Open Access Article
Open Access ArticleChemoselective enzymatic acylation of glycine as a green route to N-acyl amino acid surfactants
Dmitrii
Kurnosov
,
Andrea
Galatini
and
Luca
Banfi
 *
*
Department of Chemistry and Industrial Chemistry, University of Genova, via Dodecaneso, 31, 16146 Genova, Italy. E-mail: luca.banfi@unige.it
First published on 23rd January 2026
Abstract
N-Acyl amino acids are very important industrial surfactants, being gentle on the skin and completely biodegradable. The synthetic routes described in the literature involve the use of highly reactive acid chlorides or harsh reaction conditions. Following the latest trends in sustainable chemistry, in this work we developed an efficient, chemoselective, and environmentally friendly method for their synthesis, which involves the use of non-toxic enzymes as catalysts. Determining the most suitable starting materials and reaction conditions made it possible to achieve 97% product formation under mild reaction conditions.
Introduction
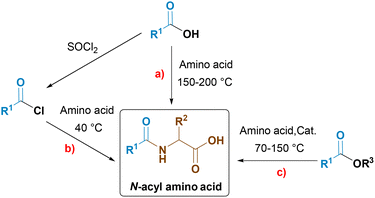
N-Acyl amino acids (NAAAs) are a class of mild amino acid-based surfactants obtained from renewable natural sources. Their structure consists of an acyl group with a long aliphatic chain directly attached via an amide bond to the nitrogen of an amino acid (Scheme 1).Due to their excellent cleansing properties,1 biodegradability2 and low toxicity,3N-acyl amino acids are widely used as the main ingredients or additives in valuable personal care products such as shampoos, shower gels, hand soaps, etc.
There are several synthetic routes for the synthesis of the desired product from fatty acids or fatty acid esters, which may in turn be derived from triglycerides (Scheme 2).
Direct condensation of fatty acids with amino acids may be achieved at high temperatures, typically at 150–200 °C. However, this method is characterized by low atom economy due to the use of an excess of fatty acids relative to amino acids (1.5–4 eq.) in order to shift an unfavourable equilibrium.4 Moreover, the use of high temperatures typically leads to coloured products, especially if unsaturated fatty acids are used.
To form an amide bond under milder conditions, the carboxyl group of the fatty acid must be converted into a good leaving group to allow attack by the amino group. Schotten–Baumann acylation of amino acids with acyl chlorides5 is the most used industrial method for the synthesis of N-acyl amino acids. The reaction takes place at low temperature in aqueous solution. However, the synthesis and use of acid chlorides are poorly sustainable, due to the toxicity of chlorine chemistry.6 Additionally, this method produces a side-product – sodium chloride, the presence of which is unfavorable in personal care products and worsens the properties of surfactants.7,8
Catalytic amidation is a direct condensation reaction between fatty acid esters and amino acids; it occurs at high temperatures (70–150 °C) in the presence of a catalyst.9,10 The disadvantages of this reaction are the long reaction times and the stoichiometric use of a base to deprotonate the amino acid.
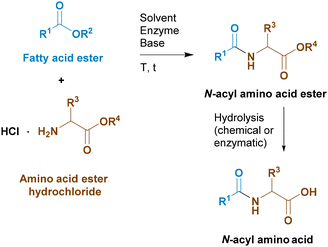
Since enzyme catalysed reactions usually proceed under mild conditions with high efficiency, selectivity, and atom economy, we decided to look for a way to synthesize N-acyl amino acids using biocatalysts. There are few reports dealing with the enzymatic condensation of carboxylic acids or esters with amino acids in water, and the main problem is the low conversion, due to an unfavourable equilibrium, and/or the use of ATP as a stoichiometric cofactor.11–13 On the other hand, the reaction of esters with amino acid esters or primary amides14 seemed more promising. However, the latter method may be troublesome due to the difficult hydrolysis of the primary amide in order to obtain the final carboxylic acid. Thus, we opted to focus on the enzyme-catalysed reaction of fatty acid esters with amino acid esters.
Thus, our plan is summarized in Scheme 3. Our idea was to use fatty acid esters and amino acid esters as starting reactants for the enzymatic synthesis of N-acyl amino acid esters in an organic solvent, followed by enzymatic or chemical hydrolysis of the resulting N-acyl amino acid ester.
The enzyme-catalysed reaction of esters with primary amines to synthesize secondary amides is definitely well known, but it has been mainly explored with the aim of achieving enantioselective kinetic resolutions.15,16 Contrary to these literature examples, the main problem in our case is that the employed amines also possess an ester moiety which too can be reactive under the amidation conditions.
Results and discussion
The activity of enzymes is highly dependent on the solvent in which the reaction is carried out.17–19 Therefore, at the beginning of this study, the effect of different types of solvents on the reaction efficiency was investigated, by studying the reaction between ethyl laurate and benzylamine, a monofunctionalized amine (Scheme 4). We tested lipophilic and lipophobic organic solvents with different polarities: ethers (diisopropyl ether (DIPE), tetramethyl tetrahydrofuran (TM-THF), tetrahydrofuran), alcohols (isopropanol, trifluoroethanol, tertiary butanol, methanol), hydrocarbons (heptane), nitriles (acetonitrile), and ketones (cyrene). We chose the commercial enzyme Novozym 435 (lipase from thermostable Candida Antarctica) as a biocatalyst because it is the most widely used enzyme in amide bond formation reactions.20,21Details are reported in the SI, but the highest conversions were obtained using tertiary butanol (75%), acetonitrile (84%), diisopropyl ether (DIPE) (88%) and tetramethyl-THF (TM-THF) (95%).
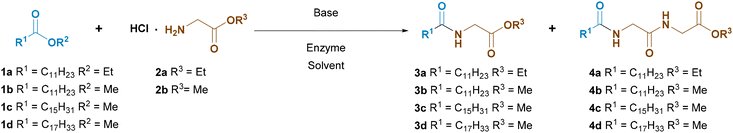
Next, we used the most suitable solvents in the reaction between ethyl laurate and glycine ethyl ester (Gly-OEt) hydrochloride. Since pure amino acid esters are not stable during storage and tend to polymerize, we chose to generate them in situ by addition of a base. Initially, 1 eq. of triethylamine was used.
As shown in Table 1, when using Novozym 435, the reactions were definitely slower than the model reaction with BnNH2, and here DIPE and TM-THF were inefficient (entries 1 and 2). The best results were achieved using tertiary butanol (35% conversion) and acetonitrile (32% conversion). However, the desired product 3a was accompanied by significant amounts of by-product 4a (entries 3 and 4).
| Entry | Solvent | R1 | R2 | R3 | Enzyme | Enzyme amount (mg mmol−1) | Base | Conc. | Time (h) | 1 | 3 | 4 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| a All reactions were carried out starting with equimolar amounts of fatty acid ester and glycine ester hydrochloride at 40 °C. | ||||||||||||
| 1 | Tetramethyl-THF | C11H23 | Et | Et | Novozym 435 | 275 | Et3N (1 eq.) | 0.25 | 3 | 100 | 0 | 0 |
| 2 | iPr2O | C11H23 | Et | Et | Novozym 435 | 275 | Et3N (1 eq.) | 0.25 | 3 | 100 | 0 | 0 |
| 3 | MeCN | C11H23 | Et | Et | Novozym 435 | 275 | Et3N (1 eq.) | 0.25 | 3 | 68 | 20 | 12 |
| 4 | tBuOH | C11H23 | Et | Et | Novozym 435 | 275 | Et3N (1 eq.) | 0.25 | 3 | 65 | 22 | 13 |
| 5 | MeCN | C11H23 | Et | Et | Lipozyme TL IM | 275 | Et3N (1 eq.) | 0.25 | 18 | 96 | 4 | 0 |
| 6 | MeCN | C11H23 | Et | Et | Amano lipase PS SD | 275 | Et3N (1 eq.) | 0.25 | 18 | 95 | 5 | 0 |
| 7 | MeCN | C11H23 | Et | Et | Amano lipase AK | 275 | Et3N (1 eq.) | 0.25 | 18 | 88 | 12 | 0 |
| 8 | MeCN | C11H23 | Me | Et | Amano lipase AK | 275 | Et3N (1 eq.) | 0.25 | 18 | 83 | 16 | 1 |
| 9 | MeCN | C11H23 | Me | Me | Amano lipase AK | 275 | Et3N (1 eq.) | 0.25 | 18 | 70 | 29 | 1 |
| 10 | MeCN | C15H31 | Me | Me | Amano lipase AK | 275 | Et3N (1 eq.) | 0.25 | 18 | 49 | 50 | 1 |
| 11 | MeCN | C15H31 | Me | Me | Amano lipase AK | 275 | KHCO3 (1 eq.) | 0.25 | 18 | 21 | 74 | 5 |
| 12 | MeCN | C15H31 | Me | Me | Amano lipase AK | 137 | KHCO3 (1.1 eq.) | 1 | 18 | 10 | 88 | 2 |
| 13 | MeCN | C15H31 | Me | Me | Amano lipase AK | 137 | Et3N (1.1 eq.) | 1 | 18 | 17 | 81 | 2 |
| 14 | MeCN | C15H31 | Me | Me | Amano lipase AK | 137 | KHCO3 (2.0 eq.) | 1 | 18 | 3 | 93 | 4 |
| 15 | MeCN | C15H31 | Me | Me | Amano lipase AK | 137 | NaHCO3 (2.0 eq.) | 1 | 18 | 10 | 86 | 4 |
| 16 | tBuOH | C 15 H 31 | Me | Me | Amano lipase AK | 137 | KHCO 3 (2.0 eq.) | 1 | 18 | 0 | 97 | 3 |
| 17 | tBuOH | C11H23 | Me | Me | Amano lipase AK | 137 | KHCO3 (2.0 eq.) | 1 | 24 | 5 | 84 | 11 |
| 18 | tBuOH | C17H33 | Me | Me | Amano lipase AK | 137 | KHCO3 (2.0 eq.) | 1 | 24 | 6 | 88 | 6 |
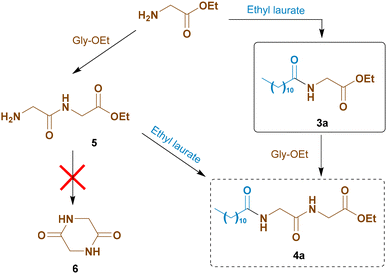
The most likely mechanism for the formation of the by-product 4a involves the formation of the N-acyl amino acid ester molecule 3a, followed by a reaction with another amino acid ester molecule (Scheme 5). Alternatively, glycine ester could first dimerize to 5. This seems less probable, since neither compound 5 nor diketopiperazine 6 was observed in the crude product.
Moreover, we could not detect larger by-products (trimers, etc.), showing that dipeptide ester 5, if present, does not react with 3a. Anyway, it is clear that Novozym 435 is highly active, but poorly chemoselective.
The next step in this study was to identify a more selective enzyme. We preferred to use MeCN instead of tBuOH in this first optimization, because we noticed that the efficiency of the reaction in tBuOH strongly depends on the dryness of this solvent. In coupling reactions between fatty acid esters and amino acid esters, we decided to focus our attention on lipases, as they are the most widely used enzymes in the formation of amide bonds, and exhibit high stability and efficiency in non-aqueous organic solvents.22
To identify selective enzyme in the synthesis of N-acyl amino acid esters, lipases of various origins were used: animal origin (porcine pancreas), plant origin (papain), fungi (Amano A-6, AYS Amano, AS Amano, M Amano, Lipozyme RM IM, Lipozyme TL IM, enzymes from Candida cylindracea, Aspergillus niger, Penicillium roqueforti), bacteria (PS Amano, AK Amano) (see the SI). Among the large number of enzymes, only three showed at least some activity: Lipozyme TL IM (entry 5), Amano lipase PS SD (entry 6), and Amano lipase AK (entry 7). Despite their low efficiency compared to Novozym 435, all three enzymes catalyzed the desired reaction with very high chemoselectivity. Due to the highest conversion, we selected Amano lipase AK as the catalyst for further improvements. Table 1 shows only selected examples, whereas full details are reported in the SI.
The length of the aliphatic chain of surfactants plays an important role in their ability to reduce surface tension,23 as well as in the critical micelle concentration, foaming ability, and foam stability.24 The ester substitute for fatty acids, in turn, affects their price due to differences in the synthesis method: starting reagents, reaction conditions, and safety considerations.25
To compare the reactivity of the starting reagents, various fatty acid esters (methyl, ethyl), various glycine esters (methyl, ethyl), and various lengths of aliphatic fatty acid chains (laurate, palmitate, oleate) were used.
First, the influence of the ester substituent was studied on lauric acid. Methyl esters were found to have a higher reactivity than ethyl esters in the reaction with glycine ethyl ester (entries 7 and 8).
Less expectedly, methyl glycinate was found to be more reactive than ethyl glycinate, although this ester does not take part directly in the reaction (entries 8 and 9). Initially we thought that glycine ethyl esters could be better from the point of view of chemoselectivity, but luckily this was not the case.
On passing from methyl laurate to methyl palmitate, conversion increased appreciably (entries 9 and 10).
This difference in rate between laurate and palmitate, although not very high, was unexpected to us. In the literature, we could find various cases where the two fatty acids (or their esters) react at different rates. In some cases the faster was lauric acid/laurate,26 in other it was the opposite.27,28 Thus, a general trend cannot be drawn. All these quoted examples refer to transesterification reactions, where probably the rate-limiting step is the formation of the acyl-enzyme adduct. In contrast, we think that in the process reported here, the rate-limiting step is the second one, that is the reaction of glycinate with the acylated enzyme. Since we used polar solvents, the hydrophobic chain of the acyl-enzyme adduct should fold in order to avoid unfavourable interactions with the solvent, staying near the lipase hydrophobic cap (lid domain).29 This could in part inhibit the access of polar glycinate to the active site. Different lengths of the chain may cause subtle differences in the interaction with the lid domain and thus in shielding the entrance to the active site. This is most likely dependent on the structure of the specific lipase employed, and therefore, a general trend of the influence of the chain length on reactivity cannot be drawn.
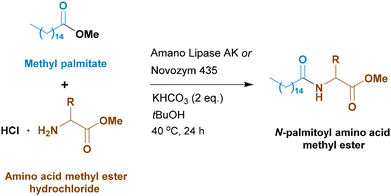
Reaction conditions, such as temperature, pH, and reactant concentrations, have a strong influence on enzymatic activity. Therefore, the next step of the work was finding the optimal reaction conditions using methyl palmitate. Amano lipase AK is an enzyme produced by Pseudomonas fluorescens and can be used in reactions in the temperature range of 5–65 °C, but it is known to exhibit maximum catalytic activity at 40–45 °C.30,31 Actually, in our case, an increase of temperature to 60 °C was deleterious (see the SI), and the optimal temperature was 40 °C.
To liberate glycine methyl ester from its hydrochloride, we initially used triethylamine as a non-nucleophilic base. However, it is not definitely a green reagent. We attempted to replace it with other less expensive and more sustainable non-nucleophilic weak bases widely used in the food industry – potassium hydrogen carbonate (KHCO3), sodium hydrogen carbonate (NaHCO3), and sodium carbonate (Na2CO3). To our delight, replacing triethylamine with KHCO3 increased the conversion from 50% to 74% (entry 11). We do not think that this improvement is due to a lower pH (KHCO3 is a weaker base than Et3N). We have actually seen that doubling the amount of base is not deleterious (compare entries 12 and 14). Neutralization of methyl glycinate hydrochloride forms just KCl, CO2 and H2O, whereas triethylammonium chloride (formed when using Et3N) may somehow interfere with the enzymatic activity.
To further raise the reaction conversion, we increased the concentration of reagents by a factor of 4: from 0.25 M to 1 M, at the same time halving the enzyme amount: from 275 mg to 137 mg per mmol of fatty acid ester. This allows the reaction conversion to be increased to 88% (entry 12). This was observed also with Et3N as the base (entry 13). A slight improvement was achieved by using 2 eq. of KHCO3 (entry 14), whereas the analogous sodium salt was less efficient (entry 15).
At this point we decided to return to tert-butyl alcohol, which is considered more sustainable than acetonitrile.32,33 As can be seen in entry 16, this solvent behaves even better than MeCN if it is adequately dried just before use.
Palmitic acid derivatives are widely used in the cosmetics industry and personal care products because they add thickness and consistency, as they are solid at room temperature and act as emulsifiers, emollients and foaming agents.34,35 However, there are two other important fatty acids that give the final compositions other properties: lauric acid derivatives could act as antimicrobial agents.36 Oleic acid derivatives have a protective effect on the skin because they have high affinity for the lipids of the stratum corneum.37 Therefore, we applied the conditions developed for methyl palmitate to reactions with methyl laurate and methyl oleate.
Again, methyl laurate showed a lower reactivity, which also leads to lower chemoselectivity (entry 17). However, the yield of the desired product 4 remains high. Methyl oleate reactivity lies in between that of laurate and palmitate (entry 18). It should be noted that synthesis of oleoyl amino acid surfactants through the traditional Schotten–Baumann process is very troublesome for the difficult preparation of oleoyl chloride.
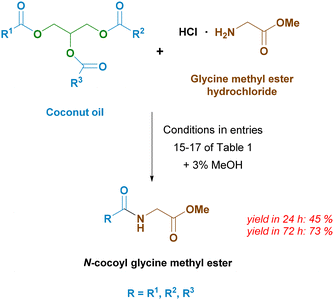
Methyl esters of fatty acids are mainly synthesized by the transesterification reaction of triglycerides in methanol in the presence of an alkaline catalyst such as hydroxide or alkoxide.38 However, transesterification can also be catalyzed by the same enzyme we used for enzymatic amidation – Amano lipase AK.39 Thus, it is possible to directly replace fatty acid methyl ester with vegetable oil; the only requirement being a small addition of methanol for transesterification (Scheme 6). Due to the numerous benefits when used in personal care products,40 we decided to use coconut oil as a source of fatty acids. This oil is particularly rich in lauric acid.
Compared to the reactions with methyl esters of fatty acids, the reaction with coconut oil proceeds more slowly. In the reaction mixture, we also found the presence of 1- and 2-monoglycerides: 30 mol% remains after 24 hours and 13 mol% after 72 hours. However, this may not be necessarily bad, since mono- and di-glycerides are widely used as emulsifiers and thus their presence in the final surfactants could be even an added value.
Glycine is a small size amino acid with minimal steric hindrance, which makes its nucleophilic nitrogen more potent at attacking the carbonyl carbon atom of a fatty acid ester. To understand the influence of different groups in the alpha position on the reaction efficiency, we attempted the synthesis of N-acyl amino acids with other amino acid esters – L-alanine methyl ester and L-glutamic acid dimethyl ester (Scheme 7).
Unfortunately, in both cases, no product was formed when using either the selective lipase Amano AK or the highly active Novozym 435; only partial hydrolysis of starting methyl palmitate to palmitic acid was observed.
In the case of L-glutamic acid dimethyl ester, enzymatically catalyzed intramolecular cyclization occurs, which results in the formation of methyl pyroglutamate (methyl 5-oxo-L-prolinate). This occurrence was demonstrated by us by carrying out an experiment under similar conditions, but without methyl palmitate. The formation of pyroglutamate was observed by GC-MS and 1H NMR of the crude product.
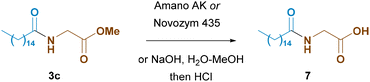
The final step in the synthesis of an N-acyl amino acid is the conversion of the ester group of the enzymatic amidation reaction product to the free carboxylic group (Scheme 8). There are two ways to do this: saponification using an alkaline base or enzymatic hydrolysis using lipases.
Amano lipase AK is a highly selective enzyme, and its active site predominantly binds to fatty acid esters rather than N-acyl amino acid esters; as a result, little formation of the N-acyl dipeptide by-product 4 was observed. This beneficial property makes it unsuited for the final hydrolysis. Only 26% of the initial material was converted within 72 hours. In contrast, Novozym 435 is active toward both fatty acid esters and N-acyl amino acid esters. When Novozym 435 was used in the reaction with the isolated compound 3c, complete conversion occurred within 24 hours. However, this does not allow us to use the same enzyme for both steps and to carry out a one-pot procedure. It is necessary to filter off the first enzyme, evaporate the solvent, and then add the hydrolysis solvent (aqueous buffer) and Novozym.
Alternatively, simple saponification with sodium hydroxide may be carried out, resulting in almost complete conversion of the starting material to the final product: 99.5% yield within 72 h.
Conclusions
In this work, we developed a highly efficient enzymatic synthesis of N-acyl amino acids from fatty acid esters and amino acid esters, which consists of two stages: amidation and hydrolysis. Initially, we identified two suitable solvents for the enzymatic amidation: tert-butyl alcohol and acetonitrile. Since the use of Novozym 435 resulted in the formation of by-products, we searched and found a more selective enzyme – Amano lipase AK. Optimization of temperature, concentration of reagents and of the enzyme in the reaction mixture, suitable base and its amount allowed the amidation reaction to be carried out with high efficiency: 97% of the product is formed using methyl palmitate; 84% using methyl laurate; and 88% using methyl oleate. The final stage – hydrolysis – was carried out with complete conversion using Novozym 435 or using an alkaline aqueous base.Overall, this optimized protocol is much more sustainable than the Schotten–Baumann reaction of acyl chlorides, which is presently used in nearly all industrial syntheses of these surfactants, since it completely avoids the toxic reagents needed for the preparation of acyl chlorides. Moreover, our procedure may be applied to oleic acid derivatives and prevents the presence of stoichiometric NaCl in the final solution of the surfactant. The only drawback is the need to use an organic solvent for the amidation reaction, but tert-butyl alcohol is considered a green solvent33 and can be recycled by distillation.
Experimental
For details on the analytical methods, see the SI.Typical procedure for the enzymatic amidation reaction: N-palmitoyl glycine methyl ester 3c
Glycine methyl ester hydrochloride (220 mg, 1.75 mmol, 1 eq.) was weighed in a round-bottom flask with methyl palmitate (473 mg, 1.75 mmol, 1 eq.), KHCO3 (350 mg, 3.50 mmol, 2 eq.) and Amano lipase AK (240 mg). After the addition of anhydrous tBuOH (1.75 mL) the mixture was magnetically stirred for 18 hours at 40 °C. The mixture was diluted with EtOAc (15 mL), further stirred for 30 min, and filtered through Celite to recover the enzyme. This organic phase was washed with saturated aqueous NH4Cl and dried with Na2SO4. After filtration, the solvent was removed under vacuum. The amounts of 1c, 3c and 4c reported in Table 1 were determined by 1H NMR analysis of this crude product.Enzymatic hydrolysis of 3c to 6
N-Acyl amino acid ester 3c (98 mg, 0.30 mmol) was suspended in a mixture of tris(hydroxymethyl)aminomethane (TRIS) buffer (pH = 7, 0.25 mol L−1, 7.2 mL, 6 eq.) and acetonitrile as the co-solvent (215 µL, 3% by volume to TRIS buffer). Then this mixture was treated with Novozym 435 (100 mg) and stirred for 24 h at 40 °C. The mixture was diluted with EtOAc and filtered through Celite. After adjusting the aqueous phase to pH 2 by addition of 1 M HCl, the phases were separated and the organic phase was washed with saturated solution of NaCl and dried with Na2SO4. After filtration, the solvent was removed under vacuum to afford pure acid 7.Chemical hydrolysis of 3c to 7
N-Acyl amino acid ester 3c (98 mg, 0.30 mmol) was suspended in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 solution of EtOH (600 µL) and 1 M NaOH (600 µL, 2 eq.). The mixture was stirred for 72 h at RT. The solution was diluted with saturated aqueous NaCl, and the pH was adjusted to <2 by the addition of 1 M HCl. Extraction with AcOEt, drying with Na2SO4, filtration and evaporation to dryness gave pure acid 7.
1 solution of EtOH (600 µL) and 1 M NaOH (600 µL, 2 eq.). The mixture was stirred for 72 h at RT. The solution was diluted with saturated aqueous NaCl, and the pH was adjusted to <2 by the addition of 1 M HCl. Extraction with AcOEt, drying with Na2SO4, filtration and evaporation to dryness gave pure acid 7.
Comments on possible challenges during scale-up
For scale-up of this protocol, we think that efficiently supporting the enzyme will be helpful, in order to make its recycling easier. The simple extractive work-up made at a low scale at the level of 6 in order to remove KCl and excess KHCO3 (mainly for analytical purposes) could be avoided, since these salts can be removed after the final step. Also the relative amount of EtOAc used for enzyme filtration can be strongly reduced if using a supported enzyme.Author contributions
Conceptualization: LB. Funding acquisition: LB. Resources: LB. Data analysis: DK, LB. Investigation: DK, AG. Supervision: LB. Writing of draft: DK. Writing: editing and review: LB.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: procedures, optimizations, analytical procedures, and NMR spectra. See DOI: https://doi.org/10.1039/d5ob01932j.Acknowledgements
We acknowledge the financial support from Lamberti SpA and from the University of Genova for a scholarship to DK. We thank Amano and Novo Nordisk for kind gift of their enzymes.References
- N. Koshti, B. Parab, R. Powale, A. Desai, K. Baral, P. Katdare, B. Sawant, S. Kadam and S. Pilli, Method to Produce N-Acyl Amino Acid Surfactants Using N-Acyl Amino Acid Surfactants or the Corresponding Anhydrides as Catalyst, EP2888226B2, 2012 Search PubMed.
- K. Holmberg, F. Bilén and R. Bordes, Development of amino acid-based surfactants: from synthesis to applications, Curr. Opin. Colloid Interface Sci., 2025, 75, 101884 CrossRef CAS.
- D. B. Tripathy, A. Mishra, J. Clark and T. Farmer, Synthesis, chemistry, physicochemical properties and industrial applications of amino acid surfactants: A review, C. R. Chim., 2018, 21, 112–130 Search PubMed.
- T. Hattori, Waschmittel und deren herstellung, EP1672055A1, 2006 Search PubMed.
- T. Hattori, N. Kitamura, N. Yamato and H. Yokota, Method for preparing N-long chain acyl neutral amino acid, US6703517B2, 2002 Search PubMed.
- P. Tundo, Chlorine-Free Synthesis for Green Chemistry: an Overview, Pure Appl. Chem., 2012, 84, 411–423 CrossRef CAS.
- Y. Akiyama, Y. Matsue, T. Mori and S. Nishijima, Effect of sodium chloride on precipitation and adsorption behavior of polymer-surfactant complex particles in aqueous solution, Colloids Surf., A, 2018, 559, 1–7 Search PubMed.
- K. Staszak, D. Wieczorek and K. Michocka, Effect of Sodium Chloride on the Surface and Wetting Properties of Aqueous Solutions of Cocamidopropyl Betaine, Surfactants Deterg., 2015, 18, 321–328 Search PubMed.
- B. Wang and G. P. Dado, Process for Preparing N-Acyl Aminoacid Salts, EP2870136B1, 2018 Search PubMed.
- A. Taussat, R. M. de Figueiredo and J.-M. Campagne, Direct Catalytic Amidations from Carboxylic Acid and Ester Derivatives: A Review, Catalysts, 2023, 13, 366 CrossRef CAS.
- K. Gramman, J. Wolter, S. Schaffer, J. Corthals, T. Hass, N. Potgrave, L. Reinecke and M. Olfert, A method for producing acyl amino acids, EP3038595B1, 2019 Search PubMed.
- M. C. Bourkaib, S. Delaunay, X. Framboisier, C. Humeau, J. Guilbot, C. Bize, E. Illous, I. Chevalot and Y. Guiavarc'h, Enzymatic synthesis of N-10-undecenoyl-phenylalanine catalysed by aminoacylases from Streptomyces ambofaciens, Process Biochem., 2020, 99, 307–315 Search PubMed.
- N. Vonderhagen, H.-C. Raths and E. Eilers, Enzymatically Catalysed N-Acylation of Amino Acids, Protein Hydrolizates and/or Derivatives Thereof, WO99/24599, 1990 Search PubMed.
- S. E. Godtfredsen and F. Biorkling, An Enzyme-Catalysed Process forn Preparing N-Acyl Amino Acids and N-Acyl Amino Acids Amides, WO90/14429, 1990 Search PubMed.
- F. Poulhès, N. Vanthuyne, M. P. Bertrand, S. Gastaldi and G. Gil, Chemoenzymatic Dynamic Kinetic Resolution of Primary Amines Catalyzed by CAL-B at 38–40 °C, J. Org. Chem., 2011, 76, 7281–7286 CrossRef PubMed.
- M. Nechab, N. Azzi, N. Vanthuyne, M. Bertrand, S. Gastaldi and G. Gil, Highly Selective Enzymatic Kinetic Resolution of Primary Amines at 80 °C: A Comparative Study of Carboxylic Acids and Their Ethyl Esters as Acyl Donors, J. Org. Chem., 2007, 72, 6918–6923 CrossRef CAS PubMed.
- M. Reslow, P. Adlercreutz and B. Mattiasson, Organic solvents for bioorganic synthesis, Appl. Microbiol. Biotechnol., 1987, 26, 1–8 Search PubMed.
- P. A. Fitzpatrick and A. M. Klibanov, How can the solvent affect enzyme enantioselectivity?, J. Am. Chem. Soc., 1991, 113, 3166–3171 CrossRef CAS.
- B. Fu and P. T. Vasudevan, Effect of Organic Solvents on Enzyme-Catalyzed Synthesis of Biodiesel, Energy Fuels, 2009, 23, 4105–4111 CrossRef CAS.
- M. Sun, K. Nie, F. Wang and L. Deng, Optimization of the Lipase-Catalyzed Selective Amidation of Phenylglycinol, Front. Bioeng. Biotechnol., 2020, 7, 486 CrossRef PubMed.
- S. R. Bansode and V. K. Rathod, An intensified technique for lipase catalysed amide synthesis, Chem. Eng. Process.: Process Intensif., 2019, 143, 107605 CrossRef.
- M. Lubberink, W. Finnigan and S. L. Flitsch, Biocatalytic amide bond formation, Green Chem., 2023, 25, 2958–2970 RSC.
- B. Smit, A. G. Schlijper, L. A. M. Rupert and N. M. van Os, Effects of chain length of surfactants on the interfacial tension: molecular dynamics simulations and experiments, J. Phys. Chem., 1990, 94, 6933–6935 CrossRef CAS.
- Y. Wang, X. Liu, L. Bai and J. Niu, Influence of alkyl chain length of alpha olefin sulfonates on surface and interfacial properties, J. Dispersion Sci. Technol., 2017, 38, 1764–1769 CrossRef CAS.
- M. F. M. Yusoff, X. Xu and Z. Guo, Comparison of Fatty Acid Methyl and Ethyl Esters as Biodiesel Base Stock: a Review on Processing and Production Requirements, J. Am. Oil Chem. Soc., 2014, 91, 525–531 CrossRef CAS.
- S. C. Tupufia, Y. J. Jeon, C. Marquis, A. A. Adesina and P. L. Rogers, Enzymatic conversion of coconut oil for biodiesel production, Fuel Process. Technol., 2013, 106, 721–726 Search PubMed.
- A. Silva, L. Soares, Á. Lima, C. Soares and R. Souza, Strategies to Reuse of Biocatalysts in the Hydrolysis and Esterification Reactions of Licuri (Syagrus coronata (Mart.) Becc.) Oil, ChemCatChem, 2022, 14, e202200448 CrossRef.
- K. Ren and B. P. Lamsal, Synthesis of some glucose-fatty acid esters by lipase from Candida antarctica and their emulsion functions, Food Chem., 2017, 214, 556–563 CrossRef CAS PubMed.
- L. Fernandez-Lopez, S. Roda, A. Robles-Martín, R. Muñoz-Tafalla, D. Almendral, M. Ferrer and V. Guallar, Enhancing the Hydrolytic Activity of a Lipase towards Larger Triglycerides through Lid Domain Engineering, Int. J. Mol. Sci., 2023, 24, 13768 CrossRef CAS PubMed.
- G. L. Christen and R. T. Marshall, Selected Properties of Lipase and Protease of Pseudomonas fluorescens 27 Produced in Four Media, J. Dairy Sci., 1984, 67, 1680–1687 CrossRef CAS.
- Y. Kojima and S. Shimizu, Purification and characterization of the lipase from Pseudomonas fluorescens HU380, J. Biosci. Bioeng., 2003, 96, 219–226 CrossRef CAS PubMed.
- F. P. Byrne, S. Jin, G. Paggiola, T. H. M. Petchey, J. H. Clark, T. J. Farmer, A. J. Hunt, C. Robert McElroy and J. Sherwood, Tools and techniques for solvent selection: green solvent selection guides, Sustainable Chem. Processes, 2016, 4, 7 CrossRef.
- M. Tobiszewski, J. Namieśnik and F. Pena-Pereira, Environmental risk-based ranking of solvents using the combination of a multimedia model and multi-criteria decision analysis, Green Chem., 2017, 19, 1034–1042 RSC.
- S. Cochran and M. Anthonavage, in Lipids and Skin Health, ed. A. Pappas, Springer International Publishing, Cham, 2015, pp. 311–319, DOI:10.1007/978-3-319-09943-9_21.
- M. Rivai, E. Hambali, A. Suryani, R. Fitria, S. Firmansyah and J. Pradesi, Synthesis of palm oil fatty acid as foaming agent for firefighting application, IOP Conf. Ser.: Earth Environ. Sci., 2017, 65, 012047 CrossRef.
- T. Nakatsuji, M. C. Kao, J.-Y. Fang, C. C. Zouboulis, L. Zhang, R. L. Gallo and C.-M. Huang, Antimicrobial Property of Lauric Acid Against Propionibacterium Acnes: Its Therapeutic Potential for Inflammatory Acne Vulgaris, J. Invest. Dermatol., 2009, 129, 2480–2488 CrossRef CAS PubMed.
- A. Kováčik, M. Kopečná, I. Hrdinová, L. Opálka, M. Boncheva Bettex and K. Vávrová, Time-Dependent Differences in the Effects of Oleic Acid and Oleyl Alcohol on the Human Skin Barrier, Mol. Pharm., 2023, 20, 6237–6245 CrossRef PubMed.
- G. Vicente, M. Martínez and J. Aracil, Integrated biodiesel production: a comparison of different homogeneous catalysts systems, Bioresour. Technol., 2004, 92, 297–305 CrossRef CAS PubMed.
- A. B. R. Moreira, V. H. Perez, G. M. Zanin and H. F. de Castro, Biodiesel Synthesis by Enzymatic Transesterification of Palm Oil with Ethanol Using Lipases from Several Sources Immobilized on Silica–PVA Composite, Energy Fuels, 2007, 21, 3689–3694 Search PubMed.
- R. Logamorthy and K. Karthikeyan, Coconut oil - Nature's own emollient, Indian J. Dermatol. Venereol. Leprol., 2025, 91, 81–83 CrossRef PubMed.
| This journal is © The Royal Society of Chemistry 2026 |