Open Access Article
Open Access ArticleFacile one-pot synthesis of 5-alkylidene-2,3,5,6,7,8-hexahydroimidazo[1,2-a]pyridines
Esra Korkmaz
Yurtoglu
 and
Metin
Zora
and
Metin
Zora
 *
*
Department of Chemistry, Faculty of Arts and Science, Middle East Technical University, 06800 Ankara, Turkey. E-mail: zora@metu.eddu.tr
First published on 3rd March 2026
Abstract
A straightforward and efficient one-pot protocol for the synthesis of 5-alkylidene-2,3,5,6,7,8-hexahydroimidazo[1,2-a]pyridines is reported. When subjected to reaction with ethylenediamine in refluxing dioxane, bis-α,β-alkynic ketones, i.e., 1,9-diarylnona-2,7-diyne-1,9-diones, afford 5-alkylidene-2,3,5,6,7,8-hexahydroimidazo[1,2-a]pyridines. Generation of three new C–N bonds, along with one C–C bond cleavage, during the reaction leads to the construction of unknown fused five- and six-membered heterocyclic ring systems. The reaction proceeds through an imidazoline intermediate. Similarly, the reaction of bis-α,β-alkynic ketones with 1,2-propylenediamine produces 2-methyl-substituted hexahydroimidazo[1,2-a]pyridines. The skeletal diversity of the synthesized hexahydroimidazo[1,2-a]pyridines may be of use in pharmaceutical and medicinal chemistry as novel molecular entities and structural leads.
Introduction
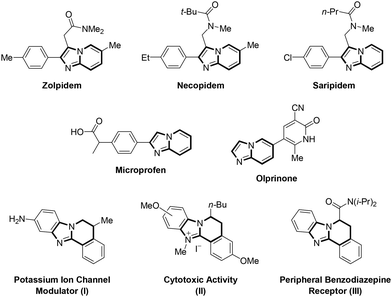
Imidazo[1,2-a]pyridine (IP) (1) and its derivatives have emerged as valuable structural features in pharmaceutical and materials science because of their easy availability, great stability and a wide range of biological and material properties they possess (Scheme 1).1 In fact, IP consists of a five-membered imidazole moiety fused with a six-membered pyridine ring and constitutes one of the most important bridgehead (ring junction) N-containing heterocyclic cores due to its common appearance in the structures of numerous natural products and drugs.2 IPs have been reported to exhibit a diverse range of biological properties, including antiulcer, anticonvulsant, antifungal, antiviral, anticancer, antiprotozoal, antibacterial, anti-inflammatory, anthelmintic, analgesic, antituberculosis, antipyretic, antiepileptic, and antitumor activities.3 They also act on various targets including enzymes (CDK, MEK, PI3K and GSK3), receptors (ALK-5, KDR, mGluR5, 5HT, MHC-R1, H3, D4 and GABAA) and infectious agents (HCMV, VZV, HSV-1, CXCR4 and CCR5).3,4 Moreover, they form the core structures of a range of drugs, including zolpidem (a hypnotic, used in the treatment of insomnia), necopidem and saripidem (sedative and anxiolytic agents), microprofen (a prostanoid signaling modulator, analgesic and NSAID) and olprinone (used for the treatment of acute heart failure) (Scheme 2).5,6 Importantly, benzo-fused and partially reduced derivatives also exhibit interesting biological activities. For instance, compound I is a useful potassium ion channel modulator for the treatment of neurological disorders.7 Compound II shows cytotoxicity against the human cancer cell line MCF-7.8 Compound III has high affinity for the peripheral benzodiazepine receptor and displays anticancer activity as well (Scheme 2).9 Therefore, over the years, many fruitful, mild and productive strategies have been developed for the synthesis of IPs, and new variants continue to emerge due to their immense importance.10 | ||
| Scheme 1 Structures of imidazo[1,2-a]pyridine (IP) and 2,3,5,6,7,8-hexahydroimidazo[1,2-a]pyridine (2,3,5,6,7,8-HIP). | ||
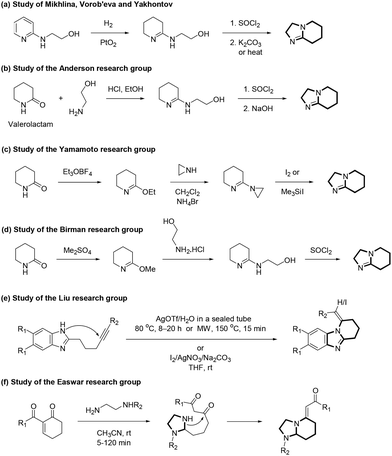
Sometimes, compared to the parent compounds, partially reduced derivatives may be more important from the point of view of biological activity and material properties. Notably, hexahydroimidazo[1,2-a]pyridines (HIPs) are less known.11 In particular, 2,3,5,6,7,8-hexahydroimidazo[1,2-a]pyridines (2,3,5,6,7,8-HIPs) (2) (Scheme 1), also called 1,7-diazabicyclo[4.3.0]non-6-enes, are very limited and, to the best of our knowledge, only a few examples of these molecules are available (Scheme 3).12–15 As shown by Mikhlina, Vorob'eva and Yakhontov, catalytic hydrogenation of 2-(pyridin-2-ylamino)ethanol provided 2-(3,4,5,6-tetrahydropyridin-2-ylamino)ethanol, which, upon chlorination with thionyl chloride, followed by intramolecular cyclization, generated 2,3,5,6,7,8-HIP (Scheme 3a).12 The Anderson research group showed that the reaction of valerolactam (piperidin-2-one) with 2-aminoethanol in the presence of hydrochloric acid afforded 2-(3,4,5,6-tetrahydropyridin-2-ylamino)ethanol, which, upon reaction with thionyl chloride, produced 2,3,5,6,7,8-HIP (Scheme 3b).13 On the other hand, the Yamamoto research group first prepared 6-ethoxy-2,3,4,5-tetrahydropyridine by the reaction of valerolactam with triethyloxonium tetrafluoroborate. Then, its reaction with aziridine afforded 6-aziridin-1-yl-2,3,4,5-tetrahydropyridine. Finally, its rearrangement with molecular iodine or trimethylsilyl iodide gave 2,3,5,6,7,8-HIP (Scheme 3c).14 Similarly, the Birman research group prepared a 6-methoxy-2,3,4,5-tetrahydropyridine derivative by the reaction of valerolactam with dimethyl sulfate, which, upon reaction with 2-aminoethanol hydrochloride, followed by treatment with thionyl chloride, yielded 2,3,5,6,7,8-HIP (Scheme 3d).15 It is noteworthy that in a similar study, Liu and coworkers synthesized alkyne-tethered benzimidazoles from the corresponding 5-hexynoic acid methyl ester, o-phenylenediamine and aryl iodides, and then investigated their intramolecular cyclizations under a few conditions.16 These reactions led to the formation of 1-alkylidene-1,2,3,4-tetrahydrobenzo[4,5]imidazo[1,2-a]pyridines or their iodo-substituted derivatives depending upon the reagents (Scheme 3e).16 Easwar and coworkers investigated the reactions of cyclic Morita–Baylis–Hillman ketones with ethylenediamine derivatives (Scheme 3f).17 The conjugate addition of diamine, followed by a retro-Mannich reaction, yielded an imidazolinyl-substituted 1,3-diketone intermediate, in which the condensation of a secondary amine with a ketone afforded octahydroimidazo[1,2-a]pyridine derivatives. Notably, the value of 2,3,5,6,7,8-HIPs has been underrepresented in medicinal chemistry due to the lack of efficient synthetic strategies. An ever-growing aspect of these studies is to find novel derivatives, which may provide a new mode of action for the treatment of specific diseases. Accordingly, there is a continuous need to develop new methodologies for the synthesis of such compounds since it may increase their significance and potential in related fields.
 | ||
| Scheme 3 Synthetic approaches for the synthesis of 2,3,5,6,7,8-hexahydroimidazo[1,2-a]pyridines and their octahydro- and benzo-fused derivatives. | ||
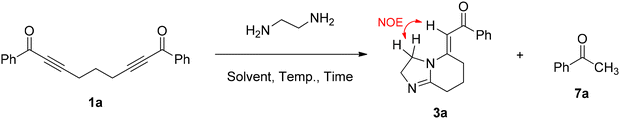
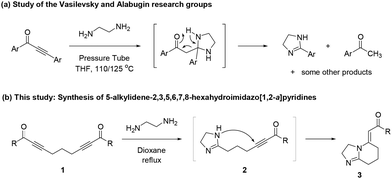
Vasilevsky and Alabugin reported that the reaction of diaryl-substituted α,β-alkynic ketones with ethylenediamine (EDA) led to the formation of 2-aryl-4,5-dihydro-1H-imidazoles and aryl methyl ketones (Scheme 4a).18 In fact, this reaction first proceeds by sequential bis-conjugate addition of EDA, yielding an imidazoline intermediate, and then by a concerted retro-Mannich reaction of the imidazoline to afford 4,5-dihydro-1H-imidazole and aryl methyl ketone.18 Due to our ongoing interest in the synthesis of new heterocyclic molecules as potential pharmaceuticals and scaffolds,19 we anticipated that the reaction of bis-α,β-alkynic ketones 1 (namely, 1,9-diarylnona-2,7-diyne-1,9-diones) with EDA might provide dihydroimidazolyl-substituted α,β-alkynic ketones 2 (i.e., 6-(4,5-dihydro-1H-imidazol-2-yl)hex-2-yn-1-ones) by a similar mechanism, which could then undergo intramolecular conjugate addition to yield 5-alkylidene-2,3,5,6,7,8-hexahydroimidazo[1,2-a]pyridines 3 (Scheme 4b). To the best of our knowledge, this approach has not been utilized previously for the synthesis of such hexahydroimidazo[1,2-a]pyridines 3. To our delight, we have found that when treated with EDA in refluxing dioxane, bis-α,β-alkynic ketones 1 produced the unknown 5-alkylidene-2,3,5,6,7,8-hexahydroimidazo[1,2-a]pyridines 3 in a one-pot manner (Scheme 4b). Notably, the formation of these molecules by the intramolecular cyclization of linear precursors also represents a valuable tool in terms of the synthetic methodology. Herein, we report the preliminary results of this study.
 | ||
| Scheme 4 Strategies for the synthesis of 4,5-dihydro-1H-imidazoles and 5-alkylidene-2,3,5,6,7,8-hexahydroimidazo[1,2-a]pyridines. | ||
Results and discussion
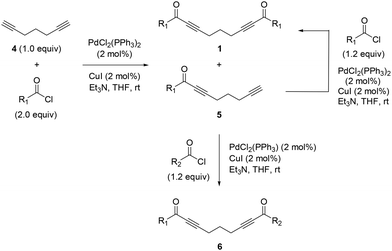
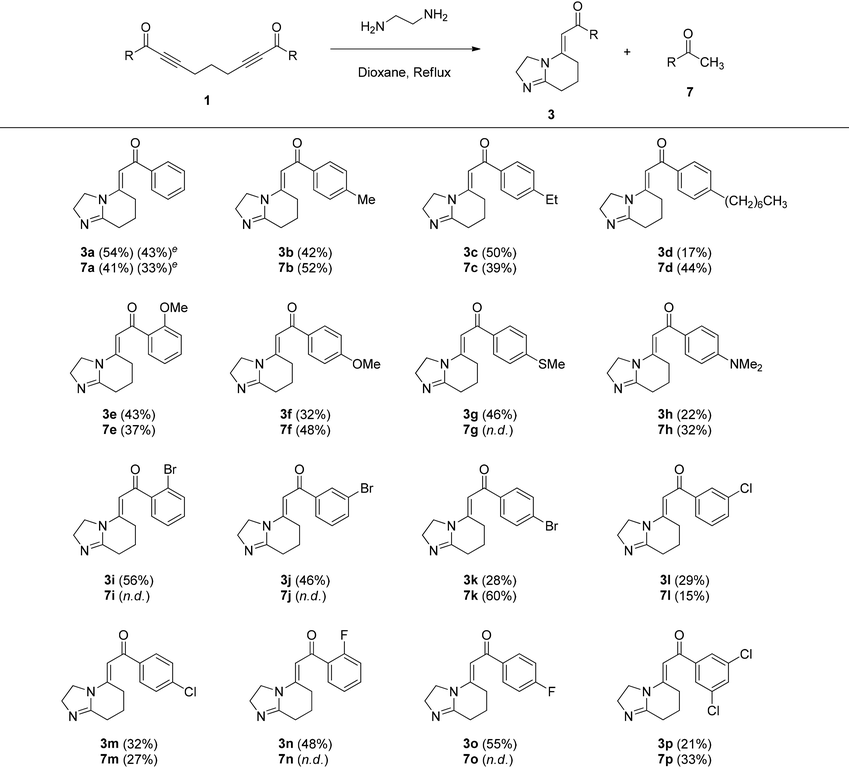
Initially, we synthesized the necessary starting materials, i.e., symmetrical and unsymmetrical bis-α,β-alkynic ketones 1 and 6, respectively, according to similar literature protocols, as depicted in Scheme 5.20 When subjected to Sonogashira coupling with aryloyl chlorides under palladium catalysis, 1,6-heptadiyne afforded doubly coupled symmetrical bis-α,β-alkynic ketones 1 and mono-coupled α,β-alkynic ketones 5. Notably, bis-α,β-alkynic ketones 1 were obtained as the major products from these reactions. We synthesized 16 derivatives of bis-α,β-alkynic ketones 1, the yields of which ranged between 33 and 64%. Mono-coupled α,β-alkynic ketones 5 were also obtained from these reactions as the minor products. Unfortunately, their formation could not be prevented. The yields of mono-coupled α,β-alkynic ketones 5 varied between 6–48% yields. When needed, mono-coupled α,β-alkynic ketones 5 could be subjected once again to Sonogashira coupling with the same aryloyl chlorides to generate symmetrical bis-α,β-alkynic ketones 1. On the other hand, Sonogashira coupling of α,β-alkynic ketones 5 with different aryloyl chlorides afforded unsymmetrical bis-α,β-alkynic ketones 6 (Scheme 5). By employing this approach, we synthesized three derivatives of unsymmetrical bis-α,β-alkynic ketones 6, the yields of which varied between 40 and 72%. For the identities and yields of the synthesized mono- and bis-α,β-alkynic ketones 1, 5 and 6, see the SI.With bis-α,β-alkynic ketones in hand, we next investigated their reactions with EDA. In order to test the reaction and optimize the conditions, we examined the reaction of 1,9-diphenylnona-2,7-diyne-1,9-dione (1a) with EDA as a model reaction, as depicted in Table 1. We first tried the reaction with 1.0, 2.0 and 3.0 equivalents of EDA in refluxing dioxane, which afforded 5-alkylidene-2,3,5,6,7,8-hexahydroimidazo[1,2-a]pyridine (HIP) 3a in 41–54% yields (Table 1, entries 1–3). Among them, the highest yield (54%) of product 3a was obtained with 2.0 equiv. of EDA. When the same reaction was carried out at room temperature, no products were obtained (Table 1, entry 4). We tested the reaction under concentrated conditions and in a pressure tube as well (Table 1, entries 5 and 6), but the yields of 3a (37 and 40%, respectively) did not improve. Then, we tried the reaction in refluxing toluene by using 1.0 and 2.0 equivalents of EDA, generating HIP 3a in 48 and 53% yields, respectively (Table 1, entries 7 and 8). Notably, the reactions with 2.0 equiv. of EDA in dioxane and toluene under reflux conditions furnished HIP 3a in very similar yields (54% vs. 53%). After these results, we continued the reactions with 2.0 equivalents of EDA. The reaction in refluxing p-xylene furnished HIP 3a in 45% yield (Table 1, entry 9). Notably, a longer reaction time at high temperature somewhat decreased the yield of HIP 3a (35%), possibly due to decomposition (Table 1, entry 10). Subsequently, we performed the reaction in acetonitrile and chloroform, yielding HIP 3a in 31 and 37% yields, respectively (Table 1, entries 11 and 12). We tested the reaction in alcohols and water as well (Table 1, entries 13–18). Among them, the highest yield (44%) of product 3a was observed in methanol. The reactions in butanol and water did not produce any products (Table 1, entries 17 and 18). Finally, we tried the reaction in N,N-dimethylformamide and dimethyl sulfoxide at 120 °C (Table 1, entries 19 and 20). The reaction in DMF produced the expected product 3a in 35% yield, but that in DMSO did not give any products. It should be mentioned that during the optimization reactions shown in Table 1, when the cyclization product HIP 3a formed, a methyl ketone derivative 7a, namely, acetophenone, was also obtained from these reactions in varying yields (11–43%). This is because the formation of the dihydroimidazole moiety of intermediate 2a occurs along with the formation of acetophenone (7a) as discussed in the mechanism. In summary, the generality and substrate coverage of the reaction were demonstrated by employing 1.0 and 2.0 equivalents of bis-α,β-alkynic ketones and EDA, respectively, in refluxing dioxane.
| Entry | Equiv. of EDAc | Solvent | Temp. (°C) | Time (h) | Yield of 3a (%) | Yield of 7ad (%) |
|---|---|---|---|---|---|---|
| a Reactions were performed on a scale of 0.25 mmol of 1,9-diphenylnona-2,7-diyne-1,9-dione (1a) in 5 mL of solvent under the indicated conditions unless noted. For workup and purification, see the Experimental section and SI. b Isolated yields. c Equivalency is given according to that of the starting bis-α,β-alkynic ketone 1a. d n.d. = yield not determined. e This reaction was carried out in a pressure tube by using 2.5 mL of solvent. f This reaction was performed in a pressure tube. g This reaction was performed in 2 mL of solvent. | ||||||
| 1 | 1.0 | Dioxane | 101 | 5 | 51 | n.d. |
| 2 | 2.0 | Dioxane | 101 | 4 | 54 | 41 |
| 3 | 3.0 | Dioxane | 101 | 7 | 41 | n.d. |
| 4 | 2.0 | Dioxane | Rt | 20 | — | — |
| 5e | 2.0 | Dioxane | 101 | 5 | 37 | 12 |
| 6f | 2.0 | Dioxane | 101 | 5 | 40 | n.d. |
| 7 | 1.0 | Toluene | 110 | 3 | 48 | n.d. |
| 8 | 2.0 | Toluene | 110 | 3 | 53 | 20 |
| 9 | 2.0 | p-Xylene | 138 | 2 | 45 | 29 |
| 10 | 2.0 | p-Xylene | 138 | 6 | 35 | n.d. |
| 11 | 2.0 | MeCN | 82 | 6 | 31 | n.d. |
| 12 | 2.0 | CHCl3 | 62 | 6 | 37 | 13 |
| 13 | 2.0 | MeOH | 65 | 6 | 44 | 11 |
| 14 | 2.0 | EtOH | 78 | 2 | 39 | 40 |
| 15 | 2.0 | n-Propanol | 97 | 7 | 15 | n.d. |
| 16 | 2.0 | Isopropanol | 82.5 | 8 | 16 | n.d. |
| 17 | 2.0 | n-BuOH | 117 | 4 | — | — |
| 18 | 2.0 | Water | 100 | 7 | — | — |
| 19g | 2.0 | DMF | 120 | 6 | 35 | 43 |
| 20 | 2.0 | DMSO | 120 | 4.5 | — | — |
Interestingly, HIP 3a was obtained as a single isomer from these reactions although in principle it could form as the E or Z isomer or a mixture of both isomers. We recorded the NOESY spectrum of 3a in order to assign the configuration of the exo double bond (see the SI). The spectrum showed a nuclear Overhauser effect (NOE) arising from a through-space interaction between vinylic hydrogen and methylene hydrogens adjacent to the tertiary nitrogen atom, as depicted in Table 1. Therefore, the configuration of the exo double bond of 3a was defined as E.
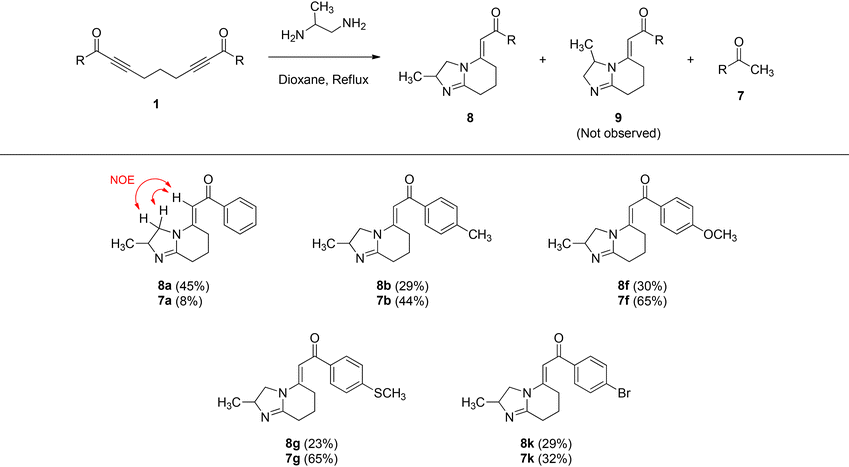
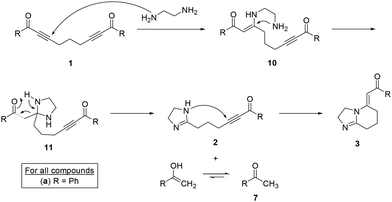
Afterwards, we explored the scope and limitations of the methodology by employing a variety of bis-α,β-alkynic ketones 1 to access 5-alkylidene-2,3,5,6,7,8-hexahydroimidazo[1,2-a]pyridines 3 (Table 2). It should be mentioned that hexahydroimidazo[1,2-a]pyridines 3 are generally quite polar compounds, and therefore, their characterization through column chromatography has been laborious, but we succeeded in their isolation in most cases. In fact, all reactions proceeded smoothly and provided the expected hexahydroimidazo[1,2-a]pyridines 3. Importantly, during the course of the reactions, three new C–N bonds were formed along with one C–C bond cleavage, which enabled the formation of new fused five- and six-membered heterocyclic ring systems 3. Overall, we synthesized 16 derivatives of hexahydroimidazo[1,2-a]pyridines 3, the yields of which changed from 17 to 56%. Halogen bonds play an important role in medicinal chemistry because of their ability to delicately influence drug–target interactions, such as binding affinity, selectivity, structural rigidity and conformation control, and absorption, distribution, metabolism and excretion (ADME) properties of a drug.21 In particular, fluorine atoms can significantly affect the biological activity, stability, and pharmacokinetics of drugs.22 Therefore, we synthesized eight derivatives (3i–p) of halogen-containing hexahydroimidazo[1,2-a]pyridines in 21–56% yields. During the synthesis of hexahydroimidazo[1,2-a]pyridines 3, aryl methyl ketones 7 were also obtained from these reactions in varying amounts (15–60%) (Table 2). Notably, aryl methyl ketones have emerged as valuable precursors and intermediates for the synthesis of a variety of medicinally important heterocycles.23 It is noteworthy that the expected products, hexahydroimidazo[1,2-a]pyridine 3 and aryl methyl ketone 7 from a particular starting bis-α,β-alkynic ketone 1, should form in a one-to-one ratio, as evident from their formation mechanism (Scheme 6). However, we were not able to obtain these products in a one-to-one ratio from column chromatography. Possibly, they decomposed and/or underwent side reactions to some extent. A similar situation was observed by Vasilevsky and Alabugin during the isolation of 4,5-dihydro-1H-imidazoles along with aryl methyl ketones (Scheme 4a).18 It is noteworthy that we carried out the reaction of bis-α,β-alkynic ketone 1a on a relatively larger scale (2.40 mmol) as well, which generated hexahydroimidazo[1,2-a]pyridine 3a in 43% yield, along with acetophenone (7a) in 33% yield (Table 2). Although the yields are slightly lower than those on a small scale, it certainly indicates the practical utility of the method as a synthetic tool.
 | ||
| Scheme 6 Proposed mechanism for the formation of 5-alkylidene-2,3,5,6,7,8-hexahydroimidazo[1,2-a]pyridines. | ||
| a Reactions were performed on a scale of 0.25 mmol of bis-α,β-alkynic ketones 1 and 0.50 mmol of ethylenediamine in 5 mL of dioxane at reflux. For workup and purification, see the Experimental section. b Isolated yields. c For the identities of aryl methyl ketones 7, see the SI. d n.d. = yield not determined. e Yield of the product obtained from the reaction performed on a 2.40 mmol scale of bis-α,β-alkynic ketones 1a (see the SI). |
|---|
|
In order to further widen the array of products, we also examined the reaction of several bis-α,β-alkynic ketones 1 with 1,2-propylenediamine (PDA) (1,2-diaminopropane) (Table 3). Although, in principle, these reactions could produce two possible products, they afforded 2-methyl-substituted 5-alkylidene-2,3,5,6,7,8-hexahydroimidazo[1,2-a]pyridines 8, along with aryl methyl ketones 7. Notably, 3-methyl-substituted hexahydroimidazo[1,2-a]pyridines 9 did not form in these reactions or formed in very low yields, which escaped isolation. The position of the methyl group was determined by an NOESY measurement. As depicted in Table 3, the NOESY spectrum of 8a showed NOE interactions between vinylic hydrogen and diastereotopic methylene hydrogens (see also the SI), proving the indicated structure of 8a. In fact, we synthesized five derivatives of 2-methyl-substituted HIPs 8 in 23–45% yields, along with the corresponding aryl methyl ketones 7 in 8–65% yields (Table 3). It should be noted that since racemic (achiral) PDA was used in these reactions, racemic (achiral) products 8 were obtained.
We investigated the reactions of a few unsymmetrical bis-α,β-alkynic ketones 6 with EDA as well. However, these reactions were not clean and produced a mixture of inseparable products, from which the expected products could not be isolated in pure form, and their clean NMR spectra could not be obtained. Consequently, we did not devote much effort to the reactions of unsymmetrical bis-α,β-alkynic ketones 6 with EDA.
The proposed mechanism for the synthesis of 5-alkylidene-2,3,5,6,7,8-hexahydroimidazo[1,2-a]pyridines 3 is given in Scheme 6. First, intermolecular conjugate (Michael-like) addition of EDA to bis-α,β-alkynic ketone 1 generates β-enaminone 10. Then, β-enaminone 10 undergoes a 5-exo-trig cyclization to afford imidazolidine 11via intramolecular conjugate (Michael-like) addition of the primary amine functionality to the enaminone moiety. Subsequently, intermediate 11 undergoes a retro-Mannich fragmentation via a six-membered transition state to produce dihydroimidazolyl-substituted α,β-alkynic ketone 2, along with aryl methyl ketone 7 after tautomerization. It should be noted that the formation of α,β-alkynic ketones 2 and aryl methyl ketones 7 in these reactions is consistent with the findings of earlier researchers.18 Finally, 6-exo-dig cyclization through another intramolecular conjugate (Michael-like) addition at 2 provides the corresponding hexahydroimidazo[1,2-a]pyridine 3 (Scheme 6).
It is noteworthy to mention that in the reaction performed by Vasilevsky and Alabugin and their coworkers, the polarized alkyne (C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C) unit is fully cleaved by the formation of final products, as depicted in Scheme 4a.18 However, in our reaction, such a cleavage of one alkyne moiety creates an advantage because the resulting intermediate 2 (dihydroimidazolyl-substituted α,β-alkynic ketone) serves as the starting material for the synthesis of 2,3,5,6,7,8-hexahydroimidazo[1,2-a]pyridine derivatives 3 (Schemes 4b and 6). We performed a few control experiments to capture the intermediate product 2a. Therefore, we carried out the reaction of 1a at various temperatures (rt, 40 and 60 °C) in THF, methanol, dioxane and toluene. At rt and 40 °C, only the first conjugate addition step (1a → 10a) took place in these solvents, as concluded from 1H NMR analysis of crude reaction mixtures, and we did not see any evidence for the formation of any cyclic intermediate products. At 60 °C, a similar trend was observed in dioxane and toluene, producing only the conjugate addition product. However, in methanol at 60 °C, hexahydroimidazo[1,2-a]pyridine 3a was also formed in addition to the conjugate addition product, but in a low amount (37%), and we could not capture the intermediate product 2a in this reaction. In brief, in these control experiments, we did not see any spectroscopic evidence for the formation of intermediate 2a. When the reaction was carried out at 100 °C, such as in dioxane, the final product 3a was obtained along with aryl methyl ketone 7a. Presumably, the second conjugate addition (10a → 11a) requires higher temperatures and proceeds around 100 °C. Furthermore, once imidazolidine 11a is formed, it undergoes immediate fragmentation to yield intermediate 2a and aryl methyl ketone 7a. So far, our observations are consistent with the findings of Vasilevsky and Alabugin on similar systems.18 In general, their experimental results and DFT calculations showed that the first conjugate addition of EDA to α,β-alkynic ketones (as in 1 → 10) easily occurs at room temperature, while the second intramolecular conjugate addition (as in 10 → 11) requires higher temperatures such as 100 °C although it is a favored 5-exo-trig closure according to Baldwin's rules.24 Moreover, the activation barriers for such 5-exo-trig ring closures have been calculated to be substantial (>26–30 kcal mol−1). They also found that the calculated activation barrier for the fragmentation step (as in 11 → 2 + 7) is slightly lower than that for the 5-exo-trig cyclization (as in 10 → 11), indicating that the second intramolecular conjugate addition (i.e., 10 → 11) is the rate-limiting step in this process. Thus, in our case, we conclude that when the reaction reaches a sufficient temperature (approximately 100 °C), the intermediate product 2 forms and immediately undergoes another conjugate addition between its secondary amine and α,β-alkynic ketone functionalities to give hexahydroimidazo[1,2-a]pyridine 3. Notably, the activation barrier for 2 → 3 is much lower than those for 10 → 11 and 11 → 2. Since the intermediate 2 contains both nucleophilic and electrophilic centers in its structure, it enables a very fast intramolecular cyclization via 6-exo-dig closure to produce the final product 3 (Scheme 6). This might be the reason why we could not observe the formation of intermediate 2.
C) unit is fully cleaved by the formation of final products, as depicted in Scheme 4a.18 However, in our reaction, such a cleavage of one alkyne moiety creates an advantage because the resulting intermediate 2 (dihydroimidazolyl-substituted α,β-alkynic ketone) serves as the starting material for the synthesis of 2,3,5,6,7,8-hexahydroimidazo[1,2-a]pyridine derivatives 3 (Schemes 4b and 6). We performed a few control experiments to capture the intermediate product 2a. Therefore, we carried out the reaction of 1a at various temperatures (rt, 40 and 60 °C) in THF, methanol, dioxane and toluene. At rt and 40 °C, only the first conjugate addition step (1a → 10a) took place in these solvents, as concluded from 1H NMR analysis of crude reaction mixtures, and we did not see any evidence for the formation of any cyclic intermediate products. At 60 °C, a similar trend was observed in dioxane and toluene, producing only the conjugate addition product. However, in methanol at 60 °C, hexahydroimidazo[1,2-a]pyridine 3a was also formed in addition to the conjugate addition product, but in a low amount (37%), and we could not capture the intermediate product 2a in this reaction. In brief, in these control experiments, we did not see any spectroscopic evidence for the formation of intermediate 2a. When the reaction was carried out at 100 °C, such as in dioxane, the final product 3a was obtained along with aryl methyl ketone 7a. Presumably, the second conjugate addition (10a → 11a) requires higher temperatures and proceeds around 100 °C. Furthermore, once imidazolidine 11a is formed, it undergoes immediate fragmentation to yield intermediate 2a and aryl methyl ketone 7a. So far, our observations are consistent with the findings of Vasilevsky and Alabugin on similar systems.18 In general, their experimental results and DFT calculations showed that the first conjugate addition of EDA to α,β-alkynic ketones (as in 1 → 10) easily occurs at room temperature, while the second intramolecular conjugate addition (as in 10 → 11) requires higher temperatures such as 100 °C although it is a favored 5-exo-trig closure according to Baldwin's rules.24 Moreover, the activation barriers for such 5-exo-trig ring closures have been calculated to be substantial (>26–30 kcal mol−1). They also found that the calculated activation barrier for the fragmentation step (as in 11 → 2 + 7) is slightly lower than that for the 5-exo-trig cyclization (as in 10 → 11), indicating that the second intramolecular conjugate addition (i.e., 10 → 11) is the rate-limiting step in this process. Thus, in our case, we conclude that when the reaction reaches a sufficient temperature (approximately 100 °C), the intermediate product 2 forms and immediately undergoes another conjugate addition between its secondary amine and α,β-alkynic ketone functionalities to give hexahydroimidazo[1,2-a]pyridine 3. Notably, the activation barrier for 2 → 3 is much lower than those for 10 → 11 and 11 → 2. Since the intermediate 2 contains both nucleophilic and electrophilic centers in its structure, it enables a very fast intramolecular cyclization via 6-exo-dig closure to produce the final product 3 (Scheme 6). This might be the reason why we could not observe the formation of intermediate 2.
The proposed mechanistic rationale for the formation of 2-methyl-substituted hexahydroimidazo[1,2-a]pyridines 8 is shown in Scheme 7. The reaction of bis-α,β-alkynic ketones 1 with PDA first produces in situ tautomeric dihydroimidazolyl-substituted α,β-alkynic ketones 12 and 13, which then give rise to the formation of 2- and 3-methyl-substituted HIPs 8 and 9, respectively, by a similar mechanism as given in Scheme 6. Notably, the secondary nitrogen atom in 12 is farther from the methyl substituent; therefore, it is more available as a nucleophile for the intramolecular conjugate addition to afford the final HIP 8. However, in 13, the methyl group increases the steric hindrance around the nearby secondary nitrogen atom, making it significantly less accessible for intramolecular conjugate addition to lead to the final HIP 9 although it is expected to be slightly more electron-rich (nucleophilic) due to the +I effect of the adjacent methyl substituent, compared to that in 12. In brief, steric accessibility is more dominant for nucleophilicity, and these reactions afforded 2-methyl-substituted HIPs 8 through the intermediacy of 12 as the major or single product as expected (Scheme 7).
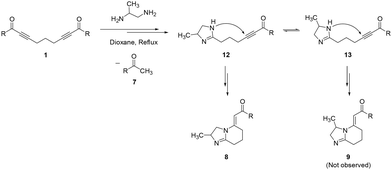 | ||
| Scheme 7 Proposed mechanistic rationale for the formation of 2-methyl-substituted 5-alkylidene-2,3,5,6,7,8-hexahydroimidazo[1,2-a]pyridines. | ||
Conclusions
In summary, we have uncovered a novel reaction of bis-α,β-alkynic ketones, i.e., 1,9-diarylnona-2,7-diyne-1,9-diones. When treated with ethylenediamine in dioxane under reflux, bis-α,β-alkynic ketones produce 5-alkylidene-2,3,5,6,7,8-hexahydroimidazo[1,2-a]pyridines in low to moderate yields. During the course of the reaction, three new C–N bonds form along with one C–C bond cleavage, resulting in the formation of unknown fused five- and six-membered heterocyclic ring systems. The reaction proceeds via the intermediacy of a five-membered imidazolidine ring. A similar trend has been observed in the reaction of bis-α,β-alkynic ketones with 1,2-propylenediamine, where only 2-methyl-substituted 5-alkylidene-2,3,5,6,7,8-hexahydroimidazo[1,2-a]pyridines were obtained. The skeletal diversity of the synthesized hexahydroimidazo[1,2-a]pyridine framework may provide many new nitrogen-based heterobicyclic systems for drug discovery and development.Experimental
General procedure for the synthesis of 1,9-diarylnona-2,7-diyne-1,9-diones 1 (Scheme 5)
To a stirred solution of the appropriate aryloyl chloride (3.0 mmol) in THF (12 mL) under argon were added PdCl2(PPh3)2 (0.03 mmol) and Et3N (1.8 mmol), and the resulting mixture was stirred for 10 min at room temperature and CuI (0.03 mmol) was added. After stirring the reaction mixture for another 10 min, 1,6-heptadiyne (4) (1.5 mmol) was added slowly, and the stirring at room temperature continued until the end of the reaction (the progress of the reaction was monitored by routine TLC analysis for the disappearance of 1,6-heptadiyne (4) using 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 hexane/ethyl acetate as the eluent). After the reaction was complete, the solvent was removed on a rotary evaporator, and ethyl acetate (50 mL) and a saturated aqueous NH4Cl solution (50 mL) were added. After the layers were separated, the aqueous layer was extracted with ethyl acetate (2 × 50 mL). The combined organic layers were dried over MgSO4 and evaporated on a rotary evaporator to give the crude product, which was purified by flash chromatography on silica gel using 9
1 hexane/ethyl acetate as the eluent). After the reaction was complete, the solvent was removed on a rotary evaporator, and ethyl acetate (50 mL) and a saturated aqueous NH4Cl solution (50 mL) were added. After the layers were separated, the aqueous layer was extracted with ethyl acetate (2 × 50 mL). The combined organic layers were dried over MgSO4 and evaporated on a rotary evaporator to give the crude product, which was purified by flash chromatography on silica gel using 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 hexane/ethyl acetate as the eluent to afford the corresponding symmetrical 1,9-diarylnona-2,7-diyne-1,9-dione 1 and 1-arylocta-2,7-diyn-1-one 5.
1 hexane/ethyl acetate as the eluent to afford the corresponding symmetrical 1,9-diarylnona-2,7-diyne-1,9-dione 1 and 1-arylocta-2,7-diyn-1-one 5.
General procedure for the synthesis of 1-aryl-2-(2,3,7,8-tetrahydroimidazo[1,2-a]pyridin-5(6H)-ylidene)ethanones 3 (Table 2)
To a stirred solution of the appropriate 1,9-diarylnona-2,7-diyne-1,9-dione 1 (0.25 mmol) in dioxane (5.0 mL) under argon was added ethylenediamine (0.50 mmol), and the resulting mixture was refluxed until the end of the reaction (the progress of the reaction was monitored by routine TLC analysis for the disappearance of bis-α,β-alkynic ketone 1 using 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 hexane/ethyl acetate as the eluent). After the reaction was complete, the solvent was removed on a rotary evaporator, and the resulting crude product was purified by flash chromatography on silica gel using 4
1 hexane/ethyl acetate as the eluent). After the reaction was complete, the solvent was removed on a rotary evaporator, and the resulting crude product was purified by flash chromatography on silica gel using 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 hexane/ethyl acetate, followed by 1
1 hexane/ethyl acetate, followed by 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 methanol/ethyl acetate, as the eluent to afford the corresponding 1-aryl-2-(2,3,7,8-tetrahydroimidazo[1,2-a]pyridin-5(6H)-ylidene)ethanone 3 and aryl methyl ketone 7.
1 methanol/ethyl acetate, as the eluent to afford the corresponding 1-aryl-2-(2,3,7,8-tetrahydroimidazo[1,2-a]pyridin-5(6H)-ylidene)ethanone 3 and aryl methyl ketone 7.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: experimental procedures, spectroscopic data and copies of NMR spectra for starting materials and products (PDF). See DOI: https://doi.org/10.1039/d5ob01892g.Acknowledgements
We thank the Scientific and Technological Research Council of Turkey (TUBITAK, Grant No. 123Z511) for financial support of this research and the Middle East Technical University (METU) for providing spectroscopic facilities.References
- (a) N. Punna, S. S. Jadav, A. Ramaraju, R. R. Donthiri and C. R. Reddy, Bicyclic 5-6 Systems With One Bridgehead (Ring Junction) Nitrogen Atom: One Extra Heteroatom 1:0, in Comprehensive Heterocyclic Chemistry IV, ed. D. StC Black, J. Cossy, C. V. Stevens and G. Masson, Elsevier Science, 2022, ch. 11.09, vol. 11, pp. 596–621 Search PubMed; (b) L. A. P. Ferreira, L. Caruso, N. F. Nadur, D. P. Franco, G. L. S. Sousa, R. B. Lacerda and A. E. Kummerle, Imidazo[1,2-a]pyridines in Medicinal Chemistry: Recent Advances in Synthesis and Biological Activities, ACS Omega, 2026, 11, 2348–2383 CrossRef CAS PubMed.
- A. Deep, R. K. Bhatia, R. Kaur, S. Kumar, U. K. Jain, H. Singh, S. Batra, D. Kaushik and P. K. Deb, Imidazo[1,2-a]pyridine Scaffold as Prospective Therapeutic Agents, Curr. Top. Med. Chem., 2017, 17, 238–250 Search PubMed.
- J. Panda, B. P. Raiguru, M. Mishra, S. Mohapatra and S. Nayak, Recent Advances in the Synthesis of Imidazo[1,2-a]pyridines: A Brief Review, ChemistrySelect, 2022, 7, e202103987 CrossRef CAS.
- C. Enguehard-Gueiffier and A. Gueiffier, Recent Progress in the Pharmacology of Imidazo[1,2-a]pyridines, Mini-Rev. Med. Chem., 2007, 7, 888–899 CrossRef CAS PubMed.
- A. K. Bagdi, S. Santra, K. Monir and A. Hajra, Synthesis of imidazo[1,2-a]pyridines: a decade update, Chem. Commun., 2015, 51, 1555–1575 Search PubMed.
- K. Pericherla, P. Kaswan, K. Pandey and A. Kumar, Recent Developments in the Synthesis of Imidazo[1,2-a]pyridines, Synthesis, 2015, 887–912 Search PubMed.
- A. B. Fulp and X. Wang, Asymmetric Benzimidazoles and Related Compouns as Potassium Channel Modulators, WO2005002503A2, 2005.
- W. Deady and T. Rodemann, Reduced Benzimidazo[2,1-a]isoquinolines. Synthesis and Cytotoxicity Studies, Aust. J. Chem., 2001, 54, 529–534 CrossRef.
- T. Okubo, R. Yoshikawa, S. Chaki, S. Okuyamac and A. Nakazato, Design, synthesis, and structure–activity relationships of novel tetracyclic compounds as peripheral benzodiazepine receptor ligands, Bioorg. Med. Chem., 2004, 12, 3569–3580 CrossRef CAS PubMed.
- For recent examples, see: (a) A. J. Zahara, B. E. Haines and S. M. Wilkerson-Hill, Programmed Heterocycle Synthesis Using Halomucononitriles as Pyridinimine Precursors, Org. Lett., 2024, 26, 2976–2981 CrossRef CAS PubMed; (b) Y. Zhang, R. Chen, Z. Wang, L. Wang and Y. Ma, I2-Catalyzed Three-Component Consecutive Reaction for the Synthesis of 3-Aroylimidazo[1,2-a]-N-Heterocycles, J. Org. Chem., 2021, 86, 6239–6246 CrossRef CAS PubMed; (c) F. Vuillermet, J. Bourret and G. Pelletier, Synthesis of Imidazo[1,2-a]pyridines: Triflic Anhydride-Mediated Annulation of 2H-Azirines with 2-Chloropyridines, J. Org. Chem., 2021, 86, 388–402 CrossRef CAS PubMed; (d) H. Okai, K. Tanimoto, R. Ohkado and H. Iida, Multicomponent Synthesis of Imidazo[1,2-a]pyridines: Aerobic Oxidative Formation of C−N and C−S Bonds by Flavin−Iodine-Coupled Organocatalysis, Org. Lett., 2020, 22, 8002–8006 CrossRef CAS PubMed; (e) S. Cacchi, A. Ciogli, N. Demitri, G. Fabrizi, F. Ghirga, A. Goggiamani, A. Iazzetti and D. Lamba, Copper-Catalyzed C–N Bond Formation via C–H Functionalization: Facile Synthesis of Multisubstituted Imidazo[1,2-a]pyridines from N,-(2-Pyridinyl)enaminones, Synthesis, 2018, 3513–3519 CAS.
- For selected examples, see: (a) S. O. Kushch, M. V. Goryaeva, E. A. Surnina, Y. V. Burgart, M. A. Ezhikova, M. I. Kodess, P. A. Slepukhin and V. I. Saloutin, Multicomponent Domino Reactions for the Synthesis of Variable Hydrogenated Imidazo[1,2-a]pyridines, Asian J. Org. Chem., 2022, 11, e202100709 CrossRef CAS; (b) H. Tan and Y. Wang, Facile Synthesis of Novel Hexahydroimidazo[1,2-a]pyridine Derivatives by One-Pot, Multicomponent Reaction under Ambient Conditions, ACS Comb. Sci., 2020, 22, 468–474 CrossRef CAS PubMed; (c) H. G. O. Alvim, J. R. Correa, J. A. F. Assumpção, W. A. da Silva, M. O. Rodrigues, J. L. de Macedo, M. Fioramonte, F. C. Gozzo, C. C. Gatto and B. A. D. Neto, Heteropolyacid-Containing Ionic Liquid-Catalyzed Multicomponent Synthesis of Bridgehead Nitrogen Heterocycles: Mechanisms and Mitochondrial Staining, J. Org. Chem., 2018, 83, 4044–4053 CrossRef CAS PubMed; (d) K. M. Wang, Y. L. Ma, X. R. Lin, S. J. Yan and J. Lin, Regioselective synthesis of pyrrolo[1,2-a]imidazoles and imidazo[1,2-a]pyridines, RSC Adv., 2015, 5, 36472–36479 RSC; (e) R. L. Wang, P. Zhu, Y. Lu, F. P. Huang and X. P. Huia, Brønsted Acid-Catalyzed Four-Component Cascade Reaction: Facile Synthesis of Hexahydroimidazo[1,2-a]pyridines., Adv. Synth. Catal., 2013, 355, 87–92 CrossRef CAS.
- E. E. Mikhlina, V. Y. Vorob'eva and L. N. Yakhontov, The Reduction of 1,7-Diazabicyclo[4,3,0]non-6-ene and of 2,3-Dihydroimidazo[1,2-a]pyridine as Azacyclic Compounds with an Amidine Structure, Chem. Heterocycl. Compd., 1970, 6, 457–459 CrossRef.
- J. Rokach, P. Hamel, N. R. Hunter, G. Reader and C. S. Rooney, Cyclic Amidine Inhibitors of Indolamine N-Methyltransferase, J. Med. Chem., 1979, 22, 237–247 CrossRef CAS PubMed.
- H. Yamamoto and K. Maruoka, Regioselective Carbonyl Amination Using Diisobutylaluminum Hydride., J. Am. Chem. Soc., 1981, 103, 4186–4194 CrossRef CAS.
- V. B. Birman, X. Li and Z. Han, Nonaromatic Amidine Derivatives as Acylation Catalysts, Org. Lett., 2007, 9, 37–40 CrossRef CAS PubMed.
- (a) X. Zhang, Y. Zhou, H. Wang, D. Guo, D. Ye, Y. Xu, H. Jiang and H. Liu, Silver-catalyzed intramolecular hydroamination of alkynes in aqueous media: efficient and regioselective synthesis for fused benzimidazoles, Green Chem., 2011, 13, 397–405 RSC; (b) X. Zhang, Y. Zhou, H. Wang, D. Guo, D. Ye, Y. Xu, H. Jiang and H. Liu, An Effective Synthetic Entry to Fused Benzimidazoles via Iodocyclization, Adv. Synth. Catal., 2011, 353, 1429–1437 CrossRef CAS.
- S. Sharma, A. K. Jha and S. Easwar, A retro-Mannich mediated transformation of Morita–Baylis–Hillman ketones to saturated imidazo[1,2-a]pyridines, Org. Chem. Front., 2024, 11, 3137–3150 RSC.
-
(a) S. Roy, M. P. Davydova, R. Pal, K. Gilmore, G. A. Tolstikov, S. F. Vasilevsky and I. V. Alabugin, Dissecting Alkynes: Full Cleavage of Polarized C≡C Moiety via Sequential Bis-Michael Addition/Retro-Mannich Cascade, J. Org. Chem., 2011, 76, 7482–7490 CrossRef CAS PubMed. For the full cleavage of C
![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C bonds, see also:
(b) S. F. Vasilevsky, D. S. Baranov, V. I. Mamatyuk, Y. V. Gatilov and I. V. Alabugin, An Unexpected Rearrangement That Disassembles Alkyne Moiety Through Formal Nitrogen Atom Insertion between Two Acetylenic Carbons and Related Cascade Transformations: New Approach to Sampangine Derivatives and Polycyclic Aromatic Amides, J. Org. Chem., 2009, 74, 6143–6150 Search PubMed;
(c) S. F. Vasilevsky, M. P. Davydova, V. I. Mamatyuk, N. Tsvetkov, A. Hughes, D. S. Baranov and I. V. Alabugin, Full Cleavage of C≡C Bond in Electron-Deficient Alkynes via Reaction with Ethylenediamine, Aust. J. Chem., 2017, 70, 421–429 CrossRef CAS.
C bonds, see also:
(b) S. F. Vasilevsky, D. S. Baranov, V. I. Mamatyuk, Y. V. Gatilov and I. V. Alabugin, An Unexpected Rearrangement That Disassembles Alkyne Moiety Through Formal Nitrogen Atom Insertion between Two Acetylenic Carbons and Related Cascade Transformations: New Approach to Sampangine Derivatives and Polycyclic Aromatic Amides, J. Org. Chem., 2009, 74, 6143–6150 Search PubMed;
(c) S. F. Vasilevsky, M. P. Davydova, V. I. Mamatyuk, N. Tsvetkov, A. Hughes, D. S. Baranov and I. V. Alabugin, Full Cleavage of C≡C Bond in Electron-Deficient Alkynes via Reaction with Ethylenediamine, Aust. J. Chem., 2017, 70, 421–429 CrossRef CAS. - (a) Y. Kelgokmen, Y. Cayan and M. Zora, Zinc Chloride Mediated Synthesis of 1,4-Oxazepines from N-Propargylic β-Enaminones, Eur. J. Org. Chem., 2017, 7167–7178 CrossRef CAS; (b) Y. Kelgokmen and M. Zora, Synthesis of 1,4-Thiazepines, J. Org. Chem., 2018, 83, 8376–8379 CrossRef CAS PubMed; (c) Y. Kelgokmen and M. Zora, A new strategy for the synthesis of pyridines from N-propargylic β-enaminothiones, Org. Biomol. Chem., 2019, 17, 2529–2541 RSC; (d) E. Korkmaz and M. Zora, Synthesis of 3-[(4-Nitrophenyl)thio]-Substituted 4-Methylene-1-pyrrolines from N-Propargylic β-Enaminones, J. Org. Chem., 2020, 85, 4937–4950 CrossRef CAS PubMed; (e) N. Kanova, B. A. Dundar, Y. Kelgokmen and M. Zora, One-Pot Synthesis of 2-Acetyl-1H-pyrroles from N-Propargylic β-Enaminones via Intermediacy of 1,4-Oxazepines, J. Org. Chem., 2021, 86, 6289–6304 CrossRef CAS PubMed; (f) N. E. Aksakal and M. Zora, InCl3-Catalyzed One-Pot Synthesis of Pyrrolo/Indolo- and Benzooxazepino-Fused Quinoxalines, ACS Omega, 2024, 9, 33251–33260 CAS; (g) E. S. Yilmaz, K. Kaya and M. Zora, Facile synthesis of unknown 6,7-dihydrofuro[3,4-c]pyridines and 3,4-diaryloylpyridines from N-homopropargylic β-enaminones, Org. Biomol. Chem., 2025, 23, 2133–2141 RSC.
- (a) R. J. Cox, D. J. Ritson, T. A. Dane, J. Berge, J. P. H. Charmant and A. Kantacha, Room temperature palladium catalysed coupling of acyl chlorides with terminal alkynes, Chem. Commun., 2005, 8, 1037–1039 RSC; (b) S. Cacchi, G. Fabrizi and E. Filisti, N-Propargylic β-Enaminones: Common Intermediates for the Synthesis of Polysubstituted Pyrroles and Pyridines, Org. Lett., 2008, 10, 2629–2632 CrossRef CAS PubMed; (c) S. Karabiyikoglu, Y. Kelgokmen and M. Zora, Facile Synthesis of Iodopyridines from N-Propargylic β-Enaminones via Iodine-Mediated Electrophilic Cyclization, Tetrahedron, 2015, 71, 4324–4333 CrossRef CAS; (d) G. Albano and L. A. Aronica, Acyl Sonogashira Cross-Coupling: State of the Art and Application to the Synthesis of Heterocyclic Compounds, Catalysts, 2020, 10, 25 CrossRef; (e) P. A. Volkov, K. O. Khrapova, I. A. Bidusenko, A. A. Telezhkin, E. Y. Schmidt, A. I. Albanov and B. A. Trofimov, Chemoselective cross-coupling of terminal propargylamines with (het)aroyl chlorides: synthesis of β-aminoacetylenic ketones bearing aromatic and heteroaromatic substituents, Russ. Chem. Bull., 2022, 71, 1514–1518 CrossRef CAS; (f) P. A. Volkov, K. O. Khrapova, A. A. Telezhkin, I. A. Bidusenko, E. Y. Schmidt, A. I. Albanov and B. A. Trofimov, Pd/Cu-Catalyzed Cross-Coupling of Densely Substituted Propargylamines with Aromatic Acyl Chlorides Followed by the Treatment with a Base: Access to Dihydro-3H-Pyrrol-3-Ones, Adv. Synth. Catal., 2023, 365, 53–67 CrossRef CAS.
- (a) M. Hernandes, S. M. Cavalcanti, D. R. Moreira, W. de Azevedo Junior and A. C. Leite, Halogen Atoms in the Modern Medicinal Chemistry: Hints for the Drug Design, Curr. Drug Targets, 2010, 11, 303–314 CrossRef CAS PubMed; (b) N. K. Shinada, A. G. de Brevern and P. Schmidtke, Halogens in Protein−Ligand Binding Mechanism: A Structural Perspective, J. Med. Chem., 2019, 62, 9341–9356 CrossRef CAS PubMed.
- (a) J. Wang, M. Sanchez-Rosello, J. L. Acena, C. del Pozo, A. E. Sorochinsky, S. Fustero, V. A. Soloshonok and H. Liu, Fluorine in Pharmaceutical Industry: Fluorine-Containing Drugs Introduced to the Market in the Last Decade (2001–2011), Chem. Rev., 2014, 114, 2432–2506 CrossRef CAS PubMed; (b) E. P. Gillis, K. J. Eastman, M. D. Hill, D. J. Donnelly and N. A. Meanwell, Applications of Fluorine in Medicinal Chemistry, J. Med. Chem., 2015, 58, 8315–8359 CrossRef CAS PubMed.
- (a) S. Rajai-Daryasarei, M. H. Gohari and N. Mohammadi, Reactions involving aryl methyl ketone and molecular iodine: a powerful tool in the one-pot synthesis of heterocycles, New J. Chem., 2021, 45, 20486–20518 RSC; (b) S. Mohammed, J. S. West and M. J. Mitton-Fry, Aryl Methyl Ketones: Versatile Synthons in the Synthesis of Heterocyclic Compounds, SynOpen, 2022, 6, 110–131 CrossRef CAS PubMed; (c) D. S. Yang, X. L. Chen and A. X. Wu, Review of application of the I2 and dimethyl sulfoxide combined reagent system to aryl methyl ketones for diverse transformations, Org. Chem. Front., 2024, 11, 2665–2692 RSC.
- (a) J. E. Baldwin, Rules for ring closure, J. Chem. Soc., Chem. Commun., 1976, 18, 734–736 RSC; (b) I. Alabugin, K. Gilmore and M. Manoharan, Rules for Anionic and Radical Ring Closure of Alkynes, J. Am. Chem. Soc., 2011, 133, 12608–12623 CrossRef CAS PubMed; (c) K. Gilmore and I. V. Alabugin, Cyclizations of Alkynes: Revisiting Baldwin's Rules for Ring Closure, Chem. Rev., 2011, 111, 6513–6556 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |