Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
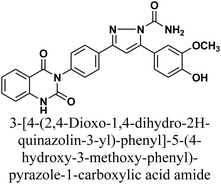
Vanillin-tethered quinazolin-2,4-dione analogues through five- and/or six-membered nitrogen-containing heterocycles as antibacterial agents: synthesis, biological evaluation and molecular docking study
Aboubakr H. Abdelmonsef *a,
Saleh M. Elnabya,
Ahmed M. Mosallama,
Huda R. M. Rashdan
*a,
Saleh M. Elnabya,
Ahmed M. Mosallama,
Huda R. M. Rashdan b,
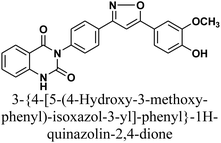
Hesham M. Alsoghier
b,
Hesham M. Alsoghier a and
Mohamed A. Raslanc
a and
Mohamed A. Raslanc
aDepartment of Chemistry, Faculty of Science, South Valley University, 83523 Qena, Egypt. E-mail: aboubakr.ahmed@sci.svu.edu.eg
bChemistry of Natural and Microbial Products Department, Pharmaceutical and Drug Industries Research Institute, National Research Centre, 33 El Buhouth St, Dokki, Giza 12622, Egypt
cDepartment of Chemistry, Faculty of Science, Aswan University, 81528 Aswan, Egypt
First published on 27th January 2026
Abstract
The escalating threat of drug-resistant bacteria confirms the urgent need to develop and identify new and potent antibacterial inhibitors. In the present research study, the molecular hybridization strategy involved the synthesis of vanillin linked to a quinazolin-2,4-dione skeleton through five- and/or six-membered nitrogen-containing heterocycles such as pyrazole, isoxazole and/or pyrimidine 1–15 and testing them for their in vitro antibacterial studies. The vanillin-derived chalcone 1 was synthesized by treatment of 3-(4-acetyl-phenyl)-1H-quinazolin-2,4-dione with 4-hydroxy-3-methoxybenzaldehyde (vanillin) through a base-catalyzed Claisen–Schmidt condensation reaction, then used as a key precursor for synthesis of a series of 14 bioactive compounds 2–15 by treatment with nitrogen nucleophiles via Michael addition reaction. Subsequently, the newly prepared compounds were structurally confirmed by well-known spectroscopic techniques such as FT-IR, 1H-NMR, 13C-NMR, mass spectroscopy, and elemental analysis. All the molecules were evaluated in vitro for their antibacterial activities, showing moderate to good potency, with compounds 2, 5 and 11 having significant activity comparable to the standard drug ciprofloxacin. Moreover, molecular docking simulations were also conducted to investigate the possible interactions of the bioactive moieties of compounds, ciprofloxacin, co-crystalized ligand and two references with the target enzyme. The binding scores correlated with the biological activity of the compounds, with energy scores ranging from −9.9 to −7.4 kcal mol−1, comparable to ciprofloxacin. Interestingly, the molecules 2, 5 and 11 formed highly stable H-bond, pi-cation and pi–sigma interactions with the amino acid residues LYS487, SER401, THR352, GLN348, SER347, and SER303, which are playing an important role in ensuring efficient binding of the ligand with glucoseamine-6-phosphate (GlcN-6-P) synthase (PDBID׃1moq). The enhanced antibacterial activity was ascribed to the presence of electron donating-groups –OH, and –OCH3 attached to pyrazole and/or isoxazole moieties, respectively. To evaluate the structural confirmation and chemical reactivity behavior of the quinazolin-2,4-dione skeleton, semiempirical studies were achieved. Overall, the findings underscore the potential of vanillin linked to quinazolin-2,4-dione skeletons through five- and/or six-membered nitrogen-containing heterocycles, particularly 2, 5 and 11, as promising candidates for development of potent antibacterial inhibitors.
1. Introduction
The rise of antimicrobial resistance (AMR) poses a serious global health threat.1–3 By 2050, drug-resistant bacterial infections will be responsible for approximately 1.9 million deaths.4,5 Overcoming the resistance problem needs the development of new and potential molecules with novel mode of action. Therefore, synthesis of vanillin linked to quinazolin-2,4-dione skeletons through five- and/or six-membered nitrogen-containing heterocycles through a molecular hybridization strategy is disclosed in this regard.Quinazolin-2,4-dione skeletons are versatile building blocks in pharmaceutical chemistry and drug discovery.6–10 They are some of the most prominent, widely used classes for preparation of various drugs with potential biological effects11 such as anticancer,12 anti-inflammatory,13 antimicrobial14,15 and anti-tuberculosis.16
Vanillin, a biologically active compound, has several pharmacological actions17 such as anti-inflammatory,18 anticancer,19 antidiabetic20 and antimicrobial effects.21 In addition, it is a promising building block for chemical and pharmaceutical industries.
Five- and/or six-membered nitrogen-containing heterocycles such as pyrazole, isoxazole and/or pyrimidine are fruitful sources of inspiration of medicinal and pharmaceutical chemists due to their spectrum of extensive biological and pharmaceutical potentials such as anticancer,22 anti-inflammatory,23 antiviral,9,24,25 antibacterial properties.26,27
Molecular hybridization strategy is a combination of different molecules to develop hybrid one with improved efficacy.28 The design strategy of newly prepared compounds 1–15 is declared in Fig. 1. Fig. 2 shows the approved marketed antibacterial drugs with quinazolin-2,4-dione structure, vanillin and five- and/or six-membered nitrogen-containing heterocycles.
 | ||
| Fig. 1 Design the concept of molecular hybridization of compounds incorporating vanillin linked to quinazolin-2,4-dione skeleton through five- and/or six-membered nitrogen-containing heterocycles. | ||
 | ||
| Fig. 2 Antibacterial drugs with quinazoline structure, vanillin with five- and/or six-membered nitrogen-containing heterocycles. | ||
Considering these results and our ongoing fascination with the synthesis of bioactive heterocycles,29–35 herein, the specific incorporation of vanillin via Claisen–Schmidt condensation and Michael addition to form particular series of fifteen quinazolin-2,4-diones was performed. Their chemical structures and purities were elucidated by spectroscopic and elemental analyses. In addition, their antibacterial efficacy was in vitro investigated against two Gram +ve strains, i.e. (Bacillus subtilis ATCC 6633 and Staphylococcus aureus NRRL B-767) and two Gram −ve strains, i.e. (Escherichia coli ATCC 25955, Pseudomonas aeruginosa ATCC 10145). Moreover, molecular docking studies of newly quinazolin-2,4-dione derivatives, ciprofloxacin, co-crystalized ligand and two references were conducted against glucoseamine-6-phosphate (GlcN-6-P) synthase, using PyRx virtual screening tool. Finally, their toxicity and pharmacokinetics were screened by the aim of AdmetSAR, and Molinspiration in order to determine the major scaffolds that possess promising antibacterial properties.
2. Results and discussion
2.1. Chemistry
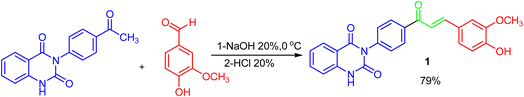
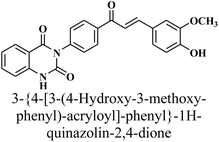
Given the urgent need for new antibacterial inhibitors, the hybrid compounds incorporating known bioactive moieties have gained increasing interest. The general procedure for the synthesis of key precursor chalcone [(E)– 3-{4-[3-(4-hydroxy-3-methoxy-phenyl)-acryloyl]-phenyl}-1H-quinazolin-2,4-dione] 1 is performed by treatment of 3-(4-acetyl-phenyl)-1H-quinazolin-2,4-dione with vanillin via Claisen–Schmidt condensation reaction, as depicted in Scheme 1. This reaction, catalyzed by amount of base NaOH, achieved yield of up to 79%. The spectral data and elemental analysis were utilized for identification of chemical structure of α–β-unsaturated carbonyl compound 1. The FT-IR spectrum showed the characteristic absorption bands at 1727 cm−1 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O carbonyl stretching) due to the conjugation of π-electrons of benzene ring and olefinic linkage. 1H-NMR spectrum showed the characteristic signals as two distinct doublets at δ 8.30 and 8.33 ppm attributed to olefinic protons adjacent to carbonyl group. The coupling constant (J value) of the doublets is 13.00 Hz characterized of an E-configuration (Trans). The other peaks appeared at δ 11.57 ppm for –NH-quinazoline, in addition to eleven phenyl protons as multiplet at δ 7.01–8.04 ppm, and a singlet at δ 3.89 ppm for methoxy group –OCH3. Also, 13C-NMR spectrum showed characteristic signals for –(C
O carbonyl stretching) due to the conjugation of π-electrons of benzene ring and olefinic linkage. 1H-NMR spectrum showed the characteristic signals as two distinct doublets at δ 8.30 and 8.33 ppm attributed to olefinic protons adjacent to carbonyl group. The coupling constant (J value) of the doublets is 13.00 Hz characterized of an E-configuration (Trans). The other peaks appeared at δ 11.57 ppm for –NH-quinazoline, in addition to eleven phenyl protons as multiplet at δ 7.01–8.04 ppm, and a singlet at δ 3.89 ppm for methoxy group –OCH3. Also, 13C-NMR spectrum showed characteristic signals for –(C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) at δ 191 ppm, δ 122 and 145 ppm for olefinic carbons, and δ 55 ppm for –OCH3. Finally, the structure of 1 was confirmed by its mass spectrum (m/z = 414) which agreed with the molecular formula (see Experimental part).
O) at δ 191 ppm, δ 122 and 145 ppm for olefinic carbons, and δ 55 ppm for –OCH3. Finally, the structure of 1 was confirmed by its mass spectrum (m/z = 414) which agreed with the molecular formula (see Experimental part).
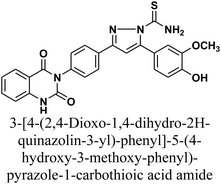
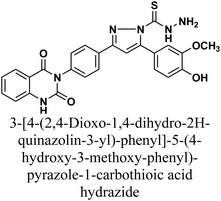
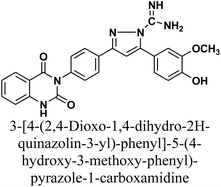
The product 1 is utilized as a good intermediate for a facile synthesis of quinazolin-2,4-dione skeleton through five- and/or six-membered nitrogen-containing heterocycles such as pyrazole, isoxazole and/or pyrimidine 2–15, as declared in Schemes 2 and 3.
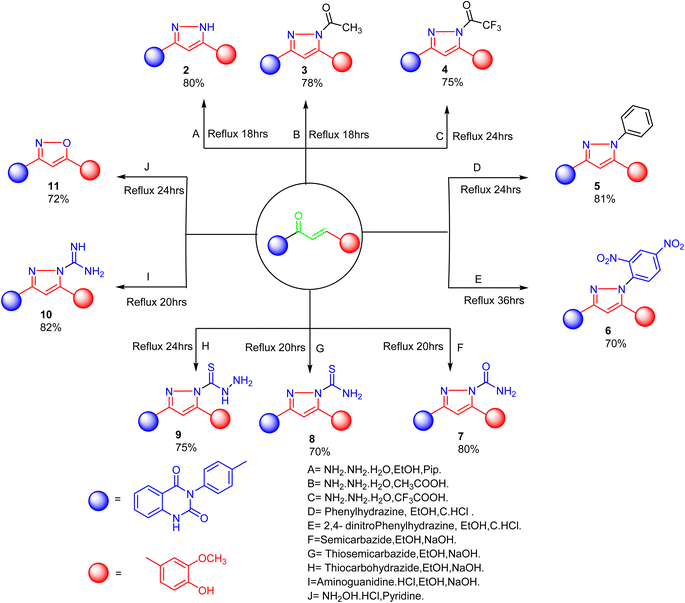 | ||
| Scheme 2 Synthesis of a series of vanillin linked to quinazolin-2,4-dione skeleton through five-membered nitrogen-containing heterocycles 2–11. | ||
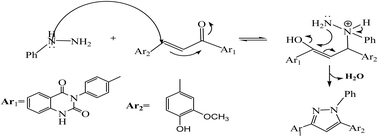
As depicted in Scheme 2, treatment of chalcone 1 with an equimolar amount of different nitrogen nucleophiles namely, hydrazine, phenyl hydrazine, 2,4-dinitro phenylhydrazine, semicarbazide, thiosemicarbazide, thiocarbohydrazide, aminoguanidine and/or hydroxylamine under thermal conditions afforded a series of vanillin linked to quinazolin-2,4-dione skeleton through five-membered nitrogen-containing heterocycles 2–11, respectively in good yields. The hydrazinolysis of chalcone 1 in ethanol under reflux afforded the compound 2. On the other hand, the addition of acetic acid and/or trifluoroacetic acid (TFA) provided the expected pyrazoles 3 and 4, respectively. The reaction of hydrazine (binucleophile) with chalcone 1 was carried out utilizing aza-Michael addition, leading to the reaction, then 1,5-exo-trig cyclization, which is followed by dehydration. For derivatives 2–4, The FT-IR spectral data exhibited absorption bands at 3210–3301 and 1597–1604 cm−1 confirming the presence of NH and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N groups of pyrazole moieties, correspondingly. In addition, 13C-NMR of compound 3 exhibited a characteristic signals at δ 26.91 ppm for –COCH3, 149.95 ppm for –C
N groups of pyrazole moieties, correspondingly. In addition, 13C-NMR of compound 3 exhibited a characteristic signals at δ 26.91 ppm for –COCH3, 149.95 ppm for –C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N and 169.45 ppm for –COCH3.
N and 169.45 ppm for –COCH3.
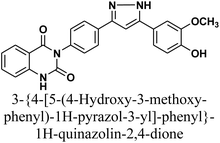
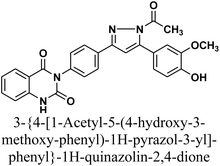
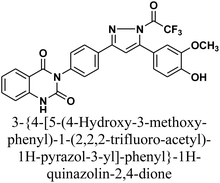
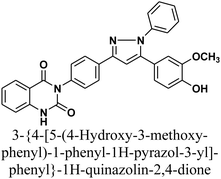
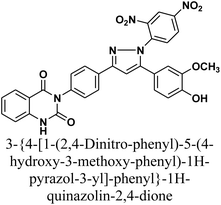
The reaction of chalcone 1 with phenyl hydrazine and/or 2,4-dinitro phenylhydrazine under reflux yielded the pyrazoles 5–6, respectively. The suggested mechanism for the synthesis of compound 5, is represented in Scheme 3. Moreover, PM6 geometrical optimization and heat of formation of two expected conformers of product 5 confirmed that the left side conformer (57.34347 kcal mol−1) is more energetically stable than the right side one (57.31583 kcal mol−1) by 27.64 cal mol−1, as declared in Fig. 3.
 | ||
| Fig. 3 Geometrical optimized structures of compound 5. Left side is more energetically favorable conformer than right side one. | ||
Additionally, treatment of compound 1 with semicarbazide, thiosemicarbazide and thiocarbohydrazide afforded the compounds 7–9, respectively. Then, the treatment of the chalcone 1 with aminoguanidine in ethanol with catalytic amount of NaOH under reflux gave compound 10. Finally, addition of hydroxylamine to compound 1 in refluxed pyridine afforded isoxazole 11. The newly prepared compounds were structurally confirmed by their spectral analyses. The FT-IR spectral data showed the disappearance of the characteristic peak for conjugated carbonyl group (–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C–CO–) at 1727 cm−1, along with C
C–CO–) at 1727 cm−1, along with C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C double bond stretching band at 1577 cm−1 and the appearance of NH stretching band at 3288 cm−1. The 1H-NMR spectra declared the absence of signals related to the CH
C double bond stretching band at 1577 cm−1 and the appearance of NH stretching band at 3288 cm−1. The 1H-NMR spectra declared the absence of signals related to the CH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH group of the compound 1, with the appearance of new signal related to olefinic CH group (pyrazole rings) in interference with aromatic proton at δ 6.52–8.26 ppm (see Experimental part).
CH group of the compound 1, with the appearance of new signal related to olefinic CH group (pyrazole rings) in interference with aromatic proton at δ 6.52–8.26 ppm (see Experimental part).
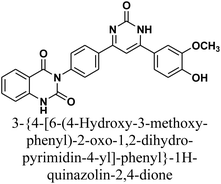
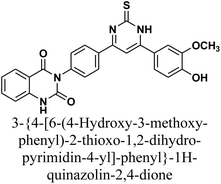
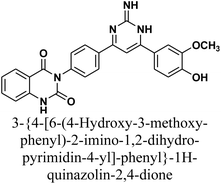
As declared in Scheme 4, cyclization of chalcone 1 with an equimolar amount of various nitrogen nucleophiles namely, urea, thiourea, guanidine and/or benzamidinium chloride yielded a series of vanillin-attached quinazolin-2,4-dione skeleton through six-membered nitrogen-containing heterocycles (pyrimidine) 12–15, respectively, in good yields. The spectral data and elemental analysis were utilized for identification of chemical structures of compounds. FT-IR spectra of these compounds exhibited characteristic absorption bands at 3315–3192 for NH, and 1720–1669 cm−1 for C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O groups. Considering the 1H-NMR spectra of new pyrimidine derivatives 12–15, NH protons of quinazolin-2,4-dione skeletons were observed as singlet signals at the range δ 11.50–11.63 ppm. In addition, the aromatic protons were observed at δ 6.58–8.39 ppm. A table of comparison with the previously published reports36,37 is included in the SI File, as Table S1. The spectral data of all prepared compounds are included in the SI File, as Fig. S1–S57.
O groups. Considering the 1H-NMR spectra of new pyrimidine derivatives 12–15, NH protons of quinazolin-2,4-dione skeletons were observed as singlet signals at the range δ 11.50–11.63 ppm. In addition, the aromatic protons were observed at δ 6.58–8.39 ppm. A table of comparison with the previously published reports36,37 is included in the SI File, as Table S1. The spectral data of all prepared compounds are included in the SI File, as Fig. S1–S57.
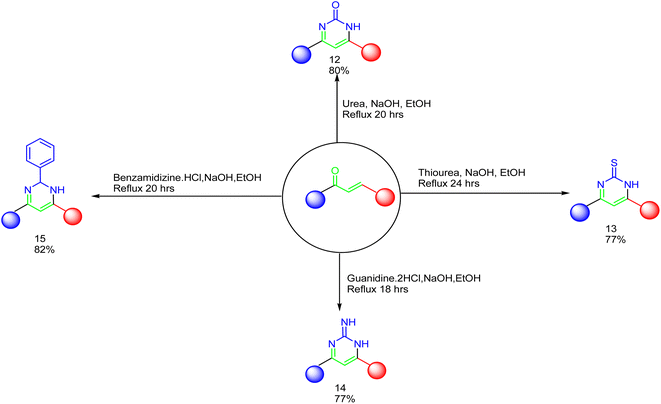 | ||
| Scheme 4 Synthesis of a series of vanillin-attached quinazolin-2,4-dione skeleton through pyrimidine ring 13–15. | ||
2.2 Antibacterial activity
Microbial chemotherapeutic resistance considered as a serious threat to global public health, reducing the effectiveness of antimicrobial drugs and causing infections to become difficult or impossible to treat. A critical strategy for combating this challenge is the development of new antimicrobials, including novel heterocyclic compounds. In our study, the antibacterial assay was performed for fifteen quinazolin-2,4-diones incorporating vanillin connecting by biologically active five- and/or six-membered nitrogen rings. These hybrid compounds were tested for their antibacterial activity against two Gram +ve strains, i.e. (Bacillus subtilis ATCC 6633 and Staphylococcus aureus NRRL B-767) and two Gram −ve strains, i.e. (Escherichia coli ATCC 25955, Pseudomonas aeruginosa ATCC 10145). The MIC of the tested novel compounds was also mentioned, as shown in Table 1.| Sample no. | Minimum inhibitory concentration (MIC, µg mL−1) | |||
|---|---|---|---|---|
| Escherichia coli | Pseudomonas aeruginosa | Bacillus subtilis | Staphylococcus aureus | |
| a Ciprofloxacin was used as standard drugs as control,38,39 ND: not determined. | ||||
| 1 | 160 ± 0.28 | 80 ± 0.14 | ND | 40 |
| 2 | 2.5 ± 0.18 | 5 ± 0.25 | 10 ± 0.22 | 2.5 ± 0.51 |
| 3 | 80 ± 0.03 | 40 ± 0.21 | ND | 16 ± 0.22 |
| 4 | 40 ± 0.15 | 20 ± 0.15 | ND | ND |
| 5 | 2.5 ± 0.08 | 5 ± 0.18 | 10 ± 0.02 | 5 ± 0.07 |
| 6 | 10 ± 0.09 | 15 ± 0.17 | 40 ± 0.18 | 160 ± 0.80 |
| 7 | 20 ± 0.11 | 5 ± 0.20 | 10 ± 0.25 | 20 ± 0.14 |
| 8 | 20 ± 0.12 | 40 ± 0.15 | 80 ± 0.45 | ND |
| 9 | ND | 160 ± 0.17 | 40 ± 0.21 | ND |
| 10 | 120 ± 0.05 | 40 ± 0.31 | 20 ± 0.05 | 15 ± 0.23 |
| 11 | 10 ± 0.04 | 5 ± 0.01 | 2.5 ± 0.08 | 5 ± 0.05 |
| 12 | ND | 16 ± 0.07 | 7 ± 0.14 | ND |
| 13 | 10 ± 0.11 | 40 ± 0.18 | 80 ± 0.16 | 120 ± 0.16 |
| 14 | 40 ± 0.14 | ND | 80 ± 0.11 | ND |
| 15 | 20 ± 0.17 | 15 ± 0.10 | 10 ± 0.24 | 10 ± 0.03 |
| Ciprofloxacin | 5 ± 0.11 | 7 ± 0.33 | 2.5 ± 0.17 | 1.25 ± 0.05 |
Minimum inhibition concentration (MIC) was defined as the average of the lowest concentrations with no observation of microbial growth. The results obtained revealed that compounds 2, 5 and 11 exhibited significant antimicrobial potency against all the tested microbes at low concentration ranging from 2.5 to 10 µg mL−1. The results showed also, that compounds 7 and 15 exhibited a considerable wide broad spectrum of antibacterial potency against all the strains of the tested pathogenic bacteria with low concentrations ranged from 5 to 20 µg mL−1. Otherwise, compound 6 revealed significant antimicrobial effect at low concentrations against the two tested Gram-negative bacteria (Escherichia coli ATCC 25955 and Pseudomonas aeruginosa ATCC 10145). Meanwhile, compound 10 showed strong antibacterial effect at relatively low concentrations against the two tested Gram-positive bacteria (Bacillus subtilis ATCC 6633 and Staphylococcus aureus NRRL B-767).
2.3. Structure–activity relationship
In this part, we summarize the effect of substituents on antibacterial activity of the newly synthesized compounds. The enhanced antibacterial activity of the molecules 2, 5 and 11 was ascribed to the presence of electron donating-groups –OH, and –OCH3 attached to pyrazole and/or isoxazole moieties, respectively. In addition, aromatic and heteroaromatic rings are favored as hydrophobic heads over aliphatic and alicyclic moieties.2.4. Computational studies
Additionally, the redocking process of 2-amino-2-deoxy-6-O-phosphono-alpha-D-glucopyranose (a co-crystallized ligand) with the active site of GlcN-6-P enzyme was performed (RMSD ˂ 2 Å) and the results exhibited similar fitness to the docked molecules. 2-Amino-2-deoxy-6-O-phosphono-alpha-D-glucopyranose showed nine H-bond interactions with CYS300, THR302, SER303, SER347, SER349, THR352 at 2.91, 3.00, 2.81, 2.80, 2.93, 2.84, 2.14, 2.15 and 2.01 Å, respectively (Table 2).
Finally, anticapsin (a reference drug 1) showed eleven HB interactions with THR302, SER303, SER347, GLN348, SER349, THR352, GLU488, and VAL399 at 3.00, 2.74, 2.87, 2.99, 2.88, 2.28, 2.01and 2.08 Å, respectively. Whereas, tetaine (a reference drug 2) exhibited five HB interactions with the active site residues of GlcN-6-P enzyme44 (Table 2).
Moreover, ADMET properties of the compounds were simulated to determine their potential as drug candidates against bacterial diseases, as declared in Table 3. The hydrogen bond donors (HBD) and hydrogen bond acceptors (HBA) of the tested compounds are in acceptable range (≤10) and (≤5), respectively; indicating high solubility of these compounds. In addition, they have good absorption properties (%HIA). Their lipophilicities (measured by MLOGP) are in an optimal value. Further, they exhibited high gastrointestinal GI absorption. None of them exhibited blood–brain barrier (BBB) permeability; indicating no side effect on central nervous system CNS. When taken as a whole, the pharmacokinetics properties of the compounds agreed with Lipinski's rule of five; confirming that could serve as antibacterial drug candidates.
| Molecular weight (g mol−1) | BBB permeant | %Human intestinal absorption (HIA+) | Log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) p p |
TPSA A2 | HBA | HBD | N rotatable | N violations | Bioavailability | GI absorption | AMES toxicity | Carcinogenicity | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
a Log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) p, logarithm of partition coefficient between n-octanol and water; n rotatable, number of rotatable bonds; TPSA, topological polar surface area; HBA, number of hydrogen bond acceptors; HBD, number of hydrogen bond donors. The acceptable ranges are as follows; Mol wt: (130–500); %Human oral absorption: >80% high, <25% low; donor HB: (0.0–6.0); accept HB: (2.0–20.0). p, logarithm of partition coefficient between n-octanol and water; n rotatable, number of rotatable bonds; TPSA, topological polar surface area; HBA, number of hydrogen bond acceptors; HBD, number of hydrogen bond donors. The acceptable ranges are as follows; Mol wt: (130–500); %Human oral absorption: >80% high, <25% low; donor HB: (0.0–6.0); accept HB: (2.0–20.0). |
|||||||||||||
| 1 | 414.41 | No | 98.21 | 2.84 | 101.39 | 5 | 2 | 5 | 0 | 0.55 | High | Nontoxic | Noncarcinogenic |
| 2 | 426.42 | No | 98.98 | 2.83 | 113.00 | 5 | 3 | 4 | 0 | 0.55 | High | Nontoxic | Noncarcinogenic |
| 3 | 468.46 | No | 99.62 | 3.21 | 119.21 | 6 | 2 | 5 | 0 | 0.55 | High | Nontoxic | Noncarcinogenic |
| 4 | 522.43 | No | 100.00 | 3.51 | 119.21 | 9 | 2 | 6 | 0 | 0.55 | Low | Nontoxic | Noncarcinogenic |
| 5 | 502.52 | No | 99.63 | 4.10 | 102.14 | 5 | 2 | 5 | 0 | 0.55 | High | Nontoxic | Noncarcinogenic |
| 6 | 592.52 | No | 98.87 | 2.53 | 193.78 | 9 | 2 | 7 | 0 | 0.55 | Low | Nontoxic | Noncarcinogenic |
| 7 | 469.45 | No | 99.67 | 2.91 | 145.23 | 6 | 3 | 5 | 0 | 0.55 | Low | Nontoxic | Noncarcinogenic |
| 8 | 485.51 | No | 98.94 | 2.87 | 160.25 | 5 | 3 | 5 | 0 | 0.55 | Low | Nontoxic | Noncarcinogenic |
| 9 | 500.53 | No | 96.65 | 2.91 | 172.28 | 6 | 4 | 6 | 0 | 0.55 | Low | Nontoxic | Noncarcinogenic |
| 10 | 468.46 | No | 99.09 | 2.91 | 152.01 | 6 | 4 | 5 | 0 | 0.55 | Low | Nontoxic | Noncarcinogenic |
| 11 | 427.41 | No | 99.43 | 2.83 | 110.35 | 6 | 2 | 4 | 0 | 0.55 | High | Nontoxic | Noncarcinogenic |
| 12 | 454.43 | No | 96.32 | 2.57 | 130.07 | 6 | 3 | 4 | 0 | 0.55 | High | Nontoxic | Noncarcinogenic |
| 13 | 470.50 | No | 78.46 | 2.53 | 145.09 | 5 | 3 | 4 | 0 | 0.55 | Low | Nontoxic | Noncarcinogenic |
| 14 | 453.45 | No | 92.55 | 2.57 | 136.85 | 6 | 4 | 6 | 0 | 0.55 | Low | Nontoxic | Noncarcinogenic |
| 15 | 514.53 | No | 98.54 | 3.85 | 110.10 | 6 | 2 | 5 | 0 | 0.55 | High | Nontoxic | Noncarcinogenic |
| Ciprofloxacin | 331.34 | No | 97.95 | 1.28 | 74.57 | 5 | 2 | 3 | 0 | 0.55 | High | Nontoxic | Noncarcinogenic |
| Glucopyranose | 259.15 | No | 86.28 | −3.64 | 172.51 | 9 | 6 | 3 | 1 | 0.11 | Low | Nontoxic | Noncarcinogenic |
| Anticapsin | 199.20 | No | 88.76 | 3.00 | 92.92 | 5 | 2 | 3 | 0 | 0.55 | High | Nontoxic | Noncarcinogenic |
| Tetaine | 270.28 | No | 86.23 | −1.21 | 122.02 | 6 | 3 | 6 | 0 | 0.55 | High | Nontoxic | Noncarcinogenic |
In addition, Fig. 5 shows the physicochemical characters such as solubility, polarity, lipophilicity, saturation, flexibility, and size of compounds 2, 5 and 12 “bioavailability radar”. The inner pink indicates the optimal values for acceptable bioavailability. The molecular weights of compounds and afatinib are in acceptable range (˂500 g mol−1). The compounds and afatinib have rotatable bonds within the allowed range (<10 bond) that enhance their flexibility. To conclude, compounds 2, 5 and 12 are predicted to have acceptable bioavailability.
3. Materials and methods
3.1. Chemistry
All chemicals and solvents were obtained from Aldrich Chemical Co. and used without purification. The melting points of the obtained compounds were investigated on capillary tubes by using MEL-TEMP II, and are uncorrected. The completion of the reactions was monitored by thin layer chromatography TLC using benzene/EtOH (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) as mobile phase, and the spots were visualized by irradiation with UV light (254 nm). FT-IR, 1H&13C-NMR and MS spectra were recorded on Shimadzu 408, Bruker Vect. 22, JEOL, and Mass 5988 Mass spectrometer, respectively. Elemental analyses were performed at Cairo University, Egypt.
1) as mobile phase, and the spots were visualized by irradiation with UV light (254 nm). FT-IR, 1H&13C-NMR and MS spectra were recorded on Shimadzu 408, Bruker Vect. 22, JEOL, and Mass 5988 Mass spectrometer, respectively. Elemental analyses were performed at Cairo University, Egypt.
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretches); 1H-NMR (400 MHz, DMSO, δ, ppm): 3.89 (s, 3H, OCH3), 7.01–8.04 (m, 13H, Ar H), 8.30 (d, 1H, CH = CO), 8.33 (d, 1H, CH = CH-Ph), 11.57 (s, 1H, NH); 13C-NMR (100 MHz, DMSO, δ, ppm): 55, 114, 115, 117, 118, 122, 127, 128, 129, 135, 139, 140, 145, 149, 162, 169, 191; MS: m/z = 414 [M+]. Elemental analysis calculated for C24H18N2O5: C, 69.56%; H, 4.38%; N, 6.76%. Found: C, 69.85%; H, 4.62%; N, 6.45%.
O stretches); 1H-NMR (400 MHz, DMSO, δ, ppm): 3.89 (s, 3H, OCH3), 7.01–8.04 (m, 13H, Ar H), 8.30 (d, 1H, CH = CO), 8.33 (d, 1H, CH = CH-Ph), 11.57 (s, 1H, NH); 13C-NMR (100 MHz, DMSO, δ, ppm): 55, 114, 115, 117, 118, 122, 127, 128, 129, 135, 139, 140, 145, 149, 162, 169, 191; MS: m/z = 414 [M+]. Elemental analysis calculated for C24H18N2O5: C, 69.56%; H, 4.38%; N, 6.76%. Found: C, 69.85%; H, 4.62%; N, 6.45%.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretches); 1H-NMR (400 MHz, DMSO-d6, δ, ppm): 3.90 (s, 3H, OCH3), 5.50 (s, 1H, NH), 7.25–8.08 (m, 12H, Ar–H + CH), 11.60 (s, 1H, NH); MS: m/z = 426 [M+]. Elemental analysis calculated for C24H18N4O4: C, 67.60%; H, 4.25%; N, 13.14%. Found: C, 67.90%; H, 4.51%; N, 12.71%.
O stretches); 1H-NMR (400 MHz, DMSO-d6, δ, ppm): 3.90 (s, 3H, OCH3), 5.50 (s, 1H, NH), 7.25–8.08 (m, 12H, Ar–H + CH), 11.60 (s, 1H, NH); MS: m/z = 426 [M+]. Elemental analysis calculated for C24H18N4O4: C, 67.60%; H, 4.25%; N, 13.14%. Found: C, 67.90%; H, 4.51%; N, 12.71%.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretches); 1H-NMR (400 MHz, DMSO-d6, δ, ppm): 1.90 (s, 3H, CH3), 3.98 (s, 3H, OCH3), 7.23–8.08 (m, 12H, Ar–H + CH), 11.62 (s, 1H, NH); 13C-NMR (100 MHz, DMSO, δ, ppm): 26, 55, 102, 114, 115, 122, 127, 128, 129, 135, 136, 139, 140, 149, 162, 169; MS: m/z = 468 [M+]. Elemental analysis calculated for C26H20N4O5: C, 66.66%; H, 4.30%; N, 11.96%. Found: C, 66.75%; H, 4.41%; N, 11.75%.
O stretches); 1H-NMR (400 MHz, DMSO-d6, δ, ppm): 1.90 (s, 3H, CH3), 3.98 (s, 3H, OCH3), 7.23–8.08 (m, 12H, Ar–H + CH), 11.62 (s, 1H, NH); 13C-NMR (100 MHz, DMSO, δ, ppm): 26, 55, 102, 114, 115, 122, 127, 128, 129, 135, 136, 139, 140, 149, 162, 169; MS: m/z = 468 [M+]. Elemental analysis calculated for C26H20N4O5: C, 66.66%; H, 4.30%; N, 11.96%. Found: C, 66.75%; H, 4.41%; N, 11.75%.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretches); 1H-NMR (400 MHz, DMSO-d6, δ, ppm): 3.95 (s, 3H, OCH3), 7.20–8.08 (m, 12H, Ar–H + CH), 11.58 (s, 1H, NH); MS: m/z = 522 [M+]. Elemental analysis calculated for C26H17F3N4O5: C, 59.77%; H, 3.28%; F, 10.91%; N, 10.72%. Found: C, 59.88%; H, 3.37%; F, 10.99%; N, 10.60%.
O stretches); 1H-NMR (400 MHz, DMSO-d6, δ, ppm): 3.95 (s, 3H, OCH3), 7.20–8.08 (m, 12H, Ar–H + CH), 11.58 (s, 1H, NH); MS: m/z = 522 [M+]. Elemental analysis calculated for C26H17F3N4O5: C, 59.77%; H, 3.28%; F, 10.91%; N, 10.72%. Found: C, 59.88%; H, 3.37%; F, 10.99%; N, 10.60%.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretches); 1H-NMR (400 MHz, DMSO-d6, δ, ppm): 3.90 (s, 3H, OCH3), 6.78–8.08 (m, 17H, Ar–H + CH), 11.59 (s, 1H, NH); 13C-NMR (100 MHz, DMSO, δ, ppm): 55, 100, 112, 114, 115, 118, 122, 125, 127, 128, 129, 135, 139, 140, 162; MS: m/z = 502 [M+]. Elemental analysis calculated for C30H22N4O4: C, 71.70%; H, 4.41%; N, 11.15%. Found: C, 71.81%; H, 4.52%; N, 11.01%.
O stretches); 1H-NMR (400 MHz, DMSO-d6, δ, ppm): 3.90 (s, 3H, OCH3), 6.78–8.08 (m, 17H, Ar–H + CH), 11.59 (s, 1H, NH); 13C-NMR (100 MHz, DMSO, δ, ppm): 55, 100, 112, 114, 115, 118, 122, 125, 127, 128, 129, 135, 139, 140, 162; MS: m/z = 502 [M+]. Elemental analysis calculated for C30H22N4O4: C, 71.70%; H, 4.41%; N, 11.15%. Found: C, 71.81%; H, 4.52%; N, 11.01%.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretches), 1495 (NO2 asymmetric stretch), 1392 (NO2 symmetric stretch); 1H-NMR (400 MHz, DMSO-d6, δ, ppm): 3.90 (s, 3H, OCH3), 7.23–8.81 (m, 15H, Ar–H + CH), 11.57 (s, 1H, NH); MS: m/z = 592 [M+]. Elemental analysis calculated for C30H20N6O8: C, 60.81%; H, 3.40%; N, 14.18%. Found: C, 60.90%; H, 3.49%; N, 14.02%.
O stretches), 1495 (NO2 asymmetric stretch), 1392 (NO2 symmetric stretch); 1H-NMR (400 MHz, DMSO-d6, δ, ppm): 3.90 (s, 3H, OCH3), 7.23–8.81 (m, 15H, Ar–H + CH), 11.57 (s, 1H, NH); MS: m/z = 592 [M+]. Elemental analysis calculated for C30H20N6O8: C, 60.81%; H, 3.40%; N, 14.18%. Found: C, 60.90%; H, 3.49%; N, 14.02%.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretches); 1H-NMR (400 MHz, DMSO-d6, δ, ppm): 3.99 (s, 3H, OCH3), 5.60 (s, 2H, NH2), 6.98–8.01 (m, 12H, Ar–H + CH), 11.57 (s, 1H, NH); MS: m/z = 469 [M+]. Elemental analysis calculated for C25H19N5O5: C, 63.96%; H, 4.08%; N, 14.92%. Found: C, 64.10%; H, 4.18%; N, 14.72%.
O stretches); 1H-NMR (400 MHz, DMSO-d6, δ, ppm): 3.99 (s, 3H, OCH3), 5.60 (s, 2H, NH2), 6.98–8.01 (m, 12H, Ar–H + CH), 11.57 (s, 1H, NH); MS: m/z = 469 [M+]. Elemental analysis calculated for C25H19N5O5: C, 63.96%; H, 4.08%; N, 14.92%. Found: C, 64.10%; H, 4.18%; N, 14.72%.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretches); 1H-NMR (400 MHz, DMSO-d6, δ, ppm): 3.89 (s, 3H, OCH3), 5.02 (s, 2H, NH2), 7.23–8.32 (m, 12H, Ar–H + CH), 11.60 (s, 1H, NH); 13C-NMR (100 MHz, DMSO, δ, ppm): 55, 102, 115, 117, 122, 125, 126, 128, 133, 136, 139, 144, 152, 155, 157, 164, 174; MS: m/z = 485 [M+]. Elemental analysis calculated for C25H19N5O4S: C, 61.85%; H, 3.94%; N, 14.42%; S, 6.60%. Found: C, 61.97%; H, 3.99%; N, 14.25%; S, 6.81%.
O stretches); 1H-NMR (400 MHz, DMSO-d6, δ, ppm): 3.89 (s, 3H, OCH3), 5.02 (s, 2H, NH2), 7.23–8.32 (m, 12H, Ar–H + CH), 11.60 (s, 1H, NH); 13C-NMR (100 MHz, DMSO, δ, ppm): 55, 102, 115, 117, 122, 125, 126, 128, 133, 136, 139, 144, 152, 155, 157, 164, 174; MS: m/z = 485 [M+]. Elemental analysis calculated for C25H19N5O4S: C, 61.85%; H, 3.94%; N, 14.42%; S, 6.60%. Found: C, 61.97%; H, 3.99%; N, 14.25%; S, 6.81%.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretches); 1H-NMR (400 MHz, DMSO-d6, δ, ppm): 3.85 (s, 3H, OCH3), 5.02 (s, 2H, NH2), 6.02 (s, 1H, NH-CS), 7.24–8.07 (m, 12H, Ar–H + CH), 11.50 (s, 1H, NH); MS: m/z = 500 [M+]. Elemental analysis calculated for C25H20N6O4S: C, 59.99%; H, 4.03%; N, 16.79%; S, 6.41%. Found: C, 60.12%; H, 4.17%; N, 16.65%; S, 6.52%.
O stretches); 1H-NMR (400 MHz, DMSO-d6, δ, ppm): 3.85 (s, 3H, OCH3), 5.02 (s, 2H, NH2), 6.02 (s, 1H, NH-CS), 7.24–8.07 (m, 12H, Ar–H + CH), 11.50 (s, 1H, NH); MS: m/z = 500 [M+]. Elemental analysis calculated for C25H20N6O4S: C, 59.99%; H, 4.03%; N, 16.79%; S, 6.41%. Found: C, 60.12%; H, 4.17%; N, 16.65%; S, 6.52%.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretches); 1H-NMR (400 MHz, DMSO-d6, δ, ppm): 3.98 (s, 3H, OCH3), 5.57 (s, 2H, NH2), 5.96 (s, 1H, =NH), 7.20–7.95 (m, 12H, Ar–H + CH), 11.58 (s, 1H, NH); MS: m/z = 468 [M+]. Elemental analysis for C25H20N6O4 calculated: C, 64.10%; H, 4.30%; N, 17.94%.Found: C, 64.22%; H, 4.41%; N, 17.72%.
O stretches); 1H-NMR (400 MHz, DMSO-d6, δ, ppm): 3.98 (s, 3H, OCH3), 5.57 (s, 2H, NH2), 5.96 (s, 1H, =NH), 7.20–7.95 (m, 12H, Ar–H + CH), 11.58 (s, 1H, NH); MS: m/z = 468 [M+]. Elemental analysis for C25H20N6O4 calculated: C, 64.10%; H, 4.30%; N, 17.94%.Found: C, 64.22%; H, 4.41%; N, 17.72%.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretches); 1H-NMR (400 MHz, DMSO-d6, δ, ppm): 3.90 (s, 3H, OCH3), 7.22–8.08 (m, 12H, Ar–H + CH), 11.58 (s, 1H, NH); MS: m/z = 427 [M+]. Elemental analysis for C24H17N3O5 calculated: C, 67.44%; H, 4.01%; N, 9.83%. Found: C, 67.55%; H, 4.21%; N, 9.75%.
O stretches); 1H-NMR (400 MHz, DMSO-d6, δ, ppm): 3.90 (s, 3H, OCH3), 7.22–8.08 (m, 12H, Ar–H + CH), 11.58 (s, 1H, NH); MS: m/z = 427 [M+]. Elemental analysis for C24H17N3O5 calculated: C, 67.44%; H, 4.01%; N, 9.83%. Found: C, 67.55%; H, 4.21%; N, 9.75%.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretches); 1H-NMR (400 MHz, DMSO-d6, δ, ppm): 3.89 (s, 3H, OCH3), 5.18 (s, 1H, NH), 7.23–8.08 (m, 12H, Ar–H + CH), 11.63 (s, 1H, NH); MS: m/z = 454 [M+]. Elemental analysis calculated for C25H18N4O5: C, 66.08%; H, 3.99%; N, 12.33%. Found: C, 66.15%; H, 4.09%; N, 12.21%.
O stretches); 1H-NMR (400 MHz, DMSO-d6, δ, ppm): 3.89 (s, 3H, OCH3), 5.18 (s, 1H, NH), 7.23–8.08 (m, 12H, Ar–H + CH), 11.63 (s, 1H, NH); MS: m/z = 454 [M+]. Elemental analysis calculated for C25H18N4O5: C, 66.08%; H, 3.99%; N, 12.33%. Found: C, 66.15%; H, 4.09%; N, 12.21%.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretches); 1H-NMR (400 MHz, DMSO-d6, δ, ppm): 3.90 (s, 3H, OCH3), 7.22–8.08 (m, 12H, Ar–H + CH), 11.58 (s, 1H, NH); MS: m/z = 470 [M+]. Elemental analysis calculated for C25H18N4O4S: C, 63.82%; H, 3.86%; N, 11.91%; S, 6.81%. Found: C, 63.90%; H, 3.97%; N, 11.75%; S, 6.92%.
O stretches); 1H-NMR (400 MHz, DMSO-d6, δ, ppm): 3.90 (s, 3H, OCH3), 7.22–8.08 (m, 12H, Ar–H + CH), 11.58 (s, 1H, NH); MS: m/z = 470 [M+]. Elemental analysis calculated for C25H18N4O4S: C, 63.82%; H, 3.86%; N, 11.91%; S, 6.81%. Found: C, 63.90%; H, 3.97%; N, 11.75%; S, 6.92%.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretches); 1H-NMR (400 MHz, DMSO-d6, δ, ppm): 3.80 (s, 3H, OCH3), 6.85–8.01 (m, 14H, Ar–H + CH+2NH), 11.50 (s, 1H, NH); 13C-NMR (100 MHz, DMSO, δ, ppm): 55, 102, 115, 118, 121, 126, 128, 129, 133, 135, 143, 152, 156, 158, 164, 169; MS: m/z = 453 [M+]. Elemental analysis calculated for C25H19N5O4: C, 66.22%; H, 4.22%; N, 15.44%. Found: C, 66.34%; H, 4.33%; N, 15.28%.
O stretches); 1H-NMR (400 MHz, DMSO-d6, δ, ppm): 3.80 (s, 3H, OCH3), 6.85–8.01 (m, 14H, Ar–H + CH+2NH), 11.50 (s, 1H, NH); 13C-NMR (100 MHz, DMSO, δ, ppm): 55, 102, 115, 118, 121, 126, 128, 129, 133, 135, 143, 152, 156, 158, 164, 169; MS: m/z = 453 [M+]. Elemental analysis calculated for C25H19N5O4: C, 66.22%; H, 4.22%; N, 15.44%. Found: C, 66.34%; H, 4.33%; N, 15.28%.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretches); 1H-NMR (400 MHz, DMSO-d6, δ, ppm): 3.89 (s, 3H, OCH3), 7.06–8.39 (m, 17H, Ar–H + CH), 11.58 (s, 1H, NH); 13C-NMR (100 MHz, DMSO, δ, ppm): 55, 100, 115, 117, 120, 121, 122, 127, 128, 129, 130, 131, 133, 135, 141, 144, 152, 162, 169; MS: m/z = 514 [M+]. Elemental analysis calculated for C31H22N4O4: C, 72.36%; H, 4.31%; N, 10.89%. Found: C, 72.56%; H, 4.41%; N, 10.79%.
O stretches); 1H-NMR (400 MHz, DMSO-d6, δ, ppm): 3.89 (s, 3H, OCH3), 7.06–8.39 (m, 17H, Ar–H + CH), 11.58 (s, 1H, NH); 13C-NMR (100 MHz, DMSO, δ, ppm): 55, 100, 115, 117, 120, 121, 122, 127, 128, 129, 130, 131, 133, 135, 141, 144, 152, 162, 169; MS: m/z = 514 [M+]. Elemental analysis calculated for C31H22N4O4: C, 72.36%; H, 4.31%; N, 10.89%. Found: C, 72.56%; H, 4.41%; N, 10.79%.3.2. Biological evaluation
Minimum inhibitory concentration (MIC) and antimicrobial efficacy of the novel derivatives were estimated against two Gram-negative bacteria (Escherichia coli ATCC 25955, Pseudomonas aeruginosa ATCC 10145) and two Gram-positive bacteria (Bacillus subtilis ATCC 6633 and Staphylococcus aureus NRRL B-767). The microbes under study were provided by Al-Azhar University, Egypt. They were cultivated in Mueller Hinton broth at 35 ± 2 °C for 24 h. The antimicrobial activity and MIC were carried out as described by Qader et al.46–483.3. In silico molecular docking study and ADMET analysis
The crystal structure of glucoseamine-6-phosphate (GlcN-6-P) synthase was retrieved from the RCSB database (PDBID׃1moq).49 The structure was prepared by removing water, heteroatoms, and co-crystalized ligand utilizing Discovery Studio 3.5 Client.50 All synthesized molecules 1–15, ciprofloxacin, co-crystalized ligand and two references were used as a library in the docking process. The 2D chemical structures of the molecules, ciprofloxacin, co-crystalized ligand and two references were sketched using ChemDraw software 16.0, and then converted to the 3D structures by using Open Babel GUI tool.51 The energy minimization for the enzyme file and ligand molecules was performed using CHARMm52 and AMBER53 Force Fields, respectively. PyRx virtual screening tool54 was utilized for the docking approach between the ligand molecules and the target. The 2D and 3D poses of the docked complexes were visualized using Discovery Studio visualizer software. Finally, the pharmacokinetics properties of the newly molecules, ciprofloxacin, co-crystalized ligand and two references were calculated using AdmetSAR, and SwissADME web-based tools. Finally, geometric optimization of the best docked molecules was investigated by a semiempirical modeling with PM6 basis set (MOPAC 2009 software).55,564. Conclusion
Herein, a new series of quinazolin-2,4-diones incorporating vanillin connecting by biologically active five- and/or six-membered nitrogen rings was rationally designed and synthesized employing Claisen–Schmidt condensation and Michael addition reactions. Structural elucidation was validated by means of spectroscopic techniques and elemental analysis. The antibacterial potentials of all molecules were evaluated, showing moderate to good potency, with compounds 2, 5 and 11 performing significant activity comparable to the standard drug ciprofloxacin. In silico molecular docking simulations were performed against glucoseamine-6-phosphate (GlcN-6-P) synthase (PDBID׃1moq), and the molecules 2, 5 and 11 formed highly binding affinities towards the target. The presence of electron donating-groups –OH, and –OCH3 attached to aromatic and heteroaromatic moieties could improve the antibacterial potentials of the compounds. When taken as a whole, the results exhibited that the molecules 2, 5 and 11 are promising lead compounds for the development of novel and potent GlcN-6-P inhibitors for bacterial treatment. Finally, in vivo evaluation of the most active compounds in appropriate animal infection models will provide critical insights into their therapeutic potentials, guiding further lead optimization toward the development of novel and potent antibacterial inhibitors.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5ra08138f.References
- E. Palma, B. Tilocca and P. Roncada, Antimicrobial Resistance in Veterinary Medicine: An Overview, Int. J. Mol. Sci., 2020, 1–21 Search PubMed.
- M. A. Salam, et al., Antimicrobial Resistance: A Growing Serious Threat for Global Public Health, Healthcare, 2023, 11, 1946 CrossRef PubMed.
- F. Prestinaci, P. Pezzotti and A. Pantosti, Antimicrobial resistance: A global multifaceted phenomenon, Pathog. Glob. Health, 2015, 109, 309–318 Search PubMed.
- S. U. Haq, et al., Exploring the intricacies of antimicrobial resistance: Understanding mechanisms, overcoming challenges, and pioneering innovative solutions, Eur. J. Pharmacol., 2025, 998, 177511 Search PubMed.
- T. R. Walsh, A. C. Gales, R. Laxminarayan and P. C. Dodd, Antimicrobial Resistance: Addressing a Global Threat to Humanity, PLoS Med, 2023, 20, 12–15 CrossRef PubMed.
- O. Babatunde, et al., Dihydroquinazolin-4(1H)-one derivatives as novel and potential leads for diabetic management, Mol. Divers., 2022, 26, 849–868 Search PubMed.
- A. Hassan, A. M. Mosallam, A. O. A. Ibrahim, M. Badr and A. H. Abdelmonsef, Novel 3-phenylquinazolin-2,4(1H,3H)-diones as dual VEGFR-2/c-Met-TK inhibitors: design, synthesis, and biological evaluation, Sci. Rep., 2023, 13, 1–17 Search PubMed.
- S. Panda and U. P. Tripathy, Quinazolone: A molecule of significant pharmacological and biological activity, Res. J. Pharm. Technol., 2013, 6, 849–855 Search PubMed.
- T. P. Selvam and P. V. Kumar, Quinazoline Marketed drugs – A Review, Res. Pharm., 2011, 1, 1–21 Search PubMed.
- P. T. Kirinde Arachchige and C. S. Yi, Synthesis of Quinazoline and Quinazolinone Derivatives via Ligand-Promoted Ruthenium-Catalyzed Dehydrogenative and Deaminative Coupling Reaction of 2-Aminophenyl Ketones and 2-Aminobenzamides with Amines, Org. Lett., 2019, 21, 3337–3341 Search PubMed.
- H. S. El-Sheshtawy, et al., Synthesis, Structural, and Theoretical Studies of Quinazoline-2,4-dione Derivatives, Polycycl. Aromat. Compd., 2017, 39, 279–286 CrossRef.
- F. Tanveer, M. F. Anwar, B. Siraj and S. Zarina, Evaluation of anti-EGFR potential of quinazoline derivatives using molecular docking: An in silico approach, Biotechnol. Appl. Biochem., 2022, 69, 1226–1237 CrossRef CAS PubMed.
- A. H. Abdelmonsef, et al., A search for antiinflammatory therapies: Synthesis, in silico investigation of the mode of action, and in vitro analyses of new quinazolin-2,4-dione derivatives targeting phosphodiesterase-4 enzyme, J. Heterocycl. Chem., 2022, 59, 474–492 Search PubMed.
- M. El-Naggar, H. R. M. Rashdan and A. H. Abdelmonsef, Cyclization of Chalcone Derivatives: Design, Synthesis, In Silico Docking Study, and Biological Evaluation of New Quinazolin-2,4-diones Incorporating Five-, Six-, and Seven-Membered Ring Moieties as Potent Antibacterial Inhibitors, ACS Omega, 2023, 8, 27216–27230 Search PubMed.
- A. H. Abdelmonsef, et al., Antimicrobial Potential of Novel Quinazolin-2,4-dione Derivatives Bearing One and/or Two Amide Moieties: In Vitro Evaluation, Molecular Docking, and Semiempirical Calculations, ChemistrySelect, 2025, 10, 1–19 Search PubMed.
- A. Dutta, et al., Design and Synthesis of Quinazolinone-Triazole Hybrids as Potent Anti-Tubercular Agents, ACS Appl. Bio Mater., 2022, 5(9), 4413–4424, DOI:10.1021/acsabm.2c00562.
- A. Olatunde, A. Mohammed, M. A. Ibrahim, N. Tajuddeen and M. N. V. Shuaibu, A food additive with multiple biological activities, Eur. J. Med. Chem. Reports, 2022, 5, 100055 CAS.
- E. Costantini, et al., Assessment of the Vanillin Anti-Inflammatory and Regenerative Potentials in Inflamed Primary Human Gingival Fibroblast, Mediators Inflamm., 2021, 2021, 1–9 Search PubMed.
- H. R. M. Rashdan and A. H. Abdelmonsef, Towards Covid-19 TMPRSS2 enzyme inhibitors and antimicrobial agents: Synthesis, antimicrobial potency, molecular docking, and drug-likeness prediction of thiadiazole-triazole hybrids, J. Mol. Struct., 2022, 1268, 133659 CrossRef CAS PubMed.
- V. F. Salau, et al., Vanillin improves glucose homeostasis and modulates metabolic activities linked to type 2 diabetes in fructose–streptozotocin induced diabetic rats, Arch. Physiol. Biochem., 2024, 130, 169–182 CrossRef CAS PubMed.
- D. J. Fitzgerald, et al., Mode of antimicrobial action of vanillin against Escherichia coli, Lactobacillus plantarum and Listeria innocua, J. Appl. Microbiol., 2004, 97, 104–113 CrossRef CAS PubMed.
- S. Lee and J. Sperry, Isolation and biological activity of azocine and azocane alkaloids, Bioorganic Med. Chem., 2022, 54, 116560 CrossRef CAS PubMed.
- C. A. Anosike, O. Obidoa and L. U. Ezeanyika, Membrane stabilization as a mechanism of the anti-inflammatory activity of methanol extract of garden egg (Solanum aethiopicum), DARU, J. Pharm. Sci., 2012, 20, 1 Search PubMed.
- C. M. M. Gómez and V. V. Kouznetsov, Recent Developments on Antimicrobial Quinoline Chemistry, Microb. Pathog. Strateg. Combat. Them Sci. Technol. Educ., 2013, 666–677 Search PubMed.
- H. A. Mohamed, et al., Discovery a novel of thiazolo[3,2-a]pyridine and pyrazolo[3,4-d]thiazole derivatives as DNA gyrase inhibitors; design, synthesis, antimicrobial activity, and some in-silico ADMET with molecular docking study, J. Mol. Struct., 2023, 1287, 135671 CrossRef CAS.
- S. M. Gomha, M. M. Edrees, R. A. M. Faty, Z. A. Muhammad and Y. N. Mabkhot, Microwave-assisted one pot three-component synthesis of some novel pyrazole scaffolds as potent anticancer agents, Chem. Cent. J., 2017, 11, 2–13 CrossRef PubMed.
- A. Masri, et al., Antibacterial effects of quinazolin-4(3h)-one functionalized-conjugated silver nanoparticles, Antibiotics, 2019, 8, 1–19 Search PubMed.
- C. Viegas-Junior, A. Danuello, V. da Silva Bolzani, E. J. Barreiro and C. A. Fraga, Molecular Hybridization: A Useful Tool in the Design of New Drug Prototypes, Curr. Med. Chem., 2007, 14, 1829–1852 CrossRef CAS PubMed.
- S. M. Gomha, et al., Thiazole-Based Thiosemicarbazones: Synthesis, Cytotoxicity Evaluation and Molecular Docking Study, Drug Des. Devel. Ther., 2021, 15, 659–677 CrossRef PubMed.
- H. R. M. Rashdan, A. H. Abdelmonsef, M. M. Abou-Krisha and T. A. Yousef, Synthesis, Identification, Computer-Aided Docking Studies, and ADMET Prediction of Novel Benzimidazo-1,2,3-triazole Based Molecules as Potential Antimicrobial Agents, Molecules, 2021, 26, 7119 CrossRef CAS PubMed.
- A. O. A. Ibrahim, et al., New quinazolin-2,4-dione derivatives incorporating acylthiourea, pyrazole and/or oxazole moieties as antibacterial agents via DNA gyrase inhibition, RSC Adv., 2024, 14, 17158–17169 RSC.
- A. H. M. Hussein, A.-B. A. El-Adasy, A. M. El-Saghier, M. Olish and A. H. Abdelmonsef, Synthesis, characterization, in silico molecular docking, and antibacterial activities of some new nitrogen-heterocyclic analogues based on a p- phenolic unit, RSC Adv., 2022, 12, 12607–12621 RSC.
- A. H. Abdelmonsef, M. Omar, H. R. M. Rashdan, M. M. Taha and A. M. Abobakr, Synthesis, characterization, computer-aided docking studies, and anti-fungal activity of two-armed quinazolin-2,4-dione derivatives, J. Mol. Struct., 2024, 1316, 1–9 CrossRef.
- A. H. Abdelmonsef, A. M. El-Saghier and A. M. Kadry, Ultrasound-assisted green synthesis of triazole-based azomethine/thiazolidin-4-one hybrid inhibitors for cancer therapy through targeting dysregulation signatures of some Rab proteins, Green Chem. Lett. Rev., 2023, 16, 2150394 CrossRef.
- A. Hassan, et al., Design and biological evaluation of 3-substituted quinazoline-2,4(1H,3H)-dione derivatives as dual c-Met/VEGFR-2-TK inhibitors, J. Enzyme Inhib. Med. Chem., 2023, 38, 2189578 CrossRef PubMed.
- A. H. Abdelmonsef and A. M. Mosallam, Synthesis, in vitro biological evaluation and in silico docking studies of new quinazolin-2,4-dione analogues as possible anticarcinoma agents, J. Heterocycl. Chem., 2020, 57, 1637–1654 CrossRef CAS.
- M. El-Naggar, H. R. M. Rashdan and A. H. Abdelmonsef, Cyclization of Chalcone Derivatives: Design, Synthesis, In Silico Docking Study, and Biological Evaluation of New Quinazolin-2,4-diones Incorporating Five-, Six-, and Seven-Membered Ring Moieties as Potent Antibacterial Inhibitors, ACS Omega, 2023, 8, 27216–27230 CrossRef CAS PubMed.
- R. V.-R. Satyendra, K. A.-A. Vishnumurthy and H. M. Vagdevi, et al., In vitro antimicrobial and molecular docking of dichloro substituted benzoxazole derivatives, Med. Chem. Res., 2012, 21, 4193–4199 CrossRef CAS.
- A. Lather, S. Sharma and A. Khatkar, Aesculin based glucosamine-6-phosphate synthase inhibitors as novel preservatives for food and pharmaceutical products: in silico studies, antioxidant, antimicrobial and preservative efficacy evaluation, BMC Chem., 2021, 15, 1–11 Search PubMed.
- O. Ebenezer, P. Awolade, N. Koorbanally and P. Singh, New library of pyrazole–imidazo[1,2-α]pyridine molecular conjugates: Synthesis, antibacterial activity and molecular docking studies, Chem. Biol. Drug Des., 2020, 95, 162–173 CrossRef CAS PubMed.
- S. Abid, A. Abdula, M. Al Marjani and Q. Abdulhameed, Synthesis, Antimicrobial, Antioxidant and Docking Study of Some Novel 3,5-Disubstituted-4,5-dihydro-1H-pyrazoles Incorporating Imine Moiety, Egypt. J. Chem., 2018, 739–749 Search PubMed.
- S. Gupta, V. Luxami and K. Paul, Unlocking the Antibacterial Potential of Naphthalimide–Coumarins to Overcome Drug Resistance with Antibiofilm and Membrane Disruption Ability against Escherichia coli, ACS Appl. Mater. Interfaces, 2025, 17, 4380–4399 CrossRef PubMed.
- S. Gupta and K. Paul, Membrane-active substituted triazines as antibacterial agents against Staphylococcus aureus with potential for low drug resistance and broad activity, Eur. J. Med. Chem., 2023, 258, 115551 CrossRef CAS PubMed.
- J. Stefaniak, M. G. Nowak, M. Wojciechowski, S. Milewski and A. S. Skwarecki, Inhibitors of glucosamine-6-phosphate synthase as potential antimicrobials or antidiabetics–synthesis and properties, J. Enzyme Inhib. Med. Chem., 2022, 37, 1928–1956 CrossRef CAS PubMed.
- S. A. A. Hussein, A. A. R. M. Kubba and A. M. Abdula, Docking study of some new 2, 5-Disubstituted-1, 3, 4-thiadiazole derivatives against glucosamine-6-phosphate synthase, Int. J. Appl. Chem., 2016, 12, 1–10 Search PubMed.
- M. M. Qader, et al., Antimicrobial and Antibiofilm Activities of the Fungal Metabolites Isolated from the Marine Endophytes Epicoccum nigrum M13 and Alternaria alternata 13A, Mar. Drugs, 2021, 19, 232 CrossRef CAS PubMed.
- M. A. El-Hashash, S. M. Sherif, A. A. Badawy and H. R. Rashdan, Synthesis of some new antimicrobial 5, 6, 7, 8-tetrahydro-pyrimido [4, 5-b] quinolone derivatives, Der Pharm. Chem, 2014, 6, 23–29 Search PubMed.
- A. Abdel-Aziem, H. R. M. Rashdan, E. Mohamed Ahmed and S. N. Shabaan, Synthesis and cytotoxic activity of some novel benzocoumarin derivatives under solvent free conditions, Green Chem. Lett. Rev., 2019, 12, 9–18 CrossRef CAS.
- H. M. Berman, et al., The protein data bank, Nucleic Acids Res., 2000, 28, 235–242 CrossRef CAS PubMed.
- A. H. Abdelmonsef, et al., Identification of Novel Antagonists for Rab38 Protein by Homology Modeling and Virtual Screening, Comb. Chem. High Throughput Screen., 2016, 19, 1–18 Search PubMed.
- N. M. O'Boyle, et al., Open Babel: An Open chemical toolbox, J. Cheminform., 2011, 3, 33 CrossRef PubMed.
- B. R. Brooks, et al., CHARMM: The biomolecular simulation program, J. Comput. Chem., 2009, 30, 1545–1614 CrossRef CAS PubMed.
- J. A. Maier, C. Martinez, K. Kasavajhala, L. Wickstrom, K. E. Hauser and C. Simmerling, et al., ff14SB: Improving the Accuracy of Protein Side Chain and Backbone Parameters from ff99SB, J. Chem. Theory Comput., 2015, 11, 3696–3713 CrossRef CAS PubMed.
- S. Dallakyan and A. J. Olson, Small-Molecule Library Screening by Docking with PyRx, in Chemical Biology, springer, 2015, vol. 1263, pp. 243–250 Search PubMed.
- J. J. P. Stewart, Optimization of parameters for semiempirical methods I. Method, J. Comput. Chem., 1989, 10, 209–220 CrossRef CAS.
- A. M. Abo-Bakr, H. M. Alsoghier and A. H. Abdelmonsef, Molecular docking, modeling, semiempirical calculations studies and in vitro evaluation of new synthesized pyrimidin-imide derivatives, J. Mol. Struct., 2022, 1249, 131548 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |