3-Methylenephthalide derivatives enable degradation of vinyl (co)polymers via simultaneous main- and side-chain cleavage
Yota
Chiba
,
Shoji
Hirabayashi
and
Yasuhiro
Kohsaka
 *
*
Faculty of Textile Science and Technology, Shinshu University, 3-15-1 Tokida, Ueda, Nagano 386-8567, Japan. E-mail: kohsaka@shinshu-u.ac.jp
First published on 16th March 2026
Abstract
The selective cleavage of carbon–carbon bonds remains a fundamental challenge for polymeric materials with carbon backbones, including many vinyl polymers. Herein, we report functional vinyl (co)polymers synthesized from 3-methylenephthalide analogs, cyclic vinyl monomers bearing various ester pendants. The (co)polymers were prepared via free-radical polymerization and exhibited tunable solubility and mechanical properties depending on the pendant ester structures. Upon hydrolysis under basic conditions, the polymers underwent simultaneous degradation of the main chain and pendant groups to afford a common backbone-derived monomer precursor. This study demonstrates a molecular design concept in which property diversification through copolymerization is decoupled from molecular convergence upon chemical degradation.
Introduction
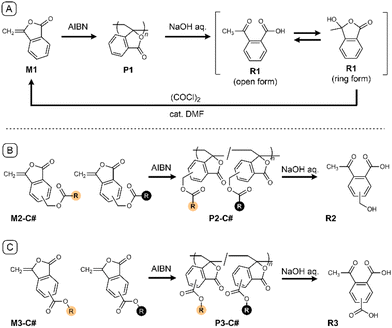
Vinyl polymers are widely used owing to their versatile synthesis and tunable properties. However, their stability, originating from the C(sp3)–C(sp3) bonds in the backbone, poses a fundamental challenge for their recycling/upcycling. To address this challenge, the incorporation of a weak covalent bond into the polymer backbone has gained attention to attain programmed chain scission.1–8 For example, radical ring-opening polymerization allows the introduction of ester bonds into vinyl polymer backbones.9–13 While these approaches are effective for homopolymers derived from single monomers, their applicability becomes limited when structural diversity is introduced. Copolymerization and pendant modification are powerful methods for tuning the properties and performance of vinyl polymers and are indispensable for fine control of thermal, mechanical, and interfacial properties in practical materials.14–18 However, such structural diversity generally leads to divergent degradation products, yielding complex mixtures whose components reflect the underlying monomer compositions.In this study, we address this limitation by proposing a molecular design strategy that intentionally eliminates structural diversity at the backbone level during chemical degradation. In our previous study,19 we reported that poly(3-methylenephthalide) (P1)20,21 undergoes hydrolysis to afford 2-acetylbenzoic acid (R1), a precursor of monomer M1 (Scheme 1A). Although P1 has potential as a chemically recyclable vinyl polymer, its inherent hardness and brittleness hinder its practical application. Copolymerization with other vinyl monomers is a typical strategy to improve these mechanical properties; however, because the degradation mechanism of P1 involves depolymerization from the chain ends, the insertion of ‘inactive’ comonomer units inhibits the reaction. In our previous study, we investigated other monomer structures, such as acyclic esters, cyclic amides, and cyclic carbonates, although the M1/P1 system was superior in terms of both polymerizability and degradability.
To address the difficulty in tuning polymer properties while maintaining degradability, we designed an alternative monomer library using the M1 skeleton and various ester substituents (Scheme 1B and C). The (co)polymerization of these monomers enables the structural diversification of polymers and the tuning of material properties. Nevertheless, these monomers share a backbone structure that can be converted to a common product through the simultaneous hydrolysis of both the main and side chains. Consequently, this strategy demonstrates that molecular information related to the copolymer composition can be intentionally erased, allowing the recovery of a unified carbon skeleton.
Results and discussion
Monomer design and synthesis
We designed an M1-derived monomer library that allows the simultaneous degradation of the main and side chains via hydrolysis. Accordingly, ester bonds were chosen as linkers for the pendant groups because they are readily hydrolyzable and allow the facile derivatization of monomers bearing a variety of substituents from common degradation products. Two types of monomers were prepared depending on the orientation of the ester linkage: M2-C# and M3-C# (Scheme 1B and C). The “C#” in the compound names denotes the number of carbon atoms in the acyloxy or alkoxy group of each monomer. Monomer designs featuring phenolic hydroxyl groups were excluded from consideration because previous studies indicated that electron-donating groups on the aromatic ring inhibit polymer degradation.
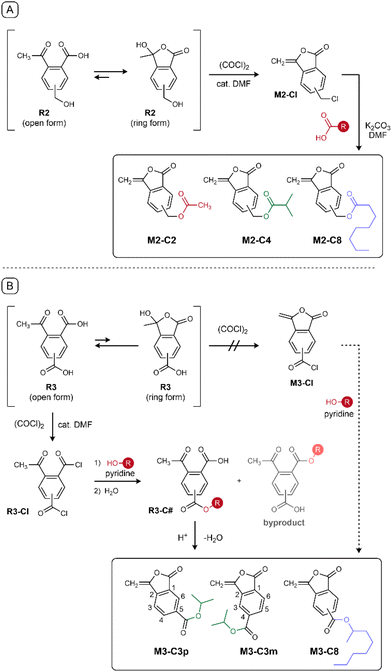
M2 was synthesized following a procedure similar to that of M1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 22 (Scheme 2A). Precursor R2 was prepared from 4-methylphthalic anhydride through three-step reactions (total yield: 30.6%; Fig. S1). Subsequent treatment with oxalyl chloride afforded M2-Cl (yield: 70.2%). Finally, the SN2 reaction with various carboxylate salts afforded M2-C# in yields of up to 98.0%. The chemical structures of the intermediates and products were determined by 1H/13C NMR and IR spectroscopy (Fig. S3–S17).
22 (Scheme 2A). Precursor R2 was prepared from 4-methylphthalic anhydride through three-step reactions (total yield: 30.6%; Fig. S1). Subsequent treatment with oxalyl chloride afforded M2-Cl (yield: 70.2%). Finally, the SN2 reaction with various carboxylate salts afforded M2-C# in yields of up to 98.0%. The chemical structures of the intermediates and products were determined by 1H/13C NMR and IR spectroscopy (Fig. S3–S17).
The synthesis of M3-C# was initially attempted in a similar manner; however, the reaction of precursor R3 with oxalyl chloride did not afford M3-Cl but the dicarboxylic acid chloride R3-Cl. In the synthesis of M1, the reaction of R1 proceeds via a cyclic tautomer.22,23 Meanwhile, an electron-withdrawing substituent on the aromatic ring, such as a carbonyl group, shifts the equilibrium toward the open form;23 the 1H NMR spectrum of R3 certainly indicated that the open form is the major tautomer (Fig. S18). Therefore, cyclization via the cyclic form was relatively inhibited. Consequently, the M3 series was synthesized using an alternative method (Fig. 2B). The obtained R3-Cl was esterified using equimolar alcohol, followed by intramolecular dehydration20 in the presence of an acidic catalyst, yielding the M3-C# series in 8.5–38.7% yields. The low yields were due to the non-selective mono-esterification of R3-Cl (Table S1), and attempts to improve the selectivity using bulkier alcohols at lower temperatures did not increase the yields. The structures of the intermediates and M3-C# were confirmed using 1H/13C NMR and IR spectroscopy (Fig. S19–S27). Although M3-C3p and M3-C3m are structural isomers, they were isolated using column chromatography.
Polymer synthesis and properties
The (co)polymerization of the prepared monomers was conducted using 2,2′-azobis(isobutyronitrile) (AIBN) in N,N-dimethyl formamide (DMF) at 65 °C, resulting in an almost complete monomer conversion regardless of the presence of ester substituents (Table 1). The 1H and 13C NMR spectra confirmed the expected chemical structures of the obtained polymers (Fig. S28–S39), while size-exclusion chromatography (SEC) calibrated using polystyrene standards indicated high number-average molar masses (Mn) of up to 85![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 300 g mol−1 (Fig. S40). In the (co)polymerization system using M2-C8 and M3-C8, the reaction system became highly viscous or solidified, and the resulting (co)polymers exhibited a bimodal molar-mass distribution with broad disparities (Đ).
300 g mol−1 (Fig. S40). In the (co)polymerization system using M2-C8 and M3-C8, the reaction system became highly viscous or solidified, and the resulting (co)polymers exhibited a bimodal molar-mass distribution with broad disparities (Đ).
| Code | M1 (mol%) | M2 (mol%) | Conv.a (%) | Yield (%) |
M
n![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) b (g mol−1) b (g mol−1) |
Đ (−) | T d5 (°C) | Solubilityd |
T
450![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) e (%) e (%) |
Tensile testf | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| DMSO | CHCl3 | Acetone | σ max (MPa) | ε max (%) | E (GPa) | |||||||||
| Polymerization was conducted using AIBN (2 mol%) in DMF (1.0 M) at 65 °C for 18 h.a Monomer conversion was determined by 1H NMR spectroscopy. For copolymerization, conversions of total monomers are shown.b Number-average molar mass and molar mass dispersity were determined by SEC (DMF, 40 °C, polystyrene standards).c 5% weight loss temperature was determined by TG–DTA under N2 flow.d 5 mg polymers in a 0.10 mL solvent. ++: soluble at 25 °C, +: soluble at 80 °C (for DMSO only), and −: insoluble.e Transmittance at 450 nm.f σmax: maximum strength. εmax: maximum elongation. E: Young's modulus. −: too fragile.g Determined by SEC (CHCl3, 40 °C, polystyrene standards).h Soluble fraction only. | ||||||||||||||
| P1 | M1 (100) | >99.0 | 81.4 | 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
2.28 | 326 | + | − | − | − | − | − | − | |
| P2-C2 | M2-C2 (100) | 97.2 | 93.2 | 72![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
2.75 | 260 | ++ | − | − | − | − | − | − | |
| P2-C4 | M2-C4 (100) | >99.0 | 63.2 | 85![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 300 300 |
2.19 | 291 | ++ | − | − | − | − | − | − | |
| P2-C8 | M2-C8 (100) | 98.8 | 68.9 | 9800g | 6.04g | 273 | + | ++ | − | 88.0 | 5.34 ± 0.89 | 11.3 ± 3.3 | 0.18 ± 0.05 | |
| P2-C250C850 | M2-C2 (50) | M2-C8 (50) | >99.0 | 75.8 | (6600)g,h | (4.66)g,h | 284 | ++ | − | − | 89.8 | − | − | − |
| P2-C225C875 | M2-C2 (25) | M2-C8 (75) | >99.0 | 87.0 | 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 900g 900g |
14.2g | 289 | ++ | ++ | − | 89.4 | 2.22 ± 0.58 | 0.64 ± 0.18 | 0.18 ± 0.03 |
| P2-C210C890 | M2-C2 (10) | M2-C8 (90) | >99.0 | 90.6 | 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 700g 700g |
33.5g | 290 | − | ++ | − | 88.3 | 6.51 ± 0.52 | 0.59 ± 0.20 | 2.38 ± 0.17 |
| P2-C425C875 | M2-C4 (25) | M2-C8 (75) | >99.0 | 87.1 | 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000g 000g |
9.00g | 294 | ++ | ++ | − | 91.8 | 4.10 ± 2.47 | 5.40 ± 2.66 | 0.14 ± 0.05 |
| P3-C3p | M3-C3p (100) | >99.0 | 56.8 | 14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200 200 |
2.19 | 283 | ++ | ++ | − | − | − | − | − | |
| P3-C3m | M3-C3m (100) | >99.0 | 72.8 | 80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200 200 |
2.40 | 280- | ++ | ++ | − | − | − | − | − | |
| P3-C8 | M3-C8 (100) | >99.0 | 40.1 | 31![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 600 600 |
3.48 | 282 | + | ++ | ++ | − | − | − | − | |
| P3-C3p50C3m50 | M3-C3p (50) | M3-C3m (50) | 91.8 | 42.8 | 31![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 800 800 |
3.60 | 267 | ++ | ++ | − | 88.1 | − | − | − |
| P3-C3p50C850 | M3-C3p (50) | M3-C8 (50) | >99.0 | 73.1 | 68![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
2.27 | 280 | ++ | ++ | + | − | − | − | − |
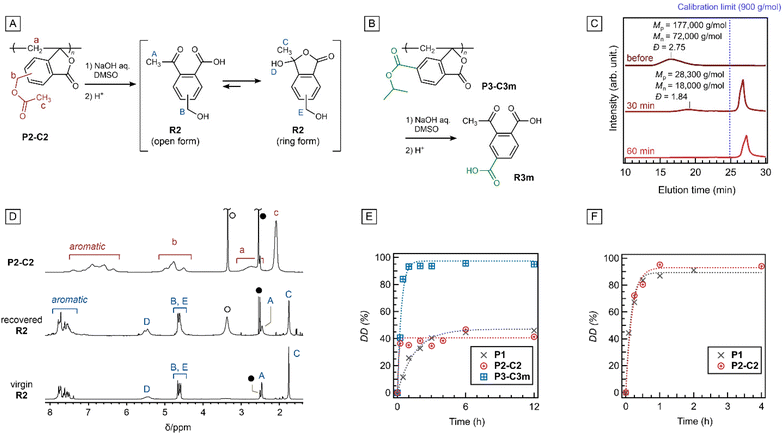
The ester pendants significantly affected the properties of the resulting polymers. For example, an endothermic peak attributable to the melting of crystalline alkyl pendant groups was observed in differential scanning calorimetry (DSC) for (co)polymers with long ester substituents such as P2-C8, P2-C210C890, P2-C225C875, and P3-C8 (Fig. S41). These polymers formed films via hot-pressing (Fig. S42), and dynamic mechanical analysis (DMA) revealed no relaxation peaks (Fig. S43). In fact, no glass transition point (Tg) was observed below the 5% weight loss temperature (Td5), as indicated by thermogravimetry–differential thermal analysis (TG–DTA; Fig. 1A for P2-C210C890 and S44 for the others) and DSC (Table 1). The substituents also affect the polymer solubility. P1, a polymer without ester substituents, was soluble in dimethyl sulfoxide (DMSO) at 80 °C, whereas P2-C2 was soluble at 25 °C. Moreover, the (co)polymers of M2-C8 and M3-C8 were soluble in chloroform (CHCl3), and the homopolymer P3-C8 was soluble in acetone. These results imply that the substituents were effective in weakening the interchain interactions. Accordingly, the effects of the substituents on the mechanical properties of the polymers were significant. Whereas polymers with no or short substituents did not form cast films owing to their brittle and fragile properties, P2-C8 afforded a self-standing soft film with the maximum stress (σmax) of 4.10 ± 2.47 MPa, the maximum elongation (εmax) of 5.40 ± 2.66%, and Young's modulus (E) of 0.14 ± 0.05 GPa. These values were smaller than those of common vinyl polymers in a glassy state, such as polystyrene (σmax ≈ 40–80 MPa, εmax ≈ 1–3%, E ≈ 3–4 GPa) or PMMA (σmax ≈ 40–80 MPa, εmax ≈ 4–10%, E ≈ 2–3 GPa).24–27 The partial introduction of short substituents increased Young's modulus, although a C2 content higher than 25 mol% resulted in a more brittle film (Fig. 1B). The cast films of the (co)polymers exhibited high transparency, with 88.0–91.8% transmittance at 450 nm (T450, Fig. 1C and Fig. S46).
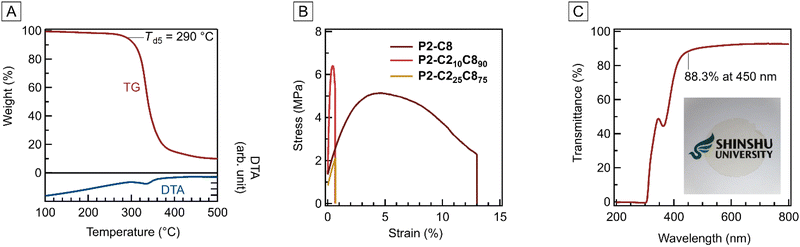 | ||
| Fig. 1 TG–DTA curves (A), stress–strain curves (B), and the UV-vis spectrum of a cast film shown in the photograph (C) of PC210C890 and the related polymers. | ||
Polymer degradation and monomer–precursor recovery
The degradability of P2-C2via hydrolysis was investigated according to our previous report (Fig. 2A).19 A 10 M aqueous solution of NaOH (3.0 molar equivalents relative to the esters) was added to a DMSO solution of P2-C2 at 70 °C, and the reaction progress was monitored by SEC. After 30 min, the Mn of the polymer fraction decreased from 72![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 g mol−1 to 18
000 g mol−1 to 18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 g mol−1, while a sharp signal indicating a small molecule was observed at 27 min of elution time (Fig. 2C). The appearance of small molecules at the early stages of degradation is a typical behavior in depolymerization. The polymer signal disappeared completely after 60 min. A similar experiment was conducted on a larger scale; after quenching the reaction with 1 M aqueous HCl, the product was extracted with ethyl acetate. The 1H NMR spectrum of the extracted product indicated the generation of R2, which was observed as a tautomeric mixture of open and ring forms (Fig. 2D), and the yield of R2 was estimated to be 70.4%. The purity of the obtained R2 was determined to be 99% by high-performance liquid chromatography (HPLC) (Fig. S47). Since R2 is the precursor of all M2-# monomers, as described above and shown in Fig. S1, the recovery of R2 suggests the chemical recyclability of P2-C2. P3-C3m also decomposed into R3 in 85.3% yield with 99% purity under the same conditions (Fig. 2B and Fig. S47 and S48).
000 g mol−1, while a sharp signal indicating a small molecule was observed at 27 min of elution time (Fig. 2C). The appearance of small molecules at the early stages of degradation is a typical behavior in depolymerization. The polymer signal disappeared completely after 60 min. A similar experiment was conducted on a larger scale; after quenching the reaction with 1 M aqueous HCl, the product was extracted with ethyl acetate. The 1H NMR spectrum of the extracted product indicated the generation of R2, which was observed as a tautomeric mixture of open and ring forms (Fig. 2D), and the yield of R2 was estimated to be 70.4%. The purity of the obtained R2 was determined to be 99% by high-performance liquid chromatography (HPLC) (Fig. S47). Since R2 is the precursor of all M2-# monomers, as described above and shown in Fig. S1, the recovery of R2 suggests the chemical recyclability of P2-C2. P3-C3m also decomposed into R3 in 85.3% yield with 99% purity under the same conditions (Fig. 2B and Fig. S47 and S48).
To investigate the effects of the substituents on degradation, hydrolysis was conducted at 25 °C. The degree of degradation (DD), defined as the area ratio of the small molecular peak (molar mass < 900 g mol−1) to the total area in the SEC curve, was plotted against time (Fig. 2E). Degradation of P2-C2 (Fig. S49) was faster than that of P1, although the DDs of both were saturated at ca. 50%. In contrast, P3-C3m underwent almost complete degradation (DD = 95%) within 1 h, even at 25 °C (Fig. S50).
To understand the different degradabilities of P2-C2 and P3-C3m, the reaction mechanisms were investigated. P1, synthesized via free-radical polymerization, exhibited a halving of Mn at the earliest stage of degradation, followed by a linear decrease in Mn with increasing DD (Fig. S51A). Therefore, the degradation of P1 was explained via main-chain scission and subsequent depolymerization (Fig. S52).19 In our previous study, degradation via a radical mechanism was ruled out because depolymerization proceeded even in the presence of excess radical scavengers. Thus, an ionic mechanism was proposed. Since M1 is a styrene derivative, the radical polymerization of M1 was expected to be terminated primarily via a combination mechanism. The head-to-head linkage formed by combination termination was sterically hindered, and the ring-opening of the lactone pendants by hydrolysis immediately led to main-chain scission via an ionic mechanism. The formed fragment chains underwent depolymerization via anionic mechanisms and retro-aldol reactions. The degradation of P1 at room temperature saturated at approximately 50% (Fig. 2E). This was attributed to the suppression of anionic depolymerization, which requires a higher activation energy. In other words, depolymerization should have proceeded exclusively via a retro-aldol reaction. To support this assumption, P1 was prepared via cationic polymerization, which typically does not provide head-to-head linkages (Table S2). For example, polymerization was initiated using BF3·OEt2 and 1-isobutoxyethyl acetate in dichloromethane (CH2Cl2) at 0 °C, yielding P1 (entry 3; yield: 29%, Mn = 59![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 300 g mol−1, Đ = 7.23). Importantly, the hydrolysis of P1, synthesized via cationic polymerization, did not exhibit a halving of Mn at the earliest stage but a linear decrease in Mn (Fig. S51B). This suggests decomposition via only a depolymerization mechanism because of the absence of head-to-head linkages (Fig. S52B). Cationic polymerization generates an unsaturated chain end via β-hydrogen abstraction termination or a carboxylic chain end via hydrolysis termination.
300 g mol−1, Đ = 7.23). Importantly, the hydrolysis of P1, synthesized via cationic polymerization, did not exhibit a halving of Mn at the earliest stage but a linear decrease in Mn (Fig. S51B). This suggests decomposition via only a depolymerization mechanism because of the absence of head-to-head linkages (Fig. S52B). Cationic polymerization generates an unsaturated chain end via β-hydrogen abstraction termination or a carboxylic chain end via hydrolysis termination.
Importantly, both chain ends were converted into β-hydroxy ketones (aldols) via hydrolysis, which undergo stepwise depolymerization via a retro-aldol reaction. Because almost complete depolymerization was achieved at 25 °C (Fig. S52B), the depolymerization driven by the retro-aldol reaction could proceed quantitatively under these conditions. In other words, the incomplete degradation of P1 and P2-C2 (DD = ca. 50%, Fig. 2E), prepared via radical polymerization, was attributed to the depolymerization via the anionic mechanism. In contrast, fast and complete decomposition was observed for P3-C3m (Fig. 2E). This was attributed to the electron-withdrawing carbonyl groups on the aromatic rings, which stabilized the carbanion species for anionic depolymerization. Notably, P1 and P2-C2 were almost completely decomposed within 1 h at 70 °C, which was a sharp contrast to that at 25 °C (Fig. 2F).
Estimation of resource circulation performance
To assess the feasibility of chemical recycling based on the present design concept, the resource circulation performance was estimated for representative P2-C2 and P3-C3 systems. The circulation efficiency per cycle was estimated for the P2-C2 and P3-C3 cases. This estimate includes the yields at each stage: the recovery of monomer precursors, R2 or R3, and the monomer synthesis from the precursors. As described previously, the isolated yield of R3 derived from P3-C3m was 85.3%, whereas the combined yield of M3-C3p (26.2%) and M3-C3m (12.5%) was 38.7%. Due to the low efficiency in the preparation of M3-Cp/m, which involved unselective mono-esterification, the overall yield of M3-C3p/m from P3-C3m was only 33.0%. In contrast, M2-C2 was obtained from R2 in a relatively high yield (68.8%). Because the isolated yield of R2 from P2-C2 was 70.4%, the overall monomer recovery ratio in one cycle was 48.4%. These results indicate that the M2-C# system provides better circulation efficiency, although it remains insufficient for practical resource circulation. Because the key step to determine the efficiency was the monomer synthesis step, rather than the degradation and recovery processes, further optimization of the monomer structure and synthetic route is required.Recovery of monomer precursors from copolymers
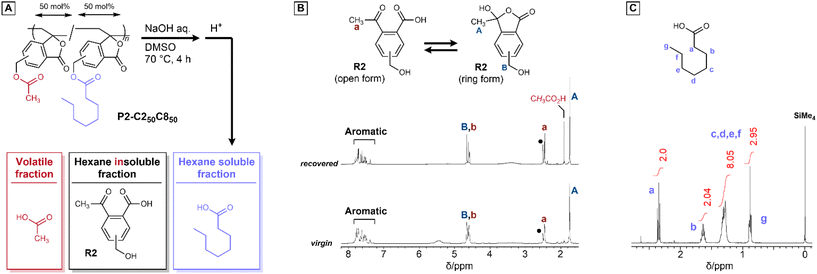
Copolymerization is a powerful method for tuning polymer properties; however, the structural diversity introduced by multiple monomer units generally complicates chemical recycling because different repeat units typically generate distinct degradation products. Methacrylate-based polymers are a notable exception, as they can undergo depolymerization to regenerate the corresponding monomers. This strategy is highly effective but relies on a specific depolymerization mechanism. In this study, we explore another concept that enables property tuning by copolymerization while allowing the unified recovery of a common precursor.As described previously, the copolymerization of M2-C2 and M2-C8 allowed tuning of the polymer solubility and mechanical properties. The hydrolysis of the copolymers led to the decomposition of both the main chain and pendant ester groups, recovering a common monomer precursor. For example, P2-C250C850 was hydrolyzed using aqueous NaOH at 70 °C (Fig. 3A), and R2 was recovered as a hexane-insoluble fraction with a yield of 54.0% (Fig. 3B). In contrast, n-octanoic acid was recovered as a hexane-soluble fraction (Fig. 3C). Because P3-C3 underwent efficient degradation even at 25 °C, the degradation of P3-C350C850 under similar conditions was also investigated. The DD growth of P3-C350C850 was slower than that of P3-C3m, P3-C3p, and P3-C3p50C3m50 (Fig. S55), probably because of the hydrophobic 2-octyl substituents. Nevertheless, the DD reached 95% after 12 h, recovering R3. These results show that the structural information in the copolymers was erased to recover a common precursor, R2 or R3, even if copolymerization introduced structural diversity.
Conclusions
In conclusion, a molecular design strategy that enables the simultaneous decomposition of the main and side chains of vinyl polymers has been demonstrated. The ester pendant groups strongly influenced the solubility and mechanical performance of the polymers, whereas all synthesized polymers decreased their molar mass upon hydrolysis while generating the corresponding monomer precursors, R2 or R3. In particular, polymer P3# was almost completely decomposed with the recovery of R3 (up to 85.3% yield), even at 25 °C. While these polymer systems possess the potential for resource circularity, high-efficiency chemical recycling cannot be ensured due to low-yield issues at each reaction stage. Nevertheless, these results show that polymers designed with diverse side-chain structures can undergo efficient main- and side-chain degradation, yielding common backbone-derived products upon hydrolysis. In other words, the present strategy enables the chemical erasure of structural diversity encoded in the polymer architecture. This study provides a conceptual framework for polymer design in which functional diversity during use is decoupled from molecular convergence upon chemical degradation.Author contributions
YC: data curation, funding acquisition, investigation (all experiments except that involving cationic polymerization), methodology, validation, visualization, and writing – original draft. SH: investigation (cationic polymerization). YK: conceptualization, funding acquisition, project administration, resources, supervision, and writing – review & editing.Conflicts of interest
There are no conflicts to declare.Abbreviations
| AIBN | 2,2′-Azobis(isobutyronitrile) |
| DMF | N,N-Dimethyl formamide |
| DSC | Differential scanning calorimetry |
| DMA | Dynamic mechanical analysis |
| TG–DTA | Thermogravimetry–differential thermal analysis |
| DMSO | Dimethyl sulfoxide |
| SEC | Size-exclusion chromatography |
| HPLC | High-performance liquid chromatography |
| DD | Degree of degradation |
Data availability
The data supporting this manuscript is available as part of the supplementary information (SI): experimental procedure, 1H and 13C NMR spectra, IR spectra, SEC profiles, TGA profiles, DSC profiles, UV spectra, rheological analysis, sample photographs, and supplemental schemes and tables (PDF). See DOI: https://doi.org/10.1039/d6py00151c.Acknowledgements
This study was supported by JSPS KAKENHI; grant numbers: JP 22H02129 and JP 23K23397 (for YK) and JP 23KJ1042 (for YC).References
- C. Lefay and Y. Guillaneuf, Prog. Polym. Sci., 2023, 147, 101764 CrossRef CAS.
- A. M. Mineo and R. Katsumata, Angew. Chem., Int. Ed., 2024, 63, e202316248 CrossRef CAS PubMed.
- S. Bag, D. Ghosh, S. Banerjee and P. De, ACS Appl. Polym. Mater., 2025, 7, 14033–14061 CrossRef CAS.
- H.-W. Lai and M. Ouchi, Chin. J. Polym. Sci., 2025, 43, 887–907 CrossRef CAS.
- Y. Chiba, S. Hirabayashi and Y. Kohsaka, Chem. Sci., 2025, 16, 12804–12811 RSC.
- S. R. Gitter, C. B. Eades, M. E. Lott, R. W. Hughes, A. M. Evans and B. S. Sumerlin, ACS Polym. Au, 2025, 5, 669–679 CrossRef CAS PubMed.
- K. R. Albanese, P. T. Morris, J. Read de Alaniz, C. M. Bates and C. J. Hawker, J. Am. Chem. Soc., 2023, 145, 22728–22734 CrossRef CAS PubMed.
- T. Maity, D. X. Bones, R. W. Hughes, K. C. Stevens, W. M. Gramlich and B. S. Sumerlin, ACS Macro Lett., 2026, 15, 17–32 CrossRef CAS PubMed.
- A. Tardy, J. Nicolas, D. Gigmes, C. Lefay and Y. Guillaneuf, Chem. Rev., 2017, 117, 1319–1406 CrossRef CAS PubMed.
- Y. Deng, F. Mehner and J. Gaitzsch, Macromol. Rapid Commun., 2023, 44, e2200941 CrossRef PubMed.
- F. Sbordone and H. Frisch, Chemistry, 2024, 30, e202401547 CrossRef CAS PubMed.
- Y. Kohsaka, K. Toyama, M. Kawauchi and K. Naganuma, ACS Macro Lett., 2024, 1016–1021 CrossRef CAS PubMed.
- Y. Kohsaka and K. Naganuma, ACS Polym. Au, 2025, 5, 481–487 CrossRef CAS PubMed.
- R. Abu Bakar, J. L. Keddie and P. J. Roth, ChemPlusChem, 2024, 89, e202400034 CrossRef CAS PubMed.
- R. Kawatani, M. Yoshino, H. Matsuzaki, T. Miyamoto and Y. Kohsaka, Macromolecules, 2024, 57, 8861–8868 CAS.
- P. Raffa, D. A. Z. Wever, F. Picchioni and A. A. Broekhuis, Chem. Rev., 2015, 115, 8504–8563 CrossRef CAS PubMed.
- T. Ohtake, H. Ito, K. Horiba, N. Toyoda and Y. Kohsaka, ACS Appl. Polym. Mater., 2023, 5, 4901–4909 CrossRef CAS.
- N. Deka, A. Bera, D. Roy and P. De, ACS Omega, 2022, 7, 36929–36944 CrossRef CAS PubMed.
- Y. Chiba, R. Kawatani and Y. Kohsaka, ACS Macro Lett., 2023, 12, 1672–1676 CrossRef CAS PubMed.
- H. Ito and M. Ueda, Macromolecules, 1990, 23, 2885–2894 CrossRef CAS.
- H. Ito, M. Ueda and T. Ito, J. Photopolym. Sci. Technol., 1990, 3, 335–345 CrossRef CAS.
- S. N. Salazkin and G. S. Chelidze, Polym. Sci., Ser. C, 2009, 51, 126–132 CrossRef.
- J. Finkelstein, T. Williams, V. Toome and S. Traiman, J. Org. Chem., 1967, 32, 3229–3230 CrossRef CAS.
- Polymer Handbook, ed. J. Brandrup and E. H. Immergut, John Wiley & Sons, Nashville, TN, 4th edn, 1999 Search PubMed.
- C. Direksilp and P. Threepopnatkul, Energy Procedia, 2014, 56, 135–141 Search PubMed.
- G. Krala, A. Ubowska and K. Kowalczyk, Polym. Eng. Sci., 2014, 54, 1030–1037 Search PubMed.
- Q. Hu, X. Luo, L. A. Ogunfowora, A. Athaley, J. S. DesVeaux, B. C. Klein, S. Xu, P. Wu, Z. Wei, C. Lin, T. Haraniya, D. Maiorano, B. Boudouris, J. Mei, M. Urgun-Demirtas, G. T. Beckham, B. M. Savoie and L. Dou, Nat. Chem. Eng., 2025, 2, 130–141 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |