Open Access Article
Open Access ArticleA supramolecular host with a cavitand core and four cholate side arms
Beijun
Cheng
 *a,
Marcos D.
García
*a,
Marcos D.
García
 b,
Yan
Tian
a,
Carlos
Peinador
b,
Yan
Tian
a,
Carlos
Peinador
 b,
Yuezhi
Cui
b,
Yuezhi
Cui
 a,
Qingqing
Lu
a,
Yuexia
Qin
c,
Zhaohua
Hou
c and
Angel E.
Kaifer
a,
Qingqing
Lu
a,
Yuexia
Qin
c,
Zhaohua
Hou
c and
Angel E.
Kaifer
 *d
*d
aSchool of Chemistry and Chemical Engineering, Qilu University of Technology (Shandong Academy of Sciences), Jinan, 250353, China. E-mail: bchengsd@163.com
bDepartamento de Química and Centro Interdisciplinar de Química e Bioloxía (CICA). Facultad de Ciencias, Universidade da Coruña, 15071 A Coruña, Spain
cSchool of Food Science and Engineering, Qilu University of Technology (Shandong Academy of Sciences), Jinan, 250353, China
dDepartment of Chemistry, University of Miami, Coral Gables, FL 33124, USA. E-mail: akaifer@miami.edu
First published on 19th February 2026
Abstract
Copper(I)-catalyzed azide–alkyne cycloaddition (CuAAC) was employed to synthesize a new cavitand (1) having four cholate groups covalently connected to the cavitand core. CuAAC between a tetrapropargyl cavitand 2 and 3α-azido-cholic acid 3 led to the isolation of pure cavitand 1, which was fully characterized by the usual complement of spectroscopic techniques. While its solubility was limited in pure aqueous media, the complexation of three 4,4′-bipyridinum (viologen) guests could be investigated, using square wave voltammetric (SWV) techniques, in a mixture of H2O/DMSO (5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 v/v) also containing 40 mM sodium tetraborate as the supporting electrolyte and further experimental evidence supporting the formation of a complex between cavitand 1 and V2+ (viologen) with a 1
2 v/v) also containing 40 mM sodium tetraborate as the supporting electrolyte and further experimental evidence supporting the formation of a complex between cavitand 1 and V2+ (viologen) with a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 stoichiometry was obtained using ITC and electrospray ionization mass spectrometry (ESI-MS). Computational studies at the GFN2-xTB/ALPB(water) semiempirical level of theory revealed that the cavitand adopts a compact globular structure in solution, while docking effectively with methylviologen as a guest without forming an inclusion complex.
1 stoichiometry was obtained using ITC and electrospray ionization mass spectrometry (ESI-MS). Computational studies at the GFN2-xTB/ALPB(water) semiempirical level of theory revealed that the cavitand adopts a compact globular structure in solution, while docking effectively with methylviologen as a guest without forming an inclusion complex.
Introduction
Cavitands are bowl-shaped compounds that constitute an important class of supramolecular hosts. Initially prepared and termed by Cram,1,2 they offer a number of possibilities for functionalization. Cram's definition of cavitands1 as ‘synthetic organic molecules with enforced cavities large enough to complex complementary organic compounds or ions’ is general enough to include many classes of hosts. However, most supramolecular chemists use the term ‘cavitands’ specifically to designate host compounds derived from the acid-catalyzed condensation of aldehydes and resorcinols, as pioneered by Cram's group. This reaction initially produces a group of macrocycles termed ‘resorcinarenes’, whose phenolic OH groups can be further interconnected by suitable bifunctional reagents, such as CH2BrCl or CH2Br2, to yield the bowl-shaped cavitands.3 Most cavitands are built from the initial condensation of four aromatic (resorcinol) groups, although larger cavitands have also been reported.4 Since the internal cavity afforded by simple cavitands is rather small, elongation of the cavity via functionalization of its upper rim is a common method to synthetically access host compounds with the ability to complex larger molecules.5,6 Numerous carcerands, hemicarcerands and other hosts based on cavitands have been prepared.7 The Gibbs group8,9 has reported extensively on a cavitand derivative functionalized with eight carboxylate units and its interesting dimerization and binding properties in aqueous media. Our own group has explored the functionalization of cavitands with redox-active 4,4′-bipyridinium (viologen) groups10 and Fréchet-type dendrons.11As a result of its facial amphiphilic nature, anionic cholate molecules aggregate in aqueous solution to form small micellar assemblies with hydrophobic cavities.12–14 Previously, we investigated the electrochemistry of a series of viologen derivatives with varying amphiphilic character in cholate micellar media.15 Our experimental data were consistent with the interaction of all surveyed viologens with cholate assemblies. Out of the three oxidation states presented by viologens [dication (V2+), cation radical (V+˙) and neutral species (V)] cholate micellar assemblies preferentially stabilized the cation radical form.15 Therefore, we decided to prepare a cavitand functionalized with four identical cholate groups connected to its upper rim (see Fig. 1) and investigate its binding interactions with several viologen derivatives. Our main goal was to compare these results with those obtained in cholate micellar media in an attempt to shed some light on the types of dynamic cavities existing in these media, since we initially thought that, given its structure, the new tetracholate cavitand would exhibit a better defined and more stable cavity, forced by the attachment of the four cholate groups to the upper cavitand rim.
As guests in this preliminary investigation we selected three viologen derivatives, namely methylviologen (MV2+), butylviologen (BV2+) and ethyl-heptylviologen (EHV2+). The first guest is the least hydrophobic on account of the two simple methyl substituents on the dicationic 4,4′-bipyridinium nucleus. The other two viologen guests are both more hydrophobic but possess different distributions of the hydrophobic groups (alkyl chain substituents) around the viologen nucleus (Fig. 2).
Results and discussion
Synthesis
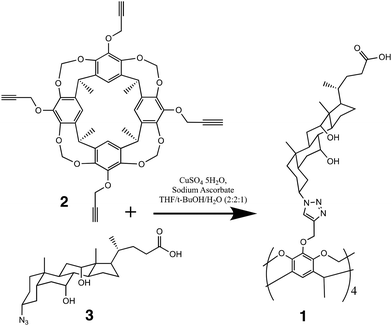
The synthesis of the target tetracholate cavitand was carried out using click chemistry, that is, copper(I)-catalyzed azide–alkyne cycloaddition16,17 (CuAAC) between the tetrapropargyl cavitand 218–22 and 3α-azido-cholic acid 323–26 (Scheme 1). Very briefly, 2 was prepared by hydroxylation of the known tetrabromo cavitand followed by treatment in refluxing acetone with propargyl bromide in the presence of K2CO3 to yield cavitand 2. Synthesis of the azide derivative 3 started by esterification of cholic acid with methanol, followed by mesitylation of the alcohol group on position 3, treatment with NaN3 to give the azide derivative of the methyl ester and, finally, hydrolysis of the ester to yield the 3α-azido-cholic acid 3. The CuAAC reaction between 2 and five equivalents of 3 was carried out in a THF/t-BuOH/H2O (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2
2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) solvent mixture in the presence of CuSO4 and sodium ascorbate. After purification, the isolated tetracholate cavitand (61% yield) was fully characterized using spectroscopic methods (see Experimental section). The viologen guests were prepared as iodide or bromide salts using the same methods already reported by our group.15
1) solvent mixture in the presence of CuSO4 and sodium ascorbate. After purification, the isolated tetracholate cavitand (61% yield) was fully characterized using spectroscopic methods (see Experimental section). The viologen guests were prepared as iodide or bromide salts using the same methods already reported by our group.15
Preliminary binding studies in solution
Not unexpectedly, cavitand 1 had limited solubility in aqueous media, even when solutions with pH > 7 were used to foster aqueous solubility through the deprotonation of the cholic acid groups (14−). After assessing the solubility of 1 in various solvent mixtures, we settled on a 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 (v/v) mixture of H2O and DMSO (containing 40 mM sodium borate for pH control) for our guest–host binding solution studies. Because of their redox properties and our previous experience with these compounds we chose a series of representative bipyridinium (viologen) derivatives as potential guests for binding interactions with cavitand 14−. The electrochemical behavior of viologen dications (V2+) consists of two fast one-electron reduction steps, which lead successively to the radical cation (V+˙) and the neutral species (V). Since these two reduction steps diminish the initial positive charge on the dication, the resulting viologen species are less water-soluble, typically leading to stronger interactions with hydrophobic cavities that might be present in the media or possible precipitation on the working electrode surface in electrochemical experiments.15
2 (v/v) mixture of H2O and DMSO (containing 40 mM sodium borate for pH control) for our guest–host binding solution studies. Because of their redox properties and our previous experience with these compounds we chose a series of representative bipyridinium (viologen) derivatives as potential guests for binding interactions with cavitand 14−. The electrochemical behavior of viologen dications (V2+) consists of two fast one-electron reduction steps, which lead successively to the radical cation (V+˙) and the neutral species (V). Since these two reduction steps diminish the initial positive charge on the dication, the resulting viologen species are less water-soluble, typically leading to stronger interactions with hydrophobic cavities that might be present in the media or possible precipitation on the working electrode surface in electrochemical experiments.15
Unfortunately, the limited solubility of cavitand 14− in aqueous media made impossible the use of 1H NMR experiments to investigate its possible host binding interactions with any of the viologen dications; and the UV-vis absorption region of cavitand 14− overlaps with those of MV2+, BV2+ and EHV2+. Therefore, we jumped directly into Square Wave Voltammetric (SWV) experiments which allow the assessment of electrochemical behavior changes experienced by the viologen dications, at concentrations as low as 0.1 mM, in the presence of similar concentrations of tetracholate cavitand. These changes offer insight on the host–guest binding interactions taking place in the solution phase. To see if any aggregates form, we measured the UV-vis dilution experiments and the absorbance of the host is perfectly linear and only starts showing some curvature at concentrations of 0.15 mM or higher (Fig. S19). Of course, at these concentrations the absorbance is already so high (A > 1.0) that errors caused by stray light and other photometric factors affect the measured values. In any instance, we did not find any evidence of aggregation at host concentrations of 0.1 mM or lower, as indicated by the linearity of the absorbance vs. concentration plot in this concentration range.
The SWV behavior of MV2+ in H2O![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DMSO (5
DMSO (5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, v/v, containing 40 mM sodium borate) consists of two cathodic peaks centered at potentials of −0.568 V and −0.976 V vs. Ag/AgCl. The first peak corresponds to the one-electron reduction of MV2+ to its radical cation (MV+˙) and the second to the one-electron reduction of the radical cation to the neutral species (MV). Addition of 1.0 equiv. of cavitand 14− leads to decreased current levels for both SWV peaks, which is consistent with the association of MV2+ with 14−, as the increased molecular weight of the supramolecular complex will result in slower diffusion rates to the electrode surface and, thus, lower currents. The presence of 1.0 equiv. of cavitand also shifts the first reduction peak to a more negative potential value (−0.580 V) while the second reduction peak shifts in the opposite direction to −0.964 V. These potential changes are small but perfectly measurable, considering that our potential values are affected by a typical error margin of ±0.003 V. Similar, although less pronounced, changes take place upon addition of an additional equiv. of cavitand. The SWV behavior of MV2+ in the absence and presence of cavitand 14− is shown in Fig. 3 and the peak potential values compiled for all three viologens are given in Table 1.
2, v/v, containing 40 mM sodium borate) consists of two cathodic peaks centered at potentials of −0.568 V and −0.976 V vs. Ag/AgCl. The first peak corresponds to the one-electron reduction of MV2+ to its radical cation (MV+˙) and the second to the one-electron reduction of the radical cation to the neutral species (MV). Addition of 1.0 equiv. of cavitand 14− leads to decreased current levels for both SWV peaks, which is consistent with the association of MV2+ with 14−, as the increased molecular weight of the supramolecular complex will result in slower diffusion rates to the electrode surface and, thus, lower currents. The presence of 1.0 equiv. of cavitand also shifts the first reduction peak to a more negative potential value (−0.580 V) while the second reduction peak shifts in the opposite direction to −0.964 V. These potential changes are small but perfectly measurable, considering that our potential values are affected by a typical error margin of ±0.003 V. Similar, although less pronounced, changes take place upon addition of an additional equiv. of cavitand. The SWV behavior of MV2+ in the absence and presence of cavitand 14− is shown in Fig. 3 and the peak potential values compiled for all three viologens are given in Table 1.
| Guest | + 0 equiv. cavitand 14− | + 1 equiv. cavitand 14− | + 2 equiv. cavitand 14− |
|---|---|---|---|
| MV2+ | −0.568/−0.976 | −0.580/−0.964 | −0.584/−0.960 |
| BV2+ | −0.520/−0.904 | −0.544/−0.920 | −0.548/−0.940 |
| EHV2+ | −0.512/−0.900 | −0.536/−0.960 | −0.528/−0.952 |
SWV peak potentials are equal to half-potential values (E1/2), which are considered an excellent approximation to the corresponding formal potentials (E°′).27 Therefore, the shifts on the measured peak potentials contain information on the relative thermodynamic stability of the three redox states of the viologen guest. For instance, with MV2+ the difference between the first and the second peak potentials decreases as the cavitand concentration increases, reflecting the decreased thermodynamic stability of the radical cation (MV+˙) compared to the dication and neutral states in the presence of the cavitand. Interestingly, this effect on the relative stability of the radical cation is the opposite to what was observed in the presence of cholate micelles.15 The more hydrophobic viologen guest BV2+ shows different SWV peak potential behavior (Fig. S20), with both peaks shifting to more negative values as the cavitand concentration increases. Finally, in the case of EHV2+ (Fig. S21), both peak potentials shift to more negative values in the presence of 1 equiv. of cavitand, but this trend partially reverses itself upon addition of an extra equiv. of cavitand host. These cavitand-induced peak potential shifts indicate that all redox states are relatively stabilized by the presence of cavitand 14−. In all cases, the addition of cavitand leads to decreased current levels, reflecting the formation of more slowly diffusing supramolecular complexes.
While the detailed interpretation of the observed peak potential shifts for the three viologen guests is complicated at this time, we notice that the shifts elicited by 1 equiv. of cavitand are much larger than those resulting from the addition of one additional equivalent. This suggests that the equilibrium concentration of complex is close to saturation when mixing viologen guest and cavitand 14− both at equal concentrations of 0.1 mM.
In order to obtain the corresponding association equilibrium constant (K), we carried out isothermal titration calorimetry (ITC) experiments. The K value obtained was 1.28 × 105 M−1 for tetracholate cavitand complexation with MV2+. Thermodynamic parameters obtained by ITC: (1) ΔH° = −36.91 kJ mol−1, (2) −TΔS° = 7.77 kJ mol−1 and (3) ΔG° = −29.14 kJ mol−1. These parameters indicate that the binding process was primarily driven by enthalpy.
Further experimental evidence supporting the formation of a complex between cavitand 1 and V2+ was obtained using electrospray ionization mass spectrometry. We detected signals for complexes of [1·MV2+]2+, [1·BV2+]2+ and [1·EHV2+]2+ (Fig. S22, S24 and Fig. 5), which confirm the formation of stable [1·V2+]2+ complexes with 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 stoichiometry. The major peak in the spectrum (Fig. 4) of a solution containing cavitand 1 and EHV2+ appears at the m/z ratio of 1413.3253, which can be assigned to the 1
1 stoichiometry. The major peak in the spectrum (Fig. 4) of a solution containing cavitand 1 and EHV2+ appears at the m/z ratio of 1413.3253, which can be assigned to the 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 complex of [1·EHV2+]2+ and the simulated isotopic distribution of the complex matches well with the experimental isotopic distribution. The same consistent isotopic distributions between the simulated one and the observed one were observed for [1·MV2+]2+ (Fig. S23) and [1·BV2+]2+ (Fig. S25).
1 complex of [1·EHV2+]2+ and the simulated isotopic distribution of the complex matches well with the experimental isotopic distribution. The same consistent isotopic distributions between the simulated one and the observed one were observed for [1·MV2+]2+ (Fig. S23) and [1·BV2+]2+ (Fig. S25).
 | ||
Fig. 4 ITC profiles for the tetracholate cavitand complexation with MV2+ at 298.15 K in H2O![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DMSO (5 DMSO (5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 v/v) containing 40 mM sodium tetraborate. 2 v/v) containing 40 mM sodium tetraborate. | ||
Computational studies
To better understand the structure of the tetracholate cavitand in aqueous solution, we employed the well-established Conformer–Rotamer Ensemble Sampling Tool (CREST), which explores the conformational space through metadynamics-based sampling in combination with genetic algorithms.28 Conformational searches were performed using standard settings together with the generic force field GFF-xTB,29 and the resulting structural ensembles were subsequently re-optimized and ranked at the efficient GFN2-xTB semiempirical level of theory (2nd generation Geometry, Frequency, Noncovalent, eXtended Tight-Binding method).30 In all cases, solvation effects in water were included using the analytically linearized Poisson–Boltzmann (ALPB) model, an implicit solvation approach parametrized for the xTB methods.31Conformational analysis of the tetracholate cavitand 14− led to the preferred, energy-minimized conformation represented in Fig. 6, which shows a globular structure with two of the cholate units blocking the cavity of the cavitand core and the other two wrapped around the macrocycle. This conformation (1p4−) can be rationalized by the inability of the cholate units to expose their hydrophilic hydroxyl-containing faces to the aqueous environment, an orientation imposed by their connection to the macrocycle through the α position of the C3 atom. Therefore, the compact, globular structure is adopted to minimize the solvation of hydrophobic surfaces by decreasing as much as possible the solvent-accessible surface area, allowing as well for the establishment of multiple stabilizing intramolecular hydrogen bonds.
In order to assess the preferred structure of the potential supramolecular species formed by the interaction of 1p4− and MV2+, docking studies were carried out at the GFN2-xTB/ALPB(water) level, using the recently-developed automated Interaction Site Screening (aISS) algorithm.32 This approach systematically explores possible binding geometries through an initial grid-based scan and force-field screening, followed by refinement and final optimization at the GFN2-xTB level. The structure proposed by the aISS workflow for the lowest-lying aggregate was found to position the guest on a lateral surface of the cavitand core, with one of the carboxylate cholate groups near enough to develop electrostatic interaction with the cationic guest, and without a significant distortion of the initial preferred conformation 1p4− for the host (Fig. 7).
Finally, the free energy of association was computed at the GFN2-xTB/ALPB(water) level of theory using the preferred conformation 1p4− as reference for the host. The obtained value for the association reveals that this process is clearly exergonic, with ΔG° = −10.00 kcal mol−1, and does not require the formation of an inclusion complex to achieve remarkable thermodynamic stability in the final adduct. In this case, the calculated K value is 2.16 × 107 M−1, which is around two orders of magnitude higher than the K value determined from our ITC measurements. The computational work was carried out in pure water, which may explain the difference in thermodynamic stability with the experimentally obtained values in H2O![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DMSO (5
DMSO (5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 v/v). In any instance, the considerable stability of the complex is well established both by our experimental and computational results, which constitutes an interesting and rare finding for an external, non-inclusion host–guest complex.
2 v/v). In any instance, the considerable stability of the complex is well established both by our experimental and computational results, which constitutes an interesting and rare finding for an external, non-inclusion host–guest complex.
Experimental
Materials
All solvents and chemicals were commercially available and used as received, except N-bromosuccinimide, which was freshly recrystallized before use. Water was purified using an Ulupure water purification system (Chengdu, Sichuan). Cavitand 2 and azido derivative 3 were prepared according to published procedures. The viologens were prepared by treatment of 4,4′-bipyridine with the appropriate iodo- or bromo-alkanes, as reported previously by our group. Synthetic schemes and other details are provided in the SI. 1H NMR (400 MHz) and 13C NMR (100 MHz) spectra were recorded on a JEOL JNM-ECZL400S Spectrometer. HRMS (ESI) was obtained on a Thermo Scientific Q Exactive Focus Orbitrap LC-MS/MS System.Synthesis of tetracholate cavitand (1)
Cavitand 2 (80.0 mg, 0.1 mmol) and azido derivative 3 (217.0 mg, 0.5 mmol) were dissolved in 8.0 mL of a 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2
2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture of THF/t-BuOH/H2O (v/v), followed by the addition of CuSO4·5H2O (12.48 mg, 0.05 mmol) and sodium ascorbate (50.49 mg, 0.255 mmol). The solution was stirred in an oil bath at 60 °C for 24 h. The solvent was removed under vacuum. The crude product was dissolved in THF (10 mL), followed by the addition of H2O (3 mL). The solution was extracted by EtOAc (10 mL). The extracted organic layer was washed with H2O (3 mL × 2) and dried over MgSO4. The solvent was removed under vacuum, then the solid material was washed with EtOAc to afford 156.2 mg (61.4%) of the final product as a white solid.
1 mixture of THF/t-BuOH/H2O (v/v), followed by the addition of CuSO4·5H2O (12.48 mg, 0.05 mmol) and sodium ascorbate (50.49 mg, 0.255 mmol). The solution was stirred in an oil bath at 60 °C for 24 h. The solvent was removed under vacuum. The crude product was dissolved in THF (10 mL), followed by the addition of H2O (3 mL). The solution was extracted by EtOAc (10 mL). The extracted organic layer was washed with H2O (3 mL × 2) and dried over MgSO4. The solvent was removed under vacuum, then the solid material was washed with EtOAc to afford 156.2 mg (61.4%) of the final product as a white solid.
1H NMR (400 MHz, (CD3)2SO): δ 11.92 (s, 4H, COOH), 8.09 (s, 4H, H-triazole), 7.46 (s, 4H, ArH), 5.96 (d, 4H, outer of OCH2O), 4.94 (d, 8, OCH2-triazole), 4.80 (q, 4H, CH3CH), 4.29 (bs, 8H, H-3β and inner of OCH2O), 4.16 (d, 4H, OH-12α), 4.11 (d, 4H, OH-7α), 3.80 (s, 4H, H-12β), 3.63 (s, 4H, H-7β), 2.86–0.96 (m, 96H), 1.82 (d, 12H, CH3CH), 0.93 (d, 12H, H-21), 0.91 (s, 12H, H-19), 0.60 (s, 3H, H-18). 13C NMR (100 MHz, (CD3)2SO): δ 174.94, 147.19, 143.72, 142.78, 139.83, 122.80, 115.70, 99.40, 71.10, 66., 66.68, 66.15, 60.43, 46.19, 45.83, 41.90, 41.42, 36.98, 35.48, 35.04, 34.51, 31.34, 30.87, 30.82, 28.53, 27.25, 26.28, 22.77, 22.58, 16.98, 15.82, 12.33.
HRMS (ESI/Q-TOF) m/z: [M + H]+ calcd for C144H197N12O28 2542.4360; found 2543.4460.
Methods
The electrochemical experiments were recorded in a single-compartment glass cell, using a CH Instruments potentiostat (Austin, TX). Typically, the cell was fitted with a glassy carbon disk working electrode (0.071 cm2), platinum auxiliary electrode, a Ag/AgCl reference electrode, and a nitrogen inlet Teflon tubing. Nitrogen gas was bubbled through the solution to remove dissolved oxygen gas. During the voltametric scans, nitrogen gas was maintained above the solution to prevent the redissolution of atmospheric oxygen. The working electrode was polished on a soft felt surface covered with an aqueous slurry of alumina (0.05 μm), and rinsed extensively with purified water.Conclusions
We have prepared a new cavitand-based molecule (1), having four cholate groups covalently attached to the cavitand core via triazole units. The final molecule was synthesized using the copper(I)-catalyzed azide–alkyne cycloaddition (CuAAC) between the tetrapropargyl cavitand 2 and 3α-azido-cholic acid 3 (Scheme 1). The new cavitand showed limited solubility in aqueous media. However, SWV electrochemical measurements showed that the tetracholate cavitand 14− effectively binds three viologen guests in a 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 (v/v) mixture of H2O and DMSO (containing 40 mM sodium borate for pH control). And ESI-MS shows direct evidence for the complexes between the cavitand 1 and the viologen guests. ITC experiments yielded a K value of 1.28 × 105 M−1 for the formation of the 1·MV2+complex. Computational studies showed that the cavitand host adopts a globular conformation in aqueous media to minimize the exposure and solvation of hydrophobic surfaces. Docking studies with the methylviologen guest reveal the formation of a very stable complex (calculated ΔG° = −10 kcal mol−1 in water and experimental ΔG° = −7.0 kcal mol−1 in H2O
2 (v/v) mixture of H2O and DMSO (containing 40 mM sodium borate for pH control). And ESI-MS shows direct evidence for the complexes between the cavitand 1 and the viologen guests. ITC experiments yielded a K value of 1.28 × 105 M−1 for the formation of the 1·MV2+complex. Computational studies showed that the cavitand host adopts a globular conformation in aqueous media to minimize the exposure and solvation of hydrophobic surfaces. Docking studies with the methylviologen guest reveal the formation of a very stable complex (calculated ΔG° = −10 kcal mol−1 in water and experimental ΔG° = −7.0 kcal mol−1 in H2O![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DMSO, 5
DMSO, 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 v/v) in which the guest attaches itself to one the external surfaces of the cavitand core. Therefore, formation of an inclusion complex is not necessary to achieve considerable stabilization of the final supramolecular adduct, which is likely to be further stabilized by the globular conformation adopted by the host.
2 v/v) in which the guest attaches itself to one the external surfaces of the cavitand core. Therefore, formation of an inclusion complex is not necessary to achieve considerable stabilization of the final supramolecular adduct, which is likely to be further stabilized by the globular conformation adopted by the host.
A recent publication has reported the preparation of a related compound containing three cholate groups.33 These results combine well with our findings on the conformational complexity of cavitand 14− as well as the relatively surprising structure of its highly stable adduct with methylviologen, and fuel our interest to continue the exploration of more aqueous soluble related host compounds with multiple cholate groups. Potential strategies to improve water solubility by introducing hydrophilic groups (e.g., sulfonate, carboxylate) on the feet of the cavitand core are under consideration.
Author contributions
The manuscript was written through contributions of all authors. All authors have given approval to the final version of the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: synthetic details, 1H, 13C NMR spectra and mass spectra of relevant compounds. SWV behavior of butylviologen and ethyl-heptylviologen. ESI-MS spectra of [1·MV2+]2+ and [1·BV2+]2+. Computational study details. See DOI: https://doi.org/10.1039/d6ob00175k.Acknowledgements
The support of this research by the Shandong Provincial Natural Science Foundation, China (to B. C., Grant ZR2020QB040), Qilu University of Technology Start-up Funds (to B. C.) and the US National Science Foundation (to A. E. K., Grant CHE-1412455) is gratefully acknowledged. M. D. G. and C. P. are thankful for the funding received from the MCIN/AEI/10.13039/501100011033 (PID2022-137361NB-I00) and the Consellería de Cultura, Educación e Universidade da Xunta de Galicia (ED431C 2022/39).References
- D. J. Cram and J. M. Cram, Container Molecules and Their Guests, The Royal Society of Chemistry, Cambridge, 1994 Search PubMed.
- D. J. Cram, Cavitands: Organic Hosts with Enforced Cavities, Science, 1983, 219, 1177 CrossRef CAS PubMed.
- P. Timmerman, W. Verboom and D. N. Reinhoudt, Resorcinarenes, Tetrahedron, 1996, 52, 2663 CrossRef CAS.
- C. Naumann, E. Román, C. Peinador, T. Ren, B. O. Patrick, A. E. Kaifer and J. C. Sherman, Expanding Cavitand Chemistry: The Preparation and Characterization of [n]Cavitands with n≥4, Chem. – Eur. J., 2001, 7, 1637 CrossRef CAS PubMed.
- R. Hooley and J. Rebek, Deep Cavitands Provide Organized Solvation Reactions, J. Am. Chem. Soc., 2005, 127, 11904 CrossRef CAS PubMed.
- S. Mosca, Y. Yu, J. Gavette, K.-D. Zhang and J. Rebek, A Deep Cavitand Templates Lactam Formation in Water, J. Am. Chem. Soc., 2015, 137, 14582 CrossRef CAS PubMed.
- A. Jasat and J. C. Sherman, Carceplexes and Hemicarceplexes, Chem. Rev., 1999, 99, 93 Search PubMed.
- C. L. D. Gibb and B. C. Gibb, Well-Defined, Organic Nanoenvironments in Water: The Hydrophobic Effect Drives a Capsular Assembly, J. Am. Chem. Soc., 2004, 126, 11408 CrossRef CAS PubMed.
- C. L. D. Gibb and B. C. Gibb, Templated Assembly of Water-Soluble Nano-Capsules: Inter-Phase Sequestration, Storage, and Separation of Hydrogen Gases, J. Am. Chem. Soc., 2006, 128, 16498 CrossRef CAS PubMed.
- C. Peinador, E. Roman, K. Abboud and A. E. Kaifer, Novel Cavitands Containing Electrochemically Active 4,4′-bipyridinium Subunits, Chem. Commun., 1999, 1887 RSC.
- R. Toba, J. M. Quintela, C. Peinador, E. Roman and A. E. Kaifer, Dendritic Cavitands: Preparation and Electrochemical Properties, Chem. Commun., 2002, 1768 RSC.
- A. Roda, A. F. Hofmann and K. J. Mysels, The Influence of Bile-Salt Structure on Self-Association in Aqueous Solutions, J. Biol. Chem., 1983, 258, 6362 Search PubMed.
- O. Rinco, M. H. Kleinman and C. Bohne, Reactivity of Benzophones in the Different Binding Sites of Sodium Cholate Aggregates, Langmuir, 2001, 17, 578 Search PubMed.
- C. Yihwa, F. H. Quina and C. Bohne, Modulation with Acetonitrile of the Dynamics of Guest Binding to the Two Distinct Binding Sites of Cholate Aggregates, Langmuir, 2004, 20, 9983 Search PubMed.
- B. Cheng and A. E. Kaifer, Electrochemistry of Viologen Dications in Cholate Media and Competition between the Cholate Assemblies and the Cucurbit[7]uril Host, Langmuir, 2015, 31, 2997 CrossRef CAS PubMed.
- V. Rostovtsev, L. Green, V. Fokin and K. B. Sharpless, A Stepwise Huisgen Cycloaddition Process: Copper(I)-Catalyzed Regioselective “Ligation” of Azides and Terminal Alkynes, Angew. Chem., Int. Ed., 2002, 41, 2596 CrossRef CAS PubMed.
- C. W. Tornoe, C. Christensen and M. Meldal, Peptidotriazoles on Solid Phase: [1,2,3]-Triazoles by Regiospecific Copper(I)-Catalyzed 1,3-Dipolar Cycloadditions of Terminal Alkynes to Azides, J. Org. Chem., 2002, 67, 3057 CrossRef CAS PubMed.
- D. J. Cram, S. Karbach, H.-E. Kim, C. B. Knobler, E. F. Maverick, J. L. Ericson and R. C. Helgeson, Host-Guest Complexation., 46. Cavitands as Open Molecular Vessels Form Solvates, J. Am. Chem. Soc., 1988, 110, 2229 CrossRef CAS.
- E. Roman, C. Peinador, S. Mendoza and A. E. Kaifer, Improved Synthesis of Cavitands, J. Org. Chem., 1999, 64, 2577 CrossRef CAS.
- J. R. Fraser, B. Borecka, J. Trotter and J. C. Sherman, An Asymmetric Carceplex and New Crystal Structure Yield Information Regarding a 1 Million-Fold Template Effect, J. Org. Chem., 1995, 60, 1207 CrossRef CAS.
- P. Timmerman, K. G. A. Nierop, E. A. Brinks, W. Verboom, F. C. J. M. van Veggel, W. P. van Hoorn and D. N. Reinhoudt, Hydrophobic Concave Surfaces and Cavities by Combination of Calix[4]arenes and Resorcin[4]arenes, Chem. – Eur. J., 1995, 1, 132 CrossRef CAS.
- D. J. Cram, R. Jaeger and K. Deshayes, Host-Guest Complexation., 65. Hemicarcerands that Encapsulate Hydrocarbons with Molecular Weights Greater than Two Hundred, J. Am. Chem. Soc., 1993, 115, 10111 Search PubMed.
- B. Dayal, J. Speck, E. Bagan, G. S. Tint and G. Salen, p-Toluenesulfonic Acid/Methanol: Mild Reagent for the Preparation of Bile Acid Methyl Esters, Steroids, 1981, 37, 239 CrossRef CAS PubMed.
- B. N. S. Thota, A. J. Savyasachi, N. Lukashev, I. Beletskaya and U. Maitra, Tripodal Bile Acid Architectures Based on a Triarylphosphine Oxide Core Obtained by Copper-Catalysed [1,3]-Dipolar Cycloaddition: Synthesis and Preliminary Aggregation Studies, Eur. J. Org. Chem., 2014, 1406 CrossRef CAS.
- Y. Zhao and Z. Zhong, Oligomeric Cholates: Amphiphilic Foldamers with Large Internal Hydrophobic Cavities, J. Am. Chem. Soc., 2005, 127, 17894 Search PubMed.
- C. Massarenti, O. Bortolini, G. Fantin, D. Cristofaro, D. Ragno, D. Perrone, E. Marchesi, G. Toniolo and A. Massi, Fluorous-tag Assisted Synthesis of Bile Acid-bisphosphonate Conjugates via Orthogonal Click Reactions: an Access to Potential Anti-resorption Bone Drugs, Org. Biomol. Chem., 2017, 15, 4907 RSC.
- A. E. Kaifer and M. Gómez-Kaifer, Supramolecular Electrochemistry, Wiley-VCH, Weinheim, 1999 Search PubMed.
- P. Pracht, F. Bohle and S. Grimme, Automated Exploration of the Low-Energy Chemical Space with Fast Quantum Chemical Methods, Phys. Chem. Chem. Phys., 2020, 22, 7169 Search PubMed.
- S. Spicher and S. Grimme, Robust Atomistic Modeling of Materials, Organometallic and Biochemical Systems, Angew. Chem., Int. Ed., 2020, 59, 15665 Search PubMed.
- C. Bannwarth, S. Ehlert and S. Grimme, S. GFN2-xTB—An Accurate and Broadly Parametrized Self-Consistent Tight-Binding Quantum Chemical Method with Multipole Electrostatics and Density-Dependent Dispersion Contributions, J. Chem. Theory Comput., 2019, 15, 1652 CrossRef CAS PubMed.
- S. Ehlert, M. Stahn, S. Spicher and S. Grimme, Robust and Efficient Implicit Solvation Model for Fast Semiempirical Methods, J. Chem. Theory Comput., 2021, 17, 4250 CrossRef CAS PubMed.
- C. Plett and S. Grimme, Automated and Efficient Generation of General Molecular Aggregate Structures, Angew. Chem., Int. Ed., 2023, 62, e202214477 CrossRef CAS PubMed.
- A. Kawka, H. Koenig, D. Nowak and T. Pospieszny, Quasi-Podands with 1,2,3-Triazoles Rings from Bile Acid Derivatives: Synthesis, and Spectroscopic and Theoretical Studies, J. Org. Chem., 2024, 89, 7561 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |