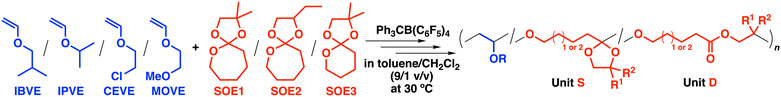
Open Access Article
Open Access ArticleSpiroorthoesters as promising comonomers for degradable polymer synthesis by cationic copolymerization with vinyl ethers
Kota Yamamoto,
Sadahito Aoshima and
Arihiro Kanazawa
and
Arihiro Kanazawa *
*
Department of Macromolecular Science, Graduate School of Science, The University of Osaka, Machikaneyama 1-1, Toyonaka, Osaka, Japan. E-mail: kanazawaa11@chem.sci.osaka-u.ac.jp
First published on 24th April 2026
Abstract
Endeavor to discover new copolymerizable monomers is of great significance for the creation of characteristic copolymers. Spiroorthoesters (SOEs) undergo cationic homopolymerization via the single ring-opening reaction of the cyclic ether ring or the double ring-opening reaction of both the cyclic ether and acetal rings, while copolymerization of SOEs with other monomers has been rarely investigated. In this study, we demonstrate that cationic copolymerization of vinyl ethers (VEs) and SOEs proceeds via very frequent crossover reactions to result in acidically degradable copolymers. 2,2-Dimethyl-1,4,6-trioxaspiro[4.6]undecane, which consists of seven-membered cyclic ether ring and five-membered cyclic acetal ring, underwent copolymerization with isobutyl VE (IBVE) by Ph3CB(C6F5)4 mostly via the single ring-opening reaction of the seven-membered ring. Homopropagation reactions of the SOE negligibly occurred during copolymerization. The average number of IBVE units per block was also very small (<1.5), indicating that the copolymers had pseudo-alternating sequences. The copolymers were degraded into low-molecular-weight compounds via the cleavage of acetal moieties that were generated by the crossover reactions from IBVE to the SOE. In addition, copolymerization of other VEs and SOEs was examined with a focus on the effects of monomer reactivities on the frequency of crossover reactions. Thermoresponsive, water-soluble copolymers with acid degradability were also synthesized from SOEs and 2-methoxyethyl VE.
Introduction
In the design of polymerizable monomers, it is required to consider various factors such as reactive groups (e.g. vinyl group, heteroatoms, etc.), generated propagating species (e.g. carbocation, carbanion, carbon radical, oxonium ion, oxyanion, etc.), resulting structures, ring strain, and steric hindrance. In particular, cyclic monomers allow considerable flexibility of structures in terms of substituents and the number of ring members.1–3 For example, cyclic monomers bearing suitable substituents undergo very efficient ring-opening reactions to generate carbon radical or carbocation that are stabilized by the substituents, as exemplified by the cyclic thioesters for benzyl-type carbon radical generation4,5 and the cyclic hemiacetal esters for alkoxy- and aryl-adjacent carbocation generation.6 Degradable polymers are also synthesized from these monomers via copolymerization with other monomers.7–9Spiroorthoesters (SOEs) are compounds consisting of two rings connected by a ring-constituting carbon atom that has three adjacent oxygen atoms.10–15 The reaction of oxirane and cyclic ester by an acid catalyst is a representative method to prepare SOEs bearing five-membered cyclic acetal and cyclic ester-derived cyclic ether rings (Scheme 1, upper right). SOEs are known to undergo cationic polymerization via single or double ring-opening reactions by Lewis acid catalysts. An orthoester moiety is generated in a polymer chain via the single ring-opening reaction, while ester and ether moieties are generated via the double ring-opening reaction. Endo and coworkers prepared a series of SOEs from cyclic esters with different ring members and systematically investigated the polymerizability of the SOEs (Scheme 1A, left).12–14 Moreover, product polymers obtained via the single ring-opening reactions were shown to undergo degradation under slightly acidic conditions, such as acidic impurities in chloroform. Recently, Mehrkhodavandi and coworkers reported copolymerization of SOEs via selective double ring-opening reactions by a indium or gallium catalyst (Scheme 1A, right).16,17 In addition, cationic photopolymerization of SOEs was applied to 3D printing.18 Cationic copolymerization of SOEs and a substituted oxetane was also reported.19
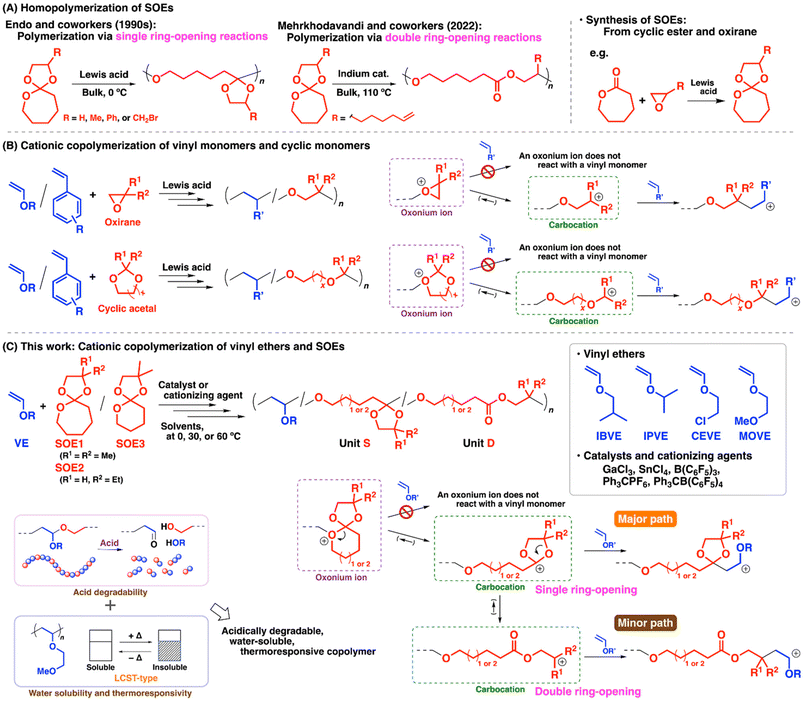 | ||
| Scheme 1 (A) Homopolymerization of SOEs; (B) cationic copolymerization of vinyl monomers and cyclic monomers; and (C) this work: cationic copolymerization of VEs and SOEs. | ||
Copolymerization of different kinds of monomers is an effective strategy for various purposes, such as the formation of degradable units in a main chain, polymerization of nonhomopolymerizable monomers, and monomer sequence control. For example, cationic copolymerization of vinyl monomers such as vinyl ethers (VEs) and styrene derivatives with cyclic monomers such as oxiranes20,21 (Scheme 1B, upper) and cyclic acetals22–24 (Scheme 1B, lower) proceeds when suitable monomer structures and initiating systems are adopted. Carbocation generation via the ring-opening reactions of oxirane- or cyclic acetal-derived oxonium ions (Scheme 1B, right) is a key to successful copolymerization of the different kinds of monomers. This is because vinyl monomers do not react with oxonium ions, while they can react with carbocations generated via the ring-opening reaction. To develop new copolymerization reactions, it is desirable to pioneer comonomers that generate suitable propagating species. SOEs potentially generate carbocations adjacent to two alkoxy groups via the single ring-opening reaction and another type of carbocations via the double ring-opening reaction (Scheme 1C, right). These carbocations are expected to react with vinyl monomers; hence, SOEs are promising comonomer candidates for cationic copolymerization with vinyl monomers such as vinyl ethers and styrene derivatives. However, there were no reports on cationic copolymerization of SOEs and vinyl monomers as far as we know. In this study, therefore, we aimed to examine cationic copolymerization of VEs and SOEs (Scheme 1C). As a result of investigation focusing on cationizing agents and reaction conditions, we achieved copolymerization that proceeds via very frequent crossover reactions between VEs and SOEs. In particular, copolymers with pseudo-alternating sequences were obtained because of both the high reactivity of SOEs in the reactions with VE-derived carbocations and the high equilibrium concentrations (low ceiling temperatures) of SOEs. The propagation reactions of SOEs occurred mostly via the single ring-opening reactions, while the amounts of the structures derived from the double ring-opening reactions were very small. In addition, the copolymers could be degraded via the cleavage of the acetal moieties derived from the crossover reactions from VEs to SOEs. Water-soluble, thermoresponsive copolymers were also obtained when a VE bearing an oxyethylene group was used for copolymerization.
Results and discussion
Cationic copolymerization of IBVE and SOE1
2,2-Dimethyl-1,4,6-trioxaspiro[4.6]undecane (SOE1), which has dimethyl groups at a carbon atom in the five-membered cyclic acetal moiety was synthesized from isobutylene oxide and ε-caprolactone using GaCl3 as a Lewis acid catalyst (Fig. S1A and S2A). This monomer design is based on our intention to promote the double-ring opening reaction due to a possible generation of a tertiary carbocation. To examine suitable catalysts or cationizing agents, we conducted cationic copolymerization of isobutyl VE (IBVE) and SOE1 using SnCl4, GaCl3, B(C6F5)3, Ph3CB(C6F5)4, or Ph3CPF6 in dichloromethane at 0 °C (entries 1–5 in Table 1). Both monomers were consumed with any catalysts or cationizing agents used, while the products exhibited differences of molecular weights (MWs) and incorporated ratios of monomers. Polymerization time was determined in order not to both result in very low monomer conversion and reach 100% conversion. In copolymerization reactions of two monomers, if one of the monomers is completely consumed, not copolymerization but homopolymerization of the other monomer potentially occurs. A polymer with a relatively high number-average molecular weight (Mn) value was obtained with GaCl3 (entry 1 in Table 1). Polymers with comparable Mn values were obtained from SnCl4 and Ph3CB(C6F5)4 (Fig. 1; entries 2 and 5). Considering the incorporated ratios of monomers (determined by 1H NMR as explained in the next paragraph), Ph3CB(C6F5)4 was more suitable than GaCl3 and SnCl4 for copolymerization via frequent crossover reactions. | ||
| Fig. 1 MWD curves of poly(IBVE-co-SOE1)s [entries (A) 2 and (B) 5 in Table 1; black: original product, pink: after purification by reprecipitation in methanol, purple: their acid hydrolysis products (hydrolysis after purification by reprecipitation)]. See the footnote of Table 1 for the polymerization conditions. [† The Mn values of the main portions (the MW region over approximately 103). The Mn values calculated from the whole region including oligomer regions are (A) 2.9 × 103 and (B) 1.8 × 103.] | ||
| Entry | IBVE | SOE1 | Catalyst or cationizing agent | Solventb | Temp. (°C) | Time | Conv.c (%) | Mn × 10−3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) d d |
Mw/Mn![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) d d |
Monomer units per blocke | |||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| IBVE | SOE1 | IBVE | Unit S | Unit D | |||||||||
| a [Catalyst or cationizing agent]0 = 3.0 (except for entry 12) or 1.0 (entry 12) mM. See Fig. 1 and Fig. S3 for the MWD curves.b dcm: dichloromethane, hex: hexane, tol: toluene.c By gas chromatography (except for entry 8) or 1H NMR analysis of the quenched reaction mixture (entry 8).d Determined by GPC (polystyrene calibration). The values of the main portions (the MW region over approximately 103).e The average number of monomer units per block. These values correspond to the average number of monomer units per the acetal moiety derived from the crossover reaction from VE to SOE. Calculated by 1H NMR after purification by reprecipitation in methanol.f v/v.g A small amount SOE1 was likely consumed; however, a polymer was not obtained. A low-MW product containing an ester moiety was obtained (see Fig. S4 for the 1H NMR spectrum).h [Ph3CB(C6F5)4]0 = 1.0 mM. | |||||||||||||
| 1 | 0.80 | 0.80 | GaCl3 | dcm | 0 | 6 h | 93 | 56 | 9.4 | 1.64 | 2.5 | 1.26 | 0.03 |
| 2 | 0.80 | 0.80 | SnCl4 | dcm | 0 | 6 h | 85 | 45 | 4.6 | 2.00 | 2.9 | 1.13 | 0.04 |
| 3 | 0.80 | 0.80 | B(C6F5)3 | dcm | 0 | 168 h | 66 | 28 | 1.1 | 1.29 | — | — | — |
| 4 | 0.80 | 0.80 | Ph3CPF6 | dcm | 0 | 20 min | 38 | 36 | 3.9 | 1.39 | 3.6 | 1.45 | 0.07 |
| 5 | 0.80 | 0.80 | Ph3CB(C6F5)4 | dcm | 0 | 45 min | 89 | 77 | 5.0 | 1.55 | 1.4 | 0.99 | 0.04 |
| 6 | 0.80 | 0.80 | Ph3CB(C6F5)4 | hex/dcm (8/2f) | 0 | 50 min | 96 | 97 | 3.7 | 1.34 | 1.4 | 0.93 | 0.08 |
| 7 | 0.80 | 0.80 | Ph3CB(C6F5)4 | tol/dcm (9/1f) | 0 | 45 min | 69 | 68 | 3.8 | 1.37 | 1.5 | 0.96 | 0.09 |
| 8 | 0.80 | 0.80 | Ph3CB(C6F5)4 | dcm | 30 | 3 min | 76 | 58 | 4.6 | 1.59 | 1.4 | 1.00 | 0.07 |
| 9 | 0.80 | 0.80 | Ph3CB(C6F5)4 | tol/dcm (9/1f) | 30 | 9 min | 81 | 65 | 5.8 | 1.70 | 1.4 | 0.98 | 0.07 |
| 10 | 0.80 | 0.80 | Ph3CB(C6F5)4 | tol/dcm (9/1f) | 60 | 30 s | 57 | 57 | 3.8 | 1.56 | 1.4 | 1.01 | 0.05 |
| 11 | 0.40 | 0.40 | Ph3CB(C6F5)4 | dcm | 0 | 30 min | 82 | 71 | 5.5 | 1.45 | 1.4 | 1.02 | 0.03 |
| 12 | 0.80 | 0.80 | Ph3CB(C6F5)4h | dcm | 0 | 90 min | 70 | 65 | 4.0 | 1.36 | 1.5 | 1.01 | 0.07 |
| 13 | — | 0.80 | Ph3CB(C6F5)4 | dcm | 0 | 3 h | — | 19g | — | — | — | — | — |
The product obtained with Ph3CB(C6F5)4 (entry 5 in Table 1) was analyzed by 1H NMR after a low-MW portion was removed by reprecipitation in methanol (Fig. 2A, upper; see Fig. S5 for the products obtained with GaCl3, SnCl4, or Ph3CPF6). A peak observed at 4.6 ppm (peak 4) was assigned to acetal moieties resulting from the crossover reaction from IBVE to SOE1. From the integral ratios of this peak and the peaks of the IBVE-derived isobutyl groups at 0.9 ppm (peak 7), the number of IBVE units per block was determined to be 1.4 (the number of monomer units per acetal corresponds to the number of monomer units per block). SOE1 potentially undergoes single or double ring-opening reactions, while an ester structure (unit D) derived from the latter reaction was obviously small in the spectrum (0.04 per acetal; peaks 16 and 17). The SOE1-derived units in polymer chains mostly had a five-membered cyclic acetal-containing structure (unit S) derived from the single ring-opening reaction. The assignments of this copolymer structure were confirmed by 13C and 2D (1H–1H COSY, 1H–13C HSQC, and 1H–13C HMBC) NMR analysis (Fig. 2B and Fig. S6–S9). The number of unit S per acetal was approximately 1.0, which indicates that homopropagation reactions of SOE1 negligibly occurred. The absence of SOE1 homosequences is consistent with the ineffective homopolymerization of SOE1 under similar conditions to those for the copolymerization (entry 13 in Table 1) due to the high equilibrium monomer concentration (low ceiling temperature).12–14 Therefore, the copolymerization proceeded via very frequent crossover reactions. The average number of IBVE/SOE1 units per block of 1.4/1.0 means that the copolymer chains have pseudo-alternating sequences. An average structure of the copolymer chains corresponds to an illustration shown in Scheme 2 (upper).
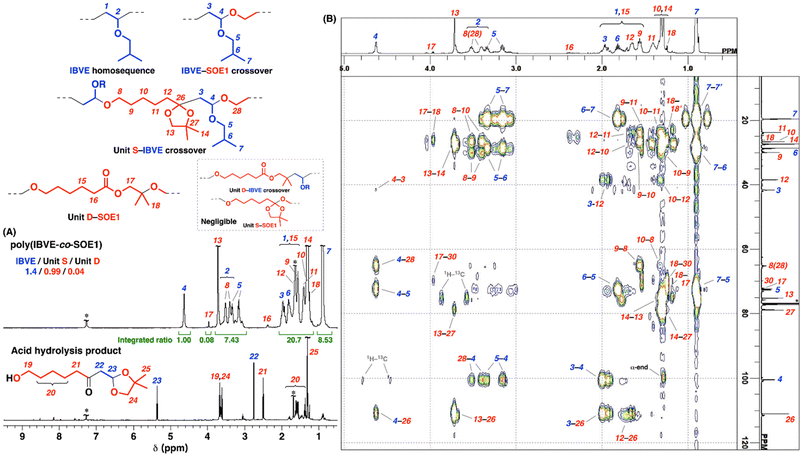 | ||
| Fig. 2 (A) 1H NMR spectra of poly(IBVE-co-SOE1) (upper; entry 5 in Table 1; after purification by reprecipitation in methanol, a pink curve in Figure 1B) and acid hydrolysis product (lower). (B) 1H–13C HMBC NMR spectrum of poly(IBVE-co-SOE1) (entry 9 in Table 1; a limited region of the spectrum is shown; see Fig. S9 for a wider region). See the footnote of Table 1 for the polymerization conditions. In CDCl3 at 30 °C. *CHCl3 or water. | ||
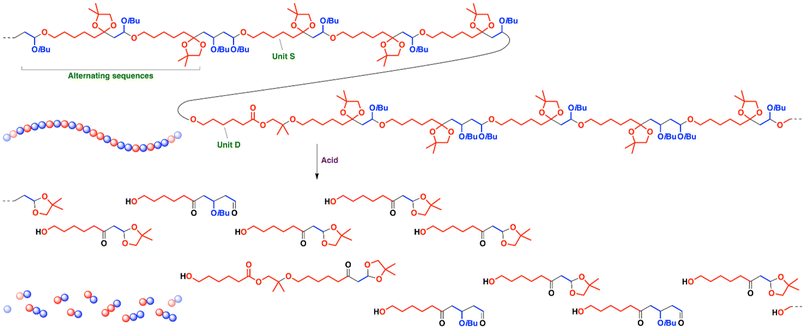 | ||
| Scheme 2 An illustration of a copolymer structure (upper) drawn based on the average number of IBVE and SOE1 units estimated by 1H NMR and its acid hydrolysis product (lower). | ||
The copolymer has acetal moieties in the main chain; hence, the copolymer was subjected to acid hydrolysis with aqueous HCl in 1,2-dimethoxyethane. As a result, a low-MW product with a very narrow molecular weight distribution (MWD) was obtained (a purple curve in Fig. 1B). The peaks observed in the 1H NMR spectrum of the product were assigned to a structure containing hydroxy group, ketone moiety, and cyclic acetal moiety (Fig. 2A, lower). A possible degradation mechanism is shown in Scheme S1 in the SI. Almost exclusive generation of this product is consistent with the pseudo-alternating structure of the copolymer (Scheme 2).
The copolymerization mechanism is summarized in Scheme 3. A trityl cation derived from Ph3CB(C6F5)4 or a proton generated from Ph3CB(C6F5)4 and adventitious water reacts with a monomer in the initiation reaction. A VE-derived carbocation preferentially reacts with SOE1 ([a] in Scheme 3) rather than IBVE ([b]) as indicated by the small number of IBVE units per block (1.4). The oxygen atom in the seven-membered ring of SOE1 likely reacts with the carbocation, considering previous studies on cationic homopolymerization of SOEs.13 The generated oxonium ion subsequently undergoes the ring-opening reaction ([c]) to result in a carbocation with two adjacent oxygen atoms. This carbocation potentially undergoes the following three reactions: the crossover reaction with IBVE ([d]), the homopropagation reaction with SOE1 ([e]), and the ring-opening reaction of the five-membered ring into a tertiary carbocation ([f]). From the NMR analysis demonstrated above, the crossover reaction from the carbocation to IBVE ([d]) preferentially occurred to generate an isolated SOE1 unit bearing a five-membered cyclic acetal moiety (unit S). An orthoester formation is unfavorable due to the instability, which is a possible reason of more preferential reaction of IBVE with the SOE-derived carbocation ([d]) than the homopropagation reaction of SOE1 ([e]). Indeed, homopolymerization of SOE1 did not proceed under the conditions similar to those for copolymerization (entry 13 in Table 1), unlike past examples of successful homopolymerization at high initial monomer concentrations.12,13 At the low monomer concentration, homopropagation reactions ([e]) were absent due to the high equilibrium concentration (low ceiling temperature) of SOEs. In addition, unlike our initial expectation, the ring-opening reaction of the five-membered ring ([f]) did not occur frequently despite the monomer design for a tertiary carbocation generation. Several factors, such as the moderate ring-strain of a five-membered cyclic acetal structure, the stability of the two oxygen atoms-adjacent carbocation, and the insufficient stability of the tertiary carbocation with dimethyl and ester-adjacent methylene groups, are likely responsible for infrequent ring-opening reaction of the five-membered ring. The monomer reactivity ratios were estimated to be 0.25 and 0.03 for IBVE and SOE1, respectively, by the Meyer–Lowry method25,26 (Fig. S10 and S11, Table S1).
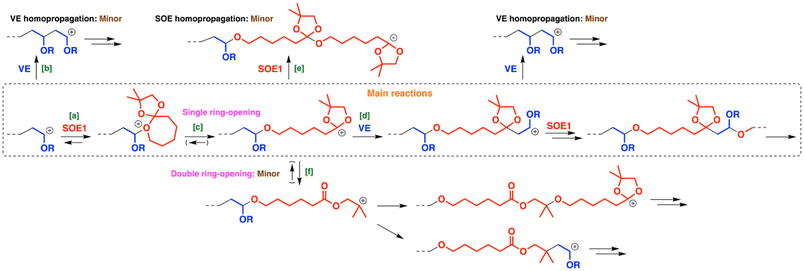 | ||
| Scheme 3 Main reactions (inside the square) and minor reactions (outside the square) in the copolymerization of VEs and SOE1. Counteranions are omitted. | ||
Copolymerization products exhibited relatively broad MWD curves with a noticeable tailing in most cases (Fig. 1 and Fig. S3). To examine the structures of the low-MW portion, we conducted electrospray ionization mass spectrometry (ESI-MS) analysis (Fig. S12). As a result, the low-MW portion most likely consisted of cyclic oligomers resulting from backbiting reactions and hydrolysis products of the cyclic oligomers. The detail of ESI-MS analysis is explained in the ESI. Side reactions, such as β-proton elimination reactions, other than backbiting reactions were not confirmed by NMR analysis. Indeed, long-lived species were likely generated as judged from the change of MWD curves of the products obtained at different polymerization time (Fig. S10). The low Mn values than those expected from the amounts of the monomers (0.80 M/0.80 M) and the cationogen (3.0 mM) are due to the generation of noticeable amounts of cyclic oligomers.
The ability of SOE1 as a promising comonomer for the cationic copolymerization with VEs is comparable to those of conjugated aldehydes and cyclic acetals. Appropriate conjugated aldehydes27 and cyclic acetals24 undergo cationic alternating copolymerization with VEs as reported in our previous studies. Complete suppression of VE homopropagation reactions is required for the synthesis of alternating copolymers from SOE1 and VEs.
To both suppress IBVE homopropagation reactions for alternating sequences and obtain higher-MW products, we conducted copolymerization of IBVE and SOE1 under various conditions. Copolymerization was examined in different solvents (entries 6 and 7 in Table 1), at 30 °C (entries 8 and 9), or at different concentrations of monomers or Ph3CB(C6F5)3 (entries 11 and 12). However, any of the conditions did not significantly affect the MWs of polymers and the incorporated ratios of monomers. In addition, the ratio of double ring-opening reaction (unit D) did not increase even when polymerization was conducted at 60 °C (entry 10) unlike the exclusive double ring-opening reaction in the homopolymerization of a SOE at high temperature in a past study.16
Cationic copolymerization of various VEs and SOEs
Other SOEs than SOE1 were also synthesized (Fig. S1 and S2) and subjected to copolymerization with IBVE. 2-Ethyl-1,4,6-trioxaspiro[4.6]undecane (SOE2), which has an ethyl group in a five-membered cyclic acetal moiety, was synthesized from 1,2-butylene oxide and ε-caprolactone. SOE2 underwent copolymerization with IBVE, resulting in a copolymer similar to that of SOE1 in terms of MW and the number of monomer units per block (entry 2 in Table 2). A higher-MW copolymer than the SOE1 and SOE2 counterparts was obtained from 2,2-dimethyl-1,4,6-trioxaspiro[4.5]decane (SOE3), which was synthesized from δ-valerolactone and thus has a six-membered cyclic ether moiety (entry 3). However, the number of IBVE units per block was obviously larger than those of the copolymers from SOE1 and SOE2, which indicates that the reactivity of SOE3 is lower than those of SOE1 and SOE2 potentially due to the lower ring strain of the six-membered ring than that of the seven-membered ring. This discussion is based on the assumption that the reaction between a VE-derived carbocation and a SOE monomer ([a] in Scheme 3) is reversible unless the ring-opening reaction of a SOE moiety ([c] in Scheme 3) occurs. Another possible reason is that the nucleophilicity of SOE3 may be lower than those of SOE1 and SOE2. The low reactivity of the six-membered cyclic ester-derived SOEs is consistent with the past study.14| Entry | VE | SOE | Conc. (M) | Time | Conv.b (%) | Mn × 10−3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) c c |
Mw/Mn![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) c c |
Monomer units per blockd | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| VE | SOE | VE | SOE | VE | Unit S | Unit D | ||||||
| a [Ph3CB(C6F5)4]0 = 3.0 mM, in toluene/dichloromethane (9/1 v/v) at 30 °C. See Fig. S13 for the MWD curves. See Fig. S14–S17 for the 1H NMR spectra.b Calculated from GC analysis.c Determined by GPC (polystyrene calibration). The values of the main portions (the MW region over approximately 103).d The average number of monomer units per block. These values correspond to the average number of monomer units per the acetal moiety derived from the crossover reaction from VE to SOE. Calculated by 1H NMR (before [entry 4] or after [except for entry 4] purification by reprecipitation in methanol).e Entry 1 corresponds to entry 9 in Table 1. | ||||||||||||
| 1e | IBVE | SOE1 | 0.80 | 0.80 | 9 min | 81 | 65 | 5.8 | 1.70 | 1.4 | 0.98 | 0.07 |
| 2 | IBVE | SOE2 | 0.80 | 0.80 | 2 min | 86 | 92 | 3.7 | 1.45 | 1.3 | 1.05 | 0.10 |
| 3 | IBVE | SOE3 | 0.80 | 0.80 | 5 min | 87 | 23 | 9.6 | 1.49 | 3.7 | 1.03 | 0.01 |
| 4 | IPVE | SOE1 | 0.80 | 0.80 | 1 min | 80 | 52 | 5.1 | 1.68 | 1.7 | 1.15 | 0.08 |
| 5 | CEVE | SOE1 | 0.80 | 0.80 | 6.5 h | 88 | 92 | 3.3 | 1.56 | 1.2 | 1.09 | 0.20 |
| 6 | MOVE | SOE1 | 0.80 | 0.80 | 1.5 min | 83 | 41 | 3.4 | 1.78 | 1.8 | 1.09 | 0.04 |
| 7 | MOVE | SOE1 | 0.80 | 0.60 | 1 min | 89 | 56 | 3.7 | 1.82 | 2.1 | 1.12 | 0.02 |
| 8 | MOVE | SOE1 | 0.80 | 0.40 | 45 s | 79 | 57 | 5.6 | 1.52 | 2.6 | 1.09 | 0.02 |
| 9 | MOVE | SOE3 | 0.80 | 0.80 | 45 s | 94 | 12 | 3.4 | 2.12 | 7.2 | 0.97 | 0.03 |
Copolymerization of isopropyl VE (IPVE), which is a more reactive VE than IBVE [the monomer reactivity ratios of 2.70 (IPVE) and 0.22 (IBVE)28], with SOE1 resulted in a copolymer with an Mn of 5.1 × 103 (entry 4 in Table 2). The average number of IPVE units per block was smaller (1.7) than expected, considering the large difference in the monomer reactivity ratios of IPVE and IBVE. 2-Chloroethyl VE (CEVE) was used as a less reactive VE [the monomer reactivity ratios of 1.90–2.17 (IBVE) and 0.34–0.70 (CEVE)]28–30 for the purpose of achieving alternating sequences (entry 5). The number of CEVE units per block in the obtained copolymer was small (1.2) although homopropagation was not completely suppressed. In the 1H NMR spectrum of the copolymer, a peak assigned to the methyl groups of the structure resulting from the double ring-opening reaction of SOE1 and subsequent crossover reaction to CEVE (unit D–CEVE) was observed at 0.9 ppm. The average number of unit D per block was 0.20. The smaller reactivity of CEVE likely resulted in more preferential double-ring opening reaction than the cases of IBVE and IPVE.
Copolymerization of MOVE and SOEs: for the synthesis of water-soluble copolymers with thermoresponsivity and degradability
A homopolymer of 2-methoxyethyl VE (MOVE) is soluble in water and exhibits lower critical solution temperature (LCST)-type thermoresponsivity.31 To obtain copolymers with water solubility, thermoresponsivity, and degradability, we conducted copolymerization of MOVE and SOE1 under similar conditions to those for the copolymerization demonstrated above. A copolymer obtained at initial MOVE/SOE1 concentrations of 0.80 M/0.80 M had a MOVE/unit S/unit D ratio of 1.8/1.09/0.04 (entry 6 in Table 2). When the initial concentration of SOE1 was decreased into 0.60 M or 0.40 M, copolymers with larger ratios of MOVE units were obtained (entries 7 and 8; MOVE/unit S/unit D ratio = 2.1/1.12/0.02 at 0.60 M and 2.6/1.09/0.02 at 0.40 M). Moreover, a copolymer with a much larger ratio of MOVE units was obtained when SOE3 was used (entry 9; MOVE/unit S/unit D ratio = 7.2/0.97/0.03).The obtained copolymers were subjected to solubility test in water after oligomer portions were removed by preparative gel permeation chromatography (GPC). The copolymers with MOVE/unit S/unit D ratios of 1.8/1.09/0.04 and 2.1/1.12/0.02 were insoluble in water even when cooled at 0 °C. The copolymer with a MOVE/unit S/unit D ratio of 2.6/1.09/0.02 was insoluble in water at room temperature, while it likely dissolved when cooled at 0 °C. Moreover, the SOE3-derived copolymer with a much larger ratio of MOVE units (MOVE/unit S/ unit D ratio = 7.2/0.97/0.03) was soluble in water (Fig. 3A, left) even at room temperature. When temperature of the copolymer solutions were varied (Fig. 3B), the MOVE–SOE1 and MOVE–SOE3 copolymers exhibited thermoresponsivity (Fig. 3A, right) at 0–10 °C (green curve) and 30–40 °C (brown curve), respectively, as indicated by the decrease in transmittance at 500 nm. These cloud points were lower than that of a MOVE homopolymer (approximately 70 °C, blue curve in Fig. 3B) due to hydrophobicity of the SOE1- and SOE3-derived units in the copolymers. Dynamic light scattering (DLS) analysis of poly(MOVE-co-SOE3) was also conducted at a concentration of 0.1 wt% (Fig. S18). A peak with a diameter of <10 nm was detected at 7 and 15 °C, while a peak assigned to aggregates with a diameter of 101–102 nm or larger was detected at 25, 35, and 45 °C. The aggregate formation at high temperature is consistent with the result of the transmittance measurement.
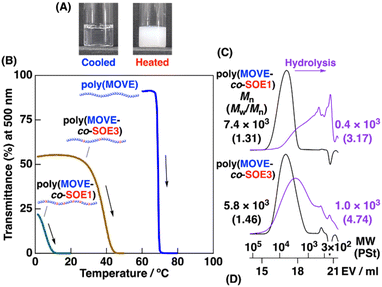 | ||
| Fig. 3 (A) Photographs taken soon after cooled in a refrigerator (8 °C; left) and heated in a water bath (50 °C; right) [poly(MOVE-co-SOE3) in water, 1 wt%]. (B) Turbidity measurement of poly(MOVE) [Mn(GPC) = 6.3 × 103] and poly(MOVE-co-SOE)s (entries 8 and 9 in Table 2) in water (polymer concentration: 1 wt%; heating rate = 1 °C min−1). (C) MWD curves of poly(MOVE-co-SOE)s (black; purified by preparative GPC) and their acid hydrolysis products (purple). | ||
The thermoresponsive copolymers exhibited acid degradability derived from the acetal moieties in the main chains. The copolymers with Mn values of 7.4 × 103 and 5.8 × 103 were degraded into products with Mn values of 0.4 × 103 and 1.0 × 103, respectively (Fig. 3C).
Thermal analysis
A copolymer obtained from IBVE and SOE1 was subjected to thermal analysis. In differential scanning calorimetry (DSC) analysis, a glass transition temperature (Tg) was detected at −32 °C (Fig. S19). This value is slightly lower than the Tg of a IBVE homopolymer (−19 °C).32 In thermogravimetric analysis (TGA), the sample gradually decomposed at approximately 200 °C on heating (Fig. S20).Conclusions
In conclusion, cationic copolymerization of VEs and SOEs successfully proceeded via very frequent crossover reactions, resulting in pseudo-alternating copolymers with acid degradability. SOEs underwent copolymerization mostly via the single ring-opening reaction of the cyclic ether moiety. The copolymers were degraded by acid due to the cleavage of the acetal moieties generated by the crossover reactions. Moreover, water-soluble, thermoresponsive copolymers with degradability were obtained from oxyethylene chain-containing VE and SOEs. The results obtained in this study demonstrate a promising ability of SOEs as comonomers for the synthesis of pseudo-alternating, degradable, and functional copolymers by cationic copolymerization. Remaining themes of SOE copolymerization are complete suppression of VE homopropagation for complete alternating monomer sequences, not single but double ring-opening reaction-dominated copolymerization, and suppression of cyclic oligomer byproducts, which will be tackled in future studies.Author contributions
K. Yamamoto: investigation and writing – original draft, S. Aoshima: writing – review & editing, A. Kanazawa: conceptualization, writing – original draft, and writing – review & editing.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: experimental section, polymerization data, NMR spectra, and ESI-MS spectrum. See DOI: https://doi.org/10.1039/d6py00306k.Acknowledgements
This work was partially supported by JSPS KAKENHI Grant 23K17946.References
- O. Nuyken and S. D. Pask, Ring-Opening Polymerization—An Introductory Review, Polymers, 2013, 5, 361–403 CrossRef.
- S. Penczek and K. Kaluzynski, in Polymer Science: A Comprehensive Reference, ed. K. Matyjaszewski and M. Möller, Elsevier B. V., Amsterdam, 2012, vol. 4, ch. 4.02 Search PubMed.
- Y. He, Z. Li, D. Zhao, Y. Shen, W. Fu and Z. Li, Ring-Opening Polymerization of Representative Carbocyclic and Oxacyclic Monomers: Versatile Platform toward Advanced Functional Polymers, Prog. Polym. Sci., 2025, 160, 101921 CrossRef CAS.
- N. M. Bingham and P. J. Roth, Degradable Vinyl Copolymers through Thiocarbonyl Addition–Ring-Opening (TARO) Polymerization, Chem. Commun., 2019, 55, 55–58 RSC.
- R. A. Smith, G. Fu, O. McAteer, M. Xu and W. R. Gutekunst, Radical Approach to Thioester-Containing Polymers, J. Am. Chem. Soc., 2019, 141, 1446–1451 CrossRef CAS PubMed.
- Y. Takahashi, A. Kanazawa and S. Aoshima, 3-Alkoxyphthalides as Nonhomopolymerizable, Highly Reactive Comonomers for ABC Pseudo-Periodic Terpolymers and Degradable Polymers via Cationic Co- and Terpolymerizations with Oxiranes and/or Vinyl Ethers, Macromolecules, 2023, 56, 4198–4207 CrossRef CAS.
- M. Dirauf, I. Muljajew, C. Weber and U. S. Schubert, Recent Advances in Degradable Synthetic Polymers for Biomedical Applications – Beyond Polyesters, Prog. Polym. Sci., 2022, 129, 101547 CrossRef CAS.
- S. Bag, D. Ghosh, S. Banerjee and P. De, Stimuli-Responsive Main-Chain Degradable Polymers: Synthesis, Applications, and Future Perspectives, ACS Appl. Polym. Mater., 2025, 7, 14033 CrossRef CAS.
- A. Kanazawa and S. Aoshima, S. Concurrent Cationic Vinyl-Addition and Ring-Opening Copolymerization of Vinyl Ethers and Oxiranes, Polym. J., 2016, 48, 679–687 CrossRef CAS.
- T. Endo and W. Bailey, Synthesis of Monomers Expanding on Polymerization, Makromol. Chem., 1975, 176, 2897–2903 CrossRef CAS.
- K. Matyjaszewski, Cationic Polymerization of 1,4,6-Trioxaspiro[4,4]-Nonane, J. Polym. Sci., Polym. Chem. Ed., 1984, 22, 29–40 CrossRef CAS.
- S. Chikaoka, T. Takata and T. Endo, A Novel Polymerization Behavior of Spiroorthoester: An Equilibrium of Polymerization, Macromolecules, 1991, 24, 331–332 CrossRef CAS.
- S. Chikaoka, T. Takata and T. Endo, New Aspects of Cationic Polymerization of Spiroorthoester: Cationic Single Ring-Opening Polymerization and Equilibrium Polymerization, Macromolecules, 1991, 24, 6557–6562 CrossRef CAS.
- S. Chikaoka, T. Takata and T. Endo, Copolymerization Utilizing an Equilibrium Polymerization System: Cationic Copolymerizations of 1,4,6-Trioxaspiro[4.6]undecane with 2-Methyl-1,4,6-trioxaspiro[4.5]decane and 2-Methyl-1,4,6-trioxaspiro[4.4]nonane, Macromolecules, 1991, 24, 6563–6566 CrossRef CAS.
- Y.-G. Hsu and Y.-S. Wan, Cationic Photopolymerization of cis-2,3-Tetramethylene-1,4,6-trioxaspiro[4,4]nonane, J. Polym. Sci., Part A: Polym. Chem., 2009, 47, 3680–3690 CrossRef CAS.
- H.-J. Jung, C. Goonesinghe and P. Mehrkhodavandi, Temperature Triggered Alternating Copolymerization of Epoxides and Lactones via Pre-sequenced Spiroorthoester Intermediates, Chem. Sci., 2022, 13, 3713–3718 RSC.
- H.-J. Jung, C. Goonesinghe, Z. Zhang, J. Chang, K. Nyamayaro, H. A. Baalbaki, S. G. Hatzikiriakos and P. Mehrkhodavandi, Synthesis of High-Molecular-Weight Poly(ether-alt-ester) by Selective Double Ring-Opening Polymerization of Spiroorthoesters, ACS Macro Lett., 2024, 13, 266–272 CrossRef CAS PubMed.
- D. Kojic, K. Ehrmann, R. Wolff, Y. Mete, T. Koch, J. Stampfl, S. Baudis and R. Liska, Stereolithographic 3D Printing of Pure Poly(ether–ester) Networks from Spirocyclic Monomers via Cationic Ring-Opening Photopolymerization at High Temperatures, Polym. Chem., 2023, 14, 4809–4818 RSC.
- T. Nagasawa, B. Ochiai and T. Endo, Infrared Thermographic Analysis on Copolymerization of Spiroorthoester with Oxetane, J. Polym. Sci., Part A: Polym. Chem., 2007, 45, 1388–1393 CrossRef CAS.
- A. Kanazawa, S. Kanaoka and S. Aoshima, Concurrent Cationic Vinyl-Addition and Ring-Opening Copolymerization Using B(C6F5)3 as a Catalyst: Copolymerization of Vinyl Ethers and Isobutylene Oxide via Crossover Propagation Reactions, J. Am. Chem. Soc., 2013, 135, 9330–9333 CrossRef CAS PubMed.
- A. Kanazawa and S. Aoshima, Cationic Copolymerization of Styrene Derivatives and Oxiranes via Concurrent Vinyl-Addition and Ring-Opening Mechanisms: Multiblock Copolymer Formation via Occasional Crossover Reactions, Macromolecules, 2020, 53, 5255–5265 CrossRef CAS.
- M. Okada, Y. Yamashita and Y. Ishii, Cationic Copolymerization of 1,3-Dioxolane with Styrene, Makromol. Chem., 1966, 94, 181–193 CrossRef CAS.
- M. Okada and Y. Yamashita, Cationic Copolymerization of Cyclic Formals and Vinyl Ethers, Makromol. Chem., 1969, 126, 266–275 CrossRef CAS.
- K. Maruyama, A. Kanazawa and S. Aoshima, Controlled Cationic Copolymerization of Vinyl Monomers and Cyclic Acetals via Concurrent Vinyl-Addition and Ring-Opening Mechanisms: The Systematic Study of Structural Effects on the Copolymerization Behavior, Polym. Chem., 2019, 10, 5304–5314 RSC.
- V. E. Meyer and G. G. Lowry, Integral and Differential Binary Copolymerization Equations, J. Polym. Sci., Part A: Gen. Pap., 1965, 3, 2843–2851 CrossRef CAS.
- N. A. Lynd, R. C. Ferrier Jr. and B. S. Beckingham, Recommendation for Accurate Experimental Determination of Reactivity Ratios in Chain Copolymerization, Macromolecules, 2019, 52, 2277–2285 CrossRef CAS.
- Y. Ishido, R. Aburaki, S. Kanaoka and S. Aoshima, Well-Defined Alternating Copolymers of Benzaldehydes with Vinyl Ethers: Precision Synthesis by Cationic Copolymerization and Quantitative Degradation to Cinnamaldehydes, Macromolecules, 2010, 43, 3141–3144 CrossRef CAS.
- T. Kelen, F. Tüdõs, B. Turcsányi and J. P. Kennedy, Analysis of the Linear Methods for Determining Copolymerization Reactivity Ratios. IV. A Comprehensive and Critical Reexamination of Carbocationic Copolymerization Data, J. Polym. Sci., Polym. Chem. Ed., 1977, 15, 3047–3074 CrossRef CAS.
- D. D. Eley and J. Saunders, The Kinetics of Ionic Polymerisations. Part VI. The Copolymerization of Isobutyl Vinyl Ethers and 2-Chloroethyl Vinyl Ether, J. Chem. Soc., 1954, 1677–1680 RSC.
- S. Okamura, N. Kanoh and T. Higashimura, Rate Constant of Propagation Reaction in Stationary State of Cationic Polymerization. Part II. Rate Constant of the Propagation Reaction of Alkyl Vinyl Ethers–Iodine Systems, Makromol. Chem., 1961, 47, 35–47 CrossRef CAS.
- S. Aoshima, H. Oda and E. Kobayashi, Synthesis of Thermally-Induced Phase Separating Polymer with Well-Defined Polymer Structure by Living Cationic Polymerization. I. Synthesis of Poly (vinyl ether)s with Oxyethylene Units in the Pendant and Its Phase Separation Behavior in Aqueous Solution, J. Polym. Sci., Part A: Polym. Chem., 1992, 30, 2407–2413 CrossRef CAS.
- R. J. Andrews and E. A. Grulke, in Polymer Handbook, ed. J. Brandrup, E. H. Immergut and E. A. Grulke, Wiley-Interscience, New York, 4th edn, 1999, ch. II, pp. 215 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |