Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Piperazine-grafted magnetic graphene oxide as a sustainable heterogeneous catalyst for solvent-free Biginelli reaction
Esmail Rezaei-Seresht *,
Faezeh Jalambadani,
Samira Cheshak
*,
Faezeh Jalambadani,
Samira Cheshak ,
Behnam Mahdavi
,
Behnam Mahdavi and
Fatemeh Tafazzoli Gazkoh
and
Fatemeh Tafazzoli Gazkoh
Department of Chemistry, School of Sciences, Hakim Sabzevari University, Sabzevar, Iran. E-mail: e.rezaei@hsu.ac.ir; Tel: +98 5144013516
First published on 24th February 2026
Abstract
Multi-component reactions (MCRs) are powerful tools for the one-pot synthesis of complex organic compounds, with the Biginelli reaction being one of the most important due to its versatility in synthesizing a wide range of dihydropyrimidine-2-ones (DHPMs) with diverse biological activities. However, traditional MCR catalysts are often homogeneous and challenging to recycle, leading to environmental concerns and high costs. In this study, we developed a novel heterogeneous catalyst for the Biginelli reaction by functionalizing graphene oxide (GO) with piperazine (NH) to form a Fe3O4@GO-NH nanocomposite. The catalyst was characterized using FT-IR, TGA, and SEM/EDX and showed high catalytic activity for the Biginelli reaction under solvent-free conditions. The optimal reaction conditions were determined by adjusting various parameters such as temperature, stability and reproducibility, time, and the molar percentage of Fe3O4@GO-NH. As a result of this eco-friendly approach, 32 Biginelli products were successfully obtained with high yields. The catalyst was also reusable, making it a more environmentally friendly and cost-effective alternative to conventional homogeneous catalysts.
1. Introduction
Multi-component reactions (MCRs) are a class of synthetic methods in which several raw materials are mixed to form a one-pot product.1 In these reactions, according to the conditions and the nature of the chemicals involved in the reaction, all the components in MCRs may react linearly (such as the Mannich reaction) or cyclize to form more complex molecules (such as the Hantzsch dihydropyridine synthesis).2 MCRs, characterized by their unique one-step nature, offer economic advantages by eliminating intermediate product separation and purification while achieving higher yields than two- or multi-step reactions.3,4 A prime example of a versatile MCR is the Biginelli reaction, discovered in 1893.3,4 This three-component condensation reaction efficiently produces dihydropyrimidinones (DHPMs), a class of molecules exhibiting diverse biological activities such as antiviral, anti-tumor, antibacterial, and anti-inflammatory properties.5–10 DHPMs are being progressively utilized to create materials like polymers, adhesives, fabric dyes, pharmaceutical compounds, and more.5 The Biginelli reaction typically involves a 1,3-dicarbonyl compound, an aromatic aldehyde, and urea/thiourea under acidic conditions.11,12 The DHPMs derived from the Biginelli reaction have attracted significant interest in drug discovery due to their potential therapeutic applications. Current research focuses on developing DHPM-based drugs for various diseases, including neurodegenerative, cardiovascular, and metabolic disorders.13–16While the Biginelli reaction is well-established, ongoing efforts seek to optimize its efficiency and sustainability. Traditional methods often involve harsh acidic conditions, long reaction times, and toxic solvents. In addition, most reported catalysts for the Biginelli reaction are homogeneous acids or metal salts, suffering from drawbacks such as difficulty in separation and recycling, catalyst leaching, and generation of toxic waste. In some cases, the reported heterogeneous catalysts showed limited activity, required high temperatures, or produced moderate yields due to poor active site accessibility or low surface area.17–20 Researchers are actively developing new catalysts and reaction conditions to address these challenges, aiming for greener and more efficient syntheses of DHPMs.21–25
Graphene oxide (GO), a single layer of sp2 hybridized carbon atoms arranged in a honeycomb lattice structure, possesses oxygen-containing functional groups such as hydroxyl, epoxide, and carboxyl groups.6 These functional groups endow GO with unique properties that make it an ideal candidate for catalytic applications in organic synthesis. Notably, GO boasts a high surface area, facilitating the adsorption of various reaction components.7 Additionally, the oxygen functionalities on GO impart Brønsted acidity, enabling it to act as a proton donor and participate in reaction mechanisms.8 These features collectively position GO as a versatile platform for developing efficient and sustainable catalysts for MCRs. GO has emerged as a versatile catalyst for various MCRs. Its unique properties, including Brønsted acidity, bio-compatibility, high surface area, inertness, and outstanding electronic, optical, thermal and mechanical properties, improve reaction efficiency across diverse MCR types.9–11 Moreover, it is derived from affordable and readily available starting materials. One notable derivative of GO is GO-NH, which is obtained through the functionalization of GO with piperazine molecules12 This functionalization process involves the nucleophilic attack of piperazine on the epoxide carbons present on the GO sheet, leading to the opening of the epoxide rings and the attachment of piperazine molecules onto the GO surface. In the Biginelli reaction, for example, GO's unique combination of acidity and adsorption properties is believed to significantly improve yields and reaction times for synthesizing biologically active DHPMs compared to traditional methods.5 This trend continues with the Knoevenagel condensation, a versatile MCR for creating valuable α,β-unsaturated carbonyl compounds. Here, π–π stacking interactions between GO and aromatic aldehydes are thought to be crucial in boosting reaction efficiency.6 GO's effectiveness further benefits the Mannich reaction, a three-component condensation for β-amino carbonyl compounds.7
To address the mentioned catalyst drawbacks in the Biginelli reaction, such as limited activity, difficulty in separation and recycling, low surface area, the generation of toxic waste, and the use of toxic solvents in the reaction. We envisaged that composite Fe3O4@GO-NH has several features that make it a promising catalyst for reactions involving multiple proton-transfer (protonation and deprotonation) steps, such as the Biginelli reaction. The Fe3O4 particles provide Lewis acid sites to activate the aldehyde. At the same time, the piperazine functionalization introduces basic sites that enhance the nucleophilicity of urea (or thiourea) and facilitate enolate formation from the β-keto ester component. Traditional catalysts often fail to manage these dual roles efficiently, leading to lower yields. Additionally, the GO component offers a high surface area, improving catalytic activity. Finally, the composite's magnetic properties enable easy separation and recycling, making it an efficient and reusable catalyst.
Therefore, this study focuses on preparing and characterizing Fe3O4@GO-NH and its application as a heterogeneous, magnetically-separable catalyst in the solvent-free Biginelli reaction for synthesizing dihydropyrimidine-2-ones.
2. Experimental
2.1. Materials and reagents
2.2. Instrumental
1H NMR spectra were taken by Bruker Advance-300 MHz in DMSO-d6 solvent. The FT-IR 8400s device made by SHIMADZU Company was used to prepare the IR spectrum of synthetic products by KBr tablets in the 400–4000 cm−1 frequency range. An Electrothermal-9200 device was used to determine the melting point of the products. In order to check the progress of the reaction, the purity and identification of the compounds were performed by TLC chromatography using a UV lamp with a wavelength of 256–365 made by Merck. Also, the Fe3O4@GO-NH catalyst was identified by the EDX device made by BuAli Institute with an accelerating voltage of 150 kV, a beam current of 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 700
700![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000
000![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 nA and a magnification of 100
000 nA and a magnification of 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000. The XRD pattern was obtained with Cu Kα radiation at 45 kV and 40 mA and at a rate of 2° min−1, using the STOE PW2773.00 device. Elemental analysis was performed on a TRUSPEC CHNS analyzer.
000. The XRD pattern was obtained with Cu Kα radiation at 45 kV and 40 mA and at a rate of 2° min−1, using the STOE PW2773.00 device. Elemental analysis was performed on a TRUSPEC CHNS analyzer.
2.3. Catalytic experiments
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) n-hexane 1
n-hexane 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). Finally, the reaction mixture was cooled at room temperature and then dissolved in 5 mL of ethanol, and after separating the catalyst by an external magnet, the remaining liquid was poured into ice-water (50 mL), and the precipitate obtained was washed with water and dried.
1). Finally, the reaction mixture was cooled at room temperature and then dissolved in 5 mL of ethanol, and after separating the catalyst by an external magnet, the remaining liquid was poured into ice-water (50 mL), and the precipitate obtained was washed with water and dried.2.3.2.1 Ethyl 4-(4-bromophenyl)-6-methyl-2-oxo-1,2,3,4-tetrahydropyrimidine-5-carboxylate (2b). Light yellow powder (0.33 g, 98%); mp 215 °C (from ethanol) (lit., 15
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 215 °C); IR (KBr) vmax/cm−1 3340 and 3120 (N–H), 1723 (C
215 °C); IR (KBr) vmax/cm−1 3340 and 3120 (N–H), 1723 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1550 (C
O), 1550 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C), 1468 (CH2), 1383 (CH3), 1240 (C–O); 1H NMR δH (300 MHz; DMSO-d6; Me4Si) 1.11 (3H, t, J = 6.3 Hz, OCH2CH3). 2.26 (3H, s, CH3), 4.01 (2H, q, J = 6.3 Hz, OCH2CH3), 5.14 (1H, s, CH), 7.21 (2H, d, J = 6.9 Hz, H-aromatic), 7.47 (2H, d, J = 6.9 Hz, H-aromatic), 7.79 (1H, s, NH), 9.26 (1H, s, NH).
C), 1468 (CH2), 1383 (CH3), 1240 (C–O); 1H NMR δH (300 MHz; DMSO-d6; Me4Si) 1.11 (3H, t, J = 6.3 Hz, OCH2CH3). 2.26 (3H, s, CH3), 4.01 (2H, q, J = 6.3 Hz, OCH2CH3), 5.14 (1H, s, CH), 7.21 (2H, d, J = 6.9 Hz, H-aromatic), 7.47 (2H, d, J = 6.9 Hz, H-aromatic), 7.79 (1H, s, NH), 9.26 (1H, s, NH).
2.3.2.2 Ethyl 4-(4-chlorophenyl)-6-methyl-2-oxo-1,2,3,4-tetrahydropyrimidine-5-carboxylate (2c). White powder (0.29 g, 97%); mp 210 °C (from ethanol) (lit., 16
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 212 °C); IR (KBr) vmax/cm−1 3318 and 3133 (N–H), 1722 (C
212 °C); IR (KBr) vmax/cm−1 3318 and 3133 (N–H), 1722 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1575 (C
O), 1575 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C), 1443 (CH2), 1314 (CH3), 1174 (C–O), 710 (C–Cl); 1H NMR δH (300 MHz; DMSO-d6; Me4Si) 1.11 (3H, t, J = 6.9 Hz, OCH2CH3). 2.27 (3H, s, CH3), 4.01 (2H, q, J = 6.9 Hz, OCH2CH3), 5.15 (1H, d, J = 3.0 Hz CH), 7.27 (2H, d, J = 7.8 Hz, H-aromatic), 7.41 (2H, d, J = 7.8 Hz, H-aromatic), 7.79 (1H, s, NH), 9.27 (1H, s, NH).
C), 1443 (CH2), 1314 (CH3), 1174 (C–O), 710 (C–Cl); 1H NMR δH (300 MHz; DMSO-d6; Me4Si) 1.11 (3H, t, J = 6.9 Hz, OCH2CH3). 2.27 (3H, s, CH3), 4.01 (2H, q, J = 6.9 Hz, OCH2CH3), 5.15 (1H, d, J = 3.0 Hz CH), 7.27 (2H, d, J = 7.8 Hz, H-aromatic), 7.41 (2H, d, J = 7.8 Hz, H-aromatic), 7.79 (1H, s, NH), 9.27 (1H, s, NH).
3. Results and discussion
3.1. Synthesis and characterization of composite Fe3O4@GO-NH
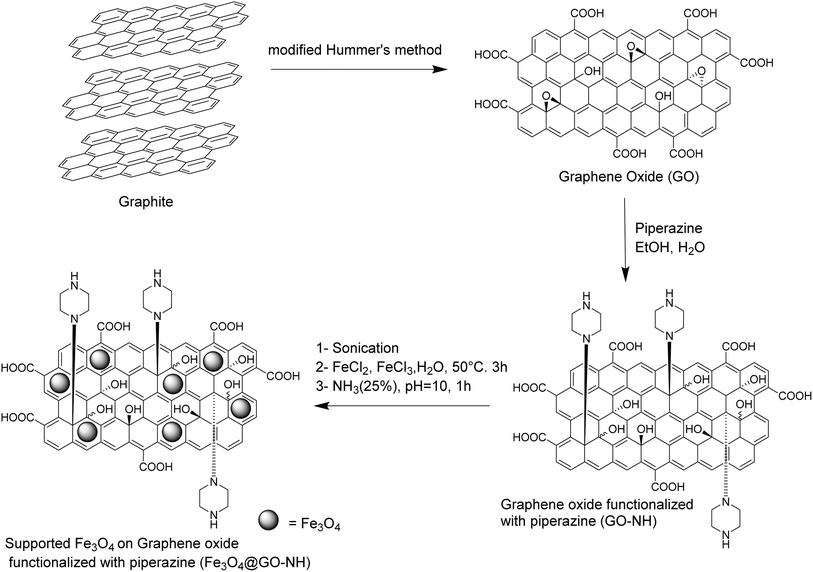
Initially, GO was synthesized from graphite using a modified version of Hammer's method.10 Subsequently, GO was functionalized with piperazine molecules (GO-NH) through a nucleophilic attack on the epoxide carbons present on the GO sheet.11 Elemental analysis of GO-NH revealed a nitrogen content of 6.46 wt%, indicating the presence of piperazine molecules. Finally, the magnetic catalyst Fe3O4@GO-NH was synthesized via the co-precipitation method, as outlined in Scheme 1. The characterization of the catalyst was confirmed using various techniques, including TGA and EDX.The thermogravimetric analysis (TGA) of the catalyst (SI) shows two main steps of weight loss, which are important for assessing its stability at the reaction temperature of 130 °C. The first weight loss (∼32%) occurs in the range of approximately 28–92 °C and is attributed to the removal of moisture and volatile surface species. The weight change between 92 °C and 130 °C is minimal (a minor weight loss of ∼2% between 91 and 130 °C). This slight loss is attributed to the evaporation of surface-adsorbed water, which is consistent with the hydrophilic nature of the GO sheets. Therefore, the catalyst retains its structural integrity and chemical stability under the reaction conditions (130 °C). The elemental composition of catalyst Fe3O4@GO-NH were determined by EDX (SI). Peak N Kα belongs to nitrogen, suggesting a notable amount of nitrogen content in the catalyst. Also, several peaks associated with iron (Fe Lα, Fe Kα, and Fe Kβ) are visible, indicating the presence of iron in the catalyst. Therefore, the EDX spectrum suggests that the catalyst primarily contains elements iron, oxygen, nitrogen and carbon in its composition.
To get an insight on the nature of acid–base sites of Fe3O4@GO-NH that are essential in catalysis of the Biginelli reaction, the point of zero charge (PZC) for catalyst Fe3O4@GO-NH was determined by mass titration method described by Noh and Schwarz.12 A PZC value of 4.70 (SI) suggests that the Fe3O4@GO-NH surface is net positively charged at pH < 4.7 and negatively charged above this value. This behavior reflects the coexistence of acidic sites derived from –COOH groups on the GO surface and basic sites from piperazine grafting.
3.2. Catalytic properties of Fe3O4@GO-NH in model reaction
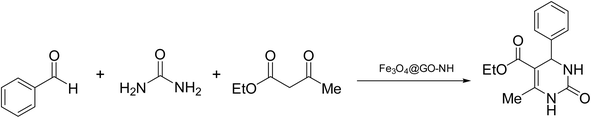
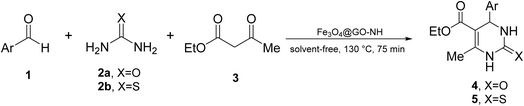
We chose the reaction of benzaldehyde (1 mmol), urea (1.3 mmol) and ethyl acetoacetate (1,3-dicarbonyl) (1 mmol) as the model reaction to evaluate the catalytic efficiency of Fe3O4@GO-NH in the Biginelli reaction, as well as to achieve optimal reaction parameters, including temperature, time, amount of the catalyst, and catalyst type (Scheme 2).In the first step, we optimized the amount of the catalyst to determine its effect on the reaction yield. As depicted in Fig. 1, the yield improves significantly as the catalyst loading increases from 0 to 11.5 wt%, based on ethyl acetoacetate. This indicates that the catalyst effectively promotes the reaction by providing active sites necessary for the reaction mechanism. Beyond 11.5 wt%, further increases in the catalyst loading (15.4 and 23.1 wt%) did not enhance the yield, suggesting that the reaction has reached a saturation point where the maximum catalytic efficiency is achieved.
 | ||
| Fig. 1 Effect of catalyst loading on the yield. Catalyst loading is expressed as weight percent of Fe3O4@GO-NH relative to ethyl acetoacetate. | ||
Further optimization focused on the reaction time parameter (Fig. 2). The figure illustrates a steady increase in the yield as the reaction time progresses from 15 to 75 minutes. At 75 minutes, the yield reached its maximum value, while further extending the reaction time to 90 and 120 minutes did not result in any further increase in the yield. This suggests that 75 minutes is the optimal reaction time for maximizing yield in the reaction.
Further investigation examined the effect of reaction temperature on the yield. Thus, six model reactions were conducted, each using 0.015 g Fe3O4@GO-NH (a catalyst loading of 11.5 wt%) for 75 minutes at temperatures between 45 °C and 140 °C (Fig. 3). The data show that 130 °C is the optimal reaction temperature, and the yield rises steadily from approximately 20% at 45 °C to 97% at 130 °C, reflecting the significant effect of temperature on the Biginelli product yield. However, higher temperatures, such as 140 °C, do not significantly improve yield. As a result, the optimized reaction conditions were determined to include a reaction time of 75 minutes, a temperature of 130 °C, and the catalyst loading of 11.5 wt%.
3.3. Efficiency of Fe3O4/GO-NH
We then evaluated several related catalysts in the model reaction under the optimized reaction conditions to compare their efficiency with that of Fe3O4@GO-NH. As shown in Table 1, Fe3O4@GO-NH provided the highest yield among the tested catalysts while offering ease of recyclability. Its superiority over Fe3O4@rGO-NH (rGO is reduced graphene oxide) stems from GO's higher density of –COOH groups, which adds a more Brønsted acidic sites together with the piperazine basic sites. These cooperative acid–base surface functionalities improve the reaction yield.| Entry | Catalyst | Yield (%) | Ref. |
|---|---|---|---|
| a Reaction conditions: benzaldehyde (1 mmol), urea (1.3 mmol), ethyl acetoacetate (1 mmol), solvent-free, catalyst loading (0.015 g, 11.5 wt%), 130 °C, 75 min. | |||
| 1 | GO | 52 | This work |
| 2 | GO-NH | 58 | This work |
| 3 | Fe3O4 | 38 | This work |
| 4 | Fe3O4 | 45 | 13 |
| 5 | Fe3O4@rGO-NH | 63 | This work |
| 6 | Fe3O4@GO-NH | 97 | This work |
Like Fe3O4@GO-NH, the PZC value was determined for Fe3O4@rGO-NH to demonstrate the expected decrease in the –COOH content of Fe3O4@rGO-NH by the reduction. A value of 5.21 for Fe3O4@rGO-NH was obtained (Fig. S9 in SI), confirming its decreased catalytic activity relative to Fe3O4@GO-NH due to less Brønsted acidic sites for the catalysis.
| Entry | ArCHO | X | Biginelli product | Yield (%) | mp (°C) | |
|---|---|---|---|---|---|---|
| Found (from ethanol) | Reported (lit. reference) | |||||
| a Reaction conditions: starting aldehyde compound (1 mmol); urea (1.3 mmol); ethyl acetoacetate (1 mmol); solvent-free; Fe3O4@GO-NH loading (0.015 g, 11.5 wt%); time = 75 min; temperature = 130 °C. | ||||||
| 1 | 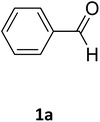 |
O | 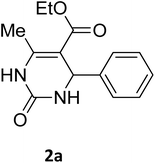 |
97 | 207 | 210 (ref. 14) |
| 2 | 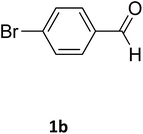 |
O | 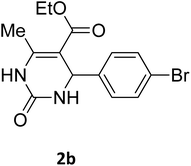 |
98 | 215 | 215 (ref. 15) |
| 3 | 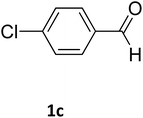 |
O | 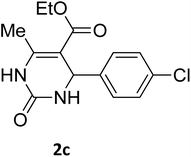 |
97 | 210 | 212 (ref. 16) |
| 4 |  |
O |  |
93 | 202 | 203 (ref. 17) |
| 5 |  |
O |  |
94 | 204 | 204 (ref. 18) |
| 6 |  |
O |  |
85 | 198 | 197 (ref. 19) |
| 7 | 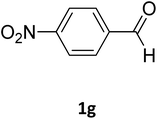 |
O | 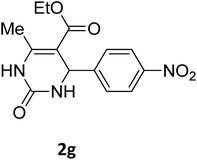 |
98 | 208 | 210 (ref. 20) |
| 8 | 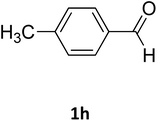 |
O | 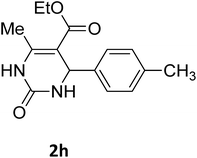 |
92 | 209 | 207 (ref. 21) |
| 9 | 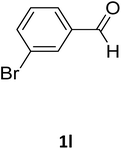 |
O | 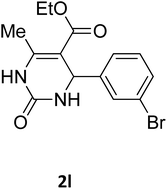 |
95 | 200 | 198 (ref. 22) |
| 10 | 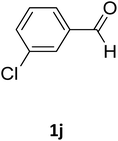 |
O | 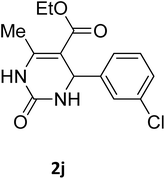 |
97 | 201 | 198 (ref. 23) |
| 11 | 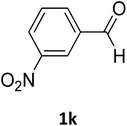 |
O | 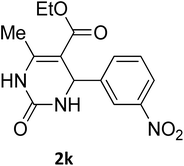 |
95 | 210 | 210 (ref. 24) |
| 12 | 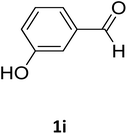 |
O | 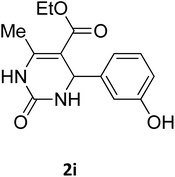 |
73 | 210 | 212 (ref. 25) |
| 13 | 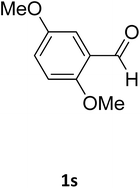 |
O | 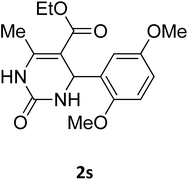 |
97 | 189 | 188 (ref. 26) |
| 14 | 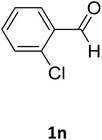 |
O | 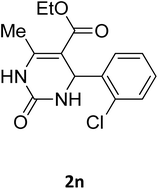 |
94 | 220 | 221 (ref. 27) |
| 15 |  |
O |  |
72 | 202 | 202 (ref. 28) |
| 16 |  |
O |  |
91 | 197 | 197 (ref. 29) |
| 17 | 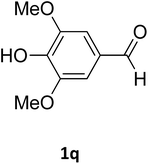 |
O | 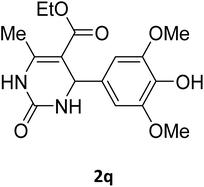 |
75 | 201 | 183 (ref. 30) |
| 18 | 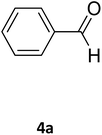 |
S | 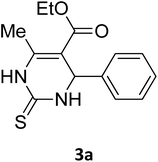 |
98 | 190 | 190 (ref. 31) |
| 19 | 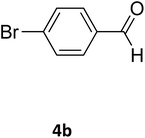 |
S | 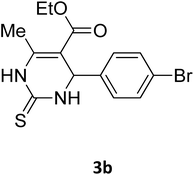 |
96 | 180 | 180 (ref. 32) |
| 20 | 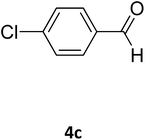 |
S | 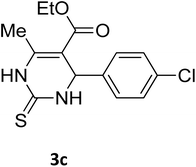 |
96 | 183 | 181 (ref. 33) |
| 21 | 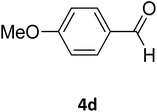 |
S | 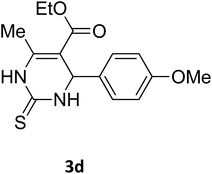 |
73 | 202 | 207 (ref. 34) |
| 22 | 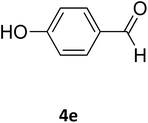 |
S | 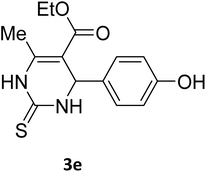 |
58 | 180 | 185 (ref. 35) |
| 23 |  |
S |  |
85 | 181 | 180 (ref. 36) |
| 24 |  |
S |  |
82 | 194 | 194 (ref. 37) |
| 25 |  |
S |  |
96 | 183 | 183 (ref. 38) |
| 26 |  |
S |  |
95 | 193 | 194 (ref. 39) |
| 27 | 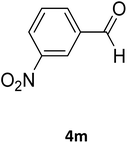 |
S | 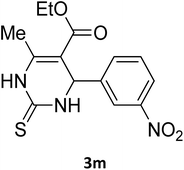 |
93 | 198 | 198 (ref. 40) |
| 28 | 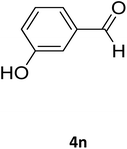 |
S | 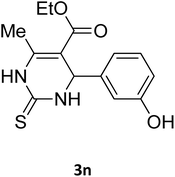 |
78 | 175 | 176 (ref. 41) |
| 29 | 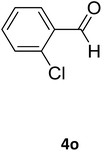 |
S | 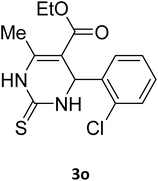 |
96 | 170 | 170 (ref. 42) |
| 30 | 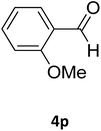 |
S | 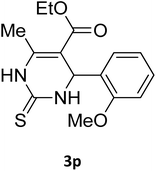 |
88 | 186 | 187 (ref. 43) |
| 31 | 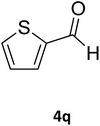 |
S | 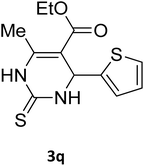 |
68 | 170 | 171 (ref. 44) |
| 32 | 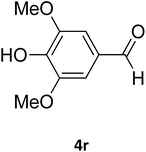 |
S | 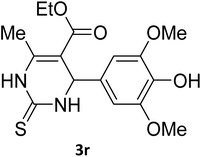 |
77 | 190 | 191 (ref. 41) |
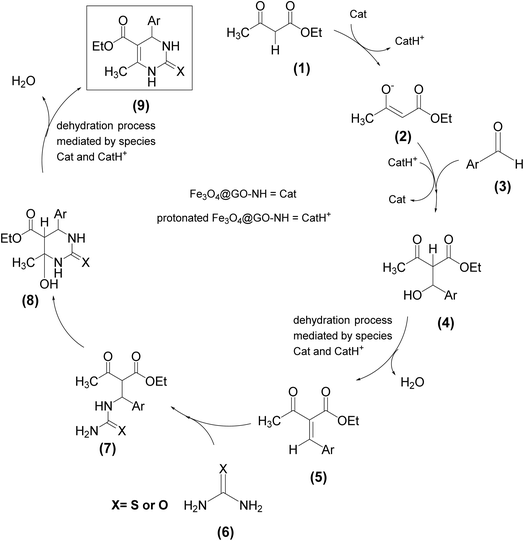
Considering the structural features of Fe3O4@GO-NH, its catalytic efficiency in the Biginelli reaction can be attributed to its ability to act as a Lewis (due to Fe3+ or Fe2+ ions present in Fe3O4) or Brønsted acid (due to the protonated piperazine moiety) and Brønsted base (due to the piperazine moiety) in multiple protonation and deprotonation reaction steps. Scheme 4 illustrates our proposed mechanism for the Biginelli reaction catalyzed by Fe3O4@GO-NH, depicting its dual role as both an acid and a base. Initially, it acts as a Brønsted base by deprotonating and activating β-ketoester 1, making it more nucleophilic for attack on aldehyde 3 activated by Fe3+ ion, Fe2+ ion or the protonated Fe3O4@GO-NH (CatH+). Moreover, the Lewis/Brønsted acidic and basic sites of the catalyst can promote the protonation and deprotonation steps in the dehydration processes, forming intermediate 5 and product 9.
| Entry | Temp. (°C) | Time (min) | Catalyst | Yield (%) | Ref. |
|---|---|---|---|---|---|
| 1 | 120 | 50 | Nano-ZrO2 sulfuric acid | 89 | 45 |
| 2 | 120 | 90 | H4[PVW11O40]/activated natural clay | 91 | 46 |
| 3 | 100 | 60 | Fe3O4@SiO2@Trz-Cu (copper 1,2,3-triazole complex) | 82 | 47 |
| 4 | 130 | 30 | Bentonite/PS-SO3H (polymer-supported sulphonic acid) | 90 | 48 |
| 5 | 130 | 75 | Fe3O4@GO-NH | 97 | This work |
4. Conclusion
In conclusion, the development of Fe3O4@GO-NH as a heterogeneous catalyst for the Biginelli reaction represents a significant advancement in sustainable catalysis. Fe3O4@GO-NH catalyst was successfully synthesized and analyzed by FT-IR, TGA, SEM/EDX. The catalyst exhibited remarkable catalytic activity under solvent-free conditions with easy isolation and excellent reusability. By optimizing reaction parameters, 32 Biginelli products were efficiently synthesized. The optimal conditions involved a solvent-free environment at 130 °C for 75 min, utilizing 0.015 g of catalyst Fe3O4@GO-NH. Furthermore, the catalyst showed superior catalytic performance compared to other catalysts, providing high yields and ease of recyclability. The approach demonstrated here offers a promising, eco-friendly alternative for the Biginelli reaction, with significant potential for broader applications in organic synthesis.Author contributions
Esmail Rezaei-Seresht: writing – review & editing, supervision, conceptualization. Samira Cheshak: writing – original draft, visualization, validation, investigation. Faezeh Jalambadani: writing – original draft, investigation, visualization, data curation. Fatemeh Tafazzoli Gazkoh: writing – original draft. Behnam Mahdavi: supervision.Conflicts of interest
There are no conflicts to declare.Data availability
Data sharing is not applicable to this article as no new data were created or analyzed in this study.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5ra05063d.
References
- J. Zhu, Q. Wang and M. Wang, Multi-component Reactions in Organic Synthesis, John Wiley & Sons, 2014 Search PubMed.
- W. Fan, K.-L. Wang, J. Li, M. Zhang and H. Liu, 5-(Hydroxymethyl)furfural and 5-(glucosyloxymethyl)furfural in multi-component reactions, in Recent Trends in Carbohydrate Chemistry, Elsevier, 2020, pp. 73 Search PubMed.
- S. Panda, P. Khanna and L. Khanna, Biginelli reaction: a green perspective, Curr. Org. Chem., 2012, 16, 507 CrossRef.
- B. Anjaneyulu and G. B. D. Rao, Mini Review: Biginelli Reaction for the Synthesis of Dihydropyrimidinones, Int. J. Eng. Technol. Res., 2015, 3, 26 Search PubMed.
- V. A. Adole, B. S. Jagdale, T. B. Pawar and B. S. Desale, Mater. Sci. Res. India, 2020, 17, 13 Search PubMed.
- D. R. Dreyer, S. Park, C. W. Bielawski and R. S. Ruoff, The chemistry of graphene oxide, Chem. Soc. Rev., 2010, 39, 228 Search PubMed.
- X. Gao, J. Jang and X. Huang, Scalable production of high-quality graphene oxide from graphene flake, J. Phys. Chem. C, 2010, 114, 3970 Search PubMed.
- E. Rodrigo, B. G. Alcubilla, R. Sainz, J. L. G. Fierro, R. Ferritto and M. B. Cid, Reduced graphene oxide supported piperazine in aminocatalysis, Chem. Commun., 2014, 50, 6270 Search PubMed.
- E. Rezaei-Seresht, S. Noroozi and B. Mahdavi, Nanocomposite Fe3O4@GO–NH as a reusable heterogeneous catalyst for the three-component synthesis of 2-amino-4H-chromenes, Org. Prep. Proced. Int., 2022, 54, 268 Search PubMed.
- N. Cao and Y. Zhang, Study of reduced graphene oxide preparation by Hummers' method and related characterization, J. Nanomater., 2015, 2015, 168125 Search PubMed.
- E. Rezaei-Seresht, F. T. Gazkoh and B. Maleki, Solvent-free Pechmann synthesis of coumarins catalysed by magnetic heterogeneous catalyst Fe3O4@rGO–NH, Carbon Lett., 2023, 33, 115 Search PubMed.
- J. S. Noh and J. A. Schwarz, Estimation of the point of zero charge of simple oxides by mass titration, J. Colloid Interface Sci., 1989, 130, 157 Search PubMed.
- A. Taravati, S. Ghorbani, M. Rahmani, R. Azadi and H. Rezazadeh, Innovative synthesis of antimicrobial Biginelli compounds using a recyclable iron oxide-based magnetic nanocatalyst, Sci. Rep., 2025, 15, 17909 Search PubMed.
- H. S. Oboudatian, H. Naeimi and M. Moradian, A Brønsted acidic ionic liquid anchored to magnetite nanoparticles as a novel recoverable heterogeneous catalyst for the Biginelli reaction, RSC Adv., 2021, 11, 7271 Search PubMed.
- G. Puthukkudy, S. Nagarajan and E. Kandasamy, Brønsted acidic 1-ethyl-1,2,4-triazolium phenylsulfonate as catalyst for Biginelli reaction, Asian J. Chem., 2018, 30, 1999 Search PubMed.
- Y. T. Wang, G. M. Tang and Y. S. Wu, A set of phenyl sulfonate metal coordination complexes triggered Biginelli reaction for the highly efficient synthesis of 3,4-dihydropyrimidin-2(1H)-ones under solvent-free conditions, Appl. Organomet. Chem., 2020, 34, e5542 Search PubMed.
- D. Patel, S. Sharma, A. Kumar and L. Singh, ZnO nanoparticles embedded on a reduced graphene oxide nanosheet (ZnO–NPs@rGO) as a proficient heterogeneous catalyst for a one-pot A3-coupling reaction, Eur. J. Inorg. Chem., 2021, 2021, 3578 CrossRef.
- E. Abbaspour-Gilandeh, A. Yahyazadeh and M. Aghaei-Hashjin, One-pot synthesis of 3,4-dihydropyrimidin-2(1H)-ones catalysed by SO3H@imine-ZCMNPs as a novel, efficient and reusable acidic nanocatalyst under solvent-free conditions, RSC Adv., 2018, 8, 40243 RSC.
- S. Ghassamipour and A. Sardarian, One-pot synthesis of dihydropyrimidinones by dodecylphosphonic acid as solid Brønsted acid catalyst under solvent-free conditions via Biginelli condensation, J. Iran. Chem. Soc., 2010, 7, 237 CrossRef.
- Z. Zhao, H. Dai and L. Shi, Synthesis of 3,4-dihydropyrimidin-2(1H)-ones catalysed by poly(ferric 2-acrylamido-2-methylpropanesulfonate), Chem. Pap., 2021, 75, 583 CrossRef.
- F. Mohamadpour, M. Bakhtiari, B. Rabiee and A. Salimi, Phthalic acid: a green, biodegradable and environmentally benign di-functional Brønsted acid catalyst for the one-pot synthesis of 3,4-dihydropyrimidin-2-(1H)-one derivatives and substituted dihydro-2-oxypyrroles, J. Chil. Chem. Soc., 2018, 63, 3811 CrossRef.
- A. Nakhaei, A. Davoodnia and S. Yadegarian, Nano isopolyoxomolybdate catalysed Biginelli reaction for one-pot synthesis of 3,4-dihydropyrimidin-2(1H)-ones and -thiones under solvent-free conditions, Russ. J. Gen. Chem., 2016, 86, 2870 CrossRef.
- M.-S. Mashhoori, R. Sandaroos and A. Zeraatkar Moghaddam, Polymeric imidazolium ionic liquid-tagged manganese Schiff base complex: an efficient catalyst for the Biginelli reaction, Res. Chem. Intermed., 2020, 46, 4939 Search PubMed.
- L. Hemmesi and H. Naeimi, Sonochemical synthesis of 1,4-dihydropyrimidinones catalysed by reduced graphene oxide encapsulated zinc oxide nanoparticles, Inorg. Nano-Met. Chem., 2023, 1, 111 Search PubMed.
- A. K. Prasad, P. Arya, S. Bhatia, R. K. Sharma, R. Singh, B. K. Singh and V. S. Parmar, Synthesis and lipase-catalysed enantioselective acylation studies on ethyl 4-aryl-3,4-dihydropyrimidin-2(1H)-ones, Indian J. Chem., Sect. B: Org. Chem. Incl. Med. Chem., 2009, 48, 1738 Search PubMed.
- N. Foroughifar, A. Mobinikhaledi and H. F. Jirandehi, Synthesis of some Biginelli compounds in solvent medium using a photochemistry method, Phosphorus, Sulfur Silicon Relat. Elem., 2003, 178, 495 CrossRef.
- F. Mohamadpour, M. Bakhtiari, D. Khalilzadeh and A. Salimi, Saccharin: a green, economical and efficient catalyst for the one-pot, multi-component synthesis of 3,4-dihydropyrimidin-2-(1H)-one derivatives and 1H-pyrazolo[1,2-b]phthalazine-5,10-dione derivatives and substituted dihydro-2-oxypyrrole, J. Iran. Chem. Soc., 2016, 13, 1549 CrossRef CAS.
- Z. T. Wang, S. C. Wang and L. W. Xu, Polymer-supported ionic-liquid-catalysed synthesis of 1,2,3,4-tetrahydro-2-oxopyrimidine-5-carboxylates via Biginelli reaction, Helv. Chim. Acta, 2005, 88, 986 CrossRef CAS.
- K. Niknam and N. Daneshvar, H4SiW12O40·xH2O as a new catalyst for the synthesis of 3,4-dihydropyrimidin-2(1H)-one, Heterocycles, 2007, 71, 373 CrossRef CAS.
- T. C. Braga, A. M. Silva, F. M. Oliveira, C. R. Pereira and L. A. Ferreira, Ionic liquid-assisted synthesis of dihydropyrimidin(thio)one Biginelli adducts and investigation of their mechanism of urease inhibition, New J. Chem., 2019, 43, 15187 RSC.
- H. Biklarian, F. K. Behbahani and Z. Fakhroueian, 22% Co/CeO2–ZrO2-catalysed synthesis of 1,2,3,4-tetrahydro-2-pyrimidinones and –thiones, Lett. Org. Chem., 2012, 9, 580 Search PubMed.
- P. K. Sahoo, A. Bose and P. Mal, Solvent-free ball-milling Biginelli reaction by subcomponent synthesis, Eur. J. Org Chem., 2015, 2015, 6994 CrossRef CAS.
- A. Poursattar Marjani, F. Asadzadeh and A. Danandeh Asl, Novel core–shell magnetic nanoparticles@zeolitic imidazolate with glycerol–nickel for the synthesis of dihydropyrimidinones, Appl. Organomet. Chem., 2023, 37, e7260 CrossRef CAS.
- J. Senapathi, R. Kumar, P. Singh, V. Reddy and A. Varma, Design, synthesis, and antiviral activity of 1,2,3,4-tetrahydropyrimidine derivatives acting as novel entry inhibitors to target the “Phe43 cavity” of HIV-1 gp120, Bioorg. Med. Chem., 2021, 52, 116526 Search PubMed.
- D. Azarifar, Y. Abbasi and O. Badalkhani, Sulfonic acid-functionalised titanomagnetite nanoparticles as recyclable heterogeneous acid catalyst for one-pot solvent-free synthesis of 3,4-dihydropyrimidin-2(1H)-ones/thiones, J. Iran. Chem. Soc., 2016, 13, 2029 CrossRef CAS.
- N. A. Abdel-Riheem, M. H. El-Banhawi, A. H. El-Syn, M. M. Abdel-Mottaleb and R. Fathy, 1,3,4-Thia- and -selenadiazole and 1,2,4-triazolo[4,3-a]pyrimidine derivatives from hydrazonoyl halides, Heteroat. Chem., 2003, 14, 421 CrossRef CAS.
- A. Rahmatpour and G. Donyapeyma, Titanium tetrachloride immobilised on cross-linked poly(N-vinyl-2-pyrrolidone) as a recyclable heterogeneous catalyst for one-pot three-component synthesis of 3,4-dihydropyrimidin-2(1H)-ones/thiones, Synth. Commun., 2022, 52, 678 CrossRef CAS.
- N. Mohammadian and B. Akhlaghinia, Punica granatum peel: an organocatalyst for green and rapid synthesis of 3,4-dihydropyrimidin-2(1H)-ones/thiones under solvent-free condition, Res. Chem. Intermed., 2017, 43, 3325 Search PubMed.
- F. Shirini, M. A. Zolfigol, J. Albadi, T. F. Rastegar and S. Sheik Mansoor, Melamine trisulfonic acid: a new, efficient and recyclable catalyst for the synthesis of 3,4-dihydropyrimidin-2(1H)-ones/thiones in the absence of solvent, Chin. Chem. Lett., 2011, 22, 318 Search PubMed.
- M. Hajjami and E. Ghiasbeygi, First catalyst- and solvent-free synthesis of 3,4-dihydropyrimidin-2(1H)-ones and –thiones, Russ. J. Org. Chem., 2016, 52, 429 CrossRef CAS.
- D. L. da Silva, R. F. da Silva, A. M. Freitas, P. J. da Silva and L. M. Silva, Free radical scavenging and antiproliferative properties of Biginelli adducts, Bioorg. Med. Chem., 2012, 20, 2645 CrossRef CAS PubMed.
- H. Moghanian, M. Rezazadeh, A. R. Ghiasinejad and S. Sadeghzadeh, Bis(p-sulfoanilino)triazine-functionalised silica-coated magnetite nanoparticles as an efficient and magnetically reusable nano-catalyst for Biginelli-type reaction, Res. Chem. Intermed., 2018, 44, 4083 CrossRef CAS.
- C. K. Khatri, S. M. Potadar and G. U. Chaturbhuj, A reactant-promoted solvent-free synthesis of 3,4-dihydropyrimidin-2(1H)-thione analogues using ammonium thiocyanate, Tetrahedron Lett., 2017, 58, 1778 CrossRef CAS.
- F. A. Attaby, M. M. Ramla and T. Harukuni, Synthesis and inhibitory activity against Epstein–Barr virus of some new 1,2,3,4-tetrahydropyrimidine-2-thiones, Phosphorus, Sulfur Silicon, 2008, 183, 2956 CrossRef CAS.
- E. Kolvari, N. Koukabi, M. M. Hosseini, M. Vahidian and E. Ghobadi, Nano-ZrO2 sulfuric acid: a heterogeneous solid acid nanocatalyst for Biginelli reaction under solvent-free conditions, RSC Adv., 2016, 6, 7419 RSC.
- K. Selvakumar, T. Shanmugaprabha, M. Kumaresan and P. Sami, Heteropoly acid supported on activated natural clay-catalyzed synthesis of 3,4-dihydropyrimidinones/thiones through Biginelli reaction under solvent-free conditions, Synth. Commun., 2018, 48, 223 CrossRef CAS.
- S. Mekki, Y. Laichi, F. Benourrad, N. Lagha, R. Chebout, K. Bachari and S. Saidi-Besbes, Copper triazole complex supported on Fe3O4@SiO2 nanoparticles as eco-friendly nanocatalysts in solvent-free Biginelli reaction, Appl. Organomet. Chem., 2023, 37, e7103 CrossRef CAS.
- R. J. Kalbasi, A. R. Massah and B. Daneshvarnejad, Preparation and characterization of bentonite/PS-SO3H nanocomposites as an efficient acid catalyst for the Biginelli reaction, Appl. Clay Sci., 2012, 55, 1 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |