Open Access Article
Open Access ArticleAl pairing in 8-membered rings drives superior methanol amination on CHA zeolites
Shuang
Liu
a,
Pan
Xu
a,
Sihui
Li
a,
Junyao
Pan
a,
Yuxin
Yan
a,
Rui
Wu
b,
Junhui
Guo
c,
Chang
Liu
*c and
Wenfu
Yan
 *a
*a
aState Key Laboratory of Inorganic Synthesis and Preparative Chemistry, College of Chemistry, Jilin University, Changchun 130012, China. E-mail: yanw@jlu.edu.cn
bChongqing Key Laboratory of Green Catalytic Materials and Technology, College of Chemistry, Chongqing Normal University, Chongqing 401331, China
cSINOPEC (Dalian) Research Institute of Petroleum and Petrochemicals Co., Ltd, Dalian 116045, P.R China. E-mail: liuchang.fshy@sinopec.com
First published on 19th January 2026
Abstract
In protonic acid-catalyzed methanol amination, achieving high selectivity toward methylamine (MMA) and dimethylamine (DMA) while suppressing dimethyl ether (DME) formation remains challenging. The catalytic performance of protonic zeolites in this reaction depends on Brønsted acidity, which is controlled by both the Si/Al ratio and the spatial distribution of framework Al. CHA zeolite is particularly attractive because of its unique pore architecture and tunable acidity. Here, we synthesized CHA zeolites with Si/Al ratios from 3.4 to 4.3 and systematically varied their Al distributions, without using organic structure-directing agents (OSDAs). The synthesis exploited the synergistic effect of alkali metal cations (Na+, Cs+) and CHA seeds with different Al contents (SAPO-34, SSZ-13, and pure silica CHA). The Al content of the seed determined the Si/Al ratio of the final product, while the seed type controlled the Al distribution. At a fixed Si/Al ratio of 3.8, the sample synthesized with a SAPO-34 seed (CHA-S34-3.8) showed the best performance. At 350 °C and 0.813 h−1, and at 400 °C and 4.3 h−1, MMA + DMA yield reached 86.6% and 87.6%, respectively, with no detectable DME after stabilization. These values represent the highest reported performance for methanol amination. Characterization revealed that enrichment of Al pairs in the eight-membered rings, combined with optimal acid density and strength, accounts for the superior activity. This study provides mechanistic insight into the role of Al distribution and offers a strategy to design high-performance zeolite catalysts for methanol amination.
1. Introduction
Monomethylamine (MMA, 3.7 × 3.9 × 4.4 Å), dimethylamine (DMA, 3.9 × 4.7 × 6.0 Å), and trimethylamine (TMA, 3.9 × 5.4 × 6.1 Å) are important intermediates in the production of solvents, agrochemicals, surfactants, and water treatment agents.1,2 Industrially, these amines are produced by reacting methanol with ammonia. However, the reaction thermodynamically favors TMA, even though market demand is greater for MMA and DMA.3,4 Improving selectivity toward MMA and DMA is therefore essential to reduce both energy consumption and downstream separation costs.Early catalysts, including γ-Al2O3 and amorphous silica–alumina, favored TMA formation and produced significant amounts of dimethyl ether (DME) as a by-product.3 To overcome these drawbacks, research has focused on developing catalysts that enhance MMA and DMA selectivity while suppressing side reactions. Zeolites are particularly attractive because of their ordered porosity, high surface area, and tunable Brønsted acidity.5,6 Modified mordenite (MOR) was the first commercial zeolite catalyst used for methanol amination in the 1980s. However, its 12-membered ring (12MR, ∼6.5 × 7.0 Å) channels are too large to restrict the diffusion and formation of TMA.7 This limitation shifted attention toward smaller-pore zeolites.
Zeolites with eight-membered rings (8MR, 3.0–4.5 Å) are especially promising because their pore sizes closely match those of low-substituted amines. Representative 8MR frameworks include LEV, MTF, DDR, UFI, CHA, AFX, PWN, RHO, PAU, KFI, and LTA.1,8–13 Their performance in methanol amination depends on both shape selectivity and Brønsted acidity. A high density of acid sites accelerates methanol conversion but also promotes over-methylation to TMA. Strongly methylated ammonium ions can also block pores, which in turn suppresses DME formation.9 Acid strength plays an equally critical role: excessively strong acidity favors TMA and DME formation, while weak acidity destabilizes methylammonium intermediates, also leading to DME. Moderate acidity strikes the right balance, favoring MMA and DMA production.3,7,8 In addition, the spatial distribution of acid sites strongly influences product selectivity.3
Therefore, methanol amination is governed by three key parameters: acid concentration, acid strength, and acid site distribution. These can be tuned by adjusting the Si/Al ratio and the distribution of framework Al.5,14 Among 8MR zeolites, the CHA framework is especially promising. Its small pores hinder the formation and diffusion of bulky by-products and coke precursors, enabling high selectivity and long catalytic lifetimes.15,16 For example, SAPO-34 and SSZ-13—both CHA-type zeolites—exhibit excellent light olefin selectivity and stability in methanol-to-olefins (MTO) reactions.17–19 These structural advantages suggest that CHA could be optimized for methanol amination by precisely controlling its Si/Al ratio and Al distribution.
Several strategies have been explored to tune Si/Al ratios and Al distributions in CHA, including variation of alkali cations,20 gel composition,21–23 seed crystals,24 and OSDAs.25 OSDA-free synthesis is both more economical and environmentally sustainable. However, under OSDA-free conditions, achieving precise control and Si/Al ratios and Al distributions—and understanding their catalytic impact—remain challenging.24,26
Another persistent challenge is the unavoidable formation of DME as a major by-product. Separating and recovering DME increases process complexity and cost.27 Although adjusting operating parameters such as the ammonia-to-methanol ratio (N/C) and space velocity can partially suppress DME formation, eliminating it through catalyst design remains an open challenge.7,11
In this study, we developed a strategy to regulate Si/Al ratios and Al distributions in CHA zeolites under OSDA-free conditions. The synthesis combined alkali cations with seed crystals of varying Al contents (SAPO-34, SSZ-13, and pure silica CHA). Cesium cations promoted CHA crystallization across a broad Si/Al range. The Al content and dosage of the seed controlled the Si/Al ratio of the final product, while the seed type determined the Al distribution.
Catalytic testing revealed that at the same Si/Al ratio (3.8), the sample synthesized with SAPO-34 seed (CHA-S34-3.8) achieved higher MMA and DMA selectivity than the sample synthesized with pure silica CHA seed (CHA-PS-3.8). Remarkably, no DME was detected under steady-state conditions. Characterization confirmed that CHA-S34-3.8 had a higher fraction of Al located in the 8MRs, as evidenced by Co2+ titration and UV–vis spectroscopy. NH3-TPD showed a high density of acid sites with moderate strength, explaining its superior performance.
Overall, this work demonstrates that tailoring Al distribution through seed selection provides an effective strategy to enhance methanol amination. By combining optimal Si/Al ratios with targeted Al placement in the CHA framework, high MMA and DMA selectivity can be achieved while completely suppressing DME formation.
2. Results and discussion
2.1 Synthesis and characterization of CHA seeds
SAPO-34, SSZ-13 and pure silica CHA (PS-CHA) seeds with different Al contents were synthesized (Fig. S1; synthesis in SI). Powder X-ray diffraction (XRD, Fig. S1a and b) confirmed that all seeds were pure CHA, both before and after calcination, yielding c-SAPO-34, c-SSZ-13, and c-PS-CHA. Thermogravimetric (TG) analysis (Fig. S1c) showed weight loss below 200 °C from physiosorbed water and between 200–800 °C from OSDA decomposition. Combined ICP-OES and TG analyses gave Al2O3 contents of 30.4% (SAPO-34), 9.4% (SSZ-13), and 0% (PS-CHA). Scanning electron microscope (SEM) images (Fig. S1d–f) revealed cubic morphologies with sizes of ∼15 μm (SAPO-34), ∼240 nm (SSZ-13), and ∼20 μm (PS-CHA).2.2 Synthesis and characterizations of CHA zeolites
Five CHA zeolites were synthesized OSDA-free using a gel of 20 SiO2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 Al2O3
1 Al2O3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6 Na2O
6 Na2O![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 Cs2O
1 Cs2O![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2000 H2O and 20 wt.% seed. Products were named CHA-S34-3.4, CHA-S13-4.1, and CHA-PS-4.3, where the final number denotes the Si/Al ratio. By tuning gel composition, CHA-S34-3.8 and CHA-PS-3.8 were also obtained.
2000 H2O and 20 wt.% seed. Products were named CHA-S34-3.4, CHA-S13-4.1, and CHA-PS-4.3, where the final number denotes the Si/Al ratio. By tuning gel composition, CHA-S34-3.8 and CHA-PS-3.8 were also obtained.
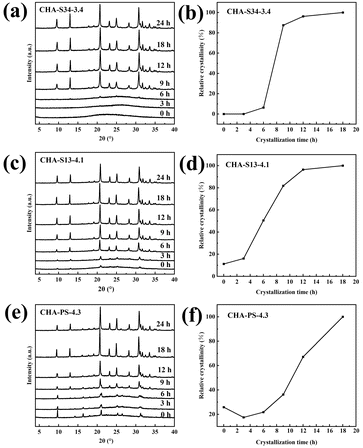
XRD confirmed high crystallinity in all samples (Fig. 1a). SEM showed aggregates of irregular grains (Fig. S2). N2 adsorption isotherms (Fig. 1b) were type I, with micropore areas of 713–745 m2 g−1 and volumes of 0.257–0.265 cm3 g−1 (Table 1), consistent with literature.2829Si MAS NMR (Fig. 1c) showed signals at −99, −105, and −110 ppm, corresponding to Si (2Si, 2Al), Si (3Si, 1Al), and Si (4Si, 0Al), respectively.29 Gaussian deconvolution yielded Si species distributions and Si/Al rations (Table S1), which matched ICP-OES results (Table 1). 27Al MAS NMR (Fig. 1d) exhibited only tetrahedral Al at 59 ppm. For CHA-S34-3.4 and CHA-S34-3.8, 31P MAS NMR (Fig. S3) and ICP-OES confirmed no phosphorus incorporation from SAPO-34 seeds.
| Sample | Si/Ala | Si/Alb |
S
BET![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) c (m2 g−1) c (m2 g−1) |
S micro (m2 g−1) | V total (cm3 g−1) |
V
micro![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) d (cm3 g−1) d (cm3 g−1) |
|---|---|---|---|---|---|---|
| a Determined by the ICP-OES analyses. b Determined by the 29Si NMR MAS analyses. c All samples are H-type. d T-plot method. | ||||||
| CHA-S34-3.4 | 3.4 | 3.5 | 716 | 703 | 0.301 | 0.258 |
| CHA-S13-4.1 | 4.1 | 4.3 | 713 | 703 | 0.305 | 0.257 |
| CHA-PS-4.3 | 4.3 | 4.3 | 745 | 731 | 0.298 | 0.265 |
| CHA-S34-3.8 | 3.8 | 4.0 | 731 | 722 | 0.294 | 0.263 |
| CHA-PS-3.8 | 3.8 | 3.8 | 731 | 722 | 0.308 | 0.263 |
Elemental analysis detected little C or N in SAPO-34- or SSZ-13-derived CHA. In contrast, CHA-PS-4.3 and CHA-PS-3.8 contained 3.51 and 2.9 wt.% C, respectively, indicating OSDA incorporation from PS-CHA seeds. TG profiles (Fig. S4) supported this conclusion. PS-CHA-seeded samples showed two weight-loss steps, including one above 400 °C from OSDA removal, while SAPO-34- and SSZ-13-derived samples displayed only dehydration.30
2.3 Crystallization process
To probe crystallization, we examined CHA-S34-3.4, CHA-S13-4.1, and CHA-PS-4.3, synthesized from identical gels but different seeds. At 0 h, XRD (Fig. 2a, c and e) showed that SSZ-13 and PS-CHA seeds retained CHA diffraction, whereas SAPO-34 appeared amorphous. By 9 h, all systems began crystallizing, and by 18 h, full CHA crystallinity was achieved. A transient GIS phase was observed in PS-CHA (3–12 h) but later disappeared. Crystallization curves showed sigmoidal growth kinetics (Fig. 2b, d and f).31SEM (Fig. S5–S7) revealed a non-classical crystallization pathway: seed dissolution at 0 h, nanoparticle formation by 3–6 h, aggregation by 6–12 h, and assembly into nanograins by 18 h.32 Notably, the grain size of the crystallized products decreases systematically with increasing Al content in the seed. This trend is attributed to the higher solubility of SAPO-34 in alkaline media, which generates a large number of nucleation sites during the early stage of crystallization.
FT-IR spectroscopy tracked short-range structural evolution (Fig. 3a–c). Bands near 780 and 720 cm−1 corresponded to Si–O–Si and Si–O–Al vibrations in single 4-membered rings (s4r). Bands at ∼650 and ∼540 cm−1 were assigned to double 6-membered rings (d6r).33 After 3 h, s4r signals appeared, indicating amorphous aluminosilicate nuclei with short-range order. Characteristic d6r signals of CHA, appeared earlier with SSZ-13 and PS-CHA seeds (3 h) but only after 9 h with SAPO-34, consistent with its slower crystallization in XRD.34
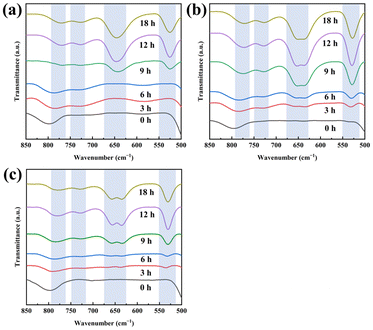 | ||
| Fig. 3 FT-IR spectra of the isolated solid samples throughout the crystallization of (a) CHA-S34-3.4, (b) CHA-S13-4.1, and (c) CHA-PS-4.3. | ||
2.4 Influence of synthesis parameters
Why do c-SAPO-34 and c-SSZ-13 still yield CHA, while c-PS-CHA fails? XRD at 0 h (Fig. S8b) showed that c-SAPO-34 and c-SSZ-13 resembled their uncalcined counterparts, retaining amorphous and CHA features. In contrast, c-PS-CHA-derived gels were fully amorphous. As shown in Fig. 2c, uncalcined PS-CHA displayed CHA peaks at 0 h, whereas c-PS-CHA did not, suggesting seed dissolution after calcination.
Previous studies indicate that negatively charged AlO4− units and OSDAs stabilize frameworks against OH− attack, preserving locally ordered species that promote crystallization.36–38 After calcination, c-SAPO-34 and c-SSZ-13 retain AlO4−, while c-PS-CHA lacks both AlO4− and OSDAs. Consequently, c-PS-CHA cannot maintain sufficient short-range order to initiate CHA crystallization. These results highlight the dual role of OSDA in PS-CHA: directing framework formation and protecting seeds from dissolution, thereby preserving precursor species essential for CHA crystallization.
2.5 Catalytic performance of CHA zeolites
The catalytic activity of five CHA zeolites—H-CHA-S34-3.4, H-CHA-S13-4.1, H-CHA-PS-4.3, H-CHA-S34-3.8, and H-CHA-PS-3.8—was tested under two conditions: (i) 350 °C, N/C = 2, WHSVMeOH = 0.813 h−1 and (ii) 400 °C, N/C = 1, WHSVMeOH = 4.3 h−1. Results are summarized in Table 2, with product trends shown in Fig. 5 and 6.| Sample | N/Ca | WHSVMeOH![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) b (h−1) b (h−1) |
T (°C) | MeOH conversion (%) | Selectivity (%) | (MMA + DMA) Yieldc (%) | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| MMA | DMA | MMA + DMA | TMA | DME | ||||||
| a Molar ratio of the feed gas composition. b Time on stream = 7 h. c MMA plus DMA yield = MeOH conversion × MMA plus DMA selectivity in all carbon base products. | ||||||||||
| H-CHA-S34-3.4 | 2 | 0.813 | 350 | 99.6 | 38.8 | 40.3 | 79.1 | 17.5 | 2.0 | 78.8 |
| H-CHA-S13-4.1 | 2 | 0.813 | 350 | 99.4 | 31.9 | 54.3 | 86.2 | 7.9 | 5.0 | 85.7 |
| H-CHA-PS-4.3 | 2 | 0.813 | 350 | 97.6 | 30.5 | 54.7 | 85.2 | 8.9 | 5.1 | 83.2 |
| H-CHA-S34-3.8 | 2 | 0.813 | 350 | 98.8 | 33.3 | 54.4 | 87.7 | 11.6 | 0 | 86.6 |
| H-CHA-PS-3.8 | 2 | 0.813 | 350 | 97.7 | 29.0 | 53.7 | 82.7 | 11.5 | 5.1 | 80.8 |
| H-CHA-S34-3.4 | 1 | 4.3 | 400 | 98.6 | 21.5 | 55.5 | 77.0 | 17.8 | 4.2 | 75.9 |
| H-CHA-S13-4.1 | 1 | 4.3 | 400 | 98.9 | 26.5 | 57.9 | 84.4 | 8.6 | 5.8 | 83.5 |
| H-CHA-PS-4.3 | 1 | 4.3 | 400 | 98.6 | 25.9 | 60.4 | 86.3 | 7.4 | 4.3 | 84.8 |
| H-CHA-S34-3.8 | 1 | 4.3 | 400 | 98.8 | 24.8 | 63.9 | 88.7 | 10.5 | 0 | 87.6 |
| H-CHA-PS-3.8 | 1 | 4.3 | 400 | 98.9 | 25.3 | 55.8 | 81.1 | 13.6 | 4.2 | 80.2 |
At 350 °C, all catalysts showed stable product distributions, whereas at 400 °C, fluctuation were more pronounced. In both cases, methanol conversion exceeded 97%. The main differences appeared in product selectivity.
For H-CHA-S34-3.4, H-CHA-S13-4.1, and H-CHA-PS-4.3, increasing the Si/Al ratio (3.4 → 4.3) enhanced MMA + DMA selectivity, lowered TMA selectivity, but increased DME formation. Thus, a higher Si/Al ratio suppressed TMA but promoted DME. Notably, H-CHA-S34-3.8 deviated from this trend: despite having a higher Si/Al ratio than H-CHA-S34-3.4, it showed reduced TMA selectivity and no detectable DME. By contrast, H-CHA-PS-3.8 (same Si/Al = 3.8) produced significant DME, indicating that Al distribution—determined by seed—was as important as the Si/Al ratio.
Previous studies suggested that reducing acid density decreases alkylammonium ion formation, leaving more void space for TMA production and diffusion. Strong acidity, on the other hand, drives continuous methylation to TMA.3 In our case, this effect was less evident, likely because the investigated Si/Al ratios covered only a narrow range.7
UV–vis spectra of Co2+-exchanged samples (Fig. 7), deconvoluted by Gaussian fitting, revealed systematic variations in Al distributions (Table 3). While the total fraction of paired Al sites was comparable across all samples, their spatial distribution depended strongly on the seed. The similar paired Al contents likely reflect thermodynamic stabilization dominated by alkali metal cations. SAPO-34-derived CHA (CHA-S34-3.4, CHA-S34-3.8) showed higher τ2Si fractions and lower ω fractions, indicating preferential Al placement in 8MRs. In contrast, PS-CHA-derived CHA (CHA-PS-4.3, CHA-PS-3.8) displayed the opposite trend. SSZ-13-derived CHA (CHA-S13-4.1) exhibited intermediate behavior. These differences arise from the greater abundance aluminosilicate species generated by SAPO-34 during the early stage of crystallization. This enhanced availability provides a kinetic advantage for the incorporation of paired Al into the CHA framework. SSZ-13 and PS-CHA seeds follow the same trend with progressively weaker effects.
 | ||
| Fig. 7 UV–vis spectra of Co2+ exchanged (a) CHA-S34-3.4, (b) CHA-S13-4.1, (c) CHA-PS-4.3, (d) CHA-S34-3.8, and (e) CHA-PS-3.8. | ||
This distinction was clearest between H-CHA-S34-3.8 and H-CHA-PS-3.8 (Si/Al = 3.8). The former contained more τ2Si Al pairs, enriching Brønsted acid sites in 8MRs. This distribution explains its unique performance: suppressed TMA selectivity and complete elimination of DME.
| Sample | Time on stream (h) | N/C | WHSVMeOH (h−1) | T (°C) | MeOH conversion (%) | Selectivity (%) | (MMA + DMA) Yield (%) | Ref. | ||
|---|---|---|---|---|---|---|---|---|---|---|
| MMA | DMA | TMA | ||||||||
| H-CHA-S34-3.8 | 7 | 2 | 0.813 | 350 | 98.8 | 33.3 | 54.4 | 11.6 | 86.6 | This work |
| H-CHA-S34-3.8 | 7 | 1 | 4.3 | 400 | 98.8 | 24.8 | 63.9 | 10.5 | 87.6 | This work |
| H-RHO-8.7 | 3 | 2 | 0.813 | 350 | 96.8 | — | — | 13.59 | 75.3 | 11 |
| H-RHO-7.3 | 3 | 2 | 0.813 | 350 | 98.1 | — | — | 12.99 | 74.1 | 11 |
| H-RHO-8.7 | 10 | 1 | 4.3 | 400 | 95.9 | — | — | 11.05 | 65.4 | 11 |
| H-RHO-7.3 | 10 | 1 | 4.3 | 400 | 96.9 | — | — | 13.31 | 69.4 | 11 |
| H-ZK-5 | 3 | 2 | 0.813 | 350 | 63.4 | 33.7 | 52.5 | 5.1 | 54.7 | 13 |
| D-20 | 12 | 2 | 0.813 | 320 | 91.4 | 22.3 | 60.9 | 11.3 | 76.0 | 19 |
| D-20–700 °C | 12 | 2 | 0.813 | 320 | 82.2 | 25.0 | 64.3 | 5.2 | 73.4 | 19 |
| D-20–800 °C | 12 | 2 | 0.813 | 320 | 65.2 | 29.6 | 55.7 | 4.1 | 55.6 | 19 |
| H-DNL-6 | 2.4 | 2 | 0.813 | 300 | 88.3 | 26.1 | 45.9 | 12.5 | 63.6 | 41 |
| M-DNL-6 | 2.4 | 2 | 0.813 | 300 | 80.0 | 24.4 | 50.7 | 17.5 | 60.1 | 41 |
| L-DNL-6 | 2.4 | 2 | 0.813 | 300 | 82.9 | 21.3 | 31.9 | 36.1 | 44.1 | 41 |
| SAPO-34 | 2.8 | 2 | 0.813 | 380 | 82.0 | 26.8 | 54.7 | 14.5 | 66.8 | 9 |
3. Conclusion
CHA zeolites were synthesized without OSDAs by combining alkali cations with seeds of varying Al contents. Cs+, owing to its low charge density, promoted CHA crystallization across a wide Si/Al ratio range. Seed type and dosage allowed precise control over Si/Al ratio and Al distribution.Catalytic testing demonstrated that both acidity and Al distribution govern methanol amination. Among all samples, H-CHA-S34-3.8 was the most effective, combining high MMA + DMA yield with negligible DME formation. This performance was linked to its high acid density, moderate acid strength, and preferential Al placement in 8MRs.
In summary, this study presents a simple and effective route to tailor Al content and distribution in CHA zeolites, offering a promising design strategy for high-performance methanol amination catalysts.
Author contributions
Shuang Liu: writing – original draft, formal analysis, data curation. Pan Xu: validation, data curation. Sihui Li: software, methodology. Junyao Pan: visualization, formal analysis, data curation. Yuxin Yan: investigation, formal analysis. Rui Wu: formal analysis, data curation. Junhui Guo: investigation, formal analysis. Chang Liu: writing – review & editing, supervision. Wenfu Yan: writing – review & editing, supervision, funding acquisition, conceptualization.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. Supplementary information: materials, synthesis, characterization, ion exchange process, catalytic tests, and additional data. See DOI: https://doi.org/10.1039/d5qi02464a.Acknowledgements
We acknowledge the financial support from the National Key Research and Development Program of China (2024YFE0201000), the National Natural Science Foundation of China (22288101), the ‘111 Center’ (B17020) and the Technology Development Contract of Sinopec (Grant No. 123092) for supporting this work.References
- D. R. Corbin, S. Schwarz and G. C. Sonnichsen, Methylamines synthesis: A review, Catal. Today, 1997, 37, 71–102 CrossRef CAS.
- K. S. Hayes, Industrial processes for manufacturing amines, Appl. Catal., A, 2001, 221, 187–195 CrossRef CAS.
- I. Mochida, A. Yasutake, H. Fujitsu and K. Takeshita, Selective synthesis of dimethylamine (DMA) from methanol and ammonia over zeolites, J. Catal., 1983, 82, 313–321 CrossRef CAS.
- K. Y. Jin, Y. Yan, J. Pan, J.-Y. Liu and W. Yan, The Application of Zeolites in the Selective Synthesis of Methylamine: A Review, Catalysts, 2025, 15, 294 CrossRef CAS.
- J. Li, M. Gao, W. Yan and J. Yu, Regulation of the Si/Al ratios and Al distributions of zeolites and their impact on properties, Chem. Sci., 2023, 14, 1935–1959 RSC.
- B. Y. Wang, Y. Yan, X. Zhou, H. Su, H. Zhang, J.-N. Zhang, J. Xu, Q. Pan and W. Yan, Mild activation of spent fluid catalytic cracking (FCC) catalysts for the pilot synthesis of zeolite a with commercial quality and excellent Co2+ removal, Chem. Eng. J., 2024, 490, 151733 CrossRef CAS.
- Y. Sasaki and M. Fukatsu, Selective catalysts for the production of nitrogen-containing compounds, Catal. Surv. Jpn., 1998, 2, 199–205 CrossRef CAS.
- H. Y. Jeon, C. H. Shin, H. J. Jung and S. B. Hong, Catalytic evaluation of small-pore molecular sieves with different framework topologies for the synthesis of methylamines, Appl. Catal., A, 2006, 305, 70–78 CrossRef CAS.
- Y. Qiao, P. Wu, X. Xiang, M. Yang, Q. Wang, P. Tian and Z. Liu, SAPO-34 synthesized with n-butylamine as a template and its catalytic application in the methanol amination reaction, Chin. J. Catal., 2017, 38, 574–582 Search PubMed.
- H. Lee, K. Lee, J. Shin and S. B. Hong, A comparative study of methylamines synthesis over zeolites H-rho and H-PST-29, Microporous Mesoporous Mater., 2020, 300, 110150 CrossRef CAS.
- Y. Wang, J. Han, K. Jin, S. Liu, Q. Li, P. Hou, S. Liu, Q. Song, Z. Wang, P. Tian and W. Yan, Fluoride-free synthesis of high-silica RHO zeolite for the highly selective synthesis of methylamine, Inorg. Chem. Front., 2024, 11, 5473–5483 RSC.
- S. H. Ahn, D. Shu and S. B. Hong, Synthesis of stable ECR-18 zeolite and its catalytic properties in methanol amination, Microporous Mesoporous Mater., 2024, 364, 112875 Search PubMed.
- R. Wu, J. Han, Y. Wang, M. Chen, P. Tian, X. Zhou, J. Xu, J.-N. Zhang and W. Yan, Exclusive SAPO-seeded synthesis of ZK-5 zeolite for selective synthesis of methylamines, Inorg. Chem. Front., 2022, 9, 5766–5773 Search PubMed.
- P. Xiao, L. Wang, H. Toyoda, Y. Wang, K. Nakamura, J. Huang, R. Osuga, M. Nishibori, H. Gies and T. Yokoi, Revealing Active Sites and Reaction Pathways in Direct Oxidation of Methane over Fe-Containing CHA Zeolites Affected by the Al Arrangement, J. Am. Chem. Soc., 2024, 146, 31969–31981 CrossRef CAS PubMed.
- M. Dusselier and M. E. Davis, Small-Pore Zeolites: Synthesis and Catalysis, Chem. Rev., 2018, 118, 5265–5329 CrossRef CAS.
- M. Sarker, N. A. Khan, D. K. Yoo, B. N. Bhadra, J. W. Jun, T. W. Kim, C. U. Kim and S. H. Jhung, Synthesis of SSZ-13 zeolite in the presence of dimethylethylcyclohexyl ammonium ion and direct conversion of ethylene to propylene with the SSZ-13, Chem. Eng. J., 2019, 377, 120116 CrossRef CAS.
- F. Bleken, M. Bjorgen, L. Palumbo, S. Bordiga, S. Svelle, K.-P. Lillerud and U. Olsbye, The Effect of Acid Strength on the Conversion of Methanol to Olefins Over Acidic Microporous Catalysts with the CHA Topology, Top. Catal., 2009, 52, 218–228 Search PubMed.
- X. Zhu, Y. Gao, H. Chen, M. Jiang, X. Wang, C. Miao, Y. Shen, Y. Ji, Z. Qin, Z. Wu, W. Song, C. Xu and B. Shen, Constructing Hierarchical Zeolites with Highly Complete Framework via Controlled Desilication, Angew. Chem., Int. Ed., 2024, 63, e202411446 Search PubMed.
- Z. Liu, Q. Wang, S. Liu, M. Yang, D. Fan, D. Zhu and P. Tian, Synthesis of SAPO-34 by utilizing spent industrial MTO catalyst and their catalytic applications, Mater. Today Sustain., 2023, 21, 100302 Search PubMed.
- W. Lv, S. Wang, P. Wang, Y. Liu, Z. Huang, J. Li, M. Dong, J. Wang and W. Fan, Regulation of Al distributions and Cu2+ locations in SSZ-13 zeolites for NH3-SCR of NO by different alkali metal cations, J. Catal., 2021, 393, 190–201 Search PubMed.
- T. Nishitoba, N. Yoshida, J. N. Kondo and T. Yokoi, Control of Al Distribution in the CHA-Type Aluminosilicate Zeolites and Its Impact on the Hydrothermal Stability and Catalytic Properties, Ind. Eng. Chem. Res., 2018, 57, 3914–3922 CrossRef CAS.
- W. Lv, P. Meng, Z. Qin, J. Li, M. Dong, J. Wang and W. Fan, A comparison of Al-rich Cu-SSZ-13 zeolites synthesized by different methods in their Al distribution, hydrothermal stability and catalytic performance in the selective catalytic reduction of NOx with NH3, Microporous Mesoporous Mater., 2021, 313, 110851 CrossRef CAS.
- K. Nakamura, P. Xiao, R. Osuga, Y. Wang, S. Yasuda, T. Matsumoto, J. N. Kondo, M. Yabushita, A. Muramatsu, H. Gies and T. Yokoi, Impacts of framework Al distribution and acidic properties of Cu-exchanged CHA-type zeolite on catalytic conversion of methane into methanol followed by lower hydrocarbons, Catal. Sci. Technol., 2023, 13, 2648–2651 RSC.
- Y. Wang, J. Han, M. Chen, W. Lv, P. Meng, W. Gao, X. Meng, W. Fan, J. Xu, W. Yan and J. Yu, Low-silica Cu-CHA Zeolite Enriched with Al Pairs Transcribed from Silicoaluminophosphate Seed: Synthesis and Ammonia Selective Catalytic Reduction Performance, Angew. Chem., Int. Ed., 2023, 62, e20230617 Search PubMed.
- J. Zhang, Y. Shan, L. Zhang, J. Du, H. He, S. Han, C. Lei, S. Wang, W. Fan, Z. Feng, X. Liu, X. Meng and F.-S. Xiao, Importance of controllable Al sites in CHA framework by crystallization pathways for NH3-SCR reaction, Appl. Catal., B, 2020, 277, 119193 CrossRef CAS.
- H. Imai, N. Hayashida, T. Yokoi and T. Tatsumi, Direct crystallization of CHA-type zeolite from amorphous aluminosilicate gel by seed-assisted method in the absence of organic-structure-directing agents, Microporous Mesoporous Mater., 2014, 196, 341–348 CrossRef CAS.
- V. A. Veefkind, C. Grundling and J. A. Lercher, Steric aspects in methylamine and dimethylether synthesis over acidic mordenites, J. Mol. Catal. A:Chem., 1998, 134, 111–119 CrossRef CAS.
- W. Wang, W. Li, Z. Wang, B. Liu, B. Wang, J. Duan and R. Zhou, Directly synthesized high-silica CHA zeolite for efficient CO2/N2 separation, J. Environ. Chem. Eng., 2024, 12, 113036 CrossRef CAS.
- M. Debost, P. B. Klar, N. Barrier, E. B. Clatworthy, J. Grand, F. Laine, P. Brazda, L. Palatinus, N. Nesterenko, P. Boullay and S. Mintova, Synthesis of Discrete CHA Zeolite Nanocrystals without Organic Templates for Selective CO2 Capture, Angew. Chem., Int. Ed., 2020, 59, 23491–23495 Search PubMed.
- L. Bing, A. Tian, F. Wang, K. Yi, X. Sun and G. Wang, Template-Free Synthesis of Hierarchical SSZ-13 Microspheres with High MTO Catalytic Activity, Chem. – Eur. J., 2018, 24, 7428–7433 CrossRef CAS.
- K. Du, X. Zhang, T. He, W. Shen, H. Xu, Y. Tang, X.-M. Cao, Z. Huang and Y. Zhang, Dynamic Evolution of Structural Ordering and Aluminum Redistribution During ZSM-5 Zeolite Crystallization, Angew. Chem., Int. Ed., 2025, 64, e202507223 CrossRef CAS PubMed.
- H. Li, J. Yu, K. Du, W. Li, L. Ding, W. Chen, S. Xie, Y. Zhang and Y. Tang, Synthesis of ZSM-5 Zeolite Nanosheets with Tunable Silanol Nest Contents across an Ultra-wide pH Range and Their Catalytic Validation, Angew. Chem., Int. Ed., 2024, 63, e202405092 CrossRef CAS PubMed.
- Z. Zhang, Y. Li, Z. Chen, F. Ji, X. Liang, H. Xuan, L. Han and P. Han, TEAOH-assisted cost-effective preparation of SSZ-13 using L zeolite as Al source, Fuel, 2024, 362, 130885 CrossRef CAS.
- S. Ghojavand, E. B. Clatworthy, A. Vicente, E. Dib, V. Ruaux, M. Debost, J. El Fallah and S. Mintova, The role of mixed alkali metal cations on the formation of nanosized CHA zeolite from colloidal precursor suspension, J. Colloid Interface Sci., 2021, 604, 350–357 CrossRef CAS PubMed.
- Z. Mi, S. Li, W. Liu, J. Wang, D. Deng, P. Liu, P. Tian, C. Liu, W. Yan, K. Zhu, Z. Wang, Z. Liu and J. Yu, Direct Synthesis of Highly Siliceous Faujasite-type Zeolite Enabled by Low Charge Density Organic Structure-directing Agents, J. Am. Chem. Soc., 2025, 147, 16922–16934 CrossRef CAS PubMed.
- K. Honda, A. Yashiki, M. Itakura, Y. Ide, M. Sadakane and T. Sano, Influence of seeding on FAU-*BEA interzeolite conversions, Microporous Mesoporous Mater., 2011, 142, 161–167 CrossRef CAS.
- J. C. Groen, J. C. Jansen, J. A. Moulijn and J. Pérez-Ramírez, Optimal aluminum-assisted mesoporosity development in MFI zeolites by desilication, J. Phys. Chem. B, 2004, 108, 13062–13065 CrossRef CAS.
- H. Zhang, B. Wang and W. Yan, The structure-directing role of heterologous seeds in the synthesis of zeolite, Green Energy Environ., 2024, 9, 792–801 Search PubMed.
- J. Han, J. Li, W. Zhao, L. Li, M. Chen, X. Ge, S. Wang, Q. Liu, D. Mei and J. Yu, Cu-OFF/ERI Zeolite: Intergrowth Structure Synergistically Boosting Selective Catalytic Reduction of NOx with NH3, J. Am. Chem. Soc., 2024, 146, 7605–7615 CrossRef CAS PubMed.
- K. Mlekodaj, J. Dedecek, V. Pashkova, E. Tabor, P. Klein, M. Urbanova, R. Karcz, P. Sazama, S. R. Whittleton, H. M. Thomas, A. V. Fishchuk and S. Sklenak, Al Organization in the SSZ-13 Zeolite. Al Distribution and Extraframework Sites of Divalent Cations, J. Phys. Chem. C, 2019, 123, 7968–7987 Search PubMed.
- P. Wu, M. Yang, W. Zhang, S. Zeng, M. Gao, S. Xu, P. Tian and Z. Liu, Silicoaluminophosphate molecular sieve DNL-6: Synthesis with a novel template, N,N’-dimethylethylenediamine, and its catalytic application, Chin. J. Catal., 2018, 39, 1511–1519 Search PubMed.
| This journal is © the Partner Organisations 2026 |