Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Polyesters with inbuilt photolabile units for degradation of PET in the natural environment
Sami
Gesslbauer
a,
Tian
Sang
a,
Daniel
Dalland
a,
Pimpisa
Titipunya
 a,
Molly
Parry
a,
Christopher
Wallis
b,
Sarah K. Y.
Ho
b,
Gavin
Hill
b,
Giovanni
Santagiuliana
a,
Molly
Parry
a,
Christopher
Wallis
b,
Sarah K. Y.
Ho
b,
Gavin
Hill
b,
Giovanni
Santagiuliana
 b and
George J. P.
Britovsek
b and
George J. P.
Britovsek
 *a
*a
aDepartment of Chemistry, Imperial College London, Molecular Sciences Research Hub, White City Campus, 82 Wood Lane, W12 0BZ, UK. E-mail: g.britovsek@imperial.ac.uk
bPolymateria Ltd, Translation & Innovation Hub, Imperial College White City Campus, 80 Wood Lane, London W12 0BZ, UK
First published on 12th February 2026
Abstract
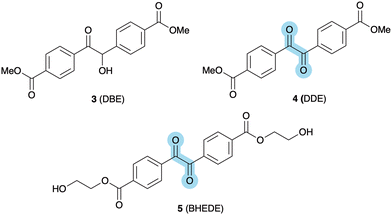
The proliferation of waste plastic is a growing environmental concern due to its harmful effects on ecosystems, wildlife and human health. Here we have investigated the introduction of photolabile carbonyl and dicarbonyl units into polyethylene terephthalate (PET) with the aim to enhance the degradability of PET in the natural environment. Single carbonyl units have been introduced using 1,3-dihydroxyacetone as the diol component in place of ethylene glycol. Studies on the thermal behaviour of molecular polymer models have shown insufficient stability of the dihydroxyacetone unit under typical PET polymerisation conditions (∼270 °C). The introduction of dicarbonyl units into PET was achieved using a dimethyl dicarbonyl ester (DDE) or di(hydroxyethyl) dicarbonyl ester (BHEDE) which have been incorporated into PET through transesterification methods. Effective removal of methanol or glycol is extremely difficult under these conditions and can lead to reductions in molecular weight due to transesterification. A series of copolymers using various ratios of additive/PET has been prepared through melt processing. The degradability of the resulting polymer films has been investigated using artificial weathering during a 14-day cycle with controlled temperature, humidity and UV irradiation. A lowering of the molecular weight was observed in all cases, most likely due to hydrolysis of the ester linkages, although oxidative cleavage of the dicarbonyl units could also have taken place, but the end-groups would be indistinguishable.
Introduction
Polyethylene terephthalate (PET) is the most commonly used thermoplastic polyester in the packaging and textile industries, with an annual global production of approximately 60 Mt in 2024.1,2 It is well known for its high impact strength, durability, and versatility and is widely used in the production of bottles for beverages, containers for food, and synthetic fibres for clothing and carpets. The success of PET is in part due to its excellent material properties, such as a high glass transition (Tg) and melting temperature (Tm), which make it highly suitable for a wide range of applications.1 PET polymer production requires purified terephthalic acid (PTA) and ethylene glycol (EG) and a common production method is via bis(2-hydroxyethyl)terephthalate (BHET), obtained by transesterification of dimethyl terephthalate (DMT) with ethylene glycol. BHET is heated under vacuum above the melting point of PET (∼280 °C) in the presence of a transesterification catalyst such as antimony oxide or titanium alkoxides to promote polycondensation. For PET resins intended for bottle manufacturing, average molecular weights are typically ∼30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 g mol−1. Due to the high melt viscosity, solid-state polymerization (SSP) processing techniques under vacuum are required, in order to remove the excess ethylene glycol.1
000 g mol−1. Due to the high melt viscosity, solid-state polymerization (SSP) processing techniques under vacuum are required, in order to remove the excess ethylene glycol.1
Of all plastics produced, PET has currently the highest recycling rate (52% in Europe and 31% in the US). Despite these recycling efforts, large amounts of plastic waste still end up in the natural environment.2 PET degradation occurs in the natural environment in the presence of oxygen, moisture and sunlight, but timescales are typically very long.3,4 In general, any polymer containing C–H bonds degrades in the natural environment through oxidative processes converting C–H into C–OH bonds and eventually carbonyl units.4,5 The reactivity of carbonyl groups toward UV radiation results in photochemical Norrish reactions which can lead to C–C bond cleavage. A consequent lowering of the polymer molecular weight, together with the formation of radicals that can undergo further reaction with oxygen ultimately leads to degradation of the entire material.6
Degradation is generally more pronounced in amorphous regions within the polymer due to better diffusion of oxygen and moisture into these regions.7 One approach to improve the degradability of polyesters, is to decrease the crystallinity within PET, and thereby increase the degree of photodegradability. To this end, various copolymers have been investigated using additional diacid or diol components in order to lower the crystallinity, such as polybutylene terephthalate (PBT), polybutylene adipate co-terephthalate (PBAT or Ecoflex™) or the use of cyclohexane dimethanol.8
An alternative approach is the introduction of carbonyl units into polymers in order to promote photo-degradability, an attractive idea that has been investigated for polyethylene (PE),9,10 and also in polyesters.11,12 Recent work by Mecking and others on the incorporation of carbonyl units in PE has shown important advances towards controllable degradability in polyolefins.9,13–16 Polymers made from alternating ethylene and CO units (polyketone) were envisaged at the end of last century as a potentially degradable alternative for polyethylene,17 but despite significant effort, notably by Shell and BP,18,19 this polymer has not yet become a high volume product in the market. Recently, a promising new polyketone product (POKETONE™) has been introduced as an engineering polymer by Hyosung at 50 Kt per annum production.20
Here we report the preparation of PET-based polymers with photolabile monocarbonyl (A) and dicarbonyl (B) units, as shown in Fig. 1. These carbonyl units have been incorporated in the ethylene glycol or the terephthalic acid parts of PET with the aim to enhance the rate of degradation of PET in the natural environment.
 | ||
| Fig. 1 Incorporation of monocarbonyl (A) and dicarbonyl (B) units in the ethylene glycol and terephthalic acid parts of PET polymer. | ||
Results and discussion
Introduction of monocarbonyl units
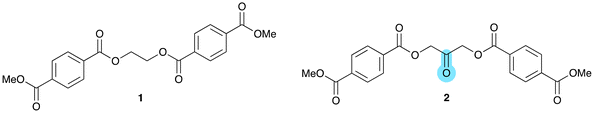
Initial experiments required the use of a molecular PET polymer model compound 1, reported by Clarke and co-workers (see Fig. 2).21 The introduction of a carbonyl unit into the ethylene glycol part was achieved using 1,3-dihydroxyacetone instead of ethylene glycol as the diol component. Polyesters based on 1,3-dihydroxyacetone have been reported before, but not in combination with terephthalic acid.22 The 1,3-dihydroxyacetone monomer is in equilibrium with its dimer at room temperature and a clean synthesis of compound 2 required initial protection via 2,2-dimethoxy-1,3-propanediol.23 Subsequent reaction with methyl terephthaloyl chloride and deprotection afforded the diester 2 in good yield (see SI for details).The thermal and photochemical reactivity of compound 2 was investigated initially under oxidative conditions, using various oxidants (H2O2, cumene hydroperoxide, urea hydrogen peroxide) and under photolytic conditions using UV light irradiation at 365 nm (see Table 1, entries 1, 2, 4 and 5). Under these conditions, conversions of 30–50% of 2 were observed, but the main reaction product according to NMR analysis was monomethyl terephthalate, resulting from hydrolysis of the ester units (see Fig. S3). The presence of an excess of d4-acetic acid (entry 3) results in H–D exchange of the enolisable methylene units. In contrast, the PET model compound 1 was stable under these conditions and showed no signs of ester hydrolysis or oxidation. The increased rate of hydrolysis observed for 2 under these conditions is promising for the potential degradation of polyesters incorporating 1,3-dihydroxyacetone. This rate enhancement is believed to be due to the known neighbouring group effect of carbonyl units on ester hydrolysis.24 1,3-Dihydroxyacetone can react with water to form a gem-diol, which undergoes intramolecular proton transfer to facilitate ester hydrolysis. However, there was no detectible decomposition related to C–C bond cleavage reactions due to irradiation using UV light at 365 nm.
| Entry | UV light (365 nm) | Temperature (°C) | Additive | [2]![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) [additive] [additive] |
Time (h) | Conversion (%) |
|---|---|---|---|---|---|---|
| All solutions in CD3CN. Additives: a: H2O2 (30%) b: CD3COOD, c: cumene hydroperoxide, d: urea hydrogen peroxide. Conversions have been determined by 1H NMR. | ||||||
| 1 | Yes | 25 | a | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 10 |
16 | 45 |
| 2 | No | 70 | a | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 10 |
1 | 50 |
| 3 | No | 70 | b | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 50 |
24 | 38 |
| 4 | Yes | 25 | c | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 4 |
28 | 31 |
| 5 | Yes | 25 | d | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 4 |
28 | 50 |
The extreme polymer processing conditions for polyesters such as PET requires monomers and additives to be thermally stable up to 280 °C. Analysis by TGA showed that 2 undergoes thermal decomposition above 230 °C (Fig. S4), which would be incompatible with typical PET polymerisation conditions of temperatures above 270 °C. Various attempts to incorporate compound 2 into PET through transesterification at 260 °C, in air or under an inert atmosphere, using various reaction conditions and transesterification catalysts such as Ti(OBu)4 or Sn(Oct)2, resulted invariably in black decomposed products, most likely due to the thermal instability of the dihydroxyacetone unit.25 This approach towards the introduction of carbonyl groups into PET was therefore not pursued further.
Introduction of dicarbonyl units
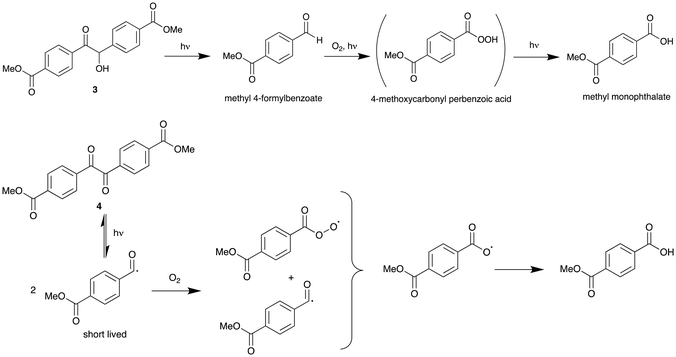
The introduction of dicarbonyl moieties as a photolabile unit within polyesters such as PET has been investigated, because the C–C bond strength in dicarbonyl units is relatively weak and these units are prone to photolytic cleavage.26,27 Furthermore, in the presence of oxygen, dicarbonyl units can be efficient triplet quenchers for the generation of highly reactive singlet oxygen, which could lead to further polymer degradation.28Dimethyl benzoin ester 3 (DBE) was prepared by the condensation of methyl 4-formyl benzoate, using 1,3,4-triphenyl-4,5-dihydro-1H-1,2,4-triazol-5-ylidene as a catalyst (Fig. 3).29,30 The dimethyl dicarbonyl ester 4 (DDE) can be prepared by oxidizing DBE (3) using a modified Swern oxidation with DMSO and aqueous HBr. Both compounds were isolated in good yield and fully characterised (see SI). It should be noted that methyl 4-formyl benzoate is readily available as a sideproduct from the industrial production of terephthalic acid.
Reactivity and mechanistic considerations
Reactivity studies have shown that DBE (3) is easily oxidised within 30 minutes at 260 °C in air to DDE (4) (see Fig. S8). The dicarbonyl compound 4 is much more stable and can be heated to 260 °C for 30 minutes without any signs of further reaction or decomposition. The UV-vis spectrum of DBE (3) in acetonitrile shows a strong absorption at 255 nm but also some weaker absorption in the visible region, responsible for its yellow colour (ε = 800 M−1 cm−1 at 400 nm, Fig. S9). Irradiation of a solution of DBE (3) in toluene in the absence of oxygen for 12 hours at 365 nm results in C–C bond cleavage of the benzoin unit and formation of methyl 4-formyl benzoate (Fig. S17), in line with similar observations for other benzoin compounds.31 In the presence of O2, oxidation generates methyl monophthalate, possibly via 4-methoxycarbonyl perbenzoic acid as an intermediate (Fig. 4).32 Some oxidation of 3 to 4 also takes place under these conditions.The UV-vis spectrum of DDE (4) shows a major absorption at 271 nm and a weaker shoulder at 232 nm, together with some residual absorption in the visible region (ε = 900 M−1 cm−1 at 400 nm, Fig. S13). Irradiation of DDE (4) at 365 nm in toluene or THF in the presence of oxygen results in the formation of methyl monophthalate (see Fig. S19 and S20), presumably via acyloxy and acylperoxy radical intermediates, as shown in Fig. 4. Irradiation under white light gave similar results but required significantly longer reaction times. EPR measurements of DDE (4) under irradiation at 405 nm for 20 minutes at room temperature shows two closely overlapping EPR signals at g = 2.0053 (Fig. S18), which are assigned to the acyloxy and acylperoxy radicals, as shown in Fig. 4. Acyl radicals are generally very short lived (less than 10−3 s), whereas acyloxy and acylperoxy radicals can last for several seconds at room temperature.33
Polymerisations reactions
Polymerisation experiments have been carried out using DDE (4) and ethylene glycol, as well as mixtures of BHET and DDE under various conditions using a catalyst combination of Sb2O3 and Ca(OAc)2. During the polymerisation, methanol and ethylene glycol are continuously removed via distillation under vacuum. The polymeric products were purified by dissolution in hot N-methylpyrrolidone (NMP) and precipitation in ethanol affording off-white/yellow polymer powders (see SI for details). A similar copolymerisation procedure using dimethyl terephthalate, ethylene glycol and DDE was reported previously with the aim to generate fluorescent polyesters.34The homo-polymerisation of DDE (4) with ethylene glycol was performed at 190 °C for 4 hours, followed by 2 hours at 250 °C under vacuum. This resulted in the new polyester PDDE, which displays two carbonyl IR stretches in an approximate 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio at 1720 and 1675 cm−1 for the ester and keto moieties, respectively (see eqn (1) in Fig. 5 and Fig. S25).
1 ratio at 1720 and 1675 cm−1 for the ester and keto moieties, respectively (see eqn (1) in Fig. 5 and Fig. S25).
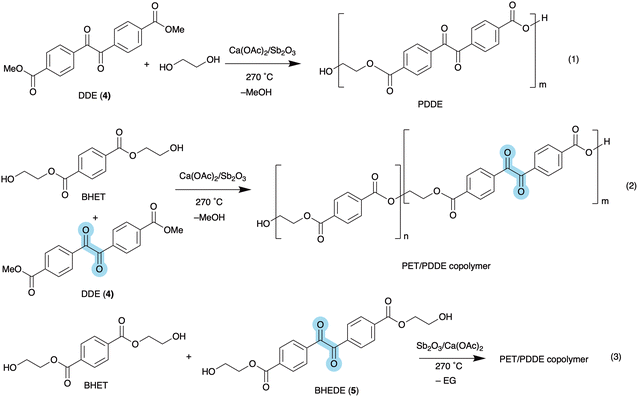 | ||
| Fig. 5 Homopolymerisation of DDE (4) and EG (1); copolymerisation of BHET and DDE (4) to PET/PDDE copolymers (2); copolymerisation of BHET and BHEDE (5) to PET/PDDE copolymers (3). | ||
Copolymers of BHET and DDE have been prepared by melt polymerisation using a mixture in a molar ratio of 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and 5
1 and 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and heating for 2 hours at 270 °C under vacuum, resulting in PET/PDDE copolymers as shown in reaction 2 in Fig. 5. Analysis by IR shows 2 carbonyl signals in different ratios (see Fig. S26). GPC analysis indicated relatively low molecular weight copolymers with Mn values of 3800 and 800 g mol−1 for BHET
1 and heating for 2 hours at 270 °C under vacuum, resulting in PET/PDDE copolymers as shown in reaction 2 in Fig. 5. Analysis by IR shows 2 carbonyl signals in different ratios (see Fig. S26). GPC analysis indicated relatively low molecular weight copolymers with Mn values of 3800 and 800 g mol−1 for BHET![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DDE ratios of 10
DDE ratios of 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and 5
1 and 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, respectively, with broad dispersity Đ of 5.5 and 10, respectively (see Fig. S60 and S61).
1, respectively, with broad dispersity Đ of 5.5 and 10, respectively (see Fig. S60 and S61).
DSC analysis of the various copolymers has shown that the introduction of 10 wt% DDE in PET leads to an increase in Tg from 74 °C (for PET) to 78 °C and the crystallisation temperature Tc increases from 107 to 129 °C, together with a lowering of the melting point from 257 to 238 °C (Fig. S28). Attempts to analyse BHET/DDE (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) copolymers by DSC gave mixed results suggesting a possible mixture of PET and PDDE copolymers and homopolymers, caused by the inhomogeneity of the highly viscous polymerisation reaction. High temperature and vacuum conditions cause DDE to sublime quite readily during the polymerisation. To avoid these problems, we prepared bis(hydroxyethyl) diketo ester 5 (BHEDE) via transesterification of DDE (4) with ethylene glycol using a combination of Sb2O3 and Ca(OAc)2 at 200 °C (Fig. 3).
1) copolymers by DSC gave mixed results suggesting a possible mixture of PET and PDDE copolymers and homopolymers, caused by the inhomogeneity of the highly viscous polymerisation reaction. High temperature and vacuum conditions cause DDE to sublime quite readily during the polymerisation. To avoid these problems, we prepared bis(hydroxyethyl) diketo ester 5 (BHEDE) via transesterification of DDE (4) with ethylene glycol using a combination of Sb2O3 and Ca(OAc)2 at 200 °C (Fig. 3).
Melt polymerisation reactions of BHET/BHEDE mixtures were carried out at 270 °C under vacuum resulting in the formation of PET/PDDE copolymers. Melt copolymerisations under vacuum, using a gastight mechanical stirrer, typically resulted in polymers with relatively low molecular weight, due to the inefficient removal of ethylene glycol. In order to obtain higher molecular weight polymers, combined with the difficulties in handling hot molten polymers at 270 °C, we changed to reactive extrusion for further polymerisation studies.
Polymer processing
In an attempt to obtain higher molecular weight copolymers, the incorporation of different amounts of diketo esters DDE (4) and BHEDE (5) into PET was investigated via reactive extrusion in a micro-compounder operating in recirculation mode at 275 °C under N2 (see SI). Commercial PET was charged into the extruder, followed by the addition of different amounts of the additive after 200 s (see Fig. 6).The change in torque force was monitored to indicate changes in viscosity of the polymer melt.35 A constant torque force is normally achieved within 2–3 minutes after the introduction of PET. Initial control experiments involved the addition of different amounts of the terephthalate esters DMT and BHET to PET at 275 °C, which revealed a rapid decrease in torque force due to a lowering of the viscosity (see Fig. 7). This suggests a very fast transesterification process under these conditions. No transesterification catalyst was needed, as these reactions are known to be fast at this temperature due to the presence of residual protons from moisture and hydroxyl end-groups in PET.36 Experiments with and without additional transesterification catalyst (Sb2O3/Ca(OAc)2) gave identical results. The release of methanol (from DMT) and ethylene glycol (from BHET) can lead to chain scission reactions, causing a reduction of the polymer molecular weight and thereby a lowering of the viscosity. As was observed during melt polymerisation reactions, transesterifications under these conditions without the continuous removal of methanol or EG under vacuum, results inevitably in a lowering of the molecular weight of the PET.
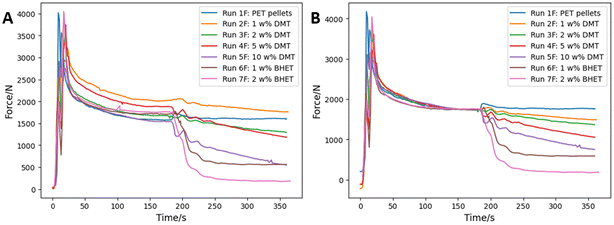 | ||
| Fig. 7 Extruder torque force versus time during PET recirculation at 275 °C and addition of DMT and BHET additives after 200 s (A). Normalised (B). | ||
It is important to note that small amounts of additive can have large effects without efficient removal of the alcohol. For example, 1 wt% DDE (M = 326 g mol−1) relative to PET (Mn ∼30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 g mol−1) is approximately the equivalent amount in moles and can therefore in theory reduce the molecular weight of PET by half, if methanol is not removed. The effect of changes in molecular weight and dispersity upon the addition of various additives are summarised in Fig. 9. The addition of 10 wt% DMT (6.70 mmol) to PET results in a relatively small change in Mn from 30
000 g mol−1) is approximately the equivalent amount in moles and can therefore in theory reduce the molecular weight of PET by half, if methanol is not removed. The effect of changes in molecular weight and dispersity upon the addition of various additives are summarised in Fig. 9. The addition of 10 wt% DMT (6.70 mmol) to PET results in a relatively small change in Mn from 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200 to 22
200 to 22![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 150 g mol−1. In this case, evaporation of some DMT due to its volatility (bp. 288 °C) under the processing conditions (275 °C) is most likely responsible for this small change.
150 g mol−1. In this case, evaporation of some DMT due to its volatility (bp. 288 °C) under the processing conditions (275 °C) is most likely responsible for this small change.
The extrusion of PET after the addition of DDE and BHEDE led to similar changes in torque force as seen with DMT and BHET (Fig. 8). The addition of 16.8 wt% (6.70 mmol) of DDE (4) to PET results in a significant lowering of Mn from 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200 to 9200. In this case, an additional melting peak is observed at 200 °C in the DSC analysis of the copolymer (see below), indicating some unreacted DDE at this high additive loading. The addition of BHEDE (5) and BHET was limited to 4.0 and 2.6 wt%, respectively (equivalent to 1.34 mmol) as higher loadings lead to increased brittleness of the copolymer product. These additive loadings resulted in a reduction of Mn by approximately half due to alcoholysis. Dispersity indexes (Đ) increased slightly with the introduction of DDE (4) or BHEDE (5) but remained in all cases under 3.
200 to 9200. In this case, an additional melting peak is observed at 200 °C in the DSC analysis of the copolymer (see below), indicating some unreacted DDE at this high additive loading. The addition of BHEDE (5) and BHET was limited to 4.0 and 2.6 wt%, respectively (equivalent to 1.34 mmol) as higher loadings lead to increased brittleness of the copolymer product. These additive loadings resulted in a reduction of Mn by approximately half due to alcoholysis. Dispersity indexes (Đ) increased slightly with the introduction of DDE (4) or BHEDE (5) but remained in all cases under 3.
 | ||
| Fig. 8 Extruder torque force versus time during PET recirculation at 275 °C and addition of DDE and BHEDE additives after 200 s (A). Normalised (B). | ||
The possibility of direct transesterification between esters under the processing conditions was considered and investigated by combining equimolar amounts of the molecular esters DMT and diethyl phthalate and heating the mixture without solvent at 265 °C for 20 minutes. Analysis of the reaction product by 1H NMR showed no methyl vs. ethyl exchange under these conditions. Therefore, a key difference between DDE/DMT versus BHEDE/BHET is that the methyl esters DDE (4) and DMT react predominantly with hydroxyethyl chain ends in PET. The methanol released in this case causes some chain scission due to methanolysis. In contrast, the hydroxyethyl esters BHEDE (5) and BHET can react directly with ester linkages along the PET chain, resulting in direct chain scission. To obtain high molecular weight PET-based copolymers, it is imperative that polymer processing conditions are used where polyester formation is carried out in the melt at 275 °C with continuous removal of ethylene glycol under vacuum.
Thermal analysis of the PET/DDE and PET/BHEDE copolymers have been performed using DSC analysis. The copolymers show very similar thermal profiles compared to PET, with Tg values of ∼80 °C, Tc ∼140–145 °C and Tm ∼245–248 °C. Tg and Tc values become increasingly difficult to detect with increased amounts of DDE additive. In the case of the PET/DDE copolymer with the highest loading of DDE of 16.8 wt%, an additional melting peak is observed at around 200 °C, attributed to unreacted DDE (m.p. 197–198 °C),37 which indicates, as suggested earlier, that DDE reacts predominantly with hydroxyethyl chain ends.
Accelerated weathering
A selection of PET copolymers with the highest additive loadings, prepared as described in the previous section from PET and the various additives listed in Tables S3 and S4 have been subjected to accelerated weathering conditions using a QUV chamber equipped with UV-B (313 nm) lamps. Copolymer samples were extruded and pressed into plates of 1 mm thickness and were analysed by GPC analysis before and after 14 days of weathering. The weathering cycle used involved irradiation at 0.71 ± 0.02 W m−2 for 8 hours at 60 °C followed by 4 hours of darkness at 50 ± 2 °C. There was no specific control of the relative humidity (generally 50% at 20 °C). Results or copolymers have been compared with pure PET, which was processed under the same conditions.From Fig. 9, it can be seen that in all cases Mn values of the copolymers have decreased after exposure to the weathering conditions. The general increase in dispersity indicates random chain scission processes. Compared to PET, the changes in Mn for the copolymers is generally less pronounced. All copolymers have lower initial molecular weights than PET due to chain scission reactions caused by alcoholysis during the reactive extrusion process. The reduction in molecular weight observed in all cases is likely the result of hydrolysis of the ester linkages during the weathering conditions, as seen by others.38 It is possible that the introduction of hydrophilic dicarbonyl units affects the rate of diffusion of water into the polymer and increases the rate of hydrolysis. Chain scission of the dicarbonyl units induced by UV-light irradiation is also possible, but this is difficult to confirm. As shown in Fig. 4, the photolytic cleavage of diketone units in DDE (4) and BHEDE (5) in solution under irradiation at 365 nm and in the presence of oxygen leads to terminal terephthalate end-groups, which will be indistinguishable from those generated via hydrolysis of the ester linkages. Exposure of DDE (4) and BHEDE (5) to the same weathering conditions as the polymers in Fig. 9, using irradiation at 313 nm UV-B light, did not show any changes. It is possible that longer UV exposure times or different wavelengths could show further chain scission.
 | ||
| Fig. 9 Molecular weight analysis (GPC) of PET copolymers before and after weathering with various additives (with wt% loadings). Samples obtained after 14 days QUV weathering are marked with *. | ||
In conclusion, we have shown that carbonyl units can be introduced into the ethylene glycol and the terephthalic acid moiety of PET. The use of 1,3-dihydroxyacetone allowed the synthesis of a polyester model compound 2, which showed degradation under oxidative and photolytic conditions, but this was mainly due to hydrolysis of the ester linkages. More importantly, the 1,3-dihydroxyacetone linker proved to be thermally unstable which prevented polyester formation.
The C–C bond strength in dicarbonyl units is relatively weak and these units are prone to photolytic cleavage. The introduction of dicarbonyl units into polyesters was achieved by synthesising the diesters DDE (4) and BHEDE (5). These diesters can be polymerised to generate the polyester PDDE or they can be incorporated into PET through copolymerisation with BHET to generate PET/PDDE copolymers. Molecular weights that can be achieved under these conditions were rather low due to inefficient removal of methanol or ethylene glycol. Alternatively, these copolymers can be prepared from PET through transesterification using reactive extrusion, which led to the preparation of a range of copolymers with different loadings of the dicarbonyl unit. Higher molecular weights are achievable under these conditions, but still lower than the original PET. Degradation of the PET copolymers was investigated using accelerated weathering conditions with controlled temperature, humidity and UV irradiation. A lowering of the molecular weight was observed in all cases, most likely due to hydrolysis of the ester linkages. Oxidative photolytic cleavage of the dicarbonyl units is also possible, but difficult to confirm due to the same carboxylic acid end-groups being generated. Further studies are underway to establish the nature of the degradation of PET in the natural environment.
Conflicts of interest
There are no conflicts of interest to declare.Data availability
All research data is available in the supplementary information (SI). Supplementary information: materials, synthesis and characterisation. See DOI: https://doi.org/10.1039/d5py00992h.Acknowledgements
The authors would like to thank Innovate UK for funding (Grant 104608). We thank Prof. Koon-Yang Lee for help with polymer extrusions. Dr Keith Whiston and Dr Clive Hamilton (Koch Technology Solutions) are thanked for helpful discussions.References
- E. Gubbels, T. Heitz, M. Yamamoto, V. Chilekar, S. Zarbakhsh, M. Gepraegs, H. Köpnick, M. Schmidt, W. Brügging, J. Rüter and W. Kaminsky, in Ullmann's Encyclopedia of Industrial Chemistry, 2018, pp. 1–30, DOI:10.1002/14356007.a21_227.pub2.
- R. Geyer, J. R. Jambeck and K. L. Law, Sci. Adv., 2017, 3, 768–771 CrossRef PubMed.
- T. Sang, C. J. Wallis, G. Hill and G. J. P. Britovsek, Eur. Polym. J., 2020, 136, 109873 CrossRef CAS.
- A. Chamas, H. Moon, J. Zheng, Y. Qiu, T. Tabassum, J. H. Jang, M. Abu-Omar, S. L. Scott and S. Suh, ACS Sustainable Chem. Eng., 2020, 8, 3494–3511 CrossRef CAS.
- A. Ammala, S. Bateman, K. Dean, E. Petinakis, P. Sangwan, S. Wong, Q. Yuan, L. Yu, C. Patrick and K. H. Leong, Prog. Polym. Sci., 2011, 36, 1015–1049 CrossRef CAS.
- G. H. Hartley and J. Guillet, Macromolecules, 1968, 1, 165–170 CrossRef CAS.
- M. Edge, M. Hayes, M. Mohammadian, N. S. Allen, T. S. Jewitt, K. Brems and K. Jones, Polym. Degrad. Stab., 1991, 32, 131–153 CrossRef CAS.
- T. Grossetête, A. Rivaton, J. L. Gardette, C. E. Hoyle, M. Ziemer, D. R. Fagerburg and H. Clauberg, Polymer, 2000, 41, 3541–3554 CrossRef.
- T. O. Morgen, M. Baur, I. Gottker-Schnetmann and S. Mecking, Nat. Commun., 2020, 11, 3693 CrossRef CAS.
- A. L. Andrady, J. Appl. Polym. Sci., 2003, 39, 363–370 CrossRef.
- A. W. White and J. R. Zoeller, Photodegradable Polyesters, Eastman, US Pat. 5395692, 1995.
- D. G. Barrett and M. N. Yousaf, Biomacromolecules, 2008, 9, 2029–2035 CrossRef CAS.
- M. Baur, N. K. Mast, J. P. Brahm, R. Habe, T. O. Morgen and S. Mecking, Angew. Chem., Int. Ed., 2023, 62, e202310990 CrossRef CAS.
- M. Baur, F. Lin, T. O. Morgen, L. Odenwald and S. Mecking, Science, 2021, 374, 604–607 CrossRef CAS PubMed.
- S. Tang, F. W. Seidel and K. Nozaki, Angew. Chem., 2021, 133, 26710–26714 CrossRef.
- L. Chen, K. G. Malollari, A. Uliana, D. Sanchez, P. B. Messersmith and J. F. Hartwig, Chemistry, 2021, 7, 137–145 CrossRef CAS.
- S. I. Kuzina, A. P. Pivovarov, A. I. Mikhailov and G. P. Belov, Eur. Polym. J., 2000, 36, 975–980 CrossRef CAS.
- E. Drent and P. H. Budzelaar, Chem. Rev., 1996, 96, 663–682 CrossRef CAS PubMed.
- C. Bianchini and A. Meli, Coord. Chem. Rev., 2002, 225, 35–66 CrossRef CAS.
- POKETONE™, https://www.poketone.com.
- J. A. Fuentes, S. M. Smith, M. T. Scharbert, I. Carpenter, D. B. Cordes, A. M. Slawin and M. L. Clarke, Chemistry, 2015, 21, 10851–10860 CrossRef CAS.
- J. N. Korley, S. Yazdi, K. McHugh, J. Kirk, J. Anderson and D. Putnam, Biomaterials, 2016, 98, 41–52 CrossRef CAS PubMed.
- P. N. Zawaneh, A. M. Doody, A. N. Zelikin and D. Putnam, Biomacromolecules, 2006, 7, 3245–3251 CrossRef CAS PubMed.
- K. Bowden, Adv. Phys. Org. Chem., 1993, 28, 171–206 CrossRef CAS.
- Y. Maeda, T. Maeda, K. Yamaguchi, S. Kubota, A. Nakayama, N. Kawasaki, N. Yamamoto and S. Aiba, J. Polym. Sci., Part A:Polym. Chem., 2000, 38, 4478–4489 CrossRef CAS.
- D. L. Bunbury and C. T. Wang, Can. J. Chem., 1968, 46, 1473–1479 Search PubMed.
- G. E. Gream, J. C. Paice and C. C. R. Ramsay, Aust. J. Chem., 1969, 22, 1229–1247 CrossRef CAS.
- W. M. Nau and J. Scaiano, J. Phys. Chem., 1996, 100, 11360–11367 CrossRef CAS.
- O. Winkelmann, C. Nather and U. Luning, Org. Biomol. Chem., 2009, 7, 553–556 RSC.
- V. Medabalmi and K. Ramanujam, J. Electrochem. Soc., 2017, 164, A1720–A1725 CrossRef CAS.
- F. D. Lewis, R. T. Lauterbach, H. G. Heine, W. Hartmann and H. Rudolph, J. Am. Chem. Soc., 1975, 97, 1519–1525 CrossRef CAS.
- N. Kawabe, K. Okada and M. Ohno, J. Org. Chem., 1972, 37, 4210–4211 Search PubMed.
- M. G. Vinogradov and G. I. Nikishin, Russ. Chem. Rev., 1971, 40, 916–932 CrossRef.
- Y. Yasujima, T. Habara and Y. Katagiri, Fluorescent Polyester Fibres, Toray Industries, JP53045394, 1978.
- C. Bascucci, I. Duretek, S. Lehner, C. Holzer, S. Gaan, R. Hufenus and A. Gooneie, Polym. Degrad. Stab., 2022, 195, 109783 CrossRef CAS.
- A. M. Kenwright, S. K. Peace, R. W. Richards, A. Bunn and W. A. MacDonald, Polymer, 1999, 40, 5851–5856 Search PubMed.
- M. H. Petersen, S. A. Gevorgyan and F. C. Krebs, Macromolecules, 2008, 41, 8986–8994 CrossRef CAS.
- S. Rostampour, R. Cook, S. S. Jhang, Y. Li, C. Fan and L. P. Sung, Polymers, 2024, 16, 2249 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |