Open Access Article
Open Access ArticleCyano-bridged {Ln2IIIFe2III} molecular squares (Ln = Gd, Tb, Dy, Ho, and Er): tuning the slow magnetic relaxation and magnetocaloric effects in zero-dimensional lanthanide Prussian blue analogues
Maria-Gabriela
Alexandru
 a,
Diana
Visinescu
a,
Diana
Visinescu
 *b,
Sergiu
Shova
c,
Nicolas
Moliner
d,
Mario
Pacheco
de,
Miguel
Julve†
d and
Francesc
Lloret
*d
*b,
Sergiu
Shova
c,
Nicolas
Moliner
d,
Mario
Pacheco
de,
Miguel
Julve†
d and
Francesc
Lloret
*d
aDepartment of Inorganic Chemistry, Physical Chemistry and Electrochemistry, Faculty of Chemical Engineering and Biotechnologies, National University of Science and Technology Politehnica of Bucharest, 1-7 Gh. Polizu Street, 011061 Bucharest, Romania
bCoordination and Supramolecular Chemistry Laboratory, Ilie Murgulescu Institute of Physical Chemistry, Romanian Academy, Splaiul Independentei 202, Bucharest 060021, Romania. E-mail: dianavisinescu@icf.ro
cPetru Poni Institute of Macromolecular Chemistry, Romanian Academy, Aleea Grigore Ghica Vodă 41-A, RO-700487 Iasi, Romania
dDepartament de Química Inorgànica/Instituto de Ciencia Molecular, Universitat de València, C/Catedrático José Beltrán 2, 46980 Paterna, València, Spain. E-mail: francisco.lloret@uv.es
eFacultad de Química, Universidad de la República, Av. Gral. Flores 2124, 11800 Montevideo, Uruguay
First published on 3rd February 2026
Abstract
An isostructural series of neutral cyano-bridged tetranuclear iron(III)–lanthanide(III) complexes of general formula {[Fe(htpzb)(CN)(μ-CN)2]2[Ln(dmbpy)(NO3)2(H2O)]2}·2CH3CN·2H2O [Ln = Gd (1), Tb (2), Dy (3), Ho (4), and Er (5); htpzb = hydrotris(pyrazolyl)borate and dmbpy = 4,4′-dimethyl-2,2′-bipyridine] was synthesized and structurally and magnetically characterized. Single-crystal X-ray analysis of 1–5 revealed the formation of neutral cyano-bridged {FeIII2LnIII2} complexes (Ln = Gd, Tb, Dy, Ho, and Er) of square-like topology that crystallize in the triclinic P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) space group. Solid-state direct-current magnetic susceptibility analysis evidenced weak intramolecular antiferromagnetic FeIII–LnIII interactions in 1 (Ln = Gd) together with large local magnetic anisotropies from the LnIII ion in 2–5 (Ln = Tb, Dy, Ho, and Er). Frequency-dependent alternating current magnetic susceptibility signals occurred for 1–5 under an applied dc magnetic field of H = 1.0 (1) or 0.5 T (2–5), indicating field-induced slow magnetic relaxation effects typical of single-molecule magnets. Depending on the non-Kramer (Tb, Ho) or Kramer (Gd, Dy, Er) nature of the LnIII ion, a single magnetic relaxation process via Orbach or Raman mechanism (2 and 4) or a multiple magnetic relaxation process that combines Orbach or Raman plus quantum tunneling of magnetization and/or direct (1, 3, and 5) mechanisms occurred along this series. 1–5 showed large magnetocaloric effects with a high to moderate maximum value of the magnetic entropy change at optimum working temperatures just above He liquefaction [−ΔSmax = 16.51 (1), 5.42 (2), 6.02 (3), 4.56 (4), and 5.86 J kg−1 K−1 (5) for H = 5 T at Topt = Tmax = 2 (1), 4 (2, 3 and 5), and 6 K (4)], as well as a high to moderate magnetocaloric index at rather low optimum working fields [MCI = 6.4 (1), 3.3 (2), 4.7 (3), 0.9 (4), and 3.6 J kg−1 K−1 T−1 (5) for Hopt = Hmax = 1.0 (1), 0.6 (2), 0.4 (3), 0.8 (4), and 0.6 T (5) at T = 2 K].
space group. Solid-state direct-current magnetic susceptibility analysis evidenced weak intramolecular antiferromagnetic FeIII–LnIII interactions in 1 (Ln = Gd) together with large local magnetic anisotropies from the LnIII ion in 2–5 (Ln = Tb, Dy, Ho, and Er). Frequency-dependent alternating current magnetic susceptibility signals occurred for 1–5 under an applied dc magnetic field of H = 1.0 (1) or 0.5 T (2–5), indicating field-induced slow magnetic relaxation effects typical of single-molecule magnets. Depending on the non-Kramer (Tb, Ho) or Kramer (Gd, Dy, Er) nature of the LnIII ion, a single magnetic relaxation process via Orbach or Raman mechanism (2 and 4) or a multiple magnetic relaxation process that combines Orbach or Raman plus quantum tunneling of magnetization and/or direct (1, 3, and 5) mechanisms occurred along this series. 1–5 showed large magnetocaloric effects with a high to moderate maximum value of the magnetic entropy change at optimum working temperatures just above He liquefaction [−ΔSmax = 16.51 (1), 5.42 (2), 6.02 (3), 4.56 (4), and 5.86 J kg−1 K−1 (5) for H = 5 T at Topt = Tmax = 2 (1), 4 (2, 3 and 5), and 6 K (4)], as well as a high to moderate magnetocaloric index at rather low optimum working fields [MCI = 6.4 (1), 3.3 (2), 4.7 (3), 0.9 (4), and 3.6 J kg−1 K−1 T−1 (5) for Hopt = Hmax = 1.0 (1), 0.6 (2), 0.4 (3), 0.8 (4), and 0.6 T (5) at T = 2 K].
Introduction
Cyano-bridged metal complexes represent a dynamic and resourceful class of multifunctional molecular materials unveiling new facets that have continuously expanded their area of application in thermo-,1–3 photo-,4,5 and electrocatalysis,6 host–guest chemistry,7 energy storage,8–10 optical sensing,11 and thermometry12 among others. More specifically, magnetic molecules containing transition metal ions linked by the light, asymmetric CN− ligand, with a strong σ-donor and π-acceptor character, have been particularly attractive in magnetochemistry, as unique examples of cyano-bridged mixed-3d/nd molecule-based magnetic materials, so-called low-dimensional Prussian blue analogues (PBA).13,14 In the past decade, however, the interest in this field is moving toward the related rare-earth hexacyanometallate derivatives, referred to as lanthanide Prussian blue analogues (Ln PBA).15–23 This family of neutral or anionic cyano-bridged mixed-3d/4f molecule-based magnetic materials combine the intrinsic chemical versatility and structural stability of PBAs with the magnetic and magnetocaloric or optical (luminescent) properties of the lanthanide ions along with the redox and/or catalytic activities of the diamagnetic or paramagnetic first-row transition metal ions (and, occasionally, the organic ligands).The high-spin ground state together with the large magnetic anisotropy exhibited by the majority of the lanthanide(III) ions, other than gadolinium(III), are crucial factors for the occurrence of the slow magnetic relaxation (SMR) effects in single-ion/molecule/chain magnets (SIM/SMM/SCM).21–23 Otherwise, the isotropic gadolinium(III) ion, with a large spin ground state (SGd = 7/2), has been intensively exploited in molecular magnetic coolants because of its large magnetocaloric effects (MCE). Lanthanide metal complexes have therefore become promising components in the development of emerging technologies like quantum information processing (QIP) or molecular spintronics,24,25 and adiabatic demagnetization refrigeration (ADR).26–29 The assembly of high-nuclearity isotropic GdIII complexes or mixed MIII–GdIII derivatives, with a large change of magnetic entropy (−ΔSm) with the magnetic field sweeps (ΔH) at ultra-low temperature (below liquid He) is an important topic in ADR.26–29 Conversely, the anisotropic Ln3+ complexes were rarely investigated in conventional ADR.30–39 Likewise, the nd/4f heterometallics were appealing for the investigation of MCE,40–42 and the cyano-bridged complexes of transition and lanthanide(III) cations provide a largely unexplored area of study.43
The rich library of heteroleptic polycyanometallate mononuclear complexes, with two-/three-/four- and five CN− groups substituted with several blocking ligands, was the subject of several review papers44–51 that highlighted their efficiency in acting as metalloligands to obtain low-dimensional heterobi-44–49 and trimetallic cyano-bridged nd/nd′ coordination compounds.50,51 In contrast, cyano-bridged 3d/4f coordination compounds were much less studied. For example, the reaction of tri- or tetracyanometallates with Ln3+ ions, capped with solvent molecules or chelating ligands, afforded a large variety of low-dimensional Ln PBAs, from: 0D oligonuclear complexes,52,53 1D54–56 and 2D56 coordination polymers. Focusing on cyano-bridged 3d/4f tetranuclear complexes, only the low-spin (LS) [FeIII(bpy)(CN)4]− (bpy = 2,2′-bipyridine)53 as well as [FeIII(htpzb)(CN)3]− complex anions [htpzb = hydrotris(pyrazolyl)borate]57–60 led to square-like {FeIII(LS)2LnIII2} motifs. Our work on cyano-bridged molecular squares brought magnetic insights for the related series of general formula {[FeIII(htpzb)]2[LnIII(NO3)x(pyim)y(Ph3PO)z]2} [Ln = La (x = 1, y = 2, z = 0); Ln = Gd, Tb, and Dy (x = 1, y = 1, z = 0); LnIII = Ce, Eu, Sm, Pr, Nd, Gd, Tb, Dy, and Er (x = 1, y = 1, z = 1); pyim = 2-(1H-imidazol-2-yl)pyridine; Ph3PO = triphenylphosphine oxide] showing a weak antiferromagnetic exchange interaction between FeIII(LS) and LnIII ions through the cyanide bridge (Ln = Eu, Sm, Nd, Gd, Tb, Dy, and Er).57–59 SMR occurred for the Tb3+, Dy3+ and Er3+ derivatives including pyim and PPh3O as capping ligands, showing how small changes in the LnIII coordination sphere can suppress quantum tunneling effects to generate 3d/4f SMMs.59
Cyano-bridged 3d/4f square-like motifs were also assembled from ion(III) hexacyanometallates and Ln(III) ions capped with bidentate o-phenatroline or tridentate 2,2′:6′,2″-terpyridine ligands.61–63 However, the simultaneous employment of two structure control elements, tricyanometallates as bis-monodentate angular connector and auxiliary bidentate blocking ligands (e.g., pyim) on coordination sites of LnIII ions to prevent polymerization, proved to be a robust approach to achieve cyano-bridged mixed-3d/4f squares as unique examples of 0D Ln PBAs. Besides, the inclusion of an additional flexible coligand (e.g., Ph3PO) allowed the fine tuning of the lanthanide(III) environment that could further influence the magnetic behavior of the tetranuclear square complex. Therefore, we envisaged to extend the cyano-bridged square-shaped family of {FeIII(LS)2LnIII2} complexes by using sterically hindered bipyridine-type chelating organic molecules as capping ligands of LnIII coordination sites. Herein, we report on the new cyano-bridged square-shaped tetranuclear complexes of general formula {[Fe(htpzb)(CN)(μ-CN)2]2[Ln(dmbpy)(NO3)2(H2O)]2}·2CH3CN·2H2O [Ln = Gd (1), Tb (2), Dy (3), Ho (4), and Er (5); htpzb = hydrotris(pyrazolyl)borate and dmbpy = 4,4′-dimetyl-2,2′-bipyridine]. Besides the static and dynamic magnetic properties, we also examined the magnetocaloric properties of 1–5 to determine the influence of the nature of the LnIII ion (Ln = Gd, Tb, Dy, Ho, and Er) on the spin dynamics and thermodynamics of the resulted {FeIII(LS)2LnIII2} complexes.
Experimental
Materials
Ln(NO3)3·6H2O, dmbpy and acetonitrile were purchased from commercial sources and used as received. Ph4P[Fe(htpzb)(CN)3]·H2O was prepared as described in the literature.64Synthesis of the compounds
1–5 were prepared by following the same general method: an acetonitrile solution (20 cm3) of Ph4P[Fe(htptz)(CN)3]·H2O (0.067 mmol) was poured into an acetonitrile solution (20 cm3) containing dmbpy (0.067 mmol), and the corresponding lanthanide salt, Ln(NO3)3·xH2O (0.067 mmol) [LnIII = Gd, 1 (x = 6); Tb, 2 (x = 6); Dy, 3 (x = 5); Ho, 4 (x = 6); Er, 5 (x = 6)]. The slow evaporation of the resulting red-orange solutions at room temperature afforded red-colored plates of 1–5 after one week. All the isolated crystalline products were air stable. Yield: 50 (1), 45 (2), 60 (3), 40 (4), 80 (5). Anal. calcd for C52H58N28B2O16Fe2Gd2 (1): C, 35.07; H, 3.26; N, 22.03; found: C, 35.31; H, 3.28; N, 22.14%. C52H58N28B2O16Fe2Tb2 (2): C, 35.00; H, 3.25; N, 22.67; found: C, 35.30; H, 3.19; N, 22.63%. C52H58N28B2O16Fe2Dy2 (3): C, 34.86; H, 3.24; N, 21.90; found: C, 35.04; H, 3.19; N, 22.06%. C52H57N28B2O15.5Fe2Ho2 (4): C, 34.94; H, 3.19; N, 21.95; found: C, 35.03; H, 3.15; N, 22.08%. C52H57B2N28O15.5Fe2Er2 (5): C, 34.85; H, 3.18; N, 21.89; found: C, 34.89; H, 3.14; N, 21.95%. Selected IR peaks (cm−1, KBr pellets): 2149(m) (1), 2153(s) and 2123(m) (2), 2153(s) and 2122(m) (3), 2155(s) and 2123(m) (4, 5) [ν(C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N)].
N)].
Physical techniques
Elemental analyses (C, H, N) and energy dispersive X-ray (EDX) analyses (Fe, Ln) for 1–5 were performed using a PerkinElmer 2400 analyzer and a Philips XL-30 scanning electron microscopy (SEM), respectively. The infrared spectra of 1–5 were recorded on a FTIR Bruker Tensor V-37 spectrophotometer using KBr pellets in the range 4000–400 cm−1. UV-Vis spectra (diffuse reflectance technique) were recorded on a 220–1000 nm domain with a JASCO V-770 spectrophotometer, using MgO as the standard. Powder X-ray diffraction (XRD) measurements of powdered samples of 1–5 were collected at room temperature on a D8 Advance A25 Bruker diffractometer using graphite-monochromated Cu-Kα radiation (λ = 1.54056 Å).Magnetic measurements
Direct current (dc) magnetic susceptibility and magnetization measurements were carried out on crushed crystals of 1–5 (mixed with grease to avoid any crystallite orientation) by using a Quantum Design MPMSXL-5 SQUID magnetometer. The dc magnetic susceptibility measurements were performed in the temperature ranges 2.0–300 K and under applied dc magnetic fields of 0.5 (T > 25 K) and 0.01 T (T ≤ 25 K) to prevent for saturation effects at low temperature. Magnetization measurements were performed in the temperature and field ranges of 2.0–10 K and 0–5 T, respectively, under steps of 1 K and 0.2 T. Alternating current (ac) magnetic susceptibility measurements were recorded with a Quantum Design Physical Property Measurement System (PPMS) in the temperature range 2.0–10 K under ±3.95 G oscillating field at frequencies in the range 1.0–10.0 kHz, both in zero dc field and under applied dc fields of 1.0 (1) or 0.5 T (2–5). The magnetic susceptibility data were corrected for the diamagnetism of the constituent atoms and the sample holder (a plastic bag).Crystallographic data collection and structure determination
Crystallographic measurements for 1–5 were carried out with an Oxford-Diffraction XCALIBUR E CCD diffractometer equipped with graphite-monochromated Mo Kα radiation. The unit cell determination and data integration were carried out using the CrysAlisPro package from Oxford Diffraction.65 The structures were solved with SHELXT program using the intrinsic phasing method and refined by the full-matrix least-squares method on F2 with SHELXL.66,67 Olex2 was used as an interface to the SHELX programs.68 Non-hydrogen atoms were refined anisotropically. Hydrogen atoms attached to carbon were added in idealized positions and refined using a riding model. The hydrogen atoms of the OH groups were located from difference Fourier maps and their positions were refined in agreement with the H-bonds parameters. The main crystallographic data together with structure solution and refinement details are summarized in Table 1 and the corresponding CIF-files. The structural images were obtained with the Diamond 4 program.69 The unit cell parameters and refinement conditions for 1–5 are given in Table 1. CCDC 2504399 (1), 2504400 (2), 2504401 (3), 2504402 (4), 2504403 (5).| 1 | 2 | 3 | 4 | 5 | |
|---|---|---|---|---|---|
| Formula | C52H58N28B2O16Fe2Gd2 | C52H58N28B2O16Fe2Tb2 | C52H58N28B2O16Fe2Dy2 | C52H57N28B2O15.5Fe2Ho2 | C52H57B2N28O15.5Fe2Er2 |
| Fw | 1779.08 | 1782.42 | 1789.58 | 1785.43 | 1790.09 |
| T [K] | 180.05(10) | 159.8(10) | 149.9(2) | 293(2) | 293(2) |
| Space group |
P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) |
P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) |
P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) |
P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) |
P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) |
| a [Å] | 11.6608(4) | 11.6335(4) | 11.6345(4) | 11.6557(3) | 11.6406(4) |
| b [Å] | 11.7464(3) | 11.7416(5) | 11.7335(4) | 11.8334(3) | 11.8371(4) |
| c [Å] | 13.5504(4) | 13.4857(4) | 13.4726(5) | 13.6036(6) | 13.5872(5) |
| α [°] | 72.921(2) | 72.912(3) | 72.844(3) | 72.838(3) | 72.779(3) |
| β [°] | 77.362(3) | 77.393(3) | 77.299(3) | 77.133(3) | 77.078(3) |
| γ [°] | 74.728(3) | 74.761(3) | 74.785(3) | 74.905(3) | 74.954(3) |
| V [Å3] | 1691.28(9) | 1678.88(12) | 1675.39(11) | 1709.13(11) | 1704.96(11) |
| Z | 1 | 1 | 1 | 1 | 1 |
| ρ calc [g cm−3] | 1.747 | 1.763 | 1.774 | 1.735 | 1.743 |
| μ [mm−1] | 2.442 | 2.591 | 2.716 | 2.790 | 2.937 |
| Crystal size [mm] | 0.25 × 0.15 × 0.05 | 0.15 × 0.1 × 0.03 | 0.3 × 0.2 × 0.1 | 0.25 × 0.15 × 0.1 | 0.3 × 0.3 × 0.25 |
| 2θ range | 1.8290–28.9430 | 1.8310–28.7070 | 1.8650–29.1090 | 1.8300–25.5280 | 1.6130–28.5870 |
| Refls collected | 14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 964 964 |
15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 418 418 |
11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 654 654 |
12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 538 538 |
12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 593 593 |
| Indep. refls, Rint | 5416, 0.0386 | 5240, 0.0443 | 5448, 0.0353 | 5028, 0.0441 | 5394, 0.0327 |
| Data/rests/params | 5892/0/464 | 5893/7/475 | 5882/0/467 | 6000/0/464 | 5952/0/464 |
| GOF | 1.059 | 1.029 | 1.074 | 1.016 | 1.039 |
| R 1, wR2 (all data) | R 1 = 0.0312 | R 1 = 0.0383 | R 1 = 0.0353 | R 1 = 0.0535 | R 1 = 0.0334 |
| wR2 = 0.0573 | wR2 = 0.0666 | wR2 = 0.0627 | wR2 = 0.0672 | wR2 = 0.0570 | |
| CCDC no. | 2504399 | 2504400 | 2504401 | 2504402 | 2504403 |
Results and discussion
Synthesis and general physicochemical characterization
The stoichiometric reaction of the anionic [FeIII(htpzb)(CN)3]− metalloligand and the preformed [LnIII(dmbpy)(NO3)2(H2O)]+ (Ln = Gd, Tb, Dy, Ho, and Er) complex cation (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 FeIII/LnIII/dmbpy molar ratio) in acetonitrile solution, at room temperature, afforded X-ray quality crystals of the isostructural series of heterometallic complexes 1–5. They were characterized by elemental (C, H, N) and EDX analyses, as well as FT-IR spectroscopy and powder XRD (see Experimental section).
1 FeIII/LnIII/dmbpy molar ratio) in acetonitrile solution, at room temperature, afforded X-ray quality crystals of the isostructural series of heterometallic complexes 1–5. They were characterized by elemental (C, H, N) and EDX analyses, as well as FT-IR spectroscopy and powder XRD (see Experimental section).
The estimated value of the Fe![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ln molar ratio for 1–5 from EDX analyses is 1
Ln molar ratio for 1–5 from EDX analyses is 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, in agreement with the proposed chemical formula determined from elemental analyses. The FT-IR spectra of 1–5 are very similar and support the formation of cyano-bridged {FeIII(LS)2LnIII2} complexes (Fig. S1–S5 in SI), through two distinct broad absorptions peaks at energies ranging from 2149–2155 and 2122–2123 cm−1 corresponding to the stretching vibrations of the bridging and terminal cyanide ligands, respectively, of the [FeIII(htpzb)(CN)(μ-CN)2]− units. The C–H stretching and ring stretching modes of htpzb and dmbpy ligands cover the range 2950–2750 cm−1 and 1650–1410 cm−1, respectively. The broad and medium intensity band in the range 2514–2553 cm−1 is characteristic for the B–H stretching vibration from the htpzb ligand. For 1–5, the coordinated nitrato ligands were also identified as a strong and broad absorption at ca. 1384–1387 cm−1, as well as a medium intensity band at ca. 812–816 cm−1.70 The solid-state UV-Vis spectra for 1–5 are shown in Fig. S6 in SI and exhibit similar profiles, with a UV band centered at approximately 300 nm, as well as a broad absorption covering the visible region. The intense UV absorption band likely envelops n/π–π* transitions of the dmbpy ligand, as well as ligand-to-metal charge transfer (LMCT) from the cyanide ligands to LSFeIII center.71 The broad and intense absorption at ca. 600 nm could be generated by LMCT transitions (htpz → LSFeIII, with a shoulder at ca. 430 nm) and metal to ligand charge transfer (MLCT) transitions within the {FeIII(htpzb)(CN)3} unit, which likely mask the characteristic, very weak, sharp absorptions of LnIII ions.
1, in agreement with the proposed chemical formula determined from elemental analyses. The FT-IR spectra of 1–5 are very similar and support the formation of cyano-bridged {FeIII(LS)2LnIII2} complexes (Fig. S1–S5 in SI), through two distinct broad absorptions peaks at energies ranging from 2149–2155 and 2122–2123 cm−1 corresponding to the stretching vibrations of the bridging and terminal cyanide ligands, respectively, of the [FeIII(htpzb)(CN)(μ-CN)2]− units. The C–H stretching and ring stretching modes of htpzb and dmbpy ligands cover the range 2950–2750 cm−1 and 1650–1410 cm−1, respectively. The broad and medium intensity band in the range 2514–2553 cm−1 is characteristic for the B–H stretching vibration from the htpzb ligand. For 1–5, the coordinated nitrato ligands were also identified as a strong and broad absorption at ca. 1384–1387 cm−1, as well as a medium intensity band at ca. 812–816 cm−1.70 The solid-state UV-Vis spectra for 1–5 are shown in Fig. S6 in SI and exhibit similar profiles, with a UV band centered at approximately 300 nm, as well as a broad absorption covering the visible region. The intense UV absorption band likely envelops n/π–π* transitions of the dmbpy ligand, as well as ligand-to-metal charge transfer (LMCT) from the cyanide ligands to LSFeIII center.71 The broad and intense absorption at ca. 600 nm could be generated by LMCT transitions (htpz → LSFeIII, with a shoulder at ca. 430 nm) and metal to ligand charge transfer (MLCT) transitions within the {FeIII(htpzb)(CN)3} unit, which likely mask the characteristic, very weak, sharp absorptions of LnIII ions.
The purity of bulk samples of 1–5 was confirmed by the agreement between their experimental and simulated powder XRD patterns (see Fig. S7–S11 in the SI).
Description of the structures
1–5 are isostructural crystallizing in the triclinic P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) space group (Table 1). Selected crystallographic and structural data for 1–5 are given in Tables 1 and 2.
space group (Table 1). Selected crystallographic and structural data for 1–5 are given in Tables 1 and 2.
| 1 | 2 | 3 | 4 | 5 | |
|---|---|---|---|---|---|
| a Symmetry codes: (a) = 1 − x, 1 − y, 1 − z (1, 3–5), 1 − x, 1 − y, 2 − z (2); (b) = 1 + x, y, z (1, 3–5), −1 + x, y, z (2); (c) = 1 − x, 1 − y, 2 − z (1, 3–5), 1 − x, 1 − y, 1 − z (2). b br – bridging. c t – terminal. | |||||
Ln1–Fe1–Ln1a![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) a (°) a (°) |
96.34 | 96.18 | 96.22 | 97.16 | 97.16 |
Fe1–Ln1–Fe1a![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) a (°) a (°) |
83.66 | 83.82 | 83.78 | 82.84 | 82.84 |
| Fe–Ccyanide (Å) | 1.914(3)–1.936(4) | 1.921(4)–1.935(5) | 1.922(4)–1.941(4) | 1.927(6)–1.933(5) | 1.929(4)–1.934(4) |
| Fe–Npyrazolyl (Å) | 1.970(2)–1.998(3) | 1.968(3)–1.994(3) | 1.971(3)–1.997(3) | 1.972(4)–1.996(4) | 1.974(3)–1.998(3) |
(Fe1–C–N)br![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) b (°) b (°) |
176.1(3)/178.3(3) | 177.9(4)/175.8(4) | 178.5(3)/175.6(4) | 175.3(5)/177.0(5) | 175.9(3)/178.6(3) |
(Fe1–C–N)t![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) c (°) c (°) |
177.1(3) | 177.1(3) | 176.5(4) | 178.5(5) | 177.4(4) |
| Ln1–Ndmbpy (Å) | 2.486(3)/2.549(2) | 2.467(3)/2.528(3) | 2.454(3)/2.515(3) | 2.448(4)/2.514(4) | 2.427(3)/2.503(3) |
| Ln1–Onitrate (Å) | 2.483(2)–2.522(2) | 2.479(3)–2.512(3) | 2.433(3)–2.508(3) | 2.416(3)–2.516(3) | 2.406(2)–2.512(2) |
| Ln1–O1 W (Å) | 2.412(2) | 2.386(3) | 2.380(2) | 2.384(3) | 2.368(2) |
(Ln1–Ncyanide)br![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) b (Å) b (Å) |
2.454(3)/2.466(3) | 2.449(3)/2.439(3) | 2.440(3)/2.422(3) | 2.409(5)/2.437(4) | 2.403(3)/2.435(3) |
(Ln1–N–C)br![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) b (°) b (°) |
168.7(3)/173.0(3) | 168.8(3)/173.6(3) | 169.3(3)/174.3(3) | 171.3(4)/175.1(4) | 171.0(3)/174.2(3) |
Fe1⋯Ln1/Fe1a⋯Ln1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) a (Å) a (Å) |
5.50 | 5.48/5.49 | 5.47/5.48 | 5.47/5.50 | 5.46/5.49 |
| (π⋯π)pyrazolyl (Å/°) | 3.60 Å/13.59 | 3.39 Å/17.37 and 17.38 | 3.39 Å/17.46 | 3.44/19.12 | 3.44/19.08 |
| (π⋯π)dmbpy (Å/°) | 3.80/23.57, 22.19 | 3.80/22.31, 23.98 | 3.80/22.64, 24.35 | 3.90/23.75, 24.76 | 3.91/24.09, 24.93 |
| (C–H⋯π)pyrazolyl (Å) | 2.72 Å | 2.78 | 2.79 | 2.87 | 2.88 |
| Fe1⋯Fe1b via (π⋯π)pyrazolyl contactsa (Å) | 7.73 | 8.01 | 8.02 | 8.13 | 8.14 |
| Ln1⋯Ln1c (H-bonds)a (Å) | 6.53 | 6.51 | 6.50 | 6.56 | 6.56 |
The crystal structure of 1–5 consists of a neutral, centrosymmetric cyano-bridged heterotetranuclear unit of square topology, {[Fe(htpzb)(CN)(μ-CN)2]2[Ln(dmbpy)(NO3)2(H2O)]2} [Ln = Gd (1), Tb (2), Dy (3), Ho (4), and Er (5)] (see Fig. 1 and Fig. S12a–S15a in SI), together with two acetonitrile and two water molecules of crystallization. Bond lengths and angles for 1–5 are gathered in Tables S1 and S2 in SI. Water and acetonitrile crystallization molecules are connected through O–H⋯O and O–H⋯N hydrogen bonds to the terminal CN− and NO3− ligands (Fig. S12a–S15a in SI).
The {FeIII2LnIII2} entity in 1–5 is assembled from two bis-monodentate, anionic [FeIII(htpzb)(CN)(μ-CN)2]− and two coordinatively unsaturated, cationic [LnIII(dmbpy)(NO3)2(H2O)]+ units. The cis-orientation of two CN− bridging ligands of the fac-[FeIII(htpzb)(CN)3]− complex anion, together with the complementary labile/free coordination sites on [LnIII(dmbpy)(NO3)2(H2O)]+ complex cation, gave rise to a cyclic distorted square-shaped molecule, the two FeIII and LnIII ions filling alternatively the corners of the square. The Ln1–Fe1–Ln1a and Fe1–Ln1–Fe1a angles in 1–5 vary in the ranges 96.18–97.16 and 82.84–83.82°, respectively, the degree of rhombic distortion being consistent with previous reports.53,57–59
Each crystallographically independent iron(III) ion from 1–5 is six-coordinated by three carbon atoms belonging to CN− ligands, disposed in a fac-arrangement, and three nitrogen atoms from the three pyrazolyl rings of the htpzb ligand, in a slightly distorted octahedral metal surrounding [CShM = 0.153 (1), 0.156 (2), 0.148 (3), 0.129 (4), and 0.126 (5), see Fig. 1b, Fig. S12b–S15b and Table S3 in SI].72,73 The Fe–C and Fe–N bond lengths are similar in 1–5, ranging from 1.914(3)–1.941(4) and 1.968(3)–1.998(3) Å, respectively (Table 2). As far as the Fe–C–N angles from the bridging and terminal cyanide ligands are concerned, their values are close to linearity in 1–5, ranging from 175.3(5)–178.6(3) and 176.5(4)–178.5(5)°, respectively (Table 2). All these values related to the iron(III) surrounding are close to those previously reported for the cyano-bearing [FeIII(htpzb)(CN)3]− moiety in the related homotetranuclear iron(III) complex.74 The two distinct FeIII⋯LnIII intermetallic distances through the CN− bridges in 1–5 range between 5.46–5.50 Å (Table 2) which are similar to previously reported cyano-bridged {LSFeIIILnIII}2 squares.53,57–59 On the other hand, each crystallographically independent lanthanide(III) ion in 1–5 is capped by two N atoms from one dmbpy and four O atoms from two nitrato chelating ligands, the coordination sphere being completed by two N atoms from the CN− bridges and one O atom from the aqua ligand (see Fig. 1c, Fig. S12c–S15c and Table S3 in SI).
The donor atoms around the LnIII ions in 1–5 describe a nine-coordinated polyhedron with a geometry close to distorted tricapped trigonal prism (TCTPR) of D3h symmetry, estimated through SHAPE 2.1 software [CShM = 1.900 (1), 1.829 (2), 1.774 (3), 1.744 (4), and 1.738 (5), see Table S3 in SI], so that the deviations from ideal TCTPR geometry decrease with increasing the number of 4f electrons along this series (n = 7–11).72,73
It is worth mentioning that, in the earlier examples of cyano-bridged {LSFeIIILnIII}2 squares, the nine-coordinated surrounding of LnIII ions is either distorted monocapped square antiprism53 or muffin-like.57–59 The bite angles of the dmbpy and nitrato chelating ligands in 1–5 are within the ranges 64.68(8)–66.22(10) and 51.38(7)–52.66(12)°, respectively (Table 2), being the main source of the polyhedron distortion from ideal tricapped trigonal prism.
The Ln1–Ndmbpy distances in 1–5 are in the range 2.427(3)–2.528(3) Å, being higher than those corresponding to the bridging cyanide ligands in the range 2.403(3)/2.466(3) Å (Table 2). Also, the Ln1–Ndmbpy bond lengths correspond well to the previously published examples including similar fragments.75–79 The two Ln1–N1–C1 and Ln1–N2a–C2a angles involving the bridging cyanide ligands in 1–5, slightly deviate from linearity, being in the ranges of 168.7(3)–175.1(4)°, as other reports of cyano-bridged {LSFeIIILnIII}2 squares.53,57–59 Also, the Ln1–O1W distance in the range 2.368(2)–2.412(2) Å is shorter than the Ln1–O bond lengths from nitrate that vary in the range 2.406(2)–2.522(2) Å (Table 2, symmetry codes (a) = 1 − x, 1 − y, 1 − z for 1, 3–5 and (a) = 1 − x, 1 − y, 2 − z for 2). Overall, the average values of the Ln1–Ndmbpy, Ln1–NCN, Ln1–Nnitrate, Ln1–Onitrate, and Ln1–OW distances decrease with the number of 4f electrons along this series (n = 7–11), as expected from the well-known “lanthanide contraction” phenomenon.
In the crystal lattice of 1–5, the {FeIII2LnIII2} entities are further interconnected through non-covalent, hydrogen bonding, π⋯π, and C–H⋯π interactions to build a three-dimensional supramolecular network (see Fig. 2 and Fig. S16–S23 in SI). The adjacent {FeIII2LnIII2} entities are linked via Oaqua–H⋯N and C–H⋯Onitrate hydrogen bonds involving the crystallization acetonitrile molecules and the peripheral aqua and nitrate ligands, respectively (Table S4 in SI), developing supramolecular double-chains running parallel to the crystallographic a axis (Fig. 2, top and Fig. S16–S19 in SI). Besides, π⋯π and C–H⋯π stacking interactions established between pyrazolyl rings of htpzb ligands belonging to neighboring {FeIII2LnIII2} entities of 1–5 consolidate the supramolecular double-chains (the centroid⋯centroid distance and the angles between the normal to the ring and the centroid–centroid vector are in the ranges 3.39–3.60 Å and 13.59–19.12°, while the C–H⋯π distances are in the range 2.72–2.88 Å, Table 2). Offset π⋯π stacking interactions are established between the pyridyl rings of the outer dmbpy ligands assembling supramolecular layers in the ac crystallographic plane. The neighboring layers interact further along c axis through H-bonds established between peripheral aqua and nitrate ligands to build a 3D supramolecular network (see Table S3, Fig. 2, bottom and Fig. S20–S23 in SI). The closest intermetallic Fe1⋯Fe1b distance in 1–5 across (π⋯π)pyrazolyl contacts is in the range 7.73–8.14 Å, whereas the smallest Ln1⋯Ln1c distance across the H-bonds is in the range 6.50–6.56 Å (Table 2).
Magnetic properties
The magnetic properties of 1–5 have been investigated, both in the dc and ac regimes, to unveil the influence of the spin ground state and magnetic anisotropy of the LnIII ion (Ln = Gd, Tb, Dy, Ho, and Er) on the static and dynamic magnetic behavior of this series of cyano-bridged heterometallic {LnIII2FeIII2} molecular squares as illustrative examples of 0D PBA (see Table S5 in SI).53,57–60 | ||
| Fig. 3 (a) Temperature dependence of χMT for 1–5. (b) Field dependence of M at 2 K for 1–5 (the solid lines are only eye-guides). | ||
At room temperature, the χMT value of 1 is 16.70 cm3 mol−1 K, a value which is close to the one expected for the sum of two magnetically isolated LS 3d5 FeIII ions (SFe = 1/2) with an unquenched orbital momentum (2T2g term in Oh symmetry) plus two orbital free 4f7 GdIII ions (SGd = 7/2) with an 8S7/2 ground state [χMT = 2 × (Nβ2gFe2/3k)SFe(SFe + 1) + 2 × (Nβ2gGd2/3k)SGd(SGd + 1) = 2 × 0.46 + 2 × 7.88 = 16.67 cm3 mol−1 K with gFe = 2.215 and gGd = 2.0] (Fig. 3a). In fact, the 2T2g term of the LS FeIII ion in Oh symmetry is split under a rhombic distortion into a ground doublet state (2A1g) and two excited doublet states (2B1g and 2B2g).
Upon cooling, χMT decreases slightly and quasi-linearly down to 20 K due to the SOC effects of the LS FeIII ions, below this temperature χMT abruptly falling down to reach a minimum value of 13.67 cm3 mol−1 K at 2.0 K. This value is well below that expected for the sum of two magnetically isolated LS FeIII ions at this low temperature plus two isotropic GdIII ions (χMT = 2 × 0.15 + 2 × 7.875 = 16.05 cm3 mol−1 K) which unambiguously supports the occurrence of an antiferromagnetic interaction between the LS FeIII and GdIII ions through the cyanide bridge in 1.
Hence, the maximum M value at 2 K for H = 5 T is 14.8Nβ (Fig. 3b), which is below that expected for the sum of two GdIII and two low-spin FeIII ions, magnetically non-interacting [M = 2 × (gFeSFe + gGdSGd)Nβ = 16.2Nβ (with gGd = 2.0 and gFe = 2.2)], being close to that of a tercet ground state [S = 2(SGd − SFe) = 6] resulting from the antiparallel alignment of the spin momenta of the LS FeIII and GdIII ions [M = gSNβ = 12.4Nβ with g = (19/28)gGd + (9/28)gFe = 2.06]. Moreover, the reduced isothermal magnetization curves from 2 up to 10 K are almost overlapped (Fig. S24a in SI), in agreement with the small, if not negligible, local magnetic anisotropy of the GdIII ions.
On the other hand, the χMT values at room temperature for 2–5 are equal to 25.17 (2), 29.10 (3), 28.70 (4), and 23.79 cm3 mol−1 K (5) (Fig. 3a). These values well correspond to the theoretical ones for the sum of two magnetically non-interacting LS 3d5 FeIII ions with a significant orbital contribution [χMT = 2 × (Nβ2gFe2/3k)SFe(SFe + 1) = 2 × 0.46 = 0.92 cm3 mol−1 K with gFe = 2.215] plus two magnetically isolated 4f8 TbIII (2), 4f9 DyIII (3), 4f10 HoIII (4), and 4f11 ErIII ions (5) with 7F6 (JTb = 6, LTb = 3, and STb = 3), 6H15/2 (JDy = 15/2, LDy = 5/2, and SDy = 5/2), 5I8 (JHo = 8, LHo = 3/2, and SHo = 2), and 4I15/2 ground states (JEr = 15/2, LEr = 3/2, and SEr = 3/2) [χMT = 2 × (Nβ2gJ2/3k)J(J + 1) = 23.64 (2), 28.34 (3), 28.14 (4), and 22.96 cm3 mol−1 K (5) with gJ = 3/2, 4/3, 5/4, and 6/5, respectively], being well-separated from the first excited states that are then unpopulated in all the temperature range investigated. Upon cooling, χMT for 2–5 (Fig. 3a) continuously decreases down to ca. 150 K, below this temperature χMT sharply dropping to attain a minimum value of 13.36 (2), 18.04 (3), 6.10 (4), and 12.75 cm3 mol−1 K (5) at 1.9 K. Overall, this magnetic behavior obeys to the occurrence of a large SOC of the magnetically anisotropic LnIII ions (Ln = Tb, Dy, Ho, and Er) in 2–5, together with an antiferromagnetic interaction between the LS FeIII and LnIII ions through the cyanide bridge, as observed in 1. In fact, the isothermal magnetization curves for 2–5 at 2.0 K are far from reaching saturation, and M attains maximum values of 10.46 (2), 10.92 (3), 11.92 (4), and 9.32Nβ (5) for H = 5 T (Fig. 3b). These values are far below those expected for the corresponding ground states [J = 2(JLn − SFe) = 11 (2), 14 (3), 15 (4), and 14 (5)] resulting from the antiparallel alignment of the total momenta of the LS FeIII and LnIII ions [M = gJJNβ = 19.15 (2), 22.88 (3), 23.76 (4), and 21.70 Nβ (5) with gJ = 1.74, 1.63, 1.58, and 1.55, respectively]. Also, the reduced isothermal magnetization curves for 2–5 from 2 to 10 K did not collapse, indicating a significant local magnetic anisotropy of the LnIII ions (Ln = Tb, Dy, Ho, and Er, see Fig. S24b–e in SI).
The 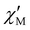 and
and 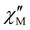 vs. T plots of 1–5 (where
vs. T plots of 1–5 (where  and
and 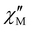 represent the in-phase and out-of-phase components of the molar ac magnetic susceptibility per {LnIII2FeIII2} unit) under an applied dc field (H) of 1.0 (1) or 0.5 T (2–5) are depicted in Fig. 4. No
represent the in-phase and out-of-phase components of the molar ac magnetic susceptibility per {LnIII2FeIII2} unit) under an applied dc field (H) of 1.0 (1) or 0.5 T (2–5) are depicted in Fig. 4. No 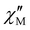 signal occurred at zero dc field for any member of this series (data not shown), as expected for a fast magnetic relaxation due to the quantum tunneling of magnetization (QTM). Under a small dc field [H = 1.0 (1) and 0.5 T (2–5)], however, very weak but not negligible frequency-dependent
signal occurred at zero dc field for any member of this series (data not shown), as expected for a fast magnetic relaxation due to the quantum tunneling of magnetization (QTM). Under a small dc field [H = 1.0 (1) and 0.5 T (2–5)], however, very weak but not negligible frequency-dependent 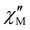 maxima appeared, which are indicative of a slow magnetic relaxation typical of field-induced SMMs. This situation contrasts with that found for the other reported examples of cyano-bridged {FeIII2LnIII2} squares, which only in a few cases show incipient frequency-dependent
maxima appeared, which are indicative of a slow magnetic relaxation typical of field-induced SMMs. This situation contrasts with that found for the other reported examples of cyano-bridged {FeIII2LnIII2} squares, which only in a few cases show incipient frequency-dependent 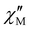 signals, but with no maxima, in the presence of a small dc field (Table S5).57–59 In particular, the isostructural series of complexes of general formula [Fe{hbpzb}(CN)(μ-CN)2Ln(NO3)2(pyim)(Ph3PO)]2·2CH3CN [Ln = Tb, Dy, and Er; hbpzb = hydrotris(pyrazolyl)borate, pyim = 2-(1H-imidazol-2-yl)pyridine, and Ph3PO = triphenylphosphine oxide] showed very incipient
signals, but with no maxima, in the presence of a small dc field (Table S5).57–59 In particular, the isostructural series of complexes of general formula [Fe{hbpzb}(CN)(μ-CN)2Ln(NO3)2(pyim)(Ph3PO)]2·2CH3CN [Ln = Tb, Dy, and Er; hbpzb = hydrotris(pyrazolyl)borate, pyim = 2-(1H-imidazol-2-yl)pyridine, and Ph3PO = triphenylphosphine oxide] showed very incipient 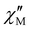 signals at H = 0.1 or 0.25 T.59
signals at H = 0.1 or 0.25 T.59
The magnetic relaxation times (τ) at H = 1.0 (1) or 0.5 T (2–5) can be alternatively calculated from the least-squares fitting of the respective 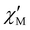 or
or 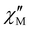 vs. ν plots through the generalized Debye model [eqn (1) and (2)] (Fig. S25 in SI).80 The τ values calculated from the
vs. ν plots through the generalized Debye model [eqn (1) and (2)] (Fig. S25 in SI).80 The τ values calculated from the 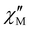 vs. ν plots are fairly more accurate than those from the
vs. ν plots are fairly more accurate than those from the 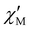 vs. ν plots, as far as the magnetic relaxation rate is equal to the angular frequency of the oscillating ac field at the
vs. ν plots, as far as the magnetic relaxation rate is equal to the angular frequency of the oscillating ac field at the 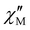 maxima (τ−1 = ωmax = 2πνmax). They are represented in Fig. 5 in the form of the ln
maxima (τ−1 = ωmax = 2πνmax). They are represented in Fig. 5 in the form of the ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) τ vs. 1/T (so-called Arrhenius) and ln
τ vs. 1/T (so-called Arrhenius) and ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) τ vs. ln
τ vs. ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) T plots. On the other hand, the
T plots. On the other hand, the 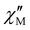 vs.
vs. (so-called Cole–Cole or Argand) plots for 2–5 in the temperature range 2.0–6.0 K give almost perfect semicircles, while those for 1 in the temperature range 2.0–9.0 K yield minor arcs in the low-temperature range and semicircles above 6 K (Fig. S26 in SI). The adiabatic (χS) and isothermal (χT) magnetic susceptibilities as well as the exponential factor (α) for 1–5 were accurately obtained from the least-squares fitting of the respective Argand plots (Fig. S27 in SI).
(so-called Cole–Cole or Argand) plots for 2–5 in the temperature range 2.0–6.0 K give almost perfect semicircles, while those for 1 in the temperature range 2.0–9.0 K yield minor arcs in the low-temperature range and semicircles above 6 K (Fig. S26 in SI). The adiabatic (χS) and isothermal (χT) magnetic susceptibilities as well as the exponential factor (α) for 1–5 were accurately obtained from the least-squares fitting of the respective Argand plots (Fig. S27 in SI).
The relatively low values of the α parameter [α = 0.18–0.65 (1)/0.15–0.41 (2)/0.32–0.41 (3)/0.23–0.47 (4)/0.20–0.31 (5); Fig. S27 in SI] supports a narrow distribution of τ values in the magnetic relaxation process for 1–5 typical of SMMs (α = 0 and 1 for single and infinite relaxation processes, respectively).
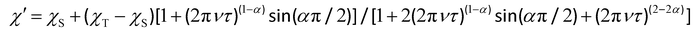 | (1) |
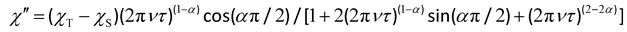 | (2) |
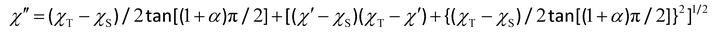 | (3) |
The Arrhenius plots for 2 and 4 give straight lines indicating an Orbach (ORB) spin–lattice relaxation mechanism [eqn (4)]. However, experimental data for 1, 3, and 5 deviate from linearity at low temperatures and they tend to saturate (3 and 5) or really saturate (1) because of the aforementioned temperature-independent QTM [eqn (5)]. Hence, the QTM process is completely or partly suppressed after applying the dc field along this series of {FeIII2LnIII2} squares, depending on the non-Kramer (Tb, Ho) or Kramer (Gd, Dy, Er) nature of the LnIII ion, respectively. Satisfactory least-squares fits were obtained through the appropriate expressions derived from a single magnetic relaxation process via ORB mechanism (2 and 4) or a double magnetic relaxation process that combines ORB plus QTM (1, 3, and 5) mechanisms (solid lines in Fig. 5a).
τ−1 = τORB−1 = τ0−1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) exp(−Ueff/kBT) exp(−Ueff/kBT) | (4) |
| τ−1 = τQTM−1 | (5) |
| τ−1 = τDIR−1 = AT | (6) |
| τ−1 = τRAM−1 = CTn | (7) |
The resulting magnetic relaxation parameters of 1–5 are given in Table 3 and they are depicted in Fig. S28 in SI. The preexponential factor of the ORB process for 1–5 is within the range observed for other SMMs [τ0 = 3 (1)/41 (2)/9.9 (3)/58 (4)/81 µs (5); Table 3] (Fig. S28b in SI). Notably, the effective energy barrier of the ORB process for the non-Kramer Tb(III) and Ho(III) derivatives 2 and 4 is one order of magnitude smaller than for the Kramer Dy(III) and Er(III) derivatives, 3 and 5 [Ueff = 0.92 (2)/8.8 (3)/0.78 (4)/3.81 cm−1 (5); Table 3] (Fig. S28c). Hence, an overall increase of the Ueff values occurs along this series in the order {FeIII2HoIII2} < {FeIII2TbIII2} < {FeIII2ErIII2} < {FeIII2DyIII2}, while for the previously reported cyano-bridged {FeIII2LnIII2} squares analogues the Er(III) derivative had the largest value of Ueff [Ueff = 13–17 (Tb)/25–28 (Dy)/38–40 cm−1 (Er); Table S5].59 The observed trend would be related to the different magnetic anisotropies of the {FeIII2LnIII2} ground state resulting from the antiparallel alignment of the spin and total momenta of the LS FeIII and LnIII ions [J = 2(JLn − SFe) = 11 (Tb), 14 (Dy), 15 (Ho), and 14 (Er)]. Otherwise, the Ueff value of 18 cm−1 for 1 is extremely large to be associated with a magnetic anisotropy energy barrier given the almost isotropic nature of the GdIII ions (see discussion above). Slow-rate spin-phonon transitions between the vibronic energy levels of the {FeIII2GdIII2} square could be the source of the slower magnetic relaxation in 1. Alternatively, the ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) τ vs. ln
τ vs. ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) T plots, which also give straight lines for 2 and 4 supporting a Raman (RAM) spin–lattice relaxation mechanism [eqn (7)]. Once again, the experimental data for 1, 3, and 5 deviate from linearity at low temperatures due to the aforementioned QTM and/or DIR mechanisms [eqn (5) and (6), respectively]. Satisfactory least-squares fits were thus obtained through the appropriate expressions derived from a single magnetic relaxation process via RAM mechanism (2 and 4) or a multiple magnetic relaxation process that combines RAM plus QTM and DIR (1, 3, and 5) mechanisms (solid lines in Fig. 5b).
T plots, which also give straight lines for 2 and 4 supporting a Raman (RAM) spin–lattice relaxation mechanism [eqn (7)]. Once again, the experimental data for 1, 3, and 5 deviate from linearity at low temperatures due to the aforementioned QTM and/or DIR mechanisms [eqn (5) and (6), respectively]. Satisfactory least-squares fits were thus obtained through the appropriate expressions derived from a single magnetic relaxation process via RAM mechanism (2 and 4) or a multiple magnetic relaxation process that combines RAM plus QTM and DIR (1, 3, and 5) mechanisms (solid lines in Fig. 5b).
| H (T) |
τ
QTM![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) a (µs) a (µs) |
τ
0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) b (µs) b (µs) |
U
eff![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) b (cm−1) b (cm−1) |
Ac (s−1 K−1) | C (s−1 K−n) | n | |
|---|---|---|---|---|---|---|---|
a Coefficient factor for the temperature-independent QTM process (τ−1 = τQTM−1).
b Pre-exponential factor and effective energy barrier for the ORB process [τ−1 = τ0−1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) exp(−Ueff/kBT)].
c Coefficient factor for the DIR process (τ−1 = AT).
d Coefficient factor and exponent for the RAM process (τ−1 = CTn). exp(−Ueff/kBT)].
c Coefficient factor for the DIR process (τ−1 = AT).
d Coefficient factor and exponent for the RAM process (τ−1 = CTn).
|
|||||||
| 1 | 1.0 | 132 | 3 | 18 | — | — | — |
| 152 | — | — | 470 | 3 | 3.9 | ||
| 126 | 3 | 16 | — | 0.04 | 3.4 | ||
| 2 | 0.5 | — | 41 | 0.92 | — | — | — |
| — | — | — | — | 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 100 100 |
0.38 | ||
| 3 | 0.5 | 85 | 9.9 | 8.8 | — | — | — |
| 109 | — | — | 1020 | 50 | 2.8 | ||
| 109 | 61 | 3.3 | — | 33 | 2.8 | ||
| 4 | 0.5 | — | 58 | 0.4 | — | — | — |
| — | — | — | — | 8200 | 0.32 | ||
| 5 | 0.5 | 50 | 81 | 3.8 | — | — | — |
| 51 | — | — | 660 | 10 | 3.6 | ||
| 94 | 95 | 0.05 | 58 | 2.7 | |||
The Raman exponent for 2 and 4 is extremely low and not physically meaningful [n = 0.38 (2)/0.32 (4); Table 3], so that a Raman (n ≥ 2) or even a direct (n = 1) process can be definitely discarded. Yet, the low n value for 1, 3, and 5 agree with a Raman process assisted by optical phonons [n = 3.9 (1)/2.8 (3)/3.6 (5); Table 3] (Fig. S28f in SI), as an alternative to the Orbach process discussed above. Indeed, satisfactory fits were also obtained by consider the simultaneous contributions of Raman, Orbach and QTM mechanisms for 1, 3, and 5, with values of the respective magnetic relaxation parameters within the normal range for SMMs (see Table 3). However, their reliability is clearly limited by the large number of fitting parameters, which could lead to overparameterization. Otherwise, the coefficient factors of the DIR and RAM processes are within the range observed for other SMMs [A = 470 (1)/1020 (3)/660 s−1 K−1 (5) and C = 3 (1)/50 (3)/10 s−1 K−n (5); Table 3], increasing in the order {FeIII2GdIII2} < {FeIII2ErIII2} < {FeIII2DyIII2} along this series (Fig. S28d and S28e in SI). On the other hand, the relaxation times of the QTM process are similar regardless of the magnetic relaxation model employed, either ORB plus QTM [τQTM = 132 (1)/85 (3)/50 µs (5); Table 3] or RAM plus QTM and DIR [τQTM = 152 (1)/109 (3)/51 µs (5); Table 3], increasing in the order {FeIII2ErIII2} < {FeIII2DyIII2} < {FeIII2GdIII2} along this series (Fig. S28a in SI).
Magnetocaloric properties
The magnetocaloric properties of 1–5 have been studied by variable-temperature (T = 2.0–10 K, with ΔT = 1 K) and variable-field (H = 0–5 T, with ΔH = 0.2 T) magnetization measurements to reveal the influence of the spin ground state and magnetic anisotropy of the LnIII ion (Ln = Gd, Tb, Dy, Ho, and Er) on the magnetothermal behavior for this series of cyano-bridged heterometallic {LnIII2FeIII2} molecular squares as illustrative examples of 0D PBA (Table S6 in SI). Selected magnetocaloric data for 1–5 are summarized in Table 4 and they are depicted in Fig. 6. | ||
| Fig. 6 Histogram plots showing the variation of the maximum values (in gravimetric units) of −ΔSm (a) and MCI (b) for 1–5 (data from Table 4). The solid lines are the corresponding optimum values of the temperature and the magnetic field. The inset shows the maximum values (in molar units) of −ΔSm (a) for 1–5. The dotted and dashed lines correspond to the calculated values for two low-spin iron(III) and two lanthanide(III) ions, magnetically isolated or antiferromagnetically coupled, respectively, considering a negligible zero-field splitting (see text). | ||
| H (T) |
T
max![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) b (K) b (K) |
−ΔSmax![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) c (J kg−1 K−1) c (J kg−1 K−1) |
T (K) |
H
max![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) e (T) e (T) |
MCIf (J kg−1 K−1 T−1) | |
|---|---|---|---|---|---|---|
| a Magnetic field value. b Temperature value of the maximum of the magnetic entropy change. c Value of the maximum of the magnetic entropy change (in gravimetric units). d Temperature value. e Magnetic field value of the maximum of the magnetocaloric index. f Value of the maximum of the magnetocaloric index (in gravimetric units). | ||||||
| 1 | 5.0 | <2.0 | 16.51 | 2.0 | 1.0 | 6.4 |
| 2 | 5.0 | 4.0 | 5.42 | 2.0 | 0.6 | 3.3 |
| 3 | 5.0 | 4.0 | 6.02 | 2.0 | 0.4 | 4.7 |
| 4 | 5.0 | 6.0 | 4.56 | 2.0 | 0.8 | 0.9 |
| 5 | 5.0 | 4.0 | 5.86 | 2.0 | 0.6 | 3.6 |
The magnetic entropy change (−ΔSm) after switching on the magnetic field (ΔH = H − H0 = H with H0 = 0) can be estimated from the M vs. H and T plots (M being the gravimetric magnetization per {LnIII2FeIII2} unit), according to the Maxwell equation.81 The corresponding −ΔSmvs. T and H color contour maps and plots for 1–5 are represented in Fig. 7a–e and Fig. S29 in SI, respectively.
Once again, the magnetothermal behavior of 2–5 are qualitatively similar, being different from that of 1, as expected from the magnetically isotropic nature of the GdIII ion compared to the other magnetically anisotropic LnIII ions (Ln = Tb, Dy, Ho, and Er).
For 1, the increase of magnetic field or the decrease of temperature determines a gradual increase of the magnetic entropy change for T > 2 K and H < 5 T (Fig. 7a, Fig. S29a and S29f in SI).
The maximum −ΔSm value (in gravimetric units) is 16.51 J kg−1 K−1 at T = 2 K for H = 5 T (Table 4), being larger than that reported for the only other example of cyano-bridged {FeIII(LS)2GdIII2} tetranuclear complex investigated to date as cryomagnetic refrigerant of formula {[Fe(htpzb)(CN)2(μ-CN)][Gd(H4L)(H2O)2]}2·8H2O·2CH3OH [H6L = N,N′-(2,6-pyridine-dicarboxyl)disalicylhydrazide] (−ΔSmax = 12.70 J kg−1 K−1 at T = 4.0 K for H = 7.0 T; Table S6).43 However, it is smaller than that reported for the related hydroxo/azido-bridged {FeIII(HS)3GdIII2} pentanuclear complex of formula TBA3[Fe3Gd2(N3)15(OH)3(tipaH3)2] (TBA = tetrabutylammonium and tipaH3 = triisopropanolamin) (−ΔSmax = 21.1 J kg−1 K−1 at T = 6.0 K for H = 7.0 T).42
The maximum −ΔSm value (in molar units) of 29.35 J mol−1 K−1 for 1 is smaller than the theoretical value for two LS FeIII and two GdIII ions, magnetically uncoupled [−ΔSm = 2R![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln(2SFe + 1) + 2R
ln(2SFe + 1) + 2R![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln(2SGd + 1) = 5.55R = 45.89 J mol−1 K−1], being somewhat above than that of a tercet ground state [S = 2(SGd − SFe) = 6] resulting from the antiparallel alignment of the spin momenta of the LS FeIII and GdIII ions [−ΔSm = R
ln(2SGd + 1) = 5.55R = 45.89 J mol−1 K−1], being somewhat above than that of a tercet ground state [S = 2(SGd − SFe) = 6] resulting from the antiparallel alignment of the spin momenta of the LS FeIII and GdIII ions [−ΔSm = R![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln(2S + 1) = 2.56R = 21.32 J mol−1 K−1]. This situation conforms to the partial thermal population of the low-lying states of higher multiplicity (S > 6) for the {FeIII(LS)2GdIII2} complex, as expected for a relatively weak antiferromagnetic interaction between the LS FeIII and two GdIII ions through the cyanide bridges found in 1.
ln(2S + 1) = 2.56R = 21.32 J mol−1 K−1]. This situation conforms to the partial thermal population of the low-lying states of higher multiplicity (S > 6) for the {FeIII(LS)2GdIII2} complex, as expected for a relatively weak antiferromagnetic interaction between the LS FeIII and two GdIII ions through the cyanide bridges found in 1.
Unlike 1, the isofield curves of the magnetic entropy change for 2–5 develop a maximum with increasing the magnetic field for H > 2.6 (2), 3.0 (3), 1.0 (4), and 3.6 T (5) (Fig. S29b–e in SI), concomitantly with an intercrossing of the isothermal curves with decreasing the temperature for T < 4.0 (2), 3.0 (3), 5.0 (4), and 3.0 K (5) (Fig. S29g–j in SI). Hence, the maximum −ΔSm values (in gravimetric units) are 5.42 (2), 6.02 (3), 4.56 (4), and 5.86 J kg−1 K−1 (5) at the optimum working temperatures Topt = Tmax = 4.0 (2), 4.0 (3), 6.0 (4), and 4.0 K (5) for H = 5 T (Table 4 and Fig. 6a). Interestingly, the maximum −ΔSm values (in gravimetric units) for 1–5 are within the range observed earlier for mixed-3d/4f 0D PBA derivatives proposed as cryomagnetic refrigerants (−ΔSmax = 3.8–17.7 J kg−1 K−1 at T = 2–8 K for H = 7 T; Table S6 in the SI).82–84 Moreover, they are comparable to those reported for the lanthanide gallium garnets of general formula Ln3Ga5O12 [−ΔSmax = 35.31 (Gd), 11.22 (Tb), and 14.52 J kg−1 K−1 (Dy) at T = 4 (Gd), 3 (Tb), and 2 K (Dy) for H = 3 (Gd), 4 (Tb), and 2 T (Dy)].85,86
The maximum −ΔSm values (in molar units) for 2–5 [−ΔSmax = 9.66 (2), 10.77 (3), 8.14 (4), and 10.48 J mol−1 K−1 (5)] are significantly smaller than for 1 (−ΔSmax = 12.70 J kg−1 K−1) (inset of (Fig. 6a)). Overall, they vary in the order: 1 (Gd) ≫ 3 (Dy) ≈ 5 (Er) > 2 (Tb) > 4 (Ho), despite the increase of the total momentum almost in the inverse order: 4 (JHo = 8) > 3 = 5 (JDy = JEr = 15/2) > 2 (JTb = 6) > 1 (JGd = 7/2). This situation is likely explained because of the increase of the local magnetic anisotropy from the GdIII ion to the other LnIII ions (Ln = Tb, Dy, Ho, and Er) along this series of cyano-bridged {FeIII(LS)2LnIII2} complexes. Hence, the −ΔSmax values for 2–5 are almost five-fold lower than the ones calculated for two low-spin iron(III) and two lanthanide(III) ions, magnetically isolated, considering a negligible zero-field splitting [−ΔSm = 2R![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln(2JFe + 1) + 2R
ln(2JFe + 1) + 2R![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln(2JLn + 1) = 54.17 (2), 57.62 (3), 58.63 (4), and 56.72 J mol−1 K−1 (5)] (dotted line in the inset of Fig. 6a).
ln(2JLn + 1) = 54.17 (2), 57.62 (3), 58.63 (4), and 56.72 J mol−1 K−1 (5)] (dotted line in the inset of Fig. 6a).
Otherwise, they are yet three-fold lower than those calculated for the ground state resulting from the antiparallel alignment of the total momenta of the LS FeIII and LnIII ions [−ΔSm = R![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln(2J + 1) = 26.07 (Tb), 28.00 (Dy), 28.55 (Ho), and 28.00 J mol−1 K−1 (Er) with J = 2(JLn − JFe) = 11 (Tb), 14 (Dy), 15 (Ho), and 14 (Er)] (dashed line in the inset of (Fig. 6a)). In fact, the large energy splitting of the ground state of the magnetically anisotropic LnIII ions into their ±mJ levels strongly minimizes the magnetic entropy at zero field, thereby reducing the maximum magnetic entropy change after switching on the magnetic field.
ln(2J + 1) = 26.07 (Tb), 28.00 (Dy), 28.55 (Ho), and 28.00 J mol−1 K−1 (Er) with J = 2(JLn − JFe) = 11 (Tb), 14 (Dy), 15 (Ho), and 14 (Er)] (dashed line in the inset of (Fig. 6a)). In fact, the large energy splitting of the ground state of the magnetically anisotropic LnIII ions into their ±mJ levels strongly minimizes the magnetic entropy at zero field, thereby reducing the maximum magnetic entropy change after switching on the magnetic field.
On the other hand, the isothermal curves for 1, 2, 3, and 5 rapidly increase monotonically, tending to saturate at lower temperatures (Fig. S29f–h and S29j in SI). In contrast, for 4, the increase of the isothermal curves (Fig. S29i in SI) is more abrupt, the magnetic entropy change diverting at higher temperatures, while a slight tendency of saturation occurred at low temperatures (2 and 3 K). At low magnetic field, H = 1 T, the maximum value of the magnetic entropy change is 5.04 (1), 2.18 (2), 3.75 (3), 1.95 (4), and 2.94 J kg−1 K−1 (5) at 2 (1, 2, 3, 5) and 4 K (4), representing 30 (1), 40 (2), 62 (3), 32 (4), and 50% (5) from the −ΔSmax value. This means that −ΔSm are significant even at weak H comparable with ceramic (0.5–1 T) or Nd-based permanent magnets (1.4 T) and, also, are of interest in cryomagnetic refrigeration applications for which high −ΔSmax values and large slope of the field dependence of −ΔSm plots are required.
In this respect, it is convenient to introduce the magnetocaloric efficiency index (MCI) as a new figure of merit for cryomagnetic refrigerants, defined as MCI = −∂(ΔSm)/∂H, which corresponds precisely to −∂M/∂T according to the Maxwell equation 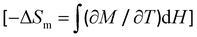 .87 Hence, the isofield and isothermal curves for 1–5 develop narrow to broad maxima of MCI which shift toward higher temperatures or fields with increasing the field or the temperature, respectively (Fig. 7e–i and Fig. S30 in SI). These MCI maxima cover a broad (1) to narrow (2–5) field range [Hmax = 1.0–3.8 (1)/0.6–1.8 (2)/0.4–1.6 (3)/0.8–2.8 (4)/0.6–2.2 T (5) for T = 2–10 K] (Fig. S30f–j in SI), so that the lowest optimum working field [Hopt = Hmax = 1.0 (1)/0.6 (2)/0.4 (3)/0.8 (4)/0.6 T (5) at T = 2 K; Table 4] along this family of cyano-bridged {FeIII(LS)2LnIII2} molecular squares decrease in the order: 1 (Gd) > 4 (Ho) > 2 (Tb) = 5 (Er) > 3 (Dy). The intensity and the location of these MCI maxima in the low-field region are both mandatory for the practical applications of mixed-3d/4f 0D PBA derivatives as magnetic refrigerants at low temperatures, just above the He liquefaction temperature.
.87 Hence, the isofield and isothermal curves for 1–5 develop narrow to broad maxima of MCI which shift toward higher temperatures or fields with increasing the field or the temperature, respectively (Fig. 7e–i and Fig. S30 in SI). These MCI maxima cover a broad (1) to narrow (2–5) field range [Hmax = 1.0–3.8 (1)/0.6–1.8 (2)/0.4–1.6 (3)/0.8–2.8 (4)/0.6–2.2 T (5) for T = 2–10 K] (Fig. S30f–j in SI), so that the lowest optimum working field [Hopt = Hmax = 1.0 (1)/0.6 (2)/0.4 (3)/0.8 (4)/0.6 T (5) at T = 2 K; Table 4] along this family of cyano-bridged {FeIII(LS)2LnIII2} molecular squares decrease in the order: 1 (Gd) > 4 (Ho) > 2 (Tb) = 5 (Er) > 3 (Dy). The intensity and the location of these MCI maxima in the low-field region are both mandatory for the practical applications of mixed-3d/4f 0D PBA derivatives as magnetic refrigerants at low temperatures, just above the He liquefaction temperature.
Hence, the MCI performance for 1 is rather high at the optimum working field (MCI = 6.4 J kg−1 K−1 T−1 for Hopt = 1.0 T at T = 2 K; Table 4). However, high to moderate MCI performances can be obtained using smaller optimum working fields for 2–5 [MCI = 3.3 (2), 4.7 (3), 0.9 (4), and 3.6 J kg−1 K−1 T−1 (5) for Hopt = Hmax = 0.6 (2), 0.4 (3), 0.8 (4), and 0.6 T (5) at T = 2 K; Table 4 and Fig. 6b], approaching the H values of the permanent magnets used commercially (ranging from 0.15 and 2 T for Alnico, a common refrigerator magnet). In this family of cyano-bridged {FeIII(LS)2LnIII2} molecular squares, a decrease of the MCI performance at the optimal working field is found with increasing the magnetic anisotropy in the order: 1 (Gd) > 3 (Dy) > 5 (Er) ≈ 2 (Tb) > 4 (Ho), so that it decreases approximately with increasing the J value in the order: 1 (JGd = 7/2) < 2 (JTb = 6) < 3 = 5 (JDy = JEr = 15/2) < 4 (JHo = 8). Otherwise, the corresponding lanthanide gallium garnets Ln3Ga5O12 show similar MCE performances than 1–5, but at higher optimum working fields [MCI = 8.47 (Gd), 4.90 (Tb), and 6.84 J kg−1 K−1 (Dy), at T = 4 K for Hopt = 2 (Gd), 1 (Tb), and 1 T (Dy)].87
Conclusions
A new isostructural series of cyano-bridged mixed-3d/4f square-shaped tetranuclear complexes was obtained through the “complex-as-ligand/complex-as-metal” strategy by employing heteroleptic iron(III) tricyanometallates with the scorpionate-like hydrotris(pyrazolyl)borate ligand (htpzb) that acts as angular connector toward coordinatively unsaturated lanthanide(III) complexes capped with chelating 4,4′-dimethyl-2,2′-bipyridine (dmbpy) and nitrate ligands.These antiferromagnetically coupled {FeIII2LnIII2} squares (Ln = Gd, Tb, Dy, Ho, and Er) constitute a unique class of field-induced single-molecule magnets (SMMs). Remarkably, {FeIII2GdIII2} is a rare example of quasi-isotropic gadolinium(III)-containing SMM that exhibit slow magnetic relaxation (SMR) under a low applied field (H = 1.0 T) at relatively high blocking temperatures (TB up to 6.0 K). The orbital contribution of the other lanthanides(III) gives rise to significant magnetic anisotropy that, together with their large magnetic moments, provide the main “ingredients” for the assembly of SMMs. The SMR properties of the resulting {FeIII2LnIII2} squares occurred, however, at lower blocking temperatures (TB < 4.0 K) under a lower applied field (H = 0.5 T), being very sensitive to the nature of the magnetically anisotropic LnIII ions (Ln = Tb, Dy, Ho, and Er). Besides, the octahedral low-spin (LS) iron(III) building block with first order orbital moment has a pronounced molecular easy axis of magnetization, being well-suited for the assembly of mixed-3d–4f SMMs.
The magneto-thermal properties along this series of {FeIII2LnIII2} squares also depends on the magnetic anisotropy of the LnIII ions (Ln = Gd, Tb, Dy, Ho, and Er), the large magnetocaloric efficiency being found for the magnetically isotropic {FeIII2GdIII2} square at optimal working temperature and field of 2.0 K and 1.0 T, respectively, while the magnetically anisotropic {FeIII2LnIII2} squares (Ln = Tb, Dy, Ho, and Er) showed moderate to high magnetocaloric efficiencies at higher optimum working temperatures (Topt = 4.0–6.0 K) and lower optimum working fields (Hopt = 0.4–1.0 T). This new family of cyano-based mixed-3d/4f SMMs belonging to the large class of zero-dimensional (0D) lanthanide Prussian blue analogues (Ln PBA) then emerges as a new route toward molecular magnetic coolers operating just above the helium liquefaction temperature. Therefore, the perspectives of exploration MCE in high-dimensional (2D and 3D) lanthanide PBA are wide and promising and could provide very interesting multifunctional molecular materials that advance the knowledge in magnetochemistry and, also, of relevance in adiabatic demagnetization refrigeration (ADR).
Conflicts of interest
There are no conflicts to declare.Data availability
The authors declare that the data supporting this article have been included as part of the supplementary information (SI).Supplementary information: FTIR spectra (Fig. S1–S5, 1–5); UV-Vis spectra for 1–5 (Fig. S6); PXRD patterns (Fig. S7–S11, 1–5); square-like units and FeIII/LnIII coordination polyhedra (Fig. S12–S15, 2–5); drawings of the crystal packing for 2–5 (Fig. S16–S23, 2–5); crystallographic data (Tables S1 and S2, 1–5), SHAPE analysis (Table S3, 1–5) and H-bond parameters (Table S4, 1–5), dc and ac magnetic parameters for cyano-bridged {LnIII2FeIII2} squares (Table S5); dc plots of M vs. HT−1 for 1–5 (Fig. S24); ac plots of χ′ and χ″ vs. ν for 1–5 (Fig. S25); Cole–Cole plots for 1–5 (Fig. S26); temperature dependence of χT, χS, and α parameters plots for 1–5 (Fig. S27); plots showing the variation of the values of τQTM (a), τ0 (b), and Ueff (c), A (d), C (e), and n (f) for the QTM, and ORB, DIR, and RAM relaxation mechanisms in 1–5 series (Fig. S28); selected magnetocaloric data for mixed-3d/nd 0D PBAs proposed as cryomagnetic refrigerants (Table S6); temperature and field dependence of −ΔSm (Fig. S29) and of MCI plots, respectively (Fig. S30) for 1–5. See DOI: https://doi.org/10.1039/d5qi02520f.
CCDC 2504399–2504403 contain the supplementary crystallographic data for this paper.88a–e
Acknowledgements
This work was supported by grants of the Ministry of Research, Innovation and Digitization, CNCS/CCCDI – UEFISCDI, project number project number PN-III-P1-1.1-TE-2019-0352, within PNCDI III and project number PN-IV-P2-2.2-MC-2024-0533, within PNCDIV, the Spanish MINECO (Projects PID2019-109735GB-I00 and Unidad de Excelencia María de Maeztu CEX2019-000919-M), and the Generalitat Valenciana (AICO/2020/183 and AICO/2021/295). M. P. acknowledges the PEDECIBA and CSIC for a postdoctoral stage. The authors would like to thank Dr Rafael Ruíz-García (Institute of Molecular Sciences, University of Valencia) for his helpful discussions on the magnetic properties of these compounds, and for his support in finalizing this manuscript.References
- T. Mwanza, G. S. Kürkçüoğlu, H. Ünver, O. Şahin and O. Yeşilel, Synthesis, spectroscopic, structural characterizations, and catalytic properties of cyanide-bridged heteronuclear metal organic frameworks with imidazole, J. Solid State Chem., 2022, 314, 123344 Search PubMed.
- A. Fonseca, A. L. Bugaev, A. Y. Pnevskaya, K. Janssens, C. Marquez and D. De Vos, Copper-cobalt double metal cyanides as green catalysts for phosphoramidate synthesis, Commun. Chem., 2023, 6, 141 Search PubMed.
- Z. Song, B. Subramaniam and R. V. Chaudhari, Transesterification of Propylene Carbonate with Methanol Using Fe–Mn Double Metal Cyanide Catalyst, Chem. Eng., 2019, 7, 5698 CAS.
- K. Y. Cohen, A. Reinhold, R. Evans, T. S. Lee, H.-Y. Kuo, D. G. Nedd, G. D. Scholes and A. B. Bocarsly, Elucidating the mechanism of photochemical CO2 reduction to CO using a cyanide-bridged di-manganese complex, Dalton Trans., 2022, 51, 17203 RSC.
- K. Y. Cohen, R. Evans, S. Dulovic and A. B. Bocarsly, Using Light and Electrons to Bend Carbon Dioxide: Developing and Understanding Catalysts for CO2 Conversion to Fuels and Feedstocks, Acc. Chem. Res., 2022, 55, 944 CrossRef CAS PubMed.
- B. Zakrzewska, A. Jabłońska, L. Adamczyk, B. Dembińska, A. Kostuch, M. Strawski, I. A. Rutkowska, P. J. Kulesza, M. Marcinek, J. A. Cox and K. Miecznikowski, Pyrolyzed cobalt hexacyanocobaltate dispersed on reduced-graphene-oxide as an electrocatalyst of the oxygen reduction reaction in an alkaline medium, J. Mater. Chem. A, 2023, 11, 7286 Search PubMed.
- M. J. Murphy, K. A. Zenere, F. Ragon, P. D. Southon, C. J. Keper and S. M. Neville, Guest Programmable Multistep Spin Crossover in a Porous 2-D Hofmann-Type Material, J. Am. Chem. Soc., 2017, 139, 1330 Search PubMed.
- J. Liu, M. Zheng, S. Wuc and L. Zhang, Design strategies for coordination polymers as electrodes and electrolytes in rechargeable lithium batteries, Coord. Chem. Rev., 2023, 483, 215084 Search PubMed.
- J.-R. Jiménez, A. Sugahara, M. Okubo, A. Yamada, L.-M. Chamoreau, L. Lisnardand and R. Lescouëzec, A [FeIII(Tp)(CN)3]− scorpionate-based complex as a building block for designing ion storage hosts (Tp: hydrotrispyrazolylborate), Chem. Commun., 2018, 54, 5189 Search PubMed.
- J. J. Zakrzewski, M. Liberka, M. Zychowicz and S. Chorazy, Diverse physical functionalities of rare-earth hexacyanidometallate frameworks and their molecular analogues, Inorg. Chem. Front., 2021, 8, 452 RSC.
- R. Jankowski, M. Wyczesany and S. Chorazy, Multifunctionality of luminescent molecular nanomagnets based on lanthanide complexes, Chem. Commun., 2023, 59, 5961 RSC.
- J.-T. Chen, T.-D. Zhou and W.-B. Sun, Multifunctional lanthanide-based single-molecule magnets exhibiting luminescence thermometry and photochromic and ferroelectric properties, Dalton Trans., 2023, 52, 4643 Search PubMed.
- D. Aguilà, Y. Prado, E. S. Koumousi, C. Mathonière and R. Clérac, Multifunctional lanthanide-based single-molecule magnets exhibiting luminescence thermometry and photochromic and ferroelectric properties, Chem. Soc. Rev., 2016, 45, 203 RSC.
- J.-H. Wang, Z.-Y. Li, M. Yamashita and X.-H. Bu, Switchable Fe/Co Prussian blue networks and molecular analogues, Coord. Chem. Rev., 2021, 428, 23617 Search PubMed.
- C. E. Plecnik, S. Liu and S. G. Shore, Recent progress on cyano-bridged transition-metal-based single-molecule magnets and single-chain magnets, Acc. Chem. Res., 2003, 36, 499 Search PubMed.
- Y.-G. Huang, F.-L. Jiang and M.-C. Hong, Magnetic lanthanide–transition-metal organic–inorganic hybrid materials: From discrete clusters to extended frameworks, Coord. Chem. Rev., 2009, 253, 2814 Search PubMed.
- S. Zhang and P. Cheng, Recent advances in the construction of lanthanide-copper heterometallic metal-organic frameworks, CrystEngComm, 2015, 17, 4250 Search PubMed.
- M. Ferbinteanu, F. Cimpoesu and S. Tanase, Metal-organic frameworks with d-f cyanide bridges: structural diversity, bonding regime, and magnetism, Struct. Bonding, 2015, 163, 185 CrossRef CAS.
- W. Shi, K. Liu and P. Cheng, Transition-lanthanide heterometal-organic frameworks: synthesis, structures, and properties, Struct. Bonding, 2015, 163, 231 Search PubMed.
- A. Dey, P. Bag, P. Kalita and V. Chandrasekhar, Heterometallic CuII–LnIII complexes: Single molecule magnets and magnetic refrigerants, Coord. Chem. Rev., 2021, 432, 213707 CrossRef CAS.
- J. Wang, C.-Y. Sun, Q. Zheng, D.-Q. Wang, Y.-T. Chen, J.-F. Ju, T.-M. Sun, Y. Cui, Y. Ding and Y.-F. Tang, Lanthanide Single-Molecule Magnets: Synthetic Strategy, Structures, Properties and Recent Advances, Chem. – Asian J., 2023, 18, e202201297 Search PubMed.
- R. Marin, G. Brunet and M. Murugesu, Shining New Light on Multifunctional Lanthanide Single-Molecule Magnets, Angew. Chem., Int. Ed., 2021, 60, 1728 Search PubMed.
- K.-Q. Hu, X. Jiang, S.-Q. Wu, C.-M. Liu, A.-L. Cui and H.-Z. Kou, Slow Magnetization Relaxation in NiIIDyIIIFeIII Molecular Cycles, Inorg. Chem., 2015, 54, 1206 Search PubMed.
- H. Weihe, J. Bendix, F. Tuna and S. Piligkos, Toward Molecular 4f Single-Ion Magnet Qubits, J. Am. Chem. Soc., 2016, 138, 5801 Search PubMed.
- M. Shiddiq, D. Komijani, Y. Duan, A. Gaita-Ariño, E. Coronado and S. Hill, Enhancing Coherence in Molecular Spin Qubits via Atomic Clock Transitions, Nature, 2016, 531, 348 Search PubMed.
- X.-Y. Zheng, X.-J. Kong, Z. Zheng, L.-S. Long and L.-S. Zheng, High-Nuclearity Lanthanide-Containing Clusters as Potential Molecular Magnetic Coolers, Acc. Chem. Res., 2018, 51, 517 Search PubMed.
- Y. Yang, Y.-X. Wang, Y.-Z. Leia and P. Cheng, Asymmetric triply bridged lanthanide binuclear clusters with distinctly different magnetic behaviors, Dalton Trans., 2024, 53, 19097 RSC.
- M. Orts-Arroyo, R. Rabelo, A. Carrasco-Berlanga, N. Moliner, J. Cano, M. Julve, F. Lloret, G. De Munno, R. Ruiz-García, J. Mayans, J. Martínez-Lillo and I. Castro, Field-induced slow magnetic relaxation and magnetocaloric effects in an oxalato-bridged gadolinium(III)-based 2D MOF, Dalton Trans., 2021, 50, 3801 RSC.
- A. Mondal, M. Raizada, P. K. Sahu and S. Konar, A new family of Fe4Ln4 (Ln = DyIII, GdIII, YIII) wheel type complexes with ferromagnetic interaction, magnetocaloric effect and zero-field SMM behavior, Inorg. Chem. Front., 2021, 8, 4625 Search PubMed.
- G. Lorusso, O. Roubeau and M. Evangelisti, Rotating Magnetocaloric Effect in an Anisotropic Molecular Dimer, Angew. Chem., Int. Ed., 2016, 55, 3360 CrossRef CAS PubMed.
- P. Konieczny, D. Czernia and T. Kajiwara, Rotating magnetocaloric effect in highly anisotropic TbIII and DyIII single molecular magnets, Sci. Rep., 2022, 1216, 601 Search PubMed.
- Y.-S. Ding and Y.-Z. Zheng, Anisotropic magnetocaloric effect in a dysprosium(III) single-molecule magnet. Commemorating the 100th anniversary of the birth of Academician Guangxian Xu, J. Rare Earths, 2021, 39, 1554 Search PubMed.
- L. Tacconi, A. S. Manvell, M. Briganti, D. Czernia, H. Weihe, P. Konieczny, J. Bendix and M. Perfetti, Exploiting High Order Magnetic Anisotropy for Advanced Magnetocaloric Refrigerants, Angew. Chem., Int. Ed., 2025, 64, e202417582 Search PubMed.
- B. Rodríguez-Barea, J. Mayans, R. Rabelo, A. Sanchis-Perucho, N. Moliner, J. Martínez-Lillo, M. Julve, F. Lloret, R. Ruiz-García and J. Cano, Holmium(III) Single-Ion Magnet for Cryomagnetic Refrigeration Based on an MRI Contrast Agent Derivative, Inorg. Chem., 2021, 60, 12719 Search PubMed.
- N. El Alouani Dahmouni, M. Orts-Arroyo, A. Sanchis-Perucho, N. Moliner, J. Mayans, M. Pacheco, I. Castro, G. De Munno, N. Marino, R. Ruíz-García and J. Martínez-Lillo, Magnetocaloric efficiency tuning through solvent-triggered 3D to 2D interconversion in holmium(III)-based dynamic MOFs, Chem. Commun., 2024, 60, 7451 Search PubMed.
- P. J. Saines, J. A. M. Paddison, P. M. M. Thygesen and M. G. Tucker, Searching beyond Gd for magnetocaloric frameworks: magnetic properties and interactions of the Ln(HCO2)3 series, Mater. Horiz., 2015, 2, 528 RSC.
- J. W. Maciel, L. H. G. Kalinke, R. Rabelo, M. E. Alvarenga, F. T. Martins, N. Moliner and D. Cangussu, Slow Relaxation of Magnetization and Magnetocaloric Effects in One-Dimensional Oxamato-Based Lanthanide(III) Coordination Polymers, Magnetochemistry, 2025, 11, 23 Search PubMed.
- Z. Li, A. Arauzo, J. G. Planas and E. Bartolomé, Magnetic properties and magnetocaloric effect of Ln = Dy, Tb carborane-based metal–organic frameworks, Dalton Trans., 2024, 53, 8969–8979 RSC.
- Z. Li, A. Arauzo, C. Roscini, J. G. Planas and E. Bartolomé, Multifunctional self-refrigerated multivariate {GdLn} (Ln = Dy, Tb, Tb/Eu) metal–organic frameworks, J. Mater. Chem. A, 2024, 12, 21971 RSC.
- J.-L. Liu, Y.-C. Chen, F.-S. Guo and M.-L. Tong, Recent advances in the design of magnetic molecules for use as cryogenic magnetic coolants, Coord. Chem. Rev., 2014, 281, 26–49 Search PubMed.
- P. Konieczny, W. Sas, D. Czernia, A. Pacanowsk, M. Fitta and R. Pełka, Magnetic cooling: a molecular perspective, Dalton Trans., 2022, 51, 12762–12780 Search PubMed.
- S. F. M. Schmidt, M. P. Merkel, G. E. Kostakis, G. Buth, C. E. Ansona and A. K. Powell, SMM behaviour and magnetocaloric effect in heterometallic 3d–4f coordination clusters with high azide: metal ratios, Dalton Trans., 2017, 46, 15661 Search PubMed.
- L.-Z. Cai, W. Zhang, Z.-X. Zhu, J. Ru and M.-X. Yao, Synthesis, structures and magnetic properties of cyano- and amide-bridged FeIII-LnIII tetranuclear heterometallic clusters, J. Coord. Chem., 2019, 72, 1097 Search PubMed.
- R. Lescouëzec, L. M. Toma, J. Vaissermann, M. Verdaguer, F. S. Delgado, C. Ruiz-Pérez, F. Lloret and M. Julve, Design of single chain magnets through cyanide-bearing six-coordinate complexes, Coord. Chem. Rev., 2005, 249, 2691 Search PubMed.
- L. M. Toma, L. D. Toma, F. S. Delgado, C. Ruiz-Pérez, J. Sletten, J. Cano, J. M. Clemente-Juan, F. Lloret and M. Julve, Trans-dicyanobis(acetylacetonato)ruthenate(III) as a precursor to build novel cyanide-bridged RuIII–MII bimetallic compounds [M = Co and Ni], Coord. Chem. Rev., 2006, 250, 2176 Search PubMed.
- S. Wang, X.-H. Ding, Y.-H. Li and W. Huang, Dicyanometalate chemistry: A type of versatile building block for the construction of cyanide-bridged molecular architectures, Coord. Chem. Rev., 2012, 256, 439 CrossRef CAS.
- Y.-H. Li, W.-R. He, X.-H. Ding, S. Wang, L.-F. Cui and W. Huang, Cyanide-bridged assemblies constructed from capped tetracyanometalate building blocks [MA(ligand)(CN)4]1−/2− (MA = Fe or Cr), Coord. Chem. Rev., 2012, 256, 2795 CrossRef CAS.
- S. Wang, X.-H. Ding, J.-L. Zuo, X.-Z. You and W. Huang, Tricyanometalate molecular chemistry: A type of versatile building blocks for the construction of cyano-bridged molecular architectures, Coord. Chem. Rev., 2011, 255, 1713 Search PubMed.
- T. Shiga, N. Mihara and M. Nihei, Cyanide-bridged assemblies with tricyanometalates, Coord. Chem. Rev., 2022, 472, 214763 CrossRef CAS.
- M. Andruh, Heterotrimetallic complexes in molecular magnetism, Chem. Commun., 2018, 54, 3559 Search PubMed.
- M.-G. Alexandru, D. Visinescu, S. Shova, M. Andruh, F. Lloret and M. Julve, Crystal Structures, and Magnetic Properties of Two Novel Cyanido-Bridged Heterotrimetallic {CuIIMnIICrIII} Complexes, Inorg. Chem., 2017, 56, 2258 CrossRef CAS PubMed.
- D. Visinescu, O. Fabelo, C. Ruiz-Perez, F. Lloret and M. Julve, Synthesis, [Fe(phen)(CN)4]−: a suitable metalloligand unit to build 3d–4f heterobimetallic complexes with mixed bpym-cyano bridges (phen = 1,10-phenantroline, bpym = 2,2′-bipyrimidine), CrystEngComm, 2010, 12, 2454 RSC.
- X.-J. Song, J.-J. Xu, Y. Chen, M. Muddassir, F. Cao, R.-M. Wei, Y. Song and X.-Z. You, Synthesis, structures and magnetic properties of cyano-bridged 3d–4f rectangular tetranuclear [FeIII2LnIII2] (Ln = Y, Tb, Dy) compounds containing [FeIII(bpy)(CN)4]− unit, Polyhedron, 2013, 66, 212 Search PubMed.
- M.-G. Alexandru, D. Visinescu, M. Andruh, N. Marino, D. Armentano, J. Cano, F. Lloret and M. Julve, Heterotrimetallic Coordination Polymers: {CuIILnIIIFeIII} Chains and {NiIILnIIIFeIII} Layers: Synthesis, Crystal Structures, and Magnetic Properties, Chem. – Eur. J., 2015, 21, 5429 Search PubMed.
- D. Visinescu, M.-G. Alexandru, D. G. Dumitrescu, S. Shova, N. Moliner, F. Lloret and M. Julve, Cyanido-bridged {FeIIILnIII} heterobimetallic chains assembled through the [FeIII{HB(pz)3}(CN)3]− complex as metalloligand: synthesis, crystal structure and magnetic properties, CrystEngComm, 2021, 23, 4615 RSC.
- D. Visinescu, L. M. Toma, O. Fabelo, C. Ruiz-Pérez, F. Lloret and M. Julve, Low-Dimensional 3d–4f Complexes Assembled, by Low-Spin [FeIII(phen)(CN)4]− Anions, Inorg. Chem., 2013, 52, 1525 CrossRef CAS PubMed.
- M.-G. Alexandru, D. Visinescu, S. Shova, W. X. C. Oliveira, F. Lloret and M. Julve, Design of 3d–4f molecular squares through the [Fe{(HB(pz)3)}(CN)3]− metalloligand, Dalton Trans., 2018, 47, 6005 RSC.
- M.-G. Alexandru, D. Visinescu, B. Braun-Cula, S. Shova, F. Lloret and M. Julve, In situ generation of Ph3PO in cyanido-bridged heterometallic {FeIIILnIII}2 molecular squares (Ln = Eu, Sm), Dalton Trans., 2019, 48, 7532 RSC.
- M.-G. Alexandru, D. Visinescu, B. Cula, S. Shova, R. Rabelo, N. Moliner, F. Lloret, J. Cano and M. Julve, A rare isostructural series of 3d–4f cyanido-bridged heterometallic squares obtained by assembling [FeIII{HB(pz)3}(CN)3]− and LnIII ions: synthesis, X-ray structure and cryomagnetic study, Dalton Trans., 2021, 50, 14640 RSC.
- M.-G. Alexandru, D. Visinescu, J. Cano, F. Lloret and M. Julve, Cyanido-Bridged Heterobimetallic Molecular Squares: Low-Dimensional Models of Prussian Blue Analogues and Beyond, Cryst. Growth Des., 2023, 23, 1288 Search PubMed.
- Y.-F. Huang, H.-H. Wei and M. Katada, A cyano-bridged hetero-tetranuclear [Sm2(o-phen)2(DMF)6(H2O)2((-CN)4Fe2(CN)8]·5H2O·CH3OH: synthesis, structure, Mössbauer spectrum, and magnetism, J. Coord. Chem., 2008, 61, 2683 CrossRef CAS.
- H. Zhou, D.-Y. Yu, H.-B. Zhou and A.-H. Yuan, Cyanide-Bridged Tetranuclear REIII2FeIII2 (RE = Y, Tb, Dy) Molecular Squares with 2,2′:6′,2"-Terpyridine as a Capping Ligand, Z. Anorg. Allg. Chem., 2015, 641, 858 Search PubMed.
- M. Muddassir, X.-J. Song, Y. Chen, F. Cao, R.-M. Wei and Y. Song, Ion-induced diversity in structure and magnetic properties of hexacyanometalate–lanthanide bimetallic assemblies, CrystEngComm, 2013, 15, 10541 Search PubMed.
- Z.-G. Gu, W. Liu, Q.-F. Yang, X.-H. Zhou, J.-L. Zuo and X.-Z. You, Cyano-Bridged FeIII2CuII3 and FeIII4NiII4 Complexes: Syntheses, Structures, and Magnetic Properties, Inorg. Chem., 2007, 46, 3236 Search PubMed.
- Rigaku Oxford Diffraction, CrysAlis Pro Software System, Rigaku Corporation, Oxford, UK, 2015; CrysAlis Pro v. 1.171.38.46 Search PubMed.
- G. M. Sheldrick, SHELXT—Integrated Space-Group and Crystal-Structure Determination, Acta Crystallogr., Sect. A: Found. Crystallogr., 2015, 71, 3 Search PubMed.
- G. M. Sheldrick, Crystal Structure Refinement with SHELXL, Acta Crystallogr., Sect. C: Struct. Chem., 2015, 71, 3 Search PubMed.
- O. Dolman, L. J. Bourhis, R. J. Gildea, J. A. K. Howard and H. Puschmann, OLEX2: A Complete Structure Solution, Refinement and Analysis Program, J. Appl. Crystallogr., 2009, 42, 339 CrossRef.
- H. Putz and K. Brandenburg, Diamond—Crystal and Molecular Structure Visualization, Crystal Impact, Bonn, Germany. Available online: https://www.crystalimpact.com/diamond Search PubMed.
- M. R. Rosenthal, The myth of the non-coordinating anion, J. Chem. Educ., 1973, 50, 331 CrossRef CAS.
- Y. Ye, P. Garrido-Barros, J. Wellauer, C. M. Cruz, R. Lescouëzec, O. S. Wenger, J. M. Herrera and J.-R. Jiménez, Luminescence and Excited-State Reactivity in a Heteroleptic Tricyanido Fe(III) Complex, J. Am. Chem. Soc., 2024, 146, 954 Search PubMed.
- M. Llunell, D. Casanova, J. Cirera, J. M. Bofill, P. Alemany, S. Alvarez, M. Pinsky and D. Avnir, SHAPE: Continuous shape measures of polygonal and polyhedral molecular fragments, 1.1b, University of Barcelona, Barcelona, 2005 Search PubMed.
- D. Casanova, M. Llunell, P. Alemany and S. Alvarez, The Rich Stereochemistry of Eight-Vertex Polyhedra: A Continuous Shape Measures Study, Chem. – Eur. J., 2005, 11, 1479 Search PubMed.
- R. Lescouëzec, J. Vaissermann, F. Lloret, M. Julve and M. Verdaguer, Ferromagnetic Coupling between Low- and High-Spin Iron(III) Ions in the Tetranuclear Complex fac-{[FeIII{HB(pz)3}(CN)2(μ-CN)]3FeIII(H2O)3}·6H2O ([HB(pz)3]− = Hydrotris(1-pyrazolyl)borate), Inorg. Chem., 2002, 41, 5943 CrossRef PubMed.
- Q.-P. Qin, Z.-F. Wang, M.-X. Tan, X.-L. Huang, H.-H. Zou, B.-Q. Zou, B.-B. Shi and S.-H. Zhang, Complexes of lanthanides(III) with mixed 2,2′-bipyridyl and 5,7-dibromo-8-quinolinoline chelating ligands as a new class of promising anti-cancer agents, Metallomics, 2019, 11, 1005 CrossRef CAS PubMed.
- F. A. Mautner, F. Bierbaumer, R. C. Fischer, A. Torvisco, R. Vicente, M. Font-Bardía, À. Tubau, S. Speed and S. S. Massoud, Diverse Coordination Numbers and Geometries in Pyridyl Adducts of Lanthanide(III) Complexes Based on β-Diketonate, Inorganics, 2021, 9, 74 CrossRef CAS.
- E. C. Queiroz, C. H. J. Franco, M. S. Ferreira, R. O. Freire and F. C. Machado, Photoluminescent complexes of Eu(III), Tb(III) and Gd(III) with 3-thiopheneacetate and 4,4′-dimethyl-2,2′-bipyridine: Synthesis, characterization and photophysical properties, J. Lumin., 2022, 249, 118990 Search PubMed.
- P. Hu, F.-P. Xiao, Y. Li, J.-F. Cao, Z.-S. Chen, L.-L. Zhu and W.-P. Huang, One mononuclear single-molecule magnet derived from Dy(III) and dmbpy (dmbpy = 4,4′-dimethyl-2,2′-dipyridyl), Inorg. Chem. Commun., 2017, 84, 207 Search PubMed.
- K. P. Zhuravlev, V. I. Tsaryuka, A. V. Vologzhanina, P. V. Dorovatovskii, Y. V. Zubavichus, V. A. Kudryashova and Z. S. Klemenkova, Structural peculiarities and luminescence of europium dipivaloylmethanates with 2,2′-bipyridine derivatives. Polymorphism of [Eu(DPM)3Bpy], Inorg. Chim. Acta, 2020, 502, 119294 Search PubMed.
- K. S. Cole and R. H. Cole, Dispersion and Absorption in Dielectrics. I. Alternating Current Characteristics, J. Chem. Phys., 1941, 9, 341–351 Search PubMed.
- L. Tocado, E. Palacios and R. Burriel, Entropy determinations and magnetocaloric parameters in systems with first order transitions: Study of MnAs, J. Appl. Phys., 2009, 105, 093918 CrossRef.
- M. Gajewski, R. Pełka, M. Fitta, Y. Miyazaki, Y. Nakazawa, M. Bałanda, M. Reczynski, B. Nowicka and B. Sieklucka, Magnetocaloric effect of high spin cluster with Ni9W6 core, J. Magn. Magn. Mater., 2016, 414, 25 Search PubMed.
- M. Arczynski, M. Rams, J. Stanek, M. Fitta, B. Sieklucka, K. R. Dunbar and D. Pinkowicz, A Family of Octahedral Magnetic Molecules Based on [NbIV(CN)8]4−, Inorg. Chem., 2017, 56, 4021 CrossRef CAS PubMed.
- W. Wen, Y.-S. Meng, C.-Q. Jiao, Q. Liu, H.-L. Zhu, Y.-M. Li, H. Oshio and T. Liu, Ferromagnetic Archimedean polyhedra {Fe24M18} (M = Fe, Ni, and Mn) with tunable electron configurations, Inorg. Chem. Front., 2021, 8, 4239 RSC.
- B. Daudin, R. Lagnier and B. Salce, Thermodynamic Properties of the Gadolinium Gallium Garnet, Gd3Ga5O12, between 0.05 and 25 K, J. Magn. Magn. Mater., 1982, 27, 315 CrossRef CAS.
- M. Kleinhans, K. Eibensteiner, J. C. Leiner, C. Resch, L. Worch, M. A. Wilde, J. Spallek, A. Regnat and C. Pfleiderer, Magnetocaloric Properties of R3Ga5O12 (R = Tb, Gd, Nd, Dy), Phys. Rev. Appl., 2023, 19, 014038 CrossRef CAS.
- N. Terada and H. Mamiya, High-efficiency magnetic refrigeration using holmium, Nat. Commun., 2021, 12, 1212 CrossRef CAS PubMed.
- (a) CCDC 2504399: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q212y; (b) CCDC 2504400: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q213z; (c) CCDC 2504401: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q2140; (d) CCDC 2504402: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q2151; (e) CCDC 2504403: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q2162.
Footnote |
| † Passed away, July 2024. |
| This journal is © the Partner Organisations 2026 |