Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Cyclisations and hydrolysis of geranyl and farnesyl halides in water facilitated by ultrasound-induced emulsification
Jan Luca
Budde
 a,
Andreas
Kirschning
a,
Andreas
Kirschning
 *ab and
Gerald
Dräger
*ab and
Gerald
Dräger
 a
a
aInstitute of Organic Chemistry and Center of Biomolecular Drug Research (BMWZ), Leibniz Universität Hannover, Schneiderberg 1B, 30167 Hannover, Germany. E-mail: andreas.kirschning@oci.uni-hannover.de
bUppsala Biomedical Center (BMC), Uppsala University, Husargatan 3, 752 37 Uppsala, Sweden
First published on 18th March 2026
Abstract
How stable are geranyl and farnesyl halides in water? Given the current increased interest in mimicking terpene synthases, we address this fundamental question by investigating their behaviour with regard to their ability to undergo hydrolysis and cyclisation reactions in the absence of a catalyst or chemical promotor. Some linear terpenoids with leaving groups, such as bromide, appear to have an intrinsic tendency to cyclise. In particular, hydrolysis or cyclisation of the respective water-insoluble bromide analogues by ultrasonic treatment was found to be effective. This technique ensured efficient dispersion of the hydrophobic oil droplets in the aqueous medium. It was found that even with short ultrasonic treatment times, the (2,3-Z)-configured terpenes neryl and Z-farnesyl bromide showed a pronounced tendency to cyclise, which in some cases led to high conversion and high selectivity. In contrast, omitting ultrasonic treatment resulted in a heterogeneous reaction environment, leading to a broader and more complex product spectrum, including the formation of dimeric ethers.
Introduction
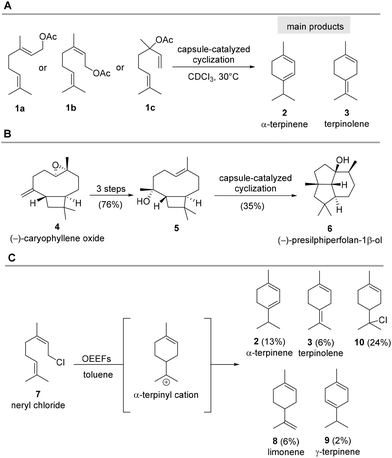
Class I terpene synthases catalyse the conversion of activated, linear terpenyl precursors into structurally complex, oligocyclic carbon scaffolds by precisely directed carbocation cascade mechanisms.1 Terpene synthases are masters of carbocation chemistry and therefore several chemically driven studies were published recently, that attempt to reproduce the active centers of such synthases. Particularly noteworthy are chemical systems with a defined three-dimensionality which can function as artificial terpene synthase mimics.2 For example, Brønsted acidic supramolecular resorcin[4]arene capsules as disclosed by Tiefenbacher et al. can be employed as artificial binding pockets for linear terpene precursors. These host systems are spontaneously formed from six resorcin monomers. In these investigations, the pyrophosphate group in geranylpyrophosphate (GPP) or farnesylpyrophosphate (FPP) was exchanged by the acetate group.3,4 Geranyl acetate 1a, neryl acetate 1b and linalool acetate 1c undergo cyclisation in the presence of these capsules in an organic solvent and besides several other by-products α-terpinene 2 and terpinolene 3 were found to be formed as major products (Scheme 1A).5 Studies were extended to cyclisations of already cyclised terpenes and as such it was possible to induce a cyclisation that leads to (−)-presilphiperfolane-1β-ol (6) from sesquiterpene 5, the latter being prepared chemically from (−)-caryophyllene oxide (4) in three steps (Scheme 1A).3Recently, cyclisations of monoterpenes, such as halogen derivative neryl chloride (7) on carbon nanotubes in electromicrofluidic devices were reported that make use of oriented external electric fields (OEEFs). It was suggested that this setup is able to direct the carbocationic cascades (Scheme 1B).6 The initial cyclisation step provides the α-terpinyl cation which leads to four elimination products including α-terpinene (2), terpinolene (3) limonene (8) and γ-terpinene (9) as well as the chloride adduct 10. Depending on the flow rate and the voltage used, the quantities of products (2, 3, 8, 9, 10) were adjustable. The work, however, was restricted to neryl chloride (7) that has a preformed bent conformation due to the presence of the (2,3-Z)-configured olefinic double bond. In fact, spontaneous cyclisations of such linear precursors have shown to occur in aqueous environments, without the need of external electric fields.7,8 As early as 1979, Bunton and colleagues investigated the solvolysis of geranyl and neryl derivatives, demonstrating the formation of limonene (8), terpinolene (3), and other monoterpenes.9,10 Interestingly, investigations with allyl chloride derivatives such as neryl chloride (7) were consistently performed in solvent systems composed of water and acetone supplemented with surfactants to increase solubility. When employing water soluble pyrophosphate analogues instead, water alone can be used for the homogeneous solvolysis. Linear pyrophosphates such as (2,3-E)-farnesyl pyrophosphate and its (2,3-Z)-isomer undergo hydrolysis in water remarkably rapid even at ambient temperature.7
Still, the heterogeneous hydrolysis of mono- and sesquiterpenyl halides solely in water in the absence of 3D template-like systems or under special chemophysical conditions has hardly been investigated. In view of the increasing interest in cationic cascades for mimicking terpene synthases, we now report a seminal study on the inherent reactivity of linear terpene halides and their principal ability to undergo cyclisations in an aqueous medium. The studies presented here will cover mono- and sesquiterpenes and are intended to serve as a guide and orientation for the development of chemical systems capable of mimicking terpene synthases.2–5
Results and discussion
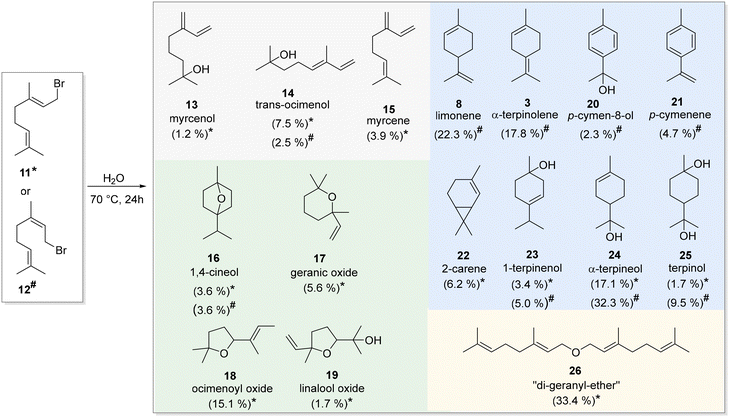
Initial hydrolysis experiments were carried out with geranyl bromide (11) and neryl bromide (12), which differ only in the configuration of the olefinic double bond at C2/C3. As such, the respective bromide was added to a microwave vial containing deionised water only. The sealed vials were stirred vigorously at 70 °C. Within 24 h, several established linear and cyclic monoterpenes were produced. The reaction mixtures, which had become acidic (pH = 2) as a result of HBr formation, were subsequently subjected to GC-MS analysis (Scheme 2). These product mixtures were analysed by comparison of their mass spectra with data base information (see SI). Furthermore, commercially available monoterpenes (3, 8, 15, 16, 19 and 24) were purchased and also used for comparing not only the mass spectra but also the retention. The (2,3-E)-configured bromide 11 gave the three linear hydrolysis products myrcenol (13), trans-ocimenol (14) and myrcene (15) while for neryl bromide (12) only one linear product 14 could be detected. In addition, geranyl bromide (11) furnished several cyclic ethers 16–19 with ocimenoyl oxide (18) and linalool oxide (19) being tetrahydrofuran derivatives. Terpenoid 18 was formed in considerable amounts and could thus be isolated chromatographically and subsequently characterised by NMR spectroscopy. 1,4-Cineole (16) was detected in the heterogeneous hydrolysis of both bromides 11 and 12.The largest and most diverse group of products is formed via initial 1,6-cyclisation, when (2,3-Z)-configured neryl bromide (12) is used. These include the known terpenes limonene (8), α-terpinolene (3), the autoxidation products p-cymene-8-ol (20) and p-cymenene (21) as well as 1-terpinenol (23), α-terpineol (24) as major product and finally terpinol (25). Despite the (2,3-E)-configuration present, heterogeneous hydrolysis of geranyl bromide (11) also provided cyclic monoterpenes 23–25. However, these were present in much smaller quantities compared to the hydrolysis products formed from bromide 12. Interestingly, also the bicyclic 2-carene (22) was generated from bromide 11 under the given conditions. Under these conditions also the dimerised geranylgeranyl ether (26) was created as the main product, which was not found when neryl bromide (12) was exposed to the same hydrolysis conditions. Based on this spectrum of products, it can be concluded that the hydrolysis of 11 leads to the formation of cyclic ethers, which originate, for example, from the linear alcohols 13 and 14. This is consistent with the use of water under solvolytic conditions, as it ‘traps’ these alcohols due to repulsive interactions, forcing them into conformations with minimal surface area. Under such circumstances it is predestined to undergo cyclisation resulting in ethers. Additionally, the constant formation of HBr, that alters the acidic aqueous medium, may further enhance this effect. Still, there is a constant competition between intra- and intermolecular ether formation which consequently can also lead to the ether dimer 26.
On the other hand, the heterogeneous hydrolysis of 12 leads to the formation of carbacycles 3, 8 and 20–25. The (2,3-Z)-configured double bond allows for an ideal orientation and close proximity of C1 to C6.
As the pH value of the reaction mixture increases, it must be assumed that the activation of the olefinic double bonds is enhanced, resulting in autocatalytic amplification. In addition, the decreasing pH value promotes proton-induced elimination reactions of tertiary alcohols. These effects contribute to a diverse product spectrum.
We commonly observed droplet formation in the aqueous phase which can be ascribed to the poor solubility of products and starting materials in water. The oil droplets of different sizes and the use of an aqueous medium in the absence of additives represent a dynamic environment yielding a more diverse number of products compared to the solvolysis conditions with surfactants employed by Bunton et al.10
We also noted that this simple two-phase system was difficult to reproduce with respect to product formation and ratios. In order to achieve more consistent results, reduce the disadvantages of the two-phase nature and achieve improved emulsification, we changed the conditions by conducting the reactions in the presence of ultrasound. In fact, sonochemistry can be utilised to exert mechanical effects, in particular shear forces, on the emulsion/solution by means of microjets and shock waves. In heterogeneous mixtures, these shear forces lead to the fragmentation of oil droplets, which results in a significantly enlarged interfacial area and can thus accelerate interface-dependent reactions.11–13 In the hydrolysis experiments with bromides conducted here, sonochemistry ought to help creating almost homogeneous conditions (through quasi-solvation of the very small oil droplets containing the bromides), as is otherwise easily achievable when linear diphosphate terpene analogues are employed in water.7
Due to the additional energy input from the ultrasonic probe, the reaction mixtures were ice-cooled during sonication. As a consequence, the reaction temperature was significantly lower than in the experiments described above. Nevertheless, we found that bromides 11 and 12 were rapidly converted within 20 minutes of pulsed sonication. Hydrolysis was carried out in a phosphate buffer (pH 7.2) as solvent to prevent corrosion of the ultrasound probe typically caused by the release of HBr. It is noteworthy that the findings obtained are in remarkably close agreement to the results of the acid-assisted hydrolysis of neryl pyrophosphate and geranyl pyrophosphate reported previously.8
Consistent with these experimental observations, ultrasound assisted hydrolysis of neryl bromide (12) mainly yields the cyclised product α-terpineol (24), whereas geranyl bromide (11) primarily generates the linear tertiary alcohol linalool (27) (Fig. 1). In general, the ultrasound accelerated hydrolysis of bromides 11 and 12 gave high conversions. Hence, terpineol (24) could be isolated in 65% yield while linalool (27) was isolated with a yield of 59%. The effect of ultrasound sonication was also evident in that emulsification of the bromides in water was achieved, as the reaction mixture became milky and opaque. Ultrasound-assisted hydrolysis of neryl bromide (12) was carried out using various output control settings on the ultrasonic device. A reduced energy input (output control set to 1) resulted in insufficient emulsification, as evidenced by the persistence of two distinct phases after sonication. These conditions led to a significantly lower conversion of bromides for the same irradiation time. In contrast, increasing the power control significantly improved the conversion efficiency and restored the formation of a homogeneous, milky and opaque emulsion (output control set to 2.5). These observations suggest that a minimum energy input is required to achieve both effective phase emulsification and optimal hydrolytic conversion.
The GC data in general show that dimerisation is now reduced to a minimum. This can be explained by the formation of smaller droplets, which reduces the likelihood of an intermolecular reaction compared to intramolecular cyclisation.
Another observation concerns the fact that the product diversity is significantly reduced. For example, no cyclic ethers could be detected, which is partly due to the lower reaction temperature and shorter hydrolysis times. In addition, the direct hydrolysis products nerol and geraniol were also formed under these conditions. It cannot be completely ruled out that the buffer is of significant importance in suppressing ether formation.
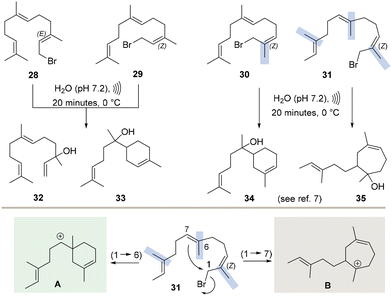
Next, also sesquiterpene bromides were included in this study. In addition to the two natural substrates (E)-farnesyl bromide (28) and (Z)-farnesyl bromide (29), we also chose the two non-natural bromide derivatives 30 and 31 as substrates. Structurally, these differ from bromides 28 and 29 in the positioning of individual methyl groups at the olefinic double bonds (Scheme 3).7 Both bromides carry a (2,3-Z)-alkene like 29, but selected methyl groups are shifted to neighbouring sp2 carbon atoms. A (2,3-Z) configuration should favour cyclisation, while the position of the methyl group on the central olefinic double bond (at C7 or C6) could influence the ring size formed. Iso-farnesol derivatives in which the methyl group at C3 is shifted to C2 are known to be more stable under hydrolytic conditions,7 which may also influence the product outcome.
Unlike geranyl bromide (11) and neryl bromide (12), sesquiterpene bromides 28–31 proved to be unstable under GC-MS analytical conditions. Various signals are found in the chromatograms that do not originate from the hydrolysis experiments, but were generated by the decomposition of the bromides inside the GC itself (Fig. 1). These signals can be used as evidence to determine whether the reactions have already run to completion. The ultrasound-assisted hydrolysis of sesquiterpene bromides also showed a lower conversion rate than that of the corresponding monoterpene bromides. After 20 minutes, products resulting from decomposition in the GC could still be found in the chromatogram.
Regardless, the product pattern remains unchanged when using C15 bromides 28 and 29 (Fig. 1), as nerolidol (32) and α-bisabolol or amylol (33)14 (ressemble C5 elongated linalool (27) and α-terpineol (24)) are formed, however, in different ratios. The (2,3-Z)-configured double bond in 29, in contrast to the (2,3-E)-configured bromide 28, greatly facilitates 1,6-cyclisation and furnishes formation of bisabolol (33) as the main product rather than the linear nerolidol (32) found as major product for 28.
The ultrasound assisted hydrolysis of E-farnesyl bromide 28 was repeated using deionised water instead of phosphate buffer, to shine light on the pH dependency of this method. Except for slight changes in the relative quantities of nerolidol (32) and bisabolol (33), ultrasound hydrolysis of 28 in water provided the same GC chromatogram. After 20 minutes of sonication, the aqueous medium turned acidic (pH = 2), similar to the very first heterogeneous hydrolysis of bromides 11 and 12 in pure water. To minimise corrosion of the ultrasound probe, this was the only hydrolysis approach using solely deionised water. Nevertheless, this additional experiment showcases, that due to the shorter reaction times and changed conditions (cooling instead of heating), the hydrolysis seems to be less pH-sensitive. Hence it is mainly driven by ultrasound sonication. Apart from natural farnesol derived bromides two unnatural derivatives (30 and 31) were also submitted to ultrasound assisted hydrolysis and showed a lower degree conversion, than natural bromides 28 and 29. The observation that shifting the methyl group from C3 to C2 leads to increased hydrolytic stability in aqueous media has already been described in another context.7 Apart from this increased stability the hydrolysis of 30 yielded the bisabolol derivative 34,7 which only differs from the natural sesquiterpene bisabolol (33) in the position of the methyl group at C2. In contrast, the ultrasonically assisted hydrolysis of substrate 31 yields product 357 with a seven-membered ring (Scheme 3).15 The methyl group now positioned at C6 instead of C7 favours 1,7-cyclisation via the more stable carbocation B compared to the cyclohexene-bearing cation A (Scheme 3). In line with previous experiments, the dimerisation of sesquiterpenes 28–31 was reduced to a minimum under ultrasonic conditions. This is evident from the GC chromatograms, which show only negligible signals at the expected retention times for C30 terpenoid core structures (see SI).16
Conclusions
The development of biomimetic systems and catalysts for terpene synthases I and II is a highly topical research field, as these enzyme classes are excellent catalysts for controlling carbocationic cascade reactions. In our view, some studies often lack detailed investigations of the cyclisation behaviour of linear terpenoid precursors in the absence of the systems and catalysts used. This may make it difficult to evaluate the actual performance of these additives. The present work can serve as a guideline for future work on biomimetic systems of terpene synthases and enable easy classification in the overall context. We show here that in aqueous environments, both monoterpene and sesquiterpene precursors do indeed undergo cyclisation reactions. Furthermore, the application of ultrasound in heterogeneous hydrolytic reactions has proven to be a suitable enabling technology17 for achieving a more controlled reaction process.Author contributions
All authors have given approval to the final version of the manuscript. The manuscript was written through contributions of all authors. Jan Luca Budde (conceptualisation, investigation, writing), Andreas Kirschning (conceptualisation, project administration, supervision, writing), and Gerald Dräger (conceptualisation).Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5ob01695a.Acknowledgements
We thank the taxpayers for funding salaries and providing an excellent infrastructure.References
- (a) J. D. Rudolf and C.-Y. Chang, Nat. Prod. Rep., 2020, 37, 425–463 RSC; (b) V. Harms, A. Kirschning and J. S. Dickschat, Nat. Prod. Rep., 2020, 37, 1080–1097 RSC.
- Q. Zhang, L. Catti, L.-D. Syntrivanis and K. Tiefenbacher, Nat. Prod. Rep., 2019, 36, 1619–1627 RSC.
- L.-D. Syntrivanis, I. Némethová, D. Schmid, S. Levi, A. Prescimone, F. Bissegger, D. T. Major and K. Tiefenbacher, J. Am. Chem. Soc., 2020, 142, 5894–5900 CrossRef CAS PubMed.
- Q. Zhang and K. Tiefenbacher, Angew. Chem., Int. Ed., 2019, 58, 12688–12695 CrossRef CAS PubMed.
- Q. Zhang and K. Tiefenbacher, Terpene cyclization catalysed inside a self-assembled cavity, Nat. Chem., 2015, 7, 197–202 CrossRef CAS PubMed.
- A. Jozeliūnaitė, S.-Y. Guo, N. Sakai and S. Matile, Angew. Chem., Int. Ed., 2025, 64, e202417333 CrossRef PubMed.
- J. L. Budde, M. Y. Çay, G. Dräger, A. Hassanin, M. D. Davari and A. Kirschning, ACS Catal., 2025, 15, 8125–8139 CrossRef.
- E. P. Brody and C. D. Gutsche, Tetrahedron, 1977, 723–729 CrossRef CAS.
- C. George-Nascimento, R. Pont-Lezica and O. Cori, Biochem. Biophys. Res. Commun., 1971, 45, 119–124 CrossRef CAS PubMed.
- C. A. Bunton, O. Cori, D. Hachey and J. P. Leresche, J. Org. Chem., 1979, 44, 3238–3244 CrossRef CAS.
- K. S. Suslick, Science, 1990, 247, 1439–1445 CrossRef CAS PubMed.
- S. M. M. Modarres-Gheisari, R. Gavagsaz-Ghoachani, M. Malaki, P. Safarpour and M. Zandi, Ultrason. Sonochem., 2019, 52, 88–105 CrossRef CAS PubMed.
- R. Fernando Martínez, G. Cravotto and P. Cintas, J. Org. Chem., 2021, 86, 13833–13856 CrossRef PubMed.
- The mass spectrum corresponds well with the data for bisabolol in the database, but we cannot completely exclude the formation of the corresponding diastereomer anymol.
- L. H. Zalkow, M. G. Glower Jr., M. M. Gordon and L. T. Gelbaum, J. Nat. Prod., 1980, 43, 382–394 CrossRef CAS.
- We took into account throughout our investigations that GC-MS analysis of sesquiterpenes must be approached with a critical eye, as reactions, particularly rearrangements, can occur under the thermal conditions of the analysis: (a) I. G. Zenkevich and T. A. Kornilova, J. Chromatogr. A, 2025, 1740, 465508 CrossRef CAS PubMed and references cited therein; (b) A. Kirschning, W. Solodenko and K. Mennecke, Chem. – Eur. J., 2006, 12, 5972–5990 CrossRef CAS PubMed.
- A. Kirschning, W. Solodenko and K. Mennecke, Chem. – Eur. J., 2006, 12, 5972–5990 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |