Open Access Article
Open Access ArticleA one-shot activating reagent of neodymium carboxylate for highly cis-1,4 specific butadiene polymerization
Yu
Jia
a,
Toru
Wada
 b,
Yuushou
Nakayama
b,
Yuushou
Nakayama
 a,
Takeshi
Shiono
a,
Takeshi
Shiono
 a and
Ryo
Tanaka
a and
Ryo
Tanaka
 *a
*a
aGraduate School of Advanced Science and Engineering, Hiroshima University, 1-4-1 Kagamiyama, Higashi-Hiroshima 739-8527, Japan. E-mail: rytanaka@hiroshima-u.ac.jp
bGraduate School of Advanced Science and Technology, Japan Advanced Institute of Science and Technology, 1-1 Asahidai, Nomi, Ishikawa 923-1292, Japan
First published on 5th February 2026
Abstract
A streamlined catalytic system to produce high-cis polybutadiene (cis-PBD), consisting of neodymium carboxylate and a halogenated MAO-derived activator, was developed. Halogenated MAO was synthesized via the replacement of methyl groups of commercial MAO with chlorides using BCl3. The polymerization of butadiene under optimal conditions (Al/Nd = 50 and Cl/Nd = 2.1) at 25 °C resulted in nearly complete conversion, producing cis-PBD with a high molecular weight (Mn > 105), a narrow molecular weight distribution (Đ ∼ 2), and a high cis-content (>98%). Cl incorporation into both the MAO framework and the active species was confirmed by X-ray total scattering analysis, which showed that halogenated MAO functions as both an alkylating and halogenating agent for the neodymium salt. Halogenated MAO may replace traditional ternary cocatalyst systems, giving a simpler catalyst composition and higher stereoregularity.
Introduction
cis-1,4-Polybutadiene (cis-PBD) is one of the most widely used elastomers, with many applications such as automotive tires or a modifier of plastics. The high chain flexibility of cis-PBD allows operation at lower temperatures and results in low hysteresis energy loss, and its highly ordered structure undergoes strain-induced crystallization, which contributes to its excellent mechanical properties. In particular, for the tire industry, highly cis-regular PBD (95% or more) is desired because it exhibits low rolling resistance. The fundamental physical properties dictating the advantages of cis-PBD, such as resistance to abrasion or tearing, tensile strength, permanent deformation, and crystallization rate, are dramatically improved with each increasing 1% with respect to cis-1,4 content.1–4The industrial production of cis-PBD is performed mainly using Ziegler–Natta catalysts containing nickel, cobalt, titanium, and lanthanide metals, typically achieving a cis-1,4 ratio of more than 90%.5 In particular, Nd-based Ziegler–Natta catalysts, discovered in the 1960s, swiftly emerged as the predominant focus in both research and industry owing to their exceptional activity and stereoselectivity in diene polymerization.6,7 Ternary systems such as Nd-versatate (Nd(vers)3)/alkylaluminum (Et3Al, iBu3Al, methylaluminoxane (MAO), or iBu2AlH)/halogen sources (Et3Al2Cl3, Et2AlCl, SiCl4, or tBuCl) are the most representative systems, typically yielding cis-1,4 isomers with approximately 95–98%.8–16
The activation mechanism of the ternary system consists of three fundamental reactions: removal of coordinating water and excess carboxylic acids from the neodymium center by alkylaluminum, alkylation of neodymium carboxylate by alkylaluminum, and chlorination of the remaining carboxylate or alkyl. The obtained active species has a mixed R/Cl first coordination shell with vacant coordination sites for the monomer and sufficient Lewis acidity (Scheme 1). These mechanisms are supported by the various reactions observed on neodymium carboxylates17–21 and halides.22–24 Generally, cis-1,4 specific polymerization is preferred when η4-pre-coordination of the diene monomer is favored. The coordination mode of the monomer can be determined by steric effects, which are mainly explained by the coordination numbers of the metal center in lanthanide chemistry.25 Therefore, active species for highly cis-specific polymerization generated in the ternary system would have a low coordination number. Indeed, X-ray absorption studies have given critical evidence for the formation of active species with less-aggregated and chlorine-coordinated species.26–28
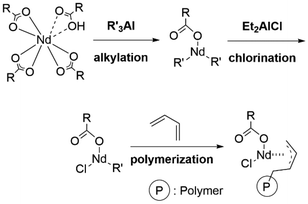 | ||
| Scheme 1 Proposed activation and propagation mechanism of Nd carboxylate-catalyzed butadiene polymerization. | ||
In this ternary system, many operational parameters like the Al/Cl ratio, residual water or acid derived from neodymium salt, addition sequence, and catalyst aging duration can all significantly alter polymerization behavior. The X-ray absorption studies mentioned above also showed that Al/Nd/Cl ratios and aging time greatly influence the coordination number of the active species. These factors make it difficult to optimize the polymerization activity and achieve precise control of the molecular weight. Therefore, the simplification of the catalyst composition and process operation while maintaining high cis-specificity remains a vital and challenging goal in this field.
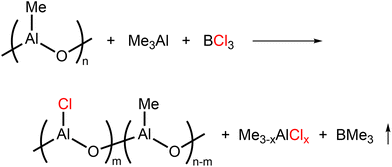
To simplify the catalyst composition, an aluminum compound containing a smaller amount of halogen per aluminum would be a promising candidate as a reagent sufficient to activate Nd carboxylate precursors by itself. Very recently, we have developed halogenated aluminoxane cocatalysts derived from commercially available MAO and BCl3.29 The resulting cocatalyst activated Ni-diimine catalysts in a similar manner to halogen-free MAO and promoted ethylene polymerization. The ethylene polymerization behavior, particularly the branching frequency, was strongly dependent on the degree of halogenation. Here, the structural replacement of methyl groups in MAO with the halogen was indicated by NMR and X-ray total scattering measurements with pair distribution function (PDF) analyses (Scheme 2). Therefore, the halogenated MAO simultaneously behaves as an alkylating reagent and a halogen donor for metal precursors.
In this study, we found that our halogenated MAO is effective for the activation of a neodymium carboxylate precursor and promotes butadiene polymerization. Quantitative monomer conversion, extremely high molecular weight, and high cis-1,4 regularity were all accomplished by tuning the Al/Cl/Nd ratio. We also performed X-ray total scattering analyses to clarify the difference between a conventional ternary catalyst system and our system, detecting the specific binding of halogenated MAO with the neodymium metal.
Experimental section
Materials
All anhydrous solvents used in the moisture and oxygen-free experiments were purchased from Kanto Chemical Co. and purified by passing through a solvent purification system fitted with activated alumina or dry molecular sieve packing. All procedures were carried out using standard Schlenk line techniques under a nitrogen atmosphere. Butadiene solution (2.4 M in toluene) and BCl3 solution (1.0 M in n-heptane) were purchased from TCI Co. Ltd and used as received. Methylaluminoxane (MAO, 2.8 M in toluene) was generously donated by Tosoh Finechem Co. and stored at −30 °C. The halogenated MAO was prepared from MAO and BCl3 according to our previous literature.29 Neodymium 2-ethylhexanoate was stored as a stock solution (0.24 mmol g−1) in n-hexane.General measurement methods
The 13C (150 MHz) NMR spectra of the obtained polymers were recorded at 130 °C using a JEOL ECZL 600 spectrometer. The signals of the reference solvent [δ = 74.7 ppm (C2D2Cl4)] were employed to calibrate the acquired spectra. The cis/trans-1,4 ratio was determined independently by the integration of two sets of diagnostic 13C signals: (i) the aliphatic region (27 ppm for cis-1,4 and 32 ppm for trans-1,4) and (ii) the olefinic region (129–130 ppm for cis-1,4 and 130–131 ppm for trans-1,4). The content reported in this work is the average of the cis values obtained from these two regions. The molecular weight and distribution of the polymers were determined by high-temperature gel permeation chromatography (GPC) using a Tosoh HLC-8321GPC/HT chromatograph (eluent: o-dichlorobenzene, temp. = 140 °C), calibrated with polystyrene standards. The concentration of the polymer samples was about 0.5 wt% and the injection volume was 200 µL. Differential scanning calorimetry (DSC) measurements were carried out using a SHIMADZU DSC-60 Plus system under a nitrogen flow (flow rate 50 mL min−1), with about 3 mg of polymer on an aluminum sample pan. The test program was set up as follows: the instrument was stabilized at 40 °C for 5 minutes, heated at 10 °C min−1 to 100 °C, cooled at 10 °C min−1 to −130 °C, and then reheated at 10 °C min−1 to 100 °C. To minimize the influence of thermal history, analyses for glass transition temperatures (Tg) and melting points (Tm) were performed using DSC curves collected during the second heating process.Total X-ray scattering measurements were conducted at the BL04B2 beamline of SPring-8. The samples were vacuum-dried previously to improve the signal-to-noise ratio. Each sample was loaded into a 1 mm quartz capillary under nitrogen-filled conditions. To prevent contamination from the atmosphere, the capillary was sealed with UV-curable glue. The sample capillary was placed in high throughput measuring equipment at the beamline and exposed to 112.8 keV X-rays for 600 seconds. Scattered X-rays were captured with a two-dimensional CdTe semiconductor detector. The scattering intensity of an empty capillary was measured under the same conditions and then subtracted as background. The sample's scattering intensity was analyzed using normal procedures to produce the atomic pair distribution function (PDF, G(r)) curve.
General procedure for butadiene polymerization
In a 20 mL Schlenk flask, halogenated MAO (4.0 mmol) was diluted with toluene (2.0 mL) under nitrogen. Neodymium 2-ethylhexanoate (80 μmol) in toluene (3.4 mL) was then added to the halogenated MAO solution. The pale purple solution was obtained after stirring for 20 minutes. The butadiene solution in toluene (2.4 M, 4.2 mL, 10 mmol) was injected and stirred to start the polymerization, which gave a light blue solution, and the system's viscosity progressively increased as the reaction proceeded. After stirring at room temperature for 2 hours, the reaction mixture was poured into acidic methanol containing 2% hydrochloric acid and the precipitated solid was recovered. The polymer was dried under vacuum overnight until it reached a constant weight.Results and discussion
Halogenated MAO was prepared according to the previous literature29 by adding various amounts of BCl3 (0.33–2.0 mol% relative to Al) to a commercial MAO solution in toluene at room temperature. The vacuum-dried halogenated MAO was redissolved in toluene, reacted with neodymium 2-ethylhexanoate, and utilized in butadiene polymerization (Table 1). Here, catalyst aging time did not affect the polymerization behavior, and the following polymerizations were started by monomer addition 20 minutes after mixing the Nd precursor and halogenated MAO.| Run | Cl/Nd | Yield (%) | M n (103) | Đ | cis (%) | T g (°C) | T m (°C) | ΔHmd (J g−1) |
|---|---|---|---|---|---|---|---|---|
| a Polymerization conditions: solvent = toluene, temp. = 25 °C, time = 2 h, Nd = 80 µmol, Al/Nd = 50, Bd = 10 mmol. b Determined by GPC calibrated with polystyrene standards. c cis-1,4 microstructure ratio determined by 13C NMR. d Determined by DSC. e Not detected. | ||||||||
| 1 | 0 | 75 | 45 | 2.0 | 70 | −103 | n.d.e | n.d.e |
| 2 | 0.5 | 87 | 55 | 1.8 | 72 | −104 | n.d.e | n.d.e |
| 3 | 1.0 | 91 | 43 | 1.9 | 79 | −105 | n.d.e | n.d.e |
| 4 | 1.5 | 90 | 68 | 2.4 | 87 | −110 | −9 | 12 |
| 5 | 2.1 | 99 | 156 | 1.7 | 98 | −111 | −7 | 33 |
| 6 | 2.5 | 99 | 108 | 1.8 | 98 | −110 | −7 | 28 |
| 7 | 3.0 | 97 | 152 | 1.8 | 98 | −110 | −7 | 26 |
Compared with the halogen-free MAO (run 1), halogenated MAO enhanced the cis-1,4 specificity and molecular weight of the polymer along with an increase of halogenation degree (runs 2–7). The microstructure of the polymer other than the cis-1,4 sequence was mostly the trans-1,4 sequence, and the 1,2-sequence was not detected by 13C NMR spectra (see SI Fig. S19–S33). As is already known, the glass transition temperature (Tg) of the obtained polybutadiene was below −100 °C and it further decreased with an increase of cis-1,4 regularity. In addition, a melting temperature of around −10 °C was observed for a highly cis-1,4 specific fraction, which is slightly lower than the reported value of perfectly cis-1,4 regular polybutadiene.30 Under these polymerization conditions, monomer conversion was not quantitative when unmodified MAO was used, whereas halogenated MAO gave the polymer almost quantitatively. These tendencies were similar to what has been observed in previously reported ternary systems. The obtained polymers were not completely soluble in THF or chloroform at ambient temperature, giving visible residues. Therefore, the evaluation of molecular weight by GPC and microstructure analysis by NMR were performed at high temperatures. All the GPC traces of the obtained polybutadiene showed a unimodal shape (Fig. S1–S15), and the molecular weight distribution (Đ) was around 2.0, which was relatively narrow for Nd-based systems. This relatively narrow dispersity suggests the formation of more uniform active species and/or improved control over chain-transfer processes compared to traditional ternary systems. The use of MAO generally contributes to narrowing the molecular weight distribution. However, both the stereospecificity and molecular weight under optimal conditions (runs 5–7) were higher than those obtained for the Nd(OiPr)3/MAO/tBuCl system.31 We further investigated the effect of polymerization conditions at a Cl/Nd ratio of 2.1 (run 5), which provided a balance between high conversion, high cis-1,4 content and high molecular weight.
The halogenated MAO system revealed a distinct low limit in the Al/Nd ratio (Table 2). A low Al/Nd ratio (Al/Nd = 30) led to insufficient activation/halogenation, producing a very small amount of polymer (run 8). Although the polymer yield was not high enough for microstructure quantification by 13C NMR, the Tg of the polymer obtained under these conditions was close to those of other highly stereoregular polybutadiene compounds, indicating high cis-specificity.
| Run | Al/Nd | Yield (%) | M n (103) | Đ | cis (%) | T g (°C) | T m (°C) | ΔHmd (J g−1) |
|---|---|---|---|---|---|---|---|---|
| a Polymerization conditions: solvent = toluene, temp. = 25 °C, time = 2 h, Nd = 80 µmol, Cl/Nd = 2.1, Bd = 10 mmol. b Determined by GPC calibrated with polystyrene standards. c cis-1,4 microstructure ratio determined by 13C NMR. d Determined by DSC. e Not determined. f Both halogenated MAO (20 equiv. to Nd) and halogen-free MAO (30 equiv. to Nd) were used to maintain the Cl/Nd ratio. | ||||||||
| 5 | 50 | 99 | 156 | 1.7 | 98 | −111 | −7 | 33 |
| 8 | 30 | 4 | 10 | 1.5 | —e | −112 | −6 | 26 |
| 9 | 70 | 97 | 30 | 2.5 | 97 | −111 | −6 | 12 |
| 10 | 20 + 30f | 99 | 151 | 1.7 | 98 | −110 | −7 | 28 |
On the other hand, a higher Al/Nd ratio caused a rapid drop in molecular weight (run 9), probably because of the increase in initiation efficiency. A mixture of halogenated MAO and halogen-free MAO (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3) (run 10) gave almost the same result as the halogenated MAO exhibiting the same Al/Cl/Nd ratio (run 5). Therefore, the distribution of chlorides seems not to affect the polymerization behavior. Polymerizations at various Bd/Nd ratios of up to 2000 all achieved almost complete conversion within 2 hours, with perfect cis-1,4 specificity (Table 3, runs 11–15). The molecular weight of the polymer showed only a slight increase at higher Bd/Nd ratios, maintaining a narrow molecular weight distribution. The results clearly demonstrate a wide operational range of our system using halogenated MAO. The molecular weight change would be mainly governed by the frequency of chain transfer to excess alkylaluminum, leading to an almost constant Mn over a broad range of Bd/Nd ratios, while at very high Bd/Nd ratios (i.e. low Al concentration), Mn slightly increased.
3) (run 10) gave almost the same result as the halogenated MAO exhibiting the same Al/Cl/Nd ratio (run 5). Therefore, the distribution of chlorides seems not to affect the polymerization behavior. Polymerizations at various Bd/Nd ratios of up to 2000 all achieved almost complete conversion within 2 hours, with perfect cis-1,4 specificity (Table 3, runs 11–15). The molecular weight of the polymer showed only a slight increase at higher Bd/Nd ratios, maintaining a narrow molecular weight distribution. The results clearly demonstrate a wide operational range of our system using halogenated MAO. The molecular weight change would be mainly governed by the frequency of chain transfer to excess alkylaluminum, leading to an almost constant Mn over a broad range of Bd/Nd ratios, while at very high Bd/Nd ratios (i.e. low Al concentration), Mn slightly increased.
| Run | Bd/Nd | Yield (%) | M n (103) | Đ | cis (%) | T g (°C) | T m (°C) | ΔHmd (J g−1) |
|---|---|---|---|---|---|---|---|---|
| a Polymerization conditions: solvent = toluene, temp. = 25 °C, time = 2 h, Al/Nd = 50, Cl/Nd = 2.1, Bd = 10 mmol. b Determined by GPC calibrated with polystyrene standards. c cis-1,4 microstructure ratio determined by 13C NMR. d Determined by DSC. | ||||||||
| 5 | 125 | 99 | 156 | 1.7 | 98 | −111 | −7 | 33 |
| 11 | 500 | 97 | 118 | 2.6 | 98 | −110 | −8 | 28 |
| 12 | 750 | 96 | 148 | 1.9 | 98 | −110 | −8 | 32 |
| 13 | 1000 | 97 | 138 | 2.1 | 98 | −110 | −9 | 31 |
| 14 | 1500 | 98 | 180 | 1.9 | 99 | −111 | −7 | 37 |
| 15 | 2000 | 98 | 214 | 1.7 | 99 | −111 | −7 | 38 |
Conventional ternary neodymium catalyst systems, based on classical formulations of an Nd salt, a cocatalyst, and a separate halide donor, typically deliver high cis-1,4 specificity but often require elevated temperatures to reach high conversion (Table 4). Nevertheless, despite qualitative comparison, achieving high molecular weight and narrow molecular weight distribution simultaneously remains challenging for many of these systems. In contrast, our Nd/BCl3-modified MAO combination matches their stereocontrol under very mild conditions with no additional chloride reagent, offering a simpler protocol with potential energy savings.
| Nd source | Cocat. | Halide donor | Bd/Nd | Temp. (°C) | Time (h) | Yield (%) | M n (104) | Đ | cis (%) |
|---|---|---|---|---|---|---|---|---|---|
| Nd(2-ethylhexanoate)3 (this work) | BCl3–MAO | — | 2000 | 25 | 2 | 98 | 21.4 | 1.7 | 99 |
Nd(neodecanate)3·(neodecanoic acid)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
iBu2AlH/iBu3Al | Et2AlCl | 42![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
60 | 2 | 90 | 23.2 | 2.6 | 99 |
Nd(vers)3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 4 |
iBu3Al | Et2AlCl | 3700 | 45 | 0.67 | 92 | 9.6 | 3.1 | 97 |
Nd(vers)3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9 9 |
iBu2AlH | SiCl4 | 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 800 800 |
70 | 1 | 81 | 4.2 | 6.5 | 97 |
Nd(vers)3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 10 |
iBu2AlH | Et3Al2Cl3 | 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 800 800 |
70 | 1 | 98 | 2.8 | 5.3 | 99 |
Nd(vers)3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 11 11 |
iBu2AlH | Et3Al2Cl3 | 9100 | 60 | 0.8 | 81 | 6.0 | 2.4 | 95 |
[Nd(Ph2HCCOO)3 (thf)2]2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 20 |
iBu3Al | Et3Al2Cl3 | 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
50 | 3 | 89 | 42.1 | 2.5 | 99 |
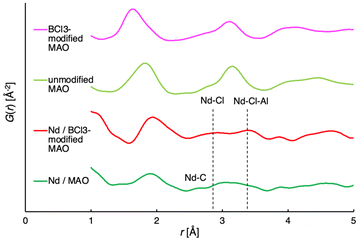
Upon achieving the outstanding performance of the catalyst system using our halogenated MAO, we are motivated to gain insight into the structure of the active species. However, an MAO-derived cocatalyst has a heterogeneous structure, and it is difficult to determine the exact structures and distributions of specific active species. Therefore, we focused on detecting the local interatomic distances in active species by X-ray total scattering measurements, combined with pair distribution function (PDF) analyses.32,33 PDF analysis provides information on average interatomic distances in the sample. For the same purpose, EXAFS played a very important role in detecting Nd–C, Nd–Cl, and Nd–Al structures, which have characteristic distances,26–28 as reported in previous literature. Our PDF analysis results of the neodymium 2-ethylhexanoate/BCl3-modified MAO (Al/Nd = 50, Cl/Nd = 2.5) showed a broad feature at around 2.9 Å, which is consistent with the presence of the Nd–Cl bond (Fig. 1). We note that the Nd–C distance expected from alkylation (∼2.6 Å) may partially overlap with this broad region. In addition, a feature at around 3.4 Å can be assigned to a secondary neighbour Nd⋯Al correlation mediated by Cl (Nd–Cl–Al). While PDF analysis reflects the ensemble-averaged environment, the value observed is notably longer than that in the Nd/unmodified MAO mixture (∼3.1 Å) and even longer than the Nd–Al distances in the Nd(vers)3/iBu3Al/iBu2AlCl (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10
10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3) system previously observed by EXAFS measurements.
3) system previously observed by EXAFS measurements.
The alkyl-bridged neodymium–aluminum binuclear complexes typically show a Nd–Al distance within the range of 3.1–3.3 Å,19,34,35 whereas chloro-bridged neodymium–aluminum multinuclear complexes often show larger distances of Nd and Al (3.6–3.8 Å)36–38 (Fig. S35). Therefore, the observed Nd–Al correlation at 3.4 Å in the BCl3-modified MAO system represents an intermediate state that reflects a higher extent of chlorination and a greater contribution of chloro-bridged environments compared to the unmodified or traditional ternary systems. Such structural characteristics may contribute to the observed high activity and relatively narrow molecular weight distribution.
Conclusions
We presented a strategy for simplifying the formulation of a cocatalyst for Nd-catalyzed butadiene polymerization by using partially halogenated aluminoxane. Moreover, the halogenated MAO gave highly cis-1,4 regular, high molecular weight polybutadiene with a narrow molecular weight distribution. Therefore, we successfully demonstrated the advantage of this catalyst system from a practical point of view. Furthermore, X-ray total scattering measurements suggested efficient chlorination at the neodymium center in this system, which may lead to high activity at ambient temperature.Author contributions
J. Y.: investigation, data curation, and writing the original draft; T. W.: methodology and investigation for PDF analyses; Y. N. and T. S.: review and editing; and R. T.: conceptualization, supervision, and validation. All authors have read and approved the final version of the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: NMR spectra of polymers, DSC thermograms and GPC traces of the obtained polymers, and raw profiles or X-ray total scattering data, examples of Nd/Al multinuclear complexes. See DOI: https://doi.org/10.1039/d6py00007j.Acknowledgements
The authors gratefully acknowledge the generous donation of methylaluminoxane from Tosoh Finechem Co. Ltd. The authors are also grateful to the Natural Science Center for Basic Research and Development (N-BARD) at Hiroshima University for high-temperature NMR measurements. The synchrotron radiation experiments were performed at the BL04B2 beamline of SPring-8 with the approval of the Japan Synchrotron Radiation Research Institute (JASRI) (T. W.).References
- G. Kwag, P. Kim, S. Han and H. Choi, Polymer, 2005, 46, 3782–3788 CrossRef CAS.
- N. M. T. Pires, A. A. Ferreira, C. H. de Lira, P. L. A. Coutinho, L. F. Nicolini, B. G. Soares and F. M. B. Coutinho, J. Appl. Polym. Sci., 2006, 99, 88–99 CrossRef CAS.
- H. Zhu, P. Chen, C.-F. Yang and Y.-X. Wu, Macromol. React. Eng., 2015, 9, 453–461 CrossRef CAS.
- M. L. Méndez-Hernández, J. L. Rivera-Armenta, U. Páramo-García, S. C. Galvan, R. García-Alamilla and B. A. Salazar-Cruz, Int. J. Polym. Sci., 2016, 2016, 7239540 Search PubMed.
- G. Ricci, G. Pampaloni, A. Sommazzi and F. Masi, Macromolecules, 2021, 54, 5879–5914 Search PubMed.
- N. G. Marina, Y. B. Monakov, Z. M. Sabirov and G. A. Tolstikov, Polym. Sci. U.S.S.R., 1991, 33, 387–417 CrossRef.
- H. Wang, J. M. O. Cue, E. L. Calubaquib, R. N. Kularatne, S. Taslimy, J. T. Miller and M. C. Stefan, Polym. Chem., 2021, 12, 6790–6823 RSC.
- D. J. Wilson and D. K. Jenkins, Polym. Bull., 1995, 34, 257–264 CrossRef CAS.
- R. P. Quirk and A. M. Kells, Polym. Int., 2000, 49, 751–756 CrossRef CAS.
- R. P. Quirk, A. M. Kells, K. Yunlu and J. P. Cuif, Polymer, 2000, 41, 5903–5908 CrossRef CAS.
- L. Friebe, O. Nuyken, H. Windisch and W. Obrecht, Macromol. Chem. Phys., 2002, 203, 1055–1064 CrossRef CAS.
- I. L. Mello and F. M. Coutinho, Eur. Polym. J., 2008, 44, 2893–2898 CrossRef CAS.
- I. L. Mello and F. M. Coutinho, J. Appl. Polym. Sci., 2009, 112, 1496–1502 CrossRef CAS.
- R. Díaz de León, F. Soriano Corral, F. J. Enríquez-Medrano, G. Bosques Ibarra, P. de León Martínez, F. Hernández Gámez, H. R. López-González and L. F. R. de Valle, Int. J. Polym. Sci., 2016, 2016, 9841896 Search PubMed.
- C. C. Gaioto, M. Schmal and J. C. da Silva Pinto, Catal. Today, 2020, 344, 84–91 CrossRef CAS.
- K. A. Tereshchenko, N. V. Ulitin, P. S. Bedrina, D. A. Shiyan, A. D. Lifanov, T. I. Madzhidov and S. I. Volfson, Ind. Eng. Chem. Res., 2022, 61, 15961–15969 CAS.
- G. Kwag, Macromolecules, 2002, 35, 4875–4879 CrossRef CAS.
- A. Fischbach, F. Perdih, P. Sirsch, W. Scherer and R. Anwander, Organometallics, 2002, 21, 4569–4571 CrossRef CAS.
- A. Fischbach, F. Perdih, E. Herfweck and R. Anwander, Organometallics, 2006, 25, 1626–1642 CrossRef CAS.
- D. M. Roitershtein, A. A. Vinogradov, A. A. Vinogradov, K. A. Lyssenko, Y. V. Nelyubina, I. V. Anan'ev, I. E. Nifant'ev, V. A. Yakovlev and N. N. Kostitsyna, Organometallics, 2013, 32, 1272–1286 Search PubMed.
- A. A. Vinogradov, M. E. Minyaev, K. A. Lyssenko and I. E. Nifant'ev, Acta Crystallogr., Sect. E: Crystallogr. Commun., 2019, 75, 456–459 CrossRef CAS.
- K. Lv and D. Cui, Organometallics, 2010, 29, 2987–2993 CrossRef CAS.
- R. Tanaka, Y. Nakayama and T. Shiono, Organometallics, 2020, 39, 1855–1860 CrossRef CAS.
- A. O. Tolpygin, A. D. Mikhailychev, T. A. Kovylina, K. A. Kozhanov, A. V. Cherkasov and A. A. Trifonov, Russ. Chem. Bull., 2023, 72, 2655–2666 CrossRef CAS.
- R. Tanaka, K. Yuuya, H. Sato, P. Eberhardt, Y. Nakayama and T. Shiono, Polym. Chem., 2016, 7, 1239–1243 RSC.
- G. Kwag, H. Lee and S. Kim, Macromolecules, 2001, 34, 5367–5369 CrossRef CAS.
- H. Guo, J. Bi, J. Wang, X. Zhang, S. Jiang and Z. Wu, Dalton Trans., 2015, 44, 9130–9139 RSC.
- H. L. Guo, J. F. Bi, Q. Y. Wu, J. Y. Wang, W. Q. Shi, X. Q. Zhang, S. C. Jiang and Z. H. Wu, RSC Adv., 2017, 7, 14413–14421 RSC.
- Y. Jia, N. Harada, T. Wada, Y. Nakayama, T. Shiono and R. Tanaka, Polym. Chem., 2025, 16, 4381–4388 RSC.
- J. C. Mitchell, J. Polym. Sci., Part B, 1963, 1, 285–288 Search PubMed.
- W. Dong, K. Endo and T. Masuda, Macromol. Chem. Phys., 2003, 204, 104–110 CrossRef CAS.
- S. Kohara, M. Itou, K. Suzuya, Y. Inamura, Y. Sakurai, Y. Oishi and M. Takata, J. Phys.: Condens. Matter, 2007, 19, 506101 Search PubMed.
- T. Wada and T. Taniike, Nanoscale, 2025, 17, 6767–6779 RSC.
- W. J. Evans, R. Anwander and J. W. Ziller, Organometallics, 1995, 14, 1107–1109 Search PubMed.
- G. Occhipinti, C. Meermann, H. M. Dietrich, R. Litlabø, F. Auras, K. W. Törnroos, C. Maichle-Mössmer, V. R. Jensen and R. Anwander, J. Am. Chem. Soc., 2011, 133, 6323–6337 CrossRef CAS.
- P. Biagini, G. Lugli, L. Abis and R. Millini, New J. Chem., 1995, 19, 713–722 CAS.
- Q. Liu, Y.-H. Lin and Q. Shen, Acta Crystallogr., Sect. C: Cryst. Struct. Commun., 1997, 53, 1579–1580 Search PubMed.
- B. Fan, Q. Shen and Y. Lin, J. Organomet. Chem., 1989, 377, 51–58 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |