Open Access Article
Open Access ArticleVisible-light-induced regioselective N-oxazolidinone radical addition to indoles and arenes via EDA complex formation
Monica Fiorenza
Boselli†
 a,
Sara
Ferrario†
a,
Sara
Ferrario†
 a,
Niccolò
Intini‡
a,
Niccolò
Intini‡
 a,
Kirsten
Zeitler
a,
Kirsten
Zeitler
 b and
Alessandra
Puglisi
b and
Alessandra
Puglisi
 *a
*a
aDipartimento di Chimica, Università degli Studi di Milano, via Golgi, 19-20133 Milano, Italy. E-mail: alessandra.puglisi@unimi.it
bInstitut für Organische Chemie, Universität Leipzig, Germany
First published on 18th March 2026
Abstract
We report the generation of N-oxazolidinone radicals via electron donor–acceptor (EDA) complex formation. Visible light irradiation of pyridinium-based radical precursors, either in the presence or in the absence of NaHCO3, enables a mild, regioselective and operationally simple N-arylation, providing direct access to N-aryloxazolidin-2-ones. The transformation can be easily converted into a continuous flow process, providing improved productivity over the corresponding batch reaction.
Oxazolidin-2-ones are important heterocyclic moieties, commonly found in several pharmaceutical compounds, and they represent the only new class of synthetic antibiotics introduced into clinical use over the past 50 years.1 Linezolid and Eperezolid are two examples of N-arylated oxazolidinones whose antibacterial activity depends on the N-aryl substituent. Tremendous advancements in the synthesis of oxazolidin-2-ones have been reported,2 however, the direct introduction is still challenging. In the past few years, we have developed mild and reliable methodologies to generate N-centered radicals under photocatalytic conditions, enabling for the direct formation of C–N bonds.3–5 In this context, we recently reported the photoredox-catalysed generation of unprecedented N-oxazolidinone radicals and their addition to variously functionalized arenes and hetarenes, starting from a novel class of N-radical precursors.6
Additionally, light-driven synthetic strategies based on the direct irradiative activation of Electron Donor–Acceptor (EDA) complexes have become widely explored in modern organic chemistry.7–9 Light excitation of the EDA complex may trigger a single-electron-transfer (SET) that can generate radical intermediates under mild, photocatalyst-free conditions.10 The use of inorganic bases as external electron donors is well documented in supramolecular chemistry11 and biological systems.12 However, their involvement in photochemical transformations has only recently begun to be explored. Exemplarily, in 2017 Leonori and co-workers developed the visible-light-mediated 5-exo-dig cyclization reactions of amidyl radicals in the presence of K2CO3 and 1,4-cyclohexadiene.13 Two years later, Pericàs and co-workers reported a light-driven amidation reaction of (hetero)aromatic systems via anion–π complex formation between N-aryloxyamides, acting as nitrogen radical precursors, and K2CO3.14 More recently, Glorius demonstrated the formation of an EDA complex between NaHCO3 and an N-aminopyridinium tetrafluoroborate salt as a nitrogen radical surrogate.15 Building on these precedents, we report here the EDA complex-mediated N-arylation of oxazolidin-2-ones.
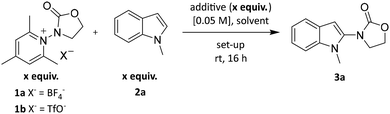
The model reaction between N-radical precursor 1a![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4,6,16 and 1-methylindole (2a) to regioselectively afford compound 3a was investigated upon blue light irradiation over 16 h under a variety of conditions, including different additives, solvents, stoichiometry and reaction set-ups. Selected results are summarised in Table 1 (for the complete survey of reaction conditions, evaluating additives, reaction time, concentration, solvent system, precursors, stoichiometry see SI).
4,6,16 and 1-methylindole (2a) to regioselectively afford compound 3a was investigated upon blue light irradiation over 16 h under a variety of conditions, including different additives, solvents, stoichiometry and reaction set-ups. Selected results are summarised in Table 1 (for the complete survey of reaction conditions, evaluating additives, reaction time, concentration, solvent system, precursors, stoichiometry see SI).
| Entry | Solvent | Additive (equiv.) | Set-up | Ratio 1a![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2a 2a |
Yielda (%) |
|---|---|---|---|---|---|
| a Isolated yield. b 1b was used as radical precursor. | |||||
| 1 | CH3CN | Na2CO3 (5) | PR-1 | 1.2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
22 |
| 2 | CH3CN | NaHCO3 (5) | PR-1 | 1.2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
60 |
| 3 | CH3CN | KHCO3 (5) | PR-1 | 1.2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
8 |
| 4 | CH3CN | NaHCO3 (5) | PR-2 | 1.2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
61 |
| 5 | Water![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CH3CN 4 CH3CN 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
NaHCO3 (5) | PR-2 | 1.2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
63 |
| 6 | Water![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CH3CN 4 CH3CN 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
NaHCO3 (1) | PR-2 | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
70 |
| 7 | CH3CN | NaHCO3 (1) | PR-2 | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
78 |
| 8b | CH3CN | NaHCO3 (1) | PR-2 | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
81 |
| 9b | CH3CN | — | PR-2 | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
66 |
| 10 | CH3CN | — | PR-2 | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
Traces |
The results were obtained by evaluating two different batch photoreactors (PRs) (see SI) to increase the efficiency of the transformation: in PR-1 the reaction vials were laterally irradiated by 455 nm blue LEDs, whereas in PR-2 irradiation was provided by a 456 nm Kessil lamp. NaHCO3 emerged as the most efficient additive when CH3CN was used as the solvent. Initial tests showed similar performance of both PRs (Table 1, entries 2 and 4). Although a large excess of NaHCO3 was initially employed during reaction optimisation (Table 1, entries 1–5), it was found out that 1 equiv. was enough to efficiently promote the radical transformation (Table 1, entry 6). A solvent system water![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CH3CN (v/v 4
CH3CN (v/v 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) was also tested, affording a homogeneous reaction and a good product yield (Table 1, entry 5). Notably, this result may allow to expand the compatibility of the methodology toward water-soluble substrates. An excess of 1-methylindole (2a) (2 equiv.), in combination with 1 equiv. of NaHCO3 upon blue irradiation with PR-2 enabled the formation of product 3a in high yield both under aqueous conditions water
1) was also tested, affording a homogeneous reaction and a good product yield (Table 1, entry 5). Notably, this result may allow to expand the compatibility of the methodology toward water-soluble substrates. An excess of 1-methylindole (2a) (2 equiv.), in combination with 1 equiv. of NaHCO3 upon blue irradiation with PR-2 enabled the formation of product 3a in high yield both under aqueous conditions water![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CH3CN (v/v 4
CH3CN (v/v 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) (70%, entry 6) as well as in CH3CN (78%, entry 7). It is worth noting that radical precursor 1b, bearing a triflate (vs. BF4−) as the pyridinium counterion, was also highly effective, delivering product 3a in 81% yield (entry 8).
1) (70%, entry 6) as well as in CH3CN (78%, entry 7). It is worth noting that radical precursor 1b, bearing a triflate (vs. BF4−) as the pyridinium counterion, was also highly effective, delivering product 3a in 81% yield (entry 8).
Control experiments revealed that the reaction does not proceed in the absence of light. Working with precursor 1b under irradiation also allowed formation of product 3a in 66% isolated yield in the absence of NaHCO3 (Table 1, entry 9), whereas the use of precursor 1a under identical conditions, only afforded trace amounts of 3a (entry 10). This interesting finding prompted us to further study the “additive-free” use of precursor 1b in CH3CN under irradiation as N-radical source. Having established different conditions to generate the N-oxazolidinone radical from precursors 1a and 1b (see SI for details), we next explored the scope of the reaction. The results are summarised in Scheme 1. Three different reaction conditions (A–C) were examined under visible-light irradiation (Kessil lamp, 456 nm) for 16 h: precursor 1a, 1 equiv. of NaHCO3, water![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CH3CN (v/v 4
CH3CN (v/v 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 0.05 M (condition A); precursor 1b, 1 equiv. of NaHCO3, CH3CN 0.05 M (condition B); precursor 1b, CH3CN 0.05 M (condition C). Differently substituted 1-methylindoles reacted under all three reaction conditions, leading to the corresponding products 3a–c in satisfactory to high yields. Other electron-rich substrates, including 1-methylpyrrole, anthracene, 1,3,5-trimethoxybenzene and 1,3-dimethoxybenzene, also underwent functionalisation to afford products 3d–g in moderate to good yields.17 Moreover, arenes 2e–g are not prone to form EDA complexes with precursor 1b (see below for mechanistic investigations). Notably, unprotected indoles also proved to be suitable substrates for the desired transformation: under the tested conditions, product 3h was isolated in 12–30% yield. Unprotected indoles bearing either electron-donating or electron-withdrawing groups also underwent the reaction both under condition A and C, although in modest yields (18% and 31% for 3i, 33% and 18% for 3j, 37% and 7% for 3k). The synthetic potential of this methodology was further demonstrated through its application to tryptophan derivatives as reaction partners. Dipeptide Boc-Trp-Phe-OMe afforded the desired product 3l in 48% yield under condition B, while tripeptide Boc-Val-Trp-Phe-OMe performed very well, delivering product 3m in 62% yield (under condition B, but irradiating at 390 nm, according to literature precedents for the light-induced modification of tryptophan residues in peptides).18–20 This highlights the pronounced selectivity of the method for the most electron-rich ring in the substrates, demonstrating its potential application in the late-stage functionalisation of complex biomolecules, under very mild conditions, thus offering further applicability to biological systems.
1) 0.05 M (condition A); precursor 1b, 1 equiv. of NaHCO3, CH3CN 0.05 M (condition B); precursor 1b, CH3CN 0.05 M (condition C). Differently substituted 1-methylindoles reacted under all three reaction conditions, leading to the corresponding products 3a–c in satisfactory to high yields. Other electron-rich substrates, including 1-methylpyrrole, anthracene, 1,3,5-trimethoxybenzene and 1,3-dimethoxybenzene, also underwent functionalisation to afford products 3d–g in moderate to good yields.17 Moreover, arenes 2e–g are not prone to form EDA complexes with precursor 1b (see below for mechanistic investigations). Notably, unprotected indoles also proved to be suitable substrates for the desired transformation: under the tested conditions, product 3h was isolated in 12–30% yield. Unprotected indoles bearing either electron-donating or electron-withdrawing groups also underwent the reaction both under condition A and C, although in modest yields (18% and 31% for 3i, 33% and 18% for 3j, 37% and 7% for 3k). The synthetic potential of this methodology was further demonstrated through its application to tryptophan derivatives as reaction partners. Dipeptide Boc-Trp-Phe-OMe afforded the desired product 3l in 48% yield under condition B, while tripeptide Boc-Val-Trp-Phe-OMe performed very well, delivering product 3m in 62% yield (under condition B, but irradiating at 390 nm, according to literature precedents for the light-induced modification of tryptophan residues in peptides).18–20 This highlights the pronounced selectivity of the method for the most electron-rich ring in the substrates, demonstrating its potential application in the late-stage functionalisation of complex biomolecules, under very mild conditions, thus offering further applicability to biological systems.
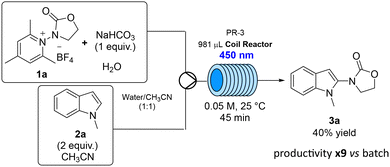
Given the well-established benefits of light-driven transformations in continuous flow,21,22 we then further explored the regioselective addition of the N-oxazolidinone radical, generated from precursor 1a, to 1-methylindole (2a) in the presence of NaHCO3 as the external donor under flow conditions using Asia Syrris system (PR-3), equipped with the photochemistry module (for further details, see SI). After extensive reaction conditions optimisation in flow, the following set-up was chosen (Scheme 2): an aqueous solution of 1a and NaHCO3 (1 equiv.) and a solution of 2a (2 equiv.) in CH3CN were fed through a T-junction into a coil of 981 µL volume. The final mixture of water and CH3CN (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 0.05 M, homogeneous solution) was irradiated at 450 nm at room temperature. The screening of different residence times (see SI), allowed to identify 45 min as a good compromise between the conversion and the productivity. After a residence time of 45 min, the desired product 3a was isolated in 40% yield. This led to an increase in productivity of about 9 times compared to the same transformation in batch (57% yield, 16 h).
1, 0.05 M, homogeneous solution) was irradiated at 450 nm at room temperature. The screening of different residence times (see SI), allowed to identify 45 min as a good compromise between the conversion and the productivity. After a residence time of 45 min, the desired product 3a was isolated in 40% yield. This led to an increase in productivity of about 9 times compared to the same transformation in batch (57% yield, 16 h).
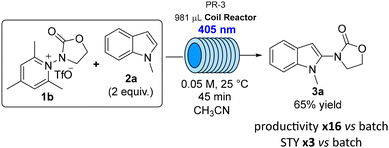
A more operationally convenient, simple single-feed flow set-up was employed for the reaction using triflate precursor 1b, with CH3CN as the solvent (0.05 M). Using the typical 450 nm irradiation, the desired product 3a was isolated in 50% yield after 45 min residence time. Increasing the residence time up to 60 min was not beneficial, since 3a was recovered in 51% yield. Gladly, a fine tuning of the reaction conditions allowed to identify 405 nm as optimal wavelength, leading to the isolation of product 3a in 65% yield in 45 min residence time, as depicted in Scheme 3. Under these flow conditions, the productivity increased of about 16 times compared to the same transformation in batch (50% yield, 16 h) while the space–time yield (STY) was approximately 3 times higher (see SI for details on reaction conditions screening and calculation of productivity and STY).
Investigations were performed in order to provide insights into the reaction mechanism. In particular, UV-Vis absorption spectroscopy15 was employed to gain insight into the photochemical reaction mechanism either in the presence or in the absence of NaHCO3. UV-Vis spectra of a mixture of pyridinium salt 1a and 1-methylindole (2a) allowed to observe a new absorption band at ca. 350 nm (Fig. 1A). The same interaction was observed for precursor 1b and 2a (Fig. 1B); a colour change was also observed in the solution of 1b and 2a (Fig. 1C). Moreover, UV-Vis spectra showed an evident bathochromic shift for the mixture of nitrogen radical precursor 1a and NaHCO3 compared to the spectrum recorded for 1a alone, thus hinting to a possible EDA complex formation between the electron-poor precursor 1a and NaHCO3 (Fig. 1D).13–15 This observation was also in agreement with the observed colour change in the solution of 1a upon addition of NaHCO3 (Fig. 1E). In contrast, mixing the nitrogen radical precursor 1b with NaHCO3 produced a noticeable, albeit less pronounced, change in the UV-Vis spectrum compared to 1b alone (Fig. 1F). This observation aligns with the less pronounced colour change observed when NaHCO3 was added to the solution of 1b (Fig. 1G). DFT calculations supported the proposal of an electron transfer from the bicarbonate ion to the pyridinium ion (see below and SI for details). Cyclic voltammetry measurements were carried out to compare the electrochemical properties of the two pyridinium salts 1a and 1b. Importantly, the different reactivity of precursors 1a and 1b can be ascribed to a difference in reaction kinetics, as highlighted by a detailed CV analysis (see SI for detailed studies).
Based on these findings, we propose a plausible mechanism illustrated in Fig. 2 for the transformation. Specifically, the EDA complex formed through the interaction between the electron-poor precursor 1b and 1-methylindole (2a) can be photoexcited under visible-light irradiation. The resulting excited state undergoes a single-electron transfer (SET), leading to the fragmentation of the complex and the subsequent cleavage of radical intermediate I to provide 2,4,6-collidine II and the N-oxazolidinone radical III. The regioselective radical addition of III to 1-methylindole (2a) affords radical intermediate IV, which can engage in a chain propagation step23,24 by reducing precursor 1b. Finally, deprotonative rearomatization of the oxidized intermediate V affords the desired functionalized indole 3a.
In the case of radical precursor 1a, the EDA complex with 2a is somehow not productive (Table 1, entry 10). In this case, the addition of NaHCO3 is beneficial to the reaction initiation, since NaHCO3 could act as an external donor for radical precursor 1a. The EDA complex can then be photoexcited under visible-light irradiation, and the resulting excited state undergoes a single-electron transfer (SET), leading to the fragmentation of the complex and the subsequent cleavage of radical intermediate I to provide 2,4,6-collidine II and the N-oxazolidinone radical III. The regioselective radical addition of III to 1-methylindole (2a) affords radical intermediate IV, which – similarly to the mechanism in Fig. 3 – can engage in a chain propagation step by reducing precursor 1a.23,24 Finally, rearomatization via deprotonation of the oxidized intermediate V affords the desired functionalized indole 3a (Fig. 3). In principle, the mechanism in Fig. 3 could be also plausible for precursor 1b.
Conclusions
In conclusion, a mild and efficient method for the direct N-arylative introduction of 2-oxazolidinone on different indoles and arenes via EDA complex formation was developed. The reaction efficiently works under a wide variety of conditions and, importantly, allows the employment of unprotected indole moieties offering the opportunity for the direct functionalisation of tryptophan dipeptides and tripeptides. Additionally, the methodology is water compatible, hence importantly expanding the applicability towards water-soluble substrates such as biomolecules. The photochemical reaction can be easily transferred to continuous flow conditions, thus greatly improving the reaction productivity. Mechanistic insights revealed a novel photochemical activation for the pyridinium-derived radical precursors in a photocatalyst-free, simplified set-up. We believe that this work will further advance N-oxazolidinone radical chemistry, providing a versatile tool for synthetic chemists aiming to achieve N-functionalised oxazolidinones under mild and metal-free conditions.Author contributions
Conceptualisation, A. P. and K. Z.; investigation, M. F. B., S. F. and N. I.; writing—original draft preparation, A. P., M. F. B. and S. F.; writing—review and editing, A. P. and K. Z.; supervision, A. P.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: description of equipment, synthesis of substrates 2k and 2l, screening of reaction conditions, mechanistic investigation. See DOI: https://doi.org/10.1039/d6ob00369a.Acknowledgements
This work was supported by: (a) MUSA – Multilayered Urban Sustainability Action – project, funded by the European Union – NextGenerationEU, under the National Recovery and Resilience Plan (NRRP) Mission 4 Component 2 Investment Line 1.5: strengthening of research structures and creation of R&D “innovation ecosystems”, set up of “territorial leaders in R&D”; (b) PSR 2025 grant “Catalytic approaches to the sustainable synthesis of high added-value fine chemicals” from the University of Milan. (c) Deutsche Forschungsgemeinschaft [DFG (German Science Foundation) grant TRR 325-444632635]. The authors thank Alessia Bianchi (Università degli Studi di Milano) for experimental activity. M. F. B. thanks the Ministero dell'Università e della Ricerca (MUR) (Project PRIN2022 “TECHNO”, financed by EU – Next Generation EU, Mission 4 Component 1 CUP G53D23003280006) for a postdoctoral fellowship. S. F. thanks Cosma SpA for cofinancing a PhD fellowship. N. I. thanks Olon SpA for cofinancing a PhD fellowship.References
- G. F. S. Fernandes, C. B. Scarim, S. H. Kim, J. Wu and D. Castagnolo, RSC Med. Chem., 2023, 14, 823–847 Search PubMed.
- F. Sun, E. V. Van der Eycken and H. Feng, Adv. Synth. Catal., 2021, 363, 5168–5195 CrossRef CAS.
- M. F. Boselli, N. Intini, A. Puglisi, L. Raimondi, S. Rossi and M. Benaglia, Eur. J. Org. Chem., 2023, e202201309 Search PubMed.
- M. F. Boselli, I. Ghosh, N. Intini, M. Fattalini, A. Puglisi, B. König and M. Benaglia, Chemistry, 2025, 31, e202404385 Search PubMed.
- E. Colombo, M. F. Boselli, M. Fattalini, V. Chiroli, S. Rossi, M. Benaglia and A. Puglisi, Org. Lett., 2025, 27, 8540–8544 Search PubMed.
- S. Ferrario, S. Rossi, N. Intini, J. Bruno-Colmenarez, M. Baumann and M. Benaglia, Org. Lett., 2025, 27, 12276–12280 CrossRef CAS PubMed.
- E. F. Hilinski, J. M. Masnovi, C. Amatore, J. K. Kochi and P. M. Rentzepis, J. Am. Chem. Soc., 2002, 105, 6167–6168 CrossRef.
- C. G. S. Lima, T. d. M. Lima, M. Duarte, I. D. Jurberg and M. W. Paixão, ACS Catal., 2016, 6, 1389–1407 CrossRef CAS.
- G. E. M. Crisenza, D. Mazzarella and P. Melchiorre, J. Am. Chem. Soc., 2020, 142, 5461–5476 CrossRef CAS PubMed.
- S. V. Rosokha and J. K. Kochi, Acc. Chem. Res., 2008, 41, 641–653 CrossRef CAS.
- C. Garau, D. Quinonero, A. Frontera, P. Ballester, A. Costa and P. M. Deya, J. Phys. Chem. A, 2005, 109, 9341–9345 Search PubMed.
- C. Estarellas, A. Frontera, D. Quinonero and P. M. Deya, Angew. Chem., Int. Ed., 2011, 50, 415–418 CrossRef CAS PubMed.
- D. F. Reina, E. M. Dauncey, S. P. Morcillo, T. D. Svejstrup, M. V. Popescu, J. J. Douglas, N. S. Sheikh and D. Leonori, Eur. J. Org. Chem., 2017, 2108–2111 CrossRef CAS.
- L. Buglioni, M. M. Mastandrea, A. Frontera and M. A. Pericàs, Chem. Eur. J., 2019, 25, 11785–11790 CrossRef CAS PubMed.
- C. C. Chintawar, R. Laskar, D. Rana, F. Schäfer, N. Van Wyngaerden, S. Dutta, C. G. Daniliuc and F. Glorius, Nat. Catal., 2024, 7, 1232–1242 CrossRef CAS.
- T. W. Greulich, C. G. Daniliuc and A. Studer, Org. Lett., 2015, 17, 254–257 CrossRef CAS PubMed.
- The modest yields of these compounds could be ascribed to the low solubility of the corresponding starting materials under condition A.
- S. J. Tower, W. J. Hetcher, T. E. Myers, N. J. Kuehl and M. T. Taylor, J. Am. Chem. Soc., 2020, 142, 9112–9118 CrossRef CAS PubMed.
- C. R. Hoopes, F. J. Garcia, A. M. Sarkar, N. J. Kuehl, D. T. Barkan, N. L. Collins, G. E. Meister, T. R. Bramhall, C. H. Hsu, M. D. Jones, M. Schirle and M. T. Taylor, J. Am. Chem. Soc., 2022, 144, 6227–6236 CrossRef CAS PubMed.
- M. Hornink, M. E. C. Thedy, E. Romero and G. A. Molander, ChemistryEurope, 2026, 4, e202500284 CrossRef CAS.
- D. Cambie, C. Bottecchia, N. J. Straathof, V. Hessel and T. Noel, Chem. Rev., 2016, 116, 10276–10341 CrossRef CAS PubMed.
- C. Sambiagio and T. Noël, Trends Chem., 2020, 2, 92–106 CrossRef CAS.
- M. A. Cismesia and T. P. Yoon, Chem. Sci., 2015, 6, 5426–5434 RSC.
- K. A. V. Miyuranga, K. E. Ashcraft and S. P. Pitre, Tetrahedron Chem, 2024, 12, 100110 CrossRef CAS.
Footnotes |
| † These authors contributed equally to this work. |
| ‡ Current address: Facultad de Quimica, Centro de Investigación Multidisciplinar Pleiades-Vitalis, Universidad de Murcia, Spain. |
| This journal is © The Royal Society of Chemistry 2026 |