Open Access Article
Open Access ArticleTungsten-catalyzed continuous-flow oxidation of sulfides to sulfones with hydrogen peroxide
Yasushi
Okamoto
,
Kazuhiro
Matsumoto
 ,
Jun-Chul
Choi
,
Jun-Chul
Choi
 ,
Yoshihiro
Kon
,
Yoshihiro
Kon
 and
Ken-ichi
Fujita
and
Ken-ichi
Fujita
 *
*
National Institute of Advanced Industrial Science and Technology (AIST), Tsukuba Central 5, 1-1-1 Higashi, Tsukuba, Ibaraki 305-8565, Japan. E-mail: k.fujita@aist.go.jp
First published on 2nd March 2026
Abstract
Continuous-flow oxidation of sulfides to sulfones was achieved with hydrogen peroxide (H2O2) using a catalyst column, which was simply packed with a mixture of oxodiperoxotungstate-2,2′-bipyridine and commercially available activated carbon. Sulfones were continuously produced in more than 90% yield for up to 160 h by passing an acetonitrile solution of sulfides and H2O2 through the catalyst column. Activated carbon played a key role in immobilizing the tungsten catalyst while the other supports, Celite or silica gel did not have such an ability to catch the catalyst. Selective oxidation to sulfone took place under continuous-flow conditions even though the sulfide substrates contained other oxidizable functional groups, such as olefinic double bond, hydroxy, pyridyl and amino functional groups.
Introduction
Sulfones are useful synthetic intermediates due to their ability to stabilize α-carbanions, and sulfonyl moieties have been incorporated into various structures, such as agrochemicals, pharmaceuticals and polymers.1 In general, sulfones can be synthesized through the oxidation of the corresponding sulfides.2 Although oxidation of the sulfide to the corresponding sulfone has been performed using a wide variety of traditional oxidizing agents, such as KMnO4,3 CrO3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 and RuO4,5 there remains a demand for environmentally and economically viable synthetic routes that do not require expensive and toxic reagents. From these points of view, hydrogen peroxide (H2O2) is a superior oxidizing agent, as water is the sole coproduct after the oxidation.6 Recently, H2O2 in combination with a metal catalyst, such as tungsten,7 iron,8 cobalt,9 or titanium10 has been shown to be effective for the oxidation of sulfides to sulfones. For recovery of the oxidation catalyst, various processes have been proposed, with immobilization of metal catalysts on a solid support being among the most promising.
4 and RuO4,5 there remains a demand for environmentally and economically viable synthetic routes that do not require expensive and toxic reagents. From these points of view, hydrogen peroxide (H2O2) is a superior oxidizing agent, as water is the sole coproduct after the oxidation.6 Recently, H2O2 in combination with a metal catalyst, such as tungsten,7 iron,8 cobalt,9 or titanium10 has been shown to be effective for the oxidation of sulfides to sulfones. For recovery of the oxidation catalyst, various processes have been proposed, with immobilization of metal catalysts on a solid support being among the most promising.
During the last few decades, organic reactions under flow conditions have attracted considerable interest in both academia and industry.11 Especially in the field of oxidation processes, flow reactions have numerous advantages over the corresponding batch reactions, including environmental compatibility due to the closed vapor-tight system, efficiency due to the small reaction volume, and relatively safer reaction conditions.12 Doherty et al. previously reported the continuous-flow oxidation of sulfides to sulfones using a fine-tuned organopolymer-immobilized peroxophosphotungstate catalyst, which was prepared from an imidazolium-based styrene monomer via multiple steps.13 They demonstrated that sulfones could be continuously provided over 8 h by the flow of sulfides and H2O2 through a catalyst-packed column. Subsequently, continuous-flow synthesis of sulfones was also achieved using electrochemical flow processes14 and microreactor systems.15 The main issue of continuous-flow reaction is the requirement of a tightly immobilized and highly efficient catalyst. With respect to oxidation catalysts, oxodiperoxotungsten and oxodiperoxomolybdenum complexes have recently been shown to be efficient.16 In 2007, Li et al. reported that an oxodiperoxotungstate-1,10-phenanthroline complex efficiently catalyzed the oxidation of dibenzothiophene to the corresponding sulfone in an ionic liquid by a batch method.17
Recently, we reported the continuous-flow selective oxidation of sulfides to sulfoxides catalyzed by iron oxide on activated carbon with H2O2.18 The iron oxide on activated carbon catalyst was selective toward sulfoxide formation and no remarkable increase in sulfone formation was observed, even though the amount of iron-activated carbon composite catalyst increased. It should be also emphasized that oxidation could be successfully performed with commercially available and less expensive materials; iron salts, H2O2 and activated carbon. Building on our previous finding that activated carbon is an effective support for metal catalysts, we explored activated carbon-supported tungsten-based catalysts. In this paper, we report the continuous-flow oxidation of sulfides to sulfones catalyzed by oxodiperoxotungstate-2,2′-bipyridine (WO(O2)2-bpy) immobilized on activated carbon.
Results and discussion
Catalytic activity of a tungsten-based catalyst for continuous-flow oxidation of sulfides
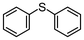
Immobilized metal–ligand catalysts have often been prepared using metallic species and solid-supported ligands. For example, peroxotungstate ions (WO42−) supported on mesoporous silica were prepared from Na2WO4 and ammonium salt-functionalized mesoporous silica by an ion-exchange process.19 On the other hand, activated carbon has recently been used as a solid support without specific ligands.20 In our previous report, we showed that iron salts were introduced onto activated carbon and that the resulting composite catalyzed the oxidation of sulfides to sulfoxides in continuous-flow reactions.18 In this study, we further explored the potential of activated carbon by investigating flow oxidations using activated carbon as a solid support for other metal catalysts. We focused on oxodiperoxotungstate-2,2′-bipyridine (WO(O2)2-bpy) 1 as shown in Fig. 1 which was prepared from tungsten oxide WO3 or the polyoxotungstate anion PW12O40− and 2,2′-bipyridine and has shown catalytic activity in the epoxidation of olefins.21,22In continuation of our studies of sulfide oxidation, we performed the continuous-flow oxidation of sulfides to sulfones with H2O2 by utilizing the tungsten complex 1 immobilized on activated carbon as a catalyst (Fig. 2). Preparation of the catalyst was straightforward. Powdered tungsten complex 1 was mixed with activated carbon, which was purchased from Strem Chemicals, Inc, and the mixture was packed into a glass column (5 mm inner diameter × 50 mm length). An acetonitrile solution of diphenyl sulfide 2a (0.1 M) containing H2O2 (2.2 eq. per 2a) was introduced into a flow reactor equipped with the packed column.23 The reaction was conducted at 40 °C under a back pressure of 0.2 MPa, with the flow rate set to 0.05 mL min−1. The molar hourly space velocity (SVmol) was 16 h−1,24 which was calculated by multiplying the flow rate by the concentration of sulfide 2a in acetonitrile, dividing by the amount of WO(O2)2 in the column, and correcting for time units.
Fig. 2a shows the time-course curve of the continuous-flow oxidation after reaching a steady state, in which the total yields of sulfide 2a, sulfoxide 3a, and sulfone 4a were over 95%, indicating that no other by-products were produced.25 The effluent was collected every hour. The sample labeled “4 h” in the graph corresponds to the fraction collected from 3 h to 4 h (hereafter, “the 4 h fraction”).
In this flow reaction, sulfide 2a was completely consumed. The gradual increase of yield of sulfone 4a accompanying the gradual decrease of yield of sulfoxide 3a was observed from 4 h to 20 h. After 10 h, over 90% yield of sulfone 4a was continuously produced up to the 110 h fraction without a decrease in the yield of 4a.26,27 At the early stage of flow reaction, fixation of catalyst 1 on activated carbon takes place. In addition, induction time may be needed to reach equilibrium stage between starting material 2a, sulfoxide 3a, sulfone 4a and activated carbon. Therefore, it was suggested that some time was essential to reach maximum catalytic efficiency.
On the other hand, the yield of sulfone 4a decreased slightly by the end of the 164 h fraction, accompanied by a slight increase in sulfoxide 3a. To clarify the reason for the decrease in catalytic activity, we performed the following experiments. First, the 51 h to 100 h fractions were collected and subjected to quantitative ICP-MS analysis to evaluate tungsten leaching. Only 0.17% of tungsten, relative to the initial amount of tungsten complex 1 loaded in the column, was detected in the collected fractions. Therefore, the decrease in catalytic activity does not appear to be attributable to the leaching of the tungsten catalytic species.
Secondly, a washout experiment was performed. After flow oxidation for 164 h, the column was washed with acetonitrile at a flow rate of 0.25 mL min−1 at 40 °C for 2 h. An acetonitrile solution of diphenyl sulfide 2a and H2O2 was again introduced into the flow reactor for 24 h. As a result, the average yield of sulfone 4a recovered to 97%, and the average yield of sulfoxide 3a was suppressed to 2% (Fig. 2b). These results suggest that the gradual decrease in the yield of sulfone 4a after 100 h (Fig. 2a) was caused by adsorption of sulfur-containing compounds on the catalytic species, namely deactivation of the catalyst. Overall, the results indicate that the decreased yield of sulfone 4a at the end stage of Fig. 2a is unlikely to be due to leaching of tungsten catalytic species from the column.
Role of activated carbon
In order to investigate the effect of activated carbon in the reaction, other solid supports were examined. The catalyst column was prepared using the same procedure, but with activated carbon replaced by Celite or silica gel. Continuous-flow oxidation reaction of diphenyl sulfide 2a using a WO(O2)2-bpy 1/Celite column was carried out for 20 h. In contrast to the 1/activated carbon catalyst, the yield of sulfone 4a decreased significantly over time (Fig. 3a). When a mixture of tungsten complex 1 and silica gel was used as a catalyst, the yield of sulfone 4a also decreased after 7 h (Fig. 3b), although the decrease was slower than in the WO(O2)2-bpy 1/Celite case. These unique results suggest that tungsten catalytic species were deactivated and/or leached from the column. As these results sharply contrast with those obtained using the WO(O2)2-bpy 1/activated carbon catalyst, we further investigated differences in tungsten leaching among the solid supports. | ||
| Fig. 3 Continuous-flow oxidation of diphenyl sulfide 2a with H2O2 using a mixture of WO(O2)2-bpy 1 and a solid support. (a) Celite; (b) silica gel. | ||
To confirm whether leached tungsten species from each solid support exhibited catalytic activity in the oxidation of sulfide 2a, we subsequently examined sulfide oxidation by a batch method using fractions collected from the flow reactions (Fig. 2a, 3a and 3b), as shown in Fig. 4. Three fractions that gave a high yield of diphenyl sulfone 4a were transferred to the reaction vessels (Fig. 2a: the 41 h–43 h fractions; Fig. 3a: the 4 h–6 h fractions, Fig. 3b: the 5 h–7 h fractions). Diphenyl sulfide 2a (final concentration, 0.1 M) and H2O2 (2.2 eq. per 2a) were added to each vessel, and the solution was stirred at 40 °C for 24 h. The yields of sulfone 4a and sulfoxide 3a were calculated based on the initial amount of sulfide 2a, which was completely converted to sulfoxide 3a and sulfone 4a under flow oxidation.
As shown in the time-course curve for oxidation of diphenyl sulfide 2a in Fig. 4, the yield of sulfone 4a did not increase when the collected fraction from the continuous-flow reaction using the WO(O2)2-bpy 1/activated carbon catalyst was subjected to further oxidation. On the other hand, a time-dependent increase in the yield of sulfone 4a was observed for cases when fractions collected from continuous-flow reactions using either WO(O2)2-bpy 1/Celite or WO(O2)2-bpy 1/silica gel were subjected to further oxidation. In particular, the increase in the yield of sulfone 4a was pronounced for the WO(O2)2-bpy 1/Celite system. These increases can be explained by catalytic oxidation by leached tungsten species present in the collected fractions. These results further indicate again that the tungsten catalytic species is firmly immobilized on activated carbon, but not on Celite or silica gel.
Continuous-flow oxidation of various sulfides
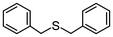
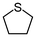
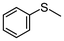
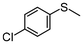
Encouraged by the results that WO(O2)2-bpy 1/activated carbon is a suitable oxidation catalyst, we then performed continuous-flow oxidation of various sulfides 2 catalyzed by WO(O2)2-bpy 1/activated carbon (Table 1). Average yields were calculated after reaching a steady state, in which the total yields of sulfide 2, sulfoxide 3, and sulfone 4 reached 90%. The time windows used for yield calculation are indicated as “fraction time” in Table 1. The procedures for catalyst preparation and the flow reaction were the same as in Fig. 2a. For diphenyl sulfide 2a (Table 1, entry 1), the data in Fig. 2a are summarized in Table 1, indicating that diphenyl sulfide 2a continuously provided sulfone 4a in a 95% average yield, with no remaining sulfide 2a, and with formation of 3% sulfoxide 3a. In the oxidation of aliphatic sulfides (dibutyl sulfide 2b, dibenzyl sulfide 2c, dodecyl methyl sulfide 2d, and thiacyclopentane 2e), the sulfides were completely consumed and the corresponding sulfones 4b–4e were continuously produced in nearly quantitative yields (Table 1, entries 2–5). Methyl phenyl sulfide 2f and the chloro- or methoxy- substituted analogues 2g and 2h also produced the corresponding sulfones 4f, 4g, and 4h in excellent average yields (>99%, >99% and 98%, respectively; Table 1, entries 6–8).| Entry | Substrate | Fraction time (h) | 3 average yieldb (%) | 4 average yieldb,c (%) | |
|---|---|---|---|---|---|
| a Reaction conditions: ca. 0.019 mmol 1, ca. 400 mg activated carbon packed into a 50 mm catalyst column (5 mm inner diameter); a 0.1 M solution of 2 in acetonitrile; a 35% (w/w) aqueous solution of H2O2 (2.2 eq. per 2); 1-tert-butyl-4-methoxybenzene as an internal standard (0.25–0.5 eq. per 2); 40 °C; flow rate: 0.05 mL min−1. b a, b, e, f, g, h, i: Determined by GC analysis using an internal standard method; c, d, j, k, l: Determined by 1H NMR analysis using an internal standard method. c Isolated yields: 4a 90% (100 h–106 h fraction); 4b 93% (15 h–20 h fraction); 4c 91% (14 h–20 h fraction); 4d 92% (5 h–9 h fraction); 4e 96% (13 h–18 h fraction); 4f 90% (16 h–24 h fraction); 4g 92% (16 h–24 h fraction); 4h 93% (16 h–24 h fraction); 4i 84% (4 h–16 h fraction); 4j 95% (16 h–24 h fraction); 4k 96% (16 h–24 h fraction); 4l 89% (8 h–22 h fraction). | |||||
| 1 |
|
2a | 3–164 | 3 | 95 |
| 2 |
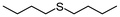
|
2b | 3–24 | 1 | 97 |
| 3 |
|
2c | 2–24 | 0 | >99 |
| 4 |
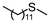
|
2d | 2–24 | 0 | >99 |
| 5 |
|
2e | 2–24 | 0 | 98 |
| 6 |
|
2f | 2–24 | 0 | >99 |
| 7 |
|
2g | 2–24 | 0 | >99 |
| 8 |
|
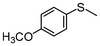
2h | 2–24 | 0 | 98 |
| 9 |
|
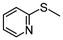
2i | 3–24 | 11 | 86 |
| 10 |
|
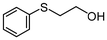
2j | 3–24 | 0 | 98 |
| 11 |
|
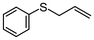
2k | 2–24 | 0 | >99 |
| 12 |
|
2l | 4–24 | 5 | 91 |
Subsequently, we performed continuous-flow oxidation of sulfides bearing easily oxidizable functional groups. In the oxidation of methyl 2-pyridyl sulfide 2i, the corresponding sulfoxide 3i and sulfone 4i were produced without N-oxidation in 11% and 86% average yields, respectively (Table 1, entry 9). These results suggest that the catalytic activity of the WO(O2)2-2,2′-bipyridine complex 1 was sustained without N-oxidation of the 2,2′-bipyridine ligand during flow oxidation. The oxidation of sulfides bearing other functional groups—2-(phenylthio)ethanol 2j and phenyl 2-propenyl sulfide 2k—gave the corresponding sulfones 4j and 4k nearly quantitatively without oxidation of the hydroxy group or the olefinic double bond (98% and >99% yield; entries 10 and 11, respectively). These results could be attributed to the short residence time under continuous-flow oxidation of sulfides (ca. 14.5 min).
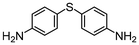
The flow oxidation of bis(4-aminophenyl) sulfide 2l efficiently afforded bis(4-aminophenyl) sulfone 4l (the antibacterial agent Dapsone) in a 91% average yield with 5% sulfoxide 3l (Table 1, entry 12). It was recently reported that sulfone 4l is synthesized by the oxidation of sulfide 2l with H2O2 using silicopolyoxotungstovanadate as a catalyst in a batch method.28 There are many reports and patents of synthetic procedures for Dapsone. One representative example starts from the coupling of 4-aminothiophenol and 4-chloro-nitrobenzene, followed by sulfide oxidation with sodium tungstate and reduction of the nitro groups.29 The continuous-flow synthesis of bis(4-aminophenyl) sulfone 4l by the oxidation of sulfide 2l would be a potential alternative to large-scale industrial synthesis of Dapsone due to its simple and safe operation.
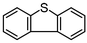
To evaluate the catalytic activity of WO(O2)2-bpy 1/activated carbon, continuous-flow oxidation of dibenzothiophene 2m was performed in comparison with WO(O2)2-1,10-phenanthroline·H2O (WO(O2)2-phen·H2O) 5 (Fig. 5). The catalyst WO(O2)2-phen·H2O 5 has been reported to effectively catalyze the oxidation of dibenzothiophene 2m to the corresponding sulfone 4m in an ionic liquid under batch conditions.17 Dibenzothiophene is known to be a refractory sulfur compound for removal from fuels by catalytic oxidation.30 Thus, we performed continuous-flow oxidation of 2m using WO(O2)2-bpy 1/activated carbon as the catalyst. The results are summarized in Table 2, together with those obtained using WO(O2)2-phen·H2O 5/activated carbon as the catalyst. When WO(O2)2-bpy 1/activated carbon catalyst was used, oxidation of dibenzothiophene 2m proceeded continuously, producing the corresponding sulfone 4m in a 92% average yield with 7% sulfoxide 3m (Table 2, entry 1). In contrast, when WO(O2)2-phen·H2O 5 catalyst was used, sulfone 4m was continuously produced in 67% yield, with 14% unreacted sulfide 2m and 19% sulfoxide 3m (Table 2, entry 2). These results suggest that WO(O2)2-bpy 1/activated carbon shows somewhat higher catalytic activity than WO(O2)2-phen·H2O 5/activated carbon under continuous-flow conditions. Therefore, continuous-flow oxidation utilizing the WO(O2)2-bpy 1/activated carbon catalyst, which maintains catalytic activity, may provide an efficient method for desulfurization.
| Entry | Substrate | Tungsten complex | Fraction time (h) | 2m average yieldb (%) | 3m average yieldb (%) | 4m average yieldb (%) | |
|---|---|---|---|---|---|---|---|
| a Reaction conditions are the same as those indicated in Table 1. b Determined by 1H NMR analysis using an internal standard method. c Isolated yield of 4m: 83% (5 h–20 h fraction). | |||||||
| 1 |
|
2m | 1 | 5–24 | 0 | 7 | 92c |
| 2 | 5 | 4–24 | 14 | 19 | 67 | ||
Conclusions
Continuous-flow oxidation of sulfides to sulfones was achieved with H2O2 using catalyst columns packed with a mixture of oxodiperoxotungstate-2,2′-bipyridine 1 and activated carbon. Sulfones were continuously produced by simply passing an acetonitrile solution of sulfides and H2O2 through the column in more than 90% yield for up to 160 h. The flow oxidation preferentially produced sulfones from sulfides, even though other functional groups, such as olefinic double bond, hydroxy, pyridyl and amino functional groups, were present in the substrates. It was also clarified that activated carbon was a superior solid support that firmly immobilized the tungsten catalytic species, whereas Celite and silica gel did not.Considering that the oxidation with H2O2 provides only H2O as a coproduct and less than 1% (w/w) H2O2 in the feed solution is sufficient for this continuous-flow oxidation of sulfides, the method we developed is an environmentally benign and safe system. In addition, preparation of the catalyst column is straightforward: the column is packed with a mixture of the tungsten complex and activated carbon. Therefore, the developed method is suitable for the continuous-flow oxidation of sulfides to sulfones.
Experimental
Materials and equipment
Oxodiperoxotungstate-2,2′-bipyridine 122a and oxodiperoxotungstate-1,10-phenanthroline·H2O 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 31 were synthesized according to the literature. Activated carbon (carbon powder, 99+%) was purchased from Strem Chemicals, Inc. Celite (Celite® No.545) was purchased from Fuji Film Wako Chemical Corporation. Silica gel (Silica Gel 60 (spherical), 40–50 μm diameter) was purchased from Kanto Chemical Co., Inc. Reagent grade acetonitrile was purchased from Fuji Film Wako Chemical Corporation. A 35% (w/w) H2O2 aqueous solution was purchased from Tokyo Chemical Industry Co. All commercial chemicals were used for all experiments without further treatment.
31 were synthesized according to the literature. Activated carbon (carbon powder, 99+%) was purchased from Strem Chemicals, Inc. Celite (Celite® No.545) was purchased from Fuji Film Wako Chemical Corporation. Silica gel (Silica Gel 60 (spherical), 40–50 μm diameter) was purchased from Kanto Chemical Co., Inc. Reagent grade acetonitrile was purchased from Fuji Film Wako Chemical Corporation. A 35% (w/w) H2O2 aqueous solution was purchased from Tokyo Chemical Industry Co. All commercial chemicals were used for all experiments without further treatment.
The flow synthesis system consisted of a degassing apparatus (Gastorr AG-42; Flom Inc.), an intelligent plunger pump (UI22-110P; Flom Inc.), a column flow reactor (MCR-1000; EYELA), a back pressure regulator (BPR-1000; EYELA), and a fraction collector (CHF100AA; Advantec Toyo Kaisha). An empty glass column (L = 50 mm, ID = 5 mm, and filter pore size = 10 μm) was purchased from EYELA.
For determination of the product yield during the continuous-flow reaction, a Shimadzu gas chromatography system GC-2014 (FID detector) was used. In some cases where GC analysis was not suitable, yields were quantified by 1H NMR analysis using a Bruker AV600 spectrometer.
Continuous-flow oxidation of sulfide 2 to sulfone 4: general procedure (Fig. 2, 3 and Tables 1, 2)
The tungsten complex 1 or 5 (0.019 mmol) and activated carbon (ca. 400 mg) were mixed and packed in a glass reactor column (L = 50 mm and ID = 5 mm), and the column was placed in the flow reaction system. Then, an acetonitrile solution containing sulfide 2 (0.1 M), 1-tert-butyl-4-methoxybenzene (an internal standard; 0.25–0.5 eq. per 2) and a 35% (w/w) aqueous H2O2 (2.2 eq. per 2), which was cooled at 0 °C to suppress the oxidation of 2 before introduction into the catalyst column, was passed through the column with a flow rate of 0.05 mL min−1 at 40 °C.The effluent was collected every hour. In the case of 2d, 2l, and 2m as substrates, the effluent was directly collected into a dichloromethane solution without using a fraction collector due to the high crystallinity of these products. Yields were determined by GC analysis using an internal standard. In some cases where GC analysis was not suitable, 1H NMR analysis was applied to determine yields, as shown in Tables 1 and 2 (note b), also using an internal standard.
Isolation of sulfone 4a from fractions of continuous-flow oxidation: typical procedure (Table 1, entry 1)
The fractions collected from 100 h to 106 h were combined to obtain a mixed acetonitrile solution. Dodecane was added as an internal standard, and GC analysis was performed. The amount of 1-tert-butyl-4-methoxybenzene contained in the mixed solution was determined by an internal standard method using the GC peak area of dodecane. From the amount of 1-tert-butyl-4-methoxybenzene, the amount of diphenyl sulfide 2a as a starting material was calculated.After the addition of sodium thiosulfate aqueous solution to the mixed acetonitrile solution, the mixture was evaporated under reduced pressure, and the residue was purified by column chromatography (hexane/ethyl acetate = 9/1–7/3 as eluent) on silica gel to give sulfone 4a (348.2 mg, 1.595 mmol) in a 90% chemical yield.
Oxidation of diphenyl sulfide 2a to diphenyl sulfone 4a using collected fractions by a batch method: general procedure (Fig. 4)
Fractions giving a high yield of diphenyl sulfone 4a under flow oxidation of diphenyl sulfide 2a (Fig. 2a, 3a and 3b) were transferred to reaction vessels (Fig. 2a (1/activated carbon): the 41 h–43 h fractions; Fig. 3a (1/Celite): the 4 h–6 h fractions; Fig. 3b (1/silica gel): the 5 h–7 h fractions). Diphenyl sulfide 2a and H2O2 (2.2 eq. per 2a) were added to each fraction, and the mixture was diluted with acetonitrile to give a 0.1 M solution of diphenyl sulfide 2a. The solution was stirred at 40 °C. Yields of sulfone 4a were determined by 1H NMR analysis with reference to an internal standard at 1 h, 2 h, 6 h and 24 h.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: time course curves of oxidation of 2a and 1H NMR spectra of products. See DOI: https://doi.org/10.1039/d6ob00125d.Acknowledgements
This article is based on results obtained from a project, JPNP19004, subsidized by the New Energy and Industrial Technology Development Organization (NEDO), Japan.References
- (a) G. H. Whitham, Organosulfur Chemistry, Oxford University Press, New York, 1995 Search PubMed; (b) S. Oae, Organic Chemistry of Sulfur, Plenium Press, New York, 1977 CrossRef; (c) C. J. M. Stirling, Organic sulphur chemistry: Structure, mechanism, and synthesis, Butterworths, London/Boston, 1975 Search PubMed; (d) S. Patai, Z. Rappoport and C. J. M. Stirling, The Chemistry of Sulphones and Sulphoxides, Wiley, New York, 1988 CrossRef.
- (a) N.-W. Liu, S. Liang and G. Manolikakes, Synthesis, 2016, 1939–1973 CAS; (b) D. J. Procter, J. Chem. Soc., Perkin Trans. 1, 2000, 835–871 RSC.
- (a) A. Shaabani, A. Rahmati, M. Sharifi, J. M. Rad, B. Aghaaliakbari, E. Farhangi and D. G. Lee, Monatsh. Chem., 2007, 138, 649–651 Search PubMed; (b) A. Shaabani, P. Mirzaei, S. Naderi and D. G. Lee, Tetrahedron, 2004, 60, 11415–11420 Search PubMed; (c) H. Rheinboldt and E. Giesbrecht, J. Am. Chem. Soc., 1946, 68, 973–978 CrossRef CAS.
- L. M. Yagupolskii, N. V. Kondratenko and V. P. Sambur, Synthesis, 1975, 721–723 CrossRef CAS.
- C. Djerassi and R. R. Engle, J. Am. Chem. Soc., 1953, 75, 3830–3840 CrossRef.
- (a) R. Goyal, O. Singh, A. Agrawal, C. Samanta and B. Sarkar, Catal. Rev.: Sci. Eng., 2022, 64, 229–285 CrossRef CAS; (b) Y. Kon, J. Jpn. Pet. Inst., 2017, 60, 159–169 CrossRef CAS; (c) R. Noyori, M. Aoki and K. Sato, Chem. Commun., 2003, 1977–1986 RSC; (d) G. Strukul, Catalytic Oxidations with Hydrogen Peroxide as Oxidant, Kluwer, 1992 CrossRef.
- (a) M. Assis, A. F. Gouveia, L. K. Ribeiro, M. A. Ponce, M. S. Churio, O. N. Oliveira Jr, L. H. Mascaro, E. Longo, R. Llusar, E. Guillamón and J. Andrés, Appl. Catal., A, 2023, 652, 119038 CrossRef CAS; (b) C. Saux, C. L. Marchena, L. R. Pizzio and L. B. Pierella, J. Porous Mater., 2016, 23, 947–956 CrossRef CAS; (c) K. Sato, M. Hyodo, M. Aoki, X.-Q. Zheng and R. Noyori, Tetrahedron, 2001, 57, 2469–2476 CrossRef CAS.
- (a) A. Rostami, S. Moradi and Z. Shokri, C. R. Chimie, 2018, 21, 80–87 CrossRef CAS; (b) E. Baciocchi, M. F. Gerini and A. Lapi, J. Org. Chem., 2004, 69, 3586–3589 CrossRef CAS PubMed; (c) A. Marques, M. Marin and M. F. Ruasse, J. Org. Chem., 2001, 66, 7588–7595 CrossRef CAS.
- Z. Chen, C. Liu, J. Liu, J. Li, S. Xi, X. Chi, H. Xu, I.-H. Park, X. Peng, X. Li, W. Yu, X. Liu, L. Zhong, K. Leng, W. Huang, M. J. Koh and K. P. Loh, Adv. Mater., 2020, 32, 1906437 CrossRef CAS.
- (a) P. Cruz, M. Fajardo, I. der Hierro and Y. Pérez, Catal. Sci. Technol., 2019, 9, 620–633 RSC; (b) M. Iwamoto, Y. Tanaka, J. Hirosumi, N. Kita and S. Triwahyono, Microporous Mesoporous Mater., 2001, 48, 271–277 CrossRef CAS.
- (a) G. Gambacorta, J. S. Sharley and I. R. Baxendale, Beilstein J. Org. Chem., 2021, 17, 1181–1312 CrossRef CAS; (b) K. Masuda, T. Ichitsuka, N. Koumura, K. Sato and S. Kobayashi, Tetrahedron, 2018, 74, 1705–1730 CrossRef CAS.
- (a) L. Wan, M. Jiang, D. Cheng, M. Liu and F. Chen, React. Chem. Eng., 2022, 7, 490–550 RSC; (b) J. P. Colomer, M. Traverssi and G. O.-Mansilla, J. Flow Chem., 2020, 10, 123–138 CrossRef CAS; (c) H. P. L. Gemoets, Y. Su, M. Shang, V. Hessel, R. Luque and T. Noël, Chem. Soc. Rev., 2016, 45, 83–117 RSC.
- (a) S. Doherty, J. G. Knight, M. A. Carroll, A. R. Clemmet, J. R. Ellison, T. Backhouse, N. Holmes, L. A. Thompson and R. A. Bourne, RSC Adv., 2016, 6, 73118–73131 Search PubMed; (b) S. Doherty, J. G. Knight, M. A. Carroll, J. R. Ellison, S. J. Hobson, S. Stevens, C. Hardacre and P. Goodrich, Green Chem., 2015, 17, 1559–1571 Search PubMed.
- (a) N. Amri and T. Wirth, J. Org. Chem., 2021, 86, 15961–15972 CrossRef CAS PubMed; (b) G. Laudadio, W. de Smet, L. Struik, Y. Cao and T. Noël, J. Flow Chem., 2018, 8, 157–165 CrossRef PubMed; (c) G. Laudadio, N. J. W. Straathof, M. D. Lanting, B. Knoops, V. Hessel and T. Noël, Green Chem., 2017, 19, 4061–4066 RSC.
- (a) C. A. Shukla, B. Udaykumar, Y. Saisivanarayana, A. Ismaili, T. Haripriya, M. M. Shinde, S. Neti, M. Uppada, V. Eda, S. Sen and S. Oruganti, J. Flow Chem., 2022, 12, 1–7 Search PubMed; (b) F. Mangiavacchi, L. Crociani, L. Sancineto, F. Marini and C. Santi, Molecules, 2020, 25, 2711 Search PubMed.
- J.-M. Bŕegeault, M. Vennat, L. Salles, J.-Y. Piquemal, Y. Mahha, E. Rriot, P. C. Bakala, A. Atlamsani and R. Thouvenot, J. Mol. Catal. A: Chem., 2006, 250, 177–189 CrossRef.
- W. Zhu, H. Li, X. Jiang, Y. Yan, J. Lu and J. Xia, Energy Fuels, 2007, 21, 2514–2516 CrossRef CAS.
- Y. Okamoto, K. Matsumoto, J.-C. Choi, Y. Kon and K. Fujita, Org. Biomol. Chem., 2025, 23, 8542–8552 RSC.
- B. Karimi and M. Khorasani, ACS Catal., 2013, 3, 1657–1664 CrossRef CAS.
- S. Ordóñez, F. V. Díez and H. Sastre, Appl. Catal., B, 2001, 31, 113–122 CrossRef.
- T. R. Amarante, M. M. Antunes, A. A. Valente, F. A. A. Paz, M. Pillinger and I. S. Gonçalves, Inorg. Chem., 2015, 54, 9690–9703 CrossRef CAS PubMed.
- (a) G. Hidalgo, G. B.-Consiglio, K. Robeyns, M. Devillers and E. M. Gaigneaux, Inorg. Chem., 2023, 62, 8576–8588 Search PubMed; (b) G. Hidalgo, G. B.-Consiglio, M. Devillers and E. M. Gaigneaux, Mol. Catal., 2022, 528, 112479 CAS.
- When using 2.0 eq. of H2O2, the flow oxidation of sulfide 2a did not completely proceed to produce sulfoxide 3a and sulfone 4a in 18% and 80% average yields over 24 h, respectively (Fig. SI S1a). Thus, 2.2 eq. of H2O2 was used as standard conditions.
- In the case of decreased amount of the catalyst where the catalyst WO(O2)2-bpy 1 was used under SVmol value of 32 which corresponds to about half amount of 1, average yield of sulfone 4a was 78% over 24 h (Fig. SI S1b).
- At the initial stage of continuous-flow reactions, no compound was detected due to the dead volume of tube and reactor equipment (0 h–1 h and the material balance (indicated as “Total” in Fig. 2a was subsequently fluctuated due to the initial specific absorption of the reaction compounds (2a, 3a and 4a and an internal standard on activated carbon (1 h–3 h).
- When using a catalyst column packed with activated carbon alone, sulfoxide 3a and sulfone 4a were continuously produced in 12% and 3% average yields over 24 h, respectively.
- When methanol was used as the alternative solvent in continuous-flow oxidation of sulfide 2a, 4% of sulfone 4a was continuously produced and the average yield of sulfoxide 3a decreased from 51% to 16% over 24 h.
- R. A. Frenzel, V. Palermo, A. G. Sathicq, A. M. Elsharif, R. Luque, L. R. Pizzio and G. P. Romanelli, Microporous Mesoporous Meter., 2021, 310, 110584 Search PubMed.
- Lundbeck Pharmaceuticals, U.S. Pat., 2009/7531694, 2009 Search PubMed.
- H. Y. Lü, J. B. Gao, Z. X. Jiang, F. Jing, Y. X. Yang, G. Wang and C. J. Li, J. Catal., 2006, 239, 369–375 Search PubMed.
- W. Zhu, H. Li, X. He, H. Shu and Y. Yan, J. Chem. Res., 2006, 774–775 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |