Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Organo-catalyzed deamination of polystyrene sulfonamide for diverse post-polymerization modification of styrenic polymers
Tulaja
Shrestha
a,
Dan My
Nguyen
 a,
Harrison
Goehrig
a,
Vidhika S.
Damani
b,
Caitlin M.
Quinn
a and
Laure V.
Kayser
a,
Harrison
Goehrig
a,
Vidhika S.
Damani
b,
Caitlin M.
Quinn
a and
Laure V.
Kayser
 *ab
*ab
aDepartment of Chemistry and Biochemistry, University of Delaware, Newark, DE 19711, USA. E-mail: lkayser@udel.edu
bDepartment of Materials Science and Engineering, University of Delaware, Newark, DE 19711, USA
First published on 1st April 2026
Abstract
Post-polymerization modification allows for the incorporation of functional groups that would otherwise be incompatible with polymerization conditions, enhancing synthetic efficiency and facilitating the creation of complex polymer architectures for specialized applications, such as biomedical devices, electronics, and advanced coatings. Herein, we report a method inspired by late-stage functionalization of small molecules for the post-polymerization modification of aromatic polymers, specifically polystyrene (PS), under mild reaction conditions. First, PS was converted to polystyrene sulfonamide (PSSNH2) with an 85% yield using established procedures. PSSNH2 was subsequently transformed into a reactive sulfinate by deamination using an N-heterocyclic carbene (NHC) catalyst and benzaldehyde. The catalytic process was optimized by varying catalysts, solvents, bases, temperatures, and reaction times. The highest degree of deamination was 88% with a bicyclic NHC and K2CO3 base in DMSO for 18 hours at 120 °C. The reactive sulfinate was then treated with various functional reagents, resulting in a library of aromatic polymers with different substituents with high degrees of functionalization ranging from 74% to 98%. Similarly, we modified expanded PS waste with trifluorobutyl iodide with a degree of functionalization of 72%, highlighting a new avenue for plastic upgrading. This approach could be used to rapidly generate functionalized polymers from PS with potential applications, including antibacterial properties and flame retardancy.
Introduction
Post-polymerization modification (PPM) is crucial in polymer chemistry because it allows for the precise tailoring of polymer properties after the initial polymerization process.1–4 This flexibility is essential for incorporating functional groups incompatible with the polymerization conditions, thereby expanding the range of possible polymer functionalities. It also enhances the efficiency of synthetic processes, enabling the introduction of desired functionalities.5 PPMs employ efficient organic synthetic strategies such as transition metal catalysis,6,7 click chemistry,8–10 photoredox catalysts,3 nucleophilic aromatic substitutions,11–15 and radical reactions.16 These innovations have enabled the creation of complex polymer architectures and high-value materials with specialized applications, such as biomedical devices,1,2 electronics,3 and advanced coatings.4 By enabling sequential modifications, PPM also allows for the stepwise introduction of multiple functional groups, leading to polymers with specific and enhanced properties.Over the past two decades, PPM has also matured into a methodology for functionalizing specialty polymers, including bio-derived materials,17 polypeptides,18 and synthetic scaffolds with thiol,19 ester, or halide handles.20 Such systems have enabled tunable properties in biomaterials and therapeutics, high-throughput functional libraries via amidation or thiol–ene reactions, and solid-state modifications of engineering thermoplastics without compromising processability.21–23 These developments illustrate the broad reach of PPM across diverse classes of polymers. PPM has also opened new avenues for repurposing commodity plastics.24–26 For example, Golder and co-workers demonstrated the functionalization of polybutadiene to its thermal and surface-wetting properties.27 Vlachos and co-workers reported a plasma-based oxidation approach for polyethylene upcycling.28 Watson and co-workers fluorinated poly(acrylic acid) in water under organophotoredox conditions using vitamin B2 as a green catalyst.29
Polystyrene (PS) is produced on a global scale exceeding 20 million metric tons annually, therefore representing an attractive target for PPM. Numerous strategies have been pursued to functionalize PS. Early approaches, such as Friedel–Crafts acylation13 and benzylic azidation with hypervalent iodine reagents,30 achieved limited conversions and often compromised molecular weight. More recently, Leibfarth and coworkers reported a photocatalytic fluoroalkylation of PS, featuring in situ generation of perfluoroalkyl species, followed by Ru(bpy)Cl2-catalyzed functionalization that afforded up to 72% conversion while preserving the polymer's molecular weight.31 Additionally, a study conducted by Xue et al. demonstrated a mild arylation strategy with a high degree of functionalization, approaching nearly 100% conversion.5 Chan and co-workers reported a two-step amination strategy to access antibacterial PS coatings,32 while Liu et al. introduced phosphorylation to tune surface and thermal properties, introducing flame-retardant properties in polystyrene.33 We previously developed a sulfonation method which preserved the integrity of the parent PS. The resulting polystyrene sulfonate (PSS) was subsequently complexed with a conjugated polymer and used in organic electrochemical transistors and silicon-based hybrid solar cells.34 These examples highlight the progress toward milder, more versatile transformations. While most of these methods have limited functional group scope, a recent study by Wang and co-workers reported the bromination of commercial-grade polystyrene under mild reaction conditions using AuCl3 as a catalyst, thereby preserving the polymeric backbone. This brominated polymer served as a lynchpin precursor that could be diversified into a range of functionalized derivatives. Such strategies underscore the value of lynchpin intermediates where a single reactive functionality enables broad diversification under mild conditions.35 Towards this goal, most studies rely on transition-metal catalysts which may be complex to remove from the final polymer motivating the development of complementary, metal-free methods.
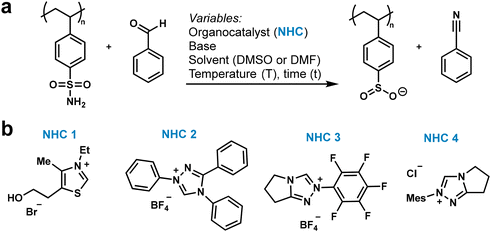
Here, we present an organocatalytic PPM strategy that installs polystyrene sulfinate (PSSO2−) as a lynchpin via N-heterocyclic carbene (NHC)-catalyzed deamination of PS sulfonamides (Scheme 1). Adapted from a small molecule synthesis originally developed by Fier et al. for small molecules, this approach generates sulfinates under mild conditions without the use of transition metals.36 The sulfinate intermediate engages with diverse electrophiles and participates in cross-coupling reactions, giving rise to alkyl and aryl sulfones, sulfonamides, and biaryls. By consolidating multiple functionalization pathways into a single precursor, this lynchpin-based, organocatalytic strategy expands the synthetic toolbox for PS modification while complementing existing metal-catalyzed approaches. The breadth of transformations accessible from this intermediate could enable the preparation of polymers for advanced applications in antifouling coatings,37,38 separation membranes,39 biosensors,40 and flame-retardant materials.41
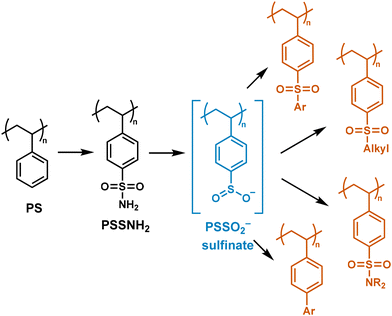 | ||
| Scheme 1 Post-polymerization modification (PPM) of polystyrene by installation of a reactive sulfinate group to access a diverse library of polymers. | ||
Results and discussion
To optimize the PPM, we focused on a model PS made by anionic polymerization with Mn = 19 kg mol−1 and low dispersity (Đ = 1.42), as measured by gel permeation chromatography (GPC) (Fig. S1), to ensure good solubility. The degree of polymerization (DP) was calculated to be 183. The PS was transformed to polystyrene sulfonamide (PSSNH2) using a commonly used sequential chlorosulfonation and amidation reactions (Scheme 1).42 PSSNH2 thus synthesized had a degree of sulfonation of >95% and degree of amidation of 85–92%, as determined by X-ray photoelectron spectroscopy (XPS). XPS and IR spectroscopy (Fig. S2) were used for characterization because PSSNH2 was insoluble in common solvents, such as toluene, trichlorobenzene, DCM, chloroform, DMF, DMSO, DMAc, pyridine, methanol, and ethyl acetate. The low solubility of the polymers prevented us from obtaining GPC data. As such, we cannot confirm that the polymer main chain did not undergo chain scission (reduction in molecular weight) or crosslinking (increase in molecular weight). In contrast to the parent polystyrene, the functionalized polymers exhibited very limited solubility in common organic solvents and only swelled in highly polar aprotic solvents such as DMSO and DMF. This behavior is likely influenced by the high density of polar functional groups introduced during post-polymerization modification, which can promote strong interchain interactions and aggregation. In addition, the incorporation of functional groups may increase chain rigidity and cohesive energy density, further suppressing molecular dissolution. While chemical crosslinking cannot be ruled out due to the absence of GPC data, the consistent observation of swelling rather than true dissolution suggests that physical association and morphological effects play a major role. XPS was chosen over elemental analysis for quantitative assessment of the degree of functionalization because it also provided a qualitative verification of the purity of the functionalized polymer (i.e., difference in binding energy in reactant versus functionalized polymer). Following the successful synthesis and characterization of PSSNH2, we proceeded to optimize the NHC-catalyzed deamination of PSSNH2 (Fig. 1a and Table 1) to form polystyrene sulfinate (PSSO2−). This reaction proceeds through the attack of the nitrogen of the sulfonamide with the aldehyde to form an N-sulfonylimine intermediate, ultimately liberating a nitrile through decomposition as the stoichiometric byproduct along with the sulfinate product.36 Following the general reaction shown in Fig. 1a of PSSNH2 with benzaldehyde, we varied the NHC catalyst (NHC 1 to NHC 4, Fig. 1b), solvent, base, temperature, and time to find the reaction conditions with the highest conversion of sulfonamide to sulfinate. The extent of deamination was calculated by XPS by measuring the nitrogen![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) sulfur ratio in the polymers purified using Soxhlet washes in chloroform, methanol, and water for 18 hours each (Table 1). The purity of the PSSNH2 for the optimized reaction conditions of the deamination step was verified qualitatively using solid-state 13C and 15N NMR to ensure the successful purification steps. The presence of sharp peaks would indicate the presence of a small molecule and, hence, residual impurities. We did not observe such impurity peaks in the 13C-NMR (Fig. S3) nor the 15N-NMR (Fig. S4).
sulfur ratio in the polymers purified using Soxhlet washes in chloroform, methanol, and water for 18 hours each (Table 1). The purity of the PSSNH2 for the optimized reaction conditions of the deamination step was verified qualitatively using solid-state 13C and 15N NMR to ensure the successful purification steps. The presence of sharp peaks would indicate the presence of a small molecule and, hence, residual impurities. We did not observe such impurity peaks in the 13C-NMR (Fig. S3) nor the 15N-NMR (Fig. S4).
| Entry | Solvent | NHC | Base | Time (h) | Temperature (°C) | % Deaminationa | Yield (%) |
|---|---|---|---|---|---|---|---|
| a Calculated from XPS. | |||||||
| 1 | DMSO | NHC 1 | K2CO3 | 24 | 100 | 5 | 72 |
| 2 | DMSO | NHC 2 | K2CO3 | 24 | 100 | 39 | 79 |
| 3 | DMSO | NHC 3 | K2CO3 | 24 | 100 | 4 | 74 |
| 4 | DMSO | NHC 4 | K2CO3 | 24 | 100 | 86 | 85 |
| 5 | DMF | NHC 4 | K2CO3 | 24 | 100 | 47 | 81 |
| 6 | DMSO | NHC 4 | Cs 2 CO 3 | 24 | 100 | 30 | 75 |
| 7 | DMSO | NHC 4 | NEt 3 | 24 | 100 | 38 | 82 |
| 8 | DMSO | NHC 4 | K2CO3 | 12 | 100 | 55 | 93 |
| 9 | DMSO | NHC 4 | K2CO3 | 18 | 100 | 67 | 91 |
| 10 | DMSO | NHC 4 | K2CO3 | 48 | 100 | 88 | 83 |
| 11 | DMSO | NHC 4 | K2CO3 | 24 | 60 | 33 | 74 |
| 12 | DMSO | NHC 4 | K2CO3 | 24 | 80 | 86 | 78 |
| 13 | DMSO | NHC 4 | K2CO3 | 24 | 120 | 88 | 79 |
| 14 | DMSO | NHC 4 | K2CO3 | 12 | 120 | 81 | 77 |
| 15 | DMSO | NHC 4 | K2CO3 | 18 | 120 | 86 | 85 |
Four NHCs were chosen for this optimization study (entries 1–4) based on their documented performance in related transformations (Fig. 1a). NHC 1 was chosen due to its reported effectiveness in facilitating Stetter-type reactions where the NHC enables aldimine umpolung, making the aldimine nucleophilic. NHC 1 was found to work better with substrates involving arylated substrates.43 NHCs 2 and 3 were selected from the work of Sun et al., where they achieved high yields (97% and 90%, respectively) in the reaction with tosylated aldimines to generate nitriles by desulfonating the imine. This mechanistic pathway is analogous to that proposed in our study.44 Although NHC 4 gave a comparatively lower yield (84%) in this article, it gave the highest yield in a separate report by Fier et al. on the deamination of sulfonamides.36 We therefore hypothesized that these four NHCs could lead to effective deamination of PSSNH2. NHC 1 (entry 1), NHC 2 (entry 2), NHC 3 (entry 3), and NHC 4 (entry 4) exhibited degrees of deamination of 5%, 39%, 4%, and 86% respectively, as determined using XPS. Similar to past reports with small molecules,36 the best NHC for the deamination was found to be NHC 4 (entry 4) with a significantly higher degree of deamination (86%) when the reaction was run at 100 °C for 24 hours in DMSO as the solvent and with K2CO3 as the base.
Next, we investigated the role of the solvent. Due to the poor solubility of PSSNH2, we could only use dimethyl sulfoxide (DMSO) and dimethylformamide (DMF) which were the only two solvents capable of swelling this parent polymer. Running the reaction in DMF (entry 5), however, led to a significantly lower degree of deamination (47%). We therefore kept DMSO as our solvent for the remainder of the study. Given that deprotonation of the NHC precatalyst is the most important step to initiate the catalytic cycle, we tried three mild bases for this optimization study—K2CO3 (entry 4), Cs2CO3 (entry 6), and NEt3 (entry 7) affording degrees of deamination of 86%, 30%, and 38%, respectively. The reaction was therefore performed in DMSO with K2CO3 as the base at 100 °C. At 12 hours (entry 8), the reaction yielded a 55% degree of deamination, which increased to 67% at 18 hours (entry 9), 86% at 24 hours (entry 4), and ultimately 88% at 48 hours (entry 10). Increasing the reaction time from 24 hours to 48 hours only increased the degree of deamination from 86% to 88%. This incremental increase was not deemed sufficient to justify an additional 24 hours of reaction time at the given temperature. We subsequently examined the effects of the temperature of the reaction on the degree of deamination. As indicated in Table 1, the degree of deamination increased proportionally with rising temperature. At 60 °C (entry 11), the degree of deamination was only 33%. However, it increased significantly to 85% at 80 °C (entry 12) and reached 88% at 120 °C (entry 13), slightly surpassing the 86% conversion achieved at 100 °C (entry 4) under the same conditions. This observation prompted us to conduct a second time study at 120 °C. As shown in entries 14 and 15 (Table 1), the degree of deamination was 81% after 12 hours, and reached 86% after 18 hours. Given that the degree of deamination only increased by 2% when the reaction time was extended from 18 hours to 24 hours at 120 °C, we concluded that the optimal reaction conditions were 120 °C for 18 hours with DMSO as the solvent, NHC 4 as the catalyst and K2CO3 as the base. The sulfinate functionalized PS (86%) was purified using Soxhlet washes in chloroform, methanol, and water.
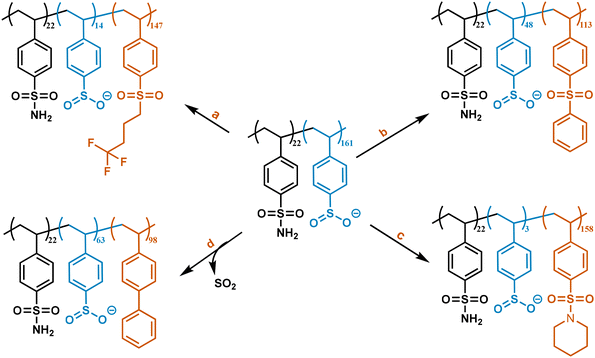
Once the deamination reaction was optimized, we proceeded to investigate the scope of potential functional groups that can be attached onto the phenyl. These functional groups included alkyl sulfones, aryl sulfones, sulfonamides, and aryls (obtained via cross-coupling) (Fig. 2). Due to the poor solubility of the resulting polymers, XPS was used throughout to quantify the degree of functionalization (Fig. S5). The conversion calculated using XPS was supplemented with solid-state 1H NMR (DFNMR) and thermogravimetric analysis (DFTGA) (Table 2). The DFNMR was generally slightly lower, but both the DFNMR and DFTGA were within the same range as the ones obtained by XPS. In the paragraphs below, we describe each functional group attachment separately.
| Entry | Final functional group | Reactants | DFXPS![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) a (%) a (%) |
DFNMR![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) b (%) b (%) |
DFTGA![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) c (%) c (%) |
T
d,10%![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) d (°C) d (°C) |
Yield (%) |
|---|---|---|---|---|---|---|---|
| a Degree of functionalization calculated from XPS. b Degree of functionalization calculated from 1H solid state NMR. c Degree of functionalization calculated from mass loss via TGA. d Decomposition temperature calculated using TGA. | |||||||
| 1 | Styrene trifluorobutyl sulfone | a: Trifluorobutyl iodide | 91 | 85 | 93 | 365 | 85 |
| 2 | Styrene phenyl sulfone | b: Phenyl iodide | 70 | 67 | 75 | 328 | 81 |
| 3 | Styrene sulfonyl piperidine | c: Piperidine | 98 | 92 | 95 | 280 | 82 |
| 4 | Phenyl styrene | d: Phenyl triflate | 74 | 70 | N/A | 360 | 75 |
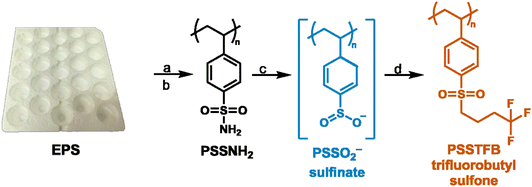
To demonstrate reactivity of the sulfinate with electrophilic alkyl iodides, we reacted purified PSSO2− with trifluorobutyl iodide to synthesize polystyrene trifluorobutyl sulfone. A solution of trifluorobutyl iodide in DMSO was added to the reaction flask with PSSO2− and the reaction was run for 24 hours at 25 °C. The pale-yellow powder thus obtained was purified using Soxhlet wash in chloroform, THF, and water for 24 hours each. The degree of functionalization was calculated to be 91% using the sulfur-to-fluorine ratio (Table 2, entry 1), which was verified by solid state 1H NMR where the degree of conversion was calculated to be 85% (Fig. S6). The purity was determined using solid-state 13C NMR (Fig. S7) and 19F NMR (Fig. S8). The purity of the polymers was verified using solid state 13C NMR by overlaying the NMR spectrum of the crude samples and that of the purified functionalized polymers. These experiments showed that sharp signals corresponding to solvent, excess reagents and/or by-products are not present in the polymers purified by Soxhlet washes, and only broad signals characteristic of polymers remain. Given these broad overlapping peaks, however, we cannot confirm the absence of impurities but the 19F NMR is pure. We selected the trifluorobutyl group to illustrate that long alkyl chains can be added to the sulfinate and also that it can provide an approach to fluorinate polymers. Additionally, the presence of the fluorine atom facilitated quantification of the degree of conversion by XPS. With efficient functionalization conditions on the model PS, we then moved on to post-consumer expanded polystyrene (EPS) packaging (Fig. 3). The degree of sulfonation and amidation on this plastic waste was calculated to be 91% and 89%, respectively. The polymer was then functionalized with trifluorobutyl iodide under the same reaction conditions as mentioned above to form a fluorinated polymer with the degree of alkylation of 72%.
The sulfinate is also reactive with aryl iodides, therefore PSSO2− was reacted with phenyl iodide in the presence of a third generation Buchwald precatalyst, Xantphos/Pd and Cs2CO3 as the base in DMSO for 24 hours at 120 °C.45 The final product was a pale yellow powder which was purified by Soxhlet washes. The degree of functionalization was calculated using the sulfur-to-carbon ratio, where the increase in carbon content compared to the XPS of PSSO2− indicated the presence of an additional phenyl group in the polymer. The degree of functionalization was determined to be 70% (Table 2, entry 2). The conversion calculated using XPS was verified using 1H NMR to be 67% (Fig. S9). Overall purity of the sample was assessed using solid-state 13C NMR (Fig. S10) showing the disappearance of sharp peaks after Soxhlet washing. Sulfinates can also react with amines in the presence of iodine to form sulfonamides by oxidative amination.46 Piperidine was chosen as the amine to show that cyclic amines can be used. For the synthesis of polystyrene sulfonyl piperidine (Table 2, entry 3), a solution of piperidine and iodine in water was reacted with PSSO2−, and the reaction was run for 24 hours at 25 °C. The pale yellow powder was purified by Soxhlet washes. The degree of functionalization, calculated using the sulfur-to-nitrogen ratio, was found to be 98%. This was cross-verified using 1H NMR to be 92% (Fig. S11), demonstrating that high degrees of amination are achievable. As before, we confirmed that small molecule impurities were washed off by solid-state 13C NMR (Fig. S12). Lastly, we demonstrated a C–C cross-coupling reaction between the sulfinate and an aryl triflate. We used phenyl triflate in the presence of a second-generation Buchwald precatalyst, XPhos-Pd-G2, in DMSO for 24 hours at 120 °C to form polyphenyl styrene, yielding a styrene polymer functionalized with phenyl group at the para position and sulfur dioxide as a byproduct.47 The decrease in the sulfur-to-carbon ratio in the XPS of the final polymer compared to that of PSSO2− suggested partial desulfonation and cross-coupling, with the degree of functionalization calculated to be 74%, as verified by 1H NMR giving a conversion of 70% (Fig. S13). Solid state 13C NMR (Fig. S14) showed that no sharp peaks were left in the sample post washing. The examples above highlight the potential of using sulfinates as lynchpins for the functionalization of PS. We expect that other alkyl and aryl iodides, amines, and aryl triflates could be used to access a variety of functional groups.
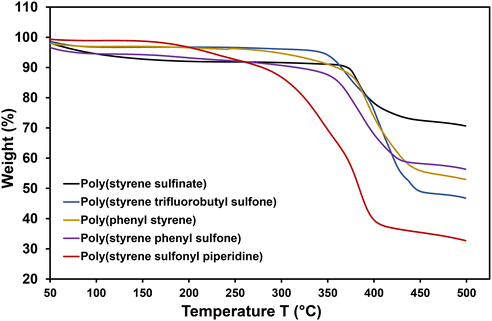
While the functionalized polymers were not soluble in any of the common and uncommon solvents, the polymers did swell up in DMSO, and hence we could hot-press the polymers into film-like structures, as shown in Fig. S15. For some materials, such as polystyrene sulfonamide, polystyrene phenyl sulfone, and polystyrene sulfonyl piperidine, it worked well, and we obtained free-standing films. In contrast, others, such as polystyrene trifluorobutyl sulfone and polyphenyl styrene, were too brittle even though they seemed moldable. We then assessed the thermal stability of the modified polymers using thermogravimetric analysis (TGA), as shown in Fig. 4. Poly(styrene sulfinate) (PSSO2−) exhibited a decomposition temperature of 372 °C. In comparison, the functionalized polymers showed lower degradation temperatures (Table 2). Poly(styrene trifluorobutyl sulfone) degraded at 365 °C (entry 1), poly(styrene phenyl sulfone) at 328 °C (entry 2), polystyrene sulfonyl piperidine at 280 °C (entry 3), and poly(phenyl styrene) at 360 °C (entry 4). The mass loss for entries 1–3 approximately corresponded to the mass attributed to the functionalized sulfone moieties, supplementing the degree of functionalization calculated using XPS. However, for poly(phenyl styrene) (entry 4), the degradation likely followed a different pathway, preventing direct verification of the degree of cross-coupling. These results demonstrate that the sulfinate functionality can serve as a versatile lynchpin for post-polymerization diversification. The distinct thermal stabilities of the resulting polymers highlight the ability to tune material properties through PPM.
Conclusions
We successfully developed a synthetic approach to modify polystyrene to a reactive polystyrene sulfinate by organocatalyzed deamination, using an N-heterocyclic carbene. This transformation efficiently converted polystyrene sulfonamide into a sulfinate intermediate with degrees of deamination over 85%. This reactive intermediate was then treated with a broad range of functional groups, including alkyl iodides, aryl iodides, amines, and aryl triflates, leading to degrees of functionalization between 74% and 98%. These polymers exhibited distinct chemical and thermal properties from the parent polystyrene, demonstrating the potential of the transformation in post-polymerization modification. This study highlights sulfinate intermediates as a versatile lynchpin to create a diverse library of functionalized styrenic polymers. The transformation was also successfully applied to post-consumer plastic waste, achieving a 72% alkylation with a trifluorobutyl group. Overall, this method proves to be a robust tool for post-polymerization modification, efficiently attaching new functional groups and yielding a range of functionalized polymers with potential applications in hydrophobic, antibacterial, or flame-retardant materials.Author contributions
T. S. and L. V. K. conceived the project idea and designed the experiments. T. S. and D. M. N synthesized the polystyrene and functionalized the polystyrene to poly(styrene sulfonamide) analyzed them. V. S. D. performed the GPC experiments and helped T. S. analyze the data. T. S. ran optimization studies for synthesis of poly(styrene sulfinate) with the help of H. G. T. S. conceived and designed the functionalization of poly(styrene sulfinate) and ran the XPS and analyzed the data. C. M. Q. ran the solid-state NMR and helped T. S. analyze the data. L. V. K. provided guidance and funding. T. S. and L. V. K. wrote the manuscript.Conflicts of interest
The authors declare no competing financial interest.Data availability
The data for this article is available freely in the Harvard Dataverse public repository at: https://doi.org/10.7910/DVN/OT2MAH. Data available: raw GPC of the model polystyrene, IR spectra, thermogravimetric analysis (TGA), NMR spectra files, and X-ray photoelectron spectroscopy of survey spectra and relevant individual elements. Processed data is available in the supplementary information (SI). Supplementary information: GPC trace of the model polystyrene, IR spectra, thermogravimetric analysis (TGA) traces, NMR spectra, and X-ray photoelectron spectroscopy of survey spectra and relevant individual elements. See DOI: https://doi.org/10.1039/d5py01024a.Acknowledgements
This project was initially funded by a seed grant to L. V. K. as part of the Center for Plastics Innovation (CPI), an Energy Frontier Research Center funded by the U.S. Department of Energy, Office of Science, Basic Energy Sciences at the University of Delaware under award # DE-SC0021166 to collect preliminary data. It was subsequently funded by an American Chemical Society Petroleum Research Fund (ACS PRF) Doctoral New Investigator award (#66259-DNI7) to L. V. K. The authors thank the Surface Analysis Facility at the University of Delaware and Dr. Xu Feng for XPS assistance. XPS analysis was performed on an instrument sponsored by the National Science Foundation (NSF) under grant no. CHE-1428149. The authors acknowledge the use of NMR facilities and instrumentation supported by the National Institutes of Health under Award Numbers P20GM104316 and S10RR026962-01. TGA analysis was done on TA Instruments Discovery Series Thermogravimetric Analyzer in the Advanced Materials Characterization Lab at the University of Delaware. The authors would also like to thank Prof. Thomas Epps at the University of Delaware for access to his GPC.References
- J. H. Kim, D. W. Kang, H. Yun, M. Kang, N. Singh, J. S. Kim and C. S. Hong, Post-synthetic modifications in porous organic polymers for biomedical and related applications, Chem. Soc. Rev., 2022, 51(1), 43–56 RSC.
- S. Roh, Y. Jang, J. Yoo and H. Seong, Surface Modification Strategies for Biomedical Applications: Enhancing Cell–Biomaterial Interfaces and Biochip Performances, BioChip J., 2023, 17(2), 174–191, DOI:10.1007/s13206-023-00104-4.
- M. Rimmele, F. Glöcklhofer and M. Heeney, Post-polymerisation approaches for the rapid modification of conjugated polymer properties, Mater. Horiz., 2022, 9(11), 2678–2697, 10.1039/d2mh00519k.
- N. Ohn and J. G. Kim, Mechanochemical post-polymerization modification: Solvent-free solid-state synthesis of functional polymers, ACS Macro Lett., 2018, 7(5), 561–565 CrossRef CAS PubMed.
- B. Xue, P.-P. Huang, M.-Z. Zhu, S.-Q. Fu, J.-H. Ge, X. Li and P.-N. Liu, Highly Efficient and para-Selective C–H Functionalization of Polystyrene Providing a Versatile Platform for Diverse Applications, ACS Macro Lett., 2022, 11(11), 1252–1257, DOI:10.1021/acsmacrolett.2c00541.
- G. Menendez Rodriguez, M. M. Díaz-Requejo and P. J. Pérez, Metal-Catalyzed Postpolymerization Strategies for Polar Group Incorporation into Polyolefins Containing C–C, C
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, and Aromatic Rings, Macromolecules, 2021, 54(11), 4971–4985 CrossRef CAS.
C, and Aromatic Rings, Macromolecules, 2021, 54(11), 4971–4985 CrossRef CAS. - L. Chen, K. G. Malollari, A. Uliana, D. Sanchez, P. B. Messersmith and J. F. Hartwig, Selective, Catalytic Oxidations of C–H Bonds in Polyethylenes Produce Functional Materials with Enhanced Adhesion, Chem, 2021, 7(1), 137–145, DOI:10.1016/j.chempr.2020.11.02.
- L. M. Campos, K. L. Killops, R. Sakai, J. M. J. Paulusse, D. Damiron, E. Drockenmuller, B. W. Messmore and C. J. Hawker, Development of thermal and photochemical strategies for thiol− ene click polymer functionalization, Macromolecules, 2008, 41(19), 7063–7070 CrossRef CAS.
- A. J. Butzelaar, S. Schneider, E. Molle and P. Theato, Synthesis and Post–Polymerization Modification of Defined Functional Poly(vinyl ether)s, Macromol. Rapid Commun., 2021, 42(13), 2100133, DOI:10.1002/marc.202100133.
- J. S. Oakdale, L. Kwisnek and V. V. Fokin, Selective and orthogonal post-polymerization modification using sulfur(VI) fluoride exchange (SuFEx) and copper-catalyzed azide–alkyne cycloaddition (CuAAC) reactions, Macromolecules, 2016, 49(12), 4473–4479 Search PubMed.
- S. Aivali, K. C. Andrikopoulos and A. K. Andreopoulou, Nucleophilic Aromatic Substitution of Pentafluorophenyl-Substituted Quinoline with a Functional Perylene: A Route to the Modification of Semiconducting Polymers, Polymers, 2023, 15(12), 2721, DOI:10.3390/polym15122721.
- A. Sarı, C. Alkan and A. Bicer, Synthesis and thermal properties of polystyrene-graft-PEG copolymers as new kinds of solid–solid phase change materials for thermal energy storage, Mater. Chem. Phys., 2012, 133(1), 87–94 CrossRef.
- Y. Gao, H.-M. Li, F.-S. Liu, X.-Y. Wang and Z.-G. Shen, Synthesis and characterization of benzoylated syndiotactic polystyrene, J. Polym. Res., 2007, 14(4), 291–296, DOI:10.1007/s10965-007-9109-7.
- M. E. Wright, E. G. Toplikar and S. A. Svejda, Details concerning the chloromethylation of soluble high-molecular-weight polystyrene using dimethoxymethane, thionyl chloride, and a Lewis acid: a full analysis, Macromolecules, 1991, 24(21), 5879–5880 Search PubMed.
- Y. Hayakawa, N. Terasawa and H. Sawada, Trifluoromethylation by bis(trifluoroacetyl) peroxide of polymers bearing benzene rings, Polymer, 2001, 42(9), 4081–4086, DOI:10.1016/s0032-3861(00)00673-x.
- T. J. Fazekas, J. W. Alty, E. K. Neidhart, A. S. Miller, F. A. Leibfarth and E. J. Alexanian, Diversification of aliphatic C–H bonds in small molecules and polyolefins through radical chain transfer, Science, 2022, 375(6580), 545–550, DOI:10.1126/science.abh4308.
- T. J. Farmer, J. W. Comerford, A. Pellis and T. Robert, Post–polymerization modification of bio–based polymers: maximizing the high functionality of polymers derived from biomass, Polym. Int., 2018, 67(7), 775–789 Search PubMed.
- Y. Li, R. Chang and Y. X. Chen, Recent Advances in Post–polymerization Modifications on Polypeptides: Synthesis and Applications, Chem. – Asian J., 2022, 17(14), e202200318 Search PubMed.
- C. E. Hoyle and C. N. Bowman, Thiol–ene click chemistry, Angew. Chem., Int. Ed., 2010, 49(9), 1540–1573 CrossRef CAS PubMed.
- J. F. Van Guyse, Y. Bernhard, A. Podevyn and R. Hoogenboom, Non–activated Esters as Reactive Handles in Direct Post–Polymerization Modification, Angew. Chem., 2023, 135(40), e202303841 CrossRef.
- J. J. Perez Bravo, C. Gerbehaye, J.-M. Raquez and R. Mincheva, Recent advances in solid-state modification for thermoplastic polymers: a comprehensive review, Molecules, 2024, 29(3), 667 CrossRef CAS PubMed.
- C. Barner-Kowollik, F. E. Du Prez, P. Espeel, C. J. Hawker, T. Junkers, H. Schlaad and W. Van Camp, “Clicking” polymers or just efficient linking: What is the difference, Angew. Chem., Int. Ed., 2011, 50(1), 60–62 Search PubMed.
- J.-F. Lutz, M. Ouchi, D. R. Liu and M. Sawamoto, Sequence-controlled polymers, Science, 2013, 341(6146), 1238149 CrossRef PubMed.
- X. Chen, Y. Wang and L. Zhang, Recent progress in the chemical upcycling of plastic wastes, ChemSusChem, 2021, 14(19), 4137–4151 CrossRef CAS PubMed.
- L. T. J. Korley, T. H. Epps, B. A. Helms and A. J. Ryan, Toward polymer upcycling—adding value and tackling circularity, Science, 2021, 373(6550), 66–69, DOI:10.1126/science.abg4503.
- R. K. Jha, B. J. Neyhouse, M. S. Young, D. E. Fagnani and A. J. McNeil, Revisiting poly(vinyl chloride) reactivity in the context of chemical recycling, Chem. Sci., 2024, 15(16), 5802–5813, 10.1039/d3sc06758k.
- M. N. Hodges, M. J. Elardo, J. Seo, A. F. Dohoda, F. E. Michael and M. R. Golder, Upcycling of Polybutadiene Facilitated by Selenium–Mediated Allylic Amination, Angew. Chem., Int. Ed., 2023, 62(20), e202303115, DOI:10.1002/anie.202303115.
- D. K. Nguyen, Z. O. G. Schyns, L. T. J. Korley and D. G. Vlachos, Sustainable upcycling of polyethylene waste to compatibilizers and valuable chemicals, Green Chem., 2025, 27(40), 12642–12658, 10.1039/d5gc02799c.
- P. S. Mehta, M. B. Levy, R. M. O’Dea, T. H. Epps and M. P. Watson, Towards the valorization of poly(acrylic acid) waste: organophotoredox-catalyzed decarboxylative fluorination, Polym. Chem., 2025, 16(38), 4233–4239, 10.1039/d5py00774g.
- N. V. Tsarevsky, Hypervalent iodine–mediated direct azidation of polystyrene and consecutive click–type functionalization, J. Polym. Sci., Part A: Polym. Chem., 2010, 48(4), 966–974 Search PubMed.
- S. E. Lewis, B. E. Wilhelmy and F. A. Leibfarth, Organocatalytic C–H fluoroalkylation of commodity polymers, Polym. Chem., 2020, 11(30), 4914–4919 RSC.
- A. O. Biying, M. J. H. Ong, W. Zhao, N. Li, H.-K. Luo and J. M. Chan, Broad-Spectrum Antimicrobial Polymer-Coated Fabrics via Commodity Polystyrene Upcycling, ACS Appl. Polym. Mater., 2024, 6(14), 8169–8177 Search PubMed.
- R. Liu, C. Lin, Y. Zou, Y. Zhang, W. Sun, Y. Yang, C. Wei, J. Zhong and L. Shen, Post-polymerization modification of polystyrene through Mn-catalyzed phosphorylation of aromatic C (sp2)–H bonds, ACS Appl. Polym. Mater., 2024, 6(18), 11633–11640 Search PubMed.
- C.-Y. Lo, K. Koutsoukos, D. M. Nguyen, Y. Wu, D. Angel Trujillo, T. Miller, T. Shrestha, E. Mackey, V. S. Damani, U. Kanbur, R. Opila, D. Martin, D. Kaphan and L. V. Kayser, Imidazolium-based sulfonating agent to control the degree of sulfonation of aromatic polymers and enable plastics-to-electronics upgrading, JACS Au, 2024, 4(7), 2596–2605, DOI:10.1021/jacsau.4c00355.
- B. Dou, Y. Xu and J. Wang, Gold-catalyzed precise bromination of polystyrene, J. Am. Chem. Soc., 2023, 145(18), 10422–10430 CrossRef CAS PubMed.
- P. S. Fier and K. M. Maloney, NHC-Catalyzed Deamination of Primary Sulfonamides: A Platform for Late-Stage Functionalization, J. Am. Chem. Soc., 2019, 141(4), 1441–1445, DOI:10.1021/jacs.8b11800.
- G. Acik, Fabrication of polypropylene fibers possessing quaternized ammonium salt based on the combination of CuAAC click chemistry and electrospinning, React. Funct. Polym., 2021, 168, 105035 CrossRef CAS.
- S. Kliewer, S. G. Wicha, A. Bröker, T. Naundorf, T. Catmadim, E. K. Oellingrath, M. Rohnke, W. R. Streit, C. Vollstedt, H. Kipphardt and W. Maison, Contact-active antibacterial polyethylene foils via atmospheric air plasma induced polymerisation of quaternary ammonium salts, Colloids Surf., B, 2020, 186, 110679 CrossRef CAS PubMed.
- L. Hager, M. Hegelheimer, J. Stonawski, A. T. Freiberg, C. Jaramillo-Hernández, G. Abellán, A. Hutzler, T. Böhm, S. Thiele and J. Kerres, Novel side chain functionalized polystyrene/O-PBI blends with high alkaline stability for anion exchange membrane water electrolysis (AEMWE), J. Mater. Chem. A, 2023, 11(41), 22347–22359 RSC.
- A. M. Debela, C. Gonzalez, M. Pucci, S. M. Hudie and I. Bazin, Surface Functionalization Strategies of Polystyrene for the Development Peptide-Based Toxin Recognition, Sensors, 2022, 22(23), 9538 CrossRef CAS PubMed.
- L. Li, X. Shao, Z. Zhao, X. Liu, L. Jiang, K. Huang and S. Zhao, Synergistic fire hazard effect of a multifunctional flame retardant in building insulation expandable polystyrene through a simple surface-coating method, ACS Omega, 2020, 5(1), 799–807 CrossRef CAS PubMed.
- M. M. Salunkhe, R. B. Mane and A. S. Kanade, Polymer-supported analogues of halogeno-sulphonamides preparation and applications in synthetic organic chemistry, Eur. Polym. J., 1991, 27(6), 461–463, DOI:10.1016/0014-3057(91)90167-m.
- H. Stetter and H. Kuhlmann, The catalyzed nucleophilic addition of aldehydes to electrophilic double bonds, Org. React., 2004, 40, 407–496 Search PubMed.
- F. Sun, T. Yin, Y. Wang, A. Feng, L. Yang, W. Wu, C. Yu, T. Li, D. Wei and C. Yao, A combined experimental and computational study of NHC-promoted desulfonylation of tosylated aldimines, Org. Chem. Front., 2020, 7(3), 578–583, 10.1039/c9qo01402k.
- S. Cacchi, G. Fabrizi, A. Goggiamani, L. M. Parisi and R. Bernini, Unsymmetrical diaryl sulfones and aryl vinyl sulfones through palladium-catalyzed coupling of aryl and vinyl halides or triflates with sulfinic acid salts, J. Org. Chem., 2004, 69(17), 5608–5614 CrossRef CAS PubMed.
- X. Pan, J. Gao, J. Liu, J. Lai, H. Jiang and G. Yuan, Synthesis of sulfonamides via I 2-mediated reaction of sodium sulfinates with amines in an aqueous medium at room temperature, Green Chem., 2015, 17(3), 1400–1403 RSC.
- C. Zhou, Q. Liu, Y. Li, R. Zhang, X. Fu and C. Duan, Palladium-catalyzed desulfitative arylation by C–O bond cleavage of aryl triflates with sodium arylsulfinates, J. Org. Chem., 2012, 77(22), 10468–10472 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |