 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
DOI: 10.1039/D6OB90044E
(Correction)
Org. Biomol. Chem., 2026, Advance Article
Correction: Synthesis of hexahydroisobenzofuran-1(3H)-ones via pyridine-boryl radical-catalyzed intramolecular cascade cyclization of alkene-tethered cyclobutyl ketones
Xiao-Ye Yang,
Hua Huang,
Qiong Wu,
Yun-Xuan Tan* and
Ping Tian*
The Research Center of Chiral Drugs, Innovation Research Institute of Traditional Chinese Medicine, Shanghai University of Traditional Chinese Medicine, 1200 Cailun Road, Shanghai 201203, China. E-mail: tanyx1993@shutcm.edu.cn; tianping@shutcm.edu.cn
Received
7th April 2026
, Accepted 7th April 2026
First published on 17th April 2026
Abstract
Correction for ‘Synthesis of hexahydroisobenzofuran-1(3H)-ones via pyridine-boryl radical-catalyzed intramolecular cascade cyclization of alkene-tethered cyclobutyl ketones’ by Xiao-Ye Yang et al., Org. Biomol. Chem., 2025, 23, 10057–10061, https://doi.org/10.1039/D5OB01566A.
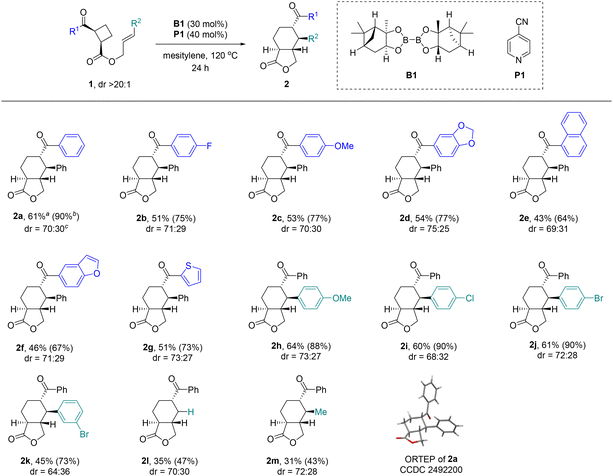
The authors regret that there was an error in the structure of compound 2f in Scheme 2 and in the SI (Pages S17, S74, and S75). The correct scheme is shown below.
The Royal Society of Chemistry apologises for these errors and any consequent inconvenience to authors and readers.
| This journal is © The Royal Society of Chemistry 2026 |

![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
: