Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
A regioselective sustainable neat approach to naphtho[2,3-b]thiophene-4,9-dione: detailed mechanistic study and DFT analysis
Soumen K. Manik a,
Sk Asraf Ali
a,
Sk Asraf Ali bc,
Nayim Sepay
bc,
Nayim Sepay d,
Aniruddha Deb
d,
Aniruddha Deb e,
Manik Shitf,
Paritosh Barikb,
Jyotirmoy Rathg,
Kankan K. Maityh,
Sudhir C. Pala,
Shubhankar Samanta
e,
Manik Shitf,
Paritosh Barikb,
Jyotirmoy Rathg,
Kankan K. Maityh,
Sudhir C. Pala,
Shubhankar Samanta *b and
Nirmal K. Hazra*a
*b and
Nirmal K. Hazra*a
aDepartment of Chemistry, Coastal Environmental Studies Research Centre, Egra Sarada Shashi Bhusan College, Vidyasagar University, Purba Medinipur, Egra, West Bengal 721429, India. E-mail: nirmalkrhazra@gmail.com
bDepartment of Chemistry, Bidhannagar College, EB 2, Sector I, Salt Lake, Kolkata-700064, West Bengal, India. E-mail: chemshubha@gmail.com
cDistrict Institute of Education and Training (DIET), Jhargram, West Bengal-721507, India
dDepartment of Chemistry, Lady Brabourne College, Kolkata-700017, West Bengal, India
eDepartment of Chemical Engineering, Birla Institute of Technology, Mesra, Ranchi, Jharkhand 835215, India
fDepartment of Chemistry, Jadavpur University, Jadavpur, Kolkata-700032, West Bengal, India
gDepartment of Physics, Coastal Environmental Studies Research Centre, Egra Sarada Shashi Bhusan College, Vidyasagar University, Purba Medinipur, Egra, West Bengal 721429, India
hDepartment of Chemistry, Belda College, Paschim Medinipur, Belda, West Bengal-721424, India
First published on 28th January 2026
Abstract
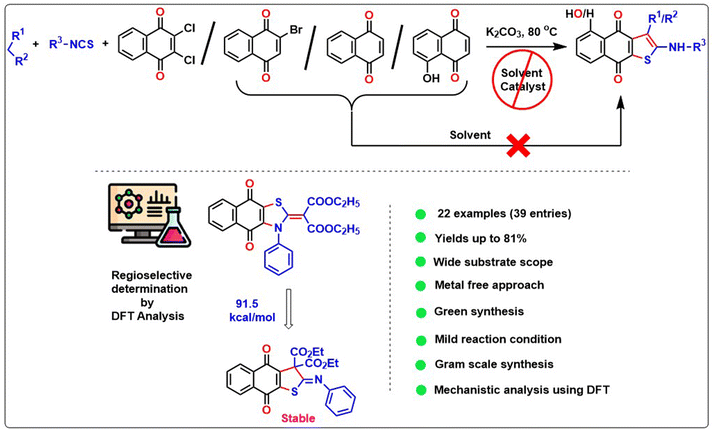
An environment friendly simple, solvent-free, and lower E-factor technique has been developed for synthesizing anticancer property-containing substituted naphtho[2,3-b]thiophene-4,9-dione derivatives via a one-pot three-component reaction using active methylene compounds, alkyl/aryl isothiocyanate, and 1,4-naphthoquinone derivatives. The reaction proceeds through the regioselective Michael addition between the di-anionic intermediate and 1,4-naphthoquinone derivatives, followed by a domino reaction, which has been established via DFT study. Finally, it undergoes a Krapcho reaction followed by aromatization in a basic environment, yielding the desired product naphtho[2,3-b]thiophene-4,9-dione. The reluctant addition of a di-anionic intermediate to the naphthalene-1,4-dione or 2-bromonaphthalene-1,4-dione in the solvent medium has been overcome using a neat approach. Practicability of the methodology has been enhanced via gram scale solvent-free cascade reaction. The wide substrate scope, biological activity, sustainability, and detailed mechanistic study make our reaction more acceptable in the current scenario.
Introduction
The naphthoquinone and thiophene moieties have shown their importance as a central core structure in various bioactive compounds. The naphthoquinone and thiophene scaffolds exhibit distinct chemical and structural properties that facilitate interactions with particular targets in biological systems. The significant therapeutic effects of naphthoquinone and thiophene-based drugs have prompted researchers to develop and evaluate numerous new molecules. Researchers have successfully developed compounds with enhanced potency and efficacy by incorporating the thiophene moiety into naphthoquinone. Therefore, naphthoquinone-fused thiophene is a significant target for synthetic and medicinal chemists due to its extensive applications across pharmaceutical, biological, and industrial domain. Interestingly, naphthoquinone-fused thiophene exhibits significant biological activity, including anticancer,1–5 antiprotozoal,6–9 and antipsoriatic effects.10–12 Naturally occurring Thioquinomycin A-D, and Serinoquinone exhibit potent cytotoxicity effects on several cancer cell lines (Fig. 1).13 FDA [Food and Drug Administration] approved drug molecules in the quinone class include doxorubicin and mitoxantrone.14 Commercially available drugs containing thiophene and benzothiophene motifs include zileuton, mobam, and sertaconazole (Fig. 1).15 These drugs have a diverse range of medicinal applications. The naphtho[2,3-b]thiophene-4,9-dione moiety has been shown to have substantial cytotoxicity against several human solid tumor cell lines that are resistant to doxorubicin and cis-platin.4Inspired by the significance of naphtho[2,3-b]thiophene-4,9-dione, researchers employed a traditional method to synthesize these scaffolds, starting with thiophene or quinone. A. Acosta et al.16 (Scheme 1) initially developed a microwave-assisted synthesis method for the fabrication of naphtho[2,3-b]thiophene-4,9-dione using an annulation approach, with thiophene as the starting material. In subsequent studies, several research groups employed two-component coupling processes to obtain the intended scaffolds. Typically, the researchers used a quinone-embedded framework that was combined with either α-enolicdithioesters/β-oxothioamides17 or the Sonogashira coupling product involving 3-bromo-2-(dimethylamino)-5-hydroxynaphthalene-1,4-dione with but-3-yne-2-ol (Scheme 1).18 Consequently, researchers are constantly encountering new obstacles in their pursuit of this objective. The three-component reaction is a novel method that enables the synthesis of naphtho[2,3-b]thiophene-4,9-dione from either naphthoquinone or bromo naphthoquinone combined with α, β-unsaturated ketones or alkynes in the presence of a sulfur donor (Scheme 1).19,20 Recently, we have established a methodology for the synthesis of naphtho[2,3-b]thiophene-4,9-dione derivatives using a traditional solvent-added approach.21
The previously reported methods required a prolonged reaction period within a sealed tube, while the subsequent method operated in the presence of a transition metal catalyst in the hazardous dioxane solvent. The significant limitations of existing methodologies have motivated us to innovate a more comprehensive, efficient, and practical approach that is also easily implemented.
In continuation of heterocyclic synthesis under sustainable conditions, we want to synthesize anti-cancer bioactive molecules under solvent-free multicomponent reaction. The multi-component reaction is a very pretty method as it produces complex molecules from single reagents without isolation of intermediates.22 It not only avoids the toxic solvent and aqueous workup but also reduces the multiple use of column chromatography for each step's separation. Nowadays, it's a challenge to develop an eco-friendly and environmentally benign synthetic strategy. Presently, the multi-component reaction has become an increasingly important method in organic synthesis. They are regarded as efficient synthetic methodologies for the synthesis of diverse functionalized complex molecules in a timely and cost-effective manner.23 MCRs are a combination of three or more reactants in one pot procedure, resulting in a new compound with high yields.24–29
Herein, we present a novel and efficient one-pot three-component regioselective synthesis of naphthothiophene (4) using active methylene compounds, alkyl/aryl isothiocyanate, and 1,4-naphthoquinone and its derivatives facilitated by K2CO3 under neat condition (Scheme 2). The limitation of the previous solvent-added reaction has been overcome using the substrates 2-bromonaphthalene-1,4-dione and naphthalene-1,4-dione for the naphtho[2,3-b]thiophene-4,9-diones formation, where we got the desired product under only neat conditions. The protocol has a low E-factor compared to the existing methods, and it furnished a broad array of the naphtho[2,3-b]thiophene-4,9-diones. Additionally, we have also introduced a brief computational mechanistic observation involving this protocol.
Results and discussion
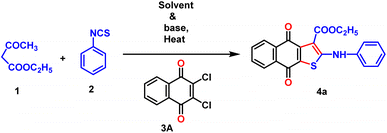
In concern to the environmental issue, we initiated our investigation by performing a model reaction utilizing ethyl acetoacetate, phenyl isothiocyanate, and 2,3-dichloro-1,4-naphthoquinone as the primary substrates (Table 1). The reaction was initiated in ethanol without a base by adding 1, 2, and 3A simultaneously at 100 °C for 9 h (entry 1). Regrettably, no required product 4a was found under these specified conditions. Moreover, the reaction was conducted using Et3N (3.5 equiv.) as a base in ethanol at a temperature of 60 °C for 9 h, but the desired product 4a was not obtained (entry 2). Similarly, in the presence of another strong base, DABCO (3.0 equiv.) was unable to produce 4a in ethanol or DMSO medium (entries 3 and 4). The lack of success in this reaction motivated us to investigate a more effective combination of base and solvent for our model reaction. Significantly, the replacement of the organic base with K2CO3 resulted in the production of the desired product 4a with a yield of 21% in DMSO (entry 5). This was achieved by adding 3A to the anion species produced from the reaction with 1 and 2 in the presence of K2CO3. By substituting DMSO with DMF, a 41% yield of 4a was obtained (entry 7). Furthermore, a 51% yield of compound 4a was obtained by substituting K2CO3 with Cs2CO3 (entry 8). The reaction was easily progressed at ambient temperature by sequentially adding 1,2, and 3A (entry 6). Nevertheless, the production of the cyclized product (35%) remains unsatisfactory even after 16 h of stirring. In concern to the environment, we want to establish our reaction under neat conditions, and surprisingly, 4a was obtained effectively with 64% yield at 80 °C (entry 9). By replacing K2CO3 with Na2CO3 or CH3COONa, the desired target product was obtained with a yield of 51% and 26%, respectively (entries 10 and 11).| Entry | Solvent | Base | Temperature (°C) | Time (h) | Yield (%) |
|---|---|---|---|---|---|
| a Reagent & conditions: substrate 1 (0.1 mmol), substrate 2 (1.1 equiv.), substrate 3 (1 equiv.), base (2.5 equiv.). | |||||
| 1 | C2H5OH | None | 100 °C | 9 | NR |
| 2 | C2H5OH | Et3N | 60 °C | 9 | NR |
| 3 | C2H5OH | DABCO | 100 °C | 9 | NR |
| 4 | DMSO | DABCO | 100 °C | 9 | NR |
| 5 | DMSO | K2CO3 | 80 °C | 9 | 21 |
| 6 | DMF | K2CO3 | RT | 16 | 35 |
| 7 | DMF | K2CO3 | 80 °C | 8 | 41 |
| 8 | DMF | CS2CO3 | 80 °C | 6 | 51 |
| 9 | Neat | K2CO3 | 80 °C | 1 | 64 |
| 10 | Neat | Na2CO3 | 80 °C | 1 | 51 |
| 11 | Neat | CH3COONa | 80 °C | 3 | 26 |
Interestingly, the visual color change in a progressive manner helps us to monitor our reaction. A yellow color is observed when substrates 1 and 2 are heated in potassium carbonate, suggesting the formation of a dianion. Subsequently, the inclusion of 3A in the generated dianion through the combination of 1 and 2 results in a color change from yellow to red, therefore indicating the formation of a significant quantity of the product (4a).
Remarkably, we have found that the yield of the desired product was increased with a constant enhancement of the K2CO3 equivalence, which became fixed after the addition of 2.5 equivalent with respect to the quinone substrate 3A, and the yield of the cyclized product was increased up to 1 h (Fig. 2). Hence, the final optimized condition for the three-component coupling reaction under neat condition is substrate 1 (0.1 mmol), substrate 2 (1.1 equiv.), substrate 3 (1 equiv.), K2CO3 (2.5 equiv.), temperature 80 °C, and time 1 h.
Furthermore, we have compared the E factor between the existing protocols with the current methodology using the equation E factor = [Σm (raw materials) + Σm (reagents) + Σm (solvents) + m (water) − m (product)]/m(product) and obtained a surprising result, which is shown in Fig. 3.30 These observations indicated that our protocol furnished lower toxic chemicals compared to the reported one.
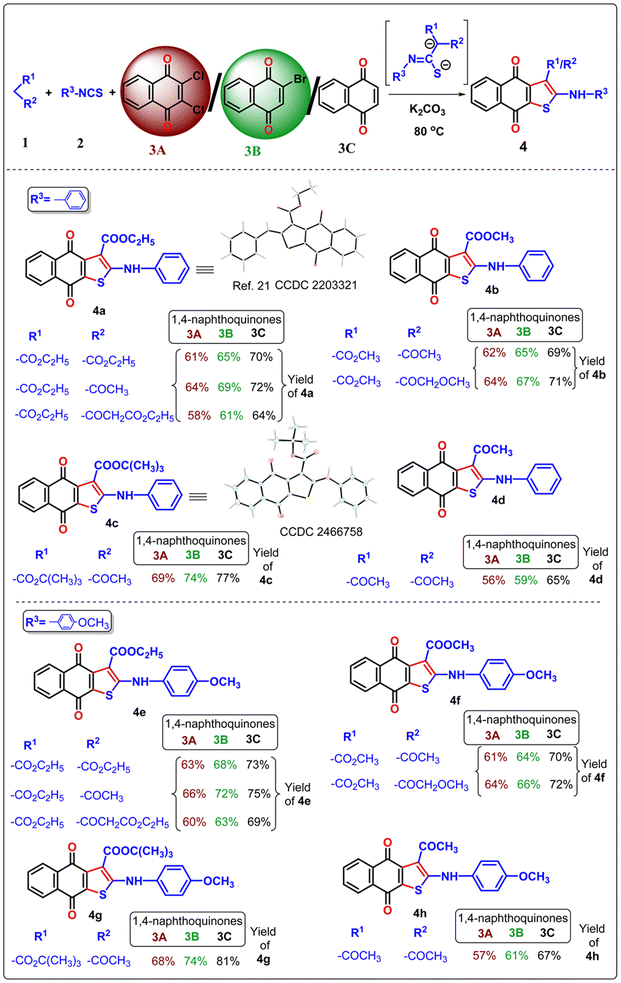
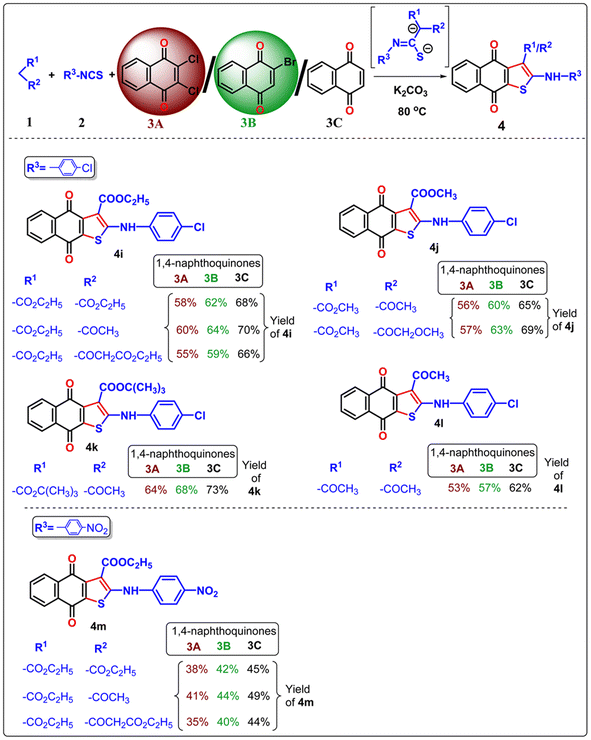
Once the optimal condition has been established (Table 1, entry 9), the synthesis of naphtho[2,3-b]thiophene-4,9-dione was investigated using different active methylene compounds, alkyl/aryl isothiocyanate, and 2,3-dichloro-1,4-naphthoquinone 3A. Several active methylene compounds easily react with various alkyl/aryl isothiocyanate derivatives and 2,3-dichloro-1,4-naphthoquinone 3A to successfully synthesize the required naphtho[2,3-b]thiophene-4,9-dione with moderate to good yields (Tables 2–4). Diethyl malonate, ethyl acetoacetate, diethyl 3-oxopentanedioate, methyl 3-oxobutanoate, methyl 4-methoxy-3-oxobutanoate, tert-butyl 3-oxobutanoate, and acetylacetone are active methylene compounds compatible with the reaction (See SI, Table S1). Tert-butyl 3-oxobutanoate exhibited superior efficacy with respect to other active methylene compounds. Phenyl isothiocyanate with an electron-donating group (p-OMe) (Table 2) provides higher reactivity, and electron-withdrawing groups (p-Cl and p-NO2) (Table 3) provide lower reactivity and yields of moderate magnitude of 4a–4m. The acetylacetone is reluctant to add with the alkyl/aryl isothiocyanate as the intermediate stabilized by the two electron-withdrawing-COMe group, and hence the yields of the ultimate products 4d and 4h are low. The −ve charge on the sulfur atom in the dianion intermediate is stabilized by the chloro (–Cl)/nitro(–NO2) group on the isothiocyanate derivatives, resulting in low yields of compound 4l–4m respectively. The beauty of this method is the removal of one ester or acyl group from the active methylene compound 1 after coupling with quinone 3 and alkyl/aryl isothiocyanate 2. During aromatization, diethylmalonate removed one ester group, while ethyl acetoacetate/diethyl 3-oxopentanedioate/methyl 3-oxobutanoate/methyl 4-methoxy-3-oxobutanoate/tert-butyl 3-oxobutanoate/acetyl acetone removed the acetyl group in the final product 4.
| a Reagent and conditions: substrate 1 (0.1 mmol), substrate 2 (1.1 equiv.), substrate 3 (1 equiv.), K2CO3 (2.5 equiv.), temperature 80 °C, time 1 h. |
|---|
 |
| a Reagent and conditions: substrate 1 (0.1 mmol), substrate 2 (1.1 equiv.), substrate 3 (1 equiv.), K2CO3 (2.5 equiv.), temperature 80 °C, time 1 h. |
|---|
 |
| a Reagent and conditions: substrate 1 (0.1 mmol), substrate 2 (1.1 equiv.), substrate 3 (1 equiv.), K2CO3 (2.5 equiv.), temperature 80 °C, time 1 h. |
|---|
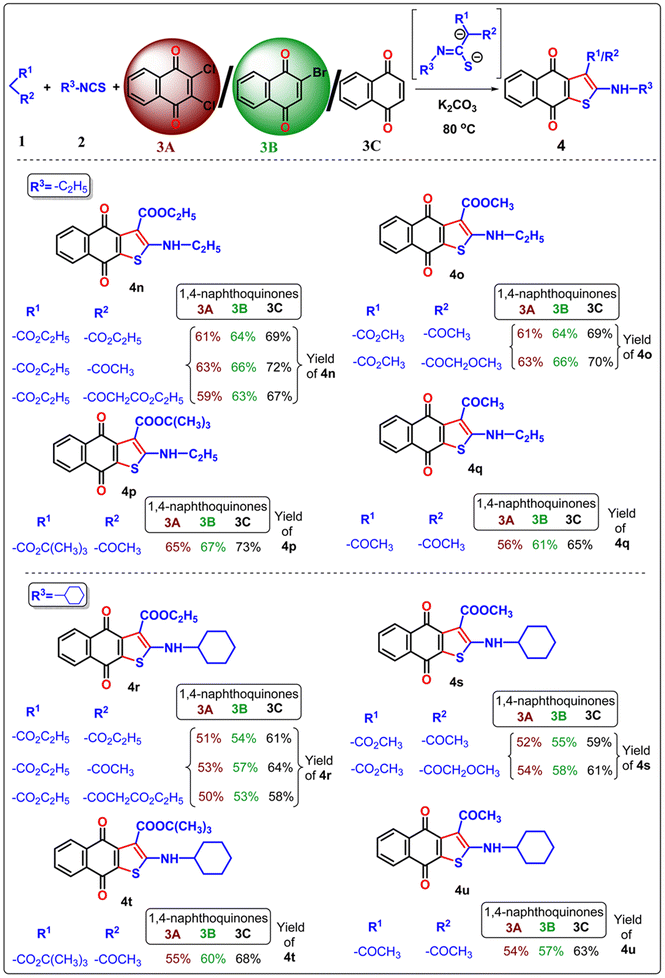 |
In the previous communications,21 we have only prepared the naphtho[2,3-b]thiophene-4,9-dione from the substrate 2,3-dichloronaphthalene-1,4-dione under solvent-added conditions, but this method has been modified via environmental benign conditions under neat conditions. Surprisingly, during the extension of the protocol in the presence of 2-bromonaphthalene-1,4-dione or naphthalene-1,4-dione, the reaction did not happen in the addition of solvent with phenyl isothiocyanate (Scheme 2). This problem has been overcome under neat conditions within 1 h. Consequently, the reaction was extended to 2-bromo-1,4-naphthoquinone 3B in the presence of alkyl/aryl isothiocyanate and active methylene compounds at 80 °C under neat conditions. With the same substituted derivatives of alkyl/aryl isothiocyanate, the established technique was well tolerated, and the yield of the desired products 4a–4v (total 22 instances) was significantly higher from 3B than the product obtained from 2,3-dichloro-1,4-naphthoquinone 3A. This observation suggests that the presence of halide substitution in the quinone ring delayed the addition–elimination process. To investigate our methodology and enhance the yield of naphtho[2,3-b]thiophene-4,9-dione, we examined the advancement of our reaction with 1,4-naphthoquinone 3C under neat conditions. We observed that the desired product 4 exhibited the best yields compared to 3A and 3B. This observation provides more evidence that the removal of the halide group from the naphthoquinone moieties initiates the addition–elimination reaction, resulting in the formation of naphtho[2,3-b]thiophene-4,9-dione derivatives. Hence, we can conclude that the presence of an electron-withdrawing group in the quinone ring lowers the yields of 4. The three components coupling product 4a and 4c were crystallized, and they are shown in Table 2 with the CCDC number.
Our protocol was also successfully applied with aliphatic alkyl isothiocyanate derivatives under neat conditions (Table 4). Ethyl isothiocyanate, cyclohexyl isothiocyanate underwent three component cyclization with significant yields and it was found that the smaller acyclic isothiocyanate furnished dianion intermediate effectively and participate Michael addition with quinone moiety more comfortably than the corresponding cyclic analogue of isothiocyanate. Consequently, the desired 4n–4q furnished with higher yields than 4r–4u.
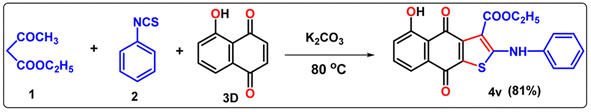
The diversity of our protocol has also been successfully applied to 5-hydroxy-1,4-naphthoquinone (juglone; obtain from natural source), ethyl acetoacetate, and phenyl isothiocyanate in the presence of K2CO3 under neat conditions with a satisfactory yield 4v (Table 5).
| a Reagent and conditions: substrate 1 (0.1 mmol), substrate 2 (1.1 equiv.), substrate 3 (1 equiv.), K2CO3 (2.5 equiv.), temperature 80 °C, time 1 h. |
|---|
 |
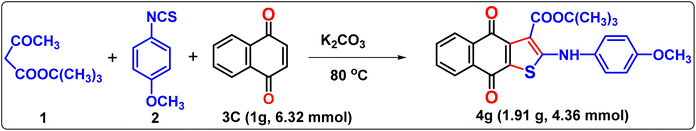
The protocol was also applicable in terms of practical aspects. From an applied standpoint, the reaction was performed on a gram scale and gave a good yield of naphthothiophene derivative 4g under neat conditions (Scheme 3). We obtained 1.91 g (4.36 mmol) product of 4g from 1 g (6.32 mmol) of 3C, yielding a 69% yield of the desired naphthothiophene at 80 °C after 1.5 h of heating. The time required for the gram-scale neat reaction was greater than that of the milligram–scale reaction.
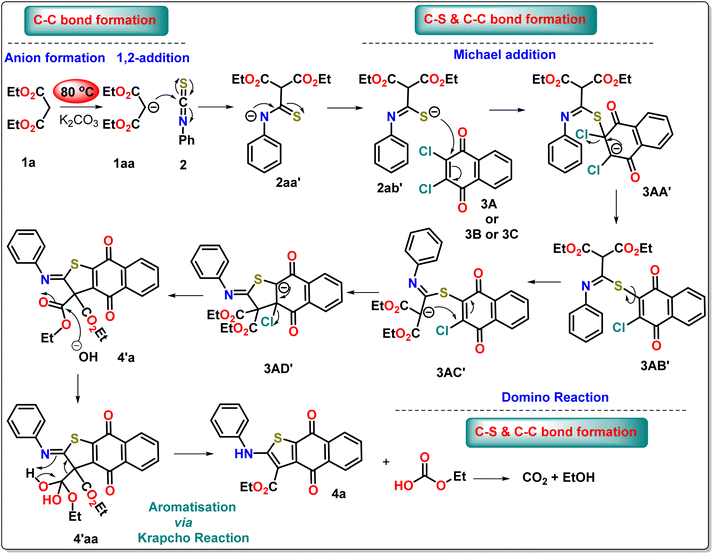
Once, we have successfully developed a synthetic strategy for the three-component neat approach, our goal is to understand the precise mechanistic pathway of the overall observation by performing a series of controlled experiments (Scheme 4). In the presence of potassium carbonate, we have taken diethylmalonate or ethyl acetoacetate 1, phenyl isothiocyanate 2, and 2,3-dichloro-1,4-napthoquinone 3A. Although we have assessed the feasibility of our method using a one-pot approach by combining all the necessary substrates 1, 2, and 3A simultaneously, we were unable to obtain the cyclized product (case 1). In this reaction, diethylmalonate easily combines with quinone to form a crystalline product 3a′ with a high yield through the replacement of one –Cl atom by an addition–elimination mechanism, which has been confirmed by X-ray crystallographic study. The phenyl isothiocyanate remains inactive in the reaction mixture. This finding offers a vital indication that the formation of dianions is the necessary step to obtain the thiophene-fused quinone derivatives. Hence, we have devised a two-step one-pot strategy where active methylene is allowed to first react with phenyl isothiocyanate in a basic medium to produce a yellow dianion intermediate 2′a within 30 minutes under neat conditions. After that, it reacts with 2,3-dichloro-1,4-napthoquinone to form a crystalline product 4′a within 30 minutes under neat conditions (case 2). Compound 4a is obtained by the Krapcho reaction of the crystalline product 4′a at 80 °C (case 3). The isolation of the intermediate 4′a is the crucial factor for understanding the complete mechanism of the reaction. Curiously, it has been observed that the intermediate cannot stabilize under neat conditions, and three substrates can be easily coupled sequentially to form the required product 4a (case 4). It has been noticed that the visual color change (yellow to red) is an interesting point to monitor the progress of the reaction (case 5).
Based on the controlled experiment and the isolation of the intermediate, we have proposed a mechanism for the three-component coupling reaction under neat conditions through density functional theory, as this study offers the best opportunity to understand the reaction dynamics (Scheme 5).
Theoretical study of the mechanism
To thoroughly elucidate the reaction leading to the formation of compound 4, we employed density functional theory (DFT). The nucleophilic attack of 1aa on the carbon center of 2 is decidedly spontaneous, yielding a free energy change of −12.2 kcal mol−1 (Fig. 4a). This reaction produces 2aa′, which possesses a negative charge on the nitrogen atom but exhibits slight instability (ΔG° = −0.4 kcal mol−1) when compared to its resonating structure 2ab′, where the sulfur atom holds the negative charge (Fig. 4b). Importantly, both resonating structures (2aa′ and 2ab′) are nearly equally poised for reaction with 3A. However, experimental results compellingly demonstrate that the reaction preferentially occurs through 2ab′, leading to the formation of intermediate 3AA′, a process requiring 35.2 kcal mol−1 of free energy. To explain the reason for this sulfur-selective nucleophilic attack, we investigated N-site attacks on 3A, resulting in 3AA″ (Fig. 4c). Notably, the conversion from 3A to 3AA″ demands a considerable 74.4 kcal mol−1 of free energy. The differences in stability between 3AA′ and 3AA″ are pivotal in this context. The π-stacking interaction between the naphthoquinone ring (blue) and the phenyl ring (wine red), along with the weak hydrogen bond between the naphthoquinone C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O and the methylene hydrogens of the diethyl malonate, significantly stabilizes 3AA′ (Fig. 4c). In stark contrast, 3AA″ lacks these stabilizing intramolecular forces, further supporting the observed selectivity.
O and the methylene hydrogens of the diethyl malonate, significantly stabilizes 3AA′ (Fig. 4c). In stark contrast, 3AA″ lacks these stabilizing intramolecular forces, further supporting the observed selectivity.
A facile chloride elimination (ΔG° = −22.7 kcal mol−1) from 3AA′ leads directly to a fully conjugated intermediate 3AB′. The deprotonation from the DEM unit of 3AC′ requires an energy input of 5.0 kcal mol−1. However, the resulting anion 3AC′ is resonance-stabilised, benefiting from resonance with both the ester groups of the DEM part and the imidothioate part. This resonance (with the negative charge on the carbon centre) enables the formation of an ambident nucleophile, having a negative charge on the nitrogen centre, both of which exhibit comparable stability. The intramolecular nucleophilic cyclisation can proceed through either the carbon centre or the nitrogen centre. Converting 3AC′ to 3AD′ demands 25.2 kcal mol−1, while cyclisation through the nitrogen centre to form 3AD″ from the canonical structure of 3AC″ requires 82.2 kcal mol−1. Notably, the anionic species 3AD″ is less stable than 3AD′ due to steric interactions between the N-phenyl ring and the ethyl groups of the malonate unit (Fig. 4d). This steric strain results in a twisted conformation, disrupting the planarity of the malonate unit and consequently impacting the degree of conjugation in 3AD″. The transformation of compound 3AD′ into 4′a via an elimination reaction significantly reduces steric congestion, yielding a conjugation-stabilised product. This reaction releases a substantial free energy of −62.2 kcal mol−1, making it spontaneous. In contrast, the elimination reaction involving 3AD″ yields product 4′ab, which shares the same destabilising structural features as 3AD″. Consequently, 4′ab is less stable compared to 4′a (Fig. 4e). In the case of compound 4′a, stabilisation is achieved through conjugation with minimal steric strain. The free energy difference between 4′a and 4′ab is 91.5 kcal mol−1. The final product, identified as compound 4, is generated through the removal of one ester group from the DEM unit via the Krapcho reaction. This mechanism involves a nucleophilic attack by a hydroxyl ion, which is produced from the reaction of K2CO3 with water, targeting one of the ester centres in compound 4′a. This interaction results in the formation of an intermediate, labelled 4′aa, which is stabilised by hydrogen bonding. The free energy required for this step is estimated at 8.5 kcal mol−1. Following this, the decomposition of compound 4′aa leads to the formation of 4a and ethyl hydrogen carbonate, with the latter releasing CO2 as a gas. This gas release not only drives the reaction forward but also increases entropy, consistent with Le Châtelier's principle. This step is spontaneous by release −12.3 kcal mol−1.
Conclusion
In conclusion, we have established a three-component sustainable cascade protocol for synthesizing 2,3-disubstituted anti-cancer naphtho[2,3-b]thiophene-4,9-diones in favourable yields utilizing active methylene compounds, alkyl/aryl isothiocyanates, and either 2,3-dichloro-1,4-naphthoquinone, 2-bromo-1,4-naphthoquinone, 1,4-naphthoquinone or 5-hydroxy-1,4-naphthoquinone. The gram-scale synthesis of fused naphtho[2,3-b]thiophene-4,9-diones is another practical application of this method. The regioselective addition of dianion intermediate to quinone system has been recognized using DFT analysis.Experimental section
Reagents, starting materials, and solvents were obtained from commercial suppliers and used without further purification. Analytical TLC was carried out on Merck silica gel GF 254 plates. The crude product was purified by column chromatography using silica gel (100–200 mesh) with a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 mixture of ethyl acetate and petroleum ether as the eluent to isolate the desired products. 1H NMR and 13C NMR spectra were recorded on a Bruker spectrometer at 300, 400, 500, 600 MHz and 75, 101, 126, 151 MHz, respectively. Chemical shifts are reported in δ values (ppm) relative to tetramethylsilane (Me4Si) as an internal standard, and coupling constants (J) are given in hertz (Hz). TOF MS ES+ spectra were recorded on a Bruker mass spectrometer.
20 mixture of ethyl acetate and petroleum ether as the eluent to isolate the desired products. 1H NMR and 13C NMR spectra were recorded on a Bruker spectrometer at 300, 400, 500, 600 MHz and 75, 101, 126, 151 MHz, respectively. Chemical shifts are reported in δ values (ppm) relative to tetramethylsilane (Me4Si) as an internal standard, and coupling constants (J) are given in hertz (Hz). TOF MS ES+ spectra were recorded on a Bruker mass spectrometer.
General procedure for the synthesis of naphtho[2,3-b]thiophene-4,9-diones (4a–4v)
Active methylene compound (0.1 mmol), alkyl/aryl isothiocyanate (1.1 equiv., 0.11 mmol), and 2.5 equiv. K2CO3 was added to a hard glass test tube. The mixture was heated at 80 °C for 30 minutes in an oil bath. Once the dianion formation was complete, then 1,4-naphthoquinone (1 equiv. 0.1 mmol) was added to the reaction mixture, and the heating was continued at 80 °C for an additional 30 minutes. Upon completion, checked by TLC, the reaction mixture was acidified with dilute HCl, extracted with ethyl acetate (3 × 10.0 mL), followed by washing with brine water (3 × 10.0 mL). The organic layer was dried over sodium sulfate (Na2SO4). After evaporating the solvent, the crude product was purified by column chromatography on silica gel (100–200 mesh) using an ethyl acetate-petroleum ether (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20) eluent to get the pure desired products 4.
20) eluent to get the pure desired products 4.
Conflicts of interest
There are no conflicts to declare.Data availability
CCDC 2203321, 2257893, 2329614 and 2466758 contain the supplementary crystallographic data for this paper.31a–dThe data supporting this article have been included as part of the supplementary information (SI). Supplementary information: experimental details, characterization and DFT data. See DOI: https://doi.org/10.1039/d5ra08362a.
Acknowledgements
We gratefully acknowledge the Department of Chemistry, Egra S. S. B. College, for financial support. The financial support from Science & Engineering Research Board (SERB TAR/2022/000070) is gratefully acknowledged. We also acknowledge the Department of Chemistry at Vidyasagar University and Jadavpur University, as well as the Central Research Facility (CRF) at IIT Kharagpur, for their instrumental support and BIT, Mesra, Ranchi, for the HRMS facility. We also acknowledge and appreciate the support from Prof. (Dr) Chittaranjan Sinha of Jadavpur University and Prof. (Dr) Anirban Bhunia of Bose Institute.References
- J. Acuna, J. Piermattey, D. Caro, S. Bannwitz, L. Barrios, J. López, Y. Ocampo, R. Vivas-Reyes, F. Aristizábal, R. Gaitán, K. Muller and L. Franco, Synthesis, Anti-Proliferative Activity Evaluation and 3D-QSAR Study of Naphthoquinone Derivatives as Potential Anti-Colorectal Cancer Agents, Molecules, 2018, 23, 186 CrossRef PubMed.
- I. Gomez-Monterrey, P. Campiglia, C. Aquino, A. Bertamino, I. Granata, A. Carotenuto, D. Brancaccio, P. Stiuso, I. Scognamiglio, M. R. Rusciano, A. S. Maione, M. Illario, P. Grieco, B. Maresca and E. Novellino, Design, Synthesis, and Cytotoxic Evaluation of Acyl a Quinone-Based System, J. Med. Chem., 2011, 54, 4077–4091 CrossRef CAS PubMed.
- J. Valderrama, O. Espinoza, J. Rodriguez and C. Theoduloz, Synthesis and Antitumor Evaluation of Thiophene Analogs of Kigelinone, Lett. Org. Chem., 2009, 6, 278–281 CrossRef CAS.
- L. J. Huang, S. C. Kuo, C. Y. Perng, Y. H. Chao, T. S. Wu, A. T. McPhail, A. Mauger, H. H. Cheng and K. H. Lee, Synthesis and Cytotoxicity of Acetyl-4H,9H-naphtho[2,3-b]thiophene-4,9-diones, Bioorg. Med. Chem. Lett., 1998, 8, 2763–2768 CrossRef CAS PubMed.
- A. P. Krapcho, M. E. Petry and M. P. Hacker, Heterosubstituted Anthracene-9,10-dione Analogues. The Synthesis and Antitumor Evaluation of 5,8-Bis[(aminoalkyl)amino]naphtho[2,3-b]thiophene-4,9-diones, J. Med. Chem., 1990, 33, 2651–2655 CrossRef CAS PubMed.
- J. A. Valderrama, C. Astudillo, R. A. Tapia, E. Prina, E. Estrabaud, R. Mahieux and A. Fournet, Studies on Quinones. Part 37. Synthesis and Biological Activity of o-Aminoester Functionalised Benzo- and Naphtho[2,3-b]thiophenequinones, Chem. Pharm. Bull., 2002, 50, 1215–1218 CrossRef CAS PubMed.
- J. Valderrama, A. Fournet, C. Valderrama, S. Bastias, C. Astudillo, A. Rojas de Arias, A. Inchausti and G. Yaluff, Synthesis and in Vitro Antiprotozoal Activity of Thiophene Ring-Containing Quinones, Chem. Pharm. Bull., 1999, 47, 1221–1226 CrossRef CAS PubMed.
- M. O. F. Goulart, C. L. Zani, J. Tonholo, F. C. de Abreu, A. B. Oliveira, D. S. Raslan, S. Starling and E. Chiari, Trypanocidal Activity and Redox Potential of Heterocyclic- and 2-Hydroxynaphthoquinones, Bioorg. Med. Chem. Lett., 1997, 7, 2043–2048 CrossRef CAS.
- C. L. Zani, E. Chiari, A. U. Krettli, S. M. F. Murta, M. L. Cunningham, A. H. Fairlamb and A. J. Romanha, Anti-Plasmodial and Anti-Trypanosomal Activity of Synthetic Naphtho[2,3-b]thiophen-4,9-quinones, Bioorg. Med. Chem., 1997, 5, 2185–2192 CrossRef CAS PubMed.
- C. Lindenschmidt, D. Krane, S. Vortherms, L. Hilbig, H. Prinz and K. Muller, 8-Halo-Substituted Naphtho[2,3-b]thiophene-4,9-diones as Redox-Active Inhibitors of Keratinocyte Hyperproliferation with Reduced Membrane-Damaging Properties, Eur. J. Med. Chem., 2016, 110, 280–290 CrossRef CAS PubMed.
- A. Basoglu, S. Dirkmann, N. Zahedi Golpayegani, S. Vortherms, J. Tentrop, D. Nowottnik, H. Prinz, R. Fröhlich and K. Müller, Oxadiazole-Substituted Naphtho[2,3-b]thiophene-4,9-diones as Potent Inhibitors of Keratinocyte Hyperproliferation. Structure-Activity Relationships of the Tricyclic Quinone Skeleton and the Oxadiazole Substituent, Eur. J. Med. Chem., 2017, 134, 119–132 CrossRef CAS PubMed.
- S. Bannwitz, D. Krane, S. Vortherms, T. Kalin, C. Lindenschmidt, N. Zahedi Golpayegani, J. Tentrop, H. Prinz and K. Muller, Synthesis and Structure-Activity Relationships of Lapacho Analogues. 2. Modification of the Basic Naphtho[2,3-b]furan-4,9-dione, Redox Activation, and Suppression of Human Keratinocyte Hyperproliferation by 8-Hydroxynaphtho[2,3-b]thiophene-4,9-diones, J. Med. Chem., 2014, 57, 6226–6239 CrossRef CAS PubMed.
- (a) D. Zhang, Y. Jiang, J. Li, W. Ding, Z. Chen and Z. Ma, Thioquinomycins A-D, novel naphthothiophenediones from the marine-derived Streptomyces sp. SS17F, Tetrahedron, 2018, 74, 6150–6154 CrossRef CAS; (b) A. S. Hirata, J. J. La Clair, P. C. Jimenez, L. V. Costa-Lotufo and W. Fenical, Preclinical development of seriniquinones as selective dermcidin modulators for the treatment of melanoma, Mar. Drugs, 2022, 20, 301 CrossRef CAS PubMed; (c) R. D. A. Miguel, A. S. Hirata, G. C. Salata, A. C. Apolinário, V. M. Barroso, K. Ishida, J. J. L. Clair, W. Fenical, T. S. Martins, L. V. Costa-Lotufo and L. B. Lopes, Topical delivery of seriniquinone for treatment of skin cancer and fungal infections is enabled by a liquid crystalline lamellar phase, Eur. J. Pharm. Sci., 2024, 192, 106635 CrossRef CAS PubMed.
- J. P. Shrestha, Y. P. Subedi, L. Chen and C. W. T. Chang, A mode of action study of cationic anthraquinone analogs: A new class of highly potent anticancer agents, MedChemComm, 2015, 6, 2012–2022 RSC.
- R. S. Keri, K. Chand, S. Budagumpi, S. B. Somappa, S. A. Patil and B. M. Nagaraja, An overview of benzo [b] thiophene-based medicinal chemistry, Eur. J. Med. Chem., 2017, 138, 1002–1033 CrossRef CAS PubMed.
- A. Acosta, P. De la Cruz, P. De Miguel, E. Diez-Barra, A. De la Hoz, F. Langa, A. Loupy, M. Majdoub, N. Martin, C. Sanchez and C. Seoane, Microwave-assisted synthesis of heterocyclic fused quinones in dry media, Tetrahedron Lett., 1995, 36, 2165–2168 CrossRef CAS.
- G. Shukla, G. K. Verma, A. Nagaraju, R. K. Verma, K. Raghuvanshi and M. S. Singh, DMAP-mediated one-pot domino thienannulation: a versatile, regioselective and green mechanochemical route to naphtho[2,3-b]thiophenes, RSC Adv., 2013, 3, 13811–13817 RSC.
- K. Ueda, M. Yamashita, K. Sakaguchi, H. Tokuda and A. Iida, Concise Synthesis of Heterocycle-Fused Naphthoquinones by Employing Sonogashira Coupling and Tandem Addition-Elimination/Intramolecular Cyclization, Chem. Pharm. Bull., 2013, 61, 648–654 CrossRef CAS PubMed.
- Y. Liu, W. Ge, J. Han, Y. Zhu, Y. Shi and H. Wu, DABCO-catalyzed three-component reaction for the synthesis of naphtho[2,3-b]thiophene-4,9-diones, Tetrahedron Lett., 2017, 58, 2665–2669 CrossRef CAS.
- D. S. Baranov, A. A. Popov, D. A. Nevostruev, A. A. Dmitriev, Y. V. Gatilov and E. S. Kobeleva, One-Pot Synthesis of 2-R-Naphtho [2,3-b] thiophene-4, 9-diones via Cyclization of 2-(R-Ethynyl)-1, 4-naphthoquinones with Na2S2O3, J. Org. Chem., 2021, 86, 11361–11369 Search PubMed.
- S. K. Manik, S. Haldar, A. Bhattachrya, U. Debnath, S. A. Ali, S. Jana, M. Shit, P. K. Haldar, S. Pradhan, D. K. Hazra, K. K. Maity, S. K. Ghorai, S. Samanta, A. K. Misra, K. Jana and N. K. Hazra, Design, synthesis, and in vitro and in vivo biological evaluation of 2-amino-naphtho[2,3-b]thiophene-4,9-dione derivatives as potent anticancer agents, Eur. J. Med. Chem., 2025, 296, 117728 Search PubMed.
- (a) M. Panda, A. Bera, D. Dandapat, P. Barik, S. Dhara, M. Ikbal, K. Pramanik, S. Pramanik and S. Samanta, Grindstone Telescopic Approach for Pyrrolodiazepines Through Pictet-Spengler-Type Reaction and Solvent-Free Synthesis of Quinazolines, Asian J. Org. Chem., 2025, e00118 Search PubMed; (b) P. Zhang, C. Liu, L. Yu, H. Hou, W. Sun and F. Ke, Synthesis of benzimidazole by mortar–pestle grinding method, Green Chem. Lett. Rev., 2021, 14, 612–619 Search PubMed.
- Y. Gu, Multicomponent reactions in unconventional solvents: state of the art, Green Chem., 2012, 14, 2091–2128 RSC.
- B. B. Toure and D. G. Hall, Natural product synthesis using multicomponent reaction strategies, Chem. Rev., 2009, 109, 4439–4486 CrossRef CAS PubMed.
- S. Zhi, X. Ma and W. Zhang, Consecutive multicomponent reactions for the synthesis of complex molecules, Org. Biomol. Chem., 2019, 17, 7632–7650 RSC.
- E. Ruijter and R. V. Orru, Multicomponent reactions–opportunities for the pharmaceutical industry, Drug Discovery Today:Technol., 2013, 10, e15–e20 CrossRef PubMed.
- (a) C. Greever, K. Ramirez-Aguilar and J. Connelly, Connections between laboratory research and climate change: what scientists and policy makers can do to reduce environmental impacts, FEBS Lett., 2020, 594, 3079–3085 CrossRef CAS PubMed; (b) S. Kar, H. Sanderson, K. Roy, E. Benfenati and J. Leszczynski, Green chemistry in the synthesis of pharmaceuticals, Chem. Rev., 2021, 122, 3637–3710 CrossRef PubMed.
- P. Anastas and N. Eghbali, Green chemistry: principles and practice, Chem. Soc. Rev., 2010, 39, 301–312 RSC.
- P. Barik, C. K. Pal, S. Chakraborty, S. A. Azad, P. Patra, R. Jana, M. R. Molla and S. Samanta, NaBH4-Induced Chemoselective Mechanochemical Neat Synthesis of Bio-Active Dihydrofuran/-Hydroxy-α, β-Unsaturated Esters and Synthetic Application to Isoindoles, Eur. J. Org Chem., 2024, 27, e202400534 CrossRef CAS.
- R. A. Sheldon, The E factor 25 years on: the rise of green chemistry and sustainability, Green Chem., 2017, 19, 18–43 RSC.
- (a) CCDC 2203321: Experimental Crystal Structure Determination, 2016, DOI:10.5517/ccdc.csd.cc2cyqwy; (b) CCDC 2257893: Experimental Crystal Structure Determination, 2016, DOI:10.5517/ccdc.csd.cc2fsj81; (c) CCDC 2329614: Experimental Crystal Structure Determination, 2016, DOI:10.5517/ccdc.csd.cc2j64vr; (d) CCDC 2466758: Experimental Crystal Structure Determination, 2016, DOI:10.5517/ccdc.csd.cc2nsvv5.
| This journal is © The Royal Society of Chemistry 2026 |