Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Evaluating the role of lactose anomeric composition in tablet disintegration and dissolution: a hidden variable in pharmaceutical formulation
Thamer
Alzoubi
ab,
Gary P.
Martin
a,
Kendal
Pitt
c,
Hetvi
Triboandas
c,
Fangyi
Chen
a,
Abhimata
Paramanandana
a and
Paul G.
Royall
 *a
*a
aInstitute of Pharmaceutical Science, King's College London, London SE1 9NH, UK. E-mail: paul.royall@kcl.ac.uk
bDepartment of Pharmaceutical Sciences, College of Health Sciences, PAAET (Public Authority for Applied Education and Training), Kuwait
cLeicester School of Pharmacy, De Montfort University, Leicester LE1 9BH, UK
First published on 5th January 2026
Abstract
Lactose, a chiral excipient, is widely used in the pharmaceutical industry as a diluent because it is safe and has developable physical properties. However, there are stability issues; β lactose powder stored at 40 °C/75% RH will epimerize from the β to the α chiral form within 7 days. The influence of lactose chiral composition on medicine stability is poorly understood but is likely to be valuable when ensuring the safety and effectiveness of the finished pharmaceutical product (FPP). Therefore, the aim of this study was to investigate the effect of the anomeric composition of lactose on tablet properties. Tablets with a higher α lactose content (79.5/20.5 α/β % w/w (α79%)) demonstrated faster disintegration times when compared to tablets formulated with a higher β content (13.5/86.5 α/β % w/w (α13%)), i.e., 27 s compared to 220 s. Differences in tablet hardness (p ≤ 0.05) were also observed, with tablets containing a higher α composition exhibiting higher hardness (i.e., 207 N as opposed to 170 N under a 40 kN compression force). The release of acetylsalicylic acid was found to be faster from tablets that were formulated with ‘aged’ or epimerised lactose (α79%) compared to those produced using β lactose, as received (α13%), the respective T90 values being 60 and 91 min. To conclude, it is apparent that the stereoisomeric effects of lactose excipients on tablets are evident, and therefore, the measurement of anomeric content is advised prior to tablet manufacture.
1. Introduction
Ensuring the safety and quality attributes of finished pharmaceutical products is essential for maintaining therapeutic performance throughout their shelf life. While the influence of active pharmaceutical ingredients on product stability is well recognized, the role of excipients remains comparatively underexplored. Excipients are often considered inert; their physicochemical properties are selected to optimise the dosage form, for example, tablet hardness, disintegration, and dissolution, thereby influencing drug bioavailability.1,2 Regulatory guidelines acknowledge that excipient characteristics impact formulation performance, yet stereochemical variability in chiral excipients is rarely addressed.3,4Lactose is one of the most widely used excipients in solid dosage forms, primarily as a diluent.5 This excipient is chiral and has two stereoisomers. It exists as two anomeric forms, α and β, which differ in solubility, crystallinity, and propensity to form hydrates.6 These anomers can interconvert under certain manufacturing and storage conditions, such as elevated temperature and humidity, leading to changes in the α/β ratio.7,8 For example, anhydrous β-lactose stored at 40 °C and 75% relative humidity undergoes epimerization to the α form within seven days, with a reported half-life of approximately 32 days.9 Despite the prevalence of lactose in pharmaceutical formulations, the implications of its anomeric composition for tablet performance have received little attention. Current compendial standards do not specify limits for anomeric content, and manufacturers typically do not quantify this parameter, even though variability between batches is common.10 For instance, lactose powders labelled as α-lactose monohydrate may contain up to 6% w/w β-anomer, while β-lactose samples may include up to 5% w/w α-anomer.9–12
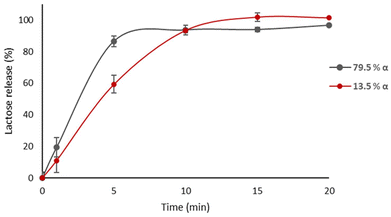
The subtlety of this stereochemical effect represents an overlooked source of variability in formulation performance. Previous studies have compared tablets composed of pure α or β lactose, but systematic evaluation across a range of anomeric ratios is lacking.8,13–16 This study addresses this gap by investigating the impact of lactose anomeric composition on key tablet properties, including disintegration, hardness, and drug release. Tablets formulated with a higher α-lactose content (79.5% α/20.5% β w/w (α79%)) demonstrated markedly faster disintegration times (27 s) compared to those with a higher β-lactose content (13.5% α/86.5% β w/w (α13%)), which disintegrated in 220 s. Differences in tablet hardness were also observed (207 N versus 170 N at 40 kN compression force), and acetylsalicylic acid release was faster from tablets containing epimerized lactose (T90 = 60 min) compared to those formulated with β-lactose as received (T90 = 91 min), Fig. 1.
The aim of this paper was to evaluate the role of lactose anomeric composition in tablet disintegration and dissolution, highlighting the complexity of common excipients used in pharmaceutical formulations. Lactose powders with defined α/β ratios were formulated into tablets and characterized using thermal and non-thermal analytical techniques, followed by compendial physical testing. The findings highlight the importance of monitoring anomeric composition as a critical quality attribute in pharmaceutical manufacturing.
2. Methods
2.1 Sample preparation
Lactose powder samples with four different ratios of α-anomer contents were used as specified in Table 1. Lactose samples II and III were produced as described previously,9 and powder batches of anhydrous β lactose (α13) (ACROS Organics (80% β and 20% α labelled content), Lot A0416062, Germany), α lactose monohydrate, α·LMH (Sigma Aldrich (≥99% α labelled content, L3625, Lot SLBX8370, USA)) and microcrystalline cellulose (MCC) (Alfa Aesar, UK, Lot 10227760) were used as received. Each lactose batch (I, II, III and IV) was placed inside a 50 mL plastic container (2.5 cm diameter and 6.5 cm height) along with 4% w/w MCC. The container was filled to less than half the volume, and the contents were mixed for 15 min using a Turbula® mixer (Glen Creston, Stanmore, UK). Each container contained 3.3 g of total powder weight, which was sufficient to produce 10 tablets + a 10% overage, to allow for possible powder loss during tablet production.| Lactose powder | α-Anomer content (% w/w) | β-Aanomer content (% w/w) | Designation | ±SD (%) | Applied modifications |
|---|---|---|---|---|---|
| I (anhydrous β-lactose) | 13.5 | 86.5 | α13% | 0.4 | As received |
| II | 50.2 | 49.8 | α50% | 0.1 | 35 days storage (40 °C/75% RH) |
| III | 79.5 | 20.5 | α79% | 0.3 | 60 days storage (40 °C/75% RH) |
| IV (α-lactose monohydrate) | 96.6 | 3.4 | α·LMH | 0.7 | As received |
2.2 Tablet production
Tablets were prepared individually using a manual hydraulic press (PerkinElmer, UK) by adding 300 mg (weighed using a Sartorius electronic balance to the nearest 0.001 mg) of each lactose/MCC mixture into a 10 mm (diameter) die punch which was assembled using alignment plates. Each tablet was flat in shape with a 10 mm diameter and 3 mm thickness (for further visual description, refer to SI 1). Four different batches of tablets were produced containing different anomeric content as specified in Table 1 under 3 different compression forces. Apart from the anomeric differences, all tablets were manufactured under the same conditions. For each batch of tablets, the compression forces used were 10, 20 and 40 kilo Newtons (kN). Upon reaching the designated force, the pressure valve was opened, minimizing dwell time. The die and punches were lightly lubricated with magnesium stearate, after the production of every 10 tablets using a small application brush.2.3 Tablets for ASA dissolution
Tablets were produced as described in section 2.2 but comprising of a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 weight ratio of aspirin (ASA) (Lot A0334368, Acros Organics) and lactose powders (α·LMH as received, α13, α50 and α79% α anomer) combined with 4% w/w MCC (i.e., 12 mg per tablet). Each tablet contained 144 mg of ASA, and every tablet weighed a total of 300 mg.
1 weight ratio of aspirin (ASA) (Lot A0334368, Acros Organics) and lactose powders (α·LMH as received, α13, α50 and α79% α anomer) combined with 4% w/w MCC (i.e., 12 mg per tablet). Each tablet contained 144 mg of ASA, and every tablet weighed a total of 300 mg.
2.4 Appearance and dimensions
Each tablet produced was visually examined for unusual characteristics. These included changes in colour, shape and chipping of tablets. The average dimensions (diameter and height) of 3 randomly selected tablets taken from every batch were recorded using a digital calliper (Duratool, Taiwan). No more than 10% deviation was deemed acceptable as per the previous study criteria.172.5 Weight uniformity
Each tablet was carefully weighed using an electronic balance (to the nearest 0.1 mg) prior to testing. The mean weight (±SD) for each batch was calculated (Table S2) with no more than 5% deviation from the average weight being acceptable for any tablet.182.6 Thermal analysis
2.7 Nuclear magnetic resonance
Sample preparation and the method for measurement of anomer composition were described previously.9 ASA + lactose tablets (n = 6) were dissolved in DMSO + 0.03% TMS (tetramethylsilane) % v/v (VWR Chemicals, USA, Lot: 210020 Batch: 0121D) and analysed 24 h after preparation using 5 mm Wilmad NMR sample tubes (Batch: 112619A). Each sample was analysed in triplicate (n = 3).2.8 Hardness testing
Hardness testing was conducted in accordance with the methods detailed in the BP (2001)19 and USP (2012).20 10 individual tablets were weighed to ensure uniformity of weight, and each, in turn, was placed in the compartment of a C50 hardness tester (Engineering Systems, Nottingham, UK). Between measurements, the jaws of the instrument were brushed to remove any residual powder or fragments. The weight (kg, converted to Newtons) causing each tablet to break was recorded. This procedure was repeated for each of the 10 tablets (n = 10) that had been prepared using 3 different compression forces (10, 20 and 40 kN) and for tablets containing lactose with different anomeric content (Table 1). The mean, minimum and maximum results for each test batch were recorded.2.9 Disintegration
Disintegration testing was carried out (DTG 4000, Copley Scientific, Nottingham, UK) in accordance with BP (2021) and USP (2019) testing procedures.21 For each batch of tablets, 6 separate tablets were carefully placed inside individual open-ended tubes (height: 75 mm, diameter: 25 mm). The tubes were enclosed within a water bath containing 800 mL of purified water at 37 °C, which served as the disintegration medium. The vertical frequency of the tubes was 30 cycles per min, and the disintegration time for each tablet was recorded as the time at which the last fragment of the tablet was lost through the mesh at the base of each tube. No plastic disks were employed in any of the tubes, in accordance with the specifications of the compendial monograph.212.10 Scanning electron microscopy
A self-adhesive carbon disc was applied to an aluminium stub. Lactose-only tablets were broken in half manually, placed on a carbon disc and coated with a 5 nm gold layer. Samples were examined using Phenom ProX software, version 4.6.4 (Thermo Scientific, USA) at a voltage of 10 kV and applying a full backscattered electron detector.2.11 Dissolution, UV and RI analyses
2.12 Similarity dissolution factor (f2)
A model-independent approach using the similarity factor (f2) was implemented, also known as the fit factor method,22 to verify the changes in dissolution profiles. This approach is recommended by the USP and FDA19,23 and is summarised in eqn (1) as follows: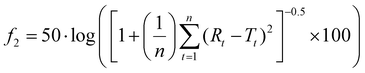 | (1) |
2.13 True density and porosity
The true density (g cm−3) and pore volume (cm3 g−1) of the powder were obtained using a helium pycnometer (Micromeritics, US). Prior to analysis, the chamber O-ring was gently greased using Dow Corning® high vacuum grease (Micromeritics Instrument Corporation, USA) to prevent gas leakage. Each powder sample was weighed (1.6 ± 0.1 g) inside the 3.5 cm3 chamber insert (AccuPyc™ Multi-Volume Calibration Standard, Micromeritics, US). The samples were subjected to 10 helium gas purges followed by 10 gas pressure cycles (19.5 psig cycle fill pressure). True density and pore volume were calculated by taking the average of the 10 cycles. Each sample was analysed in triplicate (AccuPyc II, Version 4.0 software, Micromeritics, USA).2.14 Statistical analysis
To determine statistical differences in the data, un-paired Student's t-tests were applied using Microsoft Excel 2016®. The level of significance was determined, and a significant difference was accepted if p was ≤0.05.3. Results
The results section begins with the properties of the prepared tablets and lactose powders to evidence that they were suitable to explore the complexity of lactose's anomeric composition and its effect on disintegration and dissolution. A minimum of 4% w/w microcrystalline cellulose (MCC) was mixed into all lactose powders before tableting to act as a binder and prevent lamination, which had been observed in tablets containing high levels of β-lactose without MCC25,26 (SI S1).3.1 Anomeric composition and tablet properties: weight uniformity, thermal behaviour and hardness
The tablets produced complied with compendial BP specifications for weight uniformity, remaining within the acceptable range of 95–105%, with the exception of two that were only slightly outside this limit. All tablets were entirely within the specified range for dimensions (SI S2). H1NMR and DSC analyses recorded no alteration in the anomeric content of lactose following tableting, indicating that tablet compression did not influence the anomeric composition of the original lactose powders (SI S4, Fig. 2 and Table 2). No changes in melting points were observed; however, the onset temperature for hydrate release from tablets containing α·LMH was significantly lower (p ≤ 0.05) than that of the uncompressed powder. Water loss from α·LMH occurred at 143.8 ± 0.2 °C in the powder and 131.2 ± 1.6 °C in the tablets (highlighted in red in Fig. 2, and supported by the TGA results, SI S3).| Sample | Loss of hydrate (°C) | Enthalpy of hydrate loss (J g−1) | Melting point (°C) | Enthalpy of melting (J g−1) | αAnomer content (%) |
|---|---|---|---|---|---|
| * Indicates a statistical difference (p ≤ 0.05) between the powder and tablet. | |||||
| β-Lactose powder | — | — | 235.1 ± 0.3 | 122 ± 8.75 | 13.5 ± 0.4 |
| β-Lactose tablet | — | — | 233 ± 0.6 | 121.3 ± 5 | 14.5 ± 0.7 |
| α-LMH powder | 143.8 ± 0.2* | 150.24 ± 12.2 | 213.7 ± 1.5 | 77.02 ± 3 | 96.6 ± 1.5 |
| α-LMH tablet | 131.2 ± 1.6* | 139.9 ± 11.6 | 211.7 ± 0.9 | 82.6 ± 3.2 | 97.1 ± 0.1 |
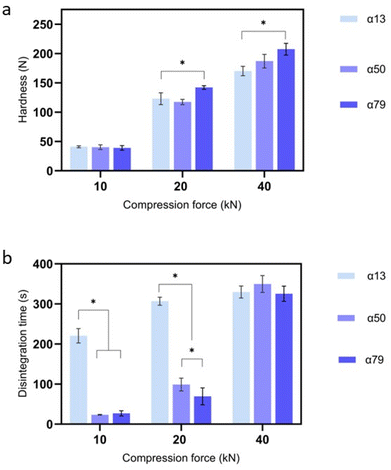
Hardness measurements further highlighted the complex influence of anomeric content on the properties of tablets containing lactose (Fig. 3a). For example, at a compression force of 20 kN, the hardness of tablets comprising β lactose (as received α13%) and α·LMH (as received α97%) was 123 ± 21 N and 78.8 ± 10.4 N, respectively (p ≤ 0.05) (SI S5). Differences in hardness were also seen for tablets prepared using β lactose powders that had been stored for different lengths of time at 40 °C/75% RH (Table 1). These batches of lactose powders contained different ratios of the α and β anomers and allowed investigation of tablets which contained 13% α anomer, 50% α anomer and 79% α anomer as determined by H1NMR (Table 1). At a compression force of 20 kN, the hardness of the tablets increased from 122.9 ± 21 to 142.1 ± 6 N, as the α anomer content increased from 13% to 79% (p ≤ 0.05). At 40 kN compression force, α13% and α79% exhibited hardness properties of 170.2 ± 16 and 207.4 ± 19 N, respectively (p ≤ 0.05). Lactose powders, α·LMH as received (α97%), and α·LMH stored at 40 °C/75% RH for 35 days (α98%) and 60 days (α98%), respectively, were also used to produce tablets. There were no significant differences between the hardness of the tablets produced using any of these lactose samples (SI S5).
3.2 Disintegration
Higher compression forces produced harder tablets, which prolonged disintegration time. However, among tablets compressed at the same force, variations in anomeric composition influenced disintegration time (Fig. 3b). For example, tablets produced with β lactose (α13%) were found to disintegrate in 220.4 ± 18 s compared to tablets containing α50% and α79%, where both disintegrated markedly faster (p ≤ 0.05) in 23.5 ± 1 and 27.8 ± 6.4 s, respectively (Table 3). In contrast, when comparing the hardness properties of these three batches of tablets (which were identical other than in the different anomer contents of lactose), no change in hardness was observed (Fig. 3(a), at a compression force of 10 kN). However, α79% containing tablets produced at 20 kN demonstrated higher crushing strength compared to β lactose (α13%) tablets but considerably shorter disintegration times, and therefore, tablet hardness was not directly correlated with disintegration time, when compression forces of 10 and 20 kN were applied.| Compression force (kN) | α13% disintegration time (s) | α50% disintegration time (s) | α79% disintegration time (s) |
|---|---|---|---|
| 10 | 220 ± 18 | 23 ± 1* | 27 ± 6.4* |
| 20 | 306 ± 10 | 98 ± 16* | 69 ± 21*,+ |
| 40 | 329 ± 15 | 349 ± 36 | 325 ± 19 |
| Compression force (kN) | α·LMH as received (α97%), disintegration time (s) | α·LMH – 35 days (α98%), disintegration time (s) | α·LMH – 60 days (α98%), disintegration time (s) |
|---|---|---|---|
| *Statistical differences (p ≤ 0.05) observed between α13% and tablets containing α50% and α79% lactose. +Statistical difference between all three samples. | |||
| 10 | 25 ± 4 | 23 ± 3 | 21 ± 3 |
| 20 | 21 ± 2 | 19 ± 3 | 20 ± 5 |
| 40 | 41 ± 9 | 32 ± 11 | 46 ± 18 |
When a 40 kN compression force was applied, there were no differences in disintegration times between the batches of tablets that contained lactose of different anomeric compositions. A similar investigation was conducted using tablets of α·LMH stored under conditions identical to the former lactose tablet batches. Table 3 shows the time taken for every batch to disintegrate. No differences (p > 0.05) were observed in the disintegration times of tablets containing α·LMH, after the tablets were stored under higher humidity.
3.3 Dissolution
| Tablet matrix | T 25 (min) | T 50 (min) | T 90 (min) |
|---|---|---|---|
| * Indicates statistical difference between the starting material of α13 + ASA and tablets comprising of epimerised lactose. α·LMH samples were used as control measures to compare the storage effect on α·LMH. + Indicates a statistically significant difference (p ≤ 0.05) between tablets produced using α13%, α50%, α79% with α·LMH tablets (as received and stored α97%). | |||
| α13% + ASA | 29 | 46.5 | 91 |
| α50% + ASA | 20* | 36* | 80* |
| α79% + ASA | 12.5* | 23* | 60* |
| α·LMH (as received α97%) + ASA | 7+ | 16+ | 60+ |
| α·LMH (stored for 60 days at 40 °C/75% RH 98% α) + ASA | 7.5+ | 17.5+ | 67+ |
3.4 Effect of tabletting and ASA incorporation on anomeric content
The hardness and disintegration tests conducted on the tablets containing ASA indicated that there were significant differences between the batches, which may be attributable to differences in the anomeric ratio of the lactose content. Initially, it was necessary to investigate whether tabletting lactose with ASA had any effect on the anomer content of lactose. H1NMR was conducted for a 24 h post-tablet production period. The NMR analysis of the tablets and pure ASA shows no effect of the API on the chemical stability of lactose (p > 0.05), as depicted in Fig. 7. Slight changes in the signals indicated that there may have been a limited alteration in the anomeric content of lactose of the tablets and the starting feed material. However, this could have been related to the presence of ASA causing the chemical shifts, which typified the lactose anomers, to become less separated (instead of doublet NMR peaks, as seen previously).9 Nevertheless, the anomeric content (%) from the starting material and post-tableting proved to be similar, indicating chemical and anomeric stability.3.5 True density and pore volume
The pore volume observed for α50% and α79% lactose powders was 0.355 and 0.353 cm3 g−1 compared to 0.367 cm3 g−1 for the starting material, α13% (as received β-lactose), with a significant statistical difference recorded (p ≤ 0.05). This was also the case for the true density, which was 1.552 and 1.546 g cm−3 (α50% and α79%, respectively) compared to 1.589 g cm−3 for α13% (p ≤ 0.05), respectively. True density and total pore volume decreased as the α content of α lactose increased due to epimerisation, as compared to β-lactose as received.4. Discussion
This study demonstrated the role of anomeric composition on the dissolution rate of aspirin tablets formulated with lactose. The following discussion explores the complexity of this behaviour, and where possible covers potential mechanisms for how anomeric content influences tablet formation, hardness, disintegration and ultimately dissolution.The 4% w/w of MCC was added to all formulations to consistently remove the potential for lamination, which was observed in the tablets containing β anhydrous lactose (α13%). The inclusion of small amounts of binders in lactose-containing tablets is common.28–30
Tablets manufactured at a low compression force (10 kN) showed no differences in hardness properties between β lactose (as received) α13%, α50%, and α79%. The force used was insufficient to cause extensive deformation of the lactose powdered particles, whereas a higher compression force would be expected to induce greater particle deformation and further stronger contact between particles within the powder.31 This assumption is supported by the data obtained from tablets prepared at higher compression forces. For example, when the tablets were compressed using forces of 20 and 40 kN, significant differences in crushing strength were recorded (p ≤ 0.05) between α13% and α79% at both forces, mirroring previous tabletting studies.8,32 Specifically for lactose tablets, anomeric composition has been observed to influence hardness, partially attributable to cohesive energy.33 Anhydrous α lactose has been observed to have a higher cohesion between its molecules which has been argued to lead to a higher tensile strength within its tablets.34 In our study (at 20 & 40 kN), the more α lactose present, the harder the tablets (Fig. 3), implying an increase in cohesion. Thermal analysis shows that this is not anhydrous α as no dehydration of α·LMH is observed as a consequence of tableting (Fig. 2).
Tablets manufactured using α·LMH powders that were stored under identical conditions to those of the tablets produced from epimerised β lactose demonstrated no difference in hardness, suggesting that the effect is caused only by the hydrate-driven epimerisation process. This justifies the assumption that the ingress of water into the as received anhydrous β lactose powder stored at 75% RH caused a shift in anomeric content and the higher α·LMH generated as a function of time results in stronger lactose tablets. It has been speculated previously that, as the compaction force is increased, the pore size within particles is diminished, with the largest pores being reduced in size first.35,36 However, using epimerised lactose, there was no evidence from the SEM images of defined pores within the powder particles9 and pore-volume data demonstrated only a slight decrease in pore volume between as received material (β lactose) and epimerised lactose powders (SI S8). Therefore, the incorporation of water molecules within the crystal structure of lactose (α79%) may have contributed to harder tablets by promoting interparticle bonding within the lactose powders and thus increasing cohesion. This suggested mechanism is supported by the related literature.32,37
Tablets containing a higher α content due to epimerisation (α50% and α79%) disintegrated more rapidly than anhydrous β lactose tablets generated by compression forces of 10 and 20 kN. Wicking may promote disintegration,38 but a potentially more convincing explanation for the influence of epimer content on disintegration is the ‘competition for water theory’.24,39 Such a mechanism also supports the observed dissolution results. At the highest compression force, there was no influence of anomer content; at this compression, penetration of water was restricted, leading to slower disintegration. The disintegration times given in section 3.4 are comparable to the times reported in the limited number of previous studies.34,40,41 For example, Patel et al.40 found that α·LMH tablets disintegrated in 45 (s), whereas van Kamp et al.41 showed that the disintegration of α·LMH and β anhydrous tablets took 14 and 205 (s), respectively, at 20 kN (using 13 mm diameter tablets). Here, the rapid disintegration of tablets α·LMH has been attributed to their ability to take up water, leading to faster disruption of bonds between lactose molecules that hold the tablet together.42 These are primarily hydrogen bonds that are weakened because of the penetration of water, with this process being dependent on the dielectric constant of the solvent.42 Such bonds are present between the lactose particles as well as the lactose molecules. Faster disintegration rates were observed with higher α content, even with increased hardness properties (SI S9). This indicated that the mechanisms discussed earlier are responsible for the tablets’ disintegration, and not the hardness properties. Nevertheless, when extremely high compression forces were used (40 kN), large differences were apparent between tablets containing α·LMH and α79%. This complex finding could be attributed to the remaining β content present in α79% contributing to the strength of the bonds formed between powder particles at extremely high compression forces (especially at the surface), promoting less elastic deformation of the β lactose particles within the α79 tablets.43
An acidic medium was used for dissolution testing, since this is viewed by Official Compendia as simulating the typical pH of gastric fluid and is regularly used for ASA tablets.44,45 Based on the hardness and disintegration data, it was decided that tablets prepared with a compression force of 20 kN might show the best differentiation between different lactose grades incorporated as an excipient in the ASA tablets.
The release of ASA might be expected to be fastest from tablets containing the highest proportion of β lactose. For example, Xiang et al. developed pollen-shaped anhydrous lactose particles with different α/β ratios, but this was based on the composition of the feed lactose grades and not the final product.46 Particles were prepared by adding an ethanolic solution of the drug which may have also induced some epimerisation (which does not appear to have been tested for). Acetaminophen release into simulated gastric fluid was recorded as being faster for tablets with a higher β anhydrous content. This was attributed in part to the higher solubility of the β analogue compared to α lactose47,48 and to the different surface structure (e.g., porosity) of the different particles produced using differing lactose anomer ratios. Confirmation of the anomeric composition of the particles prior to dissolution is required to support this conclusion. In our study, α·LMH as received produced tablets with the fastest ASA release and quickest dissolution, followed in turn by ASA tablets containing lactose with 79%, 50% and 13% α-anomer, respectively. The anomeric composition of these samples was confirmed by NMR. Xiang et al.'s lactose particles had complex structures, and the storage conditions were not reported, which further complicates the applicability of their work to the data reported in this paper.46
The principal factors affecting API dissolution from immediate-release tablets include the particle size of the tabletted powder49 as well as tablet hardness,50 although tablet hardness does not necessarily correlate inversely with the dissolution behaviour of tablets.51,52 Data for the particle size distribution (PSD) of the lactose powders obtained from9 and used in this current study showed no significant changes in this parameter, and these compared favourably with those reported previously.53,54 For example, Sebhatu and Alderborn54 reported a particle size range of 50–100 µm and a medium particle diameter of 47 µm for crystalline lactose monohydrate. Smaller particle sizes of API usually provide a faster rate of drug release because of their larger surface area.55 However, the particle size of ASA powder used in this study was identical for the starting materials prior to the tableting process. In addition, the particle size of the epimerised lactose (α50% and α79%) in the composite powders remained broadly the same as that of the β analogue, while the dissolution rate of the tablets increased as a function of increasing α lactose content. Moreover, the hardness of tablets containing ASA in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ASA + lactose powder (ranging in α/β content) did not show significant differences (SI S10), indicating that the ASA amount had no effect on the tablet strength. It was therefore concluded that particle size and tablet hardness had little influence upon the dissolution rate: lactose anomeric content controlled dissolution.
1 ASA + lactose powder (ranging in α/β content) did not show significant differences (SI S10), indicating that the ASA amount had no effect on the tablet strength. It was therefore concluded that particle size and tablet hardness had little influence upon the dissolution rate: lactose anomeric content controlled dissolution.
A competition for water mechanism may be a route to explaining the differences in ASA release from tablets containing different amounts of α and β lactose. This starts with the concept that epimerisation may take place (from β to α) upon water interaction with the tablet in the dissolution bath, for tablets containing higher β content, causing slower disintegration and dissolution at the start of the experiment (Fig. 1).
This hypothesis is based on the competition-for-water and the blockage of pores caused by recrystallisation, a theory already proposed in the literature of other tablet systems.39,56 In the case of tablets with high β lactose content (see Table 4), in the layer of aqueous dissolution media touching the surface and within tablet pores, β lactose with its higher solubility will rapidly enter solution, nearing a saturation concentration.24,39 This β lactose will epimerise and form α lactose, producing a supersaturated solution56 and resulting in the recrystallisation of α lactose monohydrate because of its lower solubility. Thus, α lactose competes for water, forming the solid monohydrate on the tablet surface and blocking further β lactose from entering the solution. The penetration of water into the β lactose tablet is reduced compared to the α lactose monohydrate tablets, because in the case of β, the activity of water, its effective concentration is reduced at the tablet surface as the water molecules are being removed to form the monohydrate. For α lactose monohydrate tablets, more water is available at the surface for penetration and to contribute to disintegration, because the hydrate is already formed. This mechanism explains the observation of higher disintegration and dissolution times for tablets prepared from lactose powders containing a high ratio of the β anomer.
The true density and pore volume data reported in section 3.5 do not appear to contradict the competition-for-water and pore blockage mechanism and are not necessarily factors causing faster disintegration and dissolution for the tablets with more α·LMH present. The β-lactose as received (α13%) had significant but only slightly higher values for density and pore volume. All values reported here (section 3.5) broadly concurred with the literature density and pore volumes for lactose powders. For example, true density values of 1.42,57 1.55, 1.55 (for a particle size range of 20–45 µm),58 1.53 (for a particle size of 45–63 µm)58 and 1.54 g cm−3 for various lactose powders59 are documented. For comparison, values of 1.38 and 1.43 g cm−3 are typical for ASA.60 Moreover, the total pore volumes of 0.35 cm3 g−1 measured here for samples containing α50% & α79% were found to be almost identical to the values reported.61 The lack of influence of density and pore volume on disintegration and dissolution reflects the proposed mechanism, as once water-induced epimerisation occurs, surface hydrate forms, reducing any influence of the powders’ original density and pore volume.
Stereoactivity and epimerisation of APIs and possible effects on the dissolution of dosage forms have been previously addressed and investigated by a number of authors, with most reporting that enantiomeric or racemic forms influence dissolution.62–64 For example, (±)-verapamil exhibited a slightly higher dissolution rate when measuring -R compared to the -S enantiomer.65 The work reported here is unusual as the dissolution of a non-chiral API has been shown to be influenced by the chirality and the complexity of the anomeric composition of an excipient frequently included in tablet formulations.
6. Conclusion
The study fulfilled the aim by evaluating how lactose anomeric composition affects tablet disintegration and dissolution. The behaviour of this common excipient was complex, and the observations were well explained by a mechanism involving competition for water between the anomers during disintegration and dissolution.Measurement of lactose anomeric composition before pharmaceutical manufacturing is advisable. Tablets prepared with different anomeric contents showed distinctive differences in tensile strength, disintegration and lamination. In tablets containing anhydrous β lactose, higher α anomer content increased hardness, while disintegration time decreased markedly as the α fraction increased. The dissolution behaviour of tablets containing ASA differed significantly with varying α content, and higher α content produced faster drug release. The similarity factor f2 between tablets made with β lactose (α13%) and α79% lactose was 23, indicating high dissimilarity. Changes in ASA release were clearly dependent on the storage of the anhydrous β lactose powders used to manufacture the tablets, with conditions such as ≥40 °C and 75% RH producing marked differences in dissolution.
Conflicts of interest
There are no conflicts to declare.Data availability
The raw data are not currently stored in an open source – but can be made available upon request to the first author.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d4pm00256c.
Acknowledgements
The authors acknowledge the financial support for Thamer Alzoubi from the Public Authority for Applied Education and Training, Kuwait, and King's College London for their PhD thesis/program.References
- A. Berardi, S. A. Rahim, L. Bisharat and M. Cespi, Swelling of Zein Matrix Tablets Benchmarked against HPMC and Ethylcellulose: Challenging the Matrix Performance by the Addition of Co-Excipients, Pharmaceutics, 2019, 11(10), 513, DOI:10.3390/pharmaceutics11100513.
- D. S. Kopp, Stability Testing of Pharmaceutical Products in a Global Environment, Regul. Aff. J., 2000, 5, 291–294 Search PubMed.
- M. A. Darji, R. M. Lalge, S. P. Marathe, T. D. Mulay, T. Fatima, A. Alshammari, H. K. Lee, M. A. Repka and S. Narasimha Murthy, Excipient, Stability in Oral Solid Dosage Forms: A Review, AAPS PharmSciTech, 2018, 19(1), 12–26, DOI:10.1208/s12249-017-0864-4.
- EMA. Guideline on Excipients in the Dossier for Application for Marketing Authorisation of a Medicinal Product, EMEA/CHMP/QWP/396951/2006. European Medicines Agency. https://www.ema.europa.eu/en/documents/scientific-guideline/guideline-excipients-dossier-application-marketing-authorisation-medicinal-product-revision-2_en.pdf (accessed 2023-07-05).
- O. S. Malallah, B. Hammond, T. Al-Adhami, A. Buanz, A. Alqurshi, W. D. Carswell, K. M. Rahman, B. Forbes and P. G. Royall, Solid-State Epimerisation and Disproportionation of Pilocarpine HCl: Why We Need a 5-Stage Approach to Validate Melting Point Measurements for Heat-Sensitive Drugs, Int. J. Pharm., 2020, 574, 118869, DOI:10.1016/j.ijpharm.2019.118869.
- R. Jawad, A. F. Drake, C. Elleman, G. P. Martin, F. J. Warren, B. B. Perston, P. R. Ellis, M. A. Hassoun and P. G. Royall, Stability of Sugar Solutions: A Novel Study of the Epimerization Kinetics of Lactose in Water, Mol. Pharm., 2014, 11, 2224–2238, DOI:10.1021/mp400509t.
- N. Maclean, E. Walsh, M. Soundaranathan, I. Khadra, J. Mann, H. Williams and D. Markl, Exploring the Performance-Controlling Tablet Disintegration Mechanisms for Direct Compression Formulations, Int. J. Pharm., 2021, 599, 120221, DOI:10.1016/j.ijpharm.2021.120221.
- S. Ziffels and H. Steckel, Influence of Amorphous Content on Compaction Behaviour of Anhydrous α-Lactose, Int. J. Pharm., 2010, 387(1–2), 71–78, DOI:10.1016/j.ijpharm.2009.12.009.
- T. Alzoubi, G. P. Martin and P. G. Royall, A Study of Solid-State Epimerisation within Lactose Powders and Implications for Milk Derived Ingredients Stored in Simulated Tropical Environmental Zones, Food Chem., 2023, 402, 134206, DOI:10.1016/J.FOODCHEM.2022.134206.
- D. Lamešić, O. Planinšek, Z. Lavrič and I. Ilić, Spherical Agglomerates of Lactose with Enhanced Mechanical Properties, Int. J. Pharm., 2017, 516(1–2), 247–257, DOI:10.1016/j.ijpharm.2016.11.040.
- R. Jawad, C. Elleman, L. Vermeer, A. F. Drake, B. Woodhead, G. P. Martin and P. G. Royall, The Measurement of the β/α Anomer Composition within Amorphous Lactose Prepared by Spray and Freeze Drying Using a Simple 1H-NMR Method, Pharm. Res., 2012, 29, 511–524, DOI:10.1007/s11095-011-0575-6.
- M. J. Altamimi, K. Wolff, A. Nokhodchi, G. P. Martin and P. G. Royall, Variability in the α and β Anomer Content of Commercially Available Lactose, Int. J. Pharm., 2019, 555, 237–249, DOI:10.1016/j.ijpharm.2018.10.061.
- H. V. van Kamp, G. K. Bolhuis, K. D. Kussendrager and C. F. Lerk, Studies on Tableting Properties of Lactose. IV. Dissolution and Disintegration Properties of Different Types of Crystalline Lactose, Int. J. Pharm., 1986, 28(2–3), 229–238, DOI:10.1016/0378-5173(86)90249-8.
- T. Alzoubi, G. P. Martin, D. J. Barlow and P. G. Royall, Stability of α-Lactose Monohydrate: The Discovery of Dehydration Triggered Solid-State Epimerization, Int. J. Pharm., 2021, 604, 120715, DOI:10.1016/j.ijpharm.2021.120715.
- G. K. Bolhuis and K. Zuurman, Tableting Properties of Experimental and Commercially Available Lactose Granulations for Direct Compression, Drug Dev. Ind. Pharm., 1995, 21(18), 2057–2071, DOI:10.3109/03639049509065890.
- H. Vromans, A. H. De Boer, G. K. Bolhuis, C. F. Lerk, K. D. Kussendrager and H. Bosch, Studies on Tableting Properties of Lactose - Part 2. Consolidation and Compaction of Different Types of Crystalline Lactose, Pharm. Weekbl. Sci., 1985, 7, 186–193, DOI:10.1007/BF02307575.
- A. N. Zaid, R. J. Al-Ramahi, A. A. Ghoush, A. Qaddumi and Y. A. Zaaror, Weight and Content Uniformity of Lorazepam Half-Tablets: A Study of Correlation of a Low Drug Content Product, Saudi Pharm. J., 2013,(1), 21, DOI:10.1016/j.jsps.2011.12.009.
- British Pharmacopoeia Commission, British Pharmacopoeia - Tablets - Uniformity of Mass (2.9.5) Appendix XII, London, 2022 Search PubMed.
- BP, British Pharmacopoeia 2016, 2016, Vol. I Search PubMed.
- FDA. Guidance for Industry: Dissolution Testing of solid oral forms of immediate release dosage. U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER). https://www.fda.gov/media/70936/download (accessed 2023-11-25).
- British Pharmacopoeia Commission, British Pharmacopoeia- Tablets, Volume III-Appendix XII A. Disintegration, TSO, London, 2022 Search PubMed.
- J. W. Moore and H. H. Flanner, Mathematical Comparison of Dissolution Profiles, Pharmaceutical Technology, 1996, vol. 20, pp. 64–74 Search PubMed.
- P. Costa, An Alternative Method to the Evaluation of Similarity Factor in Dissolution Testing, Int. J. Pharm., 2001, 220, 77–83, DOI:10.1016/S0378-5173(01)00651-2.
- L. Bisharat, H. S. AlKhatib, S. Muhaissen, J. Quodbach, A. Blaibleh, M. Cespi and A. Berardi, The Influence of Ethanol on Superdisintegrants and on Tablets Disintegration, Eur. J. Pharm. Sci., 2019, 129, 140–147, DOI:10.1016/j.ejps.2019.01.004.
- K. Papós, P. Kása, I. Ilič, S. Blatnik-Urek, G. Regdon, S. Srčič, K. Pintye-Hódi and T. Sovány, Effect of the Surface Free Energy of Materials on the Lamination Tendency of Bilayer Tablets, Int. J. Pharm., 2015,(2), 496, DOI:10.1016/j.ijpharm.2015.10.061.
- S. Mangal, I. Larson, F. Meiser and D. A. V. Morton, Particle Engineering of Polymers into Multifunctional Interactive Excipients, in Handbook of Polymers for Pharmaceutical Technologies, 2015, pp. 1–32. DOI:10.1002/9781119041412.ch1.
- J. B. Dressman, A. Nair, B. Abrahamsson, D. M. Barends, D. W. Groot, S. Kopp, P. Langguth, J. E. Polli, V. P. Shah and M. Zimmer, Biowaiver Monograph for Immediate-Release Solid Oral Dosage Forms: Acetylsalicylic Acid, J. Pharm. Sci., 2012, 101, 2653–2667, DOI:10.1002/jps.23212.
- Y. Gonnissen, J. P. Remon and C. Vervaet, Development of Directly Compressible Powders via Co-Spray Drying, Eur. J. Pharm. Biopharm., 2007, 67(1), 220–226, DOI:10.1016/j.ejpb.2006.12.021.
- B. E. Sherwood and J. W. Becker, A New Class of High-Functionality Excipients: Silicified Microcrystalline Cellulose, Pharm. Technol., 1998, 22, 78–88 CAS.
- C. de Backere, T. De Beer, C. Vervaet and V. Vanhoorne, Effect of Binder Type and Lubrication Method on the Binder Efficacy for Direct Compression, Int. J. Pharm., 2021, 607, 120968, DOI:10.1016/j.ijpharm.2021.120968.
- USP, U.S. Pharmacopoeia-National Formulary [USP 38 NF 33], United States Pharmacopeial Convention, Rockville, Md, 2015 Search PubMed.
- S. Grote and P. Kleinebudde, A Comparative Study of the Influence of Alpha-Lactose Monohydrate Particle Morphology on Granule and Tablet Properties after Roll Compaction/Dry Granulation, Pharm. Dev. Technol., 2019, 24(3), 314–322, DOI:10.1080/10837450.2018.1476977.
- S. Airaksinen, M. Karjalainen, N. Kivikero, S. Westermarck, A. Shevchenko and J. Rantanen, Excipient Selection Can Significantly Affect Solid-State Phase Transformation in Formula Wet Granulation, AAPS PharmSciTech, 2005, 6(2), 311–322, DOI:10.1208/pt060241.
- E. I. Keleb, A. Vermeire, C. Vervaet and J. P. Remon, Single-Step Granulation/Tabletting of Different Grades of Lactose: A Comparison with High Shear Granulation and Compression, Eur. J. Pharm. Biopharm., 2004, 58(1), 77–82, DOI:10.1016/j.ejpb.2004.03.007.
- G. Bockstiegel, Relations between Pore Structure and Densification Mechanism in the Compacting of Iron Powders, in Iron Powder Metallurgy, 1968, pp. 54–71. DOI:10.1007/978-1-4899-6467-0_4.
- G. Bockstiegel, The Porosity-Pressure Curve and Its Relation to the Pore-Size Distribution in Iron Powder Compacts, in Modern Developments in Powder Metallurgy, 1966, pp. 155–187. DOI:10.1007/978-1-4684-7706-1_8.
- A. L. Skelbæk-Pedersen, T. K. Vilhelmsen, V. Wallaert and J. Rantanen, Investigation of the Effects of Particle Size on Fragmentation during Tableting, Int. J. Pharm., 2020, 576, 118985, DOI:10.1016/j.ijpharm.2019.118985.
- D. Markl and J. A. Zeitler, A Review of Disintegration Mechanisms and Measurement Techniques, Pharm. Res., 2017, 34(5), 890–917, DOI:10.1007/s11095-017-2129-z.
- N. Ekmekciyan, T. Tuglu, F. El-Saleh, C. Muehlenfeld, E. Stoyanov and J. Quodbach, Competing for Water: A New Approach to Understand Disintegrant Performance, Int. J. Pharm., 2018, 548(1), 491–499, DOI:10.1016/j.ijpharm.2018.07.025.
- P. Patel, D. Telange and N. Sharma, Comparison of Different Granulation Techniques for Lactose Monohydrate, Int. J. Pharm. Sci. Drug Res., 2011, 3(3), 222–225 CrossRef CAS.
- H. V. van Kamp, G. K. Bolhuis, K. D. Kussendrager and C. F. Lerk, Studies on Tableting Properties of Lactose. IV. Dissolution and Disintegration Properties of Different Types of Crystalline Lactose, Int. J. Pharm., 1986, 28(2–3), 229–238, DOI:10.1016/0378-5173(86)90249-8.
- F. Ferrari, M. Bertoni, M. C. Bonferoni, S. Rossi, C. Caramella and C. Nyström, Investigation on Bonding and Disintegration Properties of Pharmaceutical Materials, Int. J. Pharm., 1996, 136(1–2), 71–79, DOI:10.1016/0378-5173(96)04489-4.
- Å. Adolfsson and C. Nyström, Tablet Strength, Porosity, Elasticity and Solid State Structure of Tablets Compressed at High Loads, Int. J. Pharm., 1996, 132(1–2) DOI:10.1016/0378-5173(95)04336-5.
- A. Jabbari-Gargari, J. Moghaddas, H. Jafarizadeh-Malmiri and H. Hamishehkar, Ambient Pressure Drug Loading on Trimethylchlorosilane Silylated Silica Aerogel in Aspirin Controlled-Release System, Chem. Eng. Commun., 2021, 209(12), 1612–1625, DOI:10.1080/00986445.2021.1989420.
- S. A. Ata, O. A. Tarawneh, R. H. Sejare, S. Z. Sunoqrot and R. A. Al-Qirim, Impact of Solvent Selection and Absorptivity on Dissolution Testing of Acetylsalicylic Acid Enteric-Coated Tablets, Dissolution Technol., 2021, 28(2), 22–29, DOI:10.14227/DT280221P22.
- J. Xiang, B. Wang, L. Fu, C. Chen, W. Liu and S. Tan, Tailoring α/β Ratio of Pollen-like Anhydrous Lactose as Ingredient Carriers for Controlled Dissolution Rate, Crystals, 2021, 11, 1049, DOI:10.3390/cryst11091049.
- A. Illanes, Lactose: Production and Upgrading, in Lactose-Derived Prebiotics: A Process Perspective, 2016, pp. 1–33. DOI:10.1016/B978-0-12-802724-0.00001-9.
- M. G. Gänzle, G. Haase and P. Jelen, Lactose: Crystallization, Hydrolysis and Value-Added Derivatives, Int. Dairy J., 2008, 18(7), 685–694, DOI:10.1016/j.idairyj.2008.03.003.
- R. Kumar, A. K. Thakur, P. Chaudhari and N. Banerjee, Particle Size Reduction Techniques of Pharmaceutical Compounds for the Enhancement of Their Dissolution Rate and Bioavailability, J. Pharm. Innovation, 2021, 17, 332–352, DOI:10.1007/s12247-020-09530-5.
- B. Nickerson, A. Kong, P. Gerst and S. Kao, Correlation of Dissolution and Disintegration Results for an Immediate-Release Tablet, J. Pharm. Biomed. Anal., 2018, 150, 333–340, DOI:10.1016/j.jpba.2017.12.017.
- Z. T. Chowhan, The Effect of Low– and High–humidity Ageing on the Hardness, Disintegration Time and Dissolution Rate of Dibasic Calcium Phosphate–based Tablets, J. Pharm. Pharmacol., 1980, 32(1), 10–14, DOI:10.1111/j.2042-7158.1980.tb12836.x.
- D. R. Adam, N. Al Rayes, R. Fatoum, G. Arafeh, T. R. Adam and A. Kola-Mustapha, Comparative Evaluation of Amlodipine Besylate Generic Tablet and Capsule Brands in Riyadh, Saudi Arabia, Dissolution Technol., 2022,(24), 29, DOI:10.14227/DT290422PGC2.
- C. S. Omar, R. M. Dhenge, J. D. Osborne, T. O. Althaus, S. Palzer, M. J. Hounslow and A. D. Salman, Roller Compaction: Effect of Morphology and Amorphous Content of Lactose Powder on Product Quality, Int. J. Pharm., 2015, 496(1), 63–74, DOI:10.1016/j.ijpharm.2015.06.032.
- T. Sebhatu and G. Alderborn, Relationships between the Effective Interparticulate Contact Area and the Tensile Strength of Tablets of Amorphous and Crystalline Lactose of Varying Particle Size, Eur. J. Pharm. Sci., 1999, 8(4), 235–242, DOI:10.1016/S0928-0987(99)00025-1.
- S. Dizaj, Z. Vazifehasl, S. Salatin, K. Adibkia and Y. Javadzadeh, Nanosizing of Drugs: Effect on Dissolution Rate. Research in Pharmaceutical Sciences, Res. Pharm. Sci., 2015, 10(2), 95–108 Search PubMed.
- P. H. M. Janssen, A. Berardi, J. H. Kok, A. W. Thornton and B. H. J. Dickhoff, The Impact of Lactose Type on Disintegration: An Integral Study on Porosity and Polymorphism, Eur. J. Pharm. Biopharm., 2022, 180, 251–259, DOI:10.1016/j.ejpb.2022.10.012.
- C. A. Aguilar and G. R. Ziegler, Physical and Microscopic Characterization of Dry Whole Milk with Altered Lactose Content. 2. Effect of Lactose Crystallization, J. Dairy Sci., 1994, 77(5), 1198–1204, DOI:10.3168/jds.S0022-0302(94)77058-2.
- W. Kaialy and A. Nokhodchi, Engineered Mannitol Ternary Additives Improve Dispersion of Lactose-Salbutamol Sulphate Dry Powder Inhalations, AAPS J., 2013, 15(3), 728–743, DOI:10.1208/s12248-013-9476-4.
- F. O. Costa, A. A. C. C. Pais and J. J. S. Sousa, Analysis of Formulation Effects in the Dissolution of Ibuprofen Pellets, Int. J. Pharm., 2004, 270(1–2), 9–19, DOI:10.1016/j.ijpharm.2003.10.002.
- H. Jain, K. S. Khomane and A. K. Bansal, Implication of Microstructure on the Mechanical Behaviour of an Aspirin-Paracetamol Eutectic Mixture, CrystEngComm, 2014, 16(36), 8471–8478, 10.1039/c4ce00878b.
- C. S. Omar, R. M. Dhenge, J. D. Osborne, T. O. Althaus, S. Palzer, M. J. Hounslow and A. D. Salman, Roller Compaction: Effect of Morphology and Amorphous Content of Lactose Powder on Product Quality, Int. J. Pharm., 2015, 496(1), 63–74, DOI:10.1016/j.ijpharm.2015.06.032.
- S. P. Duddu, M. Vakilynejad, F. Jamali and D. J. W. Grant, Stereoselective Dissolution of Propranolol Hydrochloride from Hydroxypropyl Methylcellulose Matrices, Pharm. Res., 1993, 10(11), 1648–1653, DOI:10.1023/A:1018937123058.
- E. Yonemochi, Y. Yoshihashi and K. Terada, Quantitative Relationship between Solubility, Initial Dissolution Rate and Heat of Solution of Chiral Drugs, Pharm. Res., 2000, 17(1), 90–93, DOI:10.1023/A:1007578811049.
- S. P. Duddu, F. K. Y. Fung and D. J. W. Grant, Effect of the Opposite Enantiomer on the Physicochemical Properties of (-) -Ephedrinium 2-Naphthalenesulfonate Crystals, Int. J. Pharm., 1993, 94(1–3), 171–179, DOI:10.1016/0378-5173(93)90021-7.
- L. Maggi, G. Massolini, E. De Lorenzi, U. Conte and G. Caccialanza, Evaluation of Stereoselective Dissolution of Verapamil Hydrochloride from Matrix Tablets Press-Coated with Chiral Excipients, Int. J. Pharm., 1996, 136(1–2), 43–51, DOI:10.1016/0378-5173(96)04487-0.
| This journal is © The Royal Society of Chemistry 2026 |