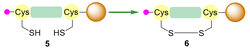Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Expanding the scope of sustainable peptide synthesis through post-linear synthesis reactions
Michael A.
Malone
 ,
Matilde
De Francisco Craparotta
,
Kirsty I. M.
Arnott
,
Andrew G.
Jamieson
,
Matilde
De Francisco Craparotta
,
Kirsty I. M.
Arnott
,
Andrew G.
Jamieson
 and
Nicola
Wade
and
Nicola
Wade
 *
*
School of Chemistry, Advanced Research Centre, University of Glasgow, 11 Chapel Lane, Glasgow, G11 6EW, UK. E-mail: Nicola.Wade@glasgow.ac.uk
First published on 30th January 2026
Abstract
The replacement of N,N-dimethylformamide (DMF) in solid-phase peptide synthesis (SPPS) is critical for improving the sustainability and safety of peptide research. While N-butyl-2-pyrrolidinone (NBP) has emerged as a viable alternative for linear peptide synthesis, its applicability to post-linear synthesis modifications remains underexplored. Here, we demonstrate the use of NBP as an alternative solvent for key on-resin transformations required in peptidomimetic synthesis. Orthogonal protecting group strategies were successfully implemented, enabling selective removal of Cys(SIT), Cys(Acm), Cys(Trt), and Lys(Dde) under mild conditions. Notably, SIT deprotection achieved 92% conversion in a single 4-hour treatment, while Acm and Trt were selectively removed using iodine or N-halosuccinimides, respectively. Lys(Dde) was efficiently cleaved in the presence of Cys(Acm) using 4% hydrazine in NBP. Furthermore, on-resin cyclisation via disulfide bond and 1,4-disubstituted triazole formation proceeded effectively in NBP, as did N-terminal acetylation under mild conditions. These findings establish NBP as a practical and greener alternative to DMF for diverse post-linear synthesis modifications, supporting the development of sustainable SPPS protocols.
Introduction
The development of solid phase peptide synthesis (SPPS) is used to access a wide variety of peptidomimetic structures through the addition of natural and synthetic amino acid moieties. Currently, N,N-dimethylformamide (DMF) is the gold standard solvent associated with SPPS, providing optimal swelling of the solid support and solvation of reagents at high concentrations.1 However, DMF is a known liver toxin in addition to being a reproductive hazard, with evidence indicating a high risk to foetal development.2,3 Subsequently, regulations sanctioned by the European Commission in 2023 have imposed DMF use restrictions; mandating lower levels of short and long term exposures.4 Thus, there is a requirement for safer solvent alternatives compatible with SPPS.5For a solvent to be suitable for SPPS, it must meet several key criteria: (i) provide adequate swelling of the solid support, (ii) effectively solubilise SPPS reagents, and (iii) maintain compatibility with coupling and deprotection chemistries.1 However, to be considered as a long-term DMF replacement, the solvent(s) must also comply with the safety standards set by the European Chemicals Agency under the Registration, Evaluation, Authorisation and Restriction of Chemicals (REACH) regulation.6 Beyond DMF, a number of solvents and binary solvent systems have been identified that satisfy these requirements.7–12
Binary solvent systems allow for the adjustment of chemical and physical features of solvents such as viscosity or polarity.11,13,14 Of recent note, a binary system of EtOAc/DMSO implemented by Polypeptide, BaChem, and Novo Nordisk has proven successful in synthesising longer peptide sequences.10,15
For singular solvents, N-butyl-2-pyrrolidinone (NBP) has drawn attention in several studies as a non-reprotoxic and biodegradable alternative to DMF (Fig. 1).16 NBP was found to limit side reactions common to linear SPPS, such as racemisation and aspartimide formation, becoming a top candidate for the synthesis of short peptides.17,18 However, potential drawbacks include a high boiling point and viscosity, while also being prone to peroxide formation.19 In order to reduce the viscosity and cost associated with NBP, binary solvent mixtures of NBP with EtOAc or DOL have also been investigated.10,20
Thus far, solvent investigations have focussed on the linear synthesis of peptides. While this is an essential advancement towards sustainable technologies, further investigations into post-linear synthesis modifications, to the best of our knowledge, have not been published. Orthogonal protecting group removal, subsequent peptide functionalisation and other post-synthesis modifications are typically achieved utilising DMF.21–23 Therefore, methodologies to facilitate on-resin peptidyl transformations, abating DMF, are essential for achieving completely sustainable SPPS protocols.
We sought to develop new methods for the post-linear synthesis modifications of peptides using a non-reprotoxic solvent. As this study was not focussed on automated linear SPPS, we elected to investigate a singular solvent rather than one of the previously mentioned binary systems. Despite the potential drawbacks of NBP, our main research interest is in relatively short peptides and so we elected to expand the reaction profile of NBP.
Herein, we describe the use of NBP as a replacement for DMF in several important post-synthesis modifications, including the selective deprotection of orthogonal cysteine and lysine protecting groups, N-terminal capping and on-resin cyclisation methods, via disulfide bond or 1,4-triazole formation. Each reaction was optimised for highest conversion to product while also considering the 12 principles of green chemistry.24,25
Experimental
General procedure for peptide synthesis
Peptides were synthesised batchwise as required on a 0.3 mmol scale using Fmoc-rink amide AM resin (0.53 mmol g−1 loading, 0.566 g). Resin swelling was achieved in NBP (5 mL) for 1 hour. Peptides were elongated in cycles of amino acid coupling followed by Fmoc removal. Fmoc-protected amino acid (3 equiv., 0.1 M in NBP) coupling was achieved by treatment with DIC (3 equiv., 0.1 M in NBP) and Oxyma (3 equiv., 0.1 M in NBP) at r.t. for 2 × 40 min. Fmoc removal was achieved by treatment with 20% piperidine in NBP (8 mL, v/v/v) at r.t. for 1 × 5 min followed by r.t. 1 × 15 min. The resin was washed with NBP following Fmoc removals (2 × 5 mL), MeOH (2 × 5 mL), and CH2Cl2 (2 × 5 mL), and after coupling NBP (2 × 5 mL).General procedure for SIT deprotections
The peptidyl resin (1 equiv.) was swollen in NBP (200 µL) at 50 °C for 20 min. The peptide was cooled to room temperature and treated with a solution of DTT (15 mg, 0.1 mmol, 10 equiv.) dissolved in 4% DIPEA, 4% H2O in NBP (1 mL, v/v/v) for 4 hours. Following DTT treatment, the resin was thoroughly washed with NBP (3 × 1 mL), MeOH (3 × 1 mL), CH2Cl2 (3 × 1 mL), and dried under vacuum.General procedure for I2 Trt/Acm removal
The peptidyl resin (1 equiv.) was swollen in NBP (200 µL) at 50 °C for 20 min. The peptide was cooled to room temperature and treated with a solution of iodine (10 mg, 0.04 mmol, 4 equiv.) dissolved in NBP (200 µL) and Na2S2O8 (5 mg, 0.02 mmol, 2 equiv.) dissolved in NBP (200 µL) for 1 hour. Following iodine treatment, the resin was thoroughly washed with NBP (3 × 1 mL), MeOH (3 × 1 mL), CH2Cl2 (3 × 1 mL), and dried under vacuum.General procedure for N-halosuccinimide Trt/Acm removal
The peptidyl resin (1 equiv.) was swollen in NBP (200 µL) at 50 °C for 20 min. The peptide was cooled to room temperature and treated with a solution of N-halosuccinimide (NCS, NBS or NIS) (4 mg, 0.022 mmol, 2.2 equiv.) dissolved in 0.5% TFA, 20% H2O in NBP (600 µL, v/v/v) for 30 min. Following NBS treatment, the resin was thoroughly washed with NBP (3 × 1 mL), MeOH (3 × 1 mL), CH2Cl2 (3 × 1 mL), and dried under vacuum.General procedure for Lys(Dde) removal
The peptidyl resin (1 equiv.) was swollen in NBP (200 µL) at 50 °C for 20 min. The peptide was cooled to room temperature and treated with a solution of 4% NH2NH2 in NBP (1 mL, v/v) for 3 × 3 min. Following NH2NH2 treatment, the resin was thoroughly washed with NBP (3 × 1 mL), MeOH (3 × 1 mL), CH2Cl2 (3 × 1 mL), and dried under vacuum.General procedure for N-chlorosuccinimide disulfide formation
The peptidyl resin (1 equiv.) was swollen in NBP (200 µL) at 50 °C for 20 min. The peptide was cooled to room temperature and treated with a solution of N-chlorosuccinimide (2 equiv.) in NBP (800 µL) for 2 × 15 min. Upon reaction completion, the resin was thoroughly washed with NBP (3 × 1 mL), MeOH (3 × 1 mL), CH2Cl2 (3 × 1 mL), and dried under vacuum.General procedure for 1,4-triazole formation
The peptidyl resin (1 equiv.) was swollen in NBP/DMSO, 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, (200 µL, v/v) at 50 °C for 20 min. To this, copper iodide (0.9 equiv.), DIPEA (5 equiv.), 2,6-lutidine (5 equiv.), NaAsc (1.5 equiv.) and TBTA (1.2 equiv.) in NBP/DMSO, 9
1, (200 µL, v/v) at 50 °C for 20 min. To this, copper iodide (0.9 equiv.), DIPEA (5 equiv.), 2,6-lutidine (5 equiv.), NaAsc (1.5 equiv.) and TBTA (1.2 equiv.) in NBP/DMSO, 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (800 µL, v/v) was added and the reaction heated (60 min, 55 °C). Upon reaction completion, the resin was washed with NBP (3 × 1 mL), MeOH (3 × 1 mL), CH2Cl2 (3 × 1 mL), and dried under vacuum.
1 (800 µL, v/v) was added and the reaction heated (60 min, 55 °C). Upon reaction completion, the resin was washed with NBP (3 × 1 mL), MeOH (3 × 1 mL), CH2Cl2 (3 × 1 mL), and dried under vacuum.
General procedure for N-terminal capping
The peptidyl resin (1 equiv.) was swollen in NBP (200 µL) at 50 °C for 20 min. The peptide was cooled to room temperature. To this acetic anhydride (3 equiv.) and DIPEA (6 equiv.) in NBP (800 µL) were added, and the reaction was agitated at r.t. for 1 hour. Upon reaction completion, the resin was thoroughly washed with NBP (3 × 1 mL), MeOH (3 × 1 mL), CH2Cl2 (3 × 1 mL), and dried under vacuum.Results and discussion
Synthesis of linear model peptides
To investigate a variety of post-synthesis modifications, model peptides with the generalised sequence *LeuTyrArgAlaLys*, where * represents a varied amino acid, were synthesised (Fig. 2).17 The choice of the model peptide was based on the LYRAX sequence reported by Hackeng et al., where X in this case is a Lys to allow for side chain protecting group variability.26 The peptides investigated herein were synthesised manually on a 0.3 mmol scale using a Fmoc SPPS strategy. The resin was swollen with NBP (5 mL) at room temperature for 1 hour. Fmoc protecting groups were removed with two treatments of 20% piperidine in NBP (5 and 15 min). Standard amino acids were coupled twice, employing 3 equivalents of amino acid, DIC and Oxyma (2 × 45 minutes) at a final concentration of 0.1 M. Arginine couplings were performed for extended time periods (2 × 1 hour) and unnatural amino acids were coupled once for 3 hours. Unless otherwise stated, the N-terminal Fmoc remained intact for orthogonal protecting group investigations using NBP. Peptides 1 and 2 contain sec-isoamyl mercaptan (SIT) and triphenylmethyl (Trt) protected cysteines respectively in the varied position, and all other amino acids utilise standard acid-labile protecting groups. Peptide 3 contains acetamidomethyl (Acm) protected cysteines in the variable position along with a 1-(4,4-dimethyl-2,6-dioxocyclohex-1-ylidene)ethyl (Dde) protected lysine in place of the standard tert-butoxycarbonyl (Boc) protection. Finally, an N-terminal propargylglycine (Pra) residue and a C-terminal azidohomoalanine (Aha) residue constitute peptide 4. The crude purity of all linear peptides was determined by analytical HPLC to be greater than 90%. | ||
| Fig. 2 Sequences of peptides (1–4) manually synthesised for post-linear synthesis modification tests in NBP. | ||
Orthogonal peptidyl transformations using sustainable solvent
Standard SIT removal is performed in DMF employing 5 equivalents of DTT per SIT-protected cysteine in a DMF/DIPEA/H2O mixture (92![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4
4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4, v/v/v) for three 20 minute treatments.27 After confirming the miscibility of water with NBP, SIT removal was attempted utilising NBP while maintaining the ratio of cleavage cocktail components.28
4, v/v/v) for three 20 minute treatments.27 After confirming the miscibility of water with NBP, SIT removal was attempted utilising NBP while maintaining the ratio of cleavage cocktail components.28
Treatment of peptide 1 under these conditions resulted in near-complete consumption of the starting material, with two major products observed: the desired product 5 and the single SIT deprotected peptide, in a 7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 56
56![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 37 ratio (Fig. 3). To improve the efficiency of the reaction, the peptide was treated with the deprotection solution in a single step for one hour. This approach yielded a comparable conversion to product 5 (47%). Encouraged by this result, the reaction was extended to 2 and 4 hours using a single set of reagents. Treatment for 2 hours resulted in an 82% conversion to product 5, with no starting material peak observed by RP-HPLC and only one of the SIT group positions being harder to remove (Fig. 3C). Extending the treatment of peptide 1 with the deprotection cocktail to 4 hours afforded the highest conversion, yielding 92% of the desired product 5.
37 ratio (Fig. 3). To improve the efficiency of the reaction, the peptide was treated with the deprotection solution in a single step for one hour. This approach yielded a comparable conversion to product 5 (47%). Encouraged by this result, the reaction was extended to 2 and 4 hours using a single set of reagents. Treatment for 2 hours resulted in an 82% conversion to product 5, with no starting material peak observed by RP-HPLC and only one of the SIT group positions being harder to remove (Fig. 3C). Extending the treatment of peptide 1 with the deprotection cocktail to 4 hours afforded the highest conversion, yielding 92% of the desired product 5.
Subjecting peptides 2 and 3 to I2 in the presence of Na2S2O8 (Fig. 4, entry A) resulted in preferential Acm removal over Trt. Peptide 3 (Acm) showed near-complete consumption of starting material. On the contrary, peptide 2 (Trt) remained largely intact despite these conditions leading to complete deprotection in DMF. Subsequently, oxidation using N-halosuccinimides was explored. Peptide 3 exhibited partial conversion for each succinimide, with the highest yield of product 7 achieved using NCS for 30 minutes (Fig. 4, entry C). Interestingly, Trt deprotection and subsequent oxidation of peptide 2 was more efficient with NIS and NBS, affording 85% and 82% of product 6 formation, respectively. Thus, in contrast to entry A, NIS and NBS provided high selectivity for Trt removal over Acm. These findings demonstrate that NBP is compatible with selective removal and oxidation of both Acm and Trt protecting groups.
Peptidomimetic cyclisation methods using sustainable solvent
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v) has been previously reported by our group.43 Due to the toxicity of TFE, we looked to adapt these conditions by searching for literature examples using other co-solvents in this reaction. We selected dimethyl sulfoxide (DMSO) as the most sustainable co-solvent and employed this at a lower ratio (NBP/DMSO, 9
1, v/v) has been previously reported by our group.43 Due to the toxicity of TFE, we looked to adapt these conditions by searching for literature examples using other co-solvents in this reaction. We selected dimethyl sulfoxide (DMSO) as the most sustainable co-solvent and employed this at a lower ratio (NBP/DMSO, 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
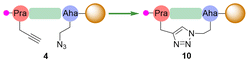
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v). Our initial investigations subjected peptide 4 to triazole forming conditions at 55 °C for 10 minutes which resulted in a 55% conversion to expected product 10. To obtain maximum yield, we repeated the reaction, increasing the time to 30 minutes or 1 hour at 55 °C. HPLC and LC/MS analysis confirmed that a 30 minutes reaction time resulted in 84% conversion, while 1 hour resulted in full conversion of starting material 4 to product 10 (Table 1, entry II) (for full HPLC spectra, see SI Fig. S25).
1, v/v). Our initial investigations subjected peptide 4 to triazole forming conditions at 55 °C for 10 minutes which resulted in a 55% conversion to expected product 10. To obtain maximum yield, we repeated the reaction, increasing the time to 30 minutes or 1 hour at 55 °C. HPLC and LC/MS analysis confirmed that a 30 minutes reaction time resulted in 84% conversion, while 1 hour resulted in full conversion of starting material 4 to product 10 (Table 1, entry II) (for full HPLC spectra, see SI Fig. S25).
Sustainable N-terminal acetylation
Following the success in protecting group removal, we aimed to investigate the effects of NBP towards peptidyl acetylation. Standard protocols employ acetic anhydride and base in DMF, depending on number of equivalents used, reaction times vary from 5 minutes (50 eq.) to multiple hours (3 eq.). Keeping in line with the reaction efficiency theme of this paper, we decided to keep the equivalents of acetic anhydride and N,N-diisopropylethylamine low (3 eq., 6 eq., respectively), trialling the reaction at three lengths. After 5 minutes, we observed a 45% conversion of the free N-terminal peptide (11) to product 12, which increased to 75% if left for 30 minutes. Following 60 minutes of treatment, full conversion to the capped peptide was observed (Table 1, Entry III) (for full HPLC spectra, see SI Fig. S26).Conclusions
This work expands the reaction profile demonstrated by NBP in peptidyl post-linear synthesis reactions. With the advantages of being non-reprotoxic and biodegradable, NBP has been identified as an appropriate sustainable alternative to DMF. Despite the challenges faced for automated synthesis due to its viscosity, NBP remains user-friendly for manual linear and post-linear synthesis workflows. Thus, we elected to develop generalised protocols for standard post-translational peptidyl reactions in NBP.First, orthogonal protecting group chemistries involving cysteine were investigated. It was determined that SIT, Acm, and Trt protecting groups can be successfully removed employing NBP as the solvent. Optimal SIT group removal was achieved in a single treatment, thus addressing one of the 12 principles of green chemistry by improving the overall reaction efficiency. Furthermore, we report orthogonal conditions for selective Acm and Trt removal in NBP. Acm groups are removed selectively via I2, while Trt is cleaved with N-bromo or N-iodosuccinimide.
Orthogonal removal of the Lys(Dde) protecting group in the presence of Cys(Acm) was achieved with a single 1 minute treatment of 4% hydrazine, without compromising the peptides alternative protecting groups.
On-resin peptide cyclisation and N-terminal acylation were successfully demonstrated in NBP. Disulfide bond formation using N-chlorosuccinimide in NBP proceeded with efficiency comparable to literature-reported DMF based conditions. Additionally, cyclic 1,4-disubstituted triazole peptides were obtained in NBP, albeit requiring extended reaction times compared to DMF. Finally, N-terminal acetylation was achieved under mild conditions, employing three equivalents of acetic anhydride for 1 hour at room temperature.
Overall, these findings establish NBP as a practical sustainable solvent for diverse post-linear synthesis modifications in peptide chemistry, offering a viable alternative to DMF. By enabling orthogonal protecting group strategies, selective transformations, key cyclisation and acylation reactions under mild conditions, this work supports the broader adoption of greener methodologies in peptidomimetic synthesis.
Author contributions
M. A. M.: experimental work, data analysis, writing original draft, M. D. F. C.: experimental work, K. I. M. A.: project conception, experimental work; A. G. J.: funding acquisition, draft editing; N. W.: funding acquisition, project conception, experimental work, data analysis, writing original draft, draft editing.Conflicts of interest
All of the authors in this study declared no conflicts of interest.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. SI contains general protocols, detailed experimental procedures and characterisation data for each crude peptide. See DOI: https://doi.org/10.1039/d5ob01890k.Acknowledgements
This work was financially supported by the RSC Sustainable Laboratories grant (L23-3258392629). M. A. M. and K. I. M. A. acknowledge the EPSRC (EP/T517896/1 and EP/W524359/1) for funding their Doctoral Scholarships. M. D. F. C. would like to acknowledge the RSC Undergraduate Research Bursary (U24-4415128223). All authors would like to acknowledge the University of Glasgow for their support. The authors acknowledge funding from the Department of the Defense, Defense Threat Reduction Agency (Research Project Grant HDTRA12210001). The content of the information does not necessarily reflect the position or the policy of the federal government, and no official endorsement should be inferred.References
- K. Wegner, D. Barnes, K. Manzor, A. Jardine and D. Moran, Green Chem. Lett. Rev., 2021, 14, 152–163 CrossRef.
- S. J. Hong, X. N. Zhang, Z. Sun and T. Zeng, J. Appl. Toxicol., 2024, 44, 1637–1646 CrossRef CAS PubMed.
- A. Gescher, Chem. Res. Toxicol., 1993, 6, 245–251 Search PubMed.
- Commission Regulation (EU) 2021/2030 of 19 November 2021 amending Annex XVII to Regulation (EC) no 1907/2006 of the European Parliament and of the Council concerning the Registration, Evaluation, Authorisation and Restriction of Chemicals (REACH) as regards N,N-dimethylformamide, Official Journal of the European Union, European Union, 2021.
- J. Sherwood, F. Albericio and B. G. de la Torre, ChemSusChem, 2024, 17, 1–6 CrossRef PubMed.
- F. P. Byrne, S. Jin, G. Paggiola, T. H. M. Petchey, J. H. Clark, T. J. Farmer, A. J. Hunt, C. R. McElroy and J. Sherwood, Sustainable Chem. Processes, 2016, 4, 1–24 Search PubMed.
- J. Lopez, S. Pletscher, A. Aemissegger, C. Bucher and F. Gallou, Org. Process Res. Dev., 2018, 22, 494–503 CrossRef CAS.
- Y. E. Jad, G. A. Acosta, T. Govender, H. G. Kruger, A. El-Faham, B. G. De La Torre and F. Albericio, ACS Sustainable Chem. Eng., 2016, 4, 6809–6814 CrossRef CAS.
- G. Vivenzio, M. C. Scala, P. Marino, M. Manfra, P. Campiglia and M. Sala, Pharmaceutics, 2023, 15, 1–12 CrossRef PubMed.
- V. Martin, S. Jadhav, P. H. G. Egelund, R. Liffert, H. Johansson Castro, T. Krüger, K. F. Haselmann, S. Thordal Le Quement, F. Albericio, F. Dettner, C. Lechner, R. Schönleber and D. S. Pedersen, Green Chem., 2021, 23, 3295–3311 RSC.
- L. Ferrazzano, D. Corbisiero, G. Martelli, A. Tolomelli, A. Viola, A. Ricci and W. Cabri, ACS Sustainable Chem. Eng., 2019, 7, 12867–12877 CrossRef CAS.
- Y. E. Jad, G. A. Acosta, S. N. Khattab, B. G. De La Torre, T. Govender, H. G. Kruger, A. El-Faham and F. Albericio, Org. Biomol. Chem., 2015, 13, 2393–2398 Search PubMed.
- J. Pawlas and J. H. Rasmussen, Green Chem., 2019, 21, 5990–5998 Search PubMed.
- Y. Ran, F. Byrne, I. D. V. Ingram and M. North, Chem. – Eur. J., 2019, 25, 4951–4964 CrossRef CAS PubMed.
- L. Ferrazzano, M. Catani, A. Cavazzini, G. Martelli, D. Corbisiero, P. Cantelmi, T. Fantoni, A. Mattellone, C. De Luca, S. Felletti, W. Cabri and A. Tolomelli, Green Chem., 2022, 24, 975–1020 RSC.
- J. Sherwood, H. L. Parker, K. Moonen, T. J. Farmer and A. J. Hunt, Green Chem., 2016, 18, 3990–3996 RSC.
- A. Kumar, M. Alhassan, J. Lopez, F. Albericio and B. G. de la Torre, ChemSusChem, 2020, 13, 5288–5294 CrossRef CAS PubMed.
- B. G. De La Torre, A. Kumar, M. Alhassan, C. Bucher, F. Albericio and J. Lopez, Green Chem., 2020, 22, 3162–3169 RSC.
- J. Pawlas, T. M. El-Dine, K. von Bargen, C. von Bargen, J. Nilsson, O. Ludemann-Hombourger and J. H. Rasmussen, Org. Process Res. Dev., 2024, 28, 3347–3367 CrossRef CAS.
- J. Pawlas, B. Antonic, M. Lundqvist, T. Svensson, J. Finnman and J. H. Rasmussen, Green Chem., 2019, 21, 2594–2600 RSC.
- A. Isidro-Llobet, M. Álvarez and F. Albericio, Chem. Rev., 2009, 109, 2455–2504 CrossRef CAS PubMed.
- R. Kowalczyk, P. W. R. Harris, G. M. Williams, S.-H. Yang and M. A. Brimble, in Peptides and Peptide-based Biomaterials and their Biomedical Applications. Advances in Experimental Medicine and Biology, ed. A. Sunna, A. Care and P. L. Bergquist, Springer, Cham, 2017, vol. 1030 Search PubMed.
- W. Tang and M. L. Becker, Chem. Soc. Rev., 2014, 43, 7013–7039 Search PubMed.
- B. W. Cue, M. Cann, D. J. C. Constable, N. D. Anastas, C. Jiménez-González, R. Wool, P. J. Dunn, R. A. Sheldon, R. Williams, D. Raynie, S. Bradley, D. C. Finster, T. Goodwin, P. T. Anastas and J. C. Warner, 12 Principles of Green Chemistry.
- P. Anastas and N. Eghbali, Chem. Soc. Rev., 2010, 39, 301–312 RSC.
- T. M. Hackeng, J. H. Griffin and P. E. Dawson, Protein synthesis by native chemical ligation: Expanded scope by using straightforward methodology, 1999, vol. 96 Search PubMed.
- A. Chakraborty, A. Sharma, F. Albericio and B. G. De La Torre, Org. Lett., 2020, 22, 9644–9647 CrossRef CAS PubMed.
- F. P. Byrne, C. M. Nussbaumer, E. J. Savin, R. A. Milescu, C. R. McElroy, J. H. Clark, B. M. A. van Vugt-Lussenburg, B. van der Burg, M. Y. Meima, H. E. Buist, E. D. Kroese, A. J. Hunt and T. J. Farmer, ChemSusChem, 2020, 13, 3212–3221 CrossRef CAS PubMed.
- Y. Xing, Y. Wang, D. Ma, S. Shen, C. Song, N. Zhang, T. Bo, T. Shi and S. Huo, Tetrahedron Lett., 2023, 120, 1–5 Search PubMed.
- N. M. Tripathi, B. K. Das, A. Chowdhury, V. Gour and A. Bandyopadhyay, JACS Au, 2025, 5, 802–810 CrossRef CAS PubMed.
- B. Kamber, A. Hartmann, K. Eisler, B. Riniker, H. Rink, P. Sieber and W. Rittel, Helv. Chim. Acta, 1980, 63, 899–915 CrossRef CAS.
- A. Chakraborty, F. Albericio and B. G. de la Torre, Int. J. Mol. Sci., 2025, 26, 1–8 Search PubMed.
- A. Chowdhury, V. Gour, B. K. Das, S. Chatterjee and A. Bandyopadhyay, Org. Lett., 2023, 25, 1280–1284 CrossRef CAS PubMed.
- B. W. Bycroft, W. C. Chan, R. Chhabra and N. D. Hone, J. Chem. Soc., Chem. Commun., 1993, 778–779 RSC.
- B. Guérin, S. Ait-Mohand, M. C. Tremblay, V. Dumulon-Perreault, P. Fournier and F. Bénard, Org. Lett., 2010, 12, 280–283 CrossRef PubMed.
- Y. Itoh, K. Aihara, P. Mellini, T. Tojo, Y. Ota, H. Tsumoto, V. R. Solomon, P. Zhan, M. Suzuki, D. Ogasawara, A. Shigenaga, T. Inokuma, H. Nakagawa, N. Miyata, T. Mizukami, A. Otaka and T. Suzuki, J. Med. Chem., 2016, 59, 1531–1544 CrossRef CAS PubMed.
- T. M. Postma and F. Albericio, Org. Lett., 2013, 15, 616–619 CrossRef CAS PubMed.
- V. V. Rostovtsev, L. G. Green, V. V. Fokin and K. B. Sharpless, Angew. Chem., Int. Ed., 2002, 41, 2596–2599 CrossRef CAS PubMed.
- C. W. Tornøe, C. Christensen and M. Meldal, J. Org. Chem., 2002, 67, 3057–3064 Search PubMed.
- A. M. White, S. J. de Veer, G. Wu, P. J. Harvey, K. Yap, G. J. King, J. E. Swedberg, C. K. Wang, R. H. P. Law, T. Durek and D. J. Craik, Angew. Chem., Int. Ed., 2020, 59, 11273–11277 CrossRef CAS PubMed.
- S. Pacifico, A. Kerckhoffs, A. J. Fallow, R. E. Foreman, R. Guerrini, J. McDonald, D. G. Lambert and A. G. Jamieson, Org. Biomol. Chem., 2017, 15, 4704–4710 Search PubMed.
- A. Knuhtsen, C. Whitmore, F. S. McWhinnie, L. McDougall, R. Whiting, B. O. Smith, C. M. Timperley, A. C. Green, K. I. Kinnear and A. G. Jamieson, Chem. Sci., 2019, 10, 1671–1676 RSC.
- O. A. Shepperson, M. A. Malone, K. I. M. Arnott, R. J. Brown and A. G. Jamieson, Org. Lett., 2025, 27, 11249–11253 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |