DOI:
10.1039/D5RA07419C
(Paper)
RSC Adv., 2026,
16, 10720-10734
Synthesis, structural refinement and luminescence properties of green-emitting Gd3GaO6:Tb3+ phosphors for display applications
Received
30th September 2025
, Accepted 9th February 2026
First published on 23rd February 2026
Abstract
Green-emitting Tb3+-doped Gd3GaO6 phosphors were fabricated through a low-temperature solution-combustion approach utilizing urea as the fuel. Powder XRD analysis suggested an orthorhombic structure with the Cmc21 space group. Crystallite size was determined using the Scherrer's formula and W–H plot. Surface topography was examined utilizing the FESEM technique, and EDX data confirmed the uniform incorporation of Tb3+ ions into the Gd3GaO6 lattice with proper stoichiometry. A bright green emission was exhibited by phosphors at 543 nm, attributed to the 5D4 → 7F5 transition upon irradiation with near ultraviolet light at 272 nm. The observed concentration quenching at 3 mol% Tb3+ in the Gd3GaO6 matrix was attributed to non-radiative energy transfer between the neighbouring Tb3+ ions, which became significant at higher doping levels. The quenching observed beyond the optimum concentration is primarily attributed to dipole–dipole interactions between adjacent Tb3+ ions. The CIE chromaticity coordinates are located the green region, and the calculated CCT value (4500–5500 K) lies in the cool temperature range, making this phosphor a suitable candidate for cool white-solid state lighting and display devices.
1. Introduction
In the past decade, inorganic phosphors with tunable luminescent colors have attracted considerable attention owing to their wide applications in bioimaging, color display panels, light-emitting diodes (LEDs), optical thermometry and white light-emitting diodes (w-LEDs).1–3 Recently, conventional fluorescent and incandescent lamps have been largely replaced by w-LEDs due to their eco-friendly nature, cost-effective production, compactness, high luminescence efficiency, long lifetime, excellent chemical and thermal stability and energy savings of approximately 70% compared with traditional sources.4,5 For the synthesis of phosphors with desired applications, the suitable choice of a host lattice is very important. Gallates have emerged as promising host lattices for rare-earth ion doping because of their eco-friendly composition, structural stability, excellent optical properties and high resistance to oxygen, acids and bases.6–9 Rare-earth-doped gallate matrices have thus found applications in waveguides, optical fibers, video displays, smart windows and control panels.10–13
In particular, Gd3GaO6 stands out as an attractive host for phosphor synthesis. It possesses a relatively narrow band gap (Eg ≈ 3.60 eV) compared with other rare-earth gallates, such as RE3GaO6 (RE = Nd, Sm, and Eu), enabling efficient absorption and energy transfer. The orthorhombic structure (space group Cmc21) provides suitable cation sites for rare-earth substitution, and the material exhibits excellent thermal stability with no structural phase transitions over a wide temperature range (298–1273 K) along with favorable thermophysical properties.14 A key advantage is the presence of Gd3+ ions, which can serve as effective sensitizers by transferring energy from their excited 6P7/2 state to dopant ions, enhancing luminescence efficiency and overcoming limitations associated with weak direct absorption in the far-UV region.15,16
Trivalent lanthanide ions are preferred activators due to their sharp emission bands in the visible region arising from the 4f–4f transitions. By varying the dopant type and concentration, multicolor luminescence can be achieved.17–19 Among these, the Tb3+ ion is a well-established green emitter, primarily due to the magnetic dipole-allowed 5D4 → 7F5 transition at ∼543 nm, which offers bright green luminescence ideal for w-LEDs, display backlights and optoelectronic devices. Tb3+ ions may also show blue emission from the 5D4 → 7F6 transition at ∼490 nm, but the relative intensity of this component depends on the host crystal field, Tb3+ ion concentration and phonon energy, which influence cross-relaxation and non-radiative processes.20,21 While Tb3+-doped phosphors are common in hosts like garnets, silicates or phosphates, many suffer from issues such as thermal quenching, concentration quenching at high doping levels or insufficient color purity due to residual blue emission. Although Gd3GaO6 has been explored as a host for other activators, primarily Bi3+ (for persistent luminescence) and Er3+ (for upconversion or color-tunable emission), systematic investigations of Tb3+-activated Gd3GaO6 remain limited or absent in the literature.22 Tb3+ doping in Gd3GaO6 is interesting due to sensitization by the host Gd3+ ion, moderate band gap, low phonon energy and orthorhombic low-symmetry sites could promote intense, high-purity green emission. Such a phosphor could offer advantages for w-LEDs and displays, including better device integration, reduced reabsorption and improved performance under operational conditions.
Traditional solid-state synthesis of inorganic phosphors often involves high-temperature treatments and repeated grinding, which can damage particle surfaces, introduces defects and reduces emission intensity.23 To address these limitations and produce uniform, high-efficiency nanophosphors, various wet-chemical methods have been developed including sol–gel, microwave-assisted, hydrothermal, precipitation and solution combustion synthesis.24–27 The solution combustion method is particularly advantageous for Tb3+-doped Gd3GaO6, as it yields homogeneous products with large surface area, high purity, nanoscale morphology and short reaction times.28 The present work, therefore, focuses on the synthesis of Tb3+-doped Gd3GaO6 nanophosphors via solution combustion. The structural and surface properties were characterized using XRD and FESEM, compositional analysis was performed by EDX and photoluminescence properties were investigated at varying dopant concentrations. These results aim to demonstrate the potential of Gd3GaO6:Tb3+ as an intense green-emitting phosphor for solid-state lighting and display applications.
2. Experimental
A series of Gd3−xGaO6:xTb3+ (x = 1–6 mol%) powdered phosphors was prepared by a facile solution combustion method. The raw materials used for the Gd, Ga and Tb sources were gadolinium nitrate [Gd(NO3)3·6H2O], gallium nitrate [Ga(NO3)3·xH2O] and terbium nitrate [Tb(NO3)3·6H2O], respectively. The precursors utilized in the solution combustion method were purchased from Sigma-Aldrich. The stoichiometric amount of metal nitrates and urea (organic fuel) was dissolved in a minimum amount of double-distilled water to obtain a homogeneous clear solution. This solution was further heated on a heating plate with uniform stirring and the temperature was maintained at 80 °C to form a gelatinous mixture. The entire solution was transferred to a crucible and placed in a muffle furnace set at 600 °C, where the resulting mixture underwent exothermic reaction, followed by decomposition and evolution of various gases. The crucible was drawn out of the furnace and the obtained voluminous foamy product was manually ground to a fine powder using an agate mortar and pestle. The samples were further calcined in an alumina crucible at 1450 °C for 4 h to improve crystallinity. Final products are brought to ambient temperature, crushed and used for characterizations. The preparation scheme of this method is illustrated in Fig. 1.
 |
| | Fig. 1 Representation of the solution combustion synthesis employed in the nanophosphor fabrication. | |
3. Characterization techniques
Powder X-ray diffraction profiles of the phosphor material were analyzed to determine phase purity by using a Rigaku Ultima-IV X-ray diffractometer equipped with a Cu-Kα source and nickel filter. Scanning was performed between 10°–70°, employing a 0.02° step size and scanning speed of 2° per minute. Microstructural and surface characteristics of the particles were examined by FESEM. The micrographs were taken on a Carl Zeiss Sigma 360 instrument. An Ametek EDX spectrometer was employed for confirming the elemental composition of the sample. Photoluminescence spectral data were recorded on a Horiba-Jobin Fluorolog spectrophotometer using an Xe lamp as an excitation source.
4. Results and discussion
4.1 XRD measurements
Phase purity and crystallinity of pure and Tb3+-doped gadolinium gallium oxide samples were analyzed by means of XRD patterns. Fig. 2(a) shows the diffraction peaks of undoped Gd3GaO6 and the Gd3−xGaO6:xTb3+ (x = 1–6 mol%) phosphors. XRD data confirmed that all peaks were indexed to the orthorhombic phase, consistent with standard JCPDS card number 53–1225, and no additional peaks were present, confirming the non-existence of an impurity phase.29 Fig. 2(b) shows an enlarged view of diffraction peaks, which revealed peak shifting toward higher 2θ values with increasing Tb3+ concentration.
 |
| | Fig. 2 (a) XRD profiles of Gd3GaO6 and Gd3−xGaO6:xTb3+ (x = 1–6 mol%) and their (b) magnified view, highlighting key features of all the samples. | |
These results were consistent with lattice compression on doping smaller Tb3+ ions. Radius difference percentage (Δr) was evaluated to confirm the replacement of the Gd3+ ion with the Tb3+ ion by eqn (1).30
| |
 | (1) |
Here,
RB(CN) = host cation radius, and
RD(CN) = dopant ion radius. The acceptable value of Δ
r for the successful inclusion of a particular cation should be lower than 15%, as reported in the literature.
31 In the present study, the value of Δ
r comes out to be 2% by substituting the radius values for Gd
3+ (
RB = 1 Å with CN = 7) and Tb
3+ (
RD = 0.98 Å with CN = 7) ions. This suggests the Tb
3+ ion successfully replaced the Gd
3+ ion without altering the crystal structure of the GGO host.
Furthermore, replacement of a bigger host cation by a smaller Tb3+ ion was supported by calculations of interplanar d-spacing values determined from the main XRD peak corresponding to various Tb3+ doping levels using Bragg's eqn (2).32
| |
nλ = 2d![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) sin sin![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θ θ
| (2) |
The computed d-spacing values are summarized in Table 1. Crystallite size (D) was estimated by employing the Debye Scherrer eqn (3) based on XRD broadening.33
| |
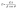 | (3) |
In this formula,
λ represents the wavelength of Cu-K
α radiations (1.54 Å),
β refers to peak broadening (FWHM) and
θ denotes Bragg's angle.
Table 1 XRD peak positions (2θ values) and corresponding interplanar spacings (d-values) of the Gd3−xGaO6:xTb3+ (x = 1–6 mol%) phosphors
| Sample |
2θ |
d-Spacing (Å) |
| GGO:0 mol% Tb3+ |
30.2062 |
2.9564 |
| GGO:1 mol% Tb3+ |
30.2416 |
2.9530 |
| GGO:2 mol% Tb3+ |
30.2633 |
2.9509 |
| GGO:3 mol% Tb3+ |
30.2952 |
2.9479 |
| GGO:4 mol% Tb3+ |
30.3052 |
2.9469 |
| GGO:5 mol% Tb3+ |
30.3269 |
2.9449 |
| GGO:6 mol% Tb3+ |
30.3491 |
2.9441 |
Crystallite size values for the host and Gd3−xGaO6:xTb3+ (x = 1–6 mol%) samples were evaluated and are presented in Table 2. Additionally, the Williamson–Hall (W–H) approach was utilized for comprehensive estimation of crystallite size using relation (4).34
| |
 | (4) |
Table 2 Computed XRD parameters (crystallite size and dislocation density) of the synthesized Gd3−xGaO6:xTb3+ (x = 1–6 mol%) phosphors
| Sample (GGO) |
2θ |
FWHM |
Crystallite size (nm) |
Dislocation density |
| Scherrer's |
W–H |
| GGO:0 mol% Tb3+ |
30.2062 |
0.34724 |
24.75 |
41.25 |
1.63 × 10−3 |
| GGO:1 mol% Tb3+ |
30.2416 |
0.30321 |
28.35 |
43.73 |
1.24 × 10−3 |
| GGO:2 mol% Tb3+ |
30.2633 |
0.27125 |
31.69 |
46.14 |
9.96 × 10−4 |
| GGO:3 mol% Tb3+ |
30.2952 |
0.37221 |
23.09 |
38.36 |
1.88 × 10−3 |
| GGO:4 mol% Tb3+ |
30.3052 |
0.40812 |
21.06 |
35.15 |
2.25 × 10−3 |
| GGO:5 mol% Tb3+ |
30.3269 |
0.32194 |
26.70 |
42.64 |
1.40 × 10−3 |
| GGO:6 mol% Tb3+ |
30.3487 |
0.29326 |
29.32 |
44.91 |
1.16 × 10−3 |
This expression can be further modified as eqn (5).35
| |
 | (5) |
In this context,
K = constant, whose value depends on the shape of particles,
ε = microstrain and all other variables have their common meanings as described earlier.
Fig. 3 illustrates a linear fitted W–H plot with 4 sin
θ plotted on the
x-axis and
β cos
θ along the
y-axis.
 |
| | Fig. 3 Williamson–Hall (W–H) plots for the Gd3GaO6 host and Gd3−xGaO6:xTb3+ (x = 1–6 mol%) phosphors. | |
Crystallite sizes for different doped samples and the host, calculated from the intercept of the linear fitted data, are given in Table 2. Slight variation in crystallite size calculated from Scherrer's formula and W–H plot arises from the assumption of Scherrer's formula, where the strain component was taken as zero and peak broadening is solely attributed to grain size. Dislocation density represented as δ was also calculated for various samples by employing eqn (6), and values are given in Table 2.36
| |
 | (6) |
4.2 Rietveld refinement and crystal structure
To further confirm the single-phase purity of the prepared phosphors, Rietveld refinement analysis was executed. The lattice constraints of representative samples were assessed via the FULLPROF software. Fig. 4(a) and (b) represent refinement plots of the undoped GGO host and the optimized Tb3+-doped GGO sample, respectively.
 |
| | Fig. 4 Rietveld-refined XRD profiles of the (a) Gd3GaO6 and (b) Gd2.97GaO6:3 mol% Tb3+ samples. | |
The observed and calculated data are shown as black crosses and red/green solid lines, respectively. Vertical (brown) lines represent the Bragg's position and the difference between observed and calculated data is indicated at the bottom by a blue line in each figure. Refinement data for Gd3GaO6 and Gd3−xGaO6:xTb3+ (x = 3 mol%) are arranged in Table 3. The reliable factors for the GGO host are Rp = 4.192%, Rwp = 5.223% and χ2 = 2.104 and for Gd2.97GaO6:3 mol% Tb3+ sample are Rp = 4.482%, Rwp = 5.613% and χ2 = 2.248. As the reliability factors lie within the 10% range, signifying strong agreement between theoretical and experimental patterns.37 The measured cell parameters for the optimized sample (a = 8.8742 Å, b = 11.3259 Å, c = 5.4636 Å, α = β = γ = 90° and V = 549.1371 Å3) are slightly below the standard host values.
Table 3 Rietveld refinement constraints of Gd3GaO6 (GGO) and the Gd2.97GaO6:3 mol% Tb3+ phosphors
| Refinement parameters |
Refinement values of Gd3GaO6 |
Refinement values of Gd2.97GaO6:3 mol% Tb3+ |
| Formula weight |
637.47 |
637.07 |
| Crystal system |
Orthorhombic |
Orthorhombic |
| 2θ interval |
10–70° |
10–70° |
| Bravais lattice |
C |
C |
| No. of space group |
36 |
36 |
| Hermann-Mauguin symbol |
Cmc21 |
Cmc21 |
| Hall symbol |
C2c-2 |
C2c-2 |
| Laue class |
mmm |
mmm |
| Point group |
mm2 |
mm2 |
| Lattice symbol |
oC |
oC |
| Formula unit (Z) |
4 |
4 |
| Unit cell parameters |
a = 8.9278 Å, b = 11.3859 Å, c = 5.5060 Å |
a = 8.8742 Å, b = 11.3259 Å, c = 5.4636 Å |
| α = β = γ = 90° |
α = β = γ = 90° |
| Volume (Å3) |
559.6906 |
549.1371 |
| R-Factors |
Rp = 4.192%, Rwp = 5.223% |
Rp = 4.482%, Rwp = 5.613% |
| Density (g cm−3) |
7.565 |
7.714 |
| χ2 |
2.104 |
2.248 |
This contraction in lattice constraints for Tb3+-doped samples is consistent with the substitution of larger Gd3+ ions by smaller Tb3+ ions. The crystal structure of the GGO lattice designed by Vesta software is depicted in Fig. 5. As represented in the structure, there are three different cationic sites: two corresponding to gadolinium [Gd(3) and Gd(4)] and one for gallium [Ga(6)]. [Gd(3), Gd(4) and Ga(6)] ions are located at the 4a, 8b and 4a Wyckoff positions, respectively. Gadolinium ions form the [Gd(3)/(4)O7] polyhedra, in which the Gd(3)/(4) ions are surrounded by seven oxygen atoms, whereas gallium ions form a tetrahedron coordinated to four oxygen atoms. Furthermore, there are four different types of oxygen atoms, i.e., O(1), O(2), O(5) and O(7), located at the 4a, 4b, 4a and 4b crystallographic positions, respectively. Table 4 shows the atomic coordinates (x, y, z) and dislocation factors of the Gd3GaO6 host.
 |
| | Fig. 5 Visualization of the crystal structure of Gd3GaO6, illustrating the coordinative environment of individual cations. | |
Table 4 Crystallographic parameters of the Gd3GaO6 (GGO) host, exhibiting an orthorhombic crystal system
| Atoms |
x |
y |
z |
Occ. |
Site |
U |
Sym. |
| Gd (3) |
0.00000 |
0.39640 |
0.42369 |
1.000 |
4a |
0.205 |
m |
| Gd (4) |
0.19300 |
0.10567 |
0.46705 |
1.000 |
8b |
0.205 |
1 |
| O (1) |
0.00000 |
0.04660 |
0.12500 |
1.000 |
4a |
0.205 |
m |
| O (2) |
0.00000 |
0.21400 |
0.67100 |
1.000 |
4a |
0.205 |
m |
| O (5) |
0.34000 |
0.01890 |
0.20600 |
1.000 |
8b |
0.205 |
1 |
| O (7) |
0.15590 |
0.26010 |
0.17000 |
1.000 |
8b |
0.205 |
1 |
| Ga (6) |
0.00000 |
0.26010 |
0.17000 |
1.000 |
4a |
0.205 |
m |
4.3 Morphological insights
To study the structural and surface characteristics of the synthesized phosphor material, FESEM is an essential tool due to its ability to provide structural information at various magnifications. Fig. 6(a) exhibits the FESEM image of the optimized Gd3−xGaO6:xTb3+ (x = 3 mol%) sample. The phosphor synthesized via the combustion approach demonstrated a porous structure and slight agglomeration, reflecting typical features associated with the exothermic nature of combustion synthesis.
 |
| | Fig. 6 (a) FESEM image of the Gd3−xGaO6:xTb3+ (x = 3 mol%) phosphor, showing its surface morphology. (b) Particle size distribution of the Gd3−xGaO6:xTb3+ (x = 3 mol%) sample. | |
Due to calcination of the sample at a higher temperature, some agglomeration was observed, and the obtained particles are slightly irregular in shape with an average particle size in the 20–50 nm range. Fig. 6(b) depicts the particle size distribution of the optimized sample.
4.4 EDX spectral analysis
EDX spectroscopy and elemental mapping of the as-prepared phosphor were conducted in order to ascertain the elemental composition.
The EDX spectra in Fig. 7(a) provide information about the presence of all expected elements: terbium (Tb), gadolinium (Gd), gallium (Ga) and oxygen (O). The elements are evenly distributed, and no indication of the presence of an extra element was observed in the synthesized sample, suggesting the successful preparation of the Tb3+-doped GGO phosphor. The inset of Fig. 7(a), along with the experimental data, illustrates each constituent element together with its atomic and weight percentages. The homogenous distribution of constituent elements was confirmed by X-ray mapping images of the Gd3−xGaO6:xTb3+ (x = 3 mol%) phosphor (Fig. 7(b)).
 |
| | Fig. 7 (a) EDX spectrum of the optimized Gd2.97GaO6:3 mol% Tb3+ nanophosphor with an inset table summarizing elemental compositions by atomic % and weight %. (b) EDX mapping images for the Gd2.97GaO6:3 mol% Tb3+ nanophosphor, displaying the spatial distribution of the Gd, Ga, Tb and O elements. | |
4.5 Study of photoluminescence behaviour
4.5.1 Analysis of excitation and emission spectra. Fig. 8 depicts the photoluminescence excitation spectra of the Gd3−xGaO6:xTb3+ (x = 3 mol%) sample assessed at an emission wavelength of 545 nm.
 |
| | Fig. 8 Excitation spectrum of the Gd2.97GaO6:3 mol% Tb3+ phosphor recorded at λem = 545 nm. | |
The trivalent gadolinium ion exhibits intra-configuration f–f transition 8S7/2 → 6I11/2 around 272 nm, which overlaps with the 4f8 → 4f74d1 transition of Tb3+ ions and is not evidently distinguishable in the spectra.38,39 There are several other peaks observed in the spectra due to the 4fn–4fn transition of the Tb3+ ion, which are assigned to the electronic transitions (7F6 → 5H6) at 306 nm, (8S7/2 → 6P7/2) at 312 nm, (7F6 → 5L6) at 343 nm, (7F6 → 5L9) at 357 nm, (7F6 → 5G6) at 380 nm and (7F6 → 5D4) at 495 nm.40–42 The emission spectra of varying concentrations of Tb3+-doped Gd3GaO6 phosphors measured at the 272 nm excitation wavelength lie in the range of 450–650 nm and is demonstrated in Fig. 9.
 |
| | Fig. 9 Emission spectra of the Gd3−xGaO6:xTb3+ (x = 1–6 mol%) phosphors. | |
The photoluminescence emission profile is mainly composed of four emission lines due to 5D4 → 7F3, 5D4 → 7F4, 5D4 → 7F5 and 5D4 → 7F6 transitions positioned at 626 nm, 590 nm, 543 nm and 490 nm, respectively.43 Among all the observed transitions, the green emission, originating from the 5D4 → 7F5 transition, exhibits the strongest intensity. The transition is distinguished as a magnetic dipole and follows the selection rule ΔJ = ±1.44
The energy level diagram represented in Fig. 10 explains the mechanism of the possibility of energy transfer from Gd3+ to the Tb3+ ion. Upon UV excitation of Gd3+, in the 6I11/2 level from the 8S7/2 state, the system relaxes to lower-energy levels through non-radiative transitions, eventually populating the 6P7/2 energy state. Energy from the 6P7/2 excited state of Gd3+ may be partially transferred to the 5H6 energy state of Tb3+ via phonon-assisted dipole–dipole interactions, which then rapidly dissipates to its 5D4 levels, thereby enhancing the luminescence of Tb3+. This state subsequently undergoes radiative transition, generating four emission peaks located at 626 nm, 590 nm, 543 nm and 490 nm, corresponding to red, orange-red, green and blue light.
 |
| | Fig. 10 Depiction of electronic transitions in Tb3+ and energy transfer from Gd3+ ions under UV excitation. | |
4.5.2 Concentration quenching. The emission intensity of phosphor materials is strongly influenced by the concentration of doped ions. Fig. 11 shows the luminescence intensity of the Gd3GaO6 phosphor at different dopant concentrations of the Tb3+ ion. The intensity of peaks in the emission spectrum first increases with an increase in Tb3+ ion concentration, reaches a maximum value at 3 mol% concentration and then decreases on further increasing the concentration of the dopant ion. This decrease is observed owing to the effect of concentration quenching. Thus, according to the above information, 3 mol% was considered the optimized Tb3+ ion doping concentration for the Gd3GaO6 lattice. The concentration quenching phenomenon is generally observed when the activator ion concentration is high, which causes reduced spacing between activator ions, leading to increased non-radiative transitions amongst luminescent centres and results in decreased luminescence intensity.45 The mechanisms accountable for concentration quenching by non-radiative energy transfer can be radiation absorption, exchange interaction or electrical multipolar interaction.46
 |
| | Fig. 11 Emission intensity dependence on Tb3+ concentration in the Gd3−xGaO6:xTb3+ (x = 1–6 mol%) phosphors. | |
Due to a lack of significant overlap between the excitation and emission spectra of the Gd3GaO6:Tb3+ phosphor, concentration quenching does not occur via the radiation absorption mechanism. Critical distance (Rc) between Tb3+ ions in the host lattice was used to better understand the exchange interactions. It was calculated by Blasse and Grabmair utilizing the formula given in eqn (7).47,48
| |
 | (7) |
Here,
V,
Z and
xc refer to unit cell volume, number of cations per unit cell and critical concentration of Tb
3+ ions, respectively. Substituting the values of
Z = 4,
xc = 0.03 and
V = 549.1371 Å
3 for the Gd
3GaO
6 system in
eqn (7), the estimated
Rc appears to be 20.596 Å. According to Van Uitert, exchange interactions are dominant between dopant ions only when the critical distance is close to 5 Å.
49 As the calculated value in the present scenario is beyond the critical limit of 5 Å, interaction
via the exchange mechanism is less likely to occur. Therefore, multipole–multipole electrostatic interaction is responsible for non-radiative energy transfer. According to Dexter and Schulman's energy transfer theories, concentration quenching of activators is attributed to energy transfer from one dopant ion (donor) to another ion, finally reaching the energy sink within the host lattice. To understand the concentration quenching mechanism of Gd
3GaO
6:Tb
3+ in detail, Dexter's
eqn (8) was utilized.
50| |
 | (8) |
In this context,
x denotes the dopant ion concentration,
I represent the intensity of emission at different Tb
3+ concentrations,
k and
β are constants and
θ indicates the nature of multipolar interactions. The values of
θ equal to 6, 8 and 10 correspond to dipole–dipole, dipole-quadrupole and quadrupole–quadrupole interactions, respectively. When
β(
x)
θ/3 ≫ 1,
eqn (8) can be transformed to
eqn (9) as follows.
51,52| |
 | (9) |
Here,
k′ is a constant. A graph of log(
I/
x)
versus log(
x) was plotted and is represented in
Fig. 12.
 |
| | Fig. 12 Graph of the synthesized materials with linear fitting applied after concentration quenching. | |
The relation was found to be almost linear with a slope of −2.17. The computed θ value is 6.51, which is approximately equal to 6. As a consequence, concentration quenching of the Gd3GaO6:Tb3+ phosphor occurs via the electric dipole–dipole interaction mechanism.
4.6 Luminescence lifetime
The decay curve of the GGO:Tb3+ phosphor at 3 mol% concentration is shown in Fig. 13(a). Measurements were performed using excitation and emission wavelengths of 272 nm and 543 nm, respectively, to obtain the luminescence lifetime.
 |
| | Fig. 13 (a) Decay lifetime curve of the Gd2.97GaO6:3 mol% Tb3+ phosphor. (b) Auzel fitting curve of all the prepared samples. | |
The decay curve was fitted with various exponential functions, and the best fit was provided by the bi-exponential eqn (10).53
| |
 | (10) |
In this equation,
τ1 and
τ2 correspond to the fast and slow components,
A1 and
A2 serve as fitting parameters and
It and
I0 refer to the luminous intensity at time
t and
t = 0, respectively. By substituting the values of
A1,
A2,
τ1 and
τ2 in
eqn (11), the average lifetime can be calculated.
54| |
 | (11) |
The fitted decay analysis reveals the average lifetimes for G3−xGaO6:xTb3+ (x = 1–6 mol%) phosphors, which are tabulated in Table 5. The rapid quenching of the average lifetime with increasing Tb3+ concentration was attributed to enhanced non-radiative energy transfer processes. Additionally, Auzel's model (Fig. 13(b)) was applied to evaluate radiative lifetimes and interpret luminescence lifetime variations.55
| |
 | (12) |
Here,
τc is defined as the lifetime at concentration
C,
τ0 is the intrinsic radiative lifetime,
C0 is a constant and
N represents the number of phonons. The intrinsic lifetime was calculated to be 4.86 ms. Furthermore, the quantum efficiencies (
η) of the activated phosphors were calculated using the ratio of the average lifetime to the radiative lifetime (
eqn (13)).
56
Table 5 Computed decay lifetimes and quantum efficiencies of the Gd3−xGaO6:xTb3+ (x = 1–6 mol%) phosphors
| Sample |
τavg (ms) |
η (%) |
| GGO:1 mol% Tb3+ |
1.657 |
83.86 |
| GGO:2 mol% Tb3+ |
1.457 |
73.73 |
| GGO:3 mol% Tb3+ |
1.396 |
70.65 |
| GGO:4 mol% Tb3+ |
1.170 |
59.21 |
| GGO:5 mol% Tb3+ |
1.055 |
53.39 |
| GGO:6 mol% Tb3+ |
0.909 |
46.00 |
The quantum efficiency (η) values obtained for the synthesized nanophosphors are tabulated in Table 5, and a gradual decrease in the values of quantum efficiencies with increasing Tb3+ content was observed owing to non-radiative energy transfer.
4.7 Temperature dependent photoluminescence
Thermal stability plays a crucial role in determining the applicability of nanophosphors. Accordingly, the temperature dependent emission spectra of Gd2.97GaO6:3 mol% Tb3+ was measured in the temperature range of 298–473 K at intervals of 25 K, as illustrated in Fig. 14(a). The data indicate a steady decrease in luminescence intensity with increasing temperature, which can be attributed to thermal quenching, a phenomenon in which the phosphor's emission intensity diminishes at an elevated temperature range. As illustrated in Fig. 14(b), the emission intensity retains 74.29% of its original value at 473 K relative to the initial temperature, confirming the good thermal stability of the developed phosphor.
 |
| | Fig. 14 (a) Temperature-dependent photoluminescence of the Gd3GaO6:3 mol% Tb3+ phosphor. (b) Variations in the relative emission intensity with temperature for the Gd2.97GaO6:3 mol% Tb3+ phosphor. | |
To further investigate the thermal quenching behavior of the synthesized material, the activation energy of the optimized GGO:3 mol% phosphor was obtained using the approach given in eqn (14).57,58
| |
 | (14) |
In this expression, A is a constant, KB represents Boltzmann's constant (8.617105 × 10−5 eV K−1) and IT and I0 correspond to the emission intensities at measured and room temperatures, respectively. Eqn (14) can be modified to eqn (15) as follows.59,60
| |
 | (15) |
The plot of 1/KBT against ln(I0/IT), as shown in Fig. 15, exhibits a linear fit with slope −0.1767 and activation energy of 0.1767 eV, which is consistent with moderate quenching behavior. Table 6 entails a comparison of the Gd3GaO6:Tb3+ phosphor with other well-known Tb3+-doped and Ce3+-co-doped green phosphors in terms of lifetime, quantum efficiency and thermal stability.61–66
 |
| | Fig. 15 Linear fit plot for the calculation of activation energy. | |
Table 6 Comparative optical performance of the synthesized GGO:Tb3+ with Tb3+-activated phosphors
| Host lattice |
Emission peak |
Luminescence lifetime |
Quantum efficiency |
Thermal stability |
References |
| Gd3GaO6:Tb3+ |
543 nm |
1.396 ms |
70.6% |
74.6% at 473 K |
This work |
| YAl3(BO3)4:Tb3+ |
543 nm |
2.248 ms |
— |
70% at 473 K |
61 |
| Li2NaBP2O8:Tb3+ |
544 nm |
0.440 ms |
34.8% |
89% at 423 K |
62 |
| SrLaAlO4:Tb3+ |
548 nm |
1.399 ms |
48% |
61.2% at 398 K |
63 |
| BiOCl:Tb3+ |
543 nm |
0.012 ms |
— |
24.6% at 423 K |
64 |
| BaY4Si5O17:Ce3+, Tb3+ |
544 nm |
20.29 ns |
94.4% |
96.1% at 423 K |
65 |
| Na3Sc2(PO4)3:Ce3+, Tb3+ |
542 nm |
18.58 ns |
65% |
85.6% at 423 K |
66 |
4.8 Colorimetric investigations
To study the color purity and accurate emission color of the synthesized green GGO:Tb3+ phosphor, the XY chromaticity coordinate diagram (XY graph) based on CIE 1931 (Commission International de I'Eclairage) was plotted. CIE triangle comprises a triangular area composed of green, red and blue colors and the location of any color within the graph is referred to as its chromaticity point.67,68 The chromaticity coordinates were evaluated from the photoluminescence emission spectra for different samples of Tb3+-doped Gd3GaO6 phosphors. Fig. 16(a) and (b) represent the CIE color coordinate chart, and the calculated values are summarized in Table 7. It is clearly observed from Fig. 16 that all the coordinates of emitted color lie in the greenish region of the triangle, signifying the single color-emitting property of the GGO:Tb3+ phosphor. The obtained CIE chromaticity coordinates (x = 0.3419, y = 0.5962) for the optimized sample are consistent with the Tb3+-activated green emission dominated by the 5D4 → 7F5 transition (∼543 nm) and are comparable to reported Tb3+-doped oxides, such as CsAlSi2O6:Tb3+, Sr2Ga2SiO7:Tb3+ and MgAl2O4:Tb3+.69–71 CIE 1976 was additionally provided for the validation of luminous characteristics of the prepared samples. According to CIE 1976, the u′, v′ coordinates (Fig. 17(a) and (b)) are computed by following eqn (16).72| |
 | (16) |
 |
| | Fig. 16 (a) Representation of chromaticity coordinates (x, y) plotted on the CIE 1931 diagram. (b) Enlarged view, showing detailed spread of coordinates. | |
Table 7 CIE (x, y and u′, v′) coordinates, CCT (K) and color purity for the GGO:Tb3+ phosphors at different Tb3+ doping concentrations
| Sample |
CIE Co-ordinates |
Color purity (%) |
CCT (K) |
| x |
y |
u′ |
v′ |
| GGO:1 mol% Tb3+ |
0.3345 |
0.5882 |
0.1425 |
0.5638 |
64.24 |
5471.81 |
| GGO:2 mol% Tb3+ |
0.3373 |
0.5864 |
0.1441 |
0.5637 |
63.81 |
5424.37 |
| GGO:3 mol% Tb3+ |
0.3419 |
0.5962 |
0.1444 |
0.5666 |
66.51 |
5351.04 |
| GGO:4 mol% Tb3+ |
0.3389 |
0.5835 |
0.1454 |
0.5632 |
63.39 |
5395.69 |
| GGO:5 mol% Tb3+ |
0.3374 |
0.5826 |
0.1449 |
0.5628 |
63.11 |
4646.13 |
| GGO:6 mol% Tb3+ |
0.3432 |
0.5952 |
0.1452 |
0.5665 |
66.30 |
5329.68 |
 |
| | Fig. 17 (a) Representation of chromaticity coordinates (u′, v′) plotted on the CIE 1976 diagram. (b) Enlarged view, showing detailed spread of coordinates. | |
In order to characterize light sources accurately, the color purity of the emitted color was determined using eqn (17).73
| |
 | (17) |
In the above equation,
xs, ys represent the CIE coordinates of the sample,
xi,
yi correspond to illuminant points and
xd,
yd signify chromaticity coordinates of the dominant wavelength. The calculated values of color purity are listed in
Table 7, which shows that the color purity of the prepared GGO:3 mol% Tb
3+ sample is 66.51%. These findings demonstrate the potential use of the Tb
3+-doped GGO material as an excellent green-emitting phosphor for designing WLEDs and photonic devices. Another key parameter that determines the color quality, along with the temperature characteristics of the light source, is correlated color temperature (CCT). Warm light is generally characterized by CCT values less than 3000 K and values more than 4000 K are associated with cool light.
74
CCT values for various Tb3+-doped samples were calculated by employing the polynomial eqn (18) given by McCamy.75,76
| | |
CCT = −437n3 + 3601n2 − 6861n + 5514.31
| (18) |
| |
 | (19) |
Here,
n signifies the anti-slope line connecting (
x,
y) and (
xe,
ye), and
xe and
ye represent chromaticity epicenters. The CCT values evaluated for all the samples are given in
Table 7, which lie in the range of 4600 K–5500 K. The results confirm the cool green light nature of the prepared samples, suggesting their applications for outdoor illumination.
5. Conclusion
This study includes the synthesis of Gd3GaO6 phosphors doped with varying concentrations of trivalent terbium via an energy-efficient solution combustion approach. Successful inclusion of the Tb3+ ion within the Gd3GaO6 matrix was confirmed by X-ray diffractogram patterns. The GGO:Tb3+ phosphors form a crystalline orthorhombic structure belonging to Cmc21 symmetry as demonstrated by Rietveld refinement analysis. FESEM micrograph, supported by the particle size distribution plot, confirmed that the synthesized phosphor possesses slightly irregular particles having a size in the nano-range (20–50 nm). EDX spectra and mapping provided insights into the distribution of Gd, Ga, O and Tb within the host lattice. Excitation at 272 nm results in multiple emission peaks equivalent to 5D4 → 7F3,4,5,6, with the strongest being the green emission at 543 nm due to the 5D4 → 7F5 electronic transition. The emission intensity increases with the rise in the Tb3+ doping concentration, reaching a maximum value at x = 3 mol% and then decreases. This decrease in intensity is caused by concentration quenching. Dipole–dipole interactions are the primary reason for the increased transfer of energy between nearby ions, leading to reduced luminescence. In addition, CIE coordinates and CCT values suggest Gd3GaO6:Tb3+ phosphors as efficient green-emitting materials for use in lighting and display technologies.
Author contributions
Reshu Kajal: data curation, writing – original draft, investigation, methodology; Devender Singh: writing – review & editing, resources, supervision; Rinki Jangra: validation; Pawan Kumar: project administration; Varun Kumar: software; Ramesh Kumar: visualization; Harish Kumar: formal analysis.
Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Data availability
Data will be made available on request.
Acknowledgements
The author (Reshu Kajal) wants to thank University Grants Commission, New Delhi, for providing the Junior Research Fellowship [221610025278].
References
- B. M. Manohara, H. Nagabhushana, D. V. Sunitha, K. Thyagarajan, B. D. Prasad, S. C. Sharma, B. M. Nagabhushana and R. P. Chakradhar, J. Alloys Compd., 2014, 592, 319–327, DOI:10.1016/j.jallcom.2014.01.003.
- S. Singh and D. Singh, J. Mater. Sci.: Mater. Electron., 2020, 31, 5165–5175, DOI:10.1007/s10854-020-03076-5.
- R. Kokate, P. Rohilla, S. Kaur, A. S. Rao and V. Singh, Optik, 2021, 243, 167391, DOI:10.1016/j.ijleo.2021.167391.
- P. Kumar, S. Singh, I. Gupta, V. Kumar and D. Singh, J. Mol. Struct., 2022, 1265, 133362, DOI:10.1016/j.molstruc.2022.133362.
- S. Cho, J. Kim, D. Hwang and S. W. Cho, Korean J. Mater. Res., 2016, 26, 67–72, DOI:10.1016/j.jallcom.2014.07.100.
- P. Kumar, D. Singh, I. Gupta, S. Singh and V. Kumar, Chem. Phys. Lett., 2023, 812, 140277, DOI:10.1016/j.cplett.2022.140277.
- G. Souadi, Ü. H. Kaynar, M. E. Ayvacikli and N. Can, Appl. Radiat. Isot., 2023, 199, 110905, DOI:10.1016/j.apradiso.2023.110905.
- R. Kajal, D. Singh, R. Jangra, P. Kumar, V. Kumar, H. Kumar and R. Kumar, J. Alloys Compd., 2025, 1050, 185688, DOI:10.1016/j.jallcom.2025.185688.
- T. P. Jyothi, K. Gopal, D. V. Sunitha, D. Kavyashree and A. G. Prakash, Ceram. Int., 2025, 51(26, Part C), 50644–50657, DOI:10.1016/j.ceramint.2025.08.293.
- B. Mari, K. C. Singh, P. Cembrero-Coca, I. Singh, D. Singh and S. Chand, Displays, 2013, 34, 346–351, DOI:10.1016/j.displa.2013.07.003.
- I. Gupta, P. Kumar, S. Singh, S. Bhagwan, S. K. Chhikara and D. Singh, Inorg. Chim. Acta, 2022, 543, 121183, DOI:10.1016/j.ica.2022.121183.
- S. Raoux, S. Anders, K. M. Yu, I. C. Ivanov and I. G. Brown, MRS Online Proc. Libr., 1995, 392, 241–246, DOI:10.1557/PROC-392-241.
- Y. Gong, L. Li, J. Chen and H. Guo, Materials, 2023, 16, 4711, DOI:10.1016/j.jallcom.2022.164016.
- O. N. Kondrat'eva, M. N. Smirnova, G. E. Nikiforova, A. V. Tyurin, V. A. Rassulov and V. A. Ketsko, J. Alloys Compd., 2024, 982, 173703, DOI:10.1016/j.jallcom.2024.173703.
- R. Kajal, R. Jangra, P. Kumar, D. Singh, R. Kumar and H. Kumar, J. Mol. Struct., 2026, 1359, 145495, DOI:10.1016/j.molstruc.2026.145495.
- M. M. Rodríguez-García, J. G. Williams and I. R. Evans, J. Mater. Chem. C, 2019, 7, 7779–7787, 10.1039/c9tc02336d.
- I. Gupta, S. Singh, S. Bhagwan and D. Singh, Ceram. Int., 2021, 47, 19282–19303, DOI:10.1016/j.ceramint.2021.03.308.
- J. He, A. A. Haider, H. Jiang, C. Liu, J. Li, W. Qian, J. Zhang and J. Zhu, Inorg. Chem. Commun., 2025, 13, e01767, DOI:10.1002/adom.202501767.
- S. Malik, K. Jakhar, D. Singh, S. Redhu, V. Aggarwal, S. Kumar, R. S. Malik and P. Kumar, Inorg. Chem. Commun., 2025, 181, 115231, DOI:10.1016/j.inoche.2025.115231.
- L. A. Rocha, R. L. Siqueira, J. Esbenshade, M. A. Schiavon and J. L. Ferrari, J. Alloys Compd., 2018, 731, 889–897, DOI:10.1016/j.jallcom.2017.10.084.
- P. Kumar, S. Singh, I. Gupta, V. Kumar and D. Singh, Optik, 2022, 267, 169709, DOI:10.1016/j.ijleo.2022.169709.
- Z. Li, Z. Lyu, P. Luo, S. Wei, C. Zhuo, D. Sun, S. Shen and H. You, Inorg. Chem. Front., 2023, 10, 6746–6753, 10.1039/D3QI01525D.
- D. Singh, V. Tanwar, A. P. Simantilleke, B. Mari and P. S. Kadyan, J. Mater. Sci.:Mater. Electron., 2016, 27, 2260–2266, DOI:10.1007/s10854-015-4020-1.
- L. A. Kolahalam, I. K. Viswanath, B. S. Diwakar, B. Govindh, V. Reddy and Y. L. Murthy, Mater. Today Proc., 2019, 18, 2182–2190, DOI:10.1016/j.matpr.2019.07.371.
- S. Singh, D. Singh, P. Siwach, I. Gupta and P. Kumar, Appl. Res., 2025, 4, e202400190, DOI:10.1002/appl.202400190.
- M. Farooq, H. Rafiq and M. H. Rasool, ECS J. Solid State Sci. Technol., 2023, 12, 126002, DOI:10.1149/2162-8777/ad1062.
- D. Singh, S. Sheoran, V. Tanwar and S. Bhagwan, J. Mater. Sci.:Mater. Electron., 2017, 28, 3243–3253, DOI:10.1007/s10854-016-5914-2.
- R. Kajal, D. Singh, R. Jangra, B. Dahiya, M. Kadian, P. Kumar, R. Kumar and H. Kumar, Opt. Mater., 2026, 174, 117942, DOI:10.1016/j.optmat.2026.117942.
- S. Choi, K. Kim, S. Nahm and H. K. Jung, Opt. Mater., 2009, 31, 1684–1687, DOI:10.1016/j.optmat.2009.04.007.
- V. Tanwar, S. Singh, I. Gupta, P. Kumar, H. Kumar, B. Mari and D. Singh, J. Mol. Struct., 2022, 1250, 131802, DOI:10.1016/j.molstruc.2021.131802.
- P. Barik, A. K. Verma, R. Kumar, V. Kumar, P. Dewangan, M. Sahu and I. P. Sahu, Appl. Phys. A, 2024, 130, 391, DOI:10.1007/s00339-024-07525-4.
- P. Kumar, S. Singh, I. Gupta, K. Nehra, V. Kumar and D. Singh, Mater. Chem. Phys., 2023, 295, 127035, DOI:10.1016/j.matchemphys.2022.127035.
- T. Ghrib, I. Massoudi, A. L. Al-Otaibi, A. Al-Malki, A. Kharma, E. Al-Hashem, R. A. Al-Ghamdi and R. A. Al-Zuraie, J. Inorg. Organomet. Polym. Mater., 2021, 31, 239–250, DOI:10.1007/s10904-020-01761-w.
- P. Kumar, D. Singh and I. Gupta, RSC Adv., 2023, 13, 7703–7718, 10.1039/D3RA00636K.
- A. S. Rao, Sens. Actuators, A, 2024, 372, 115336, DOI:10.1016/j.sna.2024.115336.
- B. M. Manohara, H. Nagabhushana, D. V. Sunitha, K. Thyagarajan, B. D. Prasad, S. C. Sharma, B. M. Nagabhushana and R. P. Chakradhar, J. Alloys Compd., 2014, 592, 319–327, DOI:10.1016/j.jallcom.2014.01.003.
- I. Gupta, D. Singh, S. Singh, P. Kumar, S. Bhagwan and V. Kumar, Chem. Phys. Lett., 2023, 814, 140350, DOI:10.1016/j.cplett.2023.140350.
- R. E. Muenchausen, L. G. Jacobsohn, B. L. Bennett, E. A. McKigney, J. F. Smith, J. A. Valdez and D. W. Cooke, J. Lumin., 2007, 126, 838–842, DOI:10.1016/j.jlumin.2006.12.004.
- P. Kumar, S. Singh, I. Gupta, A. Dalal, V. Kumar and D. Singh, J. Mater. Sci. Eng. B, 2023, 288, 116189, DOI:10.1016/j.mseb.2022.116189.
- P. K. Jisha, S. C. Prashantha and H. Nagabhushana, J. Sci.:Adv. Mater. Devices, 2017, 2, 437–444, DOI:10.1016/j.jsamd.2017.10.001.
- P. Kumar, D. Singh and H. Kumar, Mater. Sci. Semicond. Process., 2024, 174, 108162, DOI:10.1016/j.mssp.2024.108162.
- I. Gupta, D. Singh, S. Singh, P. Kumar, S. Bhagwan, V. Kumar, H. Kumar and S. K. Chhikara, Luminescence, 2023, 38, 585–599, DOI:10.1002/bio.4486.
- M. Ayvacıklı, A. Canimoglu, Y. Ü. Karabulut, Z. Kotan, L. K. Herval, M. P. De Godoy, Y. G. Gobato, M. Henini and N. Can, J. Alloys Compd., 2014, 590, 417–423, DOI:10.1016/j.jallcom.2013.12.135.
- P. Kumar, D. Singh and I. Gupta, Mater. Res. Bull., 2023, 167, 112413, DOI:10.1016/j.materresbull.2023.112413.
- Y. S. Vidya, K. Gurushantha, H. Nagabhushana, S. C. Sharma, K. S. Anantharaju, C. Shivakumara, D. Suresh, H. P. Nagaswarupa, S. C. Prashantha and M. R. Anilkumar, J. Alloys Compd., 2015, 622, 86–96, DOI:10.1016/j.jallcom.2014.10.024.
- Z. C. Wu, J. X. Shi, J. Wang, M. L. Gong and Q. Su, J. Solid State Chem., 2006, 179, 2356–2360, DOI:10.1016/j.jssc.2006.04.030.
- S. Singh and D. Singh, J. Mater. Sci.: Mater. Electron., 2021, 32, 17674–17685, DOI:10.1007/s10854-021-06303-9.
- C. Liu, Y. Wang, R. Li, G. Zhou, A. A. Haider, X. Huang, X. Zhao, Y. Qin, H. Li, H. Jiang and J. Li, Aggregate, 2025, 6, e70125, DOI:10.1002/agt2.70125.
- P. Kumar, S. Singh, I. Gupta, V. Kumar and D. Singh, Luminescence, 2022, 37, 1932–1941, DOI:10.1002/bio.4377.
- T. A. Safeera and E. I. Anila, J. Lumin., 2019, 205, 277–281, DOI:10.1016/j.jlumin.2018.09.033.
- P. Kumar, D. Singh, I. Gupta, S. Singh, S. Nehra and R. Kumar, RSC Adv., 2023, 13, 7752–7765, 10.1039/D3RA00735A.
- T. T. Chanu and N. R. Singh, Spectrochim. Acta, Part A Mol. Biomol. Spectrosc., 2018, 191, 539–546, DOI:10.1016/j.saa.2017.10.060.
- D. Singh and S. Sheoran, J. Mater. Sci.:Mater. Electron., 2016, 27, 12707–12718, DOI:10.1007/s10854-016-5405-5.
- Z. Li, P. Wang, T. Yang, H. Yu, B. Xiao and M. Zhang, J. Phys. Chem. C, 2015, 119, 27688–27694, DOI:10.1021/acs.jpcc.5b09328.
- P. Kumar, D. Singh, S. Singh, H. Kumar and R. Kumar, RSC Adv., 2024, 14, 18716–18729, 10.1039/D4RA03833A.
- M. Luo, X. Sha, B. Chen, X. Zhang, H. Yu, X. Li, J. Zhang, S. Xu, Y. Cao, Y. Wang and X. Wang, J. Am. Ceram. Soc., 2022, 105, 3353–3363, DOI:10.1111/jace.18299.
- P. Kumar, D. Singh and H. Kumar, RSC Adv., 2024, 14, 755–770, 10.1039/D3RA07175H.
- F. L. Joaquim, L. A. Rocha, C. S. Júnior, J. Esbenshade, M. A. Schiavon and J. L. Ferrari, J. Alloys Compd., 2019, 774, 694–699, DOI:10.1016/j.jallcom.2018.09.148.
- P. Kumar, D. Singh and H. Kumar, Mater. Chem. Phys., 2024, 320, 129418, DOI:10.1016/j.matchemphys.2024.129418.
- H. Li, A. A. Haider, Z. Xie, C. Liu, H. Zhang, H. Jiang, J. Li and J. Zhu, Adv. Sci., 2025, 12, 2415989, DOI:10.1002/advs.202415989.
- J. Hakami, Ü. H. Kaynar, M. E. Ayvacikli, M. B. Çoban, J. Garcia-Guinea, P. D. Townsend, M. Oglakci and N. Can, Ceram. Int., 2022, 48, 32256–32265, DOI:10.1016/j.ceramint.2022.07.167.
- J. Zhu, J. Xiang and Y. Mao, Optik, 2021, 240, 166920, DOI:10.1016/j.ijleo.2021.166920.
- X. Huang, C. He, X. Wen, Z. Huang, Y. Liu, M. Fang, X. Wu and X. Min, Opt. Mater., 2019, 95, 109191, DOI:10.1016/j.optmat.2019.109191.
- X. Huang, B. Li and H. Guo, J. Alloys Compd., 2017, 695, 2773–2780, DOI:10.1016/j.jallcom.2016.11.224.
- Z. Huang, Z. Lyu, S. Shen, S. Wang, Z. Yang, C. Chen and H. You, Inorg. Chem., 2024, 63, 6362–6369, DOI:10.1021/acs.inorgchem.4c00123.
- H. Guo, B. Devakumar, B. Li and X. Huang, Dyes Pigm., 2018, 151, 81–88, DOI:10.1016/j.dyepig.2017.12.051.
- D. Singh, V. Tanwar, A. P. Samantilleke, B. Mari, S. Bhagwan, P. S. Kadyan and I. Singh, J. Electron. Mater., 2016, 45, 2718–2724, DOI:10.1007/s11664-015-4318-z.
- V. R. Panse, N. S. Kokode, A. Shukla, A. N. Yerpude and S. J. Dhoble, Optik, 2016, 127, 5860–5864, DOI:10.1016/j.ijleo.2016.04.031.
- D. Singh, V. Tanwar, S. Bhagwan, P. S. Kadyan and B. Mari, Adv. Sci. Lett., 2014, 20, 1726–1729, DOI:10.1166/asl.2014.5736.
- Z. Liu, S. Wang, Y. Xu, J. Li, Y. Du, J. Hong and Z. Chen, Appl. Phys. A, 2025, 131, 449, DOI:10.1007/s00339-025-08518-7.
- S. Zhongxiang, S. Jun, W. Jing, L. Yang and L. Yongfu, Spectroscopy, 2023, 38, 28–34, DOI:10.56530/spectroscopy.xr3287v2.
- P. Kumar, D. Singh, S. Kadyan, H. Kumar and R. Kumar, RSC Adv., 2024, 14, 16560–16573, 10.1039/D4RA02571G.
- K. Maheshwari and A. S. Rao, Opt. Mater., 2023, 137, 113533, DOI:10.1016/j.optmat.2023.113533.
- J. Deng, Z. Wang, W. Zhou, M. Yu, J. Min, X. Jiang, Z. Xue, C. Ma, Z. Cheng and G. Luo, Ceram. Int., 2023, 49, 14478–14486, DOI:10.1016/j.ceramint.2023.01.036.
- H. E. Sekrafi, E. Hergli, S. Ferdov, P. J. Coutinho and L. Rebouta, J. Alloys Compd., 2025, 1010, 177389, DOI:10.1016/j.jallcom.2024.177389.
- P. Kumar, D. Singh, I. Gupta, S. Singh, V. Kumar, H. Kumar and S. K. Chhikara, Inorg. Chem. Commun., 2022, 145, 110064, DOI:10.1016/j.inoche.2022.110064.
|
| This journal is © The Royal Society of Chemistry 2026 |
Click here to see how this site uses Cookies. View our privacy policy here.  Open Access Article
Open Access Article *a,
Rinki Jangraa,
Pawan Kumara,
Varun Kumarb,
Ramesh Kumarc and
Harish Kumard
*a,
Rinki Jangraa,
Pawan Kumara,
Varun Kumarb,
Ramesh Kumarc and
Harish Kumard



![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) sin
sin![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θ
θ