Open Access Article
Open Access ArticleDFT study of atmospheric characteristics of CF3SO2F: the fungibility of the insulation gas of SF6
Wei Liua,
Yuanyuan Zhengb,
Yuanyuan Cuib,
Dan Lib,
Qinqin Yuanb,
Kun Wang*b and
Longjiu Cheng *b
*b
aState Grid Anhui Electric Power Co., Ltd., Electric Power Research Institute, Anhui 230601, China
bDepartment of Chemistry, Key Laboratory of Functional Inorganic Materials of Anhui Province, Anhui University, Hefei, Anhui 230601, P. R. China. E-mail: wangkun@ahu.edu.cn; clj@ustc.edu
First published on 4th February 2026
Abstract
Due to the significant greenhouse effect of SF6, CF3SO2F has emerged as a potential alternative that meets the requirements for insulation gases in high-voltage electrical equipment. Herein, the atmospheric lifetime and global warming potential (GWP) of CF3SO2F were evaluated based on its interactions with hydroxide radicals (·OH) using theoretical calculations. By employing the Monte Carlo method, we constructed molecular structures of SF6–H2O and CF3SO2F–H2O as mixed-gas systems to simulate the dissociation of these insulation gases under atmospheric conditions. The adversative efficiency (RE) of CF3SO2F was determined to be 0.177 W (m2 ppbv)−1, with an atmospheric lifetime of 52.02 years and a GWP of 4320. The reactive models, developed using density functional theory (DFT) and Car–Parrinello molecular dynamics (CPMD), not only enable the determination of the dissociation pathway in the atmosphere, but also provide detailed insights into the interactions with ·OH based on the overall dynamic behaviour.
1. Introduction
SF6 plays an essential role in the power industry due to its excellent dielectric strength, heat transfer capability, and arc-quenching ability.1 Toxic decomposition products can be formed from SF6 under electric arc conditions, despite the gas itself being non-toxic.2 SF6 is a potent greenhouse gas with a 100 year global warming potential (GWP100) of 23![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500, equivalent to 23
500, equivalent to 23![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 kg CO2 per kg of SF6, and an atmospheric lifetime of approximately 3200 years.3 Global emissions of SF6 have been increasing, reaching 9040 tons in 2018, with the power industry accounting for 80% of this total. Consequently, atmospheric concentrations of SF6 have risen from 0.66 parts per trillion (ppt) by volume in 1978 to over 11 ppt in 2022.4,5 Faced with this rapid accumulation and its permanent climate impact, aggressively implementing mitigation strategies and switching to sustainable alternatives is an urgent priority. Over the past decades, great efforts have been devoted to identifying such replacements, and several potential alternatives were reported, including C5F10O and C3F7CN.6,7 However, these gases often suffer from drawbacks, such as high liquefaction temperatures and potential toxicity. Furthermore, despite having potentially superior dielectric strength and lower GWP values than SF6, their GWP values are still relatively high, limiting their applicability as environmentally friendly insulation media.8,9
500 kg CO2 per kg of SF6, and an atmospheric lifetime of approximately 3200 years.3 Global emissions of SF6 have been increasing, reaching 9040 tons in 2018, with the power industry accounting for 80% of this total. Consequently, atmospheric concentrations of SF6 have risen from 0.66 parts per trillion (ppt) by volume in 1978 to over 11 ppt in 2022.4,5 Faced with this rapid accumulation and its permanent climate impact, aggressively implementing mitigation strategies and switching to sustainable alternatives is an urgent priority. Over the past decades, great efforts have been devoted to identifying such replacements, and several potential alternatives were reported, including C5F10O and C3F7CN.6,7 However, these gases often suffer from drawbacks, such as high liquefaction temperatures and potential toxicity. Furthermore, despite having potentially superior dielectric strength and lower GWP values than SF6, their GWP values are still relatively high, limiting their applicability as environmentally friendly insulation media.8,9
CF3SO2F has attracted considerable attention as a promising replacement for SF6 due to its excellent insulation performance, low boiling point of −22 °C, and low GWP value.10–15 Studies of field-dependent electron–molecule capture rates suggest that CF3SO2F possesses a higher dielectric strength than SF6.9 Both theoretical and experimental investigations have confirmed that the thermal stability of CF3SO2F is comparable to that of SF6.15 Furthermore, CF3SO2F has been demonstrated to exhibit a short atmospheric lifetime and a minimal contribution to smog formation,16 along with a higher breakdown voltage (32 kV) than that of SF6 (26 kV).17 These properties collectively suggest that it is more environmentally friendly and suitable for replacing SF6 in electrical equipment. Although its boiling point is higher than that of SF6 (−63.8 °C), its strong dielectric strength and high thermal stability make it applicable in most geographical regions.12 Zhang et al. reported that CF3SO2F mainly decomposes via C–S bond cleavage, yielding CF3 and SO2F radicals. These radicals can further react with free F radicals to form CF4 and SO2. Alternatively, decomposition can proceed through CF3 migration and memorization, forming the more stable intermediate CF3OSFO, which subsequently decomposes into CF2O and SOF2.18 Wang's team has conducted an in-depth analysis of CF3SO2F decomposition products.19
The atmospheric behavior and ultimate fate of an insulation gas are directly determined by its reactivity under atmospheric conditions. The most reliable method for estimating the lifetime of a trace gas involves chemical transport models (CTMs), which require accurate rate constants for reactions with ·OH as critical input parameters.13 For chemically reactive trace gases, atmospheric degradation by ·OH is often the dominant process governing their global atmospheric lifetime. Therefore, to evaluate the environmental impact of the insulation gas SF6 and its alternatives, it is essential to establish a systematic model that simulates atmospheric dissociation based on structure–property relationships. Constructing such mixed-gas models is necessary to fully elucidate the atmospheric interaction mechanisms between insulation gases and ·OH.
In this work, we first compared the electronic structures and RE of SF6 and CF3SO2F based on their optimized molecular geometries. Given that ·OH plays a key role as a reactive species interacting with greenhouse gases in the atmosphere, a co-crystal structure of CF3SO2F and H2O with a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 molar ratio was constructed using the MC method. The complete set of four dissociation pathways of CF3SO2F with ·OH in the atmosphere was revealed through Car–Parrinello molecular dynamics (CPMD) simulations. Based on the analysis of the reaction pathways, reaction rates, atmospheric lifetime, and GWP were calculated. The combined computational framework of density functional theory (DFT) and CPMD established in this study provides a reliable basis for systematically evaluating the atmospheric behavior of these gases and screening potential alternatives to SF6.
1 molar ratio was constructed using the MC method. The complete set of four dissociation pathways of CF3SO2F with ·OH in the atmosphere was revealed through Car–Parrinello molecular dynamics (CPMD) simulations. Based on the analysis of the reaction pathways, reaction rates, atmospheric lifetime, and GWP were calculated. The combined computational framework of density functional theory (DFT) and CPMD established in this study provides a reliable basis for systematically evaluating the atmospheric behavior of these gases and screening potential alternatives to SF6.
2. Computational details
To confirm the computational method, bond lengths of the optimized CF3SO2F under several DFT methods were compared with CCSD(T). Benchmark calculations show that the bond lengths values of C–F, C–S, S![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O and S–F at the M062X/def2tzvp20,21 level of theory are the closest to those at the CCSD(T)/aug-cc-pVTZ method. Hence, geometric and electronic structures of SF6, CF3SO2F and H2O were investigated at the M062X/def2tzvp level of theory, using the Gaussian 16 package.22 The original and optimized coordinates of the initial structures of SF6, CF3SO2F and H2O are presented in Tables S1 and S2, respectively.The Wiberg bond indices (WBI), atomic charges, electrostatic potentials, and polarizabilities were obtained in Multiwfn (Table 1).23
O and S–F at the M062X/def2tzvp20,21 level of theory are the closest to those at the CCSD(T)/aug-cc-pVTZ method. Hence, geometric and electronic structures of SF6, CF3SO2F and H2O were investigated at the M062X/def2tzvp level of theory, using the Gaussian 16 package.22 The original and optimized coordinates of the initial structures of SF6, CF3SO2F and H2O are presented in Tables S1 and S2, respectively.The Wiberg bond indices (WBI), atomic charges, electrostatic potentials, and polarizabilities were obtained in Multiwfn (Table 1).23
| C–F | C–S | S![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O O |
S–F | |
|---|---|---|---|---|
| B3LYP/def2tzvp | 1.33 | 1.87 | 1.42 | 1.57 |
| BP86/def2tzvp | 1.33 | 1.87 | 1.42 | 1.57 |
| WB97XD/def2tzvp | 1.32 | 1.86 | 1.41 | 1.55 |
| M06-2X/def2tzvp | 1.32 | 1.84 | 1.41 | 1.55 |
| MP2/aug-cc-pVTZ | 1.32 | 1.85 | 1.43 | 1.58 |
| CCSD(T)/aug-cc-pVTZ | 1.32 | 1.85 | 1.41 | 1.55 |
To obtain the reasonable interaction pathways for the dissociation of SF6 and CF3SO2F with hydroxyl radial in atmosphere. We constructed the crystal (SF6, SF6–H2O, CF3SO2F, and CF3SO2F–H2O) using the Monte Carlo (MC) method in Materials Studio (MS) software.24 Prior to prediction, the Universal Force Field (UFF)25,26 in the Forcite module was applied to assign force field parameters (Fig. S1). Subsequently, the Polymorph module27 was used to predict the periodic crystal structures of the molecules (Fig. S2). And the optimized coordinates of the crystal structures of SF6, SF6–H2O, CF3SO2F, and CF3SO2F–H2O are presented in Table S3.
According to Berskey's theory,28 88.6% of organic molecular crystal structures belong to the space groups P21/c, P212121, P1, P21, and C2/c in Table S4. The final structures with the lowest total energies are chosen among the five different space groups. And the final coordinates of the lowest-energy structures of SF6, SF6–H2O, CF3SO2F, and CF3SO2F–H2O are presented in Table S5. Based on these lowest-energy structures, the predicted crystal structures of SF6, SF6–H2O, CF3SO2F, and CF3SO2F–H2O were further optimized using the CASTEP module29 in MS with the GGA-PBE functional.30 Ultrasoft pseudopotentials were used to describe the valence states of elements.31 Convergence tests for the plane-wave cutoff energy and k-points were performed. The cutoff energies for SF6, SF6–H2O, CF3SO2F, and CF3SO2F–H2O were set to 300, 400, 500, and 600 eV, respectively. The Brillouin zones32 were sampled using k-point grids of 2 × 1 × 2, 1 × 1 × 1, 3 × 1 × 1, and 1 × 3 × 5, respectively. All the test results are summarized in Table S6.
Based on the optimized crystal structures, CPMD simulations were performed. We first carried out the convergence of the wave function and the structural optimization under the NVT ensemble,33 ensuring that the ion temperature, the virtual kinetic energy of electrons, and the total electronic energy were all converged (Fig. S3). Subsequently, the four systems were directly heated to 2500 K and maintained at this temperature for 5 ps to allow the system to reach equilibrium. Then the temperature has been continuously increased to 3000 K for another 21 ps. The final temperature is converged at 3000 K. The total length of 26 ps (260![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 steps) is enough for obtaining the interaction pathways.34,35
000 steps) is enough for obtaining the interaction pathways.34,35
Finally, the active energies in the typical four dissociation pathways are obtained based on the CPMD trajectories. kOH36 is the rate constant of reaction between the greenhouse gas and ·OH, which has been obtained by eqn (1)
 | (1) |
 | (2) |
3. Results and discussion
3.1 The electronic structures of gas phase
The optimized structures along with the corresponding charge distribution maps are summarized in Fig. 1. SF6 is the most common insulation gas (Fig. 1a) used in high-voltage equipment. SF6 adopts equivalent sp3d2 hybridization to bond with six fluorine atoms, resulting in an S–F bond length of 1.59 Å. The candidate insulation gas CF3SO2F (Fig. 1b) has a highly polarized S![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bond, whose bond dissociation energy (BDE) of 163.91 kcal mol−1 exceeds that of the S–F bond in SF6 (110.23 kcal mol−1). The S–C bond in CF3SO2F has the lowest BDE, making it the weakest bond and the most probable initial site for bond cleavage during dissociation. As shown in Fig. 1c and d, the electrostatic potential (ESP) values are labeled. SF6, as a highly symmetrical molecule, exhibits a relatively uniform electrostatic potential distribution with minimal variation. In contrast, CF3SO2F has a larger electrostatic potential gradient. A uniform electrostatic potential distribution and a low electrostatic potential gradient contribute to improved insulation performance. Therefore, CF3SO2F may be slightly inferior in terms of insulation performance due to its significant variation in electrostatic potential.
O bond, whose bond dissociation energy (BDE) of 163.91 kcal mol−1 exceeds that of the S–F bond in SF6 (110.23 kcal mol−1). The S–C bond in CF3SO2F has the lowest BDE, making it the weakest bond and the most probable initial site for bond cleavage during dissociation. As shown in Fig. 1c and d, the electrostatic potential (ESP) values are labeled. SF6, as a highly symmetrical molecule, exhibits a relatively uniform electrostatic potential distribution with minimal variation. In contrast, CF3SO2F has a larger electrostatic potential gradient. A uniform electrostatic potential distribution and a low electrostatic potential gradient contribute to improved insulation performance. Therefore, CF3SO2F may be slightly inferior in terms of insulation performance due to its significant variation in electrostatic potential.
As the insulation gas, the most important factor is the molecular stability in the arc extinction, which is determined by the electronic structural properties. In this paper, the stability is evaluated through multiple aspects, including bond dissociation energy, HOMO–LUMO gap, and vertical ionization energy.40–42 The electronic structure properties of SF6 and CF3SO2F are presented in Table 2. The HOMO–LUMO gap of SF6 is 15.24 eV. Its VIE and EA are 367.76 kcal mol−1 and 1.67 eV, respectively. These values indicate the high stability of SF6 in high-voltage electric fields regarding its ability to accept and donate electrons. CF3SO2F has a molecular volume of 104.74 Å3, which is larger than that of SF6 (87.53 Å3). A larger molecular size is an effective factor in improving electron absorption efficiency. In CF3SO2F, the S![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bond is the most polarized, with a WBI of 2.51. This bond is more stable than the S–F bond in SF6, which has a WBI of 0.72. The VIE and EA of CF3SO2F are 310.64 kcal mol−1 and 2.33 eV, respectively. Compared with SF6, CF3SO2F exhibits stronger electron affinity and a larger molecular dipole moment, making it an outstanding insulation gas in the arc extinction process, despite having a lower VIE.
O bond is the most polarized, with a WBI of 2.51. This bond is more stable than the S–F bond in SF6, which has a WBI of 0.72. The VIE and EA of CF3SO2F are 310.64 kcal mol−1 and 2.33 eV, respectively. Compared with SF6, CF3SO2F exhibits stronger electron affinity and a larger molecular dipole moment, making it an outstanding insulation gas in the arc extinction process, despite having a lower VIE.
| Gas | V | EH–L | VIE | EA | DM | WBI |
|---|---|---|---|---|---|---|
| SF6 | 87.53 | 15.24 | 367.76 | 1.67 | 0 | S–F: 0.72 |
| CF3SO2F | 104.74 | 13.4 | 310.64 | 2.33 | 1.46 | S![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O: 2.51; S–F: 1.54 O: 2.51; S–F: 1.54 |
| S–C: 0.92; C–F: 1.85 |
3.2 The RE of insulation gases
RE is a physical quantity used to measure the extent of the impact of changes in the concentration of gases in the atmosphere on radiative forcing, and its variations can directly affect the magnitude of radiative forcing.38 The radiative forcing (RF) resulting from variations in greenhouse gas concentrations is a crucial concept in climate science, which indicates the magnitude and signs of the drivers of climate change.43 RE can be accurately predicted through the formula (3) provided by Shine and Myhre,44
 | (3) |
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching vibration appeared in the range of 1460–1360 cm−1, CF3 symmetric bending vibration at 780–760 cm−1, and S–F stretching vibration at 880–850 cm−1; for SF6, the symmetric F–S–F stretching vibration was observed at 810–790 cm−1, with degenerate bending vibrations at 640–620 cm−1. Then we selected the Raman wavenumber corresponding to the infrared intensity value (data shown in Table 3), and retrieved the corresponding Fi value from the data calculated by Pinnock et al. σi is the high-resolution absorption cross-section data of the target gas in the band (500–3000 cm−1), with a value of 0.015 cm2−molecule−1 obtained from the HITRAN database. This value has been calibrated using vscale = 29.4 + 0.9475Vcal. where Vcal is the theoretical value obtained from DFT calculations and Vscal represents the final calibrated value used for GWP estimation. For Δυ, by combining the instantaneous radiative forcing (IRF) calculated using the high-resolution line-by-line model (OLBL IRF) and the STA adjustment factor (RF/IRF ratio) calculated by the narrow-band model (NBM), we obtained Δυ equals to 1 cm−1.47,48 Compared with the RE of SF6,49 which is 0.57 W (m2 ppbv)−1, CF3SO2F has an RE value of 0.177 W (m2 ppbv)−1, indicating significantly lower RE.
O stretching vibration appeared in the range of 1460–1360 cm−1, CF3 symmetric bending vibration at 780–760 cm−1, and S–F stretching vibration at 880–850 cm−1; for SF6, the symmetric F–S–F stretching vibration was observed at 810–790 cm−1, with degenerate bending vibrations at 640–620 cm−1. Then we selected the Raman wavenumber corresponding to the infrared intensity value (data shown in Table 3), and retrieved the corresponding Fi value from the data calculated by Pinnock et al. σi is the high-resolution absorption cross-section data of the target gas in the band (500–3000 cm−1), with a value of 0.015 cm2−molecule−1 obtained from the HITRAN database. This value has been calibrated using vscale = 29.4 + 0.9475Vcal. where Vcal is the theoretical value obtained from DFT calculations and Vscal represents the final calibrated value used for GWP estimation. For Δυ, by combining the instantaneous radiative forcing (IRF) calculated using the high-resolution line-by-line model (OLBL IRF) and the STA adjustment factor (RF/IRF ratio) calculated by the narrow-band model (NBM), we obtained Δυ equals to 1 cm−1.47,48 Compared with the RE of SF6,49 which is 0.57 W (m2 ppbv)−1, CF3SO2F has an RE value of 0.177 W (m2 ppbv)−1, indicating significantly lower RE.
| ε | Fi [W m−2 cm (cm2 molecule−1)−1] | σi (cm molecule−1) |
|---|---|---|
| 1525 | 0.0341 | 0.015 |
| 1319 | 0.3335 | 0.015 |
| 1303 | 0.207 | 0.015 |
| 1189 | 1.52 | 0.015 |
| 869 | 3.135 | 0.015 |
| 796 | 2.74 | 0.015 |
| 630 | 0.6145 | 0.015 |
| 574 | 2.13 | 0.015 |
| 500 | 1.67 | 0.015 |
3.3 Optimized structures in simulated atmospheric environments
A crucial factor for the continuous dissociation of insulation gas is the interactions between insulation gas and ·OH.50 Since H2O is the source of ·OH in the atmospheric environment, both SF6 and CF3SO2F are mixed with H2O to acquire their static reactions. Based on the predicted structures with the lowest total energy based on Monte-Carlo methods in polymorph code, the four structures are further optimized in CASTEP with GGA-PBE method in Fig. 3. | ||
| Fig. 3 The two predicted crystal structures. Top left is SF6(a), Top right is SF6–H2O(b), lower left is CF3SO2F (c), lower right is CF3SO2F–H2O (d). | ||
The optimized structures with the corresponding DOS maps are shown in Fig. 4. Using the HSE06 hybrid functional, we calculated the band gaps of the electronic structures for SF6 and SF6–H2O. The crystalline SF6 adopts P21-c space group with the band gap of 7.56 eV, where the crystal parameters are a = 4.59 Å, b = 8.69 Å, c = 9.12 Å, α = 90.0°, β = 120.9°, γ = 90.0° (Fig. 4a). As the mixed model of the crystalline SF6–H2O in Fig. 4b, the band gap is decreased to 4.43 eV compared with SF6. The crystal parameters are a = b = 4.65 Å, c = 23.42 Å, α = 113.63°, β = 120.9°, γ = 67.5° with the space group of C2-c. The density of states of SF6 and SF6–H2O are compared in Fig. 4c and d. S 3s and 3p states occupy the lowest energy area in both structures. In the area of −5 eV to Fermi level, the most active electron states are contributed from O 2p and H 1 s states. In the structure of SF6–H2O, we notice that the occupied energy level of both S 3s/3p and F 2p states is decreased. However, the energy level of O 2p and H 1s states are both centered around Fermi level, indicating that the doped H2O increase the reactivity of SF6 caused by the highly active O 2p and H 1s electronic states.
 | ||
| Fig. 4 The predicted crystal SF6 (a) and SF6–H2O (b), and the corresponding DOS maps for SF6 (c) and SF6–H2O (d). | ||
As for CF3SO2F, corresponding band gaps were obtained through independent HSE06 calculations of the electronic structures; the predicted crystal CF3SO2F adopts P21 space group with the band gap of 6.56 eV. The crystal parameters are a = 15.39 Å, b = 25.92 Å, c = 18.54 Å, α = 90.0°, β = 100.61°, γ = 90° (Fig. 5a), where the most active electron states are composed of C 2p, F 2p and O 2p states closed to the Fermi level of DOS map in Fig. 5b. Crystal CF3SO2F–H2O adopts P1 space group with the band gap of 2.47 eV and the crystal parameters of a = 4.59 Å, b = 8.69 Å, c = 9.12 Å, α = 90.0°, β = 120.9°, γ = 90.0° (Fig. 5c). Based on the DOS map in Fig. 5d, with introducing H2O, the high active O 2p and H 1s electronic states occupy the area closed to the Fermi level. Compared with Fig. 5c, the energy level of F 2p, S 3p, and C 2p states of CF3SO2F are decreased to overlap with the occupied energy area of O 2p and H 1s states, indicating that the doped ·OH groups (molecular water) increase the reactivity of CF3SO2F with forming CF3SO2F–H2O interaction. Moreover, compared with SF6–H2O, more kinds of active electronic states around Fermi level indicate the possible high reactivity of CF3SO2F–H2O, which is the determining factor for the dissociation of insulation gases in air.
 | ||
| Fig. 5 The predicted crystal CF3SO2F (a) and CF3SO2F–H2O (b), and the corresponding DOS maps for CF3SO2F (c) and CF3SO2F–H2O (d). | ||
Based on the optimized solid-state crystal structures, we analyzed the electron distribution by comparing the charge density difference shown in Fig. 6, where the green part represents the electron-rich area and the red part is the electron-deficient area. The charge density difference plot reveals electron redistribution between atoms during chemical bond formation. The charge density difference (Δρ = ρ(AB) – ρ(A) – ρ(B)) quantifies electron redistribution during chemical bonding, visualized through isosurface plots that reveal regions of electron accumulation (Δρ > 0) and depletion (Δρ < 0).51 Without introducing H2O, electrons in SF6 are primarily distributed on the surface of F atoms, with charge transferring from S to F. In CF3SO2F, the S![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bonds exhibit the highest polarizability and appear to have the strongest electron-withdrawing effect, resulting in a polarized structure with charge transfer from the C–F bond to the S
O bonds exhibit the highest polarizability and appear to have the strongest electron-withdrawing effect, resulting in a polarized structure with charge transfer from the C–F bond to the S![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bonds. With the introduction of H2O, charge from F atoms in SF6 and O atom in CF3SO2F transfers to H, forming hydrogen bonds (F⋯H–O and O⋯H–O, respectively). This is consistent with the results from DOS analysis, which indicates that H2O enhances the reactivity of insulation gas due to the high active O 2p and H 1s electronic states, leading to the continuous dissociation. Furthermore, the stronger interaction between the insulation gas and water facilitates dissociation in atmospheric environments. To quantify this, we compare the interaction between water and the insulation gases using the reaction: SF6/CF3SO2F + H2O → SF6–H2O/CF3SO2F–H2O. For SF6–H2O, the interaction strength between SF6 and H2O is 0.22 eV, which is lower than that of CF3SO2F–H2O (0.25 eV), indicating the competitive structure of S–F⋯H–OH is easier to form for CF3SO2F in the atmosphere.
O bonds. With the introduction of H2O, charge from F atoms in SF6 and O atom in CF3SO2F transfers to H, forming hydrogen bonds (F⋯H–O and O⋯H–O, respectively). This is consistent with the results from DOS analysis, which indicates that H2O enhances the reactivity of insulation gas due to the high active O 2p and H 1s electronic states, leading to the continuous dissociation. Furthermore, the stronger interaction between the insulation gas and water facilitates dissociation in atmospheric environments. To quantify this, we compare the interaction between water and the insulation gases using the reaction: SF6/CF3SO2F + H2O → SF6–H2O/CF3SO2F–H2O. For SF6–H2O, the interaction strength between SF6 and H2O is 0.22 eV, which is lower than that of CF3SO2F–H2O (0.25 eV), indicating the competitive structure of S–F⋯H–OH is easier to form for CF3SO2F in the atmosphere.
3.4 Atmospheric behaviours of CF3SO2F based on CPMD simulation
To understand the dissociation of CF3SO2F in atmospheric environments, we employed CPMD to simulate the dynamic pathways between CF3SO2F and ·OH, the latter being generated from H2O in the simulation.33 We first determined the decomposition pathways of pure CF3SO2F in Scheme 1. For comparison, decomposition pathways of pure SF6 in Scheme S1. To accelerate the decomposition, we set the temperature to 3000 K in equilibrium systems to observe the trajectories. This approach is also motivated by the fact that protective gases in high-voltage insulation equipment undergo arc discharge, where central temperatures can reach 5000 to 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 K.52,53 Using elevated temperatures thus helps elucidate dissociation mechanisms under these extreme conditions and continuous atmospheric decomposition pathways. In the case of CF3SO2F, no cleavage of the S
000 K.52,53 Using elevated temperatures thus helps elucidate dissociation mechanisms under these extreme conditions and continuous atmospheric decomposition pathways. In the case of CF3SO2F, no cleavage of the S![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bonds was observed. Instead, CF3· and fluorine radicals dissociated sequentially via breaking of the S–C bonds, consistent with the BDE results in Fig. 1, which identify the S–C bond as the weakest in CF3SO2F. At ultrahigh temperatures, the generated active radicals can recombine, potentially forming products such as SO2 or CF4, as outlined in Scheme 1.
O bonds was observed. Instead, CF3· and fluorine radicals dissociated sequentially via breaking of the S–C bonds, consistent with the BDE results in Fig. 1, which identify the S–C bond as the weakest in CF3SO2F. At ultrahigh temperatures, the generated active radicals can recombine, potentially forming products such as SO2 or CF4, as outlined in Scheme 1.
 | ||
| Scheme 1 The summarized decomposition reaction (in the box) with the corresponding pathway of pure CF3SO2F at 3000 K from the initial decomposition of 6.72 ps. | ||
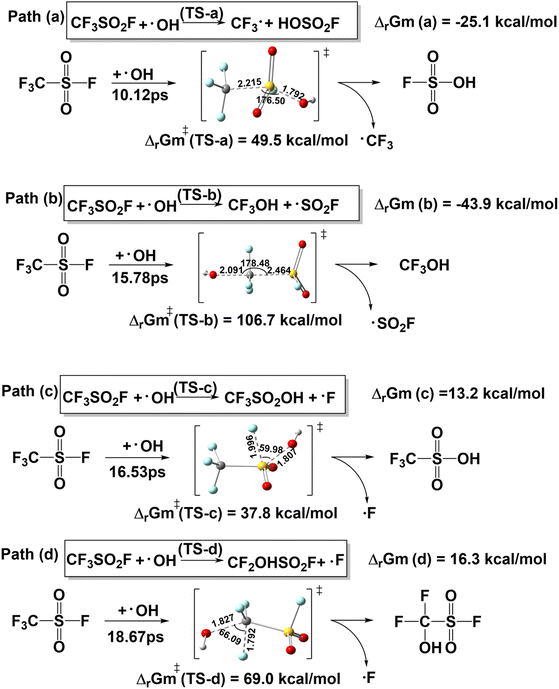
Given that the primary process for the removal of greenhouse gases involves their reaction with tropospheric ·OH,50 the reaction between CF3SO2F and ·OH were analyzed via four distinct pathways based on the results of CPMD, denoted as path (a) to path (d) in Scheme 2. It was reported that the precision of energy and rate constants can be achieved to one decimal place.54 Therefore, the calculated energies and reaction rate constants are rounded to one significant figure. The atomic coordinates of the transition state structure are presented in Table S7. And the reaction energy barrier was comput ed as the difference between the transition state and reactant energies, while the Gibbs free energy change (ΔGm) was derived from the energy difference between products and reactants, which is in Table 4. These pathways are consistent with previously reported results from designed reactions.55,56 Path (a) is a nucleophilic substitution reaction in which ·OH, acting as a highly reactive electrophilic reagent, attacks the central sulfur atom. The transition state (TS-a) is reached at 10.12 ps, leading to the formation of HOSO2F and CF3·. This pathway crosses an energy barrier of 49.5 kcal mol−1 and releases 25.1 kcal mol−1 of energy. In Path (b), ·OH attacks the electron-deficient carbon atom in the CF3SO2F molecule, resulting in cleavage of the S–C bond via transition state (TS-b) at 15.78 ps. This step produces CF3OH and ·SO2F, with an energy barrier of 106.7 kcal mol−1 and a Gibbs free energy change (ΔG) of 43.9 kcal mol−1. In paths (c) and (d), F· are generated, but the attack sites of the ·OH are different. Although paths (c) and (d) both generate F·, their mechanisms differ in the site where the ·OH attacks. In path (c), the reaction proceeds through transition state (TS-c) at 16.53 ps, yielding CF3SO2OH and F·. The energy barrier is 37.6 kcal mol−1, and the Gibbs free energy of reaction is 13.2 kcal mol−1. Path (d) proceeds through transition state (TS-d) at 18.67 ps, forming CF2OHSO2F and F·, with an energy barrier of 69.0 kcal mol−1 and Gibbs free energy of reaction of 16.3 kcal mol−1. For comparison, the reaction between SF6 and ·OH were analyzed in Scheme S2, and the Gibbs free energy change was showed in Table S8.
| Pathways | ΔrGm | ΔrGm≠ | k·OH | τ |
|---|---|---|---|---|
| Path (a) | −25.1 | 49.5 | 2.9 × 10−24 | 2.28 × 1010 |
| Path (b) | −43.9 | 106.7 | 3.6 × 10−66 | 1.87 × 1052 |
| Path (c) | 13.2 | 37.8 | 1.3 × 10−15 | 52.02 |
| Path (d) | 16.3 | 69.0 | 1.5 × 10−38 | 4.56 × 1024 |
In the four pathways, the most possible dissociation pathway is path (c), triggered by the bond splitting of the weakest SO2–F bond. The corresponding energy barrier of TS-(c) is the lowest of the four. In the atmosphere, the lifetime of CF3SO2F is dominated by the rate-determining step in the interaction with ·OH, which should be the dissociation path (c) with the lowest energy barrier (37.8 kcal mol−1) of the four, rather than the other paths.
To further assess the applicability of M062X/def2tzvp method, the MP2/def2tzvp theoretical approach was employed to compute Path (c) of the reaction between CF3SO2F and ·OH (see Table S9). The relative energy of the products and energy barrier obtained using the MP2/def2tzvp method are 1.6 kcal mol−1 and 4.9 kcal mol−1 higher than those derived from the M062X/def2tzvp method, respectively. The rate constant (k·OH) calculated at the MP2/def2tzvp level of theory is 3.9 × 10−19 mol−1 cm3 s−1, whereas the k·OH value obtained at the M062X/def2tzvp method level is 1.3 × 10−15 mol−1 cm3 s−1. This discrepancy arises from the intrinsic characteristics of the respective theoretical methods and falls within the normal range of computational deviations. Therefore, the results obtained at the M062X/def2tzvp level of theory are reasonable.
In the troposphere, the primary source of ·OH is water vapor,56 with its concentration significantly lower than that of the latter. Therefore, the reaction between CF3SO2F and H2O also merits consideration. The reaction leading to the formation of CF3SO2OH and HF (Scheme S3) exhibits the lowest energy barrier (57.2 kcal mol−1), which is still higher than that of Path (c). Consequently, reaction between CF3SO2F and H2O proceeds sluggishly, consistent with previous experimental and theoretical studies.57,58 Besides, the reaction rate constant (kH2O) for the reaction between CF3SO2F and H2O is 6.5 × 10−30 mol−1 cm3 s−1,while k·OH for Path (c) is 1.3 × 10−15 mol−1 cm3 s−1, suggesting that the high concentration of water cannot compensate for its deficiency in reaction activity. Hence, CF3SO2F remains low reactivity toward water.
The lifetime of CF3SO2F is 52.02 years, which can be obtained by eqn (2), close to the predicted lifetime of 40 years.59 As a comparison, the lifetime of SF6 in Table S8. The theoretical lifetime of SF6 is 3296 years, which is reasonable with the experimental value of ∼3400 years reported in 2017.60,61 Finally, we have calculated the GWP value based on RE results (0.177 W (m2 ppbv)−1) and lifetime (52.02 years) of CF3SO2F based on eqn (3), which is the most important parameter to evaluate the possibility of application of new insulation gas.
The most widely used metric is the GWP with a 100 year time-horizon (hereafter GWP(100)), which is based on the time-integrated RF due to a pulse emission of a unit mass of gas, normalized by the reference gas CO2, and was introduced in the first assessment report of the Intergovernmental Panel on Climate Change (IPCC).62 Based on eqn (4),
 | (4) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 845 for SF6, while the IPCC reported GWP value for SF6 is 23
845 for SF6, while the IPCC reported GWP value for SF6 is 23![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500. Therefore, the simulation provides a simple and direct method to understand the atmospheric behaviours of insulation gas based on the reasonable trajectories based on DFT method and Ab initio molecular dynamics simulation.
500. Therefore, the simulation provides a simple and direct method to understand the atmospheric behaviours of insulation gas based on the reasonable trajectories based on DFT method and Ab initio molecular dynamics simulation.
3.5 Conclusion
This study provides an effective approach using DFT and CPMD simulations to understand the relationship between the structure and dynamic atmospheric behavior of insulation gases, evaluating CF3SO2F as a potential alternative to SF6. The highly polarized C–F, S–F, and S![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bonds in CF3SO2F effectively absorb electrons during arc extinction, while its stronger interaction with ·OH results in faster dissociation and a shorter atmospheric lifetime (50.02 years) compared to that of SF6 (3296 years). Three dissociation pathways—including synergistic and stepwise mechanisms—were identified, with the rate-determining step involving a synergistic mechanism and an energy barrier of 37.6 kcal mol−1. The GWP of CF3SO2F (4320) is significantly lower than that of SF6 (22
O bonds in CF3SO2F effectively absorb electrons during arc extinction, while its stronger interaction with ·OH results in faster dissociation and a shorter atmospheric lifetime (50.02 years) compared to that of SF6 (3296 years). Three dissociation pathways—including synergistic and stepwise mechanisms—were identified, with the rate-determining step involving a synergistic mechanism and an energy barrier of 37.6 kcal mol−1. The GWP of CF3SO2F (4320) is significantly lower than that of SF6 (22![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 845), indicating that CF3SO2F is a promising and environmentally friendly alternative gas for high-voltage equipment. Based on mixed-gas models and systematic trajectory analysis, this approach offers a robust framework for evaluating the atmospheric behavior of candidate insulation gases and extends its applicability to the selection of protective gases in power equipment.
845), indicating that CF3SO2F is a promising and environmentally friendly alternative gas for high-voltage equipment. Based on mixed-gas models and systematic trajectory analysis, this approach offers a robust framework for evaluating the atmospheric behavior of candidate insulation gases and extends its applicability to the selection of protective gases in power equipment.
Conflicts of interest
The authors declare no competing financial interest.Data availability
The data supporting this article have been included as part of the manuscript. For requests about the original data, please do not hesitate to contact the corresponding author.Supplementary information (SI): structures of molecules and crystals; convergence statements; decomposition pathways; coordinates and parameters of crystals. See DOI: https://doi.org/10.1039/d5ra04357c.
Acknowledgements
The calculations are carried out at the High-Performance Computing Center of Anhui University and Hefei Advanced Computing Center. This work is financed by Anhui Provincial Natural Science Foundation (2208085UD16), the National Natural Science Foundation of China (21701001) and by the Natural Science Research Project of Anhui Province (KJ2020ZD04).References
- Z. Cui, Y. Li, S. Xiao, S. Tian, J. Tang, Y. Hao and X. Zhang, Recent progresses, challenges and proposals on SF6 emission reduction approaches, Sci. Total Environ., 2024, 906, 167347 CrossRef CAS PubMed.
- H. C. Hodge, W. G. Swalbach and H. Rahn, Toxicity Studies of Sulfur Hexafluoride (SF6) in Pneumoperitoneum, J. Am. Pharm. Assoc., 1958, 47, 197–201 CrossRef CAS PubMed.
- P. Billen, B. Maes, M. Larrain and J. Braet, Replacing SF6 in electrical gas-insulated switchgear: technological alternatives and potential life cycle greenhouse gas savings in an EU-28 perspective, RSC Adv., 2020, 10, 12345–12356 Search PubMed.
- H. Jiang, C. Xie, Y. Liu, C. Xiao, W. Zhang, H. Li, B. Long, W. Dong, D. G. Truhlar, X. Yang and J. Am, Criegee Intermediates Significantly Reduce Atmospheric (CF3)2CFCN, Chem. Soc., 2025, 147, 99 Search PubMed.
- C. Man, J. Wang, K. Gao, Y. Li, Y. Zhang and X. Pei, Detection of Transient Decomposition Products of CF3SO2F and C4F7N via OES Under Spark and Glow Discharges, High Volt., 2025, 2397–7264 Search PubMed.
- Y. Kieffel, F. Biquez, D. Vigouroux, P. Ponchon, A. Schlernitzauer, R. Magous, G. Cros and J. G. Owens, Characteristics of g3–an alternative to SF6, CIRED, 2017, 24, 54–57 Search PubMed.
- A. Beroual and A. Haddad, Recent advances in the quest for a new insulation gas with a low impact on the environment to replace sulfur hexafluoride (SF6) gas in high-voltage power network applications, Energies, 2017, 10, 1216 CrossRef.
- Y. Fu, A. Yang, X. Wang, A. B. Murphy, X. Li, D. Liu, Y. Wu and M. Rong, Theoretical study of the neutral decomposition of SF6 in the presence of H2O and O2 in discharges in power equipment, J. Phys. D Appl. Phys., 2016, 49, 385203 Search PubMed.
- Y. Long, L. Guo, Y. Wang, C. Chen, Y. Chen, F. Li and W. Zhou, Electron swarms parameters in CF3SO2F as an alternative gas to SF6, Ind. Eng. Chem. Res., 2020, 59, 11355–11358 CrossRef CAS.
- X. Yu, H. Hou and B. Wang, A priori theoretical model for discovery of environmentally sustainable perfluorinated compounds, J. Phys. Chem. A, 2018, 122(13), 3462–3469 CrossRef CAS PubMed.
- Y. Wang, X. Lin, M. Wang and X. Li, Properties of CF3SO2F under the influence of external electric field: A DFT study, Results Phys., 2023, 45, 106248 CrossRef.
- Y. Wang, Z. Gao, B. Wang, W. Zhou, P. Yu and Y. Luo, Synthesis and dielectric properties of trifluoromethanesulfonyl fluoride: an alternative gas to SF6, Ind. Eng. Chem. Res., 2019, 58, 21913–21920 CrossRef CAS.
- M. P. Sulbaek Andersen, O. J. Nielsen, B. Karpichev, T. J. Wallington and S. P. Sander, Atmospheric chemistry of isoflurane, desflurane, and sevoflurane: kinetics and mechanisms of reactions with chlorine atoms and OH radicals and global warming potentials, J. Phys. Chem. A, 2012, 116(24), 5806–5820 Search PubMed.
- S. Hu, Y. Wang, W. Zhou, R. Qiu, Y. Luo and B. Wang, Dielectric properties of CF3SO2F/N2 and CF3SO2F/CO2 mixtures as a substitute to SF6, Ind. Eng. Chem. Res., 2020, 59, 15796–15804 CrossRef.
- M. Zhang, H. Hou and B. Wang, Mechanistic and kinetic investigations on decomposition of trifluoromethanesulfonyl fluoride in the presence of water vapor and electric field, J. Phys. Chem. A, 2023, 127, 671–684 Search PubMed.
- M. K. Vollmer, B. R. Miller, M. Rigby, S. Reimann, J. Kim, T. S. Rhee and R. F. Weiss, Atmospheric histories and global emissions of the anthropogenic hydrofluorocarbons HFC-365mfc, HFC-245fa, HFC-227ea, and HFC-236fa, J. Geophys. Res., 2011, 116, 2156–2202 Search PubMed.
- A. Beroual, U. Khaled and M.-L. Coulibaly, Experimental Investigation of the Breakdown Voltage of CO2, N2, and SF6 Gases, and CO2–SF6 and N2–SF6 Mixtures under Different Voltage Waveforms 11, 2018, p. 902 Search PubMed.
- S. Papasavva, D. J. Luecken, R. L. Waterland, K. N. Taddonio and S. O. Andersen, Estimated 2017 refrigerant emissions of 2, 3, 3, 3-tetrafluoropropene (HFC-1234yf) in the United States resulting from automobile air conditioning, Environ. Sci. Technol., 2009, 43, 9252–9259 Search PubMed.
- C. Wang, F. Xie, X. Liu, P. Zhang and X. Wang, Adsorption and gas sensing properties of Cr and Mo modified TaS2 for CF3SO2F decomposition products, Comput. Theor. Chem., 2024, 1240, 114838 Search PubMed.
- Y. Zhao and D. G. Truhlar, The M06 suite of density functionals for main group thermochemistry, thermochemical kinetics, noncovalent interactions, excited states, and transition elements: two new functionals and systematic testing of four M06-class functionals and 12 other functionals, Theor. Chem. Acc., 2008, 120, 215–241 Search PubMed.
- F. Weigend and R. Ahlrichs, Balanced basis sets of split valence, triple zeta valence and quadruple zeta valence quality for H to Rn: Design and assessment of accuracy, Phys. Chem. Chem. Phys., 2005, 7, 3297–3305 Search PubMed.
- M. Frisch, G. Trucks, H. Schlegel, G. Scuseria, M. Robb, J. Cheeseman, G. Scalmani, V. Barone, G. Petersson and H. Nakatsuji, Gaussian 16, Gaussian Inc., Wallingford, CT, 2016, vol. 1, p. 572 Search PubMed.
- K. B. Wiberg, Application of the pople-santry-segal CNDO method to the cyclopropylcarbinyl and cyclobutyl cation and to bicyclobutane, Tetrahedron, 1968, 24, 1083–1096 CrossRef CAS.
- R. L. C. Akkermans, N. A. Spenley and S. H. Robertson, Monte Carlo methods in Materials Studio, Mol. Simul., 2013, 39, 14–15 Search PubMed.
- T. Lu and F. Chen, Multiwfn: A multifunctional wavefunction analyzer, J. Comput. Chem., 2012, 33, 580–592 CrossRef CAS PubMed.
- A. K. Rappé, C. J. Casewit, K. Colwell, W. A. Goddard III and W. M. Skiff, UFF, a full periodic table force field for molecular mechanics and molecular dynamics simulations, J. Am. Chem. Soc., 1992, 114, 10024–10035 Search PubMed.
- W. A. Trzciński, S. Cudziło and L. Szymańczyk, Studies of detonation characteristics of aluminum enriched rdx compositions, Propellants, Explos., Pyrotech., 2007, 32, 392–400 Search PubMed.
- P. Zorkii, A. Razumaeva and V. Belsky, The Systematization of Molecular Crystal Structures, Acta Crystallogr. A, 1977, 33, 1001–1004 Search PubMed.
- M. Segall, P. J. Lindan, M. Probert, C. J. Pickard, P. J. Hasnip, S. Clark and M. Payne, First-principles simulation: ideas, illustrations and the CASTEP code, J. Phys. Condens. Matter, 2002, 14, 2717 Search PubMed.
- S. Grimme, J. Antony, S. Ehrlich and H. Krieg, A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu, J. Chem. Phys., 2010, 132, 15 Search PubMed.
- R. Car and M. Parrinello, Unified approach for molecular dynamics and density-functional theory, Phys. Rev. Lett., 1985, 55, 2471 CrossRef CAS PubMed.
- H. J. Monkhorst and J. D. Pack, Special points for Brillouin-zone integrations, Phys. Rev. B, 1976, 13, 5188 Search PubMed.
- S. I. NOSÉ, A molecular dynamics method for simulations in the canonical ensemble, Mol. Phys., 2002, 100, 191–198 Search PubMed.
- J. Crapse, N. Pappireddi, M. Gupta, S. Y. Shvartsman, E. Wieschaus and M. Wühr, Evaluating the Arrhenius equation for developmental processes, Mol. Syst. Biol., 2021, 17, e9895 Search PubMed.
- Q. Guo, N. Zhang, T. Uchimarub, L. Chen, H. Quan and J. Mizukado, Kinetics, products, and mechanism for the reaction of E−CF3CH=CHC2F5 with OH radicals in gas phase and atmospheric implications, Atmos. Environ., 2018, 185, 7–14 Search PubMed.
- V. C. Papadimitriou and J. B. Burkholder, OH radical reaction rate coefficients, infrared spectrum, and global warming potential of (CF3)2CFCH=CHF (HFO-1438ezy(E)), J. Phys. Chem. A, 2016, 120, 6618–6628 Search PubMed.
- A. E. Heathfield, C. Anastasi, A. McCulloch and F. M. Nicolaisen, Integrated infrared absorption coefficients of several partially fluorinated ether compounds: CF3OCF2H, CF27HOCF2H, CH3OCF2CF2H, CH3OCF2CFClH, CH3CH2OCF2CF2H, CF3CH2OCF2CF2H and CH2=CHCH2OCF2CF2H, Atmos. Environ., 1998, 32, 2825–2833 Search PubMed.
- A. E. Heathfield, C. Anastasi, P. Pagsberg and A. McCulloch, Atmospheric lifetimes of selected fluorinated ether compounds, Atmos. Environ., 1998, 32, 711–717 Search PubMed.
- M. Rabie, D. A. Dahl, S. M. A. Donald, M. Reiher and C. M. Franck, Predictors for gases of high electric strength, IEEE Trans. Dielectr. Electr. Insul., 2013, 20, 856–863 Search PubMed.
- M. Rabie, D. A. Dahl, S. M. A. Donald, M. Reiher and C. M. Franck, Predictors for gases of high electric strength, IEEE Trans. Dielectr. Electr. Insul., 2013, 20, 856–863 Search PubMed.
- M. Rabie and C. M. Franck, Computational screening of new high-voltage insulation gases with low global warming potential, IEEE Trans. Dielectr. Electr. Insul., 2015, 22, 296–302 Search PubMed.
- X. Yu, H. Hou and B. Wang, A priori theoretical model for discovery of environmentally sustainable perfluorinated compounds, J. Phys. Chem. A, 2018, 122, 3462–3469 CrossRef CAS PubMed.
- K. P. Shine and G. Myhre, The Spectral Nature of Stratospheric Temperature Adjustment and its Application to Halocarbon Radiative Forcing, Geophys. Res. Lett., 2020, 12, 1942–2466 Search PubMed.
- K. P. Shine and G. Myhre, The spectral nature of stratospheric temperature adjustment and its application to halocarbon radiative forcing, J. Adv. Model. Earth Syst., 2020, 12, e2019MS001951 CrossRef.
- S. Pinnock, M. D. Hurley, K. P. Shine, T. J. Wallington and T. J. Smyth, Radiative forcing of climate by hydrochlorofluorocarbons and hydrofluorocarbons, J. Geophys. Res., 1995, 100, 23227–23238 CrossRef CAS.
- M. H. Jamróz and J. Cz. Dobrowolski, Potential energy distribution (PED) analysis of DFT calculated IR spectra of the most stable Li, Na, and Cu(I) diformate molecules, J. Mol. Struct., 2001, 565–566, 475–480 CrossRef.
- R. Pincus, S. A. Buehler, M. Brath, C. Crevoisier, O. Jamil, K. Franklin Evans, J. Manners, R. L. Menzel, E. J. Mlawer, D. Paynter, R. L. Pernak and Y. Tellier, Benchmark calculations of radiative forcing by greenhouse gases, J. Geophys. Res.: Atmos., 2020, 125, e2020JD033483 Search PubMed.
- Ø. Hodnebrog, M. Etminan, J. Fuglestvedt, G. Marston, G. Myhre, C. Nielsen, K. P. Shine and T. Wallington, Global warming potentials and radiative efficiencies of halocarbons and related compounds: A comprehensive review, Rev. Geophys., 2013, 51, 300–378 CrossRef.
- X. Wan, W. Yu, A. Wang, X. Wang, J. Robertson, Z. Zhang and Y. Guo, High-throughput screening of gas sensor materials for decomposition products of eco-friendly insulation medium by machine learning, ACS Sens., 2023, 8, 2319–2330 CrossRef CAS PubMed.
- P. Price, B. Bottorff, J. Jenkins, W. H. Brune and P. S. Stevens, Re-assessing hydroxyl radical chemistry in the atmosphere: Instrument interferences may explain previous measurement discrepancies, Commun. Earth Environ., 2025, 6, 325 CrossRef.
- M. A. A. Ibrahim, N. K. M. Ahmed, A. H. M. Mahmoud, M. A. El-Tayeb, A. M. M. Abdelbacki, S. Khan, M. E. S. Soliman and T. Shoeib, RuC nanosheet as a promising biosensing material for detecting the aromatic amino aciacids: a DFT study, Nanoscale Adv., 2024, 6, 6398–6407 RSC.
- Q. Gao, X. Wang, H. Sun, A. Yang and C. Niu, Calculation of the decomposition products of C5F10O-Air mixtures from 500 K to 3500 K with a chemical kinetic model, Plasma Chem. Plasma Process., 2024, 44, 1883–1903 CrossRef CAS.
- K. Li, H. Javed, G. Zhang and A. T. Plesca, Analysis of air decomposition by-products under four kinds of partial discharge defects, IEEE Trans. Dielectr. Electr. Insul., 2017, 24(6), 3713–3721 Search PubMed.
- A. Wang, R. Kingsbury and M. McDermott, et al., A framework for quantifying uncertainty in DFT energy corrections, Sci. Rep., 2021, 11(1), 15496 CrossRef CAS PubMed.
- C. Man, J. Wang, K. Gao, Y. Li, Y. Zhang and X. Pei, Detection of Transient Decomposition Products of CF3SO2F and C4F7N via OES Under Spark and Glow Discharges, High Volt., 2025, 2397–7264 Search PubMed.
- D. C. Anderson, B. N. Duncan and A. M. Fiore, et al., Spatial and temporal variability of the hydroxyl radical: Understanding the role of large-scale climate features and their influence on OH through its dynamical and photochemical drivers, Atmos. Chem. Phys. Discuss., 2020, 2020, 1–32 Search PubMed.
- T. Gramstad and R. N. Haszeldine, 33. Perfluoroalkyl derivatives of sulphur. Part IV. Perfluoroalkanesulphonic acids, J. Chem. Soc., 1956, 173–180 RSC.
- M. Zhang, H. Hou and B. Wang, Mechanistic and Kinetic Investigations on Decomposition of Trifluoromethanesulfonyl Fluoride in the Presence of Water Vapor and Electric Field, J. Phys. Chem. A., 2023, 127, 671–684 CrossRef CAS PubMed.
- Y. Long, L. Guo, Y. Wang, C. Chen, Y. Chen, F. Li and W. Zhou, Electron swarms parameters in CF3SO2F as an alternative gas to SF6, Ind. Eng. Chem. Res., 2020, 59(24), 11355–11358 CrossRef CAS.
- M. P. Sulbaek Andersen, M. Kyte, S. T. Andersen, C. J. Nielsen and O. J. Nielsen, Atmospheric Chemistry of (CF3)2CF–CN: A Replacement Compound for the Most Potent Industrial Greenhouse Gas, SF6, Environ. Sci. Technol., 2017, 51, 1321–1329 Search PubMed.
- G. Zhao, H. Kim, C. Yang and Y. G. Chung, Leveraging machine learning to predict the atmospheric lifetime and the global warming potential of SF6 replacement gases, J. Phys. Chem. A, 2024, 128(12), 2399–2408 Search PubMed.
- C. Smith, Z. R. J. Nicholls, K. Armour, W. Collins, P. Forster, M. Meinshausen, M. D. Palmer, M. Watanabe, The Earth's Energy Budget, Climate Feedbacks, and Climate Sensitivity Supplementary Material, in Climate Change 2021: the Physical Science Basis. Contribution of Working Group I to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change, 2021 Search PubMed.
- Ø. Hodnebrog, B. Aamaas, J. S. Fuglestvedt, G. Marston, G. Myhre, C. J. Nielsen, M. Sandstad, K. P. Shine and T. J. Wallington, Updated global warming potentials and radiative efficiencies of halocarbons and other weak atmospheric absorbers, Rev. Geophys., 2020, 58, e2019RG000691 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |