Open Access Article
Open Access ArticleSynthesis of macro-rotaxanes via direct trapping of multicyclic poly(n-butyl acrylate) in a cross-linked network
Kotaro
Ibe
a,
Yamato
Ebii
a,
Minami
Ebe
 b,
Kazushige
Suzuki
b,
Takayuki
Kurokawa
b,
Kazushige
Suzuki
b,
Takayuki
Kurokawa
 c,
Takuya
Yamamoto
c,
Takuya
Yamamoto
 b,
Feng
Li
b,
Feng
Li
 b,
Takuya
Isono
b,
Takuya
Isono
 *b and
Toshifumi
Satoh
*b and
Toshifumi
Satoh
 *bde
*bde
aGraduate School of Chemical Sciences and Engineering, Hokkaido University, Kita 13, Nishi 8, Kita-ku, Sapporo, Hokkaido 060-8628, Japan
bFaculty of Engineering, Hokkaido University, Kita 13, Nishi 8, Kita-ku, Sapporo, Hokkaido 060-8628, Japan. E-mail: isono.t@eng.hokudai.ac.jp; satoh@eng.hokudai.ac.jp
cDivision of Soft Matter, Faculty of Advanced Life Science, Hokkaido University, Sapporo 001-0021, Japan
dList Sustainable Digital Transformation Catalyst Collaboration Research Platform, Institute for Chemical Reaction Design and Discovery, Hokkaido University, Kita 21, Nishi 10, Kita-ku, Sapporo, Hokkaido 001-0021, Japan
eDepartment of Chemical & Materials Engineering, National Central University, Taoyuan 320317, Taiwan
First published on 31st March 2026
Abstract
Macro-rotaxanes, in which both the linear and cyclic components are polymers, remain largely unexplored in terms of their formation and potential material applications due to the synthetic challenges associated with cyclic polymers. In this study, macro-rotaxane formation was investigated by means of the photoradical polymerization of n-butyl acrylate (BA) and a cross-linker in the presence of multicyclic poly(n-butyl acrylate) (mc-PBA). For this purpose, mc-PBAs with different average numbers of cyclic units (Nring) and cyclic unit molecular weights (Mn,ring) were synthesized via the cyclopolymerization of an α,ω-dinorbornenyl-functionalized PBA macromonomer. Subsequently, polymer networks incorporating mc-PBA were prepared and the trapping efficiency of mc-PBA within these networks was evaluated. The fraction of mc-PBA trapped as the macro-rotaxane increased with increasing Nring and Mn,ring. Additionally, dynamic viscoelastic measurements revealed that the incorporation of mc-PBA enhanced the damping properties of the resulting PBA networks. Notably, these properties were retained even after the removal of untrapped mc-PBA by solvent washing, demonstrating that mc-PBA functioned effectively as a non-leaching additive.
Introduction
Rotaxanes are supramolecular structures in which a linear molecule penetrates a cyclic component and is stabilized in the presence of bulky stoppers (Fig. 1a). Owing to the presence of such mechanical bonds, rotaxanes exhibit molecular mobility that cannot be achieved through covalent bonding, thereby reflecting one of their most distinctive features. By exploiting the mobility of their cyclic component, rotaxanes have been extensively studied for applications in molecular machines1–3 and drug delivery systems,4–6 with interest in these structures recently extending to polymer materials. In particular, the use of topologically cross-linked polymers incorporating rotaxanes as movable cross-linking points has emerged as an effective strategy for enhancing the mechanical properties of conventional materials owing to their superior stress dissipation capability.7–9Various strategies for constructing rotaxanes have been reported, most of which employ small cyclic molecules, such as crown ethers10–12 or cyclodextrins,13–15 as cyclic components. For example, Stoddart et al.10 demonstrated that rotaxanes can be formed by exploiting ion–dipole interactions and hydrogen bonding between crown ethers and ammonium-containing linear components. Additionally, Harada et al.13 established a method in which hydrophobic interactions between cyclodextrins and linear components serve as the driving force for rotaxane formation (Fig. 1b). In principle, the self-assembly of cyclic and linear components to form pseudorotaxanes, which are precursors of rotaxanes, is entropically unfavorable. Thus, strong intermolecular interactions, including ion–dipole interactions, hydrogen bonding, hydrophobic interactions, or π–π stacking,16–19 are indispensable as driving forces in (pseudo)rotaxane formation. Consequently, the accessible combinations of cyclic and linear components remain limited.
Our group recently investigated a new class of rotaxanes, termed macro-rotaxanes, in which both the linear and cyclic components consist of polymers. In contrast to small-molecule-based rotaxanes, macro-rotaxane formation does not require specific intermolecular attractive interactions to generate the pseudorotaxane, which is expected to allow a broader range of combinations of cyclic and linear components. When both the linear and cyclic components are composed of the same polymer, the linear component spontaneously threads through the polymer ring, resulting in the formation of a pseudomacro-rotaxane. Such spontaneous threading has been supported by both experimental20,21 and computational studies.22 From an experimental perspective, it was demonstrated that the addition of a small amount of linear polystyrene (PS) to cyclic PS leads to a substantial increase in viscoelasticity, indicating that cyclic PS acts as a dynamic cross-linking point through threading with the linear PS.20 In another study,23 the possibility of pseudomacro-rotaxane formation using a linear polymer in combination with a monocyclic polymer was experimentally suggested using small-angle neutron scattering. Moreover, computational studies based on molecular dynamics simulations further demonstrated that blends of cyclic and linear polymers exhibit a negative Flory–Huggins interaction parameter, indicating that pseudomacro-rotaxane formation could occur spontaneously.22
The threading structures generated from cyclic and linear polymers have been discussed historically in terms of “topological trapping”.24–30 For instance, Semlyen et al.24 reported that up to 94% of monocyclic polydimethylsiloxane (PDMS) molecules were trapped within a polymer network via the cross-linking of end-functionalized PDMS chains. Additionally, Waymouth et al.25 performed the radical polymerization of 2-hydroxyethyl methacrylate in the presence of monocyclic poly(alkylene phosphate), revealing that 36% of the cyclic polymer was incorporated into the resulting network. These networks satisfy the structural requirements for a rotaxane architecture, wherein the cross-linking points serve as stoppers to produce macro-rotaxanes. Although such macro-rotaxanes have been realized in practice, studies have primarily focused on assessing the presence and formation efficiency of cyclic polymers, and their potential as functional material components remains largely unexplored. This stems from the fact that previous studies have primarily focused on developing cyclic polymer synthesis methods rather than investigating the material properties.
Our group previously reported a cyclopolymerization approach for the facile synthesis of various well-defined multicyclic polymers, in which linear macromonomers functionalized at both ends underwent successive reactions to generate multiple rings in a single step.31 This method relies on ring-opening metathesis polymerization (ROMP) initiated by a third-generation Grubbs catalyst (G3), which serves as the elementary ring-forming process. Specifically, α,ω-dinorbornenyl-functionalized linear polymers were employed as macromonomers, and ROMP conducted under high-dilution conditions promoted alternating intramolecular cyclization and intermolecular propagation to afford well-defined multicyclic architectures. This approach was applicable to a wide variety of polymer species, including poly(L-lactic acid), PDMS,32 and PS.33
Furthermore, by combining this facile cyclopolymerization approach with topological trapping based on the cross-linking of linear polymers, our group demonstrated that macro-rotaxanes can be efficiently formed when multicyclic PDMS (mc-PDMS) is employed as the cyclic component (Fig. 1c).32 These results provided concrete experimental validation of the macro-rotaxane concept. Remarkably, mc-PDMS exhibited superior efficiency in macro-rotaxane formation compared with its monocyclic counterpart. Additionally, using coarse-grained molecular dynamics simulations, Hagita et al.34 demonstrated that the extent of linear polymer threading increased from monocyclic to bicyclic and tricyclic polymers. Beyond the fundamental interest in macro-rotaxane formation, the resulting networks were useful as damping materials, as the cyclic components relaxed independently from the cross-linked network. Furthermore, the macro-rotaxanes composed of mc-PDMS toughened the polymer network.35 These reports demonstrate the advantages of multicyclic polymers as cyclic components for macro-rotaxanes. However, despite such advances, the formation of macro-rotaxanes using multicyclic polymers remains largely unexplored.
The aim of the current study is therefore to extend the scope of this cyclopolymerization approach to the formation of macro-rotaxanes using a method distinct from our previous end-cross-linking strategy, thereby opening additional possibilities for their application (Fig. 2). For this purpose, poly(n-butyl acrylate) (PBA), a polymer widely used in adhesives and damping materials, was selected as the model system. Additionally, α,ω-dinorbornenyl-functionalized PBA (NB–PBA–NB) was used as a macromonomer to synthesize multicyclic PBA (mc-PBA) with varying numbers of cyclic units and molecular weights. Thus, mc-PBA-blended PBA networks, in which mc-PBA was topologically trapped within the cross-linked PBA network, were constructed via the photoradical polymerization of n-butyl acrylate (BA) and a cross-linker in the presence of mc-PBA. Systematic evaluation of the macro-rotaxane formation efficiency was subsequently performed to investigate the influence of different molecular parameters. Furthermore, dynamic viscoelastic measurements were carried out to reveal the effects of mc-PBA on damping properties and as a non-leaching additive to facilitate the development of materials exhibiting stable performance over extended periods.
 | ||
| Fig. 2 (a) Synthesis of mc-PBA via cyclopolymerization. (b) Macro-rotaxane formation via photoradical polymerization of BA in the presence of mc-PBA. | ||
Results and discussion
Synthesis of the macromonomer
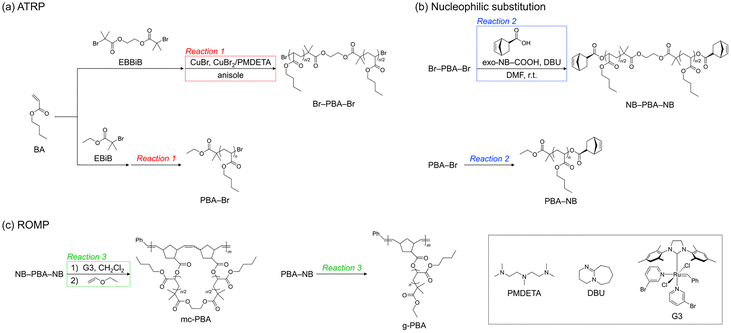
NB–PBA–NB, serving as a macromonomer for mc-PBA, was synthesized via atom transfer radical polymerization (ATRP) of BA, followed by nucleophilic substitution of the bromo-terminals with exo-5-norbornenecarboxylic acid (exo-NB–COOH) (Schemes 1(a) and (b)).Initially, dibromo-terminated PBA (Br–PBA–Br) was prepared via ATRP, targeting a number-average molecular weight (Mn) of ∼10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000. Ethylene bis(2-bromoisobutyrate) (EBBiB), copper(I) bromide (CuBr), N,N,N′,N″,N″-pentamethyldiethylenetriamine (PMDETA), and copper(II) bromide (CuBr2) were employed as the initiator, catalyst, ligand, and deactivator, respectively ([BA]0/[EBBiB]0/[CuBr]/[CuBr2]/[PMDETA] = 224
000. Ethylene bis(2-bromoisobutyrate) (EBBiB), copper(I) bromide (CuBr), N,N,N′,N″,N″-pentamethyldiethylenetriamine (PMDETA), and copper(II) bromide (CuBr2) were employed as the initiator, catalyst, ligand, and deactivator, respectively ([BA]0/[EBBiB]0/[CuBr]/[CuBr2]/[PMDETA] = 224![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.00
1.00![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.95
0.95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.05
0.05![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.10). Polymerization was conducted in a mixed solvent of anisole and dry N,N-dimethylformamide (DMF) at 70 °C for 2.5 h under an argon atmosphere (Table 1, run 1). The Mn of the resulting Br–PBA–Br was determined by proton nuclear magnetic resonance (1H NMR) spectroscopy (Mn,NMR), giving a value of 11
1.10). Polymerization was conducted in a mixed solvent of anisole and dry N,N-dimethylformamide (DMF) at 70 °C for 2.5 h under an argon atmosphere (Table 1, run 1). The Mn of the resulting Br–PBA–Br was determined by proton nuclear magnetic resonance (1H NMR) spectroscopy (Mn,NMR), giving a value of 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 600, while size-exclusion chromatography (SEC) using polystyrene standards (Mn,SEC) yielded a value of 10
600, while size-exclusion chromatography (SEC) using polystyrene standards (Mn,SEC) yielded a value of 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 400, with a molecular weight dispersity (Đ) of 1.07 (Fig. S1 and S2).
400, with a molecular weight dispersity (Đ) of 1.07 (Fig. S1 and S2).
| Run | Sample | Time [h] |
M
n,NMR![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) c c |
M
n,SEC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) d d |
Đ![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) d d |
M
n,MALS![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) e e |
Đ
MALS![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) e e |
|---|---|---|---|---|---|---|---|
a Polymerization conditions: Ar atmosphere; solvent, anisole/DMF; [BA]0/[initiator]0/[CuBr]/[CuBr2]/[PMDETA] = 224![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.00 1.00![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.95 0.95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.05 0.05![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.10 (run 1), 511 1.10 (run 1), 511![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.00 1.00![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.95 0.95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.05 0.05![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.10 (run 3), and 770 1.10 (run 3), and 770![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.00 1.00![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.95 0.95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.05 0.05![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.10 (run 5).
b Reaction conditions: solvent, dry DMF; temperature, r.t.; [Br–PBA–Br]0/[exo-NB–COOH]0/[DBU] = 1.0 1.10 (run 5).
b Reaction conditions: solvent, dry DMF; temperature, r.t.; [Br–PBA–Br]0/[exo-NB–COOH]0/[DBU] = 1.0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4.0 4.0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4.0 (run 2), 1.0 4.0 (run 2), 1.0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4.3 4.3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4.1 (run 4), and 1.0 4.1 (run 4), and 1.0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4.2 4.2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4.1 (run 6); [Br–PBA–Br]0 = 100 g L−1.
c Determined by 1H NMR spectroscopy performed in CDCl3 (Fig. S1, S3, S5, S11, S13, and S15).
d Determined by SEC in THF using PS standards (Fig. S2, S4, S6, S12, S14, and S16).
e Calculated using multi-angle light scattering. 4.1 (run 6); [Br–PBA–Br]0 = 100 g L−1.
c Determined by 1H NMR spectroscopy performed in CDCl3 (Fig. S1, S3, S5, S11, S13, and S15).
d Determined by SEC in THF using PS standards (Fig. S2, S4, S6, S12, S14, and S16).
e Calculated using multi-angle light scattering.
|
|||||||
| 1 | Br−PBA10k−Br | 2.5 | 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 600 600 |
10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 400 400 |
1.07 | — | — |
| 2 | NB–PBA10k–NB | 166 | 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 900 900 |
1.07 | 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200 200 |
1.15 |
| 3 | Br−PBA19k−Br | 7.0 | 23![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 400 400 |
20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 900 900 |
1.07 | — | — |
| 4 | NB–PBA19k–NB | 161 | 24![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 300 300 |
21![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 500 |
1.08 | 19![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200 200 |
1.05 |
| 5 | Br−PBA32k−Br | 13.0 | 41![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 900 900 |
35![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
1.10 | — | — |
| 6 | NB–PBA32k–NB | 138 | 39![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 700 700 |
37![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 100 100 |
1.10 | 31![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 500 |
1.03 |
Subsequently, NB–PBA–NB was synthesized via the nucleophilic substitution of Br–PBA–Br with exo-NB–COOH. This reaction was carried out according to a previously reported method for similar end-group functionalization of polyacrylates.36 The reaction was performed at room temperature in dry DMF for 166 h, employing 1,8-diazabicyclo[5.4.0]-7-undecene (DBU) as the base ([Br–PBA–Br]0/[exo-NB–COOH]0/[DBU] = 1.0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4.0
4.0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4.0, [Br–PBA–Br]0 = 100 g L−1; Table 1, run 2). 1H NMR analysis confirmed the quantitative conversion of Br–PBA–Br into NB–PBA–NB, as evidenced by a downfield shift of the chain-end methine protons from 4.29–3.84 to 5.03–4.90 ppm, together with the appearance of characteristic norbornenyl olefinic and bridgehead proton signals at 6.21–6.06 and 3.21–2.88 ppm, respectively. The expected integration ratios were calculated for all characteristic peaks of NB–PBA–NB, confirming sufficient end-group fidelity (Fig. S11). Additionally, SEC revealed a monomodal elution peak both before and after the reaction, indicating the absence of side reactions and confirming the selective progression of the desired substitution (Fig. S12). The absolute Mn (Mn,MALS) of the product, determined by SEC coupled with multi-angle light scattering and viscometry (SEC-MALS-Visco), was 10
4.0, [Br–PBA–Br]0 = 100 g L−1; Table 1, run 2). 1H NMR analysis confirmed the quantitative conversion of Br–PBA–Br into NB–PBA–NB, as evidenced by a downfield shift of the chain-end methine protons from 4.29–3.84 to 5.03–4.90 ppm, together with the appearance of characteristic norbornenyl olefinic and bridgehead proton signals at 6.21–6.06 and 3.21–2.88 ppm, respectively. The expected integration ratios were calculated for all characteristic peaks of NB–PBA–NB, confirming sufficient end-group fidelity (Fig. S11). Additionally, SEC revealed a monomodal elution peak both before and after the reaction, indicating the absence of side reactions and confirming the selective progression of the desired substitution (Fig. S12). The absolute Mn (Mn,MALS) of the product, determined by SEC coupled with multi-angle light scattering and viscometry (SEC-MALS-Visco), was 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200. For clarity, the polymers were denoted as Br–PBA10k–Br and NB–PBA10k–NB, based on the Mn,MALS values of NB–PBA–NB. Furthermore, by varying the feed ratios of [BA]0/[EBBiB]0/[CuBr]/[CuBr2]/[PMDETA] and [Br–PBA–Br]0/[exo-NB–COOH]0/[DBU], a series of PBA macromonomers with different molecular weights (i.e., 19k and 32k) were synthesized, as summarized in Table 1 (runs 3–6; Fig. S3–S6 and S13–S16).
200. For clarity, the polymers were denoted as Br–PBA10k–Br and NB–PBA10k–NB, based on the Mn,MALS values of NB–PBA–NB. Furthermore, by varying the feed ratios of [BA]0/[EBBiB]0/[CuBr]/[CuBr2]/[PMDETA] and [Br–PBA–Br]0/[exo-NB–COOH]0/[DBU], a series of PBA macromonomers with different molecular weights (i.e., 19k and 32k) were synthesized, as summarized in Table 1 (runs 3–6; Fig. S3–S6 and S13–S16).
Synthesis of mc-PBA via cyclopolymerization
mc-PBA was synthesized via the cyclopolymerization of NB–PBA10k–NB (Table 2, run 1; Scheme 1c). The reaction was performed under an argon atmosphere in dry CH2Cl2 at room temperature for 3.5 h. To suppress intermolecular reactions and promote intramolecular cyclization, high-dilution conditions were employed, with [NB–PBA10k–NB]0 = 0.11 mmol L−1. To obtain mc-PBA10k containing eight cyclic units, an [NB–PBA10k–NB]0/[G3]0 feed ratio of 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 was used. Even after completion of the reaction, no gelation was observed, and only soluble products were obtained. The SEC profile of the crude product revealed a shoulder peak in the lower-molecular-weight region arising from the mc-PBA species containing fewer cyclic units, which were removed by preparative SEC (Fig. S20). The 1H NMR spectrum of the purified product showed complete disappearance of the signals corresponding to the terminal norbornenyl group of NB–PBA10k–NB (Ha, Hb, 6.21–6.06 ppm; Hc, Hd, 3.21–2.88 ppm), while the overlapping broad signals (5.62–4.74 and 4.36–2.00 ppm) corresponded to protons of the polynorbornene backbone (Fig. 3a), indicating successful ROMP. SEC demonstrated that the elution peak of the product shifted to a higher-molecular-weight region compared with NB–PBA10k–NB (Mn,SEC = 48
1 was used. Even after completion of the reaction, no gelation was observed, and only soluble products were obtained. The SEC profile of the crude product revealed a shoulder peak in the lower-molecular-weight region arising from the mc-PBA species containing fewer cyclic units, which were removed by preparative SEC (Fig. S20). The 1H NMR spectrum of the purified product showed complete disappearance of the signals corresponding to the terminal norbornenyl group of NB–PBA10k–NB (Ha, Hb, 6.21–6.06 ppm; Hc, Hd, 3.21–2.88 ppm), while the overlapping broad signals (5.62–4.74 and 4.36–2.00 ppm) corresponded to protons of the polynorbornene backbone (Fig. 3a), indicating successful ROMP. SEC demonstrated that the elution peak of the product shifted to a higher-molecular-weight region compared with NB–PBA10k–NB (Mn,SEC = 48![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 600, Đ = 1.16), as depicted in Fig. 3b. Subsequently, the average number of cyclic units (Nring) was calculated by dividing the Mn,MALS value of mc-PBA by that of NB–PBA–NB. For NB–PBA10k–NB, the Mn,MALS of mc-PBA was 83
600, Đ = 1.16), as depicted in Fig. 3b. Subsequently, the average number of cyclic units (Nring) was calculated by dividing the Mn,MALS value of mc-PBA by that of NB–PBA–NB. For NB–PBA10k–NB, the Mn,MALS of mc-PBA was 83![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200; therefore, the resulting mc-PBA exhibited an Nring value of ∼8 and was denoted as 8c-PBA10k. As reported previously, Nring can be tuned by varying the [macromonomer]0/[G3]0 feed ratio during cyclopolymerization. Accordingly, increasing the [NB–PBA10k–NB]0/[G3]0 ratio to 48
200; therefore, the resulting mc-PBA exhibited an Nring value of ∼8 and was denoted as 8c-PBA10k. As reported previously, Nring can be tuned by varying the [macromonomer]0/[G3]0 feed ratio during cyclopolymerization. Accordingly, increasing the [NB–PBA10k–NB]0/[G3]0 ratio to 48![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 enabled the synthesis of mc-PBA10k with a greater Nring (34c-PBA10k; Mn,MALS = 348
1 enabled the synthesis of mc-PBA10k with a greater Nring (34c-PBA10k; Mn,MALS = 348![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 300, ĐMALS = 1.62), as detailed in Table 2 (run 2; Fig. S21 and S22).
300, ĐMALS = 1.62), as detailed in Table 2 (run 2; Fig. S21 and S22).
| Run | Sample | Macromonomer | [MM]0/[G3]0 | [MM]0 [mmol L−1] | Time [h] |
M
n,SEC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) b b |
Đ![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) b b |
M
n,MALS![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) c c |
Đ
MALS![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) c c |
M
n,ring![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) c c |
N ring | Yield [%] |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| a Polymerization conditions: solvent, dry CH2Cl2; temperature, r.t. b Determined by SEC in THF using PS standards (Fig. S20, S22, S24, S26, S28 and S30). c Calculated using multi-angle light scattering. d N ring was calculated for the obtained mc-PBA using the expression: (Mn,MALS of mc-PBA)/(Mn,ring). | ||||||||||||
| 1 | 8c-PBA10k | NB–PBA10k–NB | 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
0.11 | 3.5 | 48![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 600 600 |
1.16 | 83![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200 200 |
1.15 | 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200 200 |
8 | 69.0 |
| 2 | 34c-PBA10k | NB–PBA10k–NB | 48![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
0.11 | 27.0 | 110![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200 200 |
1.34 | 348![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 300 300 |
1.62 | 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200 200 |
34 | 50.0 |
| 3 | 12c-PBA19k | NB–PBA19k–NB | 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
0.10 | 27.0 | 108![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 300 300 |
1.40 | 222![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 400 400 |
1.75 | 19![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200 200 |
12 | 68.0 |
| 4 | 5c-PBA32k | NB–PBA32k–NB | 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
0.07 | 8.5 | 118![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 100 100 |
1.39 | 162![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 100 100 |
1.27 | 31 500 | 5 | 61.7 |
| 5 | 8c-PBA32k | NB–PBA32k–NB | 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
0.07 | 7.5 | 159![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
1.43 | 256![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 100 100 |
1.47 | 31![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 500 |
8 | 56.0 |
| 6 | 13c-PBA32k | NB–PBA32k–NB | 19![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
0.07 | 25.0 | 221![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 700 700 |
1.58 | 403![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 800 800 |
2.38 | 31![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 500 |
13 | 45.7 |
To systematically evaluate the effects of Nring and the cyclic unit molecular weight (Mn,ring) on the efficiency of macro-rotaxane formation, mc-PBAs with different Nring values ranging from 5 to 13 and Mn,ring values of 19k and 32k were synthesized and prepared via the cyclopolymerization of NB–PBA19k–NB and NB–PBA32k–NB (Table 2, runs 3–6; Fig. S23–S30).
Synthesis of the reference samples
Linear PBA specimens with a high Mn content and grafted PBA were prepared as reference samples for mc-PBA (Scheme 1; Fig. S7–S10, S17, S18, S31, and S32). The linear PBA was designed to possess an Mn comparable to that of mc-PBA. In contrast, the graft PBA was designed such that the Mn of the graft side chain units was approximately half that (Mn,ring/2) of the cyclic units in mc-PBA, whereas the number of graft units was set to approximately twice the number of cyclic units (2Nring). Linear PBA was synthesized via ATRP using a [BA]0/[EBBiB]0/[CuBr]/[CuBr2]/[PMDETA] feed ratio of 5735![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.00
1.00![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5.69
5.69![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.32
0.32![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6.67, as described above. For the graft PBA, ω-norbornenyl-functionalized PBA (PBA-NB) was prepared by ATRP using ethyl 2-bromoisobutyrate (EBiB) followed by treatment with exo-NB–COOH, and subsequent ROMP afforded the desired product. The resulting final products were denoted as Br–PBA175k–Br and 19g-PBA15k based on their Mn,MALS values. For 19g-PBA15k, the prefix “19g” denotes the average number of graft units, which was calculated by dividing Mn,MALS by the corresponding value for the macromonomer.
6.67, as described above. For the graft PBA, ω-norbornenyl-functionalized PBA (PBA-NB) was prepared by ATRP using ethyl 2-bromoisobutyrate (EBiB) followed by treatment with exo-NB–COOH, and subsequent ROMP afforded the desired product. The resulting final products were denoted as Br–PBA175k–Br and 19g-PBA15k based on their Mn,MALS values. For 19g-PBA15k, the prefix “19g” denotes the average number of graft units, which was calculated by dividing Mn,MALS by the corresponding value for the macromonomer.
Synthesis and characterization of the mc-PBA-blended PBA network
Using mc-PBA specimens with various Nring and Mn,ring values, macro-rotaxanes were generated via photoradical polymerization in the presence of BA, together with ethylene glycol dimethacrylate (EGDM) as the cross-linker and 2,2-diethoxyacetophenone (DEAP) as the photoinitiator. The reaction was carried out in a quartz vessel under ultraviolet (UV) irradiation (352 nm) at room temperature for 1 h. mc-PBA was incorporated into a BA/EGDM/DEAP mixture (97![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3
3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, w/w/w) at loadings of 20, 40, and 60 wt%. It is anticipated that the PBA chains formed during radical polymerization thread through the mc-PBA rings. Subsequently, the cross-linking points, generated by the copolymerization with EGDM, act as end-capping agents to prevent dethreading of mc-PBA, thereby successfully constructing the target macro-rotaxane. Indeed, a control experiment without adding EGDM confirmed the dethreading of mc-PBA and linear PBA (Fig. S34). Assuming the successful formation of the macro-rotaxane, the mc-PBA rings would remain trapped within the network, even after solvent swelling. The degree of macro-rotaxane formation was therefore assessed by determining the fraction of unextracted mc-PBA. Specifically, Soxhlet extractions with toluene were carried out for the 60 wt% mc-PBA-blended PBA networks over 4 days. The soluble fractions obtained by subsequent solvent evaporation were assumed to contain both free mc-PBA and the PBA sol fraction that was not incorporated into the network. To estimate the sol fraction, the same extraction procedure was applied to pristine PBA networks prepared without mc-PBA. As a result, 61.7 mg of the PBA sol fraction was extracted per gram of pristine PBA network (average of three independent experiments). Using this value, the fraction of mc-PBA trapped in the networks (%Trapped) was calculated to be 20–85% according to the following equation (Table 3, runs 1–7):
1, w/w/w) at loadings of 20, 40, and 60 wt%. It is anticipated that the PBA chains formed during radical polymerization thread through the mc-PBA rings. Subsequently, the cross-linking points, generated by the copolymerization with EGDM, act as end-capping agents to prevent dethreading of mc-PBA, thereby successfully constructing the target macro-rotaxane. Indeed, a control experiment without adding EGDM confirmed the dethreading of mc-PBA and linear PBA (Fig. S34). Assuming the successful formation of the macro-rotaxane, the mc-PBA rings would remain trapped within the network, even after solvent swelling. The degree of macro-rotaxane formation was therefore assessed by determining the fraction of unextracted mc-PBA. Specifically, Soxhlet extractions with toluene were carried out for the 60 wt% mc-PBA-blended PBA networks over 4 days. The soluble fractions obtained by subsequent solvent evaporation were assumed to contain both free mc-PBA and the PBA sol fraction that was not incorporated into the network. To estimate the sol fraction, the same extraction procedure was applied to pristine PBA networks prepared without mc-PBA. As a result, 61.7 mg of the PBA sol fraction was extracted per gram of pristine PBA network (average of three independent experiments). Using this value, the fraction of mc-PBA trapped in the networks (%Trapped) was calculated to be 20–85% according to the following equation (Table 3, runs 1–7):| Run | Additive | Mass of soluble fraction per g of network [mg] | %Trapped [%] |
|---|---|---|---|
a Polymerization conditions: temperature, r.t.; BA/EGDM/DEAP = 97![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (w/w/w); mixture of BA, EGDM, and DEAP/additive = 100 1 (w/w/w); mixture of BA, EGDM, and DEAP/additive = 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 60 (w/w). 60 (w/w).
|
|||
| 1 | — | 61.7 ± 8.0 | — |
| 2 | 8c-PBA10k | 337.2 | 20.4 |
| 3 | 34c-PBA10k | 259.8 | 41.0 |
| 4 | 12c-PBA19k | 169.2 | 65.2 |
| 5 | 5c-PBA32k | 198.4 | 57.3 |
| 6 | 8c-PBA32k | 147.4 | 70.9 |
| 7 | 13c-PBA32k | 96.4 | 84.6 |
| 8 | Br–PBA175k–Br | 456.8 | <0.1 |
| 9 | 19g-PBA15k | 456.8 | <0.1 |
Despite sub-quantitative macro-rotaxane formation, the data demonstrated that threading took place to some extent, with the efficiency governed by the Mn,ring and Nring values of mc-PBA.
To further confirm macro-rotaxane formation, the same procedure was applied to the PBA networks prepared in the presence of 60 wt% Br–PBA175k–Br and 19g-PBA15k (Table 3, runs 8 and 9). Owing to their acyclic nature, the added PBA chains are unable to mechanically interlock with the PBA network and should be completely extracted during solvent swelling. As expected, Soxhlet extraction revealed trapped fractions of <0.1% for the Br–PBA175k–Br and 19g-PBA15k-blended PBA networks. Focusing on the Br–PBA175k–Br- and 5c-PBA32k-blended PBA networks, it was found that although both additives had comparable Mn values, the added Br–PBA175k–Br was completely extracted, whereas 57.3% of 5c-PBA32k remained trapped within the network (Table 3, runs 5 and 8). Furthermore, despite the similar molecular architectures and Mn,MALS values of the 19g-PBA15k and 8c-PBA32k-blended PBA networks, 19g-PBA15k was completely extracted due to its acyclic structure, whereas 70.9% of 8c-PBA32k remained trapped within the network (Table 3, runs 6 and 9). These results confirm that the presence of a cyclic architecture is essential for entrapment within the network. Therefore, the observed retention of mc-PBA provides strong evidence for successful macro-rotaxane formation.
It is also possible that entrapment of high-molecular-weight mc-PBA arises from simple physical constraints within the network lattice rather than the expected macro-rotaxane formation. To examine this possibility, a series of PBA networks were prepared containing 12c-PBA19k (60 wt%) with varying cross-link densities. This was achieved by systematically altering the feed ratio of the EGDM cross-linker from 1 to 6 wt%. The theoretical molecular weights between the cross-linking points (Mc) determined by the feed ratio were ∼9800, 3200, and 1600 for 1, 3, and 6 wt% EGDM, respectively. Subsequently, Soxhlet extractions were performed on the obtained PBA networks, and the trapped fraction was evaluated (Table 4 and Fig. 4a). The 12c-PBA19k-blended PBA network prepared using 1 wt% EGDM exhibited a slightly lower trapped fraction (52.2%) than those prepared using 3 and 6 wt% EGDM, due to the increased number of network defects caused by the lower cross-link density. Nevertheless, all three samples exhibited similar trapped fractions (52.2–65.2%). In particular, the 12c-PBA19k-blended PBA network prepared using 6 wt% EGDM exhibited a value similar to that of the network prepared using 3 wt% EGDM, despite the cross-link density being doubled and the physical constraints imposed on mc-PBA being significantly increased. These results confirm that mc-PBA formed macro-rotaxanes that dominated the trapping behavior within the PBA network.
| Run | Additive | BA/EGDM/DEAP (w/w/w) |
M
c![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) b b |
Mass of soluble fraction per g of network [mg] | %Trapped [%] |
|---|---|---|---|---|---|
a Polymerization conditions: temperature, r.t.; mixture of BA, EGDM, and DEAP/additive = 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 60 (w/w).
b Determined from the BA/EGDM feed ratio. 60 (w/w).
b Determined from the BA/EGDM feed ratio.
|
|||||
| 1 | 99![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
9800 | 115.4 ± 10.4 | — | |
| 2 | — | 97![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
3200 | 61.7 ± 8.0 | — |
| 3 | 94![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
1600 | 34.5 ± 5.9 | — | |
| 4 | 99![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
9800 | 251.8 | 52.2 | |
| 5 | 12c-PBA19k | 97![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
3200 | 169.2 | 65.2 |
| 6 | 94![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
1600 | 169.7 | 60.6 | |
To investigate the effect of Nring on the efficiency of macro-rotaxane formation, the trapped fractions of a series of mc-PBAs with identical Mn,ring values (32k) were also examined (Fig. 4b), revealing an increase in trapping with increasing Nring (i.e., from 57.3 to 84.6%). Subsequently, to assess the influence of Mn,ring, the trapped fractions of mc-PBAs with comparable Nring (8–12) and different Mn,ring values (10k, 19k, and 32k) were examined (Fig. 4c). Again, the trapped fraction increases with increasing Mn,ring, demonstrating that macro-rotaxane formation becomes more efficient with increasing Nring and Mn,ring values.
Our previous work demonstrated 100% trapping when macro-rotaxane formation was performed via end-cross-linking of linear PDMS in the presence of mc-PDMS with Nring and Mn,ring values of 6 and 26k, respectively. In contrast, in the present work, the maximum trapped fraction for mc-PBA remained around 85% even at high Nring and Mn,ring values. This could be attributed to several factors. First, the longer side chains of PBA compared with those of PDMS may have limited the network penetration in mc-PBA. Indeed, the entanglement molecular weight (Me) of PDMS37 (Me = 10k) is lower than that of PBA38 (Me = 32k). Second, photoradical polymerization inherently produces networks with structural heterogeneity, such as dangling chains, which can partially form pseudomacro-rotaxanes rather than true macro-rotaxanes. These factors reasonably account for the fact that the trapped fractions of the mc-PBA-blended PBA networks did not reach 100%.
Subsequently, swelling experiments were performed on the network samples after Soxhlet extraction (Table S1). The 60 wt% 5c-, 8c-, and 13c-PBA-blended PBA network samples exhibited approximately threefold Mc compared to the pristine PBA network. This result indicates that the mc-PBA interferes with the network formation process, leading to a decrease in the effective cross-link density and increased structural heterogeneity in the PBA network. These findings further support the observation that the trapped fraction did not reach 100%. On the other hand, a slight decrease in the Mc was observed with increasing Nring. This trend suggests that as the Nring increases, a greater number of network chains are threaded through the cyclic units, and the mc-PBA may partially act as additional cross-linking points.
Mechanical properties
Having established the successful formation of a macro-rotaxane network, attention was directed toward its damping performance to demonstrate its unique application potential. In various fields, such as automotive and construction engineering, vibration suppression using damping materials is essential for preventing fatigue failure and mitigating environmental noise. Liu et al.39 recently demonstrated that the incorporation of a linear PBA into a PBA network significantly enhanced its damping properties, giving a tunable effective frequency range. In this system, the added linear polymer exhibited reptation behavior in the high-elastic region of the polymer network, leading to a substantial increase in energy dissipation. However, such linear additives could potentially leach out over time, compromising long-term stability. It was therefore envisaged that mc-PBA could yield a sustained damping performance due to topological trapping of the cyclic units within the macro-rotaxane architecture, effectively preventing leaching while maintaining the desired damping enhancement.Thus, the effect of mc-PBA addition on the damping properties was investigated by dynamic viscoelastic measurements of PBA networks containing 20, 40, and 60 wt% 5c-PBA32k using a rotational rheometer with a parallel-plate geometry. Measurements were performed in the linear viscoelastic region (γ = 0.1%) (Fig. S36) and master curves of the storage modulus (G′), loss modulus (G″), and loss factor (tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ) were created by time–temperature superposition using a reference temperature of 25 °C (Fig. 5). The tan
δ) were created by time–temperature superposition using a reference temperature of 25 °C (Fig. 5). The tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ value is a key parameter reflecting energy dissipation in the material. Focusing on the pristine PBA network, a distinct peak in tan
δ value is a key parameter reflecting energy dissipation in the material. Focusing on the pristine PBA network, a distinct peak in tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ associated with the glass transition was observed in the high-frequency region, whereas a rubbery plateau region originating from the cross-linked structure was evident in the low-frequency region. In the low-frequency region (ω → 0), G′ decreased with increasing mc-PBA content, while tan
δ associated with the glass transition was observed in the high-frequency region, whereas a rubbery plateau region originating from the cross-linked structure was evident in the low-frequency region. In the low-frequency region (ω → 0), G′ decreased with increasing mc-PBA content, while tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ increased. This decrease in G′ was attributed to a decrease in PBA cross-link density due to the interference of mc-PBA with network formation and dilution of the PBA network fraction by the added mc-PBA. In contrast, the increase in tan
δ increased. This decrease in G′ was attributed to a decrease in PBA cross-link density due to the interference of mc-PBA with network formation and dilution of the PBA network fraction by the added mc-PBA. In contrast, the increase in tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ was ascribed to mc-PBA chain relaxation, which enhances energy dissipation within the network. These results clearly demonstrate that mc-PBA incorporation improves the damping properties of the PBA network, irrespective of macro-rotaxane formation. The increase in tan
δ was ascribed to mc-PBA chain relaxation, which enhances energy dissipation within the network. These results clearly demonstrate that mc-PBA incorporation improves the damping properties of the PBA network, irrespective of macro-rotaxane formation. The increase in tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ upon the addition of this polymer is consistent with the results reported by Liu et al., indicating that linear and cyclic polymers exhibit similar energy dissipation effects.
δ upon the addition of this polymer is consistent with the results reported by Liu et al., indicating that linear and cyclic polymers exhibit similar energy dissipation effects.
Subsequently, to clarify the influence of Nring on the damping properties, PBA networks containing a series of mc-PBAs (60 wt%) with identical Mn,ring values (32k) were investigated (Fig. 6a). It was found that tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ decreased as Nring increased from 5 to 13, potentially due to the progressive increase in mc-PBA relaxation time with increasing Nring. To support this interpretation, viscoelastic measurements were performed on neat mc-PBA, revealing that the crossover point of G′ and G″ in the terminal flow region shifted toward lower frequencies. This shift confirms that the relaxation time of mc-PBA increases with higher Nring values (Fig. 6b). The reduction in tan
δ decreased as Nring increased from 5 to 13, potentially due to the progressive increase in mc-PBA relaxation time with increasing Nring. To support this interpretation, viscoelastic measurements were performed on neat mc-PBA, revealing that the crossover point of G′ and G″ in the terminal flow region shifted toward lower frequencies. This shift confirms that the relaxation time of mc-PBA increases with higher Nring values (Fig. 6b). The reduction in tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ could also be related to the increased fraction of mc-PBA trapped in the network at higher Nring values (%Trapped = 57.3–84.6%). Macro-rotaxane-trapped mc-PBA is expected to exhibit more strongly restricted molecular motion than its untrapped counterparts. Consequently, the fluid-like behavior of mc-PBA is weakened, leading to suppressed energy dissipation in the network and a corresponding decrease in tan
δ could also be related to the increased fraction of mc-PBA trapped in the network at higher Nring values (%Trapped = 57.3–84.6%). Macro-rotaxane-trapped mc-PBA is expected to exhibit more strongly restricted molecular motion than its untrapped counterparts. Consequently, the fluid-like behavior of mc-PBA is weakened, leading to suppressed energy dissipation in the network and a corresponding decrease in tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ.
δ.
Finally, the potential of mc-PBA as a non-leaching additive was evaluated by examining changes in tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ before and after solvent washing to remove any untrapped mc-PBA. Dynamic viscoelastic measurements were conducted on networks blended with 60 wt% 13c-PBA32k, which exhibited the highest trapping performance, and with 19g-PBA15k, a control sample without cyclic structures. Since the samples used for Soxhlet extraction were damaged during the process, alternative samples immersed in toluene for 2 d were employed. The calculated trapped fractions were 89.8 and <0.1% for the 13c-PBA32k and 19g-PBA15k-blended PBA networks, respectively. As expected, the 13c-PBA32k-blended PBA network exhibited similar tan
δ before and after solvent washing to remove any untrapped mc-PBA. Dynamic viscoelastic measurements were conducted on networks blended with 60 wt% 13c-PBA32k, which exhibited the highest trapping performance, and with 19g-PBA15k, a control sample without cyclic structures. Since the samples used for Soxhlet extraction were damaged during the process, alternative samples immersed in toluene for 2 d were employed. The calculated trapped fractions were 89.8 and <0.1% for the 13c-PBA32k and 19g-PBA15k-blended PBA networks, respectively. As expected, the 13c-PBA32k-blended PBA network exhibited similar tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ profiles before and after solvent washing, whereas the 19g-PBA15k-blended PBA network showed a marked decrease in tan
δ profiles before and after solvent washing, whereas the 19g-PBA15k-blended PBA network showed a marked decrease in tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ after washing (Fig. S37). These findings clearly demonstrate that mc-PBA acts as an effective non-leaching additive in macro-rotaxane systems.
δ after washing (Fig. S37). These findings clearly demonstrate that mc-PBA acts as an effective non-leaching additive in macro-rotaxane systems.
In addition, indentation tests were performed to investigate the effect of mc-PBA on Young's modulus (E) (Fig. S38 and Table S2). The E value was calculated from the initial loading region based on Hertzian contact theory. The results revealed that the E value decreased upon the addition of mc-PBA, which is attributed to a reduction in the effective cross-link density. Furthermore, the E value was found to depend on both the Nring and the Mn,ring of the added mc-PBA, indicating that the mechanical properties can be tuned through the molecular parameters of mc-PBA and its fraction.
Conclusions
Macro-rotaxanes were formed via the photoradical polymerization of BA and a cross-linker in the presence of mc-PBA. Unlike conventional approaches based on the cross-linking of end-functionalized linear polymers, this method allowed the monomer itself to be used as the solvent and did not require time-consuming procedures such as solvent casting, thereby enabling the rapid formation of polymer networks incorporating macro-rotaxanes. Notably, increasing both Nring and Mn,ring led to a maximum macro-rotaxane formation efficiency of 84.6%. The damping properties of the mc-PBA-blended PBA networks were subsequently evaluated to assess the applicability of mc-PBA as a functional material. The results revealed that tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ increased owing to the enhanced relaxation behavior of mc-PBA compared with that of pristine PBA network. Furthermore, the enhanced tan
δ increased owing to the enhanced relaxation behavior of mc-PBA compared with that of pristine PBA network. Furthermore, the enhanced tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ was retained even after washing with toluene, demonstrating the potential of mc-PBA as a non-leaching additive. Overall, this synthetic methodology and systematic evaluation provide a foundation for extending the present approach to a wide range of radically polymerizable monomers for macro-rotaxane formation and functional material development.
δ was retained even after washing with toluene, demonstrating the potential of mc-PBA as a non-leaching additive. Overall, this synthetic methodology and systematic evaluation provide a foundation for extending the present approach to a wide range of radically polymerizable monomers for macro-rotaxane formation and functional material development.
Author contributions
Kotaro Ibe: investigation, writing – original draft, methodology, visualization, and data curation; Yamato Ebii: investigation and methodology; Minami Ebe: investigation and methodology; Kazushige Suzuki: investigation; Takayuki Kurokawa: investigation; Takuya Yamamoto: investigation and supervision; Feng Li: investigation and supervision; Takuya Isono: investigation, supervision, conceptualization, writing – review and editing, and funding acquisition; Toshifumi Satoh: supervision, conceptualization, writing – review and editing, and funding acquisition.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d6py00144k.Acknowledgements
This work was financially supported by JST CREST (JPMJCR25S2 and JPMJCR19T4), the MEXT Grant-in-Aid for Challenging Exploratory Research (19K22209 and 24K21788), the Eno Scientific Foundation, the Tokyo Ohka Foundation for the Promotion of Science and Technology, the Sumitomo Foundation, the Photoexcitonix Project at Hokkaido University, the Co-Creation Core for Soft Materials Aspiring Research & Translation (C3-SMART), and the JSPS Program for Forming Japan's Peak Research Universities, namely the “Soft Materials Platform Aspiring the Unique Properties from Natural Polymers” (J-PEAKS; JPJS00420230001).References
- J. Li, Y. Li, Y. Guo, J. Xu, J. Lv, Y. Li, H. Liu, S. Wang and D. Zhu, Chem. – Asian J., 2008, 3, 2091–2096 CrossRef CAS PubMed.
- G. De Bo, M. A. Y. Gall, M. O. Kitching, S. Kuschel, D. A. Leigh, D. J. Tetlow and J. W. Ward, J. Am. Chem. Soc., 2017, 139, 10875–10879 CrossRef CAS PubMed.
- J. J. Yu, L. Y. Zhao, Z. T. Shi, Q. Zhang, G. London, W. J. Liang, C. Gao, M. M. Li, X. M. Cao, H. Tian, B. L. Feringa and D. H. Qu, J. Org. Chem., 2019, 84, 5790–5802 CrossRef CAS PubMed.
- L. Chen, R. Nixon and G. De Bo, Nature, 2024, 628, 320–325 CrossRef CAS PubMed.
- R. Barat, T. Legigan, I. Tranoy-Opalinski, B. Renoux, E. Péraudeau, J. Clarhaut, P. Poinot, A. E. Fernandes, V. Aucagne, D. A. Leigh and S. Papot, Chem. Sci., 2015, 6, 2608–2613 RSC.
- G. Yu, Z. Yang, X. Fu, B. C. Yung, J. Yang, Z. Mao, L. Shao, B. Hua, Y. Liu, F. Zhang, Q. Fan, S. Wang, O. Jacobson, A. Jin, C. Gao, X. Tang, F. Huang and X. Chen, Nat. Commun., 2018, 9, 766 CrossRef PubMed.
- Y. Okumura and K. Ito, Adv. Mater., 2001, 13, 485–487 CrossRef CAS.
- C. Liu, N. Morimoto, L. Jiang, S. Kawahara, T. Noritomi, H. Yokoyama, K. Mayumi and K. Ito, Science, 2021, 372, 1078–1081 CrossRef CAS PubMed.
- K. Kato, M. Taniguchi and K. Ito, Macromolecules, 2023, 56, 1810–1817 CrossRef CAS.
- P. R. Ashton, P. T. Glink, J. F. Stoddart, P. A. Tasker, A. J. P. White and D. J. Williams, Chem. – Eur. J., 1996, 2, 729–736 CrossRef CAS.
- C. Zhang, S. Li, J. Zhang, K. Zhu, N. Li and F. Huang, Org. Lett., 2007, 9, 5553–5556 CrossRef CAS PubMed.
- S. Y. Hsueh, K. W. Cheng, C. C. Lai and S. H. Chiu, Angew. Chem., Int. Ed., 2008, 47, 4436–4439 CrossRef CAS PubMed.
- A. Hashidzume, H. Yamaguchi and A. Harada, Eur. J. Org. Chem., 2019, 3344–3357 CrossRef CAS.
- H. Ogino, J. Am. Chem. Soc., 1981, 103, 1303–1304 CrossRef CAS.
- R. S. Wylie and D. H. Macartney, J. Am. Chem. Soc., 1992, 114, 3136–3138 CrossRef CAS.
- A. N. Basuray, H. P. Jacquot de Rouville, K. J. Hartlieb, T. Kikuchi, N. L. Strutt, C. J. Bruns, M. W. Ambrogio, A. J. Avestro, S. T. Schneebeli, A. C. Fahrenbach and J. F. Stoddart, Angew. Chem., Int. Ed., 2012, 51, 11872–11877 CrossRef CAS PubMed.
- D. B. Amabilino, P. R. Ashton, V. Balzani, C. L. Brown, A. Credi, J. M. J. Fréchet, J. W. Leon, F. M. Raymo, N. Spencer, J. F. Stoddart and M. Venturi, J. Am. Chem. Soc., 1996, 118, 12012–12020 CrossRef CAS.
- Q. S. Zong, C. Zhang and C. F. Chen, Org. Lett., 2006, 8, 1859–1862 CrossRef CAS PubMed.
- F. Huang, D. S. Nagvekar, C. Slebodnick and H. W. Gibson, J. Am. Chem. Soc., 2005, 127, 484–485 CrossRef CAS PubMed.
- D. Parisi, J. Ahn, T. Chang, D. Vlassopoulos and M. Rubinstein, Macromolecules, 2020, 53, 1685–1693 CrossRef CAS PubMed.
- M. Kapnistos, M. Lang, D. Vlassopoulos, W. Pyckhout-Hintzen, D. Richter, D. Cho, T. Chang and M. Rubinstein, Nat. Mater., 2008, 7, 997–1002 CrossRef CAS PubMed.
- G. S. Grest, T. Ge, S. J. Plimpton, M. Rubinstein and T. C. O'Connor, ACS Polym. Au, 2023, 3, 209–216 CrossRef CAS PubMed.
- M. Kruteva, M. Monkenbusch, J. Allgaier, W. Pyckhout-Hintzen, L. Porcar and D. Richter, Macromolecules, 2023, 56, 4835–4844 CrossRef CAS.
- S. J. Clarson, J. E. Mark and J. A. Semlyen, Polym. Commun., 1986, 27, 244–245 CAS.
- T. S. Stukenbroeker, D. Solis-Ibarra and R. M. Waymouth, Macromolecules, 2014, 47, 8224–8230 CrossRef CAS.
- L. Garrido, J. E. Mark, S. J. Clarson and J. A. Semlyen, Polym. Commun., 1985, 26, 53–55 CAS.
- L. C. Debolt and J. E. Mark, Macromolecules, 1987, 20, 2369–2374 CrossRef CAS.
- B. R. Wood, S. J. Joyce, G. Scrivens, J. A. Semlyen, P. Hodge and R. O'Dell, Polymer, 1993, 34, 3059–3063 CrossRef CAS.
- W. Huang, H. L. Frisch, Y. Hua and J. A. Semlyen, J. Polym. Sci., Part A:Polym. Chem., 1990, 28, 1807–1812 CrossRef CAS.
- T. J. Fyvie, H. L. Frisch, J. A. Semlyen, S. J. Clarson and J. E. Mark, J. Polym. Sci., Part A:Polym. Chem., 1987, 25, 2503–2509 CrossRef CAS.
- T. Isono, T. Sasamori, K. Honda, Y. Mato, T. Yamamoto, K. Tajima and T. Satoh, Macromolecules, 2018, 51, 3855–3864 CrossRef CAS.
- M. Ebe, A. Soga, K. Fujiwara, B. J. Ree, H. Marubayashi, K. Hagita, A. Imasaki, M. Baba, T. Yamamoto, K. Tajima, T. Deguchi, H. Jinnai, T. Isono and T. Satoh, Angew. Chem., Int. Ed., 2023, 62, e202304493 CrossRef CAS PubMed.
- Y. Ebii, Y. Mato, F. Li, K. Tajima, T. Yamamoto, T. Isono and T. Satoh, Polym. Chem., 2023, 14, 3099–3109 RSC.
- K. Hagita and T. Murashima, Polymer, 2021, 223, 123705 CrossRef CAS.
- H. Marubayashi, M. Ebe, A. Imasaki, K. Fujiwara, N. Mashita, K. Hagita, T. Murashima, S. Mori, T. Isono, T. Satoh and H. Jinnai, Polymer, 2024, 292, 126607 CrossRef CAS.
- N. D. Ogbonna, M. Dearman, C.-T. Cho, B. Bharti, A. J. Peters and J. Lawrence, JACS Au, 2022, 2, 898–905 CrossRef CAS PubMed.
- L. J. Fetters, D. J. Lohse, D. Richter, T. A. Witten and A. Zirkel, Macromolecules, 1994, 27, 4639–4647 CrossRef CAS.
- N. Jullian, F. Leonardi, B. Grassl, J. Peyrelasse and C. Derail, Appl. Rheol., 2010, 20, 33685 Search PubMed.
- J. Huang, Y. Xu, S. Qi, J. Zhou, W. Shi, T. Zhao and M. Liu, Nat. Commun., 2021, 12, 3610 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |