Open Access Article
Open Access ArticleCycle contraction and symmetrisation in redox-active ligands: from alloxazine to isoimidazolonequinoxaline derivatives and their electrochemical and coordination studies
Jaison
Casas
a,
Shaban Raja
Muhammad
a,
David
Pianca
 b,
Nolwenn
Le Breton
b,
Sylvie
Choua
b,
Nathalie
Kyritsakas
c,
Christophe
Gourlaouen
b,
Nolwenn
Le Breton
b,
Sylvie
Choua
b,
Nathalie
Kyritsakas
c,
Christophe
Gourlaouen
 a,
Abdelaziz
Jouaiti
a,
Abdelaziz
Jouaiti
 a and
Sylvie
Ferlay
a and
Sylvie
Ferlay
 *a
*a
aUniversité de Strasbourg, CNRS, CMC UMR 7140, F-67000 Strasbourg, France. E-mail: ferlay@unistra.fr
bInstitut de Chimie, UMR CNRS 7177, Université de Strasbourg, Institut Le Bel, Strasbourg 67000, France
cService de radiocristallographie de la Fédération de Chimie Le Bel-UAR 2042, Université de Strasbourg and CNRS, F-67000 Strasbourg, France
First published on 28th January 2026
Abstract
The formation of two parent divergent ligands derived from 1,4-bis(pyrid-3-yl)benzene is reported. The synthetic route involves condensation of alloxan with a dibromodiamine precursor, followed by benzylation, leading, after the Suzuki–Miyaura cross-coupling reaction, to the formation of two ligands: L1, bearing the well-known pteridine-dione moiety, and L2, in which ring contraction and symmetrisation occur, resulting in an imidazopyrazinone core. The synthesis of L1 and L2, along with their characterization in solution and in the solid state, is reported. Electrochemical studies of L1 and L2 solutions revealed analogous two-electron reduction processes, with the first reduction step leading to radical species, as confirmed by EPR spectroelectrochemistry. For L1, the first and second reductions occur at ERed1 = −0,80 V and ERed2 = −1.35 V vs. Ag/AgCl, in agreement with the values reported for other pteridine-dione species, whereas L2 displays more negative reduction potentials, shifted by approximately 0.7 V. These observations were confirmed by DFT calculations. The coordination abilities of L1 and L2 were investigated. Single-crystal X-ray diffraction (SCXRD) revealed the formation of a pillared 3D compound, L2-Zn, obtained by mixing L2, 4,4′-biphenyldicarboxylic acid (H2bpdc) and a Zn2+ salt under solvothermal conditions. A series of powdered isostructural L2-M compounds (M = Zn, Ni, Co) was synthesized and characterized by powder X-ray diffraction (PXRD). Under the same conditions, using L1 instead of L2 led to the formation of poorly diffracting crystals, which nevertheless exhibited a three-dimensional pillared architecture. A complete series of powdered isostructural L1-M compounds (M = Co, Ni, Cu and Zn) was evidenced. The solid-state electrochemical behavior of the L2-M analogues (M = Zn and Co) was preliminarily investigated, revealing ligand-based reduction processes occurring within the three-dimensional pillared structure for both L2-Zn and L2-Co.
Introduction
Redox-active MOFs (RAMOFs),1–3 a subclass of Metal Organic Frameworks (MOFs),4–6 molecular crystalline materials, have received considerable attention due to their wide range of potential applications, including gas separation and purification, (electrochemical) sensing, photochromism, (photo)catalysis, electronic conductivity, magnetism, and energy storage devices such as rechargeable batteries and supercapacitors.7 The specific feature of RAMOFs lies in their ability to finely tune physical properties through electron exchange processes, making them interesting multifunctional materials,7 for example by improving charge transport within the network and enhancing the electrochemical response, as observed in electrochromic materials.8 Different methodologies can be applied for the formation of RAMOFs, comprising the use of metal centers with different valence states,2 the utilization of redox-active ligands,9 the incorporation of organic redox-active guests, or combinations of these approaches. In RAMOFs, redox-active ligands serve as electron donors or acceptors and participate in oxidation–reduction processes. Integrating redox-active species into MOFs facilitates charge delocalization throughout the three-dimensional coordination network, a key factor for effective charge transport.Among the various redox-active species used as ligands or guests for the formation of RAMOFs, one can cite tetrathiafulvalene (TTF),10,11 anthraquinone (AQ),12 tetracyanoquinodimethane (TCNQ),13 naphthalene diimide (NDI),14 perylene diimide (PDI),15 triphenylene (TP),16 pyrene, methyl viologen (MV2+),17 tetraphenylethylene (TPE),18 porphyrin (Por),19,20 2,2,6,6-tetramethylpiperidin N-oxyl radical (TEMPO)21 or perchlorotriphenylmethyl radical (PTM).22,23 These compounds offer several advantages including their easy chemical functionalization, chemical and electrochemical stability and relatively low cost production, making them suitable for their implementation in different types of devices: electronic or energy storage, for example.
More recently, a new class of redox-active compounds, alloxazine, isomers of isoalloxazine related to flavins,24 has emerged as promising materials to be used in energy storage devices.25,26 They belong to a significant class of biomolecules characterized by three distinct protonation and redox states, including an accessible intermediate radical state. We recently reported a groundbreaking redox-active divergent ligand derived from isoalloxazine, together with the formation of related coordination polymers27 and porous materials28,29 derived from pillared MOFs,30,31 that exhibit redox activity.
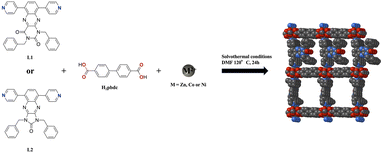
In the context of developing stable redox-active compounds for electrochemical energy storage,32 we report here two redox-active ligands based on 1,4-bis(pyrid-3-yl)benzene that are suitable for the construction of pillared metal–organic frameworks (MOFs):28,29L1 is derived from the pteridine-dione core (pyrimidine-2,4-dione, 6 membered ring) and L2 is derived from the imidazopyrazinone core (imidazolidin-2-one, 5 membered ring). In order to increase the solubility and stability of such compounds, particularly in aqueous solution, benzyl moieties were introduced on the core for L1 and L2 (Scheme 1). To the best of our knowledge, ligands based on isoimidazolonequinoxaline and their electrochemical properties have not been previously reported. The synthesis, characterization, electrochemical behaviours and coordination abilities of ligands L1 and L2 are reported. Herein, we present a comparative investigation of the electrochemical and coordination properties of two closely related redox-active ligands, one of which has undergone ring contraction and symmetrisation.
Results and discussion
Synthesis and formation of L1 and L2
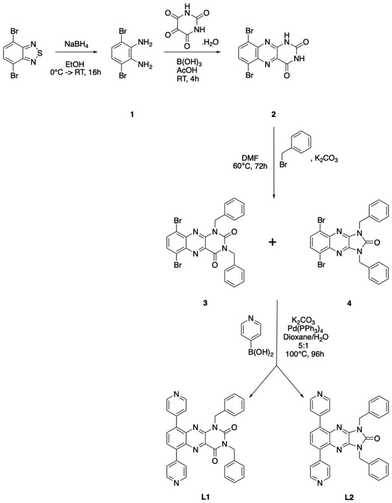
L1 and L2 were synthesized in a four-step sequence, adapted from the methodology recently reported (Scheme 2).27 The dibromodiamine precursor 1 was first prepared by reduction of a dibrominated thiadiazole derivative; then 1 was condensed with alloxan under acidic conditions, leading to the formation of 2, a dibromo-alloxazine. 2 was then appended on the nitrogen atoms of the alloxan moiety by the benzyl group, and during this process, performed in the presence of oxygen, two dibromo compounds were obtained: 3 (29% yield), presenting an alloxazine core and the symmetrical 4, displaying an imidazoquinoxalinone 5-membered ring (56% yield), evidencing a ring reduction during this step. It is noteworthy that compound 3 can be isolated in higher yield under anaerobic conditions. On both 3 and 4, the pyridyl moiety was then introduced via a Suzuki–Miyaura cross-coupling, affording L1 and L2 in 39% and 37% yield, respectively (see the Experimental part).During step 3 of the reaction, the nucleophilic substitution with bromobenzyl under aerobic conditions, a ring reduction to imidazo[4,5-b]quinoxaline and thus symmetrisation of the moiety occurred. Similar results were observed under anaerobic conditions; however, the yields were lower. This phenomenon was already described in the literature, while performing the alkylation of the alloxazine disodium salt33 or the conversion of 1,3-dialkyl-7-azapteridines into 1,3-dialkyl-6-azapurines.34 The literature reports a partial mechanism35–38 of ring contraction under basic conditions, proceeding through ring opening and decarboxylation to form a quinoxalinone intermediate.39 In addition, the photochemical reactivity of imidazo[4,5-b]quinoxaline derivatives has been reported only very recently39 and to the best of our knowledge, the redox properties of such species have not been exploited yet; only those of pyrido-[1′,2′:1,2]imidazo[4,5-b]quinoxaline have been reported.40
As a consequence of this phenomenon, L2 ligand (Fig. 1) displays a symmetrical redox-active core (imidazopyrazinone), whereas L1 contains a redox-active pteridine-dione unsymmetrical core. L1 and L2 have been characterized thoroughly in solution (see the Experimental section), and optical and electrochemical properties are reported here.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 450 L mol−1 cm−1) and 393 nm (ε = 9650 L mol−1 cm−1), which can be assigned to π–π* and n–π* transitions, respectively. L2 exhibits two closely spaced absorption bands at 338 nm (ε = 24
450 L mol−1 cm−1) and 393 nm (ε = 9650 L mol−1 cm−1), which can be assigned to π–π* and n–π* transitions, respectively. L2 exhibits two closely spaced absorption bands at 338 nm (ε = 24![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 920 L mol−1 cm−1) and 354 nm (ε = 24
920 L mol−1 cm−1) and 354 nm (ε = 24![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 360 L mol−1 cm−1).
360 L mol−1 cm−1).
Both compounds are emissive in solution (λexc = 320 nm for L1 and λexc = 349 nm for L2) and the emission spectrum of L1 displays two maxima at 382 nm and 457 nm, whereas L2 exhibits only a single emission peak at 382 nm (Fig. S1).
Through this study, it appeared that L2 is less oxidizing, with a significant shift towards low potentials, in the ground state. This can be explained by the missing carbonyl group on the imidazolidin-2-one 5 membered ring. The first reduction potential value of L2 is close to those of the quinoxaline radical anion bearing an electron-withdrawing substituent.43,44 Although the π-system of L2 is slightly more extended, the presence of two trivalent nitrogen atoms acting as π-donors, together with the absence of the carbonyl group on its five-membered ring compared to L1, lowers the reduction potential. This effect likely arises from increased electron density within the aromatic core (vide infra). Furthermore, this behavior is associated with charge delocalization along the rings of L1 and L2, as supported by the DFT calculations presented here.
As a conclusion, L1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 45 and L2 exhibit distinct two-electron redox profiles and the proposed redox states are depicted in Scheme S1 for L2 and admitted for L1.
45 and L2 exhibit distinct two-electron redox profiles and the proposed redox states are depicted in Scheme S1 for L2 and admitted for L1.
These data confirm the hypothetic electron exchange pathways proposed for L2 in Scheme S1, with the radical fully delocalized on the quinoxaline and imidazolone rings.
The lowest unoccupied molecular orbital (LUMO) of L2 (and L1) and the singly occupied molecular orbital (SOMO) of L2˙− (and L1˙−) depicted in Fig. S2 are almost identical. This indicates that the added electron occupies the LUMO of the initially oxidized ligand, effectively converting it into the SOMO upon reduction.
The L1 and L2 scaffolds are planar and their SOMO is of π-type. The two nitrogen atoms of the imidazolone core on L2 lie in the nodal plane of the quinoxaline π-electron system facilitating the extension of conjugation and suggesting a hyperfine coupling mechanism based on spin polarization. The condensed Mulliken spin densities of both L2˙− and L1˙− indicate that the added electron is fully delocalized over L2˙− with a higher spin density concentrated on the imidazolone core (Fig. 3). This increase in electron density within the fully aromatic core raises the reduction potential, thereby making reduction more difficult. The computed HOMO–LUMO energy gaps of the two ligands follow the same trend (3.652 eV for L1vs. 4.117 eV for L2). This confirms the Electrochemical and EPR data.
 | ||
| Scheme 3 Synthetic pathways for the formation of L1-M (M = Co, Ni, Cu and Zn) and L2-M (M = Co, Ni and Zn). | ||
Solvothermal reactions were conducted using L2 and H2bpdc (4,4′-biphenyldicarboxylic acid, H2bpdc) with three divalent metal salts. Reactions involving Ni2+ and Zn2+ salts led to the formation of single crystals which were analysed by single crystal X-ray diffraction (see Table S2). Both compounds crystallise in the monoclinic C2/c space group, are isometric, and present the formula 2((C14H8O4)2C33H24N6OM2), nC3H7NO [+solvent] (M = Co or Zn and n = 1 for M = Ni and n = 2 for M = Zn) or more generally (bpdc)2M2L2·nDMF (L2-M). The description of the structure will be performed for L2-Zn, since for L2-Ni the structure was poorly refined, but both compounds are isostructural, as seen from the crystallographic data (Table S2). The asymmetric unit is composed of two bpdc2− ligands, one ligand L2, two M2+ cations and one (M = Ni) or 2 (M = Zn) and free DMF molecule (see Fig. S4). In L2-Zn, disorder is observed both on the pyridyl rings and on the benzyl moieties appended to L2. As already observed,28,29 the connectivity in L2-Zn can be described as the formation of Zn2 paddlewheels (short Zn–Zn distance of 2.9186(5) Å), (Table S3), where the Zn2+ cations adopt a distorted square pyramidal geometrical environment. These Zn2 paddlewheels (see Fig. S3) are the corner square connecting units surrounded by four disordered pbdc2− ligands, ensuring the 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 metal-to-ligand stoichiometric ratio: this results in the formation of a 2D lozenge-shaped lattice (close to a distorted square grid), with sides measuring 15.170(8) Å and an angle of 77.63° (Fig. 4a). These 2D “grids” stack into layers interconnected by L2 ligands, resulting in a pillared 3D network with a metal
1 metal-to-ligand stoichiometric ratio: this results in the formation of a 2D lozenge-shaped lattice (close to a distorted square grid), with sides measuring 15.170(8) Å and an angle of 77.63° (Fig. 4a). These 2D “grids” stack into layers interconnected by L2 ligands, resulting in a pillared 3D network with a metal![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) pbdc
pbdc![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) L2 stochiometric ratio of 2
L2 stochiometric ratio of 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2
2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, as shown in Fig. 4b. Refined DMF molecules are also present in the network. The crystal structure contains two interpenetrating 3D networks as shown in Fig. S3, leading to channels with a reduced diameter of ca. 8.5 Å. The two networks interact with each other through π–π stacking between the imidazolonequinoxaline (L2) cores and the phenyl groups from the pbdc2− ligands as shown in Fig. 4c.
1, as shown in Fig. 4b. Refined DMF molecules are also present in the network. The crystal structure contains two interpenetrating 3D networks as shown in Fig. S3, leading to channels with a reduced diameter of ca. 8.5 Å. The two networks interact with each other through π–π stacking between the imidazolonequinoxaline (L2) cores and the phenyl groups from the pbdc2− ligands as shown in Fig. 4c.
For both compounds L2-Zn and L2-Ni, the squeeze command of Platon48 was used for the structure refinement. The residual electron density (222 for L2-Ni or 81 for L2-Zn) was assigned to five (L2-ZNi) or two (L2-Zn) additional molecules of the N,N-dimethylformamide solvent per asymmetric unit, which could not be refined.
Attempts to obtain an isostructural compound of L2-Ni or L2-Zn were made using the same conditions and the Co2+ cation. A powdered compound was obtained (L2-Co) that was analysed using PXRD, as shown in Fig. S5. It evidenced the formation of a series of 3 isostructural polycrystalline compounds L2-Ni, L2-Zn and L2-Co.
Solvothermal reactions, under the same conditions but involving L1, H2bpdc (4,4′-biphenyldicarboxylic acid, H2bpdc) and metallic salts, were performed with three divalent metal salts. Poorly diffracting single crystals were obtained using a Co2+ salt, leading to a compound named L1-Co. The cell parameters could be estimated by SCXRD (see Table S4). Despite analysis of the single crystals at a synchrotron facility, only a preliminary structure could be obtained, which could not be completely solved. Preliminarily diffraction data showed that L1-Co adopts the same arrangement as observed in parent compounds, Fig. S4.28,29 Pictures of the preliminarily solved structure are shown in the SI, revealing a strong disorder for L1. A series of 4 powdered isostructural compounds could thus be obtained starting from Co2+, Ni2+, Cu2+ or Zn2+ salts, as shown by the PXRD diagrams, leading to a whole series of polycrystalline isostructural compounds L1-M (M = Co, Ni, Cu and Zn) (Fig. S6).
For L2-M (M = Zn, Ni and Co) and L1-M (M = Zn, Ni, Co and Cu), the TGA traces reveal a stability depending on the nature of the metal: the decomposition temperature around 400 °C for L1-M (M = Co, Ni and Zn) and L2-Co and L2-Zn, 350 °C for L1-Cu and 320 °C for L2-Ni (Fig. S6). The measurements revealed significant solvent losses, making it impossible to accurately determine the exact number of solvent molecules present in each sample (i.e., adsorbed H2O and DMF).
For L1-M (M = Co and Zn), after careful activation (see the Experimental section), N2 adsorption measurements were performed. The compounds indicate a type I sorption profile (see Fig. S7), and using the BJH model,49 the highest surface areas of 698 m2 g−1 and 641 m2 g−1 were determined, with the pore size estimated to be 12 and 14 Å for L1-Co and L1-Zn respectively.
For the L2-M compounds, an insufficient amount of material was available to carry out the adsorption measurements.
Unfortunately, due to a small amount of reduced compounds in the solid state (films), further characterization (PXRD, gas sorption or TGA) of the reduced MOF species could not be performed.
Experimental
Synthetic procedures
L1 and L2 have been obtained using a synthetic pathway already reported by us (see Scheme 2), and 1 and 2 intermediates have been already described.27![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1).
1).
3: yellow solid in 29% yield.
Compound 3 could be synthesized under the same precedent conditions with a higher yield (up to 90%) by carrying out the reaction under argon.
1 H NMR (CDCl 3 , 500 MHz, 25 °C) δ ppm: 8.03 (d, J = 8.1 Hz, 1H), 7.90 (d, J = 8.1 Hz, 1H), 7.80–7.74 (m, 2H), 7.64–7.58 (m, 2H), 7.36–7.27 (m, 6H), 5.68 (s, 2H), 5.37 (s, 2H).
13 C NMR (CDCl 3 , 126 MHz, 25 °C) δ ppm: 136.81, 132.51, 129.84, 129.60, 128.60, 128.57, 128.24, 128.15, 46.33, 46.01.
ESI-MS: m/z [M + H]+ calcd for C24H17N4O4Br2 [M + H]+ 572.9532; found 572.9528.
4: white solid, 0.2 g in 56% yield.
1 H NMR (CDCl 3 , 500 MHz, 25 °C) δ ppm:.7.75 (s, 2H), 7.69 (m, 4H), 7.34 (m, 6H), 5.24 (s, 4H).
13 C NMR (CDCl 3 , 126 MHz, 25 °C) δ ppm: 153.8, 140.3, 137.8, 135.2, 130.5, 129.6, 128.8, 128.4, 121.9, 50.9.
ESI-MS: m/z [M + H]+ calcd for C23H17N4O4Br2 [M + H]+ 522.9763; found 522.9759.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 60 mL) together with 4-pyridylboronic acid (0.408 g, 3.31 mmol) and K2CO3 (0.600 g, 4.34 mmol). The flask was placed under argon, and Pd(PPh3)4 (68 mg, 0.059 mmol, ∼5 mol%) was added, resulting in a homogeneous orange solution. The reaction mixture was stirred at 100 °C for 4 days under an inert atmosphere. After cooling to room temperature, the solvent was evaporated under reduced pressure. The resulting solid was dissolved in CH2Cl2 (50 mL) and washed with brine (3 × 50 mL). The organic phase was then concentrated and the crude product was purified by silica gel column chromatography (CH2Cl2/CH3OH 99.5
1, 60 mL) together with 4-pyridylboronic acid (0.408 g, 3.31 mmol) and K2CO3 (0.600 g, 4.34 mmol). The flask was placed under argon, and Pd(PPh3)4 (68 mg, 0.059 mmol, ∼5 mol%) was added, resulting in a homogeneous orange solution. The reaction mixture was stirred at 100 °C for 4 days under an inert atmosphere. After cooling to room temperature, the solvent was evaporated under reduced pressure. The resulting solid was dissolved in CH2Cl2 (50 mL) and washed with brine (3 × 50 mL). The organic phase was then concentrated and the crude product was purified by silica gel column chromatography (CH2Cl2/CH3OH 99.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.5) to afford L1 as a yellow solid (0.232 g, 0.42 mmol, 38.8%).
0.5) to afford L1 as a yellow solid (0.232 g, 0.42 mmol, 38.8%).
1 H NMR (CDCl 3 , 500 MHz, 25 °C) δ ppm: 8.92–8.76 (m, 2H), 8.70–8.60 (m, 2H), 8.15 (d, J = 7.9 Hz, 1H), 7.93 (d, J = 7.4 Hz, 1H), 7.84 (d, J = 7.5 Hz, 1H), 7.78 (d, J = 7.9 Hz, 1H), 7.51–7.33 (m, 4H), 7.26–7.17 (m, 3H), 7.07 (td, J = 8.9, 4.6 Hz, 3H), 6.98 (d, J = 7.2 Hz, 2H), 5.37 (s, 2H), 5.27 (s, 2H).
13 C NMR (CDCl 3 , 126 MHz, 25 °C) δ ppm: 158.84, 150.70, 150.66, 150.47, 149.42, 149.17, 144.53, 141.40, 138.84, 138.69, 138.05, 137.82, 136.26, 136.12, 135.77, 133.86, 133.56, 132.93, 132.16, 132.08, 129.55, 129.37, 129.07, 128.59, 128.45, 128.35, 128.06, 127.76, 123.13, 45.89.
IR bands (cm −1 ): 1722 (medium), 1677 (strong), 1585 (weak), 1555 (strong), 1496 (medium), 1472 (medium), 1397 (strong), 1327 (medium), 1280 (medium), 1239 (strong), 1224 (strong), 1075 (weak), 1025 (medium), 802 (medium), 747 (medium), 700 (strong), 632 (medium), 606 (medium), 554 (weak), 508 (strong), 453 (weak).
MS (ESI-MS): m/z calculated for C34H24N6O2, 548.20; found, 549.20 [M + H]+.
UV: ε (351 nm) = 17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 450 L mol−1 cm−1; ε (393 nm) = 9650 L mol−1 cm−1 (see Fig. S1).
450 L mol−1 cm−1; ε (393 nm) = 9650 L mol−1 cm−1 (see Fig. S1).
Elemental analysis: Anal. calc. for C37H30N6O3 (C34H24N6O2·CH3COCH3), N, 13.85%; C, 73.25%; H, 4.98%; found, N, 13.85%, C, 73.25%; H, 4.98%.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 60 mL). The flask was placed under argon and Pd(PPh3)4 (62.4 mg, 0.054 mmol, 5 mol%) was added. The dissolution of all components was observed at 65 °C, resulting in a clear orange-red solution. The reaction mixture was stirred at 100 °C for 4 days. At the end of the reaction, the dark rust-colored mixture was cooled to room temperature and extracted with CH2Cl2 (3 × 50 mL), and the organic layers were washed with brine (3 × 50 mL). The organic phase evaporated under reduced pressure. The crude product was purified by column chromatography (silica gel, CH2Cl2/methanol 99.5
1, 60 mL). The flask was placed under argon and Pd(PPh3)4 (62.4 mg, 0.054 mmol, 5 mol%) was added. The dissolution of all components was observed at 65 °C, resulting in a clear orange-red solution. The reaction mixture was stirred at 100 °C for 4 days. At the end of the reaction, the dark rust-colored mixture was cooled to room temperature and extracted with CH2Cl2 (3 × 50 mL), and the organic layers were washed with brine (3 × 50 mL). The organic phase evaporated under reduced pressure. The crude product was purified by column chromatography (silica gel, CH2Cl2/methanol 99.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.5) to afford L2 as a yellow solid (0.205 g, 0.40 mmol, 37.4%).
0.5) to afford L2 as a yellow solid (0.205 g, 0.40 mmol, 37.4%).
1 H NMR (CDCl 3 , 300 MHz, 25 °C) δ ppm: 8.75 (d, J = 5.7 Hz, 4H), 7.68 (s, 2H), 7.54 (d, J = 5.7 Hz, 4H), 7.43 (dd, J = 9.6, 1.8 Hz, 4H), 7.35–7.30 (m, 6H), 5.093 (s, 4H).
IR bands (cm −1 ): 3031 (weak), 1723 (medium), 1672 (strong), 1590 (medium), 1560 (strong), 1495 (weak), 1396 (strong), 1327 (medium), 1280 (medium), 1239 (strong), 1224 (strong), 1075 (weak), 1026 (weak), 811 (strong), 746 (medium), 690 (strong), 626 (medium), 509 (strong), 488 (medium).
Elemental analysis: Anal. calc. for C33.2H24.8N6O1.2 (C33H24N6O·0.2MeOH), N, 15.95%, C, 75.66%; H, 4.74%; found, N, 15.93%; C, 75.53%; H, 4.64%.
UV: ε (338 nm) = 24![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 920 Lmol−1cm−1; ε (354 nm) = 24
920 Lmol−1cm−1; ε (354 nm) = 24![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 360 L mol−1 cm−1 (see Fig. S1).
360 L mol−1 cm−1 (see Fig. S1).
MS (ESI-MS): m/z calculated for C33H24N6O, 520.60; found, 521.21 [M + H]+.
Anal. calc. for (C14H8O2)2(C33H24N6O)Ni2·(C3H7NO)8(H2O): N, 7.71%, C, 60.45%; H, 4.99%; found, N, 7.62%; C, 62.56%; H, 4.93%.
Anal. calc. for (C14H8O2)2(C33H24N6O)Zn2·4(C3H7NO) 3(H2O): N, 9.90%, C, 61.99%; H, 5.27%; found, N, 9.85%; C, 62.43%; H, 5.15%.
Anal. calc. for (C14H8O2)2(C33H24N6O)Co2·(C3H7NO) 6(H2O): N, 7.93%, C, 62.19%; H, 4.81%; found, N, 7.86%; C, 62.48%; H, 4.85%.
Anal. calc. for (C14H8O2)2(C34H24N6O2)Co2·6(C3H7NO)4(H2O): N, 10.55%, C, 60.30%; H, 5.7%; found, N, 10.60%; C, 61.56%; H, 5.60%.
Anal. calc. for (C14H8O2)2(C34H24N6O2)Ni2·5(C3H7NO): N, 10.64%, C, 63.87%; H, 5.22%; found, N, 10.55%; C, 62.49%; H, 5.30%.
Anal. calc. for (C14H8O2)2(C34H24N6O2)Zn2·10(C3H7NO)4(H2O): N, 11.80%, C, 58.19%; H, 6.26%; found, N, 11.72%; C, 60.26%; H, 6.24%.
Anal. calc. for (C14H8O2)2(C34H24N6O2)Cu2·6(C3H7NO)3(H2O): N, 10.60%, C, 60.63%; H, 5.60%; found, N, 10.42%; C, 60.42%; H, 5.58%.
For the 4 compounds, IR (cm−1) ν: 1659 (s), 1610 (m), 1093 (w), 826 (w), 771 (s), 680 (m), 661 (m), 468 (s).
Materials and methods
FT-IR spectra of the powdered microcrystalline samples were recorded on a PerkinElmer Spectrum Two FTIR-UATR in the wavenumber interval of 4000–400 cm−1. Absorbance spectra were recorded on a PerkinElmer Lambda 650 spectrometer. Emission spectra were recorded on a PerkinElmer LS55 spectrophotometer. Low-pressure gas sorption measurements (N2) were obtained on a Micromeritics ASAP 2020 analyser. Powdered microcrystalline samples were activated under dynamic vacuum at a temperature of 160 °C over 12 h. TGA measurements were performed on powdered microcrystalline compounds on a Pyris 6 TGA Lab System (PerkinElmer), using a N2 flow of 20 mL min−1 and a heating rate of 4 °C min−1.L2-Co and L2-Zn have been investigated in the solid state: the deposition on the glassy carbon electrode was performed by suspending microcrystalline powder of L2-Co (or L2-Zn) in an EtOH solution, then the solution was drop-cast on the surface of the electrode and dried in air, in order to cover the entire glassy carbon disk.
The ORCA package57,58 was used to calculate EPR properties through a single point on ADF-optimized geometries using the B3LYP/G functional in combination with 6-31g* basis sets.
SCXRD. Intensity data were collected for L2-Ni, L1-Co and L2-Zn, using a 4-circle Bruker PHOTON III diffractometer equipped with two micro-sources IμS Mo and IμS Diamond Cu, along with an Oxford Cryosystem 800 for low temperature measurements. The cell parameters were determined using APEX3 software,59 and the structures were solved using the program SHELXT-2014.60 The refinement and all further calculations were carried out using SHELXL-2018.61 Hydrogen atoms were included in calculated positions and treated as riding atoms using SHELXL default parameters. The non-H atoms were refined anisotropically, using weighted full-matrix least-squares on F2. A semi-empirical absorption correction was applied using SADABS in APEX3.
CCDC for L2-Zn: 2515971.
The cif file of poorly refined L2-Ni and L1-Co is provided as an example in the SI.
PXRD. Diffractograms for the air-dried microcrystalline samples were recorded at room temperature (293(2) K), on a Bruker D8 diffractometer using monochromatic Cu-Kα radiation. A scanning range between 2° and 40° with a scan step size of 2° min−1 was used, and the sample holder was rotated at 15 rpm.
Conclusions
The formation of L1 and L2 demonstrates the feasibility of generating redox-active ligands derived from 1,4-bis(pyrid-3-yl)benzene. These ligands can be investigated from an electrochemical perspective and employed in the construction of three-dimensional pillared networks, thereby providing a synthetic strategy for the development of new solid-state materials, supported by a comprehensive range of characterisation techniques. Both ligands are appended with benzyl groups and display redox-active cores: pteridine-dione for L1 (pyrimidine-2,4-dione, 6 membered ring) and imidazopyrazinone for L2 (imidazolidin-2-one, 5 membered ring), which is thus a symmetrical core. In CH2Cl2 solution, both L1 and L2 exhibit reversible reduction processes, occurring at lower potentials for L1. DFT calculations confirm the observed electrochemical trends, and spectroelectrochemical studies involving EPR, evidenced, in both cases, the formation of the radical anions and the density of spins on the different rings. This is the first study evidencing the electrochemical properties of imidazolonequinoxaline derivatives.The coordination abilities of both pillars L1 and L2 were investigated and it was revealed that in the presence of a metal and a dicarboxylic acid, interpenetrated 3D pillared MOFs are formed, that are named L1-M (M = Co, Ni, Cu and Zn) and L2-M (M = Co, Ni and Zn). The electrochemical ability of L2-Zn and L2-Co was checked using solid-state electrochemistry, and a reduction process was found centered on the ligands.
This comparative study provides new insight into the electrochemical behavior of the two parent species and their ability to coordinate to transition metals. These investigations demonstrate the feasibility of developing new, robust redox-active ligands that can be incorporated into molecular networks. This provides new perspectives for the development of new materials for energy storage applications, for example.
Author contributions
Jaison Casas: formal analysis, investigation and methodology; Shaban Raja Muhammad: formal analysis and investigation; David Pianca: formal analysis, investigation and methodology; Nolwenn Le Breton: investigation and data curation; Sylvie Choua: investigation, data curation, writing, reviewing and supervision; Nathalie Kyritsakas: investigation and data curation; Christophe Gourlaouen: investigation and data curation; Abdelaziz Jouaiti: methodology, reviewing and supervision; Sylvie Ferlay: methodology, funding, writing, reviewing, editing and supervision.All authors have given approval to the final version of the manuscript.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: UV characterization of L1 and L2 in solution, proposed redox-behaviour from electrochemical studies in the solid state, the numbering scheme for L1 and L2, DFT calculations for L1 and L2, representation of L2-Zn from the crystallographic data, crystallographic data, selected bond lengths, representation and description of L1-Co from preliminarily crystallographic data, PXRD patterns and TGA traces for L2-M (M = Zn, Ni and Co) and L1-M (M = Zn, Ni, Co and Cu) and N2 adsorption for L1-M (M = Co and Zn). See DOI: https://doi.org/10.1039/d5qi02572a.CCDC 2515971 (L2-Zn) contains the supplementary crystallographic data for this paper.62
Acknowledgements
Financial support from the University of Strasbourg, Unistra, and the CNRS (INC) is acknowledged. For the XRD resolution of the structures, the Service de radiocristallographie de la Fédération de Chimie Le Bel – UAR 2042 is warmly acknowledged. We thank the Agence Nationale de la Recherche (ANR) through the BattAllox Project ANR-20-CE05-0005. Sidonie Brillard is warmly acknowledged for performing some CV measurements. The French research infrastructure INFRANALYTICS FR2054 is acknowledged for its support.References
- J. Su, S. Yuan, H. Y. Wang, L. Huang, J. Y. Ge, E. Joseph, J. Qin, T. Cagin, J. L. Zuo and H. C. Zhou, Redox-switchable breathing behavior in tetrathiafulvalene-based metal-organic frameworks, Nat. Commun., 2017, 8, 2008, DOI:10.1038/s41467-017-02256-y.
- D. M. D'Alessandro, Exploiting redox activity in metal-organic frameworks: concepts, trends and perspectives, Chem. Commun., 2016, 52, 8957–8971, 10.1039/C6CC00805D.
- J. J. Calbo, M. J. Golomb and A. Walsh, Redox-active metal-organic frameworks for energy conversion and storage, J. Mater. Chem. A, 2019, 7, 16571–16579, 10.1039/C9TA04680A.
- H. C. Zhou, J. R. Long and O. M. Yaghi, Chem. Rev., 2012, 112 DOI:10.1021/cr300014x . Metal-organic frameworks special issue.
- H.-C. Zhou and S. Kitagawa, Metal-Organic Frameworks (MOFs), Chem. Soc. Rev., 2014, 43, 5415–5418, 10.1039/C4CS90059F.
- M. Dincă and J. R. Long, Porous framework chemistry special issue, Chem. Rev., 2020, 120, 673–674, DOI:10.1021/acs.chemrev.0c00836.
- S. A. A. Razavi, W. Chen, H.-C. Zhou and A. Morsali, Tuning redox activity in Metal-Organic Frameworks: from structure to application, Coord. Chem. Rev., 2024, 517, 216004, DOI:10.1016/j.ccr.2024.216004.
- B. Ding, M. B. Solomon, C. F. Leong and D. M. D'Alessandro, Redox-active ligands: recent advances towards their incorporation into coordination polymers and metal–organic frameworks, Coord. Chem. Rev., 2021, 439, 213891, DOI:10.1016/j.ccr.2021.213891.
- M. Souto, K. Strutynski, M. Melle-Franco and J. Rocha, Electroactive organic building blocks for the chemical design of functional porous frameworks (MOFs and COFs) in electronics, Chem. – Eur. J., 2020, 26, 10912–10935, DOI:10.1002/chem.202001211.
- Y.-G. Weng, Z.-H. Ren, Z.-R. Zhang, J. Shao and Q.-Y. Zhu, Tetrathiafulvalene-cobalt metal–organic frameworks for lithium-ion batteries with superb rate capability, Inorg. Chem., 2021, 60, 17074–17082, DOI:10.1021/acs.inorgchem.1c02304.
- F. Solano, P. Auban-Senzier, I. Olejniczak, B. Barszcz, T. Runka, P. Alemany, E. Canadell, N. Avarvari and N. Zigon, Bis(vinylenedithio)-tetrathiafulvalene-based coordination networks, Chem. – Eur. J., 2023, 29, e202203138, DOI:10.1002/chem.202203138.
- L. Zhao, W. Cai, G. Ji, J. Wei, Z. Du, C. He and C. Duan, Anthraquinone-based metal-organic frameworks as a bifunctional photocatalyst for C–H activation, Inorg. Chem., 2022, 61, 9493–9503, DOI:10.1021/acs.inorgchem.2c00441.
- Y. Y. Grisan Qiu, S. Mearini, D. Baranowski, I. Cojocariu, M. Jugovac, G. Zamborlini, P. Gargiani, M. Valvidares, V. Feyer and C. M. Schneider, Robust high-spin Fe2+ centers in 2D TCNQ-based metal–organic frameworks, Inorg. Chem. Front., 2025, 12, 2334–2340, 10.1039/D4QI03123G.
- L. S. Xie, G. Skorupskii and M. Dincă, Electrically conductive metal–organic frameworks, Chem. Rev., 2020, 120, 8536–8580, DOI:10.1021/acs.chemrev.9b00766.
- J. Wang, H. Wei, J. Guan, K. Müllen and M. Yin, Perylene- and, Perylene Diimide-based Framework Materials Constructed through Metal Coordination, Chem. – Eur. J., 2025, 31, e202403234, DOI:10.1002/chem.202403234.
- N. Contreras-Pereda, S. Pané, J. Puigmartí-Luis and D. Ruiz-Molina, Conductive properties of triphenylene MOFs and COFs, Coord. Chem. Rev., 2022, 460, 214459, DOI:10.1016/j.ccr.2022.214459.
- W.-Q. Kan, S.-Z. Wen, Y.-C. He and C.-Y. Xu, Viologen-Based Photochromic Coordination Polymers for Inkless and Erasable Prints, Inorg. Chem., 2017, 56, 14926–14935, DOI:10.1021/acs.inorgchem.7b02206.
- S. Wang, L. Zhao, H. Sun, Y. Wu, R. Wang, S. Zhang, L. Du and Q.-H. Zhao, Two novel three-dimensional tetraphenylethylene-based rare earth MOFs with ultra-high proton conductivity and performance stability, Chem. – Eur. J., 2022, 28, e202202154, DOI:10.1002/chem.202202154.
- S. R. Ahrenholtz, C. C. Epley and A. J. Morris, Solvothermal preparation of an electrocatalytic metalloporphyrin MOF thin film and its redox hopping charge-transfer mechanism, J. Am. Chem. Soc., 2014, 136, 2464–2472, DOI:10.1021/ja410684q.
- D. Micheroni, G. Lan and W. Lin, Efficient electrocatalytic proton reduction with carbon nanotube-supported metal–organic frameworks, J. Am. Chem. Soc., 2018, 140, 15591–15595, DOI:10.1021/jacs.8b09521.
- L. Li, R. Matsuda, I. Tanaka, H. Sato, P. Kanoo, H. J. Jeon, M. L. Foo, A. Wakamiya, Y. Murata and S. Kitagawa, A crystalline porous coordination polymer decorated with nitroxyl radicals catalyzes aerobic oxidation of alcohols, J. Am. Chem. Soc., 2014, 136, 7543–7546, DOI:10.1021/ja5019095.
- G. Valente, P. Ferreira, M. A. Hernández-Rodríguez, C. D. S. Brites, J. S. Amaral, P. Zelenovskii, F. A. Almeida Paz, S. Guieu, J. Rocha and M. Souto, Exploring the luminescence, redox, and magnetic properties in a multivariate metal–organic radical framework, Chem. Mater., 2024, 36, 1333–1341, DOI:10.1021/acs.chemmater.3c02460.
- D. D. Maspoch, D. Ruiz-Molina, K. Wurst, N. Domingo, M. Cavallini, F. Biscarini, J. Tejada, C. Rovira and J. Veciana, A nanoporous molecular magnet with reversible solvent-induced mechanical and magnetic properties, Nat. Mater., 2003, 2, 190–195, DOI:10.1038/nmat834.
- C. Walsh, Naturally occurring 5-deazaflavin coenzymes: biological redox roles, Acc. Chem. Res., 1986, 19, 216–221, DOI:10.1021/ar00127a004.
- J. Hong, M. Lee, B. Lee, D.-H. Seo, C. B. Park and K. Kang, Biologically inspired pteridine redox centres for rechargeable batteries, Nat. Commun., 2014, 5, 5335, DOI:10.1038/ncomms6335.
- K. Lin, R. Gomez-Bombarelli, E. S. Beh, L. Tong, Q. Chen, A. Valle, A. Aspuru-Guzik, M. J. Aziz and R. G. Gordon, A redox-flow battery with an alloxazine-based organic electrolyte, Nat. Energy, 2016, 1, 16102, DOI:10.1038/nenergy.2016.102.
- J. Casas, D. Pianca, N. L. Breton, A. Jouaiti, C. Gourlaouen, M. Desage-El Murr, S. Le Vot, S. Choua and S. Ferlay, Alloxazine-based ligands appended with coordinating groups: synthesis, electrochemical studies, and formation of coordination polymers, Inorg. Chem., 2024, 63, 4802–4806, DOI:10.1021/acs.inorgchem.3c04550.
- J. Casas, A. I. Vicatos, L. J. Barbour, N. Kyritsakas, A. Jouaiti and S. Ferlay, Series of microporous redox-active pillared MOFs based on alloxazine ligands, ChemistryOpen, 2025, e202500461, DOI:10.1002/open.202500461.
- J. Casas, A. I. Vicatos, N. Kyritsakas, S. Le Vot, A. Jouaiti and S. Ferlay, Rational formation of porous pillared alloxazine based MOFs: pore opening effect and redox properties, 2025, submitted.
- V. Guillerm, D. Kim, J. F. Eubank, R. Luebke, X. Liu, K. Adil, M. S. Lah and M. Eddaoudi, A supermolecular building approach for the design and construction of metal-organic frameworks, Chem. Soc. Rev., 2014, 43, 6141–6172, 10.1039/C4CS00135D.
- F. Zarekarizi, M. Joharian and A. Morsali, Pillar-layered MOFs: functionality, interpenetration, flexibility and applications, J. Mater. Chem. A, 2018, 6, 19288–19329, 10.1039/C8TA03306D.
- J. J. M. Cameron, C. Holc, A. J. Kibler, C. L. Peake, D. A. Walsh, G. N. Newton and L. R. Johnson, Molecular redox species for next-generation batteries, Chem. Soc. Rev., 2021, 50, 5863–5883, 10.1039/D0CS01507E.
- V. E. Semenov, R. K. Giniyatullin, D. R. Sharafutdinova and V. S. Reznik, Russ. J. Org. Chem., 2010, 46, 439–443, DOI:10.1134/S1070428010030243.
- F. Yoneda, M. Noguchi, M. Noda and Y. Nitta, Synthesis of imidazo[4,5-e]-as-triazine (6-azapurine) derivatives, Chem. Pharm. Bull., 1978, 26, 3154–3160, DOI:10.1248/cpb.26.3154.
- S. B. Smith and T. C. Bruice, Mechanisms of isoalloxazine (flavine) hydrolysis, J. Am. Chem. Soc., 1975, 97, 2875–2881, DOI:10.1021/ja00843a043.
- H. I. X. Mager, Activation and transfer of oxygen-XII: alkoxy adducts derived from I,3,10-trimethylalloxazinium (1,3-dimethylflavinium) cations, Tetrahedron, 1977, 33, 981–989, DOI:10.1016/0040-4020(77)80212-3.
- T. Harayama, Y. Tezuka, T. Taga and F. Yoneda, Hydrolysis products of flavins (isoalloxazines), J. Chem. Soc., Perkin Trans. 1, 1987, 75–83, 10.1039/P19870000075.
- V. E. Semenov, R. K. Giniyatullin, D. R. Sharafutdinova and V. S. Rezni, Ring Contraction of 1,3-Bis(5-bromopentyl)alloxazine, Russ. J. Org. Chem., 2010, 46, 439–443, DOI:10.1134/S1070428010030243.
- A. Rehpenn, S. Hindelang, K.-N. Truong, A. Pöthig and G. Storch, Enhancing flavins photochemical activity in hydrogen atom abstraction and triplet sensitization through ring-contraction, Angew. Chem., Int. Ed., 2024, 63, e202318590, DOI:10.1002/anie.202318590.
- S. Iwata and K. Tanaka, Electrochemical properties of pyrido[1′,2′:1,2]imidazo[4,5-b]pyrazine and pyrido[1′,2′:1,2]imidazo[4,5-b]quinoxaline derivatives, J. Heterocycl. Chem., 1998, 35, 939–941, DOI:10.1002/jhet.5570350426.
- M. J. Clarke and M. G. Dowling, Cyclic voltammetry studies of metalloflavin complexes in aqueous solution, Inorg. Chem., 1981, 20, 3506–3514, DOI:10.1021/ic50224a070.
- F. M. Hornung, O. Heilmann, W. Kaim, S. Zalis and J. Fiedler, Metal vs ligand reduction in complexes of 1,3-dimethylalloxazine (DMA) with copper(I), ruthenium(II), and tungsten(VI): crystal structures of (DMA)WO2Cl2 and (Bis(1-methylimidazol-2-yl)ketone)WO2Cl2, Inorg. Chem., 2000, 39, 4052–4058, DOI:10.1021/ic0001816.
- J. R. Ames, M. A. Houghtaling and D. L. Terrian, Cyclic voltammetry of some quinoxaline di-N-oxides and quinoxalines in dimethylformamide, Electrochim. Acta, 1992, 37, 1433–1436, DOI:10.1016/0013-4686(92)87018-U.
- M. Maruyama and K. Murakami, Electrochemical behavior of nitrogen heterocycles in N,N-dimethylformamide-water and aqueous buffer solution, J. Electroanal. Chem., 1979, 102, 221–235, DOI:10.1016/S0022-0728(79)80393-9.
- Flavins: Photochemistry and Photobiology, ed. E. Silva and A. M. Edwards, RSC Publishing, 2006 Search PubMed.
- W. Kaim, B. Schwederski, O. Heilmann and F. M. Hornung, Coordination compounds of pteridine, alloxazine and flavin ligands: structures and properties, Coord. Chem. Rev., 1999, 182, 323–342, DOI:10.1016/S0010-8545(98)00193-3.
- A. Das, H. Jobelius, J. Schleinitz, S. Gamboa-Ramirez, G. Creste, G. Kervern, J. Raya, N. L. Breton, A. Guénet, Z. Boubegtiten-Fezoua, L. Grimaud, M. Orio, G. Rogez, P. Hellwig, S. Choua, S. Ferlay and M. Desage-El Murr, A hybrid bioinspired catechol-alloxazine triangular nickel complex stabilizing protons and electrons, Inorg. Chem. Front., 2021, 8, 5286–5298, 10.1039/D1QI01131F.
- A. L. Spek, Single-crystal structure validation with the program PLATON, J. Appl. Crystallogr., 2003, 36, 7–13, DOI:10.1107/S0021889802022112.
- E. P. Barrett, L. G. Joyner and P. P. Halenda, The Determination of Pore Volume and Area Distributions in Porous Substances. I. Computations from Nitrogen Isotherms, J. Am. Chem. Soc., 1951, 73, 373–380, DOI:10.1021/ja01145a126.
- S. Stoll and A. J. Schweiger, EasySpin, a comprehensive software package for spectral simulation and analysis in EPR, Magn. Reson., 2006, 178, 42–55, DOI:10.1016/j.jmr.2005.08.013.
- G. te Velde, F. M. Bickelhaupt, E. J. Baerends, C. F. Guerra, S. J. A. van Gisbergen, J. G. Snijders and T. Ziegler, Chemistry with ADF, J. Comput. Chem., 2001, 22, 931–967, DOI:10.1002/jcc.1056.
- P. J. Stephens, F. J. Devlin, C. F. Chabalowski and M. J. Frisch, Ab initio calculation of vibrational absorption and circular dichroism spectra using density functional force fields, J. Phys. Chem., 1994, 98, 11623–11627, DOI:10.1021/j100096a001.
- S. Grimme, J. Anthony, S. Ehrlich and H. Krieg, A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H–Pu, J. Chem. Phys., 2010, 132, 154104, DOI:10.1063/1.3382344.
- C. Lee, W. Yang and R. G. Parr, Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density, Phys. Rev. B:Condens. Matter Mater. Phys., 1988, 37, 785–789, DOI:10.1103/physrevb.37.785.
- A. D. Becke, Density-functional thermochemistry. III. The role of exact exchange, J. Chem. Phys., 1993, 98, 5648–5652, DOI:10.1063/1.464913.
- C. Pye and T. Ziegler, An implementation of the conductor-like screening model of solvation within the Amsterdam density functional package, Theor. Chem. Acc., 1999, 101, 396–408, DOI:10.1007/s002140050457.
- F. Neese, ORCA: An ab initio, DFT and semiempirical electronic structure package, version 2.9.1, Universität Bonn, Bonn, Germany, 2012 Search PubMed.
- F. Neese, Calculation of the zero-field splitting tensor on the basis of hybrid density functional and Hartree-Fock theory, J. Chem. Phys., 2007, 127, 164112, DOI:10.1063/1.2772857.
- M86-EXX229V1 APEX3 User Manual, Bruker AXS Inc., Madison, USA, 2016 Search PubMed.
- G. M. Sheldrick, SHELXT – Integrated space-group and crystal-structure determination, Acta Crystallogr., Sect. A: Found. Adv., 2015, 71, 3–8, DOI:10.1107/S2053273314026370.
- G. M. Sheldrick, Crystal structure refinement with SHELXL, Acta Crystallogr., Sect. C: Struct. Chem., 2015, 71, 3–8, DOI:10.1107/S2053229614024218.
- CCDC 2515971: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qg2cn.
| This journal is © the Partner Organisations 2026 |