Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Transaminase-triggered synthesis of 2,5-disubstituted pyrrolidines
Aoife
Martin
,
Lisa
Kennedy
,
Ishita
Solanki
,
Patricia
Fleming
,
Julia
Bruno
 ,
Paul
Evans
,
Marianne Bore
Haarr
,
Paul
Evans
,
Marianne Bore
Haarr
 * and
Elaine
O'Reilly
* and
Elaine
O'Reilly
 *
*
School of Chemistry, University College Dublin, Belfield, Dublin 4, Ireland. E-mail: elaine.oreilly@ucd.ie; marianne.haarr@ucd.ie
First published on 4th March 2026
Abstract
2,5-Disubstituted pyrrolidines were synthesised from ketoenone substrates using a transaminase-triggered intramolecular aza-Michael reaction in moderate to good yields. The pyrrolidines were isolated as mixtures of diastereoisomers and a novel epimerisation was developed to isolate cis-2,5-N-nosyl-protected pyrrolidines as the major product in all examples, apart from the ester derivative, with high levels of diastereoselectivity and moderate yields.
Pyrrolidines are ubiquitous in Nature and have been well-represented in US FDA-approved drugs in the last 10 years. The 2,5-disubstituted derivatives also have several synthetic applications in the field of asymmetric organocatalysis.1–4 Given their importance, numerous routes for their synthesis have been reported in the literature, and they often involve starting from an existing pyrrolidine ring,5,6 exploiting 1,3-dipolar cycloadditions7,8 or relying on intramolecular cyclisations.9–13 The intramolecular aza-Michael reaction (IMAMR) is one of the most direct approaches to form a new carbon–nitrogen bond and was first reported over a century ago. The reaction requires the presence of a nitrogen nucleophile and a Michael acceptor in the same molecule and the strategy has been widely adopted for the preparation of N-heterocycles,10,11,14 including asymmetric approaches.15 The availability of amine synthesis enzymes and their unrivalled stereo- and regioselectivity has seen considerable attention devoted to the development of biocatalytic approaches for the preparation of N-heterocycles, including substituted piperidines16–20 and pyrrolidines.21–23 Kroutil and co-workers published a chemoenzymatic route towards the synthesis of 2,6-disubstiuted piperidines from 1,5-diketones that employed an amine transaminase (ATA) to install a chiral amine, followed by diastereoselective hydrogenation using Pd/C.16 However, this synthetic route could not be utilised for the five-membered counterparts due to poor diastereoselectivity in the reduction of the pyrroline. The Turner group reported an efficient cascade for the synthesis of 2,5-disubstituted pyrrolidines that involved the ATA-mediated amination of 1,4-diketone to form a chiral pyrroline, followed by a diastereoselective chemoenzymatic cascade, which utilised a monoamine oxidase (MAO-N) and ammonia borane (NH3·BH3).21 There have also been several recent reports of enzymatic cascades to access these disubstituted heterocycles with many relying on the initial generation of a cyclic imine followed by selective reduction to the pyrrolidine.18 ATA-triggered reactions have been particularly useful in the biocatalytic synthesis of N-heterocycles and involve the functional group interconversion between carbonyls and amines followed by an inter- or intramolecular reaction.24 Our research group first reported an ATA-triggered aza-Michael strategy for the synthesis of disubstituted piperidines in 2016,17 and since then we have developed this methodology further for the synthesis of cyclic β-enaminones and more complex tricyclic N-heterocycles.25,26
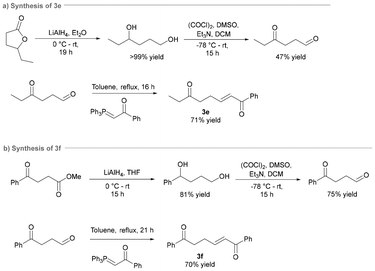
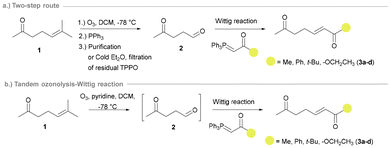
To expand the scope of the ATA-triggered aza-Michael approach for the synthesis of disubstituted pyrrolidines, a panel of six ketoenone substrates were synthesised. The synthetic route to access ketoenones 3a–d in Scheme 1 was reported previously by our group and involved the ozonolysis of 6-methyl-5-hepten-2-one (1) to ketoaldehyde 2, followed by a Wittig reaction with a stabilized ylide to access the target (E)-ketoenone substrate (Scheme 1a).27
 | ||
| Scheme 1 (a) Two-step synthetic route towards the synthesis of the methyl ketoenone substrates previously reported by our group.26,27 (b) In this work, the ketoenones were synthesised using a tandem ozonolysis-Wittig reaction that utilised pyridine as a ‘reducing agent’. | ||
However, yields were lower than expected using this route, likely due to issues arising during the purification of ketoaldehyde 2, owing to the generation of stoichiometric triphenylphosphine oxide (TPPO). For the current work, we accessed the ketoenone substrates by a modified version of this methodology developed in the Dussault group (Scheme 1b).28 The tandem ozonolysis-Wittig reaction gave improved yields, faster reaction times, less purification steps, and avoided the accumulation of the unstable ozonide intermediate. Alternatively, a modified two-step approach can be used that enables the isolation of the common ketoaldehyde intermediate 2 with minimal purification (filtration of recrystallised TPPO), allowing for its application in multiple Wittig reactions.26 Ketoenones 3e and 3f, bearing more sterically demanding substituents, were accessed in good yields via the novel combination of previously reported synthetic steps (Scheme 2).
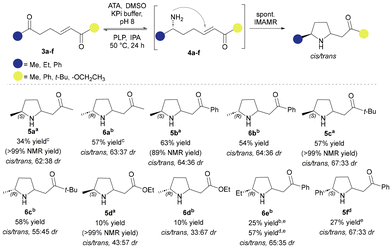
A selection of commercially available ATAs from Codexis® were screened for their ability to accept and aminate ketoenone substrates 3a–f to form aza-Michael intermediates 4a–f, employing a colorimetric assay involving o-xylylenediamine.29 There were several positive hits from this assay (see SI) and a selection of ATAs were tested in small-scale ATA-triggered aza-Michael reactions. Two enantiocomplementary enzymes, (S)-selective ATA256 and (R)-selective ATA025, successfully converted selected ketoenones to their corresponding pyrrolidine products 5a–d and 6a–d (Scheme 3). It was notable that both ATA251 and ATA025 allowed high conversion of 3e to pyrrolidine 6e using 2 equivalents of isopropylamine (IPA). Additionally, ATA251 efficiently converted ketoenone 3f to the corresponding pyrrolidine (5f), which was pleasing, given ATAs traditionally display low activity towards substrates bearing two bulky substituents. Unfortunately, we were unable to access the (R)-enantiomer of 5f as no conversion of ketoenone 3f was observed with ATA025. As expected, NMR analysis of the biotransformation products revealed the presence of diastereoisomers, resulting from the non-selective, spontaneous IMAMR. While many of the NMR signals overlap in the spectra, H2 and H5 were distinct allowing the ratio of diastereoisomers to be identified for each reaction. A clear NOE between H2 and H5, which was absent in the trans-isomers, and a distinct pattern in the order of appearance of each proton revealed that the cis-isomer was present in slight excess (approx. 60![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 40 dr) in all cases, apart from the ester derivatives 5d and 6d, where the trans-isomer was favoured.
40 dr) in all cases, apart from the ester derivatives 5d and 6d, where the trans-isomer was favoured.
Water suppression NMR spectroscopy was used to accurately measure the conversion to (S)-methyl derivatives 5a–d directly from the biotransformation mixture. Maleic acid was used as an internal standard, to circumvent any extraction issues and to gain a complete insight into the progress of the reaction. Interestingly, the cis/trans ratio of pyrrolidine 5b was 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 when water suppression NMR was performed. However, after a basic extraction, isolated 5b showed a bias towards the cis-isomer. The ratio differences are consistent with a dynamic diastereo-equilibration process (see below for more discussion).
1 when water suppression NMR was performed. However, after a basic extraction, isolated 5b showed a bias towards the cis-isomer. The ratio differences are consistent with a dynamic diastereo-equilibration process (see below for more discussion).
Despite reproducibly good to excellent conversions (i.e., NMR yields), isolated yields were low, which suggested that the pyrrolidine products extracted poorly, or that the basic conditions of the work-up were leading to side-reactions/decomposition (although no side-products were detected). Particularly notable in this respect were the biotransformations forming ester derivatives 5d and 6d. Following extraction, the novel disubstituted pyrrolidines synthesised by the ATA-triggered aza-Michael reaction were purified by column chromatography using alumina as the stationary phase, as the compounds undergo decomposition in the presence of silica (Scheme 3).
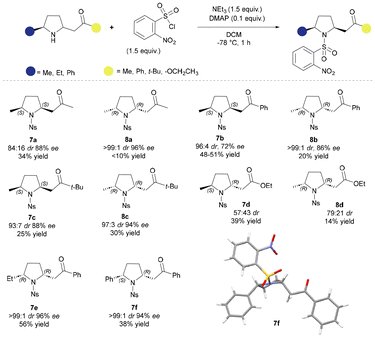
The disubstituted pyrrolidines 5 and 6 were all isolated as inseparable mixtures of diastereoisomers and we were keen to explore the possibility of using chemical or biocatalytic epimerisation approaches. Unlike their 6-membered relatives, the 2,6-disubstituted piperidines,17 attempted epimerisation by stirring in methanol, or exposure to various bases, was unsuccessful and often led to uncharacterised decomposition products (Table S1 in the SI). We hypothesise that this is due to minimal energy difference between the cis and trans pyrrolidine isomers. Previous reports on similar compounds suggested that a protecting group was necessary for base-mediated epimerisation,30,31 but there was no change observed in the cis/trans ratio upon N-propionyl or N-Boc protection of compound 5b, nor after attempted base epimerisation of N-Boc 5b. However, when 5b was reacted with 2-nosyl chloride at room temperature to give the sulfonamide derivative, a change in the diastereomeric ratio (dr) (from 64![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 36 to 80
36 to 80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20) was observed by 1H-NMR analysis of the crude reaction product 7b (Scheme 4). Additionally, these nosyl-protected derivatives were significantly more stable than the unprotected pyrrolidines and could be purified on silica, although separation of the cis/trans-stereoisomers was not possible.
20) was observed by 1H-NMR analysis of the crude reaction product 7b (Scheme 4). Additionally, these nosyl-protected derivatives were significantly more stable than the unprotected pyrrolidines and could be purified on silica, although separation of the cis/trans-stereoisomers was not possible.
We initially speculated that the change in the dr recorded upon nosyl protection was due to a retro-aza-Michael reaction of the product sulfonamide; therefore, we screened several typical epimerisation conditions (Table S1). However, aside from some observed decomposition under certain conditions, there was no notable change in the diastereomeric ratio, i.e., once formed the sulfonamide is not subject to any change in stereochemistry. We, therefore, considered whether the adjustment in diastereomeric ratio upon nosyl protection was due to epimerisation and then a selective protection of pyrrolidine 5b. The nosylation reaction was thus tested at various temperatures (Table S2). When the reaction was performed at 0 °C and −20 °C the 80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 ratio was maintained but at −78 °C we observed a 94
20 ratio was maintained but at −78 °C we observed a 94![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6 ratio of cis/trans isomers. We applied these conditions across our panel of disubstituted pyrrolidines (5a–f and 6a–e) and were able to isolate the cis-nosyl-derivative as the major/exclusive stereoisomer in all examples, apart from the ester derivative 7d (Scheme 4). The relative stereochemistry of these novel compounds was determined by 1H-NMR spectroscopy and the single crystal X-ray structure of 7f confirmed the absolute stereochemistry of the compound.
6 ratio of cis/trans isomers. We applied these conditions across our panel of disubstituted pyrrolidines (5a–f and 6a–e) and were able to isolate the cis-nosyl-derivative as the major/exclusive stereoisomer in all examples, apart from the ester derivative 7d (Scheme 4). The relative stereochemistry of these novel compounds was determined by 1H-NMR spectroscopy and the single crystal X-ray structure of 7f confirmed the absolute stereochemistry of the compound.
We propose that the unprecedented levels of diastereoselectivity in the sulfonamide products observed at −78 °C was due to a rapid background retro-aza-Michael-mediated epimerisation that relied on the selective reaction of 2-nosyl chloride with the cis-isomer. We suggest that the high levels of diastereoselectivity observed at −78 °C, in comparison to the other temperatures, is because the rate of the nosyl protection is significantly slower than the rate of the aza-Michael epimerisation reaction at this temperature. The excellent diastereoselectivities observed for some of the cis-sulfonamide compounds formed in this reaction support the proposed epimerisation mechanism; however, isolated yields were low. Despite these low yields, the sulfonamide products were the major compounds present in the crude 1H-NMR spectra following these reactions, and there were no cis- or trans-pyrrolidine starting materials remaining. While we are unable to detect any significant impurities by NMR spectroscopy, there is a mass balance issue most likely originating from unstable starting material. This was evidenced by a measured discrepancy in the concentration of model substrate 5b added to the reaction (see SI section 13 for details).
Unsurprisingly, deprotection of 7b (cis![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) trans = 96
trans = 96![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4) led to a mixture of diastereoisomers of 5b consistent with the original ratio (cis
4) led to a mixture of diastereoisomers of 5b consistent with the original ratio (cis![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) trans = 64
trans = 64![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 36) observed prior to nosyl-protection and reinforced the theory that a retro-aza-Michael reaction was mediating the epimerisation. We also reduced 7b to the corresponding alcohol using L-Selectride® (see SI for more info.), and while the reduction was not particularly stereoselective (2
36) observed prior to nosyl-protection and reinforced the theory that a retro-aza-Michael reaction was mediating the epimerisation. We also reduced 7b to the corresponding alcohol using L-Selectride® (see SI for more info.), and while the reduction was not particularly stereoselective (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 dr) there was no change in the diastereomeric ratio upon deprotection of this alcohol, indicating that the ketone moiety was necessary for epimerisation and again supported the retro-aza-Michael route.
1 dr) there was no change in the diastereomeric ratio upon deprotection of this alcohol, indicating that the ketone moiety was necessary for epimerisation and again supported the retro-aza-Michael route.
These results support our proposed epimerisation mechanism for the synthesis of cis-nosyl-protected pyrrolidines. Additionally, this work demonstrates that both diastereoisomers of the unprotected pyrrolidine can interconvert via a retro-aza-Michael reaction. In conclusion, we have expanded the scope of the ATA-triggered aza-Michael reaction for the synthesis of 2,5-disubstiuted pyrrolidines. Owing to the unique chemical reactivity of pyrrolidine compounds they could not be epimerised as readily as their 6-membered counterparts in their unprotected form, but a novel epimerisation strategy was uncovered involving a retro-aza-Michael reaction and a selective reaction between 2-nosyl chloride and the cis-disubstituted pyrrolidine at −78 °C. We propose that this epimerisation is not limited to 2-nosyl chloride and that future work could involve the development of similar reactions with other protecting groups if the resolution (protection) step was significantly slower than the retro-aza-Michael reaction, allowing high levels of diastereoselectivity to be achieved.
Conflicts of interest
There are no conflicts of interest to declare.Data availability
The supporting data has been provided as part of the supplementary information (SI), and includes additional data gathered that is not discussed in detail in the main paper.Supplementary information is available. See DOI: https://doi.org/10.1039/d6ob00264a.
CCDC 2506209 contains the supplementary crystallographic data for this paper.32
Acknowledgements
We acknowledge financial support from Taighde Éireann – Research Ireland (GOIPG/2023/2755), the UCD School of Chemistry Sir Walter Hartley Fellowship (AM), and support from a Research Ireland Laureate Award (IRCLA/2022/2810). We also acknowledge RI grants supporting NMR (12/RI/2341) and mass spectrometry (18/RI/5702) facilities, and equipment and facilities funded by HEA PRTLI Cycle 3. We thank Dr Jimmy Muldoon for recording HRMS.References
- G. Li Petri, M. V. Raimondi, V. Spanò, R. Holl, P. Barraja and A. Montalbano, Top. Curr. Chem., 2021, 379, 34 CrossRef CAS PubMed.
- P. Melchiorre, M. Marigo, A. Carlone and G. Bartoli, Angew. Chem., Int. Ed., 2008, 47, 6138–6171 CrossRef CAS PubMed.
- A. Claraz, G. Sahoo, D. Berta, Á. Madarász, I. Pápai and P. M. Pihko, Angew. Chem., 2016, 128, 679–683 CrossRef.
- S. Singh, R. Kumar, N. N. Dubey and C. Appayee, Chem. Commun., 2024, 60, 8768–8771 RSC.
- K. R. Campos, A. Klapars, J. H. Waldman, P. G. Dormer and C.-y. Chen, J. Am. Chem. Soc., 2006, 128, 3538–3539 CrossRef CAS.
- M. He, L. J. Xie, L. Chen and S. Xu, ACS Catal., 2024, 14, 18701–18707 CrossRef CAS.
- J. M. Longmire, B. Wang and X. Zhang, J. Am. Chem. Soc., 2002, 124, 13400–13401 CrossRef CAS PubMed.
- A. S. Gothelf, K. V. Gothelf, R. G. Hazell and K. A. Jørgensen, Angew. Chem., Int. Ed., 2002, 41, 4236–4238 CrossRef CAS PubMed.
- Y. Lazib, J. G. Naves, A. Labande, P. Dauban and T. Saget, ACS Org. Inorg. Au, 2023, 3, 120–129 CrossRef CAS PubMed.
- M. Sánchez-Roselló, J. L. Aceña, A. Simón-Fuentes and C. del Pozo, Chem. Soc. Rev., 2014, 43, 7430–7453 RSC.
- R. W. Bates, W. Ko and V. Barát, Org. Biomol. Chem., 2020, 18, 810–829 RSC.
- S. Munnuri, A. M. Adebesin, M. P. Paudyal, M. Yousufuddin, A. Dalipe and J. R. Falck, J. Am. Chem. Soc., 2017, 139, 18288–18294 CAS.
- P. Laohapaisan, I. Roy and D. A. Nagib, Chem. Catal., 2024, 4, 101149 CAS.
- Y. X. Song and D. M. Du, Adv. Synth. Catal., 2021, 363, 4667–4694 CrossRef CAS.
- M. Sánchez-Roselló, M. Escolano, D. Gaviña and C. Del Pozo, Chem. Rec., 2022, 22, e202100161 CrossRef PubMed.
- R. C. Simon, B. Grischek, F. Zepeck, A. Steinreiber, F. Belaj and W. Kroutil, Angew. Chem., Int. Ed., 2012, 51, 6713–6716 CrossRef CAS PubMed.
- J. Ryan, M. Šiaučiulis, A. Gomm, B. Macia, E. O'Reilly and V. Caprio, J. Am. Chem. Soc., 2016, 138, 15798–15800 CrossRef CAS PubMed.
- S. P. France, S. Hussain, A. M. Hill, L. J. Hepworth, R. M. Howard, K. R. Mulholland, S. L. Flitsch and N. J. Turner, ACS Catal., 2016, 6, 3753–3759 CrossRef CAS.
- S. Parisotto, M. Blangetti and C. Prandi, Org. Biomol. Chem., 2025, 23, 9380–9387 RSC.
- R. C. Simon, F. Zepeck and W. Kroutil, Chem. – Eur. J., 2013, 19, 2859–2865 CrossRef CAS PubMed.
- E. O'Reilly, C. Iglesias, D. Ghislieri, J. Hopwood, J. L. Galman, R. C. Lloyd and N. J. Turner, Angew. Chem., Int. Ed., 2014, 53, 2447–2450 CrossRef PubMed.
- B. Z. Costa, J. L. Galman, I. Slabu, S. P. France, A. J. Marsaioli and N. J. Turner, ChemCatChem, 2018, 10, 4733–4738 CrossRef CAS.
- C. M. Heckmann and C. E. Paul, JACS Au, 2023, 3, 1642–1649 CrossRef CAS PubMed.
- A. Martin, I. Solanki, M. B. Haarr and E. O’Reilly, Eur. J. Org. Chem., 2023, 26, e20230085 CrossRef.
- F. Taday, J. Ryan, S. P. Argent, V. Caprio, B. Maciá and E. O'Reilly, Chem. – Eur. J., 2020, 26, 3729–3732 CrossRef CAS.
- A. O'Connell, M. B. Haarr, J. Ryan, X. Xu, A. Martin, S. N. Smith, N. Elghobashi-Meinhardt, P. Fleming, B. Maciá, V. Caprio and E. O'Reilly, Angew. Chem., Int. Ed., 2025, 64, e202422584 CrossRef PubMed.
- H. Eastman, J. Ryan, B. Maciá, V. Caprio and E. O'Reilly, ChemCatChem, 2019, 11, 3760–3762 CrossRef CAS.
- R. Willand-Charnley and P. H. Dussault, J. Org. Chem., 2013, 78, 42–47 CrossRef CAS PubMed.
- A. P. Green, N. J. Turner and E. O'Reilly, Angew. Chem., Int. Ed., 2014, 53, 10714–10717 CrossRef CAS.
- C. Enkisch and C. Schneider, Eur. J. Org. Chem., 2009, 5549–5564 CrossRef CAS.
- S. Fustero, L. Herrera, R. Lázaro, E. Rodríguez, M. A. Maestro, N. Mateu and P. Barrio, Chem. – Eur. J., 2013, 19, 11776–11785 CrossRef CAS PubMed.
- CCDC 2506209: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q3xg7.
| This journal is © The Royal Society of Chemistry 2026 |