Open Access Article
Open Access ArticleFrom experimental data to thermophysical insight: characterizing choline-based ionic liquids using advanced data analysis
Mahasoa-Salina Souvenir-Zafindrajaonaabc,
Jean-Pierre Mbakidib,
Zdeněk Wagnerc,
Sandrine Bouquillon b and
Magdalena Bendová
b and
Magdalena Bendová *ac
*ac
aDepartment of Physical Chemistry, University of Chemistry and Technology Prague, Technická 5, 166 28 Prague 6 – Dejvice, Czech Republic. E-mail: bendovam@vscht.cz; Tel: +420 22044 4297
bInstitut de Chimie Moléculaire de Reims UMR CNRS 7312, Université de Reims Champagne-Ardenne, Boîte n° 44, B.P. 1039, F-51687 Reims, France
cInstitute of Chemical Process Fundamentals CAS, v. v. i., Rozvojová 135/1, 165 02 Prague 6 – Lysolaje, Czech Republic
First published on 19th January 2026
Abstract
Extracting valuable compounds from biomass requires the use of efficient dissolution processes, preferably at mild conditions. In the present study we examine the impact of hydration on the thermophysical properties of four choline-based ionic liquids (CHILs) that were shown to dissolve lignin efficiently in our previous work. Density and isobaric heat capacity across various temperatures and levels of hydration was measured. Due to the hygroscopic nature of these ionic liquids, careful control of the water content and extrapolation to zero water content were performed to derive their pure-phase properties. The extrapolation as well as the measurement uncertainty and outlier detection was carried out by means of a robust non-statistical approach based on mathematical gnostics. Derived apparent molar properties reveal distinct ion–water interactions, underpinning the role of hydration in modulating CHIL performance. These insights advance the fundamental understanding of CHIL–water mixtures and support their development for sustainable biomass processing and other green chemical technologies.
1 Introduction
Choline-derived ionic liquids have been found to show a pronounced application potential, for example as solvents in one-pot saccharification process combining pretreatment and enzymatic hydrolysis of biomass, influencing the catalytic activity of cellulases,1 tribology, gas sorption or CO2 capture,2,3 or drug delivery,4,5 to name but a few. Choline acetate was found to be an efficient, inexpensive and bio-based organocatalyst for the hydroboration of several carbonyl compounds,6 while poly-ionic liquids based on choline were found to significantly improve the performance of LiS battery systems, allowing for a better confinement of polysulfide species7 within the cathodic compartment. In our recent publication,8 we proposed a novel approach towards synthesis of choline based ionic liquids (CHILs) with lactate and levulinate anions, to facilitate their utilization in green processes, such as the treatment of lignocellulosic mass. We compared a classical cross-metathesis and a microwave-assisted cross metathesis of structures based on choline, monocatenar choline esters and bolaform esters to propose accessible and environmentally friendly synthetic pathways to obtain these structures. To take the present work further and design a successful process for lignin dissolution, it is essential to understand the physicochemical properties of the studied ionic liquids. The proposed structures with lactate and levulinate anions are inherently hygroscopic. For this reason, it turned out to be necessary to study their mixtures with water. Indeed, as shown by Tanner et al.9 for choline and geranic acid ionic liquids, water has a pronounced impact on the CHIL properties, ions undergoing reorganization in the presence of water molecules. This is particularly important for the design of processes utilizing ionic liquids in general, and CHILs in particular. For example, hydrated ionic liquids with the addition of salts have been shown to be suitable media for solubilizing and stabilizing proteins. For example, choline and amino acid based ionic liquids were studied by modelling to explore their hydration and specific interactions with NaCl by Kruchinin and Fedotova.10 In the present work, we found that it was challenging to dry the ionic liquids sufficiently to reduce their water content and to characterize them as pure compounds, due to the presence of highly hygroscopic lactate and levulinate anions. However, measuring their thermodynamic and thermophysical properties as a function of water content enabled us to obtain the properties of pure and hydrated CHILs, covering a range of water concentration present in samples used in real applications, along with a fundamental insight into the interaction between CHIL ions and water molecules via derived apparent molar properties. Additionally, the uncertainty in the density and molar heat capacity values extrapolated to zero water content will be discussed in terms of the influence of property measurement uncertainty and uncertainty in composition determination.2 Experimental
2.1 Materials and methods
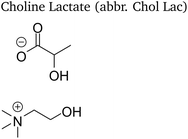
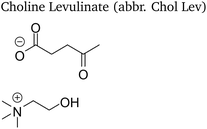
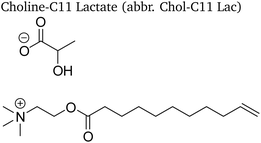
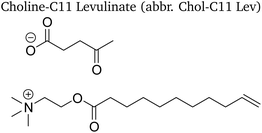
Four choline-based ionic liquids (CHILs) with lactate and levulinate anions synthesized in our previous work8 were studied and are listed in Table 1. A general synthesis procedure and characterization of the studied CHILs is provided in the Electronic Supporting Material. 1-Butanol anhydrous (purity 99.8%, Sigma-Aldrich) was used as received without any additional purification as a calorimetric standard. Throughout the experiments the amount of water in 1-butanol was checked by means of the Karl-Fischer titration and kept lower than 0.075 wt%. In density measurements, tetrachlorethylene and dodecane (H&D Fitzgerald density standards) were used in calibration of the density meter.2.2 Isobaric heat capacity
The isobaric heat capacity as a function of temperature at 0.1 MPa was measured by means of the differential scanning calorimetry. A Setaram µDSC 3 Evo microcalorimeter based on the Tian–Calvet principle using batch cells was used. The continuous method with the heating rate of 0.3 K min−1 was used in this work in the temperature range of 8 °C to 30 °C (281.15 K to 303.15 K): the initial and final temperatures of the scans being held constant for 20 min in the same way as in ref. 11. The calibration data of 1-butanol that was used as the calibration standard were taken from the monograph by Zábranský et al.12 The heat capacity was then calculated by means of the Calisto software. The standard and samples of pure ionic liquids, respectively, were prepared into the measuring cell by weighing using a SCALTEC SBC 21 and KERN & Sohn GmbH balances with an accuracy of 1 × 10−5 g. The typical uncertainty of the experimental isobaric molar heat capacity was found in ref. 11 to be 1.1 J K−1 mol−1. In this work, measurement uncertainty was obtained on data analysis described in Section 3.2.3 Density
The density as a function of temperature at 0.1 MPa was measured by means of an Anton Paar DSA5000 Density and Sound Velocity Analyzer within the temperature range from (288.15 to 343.15) K. The apparatus was calibrated using degassed redistilled water (ρ = (0.997043 ± 0.00001) g cm−3 at T = 298.15 K),13 dodecane standard (ρ = (0.745566 ± 0.00001) g cm−3 at T = 298.15 K) and a high-density standard tetrachloroethylene (ρ = (1.61420 ± 0.00001) g cm−3 at T = 298.15 K), all supplied by H&D Fitzgerald Ltd. The apparatus is kept at the experimental temperature within 0.001 K by means of a built-in solid-state thermostat. The uncertainty of the device stated by the manufacturer is of 0.00001 g cm−3 which was confirmed in our previous work,11,14 however, due to the large viscosity of the measured CHILs it may be as low as 0.0002 g cm−3 as shown in Section 3.2.4 Sample preparation procedure for choline lactate and choline levulinate
After synthesis, the solvents used during synthesis were removed in vacuo and the purity of the studied ionic liquids was verified by 13C and 1H NMR, IR spectroscopy, and elemental analysis. An AC 250 Bruker and a Bruker Alpha-T FTIR spectrometer were used for 1H and IR spectra of liquid and solid compounds at room temperature, respectively. Elemental analyses (C, H and N) were carried out using a PerkinElmer 2400 C, H, N, S element analyser. Details of this characterization are given in the SI. The water content was then determined using the Karl-Fischer titration. Usually, the water content after this initial drying ranged between 10 and 15% by mass.Samples of ionic liquids were then dried under vacuum (approx. 2 mbar) to obtain samples with water contents ranging from <1 to 15% by mass. The water content was determined using the Karl Fischer titration after each drying. For samples in which water concentration was higher than 15% by mass, a known mass of water was added to the ionic liquids with a known water content, the mixture was homogenized by stirring, and the water content was then checked again by KF titration.
The studied choline ionic liquids show a large viscosity at room temperature—199 mPa s and 79 mPa s for choline lactate and levulinate, respectively—which made their sampling challenging. Therefore, they were degassed prior to sampling by an ultrasonic bath and heated at 50 °C for 30 minutes to 1 hour to decrease their viscosity. This allowed their efficient sampling for density and heat capacity measurements.
2.5 Sample preparation procedure for choline-C11 lactate and choline-C11 levulinate
For the choline ester based ILs, the above-described procedure had to be modified, due to their high viscosity. To determine the water content of Chol-C11 ionic liquids, a mixture of the ionic liquid with a solvent of known water content was prepared, with a known composition. The overall water content of the mixture was determined by KF titration, after which the water content of the ionic liquid was calculated.The dry choline esters studied in this work have a viscous oil consistency and are even more viscous at room temperature than the simple choline ionic liquids studied in this work. For this reason, they were first heated for 15 minutes at 50 °C to liquefy them, after which samples with different water contents could be prepared. After that, a defined mass of water was added, and the mixture was heated to 40 °C for 15–30 minutes. The resulting mixture was homogenised by stirring at room temperature for 24 hours.
In this work, the specific heat capacity of all the four studied ionic liquids was measured. However, density was only measured for the Chol Lac and Chol Lev. Although homogenizing the choline ester samples by adding water at higher temperatures and feeding them into the oscillation tube was possible, preventing the formation of bubbles that interfered with the measurements was difficult. Moreover, the samples with higher water contents took on a waxy appearance on cooling to the room temperature. Due to this, it was only possible to prepare samples for heat capacity measurements into the standard batch cells, as the change in viscosity on temperature change appears to have little to no influence on the measurement uncertainty.
3 Data analysis
Experimental data are traditionally analyzed by methods of mathematical statistics assuming normal distribution of errors. This assumption is either violated or there are not enough experimental data to either confirm or reject their normality. Our previous works demonstrated this, as illustrated by the analysis of heat capacity data.15 In certain cases it is impossible to avoid outliers which make statistical data analysis even more difficult. Such data can be analyzed by methods of mathematical gnostics11,15–17 where no prior assumption on the distribution of measurement errors are needed and the methods are robust, i.e. not sensitive to outliers. Marginal analysis of repeated measurements is thus used to detect and remove outliers and to obtain the most probable value of a given data set and to estimate the repeatability of the measurements. Then, to correlate the heat capacities and densities functions of temperature a robust linear regression along a gnostic influence function was used. This enabled us to obtain reliable regression parameters and to derive apparent molar properties discussed in this work. More details on the application of both methods can be found in the recent paper by Parmar et al. and references therein.15The choice of a polynomial as a regression model is based on an expansion to the Taylor series. There are two different strategies depending on the main purpose, smoothing for interpolation or for extrapolation. Each purpose requires a different strategy for choosing an appropriate polynomial order. If a polynomial is used for interpolation, the order is increased while it improves the residuals. A statistical goodness-of-fit test cannot be used because it assumes normal distribution of errors but the residuals show that this assumption is violated. We should not examine just the sum of squares of residuals as in the F-test of χ2-test but compare individual residuals and a posteriori weights resulting from the robust regression. Simple achievement of lowest possible residuals is not the best strategy if stable extrapolation to zero is required. Polynomials are not orthogonal. This means that addition of another parameter changes the values of all parameters estimated by fitting the polynomial of a lower degree. In addition, if too many parameters are used, monotonicity of the dependence is lost and most often a local extremum between zero and the lowest measured concentration occurs. For this reason we stop increasing the polynomial order before the intercept starts changing significantly in which case the intercept would rather reflect the curvature caused by the scattered data. Water mass fraction was thus selected as a concentration unit; a linear model was used for regression of densities and a quadratic model for regression of specific heat capacities.
In case of a statistical linear regression using an assumption of normally distributed errors with known variance, the uncertainty of the model parameters can be calculated. This is not that simple if mathematical gnostics is applied. A gnostic distribution function is a nonparametric kernel estimate transformed from the infinite data support to the finite one. An equation for calculation of uncertainty of the parameters based on the distribution functions of the uncertain data thus cannot be derived. Gnostic distribution functions are available for heat capacities only, as we do not have repeated measurements of densities. Their uncertainties were estimated based on our previous work18 where the repeatability and viscosity correction are expressed as Type B uncertainty approximated with the normal distribution. Even in this case the statistical formula cannot be used. Regression along an influence function is similar to the method of weighted least squares but the weights are not constant, they are iteratively reweighted. Although the model is linear in parameters, the optimization algorithm is nonlinear. The use of simulation, as outlined in the ref. 14, is necessary. We used 15 sets of pseudorandom data with the gnostic distribution for heat capacities and normal distribution of densities. Together with the original experimental data we obtained 16 sets of parameters. Their marginal analysis than gave their uncertainties. The extrapolated value is equal to the intercept, thus the correlation between the parameters is irrelevant.
The total uncertainty budget consists of a random error and an unknown part of a systematic error, the known part of the systematic error was compensated by calibration of the instruments. The random error of the regression parameters were estimated by the above described simulation with the pseudorandom sets. The systematic error caused by presence of impurities can be approximated by method suggested by Chirico et al.19 The measured system is considered a pseudobinary mixture of the studied compound and the pseudocomponent consisting of all impurities. Any extensive quantity Z of such a mixture can be expressed as
| Z = (1 − x)Z* + xZ*(1 ± δ) + ZE | (1) |
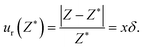 | (2) |
Chirico et al.19 suggest δ = 0.1 and in our case x ≤ 0.05 for all ILs studied. The relative systematic errors of both molar isobaric heat capacities and molar volumes is 0.005, i.e. 0.5%. The density is calculated as M/Vm and thus from application of the error propagation law we get the same relative systematic error for the density.
The ± sign in eqn (1) means that we can approximate δ as an absolute value but do not know its sign. The sign is equal for all measured values because the error is systematic. If we expressed it as a Type B20 uncertainty with normal distribution and combined it into all values of the pseudorandom sets, we would increase the spread of the data in an incorrect way. All data should be shifted in the same direction but we do not know whether to increase or decrease them. Regression of the shifted data will affect the intercept only and the intercept is the required value extrapolated to the zero content of water. The combined uncertainty20 of the extrapolated value is thus obtained by application of the error propagation law. The random errors are independent of the systematic error caused by impurities which means that the covariance is zero. The relative combined uncertainty is thus a square root of sum of squares of individual relative errors. The random error of the extrapolated density is 0.1%, the relative error of the extrapolated isobaric molar heat capacity is 0.5%. Combining them with the systematic error, 0.5% in both cases, we get 0.5% for ρ and 0.7% for Cp.
4 Results and discussion
4.1 Volumetric properties
The experimental density data as a function of water mole fraction for choline levulinate and choline lactate (Tables SI-1 and SI-2) were fitted with a linear equation for all the experimental temperatures. Density extrapolated to zero water content obtained from this fit is then listed as a function of temperature in Table 2 and represented in Fig. 1.| T (K) | Chol Lac | Chol Lev |
|---|---|---|
| ρ (g cm−3) | ρ (g cm−3) | |
| a u(T) = 0.01 K, u(ρ) = 0.0002 g cm−3. | ||
| 287.15 | 1.1594 | 1.1311 |
| 289.15 | 1.1581 | 1.1298 |
| 291.15 | 1.1568 | 1.1286 |
| 293.15 | 1.1555 | 1.1273 |
| 295.15 | 1.1542 | 1.1261 |
| 297.15 | 1.1529 | 1.1248 |
| 299.15 | 1.1516 | 1.1235 |
| 301.15 | 1.1503 | 1.1223 |
| 303.15 | 1.1490 | 1.1210 |
| 305.15 | 1.1478 | 1.1198 |
| 307.15 | 1.1465 | 1.1185 |
| 309.15 | 1.1452 | 1.1172 |
| 311.15 | 1.1439 | 1.1160 |
| 313.15 | 1.1426 | 1.1146 |
 | ||
| Fig. 1 Density of Chol Lac and Chol Lev as a function of temperature. Data extrapolated to zero water content. Lines represent the data fit with eqn (3). | ||
It appears from the experimental data that the density of both choline-based ionic liquids at zero water content is a linear function of temperature:
| ρ = A + B·T | (3) |
| Ionic liquid | A (g cm−3) | B 104 (g cm−3 K−1) | SSE 105 |
|---|---|---|---|
| Chol Lac | 1.3446 | −6.4499 | 2.4373 |
| Chol Lev | 1.3118 | −6.2937 | 3.9782 |
The presence of the lactate anion has been demonstrated to result in a higher density of the studied choline-based ionic liquids in comparison to the levulinate anion. This phenomenon can be attributed to a more compact arrangement between the choline cation and a smaller lactate anion. On the other hand, the additional methylene group and the shift from a hydroxyl substituent to a γ-keto group on the levulinate anion seems to create a looser molecular arrangement due to steric effects.
Apparent molar volumes of ionic liquids in water and water in ionic liquids are listed in Tables SI-7 to SI-11 and shown in Fig. 2 and 3 as a function of water mole fraction x2. They were derived from the experimental density both for the ionic liquids and water in the mixture as
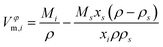 | (4) |
The apparent molar volume, Vφm,1, of both Chol Lac (Fig. 2a) and Chol Lev (Fig. 2b) appears to be slightly lower than their corresponding molar volume and a decreasing function of water mole fraction and temperature. The apparent molar volume of water, Vφm,2, in choline lactate (Fig. 3a) and in Chol Lev (Fig. 3b) appears to be a relatively constant function of water content within the uncertainty of the measurements.
Outliers were detected in density datasets for both ionic liquids. Tables SI-1, SI-2 and SI-7 to SI-11 contain all of the measured and derived data, whereas Fig. 1 and 2 show datasets with removed outliers. Apparent molar properties are sensitive to both density measurement uncertainty and uncertainty in composition. The uncertainty of the mass fraction of water in the samples was mostly determined by the uncertainty of KF titration measurements and was found to be 0.05 wt% which translates to approx. 0.015 mole fraction units. The resulting median uncertainty in apparent molar volume of ionic liquids in water was then obtained from the error propagation law as 0.44 mol cm−3 and 1.04 mol cm−3 for choline lactate and choline levulinate, respectively. However, unknown sample preparation errors may be also reflected in the measurements. For instance, a change in the uncertainty in water content determination by KF titration from 0.05 to 0.1 wt% increases the resulting uncertainty in apparent molar volume by approx. 50%. Measurements carried out with great care are therefore required and outliers should be removed from the datasets before obtaining the derived volumetric properties. In addition, the measurement uncertainty in derived properties is also influenced by the presence of impurities as discussed in Section 3. Outliers detected in original datasets on the extrapolation of the data to zero water content were thus removed from the derived data to avoid misinterpretation of the discussed properties.
Apparent molar properties in general provide useful insight into solvation of the studied structures in solution. Specifically, the apparent molar volume is a difference between the total volume of the solution and the hypothetical volume the solvent, if it were not affected by the presence of the salt, both per mol of the salt present, and is thus the intrinsic volume of the solvated molecule.21 The trends in the measured Vφm,1, i.e. the fact that they are lower than the corresponding molar volume of the pure CHILs and a decreasing function of water content for both CHILs, indicate a shift towards tighter hydration and dense water.22 The dependence of the apparent molar volume of CHILs on water molality further appears to be linear, indicating an overlapping of hydration shells around the ions resulting in a stronger electrostrictive effect on water molecules per ion.23 Indeed, charged groups strongly attract neighbouring water molecules, leading to an increase in local density and smaller partial molar volume of the studied CHILs.24 Comparing the effect of the anions on the studied apparent molar volume it appears that Vφm,1 of lactate is larger than that of levulinate. This is in good agreement with the expectation that a smaller anion would have a smaller effective charge density.21,25,26
To further quantify these effects and to provide a mechanistic insight into the interaction of the studied CHILs with water, the apparent molar volume for both CHILs in water was then correlated with the Redlich–Rosenfeld–Mayer equation:27
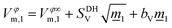 | (5) |
![[m with combining low line]](https://www.rsc.org/images/entities/i_char_006d_0332.gif) 1 is the molality of the ionic liquid in mol kg−1, Vφ∞m,1 is the apparent molar volume of the CHIL at infinite dilution, SDHV is the Debye–Hückel term, and bV represents the deviation from the Debye–Hückel limiting law.
1 is the molality of the ionic liquid in mol kg−1, Vφ∞m,1 is the apparent molar volume of the CHIL at infinite dilution, SDHV is the Debye–Hückel term, and bV represents the deviation from the Debye–Hückel limiting law.
This semitheoretical equation was derived by applying the interionic attraction theory of Debye and Hückel to the concentration dependence of the apparent molar volumes of electrolytes in solution.28–30 The theoretical limiting slope SDHV is a characteristic of the specific solvent for a given electrolyte charge type if the Debye–Hückel theory is obeyed. Thus, for 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 electrolytes, the limiting slope for water is 1.85 cm3 kg1/2 mol−3/2. However, this equation is also a limiting law, therefore, for the parameters to have the theoretical meaning, the concentrations of the solute should not be higher than 1 mol kg−1.30 Because the range of molalities of the studied CHILs in this work (15 to 300 mol kg−1) cannot be considered as even close to infinite dilution, we decided to consider the parameters of eqn (5) as phenomenological and to optimize all of them. Indeed, as shown by Vosburgh et al.,31 different limiting slopes might be obtained for the electrolytes of the same valency in the same solvent at moderately high concentrations, due to the departure from the Debye–Hückel limiting law. In our case, the effect of relatively large ions and possibly weak electrolytes should also be taken into account. However, approximates of the limiting apparent molar volumes that are equal to partial molar volumes at infinite dilution, and a tentative insight into the interactions between the ions and water molecules may still be obtained using Redlich–Rosenfeld–Mayer equation, especially considering that the parameters of the equation were obtained by the robust regression along the gnostic influence function which provides reliable fits for a number of different datasets and regression conditions.
1 electrolytes, the limiting slope for water is 1.85 cm3 kg1/2 mol−3/2. However, this equation is also a limiting law, therefore, for the parameters to have the theoretical meaning, the concentrations of the solute should not be higher than 1 mol kg−1.30 Because the range of molalities of the studied CHILs in this work (15 to 300 mol kg−1) cannot be considered as even close to infinite dilution, we decided to consider the parameters of eqn (5) as phenomenological and to optimize all of them. Indeed, as shown by Vosburgh et al.,31 different limiting slopes might be obtained for the electrolytes of the same valency in the same solvent at moderately high concentrations, due to the departure from the Debye–Hückel limiting law. In our case, the effect of relatively large ions and possibly weak electrolytes should also be taken into account. However, approximates of the limiting apparent molar volumes that are equal to partial molar volumes at infinite dilution, and a tentative insight into the interactions between the ions and water molecules may still be obtained using Redlich–Rosenfeld–Mayer equation, especially considering that the parameters of the equation were obtained by the robust regression along the gnostic influence function which provides reliable fits for a number of different datasets and regression conditions.
The parameters of the Redlich–Rosenfeld–Mayer equation (eqn (5)) are then summarized for all the measured temperatures in Table 4. The limiting apparent molar volumes range between 155 and 159 cm3 mol−1 and 186 and 190 cm3 mol−1 for choline lactate and choline levulinate, respectively. This is in agreement with the finding that the apparent molar volumes of the studied CHILs are lower than their respective pure molar volumes. Apparent densities of both ILs can then be approximated from these values e.g. at 293.15 K as M1/Vφ∞m,1 to be ≈1.24 g cm−3 and ≈1.17 g cm−3 for the choline lactate and choline levulinate, respectively, pointing towards a better and tighter hydration of the choline lactate, i.e. the CHIL with a smaller and more hydrophilic anion.
| T (K) | Vφ∞m,1 (mol cm−3) | SDHV (cm3 kg1/2 mol−3/2) | bV (kg cm3 mol−2) |
|---|---|---|---|
| Chol Lac | |||
| 313.15 | 159.301 | 2.037 | −0.090 |
| 311.15 | 159.052 | 2.045 | −0.090 |
| 309.15 | 158.782 | 2.058 | −0.091 |
| 307.15 | 158.501 | 2.073 | −0.091 |
| 305.15 | 158.225 | 2.087 | −0.092 |
| 303.15 | 157.940 | 2.103 | −0.092 |
| 301.15 | 157.648 | 2.121 | −0.093 |
| 299.15 | 157.351 | 2.139 | −0.094 |
| 297.15 | 157.047 | 2.159 | −0.095 |
| 295.15 | 156.733 | 2.181 | −0.096 |
| 293.15 | 156.412 | 2.204 | −0.097 |
| 291.15 | 156.085 | 2.229 | −0.098 |
| 289.15 | 155.748 | 2.256 | −0.099 |
| 287.15 | 155.400 | 2.285 | −0.100 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
|||
| Chol Lev | |||
| 313.15 | 189.993 | 0.835 | −0.026 |
| 311.15 | 189.736 | 0.840 | −0.027 |
| 309.15 | 189.469 | 0.846 | −0.027 |
| 307.15 | 189.201 | 0.854 | −0.027 |
| 305.15 | 188.921 | 0.863 | −0.027 |
| 303.15 | 188.635 | 0.874 | −0.028 |
| 301.15 | 188.340 | 0.887 | −0.028 |
| 299.15 | 188.042 | 0.901 | −0.029 |
| 297.15 | 187.743 | 0.914 | −0.029 |
| 295.15 | 187.439 | 0.928 | −0.030 |
| 293.15 | 187.130 | 0.943 | −0.030 |
| 291.15 | 186.819 | 0.959 | −0.031 |
| 289.15 | 186.509 | 0.974 | −0.031 |
| 287.15 | 186.192 | 0.990 | −0.032 |
The limiting Debye–Hückel slopes range between 2.04 and 2.29 m3 kg1/2 mol−3/2 and 0.84 and 0.99 m3 kg1/2 mol−3/2 for choline lactate and choline levulinate, respectively. Based on those values, it could be hypothesized that choline levulinate presents a less ideal behaviour than lactate and weaker long-range interactions between the ions and water molecules. This is somewhat supported by the obtained values bV parameter, representing the deviation from the Debye–Hückel law that then appear to show weakly negative values, approx. three times more negative for choline lactate. Strongly negative values of bV being tied to hydrophobic electrolytes32 and to mutual salting out of the ions,33 the present values seem to corroborate the observed weakly hydrophilic behaviour of the studied CHILs, choline levulinate showing a slightly more amphiphilic character.
Further refinement of the understanding of hydration of the studied CHILs is brought by the apparent molar volume of water, Vφm,2. In both Chol Lac and Chol Lev, the apparent molar volume appears to take on a relatively constant value within the measurement uncertainty (1–1.5 cm3 mol−1, median), pointing towards an amphiphilic (or weakly hydrophilic) character of both anions. The added water thus behaves like bulk solvent, producing little net change in per-mole contributions. The local packing environment thus seems to stabilize quickly; the electrostriction effect being already saturated at very low water mole fraction.
Considering these observations, both anions thus exhibit weakly hydrophilic characteristics and structure making behaviour, with Chol Lev showing slightly more amphiphilic character. Such differences have clear implications for solvent performance in biomass-processing applications: lactate-based CHILs may tend towards stronger local ion–water interactions, while levulinate-based CHILs could afford more homogeneous solvent environments. Indeed, as discussed by Reynolds,34 the apparent molar volume values can be linked to Lewis acidity/basicity in the solution, structure making ions being considered as stronger Lewis acids than the water molecules that they are displacing. Consequently, they are good hydrogen bond acceptors and will be able to compete effectively with –OH⋯OH– bonds in lignocellulose. At small water content, dissolution or swelling of biomass in CHILs may be improved, however at higher water concentration, water will dominate the network and CHILs will lose their dissolution power.
4.2 Isobaric heat capacity
The experimental isobaric molar heat capacity for Chol Lac, Chol Lev, Chol-C11 Lac, and Chol-C11 Lev as a function of water mass fraction x2 (Tables SI-3 to SI-6) were fitted with a quadratic equation (Table 5) for all the experimental temperatures (see Data analysis section for more details).| CHIIL | a (J K−1 mol−1) | b (J K−1 mol−1 K−1) | c (J K−1 mol−1 K−2) | d (J K−1 mol−1 K−3) | SSE |
|---|---|---|---|---|---|
| Chol Lac | 314.750 | 0.324 | 8.11 × 10−7 | ||
| Chol Lev | 318.692 | 0.437 | 2.20 × 10−5 | ||
| Chol-C11 Lac | 0.101 | 9.815 | −0.050 | 0.000086 | 1.50 × 10−4 |
| Chol-C11 Lev | 6.879 | 2.6429 | 2.67 × 10−6 |
As the primary output of the heat capacity measurements is the specific heat capacity, extrapolation to zero water content was obtained by fitting the specific heat capacity, rather than the molar heat capacity, as a function of the water mass fraction. The calculated molar heat capacity at zero water content is then listed as a function of temperature in Table 6 for Chol Lac, Chol Lev, Chol-C11 Lac and Chol-C11 Lev and represented in Fig. 4. Molar heat capacity as a function of temperature and water content is then listed for all the studied CHILs in Tables SI-3 to SI-6.
| T (K) | Chol Lac | Chol Lev | Chol-C11 Lac | Chol-C11 Lev |
|---|---|---|---|---|
| Cpm (J K−1 mol−1) | Cpm (J K−1 mol−1) | Cpm (J K−1 mol−1) | Cpm (J K−1 mol−1) | |
| a u(T) = 0.01 K, u(Cpm) = 0.1 J K−1 mol−1. | ||||
| 283.15 | 441.52 | 730.85 | 755.53 | |
| 284.15 | 442.86 | 737.75 | 758.35 | |
| 285.15 | 407.08 | 443.23 | 742.71 | 760.47 |
| 286.15 | 407.49 | 443.60 | 745.30 | 762.89 |
| 287.15 | 407.74 | 444.02 | 748.07 | 764.63 |
| 288.15 | 408.06 | 444.66 | 751.27 | 765.98 |
| 289.15 | 408.35 | 445.05 | 754.54 | 771.45 |
| 290.15 | 408.78 | 445.53 | 758.24 | 771.37 |
| 291.15 | 409.11 | 444.66 | 760.00 | 773.87 |
| 292.15 | 409.42 | 446.13 | 760.15 | 778.15 |
| 293.15 | 409.73 | 446.63 | 762.48 | 780.54 |
| 294.15 | 410.89 | 447.55 | 764.97 | 784.93 |
| 295.15 | 411.10 | 447.88 | 768.13 | 786.66 |
| 296.15 | 411.42 | 448.30 | 770.97 | 789.52 |
| 297.15 | 411.75 | 448.62 | 774.28 | 792.06 |
| 298.15 | 412.13 | 449.02 | 776.72 | 795.06 |
| 299.15 | 412.43 | 449.44 | 780.46 | 798.15 |
| 300.15 | 412.70 | 449.72 | 785.10 | 800.19 |
| 301.15 | 413.04 | 450.27 | 791.17 | 803.66 |
| 302.15 | 413.36 | 450.47 | 799.01 | 804.89 |
| 303.15 | 413.66 | 451.02 | 809.08 | 809.82 |
 | ||
| Fig. 4 Heat capacity of Chol Lac and Chol Lev (a), and Chol-C11 Lac and Chol-C11 Lev (b) as a function of temperature. Data extrapolated to zero water mass fraction. Lines represent the data fit with eqn (6) and (7). | ||
The extrapolated heat capacity data were then fitted with a linear equation
| Cpm = a + b·T | (6) |
| Cpm = a + b·T + c·T2 + d·T3 | (7) |
Although the cubic equation does not seem to describe the heat capacity of Chol-C11 lactate accurately, higher-order polynomials did not provide an overall better fit. A polynomial of the sixth order was found to provide a seemingly more accurate description of the data using a regression along the gnostic influence function, however, extrapolation to higher and lower temperatures outside the experimentally studied range does not appear to be thermodynamically sound. Indeed, as stated by Zábranský et al.,12 third-order polynomials are usually sufficient to describe heat capacity as a function of temperature and it is preferable to sacrifice some of the fit accuracy for the sake of a meaningful extrapolation. The equation parameters were again optimized using the robust regression along a gnostic influence function, providing us with a reliable fit.
Apparent molar heat capacity listed in Tables SI-13 to SI-18 and shown in Fig. 5 and 6 was then derived from the experimental data for the ionic liquids and water as
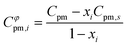 | (8) |
It should be noted that heat capacities do not reflect interaction energies between the solute and solvent molecules in themselves, but rather the temperature derivatives of these quantities. It appears from the present data that the derived Cφpm,1 show large positive values close to the corresponding molar heat capacity of the pure CHIL and a relatively constant function over a large range of the water content for Chol Lac and Chol Lev (Fig. 5a and c, respectively), Chol Lev being a slowly decreasing function of water content. This corroborates our finding that both choline ionic liquids are hydrophilic,22 indicating strong ion–water interactions that suppress fluctuations in the hydration shell. This lowers the system's ability to absorb heat at constant pressure and is characteristic of “constraining” hydration, where solvent molecules lose configurational freedom. The increasingly tighter hydration (especially at low water mole fractions) thus reduces the incremental heat capacity.
To estimate the derivative Cφpm,1 with respect to temperature, the apparent molar heat capacities were then fitted with a linear equation
| Cφpm,1 = i + s·T | (9) |
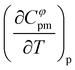 and are summarized in Table SI-19 of. It appears that Cφpm,1 is an increasing function of temperature for all the four CHILs studied in the present work. Matakhadze et al.35 discuss the apparent molar heat capacity derivatives with respect to temperature in terms of solute hydrophobicity or hydrophilicity, linking positive
and are summarized in Table SI-19 of. It appears that Cφpm,1 is an increasing function of temperature for all the four CHILs studied in the present work. Matakhadze et al.35 discuss the apparent molar heat capacity derivatives with respect to temperature in terms of solute hydrophobicity or hydrophilicity, linking positive 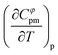 to hydrophilic solutes. Cφpm,1 of choline levulinate then appears to be a more quickly increasing function of temperature than that of choline lactate, which is in agreement with its slightly amphiphilic character.
to hydrophilic solutes. Cφpm,1 of choline levulinate then appears to be a more quickly increasing function of temperature than that of choline lactate, which is in agreement with its slightly amphiphilic character.
The apparent heat capacity of water, Cφpm,2, in choline lactate is then a slowly decreasing function of water content (Fig. 5b), whereas for choline levulinate, Cφpm,2 appears to take on an almost constant value (Fig. 5d). In both cases the values are close to the molar heat capacity of pure water. This reflects the behaviour observed in apparent molar volumes, confirming the hydrophilic nature of both anions, with levulinate showing a more amphiphilic character.
The situation appears to be more complex in Chol-C11 CHILs (Fig. 6). Similarly to choline levulinate and choline lactate, Cφpm,1 appears to show large positive values that are higher than the corresponding “dry” ionic liquid molar heat capacity, a weak dependence on the water content, and a positive derivative of the apparent molar heat capacity with respect to temperature (Fig. 6a and c, respectively, and Table SI-19). At the same time, the apparent molar heat capacity of water, Cφpm,2, in both Chol-C11 CHILs appears to show a very weak concentration dependence within the measured concentration range (Fig. 6b and d). This points to a more rigid arrangement on the molecular level. The bulky Chol-C11 cation shows an amphiphilic character, with a localized headgroup that strongly interacts with water, through electrostatic forces and hydrogen bonding via ether/ester groups. The hydrophobic long alkyl chain is hydrophobic then excludes water, forcing structuring around it through low configurational entropy. Water molecules near the charged head are tightly bound, while around the alkyl chain, water is structured into “clathrate-like” cages, which reduces the number of accessible microstates.36
Considering the influence of the anion on the solution molecular structure it appears that the combination of the slightly amphiphilic levulinate with the amphiphilic cation is reflected in the stronger concentration dependence of Cφpm,1 than for in the Chol-C11 Lac, indicating a more homogeneous solution structure. It should be noted that because of the large difference between the heat capacity of the pure CHILS and water, the uncertainty of the apparent molar heat capacity shows a strong dependence on the water content, ranging between 5 and 9 J K−1 mol−1 for Chol Lac and Chol Lev, respectively, and 5 and 17 J K−1 mol−1, respectively, for Chol-C11 Lac and Chol-C11 Lev. The sensitivity to the uncertainty in water content determination is then similarly pronounced as in apparent molar volumes discussed above. This means a discussion of the influence of the water content on the hydration properties of the studied mixtures using heat capacity data from this work may be considered as purely tentative.
5 Conclusions
An analysis of the volumetric and thermal properties of four choline-based ionic liquids that differ in structure and level of hydration is provided in the present work. Estimates of the density and molar heat capacity of the studied CHILs were obtained using advanced data analysis by extrapolation from hydrated samples. The data analysis applied in this work shows the importance of a careful determination of the uncertainty budget in determining of derived thermophysical properties, such as apparent molar properties. This is especially important for hygroscopic and viscous compounds such as the CHILs studied in this work, that present additional difficulties on sample preparation. The importance of a judicious choice of the extrapolation methods, extrapolating functions, and concentration units is also discussed. Indeed, the curvature of the function from which the extrapolated values should be obtained must be taken into account to obtain reliable and physically meaningful extrapolated values. The obtained results show and quantify the significant impact of water on the thermophysical behaviour of CHILs. The apparent molar volumes derived from the measured densities of Chol Lev and Chol Lac CHILs show a pronounced hygroscopicity, pointing towards an amphiphilic behaviour of the levulinate CHIL. The apparent molar heat capacities then bring and additional possible interpretation of the hydration of the studied CHILs, although it must be noted that the uncertainty of the measurements allow only for a tentative discussion of the present systems. The present results then open further avenues for the application of the studied CHILs in environmentally friendly processes, especially in biomass treatment. The insights gained here enhance our fundamental understanding of CHIL–water interactions deserve to be explored in future works, especially as to the transport properties of the present CHILs. A broader application of CHILs in sustainable chemical technologies may then be possibly developed. The present study will therefore be further developed in a subsequent article in which the relationship between the CHIL structure and hydration will be discussed in the light of acoustic and transport properties with the support of molecular simulations.Author contributions
Magdalena Bendová (conceptualisation, project funding, supervision, data analysis, draft writing and editing), Zdeněk Wagner (data analysis), Sandrine Bouquillon (conceptualisation, supervision, project funding, draft writing and editing), Mahasoa-Salina Souvenir-Zafindrajaona (synthesis of the compounds, data acquisition), Jean-Pierre Mbakidi (supervision, synthesis).Conflicts of interest
There are no conflicts to declare.Data availability
Due to the large amount of raw data it is impossible to include all the experimental data in the manuscript. These data will be made available on demand. The robust non-statistical data analysis by means of mathematical gnostics was carried out by in-house Octave codes that may also be provided on demand, as well as the entirety of the extrapolation results.Supplementary information: Tables SI1–SI19 as an Excel file and details of synthesis of the studied ionic liquids. See DOI: https://doi.org/10.1039/d5ra07884a.
Acknowledgements
This work was supported by the URCA and the Barrande Fellowship of the French Government (PhD of M. S. S. Z.), and the FEDER for material funds. M. B. gratefully acknowledges funding from the Czech Ministry of Education, Youth and Sports Mobility project no. 8J21FR0027.References
- K. K. Chan, K. J. Yong, Y. K. Ho, T. Y. Wu and C. W. Ooi, Int. J. Biol. Macromol., 2025, 307, 141697 CrossRef CAS.
- A. R. Shaikh, A. Grillo, M. C. D'Alterio, J. J. Pajski, S. I. Amran, H. Karim, M. Chawla, G. Talarico, A. Poater and L. Cavallo, J. Mol. Liq., 2025, 424, 127084 CrossRef CAS.
- B. L. Gadilohar and G. S. Shankarling, J. Mol. Liq., 2017, 227, 234–261 CrossRef CAS.
- Z. Dong, L. Zhang, G. Li, Y. Li, H. He, Y. Lu, W. Wu and J. Qi, J. Controlled Release, 2024, 375, 812–828 Search PubMed.
- X. Li, N. Ma, L. Zhang, G. Ling and P. Zhang, Int. J. Pharm., 2022, 612, 121366 Search PubMed.
- B. Krupa, M. Nowicki, P. Huninik, J. Szyling and J. Walkowiak, ChemRxiv, 2024 DOI:10.26434/chemrxiv-2024-z8lqq.
- A. Hernández-Sánchez, J. J. Alcaraz-Espinoza, C. S. Thomas, E. Jiménez-Regalado, A. Mayrén, I. González and G. R. Sánchez, J. Energy Storage, 2025, 106, 114746 CrossRef.
- M.-S. Souvenir-Zafindrajaona, S. Nadir, J.-P. Mbakidi, V. Jandová, M.-A. Ndong, V. Lequart, P. Martin, M. Bendova and S. Bouquillon, J. Mol. Liq., 2025, 429, 127642 CrossRef CAS.
- E. E. L. Tanner, K. M. Piston, H. Ma, K. N. Ibsen, S. Nangia and S. Mitragotri, ACS Biomater. Sci. Eng., 2019, 5, 3645–3653 CrossRef CAS.
- S. E. Kruchinin and M. V. Fedotova, J. Mol. Liq., 2025, 428, 127532 CrossRef CAS.
- A. Andresová, M. Bendová, J. Schwarz, Z. Wagner and J. Feder-Kubis, J. Mol. Liq., 2017, 242, 336–348 CrossRef.
- M. Zábranský, Z. Kolská, V. Růžička and E. S. Domalski, J. Phys. Chem. Ref. Data, 2010, 39(1), 013103 CrossRef.
- F. Spieweck and H. Bettin, Tech. Mess., 1992, 59, 285–292 CAS.
- Z. Wagner, M. Bendová, J. Rotrekl, A. Sýkorová, M. Čanji and N. Parmar, J. Mol. Liq., 2021, 329, 115547 CrossRef CAS.
- N. Parmar, M. Bendová and Z. Wagner, J. Therm. Anal. Calorim., 2024, 150, 313–325 CrossRef.
- P. Kovanic, Mathematical Gnostics: Advanced Data Analysis for Research and Engineering Practice, CRC Press, 2023 Search PubMed.
- P. Kovanic and M. B. Humber, The Economics of Information (Mathematical Gnostics for Data Analysis), 2015, https://www.math-gnostics.eu/books/ Search PubMed.
- Z. Wagner, M. Bendová, J. Rotrekl, A. Sýkorová, M. Čanji and N. Parmar, J. Mol. Liq., 2021, 329, 115547 CrossRef CAS.
- R. D. Chirico, M. Frenkel, J. W. Magee, V. Diky, C. D. Muzny, A. F. Kazakov, K. Kroenlein, I. Abdulagatov, G. R. Hardin, W. E. Acree, J. F. Brenneke, P. L. Brown, P. T. Cummings, T. W. de Loos, D. G. Friend, A. R. H. Goodwin, L. D. Hansen, W. M. Haynes, N. Koga, A. Mandelis, K. N. Marsh, P. M. Mathias, C. McCabe, J. P. O'Connell, A. Pádua, V. Rives, C. Schick, J. P. M. Trusler, S. Vyazovkin, R. D. Weir and J. Wu, J. Chem. Eng. Data, 2013, 58, 2699–2716 CrossRef CAS.
- Joint Committee for Guides in Metrology (JCGM), International Vocabulary of Metrology – Basic and general concepts and associated terms (VIM), 3rd edn, 2008, version with minor corrections, JCGM 200:2012, Bureau International des Poids et Mesures (BIPM), Sévres, 2012https://www.bipm.org/documents/20126/41373499/JCGM_200_2012.pdf/f0e1ad45-d337-bbeb-53a6-15fe649d0ff1, (accessed January 15 2026) Search PubMed.
- Y. Marcus, Biophys. Chem., 2006, 124, 200–207 CrossRef CAS PubMed.
- C. De Visser, W. J. M. Heuvelsland, L. A. Dunn and G. Somsen, J. Chem. Soc., Faraday Trans. 1: Phys. Chem. Cond. Phases, 1978, 74, 1159 RSC.
- C. Tondre and R. Zana, J. Phys. Chem., 1972, 76, 3451–3459 CrossRef CAS.
- M. Lukšič and B. Hribar-Lee, J. Mol. Liq., 2017, 228, 126–132 CrossRef.
- A. Y. Payumo, R. M. Huijon, D. D. Mansfield, L. M. Belk, A. K. Bui, A. E. Knight and D. K. Eggers, J. Phys. Chem. B, 2011, 115, 14784–14788 CrossRef CAS PubMed.
- Y. Marcus, Chem. Rev., 2009, 109, 1346–1370 CrossRef CAS PubMed.
- O. Redlich and D. M. Meyer, Chem. Rev., 1964, 64, 221–227 CrossRef CAS.
- O. Redlich, J. Phys. Chem., 1940, 44, 619–629 CrossRef CAS.
- O. Redlich and P. Rosenfeld, Z. Elektrochem., 1931, 37, 705–711 CrossRef CAS.
- F. J. Millero, Chem. Rev., 1971, 71, 147–176 CrossRef CAS.
- W. C. Vosburgh, L. C. Connell and J. A. V. Butler, J. Chem. Soc., 1933, 933–942 RSC.
- D. Warmińska and M. Śmiechowski, J. Mol. Liq., 2022, 354, 118876 CrossRef.
- J. Desnoyers and M. Arel, Can. J. Chem., 1967, 45, 359–366 CrossRef.
- J. G. Reynolds, J. Mol. Liq., 2024, 395, 123824 CrossRef CAS.
- G. Makhatadze, S. Gill and P. Privalov, Biophys. Chem., 1990, 38, 33–37 Search PubMed.
- J. E. Desnoyers, Pure Appl. Chem., 1982, 54, 1469–1478 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |