and a challenge for MXene-based technologies, and this systematic progression has not yet been extensively explored. In terms of technological readiness level (TRL), current MXene research is generally considered to be at TRL 1–2, with some studies approaching TRL 3–4, as no commercially available devices or large-scale applications based on MXenes have yet reached the market. Therefore, replacing conventional materials or integrating MXenes into existing systems represents an important step toward advancing their technological maturity. Achieving higher TRLs will require systematic prototyping and comprehensive evaluation of complete systems or components to demonstrate clear advantages over state-of-the-art commercial materials. Furthermore, establishing clearer commercialization pathways will depend on coordinated progress in synthesis strategies, surface termination control, structural and electronic property optimization, and application-specific performance, which together form the foundation for further advancement of MXene technologies along the TRL scale.32
Looking forward, meaningful progress toward commercialization will depend on coordinated advances in synthesis standardization, lifecycle assessments, and device-level validation rather than incremental performance improvements alone. Currently, Ti3C2Tx MXenes are unlikely to fully replace traditional materials such as graphene; continued improvements in stability, cost-efficiency, and reproducibility could enable their adoption in high-value applications in energy harvesting and environmental protection (reducing the carbon footprint) within the next one to two decades. Overall, this review highlights both the opportunities and the limitations of Ti3C2Tx MXenes and provides a fundamental framework for future research aimed at advancing MXene-based technologies toward reliable, sustainable, and application-ready systems.
Author contributions
Ly Tan Nhiem: figure organization, manuscript preparation.Conflicts of interest
There are no conflicts to declare.Data availability
No primary research results, software or code have been included, and no new data were generated or analysed as part of this review.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5ra08486e.
Acknowledgements
The author would like to thank Ho Chi Minh City University of Technology and Engineering for its support.References
- T. K. Pardhi, V. Balaji, S. Sunil, G. Arumugam, V. Naveen, P. Porkodi, A. J. Kottiyatil, K. Madhavan and P. Ghosh, Sci. Cult., 2025, 91, 37–48 CrossRef.
- E. Pavón, R. Martín-Rodríguez, A. C. Perdigón and M. D. Alba, Inorganics, 2021, 9, 43 CrossRef.
- N. Veeramani, M. K. Chaithra, G. Santhana Krishnan, Kalavati and N. T. Manikandanath, Polym. Eng. Sci., 2025, 66, 1091–1098 CrossRef.
- D. S. Nakonieczny, G. S. Martynková, M. Hundáková, G. Kratošová, S. Holešová, J. Kupková, L. Pazourková and J. Majewska, Materials, 2022, 15, 1390 CrossRef CAS PubMed.
- V. Naveen, A. P. Deshpande and S. Raja, RSC Adv., 2020, 10, 33178–33188 RSC.
- M. Naguib, O. Mashtalir, J. Carle, V. Presser, J. Lu, L. Hultman, Y. Gogotsi and M. W. Barsoum, ACS Nano, 2012, 6, 1322–1331 CrossRef CAS PubMed.
- X.-F. Yu, Y.-C. Li, J.-B. Cheng, Z.-B. Liu, Q.-Z. Li, W.-Z. Li, X. Yang and B. Xiao, ACS Appl. Mater. Interfaces, 2015, 7, 13707–13713 CrossRef CAS PubMed.
- W. Bai, L. Shi, Z. Li, D. Liu, Y. Liang, B. Han, J. Qi and Y. Li, Mater. Today Energy, 2024, 41, 101547 CrossRef CAS.
- S. Vallem, S. Venkateswarlu, S. Song, J. Kim, Y. Li and J. Bae, Coord. Chem. Rev., 2024, 510, 215778 CrossRef CAS.
- M. Ghidiu, M. R. Lukatskaya, M. Q. Zhao, Y. Gogotsi and M. W. Barsoum, MXenes: from Discovery to Applications of Two-Dimensional Metal Carbides and Nitrides, 2023, pp. 379–399 Search PubMed.
- X. Li, Y. Bai, X. Shi, N. Su, G. Nie, R. Zhang, H. Nie and L. Ye, Mater. Adv., 2021, 2, 1570–1594 RSC.
- L. Verger, V. Natu, M. Carey and M. W. Barsoum, Trends Chem., 2019, 1, 656–669 CrossRef CAS.
- V. T. T. Linh, N. P. T. Trieu, Q. T. H. Ta, N. N. Tri, V. N. Huynh, D. T. Y. Oanh, N. H. Hieu and S. M. Seo, Vietnam J. Chem., 2026, 1 DOI:10.1002/vjch.70129.
- G. Deysher, C. E. Shuck, K. Hantanasirisakul, N. C. Frey, A. C. Foucher, K. Maleski, A. Sarycheva, V. B. Shenoy, E. A. Stach, B. Anasori and Y. Gogotsi, ACS Nano, 2020, 14, 204–217 CrossRef CAS PubMed.
- M. A. Zaed, K. H. Tan, R. Saidur, N. Abdullah and A. K. Pandey, J. Mater. Sci., 2024, 59, 7575–7594 CrossRef CAS.
- N. M. Tran, Q. T. H. Ta, A. Sreedhar and J.-S. Noh, Appl. Surf. Sci., 2021, 537, 148006 CrossRef.
- Y. Fan, Z. Yuan, G. Zou, Q. Zhang, B. Liu and Q. Peng, Catal. Today, 2018, 318, 167–174 CrossRef CAS.
- X. Zhao, A. Vashisth, E. Prehn, W. Sun, S. A. Shah, T. Habib, Y. Chen, Z. Tan, J. L. Lutkenhaus, M. Radovic and M. J. Green, Matter, 2019, 1, 513–526 CrossRef.
- W. Y. Chen, S.-N. Lai, C.-C. Yen, X. Jiang, D. Peroulis and L. A. Stanciu, ACS Nano, 2020, 14, 11490–11501 CrossRef CAS PubMed.
- W. Cao, J. Nie, Y. Cao, C. Gao, M. Wang, W. Wang, X. Lu, X. Ma and P. Zhong, Chem. Eng. J., 2024, 496, 154097 CrossRef CAS.
- A. Sreedhar, P. Ravi and J.-S. Noh, J. Mater. Sci. Technol., 2024, 203, 237–254 CrossRef CAS.
- M. A. Zaed, J. Paul, S. Aktar, J. Jacob, K. H. Tan and P. Thomas, Energy Nexus, 2026, 21, 100625 CrossRef CAS.
- P. Kuang, J. Low, B. Cheng, J. Yu and J. Fan, J. Mater. Sci. Technol., 2020, 56, 18–44 CrossRef CAS.
- S. Myhra, J. A. A. Crossley and M. W. Barsoum, J. Phys. Chem. Solids, 2001, 62, 811–817 CrossRef CAS.
- M. Naguib, M. Kurtoglu, V. Presser, J. Lu, J. Niu, M. Heon, L. Hultman, Y. Gogotsi and M. W. Barsoum, Adv. Mater., 2011, 23, 4248–4253 CrossRef CAS PubMed.
- L. Yang, D. Kan, C. Dall'Agnese, Y. Dall'Agnese, B. Wang, A. K. Jena, Y. Wei, G. Chen, X. F. Wang, Y. Gogotsi and T. Miyasaka, J. Mater. Chem. A, 2021, 9, 5016–5025 RSC.
- P. Das and Pooja, Hazardous Gases: Risk Assessment on the Environment and Human Health, 2021, pp. 153–167 Search PubMed.
- D. Masekela, P. J. Mafa, T. L. Yusuf, S. A. Balogun, A. T. Kuvarega and K. D. Modibane, Coord. Chem. Rev., 2026, 549, 217270 CrossRef CAS.
- A. Mashele, N. S. Seroka and L. Khotseng, Renewable Sustainable Energy Rev., 2026, 226, 116373 CrossRef CAS.
- A. Iqbal, T. Hassan, S. M. Naqvi, Y. Gogotsi and C. M. Koo, Nat. Rev. Electr. Eng., 2024, 1, 180–198 CrossRef.
- M. Downes, C. E. Shuck, B. McBride, J. Busa and Y. Gogotsi, Nat. Protoc., 2024, 19, 1807–1834 CrossRef CAS PubMed.
- C. E. Shuck, A. Sarycheva, M. Anayee, A. Levitt, Y. Zhu, S. Uzun, V. Balitskiy, V. Zahorodna, O. Gogotsi and Y. Gogotsi, Adv. Eng. Mater., 2020, 22, 1901241 CrossRef CAS.
- A. Lipatov, M. Alhabeb, M. R. Lukatskaya, A. Boson, Y. Gogotsi and A. Sinitskii, Adv. Electron. Mater., 2016, 2, 1600255 CrossRef.
- C. J. Zhang, S. Pinilla, N. McEvoy, C. P. Cullen, B. Anasori, E. Long, S. H. Park, A. Seral-Ascaso, A. Shmeliov, D. Krishnan, C. Morant, X. Liu, G. S. Duesberg, Y. Gogotsi and V. Nicolosi, Chem. Mater., 2017, 29, 4848–4856 CrossRef CAS.
- M. Alhabeb, K. Maleski, B. Anasori, P. Lelyukh, L. Clark, S. Sin and Y. Gogotsi, Chem. Mater., 2017, 29, 7633–7644 CrossRef CAS.
- A. Jawaid, A. Hassan, G. Neher, D. Nepal, R. Pachter, W. J. Kennedy, S. Ramakrishnan and R. A. Vaia, ACS Nano, 2021, 15, 2771–2777 CrossRef CAS PubMed.
- N. Kumar, Q. V. Hoang, B. Mohammad, K. R. Kaja, P. H. Nguyen, Q. L. Van, V. Vien, V. T. T. Linh, P. K. T. Nguyen and Q. T. H. Ta, J. Sci.:Adv. Mater. Devices, 2025, 4, 101034 Search PubMed.
- A. Lipatov, A. Goad, M. J. Loes, N. S. Vorobeva, J. Abourahma, Y. Gogotsi and A. Sinitskii, Matter, 2021, 4, 1413–1427 CrossRef CAS.
- Q. Xue, H. Zhang, M. Zhu, Z. Pei, H. Li, Z. Wang, Y. Huang, Y. Huang, Q. Deng, J. Zhou, S. Du, Q. Huang and C. Zhi, Adv. Mater., 2017, 29, 1604847 CrossRef PubMed.
- J. Zhang, N. Kong, S. Uzun, A. Levitt, S. Seyedin, P. A. Lynch, S. Qin, M. Han, W. Yang, J. Liu, X. Wang, Y. Gogotsi and J. M. Razal, Adv. Mater., 2020, 32, 2001093 CrossRef CAS PubMed.
- C. Zhang, B. Anasori, A. Seral-Ascaso, S.-H. Park, N. McEvoy, A. Shmeliov, G. S. Duesberg, J. N. Coleman, Y. Gogotsi and V. Nicolosi, Adv. Mater., 2017, 29, 1702678 CrossRef PubMed.
- M. W. Barsoum and Y. Gogotsi, Ceram. Int., 2023, 49, 24112–24122 CrossRef CAS.
- V. Natu, M. Sokol, L. Verger and M. W. Barsoum, J. Phys. Chem. C, 2018, 122, 27745–27753 CrossRef CAS.
- Z. Khalid, F. Hadi, J. Xie, V. Chandrabose and J.-M. Oh, Small, 2025, 21, 2407856 CrossRef CAS PubMed.
- Z. Cai and H. Kim, npj 2D Mater. Appl., 2025, 9, 66 CrossRef CAS.
- K. K. Swain, S. K. Pradhan and D. J. Late, ChemistrySelect, 2025, 10, e202404885 CrossRef CAS.
- S. Lee, E. Yang, J. Lee, T. Y. Ko and S. J. Kim, Adv. Mater., 2025, 37, 2502440 CrossRef CAS PubMed.
- G. Bhuvaneshwari, N. Ponpandian and C. Viswanathan, Surf. Interfaces, 2025, 67, 106583 CrossRef CAS.
- M. Y. Bhat, W. A. Adeosun, K. Prenger, Y. A. Samad, K. Liao, M. Naguib, S. Mao and A. Qurashi, Adv. Compos. Hybrid Mater., 2024, 8, 52 CrossRef.
- C. I. Idumah, Polym.-Plast. Technol. Mater., 2023, 62, 443–466 CAS.
- M. Safarkhani, B. F. Far, Y. S. Huh and N. Rabiee, ACS Biomater. Sci. Eng., 2023, 9, 6516–6530 CrossRef CAS PubMed.
- C. Qiao, H. Wu, X. Xu, Z. Guan and W. Ou-Yang, Adv. Mater. Interfaces, 2021, 8, 2100903 CrossRef CAS.
- C. J. Zhang, M. P. Kremer, A. Seral-Ascaso, S.-H. Park, N. McEvoy, B. Anasori, Y. Gogotsi and V. Nicolosi, Adv. Funct. Mater., 2018, 28, 1705506 CrossRef.
- M. Dadashi Firouzjaei, S. K. Nemani, M. Sadrzadeh, E. K. Wujcik, M. Elliott and B. Anasori, Adv. Mater., 2023, 35, 2300422 CrossRef CAS PubMed.
- S. Jolly, M. P. Paranthaman and M. Naguib, Mater. Today Adv., 2021, 10, 100139 CrossRef CAS.
- Y. Zhong, S. Huang, Z. Feng, Y. Fu and A. Mo, J. Biomed. Mater. Res., Part A, 2022, 110, 1840–1859 CrossRef CAS PubMed.
- D. Ayodhya, Diamond Relat. Mater., 2023, 132, 109634 CrossRef CAS.
- G. Perini, A. Rosenkranz, G. Friggeri, D. Zambrano, E. Rosa, A. Augello, V. Palmieri, M. De Spirito and M. Papi, Biomed. Pharmacother., 2022, 153, 113496 CrossRef CAS PubMed.
- G. Yang, F. Liu, J. Zhao, L. Fu, Y. Gu, L. Qu, C. Zhu, J.-J. Zhu and Y. Lin, Coord. Chem. Rev., 2023, 479, 215002 CrossRef CAS.
- S. Iravani and R. S. Varma, Nano-Micro Lett., 2022, 14, 213 CrossRef CAS PubMed.
- S. Iravani, E. Nazarzadeh Zare and P. Makvandi, ACS Biomater. Sci. Eng., 2024, 10, 1892–1909 CrossRef CAS PubMed.
- X. Qu, Y. Guo, C. Xie, S. Li, Z. Liu and B. Lei, ACS Nano, 2023, 17, 7229–7240 CrossRef CAS PubMed.
- Y. Wei, P. Zhang, R. A. Soomro, Q. Zhu and B. Xu, Adv. Mater., 2021, 33, 2103148 CrossRef CAS PubMed.
- B. Lu, Z. Zhu, B. Ma, W. Wang, R. Zhu and J. Zhang, Small, 2021, 17, 2100946 CrossRef CAS PubMed.
- R. Deng, M. Chang, Y. Chen and Y. Zhou, Nanophotonics, 2022, 11, 4995–5017 CrossRef CAS PubMed.
- B. Xu and Y. Gogotsi, Adv. Funct. Mater., 2020, 30, 47 Search PubMed.
- Z. Gao, W. Feng, M. Zhang, S. Xu, J. Zhang, J. Jiang, J. Li, B. Li, L. Xie and W. Zhao, Appl. Mater. Today, 2025, 47, 102949 CrossRef.
- L. Mao, S. Hu, Y. Gao, L. Wang, W. Zhao, L. Fu, H. Cheng, L. Xia, S. Xie, W. Ye, Z. Shi and G. Yang, Adv. Healthcare Mater., 2020, 9, 2000872 CrossRef CAS PubMed.
- C.-Y. Lee, Y.-T. Hsu, Q.-C. Huang, P. Subramaniyan, A. K. Paulose, C.-C. Huang and T.-E. Lin, Small, 2025, 22, e08633 CrossRef PubMed.
- X. Wang, Y. Sang, H. Zhang, B. Zheng, B. Wang, Y. Hu and R. Wu, Int. J. Biol. Macromol., 2026, 339, 149645 CrossRef CAS PubMed.
- K. Diedkova, A. D. Pogrebnjak, S. Kyrylenko, K. Smyrnova, V. V. Buranich, P. Horodek, P. Zukowski, T. N. Koltunowicz, P. Galaszkiewicz, K. Makashina, V. Bondariev, M. Sahul, M. Caplovicova, Y. Husak, W. Simka, V. Korniienko, A. Stolarczyk, A. Blacha-Grzechnik, V. Balitskyi, V. Zahorodna, I. Baginskiy, U. Riekstina, O. Gogotsi, Y. Gogotsi and M. Pogorielov, ACS Appl. Mater. Interfaces, 2023, 15, 14033–14047 CAS.
- C. Liang, F. Ai, P. Wang, J. kang, H. Deng, Z. Fan, Z. Lin, G. Liu, Y. Zhu and J. Tao, Int. J. Biol. Macromol., 2026, 335, 149207 CrossRef CAS PubMed.
- K. Chen, Y. Chen, Q. Deng, S. H. Jeong, T. S. Jang, S. Du, H. E. Kim, Q. Huang and C. M. Han, Mater. Lett., 2018, 229, 114–117 CrossRef CAS.
- S. H. Lee, S. Jeon, X. Qu, M. S. Kang, J. H. Lee, D. W. Han and S. W. Hong, Nano Convergence, 2022, 9, 38 CrossRef CAS PubMed.
- S. De, S. Acharya, C. K. Maity and G. C. Nayak, ACS Appl. Nano Mater., 2023, 6, 11175–11186 CrossRef CAS.
- X. Zhang, M. S. Javed, H. Ren, X. Zhang, S. Ali, K. Han, A. Ahmad, A. M. Tighezza, W. Han and K.-Q. Peng, Mater. Today Energy, 2024, 40, 101496 CrossRef CAS.
- X. Zhang, M. S. Javed, S. Ali, A. Ahmad, S. S. A. Shah, I. Hussain, D. Choi, A. M. Tighezza, E. Tag-Eldin and C. Xia, Nano Energy, 2024, 120, 109108 CrossRef CAS.
- D. B. Tripathy, S. Pradhan, P. Agarwal and D. Jain, Sustainable Energy Fuels, 2026, 2, 447–487 RSC.
- M. A. Zaed, K. H. Tan, N. Abdullah, R. Saidur, A. K. Pandey and A. M. Saleque, Open Ceram., 2024, 17, 100526 CrossRef CAS.
- Graphite Market Analysis: Projected to Reach USD 13.35 Billion by 2032 with a CAGR of 6.9% - EIN Presswire, https://www.einnews.com/pr_news/789993641/graphite-market-analysis-projected-to-reach-usd-13-35-billion-by-2032-with-a-cagr-of-6-9, accessed 8 January 2026.
- MXene Market Size Report, 2022-2027, https://www.industryarc.com/Report/20088/MXene-market.html, accessed 8 January 2026.
- S. H. Hosseini-Shokouh, J. Zhou, E. Berger, Z.-P. Lv, X. Hong, V. Virtanen, K. Kordas and H.-P. Komsa, ACS Appl. Mater. Interfaces, 2023, 15, 7063–7073 CrossRef CAS PubMed.
- Q. T. H. Ta, A. Sreedhar, N. N. Tri and J.-S. Noh, Ceram. Int., 2024, 50, 27227–27236 CrossRef CAS.
- Q. T. H. Ta, N. N. Tri and J.-S. Noh, Appl. Surf. Sci., 2022, 604, 154624 CrossRef.
- A. Sreedhar, Q. T. H. Ta and J.-S. Noh, J. Electroanal. Chem., 2023, 940, 117509 CrossRef CAS.
- S. Zhang, Y. Xiao, H. Chen, Y. Zhang, H. Liu, C. Qu, H. Shao and Y. Xu, ACS Appl. Mater. Interfaces, 2023, 15, 13802–13812 CrossRef CAS PubMed.
- Y. Liu, Z. Shi, T. Liang, D. Zheng, Z. Yang, Z. Wang, J. Zhou and S. Wang, InfoMat, 2024, 6, e12536 CrossRef CAS.
- A. Kumar, R. K. R. Kumar, M. O. Shaikh, J.-Y. Yang, A. M. Bharti, B.-Y. Huang, H.-L. Chang, D.-H. Lee and C.-H. Chuang, Mater. Today Chem., 2024, 37, 102024 CrossRef CAS.
- P. Wang, T. Mai, W. Zhang, M. Qi, L. Chen, Q. Liu and M. Ma, Small, 2024, 20, 2304914 CrossRef CAS PubMed.
- L. Huang, L. Ding, J. Caro and H. Wang, Angew. Chem., Int. Ed., 2023, 62, e202311138 CrossRef CAS PubMed.
- C. Yao, W. Zhang, L. Xu, M. Cheng, Y. Su, J. Xue, J. Liu and S. Hou, Sep. Purif. Technol., 2021, 263, 118365 CrossRef CAS.
- H. Zhang, Y. Zheng, H. Zhou, S. Zhu and J. Yang, Sep. Purif. Technol., 2023, 305, 122425 CrossRef CAS.
- G. Zeng, Z. He, T. Wan, T. Wang, Z. Yang, Y. Liu, Q. Lin, Y. Wang, A. Sengupta and S. Pu, Sep. Purif. Technol., 2022, 292, 121037 CrossRef CAS.
- A. A. Ghani, A. Shahzad, M. Moztahida, K. Tahir, H. Jeon, B. Kim and D. S. Lee, Chem. Eng. J., 2021, 421, 127780 CrossRef.
- Q. Long, S. Zhao, J. Chen, Z. Zhang, G. Qi and Z. Q. Liu, J. Membr. Sci., 2021, 635, 119464 CrossRef CAS.
- Y. Wang, J. Chen, M. Que, Q. Wu, X. Wang, Y. Zhou, Y. Ma, Y. Li and X. Yang, Appl. Surf. Sci., 2023, 639, 158270 CrossRef CAS.
- W. Zhang, Q. Zhang, X. Yang, M. Ran, Y. Ding and P. La, Mater. Sci. Eng., B, 2026, 325, 119120 CrossRef CAS.
- Z. Gao, D. Wang, B. Zou, Y. Liu, X. Yu, Q. Li, J. Ma, L. Liu, C. Liu, Z. Tong and J. Hu, J. Taiwan Inst. Chem. Eng., 2026, 182, 106584 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |
